Published online Mar 25, 2026. doi: 10.5527/wjn.v15.i1.114142
Revised: October 27, 2025
Accepted: January 8, 2026
Published online: March 25, 2026
Processing time: 183 Days and 8.5 Hours
Non-steroidal anti-inflammatory drugs (NSAIDs) are widely used for pain management, but they can cause nephrotoxicity, particularly in patients with existing renal impairment. Cerium oxide (CeO2), an antioxidant nanoparticle, may provide protection against renal damage caused by NSAIDs.
To assess the protective effects of CeO2 against ibuprofen-induced renal injury in a rat model of unilateral ureteral obstruction (UUO).
Thirty male Wistar albino rats, weighing between 300 g and 425 g, were randomly divided into five groups (n = 6 each): Control (sham), UUO only, UUO + ibupro
In the right (non-obstructed) kidneys, the UUO + CeO2 group exhibited significantly lower levels of GV, tubular hyaline casts, and tubular desquamation compared to the UUO + ibuprofen group (P < 0.05). In the left (obstructed) kidneys, the CeO2-treated group also exhibited significant reductions in GV, vascular hypertrophy, and tubular hyaline casts (P < 0.05). Furthermore, malondialdehyde levels were markedly decreased in the CeO2 groups, whereas nitric oxide levels were elevated in the ibuprofen group. Blood urea nitrogen and creatinine levels increased across all UUO groups, but no significant differences were observed between the treatment groups.
CeO2 attenuates ibuprofen-induced renal injury in rats with UUO, likely through antioxidative mechanisms. These findings suggest that CeO2 could serve as a potential nephroprotective adjunct during NSAID therapy in patients with obstructive renal conditions.
Core Tip: This experimental study explores the protective effects of cerium oxide (CeO2), a powerful antioxidant nanoparticle, against renal injury induced by ibuprofen in rats with unilateral ureteral obstruction. Histopathological and biochemical analyses demonstrated that CeO2 significantly reduced tubular degeneration, glomerular vacuolization, and oxidative stress markers in both the obstructed and contralateral kidneys. These results indicate that CeO2 may function as a nephroprotective adjunct during non-steroidal anti-inflammatory drug therapy, potentially mitigating renal damage associated with obstructive uropathy.
- Citation: Toruk S, Babacan A, Arslan M, Şıvgın V, Sezen ŞC, Boyunağa H. Protective effects of cerium oxide on ibuprofen-induced renal injury in rats with ureteral obstruction. World J Nephrol 2026; 15(1): 114142
- URL: https://www.wjgnet.com/2220-6124/full/v15/i1/114142.htm
- DOI: https://dx.doi.org/10.5527/wjn.v15.i1.114142
Renal failure is characterized by the kidney’s inability to maintain fluid-electrolyte balance, as well as metabolic and endocrine functions, due to a decrease in glomerular filtration. Given its complex pathophysiology and treatment challenges, renal failure is a clinically significant condition that leads to substantial morbidity and mortality. It is categorized into two types: Acute and chronic. Acute renal failure is characterized by the rapid accumulation of nitrogenous waste products such as urea and creatinine due to the rapid deterioration of kidney function over hours or days. Conversely, chronic renal failure involves a progressive loss of kidney function that occurs over weeks to months. Kidney diseases rank among the leading causes of morbidity and mortality worldwide. Chronic kidney disease (CKD), which is increasingly recognized in clinical settings, poses a significant global health issue. Estimates in 2014 indicated that over 10% of adults in the United States had CKD, while an even larger percentage experienced acute kidney injury and a smaller group presented with severe renal dysfunction[1].
In the perioperative setting, the primary objectives of pain management are to alleviate pain, facilitate early mobilization, shorten hospital stays, and improve patient satisfaction. To achieve these objectives, a wide variety of analgesic agents are commonly used. However, concerns about potential adverse effects, particularly renal complications, may result in inadequate pain management in the postoperative period[2]. Although many commonly used drugs in clinical practice have a relatively low incidence of nephrotoxicity, administering high doses of nephrotoxic agents to elderly or asymptomatic patients, whose impaired renal function may go unrecognized, can result in renal injury[3].
Non-steroidal anti-inflammatory drugs (NSAIDs) are among the most frequently utilized agents in pain management. By inhibiting the cyclooxygenase (COX) enzyme, NSAIDs suppress renal prostaglandin synthesis, which subsequently reduces levels of prostaglandin I2 and prostaglandin E2. NSAID metabolites are excreted through the kidneys, and their administration may increase renal vasoconstrictors, thereby decreasing renal blood flow and the glomerular filtration rate. Consequently, administration of NSAIDs may result in edema, electrolyte imbalances, and both acute renal failure and chronic renal failure. Clinically, renal complications develop in approximately 1% of patients treated with NSAIDs. However, research on the acute renal effects of short-term postoperative parenteral NSAID use remains limited[4,5]. NSAIDs are classified based on their inhibitory effects on COX isoenzymes into selective COX-1 inhibitors (e.g., low-dose acetylsalicylic acid), non-selective COX inhibitors (e.g., indomethacin), selective COX-2 inhibitors (e.g., meloxicam, etodolac), and highly selective COX-2 inhibitors (e.g., celecoxib, rofecoxib). COX-1 is present in many cells and is involved in hemostasis and platelet aggregation, whereas COX-2 is typically absent in most tissues and is induced during inflammation by various mediators such as bacterial lipopolysaccharides, interleukin-1, tumor necrosis factor-alpha, interferon, epidermal growth factor, and platelet-derived growth factor[6,7].
Ibuprofen, a derivative of phenylpropionic acid, is a widely used NSAID known for its analgesic, anti-inflammatory, and antipyretic properties. It is available in various formulations, including tablets, oral suspension, capsules, drops, and intravenous forms[8]. Ibuprofen is frequently prescribed for mild to moderate pain relief. It works by reversibly inhibiting both COX-1 and COX-2 enzymes, thereby blocking the synthesis of prostaglandins from arachidonic acid. Its analgesic effects are primarily attributed to the inhibition of prostaglandin E2, which, along with prostaglandin I2, plays a critical role in inflammation, particularly in the development of edema and leukocyte migration[9].
Recently, nanoparticle-based agents have attracted attention for their therapeutic potential in diseases associated with oxidative stress, such as neurodegenerative, cardiovascular, and inflammatory disorders, as well as cancer. Beyond their biomedical applications, these nanoparticles are also utilized in solar cells, fuel cells, gas sensors, and oxygen pumps[10,11]. Cerium oxide (CeO2) is a therapeutic antioxidant nanoparticle with a wide range of potential applications. Distinctive for its antioxidant properties, CeO2 serves as an efficient free radical scavenger and is considered suitable for medical applications. It is one of five nanoparticles currently under investigation by the United States Environmental Protection Agency for potential therapeutic use[12,13]. In this study, we aimed to investigate the effects of CeO2, an antioxidant nanoparticle, on renal tissue following ibuprofen administration in a rat model of renal failure induced by unilateral ureteral obstruction (UUO).
A total of 30 male Wistar Albino rats, aged four months and weighing 300-425 g, were included in the study. The rats were housed under controlled conditions (20-21 °C, 12 hours light-dark cycle) and provided food ad libitum until 2 hours before anesthesia. They were randomly assigned to five experimental groups (n = 6 per group) as follows: (1) Control (group C) - sham operation; (2) UUO (group UO) - UUO; (3) UUO + ibuprofen (group UOI); (4) UUO + CeO2 (group UOCO); and (5) UUO + ibuprofen + CeO2 (group UOICO).
Anesthesia was administered intramuscularly using ketamine hydrochloride (50 mg/kg) and xylazine hydrochloride (10 mg/kg). Except for the control group, all rats underwent left distal ureter ligation using 2.0 Mersilene suture through a lower abdominal incision to induce UUO. The control group received a sham operation without ureter ligation.
Postoperative analgesia was maintained using subcutaneous morphine sulfate (0.5 mg/kg) every 8 hours for a total of six doses. The treatment was administered according to group assignment as follows: Group UOI received 30 mg/kg/day ibuprofen intraperitoneally for 21 consecutive days. Group UOCO received 0.5 mg/kg/day CeO2 intraperitoneally for 21 days. Group UOICO received both agents sequentially at the same doses and duration. Twenty-four hours after treatment, the animals were euthanized.
Euthanasia was performed under anesthesia by intracardiac blood collection. Both kidneys were carefully removed to preserve structural integrity. The tissues were fixed in 10% formalin for histological analysis. Blood samples were used to measure blood urea nitrogen (BUN), creatinine, malondialdehyde (MDA), and nitric oxide (NO) levels.
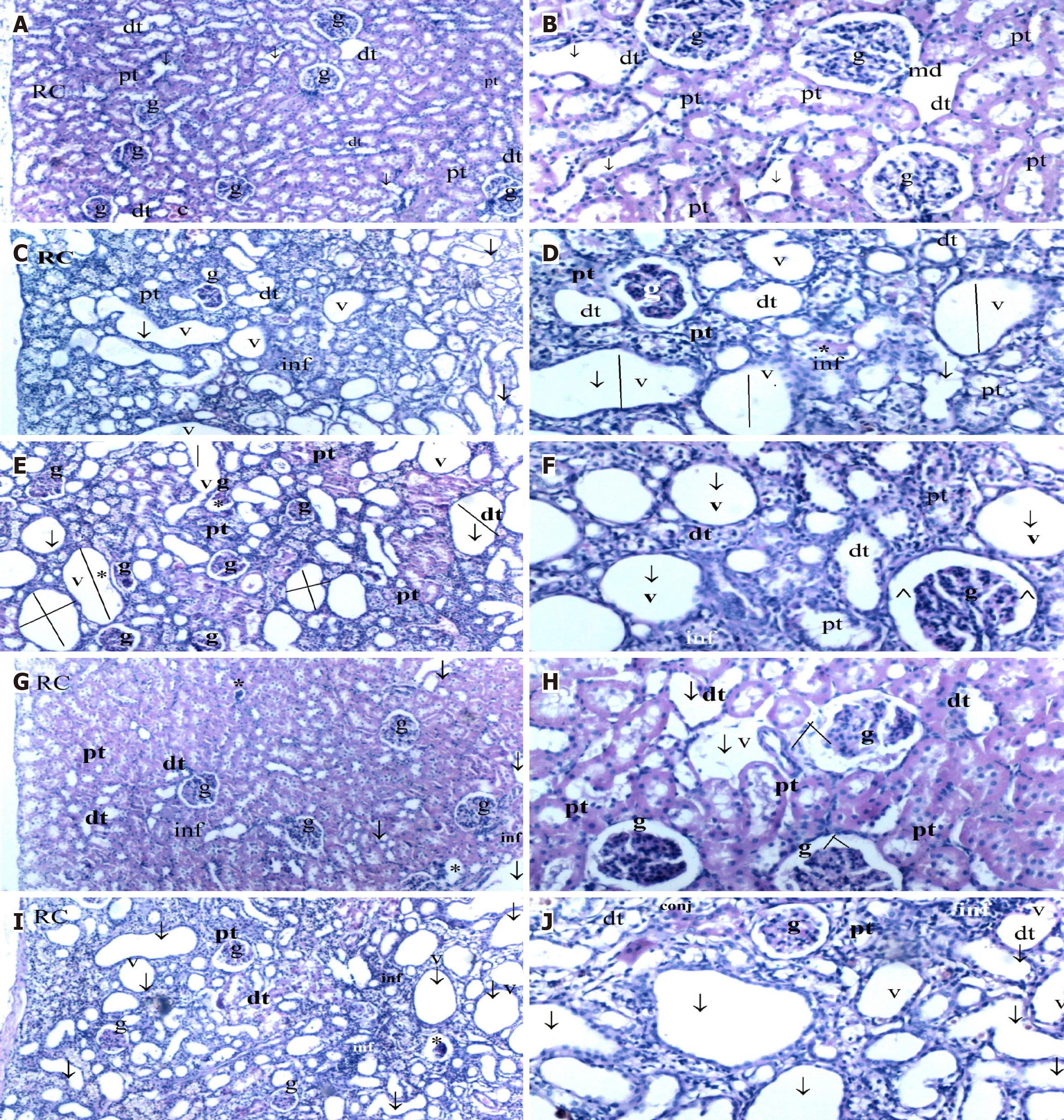
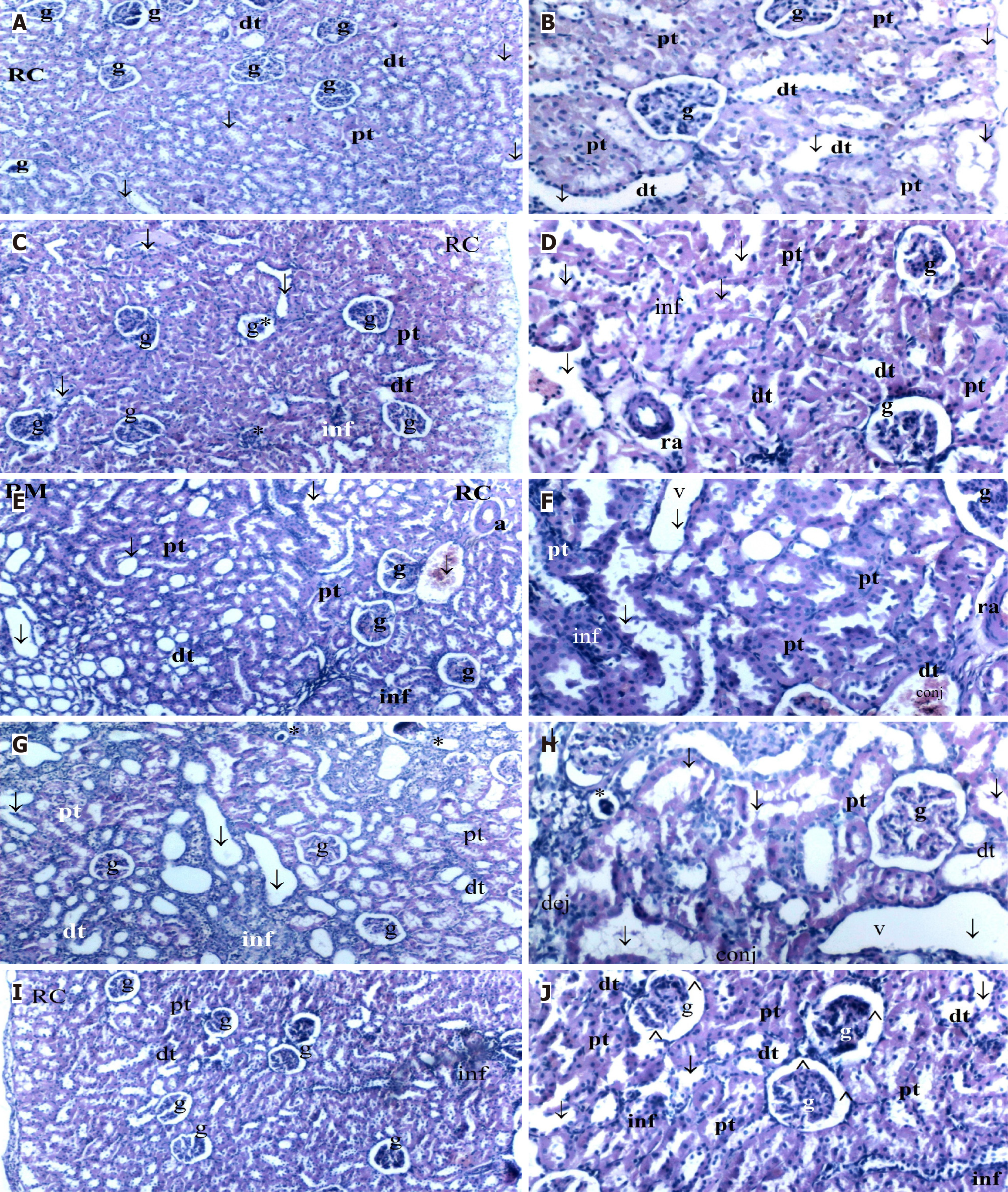
Tissue processing and histological analyses were conducted at the Department of Histology and Embryology, Faculty of Medicine, Kırıkkale University. Kidney samples were embedded in paraffin blocks, sectioned at 5 μm, and stained with hematoxylin and eosin. A blinded pathologist assessed the following parameters using a semi-quantitative 4-point scoring system (0: None, 1: Mild, 2: Moderate, and 3: Severe): Glomerular vacuolization (GV), tubular dilation (TD), vascular vacuolization and hypertrophy (VVH), tubular cell degeneration and necrosis (THDN), Bowman’s space dilation (BSD), hyaline cast formation, lymphocyte infiltration (LI), and tubular cell desquamation (TCD)[14].
Biochemical analyses were performed at the Department of Medical Biochemistry, Faculty of Medicine, Kırıkkale University. Serum samples were stored at -20 °C until analysis.
MDA measurement: Lipid peroxidation was quantified using the Esterbauer method. MDA reacted with thiobarbituric acid at 90-95 °C to form a pink chromogen, which was measured spectrophotometrically at 532 nm.
NO measurement: NO levels were determined based on the concentration of stable oxidative metabolites (NO2-and NO3-) using the Griess reaction.
Statistical analyses were conducted using SPSS for Windows, version 22.0. Data distribution was evaluated using visual (histograms and probability plots) and analytical (Kolmogorov-Smirnov/Shapiro-Wilk) methods. Normally distributed variables were expressed as mean ± SD, whereas non-normally distributed data were expressed as median and interquartile range. Categorical variables were presented as n (%). The χ2 test was used for categorical comparisons. The Kruskal-Wallis test was applied to compare non-normally distributed numerical variables among three or more groups, followed by the Mann-Whitney U test with Bonferroni correction for pairwise comparisons. P < 0.05 was considered statistically significant.
The results are expressed as mean ± SEM. Intergroup comparisons were performed using the Kruskal-Wallis test followed by post hoc pairwise analyses. Significant differences were observed among groups for multiple histopathological parameters, oxidative stress markers, and renal function indicators (Tables 1, 2, 3, and 4, Figures 1 and 2).
| Group C (n = 6) | Group UO (n = 6) | Group UOI (n = 6) | Group UOCO (n = 6) | Group UOICO (n = 6) | 1P value | |
| Glomerular vacuolization | 0.17 ± 0.17 | 1.00 ± 0.26a | 1.17 ± 0.17a | 0.50 ± 0.22b | 0.83 ± 0.17a | 0.012 |
| Tubular dilatation | 0.17 ± 0.17 | 1.17 ± 0.31a | 1.00 ± 0.00a | 0.50 ± 0.22 | 0.83 ± 0.31 | 0.037 |
| Vascular vacuolization and hypertrophy | 0.33 ± 0.21 | 1.00 ± 0.26 | 0.83 ± 0.17 | 0.50 ± 0.22 | 0.83 ± 0.31 | 0.289 |
| Tubular cell degeneration and necrosis | 0.33 ± 0.21 | 0.83 ± 0.17 | 0.83 ± 0.17 | 0.50 ± 0.22 | 0.67 ± 0.21 | 0.330 |
| Bowman’s space dilatation | 0.33 ± 0.21 | 0.83 ± 0.31 | 1.33 ± 0.21 | 0.83 ± 0.31 | 1.00 ± 0.37 | 0.209 |
| Tubular hyaline casts | 0.17 ± 0.17 | 0.83 ± 0.17 | 1.33 ± 0.33a | 0.50 ± 0.22b | 0.67 ± 0.21 | 0.020 |
| Lymphocyte infiltration | 0.33 ± 0.21 | 1.00 ± 0.37 | 1.33 ± 0.21 | 0.67 ± 0.21 | 0.83 ± 0.17 | 0.081 |
| Tubular cell desquamation | 0.33 ± 0.21 | 0.83 ± 0.31 | 1.33 ± 0.21a | 0.50 ± 0.22b | 0.83 ± 0.17 | 0.047 |
| Group C (n = 6) | Group UO (n = 6) | Group UOI (n = 6) | Group UOCO (n = 6) | Group UOICO (n = 6) | 1P value | |
| Glomerular vacuolization | 0.17 ± 0.17 | 1.33 ± 0.21a | 1.50 ± 0.34a | 0.67 ± 0.21b | 1.00 ± 0.26a | 0.005 |
| Tubular dilatation | 0.33 ± 0.21 | 1.67 ± 0.33a | 1.83 ± 0.40a | 1.00 ± 0.26 | 1.67 ± 0.33a | 0.012 |
| Vascular vacuolization and hypertrophy | 0.33 ± 0.21 | 1.33 ± 0.21a | 1.83 ± 0.40a | 0.83 ± 0.17b | 1.17 ± 0.40 | 0.019 |
| Tubular cell degeneration and necrosis | 0.33 ± 0.21 | 1.33 ± 0.21a | 2.00 ± 0.37a | 1.17 ± 0.40 | 1.17 ± 0.31 | 0.017 |
| Bowman’s space dilatation | 0.33 ± 0.21 | 1.67 ± 0.33a | 1.83 ± 0.31a | 1.17 ± 0.31 | 1.50 ± 0.43a | 0.025 |
| Tubular hyaline casts | 0.33 ± 0.21 | 1.33 ± 0.33a | 2.00 ± 0.36a | 0.83 ± 0.31b | 1.17 ± 0.17 | 0.006 |
| Lymphocyte infiltration | 0.33 ± 0.21 | 1.17 ± 0.31 | 1.83 ± 0.40a | 1.17 ± 0.31 | 1.00 ± 0.26 | 0.033 |
| Tubular cell desquamation | 0.17 ± 0.17 | 1.00 ± 0.37a | 2.00 ± 0.45a | 0.83 ± 0.17b | 1.00 ± 0.00a | 0.002 |
Right kidney (non-obstructed side): Histopathological analysis of the right kidneys revealed significant intergroup differences in several parameters (Table 1, Figure 2). GV scores differed significantly among the groups (P = 0.012), being higher in the UO, UOI, and UOICO groups and lower in the UOCO group compared with UOI (P < 0.05). Tubular hyaline casts (THC) formation differed significantly among the groups (P = 0.020), showing an increase in the UOI group and a reduction in the UOCO group compared with UOI (P < 0.05). TCD varied significantly among the groups (P = 0.047), showing an increase in the UOI group and a decrease in the UOCO group compared with UOI (P < 0.05).
Left kidney (obstructed side): Histopathological evaluation of the left kidneys (with ureteral obstruction) also revealed significant intergroup differences (Table 2, Figure 1). GV scores differed significantly among the groups (P = 0.005), with higher values in the UO, UOI, and UOICO groups and lower in the UOCO group compared with UOI (P < 0.05). VVH differed significantly among the groups (P = 0.019), showing an increase in the UO and UOI groups and a reduction in the UOCO group compared with UOI (P < 0.05).
THDN varied significantly among the groups (P = 0.017), with higher scores in the UO and UOI groups compared with controls. BSD differed significantly among the groups (P = 0.025), showing higher scores in the UO, UOI, and UOICO groups compared with controls. THC formation differed significantly among the groups (P = 0.006), being elevated in the UO and UOI groups and reduced in the UOICO group compared with UOI. LI differed significantly among the groups (P = 0.033), showing increased infiltration in the UOI group compared with controls. TCD scores differed significantly among the groups (P = 0.002), being elevated in the UO, UOI, and UOICO groups and reduced in the UOCO group compared with UOI.
Serum MDA and NO levels differed significantly among the groups (P < 0.001). Both markers were elevated in all experimental groups compared with controls, whereas the UOCO group showed lower values than the UOI group (Table 3).
Serum urea and creatinine levels showed significant intergroup differences (P < 0.001). Both parameters were elevated in all experimental groups compared with controls, confirming renal impairment. Detailed statistical results are presented in Table 4. Pathological findings include TD, glomerular and tubular degeneration, inflammatory infiltration, and BSD across different treatment groups.
In this experimental study, the effects of CeO2, an antioxidant nanoparticle, on renal tissue were investigated following ibuprofen administration in rats with UUO-induced renal failure. Ibuprofen administration led to alterations in serum BUN, creatinine, NO, and MDA levels, as well as histopathological changes in tubular and glomerular structures of both obstructed and contralateral kidneys.
Previous studies have demonstrated that ibuprofen induces glomerular and tubular alterations in the kidney[15,16]. In a study by Kent et al[15], neonatal rats were administered indomethacin, ibuprofen, or gentamicin, and renal changes were evaluated using electron and light microscopy after two weeks. In the indomethacin and ibuprofen groups, light microscopy revealed vacuolization of proximal tubular epithelial cells, interstitial edema, and intratubular protein accumulation, and electron microscopy demonstrated glomerular enlargement and irregularities in the glomerular basement membrane[15]. Similarly, in another study using Sprague-Dawley rats, antenatal ibuprofen administration for 15 days postnatally resulted in glomerular hypoplasia[17]. Consistent with the findings of Kent et al[15], our histopathological examination of the contralateral kidney revealed glomerular and tubular changes, supporting the potential nephrotoxic effects of ibuprofen. Based on this evidence, we aimed to evaluate the possible protective effects of CeO2 against ibuprofen-induced renal impairment.
CeO2 is a potent free radical scavenger with established antioxidant properties suitable for medical use. It is one of five nanoparticles currently under investigation by the United States Environmental Protection Agency[18]. In our study, histopathological evaluation of the contralateral kidney revealed significantly higher TD in the UO and UOI groups compared with controls. By contrast, the CeO2-treated group (UOCO) showed significantly lower GV, TCD, and THC formation compared with the UOI group. This reduction may be attributed to the antioxidant activity of CeO2; however, larger-scale studies are required to determine its clinical significance.
Untreated urinary obstruction leads to tubular atrophy, interstitial fibrosis, and inflammation, eventually resulting in irreversible renal damage[19,20]. In cases of complete obstruction, the obstructed kidney rapidly ceases urine production, and acute renal injury develops. Continuation of obstruction for one to two weeks produces histopathological features of CKD[21]. UUO remains one of the most widely used experimental models for studying renal failure and testing potential therapeutic agents. In our study, histopathological evaluation of the obstructed kidney revealed significantly lower GV, VVH, and THC scores in the UOCO group compared with the UOI group. The milder severity of these alterations in the CeO2-treated group supports its protective antioxidant effect. However, no significant improvements were observed in the UOICO group, indicating that further comprehensive studies are needed to clarify the mechanisms of ibuprofen-induced damage and the potential clinical benefits of CeO2.
Ucero et al[22] reported that tubular cell death exceeds proliferation in early acute kidney injury and CKD, leading to tubular cell loss and chronic tubular atrophy. Histological evaluation two weeks after UUO showed tubular dilatation and apoptosis in distal tubules and collecting ducts across both cortical and medullary regions. Similarly, Grande and López-Novoa[23] demonstrated that mild fibrosis appeared by the end of the first week and became more pronounced by the second week in rat models of experimental renal failure. Xu et al[24] observed tubular epithelial desquamation, dilatation, and atrophy in rats with UUO. Li et al[25] also reported interstitial fibrosis, cellular infiltration, tubular ectasia, atrophy, necrosis, and epithelial degeneration. In our study, THC was observed less frequently in the UO and UOI groups compared with controls, whereas TD, BSD, TCD, and THDN were more frequent. These findings are consistent with those in the literature. The absence of significant TD changes in the UOCO group compared with the control group may reflect the antioxidant properties of CeO2. Larger studies evaluating varying CeO2 doses are required to confirm these findings and determine their clinical importance.
LI is another important indicator of renal injury. In our study, LI was more pronounced in the UOI group compared with controls. Previous research has shown that cytokines, chemokines, and adhesion molecules released by injured renal and endothelial cells recruit leukocytes to the peritubular interstitium, which enhances inflammation and promotes fibrosis and tubular cell damage. Interstitial leukocyte infiltration begins as early as 12 hours after obstruction and increases progressively until day 14[26,27]. The absence of significant LI in the CeO2-treated groups suggests that CeO2 may exert anti-inflammatory effects via its antioxidant properties.
Oxidative stress is considered a crucial mechanism underlying renal injury in UUO. Continuous oxidative stress is induced by stretching, stress, and local hypoxia within the renal interstitium[28]. Overproduction of reactive oxygen species (ROS) contributes to the progression of renal fibrosis[29]. ROS promote nephropathy by inducing phenotypic changes, collagen synthesis, cell proliferation, and interstitial cell accumulation[30]. They also contribute to lipid peroxidation, leukocyte activation, DNA damage, protein oxidation, apoptosis, and overall tissue damage[31]. CeO2 has been demonstrated to scavenge ROS and to suppress inflammatory mediators in experimental models of intra-abdominal infection, thereby alleviating acute kidney injury[32]. In addition, CeO2 has been demonstrated to enhance liver regeneration following partial hepatectomy and reduce early acetaminophen-induced liver injury in rats[33].
MDA, formed by oxidation of polyunsaturated fatty acids in cell membranes, is an important marker of lipid peroxidation, although it lacks specificity[34]. In our study, serum MDA levels were significantly elevated in all experimental groups compared with controls but were markedly lower in the UOCO group compared with the UOI group. Zhou et al[35] reported similar findings, showing progressively increased oxidative stress, accompanied by reduced antioxidant activity and elevated MDA levels, after four weeks of UUO, thereby indicating lipid peroxidation. Similarly, Grande and López-Novoa[23] observed increased MDA levels in rats with experimental renal failure. The lower MDA levels in our UOCO group may be attributed to the antioxidant effects of CeO2.
NO, an uncharged diatomic free radical gas, is a rapid intracellular signaling molecule. It exists in three isoforms of NO synthase (NOS): Endothelial NOS, neuronal NOS, and inducible NOS (iNOS). iNOS is present in nearly all nucleated mammalian cells and is induced by immunologic stimuli. NO regulates renal physiology, including renal blood flow, autoregulation, tubuloglomerular feedback, renin release, natriuresis, and tubular function. However, NO may react with ROS to form peroxynitrite, a highly cytotoxic molecule that damages lipids, DNA, and proteins[36]. In inflammation-related pathologies, excessive NO production by iNOS contributes to tissue injury. Following UUO, iNOS and endothelial NOS expression increase around days 7-10 and continue to increase until day 21[37]. In our study, NO enzyme activity was significantly increased in all experimental groups compared with controls, as expected in renal injury. However, NO activity was significantly lower in the UOCO group compared with the UOI group, suggesting a protective effect of CeO2. Clinical studies have similarly shown elevated NO levels in hemodialysis patients, where NO acts as a cytotoxic mediator contributing to dialysis complications; NO, alongside creatinine, may serve as prognostic markers in CKD. Kiliç et al[38] demonstrated that the effects of NO vary depending on the balance between its beneficial hemodynamic properties and its cytotoxic effects on renal and extrarenal tissues. In another study, NO enzyme activity was found to be decreased in ureteral obstruction models compared with controls, which aligns with our findings[37]. Collectively, the increased NO activity observed in our study supports existing literature, and the lower activity in the UOCO group suggests that CeO2 may have a protective role. However, further studies are warranted to confirm its clinical relevance.
Serum creatinine remains a standard marker of renal function, although significant increases typically occur only after glomerular filtration rate falls below 50 mL/minute/m2. Its levels are also influenced by factors such as age, sex, muscle mass, and protein intake[39]. In our study, serum BUN and creatinine levels were significantly elevated in all experimental groups compared with controls, which is consistent with previous findings in UUO models. Similarly, Li et al[25] reported increased serum creatinine and BUN levels in rats with UUO, reflecting impaired renal excretion. These elevations confirm renal impairment, while the reduction in the CeO2-treated group suggests possible nephroprotection. Future studies incorporating varied dosing regimens and larger sample sizes are required to validate these findings.
Finally, in addition to the biochemical and histopathological parameters evaluated in the present study, future investigations should include molecular analyses such as polymerase chain reaction and Western blotting to further clarify the underlying antioxidant and anti-inflammatory mechanisms of CeO2. These methods can provide valuable insights into gene and protein expression pathways affected by CeO2 and its potential regulatory roles in oxidative stress and inflammation.
In conclusion, the increasing prevalence of kidney diseases underscores the importance of understanding the renal effects of NSAIDs, widely used medications with known nephrotoxic potential. In this experimental study, ibuprofen administration caused tubular and glomerular alterations in both obstructed and non-obstructed renal tissues of rats. CeO2, a potent antioxidant, significantly attenuated the histopathological and biochemical changes observed in the ibuprofen-treated groups. These results suggest that CeO2 exerts a protective effect against ibuprofen-induced renal injury.
To the best of our knowledge, this is the first experimental study to investigate the potential renoprotective effects of CeO2 against NSAID-induced nephrotoxicity. Future studies should explore the influence of factors such as age, sex, and repeated NSAID exposure on oxidative stress biomarkers to further elucidate and validate the therapeutic potential of CeO2 in renal protection.
| 1. | Coresh J, Selvin E, Stevens LA, Manzi J, Kusek JW, Eggers P, Van Lente F, Levey AS. Prevalence of chronic kidney disease in the United States. JAMA. 2007;298:2038-2047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3821] [Cited by in RCA: 3465] [Article Influence: 182.4] [Reference Citation Analysis (3)] |
| 2. | Kenny GN. Ketorolac trometamol--a new non-opioid analgesic. Br J Anaesth. 1990;65:445-447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 17] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 3. | Thatte L, Vaamonde CA. Drug-induced nephrotoxicity: the crucial role of risk factors. Postgrad Med. 1996;100:83-84, 87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 9] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 4. | Jaquenod M, Rönnhedh C, Cousins MJ, Eckstein RP, Jordan V, Mather LE, Power I. Factors influencing ketorolac-associated perioperative renal dysfunction. Anesth Analg. 1998;86:1090-1097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (1)] |
| 5. | Schnitzer TJ. Non-NSAID pharmacologic treatment options for the management of chronic pain. Am J Med. 1998;105:45S-52S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 34] [Article Influence: 1.2] [Reference Citation Analysis (3)] |
| 6. | Jackson LM, Hawkey CJ. COX-2 selective nonsteroidal anti-Inflammatory drugs: do they really offer any advantages? Drugs. 2000;59:1207-1216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 93] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 7. | Fu S, Ramanujam KS, Wong A, Fantry GT, Drachenberg CB, James SP, Meltzer SJ, Wilson KT. Increased expression and cellular localization of inducible nitric oxide synthase and cyclooxygenase 2 in Helicobacter pylori gastritis. Gastroenterology. 1999;116:1319-1329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 320] [Cited by in RCA: 284] [Article Influence: 10.5] [Reference Citation Analysis (1)] |
| 8. | Rao P, Knaus EE. Evolution of nonsteroidal anti-inflammatory drugs (NSAIDs): cyclooxygenase (COX) inhibition and beyond. J Pharm Pharm Sci. 2008;11:81s-110s. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 391] [Cited by in RCA: 450] [Article Influence: 25.0] [Reference Citation Analysis (1)] |
| 9. | Smyth EM, Grosser T, Wang M, Yu Y, FitzGerald GA. Prostanoids in health and disease. J Lipid Res. 2009;50 Suppl:S423-S428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 432] [Cited by in RCA: 388] [Article Influence: 22.8] [Reference Citation Analysis (1)] |
| 10. | Dahle JT, Arai Y. Environmental geochemistry of cerium: applications and toxicology of cerium oxide nanoparticles. Int J Environ Res Public Health. 2015;12:1253-1278. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 213] [Cited by in RCA: 189] [Article Influence: 17.2] [Reference Citation Analysis (1)] |
| 11. | Cassee FR, van Balen EC, Singh C, Green D, Muijser H, Weinstein J, Dreher K. Exposure, health and ecological effects review of engineered nanoscale cerium and cerium oxide associated with its use as a fuel additive. Crit Rev Toxicol. 2011;41:213-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 259] [Cited by in RCA: 203] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 12. | Colon J, Herrera L, Smith J, Patil S, Komanski C, Kupelian P, Seal S, Jenkins DW, Baker CH. Protection from radiation-induced pneumonitis using cerium oxide nanoparticles. Nanomedicine. 2009;5:225-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 174] [Article Influence: 10.2] [Reference Citation Analysis (1)] |
| 13. | Niu J, Azfer A, Rogers LM, Wang X, Kolattukudy PE. Cardioprotective effects of cerium oxide nanoparticles in a transgenic murine model of cardiomyopathy. Cardiovasc Res. 2007;73:549-559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 321] [Cited by in RCA: 293] [Article Influence: 14.7] [Reference Citation Analysis (1)] |
| 14. | Bostan H, Kalkan Y, Tomak Y, Tumkaya L, Altuner D, Yılmaz A, Erdivanli B, Bedir R. Reversal of rocuronium-induced neuromuscular block with sugammadex and resulting histopathological effects in rat kidneys. Ren Fail. 2011;33:1019-1024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 17] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 15. | Kent AL, Maxwell LE, Koina ME, Falk MC, Willenborg D, Dahlstrom JE. Renal glomeruli and tubular injury following indomethacin, ibuprofen, and gentamicin exposure in a neonatal rat model. Pediatr Res. 2007;62:307-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 41] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 16. | Fong HJ, Cohen AH. Ibuprofen-induced acute renal failure with acute tubular necrosis. Am J Nephrol. 1982;2:28-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 27] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 17. | Akinola O, Noronha C, Oremosu A, Kusemiju O, Okanlawon OA. The effect of the cyclooxygenase blockers, ibuprofen on the development of glomeruli in Sprague-Dawley rats. Niger Postgrad Med J. 2003;10:46-50. [PubMed] [DOI] [Full Text] |
| 18. | Carvajal S, Perramón M, Casals G, Oró D, Ribera J, Morales-Ruiz M, Casals E, Casado P, Melgar-Lesmes P, Fernández-Varo G, Cutillas P, Puntes V, Jiménez W. Cerium Oxide Nanoparticles Protect against Oxidant Injury and Interfere with Oxidative Mediated Kinase Signaling in Human-Derived Hepatocytes. Int J Mol Sci. 2019;20:5959. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 19. | Better OS, Arieff AI, Massry SG, Kleeman CR, Maxwell MH. Studies on renal function after relief of complete unilateral ureteral obstruction of three months' duration in man. Am J Med. 1973;54:234-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 56] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 20. | Sacks SH, Aparicio SA, Bevan A, Oliver DO, Will EJ, Davison AM. Late renal failure due to prostatic outflow obstruction: a preventable disease. BMJ. 1989;298:156-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 50] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 21. | Ucero AC, Benito-Martin A, Fuentes-Calvo I, Santamaria B, Blanco J, Lopez-Novoa JM, Ruiz-Ortega M, Egido J, Burkly LC, Martinez-Salgado C, Ortiz A. TNF-related weak inducer of apoptosis (TWEAK) promotes kidney fibrosis and Ras-dependent proliferation of cultured renal fibroblast. Biochim Biophys Acta. 2013;1832:1744-1755. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 82] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 22. | Ucero AC, Benito-Martin A, Izquierdo MC, Sanchez-Niño MD, Sanz AB, Ramos AM, Berzal S, Ruiz-Ortega M, Egido J, Ortiz A. Unilateral ureteral obstruction: beyond obstruction. Int Urol Nephrol. 2014;46:765-776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 143] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 23. | Grande MT, López-Novoa JM. Fibroblast activation and myofibroblast generation in obstructive nephropathy. Nat Rev Nephrol. 2009;5:319-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 221] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 24. | Xu B, Zhang YB, Li ZZ, Yang MW, Wang S, Jiang DP. Hydrogen-rich saline ameliorates renal injury induced by unilateral ureteral obstruction in rats. Int Immunopharmacol. 2013;17:447-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 25. | Li SS, Ye JM, Deng ZY, Yu LX, Gu XX, Liu QF. Ginsenoside-Rg1 inhibits endoplasmic reticulum stress-induced apoptosis after unilateral ureteral obstruction in rats. Ren Fail. 2015;37:890-895. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 26. | Diamond JR, Kees-Folts D, Ricardo SD, Pruznak A, Eufemio M. Early and persistent up-regulated expression of renal cortical osteopontin in experimental hydronephrosis. Am J Pathol. 1995;146:1455-1466. [PubMed] |
| 27. | Yoo KH, Thornhill BA, Forbes MS, Coleman CM, Marcinko ES, Liaw L, Chevalier RL. Osteopontin regulates renal apoptosis and interstitial fibrosis in neonatal chronic unilateral ureteral obstruction. Kidney Int. 2006;70:1735-1741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 72] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 28. | Kamijo-Ikemori A, Sugaya T, Matsui K, Yokoyama T, Kimura K. Roles of human liver type fatty acid binding protein in kidney disease clarified using hL-FABP chromosomal transgenic mice. Nephrology (Carlton). 2011;16:539-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 39] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 29. | Manucha W, Carrizo L, Ruete C, Molina H, Vallés P. Angiotensin II type I antagonist on oxidative stress and heat shock protein 70 (HSP 70) expression in obstructive nephropathy. Cell Mol Biol (Noisy-le-grand). 2005;51:547-555. [PubMed] |
| 30. | Djamali A, Reese S, Yracheta J, Oberley T, Hullett D, Becker B. Epithelial-to-mesenchymal transition and oxidative stress in chronic allograft nephropathy. Am J Transplant. 2005;5:500-509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 90] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 31. | Filho DW, Torres MA, Bordin AL, Crezcynski-Pasa TB, Boveris A. Spermatic cord torsion, reactive oxygen and nitrogen species and ischemia-reperfusion injury. Mol Aspects Med. 2004;25:199-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 239] [Article Influence: 10.9] [Reference Citation Analysis (1)] |
| 32. | Manne ND, Arvapalli R, Nepal N, Shokuhfar T, Rice KM, Asano S, Blough ER. Cerium oxide nanoparticles attenuate acute kidney injury induced by intra-abdominal infection in Sprague-Dawley rats. J Nanobiotechnology. 2015;13:75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 53] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 33. | Córdoba-Jover B, Arce-Cerezo A, Ribera J, Pauta M, Oró D, Casals G, Fernández-Varo G, Casals E, Puntes V, Jiménez W, Morales-Ruiz M. Cerium oxide nanoparticles improve liver regeneration after acetaminophen-induced liver injury and partial hepatectomy in rats. J Nanobiotechnology. 2019;17:112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 50] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 34. | Halliwell B, Chirico S. Lipid peroxidation: its mechanism, measurement, and significance. Am J Clin Nutr. 1993;57:715S-724S; discussion 724S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1723] [Cited by in RCA: 1414] [Article Influence: 42.8] [Reference Citation Analysis (0)] |
| 35. | Zhou TB, Qin YH, Lei FY, Huang WF, Drummen GP. Association of prohibitin-1 and 2 with oxidative stress in rats with renal interstitial fibrosis. Mol Biol Rep. 2014;41:3033-3043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 36. | Amore A, Coppo R. Immunological basis of inflammation in dialysis. Nephrol Dial Transplant. 2002;17 Suppl 8:16-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 66] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 37. | Chuang YH, Chuang WL, Huang SP, Huang CH. Roles of nitric oxide and nitric oxide synthases in tissue damage of obstructed ureters in rats. Scand J Urol Nephrol. 2005;39:187-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (1)] |
| 38. | Kiliç I, Kiliç BA, Güven C, Demirpençe E, Akşit MA. Role of nitric oxide in hypoxia-induced changes in newborn rats. Biol Neonate. 2000;78:191-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 39. | van Meer L, Moerland M, Cohen AF, Burggraaf J. Urinary kidney biomarkers for early detection of nephrotoxicity in clinical drug development. Br J Clin Pharmacol. 2014;77:947-957. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (1)] |