Published online Apr 19, 2026. doi: 10.5498/wjp.v16.i4.115300
Revised: November 17, 2025
Accepted: December 18, 2025
Published online: April 19, 2026
Processing time: 168 Days and 1.2 Hours
Major depressive disorder (MDD) is among the most common mental disorders, with clinical symptoms involving multiple aspects such as mood, cognition, phy
Core Tip: Major depressive disorder (MDD) is a complex disorder involving multiple neurotransmitters and brain regions. In patients with MDD, alterations may occur in the stability and functionality of these neural networks, extending beyond mere chemical imbalances. Antidepressants have neurotrophic effects that can enhance synaptic connections and stimulate the growth of new neurons, thus helping restore the function of neural networks. Synaptic and neurogenic enhancements induced by antidepressants require time to manifest and develop. These advances may yield more effective and safer antidepressants and improve the use of existing medications for patients with MDD.
- Citation: Jing P, Liu XC, Yin XP, Fu Y, Tian Q, Zhang XB. Progress on the neurotrophic effects of antidepressant drugs. World J Psychiatry 2026; 16(4): 115300
- URL: https://www.wjgnet.com/2220-3206/full/v16/i4/115300.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i4.115300
Major depressive disorder (MDD) is a type of mood disorder characterized primarily by persistent and significant low mood, slowed thinking, reduced volitional activity, and physical discomfort[1]. The primary clinical symptoms of MDD include a depressed mood, a lack of interest, and anhedonia, which is the inability to feel pleasure. These are often accompanied by other symptoms, such as reduced appetite, waking up early in the morning, and unintentional weight loss. Furthermore, patients with MDD frequently experience cognitive dysfunction, along with somatic symptoms, suicidal ideation, and suicidal behaviors[2]. Studies indicate that more than 19 million adults in the United States experience depression, with the costs for both direct and indirect treatments surpassing 30 billion dollar[3]. In China, more than 26 million people are affected by depression, with an incidence rate of about 2%. It is concerning that 90% of individuals are unaware of their condition. Moreover, the annual number of suicide deaths attributed to depression is estimated to be 1 million[4]. MDD not only has a profound psychological impact but also heightens the risk of developing other health conditions, such as cardiovascular disorders, diabetes, obesity, Alzheimer’s disease, and cancer. Consequently, the overall burden of disease increases, compounding the challenges faced by individuals, families, and society at large[5].
The existing clinical treatment for MDD still primarily relies on pharmacological intervention. Commonly used antidepressants include the following: Tricyclic antidepressants, such as imipramine, amitriptyline, and doxepin; selective serotonin reuptake inhibitors (SSRIs), such as fluoxetine, paroxetine, and sertraline; serotonin-norepinephrine reuptake inhibitors, such as venlafaxine and milnacipran; noradrenergic and specific serotonergic antidepressants, such as mirtazapine[6]. Although antidepressants can immediately increase neurotransmitter concentrations in the synaptic cleft, their therapeutic effects typically take 2-3 weeks to manifest. Moreover, treatment efficacy varies depending on factors such as gender, age, and the specific drug used, suggesting the limitations of the monoamine hypothesis. Furthermore, existing antidepressants may act on other molecular targets beyond monoamine modulation[7,8].
Recent advances in understanding MDD have led to the formulation of the neurotrophic hypothesis[9,10]. This hypothesis posits that, beyond changes in neurotransmitters, the onset of MDD is primarily associated with decreased expression of neurotrophic factors and reduced adult hippocampal neurogenesis. These changes may lead to hippocampal atrophy in patients experiencing depression. Conversely, antidepressant treatment can elevate the expression of neurotrophic factors and enhance adult hippocampal neurogenesis, thereby reversing or blocking the effects of stress on hippocampal atrophy and improving depressive symptoms. Clinical studies have demonstrated that patients with MDD exhibit reduced hippocampal volume. The extent of reduction is directly linked to MDD duration, which might be associated with disrupted hippocampal neurogenesis. Consequently, promoting hippocampal neu
Neurotrophic factors are a group of polypeptide molecules that exert specific trophic effects on neural cells. They include brain-derived neurotrophic factor (BDNF), glial cell line-derived neurotrophic factor (GDNF), nerve growth factor, neurotrophin-3 (NT-3), neurotrophin-4, and more than 20 others. These factors are vital for preserving nervous system integrity, guiding neural differentiation, supporting neuronal survival, driving development and maturation, regulating synapse formation, and modulating synaptic plasticity[13].
In summary, current research mainly focuses on single pathways or lacks systematic integration between preclinical and clinical studies. Therefore, this article aims to address this research gap through synthesizing evidence on the neurotrophic effects of antidepressants, reviewing key signaling pathways such as BDNF/tropomyosin receptor kinase B (TrkB) and mammalian target of rapamycin (mTOR)-BDNF, and exploring how these mechanisms contribute to the depressive symptom improvement. Ultimately, providing insights for developing future depression treatment strategies based on neurotrophic mechanisms.
Among the neurotrophic factors studied in MDD, BDNF has received the most attention. BDNF, one of the most extensively distributed neurotrophic factors, is abundantly present in the mammalian brain, especially within the cerebral cortex and hippocampus. BDNF is vital for encouraging synaptic development, enhancing synaptic connections, adjusting neuronal plasticity, and providing neuroprotective benefits while aiding in neuronal recovery following injury[14,15]. BDNF and NT-3 improve the function and growth of serotonergic neurons in the adult brain. Chronic infusion of BDNF and NT-3 into the midbrain of rats increases serotonin turnover and norepinephrine levels in several forebrain regions, including the neocortex, basal ganglia, and hippocampus[16,17]. The significant effects of BDNF and NT-3 on the function, growth, and regeneration of serotonergic neurons suggest a potential link between neurotrophic factors and MDD[18]. In animal models subjected to stress, such as those used in learned helplessness and forced swim tests, the continuous infusion of BDNF into the dorsal raphe nucleus (a region where serotonin-producing neurons are concentrated) results in antidepressant-like effects akin to those observed with conventional antidepressants[19]. While exogenous BDNF administration mimics antidepressant effects, it remains unclear whether endogenous neurotrophic factors are involved in MDD treatment.
Escitalopram treatment significantly increased hippocampus neurotransmitter and neurotrophic factor levels, stimulated hippocampus neurogenesis, and relieved central nervous system (CNS) microglial overactivation in chronic mild stress mice[20]. Duman et al[21] demonstrated that several classes of antidepressants, including serotonin or norepinephrine reuptake inhibitors and monoamine oxidase inhibitors, increase BDNF mRNA expression in the rat hippocampus. Saliently, this upregulation occurs only after three weeks of treatment, consistent with the delayed therapeutic onset observed in clinical MDD treatment. These antidepressants also elevate mRNA expression of TrkB, the high-affinity tyrosine kinase receptor for BDNF[22]. Unlike antidepressants, the prolonged use of psychostimulants, such as cocaine or other psychoactive substances, does not lead to an increase in BDNF or TrkB levels[23]. In rats treated with a nanotransferosomal gel containing paroxetine, improved behavior was observed, characterized by a decrease in immobility time and the presence of clearly defined neuronal morphology in Nissl-stained brain tissues[24]. Fluoxetine acts on astrocytes and increases exocytosis of ATP. This has therapeutic effects via BDNF-dependent mechanisms[25].
Electroconvulsive therapy is clinically employed to treat severe depression. In animal studies simulating this therapy, seizures induced either chemically, such as with pilocarpine, or electrically, led to an increase in BDNF mRNA levels in the hippocampus, cortex, and various other brain regions[26,27]. Similar to antidepressants, electroconvulsive therapy prevents stress-induced decreases in hippocampal BDNF mRNA. Lithium and valproate are mood stabilizers commonly used to treat and prevent mood disorders. After chronic administration, they increase BDNF expression in the rat brain, especially in the hippocampus and prefrontal cortex. They also promote hippocampal neurogenesis and partial synaptic remodeling in rodents[28].
GDNF is a member of the transforming growth factor-β superfamily and is widely distributed in the mammalian brain. It promotes the survival of various CNS neurons, including midbrain dopaminergic neurons, motor neurons, and norepinephrine neurons in the locus coeruleus. GDNF also enhances the survival of peripheral neurons, including sympathetic, parasympathetic, sensory, and enteric neurons. GDNF exerts protective effects on cultured or developing cholinergic motor neurons, increases cholinesterase activity, and is recognized as the most potent neurotrophic factor for cholinergic motor neurons identified to date[29]. In animal studies, antidepressants have not been found to affect GDNF or its receptor mRNA expression[30]. However, lithium has been reported to increase GDNF protein levels in the brains of depressive rat models[31]. Research on animal models of electroconvulsive therapy has yielded mixed results. While some studies report that electroconvulsive stimulation decreased GDNF concentration in the rat hippocampus and striatum[32], others found no significant effect on hippocampal GDNF mRNA expression. However, they observed a marked increase in GDNF receptor mRNA expression[30]. In vitro studies using C6 glioma cell lines and cultured rat astrocytes have demonstrated that serotonin, antidepressants, and mood stabilizers (lithium and valproate) can significantly increase GDNF secretion in a dose- and time-dependent manner[33-36]. These inconsistent results likely reflect differences in patients populations, disease stages and drug exposure. For instance, peripheral GDNF levels may fluctuate dynamically with emotional states. Studies indicate that levels decrease during remission or chronic phases but increase during acute emotional episodes[37,38]. Meanwhile, variations in biological samples, such as serum and plasma, and detection methods further contribute to result heterogeneity[39]. Additionally, some studies suggest that blocking the extracellular signal-regulated kinase pathway partially attenuates fluoxetine-induced upregulation of GDNF mRNA expression in astrocytes, highlighting the important role of extracellular signal-regulated kinase in cell growth and survival[40]. Overall, changes in GDNF levels may reflect state or treatment dependent phenomena rather than stable trait markers.
Combining findings from animal and cellular studies, both BDNF and GDNF play crucial roles in maintaining neuronal survival, synaptic plasticity, and stress recovery. BDNF primarily promotes dendritic growth and synaptic remodeling through the TrkB-mTOR signaling pathway, while GDNF exerts antioxidant effects. These findings provide evidence for understanding the mechanistic basis of neurotrophic factors in antidepressant actions and establish a crucial bridge for subsequent clinical translation.
Clinical studies have found that serum BDNF levels in patients with MDD are lower than in healthy controls[41,42]. BDNF is abundant in human plasma, with a significant portion localized within platelets. Studies indicate that a reduction in serum BDNF levels in individuals with MDD could be linked to a deficiency in BDNF release from platelets[43]. BDNF is linked to how individuals with mood disorders respond to antidepressant medications[44]. Aydemir et al[45] found that patients with untreated MDD exhibited lower serum BDNF than controls. Following 12 weeks of antidepressant treatment, BDNF returned to the normal range. Similarly, Gonul et al[46] reported that serum BDNF in patients with MDD returned to normal after eight weeks of antidepressant therapy. Factors such as recurrent depressive episodes, suicidal behavior, and psychotic symptoms may also influence plasma BDNF levels[47-50]. Plasma BDNF levels remained unchanged before and after infusion with non-antidepressants such as scopolamine[51]. BDNF lysis pathway could play a role in MDD pathogenesis and the processes involved in the therapeutic efficacy of antidepressant treatments. BDNF could serve as a diagnostic biomarker panel for MDD[52]. A notable finding was that serum BDNF levels increased in all patients treated with antidepressants, regardless of clinical improvement. Furthermore, the magnitude of increase in BDNF levels did not differ significantly between responders and non-responders[53]. Studies have demonstrated that the BDNF/TrkB signaling pathway plays a crucial role in the effectiveness of antidepressants and partly influences how sensitive individuals are to SSRIs and other commonly used antidepressant medications[54]. This suggests that enhancing BDNF/TrkB pathway signaling may represent the key mechanism underlying antidepressant efficacy.
Takebayashi et al[37] measured serum GDNF in patients with MDD in partial or full remission and in bipolar disorder, and found significantly lower levels than in healthy controls. Conversely, Rosa et al[38] reported increased serum GDNF protein concentrations during both manic and depressive episodes in bipolar disorder patients. Otsuki et al[55] reported reduced peripheral blood GDNF mRNA expression during depressive episodes in patients with MDD; in remission, expression did not differ from controls. Michel et al[56] observed that patients with MDD exhibited higher GDNF levels in the parietal cortex but lower levels in the hippocampus when compared with matched controls. A strong body of evidence indicates an association between GDNF and MDD. However, clinical studies have predominantly focused on serum assays. In contrast to BDNF, GDNF is a sizable protein that is unable to pass through the blood-brain barrier. Although serum GDNF does not directly mirror brain concentrations, animal studies indicate neuroprotective effects. In rats, the striatal injection of recombinant human GDNF increases the activity of superoxide dismutase, catalase, and glutathione peroxidase[57]. These findings imply that peripheral GDNF may confer protection against oxidative damage and influence depression pathogenesis through indirect antioxidant effects in the CNS.
Recent therapeutic advances have extended beyond traditional SSRIs/serotonin-norepinephrine reuptake inhibitors to include N-methyl-D-aspartate (NMDA) receptor antagonists such as esketamine. These agents demonstrate rapid clinical benefits in treatment-resistant depression (TRD) and play a role in neurotrophic mechanisms distinct from classical monoaminergic drugs. Esketamine transiently blocks NMDA receptors, causing a surge in cortical glutamate release. This promotes BDNF release and TrkB signaling cascades. These changes rapidly activate mTOR-dependent protein synthesis pathways, promote synaptic formation in prefrontal-hippocampal circuits, and correlate with behavioral improvements[58,59]. Additionally, TrkB agonists have demonstrated neurotrophic targeting potential in current research. Among these, 7,8-dihydroxyflavone has been identified as a selective TrkB agonist capable of directly activating the BDNF-TrkB signaling pathway, thereby enhancing synaptic plasticity and exhibiting antidepressant-like effects in preclinical models[60].
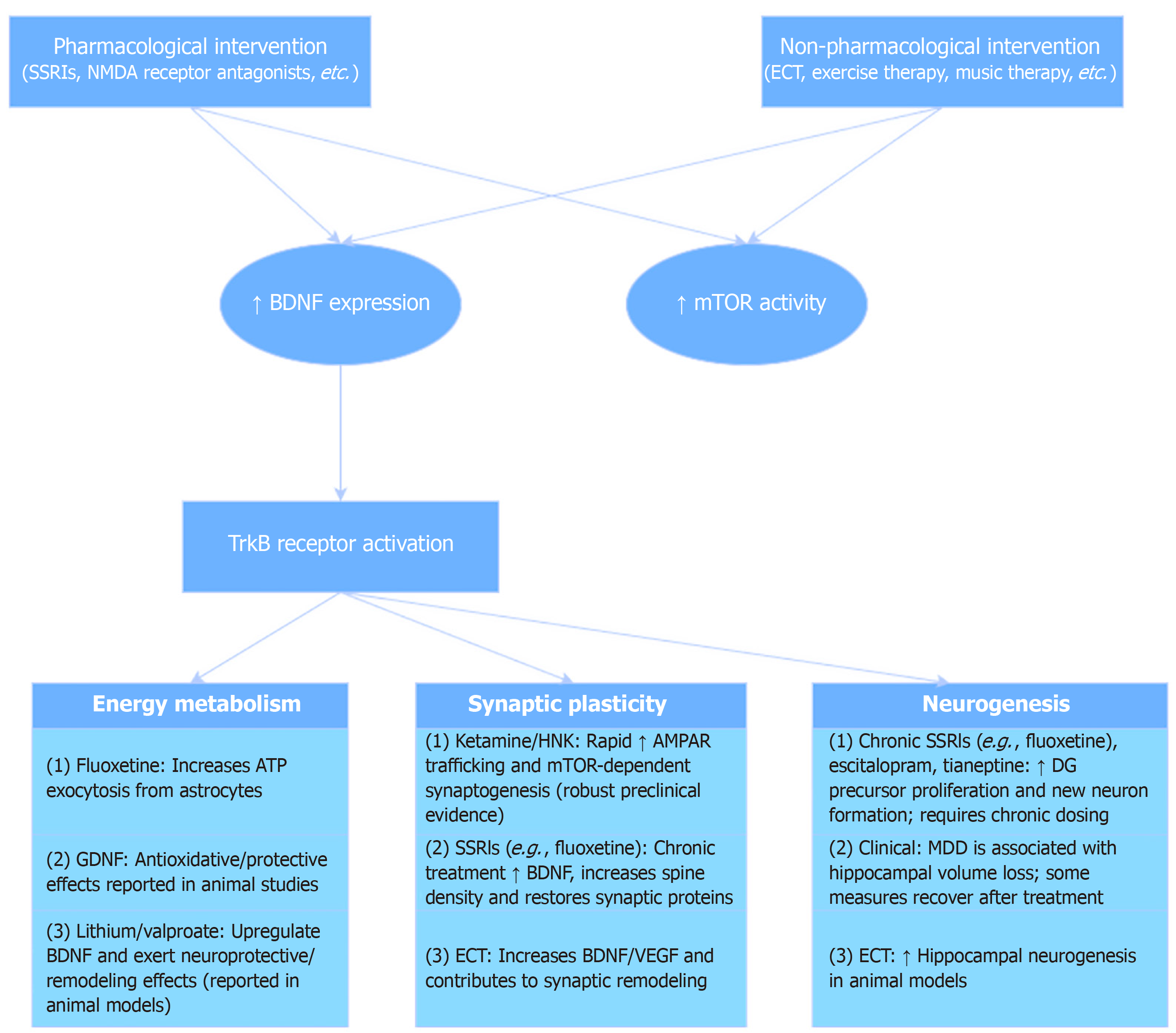
Similar to pharmacological interventions, an increasing number of studies are delving into non-pharmacological interventions, with multiple non-pharmacological approaches demonstrated to participate in neurotrophic and neuroplastic processes. For instance, electroconvulsive therapy enhances central and peripheral neurotrophic markers, such as BDNF and vascular endothelial growth factor[61], and promotes hippocampal neurogenesis[62]. These effects may underpin the mechanism by which this treatment exhibits higher efficacy in TRD. Additionally, more environment-based adjunct therapies, such as exercise intervention and music therapy, have also been shown to exert effects through neurotrophic mechanisms. For instance, exercise intervention can stably increase peripheral BDNF levels and is associated with improved hippocampal function and mood[63,64]. Notably, non-pharmacological and pharmacological interventions exhibit overlapping mechanisms across pathways such as BDNF-TrkB and mTOR. Therefore, combining both approaches may yield synergistic effects in ameliorating depressive symptoms. To integrate these convergence mechanisms, we have introduced a new mechanism integration diagram (Figure 1).
It is particularly noteworthy that the neurotrophic mechanisms differ across clinical subtypes. For instance, patients with TRD often exhibit lower BDNF levels[65], and this population typically responds more strongly to interventions that potently induce synaptogenesis, such as electroconvulsive therapy or ketamine/dextroketamine[66,67]. In contrast, MDD with anxiety frequently presents with hyperreactivity in amygdala/striatal-limbic regions and abnormal hypothalamic-pituitary-adrenal axis dynamics. These alterations suppress BDNF expression and shift plasticity toward stress-related remodeling rather than reparative synaptogenesis[68,69]. These distinct mechanisms suggest that future trials should pre-specify subtype analyses and incorporate mechanistic biomarkers, such as BDNF/TrkB phosphorylation, mTOR activity.
In the normal nervous system, synapses interconnect with each other, establishing connections between isolated neurons and forming intricate neural pathways. Excitatory and inhibitory synapses in the brain mutually regulate each other to maintain the homeostasis of signal transmission. When the body is subjected to intense or prolonged external stimuli, this homeostasis is disrupted, leading to the reformation of synapses with different properties. The resulting facilitation or inhibition of signal transmission is referred to as synaptic plasticity[70]. Synaptic plasticity manifests in two forms: Structural plasticity and functional plasticity. Structural plasticity refers to changes in the morphology, structure, and number of synapses that occur following neuronal stimulation, resulting in synaptic remodeling that influences brain function. Functional plasticity can be further divided into two categories: Long-term depression, a sustained decrease in synaptic transmission efficiency that occurs after repeated neuronal stimulation, and long-term potentiation (LTP), a sustained increase in synaptic transmission efficiency that occurs after repeated neuronal stimulation. LTP: A persistent enhancement of synaptic transmission efficiency due to increased postsynaptic potential after stimulating presynaptic neuron terminals. LTP is induced when presynaptic stimulation coincides with postsynaptic depolarization, and is generated and maintained through a combination of presynaptic and postsynaptic mechanisms[71]. LTP and long-term depression are critical manifestations of synaptic plasticity, serving as key physiological mechanisms underlying cognition and memory[72]. Antidepressant drugs promote neuronal plasticity, and activation of BDNF signaling through its receptor, neuronal receptor tyrosine kinase 2, is among the critical steps in this process[73].
Prolonged transmission of nociceptive signals induces structural and functional plasticity changes in higher brain regions, such as the prefrontal cortex[74], amygdala[75], dentate gyrus[76], and anterior cingulate cortex[77]. These changes include neuronal atrophy, reduced dendritic complexity, and decreased dendritic spine density, resulting in depression-like symptoms[78,79]. The size, number, and morphology of dendritic spines play a crucial role in synaptic plasticity, learning, and memory[80]. Depressive symptoms, such as anhedonia (loss of pleasure), and overall severity are closely linked to brain volume reductions and changes in synaptic plasticity in patients with MDD[81]. Stress-induced neuronal atrophy and dysfunction in glutamatergic synaptic transmission are key factors in depressive pathology[4,82]. Studies have found that when the gamma-aminobutyric acid B receptor agonist baclofen is applied to neurons treated with NMDA receptor antagonists, it enhances resting dendritic calcium signaling via gamma-aminobutyric acid B receptor activation, subsequently upregulating the mTOR activity[83]. The mTOR-BDNF signaling pathway, a critical regulator of synaptic plasticity, plays a pivotal role in ketamine’s antidepressant effects[84,85]. Ketamine activates mTOR, which in turn stimulates downstream ribosomal protein S6 kinase and eukaryotic initiation factor 4E-binding protein, promoting the synthesis of BDNF and synaptic proteins (glutamate receptor 1 and postsynaptic density protein 95). As a key regulator of synaptogenesis, BDNF binds to and activates TrkB, fostering dendritic spine growth and maintaining synaptic plasticity[86,87]. By blocking NMDA receptors, ketamine decreases the activity of GABAergic neurons, leading to an increase in glutamate release. This process boosts synaptic plasticity and promotes neuronal growth, thereby alleviating depressive symptoms[88,89].
Zanos et al[90] further discovered that (2R,6R)-hydroxynorketamine can induce a rapid enhancement of glutamatergic signaling. This involves increased presynaptic glutamate release and its binding to postsynaptic α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor-gated channels, allowing sodium ions to enter the cell[91]. This process boosts the activity of hippocampal afferents, increases the levels of glutamate A1 and glutamate A2 in hippocampal synapses, and markedly alleviates depressive symptoms. Antidepressants, such as venlafaxine, mirtazapine, fluoxetine, and desipramine, are capable of reversing the reduction in BDNF protein levels, encouraging synaptic development, promoting neurogenesis, and leading to an enhancement of mood[20,92]. Specifically, fluoxetine acts on multiple aspects of neuroplasticity mechanisms by enhancing the secretion of various neurotrophic factors, facilitating the formation of new synapses, and promoting neuronal regeneration, all of which contribute to neural recovery[93-95]. Furthermore, after undergoing antidepressant therapy, notable alterations in the levels of proteins associated with synapses often occur, including synaptophysin-1, microtubule-associated protein-2, synaptotagmib-1, and postsynaptic density protein 95[96-100]. These proteins serve as biomarkers for neuronal synaptic plasticity alterations and are likely involved in both the pathogenesis and recovery of mood and cognitive-related psychiatric disorders, including depression. Emerging evidence suggests that depression arises from dysregulation not only in central neurotransmitter systems but also in postsynaptic signaling cascades and transcriptional regulation, resulting in region-specific neurotrophic deficits and compromised neural plasticity[101]. Importantly, BDNF appears to function as a molecular transducer that mechanistically links antidepressant efficacy with the restoration of neuroplasticity in depression pathophysiology[102,103].
Neurogenesis refers to the self-repair or functional reconstruction process of the nervous system after damage, encompassing the regeneration of neurons (nerve cells), regrowth of axons (nerve fibers), and re-establishment of synaptic connections. In patients with MDD, neurogenic capacity is typically suppressed, particularly in key brain regions such as the hippocampus and prefrontal cortex. Convergent evidence indicates that MDD is closely correlated with reduced neuroplasticity, neuronal atrophy, and diminished neurogenesis (formation of new neurons)[104].
Neuroimaging studies have revealed reduced cerebral blood flow, decreased metabolic activity, and diminished volumes in the limbic system, hippocampus, prefrontal cortex, and amygdala among patients with MDD[105-109]. Numerous studies have consistently demonstrated a reduction in hippocampal volume in MDD[110-113]. Antidepressant drugs, such as fluoxetine, have been demonstrated to reverse or prevent this hippocampal atrophy in patients with MDD[114,115]. Stockmeier et al[116] reported increased neuronal and glial cell density in the dentate gyrus and cornuammonis regions of the hippocampus in patients with MDD. The observed hippocampal volume reduction in depression may contribute to this elevated cellular packing density within hippocampal structures.
In adult mammalian brains, neural stem cell populations are primarily located in the hippocampal dentate gyrus, ventricular and subventricular zones, as well as the region connecting the lateral ventricles to the olfactory bulb. Neural stem cells can be isolated from both the dentate gyrus of the hippocampus and the lateral walls of the ventricular system in adult mammals. The cells in the ventricular zone and subventricular zone of the lateral ventricle walls generate neural progenitor cells that migrate to the olfactory bulb. In contrast, neural stem cells originating from the subgranular zone of the dentate gyrus migrate into the granule cell layer[117]. The hippocampus serves as a central hub for emotional in
Antidepressant drugs such as fluoxetine[120,121], escitalopram[122], and tianeptine[123] can reverse stress-induced reductions in adult neurogenesis[124,125]. With the exception of one study demonstrating that repetitive transcranial magnetic stimulation fails to counteract the effects of chronic psychological stress on hippocampal neurogenesis[126], all other antidepressant treatments investigated to date have been found to enhance hippocampal neurogenesis and/or reverse stress-induced impairments[127,128]. These findings form the foundation of the neurogenic hypothesis of antidepressant action. Antidepressant-induced neurogenesis requires chronic dosing and is confined to the hippocampus, with unnoticeable changes in proliferation rates in the cerebral cortex. Notably, chronic fluoxetine treatment (a serotonin re
The hippocampus is a key structure for memory processes, and reduced hippocampal neurogenesis may contribute to memory impairments in patients with MDD[142]. Studies report that when adult hippocampal neurogenesis is inhibited, hippocampal-dependent learning and memory functions are impaired[143-146]. In animal models of depression (forced swim test and learned helplessness), decreased proliferation of granule layer precursor cells has been observed, leading to reduced numbers of newborn neurons in the dentate gyrus. Hippocampal volume reduction has also been documented in patients with MDD[147]. Chen et al[148] found that various antidepressants can increase the number of precursor cells in the hippocampal dentate gyrus, leading to a gradual improvement in clinical symptoms. Czéh et al[149] reported that antidepressant treatment can protect against stress-induced hippocampal atrophy.
Malberg et al[150] further demonstrated that repeated courses of antidepressants enhance hippocampal cell proliferation. To quantify this effect, 5-bromo-2’-deoxyuridine (BrdU) was injected following the last treatment, and proliferating cells were identified by immunohistochemistry 24 hours after BrdU administration. The elevated number of BrdU-positive cells at this time point indicates increased cell proliferation. This cell proliferation was observed using multiple classes of antidepressants, including a monoamine oxidase inhibitor (tranylcypromine), a SSRI (fluoxetine), and a selective norepinephrine reuptake inhibitor (reboxetine). It was further demonstrated that long-term (2 weeks), but not short-term (1-5 days), drug administration was necessary for the fluoxetine-induced increase in cell proliferation and neurogenesis. An increased count of BrdU-positive cells was still evident at one month. Most of these cells had become neurons, indicating a net increase in neurogenesis[151]. These findings collectively provide compelling evidence for a close relationship between depression and impaired neurogenesis and defective plasticity. Applying these mechanisms to clinical indications may enhance the long-term remission and functional recovery outcomes of conventional treatments through drugs, behavioral interventions, or neuromodulation therapies that promote neural regeneration.
In summary, evidence from molecular, basic, and clinical levels collectively demonstrates that neurotrophic signaling plays a central role in the pathogenesis and treatment of depression. BDNF and GDNF, as key modulators linking stress, synaptic plasticity, and neuronal survival, provide a framework for understanding the unified mechanism of antidepressants. Future research must overcome several limitations, including standardizing peripheral neurotrophic biomarker measurements, further clarifying causal relationships, and conducting clinical validation based on neurotrophic factors. With advances in gene regulation, neuroimaging, and personalized medicine, strategies targeting neurotrophic signaling hold promise for translating mechanistic research into precise clinical interventions.
MDD is a complex disorder involving multiple neurotransmitters and brain regions. Earlier studies into antidepressants concentrated on their influence on brain neurotransmitters. However, more recent research has focused on investigating how these drugs impact intracellular signaling pathways and the growth of neurons in neural cells. The primary role of the nervous system is to store and process information, a task that cannot be achieved by individual neurons or synapses alone. Alternatively, it is accomplished through dynamic neural networks, which are created by interconnected neurons. In patients with MDD, alterations may occur in the stability and functionality of these neural networks, extending beyond mere chemical imbalances. Antidepressants have neurotrophic effects that can enhance synaptic connections and stimulate the growth of new neurons, thus helping restore the function of neural networks. Synaptic and neurogenic enhancements induced by antidepressants require time to manifest and develop. This explains the typical 2-3 weeks delay in clinical efficacy. By identifying key neural networks and characterizing their neurobiology, clinicians can optimize combinations of pharmacological and non-pharmacological treatments. These advances may yield more effective and safer antidepressants and improve the use of existing medications for patients with MDD.
We are grateful to all the psychiatrists and nurses who participated in our current study.
| 1. | Huang Y, Wang Y, Wang H, Liu Z, Yu X, Yan J, Yu Y, Kou C, Xu X, Lu J, Wang Z, He S, Xu Y, He Y, Li T, Guo W, Tian H, Xu G, Xu X, Ma Y, Wang L, Wang L, Yan Y, Wang B, Xiao S, Zhou L, Li L, Tan L, Zhang T, Ma C, Li Q, Ding H, Geng H, Jia F, Shi J, Wang S, Zhang N, Du X, Du X, Wu Y. Prevalence of mental disorders in China: a cross-sectional epidemiological study. Lancet Psychiatry. 2019;6:211-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1960] [Cited by in RCA: 1586] [Article Influence: 226.6] [Reference Citation Analysis (1)] |
| 2. | Marx W, Penninx BWJH, Solmi M, Furukawa TA, Firth J, Carvalho AF, Berk M. Major depressive disorder. Nat Rev Dis Primers. 2023;9:44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 543] [Cited by in RCA: 428] [Article Influence: 142.7] [Reference Citation Analysis (1)] |
| 3. | Baglioni C, Battagliese G, Feige B, Spiegelhalder K, Nissen C, Voderholzer U, Lombardo C, Riemann D. Insomnia as a predictor of depression: a meta-analytic evaluation of longitudinal epidemiological studies. J Affect Disord. 2011;135:10-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2264] [Cited by in RCA: 1845] [Article Influence: 123.0] [Reference Citation Analysis (3)] |
| 4. | Duman RS, Aghajanian GK, Sanacora G, Krystal JH. Synaptic plasticity and depression: new insights from stress and rapid-acting antidepressants. Nat Med. 2016;22:238-249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1532] [Cited by in RCA: 1330] [Article Influence: 133.0] [Reference Citation Analysis (0)] |
| 5. | Han LKM, Aghajani M, Clark SL, Chan RF, Hattab MW, Shabalin AA, Zhao M, Kumar G, Xie LY, Jansen R, Milaneschi Y, Dean B, Aberg KA, van den Oord EJCG, Penninx BWJH. Epigenetic Aging in Major Depressive Disorder. Am J Psychiatry. 2018;175:774-782. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 232] [Cited by in RCA: 204] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 6. | Dionisie V, Filip GA, Manea MC, Manea M, Riga S. The anti-inflammatory role of SSRI and SNRI in the treatment of depression: a review of human and rodent research studies. Inflammopharmacology. 2021;29:75-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 90] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 7. | Andersen SL, Navalta CP. Annual Research Review: New frontiers in developmental neuropharmacology: can long-term therapeutic effects of drugs be optimized through carefully timed early intervention? J Child Psychol Psychiatry. 2011;52:476-503. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 8. | Xie W, Meng X, Zhai Y, Zhou P, Ye T, Wang Z, Sun G, Sun X. Panax Notoginseng Saponins: A Review of Its Mechanisms of Antidepressant or Anxiolytic Effects and Network Analysis on Phytochemistry and Pharmacology. Molecules. 2018;23:940. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 102] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 9. | Sampath D, McWhirt J, Sathyanesan M, Newton SS. Carbamoylated erythropoietin produces antidepressant-like effects in male and female mice. Prog Neuropsychopharmacol Biol Psychiatry. 2020;96:109754. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 10. | Pekas NJ, Petersen JL, Sathyanesan M, Newton SS. Design and Development of a Behaviorally Active Recombinant Neurotrophic Factor. Drug Des Devel Ther. 2020;14:5393-5403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 11. | Santarelli L, Saxe M, Gross C, Surget A, Battaglia F, Dulawa S, Weisstaub N, Lee J, Duman R, Arancio O, Belzung C, Hen R. Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science. 2003;301:805-809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3567] [Cited by in RCA: 3236] [Article Influence: 140.7] [Reference Citation Analysis (5)] |
| 12. | Almeida FB, Nin MS, Barros HMT. The role of allopregnanolone in depressive-like behaviors: Focus on neurotrophic proteins. Neurobiol Stress. 2020;12:100218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 13. | Castrén E, Antila H. Neuronal plasticity and neurotrophic factors in drug responses. Mol Psychiatry. 2017;22:1085-1095. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 259] [Cited by in RCA: 206] [Article Influence: 22.9] [Reference Citation Analysis (0)] |
| 14. | Castrén E, Monteggia LM. Brain-Derived Neurotrophic Factor Signaling in Depression and Antidepressant Action. Biol Psychiatry. 2021;90:128-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 440] [Cited by in RCA: 360] [Article Influence: 72.0] [Reference Citation Analysis (1)] |
| 15. | Notaras M, van den Buuse M. Neurobiology of BDNF in fear memory, sensitivity to stress, and stress-related disorders. Mol Psychiatry. 2020;25:2251-2274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 336] [Cited by in RCA: 261] [Article Influence: 43.5] [Reference Citation Analysis (0)] |
| 16. | Martin-Iverson MT, Todd KG, Altar CA. Brain-derived neurotrophic factor and neurotrophin-3 activate striatal dopamine and serotonin metabolism and related behaviors: interactions with amphetamine. J Neurosci. 1994;14:1262-1270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 120] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 17. | Xu D, Gao LN, Song XJ, Dong QW, Chen YB, Cui YL, Wang Q. Enhanced antidepressant effects of BDNF-quercetin alginate nanogels for depression therapy. J Nanobiotechnology. 2023;21:379. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 46] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 18. | Lenart L, Balogh DB, Lenart N, Barczi A, Hosszu A, Farkas T, Hodrea J, Szabo AJ, Szigeti K, Denes A, Fekete A. Novel therapeutic potential of angiotensin receptor 1 blockade in a rat model of diabetes-associated depression parallels altered BDNF signalling. Diabetologia. 2019;62:1501-1513. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 19. | Siuciak JA, Lewis DR, Wiegand SJ, Lindsay RM. Antidepressant-like effect of brain-derived neurotrophic factor (BDNF). Pharmacol Biochem Behav. 1997;56:131-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 677] [Cited by in RCA: 615] [Article Influence: 21.2] [Reference Citation Analysis (1)] |
| 20. | Pan SM, Yin XY, Dai DM, Zhang LW, Qi Q, Wang PJ, Hui L, Zhu ZH. Unraveling the potential of Morinda officinalis oligosaccharides as an adjuvant of escitalopram in depression treatment and exploring the underlying mechanisms. J Ethnopharmacol. 2024;328:118124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 21. | Duman RS, Heninger GR, Nestler EJ. A molecular and cellular theory of depression. Arch Gen Psychiatry. 1997;54:597-606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1688] [Cited by in RCA: 1441] [Article Influence: 49.7] [Reference Citation Analysis (5)] |
| 22. | Wang G, Lei C, Tian Y, Wang Y, Zhang L, Zhang R. Rb1, the Primary Active Ingredient in Panax ginseng C.A. Meyer, Exerts Antidepressant-Like Effects via the BDNF-Trkb-CREB Pathway. Front Pharmacol. 2019;10:1034. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 23. | Nibuya M, Morinobu S, Duman RS. Regulation of BDNF and trkB mRNA in rat brain by chronic electroconvulsive seizure and antidepressant drug treatments. J Neurosci. 1995;15:7539-7547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1706] [Cited by in RCA: 1488] [Article Influence: 48.0] [Reference Citation Analysis (2)] |
| 24. | Tahir A, Aslam S, Sohail S, Ud Din F, Alamri AH, Lahiq AA, Alsharif ST, Asiri A. Development of paroxetine loaded nanotransferosomal gel for intranasal delivery with enhanced antidepressant activity in rats. Colloids Surf B Biointerfaces. 2025;246:114351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 25. | Koizumi S. Glial Purinergic Signals and Psychiatric Disorders. Front Cell Neurosci. 2021;15:822614. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 26. | Schmidt-Kastner R, Humpel C, Wetmore C, Olson L. Cellular hybridization for BDNF, trkB, and NGF mRNAs and BDNF-immunoreactivity in rat forebrain after pilocarpine-induced status epilepticus. Exp Brain Res. 1996;107:331-347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 71] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 27. | Smith MA, Zhang LX, Lyons WE, Mamounas LA. Anterograde transport of endogenous brain-derived neurotrophic factor in hippocampal mossy fibers. Neuroreport. 1997;8:1829-1834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 92] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 28. | Rantamäki T, Knuuttila JE, Hokkanen ME, Castrén E. The effects of acute and long-term lithium treatments on trkB neurotrophin receptor activation in the mouse hippocampus and anterior cingulate cortex. Neuropharmacology. 2006;50:421-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 29. | Lapchak PA, Miller PJ, Jiao S. Glial cell line-derived neurotrophic factor induces the dopaminergic and cholinergic phenotype and increases locomotor activity in aged Fischer 344 rats. Neuroscience. 1997;77:745-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 52] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 30. | Kokaia Z, Airaksinen MS, Nanobashvili A, Larsson E, Kujamäki E, Lindvall O, Saarma M. GDNF family ligands and receptors are differentially regulated after brain insults in the rat. Eur J Neurosci. 1999;11:1202-1216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 89] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 31. | Angelucci F, Aloe L, Jiménez-Vasquez P, Mathé AA. Lithium treatment alters brain concentrations of nerve growth factor, brain-derived neurotrophic factor and glial cell line-derived neurotrophic factor in a rat model of depression. Int J Neuropsychopharmacol. 2003;6:225-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 102] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 32. | Angelucci F, Aloe L, Jiménez-Vasquez P, Mathé AA. Electroconvulsive stimuli alter the regional concentrations of nerve growth factor, brain-derived neurotrophic factor, and glial cell line-derived neurotrophic factor in adult rat brain. J ECT. 2002;18:138-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 46] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 33. | Hisaoka K, Nishida A, Koda T, Miyata M, Zensho H, Morinobu S, Ohta M, Yamawaki S. Antidepressant drug treatments induce glial cell line-derived neurotrophic factor (GDNF) synthesis and release in rat C6 glioblastoma cells. J Neurochem. 2001;79:25-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 96] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 34. | Bartholomä P, Erlandsson N, Kaufmann K, Rössler OG, Baumann B, Wirth T, Giehl KM, Thiel G. Neuronal cell death induced by antidepressants: lack of correlation with Egr-1, NF-kappa B and extracellular signal-regulated protein kinase activation. Biochem Pharmacol. 2002;63:1507-1516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 35. | Hisaoka K, Nishida A, Takebayashi M, Koda T, Yamawaki S, Nakata Y. Serotonin increases glial cell line-derived neurotrophic factor release in rat C6 glioblastoma cells. Brain Res. 2004;1002:167-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 30] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 36. | Castro LM, Gallant M, Niles LP. Novel targets for valproic acid: up-regulation of melatonin receptors and neurotrophic factors in C6 glioma cells. J Neurochem. 2005;95:1227-1236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 88] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 37. | Takebayashi M, Hisaoka K, Nishida A, Tsuchioka M, Miyoshi I, Kozuru T, Hikasa S, Okamoto Y, Shinno H, Morinobu S, Yamawaki S. Decreased levels of whole blood glial cell line-derived neurotrophic factor (GDNF) in remitted patients with mood disorders. Int J Neuropsychopharmacol. 2006;9:607-612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 103] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 38. | Rosa AR, Frey BN, Andreazza AC, Ceresér KM, Cunha AB, Quevedo J, Santin A, Gottfried C, Gonçalves CA, Vieta E, Kapczinski F. Increased serum glial cell line-derived neurotrophic factor immunocontent during manic and depressive episodes in individuals with bipolar disorder. Neurosci Lett. 2006;407:146-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 70] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 39. | Lin PY, Tseng PT. Decreased glial cell line-derived neurotrophic factor levels in patients with depression: a meta-analytic study. J Psychiatr Res. 2015;63:20-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 77] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 40. | Mercier G, Lennon AM, Renouf B, Dessouroux A, Ramaugé M, Courtin F, Pierre M. MAP kinase activation by fluoxetine and its relation to gene expression in cultured rat astrocytes. J Mol Neurosci. 2004;24:207-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 120] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 41. | Karege F, Perret G, Bondolfi G, Schwald M, Bertschy G, Aubry JM. Decreased serum brain-derived neurotrophic factor levels in major depressed patients. Psychiatry Res. 2002;109:143-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 978] [Cited by in RCA: 899] [Article Influence: 37.5] [Reference Citation Analysis (0)] |
| 42. | Hsieh MT, Lin CC, Lee CT, Huang TL. Abnormal Brain-Derived Neurotrophic Factor Exon IX Promoter Methylation, Protein, and mRNA Levels in Patients with Major Depressive Disorder. J Clin Med. 2019;8:568. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 43. | Karege F, Schwald M, Cisse M. Postnatal developmental profile of brain-derived neurotrophic factor in rat brain and platelets. Neurosci Lett. 2002;328:261-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 668] [Cited by in RCA: 650] [Article Influence: 27.1] [Reference Citation Analysis (0)] |
| 44. | Ochi T, Vyalova NM, Losenkov IS, Levchuk LA, Osmanova DZ, Mikhalitskaya EV, Loonen AJM, Bosker FJ, Simutkin GG, Bokhan NA, Wilffert B, Ivanova SA. Investigating the potential role of BDNF and PRL genotypes on antidepressant response in depression patients: A prospective inception cohort study in treatment-free patients. J Affect Disord. 2019;259:432-439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 45. | Aydemir O, Deveci A, Taneli F. The effect of chronic antidepressant treatment on serum brain-derived neurotrophic factor levels in depressed patients: a preliminary study. Prog Neuropsychopharmacol Biol Psychiatry. 2005;29:261-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 210] [Article Influence: 10.0] [Reference Citation Analysis (3)] |
| 46. | Gonul AS, Akdeniz F, Taneli F, Donat O, Eker C, Vahip S. Effect of treatment on serum brain-derived neurotrophic factor levels in depressed patients. Eur Arch Psychiatry Clin Neurosci. 2005;255:381-386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 284] [Cited by in RCA: 259] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 47. | Lee BH, Kim H, Park SH, Kim YK. Decreased plasma BDNF level in depressive patients. J Affect Disord. 2007;101:239-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 244] [Cited by in RCA: 227] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 48. | Kim YK, Lee HP, Won SD, Park EY, Lee HY, Lee BH, Lee SW, Yoon D, Han C, Kim DJ, Choi SH. Low plasma BDNF is associated with suicidal behavior in major depression. Prog Neuropsychopharmacol Biol Psychiatry. 2007;31:78-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 237] [Cited by in RCA: 212] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 49. | Mishra BR, Mohapatra D, Biswas T, Mishra A, Panigrahi S, Maiti R. Comparative efficacy of antidepressant augmentation with amantadine vs pramipexole in treatment-resistant unipolar depression: A randomised controlled trial. J Affect Disord. 2025;390:119891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 50. | Chiou YJ, Huang TL. Serum Brain-Derived Neurotrophic Factors in Taiwanese Patients with Drug-Naïve First-Episode Major Depressive Disorder: Effects of Antidepressants. Int J Neuropsychopharmacol. 2017;20:213-218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 24] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 51. | Park L, Furey M, Nugent AC, Farmer C, Ellis J, Szczepanik J, Lener MS, Zarate CA Jr. Neurophysiological Changes Associated with Antidepressant Response to Ketamine Not Observed in a Negative Trial of Scopolamine in Major Depressive Disorder. Int J Neuropsychopharmacol. 2019;22:10-18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 52. | Jiang H, Chen S, Li C, Lu N, Yue Y, Yin Y, Zhang Y, Zhi X, Zhang D, Yuan Y. The serum protein levels of the tPA-BDNF pathway are implicated in depression and antidepressant treatment. Transl Psychiatry. 2017;7:e1079. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 125] [Article Influence: 13.9] [Reference Citation Analysis (1)] |
| 53. | Brunoni AR, Lopes M, Fregni F. A systematic review and meta-analysis of clinical studies on major depression and BDNF levels: implications for the role of neuroplasticity in depression. Int J Neuropsychopharmacol. 2008;11:1169-1180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 770] [Cited by in RCA: 700] [Article Influence: 38.9] [Reference Citation Analysis (0)] |
| 54. | Li Y, Luikart BW, Birnbaum S, Chen J, Kwon CH, Kernie SG, Bassel-Duby R, Parada LF. TrkB regulates hippocampal neurogenesis and governs sensitivity to antidepressive treatment. Neuron. 2008;59:399-412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 527] [Cited by in RCA: 491] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 55. | Otsuki K, Uchida S, Watanuki T, Wakabayashi Y, Fujimoto M, Matsubara T, Funato H, Watanabe Y. Altered expression of neurotrophic factors in patients with major depression. J Psychiatr Res. 2008;42:1145-1153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 140] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 56. | Michel TM, Frangou S, Camara S, Thiemeyer D, Jecel J, Tatschner T, Zoechling R, Grünblatt E. Altered glial cell line-derived neurotrophic factor (GDNF) concentrations in the brain of patients with depressive disorder: a comparative post-mortem study. Eur Psychiatry. 2008;23:413-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 61] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 57. | Chao CC, Lee EH. Neuroprotective mechanism of glial cell line-derived neurotrophic factor on dopamine neurons: role of antioxidation. Neuropharmacology. 1999;38:913-916. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 63] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 58. | Duman RS, Li N, Liu RJ, Duric V, Aghajanian G. Signaling pathways underlying the rapid antidepressant actions of ketamine. Neuropharmacology. 2012;62:35-41. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 460] [Cited by in RCA: 423] [Article Influence: 30.2] [Reference Citation Analysis (0)] |
| 59. | Abdallah CG, Adams TG, Kelmendi B, Esterlis I, Sanacora G, Krystal JH. Ketamine's mechanism of action: A path to rapid-acting antidepressants. Depress Anxiety. 2016;33:689-697. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 179] [Cited by in RCA: 164] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 60. | Jang SW, Liu X, Yepes M, Shepherd KR, Miller GW, Liu Y, Wilson WD, Xiao G, Blanchi B, Sun YE, Ye K. A selective TrkB agonist with potent neurotrophic activities by 7,8-dihydroxyflavone. Proc Natl Acad Sci U S A. 2010;107:2687-2692. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 604] [Cited by in RCA: 559] [Article Influence: 34.9] [Reference Citation Analysis (0)] |
| 61. | Maffioletti E, Carvalho Silva R, Bortolomasi M, Baune BT, Gennarelli M, Minelli A. Molecular Biomarkers of Electroconvulsive Therapy Effects and Clinical Response: Understanding the Present to Shape the Future. Brain Sci. 2021;11:1120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 62. | van Buel EM, Patas K, Peters M, Bosker FJ, Eisel UL, Klein HC. Immune and neurotrophin stimulation by electroconvulsive therapy: is some inflammation needed after all? Transl Psychiatry. 2015;5:e609. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 85] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 63. | Jemni M, Zaman R, Carrick FR, Clarke ND, Marina M, Bottoms L, Matharoo JS, Ramsbottom R, Hoffman N, Groves SJ, Gu Y, Konukman F. Exercise improves depression through positive modulation of brain-derived neurotrophic factor (BDNF). A review based on 100 manuscripts over 20 years. Front Physiol. 2023;14:1102526. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 37] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 64. | Sanaeifar F, Pourranjbar S, Pourranjbar M, Ramezani S, Mehr SR, Wadan AS, Khazeifard F. Beneficial effects of physical exercise on cognitive-behavioral impairments and brain-derived neurotrophic factor alteration in the limbic system induced by neurodegeneration. Exp Gerontol. 2024;195:112539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 50] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 65. | Meshkat S, Alnefeesi Y, Jawad MY, D Di Vincenzo J, B Rodrigues N, Ceban F, Mw Lui L, McIntyre RS, Rosenblat JD. Brain-Derived Neurotrophic Factor (BDNF) as a biomarker of treatment response in patients with Treatment Resistant Depression (TRD): A systematic review & meta-analysis. Psychiatry Res. 2022;317:114857. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 47] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 66. | Vasiliu O. Esketamine for treatmentresistant depression: A review of clinical evidence (Review). Exp Ther Med. 2023;25:111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 47] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 67. | Xie XH, Xu SX, Yao L, Chen MM, Zhang H, Wang C, Nagy C, Liu Z. Altered in vivo early neurogenesis traits in patients with depression: Evidence from neuron-derived extracellular vesicles and electroconvulsive therapy. Brain Stimul. 2024;17:19-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 68. | Keller J, Gomez R, Williams G, Lembke A, Lazzeroni L, Murphy GM Jr, Schatzberg AF. HPA axis in major depression: cortisol, clinical symptomatology and genetic variation predict cognition. Mol Psychiatry. 2017;22:527-536. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 795] [Cited by in RCA: 688] [Article Influence: 76.4] [Reference Citation Analysis (0)] |
| 69. | Nawijn L, Dinga R, Aghajani M, van Tol MJ, van der Wee NJA, Wunder A, Veltman DJ, Penninx BWHJ. Neural correlates of anxious distress in depression: A neuroimaging study of reactivity to emotional faces and resting-state functional connectivity. Depress Anxiety. 2022;39:573-585. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 70. | Wiera G, Mozrzymas JW. Extracellular proteolysis in structural and functional plasticity of mossy fiber synapses in hippocampus. Front Cell Neurosci. 2015;9:427. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 44] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 71. | Amtul Z, Atta-Ur-Rahman. Neural plasticity and memory: molecular mechanism. Rev Neurosci. 2015;26:253-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 72. | Chen Q, Ren L, Min S, Hao X, Chen H, Deng J. Changes in synaptic plasticity are associated with electroconvulsive shock-induced learning and memory impairment in rats with depression-like behavior. Neuropsychiatr Dis Treat. 2018;14:1737-1746. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 73. | Brunello CA, Cannarozzo C, Castrén E. Rethinking the role of TRKB in the action of antidepressants and psychedelics. Trends Neurosci. 2024;47:865-874. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 20] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 74. | Mecca CM, Chao D, Yu G, Feng Y, Segel I, Zhang Z, Rodriguez-Garcia DM, Pawela CP, Hillard CJ, Hogan QH, Pan B. Dynamic Change of Endocannabinoid Signaling in the Medial Prefrontal Cortex Controls the Development of Depression After Neuropathic Pain. J Neurosci. 2021;41:7492-7508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 75. | Jiang H, Liu JP, Xi K, Liu LY, Kong LY, Cai J, Cai SQ, Han XY, Song JG, Yang XM, Wan Y, Xing GG. Contribution of AMPA Receptor-Mediated LTD in LA/BLA-CeA Pathway to Comorbid Aversive and Depressive Symptoms in Neuropathic Pain. J Neurosci. 2021;41:7278-7299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 76. | Vega-Rivera NM, González-Trujano ME, Luna-Angula A, Sánchez-Chapul L, Estrada-Camarena E. Antidepressant-like effects of the Punica granatum and citalopram combination are associated with structural changes in dendritic spines of granule cells in the dentate gyrus of rats. Front Pharmacol. 2023;14:1211663. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 77. | Li XH, Matsuura T, Xue M, Chen QY, Liu RH, Lu JS, Shi W, Fan K, Zhou Z, Miao Z, Yang J, Wei S, Wei F, Chen T, Zhuo M. Oxytocin in the anterior cingulate cortex attenuates neuropathic pain and emotional anxiety by inhibiting presynaptic long-term potentiation. Cell Rep. 2021;36:109411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 149] [Article Influence: 29.8] [Reference Citation Analysis (0)] |
| 78. | Evans JW, Szczepanik J, Brutsché N, Park LT, Nugent AC, Zarate CA Jr. Default Mode Connectivity in Major Depressive Disorder Measured Up to 10 Days After Ketamine Administration. Biol Psychiatry. 2018;84:582-590. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 150] [Cited by in RCA: 135] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 79. | Dong C, Zhang JC, Yao W, Ren Q, Ma M, Yang C, Chaki S, Hashimoto K. Rapid and Sustained Antidepressant Action of the mGlu2/3 Receptor Antagonist MGS0039 in the Social Defeat Stress Model: Comparison with Ketamine. Int J Neuropsychopharmacol. 2017;20:228-236. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 60] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 80. | Bailey CH, Kandel ER, Harris KM. Structural Components of Synaptic Plasticity and Memory Consolidation. Cold Spring Harb Perspect Biol. 2015;7:a021758. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 335] [Cited by in RCA: 278] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 81. | Hamm JP, Peterka DS, Gogos JA, Yuste R. Altered Cortical Ensembles in Mouse Models of Schizophrenia. Neuron. 2017;94:153-167.e8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 181] [Cited by in RCA: 149] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 82. | Kraeuter AK, Guest PC, Sarnyai Z. The Y-Maze for Assessment of Spatial Working and Reference Memory in Mice. Methods Mol Biol. 2019;1916:105-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1090] [Cited by in RCA: 906] [Article Influence: 129.4] [Reference Citation Analysis (0)] |
| 83. | Li ZL, Wang Y, Zou HW, Jing XY, Liu YJ, Li LF. GABA(B) receptors within the lateral habenula modulate stress resilience and vulnerability in mice. Physiol Behav. 2021;230:113311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 84. | Ignácio ZM, Réus GZ, Arent CO, Abelaira HM, Pitcher MR, Quevedo J. New perspectives on the involvement of mTOR in depression as well as in the action of antidepressant drugs. Br J Clin Pharmacol. 2016;82:1280-1290. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 140] [Cited by in RCA: 129] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 85. | Choudhury D, Autry AE, Tolias KF, Krishnan V. Ketamine: Neuroprotective or Neurotoxic? Front Neurosci. 2021;15:672526. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 86. | Li YJ, Li YJ, Yang LD, Zhang K, Zheng KY, Wei XM, Yang Q, Niu WM, Zhao MG, Wu YM. Silibinin exerts antidepressant effects by improving neurogenesis through BDNF/TrkB pathway. Behav Brain Res. 2018;348:184-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 87. | Duman CH, Duman RS. Spine synapse remodeling in the pathophysiology and treatment of depression. Neurosci Lett. 2015;601:20-29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 241] [Cited by in RCA: 206] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 88. | Molero P, Ramos-Quiroga JA, Martin-Santos R, Calvo-Sánchez E, Gutiérrez-Rojas L, Meana JJ. Antidepressant Efficacy and Tolerability of Ketamine and Esketamine: A Critical Review. CNS Drugs. 2018;32:411-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 197] [Article Influence: 24.6] [Reference Citation Analysis (0)] |
| 89. | Faustino Martins AC, Badenoch B, da Silva Gomes R. Insights for the Next Generation of Ketamine for the Treatment of Depressive Disorder. J Med Chem. 2025;68:944-952. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 90. | Zanos P, Moaddel R, Morris PJ, Georgiou P, Fischell J, Elmer GI, Alkondon M, Yuan P, Pribut HJ, Singh NS, Dossou KS, Fang Y, Huang XP, Mayo CL, Wainer IW, Albuquerque EX, Thompson SM, Thomas CJ, Zarate CA Jr, Gould TD. NMDAR inhibition-independent antidepressant actions of ketamine metabolites. Nature. 2016;533:481-486. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1440] [Cited by in RCA: 1257] [Article Influence: 125.7] [Reference Citation Analysis (0)] |
| 91. | Hashimoto K. Molecular mechanisms of the rapid-acting and long-lasting antidepressant actions of (R)-ketamine. Biochem Pharmacol. 2020;177:113935. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 179] [Article Influence: 29.8] [Reference Citation Analysis (0)] |
| 92. | Zhang Y, Gu F, Chen J, Dong W. Chronic antidepressant administration alleviates frontal and hippocampal BDNF deficits in CUMS rat. Brain Res. 2010;1366:141-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 99] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 93. | Hrdina PD, Vu TB. Chronic fluoxetine treatment upregulates 5-HT uptake sites and 5-HT2 receptors in rat brain: an autoradiographic study. Synapse. 1993;14:324-331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 106] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 94. | Park SH, Lee YS, Yang HJ, Song GJ. Fluoxetine Potentiates Phagocytosis and Autophagy in Microglia. Front Pharmacol. 2021;12:770610. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 95. | Scabia G, Barone I, Mainardi M, Ceccarini G, Scali M, Buzzigoli E, Dattilo A, Vitti P, Gastaldelli A, Santini F, Pizzorusso T, Maffei L, Maffei M. The antidepressant fluoxetine acts on energy balance and leptin sensitivity via BDNF. Sci Rep. 2018;8:1781. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 37] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 96. | Hedegaard C, Kjaer-Sorensen K, Madsen LB, Henriksen C, Momeni J, Bendixen C, Oxvig C, Larsen K. Porcine synapsin 1: SYN1 gene analysis and functional characterization of the promoter. FEBS Open Bio. 2013;3:411-420. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 16] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 97. | Bianchi M, Baulieu EE. 3β-Methoxy-pregnenolone (MAP4343) as an innovative therapeutic approach for depressive disorders. Proc Natl Acad Sci U S A. 2012;109:1713-1718. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 52] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 98. | Nishimoto T, Kadoyama K, Taniguchi T, Takano M, Otani M, Nakamura-Hirota T, Lu Y, Matsumoto A, Matsuyama S. Synaptotagmin1 synthesis induced by synaptic plasticity in mouse hippocampus through activation of nicotinic acetylcholine receptors. Neurosci Lett. 2011;489:25-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 99. | Marco EM, Valero M, de la Serna O, Aisa B, Borcel E, Ramirez MJ, Viveros MP. Maternal deprivation effects on brain plasticity and recognition memory in adolescent male and female rats. Neuropharmacology. 2013;68:223-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 94] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 100. | Reinés A, Cereseto M, Ferrero A, Sifonios L, Podestá MF, Wikinski S. Maintenance treatment with fluoxetine is necessary to sustain normal levels of synaptic markers in an experimental model of depression: correlation with behavioral response. Neuropsychopharmacology. 2008;33:1896-1908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 58] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 101. | Zanos P, Thompson SM, Duman RS, Zarate CA Jr, Gould TD. Convergent Mechanisms Underlying Rapid Antidepressant Action. CNS Drugs. 2018;32:197-227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 153] [Cited by in RCA: 135] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 102. | Haroon E, Miller AH. Inflammation Effects on Brain Glutamate in Depression: Mechanistic Considerations and Treatment Implications. Curr Top Behav Neurosci. 2017;31:173-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 107] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 103. | Fang W, Zhang J, Hong L, Huang W, Dai X, Ye Q, Chen X. Metformin ameliorates stress-induced depression-like behaviors via enhancing the expression of BDNF by activating AMPK/CREB-mediated histone acetylation. J Affect Disord. 2020;260:302-313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 111] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 104. | Mosiołek A, Pięta A, Jakima S, Zborowska N, Mosiołek J, Szulc A. Effects of Antidepressant Treatment on Peripheral Biomarkers in Patients with Major Depressive Disorder (MDD). J Clin Med. 2021;10:1706. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 105. | Sheline YI. Neuroimaging studies of mood disorder effects on the brain. Biol Psychiatry. 2003;54:338-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 494] [Cited by in RCA: 425] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 106. | Bremner JD, Vythilingam M, Vermetten E, Nazeer A, Adil J, Khan S, Staib LH, Charney DS. Reduced volume of orbitofrontal cortex in major depression. Biol Psychiatry. 2002;51:273-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 421] [Cited by in RCA: 364] [Article Influence: 15.2] [Reference Citation Analysis (3)] |
| 107. | Campbell S, MacQueen G. An update on regional brain volume differences associated with mood disorders. Curr Opin Psychiatry. 2006;19:25-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 141] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 108. | Drevets WC. Neuroimaging studies of mood disorders. Biol Psychiatry. 2000;48:813-829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 999] [Cited by in RCA: 866] [Article Influence: 33.3] [Reference Citation Analysis (0)] |
| 109. | Videbech P, Ravnkilde B. Hippocampal volume and depression: a meta-analysis of MRI studies. Am J Psychiatry. 2004;161:1957-1966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1290] [Cited by in RCA: 1139] [Article Influence: 51.8] [Reference Citation Analysis (6)] |
| 110. | Sheline YI, Gado MH, Price JL. Amygdala core nuclei volumes are decreased in recurrent major depression. Neuroreport. 1998;9:2023-2028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 368] [Cited by in RCA: 318] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 111. | Sheline YI, Wang PW, Gado MH, Csernansky JG, Vannier MW. Hippocampal atrophy in recurrent major depression. Proc Natl Acad Sci U S A. 1996;93:3908-3913. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1484] [Cited by in RCA: 1321] [Article Influence: 44.0] [Reference Citation Analysis (4)] |
| 112. | Sheline YI, Sanghavi M, Mintun MA, Gado MH. Depression duration but not age predicts hippocampal volume loss in medically healthy women with recurrent major depression. J Neurosci. 1999;19:5034-5043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1061] [Cited by in RCA: 882] [Article Influence: 32.7] [Reference Citation Analysis (3)] |
| 113. | Mervaala E, Föhr J, Könönen M, Valkonen-Korhonen M, Vainio P, Partanen K, Partanen J, Tiihonen J, Viinamäki H, Karjalainen AK, Lehtonen J. Quantitative MRI of the hippocampus and amygdala in severe depression. Psychol Med. 2000;30:117-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 297] [Cited by in RCA: 256] [Article Influence: 9.8] [Reference Citation Analysis (3)] |
| 114. | Sheline YI, Gado MH, Kraemer HC. Untreated depression and hippocampal volume loss. Am J Psychiatry. 2003;160:1516-1518. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 929] [Cited by in RCA: 799] [Article Influence: 34.7] [Reference Citation Analysis (5)] |
| 115. | Jin L, Gao LF, Sun DS, Wu H, Wang Q, Ke D, Lei H, Wang JZ, Liu GP. Long-term Ameliorative Effects of the Antidepressant Fluoxetine Exposure on Cognitive Deficits in 3 × TgAD Mice. Mol Neurobiol. 2017;54:4160-4171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 42] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 116. | Stockmeier CA, Mahajan GJ, Konick LC, Overholser JC, Jurjus GJ, Meltzer HY, Uylings HB, Friedman L, Rajkowska G. Cellular changes in the postmortem hippocampus in major depression. Biol Psychiatry. 2004;56:640-650. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 589] [Cited by in RCA: 544] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 117. | Moskowitz MA, Lo EH. Neurogenesis and apoptotic cell death. Stroke. 2003;34:324-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 34] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 118. | van Praag H, Kempermann G, Gage FH. Neural consequences of environmental enrichment. Nat Rev Neurosci. 2000;1:191-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2005] [Cited by in RCA: 1745] [Article Influence: 67.1] [Reference Citation Analysis (0)] |
| 119. | Malberg JE, Hen R, Madsen TM. Adult Neurogenesis and Antidepressant Treatment: The Surprise Finding by Ron Duman and the Field 20 Years Later. Biol Psychiatry. 2021;90:96-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 120. | Malberg JE, Duman RS. Cell proliferation in adult hippocampus is decreased by inescapable stress: reversal by fluoxetine treatment. Neuropsychopharmacology. 2003;28:1562-1571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 653] [Cited by in RCA: 589] [Article Influence: 25.6] [Reference Citation Analysis (0)] |
| 121. | Banasr M, Valentine GW, Li XY, Gourley SL, Taylor JR, Duman RS. Chronic unpredictable stress decreases cell proliferation in the cerebral cortex of the adult rat. Biol Psychiatry. 2007;62:496-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 296] [Cited by in RCA: 282] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 122. | Jayatissa MN, Bisgaard C, Tingström A, Papp M, Wiborg O. Hippocampal cytogenesis correlates to escitalopram-mediated recovery in a chronic mild stress rat model of depression. Neuropsychopharmacology. 2006;31:2395-2404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 305] [Cited by in RCA: 279] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 123. | Czéh B, Michaelis T, Watanabe T, Frahm J, de Biurrun G, van Kampen M, Bartolomucci A, Fuchs E. Stress-induced changes in cerebral metabolites, hippocampal volume, and cell proliferation are prevented by antidepressant treatment with tianeptine. Proc Natl Acad Sci U S A. 2001;98:12796-12801. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 861] [Cited by in RCA: 778] [Article Influence: 31.1] [Reference Citation Analysis (4)] |
| 124. | Warner-Schmidt JL, Duman RS. Hippocampal neurogenesis: opposing effects of stress and antidepressant treatment. Hippocampus. 2006;16:239-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 624] [Cited by in RCA: 573] [Article Influence: 28.7] [Reference Citation Analysis (0)] |
| 125. | Dranovsky A, Hen R. Hippocampal neurogenesis: regulation by stress and antidepressants. Biol Psychiatry. 2006;59:1136-1143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 529] [Cited by in RCA: 485] [Article Influence: 24.3] [Reference Citation Analysis (0)] |
| 126. | Czéh B, Welt T, Fischer AK, Erhardt A, Schmitt W, Müller MB, Toschi N, Fuchs E, Keck ME. Chronic psychosocial stress and concomitant repetitive transcranial magnetic stimulation: effects on stress hormone levels and adult hippocampal neurogenesis. Biol Psychiatry. 2002;52:1057-1065. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 273] [Cited by in RCA: 239] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 127. | Duman RS. Depression: a case of neuronal life and death? Biol Psychiatry. 2004;56:140-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 475] [Cited by in RCA: 430] [Article Influence: 19.5] [Reference Citation Analysis (3)] |
| 128. | Manji HK, Duman RS. Impairments of neuroplasticity and cellular resilience in severe mood disorders: implications for the development of novel therapeutics. Psychopharmacol Bull. 2001;35:5-49. [PubMed] |
| 129. | D'Sa C, Duman RS. Antidepressants and neuroplasticity. Bipolar Disord. 2002;4:183-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 415] [Cited by in RCA: 356] [Article Influence: 14.8] [Reference Citation Analysis (3)] |
| 130. | Duman CH, Schlesinger L, Kodama M, Russell DS, Duman RS. A role for MAP kinase signaling in behavioral models of depression and antidepressant treatment. Biol Psychiatry. 2007;61:661-670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 306] [Cited by in RCA: 290] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 131. | Schechter LE, Ring RH, Beyer CE, Hughes ZA, Khawaja X, Malberg JE, Rosenzweig-Lipson S. Innovative approaches for the development of antidepressant drugs: current and future strategies. NeuroRx. 2005;2:590-611. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 162] [Cited by in RCA: 135] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 132. | Zarate CA Jr, Payne JL, Quiroz J, Sporn J, Denicoff KK, Luckenbaugh D, Charney DS, Manji HK. An open-label trial of riluzole in patients with treatment-resistant major depression. Am J Psychiatry. 2004;161:171-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 264] [Cited by in RCA: 229] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 133. | Sanacora G, Kendell SF, Fenton L, Coric V, Krystal JH. Riluzole augmentation for treatment-resistant depression. Am J Psychiatry. 2004;161:2132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 51] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 134. | Katoh-Semba R, Asano T, Ueda H, Morishita R, Takeuchi IK, Inaguma Y, Kato K. Riluzole enhances expression of brain-derived neurotrophic factor with consequent proliferation of granule precursor cells in the rat hippocampus. FASEB J. 2002;16:1328-1330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 139] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 135. | Duman RS. Role of neurotrophic factors in the etiology and treatment of mood disorders. Neuromolecular Med. 2004;5:11-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 432] [Cited by in RCA: 372] [Article Influence: 16.9] [Reference Citation Analysis (1)] |
| 136. | Jin K, Zhu Y, Sun Y, Mao XO, Xie L, Greenberg DA. Vascular endothelial growth factor (VEGF) stimulates neurogenesis in vitro and in vivo. Proc Natl Acad Sci U S A. 2002;99:11946-11950. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1271] [Cited by in RCA: 1177] [Article Influence: 49.0] [Reference Citation Analysis (0)] |
| 137. | Louissaint A Jr, Rao S, Leventhal C, Goldman SA. Coordinated interaction of neurogenesis and angiogenesis in the adult songbird brain. Neuron. 2002;34:945-960. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 620] [Cited by in RCA: 559] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 138. | Meirer R, Gurunluoglu R, Siemionow M. Neurogenic perspective on vascular endothelial growth factor: review of the literature. J Reconstr Microsurg. 2001;17:625-630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 139. | Warner-Schmidt JL, Duman RS. VEGF is an essential mediator of the neurogenic and behavioral actions of antidepressants. Proc Natl Acad Sci U S A. 2007;104:4647-4652. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 367] [Cited by in RCA: 349] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 140. | Heine VM, Zareno J, Maslam S, Joëls M, Lucassen PJ. Chronic stress in the adult dentate gyrus reduces cell proliferation near the vasculature and VEGF and Flk-1 protein expression. Eur J Neurosci. 2005;21:1304-1314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 159] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 141. | Deyama S, Aoki S, Sugie R, Fukuda H, Shuto S, Minami M, Kaneda K. Intranasal Administration of Resolvin E1 Produces Antidepressant-Like Effects via BDNF/VEGF-mTORC1 Signaling in the Medial Prefrontal Cortex. Neurotherapeutics. 2023;20:484-501. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 142. | Li JM, Zhao Y, Sun Y, Kong LD. Potential effect of herbal antidepressants on cognitive deficit: Pharmacological activity and possible molecular mechanism. J Ethnopharmacol. 2020;257:112830. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 143. | Shors TJ, Miesegaes G, Beylin A, Zhao M, Rydel T, Gould E. Neurogenesis in the adult is involved in the formation of trace memories. Nature. 2001;410:372-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1571] [Cited by in RCA: 1458] [Article Influence: 58.3] [Reference Citation Analysis (0)] |
| 144. | Leuner B, Gould E, Shors TJ. Is there a link between adult neurogenesis and learning? Hippocampus. 2006;16:216-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 421] [Cited by in RCA: 387] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 145. | Kempermann G, Wiskott L, Gage FH. Functional significance of adult neurogenesis. Curr Opin Neurobiol. 2004;14:186-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 491] [Cited by in RCA: 460] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 146. | Shors TJ, Townsend DA, Zhao M, Kozorovitskiy Y, Gould E. Neurogenesis may relate to some but not all types of hippocampal-dependent learning. Hippocampus. 2002;12:578-584. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 664] [Cited by in RCA: 636] [Article Influence: 26.5] [Reference Citation Analysis (0)] |
| 147. | MacQueen GM, Campbell S, McEwen BS, Macdonald K, Amano S, Joffe RT, Nahmias C, Young LT. Course of illness, hippocampal function, and hippocampal volume in major depression. Proc Natl Acad Sci U S A. 2003;100:1387-1392. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 754] [Cited by in RCA: 680] [Article Influence: 29.6] [Reference Citation Analysis (4)] |
| 148. | Chen SJ, Kao CL, Chang YL, Yen CJ, Shui JW, Chien CS, Chen IL, Tsai TH, Ku HH, Chiou SH. Antidepressant administration modulates neural stem cell survival and serotoninergic differentiation through bcl-2. Curr Neurovasc Res. 2007;4:19-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 149. | Czéh B, Simon M, Schmelting B, Hiemke C, Fuchs E. Astroglial plasticity in the hippocampus is affected by chronic psychosocial stress and concomitant fluoxetine treatment. Neuropsychopharmacology. 2006;31:1616-1626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 368] [Cited by in RCA: 346] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 150. | Malberg JE, Eisch AJ, Nestler EJ, Duman RS. Chronic antidepressant treatment increases neurogenesis in adult rat hippocampus. J Neurosci. 2000;20:9104-9110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2538] [Cited by in RCA: 2219] [Article Influence: 85.3] [Reference Citation Analysis (3)] |
| 151. | Madsen TM, Treschow A, Bengzon J, Bolwig TG, Lindvall O, Tingström A. Increased neurogenesis in a model of electroconvulsive therapy. Biol Psychiatry. 2000;47:1043-1049. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 629] [Cited by in RCA: 552] [Article Influence: 21.2] [Reference Citation Analysis (0)] |