Published online Mar 25, 2026. doi: 10.5501/wjv.v15.i1.118225
Revised: January 25, 2026
Accepted: March 5, 2026
Published online: March 25, 2026
Processing time: 76 Days and 19.4 Hours
Chronic viral infections, such as human immunodeficiency virus (HIV), hepatitis B (HBV), and hepatitis C (HCV), negatively impact bone health leading to osteopenia/osteoporosis and increased bone fragility. This narrative review aims to provide a comprehensive overview of the contemporary literature on bone changes in patients with chronic viral infections (HIV, HBV, HCV mono-infection, and coinfections), focusing on their clinical implications and the importance of assessing multiscale bone properties to evaluate bone strength decline in these individuals. Previous studies suggest that skeletal alterations in these subjects may arise from direct viral effects on bone cells and from indirect mechanisms involving systemic inflammation, immune dysregulation, therapy-related effects, and distant organ failure (e.g., liver disease). It has been reported that HBV/HCV co-infection in people living with HIV produces the most severe phenotype through additive inflammatory, hepatic, and metabolic insults. Further, an increased risk of developing osteonecrosis of multiple joints has also been reported among people living with HIV. Given the limited contemporary data, future studies should focus on investigating hierarchical alterations in bone structure to deepen our understanding of the complex skeletal changes in patients with chronic viral infections, thereby providing a solid foundation for advancing clinical management. As the population living with chronic viral infections ages, total joint arthroplasty will increasingly become a standard procedure, requiring a deeper understanding of how various hierarchical bone morpho-structural changes affect implant stability and longevity in these patients.
Core Tip: Chronic viral infections, such as human immunodeficiency virus, hepatitis B and hepatitis C mono- and coinfection, negatively impact bone health leading to osteopenia, osteoporosis, and increased bone fragility at an earlier age. However, many questions remain unanswered, underscoring the need for further research into the etiopathogenetic mechanisms underlying multiscale determinants of bone strength to guide more effective, personalized treatments and preventive modalities for these individuals.
- Citation: Jadzic JS, Baljozovic AB, Simic LD. Bone health and chronic viral infections: A narrative literature review. World J Virol 2026; 15(1): 118225
- URL: https://www.wjgnet.com/2220-3249/full/v15/i1/118225.htm
- DOI: https://dx.doi.org/10.5501/wjv.v15.i1.118225
Osteoporosis is a silent, progressive skeletal condition defined by low bone mineral density (BMD) and altered bone microarchitecture, which ultimately predisposes patients to fractures. In an etiopathogenetic sense, two main types of osteoporosis have been described: Primary and secondary osteoporosis[1]. Primary osteoporosis is associated with aging and declining sex hormones (i.e., postmenopausal osteoporosis), while secondary osteoporosis is caused by systemic diseases, organ dysfunctions, substance abuse, toxins, and medications[2,3]. Recent data suggest that secondary oste
Thus, this study aimed to provide an overview of the contemporary literature on skeletal changes in patients with chronic viral infections (HIV, HCV, and HBV mono- or coinfection), focusing on their clinical implications and the importance of using assessment of multiscale bone properties to evaluate bone strength decline in these individuals.
An electronic literature search was conducted in November 2025 and December 2025 using the PubMed/MEDLINE, Cochrane, and Web of Science databases. To identify published articles on bone health in patients with chronic viral infections, we used the following search terms: “viral infections” OR “HIV” OR “PLWH” OR “Hepatitis C” OR “HCV” OR “Hepatitis B” OR “HBV” AND ”bone” OR ”osteopenia” OR “osteoporosis” OR ”fracture” OR ”osteonecrosis” OR “bone mineral density” OR ”BMD” OR ”DXA” OR ”FRAX” OR “bone quality” OR ”bone microarchitecture” OR “pQCT”. Three authors independently conducted the literature search. This review included only human studies from basic, translational, and clinical fields, written in English. Studies involving pediatric patients living with HIV, HCV, and/or HBV were out of the scope of this review. Further, in vitro studies using cell lines to assess the etiopathogenetic mechanisms underlying bone alterations associated with viral infections were beyond the scope of this review. Our review focused on the evaluation of the postcranial skeleton in patients with HIV, HCV, and HBV infection, meaning that dental research in patients with chronic viral infections was not included. In the event of any discrepancies, the dilemma was resolved through discussion and all authors agreed on the final pool of studies included in this review.
Over the past two decades, a significant increase in life expectancy of patients with HIV, HCV, and HBV occurred due to major advances in the available treatment options. As a result, a new population of elderly patients with long-term viral infections is emerging. Nowadays, it is commonly debated whether bone health is affected in these individuals and whether viral infection itself or long-term exposure to antiviral drugs contributes to skeletal decline, revealing key issues for the clinical management of bone fragility in these patients[17]. Large epidemiological studies have revealed an increased fracture rate in PLWH compared with noninfected individuals[25,26], with a nearly threefold increase observed in the Danish register[27]. A most recent meta-analysis reported that 21% of PLWH experienced fragility fractures, indicating a significant prevalence of bone-related complications[28]. Overall fracture prevalence of 4.08% vs 0.44% and fragility fracture prevalence of 2.66% vs 2.19% have been noted in PLWH compared with controls, revealing relative risks for all fractures and fragility fractures of 1.91 and 1.68, respectively[7]. A high prevalence of subclinical vertebral fractures has been reported in PLWH (12%-46%)[29,30]. Since prior vertebral fracture [independent of Dual-energy X-ray absorptiometry (DXA) findings] is a risk factor for further fractures, screening with novel vertebral fracture assessment tools may help assess fracture risk, especially in osteopenic PLWH[29-31].
Regarding patients with chronic viral hepatitis, studies are also showing an increased risk of low-trauma fractures[32]. The DANVIR study[33] found an increased risk for all fracture localizations, with no significant difference between chronic and cleared HCV infection. Consistently, population-based analyses in postmenopausal women with HCV report a significantly higher predicted 10-year fracture risk (major osteoporotic fracture risk of 11.3% vs 9.0%, and hip fracture risk of 3.4% vs 2.3%) compared with HCV-seronegative individuals[34]. HIV/HCV co-infection is linked to a higher risk of fractures compared to HIV infection alone, which may only be partly attributable to the severity of liver disease. In a systematic review and meta-analysis of 15 studies, fracture incidence was increased (incidence rate ratio 1.77), and fracture risk was nearly threefold higher in HIV/HCV coinfected patients compared with uninfected controls[35]. Further, studies reported that HIV/HCV coinfection is linked to a 1.2-2.4-fold higher risk compared with HIV mono-infection, indicating that the detrimental impact of HCV on bone health may occur independently of advanced liver disease[18]. Bedimo et al[18] also reported markedly reduced bone turnover markers in HIV/HCV-co-infected patients who received pegylated interferon-α and ribavirin. However, it is unknown whether this is a direct effect of interferon or a result of HCV viral clearance, and whether the reduction of bone turnover markers will result in improved BMD[17]. In contrast, no difference in hip fracture incidence was noted for chronic HBV infection in comparison with the uninfected population, while in cases of hepatic decompensation, there was an increased incidence of hip fractures in individuals with HBV[10]. Comparable to HCV, patients with HIV/HBV co-infection who are receiving antiviral treatment face a higher risk of hip fracture compared to those treated for HIV alone or uninfected individuals[10]. In long-term follow-up, tenofovir disoproxil fumarate (TDF) therapy has also been associated with an increased risk of fractures in elderly individuals with HBV infection, whereas entecavir demonstrates a more favorable bone safety profile[36].
In summary, contemporary data suggest that clinical fracture risk evaluation is advised for individuals with chronic viral infections who exhibit specific clinical indicators (males aged 50 or above, postmenopausal women, and patients with prior fragility fractures)[37]. However, distinct universally accepted and applied guidelines for clinical fracture risk assessment in patients with chronic viral infections are yet to be established.
Large population-based cohorts and meta-analyses consistently demonstrate reduced BMD in patients living with HIV, HBV, and HCV infections. These associations are observed in both mono-infection and coinfection settings, with greater skeletal impact in coinfected individuals and important implications for long-term morbidity[7,11]. Reduced BMD of the lumbar spine and femoral neck, with a higher prevalence of osteoporosis in PLWH compared with uninfected controls, has recently been reported[8]. Namely, a cross-sectional cohort study in China[38] (n = 706 PLWH) showed a prevalence of low BMD (osteopenia/osteoporosis) of 13.9% overall, which markedly increased to 65.3% in PLWH aged ≥ 50 years compared with 38.2% in age-matched controls, which is further supported by conclusion of meta-analysis conducted by Chang et al[7]. Identified risk factors include older age, low body mass index, chronic inflammation, immunodeficiency, and prolonged ART[38,39]. Previous studies also revealed that most bone loss in PLWH occurs during the early stages of the disease and is exacerbated by the ART initiation, highlighting the interplay among the immune system, inflammation, and increased osteoclast activity[40]. Namely, multiple studies indicate that ART initiation is associated with 2%-6% decline in BMD during the first 1-2 years after starting treatment, particularly at trabecular-rich sites such as the lumbar spine. This effect is most pronounced with TDF-containing regimens and has also been reported with protease inhibitor (PIs)-based therapies[3]. Treatment with TDF has been associated with greater decreases in BMD, particularly at the lumbar spine (approximately -2.1% vs -0.8% with entecavir), and with a higher prevalence of osteopenia compared with entecavir-treated PLWH[41,42]. Switching to tenofovir alafenamide is associated with smaller declines or modest increases in spine and hip BMD compared to continued TDF-based therapy in these patients[43]. Thus, current data suggest that it may be advisable to avoid using TDF or boosted PIs in PLWH with a high likelihood of skeletal alterations.
Chronic HBV infection is increasingly recognized as a condition accompanied by reduced bone mass. Data from case-control studies and cohort analyses show that patients with chronic HBV have lower BMD at the lumbar spine and femoral neck compared with healthy controls of similar age[17,44]. Large population-based cohort data further demonstrate that chronic HBV infection is independently associated with an increased risk of osteoporosis that may occur at an earlier age. In a retrospective cohort analysis[11], the 5-year cumulative incidence of osteoporosis was higher in HBV patients than in matched controls (2.9% vs 1.6%), with a corresponding hazard ratio of 1.76 for osteoporosis, and a more pronounced association was observed in males. Also, chronic HCV infection is associated with reduced BMD and an increased risk of osteoporosis, as demonstrated in large population-based cohorts and meta-analyses[11,15]. This impact is further supported by findings of DXA studies of treatment-naïve, non-cirrhotic HCV patients, which demonstrate lower lumbar spine and femoral neck BMD, particularly among postmenopausal women, supporting HCV infection as an independent risk factor for osteoporosis[45]. Still, it is important to note that the direct bone effects of anti-HCV therapy, including direct-acting antivirals, remain incompletely characterized, as clinical studies have not thoroughly examined how direct-acting antiviral-mediated HCV cure affects bone outcomes[46,47]. Furthermore, patients with HIV/HCV coinfection have greater reductions in BMD compared with HIV mono-infected or uninfected individuals[18,35]. In a systematic review and meta-analysis of 15 studies[35], the prevalence of osteoporosis in HIV/HCV coinfected patients was approximately 22%, with an odds ratio of 1.63 compared with HIV mono-infection. Recent DXA-based studies demonstrate significantly lower BMD and Z-scores in HIV/HCV coinfected individuals compared with controls, as well as a higher prevalence of bone demineralization (31.7%) compared with HIV (23.8%) or HCV (7.5%) mono-infected patients, with more frequent lumbar spine demineralization in HIV/HCV coinfection[48]. Still, data on BMD in HBV/HCV coinfection are limited, with only one cross-sectional study demonstrating that HBV/HCV coinfection was more strongly associated with reduced BMD than HBV mono-infection, with the association remaining significant only in males, not in females[49].
In summary, contemporary data support the notion that chronic viral infections represent a relevant and underrecognized cause of secondary osteoporosis, with HIV, HBV, HCV, and their coinfections being associated with clinically significant reductions in BMD and increased fracture risk[7,8,11,17,21,38]. However, a substantial proportion of fragility fractures occur in individuals with BMD values above osteoporotic thresholds defined by DXA, underscoring the need for generating new integrated clinical assessments beyond BMD alone. Optimization of calcium, protein, and vitamin D intake has been recommended, with evidence indicating that bone loss at the initiation of ART can be attenuated through supplementation with high doses of vitamin D and calcium[40,50]. Recent data indicate that alendronate and zoledronate increase BMD and are well tolerated in PLWH[19,51], while raloxifene has been reported to enhance the effectiveness of antiviral therapy in postmenopausal women suffering from HCV[52]. This may suggest it could serve as a beneficial adjunct to standard antiviral regimens in osteoporotic women with HCV[53], warranting further confirmation.
Apart from osteoporosis and bone fragility, it is important to recognize other specific skeletal abnormalities in individuals living with chronic viral infections. One of these is osteonecrosis[54] - an ischemic or cytotoxic necrosis affecting the epiphyseal bone, leading to gradual destruction and collapse of the subchondral bone, which can cause secondary osteoarthritis[55,56]. It is a debilitating and progressive condition that is most often irreversible without treatment. Symptoms vary depending on the affected bone and the stage of the disease. Most patients experience sudden pain as the initial symptom. While joint mobility is usually maintained in the early stages, it tends to decline as the disease advances, eventually leading to painful joint contracture[56]. Osteonecrosis of the femoral head is the most frequent[22], with the humeral head being the second most frequent, followed by the knee, ankle, and elbow joint[57]. When present in PLWH, the condition is often bilateral, with multiple joint involvement not uncommon[55,58]. Osteonecrosis is a multifactorial condition with known risk factors including systemic corticosteroids, alcoholism, hyperlipidemia, sickle cell anemia, coagulopathies, autoimmune disease, obesity, and smoking. Still, PLWH experience symptomatic osteonecrosis more frequently than the general population[56], indicating that these individuals may have some additional risk factors for osteonecrosis. Indeed, previous studies revealed that dyslipidemia, the use of megestrol acetate and steroids, testosterone replacement, vasculitis, anti-phospholipid syndrome, and specific ART treatment, including regimens with PIs, have been associated with osteonecrosis in PLWH[22,59]. Various theories have been proposed regarding the development of multifocal osteonecrosis in PLWH, including direct damage through necrotizing vasculitis and hyperlipidemia-caused ischemia induced by PIs[54]. Further, a significant correlation has been identified between TDF-based regimens and osteonecrosis in PLWH, but the exact mechanisms remain to be fully elucidated[59]. In PLWH, osteonecrosis may progress rapidly and be accompanied by severe symptoms, often requiring multiple joint replacement procedures, apart from non-surgical therapeutic approaches[56]. Previous studies indicate that joint replacement in PLWH is safe and effective, with complication risks similar to those in the general population[60,61]. It is important to note that previous studies focus on HIV alone[62], while the incidence of osteonecrosis in patients with HCV and HBV mono-infection and HIV-coinfection is still unknown, requiring further research.
Contemporary clinical and epidemiological literature indicates that increased bone fragility cannot be captured solely by DXA and cannot be explained solely by a decrease in BMD[39,63]. It is well known that only up to one-third of fragility fractures are associated with low BMD, and DXA findings cannot explain the remainder[64,65]. Moreover, recent studies are suggesting that the clinically common applicable fracture risk assessment tool (FRAX) underestimates fracture risk in people with chronic viral infections (especially in HIV and HCV-infected individuals)[66-69]. FRAX overlooks infection-related factors such as HIV or HCV co-infection, duration of antiviral treatment, nutritional status, frailty, fall propensity, and it is not validated for patients under 40. Emerging evidence indicates that a modified FRAX[70], incorporating HIV as a secondary cause of osteoporosis, enhances predictive accuracy in clinical practice, which may indicate that applying this modified FRAX tool yields more reliable guidance for therapeutic interventions targeting skeletal disturbances associated with both the virus and ART. These data support the importance of assessing other factors (especially intrinsic bone features, referred to as bone quality[71,72]). Evaluating these bone features is crucial because it may lead to the development of new and more effective treatment methods, as some anti-osteoporosis medications have been shown to enhance bone strength and reduce fracture risk without elevating BMD[73].
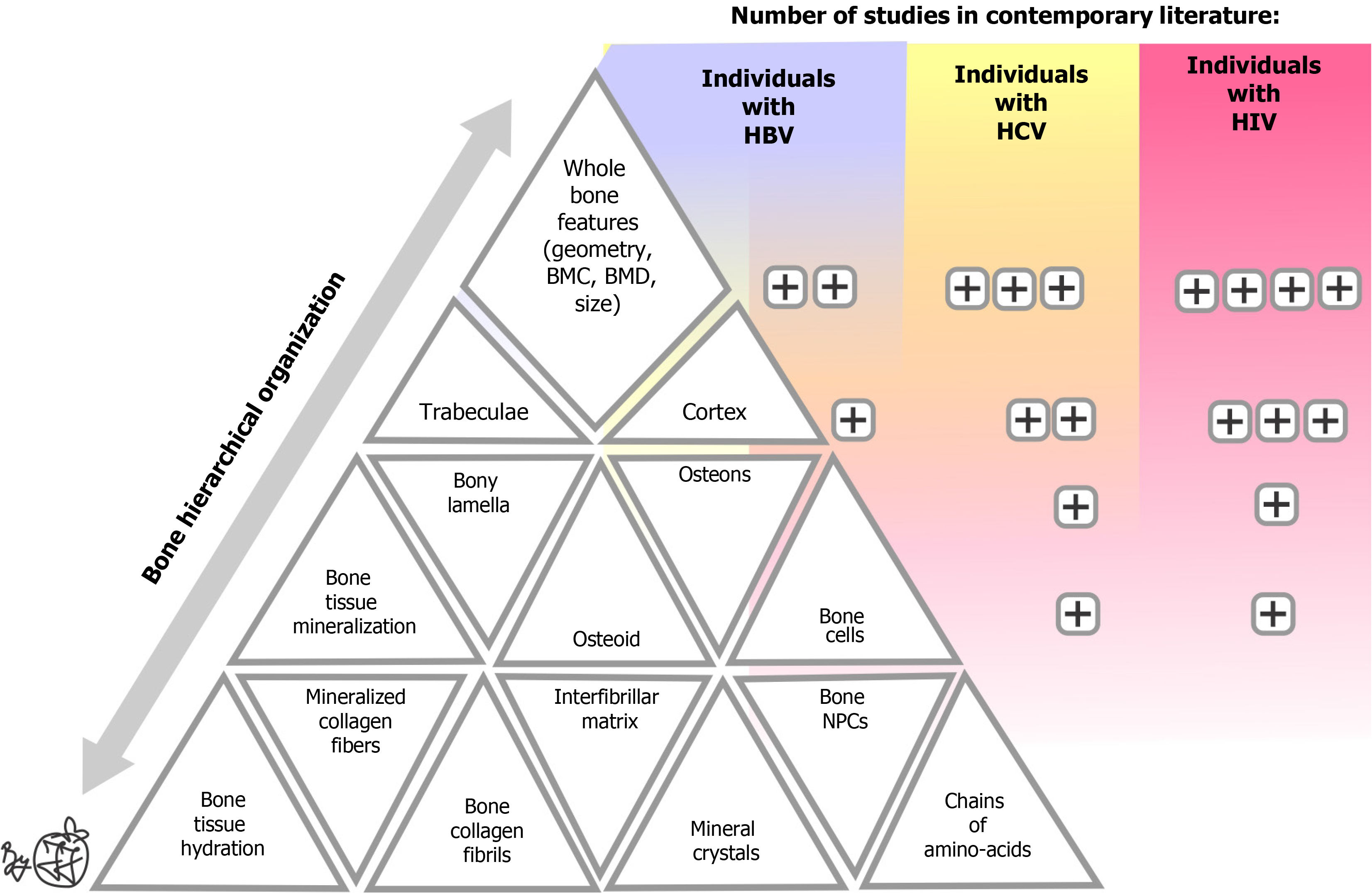
Peripheral quantitative computed tomography (pQCT) is a noninvasive 3D method for the clinical assessment of bone microarchitecture at the distal radius and tibia, developed to overcome limitations of DXA[74-77]. Most frequently, recent studies have used pQCT to assess bone microarchitecture in PLWH[78-80]. Recently, our team published a detailed evaluation of the studies on bone microarchitecture in PLWH[8]. In short, previous studies reported deficits in trabecular and cortical bone parameters (e.g., 6%-19% lower volumetric BMD, thinner cortices, reduced trabecular number) in HIV-infected patients compared to uninfected controls, often independent of areal BMD by DXA, and linked to HIV duration, ART (especially tenofovir), and inflammation[74-80]. Thus, in this review, we will focus more on the rare studies involving intrinsic bone alterations in individuals with HBV and HCV mono-infections and their coinfections with HIV (Figure 1). Pioneering studies[81-83] used bone histomorphometry on transiliac bone biopsy samples to evaluate skeletal changes in individuals with chronic HCV or HBV infection. These studies are primarily case reports or small series, with exclusive involvement of female patients. Findings in HCV patients center on HCV-associated osteosclerosis, characterized by markedly increased bone turnover with net bone gain (elevated bone volume and osteoid volume, thicker trabeculae), and slightly delayed mineralization[81-83]. Moreover, larger pioneering studies were focused on bone histomorphometry of transiliac bone biopsy samples from individuals with viral liver disease and liver cirrhosis. A cross-sectional study by Diamond et al[84] showed reduced trabecular bone volume, due to reduced trabecular thickness, coupled with prolonged mineralization lag time and low bone formation rates, noted in 20 individuals with chronic viral cirrhosis compared to control individuals. A cross-sectional study by Stellon et al[85], revealed low-turnover osteoporosis driven by impaired bone formation, prolonged osteoid maturation, and mild resorption increase in 34 women with corticosteroid-treated chronic viral hepatitis. Moreover, Klco et al[86] reported a wide range of bone marrow alterations in individuals living with HCV, without correlating these observations with possible skeletal changes. On the other hand, bone histomorphometry of transiliac bone biopsy samples from individuals with HBV showed drug-induced (adefovir or tenofovir) hypophosphatemic osteomalacia with impaired mineralization and low bone turnover[87]. These findings were recently supported by the pQCT study by Wakolbinger et al[88], which involved eight patients with liver cirrhosis of viral origin (HCV and HBV). These studies suggest that trabecular bone is preferentially affected due to its higher cytokine sensitivity and bone turnover rate, while cortical bone is relatively spared except in advanced liver disease with secondary hyperparathyroidism. Some more recent studies consistently demonstrate that individuals with HIV/HCV coinfection have mild deficits in trabecular and cortical parameters of distal radius and tibia (lower trabecular bone volume, thinner trabeculae, thinner and more porous cortices) compared to HIV-mono-infected or uninfected controls[16,80]. It is important to note the findings of the latest study[89], which report the absence of significant changes in trabecular/cortical microstructure, stiffness, or failure load before and 18 months post-cure in HCV-positive individuals.
Contemporary literature on bone quality alterations in individuals with chronic viral infection is based on small-scale cross-sectional studies, which fairly limits the statistical power, warranting further research using large-scale prospective studies with specific stratification strategies to reveal the true nature of these changes. Given the scarcity of data in contemporary literature (especially in individuals with HBV, Figure 1), future research should prioritize the analysis of the internal organization of bone tissue (with distinct focus on the Haversian system and bony lamellae analysis), morpho-structural and functional characteristics of bone cells, the compositional evaluation of the mineral and organic parts of bone extracellular matrix, and the functional examination of bone marrow to clarify its impact on bone fragility in individuals living with HIV, HBV and HCV. The overall significance of these investigations could be enhanced by employing multiple advanced techniques to examine various hierarchical structural bone properties within the same bone sample from an individual patient[71]. Most importantly, these bone-assessing techniques should be combined with computational methods[90,91] to fully describe bone alterations in individuals with chronic viral infections[8]. Ultimately, in combination with clinical data, these new findings may lay a solid foundation for the development of personalized algorithms to predict fracture risk in these individuals.
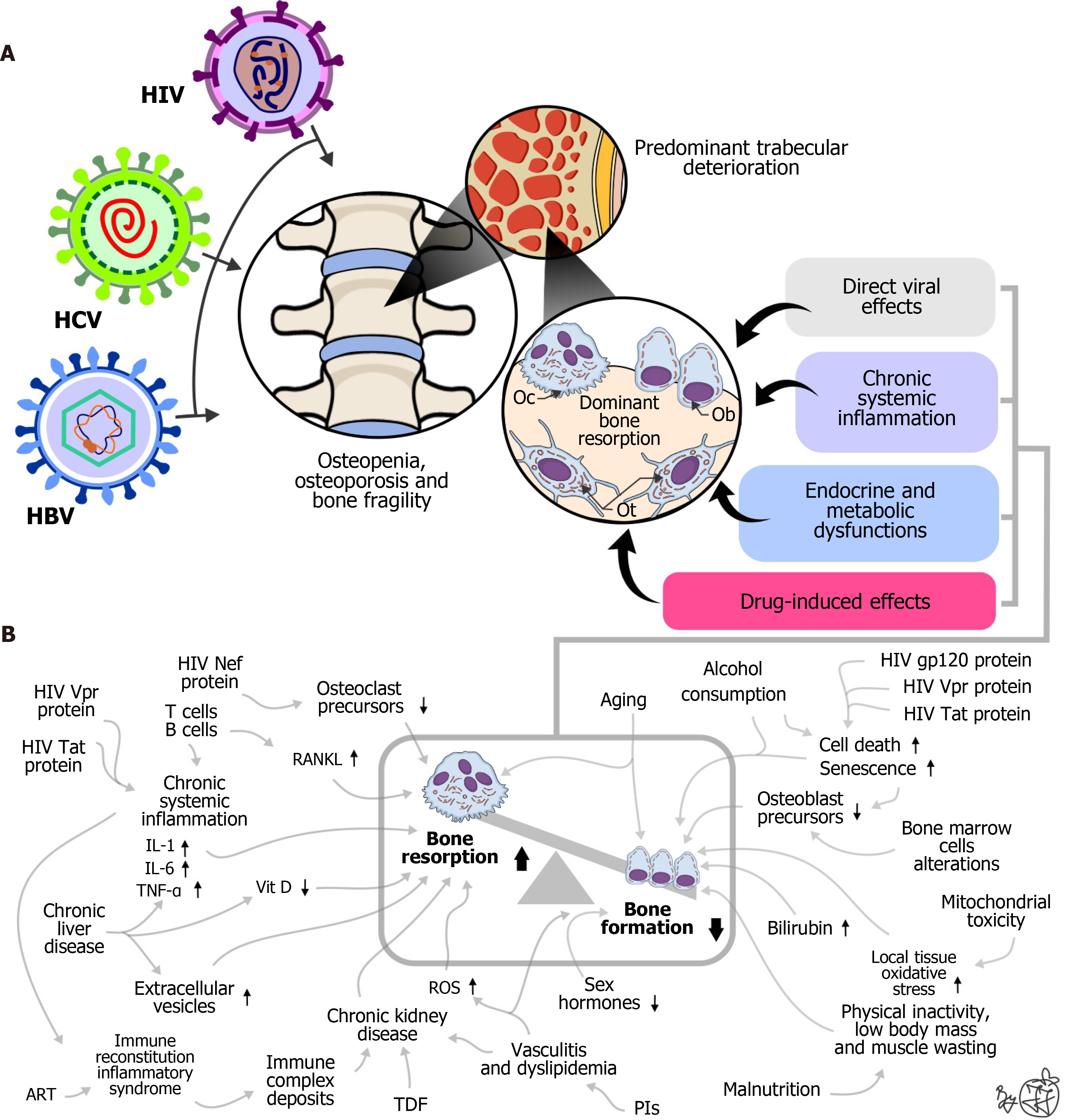
This review has several limitations that should be acknowledged. Our review was restricted to studies published in English and available in full text, which may introduce publication bias, indicating that future reviews might benefit from searching the literature in other languages to provide a more comprehensive and population-specific overview of the topic. Since this is a narrative rather than a systematic review, we did not quantify the methodological quality of the cited studies and thus did not evaluate publication bias. Moreover, our review extrapolates findings from a limited, heterogeneous pool of studies, particularly regarding micro-scale bone features in individuals with chronic viral infections. Lastly, a detailed evaluation of studies on the etiopathogenetic mechanisms of skeletal alterations in individuals with chronic viral infections would require a more systematic approach and has been previously elaborated in great detail elsewhere[8,21,39,92,93]. To avoid repetition of previous work, we should state that these studies suggest that bone loss in HIV, HBV, and HCV, and especially its coinfection, is driven by synergistic inflammatory, viral, endocrine, and iatrogenic mechanisms that disproportionately target trabecular microarchitecture and osteoclast function, resulting in accelerated skeletal fragility at an earlier age than in the general population. Thus, in addition to aging-related bone loss, other unique factors contribute to bone loss in individuals with chronic viral infections (Figure 2). It is important to note that these etiopathogenetic pathways disproportionately affect trabecular bone due to their high metabolic demands, which may explain the findings of previous studies on inter-site differences and the predominance of vertebral fractures in these patients[94-97]. In short, bone loss in individuals living with chronic viral conditions results from the convergence of four major pathophysiological pathways (Figure 2): Chronic systemic inflammation and immune activation, direct viral effects on bone cells, endocrine and metabolic disturbances, and drug-induced mechanisms (ART and historical HCV therapies). It has been reported that HIV and HCV core proteins inhibit osteoblast differentiation while promoting osteoclast activity by upregulating receptor activator of nuclear factor-κB and pro-inflammatory cytokines [tumor necrosis factor-α, interleukin-6 (IL)-6, IL-1β], disrupting the receptor activator of nuclear factor-κB/osteoprotegerin axis, and accelerating bone resorption[17,97-100]. Studies revealed that most bone loss in PLWH occurs during the early stages of the disease and is exacerbated by the initiation of antiviral therapy, highlighting the interplay among the immune system, inflammation, and osteoclast activity[40]. In patients with chronic HCV infection, persistent arthralgia and bone loss are associated with elevated IL-6 and a skewing of mesenchymal stem cell fate toward adipogenesis rather than osteogenesis[100]. HBV and HIV/HBV co-infection accelerate BMD loss and fractures via insulin-like growth factor 1 deficiency, cytokine excess (IL-1, tumor necrosis factor-α), and ART toxicity, with trabecular predominance and males at heightened risk[101,102]. Taken together, contemporary literature on the etiopathogenetic mechanisms of bone loss in chronic viral infections cannot fully explain the clinical presentation, underscoring the need for further research in the years to come.
Skeletal damage is common in individuals living with HIV, HBV, and HCV. Still, the influence of these viral infections on the musculoskeletal system may have been underestimated and frequently overlooked. Previous studies have contributed to our understanding of skeletal alterations in these individuals, but numerous questions remain un
| 1. | Amarnath SS, Kumar V, Das SL. Classification of Osteoporosis. Indian J Orthop. 2023;57:49-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 31] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 2. | Sözen T, Özışık L, Başaran NÇ. An overview and management of osteoporosis. Eur J Rheumatol. 2017;4:46-56. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1833] [Cited by in RCA: 1466] [Article Influence: 162.9] [Reference Citation Analysis (1)] |
| 3. | Ebeling PR, Nguyen HH, Aleksova J, Vincent AJ, Wong P, Milat F. Secondary Osteoporosis. Endocr Rev. 2022;43:240-313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 229] [Article Influence: 57.3] [Reference Citation Analysis (1)] |
| 4. | Jähn-Rickert K, Tomšić KZ, Anastasilakis AD, Bertocchio JP, Brandi ML, Fassnacht M, Cheung MS, Gevers E, Gittoes N, Mäkitie O, Martos-Moreno GÁ, Milovanovic P, Raimann A, Tencerova M, Tsourdi E, Vlug AG, Högler W, Kamenický P. EndoCompass Project: Research Roadmap for Calcium and Bone Endocrinology. Horm Res Paediatr. 2025;98:29-43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 5. | Sobh MM, Abdalbary M, Elnagar S, Nagy E, Elshabrawy N, Abdelsalam M, Asadipooya K, El-Husseini A. Secondary Osteoporosis and Metabolic Bone Diseases. J Clin Med. 2022;11:2382. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 93] [Article Influence: 23.3] [Reference Citation Analysis (1)] |
| 6. | Mirza F, Canalis E. Management of endocrine disease: Secondary osteoporosis: pathophysiology and management. Eur J Endocrinol. 2015;173:R131-R151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 228] [Article Influence: 20.7] [Reference Citation Analysis (1)] |
| 7. | Chang CJ, Chan YL, Pramukti I, Ko NY, Tai TW. People with HIV infection had lower bone mineral density and increased fracture risk: a meta-analysis. Arch Osteoporos. 2021;16:47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 8. | Jadzic J, Dragovic G, Lukic R, Obradovic B, Djuric M. Bone Tissue Changes in Individuals Living with HIV/AIDS: The Importance of a Hierarchical Approach in Investigating Bone Fragility. J Pers Med. 2024;14:791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 9. | Pramukti I, Lindayani L, Chen YC, Yeh CY, Tai TW, Fetzer S, Ko NY. Bone fracture among people living with HIV: A systematic review and meta-regression of prevalence, incidence, and risk factors. PLoS One. 2020;15:e0233501. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 46] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 10. | Byrne DD, Newcomb CW, Carbonari DM, Nezamzadeh MS, Leidl KB, Herlim M, Yang YX, Hennessy S, Kostman JR, Leonard MB, Localio AR, Lo Re V 3rd. Increased risk of hip fracture associated with dually treated HIV/hepatitis B virus coinfection. J Viral Hepat. 2015;22:936-947. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 11. | Loosen SH, Killer A, Bock HH, Luedde T, Roderburg C, Kostev K. Association between Chronic Hepatitis B/C and Incidence of Osteoporosis and Bone Fractures: Results from a Retrospective Cohort Study. J Clin Med. 2024;13:6152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 12. | Shin YE, Kim JY, Kim H, Yoo JJ, Kim SG, Kim YS. Comparative risk of osteoporosis and fractures in chronic hepatitis B patients: Tenofovir disoproxil fumarate vs. entecavir in a Korean nationwide cohort. JHEP Rep. 2025;7:101489. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 13. | Chen CH, Lin CL, Kao CH. Relation Between Hepatitis C Virus Exposure and Risk of Osteoporosis: A Nationwide Population-Based Study. Medicine (Baltimore). 2015;94:e2086. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 22] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 14. | Lin MS, Chen PH, Wang PC, Lin HS, Huang TJ, Chang ST, Chiu WN, Chen MY. Association between hepatitis C virus infection and osteoporotic fracture risk among postmenopausal women: a cross-sectional investigation in Taiwan. BMJ Open. 2019;9:e021990. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 15. | Wijarnpreecha K, Thongprayoon C, Panjawatanan P, Ungprasert P. Hepatitis C virus infection and risk of osteoporotic fracture: A systematic review and meta-analysis. J Evid Based Med. 2018;11:20-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 16. | Lo Re V 3rd, Lynn K, Stumm ER, Long J, Nezamzadeh MS, Baker JF, Hoofnagle AN, Kapalko AJ, Mounzer K, Zemel BS, Tebas P, Kostman JR, Leonard MB. Structural Bone Deficits in HIV/HCV-Coinfected, HCV-Monoinfected, and HIV-Monoinfected Women. J Infect Dis. 2015;212:924-933. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 17. | Biver E, Calmy A, Rizzoli R. Bone health in HIV and hepatitis B or C infections. Ther Adv Musculoskelet Dis. 2017;9:22-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 36] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 18. | Bedimo R, Maalouf NM, Lo Re V 3rd. Hepatitis C virus coinfection as a risk factor for osteoporosis and fracture. Curr Opin HIV AIDS. 2016;11:285-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 19. | Wang JC, Chung SM, Wu PT, Tu YK, Lai PC, Tai TW, Wu CH, Chang YF, Kuan FC, Hsu KL, Fang CJ, Li CW, Chen PL, Shih CA. Optimizing bone health in people living with HIV: insights from a network meta-analysis of randomized controlled trials. EClinicalMedicine. 2025;81:103103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 20. | Bolland MJ, Grey A, Reid IR. Skeletal health in adults with HIV infection. Lancet Diabetes Endocrinol. 2015;3:63-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 35] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 21. | Jadzic J, Djonic D. Bone loss in chronic liver diseases: Could healthy liver be a requirement for good bone health? World J Gastroenterol. 2023;29:825-833. [PubMed] [DOI] [Full Text] |
| 22. | Munhoz Lima ALL, Oliveira PR, Carvalho VC, Godoy-Santos AL, Ejnisman L, Oliveira CR, Uip DE, Duarte MIS. Osteonecrosis of the femoral head in people living with HIV: anatomopathological description and p24 antigen test. HIV AIDS (Auckl). 2018;10:83-90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 23. | Pietrzak JRT, Maharaj Z, Mokete L, Sikhauli N. Human immunodeficiency virus in total hip arthroplasty. EFORT Open Rev. 2020;5:164-171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 24. | Sidiropoulos K, Christofilos SI, Tsikopoulos K, Kitridis D, Drago L, Meroni G, Romanò CL, Kavarthapu V. Viral infections in orthopedics: A systematic review and classification proposal. World J Orthop. 2022;13:1015-1028. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 3] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 25. | Güerri-Fernandez R, Vestergaard P, Carbonell C, Knobel H, Avilés FF, Castro AS, Nogués X, Prieto-Alhambra D, Diez-Perez A. HIV infection is strongly associated with hip fracture risk, independently of age, gender, and comorbidities: a population-based cohort study. J Bone Miner Res. 2013;28:1259-1263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 105] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 26. | Triant VA, Brown TT, Lee H, Grinspoon SK. Fracture prevalence among human immunodeficiency virus (HIV)-infected versus non-HIV-infected patients in a large U.S. healthcare system. J Clin Endocrinol Metab. 2008;93:3499-3504. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 428] [Cited by in RCA: 390] [Article Influence: 21.7] [Reference Citation Analysis (1)] |
| 27. | Hansen AB, Gerstoft J, Kronborg G, Larsen CS, Pedersen C, Pedersen G, Obel N. Incidence of low and high-energy fractures in persons with and without HIV infection: a Danish population-based cohort study. AIDS. 2012;26:285-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 121] [Article Influence: 8.6] [Reference Citation Analysis (1)] |
| 28. | Afraie M, Zamani K, Moradi G, Khateri S, Nikbakht N, Moradi Y. Osteoporosis and fracture risk among individuals with HIV: a systematic review and meta-analysis. Discov Public Health. 2025;22. [DOI] [Full Text] |
| 29. | Ilha TASH, Comim FV, Copes RM, Compston JE, Premaor MO. HIV and Vertebral Fractures: a Systematic Review and Metanalysis. Sci Rep. 2018;8:7838. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 30. | Dalla Grana E, Rigo F, Lanzafame M, Lattuada E, Suardi S, Mottes M, Valenti MT, Dalle Carbonare L. Relationship Between Vertebral Fractures, Bone Mineral Density, and Osteometabolic Profile in HIV and Hepatitis B and C-Infected Patients Treated With ART. Front Endocrinol (Lausanne). 2019;10:302. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 31. | Stephens KI, Rubinsztain L, Payan J, Rentsch C, Rimland D, Tangpricha V. Dual-energy x-ray absorptiometry and calculated frax risk scores may underestimate osteoporotic fracture risk in vitamin d-deficient veterans with hiv infection. Endocr Pract. 2016;22:440-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 32. | Lo Re V 3rd, Volk J, Newcomb CW, Yang YX, Freeman CP, Hennessy S, Kostman JR, Tebas P, Leonard MB, Localio AR. Risk of hip fracture associated with hepatitis C virus infection and hepatitis C/human immunodeficiency virus coinfection. Hepatology. 2012;56:1688-1698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 65] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 33. | Hansen AB, Omland LH, Krarup H, Obel N; DANVIR cohort study. Fracture risk in hepatitis C virus infected persons: results from the DANVIR cohort study. J Hepatol. 2014;61:15-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 34. | Maalouf NM, Zhang S, Drechsler H, Brown GR, Tebas P, Bedimo R. Hepatitis C co-infection and severity of liver disease as risk factors for osteoporotic fractures among HIV-infected patients. J Bone Miner Res. 2013;28:2577-2583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 35] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 35. | Dong HV, Cortés YI, Shiau S, Yin MT. Osteoporosis and fractures in HIV/hepatitis C virus coinfection: a systematic review and meta-analysis. AIDS. 2014;28:2119-2131. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 56] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 36. | Yip TC, Lai JC, Yam TF, Tse YK, Hui VW, Lai MS, Chan HL, Wong VW, Wong GL. Long-term use of tenofovir disoproxil fumarate increases fracture risk in elderly patients with chronic hepatitis B. J Hepatol. 2024;80:553-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 32] [Article Influence: 16.0] [Reference Citation Analysis (1)] |
| 37. | Ahmed M, Mital D, Abubaker NE, Panourgia M, Owles H, Papadaki I, Ahmed MH. Bone Health in People Living with HIV/AIDS: An Update of Where We Are and Potential Future Strategies. Microorganisms. 2023;11:789. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 38. | Shao Y, Sun J, Kong K, Zhang R, Zhang R, Liu L, Wang J, Shen Y, Wu L, Zheng Z, Sun M, Qi T, Wang Z, Tang Y, Sun J, Song W, Yang J, Xu S, Zhao B, Shan F, Qin A, Lu H, Chen J. Prevalence and associated factors of low bone mineral density in people living with HIV: a cross-sectional study. Arch Osteoporos. 2024;19:56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 39. | Delpino MV, Quarleri J. Influence of HIV Infection and Antiretroviral Therapy on Bone Homeostasis. Front Endocrinol (Lausanne). 2020;11:502. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 42] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 40. | Biver E. Osteoporosis and HIV Infection. Calcif Tissue Int. 2022;110:624-640. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 65] [Article Influence: 16.3] [Reference Citation Analysis (1)] |
| 41. | Liu Z, Zhao Z, Ma X, Liu S, Xin Y. Renal and bone side effects of long-term use of entecavir, tenofovir disoproxil fumarate, and tenofovir alafenamide fumarate in patients with Hepatitis B: a network meta-analysis. BMC Gastroenterol. 2023;23:384. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 42. | Kahraman R, Şahin A, Öztürk O, Çalhan T, Sayar S, Kanat E, Doğanay L, Özdil K. Effects of Long-Term Tenofovir and Entecavir Treatment on Bone Mineral Density in Patients with Chronic Hepatitis B. Turk J Gastroenterol. 2022;33:35-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 43. | Shao Y, Yang X, Yu J, Wang X, Wang J, Liu M, Yang Z, Han J, Zhang R, Liu L, Shen Y, Sun M, Wu L, Zheng Z, Tang Y, Yang J, Wang Z, Qi T, Xu S, Xun J, Sun J, Song W, Chen J. Bone Mineral Density Changes in People with HIV Who had Immediate Switch Versus Deferred Switch from Tenofovir Disoproxil Fumarate-Based Regimens to Bictegravir/Emtricitabine/Tenofovir Alafenamide: A Multicenter, Open-Label, Randomized Clinical Trial. Infect Dis Ther. 2026;15:165-181. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 44. | Huang Z, Wei H, Cheng C, Yang S, Wang J, Liu X. Low bone mineral density in chronic hepatitis B virus infection: A case-control study. Pak J Med Sci. 2017;33:457-461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 45. | Olmos-Martínez JM, Hernández JL, Fábrega E, Olmos JM, Crespo J, González-Macías J. Bone mineral density and trabecular bone score in treatment-naïve patients with non-cirrhotic hepatitis C virus infection. Arch Osteoporos. 2020;15:72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 46. | Psomas CK, Lellouche-Slama E, Langan S, Sulkowski M, Moseley KF, Pena-Dias J, Khati S, Sun J, Abraham AG, Brown TT. Impact of HCV cure on systemic inflammation and bone density, quality, and turnover. Front Immunol. 2025;16:1626875. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 47. | Ichikawa T, Yamashima M, Yamamichi S, Koike M, Nakano Y, Honda T, Yajima H, Miyazaki O, Kuribayashi Y, Ikeda T, Okamura T, Nakao K. Directacting antiviral treatment decreases serum undercarboxylated osteocalcin in male patients with chronic hepatitis C. Biomed Rep. 2022;17:84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 48. | Barbu EC, Moroti-Constantinescu VR, Lazar M, Chitu CE, Olariu CM, Bojinca M, Ion DA. Body composition changes in men with HIV/HCV coinfection, HIV monoinfection, and HCV monoinfection. Acta Endocrinol (Buchar). 2022;18:442-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 49. | Chen YY, Fang WH, Wang CC, Kao TW, Chang YW, Yang HF, Wu CJ, Sun YS, Chen WL. Crosssectional Assessment of Bone Mass Density in Adults with Hepatitis B Virus and Hepatitis C Virus Infection. Sci Rep. 2019;9:5069. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 50. | Overton ET, Chan ES, Brown TT, Tebas P, McComsey GA, Melbourne KM, Napoli A, Hardin WR, Ribaudo HJ, Yin MT. Vitamin D and Calcium Attenuate Bone Loss With Antiretroviral Therapy Initiation: A Randomized Trial. Ann Intern Med. 2015;162:815-824. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 103] [Article Influence: 9.4] [Reference Citation Analysis (2)] |
| 51. | Carr A, Kerr SJ, Richardson R, Ebeling P, Pocock N, Rojas J, Martinez E, Hoy J; ZEST study Investigators. Prolonged Effect of Zoledronic Acid on Bone Mineral Density and Turnover in HIV-Infected Adults on Tenofovir: A Randomized, Open-Label Study. J Bone Miner Res. 2019;34:2192-2197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 52. | Takeda M, Ikeda M, Mori K, Yano M, Ariumi Y, Dansako H, Wakita T, Kato N. Raloxifene inhibits hepatitis C virus infection and replication. FEBS Open Bio. 2012;2:279-283. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 53. | Hong S, Chang J, Jeong K, Lee W. Raloxifene as a treatment option for viral infections. J Microbiol. 2021;59:124-131. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (1)] |
| 54. | Bayard C, Ledergerber B, Flepp M, Lecompte T, Moulin E, Hoffmann M, Weber R, Staehelin C, Di Benedetto C, Fux CA, Tarr PE, Hasse B; Swiss HIV Cohort Study. Associations Between Antiretroviral Treatment and Avascular Bone Necrosis: The Swiss HIV Cohort Study. Open Forum Infect Dis. 2017;4:ofx177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 55. | Kawiche GS, Mahsein M, Mwanga DR, Ringo TA, Mrimba PM, Mandari FN. HIV positive patient with bilateral osteonecrosis of the femoral head, a case report. Int J Surg Case Rep. 2024;119:109660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 56. | Pisanu F, Fiore V, Corradi M, Esposito E, Maida I, Caggiari G, Ciatti C, Manunta AF, Doria C. Joint replacement for avascular necrosis in people living with HIV. Acta Biomed. 2022;92:e2021560. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 57. | Jeong HJ, Park JW, Lee YK, Koo KH, Oh JH. Comparison between osteonecrosis of the humeral and femoral heads - epidemiological analysis of the surgical trend using the nationwide claims database of the republic of Korea. BMC Musculoskelet Disord. 2023;24:878. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 58. | González García A, Sifuentes Giraldo WA, Blázquez Cañamero MÁ, Ahijón Lana M, Navas Elorza E, Vázquez Díaz M. Multifocal osteonecrosis associated with human immunodeficiency virus infection. Reumatol Clin. 2012;8:361-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 59. | Green KR, Hernandez-Jimenez JM, Isache CL, Jacob R. Avascular necrosis: a growing concern for the HIV population. BMJ Case Rep. 2018;2018:bcr2017221678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 60. | Graham SM, Lubega N, Mkandawire N, Harrison WJ. Total hip replacement in HIV-positive patients. Bone Joint J. 2014;96-B:462-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 35] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 61. | Zhang C, Li Y, Lin Y, Zhang X, Huang Z, Bai G, Wan Y, Zhang W, Fang X, Li W. Joint Replacement Surgeries Can Be Safely Performed in HIV Patients. J Clin Med. 2022;12:293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 62. | Morse CG, Mican JM, Jones EC, Joe GO, Rick ME, Formentini E, Kovacs JA. The incidence and natural history of osteonecrosis in HIV-infected adults. Clin Infect Dis. 2007;44:739-748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 82] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 63. | Ott SM. Bone strength: more than just bone density. Kidney Int. 2016;89:16-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 64. | Binkley N, Morin SN, Martineau P, Lix LM, Hans D, Leslie WD. Frequency of normal bone measurement in postmenopausal women with fracture: a registry-based cohort study. Osteoporos Int. 2020;31:2337-2344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 65. | Stone KL, Seeley DG, Lui LY, Cauley JA, Ensrud K, Browner WS, Nevitt MC, Cummings SR; Osteoporotic Fractures Research Group. BMD at multiple sites and risk of fracture of multiple types: long-term results from the Study of Osteoporotic Fractures. J Bone Miner Res. 2003;18:1947-1954. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 856] [Cited by in RCA: 730] [Article Influence: 31.7] [Reference Citation Analysis (2)] |
| 66. | Leslie WD, Luo Y, Yang S, Goertzen AL, Ahmed S, Delubac I, Lix LM. Fracture Risk Indices From DXA-Based Finite Element Analysis Predict Incident Fractures Independently From FRAX: The Manitoba BMD Registry. J Clin Densitom. 2019;22:338-345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 67. | Yin MT, Shiau S, Rimland D, Gibert CL, Bedimo RJ, Rodriguez-Barradas MC, Harwood K, Aschheim J, Justice AC, Womack JA. Fracture Prediction With Modified-FRAX in Older HIV-Infected and Uninfected Men. J Acquir Immune Defic Syndr. 2016;72:513-520. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 43] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 68. | Yang J, Sharma A, Shi Q, Anastos K, Cohen MH, Golub ET, Gustafson D, Merenstein D, Mack WJ, Tien PC, Nieves JW, Yin MT. Improved fracture prediction using different fracture risk assessment tool adjustments in HIV-infected women. AIDS. 2018;32:1699-1706. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 69. | Gazzola L, Comi L, Savoldi A, Tagliabue L, Del Sole A, Pietrogrande L, Bini T, d'Arminio Monforte A, Marchetti G. Use of the FRAX equation as first-line screening of bone metabolism alteration in the HIV-infected population. J Infect Dis. 2010;202:330-1; author reply 331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 70. | Castro AP, Brito KB, Oliva TDR, Silva IMS, Kato BS, Caldeira GAM, Santos FM, Libonati RMF. Fracture Risk with Modified FRAX in Men Living with HIV. J Clin Densitom. 2024;27:101460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 71. | Jadžić J, Đurić M. Structural basis of increased bone fragility in aged individuals: Multi-scale perspective. Med Istraživanja. 2024;57:67-74. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 72. | Olali AZ, Carpenter KA, Myers M, Sharma A, Yin MT, Al-Harthi L, Ross RD. Bone Quality in Relation to HIV and Antiretroviral Drugs. Curr HIV/AIDS Rep. 2022;19:312-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (1)] |
| 73. | Sarkar S, Mitlak BH, Wong M, Stock JL, Black DM, Harper KD. Relationships between bone mineral density and incident vertebral fracture risk with raloxifene therapy. J Bone Miner Res. 2002;17:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 358] [Cited by in RCA: 284] [Article Influence: 11.8] [Reference Citation Analysis (1)] |
| 74. | Yin MT, Shu A, Zhang CA, Boutroy S, McMahon DJ, Ferris DC, Colon I, Shane E. Trabecular and cortical microarchitecture in postmenopausal HIV-infected women. Calcif Tissue Int. 2013;92:557-565. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 75. | Calmy A, Chevalley T, Delhumeau C, Toutous-Trellu L, Spycher-Elbes R, Ratib O, Zawadynski S, Rizzoli R. Long-term HIV infection and antiretroviral therapy are associated with bone microstructure alterations in premenopausal women. Osteoporos Int. 2013;24:1843-1852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 37] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 76. | Biver E, Calmy A, Delhumeau C, Durosier C, Zawadynski S, Rizzoli R. Microstructural alterations of trabecular and cortical bone in long-term HIV-infected elderly men on successful antiretroviral therapy. AIDS. 2014;28:2417-2427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 77. | Sellier P, Ostertag A, Collet C, Trout H, Champion K, Fernandez S, Lopes A, Morgand M, Clevenbergh P, Evans J, Souak S, de Vernejoul MC, Bergmann JF. Disrupted trabecular bone micro-architecture in middle-aged male HIV-infected treated patients. HIV Med. 2016;17:550-556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 78. | Foreman SC, Wu PH, Kuang R, John MD, Tien PC, Link TM, Krug R, Kazakia GJ. Factors associated with bone microstructural alterations assessed by HR-pQCT in long-term HIV-infected individuals. Bone. 2020;133:115210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 79. | Macdonald HM, Maan EJ, Berger C, Dunn RA, Côté HCF, Murray MCM, Pick N, Prior JC. Deficits in bone strength, density and microarchitecture in women living with HIV: A cross-sectional HR-pQCT study. Bone. 2020;138:115509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 80. | Tan DH, Raboud J, Szadkowski L, Szabo E, Hu H, Wong Q, Cheung AM, Walmsley SL. Novel imaging modalities for the comparison of bone microarchitecture among HIV+ patients with and without fractures: a pilot study. HIV Clin Trials. 2017;18:28-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 81. | Diamond T, Depczynski B. Acquired osteosclerosis associated with intravenous drug use and hepatitis C infection. Bone. 1996;19:679-683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 82. | Serraino C, Melchio R, Silvestri A, Borretta V, Pomero F, Fenoglio L. Hepatitis C-associated osteosclerosis: a new case with long-term follow-up and a review of the literature. Intern Med. 2015;54:777-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 83. | Miyamura N, Nishida S, Itasaka M, Matsuda H, Ohtou T, Yamaguchi Y, Inaba D, Tamiya S, Nakano T. A case of hepatitis C-associated osteosclerosis: accelerated bone turnover controlled by pulse steroid therapy. Endocrinol Diabetes Metab Case Rep. 2016;2016:16-0097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 84. | Diamond TH, Stiel D, Lunzer M, McDowall D, Eckstein RP, Posen S. Hepatic osteodystrophy. Static and dynamic bone histomorphometry and serum bone Gla-protein in 80 patients with chronic liver disease. Gastroenterology. 1989;96:213-221. [PubMed] |
| 85. | Stellon AJ, Webb A, Compston JE. Bone histomorphometry and structure in corticosteroid treated chronic active hepatitis. Gut. 1988;29:378-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 50] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 86. | Klco JM, Geng B, Brunt EM, Hassan A, Nguyen TD, Kreisel FH, Lisker-Melman M, Frater JL. Bone marrow biopsy in patients with hepatitis C virus infection: spectrum of findings and diagnostic utility. Am J Hematol. 2010;85:106-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 27] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 87. | Hiramatsu R, Ubara Y, Sawa N, Hasegawa E, Kawada M, Imafuku A, Sumida K, Hoshino J, Takaichi K. Bone Histology of Two Cases with Osteomalacia Related to Low-dose Adefovir. Intern Med. 2016;55:3013-3019. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (1)] |
| 88. | Wakolbinger R, Muschitz C, Scheriau G, Bodlaj G, Kocijan R, Feichtinger X, Schanda JE, Haschka J, Resch H, Pietschmann P. Bone microarchitecture and bone turnover in hepatic cirrhosis. Osteoporos Int. 2019;30:1195-1204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 89. | Lo Re V 3rd, Carbonari DM, Newcomb CW, Torgersen J, Weinstein EJ, Smith SM, Brecker KL, Liu XS, Kostman JR, Trooskin S, Hubbard RA, Baker JF, Zemel BS, Leonard MB. Changes in Bone Microarchitecture and Inflammatory Cytokines After Cure of Chronic Hepatitis C Infection With Direct-Acting Antiviral Therapy. Open Forum Infect Dis. 2025;12:ofaf571. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 90. | Ravazzano L, Colaianni G, Tarakanova A, Xiao YB, Grano M, Libonati F. Multiscale and multidisciplinary analysis of aging processes in bone. NPJ Aging. 2024;10:28. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 23] [Reference Citation Analysis (1)] |
| 91. | Micheletti C, Shah FA. Bone hierarchical organization through the lens of materials science: Present opportunities and future challenges. Bone Rep. 2024;22:101783. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 92. | Spirlandeli AL, Dick-de-Paula I, Zamarioli A, Jorgetti V, Ramalho LNZ, Nogueira-Barbosa MH, Volpon JB, Jordão AA, Cunha FQ, Fukada SY, de Paula FJA. Hepatic Osteodystrophy: The Mechanism of Bone Loss in Hepatocellular Disease and the Effects of Pamidronate Treatment. Clinics (Sao Paulo). 2017;72:231-237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 93. | Bedimo R, Cutrell J, Zhang S, Drechsler H, Gao A, Brown G, Farukhi I, Castanon R, Tebas P, Maalouf NM. Mechanisms of bone disease in HIV and hepatitis C virus: impact of bone turnover, tenofovir exposure, sex steroids and severity of liver disease. AIDS. 2016;30:601-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 94. | Ó Breasail M, Madanhire T, Kahari C, Ebeling PR, Simms V, Micklesfield LK, Ferrand RA, Gregson CL, Ward KA. Trabecular bone deficits predominate in the appendicular skeleton of midlife women living with HIV: findings from a cross-sectional study in Zimbabwe. J Bone Miner Res. 2025;40:454-462. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 95. | Hileman CO, Labbato DE, Storer NJ, Tangpricha V, McComsey GA. Is bone loss linked to chronic inflammation in antiretroviral-naive HIV-infected adults? A 48-week matched cohort study. AIDS. 2014;28:1759-1767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 55] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 96. | Du ZX, Wang L, Chen MY, Hu YF, Zhong YD, Xiong QF, Li Y, Yang YF. Association between Chronic Hepatitis B and Osteoporosis: a two-sample mendelian randomization study. J Infect Dev Ctries. 2025;19:446-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 97. | Weinstein EJ, Carbonari DM, Newcomb CW, Torgersen J, Smith SM, Brecker KL, Liu XS, Kostman JR, Trooskin S, Hubbard RA, Baker JF, Zemel BS, Leonard MB, Lo Re V 3rd. Abnormal Trabecular and Cortical Bone Microarchitecture in Chronic Hepatitis C Infection and Associations With Select Inflammatory Cytokines. Open Forum Infect Dis. 2025;12:ofaf102. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 98. | Xia S, Qin X, Wang J, Ren H. Advancements in the pathogenesis of hepatic osteodystrophy and the potential therapeutic of mesenchymal stromal cells. Stem Cell Res Ther. 2023;14:359. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 99. | Zampino R, Marrone A, Restivo L, Guerrera B, Sellitto A, Rinaldi L, Romano C, Adinolfi LE. Chronic HCV infection and inflammation: Clinical impact on hepatic and extra-hepatic manifestations. World J Hepatol. 2013;5:528-540. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 179] [Cited by in RCA: 179] [Article Influence: 13.8] [Reference Citation Analysis (1)] |
| 100. | Aktas GE, Sarikaya A, Kandemir O. Hepatitis C Virus-related Arthritis: Bone Scintigraphic Appearances. Indian J Nucl Med. 2017;32:30-32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (1)] |
| 101. | Singh KP, Crane M, Audsley J, Avihingsanon A, Sasadeusz J, Lewin SR. HIV-hepatitis B virus coinfection: epidemiology, pathogenesis, and treatment. AIDS. 2017;31:2035-2052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 198] [Article Influence: 22.0] [Reference Citation Analysis (2)] |
| 102. | Cheng Z, Lin P, Cheng N. HBV/HIV Coinfection: Impact on the Development and Clinical Treatment of Liver Diseases. Front Med (Lausanne). 2021;8:713981. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (1)] |