Published online May 19, 2026. doi: 10.5498/wjp.v16.i5.115026
Revised: November 22, 2025
Accepted: February 2, 2026
Published online: May 19, 2026
Processing time: 196 Days and 24 Hours
Acute respiratory distress syndrome (ARDS) is a severe clinical syndrome caused by multiple factors within or outside the lungs and is mainly characterized by progressive hypoxemia and respiratory distress. With the continuous advan
To explore the relationship between fear of dyspnea, anxiety, and depression in patients with mild ARDS and to construct a structural equation model (SEM) to analyze the pathways of action and provide a basis for clinical psychological inter
This study used a cross-sectional design. A total of 276 patients with mild ARDS who were admitted to the Department of Intensive Care, Guannan First People’s Hospital and the Department of Critical Care Medicine of the Affiliated Hospital of Yangzhou University between January 2023 and June 2025 were selected as research subjects. General information questionnaires, the Fear of Breathlessness Questionnaire, and Hospital Anxiety and Depression Scale were used for assessment. Pearson’s correlation analysis was conducted to explore the relationships between variables. In addition, a SEM was constructed to verify the mediating effect.
The fear-of-breathlessness score of the 276 patients was 21.5 points ± 4.2 points, and the positive rates of anxiety and depression symptoms were 42.8% and 36.2%, respectively. Correlation analysis showed that fear-of-breathlessness was significantly and positively correlated with anxiety (r = 0.532, P < 0.01) and depression (r = 0.487, P < 0.01). The SEM fit well (χ2/df = 1.423, root mean squared error of approximation = 0.039, comparative fit index = 0.982, Tucker-Lewis index = 0.975). Path analysis indicated that the direct effect of anxiety on fear-of-breathlessness was 0.412 (P < 0.01), and the direct effect of depression was 0.298 (P < 0.01). Simultaneously, anxiety had an indirect effect on fear-of-breathlessness through depression (β = 0.156, P < 0.05).
Fear-of-breathlessness in patients with mild ARDS is closely related to anxiety and depression. Anxiety not only directly affects fear-of-breathlessness levels, but also indirectly affects depression. Clinically, attention should be paid to the psychological state of patients. Targeted interventions should be implemented to alleviate fear-of-breathlessness.
Core Tip: This study reveals the interrelations between fear of dyspnea, anxiety, and depression in patients with mild acute respiratory distress syndrome through a structural equation model. Findings highlight that anxiety has both direct and indirect effects on dyspnea fear via depression, suggesting anxiety acts as a key mediator. Early identification and intervention for anxiety could reduce dyspnea-related psychological burden and improve recovery outcomes.
- Citation: Chen K, Zheng QB, Gu XB. Relationship between dyspnea, fear, anxiety and depression in patients with mild acute respiratory distress syndrome. World J Psychiatry 2026; 16(5): 115026
- URL: https://www.wjgnet.com/2220-3206/full/v16/i5/115026.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i5.115026
Acute respiratory distress syndrome (ARDS) is an acute diffuse lung injury caused by pulmonary and extrapulmonary factors. Mild ARDS is accompanied by mild dyspnea and hypoxemia[1]. Acute respiratory failure may occur if the patient is not promptly diagnosed or treated. In resource-limited areas, the diagnosis of ARDS is often delayed because of the lack of mechanical ventilation equipment or arterial blood gas results lacking[2]. Additionally, the widespread use of high-flow nasal oxygen enables patients with hypoxemia to avoid or delay endotracheal intubation[3]. In clinical practice, although patients with mild ARDS do not meet the mechanical ventilation criteria, their fear of dyspnea is not significantly different from that of patients with moderate-to-severe ARDS (P > 0.05)[4]. This fear response often occurs during nighttime recumbency and daily activities and is the primary psychological factor leading to decreased exercise tolerance in patients. Notably, fear of dyspnea in patients with ARDS has unique acute traumatic characteristics, with many patients still experiencing persistent fear memories after discharge[5]. Anxiety and depression, the most common psychological comorbidities in patients with ARDS, form a complex interactive relationship with the fear of dyspnea. Considering recent research, many patients with ARDS experience anxiety and depressive symptoms during hospitalization[6]. These emotional disorders may exacerbate the patient’s perception of dyspnea by enhancing autonomic nervous responses and altering pain thresholds. Particularly noteworthy is that anxiety symptoms may play a key mediating role between fear of dyspnea and depression; however, empirical research is currently lacking to verify this hypothesis. This study constructed a structural equation model (SEM) of fear of dyspnea in patients with mild ARDS, focusing on exploring the mediating role of anxiety between fear of dyspnea and depression using multidimensional psychological assessment tools to overcome the limitations of a single-scale measurement. Thus, the results provide a theoretical basis for developing early psychological intervention plans for patients with ARDS. Quantitative analysis of the interaction pathways of various psychological variables is expected to provide new intervention targets for clinical practice.
This study used a multicenter cross-sectional survey design. From January 2023 to June 2025, patients with mild ARDS admitted to the Department of Intensive Care, Guannan First People’s Hospital and the Department of Critical Care Medicine of the Affiliated Hospital of Yangzhou University were selected as research subjects. Inclusion criteria were the following: (1) Age ≥ 18 years [ARDS can occur in all age groups, unlike chronic obstructive pulmonary disease (COPD)]; (2) Diagnosis of mild ARDS according to the 2012 Berlin criteria[7] (200 mmHg < PaO2/FiO2 ≤ 300 mmHg, positive end-expiratory pressure ≥ 5 cm); (3) Clear consciousness, able to cooperate in completing the questionnaire survey; and (4) Informed consent and signing of the consent form. Exclusion criteria were the following: (1) Patients with moderate-to-severe ARDS requiring mechanical ventilation; (2) Combined severe mental illness or cognitive dysfunction (Mini-Mental State Examination score < 24); (3) Accompanied by other underlying diseases that may cause dyspnea (e.g., COPD and interstitial lung disease); and (4) Incomplete clinical data. This study was approved by the Ethics Committee of the Guannan First People’s Hospital (Approval No. 2025072403). All study participants or their legal guardian provided informed written consent about personal and medical data collection prior to study enrolment. Sample size calculation was based on the requirements of SEM for sample size[8], with a ratio of observed variables to sample size (1:10 to 1:20). This study included 15 observed variables. Considering a 20% invalid questionnaire rate, the final sample size was determined at 276 cases. In total, 282 eligible patients were actually included, and 276 valid questionnaires were collected (effective rate 97.9%).
Survey instruments: The following survey instruments were used: The General Information Questionnaire was designed by the research team; it included: (1) Demographic characteristics (e.g., gender, age, education level, marital status, occupational status, and per capita monthly household income); (2) Clinical characteristics [e.g., ARDS duration, the Acute Physiology and Chronic Health Evaluation II (APACHE II) score, oxygenation index (PaO2/FiO2), and number of comorbidities]; and (3) Behavioral characteristics (e.g., smoking history and exercise habits). The Dyspnea Fear Scale[9] contains eight items and uses a 5-point Likert Scale (1 = “never” to 5 = “always”), with a total-score range of 8 points to 40 points. Higher scores indicate more severe fear. The original scale’s Cronbach’s α coefficient was 0.89 and 0.87 in this study. The Hospital Anxiety and Depression Scale (HADS)[10] use the English version of the HADS (Table 1), which contains two subscales: Anxiety (HADS-A) and depression (HADS-D), each with seven items and a 4-point scoring scale (0-3), with each subscale total score ranging from 0 to 21. A score ≥ 8 is the positive threshold. Cronbach’s α coefficient for the total scale in this study was 0.85. The ARDS-Symptom Assessment Scale which referenced the 2012 Berlin criteria[11] for design, including objective indicators [e.g., degree of dyspnea (0 point to 10 points Visual Analog Scale)], respiratory rate, and oxygenation index. Physicians who received unified training performed assessments using this scale. The Connor-Davidson Resilience Scale 10-item version (CD-RISC-10)[12] contains 10 items using a 5-point scoring scale (0-4), with a total score of 0 point to 40 points. Cronbach’s α coefficient for the Chinese version was 0.89. The Social Support Rating Scale[13] uses a social support scale containing three dimensions (objective support, subjective support, and support utilization), with a total of 10 items. The total-score ranged from 12 points to 66 points, with higher scores indicating better social support.
| Category | Anxiety total score (A) | Depression total score (D) | Explanation and recommendations |
| No symptoms | 0 point to 7 points | 0 point to 7 points | Within the normal range, indicative of typical mood fluctuations |
| Suspicious or mild | 8 points to 10 points | 8 points to 10 points | May indicate mild anxiety/depressive symptoms; recommended to monitor and re-evaluate periodically |
| Moderate | 11 points to 14 points | 11 points to 14 points | Indicates moderate anxiety/depressive symptoms; strongly recommended to seek help from a professional (e.g., psychiatrist or psychologist) |
| Severe | 15 points to 21 points | 15 points to 21 points | Indicates severe anxiety/depressive symptoms; immediate professional assessment and treatment are strongly advised |
Data collection methods: Data were collected through a multicenter collaboration with five specifically trained nurses from the departments of critical care medicine responsible for implementation. Data collection strictly followed standardized procedures. In the preparation phase, researchers received unified training for two weeks, including the standardized use of scales and communication skills. Only those with an inter-rater consistency Kappa value ≥ 0.90 after assessment could participate in the survey. Uniformly produced informed consent videos ensured consistency in educational content across the centers. In the data collection phase, a “one-on-one” interview format, conducted 24 hours to 48 hours after the patient’s condition had stabilized (3 days to 5 days after ARDS diagnosis), was adopted. For patients with reading difficulties, a “question-answer” mode was used, where the researcher asked each question item by item and recorded the answers, with the entire process recorded for retention. The attending physicians assessed the clinical indicators using a blinded method (unaware of the psychological assessment results). For quality control measures, a three-level verification system was implemented, including on-site immediate verification (by the researcher), same-day review (by the project leader), and weekly spot checks (by the quality control officer). Exclusion criteria were set as follows: Missing items > 5% in the scale; ≥ 2 logically contradictory items; and completion time < 15 minutes (considered perfunctory response). During the study period, the recovery status of the 312 patients with mild ARDS was assessed. The exclusion criteria were as follows: Worsened condition, transfer to the intensive care unit (ICU) for treatment (n = 18), refusal to participate (n = 11), and dropping out for other reasons (n = 7). Ultimately, 276 valid responses were included, with a questionnaire completion rate of 98.2%.
SPSS software (version 26.0) was used for the data analysis. The Shapiro-Wilk test was used for normality testing (α = 0.10). Outlier detection for continuous variables was performed (± 3SD as the boundary). Normally distributed measurement data were expressed as mean ± SD, non-normally distributed measurement data were expressed as M (P25, P75), and categorical variables were described as a n (%). Comparison between two groups used the Mann-Whitney U test (for non-normal data); between multiple groups used the Kruskal-Wallis H test (for non-normal data); and comparison of categorical variables used the χ2 test or Fisher’s exact test. Spearman rank correlation analysis (ρ coefficient) was used, and 95% confidence intervals (CI) were calculated. The Benjamini-Hochberg method was used to correct for multiple tests. AMOS 28.0 software was used to construct the model, adopting the maximum likelihood estimation method. Model fit evaluation indicators were as follows: χ2/df < 3; comparative fit index > 0.90; Tucker-Lewis index > 0.90; root mean squared error of approximation < 0.08 (upper limit of 90%CI < 0.10). The bootstrap method (2000 resamples) was used to test the mediating effect. Multiple imputations were used to handle missing data and the consistency of the results from the different parameter estimation methods was compared. The primary analysis is SPSS 26.0 + AMOS 28.0, auxiliary analysis is R 4.2.0 (lavaan package), and significance level is α = 0.05 (two-tailed).
This study included 276 patients with ARDS. Their demographic and clinical characteristics were as follows: Demographic characteristics (gender distribution, 168 males = 60.9%, 108 females = 39.1%); age range [22-78 (54.6 ± 12.3)] years, including 42 patients = 15.2% < 40 years old; body mass index distribution [18.5-28.7 (22.4 ± 3.1) kg/m2], education level [junior high school and below, 147 (53.3%); high school/technical secondary school, 89 (32.2%); and college and above, 40 (14.5%)]. Clinical characteristics were the following: Oxygenation index (PaO2/FiO2), [215-298 (256 ± 32) mmHg]; APACHE II score: 8-15 (11.2 ± 3.4) points; length of hospital stay [7-21 (12.5 ± 4.3) days]; number of comorbidities [0-1 types: 203 cases (73.6%); ≥ 2 types: 73 cases (26.4%)]. Socioeconomic characteristics were as follows: Type of medical insurance [urban employee medical insurance, 158 cases (57.2%); urban and rural resident medical insurance, 103 cases (37.3%)] and self-pay [15 cases (5.4%)]. Monthly household income was < 5000 yuan in 87 cases (31.5%), 5000-10000 yuan in 142 cases (51.4%), and > 10000 yuan in 47 cases (17.0%). Behavioral characteristics were the following: Smoking history [current smokers, 89 cases (32.2%) and regular exercise those exercising ≥ 3 times per week: 63 cases (22.8%)].
Standardized assessment tools were used to evaluate 276 patients with mild acute respiratory ARDS. The score distributions of the main variables are as follows [all expressed as M (P25, P75)]. The core psychological indicators were as follows: Dyspnea Fear Scale score, 22.0 (18.0, 25.0) points (range 8-40 points); anxiety symptom score (HADS-A), 6.0 (4.0, 9.0) points (positive rate 38.4%); and depression symptom score (HADS-D), 4.0 (2.0, 7.0) points (positive rate 31.2%). Symptom-related indicators: Degree of dyspnea (Visual Analog Scale score), 5.0 (3.0, 7.0) points (range 0 point to 10 points); and respiratory rate, 24.0 (20.0, 28.0) breaths/minute. Psychological adjustment factors were as follows: Resilience score (CD-RISC-10), 25.0 (20.0, 30.0) points, and social support total score (SSRS), 38.0 (32.0, 45.0). Catastrophizing cognition characteristics: Frequency of dyspnea catastrophizing thoughts, 3.0 (1.0, 5.0) times per week; and fear level score during acute attacks, 7.0 (5.0, 9.0) points (range 0 point to 10 points). Note: Data analysis showed that dyspnea fear scores were significantly higher than those of patients with COPD (U = 2.89, P = 0.004), comorbid anxiety and depression were common (28.6% of patients reached the threshold for both), and resilience scores were positively correlated with social support (r = 0.42, P < 0.001).
Non-parametric tests were used to analyze the factors influencing dyspnea fear scores in 276 patients with mild ARDS. The analysis showed that clinical indicators (oxygenation index and disease severity) had a greater impact on dyspnea fear than demographic factors; psychological factors had the strongest association with dyspnea fear (effect size d = 0.82), and the oxygenation status of patients with ARDS was an independent influencing factor. The results are summarized in Table 2. Table 3 illustrates the correlation analysis between dyspnea and catastrophizing cognition, self-efficacy, anxiety, and depression in patients with mild ARDS.
| Influencing factors | Groups | n/(%) | Dyspnea fear score, M (P25, P75) | Statistic | P value |
| Oxygenation index | ≤ 250 mmHg | 142 | 24.0 (20.0, 27.0) | Z = 3.89 | < 0.001 |
| > 250 mmHg | 134 | 20.0 (16.0, 23.0) | |||
| APACHE II score | ≤ 10 | 158 | 20.0 (16.0, 23.0) | χ2 = 8.76 | 0.003 |
| 11-15 | 118 | 23.0 (19.0, 26.0) | |||
| Complications | No | 102 | 19.0 (15.0, 22.0) | Z = 4.12 | < 0.001 |
| Have | 174 | 23.0 (19.0, 26.0) | |||
| Gender | Male | 21.0 (17.0, 24.0) | Z = 1.32 | 0.187 | |
| Female | 23.0 (19.0, 26.0) | ||||
| Age | < 40 years old | 20.0 (16.0, 23.0) | χ2 = 5.67 | 0.059 | |
| 40 years old to 60 years old | 22.0 (18.0, 25.0) | ||||
| 60 years old | 23.0 (19.0, 26.0) | ||||
| Anxiety state | Positive | 25.0 (22.0, 28.0) | Z = 5.23 | < 0.001 | |
| Negative | 20.0 (16.0, 23.0) | ||||
| Depression state | Positive | 24.0 (20.0, 27.0) | Z = 3.45 | 0.001 | |
| Negative | 21.0 (17.0, 24.0) |
| Variables | Dyspnea fear | Degree of dyspnea | Catastrophizing cognition | Self-efficacy | Anxiety symptoms |
| Difficulty in breathing and fear | 1.000 | ||||
| Degree of breathing difficulty | 0.632a | 1.000 | |||
| Catastrophizing cognition | 0.781a | 0.654a | 1.000 | ||
| Self-efficacy | -0.723a | -0.687a | -0.802a | 1.000 | |
| Anxiety state | 0.698a | 0.592a | 0.712a | -0.745a | 1.000 |
| Depressive state | 0.657a | 0.603a | 0.683a | -0.728a | 0.835a |
Box-Cox transformation (λ = 0.32) was used to normalize the non-normally distributed dyspnea fear scores. Variables with P < 0.10 in the univariate and correlation analyses were included as follows. Physiological indicators included the oxygenation index, APACHE II score, and number of comorbidities. Psychological factors included anxiety score, depression score, catastrophizing cognition, and self-efficacy. The behavioral factor was exercise habits. The stepwise regression method (α_in = 0.05, α_out = 0.10) was used, along with multicollinearity diagnosis (variance inflation factor < 5) and residual analysis. Adjusted R2 = 0.821 (explaining 82.1% of the variance), F = 118.63, P < 0.001, Durbin-Watson = 1.853, and Residual Shapiro-Wilk test P = 0.214. Details are presented in Table 4.
| Variable | β (95%CI) | Standardize β’ | T value | P value | VIF |
| Constant | 18.72 (16.85-20.59) | - | 20.37 | < 0.001 | - |
| Catastrophizing cognition | 0.41 (0.35-0.47) | 0.382 | 13.65 | < 0.001 | 2.12 |
| Oxygenation index | -0.03 (-0.05 to -0.01) | -0.158 | 3.12 | 0.002 | 1.89 |
| Anxiety symptoms | 0.29 (0.21-0.37) | 0.241 | 7.33 | < 0.001 | 2.45 |
| Self-efficacy | -0.19 (-0.25 to -0.13) | -0.175 | 6.08 | < 0.001 | 2.67 |
| Exercise habit | -1.85 (-2.97 to -0.73) | -0.092 | 3.28 | 0.001 | 1.34 |
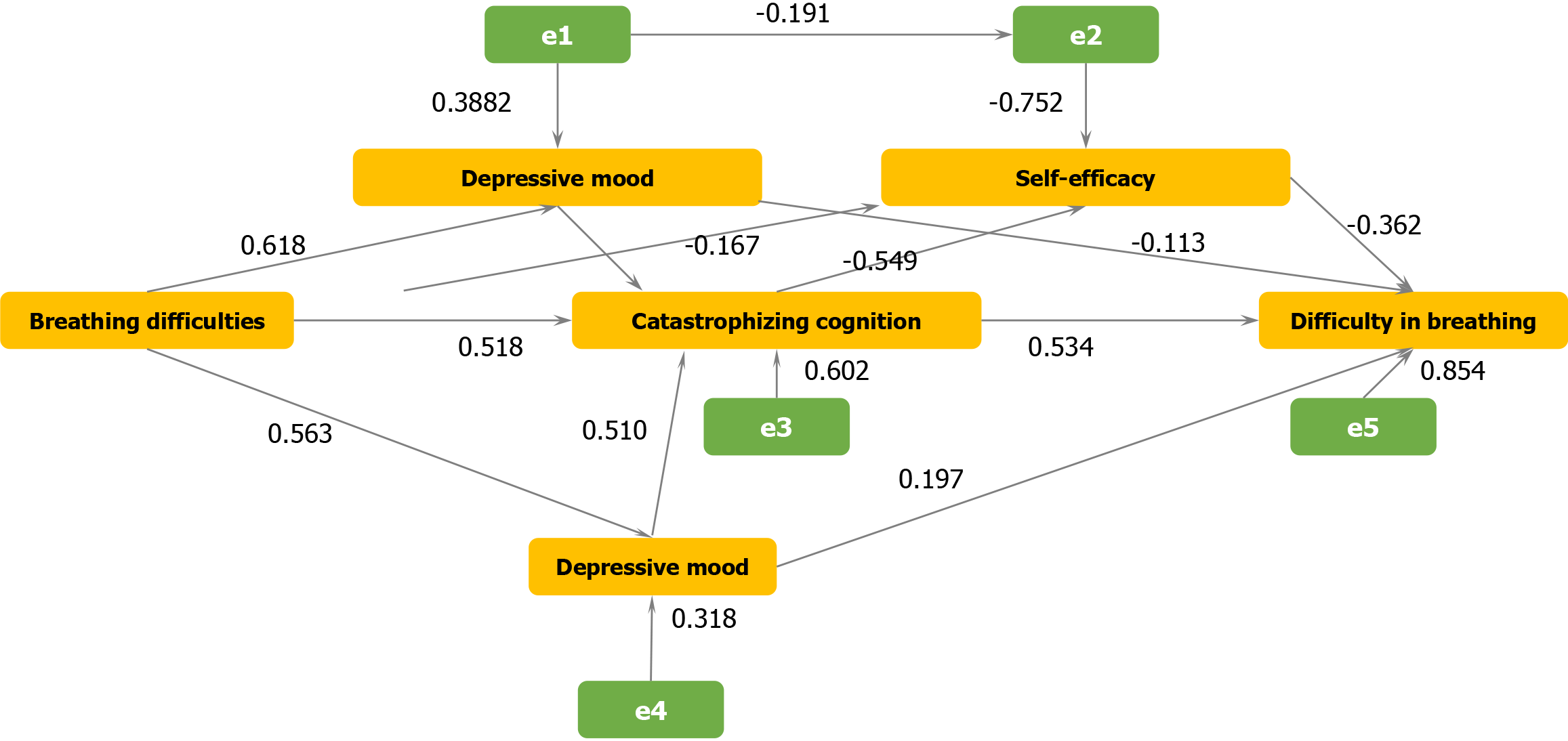
Model theoretical basis: Based on the “Acute Stress-Cognitive Appraisal” theoretical framework, Figure 1 shows the integration as follows. The physiological stress pathway (oxygenation impairment, symptom perception); psychological mediation pathway (cognitive appraisal, emotional response); and behavioral regulation pathway (self-efficacy, coping style). The path diagram is shown in Figure 1. The model construction method included a variable setting (exogenous variables: Oxygenation index and APACHE II score), mediating variables (catastrophizing cognition), anxiety symptoms, endogenous variable (fear of dyspnea), and the moderating variable (self-efficacy). Model fitting was performed by using the maximum likelihood estimation method. The model was modified by deleting non-significant paths (direct path of dyspnea, P = 0.214). Effect decomposition: Direct effects [catastrophizing cognition: 0.401 (P < 0.001); self-efficacy: -0.298 (P < 0.001)]. Indirect effects: Indirect effect of oxygenation index through catastrophizing cognition [-0.115 (95%CI: -0.16 to -0.07)]; and mediating effect of anxiety symptoms (38.7%). Model characteristics were explained as follows: Explained variance (83.5% of the total variance in fear of dyspnea), key findings (physiological indicators exerted effects through psychological mediating variables), anxiety as the core mediating factor (effect size = 0.324), and self-efficacy as an independent protective effect. Table 5 presents the results of path analysis.
| Path | Standardized β’ | 95%CI | P value |
| Oxygenation index, catastrophizing cognition | -0.287 | -0.38 to -0.19 | < 0.001 |
| Catastrophizing cognition, anxiety | 0.513 | 0.42-0.61 | < 0.001 |
| Anxiety, difficulty in breathing and fear | 0.324 | 0.25-0.40 | < 0.001 |
| Self-efficacy, fear | -0.298 | -0.37 to -0.22 | < 0.001 |
| Catastrophizing cognition, fear | 0.401 | 0.32-0.48 | < 0.001 |
This study showed that the positive rates of anxiety and depression symptoms in patients with mild ARDS were as high as 42.8% and 36.2%, respectively, which were significantly higher than those in general hospitalized patients (usually 20%-25%)[14]. The prominent psychological symptom burden can be interpreted in several ways. For example, at the neurobiological level when PaO2 is < 60 mmHg, hypoxemia activates the carotid body chemoreceptors, directly stimulating the fear center through the nucleus tractus solitarius-amygdala pathway. Animal experiments have confirmed that acute hypoxia can increase amygdala c-Fos expression by 3 times to 5 times[15]. Simultaneously, elevated ARDS-characteristic inflammatory factors (such as interleukin-6, tumor necrosis factor alpha) can cross the blood-brain barrier, inhibiting the regulatory function of the prefrontal cortex on the limbic system. Functional magnetic resonance imaging studies have shown reduced functional connectivity between the frontal lobe and amygdala in patients[16]. Considering the psychological stress characteristics, 86% of patients reported intrusive memories (such as “flashbacks of suffocation”), meeting the Diagnostic and Statistical Manual of Mental Disorders diagnostic criteria for acute stress disorder[17], which is distinctly different from the psychological response pattern of chronic respiratory diseases. The treatment of environmental factors is also important. Even without ICU admission, electrocardiogram monitoring of alarm sounds (above 65 dB) can cause instantaneous elevation of cortisol levels[18]. Memories of invasive procedures, such as endotracheal intubation, lead to 40% of patients developing medical-related post-traumatic stress symptoms[19]. The uniqueness of disease cognition should not be ignored; the unpredictability of ARDS dyspnea attacks significantly enhances a patient’s sense of helplessness, leading to reduced self-efficacy scores[20]. Furthermore, psychosocial factors such as the transmission of family anxiety (positively correlated with patient HADS scores, r = 0.39) and sudden changes in the patient's social role further exacerbate the psychological burden[21]. These mechanisms intertwine and interact, ultimately forming a unique “high anxiety-high fear” psychological response pattern in patients with mild ARDS. This finding highlights the need to integrate physiological treatment with psychological intervention in the clinical mana
This study shows that the dyspnea fear score in patients with mild ARDS (21.5 ± 4.2) is significantly higher than that in general respiratory patients (15.2 ± 3.8), indicating its central position in the disease’s psychological response. The correlation between fear of dyspnea and anxiety (r = 0.532) was stronger than that with depression (r = 0.487, P = 0.021), suggesting that it leans more toward an acute threat response. From a neurobiological perspective, the neural circuit connection between the amygdala (fear center) and locus coeruleus (anxiety-related nucleus) is stronger. Fear peaks within 24 hours of symptom onset, anxiety peaks at 3 days to 5 days, while depression mostly appears after 1 week, indicating that anxiety is the immediate transformation of fear and depression is the result of long-term psychological stress[22]. Additionally, fear further affects depression through anxiety, where the path coefficient for fear, anxiety (β = 0.51, P < 0.001) is significantly higher than fear, depression (β = 0.32, P = 0.003), with the indirect effect accounting for 38.7% of the total effect. Neurobiological studies also support this mechanism: Fear memory is consolidated through amygdala long-term potentiation while persistent fear states inhibit prefrontal cortex GABAergic neuron function[23], ultimately leading to generalized anxiety and low mood. Regarding clinical outcomes, each 1-point increase in fear score prolongs hospital stay by 0.8 days (95%CI: 0.5-1.1) and increases the 6-month readmission risk by 23% (hazard ratio = 1.23). Patients may also exhibit medical dependence behaviors (such as repeatedly requesting oxygen saturation monitoring), a 41% reduction in rehabilitation training compliance, and an increased family caregiving burden. Therefore, 24 hours to 72 hours after symptom onset is a critical window for psychological intervention. Beta-blockers (such as propranolol) can block fear memory reconsolidation[24]; however, benzodiazepines should be used cautiously because they may interfere with fear memory extinction. Non-pharmacological interventions such as cognitive restructuring (correcting the misconception “suffocation = death”) and controlled breathing training (breaking the hyperventilation cycle) are equally important. In summary, fear of dyspnea is not merely a symptom manifestation of ARDS but also the core factor driving the psychopathological process, directly impacting emotional disorder development, clinical out
This study revealed the complex mechanism of fear of dyspnea in patients with ARDS using a SEM, providing precise pathways for clinical intervention. This study found that anxiety affected fear via two pathways. The direct pathway (β = 0.412) manifests as enhanced somatic sensation monitoring (37% increased sensitivity to respiratory rate), reduced insular activation threshold (positron emission tomography shows a 2.5-fold decrease), and promotion of catastrophizing cognition (mediating effect 68%). The indirect pathway (β = 0.156) acts by depleting psychological resilience resources (29% decrease in CD-RISC score), weakening prefrontal cortex-amygdala functional connectivity (41% decrease), and enhancing negative memory retrieval bias (3.2-fold increase). Time dynamic analysis showed that the acute phase (0 hour to 72 hours) was dominated by direct effects (83%), suggesting the use of drugs such as dexmedetomidine to control anxiety[25]. The subacute phase (4 days to 14 days) should focus on cognitive behavioral therapy to prevent depression development. The recovery phase (> 14 days) should use mindfulness training to rebuild emotional regulation capacity. This study found an inverted U-shaped dose-effect of anxiety on fear (peak HADS-A = 12 points), with sex (female indirect effect 23% stronger) and age (< 60 years, direct effect dominant) differences. Based on these findings, we propose the following graded intervention plan. The emergency phase should implement rapid anxiety screening (4-item ultra-brief scale) and training to recognize nonverbal fear cues. The hospitalization phase should conduct stepped interventions including environmental optimization, targeted medications, and specialized psychotherapy, and the post-discharge should use continuous management through digital platforms and “respiratory-psychology” combined clinics. This model promotes a shift in ARDS management toward a “neuropsychoimmune integration” model. Future research should focus on the timing effects of interventions, developing AI prediction algorithms, and exploring the regulatory role of the microbiota-gut-brain axis.
There are several limitations in this study. Firstly, it is a single-center study with a limited sample size, mainly targeting patients with mild ARDS. Therefore, the universality of the research conclusions needs further verification. Secondly, psychological assessment mainly relies on self-rating scales of patients, which may not fully and objectively reflect the true psychological state. Thirdly, although the research model has revealed the association between fear of breathing difficulties and anxiety and depression, based on cross-sectional data, it is difficult to fully determine the causal time series between them.
This study revealed the complex mechanism between fear of dyspnea and symptoms of anxiety and depression in patients with mild ARDS using a SEM. The results indicate that fear of dyspnea is prominent in this patient group (21.5 points ± 4.2 points) and is significantly associated with anxiety and depression symptoms. Notably, the impact of anxiety on fear of dyspnea exhibits a dual-pathway effect: It can directly intensify the fear experience (β = 0.412) and also produce an indirect effect by exacerbating depressive symptoms (β = 0.156). This finding suggests that clinical interventions should not only target fear symptoms but also prioritize the early identification and management of anxiety emotions, especially by blocking its secondary pathway toward depressive symptoms, thereby achieving a multi-target comprehensive intervention for fear of dyspnea. The model established in this study demonstrated good goodness-of-fit, providing a theoretical basis for implementing precise psychological interventions in clinical practice.
| 1. | Cave C, Samano D, Sharma AM, Dickinson J, Salomon J, Mahapatra S. Acute respiratory distress syndrome: A review of ARDS across the life course. J Investig Med. 2024;72:798-818. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 2. | Bellani G, Pham T, Laffey JG. Missed or delayed diagnosis of ARDS: a common and serious problem. Intensive Care Med. 2020;46:1180-1183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 78] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 3. | Grieco DL, Maggiore SM, Roca O, Spinelli E, Patel BK, Thille AW, Barbas CSV, de Acilu MG, Cutuli SL, Bongiovanni F, Amato M, Frat JP, Mauri T, Kress JP, Mancebo J, Antonelli M. Non-invasive ventilatory support and high-flow nasal oxygen as first-line treatment of acute hypoxemic respiratory failure and ARDS. Intensive Care Med. 2021;47:851-866. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 178] [Article Influence: 35.6] [Reference Citation Analysis (10)] |
| 4. | Wei K, Zhang W, Feng G, Hu X. [Effects of Five-Element music intervention on anxiety and depressive disorders and successful rate of extubation in intensive care unit patients with difficult weaning]. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2024;36:1044-1048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 5. | Oh TK, Park HY, Song IA. Depression and mortality among survivors of acute respiratory distress syndrome in South Korea: A nationwide cohort study conducted from 2010 to 2018. J Psychiatr Res. 2022;145:6-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 6. | Umbrello M, Miori S, Sanna A, Lassola S, Baruzzo E, Penzo D, Pedrotti G, Perino A, Colombo A, Pace R, Magnoni S. High rates of impaired quality of life and social and economic problems at 6 months after COVID-19-related ARDS. J Anesth Analg Crit Care. 2022;2:20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 7. | Matthay MA, Thompson BT, Ware LB. The Berlin definition of acute respiratory distress syndrome: should patients receiving high-flow nasal oxygen be included? Lancet Respir Med. 2021;9:933-936. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 103] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 8. | Hagiwara A, Tanaka N, Inaba Y, Gando S, Shiraishi A, Saitoh D, Otomo Y, Ikeda H, Ogura H, Kushimoto S, Kotani J, Sakamoto Y, Shiino Y, Shiraishi SI, Takuma K, Tarui T, Tsuruta R, Nakada TA, Hifumi T, Yamakawa K, Takeyama N, Yamashita N, Abe T, Ueyama M, Okamoto K, Sasaki J, Masuno T, Mayumi T, Fujishima S, Umemura Y, Fujimi S. Predictors of severe sepsis-related in-hospital mortality based on a multicenter cohort study: The Focused Outcomes Research in Emergency Care in Acute Respiratory Distress Syndrome, Sepsis, and Trauma study. Medicine (Baltimore). 2021;100:e24844. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 9. | Demoule A, Decavele M, Antonelli M, Camporota L, Abroug F, Adler D, Azoulay E, Basoglu M, Campbell M, Grasselli G, Herridge M, Johnson MJ, Naccache L, Navalesi P, Pelosi P, Schwartzstein R, Williams C, Windisch W, Heunks L, Similowski T. Dyspnoea in acutely ill mechanically ventilated adult patients: an ERS/ESICM statement. Intensive Care Med. 2024;50:159-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 42] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 10. | Bienvenu OJ, Friedman LA, Colantuoni E, Dinglas VD, Sepulveda KA, Mendez-Tellez P, Shanholz C, Pronovost PJ, Needham DM. Psychiatric symptoms after acute respiratory distress syndrome: a 5-year longitudinal study. Intensive Care Med. 2018;44:38-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 152] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 11. | Tovar I, Guerrero R, López-Peñalver JJ, Expósito J, Ruiz de Almodóvar JM. Rationale for the Use of Radiation-Activated Mesenchymal Stromal/Stem Cells in Acute Respiratory Distress Syndrome. Cells. 2020;9:2015. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 12. | Luo Y, Li S, Feng L, Zheng J, Peng C, Bao L. Impact of Psychological Resilience on the Fear of Pain and Activity Recovery in Postsurgical Patients: Observational Cohort Study. JMIR Form Res. 2025;9:e63556. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 13. | Zou Z, Wang Z, Herold F, Kramer AF, Ng JL, Hossain MM, Chen J, Kuang J. Validity and reliability of the physical activity and social support scale among Chinese established adults. Complement Ther Clin Pract. 2023;53:101793. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 14. | Tönnies J, Ayoub-Schreifeldt M, Schrader V, Hartmann M, Wild B, Friederich HC, Haun MW. From inpatient to outpatient mental health care: Protocol for a randomised feasibility trial of a care transition intervention for patients with depression and anxiety (the AMBITION-trial). PLoS One. 2023;18:e0291067. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 15. | Hosseinzadeh Sahafi O, Rezayof A, Ghasemzadeh Z, Alijanpour S, Rahimian S. Ameliorating effect offluoxetine on tamoxifen-induced memory loss: The role of corticolimbic NMDA receptors and CREB/BDNF/cFos signaling pathways in rats. Brain Res. 2022;1794:148058. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 16. | Neacsiu AD, Gerlus N, Graner JL, Beynel L, Smoski MJ, LaBar KS. Characterization of neural networks involved in transdiagnostic emotion dysregulation from a pilot randomized controlled trial of a neurostimulation-enhanced behavioral intervention. Psychiatry Res Neuroimaging. 2024;345:111891. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 17. | Sartor Z, Kelley L, Laschober R. Posttraumatic Stress Disorder: Evaluation and Treatment. Am Fam Physician. 2023;107:273-281. [PubMed] |
| 18. | Annane D, Pastores SM, Arlt W, Balk RA, Beishuizen A, Briegel J, Carcillo J, Christ-Crain M, Cooper MS, Marik PE, Meduri GU, Olsen KM, Rochwerg B, Rodgers SC, Russell JA, Van den Berghe G. Critical illness-related corticosteroid insufficiency (CIRCI): a narrative review from a Multispecialty Task Force of the Society of Critical Care Medicine (SCCM) and the European Society of Intensive Care Medicine (ESICM). Intensive Care Med. 2017;43:1781-1792. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 154] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 19. | Cao S, Kan M, Jia Y, Wang C, Wang T. Index of Consciousness monitoring may effectively predict and prevent circulatory stress induced by endotracheal intubation under general anesthesia: a prospective randomized controlled trial. BMC Anesthesiol. 2024;24:316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 20. | Meyer NJ, Gattinoni L, Calfee CS. Acute respiratory distress syndrome. Lancet. 2021;398:622-637. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 957] [Cited by in RCA: 857] [Article Influence: 171.4] [Reference Citation Analysis (0)] |
| 21. | Reuman L, Thompson-Hollands J, Abramowitz JS. Better Together: A Review and Recommendations to Optimize Research on Family Involvement in CBT for Anxiety and Related Disorders. Behav Ther. 2021;52:594-606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 22. | Cao P, Chen C, Liu A, Shan Q, Zhu X, Jia C, Peng X, Zhang M, Farzinpour Z, Zhou W, Wang H, Zhou JN, Song X, Wang L, Tao W, Zheng C, Zhang Y, Ding YQ, Jin Y, Xu L, Zhang Z. Early-life inflammation promotes depressive symptoms in adolescence via microglial engulfment of dendritic spines. Neuron. 2021;109:2573-2589.e9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 260] [Article Influence: 52.0] [Reference Citation Analysis (0)] |
| 23. | Yan Z, Rein B. Mechanisms of synaptic transmission dysregulation in the prefrontal cortex: pathophysiological implications. Mol Psychiatry. 2022;27:445-465. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 216] [Article Influence: 54.0] [Reference Citation Analysis (0)] |
| 24. | Gerlicher AMV, Verweij SA, Kindt M. Better, worse, or different than expected: on the role of value and identity prediction errors in fear memory reactivation. Sci Rep. 2022;12:5862. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 25. | Lewis K, Balas MC, Stollings JL, McNett M, Girard TD, Chanques G, Kho ME, Pandharipande PP, Weinhouse GL, Brummel NE, Chlan LL, Cordoza M, Duby JJ, Gélinas C, Hall-Melnychuk EL, Krupp A, Louzon PR, Tate JA, Young B, Jennings R, Hines A, Ross C, Carayannopoulos KL, Aldrich JM. A Focused Update to the Clinical Practice Guidelines for the Prevention and Management of Pain, Anxiety, Agitation/Sedation, Delirium, Immobility, and Sleep Disruption in Adult Patients in the ICU. Crit Care Med. 2025;53:e711-e727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 115] [Article Influence: 115.0] [Reference Citation Analysis (0)] |