Published online Mar 25, 2026. doi: 10.5527/wjn.v15.i1.114185
Revised: November 24, 2025
Accepted: January 16, 2026
Published online: March 25, 2026
Processing time: 182 Days and 13.5 Hours
Renal emergencies include a wide variety of pathologies requiring immediate diagnosis and management to reduce morbidity and mortality. Imaging plays a crucial role in identifying the underlying condition, which guides clinical de
Core Tip: Imaging aids in prompt diagnosis and management of emergent renal conditions of various etiologies such as traumatic, vascular, infectious, and obstructive. Thereby, it plays a major role in preserving the renal function and preventing the progression to end stage disease. Our article serves as a guide for the residents to recognize these conditions and guide the diagnosis.
- Citation: Vulasala SSR, Robinson J, Engel C, Zulia Y, Sharma A, Gopireddy DR, Adler G, Virarkar M. Computed tomography of renal emergencies: A comprehensive diagnostic guide for radiology residents. World J Nephrol 2026; 15(1): 114185
- URL: https://www.wjgnet.com/2220-6124/full/v15/i1/114185.htm
- DOI: https://dx.doi.org/10.5527/wjn.v15.i1.114185
Renal emergencies are among the most encountered pathologies in the emergency department. Prompt diagnosis is crucial for minimizing morbidity and mortality and preserving renal function. Ultrasound is the first-line imaging modality due to its wide availability and absence of radiation exposure. However, interobserver variability and large body habitus limit the exam and reduce the sensitivity of ultrasound. With the evolution of imaging technology allowing for better resolution and multiplanar reformatting, computed tomography (CT) has become the primary modality for evaluating suspected renal emergencies. In this review, we discuss the CT findings of a diverse spectrum of renal pathologies under traumatic, vascular, obstructive, and infectious etiologies.
Retroperitoneum is divided by anterior (Gerota) and posterior (Zuckerkandl) perirenal fascia into anterior pararenal space, posterior pararenal space, and perirenal/perinephric space[1]. The anterior pararenal space is bounded anteriorly by the anterior pararenal fascia and posteriorly by the Gerota fascia. Posterior pararenal space is bounded anteriorly by the Zuckerkandl fascia, laterally by the lateroconal fascia, and posteriorly by the posterior pararenal fascia. The anterior pararenal fascia and posterior pararenal fascia are not single fascial layers but rather composed of multiple mesenteric layers, creating a space in between the fascial layers. The space within the anterior pararenal fascia is the retromesenteric plane, and the space within the posterior pararenal fascia is the retrorenal plane. The anterior and posterior pararenal fasciae fuse laterally to become a lateroconal plane and fuse inferiorly to become a combined interfascial plane[2].
Understanding the retroperitoneal anatomy is crucial for describing radiologic findings and recognizing the pattern of pathologic spread, such as hemorrhage or infection. For instance, the hemorrhage contained within the perirenal space is classified as American Association for the Surgery of Trauma (AAST) grade III, and any hemorrhage extending beyond the perirenal fascia is upgraded to grade IV. The detailed findings of the renal trauma grading are discussed in further sections. In patients with ruptured pyonephrosis, the purulent fluid extends along the interfascial planes into the peritoneum, resulting in fistula formation between the renal system and gastrointestinal tract.
Typically, each kidney is supplied by a single renal artery, which serves the right and left kidney, respectively. They originate from the abdominal aorta below the superior mesenteric artery origin. Main renal arteries divide into segmental arteries at the renal hilum, which further branch into lobar arteries at the renal sinuses, then into interlobar arteries, arcuate arteries, and interlobular arteries. Venous drainage is through interlobular veins, arcuate veins, lobar veins, the central renal vein, and the inferior vena cava. Around 30% of the patients may have supernumerary renal arteries, and 15%-30% may have supernumerary renal veins. It is essential to recognize the importance of renal vascular anatomy in guiding endovascular or surgical procedures. For example, the AAST grading of renal trauma varies based on whether the segmental or main renal artery or vein is involved, which determines the clinical management.
Kidneys are the third most commonly injured abdominal organs (10%-20%) in blunt abdominal trauma and the most frequently involved genitourinary organs (60%)[2,3]. Given their location in the retroperitoneum, these solid organs are relatively well protected, in the adult population, by the peritoneum, retroperitoneal fat, paravertebral musculature, and rib cage[4]. The pediatric population is more prone to renal trauma due to multiple factors, including: (1) Larger kidneys relative to the size of the abdominal cavity; (2) Weaker connective tissues and less ossified ribs; (3) Increased mobility on the vascular pedicle; and (4) Higher sustained force of impact secondary to smaller body surface area[2]. Renal trauma comprises 5% of the trauma cases, and approximately 245000 cases are encountered per year worldwide[2]. Amongst these cases, 90% are secondary to blunt abdominal injury from falls, motor vehicle collisions, and assaults, whereas 10% constitute penetrating injuries from gunshot and stab wounds[2,5]. Although less common, the penetrating trauma is associated with high-grade renal injury compared to the blunt trauma (68% vs 25%)[4].
Patients typically present with microscopic or gross hematuria, flank pain, and abdominal wall bruising. At our institution, the general trauma imaging protocol consists of contrast-enhanced CT in both the portal venous and excretory phases for the comprehensive evaluation of all solid organ injuries. If there is an imaging concern regarding active bleeding, a follow-up contrast-enhanced CT scan can be obtained in the arterial phase. The kidneys enhance uniformly during the portal venous phase, making the parenchymal injuries more conspicuous. The excretory phase evaluates for hilar injury involving the renal collecting system, ureters, and urinary bladder. Renal trauma is graded based on the AAST-Organ Injury Scale, which was revised in 2018 (Table 1)[4]. Grades I-III are classified as low-grade injuries, while grades IV-V are the high-grade injuries.
| Grade | Description | Imaging findings |
| I | Subcapsular hematoma +/- renal parenchymal contusion without definite laceration | Contusion: Ill-defined area of hypoattenuation on nephrographic phase; hyperdense on excretory phase due to retained parenchymal contrast. Subcapsular hematoma: Iso-to-hyperdense renal subcapsular collection |
| II | Renal parenchymal laceration of ≤ 1 cm depth with perirenal hematoma; absent urinary extravasation | Laceration: Linear area of hypoattenuation of ≤ 1 cm in length. Perirenal hematoma: Ill-defined stranding or hyperdense collection contained within the Gerota fasci. Absent urinary extravasation on excretory phase |
| III | Renal parenchymal laceration of > 1 cm depth without urine extravasation, active kidney bleeding contained within Gerota fascia, renal vascular pseudoaneurysm, or an AVF contained within Gerota fascia | Laceration: Linear area of hypoattenuation of > 1 cm in length. Vascular findings contained within the Gerota fascia: Pseudoaneurysm: Focal contrast-filled outpouching. AVF: Early renal venous opacification. Absent urinary extravasation on excretory phase |
| IV | Parenchymal laceration extending to the renal collecting system with urine extravasation, renal pelvic laceration +/- complete ureteropelvic disruption, segmental renal vein or artery AVF or pseudoaneurysm, active bleeding beyond Gerota fascia into retroperitoneal or peritoneal cavity, segmental or complete kidney infarction due to vessel thrombosis but without active bleeding | Urine extravasation on excretory phase. Segmental vascular involvement with findings similar to grade III, however, extending beyond Gerota fascia. Wedge-shaped hypoattenuations indicating renal infarcts |
| V | Shattered kidney with loss of identifiable parenchymal anatomy, devascularized kidney with or without active bleeding, lacerated main renal artery or vein, or an avulsed renal hilum | Active contrast extravasation on early phase with blooming on delayed phases indicating active vascular bleeding. Extensive ill-defined renal parenchymal hypoattenuation with disrupted anatomy |
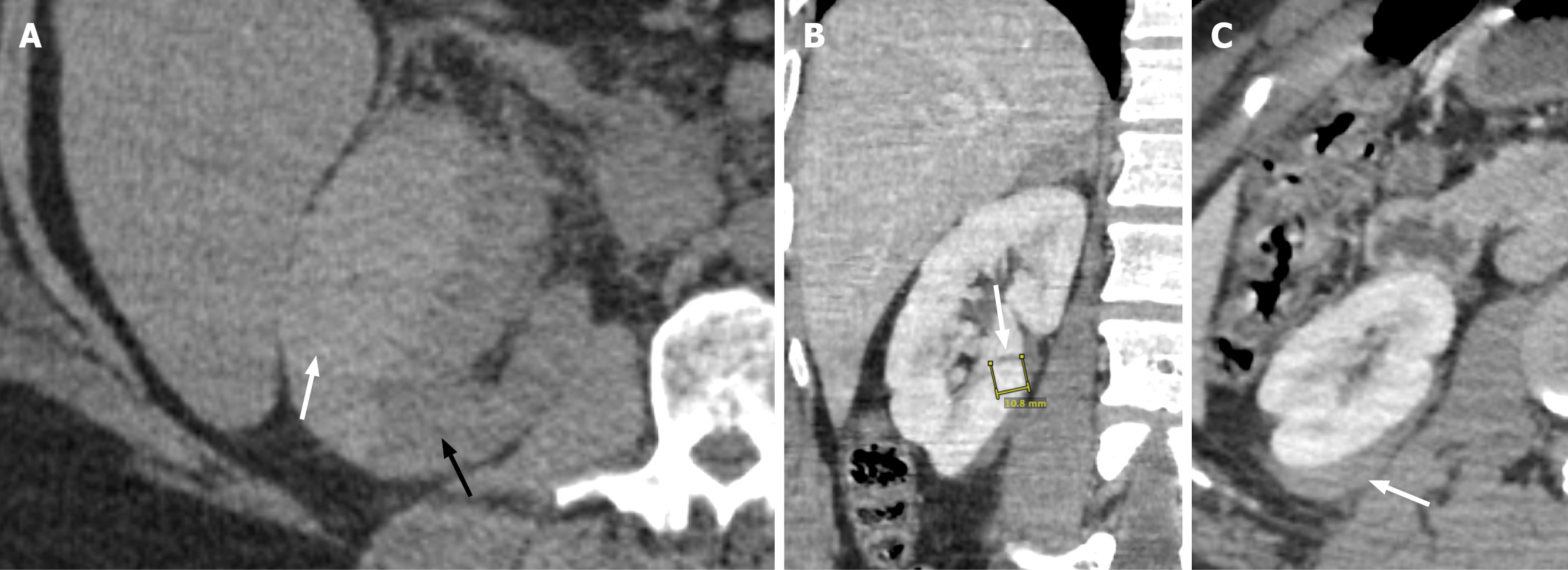
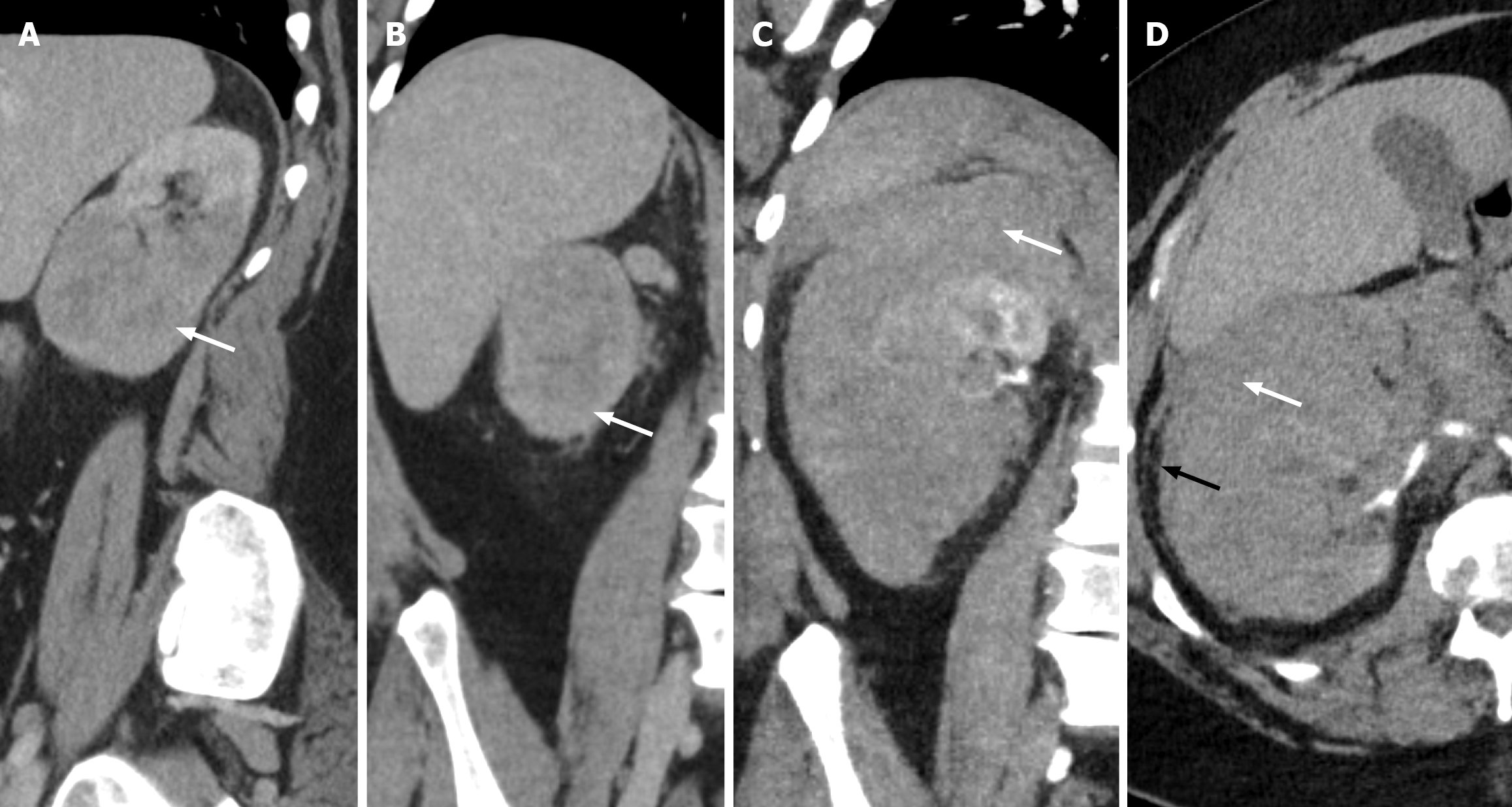
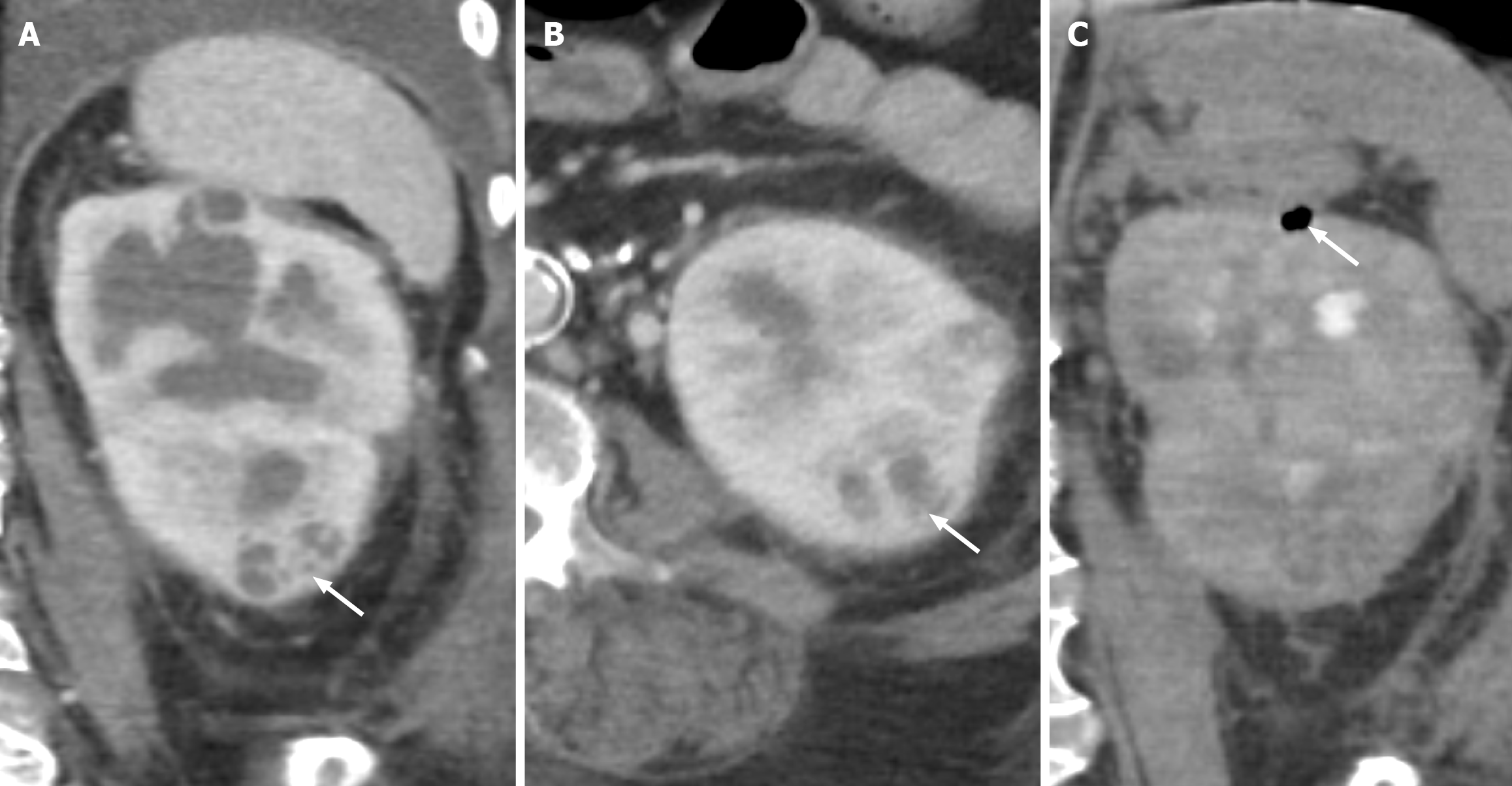
Isolated parenchymal contusion without laceration and/or subcapsular hematoma are the imaging signs of grade I renal injury. These injuries comprise 75%-85% of all renal trauma[2]. Parenchymal contusion demonstrates a focal region of ill-defined hypoenhancement on the nephrogenic phase and is less apparent on the corticomedullary phase due to suboptimal renal pyramid enhancement. Retained parenchymal contrast can be noted in the area of renal contusion on the excretory phase. Acute subcapsular hematomas appear as hyperdense (50-70 Hounsfield units) crescentic or lentiform collections between the renal cortex and fibrous renal capsule (Figure 1A)[4,6]. In hyperacute subcapsular hematoma, the unclotted blood products appear isodense, with a mean attenuation ranging from 30 Hounsfield units to 50 Hounsfield units[4]. With the evolving blood products, subacute to chronic subcapsular hematomas become iso- to hypodense relative to the renal parenchyma. Large subcapsular hematomas cause a mass effect on the adjacent renal cortex, resulting in increased arterial resistance and activation of the renin-angiotensin-aldosterone system, known as a Page kidney[7]. The Page kidney is discussed in detail under the section on spontaneous renal hemorrhage (SRH).
Renal parenchymal laceration of ≤ 1 cm depth with associated perirenal hematoma is classified as grade II renal injury. The renal collecting system is not involved in this category. The laceration appears as a linear hypoattenuation within the renal parenchyma. Unlike a subcapsular hematoma, the perirenal hematoma is not confined by the renal capsule. It can involve the perinephric fat as an ill-defined stranding or collection between the renal capsule and Gerota fascia (Figure 1B and C). The collection may cross midline and extend inferiorly to the pelvis while still being contained within the Gerota fascia.
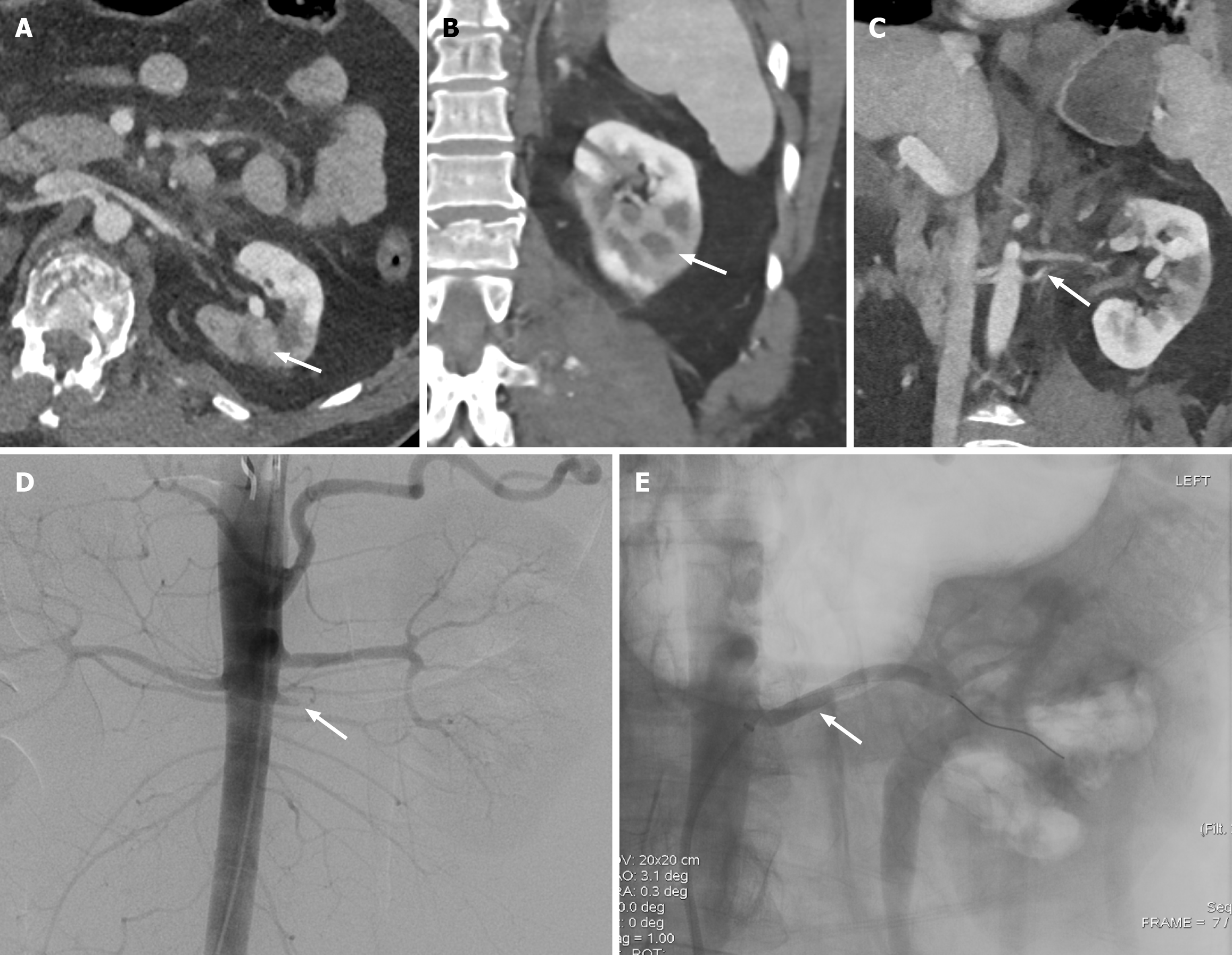
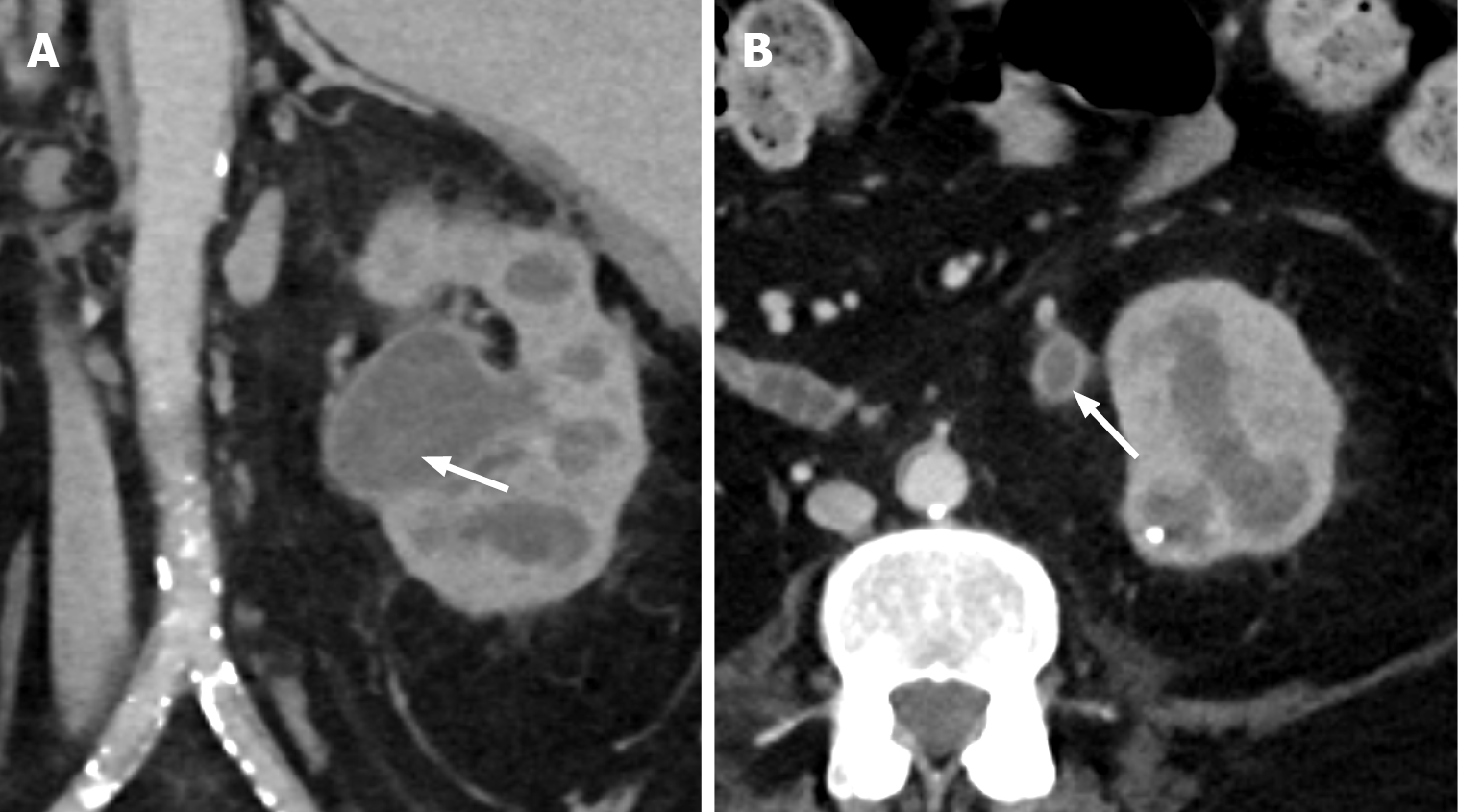
Renal parenchyma laceration of greater than 1 cm depth without involvement of the renal collecting system, or any low-grade (I-III) renal injury with signs of vascular injury or active bleeding contained within Gerota fascia, is classified under Grade III renal injury (Figure 2). The signs of vascular injury include pseudoaneurysm or an arterio-venous malformation, which are more common with penetrating injuries compared with blunt abdominal trauma (Figure 3)[6]. In grade III, the pseudoaneurysm or arterio-venous malformation is observed within the arteries or veins distal to the level of segmental branches[8]. If the segmental branches are involved, the injury is upgraded to a high-grade category (IV or V)[8]. The pseudoaneurysm appears as a focal outpouching along the vasculature with attenuation similar to that of the blood pool[6]. The size of the pseudoaneurysm remains constant on delayed-phase images, unlike active contrast extravasation[6]. Arteriovenous malformation demonstrates early opacification of the renal vein with an increased venous caliber. Active bleeding in grade III injury is contained within the Gerota fascia, and if the fascial limit is breached, the injury is upgraded to grade IV[8].
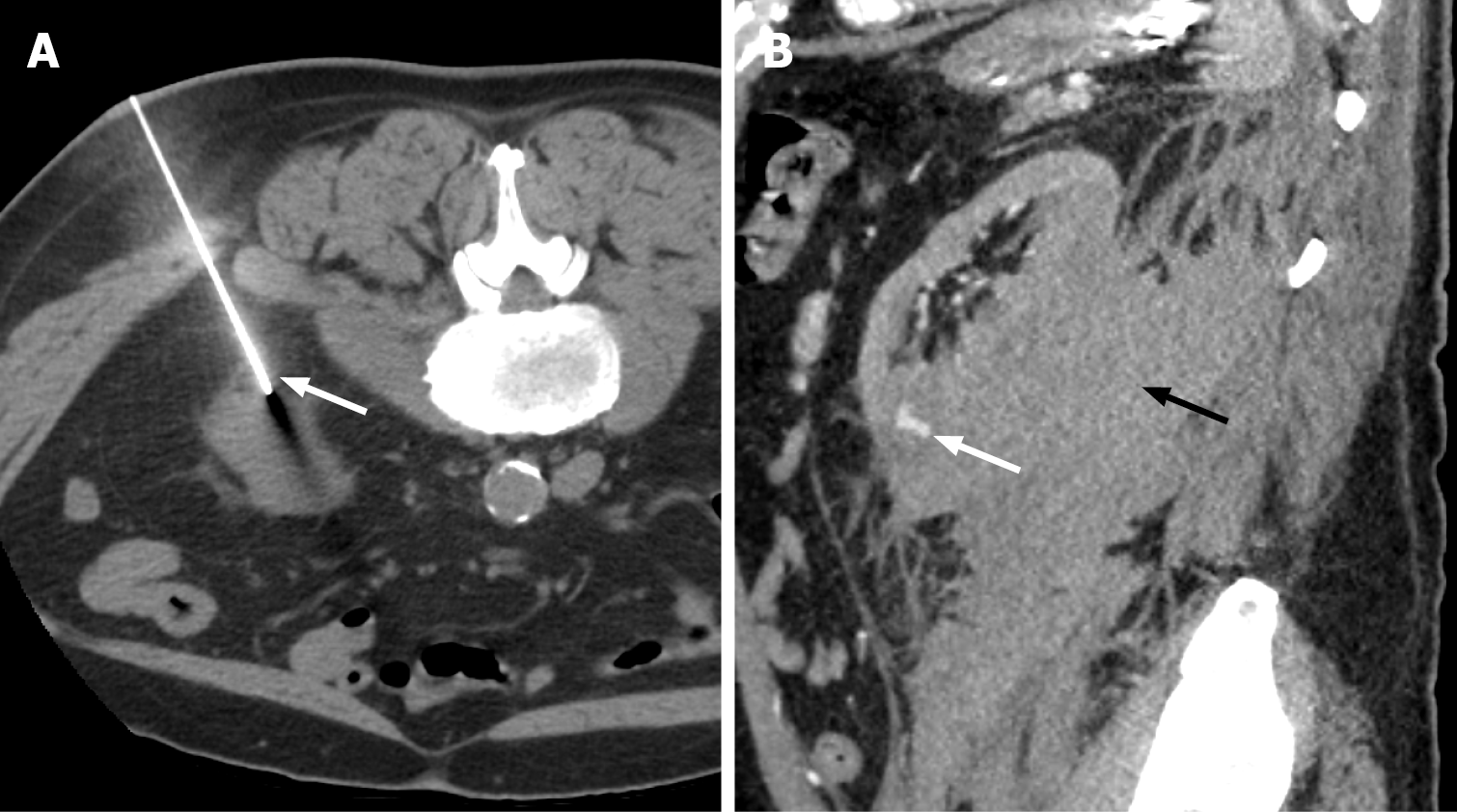
Grade IV and V are considered high-grade renal injuries with varying degrees of renal parenchymal, collecting system, and vascular involvement (Table 1)[4]. The involvement of the renal collecting system is one of the vital imaging features that upgrades renal injury from low-grade to high-grade. It can occur either through the extension of a renal parenchymal laceration to the collecting system or through an isolated renal pelvic laceration or ureteropelvic disruption. The involvement of the collecting system can be confirmed in the excretory phase if there is active extravasation of the excreted contrast (Figure 4). The injury can be a partial or complete avulsion. In case of partial ureteropelvic or pelvic disruption, the excreted contrast may still be visible within the ureters distal to the injury site. The absence of comparison with the ureters distal to the injury site is highly concerning for complete ureteropelvic avulsion.
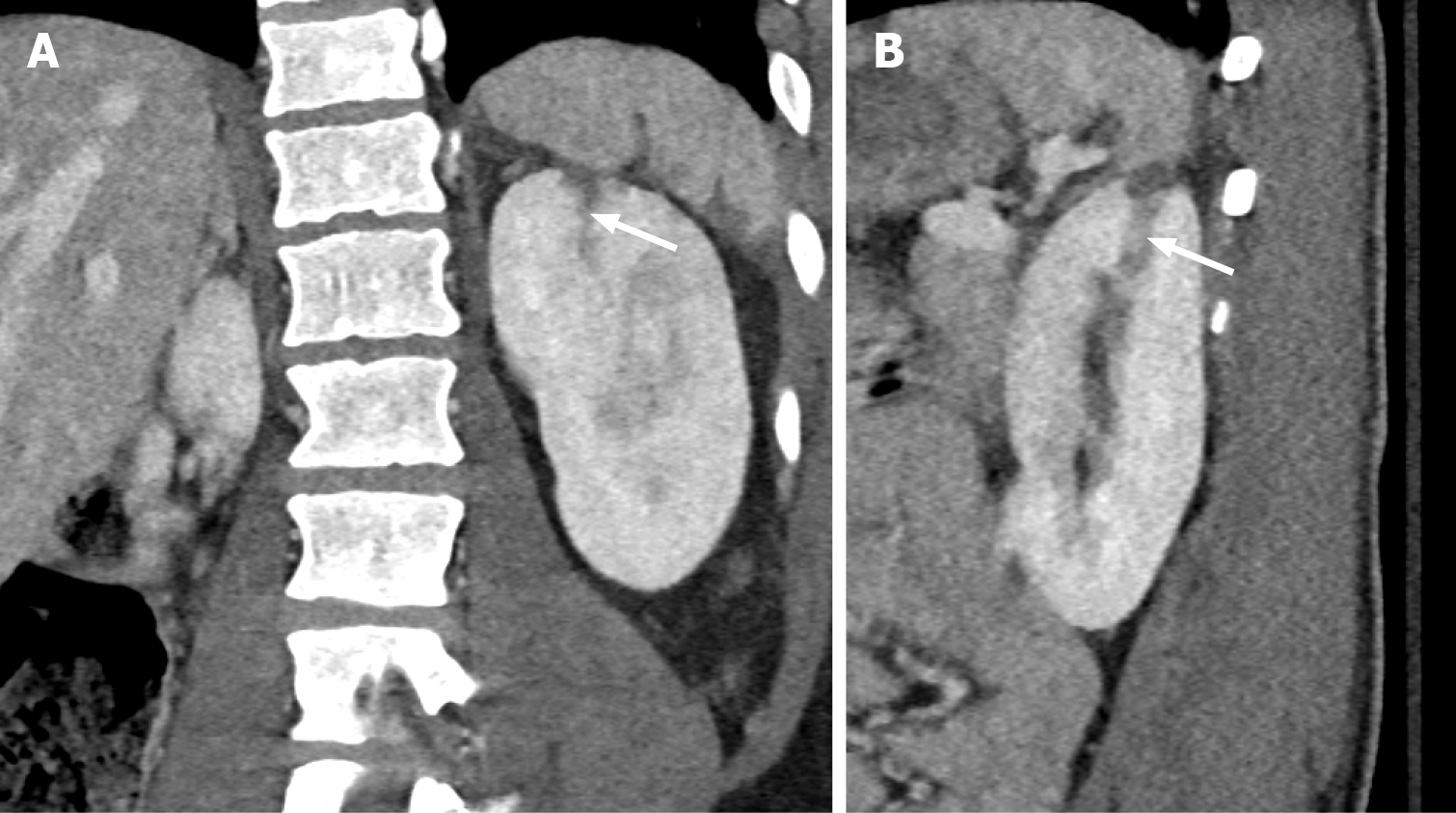
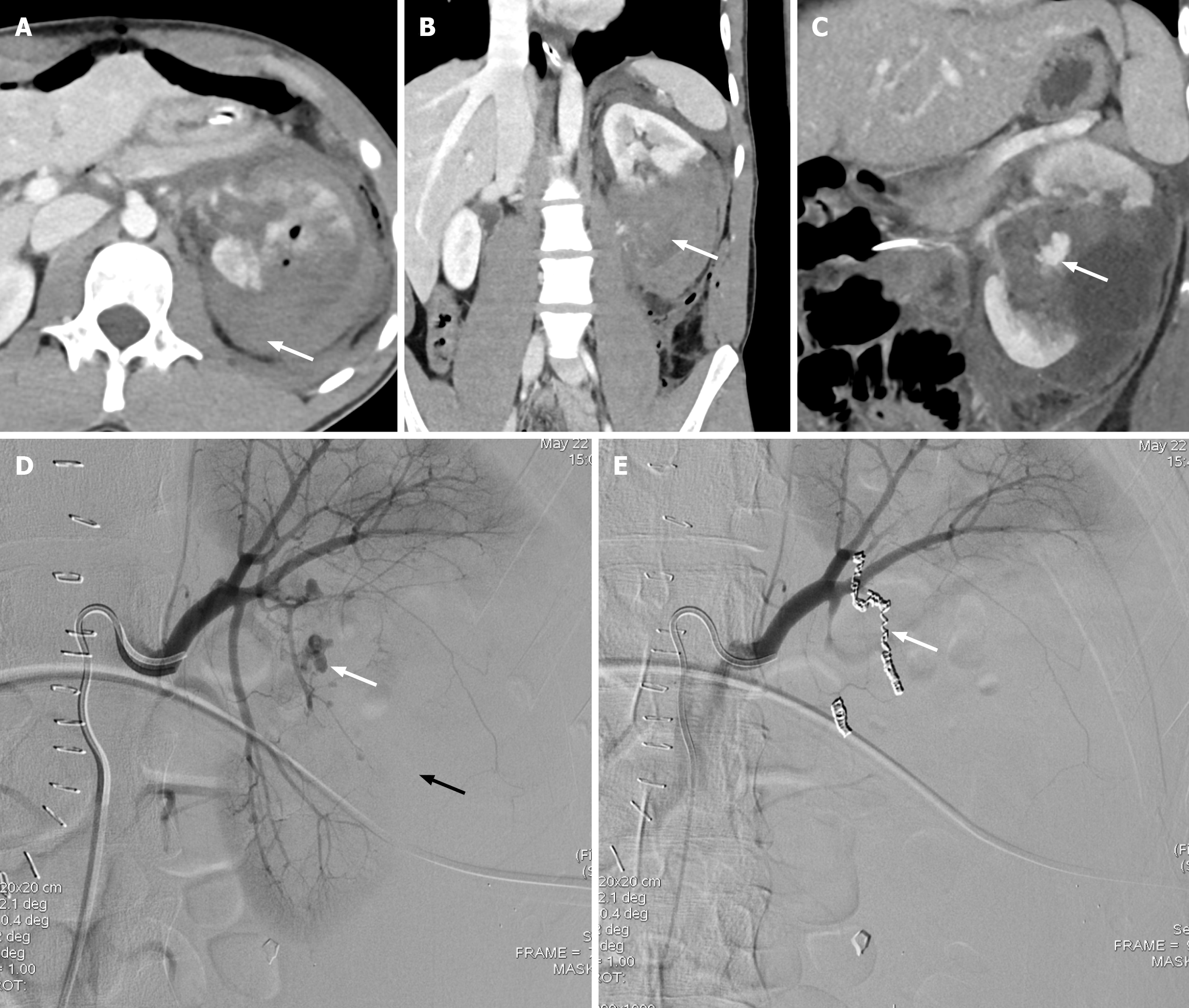
Vascular involvement with bleeding beyond the Gerota fascia is characteristic of high-grade renal injuries. In grade IV, the segmental or main renal artery is involved, resulting in uncontained hemorrhage within the retroperitoneum and peritoneum. As a consequence, the patients become hemodynamically unstable and rapidly progress towards death. Other forms of vascular involvement include intimal tear, dissection, and thrombosis without signs of active bleeding, which ultimately can result in segmental or complete renal infarction (Figure 5). An infarct can be differentiated from a contusion by its wedge-shaped hypoattenuation and sharp margins, whereas a contusion has ill-defined margins. A detailed imaging evaluation of the renal infarcts is discussed in a separate section of this article.
Grade V renal injury is the severe form with involvement of the renal parenchyma and vasculature, and accounts for only 5% of renal trauma cases[4]. The avulsion of the renal hilum with laceration of the main renal artery or vein results in a completely devascularized kidney with signs of active bleeding (Figure 6). Emergent nephrectomy is warranted in these patients. Numerous parenchymal lacerations result in the loss of renal integrity and are termed a shattered kidney.
The primary role of follow-up imaging is to assess complications such as urinoma, active hemorrhage, or arteriovenous fistula. Follow-up imaging is not necessary in low-grade injuries, as it has no effect on the outcome. These patients are typically managed with adequate bed rest, analgesia, and hydration. In low-grade and high-grade injuries, vascular complications can be managed with endovascular embolization, thereby sparing the need for nephrectomy. Superselective renal artery embolization minimizes the amount of renal parenchyma that needs to be sacrificed and preserves renal function[3].
Renal collecting system injuries typically heal spontaneously in 80% of cases, unless there is involvement of the renal pelvis or ureteropelvic junction. Percutaneous nephrostomy or urinary diversion with stent placement can be performed in patients with persistent urinary extravasation. Persistent urine leak can lead to a urinoma, with the majority demonstrating spontaneous resolution. In some cases, urinomas may become superinfected, requiring surgical or percutaneous drainage catheter placement[6].
A segmental renal infarct can result from renal artery injury or thrombosis. These can manifest as wedge-shaped hypoattenuation within the renal parenchyma, with the base directed away from the renal hilum and the apex towards the renal hilum. Unlike renal contusion, the borders of renal infarct are well-defined and confined to the vascular plane. The extent of the renal infarct depends on the size of the occluded artery, with the more proximal central renal artery occlusion resulting in an ipsilateral devascularized kidney. Small segmental infarcts do not compromise renal function[8]. The devascularized kidney demonstrates absent enhancement on arterial and portal-venous phases. Urinary contrast excretion is not evident on delayed phase images due to loss of renal function. A characteristic cortical rim sign has been described in the literature, characterized by the development of collateral circulation from capsular, periureteric, and peripelvic arteries[8]. This finding is evident on the follow-up imaging obtained more than 8 hours post-traumatic insult, when collaterals have had time to develop. The cortical rim sign also aids in determining the age of a renal infarct, which guides clinical management[8]. An additional characteristic finding is the retrograde filling of the renal vein through the inferior vena cava, resulting from renal hypoperfusion and low renal venous pressures. This finding is more pronounced in the right kidney relative to the left, due to its shorter course and lack of significant collaterals. On the left, the renal vein receives drainage from the adrenal, gonadal, and lumbar veins[8].
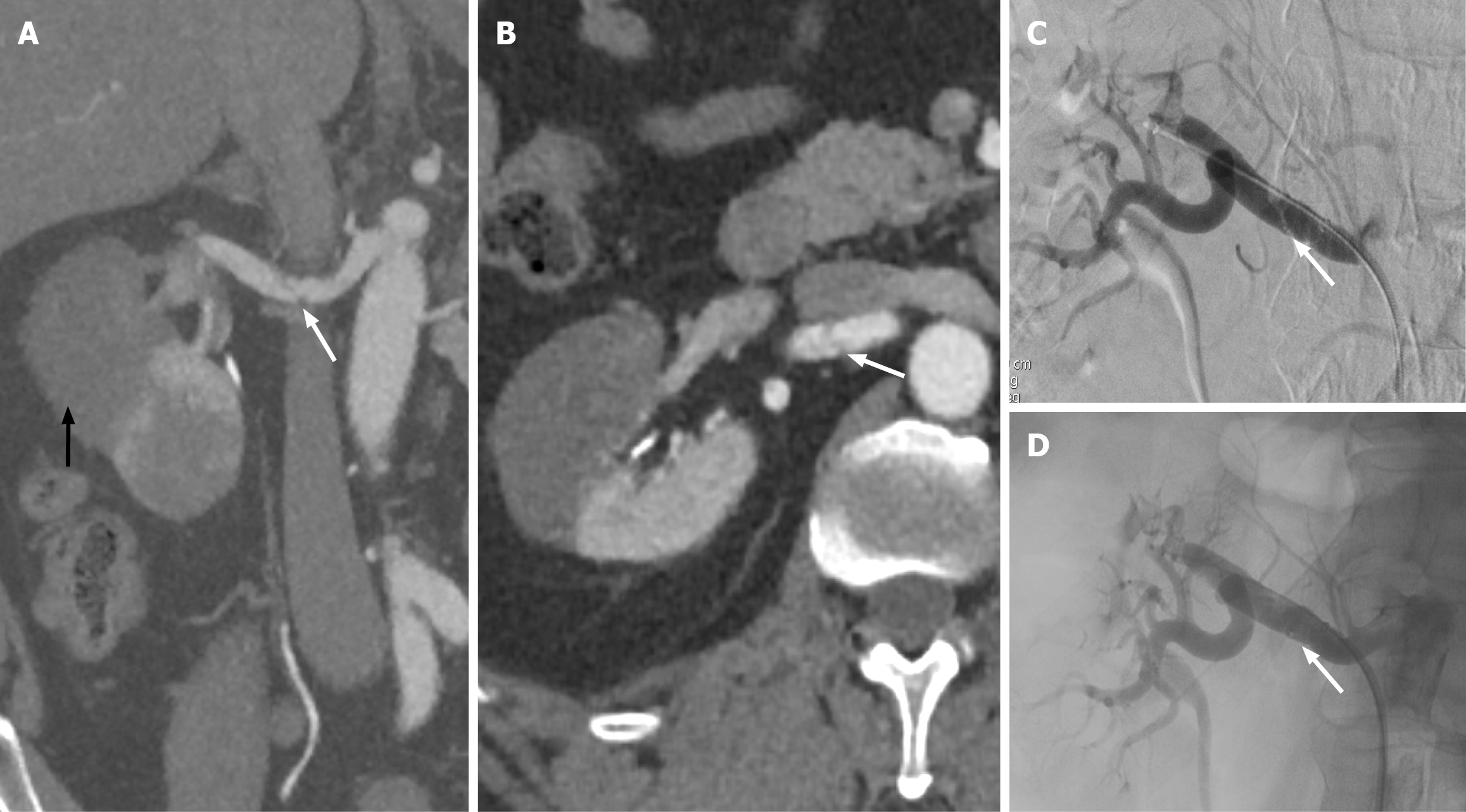
Rapid deceleration injuries can cause arterial stretching, leading to subintimal dissection and thrombosis (Figure 7). More proximal renal artery dissection and associated thrombosis result in a devascularized kidney. The patients with a unilateral devascularized kidney can be managed with close follow-up for the development of renovascular hypertension. Endovascular thrombectomy can be performed in patients with bilateral renal artery thrombosis or in a patient with a solitary kidney and unilateral renal artery thrombosis[8]. Studies show that the revascularization performed within 4 hours of trauma has a high chance of restoring renal function[8].
The renal artery dissection/thrombosis should be differentiated from renal hilar avulsion and renal artery laceration injuries because the latter would upgrade the patient to grade V renal injury, and the management would differ. The absence of a hematoma surrounding the renal artery in the perinephric space, the ability to follow the entire course of the renal artery, and the lack of active contrast extravasation favor a diagnosis of renal artery dissection/thrombosis. If a diagnosis of renal hilar avulsion or renal artery laceration is made, endovascular management can be performed in hemodynamically stable patients. At the same time, surgical management is preferred if the patient is hemodynamically unstable[8].
The SRH is defined as renal hemorrhage in the absence of trauma (Figure 8)[9]. Most often associated with underlying renal neoplasms, such as benign angiomyolipoma (AML) and malignant renal cell carcinoma (RCC), as well as vascular pathologies like vasculitis and vascular malformation, systemic hematologic conditions including hemophilia, polyarteritis nodosa, and systemic lupus erythematosus, and obstructive diseases of the pelvicalyceal system. Pregnancy is also a risk factor for SRH due to increased gonadal vein and renal vein pressure. Approximately 20% of the patients present with a characteristic “Lenk’s triad” comprising flank pain, flank mass, and hypovolemic shock, and is termed as Wunderlich syndrome[10]. The remaining patient population may present with isolated symptoms, with the flank pain being the most common (67%), followed by hematuria, flank mass, and hypovolemic shock[10]. The SRH may compress the renal parenchyma extrinsically, increasing renal arteriolar resistance and decreasing renal blood flow. This would further activate the renin-angiotensin-aldosterone system, resulting in systemic hypertension, also known as Page kidney. Early diagnosis is crucial due to its high mortality rate, running from 2.3%-14%[9].
CT is often the initial imaging modality of choice in the current clinical practice and aids in detecting the size and extent of involvement. If it occurs, the hemorrhage is confined within the renal parenchyma or spreads to the subcapsular region, perinephric space, and retroperitoneal space (anterior pararenal space). The SRH demonstrates high attenuation on non-contrast CT. Active extravasation on contrast-enhanced arterial-phase images and blooming on delayed-phase images indicate active bleeding. The hematocrit effect - manifested as a fluid-fluid level resulting from sedimentation of blood cells beneath a plasma-rich supernatant - is a radiologic sign suggestive of SRH, particularly in the setting of coagulation abnormalities[11].
Renal neoplasms comprise two-thirds of cases of SRH, with AML (40%) being the most common, followed by RCC (30%-35%). Patients with AML have elastin-poor vessel walls, which are prone to aneurysms and rupture. An AML of greater than 4 cm in size or an aneurysm of greater than 5 mm in size has an increased risk for rupture[12]. The presence of macroscopic intratumoral fat is characteristic of an AML. Around 5% of AML may not exhibit detectable fat on imaging[12]. At the time of acute SRH presentation, CT is only 57% sensitive in identifying the underlying neoplasm due to obscuration by extensive hemorrhage. Prophylactic transarterial embolization is the treatment of choice for patients presenting with active bleeding. It is helpful in controlling bleeding as well as achieving a tumor-reduction effect, especially in tumors less than 7 cm[10]. Nephron-sparing surgery can be performed in patients with failed transarterial embolisation. Total nephrectomy is the ultimate treatment option in patients with large tumors or uncontrollable bleeding[10].
Only 1% of RCCs are associated with SRH, particularly the clear cell type, due to its hypervascularity and rapid growth[10]. Additional risk factors include intratumoral necrosis, arteriovenous shunting, and renal vascular invasion. If not obscured by hemorrhage, RCC would typically appear as a heterogeneously enhancing mass with areas of necrosis or hemorrhage. They enhance during the corticomedullary phase with washout on nephrographic and delayed phase imaging. In patients with an obscured mass, follow-up imaging is recommended after the hematoma has evolved. Acute management depends on the patient’s hemodynamic status and the size of the hematoma. Prophylactic transarterial embolisation can be performed to achieve a temporary halt in hemorrhage, followed by definitive treatment depending on the tumoral staging[10].
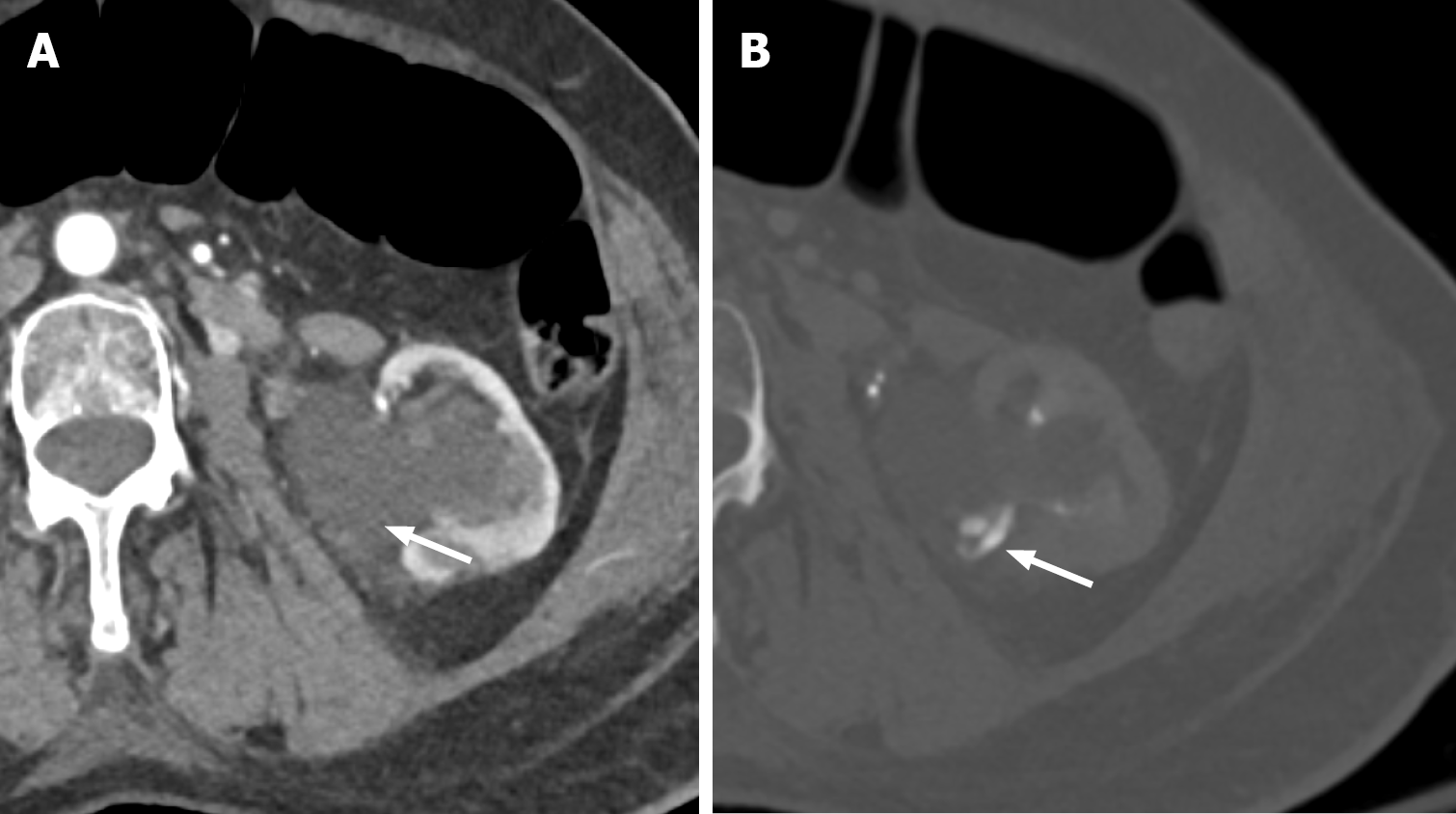
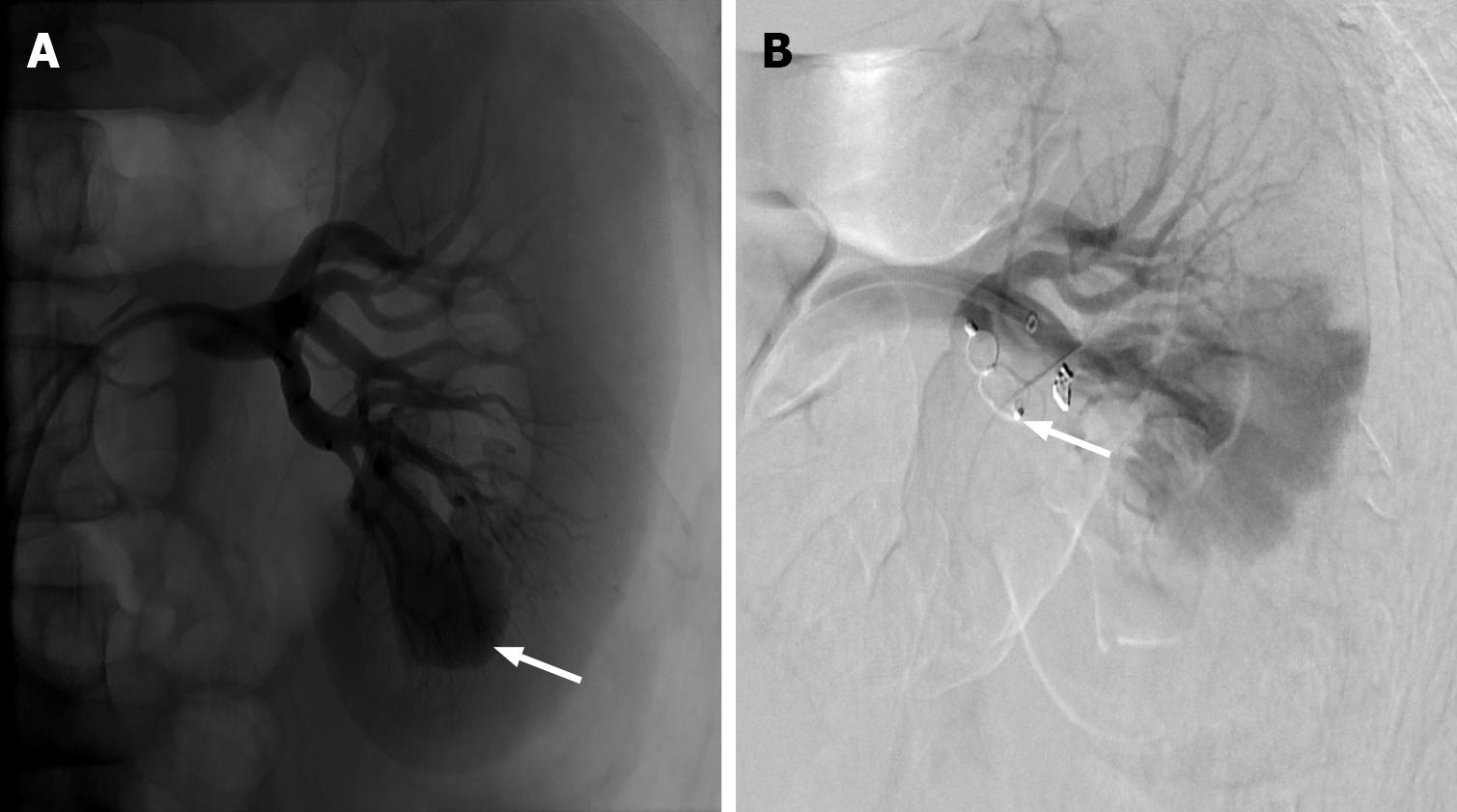
Traumatic renal arterio-venous fistula (AVF) can occur secondary to renal injury due to penetrating or blunt trauma, renal biopsy, or surgery (Figure 9). Around 7.4%-14% of renal AVF develop following renal biopsies, and most cases resolve spontaneously within 2 years[13,14]. If symptomatic, the AVF requires endovascular or surgical repair. The symptoms vary from mild flank pain, hematuria, and hypertension to severe high-output cardiac failure. The fistula diverts the blood supply away from the normal renal parenchyma, resulting in renal ischemia and activation of the renin-angiotensin-aldosterone system, leading to secondary hypertension.
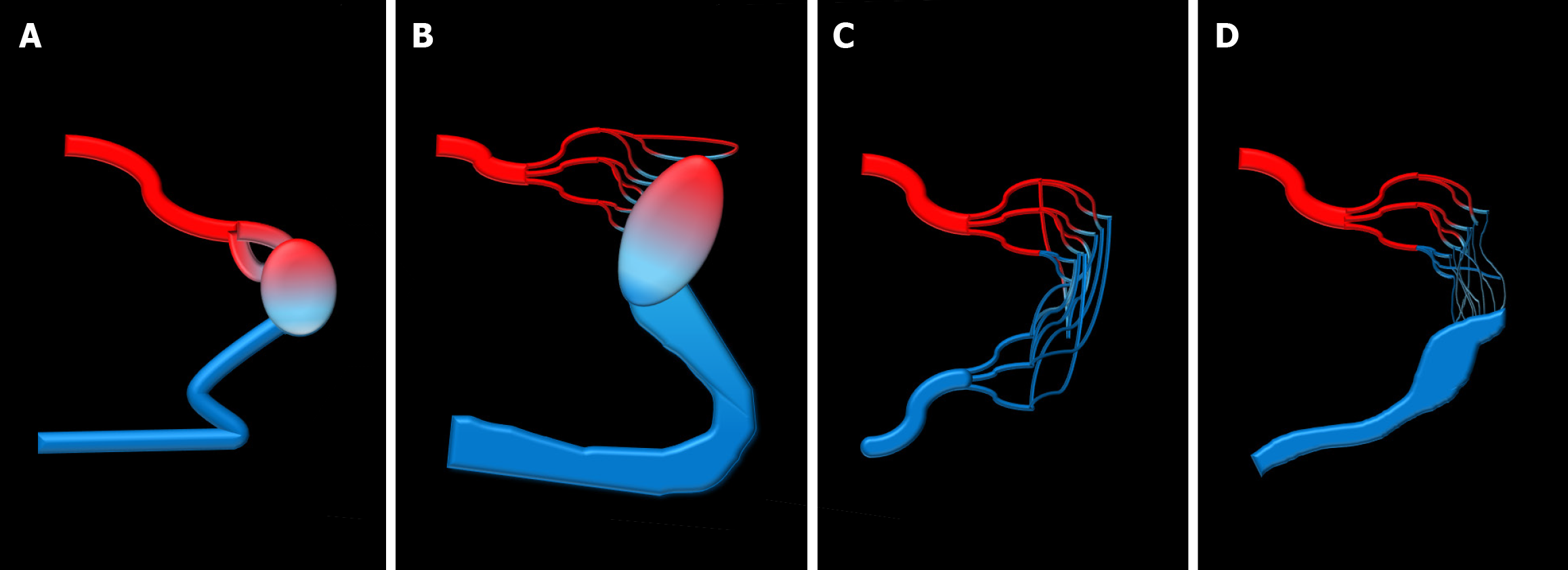
Imaging with CT, using intravenous contrast, demonstrates early enhancement of the ipsilateral renal veins and inferior vena cava in the corticomedullary phase[15]. CT helps in identifying the location and size of the fistula for further treatment planning. The AVF can be classified into type I, II, IIIa, and IIIb based on Cho’s classification (Figure 10 and Table 2)[13]. Digital subtraction angiography is the gold standard technique for diagnosing AVF; however, it is an invasive procedure. With the increased usage of CT, digital subtraction angiography is limited to cases with equivocal CT findings or for therapeutic evaluation. Years ago, surgical ligation of the feeding artery with or without nephrectomy was the preferred treatment of any renal AVF. With the advancements of endovascular techniques, embolization is primarily considered due to the minimally invasive nature, the ability to preserve renal function, and fewer surgical complications/morbidity. Nonetheless, surgical ligation is still preferred in cases of a large, high-flow AVF due to the risk of incomplete embolization and the potential migration of the embolization material[16].
| Type | Description |
| I | Less than four feeding arteries supplying a single dilated vein (arterio-venulous fistula) |
| II | Multiple feeding arterioles supplying a single dilated vein (arterio-venulous fistula) |
| IIIa | Multiple nondilated feeding arterioles supplying multiple nondilated venules (non-dilated arterio-venulous fistula) |
| IIIb | Multiple dilated feeding arterioles supplying multiple dilated venules (dilated arterio-venulous fistula) |
Urolithiasis is a common clinical entity. Patients typically present clinically with flank pain with or without radiation to the groin region, as well as hematuria. Renal tubular acidosis, hypercalciuria, gout, and recurrent urinary tract infections (UTIs) are the most common risk factors associated with the formation of renal calculi. Imaging plays a crucial role in identifying the size and location of calculus, as well as signs of infection.
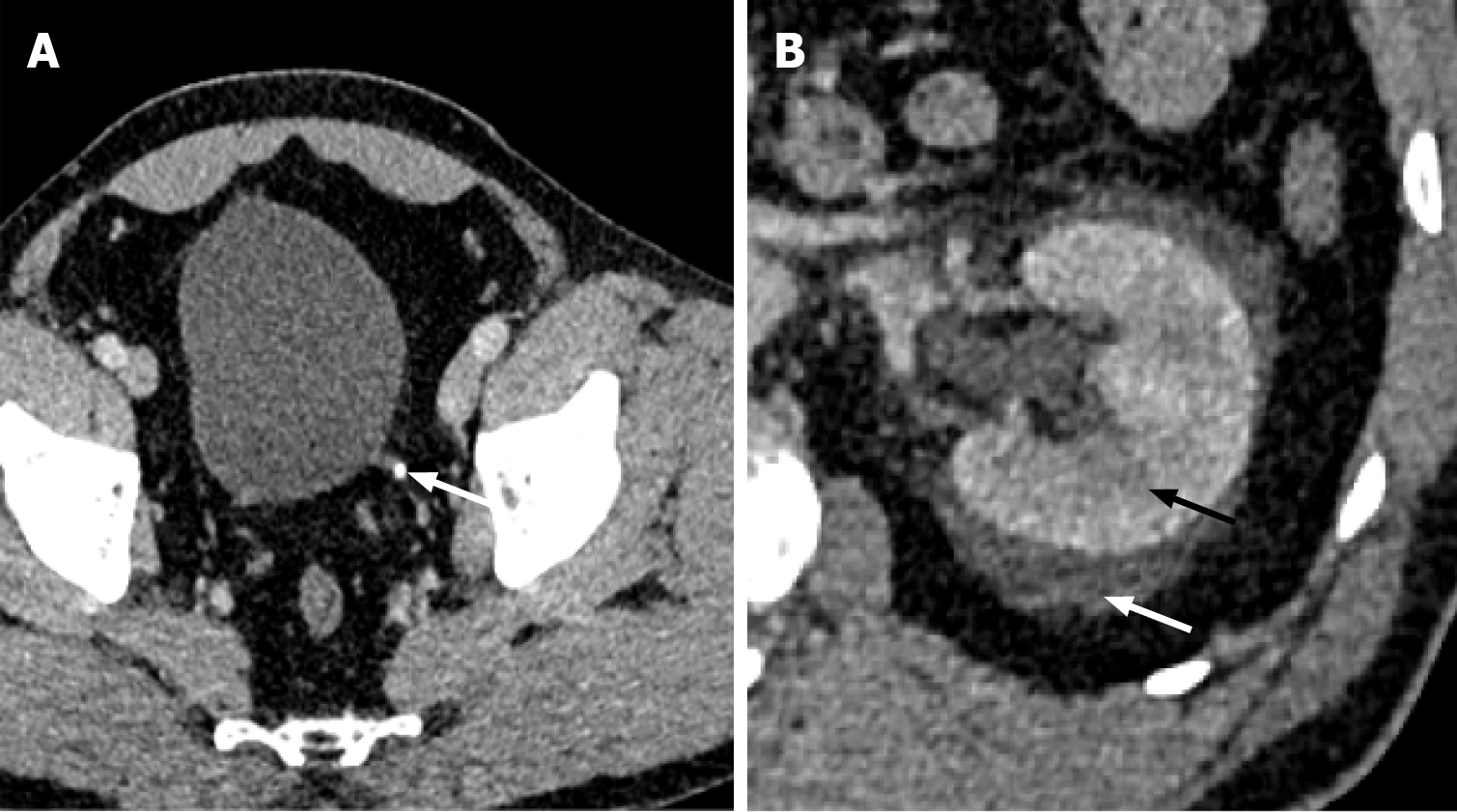
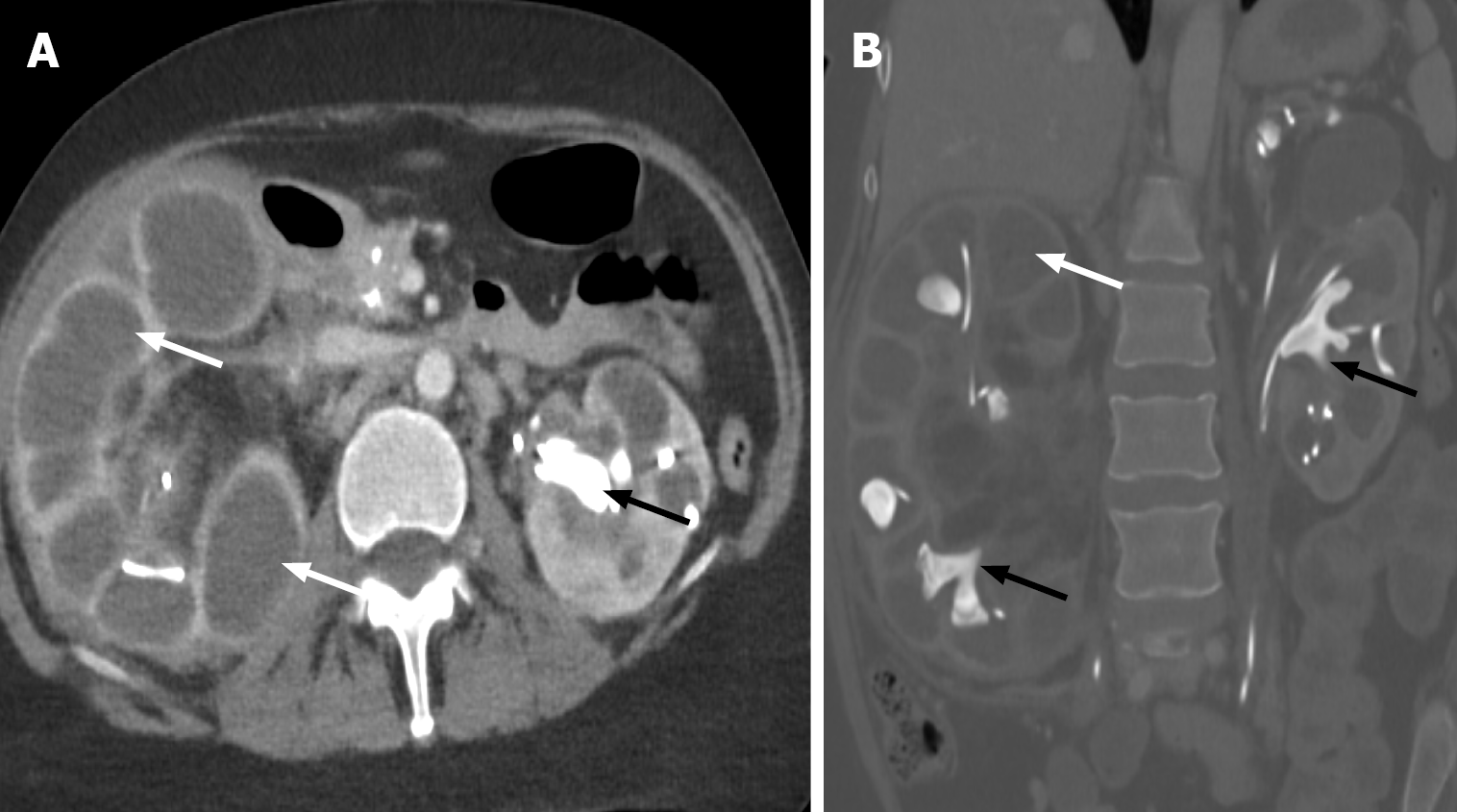
Ultrasound is a readily available imaging modality that shows calculi as an echogenic focus on grayscale ultrasound, with twinkling artifacts on color Doppler ultrasound. In current clinical practice, ultrasound has been replaced by CT; however, it remains the imaging modality of choice in pregnant and pediatric patients due to concerns about ionizing radiation. CT provides additional relevant information, such as the size of the calculus, its location, signs of obstruction or infection, and other potential sources of pain that may not be related to the calculus. Secondary signs of obstruction include hydronephrosis, perinephric edema, unilateral renal enlargement, asymmetric renal parenchymal enhancement, and periureteral edema (Figure 11). Abdominal and pelvic calcifications such as phleboliths are common along the course of the ureters and may be mistaken for ureterolithiasis. The soft-tissue rim sign is indicative of ureterolithiasis and manifests as soft-tissue attenuation surrounding the calculus, resulting from ureteral wall edema. The degree of edema depends on the size of the calculus. On the other hand, phleboliths demonstrate a comet-tail sign, which appears as tapering soft tissue attenuation adjacent to the calcification representing the scarred parent vein[17].
Management of urolithiasis depends on the symptomatology, stone burden, and degree of obstruction. Treatment options include shockwave lithotripsy, ureteroscopy with lithotripsy, and percutaneous nephrolithotomy. The size and volume of the stone help in determining the mode of treatment. Studies show that a stone volume of > 700 mm3 has a high risk for shock wave lithotripsy failure. Hence, as radiologists, we can aid urologists in deciding on the treatment modality by providing the accurate size and volume of the calculus.
Acute pyelonephritis is a clinical and laboratory diagnosis characterized by fever, chills, leukocytosis, bilateral costovertebral angle tenderness, dysuria, and positive urine cultures. However, in certain patients with inadequate treatment response and who are prone to complications due to pre-existing risk factors such as an immunocompromised state, prior renal surgery, and urolithiasis, imaging plays a crucial role. Additionally, imaging aids in assessing the extent of disease and informs future therapy or interventions.
CT with intravenous contrast, performed in the corticomedullary or early nephrographic phase, is the first-line imaging modality (Figure 12 and Table 3). Bacterial pyelonephritis can be secondary to an ascending UTI or hematogenous spread of infection. Pyelonephritis from ascending UTI can manifest as focal or multifocal wedge-shaped areas of hypoattenuation extending from the renal papilla to the cortex. The hypoattenuation is secondary to congestion from obstructing inflammatory debris in the tubular collecting system, interstitial inflammation, and vasospasm. These imaging findings mimic a renal infarct and can be differentiated by the presence of a cortical rim sign. The cortical rim sign is characterized by relative sparing of the cortex, which can be observed in up to 50% of renal infarcts.
| Pathology | Imaging findings | Management |
| Pyelonephritis | Multifocal wedge-shaped hypoattenuation extending from the renal papilla to cortex. Round hypoattenuation rim-enhancing collections indicate abscess formation | Antibiotics and percutaneous or surgical abscess drainage |
| Emphysematous pyelonephritis | Gas locules within the renal collecting system with or without extension to the renal parenchyma, perinephric space, or pararenal space | Grade I, II: Antibiotics, percutaneous drainage of abscess, and decompression of urinary tract obstruction, if any. Grade III: Immediate nephrectomy and antibiotics. Grade IV: Same as grade I and II; nephrectomy if the initial treatment fails |
| Xanthogranulomatous pyelonephritis | Enlarged kidney with dilated renal calyces and a large staghorn calculus | Nephron-sparing nephrectomy in focal disease and total nephrectomy in diffuse involvement |
| Pyonephrosis | Complex fluid within the renal collecting system with or without internal septations | Urgent decompression with percutaneous or retrograde stent placement. Nephrectomy in advanced cases with irreversible renal damage |
In contrast, in pyelonephritis, the wedge of necrosis extends from the renal pyramids all the way to the renal capsule. There is evidence of delayed contrast excretion from the infected parenchyma; hence, these areas appear hyperattenuating even after 3-6 hours of contrast administration. Pyelonephritis from hematogenous seeding can appear as round lesions with peripheral hypoattenuation. Often, it is not possible to differentiate between ascending and hematogenous pyelonephritis based on imaging findings alone, thereby mandating clinical correlation.
CT helps in evaluating complications such as intraparenchymal or extraparenchymal abscess formation (Figure 12). In early stages, abscesses lack a wall or pseudocapsule and can present as an ill-defined hypoattenuating area. A well-formed abscess is a round, hypoattenuating collection with a peripheral, thick, enhancing rim. The rupture of an abscess may result in extra-renal extension of pyelonephritis complicated by perirenal abscess. Percutaneous or surgical drainage is recommended.
In immunocompromised patients, pyelonephritis can be complicated by severe necrosis of the renal parenchyma, known as emphysematous pyelonephritis (EP) (Figure 13). It is a life-threatening condition caused by organisms such as Escherichia coli, Klebsiella pneumonia, and Proteus mirabilis[18]. If not treated promptly, EP may lead to fulminant sepsis and death[18].
Huang and Tseng classified the imaging findings of EP into four types: (1) Type 1: Gas in the collecting system only, with a good prognosis with medical management; (2) Type II: Gas confined to the renal parenchyma requiring parenteral antibiotics and percutaneous drainage catheter placement. Obstructing etiology must be treated, if any; (3) Type IIIa: Gas within renal parenchyma extending to the perinephric space. Type IIIb: Gas within renal parenchyma extending beyond the perinephric space into the pararenal space. Management of type III includes antibiotics and placement of a percutaneous drainage catheter, or nephrectomy, depending on the risk factors and clinical condition; and (4) Type IV: Gas within bilateral renal parenchyma or within unilateral solitary renal parenchyma. Management includes antibiotics and percutaneous catheter drainage; if these fail, nephrectomy is recommended as the next step in management (Table 3)[19-21].
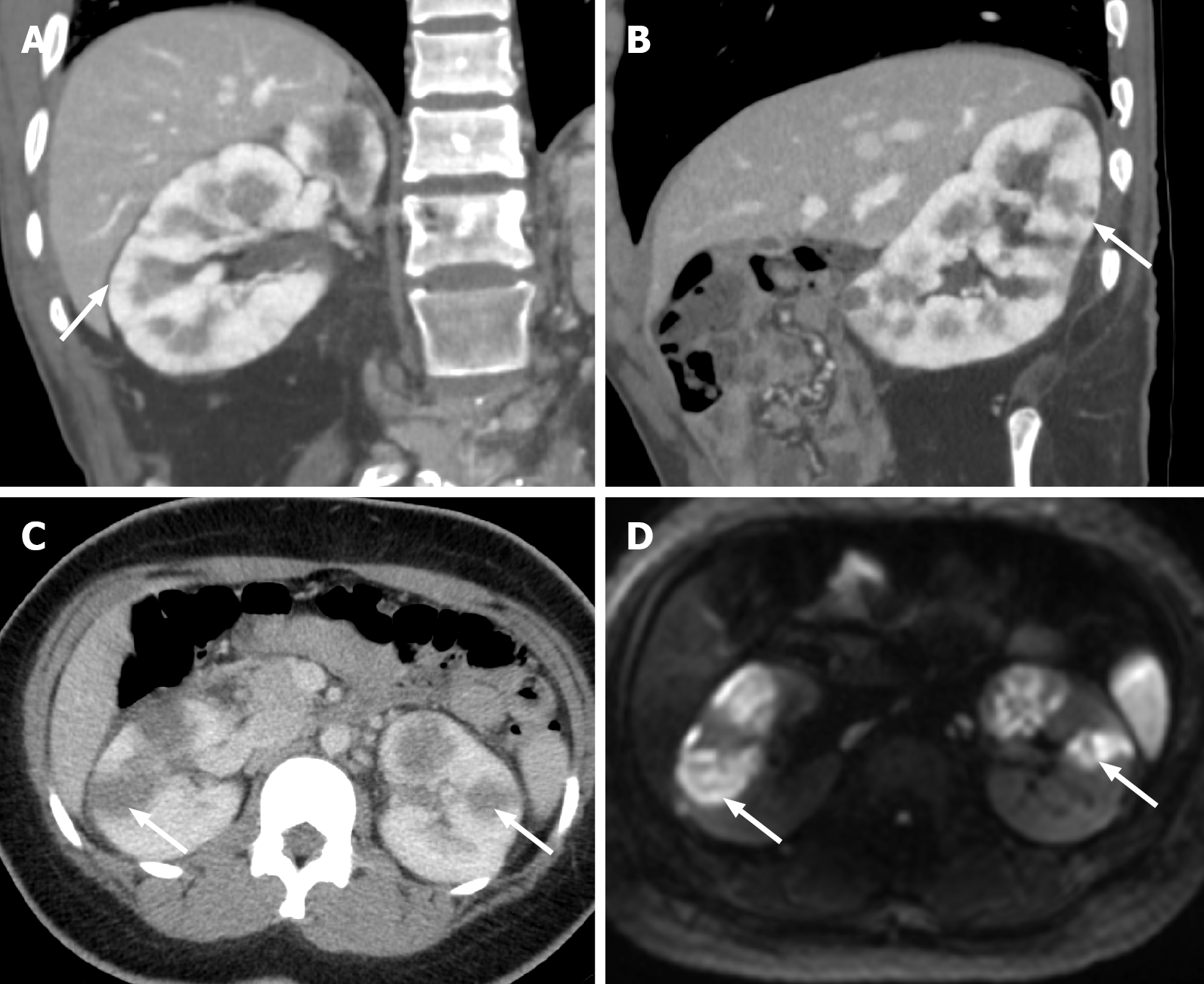
Recurrent UTIs caused by urease-producing organisms, such as Proteus mirabilis and Escherichia coli, lead to urinary alkalinization and increase the risk of struvite stone formation. The struvite stones are composed of calcium, magnesium, phosphate, and ammonium, and have a branching pattern that fills the renal calyces, termed a staghorn calculus[22]. Due to a high risk of recurrent UTI, females are more prone to staghorn calculi formation and obstruction. As part of a cascade of immune responses, the renal parenchyma is replaced with lipid-laden macrophages, known as “xanthoma cells”[23]. CT shows an enlarged kidney with dilated renal calyces, renal cortical thinning, and atrophy, giving the “classic bear paw sign” (Figure 14 and Table 3)[24]. A large staghorn calculus fills and obstructs the renal collecting system. The inflammation may extend to the perirenal and pararenal soft tissues. The involved kidney usually becomes nonfunctional, and nephrectomy is indicated[25].
Pyonephrosis is the infected hydronephrosis with a purulent collection within the renal pelvicalyceal system. Predisposing factors to pyonephrosis include neurogenic bladder, indwelling catheters or drainage devices, calculi, and congenital uropathy. The history of diabetes, renal transplant, renal failure, and other immunocompromised conditions further increases the risk of developing pyonephrosis[26]. It can be associated with destruction of the adjacent renal parenchyma, thereby compromising renal function and increasing the risk for nephrectomy. In addition, intraperitoneal or retroperitoneal rupture of pyonephrosis can provoke the systemic inflammatory response resulting in sepsis, multiorgan dysfunction, and death. Hence, pyonephrosis is considered a urologic emergency, and an immediate decom
Ultrasound is a readily available imaging modality that can show a dilated renal collecting system with echogenic debris and fluid-fluid levels. These findings have a sensitivity, specificity, and accuracy of 90%, 97%, and 96% respectively[18]. Dirty shadowing indicates internal foci of air within the collection. Differentiating between hydronephrosis and pyonephrosis on CT can be challenging. The density of the fluid can help in determining whether it is hydronephrosis or pyonephrosis, with the former being characterized by simple fluid attenuation and the latter by complex fluid attenuation[18]. Additional secondary signs of infection on CT with intravenous contrast include pelvicalyceal wall thickening (greater than 2 mm), wall enhancement, air-fluid or fluid-fluid levels, perinephric fat stranding, and urinary tract obstruction (Figure 15). On the excretory phase, the dense purulent fluid is observed layering in the dependent portions while the excreted contrast material is identified anteriorly[18]. If untreated, the pressure in the collecting system increases, causing rupture into the intraperitoneal and retroperitoneal spaces. The rupture can be further complicated by the formation of a fistula between the kidney and gastrointestinal structures (Table 3)[26].
Renal emergencies may manifest with wide clinical and imaging findings prompting timely recognition crucial for diagnosis and management. CT is the first line imaging modality due to its wide availability and rapid acquisition. Familiarity with the characteristic imaging findings is important for radiology residents in training to enhance diagnostic confidence and improve patient triage. Early diagnosis and treatment reduces the risk of progression to irreversible renal damage, thereby improving morbidity and mortality.
| 1. | Quaia E, Martingano P, Cavallaro M, Premm M, Angileri R. Normal radiological anatomy and anatomical variants of the kidney. In: Radiological imaging of the kidney. Heidelberg: Springer, 2014: 17-74. |
| 2. | Chong ST, Cherry-Bukowiec JR, Willatt JM, Kielar AZ. Renal trauma: imaging evaluation and implications for clinical management. Abdom Radiol (NY). 2016;41:1565-1579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 3. | Stein R, Bashir S, Kee-Sampson J. Interventional radiology in renal emergencies: a pictorial essay. Emerg Radiol. 2022;29:409-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 4. | Chien LC, Vakil M, Nguyen J, Chahine A, Archer-Arroyo K, Hanna TN, Herr KD. The American Association for the Surgery of Trauma Organ Injury Scale 2018 update for computed tomography-based grading of renal trauma: a primer for the emergency radiologist. Emerg Radiol. 2020;27:63-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 5. | Dixe de Oliveira Santo I, Sailer A, Solomon N, Borse R, Cavallo J, Teitelbaum J, Chong S, Roberge EA, Revzin MV. Grading Abdominal Trauma: Changes in and Implications of the Revised 2018 AAST-OIS for the Spleen, Liver, and Kidney. Radiographics. 2023;43:e230040. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 6. | Heller MT, Schnor N. MDCT of renal trauma: correlation to AAST organ injury scale. Clin Imaging. 2014;38:410-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 7. | Gross JA, Lehnert BE, Linnau KF, Voelzke BB, Sandstrom CK. Imaging of Urinary System Trauma. Radiol Clin North Am. 2015;53:773-788, ix. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 8. | Nair AD, Kumar A, Gamanagatti S, Gupta A, Kumar S. CT findings in renovascular injuries following abdominal trauma: a pictorial review. Emerg Radiol. 2022;29:595-604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 9. | Pinto DS, Clode H, Madrazo BL, Paes FM, Alessandrino F. Imaging review of spontaneous renal hemorrhage. Emerg Radiol. 2024;31:515-528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 10. | Shah JN, Gandhi D, Prasad SR, Sandhu PK, Banker H, Molina R, Khan S, Garg T, Katabathina VS. Wunderlich Syndrome: Comprehensive Review of Diagnosis and Management. Radiographics. 2023;43:e220172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 26] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 11. | Huynh AD, Sweet DE, Feldman MK, Remer EM. Imaging of renal emergencies: Review of infectious, hemorrhagic, vascular, and traumatic etiologies. Br J Radiol. 2022;95:20211151. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 12. | White RD, Moore KS, Salahia MG, Thomas WR, Gordon AC, Williams IM, Wood AM, Zealley IA. Renal Arteries Revisited: Anatomy, Pathologic Entities, and Implications for Endovascular Management. Radiographics. 2021;41:909-928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 13. | Maruno M, Kiyosue H, Tanoue S, Hongo N, Matsumoto S, Mori H, Sagara Y, Kashiwagi J. Renal Arteriovenous Shunts: Clinical Features, Imaging Appearance, and Transcatheter Embolization Based on Angioarchitecture. Radiographics. 2016;36:580-595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 54] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 14. | Leckie A, Tao MJ, Narayanasamy S, Khalili K, Schieda N, Krishna S. The Renal Vasculature: What the Radiologist Needs to Know. Radiographics. 2022;42:E80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 15. | Chaurasia A, Singh S, Homayounieh F, Gopal N, Jones EC, Linehan WM, Shyn PB, Ball MW, Malayeri AA. Complications after Nephron-sparing Interventions for Renal Tumors: Imaging Findings and Management. Radiographics. 2023;43:e220196. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 16. | Duc VT, Duong N, Phong NT, Nam NH, Quoc DA, Cuong T, Huy NH, Duy TL, Chien PC. Large renal arteriovenous fistula treated by embolization: a case report. Radiol Case Rep. 2021;16:2289-2294. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 17. | Kambadakone AR, Eisner BH, Catalano OA, Sahani DV. New and evolving concepts in the imaging and management of urolithiasis: urologists' perspective. Radiographics. 2010;30:603-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 102] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 18. | Craig WD, Wagner BJ, Travis MD. Pyelonephritis: radiologic-pathologic review. Radiographics. 2008;28:255-77; quiz 327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 380] [Cited by in RCA: 229] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 19. | Kim N, Machnicki S. Emphysematous pyelonephritis. Appl Radiol. 2015;44. |
| 20. | Huang JJ, Tseng CC. Emphysematous pyelonephritis: clinicoradiological classification, management, prognosis, and pathogenesis. Arch Intern Med. 2000;160:797-805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 575] [Cited by in RCA: 456] [Article Influence: 17.5] [Reference Citation Analysis (2)] |
| 21. | Sureka B, Thukral BB. Emphysematous infections of the urinary tract: A radiological perspective. Indian J Nephrol. 2012;22:395-397. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Black JC, Dyer RB. The "staghorn" calculus. Abdom Imaging. 2015;40:2908-2909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 23. | Demertzis J, Menias CO. State of the art: imaging of renal infections. Emerg Radiol. 2007;14:13-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 51] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 24. | El-Ghar MA, Farg H, Sharaf DE, El-Diasty T. CT and MRI in Urinary Tract Infections: A Spectrum of Different Imaging Findings. Medicina (Kaunas). 2021;57:32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 25. | Vernuccio F, Patti D, Cannella R, Salvaggio G, Midiri M. CT imaging of acute and chronic pyelonephritis: a practical guide for emergency radiologists. Emerg Radiol. 2020;27:561-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 26. | Tamburrini S, Lugarà M, Iannuzzi M, Cesaro E, De Simone F, Del Biondo D, Toto R, Iulia D, Marrone V, Faella P, Liguori C, Marano I. Pyonephrosis Ultrasound and Computed Tomography Features: A Pictorial Review. Diagnostics (Basel). 2021;11:331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |