Published online Apr 19, 2026. doi: 10.5498/wjp.v16.i4.115109
Revised: November 20, 2025
Accepted: December 23, 2025
Published online: April 19, 2026
Processing time: 173 Days and 17.1 Hours
Early-life stress (ELS) constitutes a significant risk factor for the development of neuropsychiatric disorders, including depression, anxiety, and schizophrenia, through its enduring impact on neural circuitry, endocrine function, and epige
Core Tip: Early-life stress increases lifelong risk for neuropsychiatric disorders via lasting neural, endocrine, and epigenetic changes. Oxytocin, a key neuropeptide, shows promise in mitigating these effects by normalizing amygdala-prefrontal connectivity, reducing hypothalamic-pituitary-adrenal axis hyperactivity, promoting hippocampal neurogenesis, and reversing stress-associated epigenetic marks. However, its efficacy is context-dependent and influenced by factors like sex, timing, and administration route. Future oxytocin-based therapies should be integrated with psychosocial support and tailored through a precision medicine framework to effectively promote resilience in the affected individuals.
- Citation: Zhang Y, Wang S, Hei MY. Attenuating neuropsychiatric disorders of early-life stress: The protective role of oxytocin. World J Psychiatry 2026; 16(4): 115109
- URL: https://www.wjgnet.com/2220-3206/full/v16/i4/115109.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i4.115109
Early-life stress (ELS) refers to a spectrum of adverse experiences occurring during sensitive developmental periods from the prenatal stage through adolescence, including neglect, abuse, socioeconomic deprivation, and disrupted caregiving[1-3]. Epidemiological studies indicate that a significant proportion of children worldwide are exposed to at least one form of ELS, with heightened prevalence among marginalized populations[4-7]. The Adverse Childhood Experiences study[5] and subsequent research have consistently demonstrated that ELS is associated with lifelong vulnerabilities to psychiatric disorders such as depression, anxiety, and schizophrenia, as well as impairments in social cognition and emotional regu
The neurobiological sequelae of ELS are multifaceted, involving dysregulation of the hypothalamic-pituitary-adrenal (HPA) axis[13,14], epigenetic modifications in stress-related genes[15,16], aberrant synaptic plasticity[17,18], and chronic neuroinflammation[19,20]. Key brain regions implicated in emotional and cognitive processing [such as the amygdala, prefrontal cortex (PFC), and hippocampus] undergo structural and functional alterations that contribute to increased psychiatric risk[21,22]. Despite advances in characterizing these mechanisms, translational interventions that effectively prevent or reverse the long-term effects of ELS remain limited[23]. Most current therapeutic approaches focus on symptom management rather than targeting the underlying neurodevelopmental disruptions induced by early adversity[24].
Oxytocin, a neuropeptide produced in the hypothalamus, has gained increasing attention for its potential role in mitigating the effects of stress and promoting adaptive social and emotional functions[25-27]. Beyond its well-established functions in parturition, lactation, and social bonding, oxytocin exerts profound effects on stress circuitry, neural plasticity, and inflammatory processes[28-31]. Preclinical studies indicate that oxytocin administration can normalize HPA axis activity, enhance GABAergic inhibition in the amygdala, and stimulate neurogenesis in the hippocampus[32,33]. Furthermore, oxytocin has been shown to modulate DNA methylation patterns of stress-related genes, suggesting a role in epigenetic regulation[34,35].
In humans, intranasal oxytocin has demonstrated promise in improving social cognition and reducing amygdala hyperreactivity to negative emotional stimuli, particularly among individuals with histories of childhood trauma[36,37]. Nevertheless, the therapeutic application of oxytocin is not without controversy. Questions such as context-dependent effects, sex differences, optimal dosing, and long-term safety require further investigation[28]. Moreover, emerging evidence suggests that the efficacy of oxytocin may be enhanced when combined with psychosocial interventions, highlighting the importance of an integrated treatment approach[38-40].
This review synthesizes contemporary evidence from animal and human studies on the protective effects of oxytocin against ELS-induced neuropsychiatric impairments. We examine the mechanisms through oxytocin modulates neural, endocrine, and immune systems, and discuss its potential to promote resilience following early adversity. Finally, we identify critical gaps in current knowledge and propose directions for future research aimed at developing oxytocin-based therapeutic strategies within a precision medicine framework.
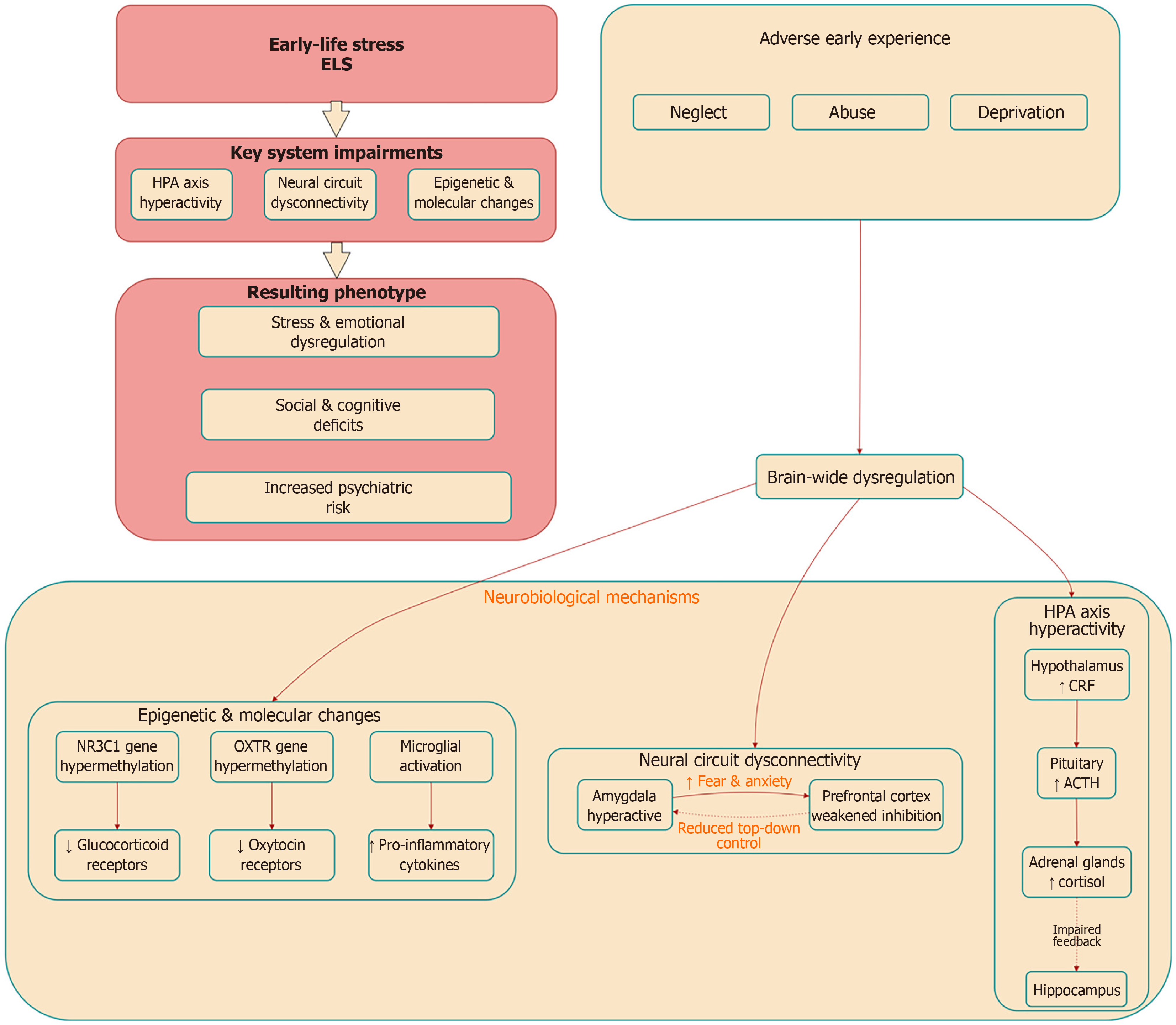
Exposure to ELS induces profound and often persistent alterations in brain development and function, impacting neural circuits critical for emotional regulation, threat processing, and cognitive control[1-3,41,42]. One of the most consistently reported neural sequelae of ELS is the dysregulation of amygdala-PFC connectivity[43,44]. ELS leads to amygdala hyperactivity, characterized by heightened reactivity to threat-related stimuli, coupled with a reduction in top-down inhibitory control from the PFC[43,44]. This imbalance disrupts the typical modulation of emotional responses, resulting in increased anxiety-like behaviors, impaired fear extinction, and a heightened risk for anxiety disorders later in life[45,46]. Neuroimaging studies in humans corroborate these findings, showing altered functional connectivity between the amygdala and medial PFC (mPFC) in adults with histories of childhood trauma[47-49].
The HPA axis represents another core system vulnerable to the impact of ELS[50-52]. Chronic or severe early adversity frequently leads to a hyperactive HPA axis phenotype, marked by exaggerated cortisol release in response to stress and impaired negative feedback inhibition[53,54]. This dysregulation arises from several mechanisms, including reduced expression of glucocorticoid receptors (GRs) in the hippocampus and altered corticotropin-releasing factor (CRF) signaling in the hypothalamus[55,56]. Prolonged elevation of glucocorticoids contributes to structural changes in the brain, most notably hippocampal atrophy, through the suppression of neurogenesis in the dentate gyrus and the promotion of dendritic retraction in CA3 pyramidal neurons[25,57]. These morphological changes are associated with deficits in memory and contextual fear processing[58,59].
At the epigenetic level, ELS can embed long-lasting molecular marks that alter gene expression and neuronal function without changing the underlying DNA sequence[15,60,61]. A well-characterized mechanism involves the methylation of the NR3C1 gene, which encodes the GR[62,63]. Increased methylation of specific promoter regions of NR3C1 is associated with reduced GR expression, impaired HPA axis feedback, and a sustained stress-reactive phenotype[64,65]. Similarly, ELS has been linked to epigenetic modifications of the OXTR gene[66,67]. Hypermethylation of OXTR promoter regions results in decreased OXTR availability in limbic and cortical regions, potentially compromising social reward processing, stress buffering, and affiliative behaviors[68,69]. These epigenetic alterations are thought to represent adaptive responses to a hostile early environment that may become maladaptive in later, more supportive contexts, thereby increasing long-term vulnerability to psychopathology[68,69].
Furthermore, ELS promotes a state of low-grade neuroinflammation, characterized by elevated pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α[19,20]. This inflammatory milieu can exacerbate neuronal damage, inhibit neurotrophic factor signaling, and further disrupt neuroendocrine homeostasis[70]. The convergence of these neural, endocrine, epigenetic, and inflammatory changes creates a biological substrate that is predisposed to maladaptive stress responses and psychiatric disorders, underscoring the need for interventions that can target these multifaceted mechanisms[14,71]. The convergence of these neural, endocrine, epigenetic, and inflammatory changes creates a biological substrate predisposed to maladaptive stress responses, as summarized in Figure 1.
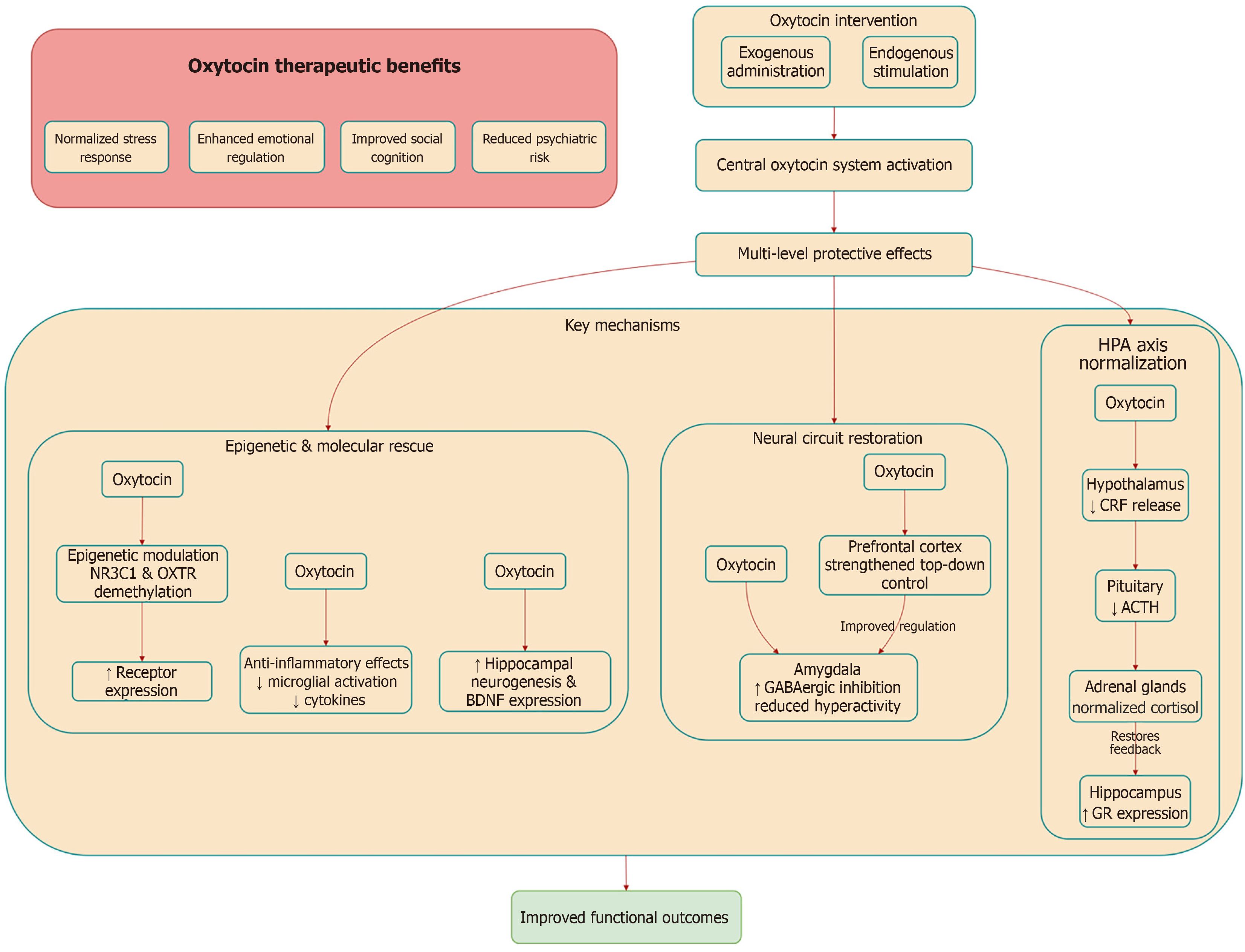
Oxytocin demonstrates a remarkable capacity to counteract the multifaceted neural and molecular alterations induced by ELS, functioning through a series of complementary and often synergistic mechanisms[25,26,31]. Preclinical studies, particularly those utilizing rodent models of maternal separation (MS) or fragmented care, have provided evidence that exogenous oxytocin administration during critical developmental windows can rescue many of the negative effects of ELS[72-74]. A key mechanism involves the restoration of amygdala-PFC connectivity, where oxytocin dampens amygdala hyperactivity by enhancing local GABAergic inhibition[75,76] while concurrently strengthening top-down regulatory input from the PFC, potentially via dopaminergic modulation[77,78]. This neural reorganization is correlated with measurable behavioral improvements, including reduced anxiety-like behaviors and the restoration of social preference in adulthood[79,80].
Oxytocin potently normalizes HPA axis hyperactivity by attenuating the exaggerated corticosterone response to stress[28,32,39,81]. This is mediated via OXTRs in key limbic and hypothalamic regions, inhibiting CRF release and facilitating the stress-buffering effects of social support[82-84]. OXTRs are densely expressed in key limbic and hypothalamic regions, and their activation inhibits the release of CRF and facilitates the stress-buffering effects of social support[83,84]. Furthermore, oxytocin promotes resilience by stimulating neuroplasticity within the hippocampus[85,86]. It enhances hippocampal neurogenesis, synaptogenesis, and the expression of brain-derived neurotrophic factor (BDNF), which collectively help to reverse dendritic atrophy and volume loss associated with chronic stress and high glucocorticoid exposure[87,88].
Another significant protective mechanism involves oxytocin’s potent anti-inflammatory properties[28]. ELS can trigger a persistent state of low-grade neuroinflammation, characterized by activated microglia and elevated levels of pro-inflammatory cytokines. Oxytocin counteracts this process by reducing microglial activation and suppressing the production of cytokines such as IL-1β, IL-6, and TNF-α, while simultaneously promoting anti-inflammatory signaling pathways[19]. This immunomodulatory action helps restore neuroendocrine-immune homeostasis, which is frequently disrupted following early adversity.
Perhaps most intriguing is emerging evidence suggesting that oxytocin can influence the epigenetic landscape altered by ELS[89]. Animal studies indicate that oxytocin administration can modulate DNA methylation patterns of stress-related genes. For instance, oxytocin has been shown to normalize methylation levels of the GR gene (NR3C1) in the hippocampus, leading to increased GR expression and improved HPA feedback sensitivity[90]. Similarly, oxytocin can upregulate OXTR gene expression in stress-sensitive brain regions, potentially by altering the methylation status of the OXTR promoter[91]. These findings posit that oxytocin may contribute to a kind of epigenetic “reprogramming”, helping to reverse the molecular scars of early adversity and open windows of plasticity for therapeutic intervention.
Collectively, these mechanisms position oxytocin as a unique therapeutic agent capable of targeting ELS-induced dysregulation across multiple levels from neural circuits and endocrine systems to inflammatory responses and epigenetic modifications[92]. However, the efficacy of these protective effects is highly dependent on factors such as the timing of administration, dosage, and the context in which oxytocin is delivered, underscoring the necessity for further research to optimize its translational potential[28,93]. Collectively, these multi-level mechanisms position oxytocin as a unique therapeutic agent capable of targeting ELS-induced dysregulation, with its integrative actions summarized in Figure 2.
The following sections synthesize translational evidence from animal and human research, with key comparative findings across species, behavioral domains, and neural systems detailed in Table 1.
| Domain/system | Preclinical findings (rodent models) | Human studies (intranasal oxytocin) | Key convergences & divergences |
| Amygdala reactivity & connectivity | Reduces BLA hyperactivity; strengthens amygdala-mPFC connectivity[77,78] | Attenuates amygdala hyperreactivity to threat; enhances amygdala-PFC functional connectivity[110,114,117] | Strong convergence: OT consistently normalizes amygdala-centric threat circuits across species |
| HPA axis function | Attenuates stress-induced corticosterone release; improves GR feedback sensitivity[32,98] | Context-dependent blunting of cortisol response; effects more pronounced with supportive context[28,82,167] | Divergence/complexity: Effects in humans are less robust and more dependent on psychological context |
| Social behavior | Rescues social preference and social memory deficits[101,102] | Enhances trust, eye contact, and emotion recognition; effects strongest in ELS-exposed[36,37,127] | Convergence: OT promotes prosocial behaviors. Divergence: Human effects are highly sensitive to perceived trustworthiness of others |
| Epigenetic modulation | Normalizes methylation of NR3C1 and OXTR genes in limbic regions[90,91] | OXTR methylation status moderates behavioral and neural response to OT[121,122] | Emerging convergence: Epigenetic state of the oxytocin system is a key moderator of treatment response |
| Key moderators | Sex, timing, dose, specific ELS paradigm[32,154] | Sex, ELS type, OXTR genotype/methylation, therapeutic context[28,93,124,157] | Strong Convergence: Efficacy is universally moderated by individual differences and context, not a one-size-fits-all |
| Amygdala reactivity & connectivity | Reduces BLA hyperactivity; strengthens amygdala-mPFC connectivity[77,78] | Attenuates amygdala hyperreactivity to threat; enhances amygdala-PFC functional connectivity[110,114,117] | Strong convergence: OT consistently normalizes amygdala-centric threat circuits across species |
| HPA axis function | Attenuates stress-induced corticosterone release; improves GR feedback sensitivity[32,98] | Context-dependent blunting of cortisol response; effects more pronounced with supportive context[28,82,167] | Divergence/Complexity: Effects in humans are less robust and more dependent on psychological context |
Preclinical models have been instrumental in elucidating how exogenous oxytocin administration can counteract the neurobiological alterations induced by ELS, as outlined in Oxytocin’s protective mechanisms section. Extensive pre
A key insight from this body of work is that oxytocin intervention delivered during the early postnatal period can prevent the emergence of behavioral and neuroendocrine deficits in adulthood[96]. For instance, in rat pups subjected to prolonged MS, daily subcutaneous or intranasal administration of oxytocin during the first weeks of life has been shown to mitigate the development of anxiety- and depression-like behaviors in adulthood[96]. These behavioral improvements are correlated with the rescue of ELS-induced basolateral amygdala hyperactivation and the strengthening of amygdala-mPFC connectivity, resulting in improved fear extinction and emotional regulation[77,78,97].
Consistent with its established neuroendocrine role in above, oxytocin administration in ELS models buffers HPA axis dysregulation, resulting in an attenuated corticosterone response to acute stressors[32,98]. Furthermore, oxytocin promotes neural resilience by stimulating structural and functional plasticity within the hippocampus[86,99]. Studies report that oxytocin reverses the suppression of hippocampal neurogenesis typically seen in MS models and increases the expression of synaptic markers and BDNF, thereby supporting cognitive functions such as learning and memory that are often impaired by ELS[87,88,100].
The prosocial and cognitive benefits of oxytocin are also evident. Oxytocin-treated rats that experienced ELS show enhanced social preference compared to saline-treated controls, and demonstrate improvements in social memory tests[101-104]. These behavioral changes are supported by oxytocin’s ability to modulate dopaminergic reward pathways and increase OXTR binding in regions critical for social behavior, such as the nucleus accumbens and the anterior cingulate cortex (ACC)[103-105].
Importantly, research points to a dose-dependent and often biphasic effect of oxytocin, where moderate doses produce optimal therapeutic outcomes while very high doses may be ineffective or even anxiogenic[97]. The concept of “priming” the oxytocinergic system is also prominent; early oxytocin exposure can upregulate central OXTR expression, creating a more resilient phenotype that is better equipped to handle future stressors[106]. However, it is crucial to note that the effects of oxytocin are not uniform and can be influenced by factors such as the sex of the animal, the specific nature and timing of the ELS paradigm, and the social context, underscoring the complexity of translating these findings into therapeutic applications[32,34].
The promising findings from preclinical models provide a compelling neurobiological framework and identify key mechanistic targets for oxytocin intervention[3,94,95]. These models predict that oxytocin should normalize threat-related amygdala hyperactivity, strengthen prefrontal regulatory control, and buffer HPA axis stress responses in humans with ELS histories[43,44]. They further suggest that the efficacy of oxytocin may be influenced by the individual's oxytocin system state, potentially reflected by epigenetic markers such as OXTR methylation[93]. Translational research in humans provides compelling, though complex, evidence supporting the potential of oxytocin as a therapeutic intervention for individuals with histories of ELS[92,107]. A primary focus has been on the effects of intranasal oxytocin administration, a non-invasive method believed to facilitate central nervous system delivery, on emotional processing, neural activity, and social cognition[36,108,109]. The following section evaluates the translational evidence for these predictions in human studies. While human research largely supports these core mechanisms, it also introduces critical layers of complexity that are less easily captured in controlled animal experiments but are paramount for therapeutic application.
A consistent finding from neuroimaging studies is that intranasal oxytocin modulates limbic reactivity, particularly amygdala hyperactivity, which is a well-documented neural correlate of ELS[110,111]. Adults who experienced childhood trauma often exhibit heightened amygdala responses to threatening or negative social stimuli[112,113]. Single-dose administration of intranasal oxytocin in these individuals has been shown to attenuate this exaggerated amygdala activity, effectively normalizing the neural response to threat[114-116]. Furthermore, oxytocin enhances functional con
Beyond acute neural effects, research has begun to illuminate how an individual’s oxytocin system interacts with their early environment. Epigenetic studies reveal that the methylation status of the OXTR gene serves as a molecular link between ELS and psychiatric vulnerability[67]. Higher levels of OXTR promoter methylation, which typically suppresses gene expression and receptor availability, have been associated with more severe childhood adversity and greater symptom severity in disorders like anxiety and depression[2,67,120]. Critically, emerging evidence suggests that the efficacy of intranasal oxytocin may be influenced by this pre-existing epigenetic landscape[121]. Some studies propose that individuals with higher OXTR methylation may demonstrate a more pronounced beneficial response to exogenous oxytocin administration, a concept often referred to as “compensation”[122,123]. However, this interaction is nuanced and may be further modulated by factors such as the specific type of childhood trauma, sex, and current psychosocial context[28,124].
Behaviorally, intranasal oxytocin has been found to enhance the processing of positive social cues, increase trust and eye contact, and improve the recognition of emotional states in others[36,37]. These prosocial and cognitive effects are particularly relevant for ELS-exposed populations, who frequently struggle with social withdrawal and misinterpretation of social intentions[125,126]. For instance, individuals with borderline personality disorder and a history of childhood trauma have shown reduced amygdala activation and felt less mistrustful after oxytocin administration when viewing angry faces[127].
Despite these promising findings, clinical trials are characterized by significant variability. Factors such as dose, frequency of administration, sex, diagnostic comorbidity, and the surrounding social and therapeutic context appear to critically moderate oxytocin’s effects[28,128,129]. In some cases, oxytocin has been shown to enhance the salience of both positive and negative social stimuli, meaning that without a supportive environment, its effects might not be uniformly beneficial[93,130,131]. Moreover, oxytocin is not a simple “anti-anxiety” drug but rather a neuropeptide that facilitates social engagement, which can be therapeutic in a safe context but may potentially exacerbate distress in a negative one[93,132-134].
In summary, clinical studies lend support to the premise that oxytocin can modulate key neural circuits dysregulated by ELS and can interact with the epigenetic footprints of early adversity. However, they also highlight that its therapeutic application is controversial. Future research must prioritize individualized approaches that account for personal history, neurobiological phenotype, and the crucial role of context to fully realize oxytocin’s potential in mitigating the long-term consequences of ELS.
A significant and recurring theme in the oxytocin literature is that its effects are not uniform or universally beneficial but are profoundly modulated by the context in which it is administered and the individual’s internal state and past experiences[25,28,134-136]. This context-dependency is crucial for understanding the mixed results observed in both preclinical and human studies and poses a major consideration for its therapeutic application.
Rather than functioning as a simple anxiolytic or prosocial agent, oxytocin appears to amplify the salience or pro
Conversely, in the absence of a supportive environment, or in situations perceived as threatening, the same mechanism of increased social salience can lead to unintended adverse outcomes[140]. For example, several studies have shown that in individuals who have experienced significant social trauma or adversity, intranasal oxytocin can sometimes intensify feelings of social threat, enhance memories of negative events, or increase out-group mistrust and anxiety[27,137,140]. This suggests that oxytocin may exacerbate existing negative emotional states if administered without regard to the surrounding psychosocial context[29,93,141]. Oxytocin does not create a new emotional state but rather potentiates the processing of the existing social and emotional environment[87,142,143].
This paradox underscores that oxytocin’s efficacy is not merely a function of the external environment but is de
Furthermore, the type of ELS experienced may shape the specific ‘contexts’ that are most therapeutic. For example, oxytocin administered during exposure therapy might be most beneficial for individuals whose ELS involved explicit threat (e.g., physical abuse), as it could facilitate fear extinction by modulating amygdala-prefrontal circuitry[110,115]. In contrast, for individuals whose primary adversity was emotional neglect and social deprivation, oxytocin might be most effective when paired with interventions that explicitly train social reward processing and build affiliative skills, potentially by targeting striatal pathways[103,105]. Sex further moderates these interactions, as organizational and activational effects of gonadal hormones influence OXTR expression and binding in key brain regions, leading to divergent neurobehavioral responses to oxytocin in males and females, even with similar ELS histories[28,124]. Therefore, the ‘context’ is not a monolithic entity but is filtered through a lens shaped by genetics, epigenetics, sex, and the specific nature of early adversity, ultimately determining whether oxytocin's salience-enhancing properties will have a therapeutic or detrimental effect.
Therefore, the simplistic notion of oxytocin as a “love hormone” or a universal social lubricant has been largely superseded by a more nuanced understanding of it as a “social salience” hormone[27,121]. Its ultimate effect depends on a complex interplay between the drug, the individual’s neurobiological and epigenetic background (e.g., OXTR methylation), and the environment[93,120]. This underscores a critical limitation in its current investigational use: Administering oxytocin as a standalone pharmaceutical agent, divorced from a structured psychosocial intervention, may fail to yield consistent benefits and could, in some cases, be counterproductive. Therefore, future research must move beyond monotherapy models and rigorously explore oxytocin as an adjunctive treatment, specifically examining how it can potentiate the effects of evidence-based psychotherapy in a safe and controlled setting[28,149]. Addressing this requires the development of oxytocin-integrated psychotherapeutic protocols that provide a structured and positive context to harness its salience-enhancing properties therapeutically.
Despite the growing body of evidence supporting oxytocin’s potential in mitigating the effects of ELS, several fundamental questions remain unresolved, presenting significant barriers to its translation into routine clinical practice.
A primary area of uncertainty pertains to optimal dosing and treatment regimens. Existing studies have employed a wide range of single and repeated doses of intranasal oxytocin, yet no consensus exists on what constitutes an effective dose for specific populations or outcomes[36,129,140]. The question of whether chronic, intermittent, or acute administration is most beneficial for long-term neuroplastic changes is still open. Furthermore, the pharmacokinetics of intranasal oxytocin, which include its precise bioavailability in the central nervous system, the timing of its effects, and the potential for receptor desensitization with prolonged use, are not fully understood[150,151]. The field lacks well-established dose-response curves, making it difficult to determine the window of efficacy and avoid potential biphasic or inverted-U-shaped effects, where both insufficient and excessive doses may be ineffective[152,153]. Addressing these fundamental pharmacokinetic and dosing uncertainties demands the establishment of well-defined dose-response curves through rigorous dose-finding studies.
A second critical unresolved issue is the nature and extent of sex-specific responses. Preclinical research robustly indicates that the effects of oxytocin often vary significantly between males and females, influenced by the effects of gonadal hormones like estrogen[154-156]. However, human studies have frequently underpowered analyses to detect sex differences or have predominantly enrolled male participants[157]. Consequently, it is unclear whether people with ELS experience require different dosing strategies or might experience distinct therapeutic outcomes[158,159]. Understanding these sex-dependent mechanisms through purposefully designed studies is an essential prerequisite for developing equitable and effective treatments[153].
The long-term safety and consequences of chronic oxytocin administration represent another major gap. Most human trials have focused on acute effects following single or short-term dosing[109,135,160]. The implications of sustained oxytocin exposure on social behavior, neuroendocrine function, and emotional regulation over months or years are unknown[36,140]. Particularly concerning is the theoretical risk that chronic administration in certain contexts could potentially reinforce maladaptive social patterns or lead to dysfunctional dependency[161-163]. The safety profile in adolescents is especially understudied. Clarifying the long-term safety profile, particularly in adolescents, necessitates large-scale, longitudinal studies that monitor a range of behavioral and physiological outcomes.
Finally, the optimal method of delivery remains a subject of investigation. While intranasal administration is the current standard for human research, its efficiency in delivering sufficient concentrations to the brain is debated[150,164]. Questions surround the influence of nasal physiology, the specific brain regions reached, and the consistency of delivery across individuals[153]. Exploring alternative delivery systems, such as slow-release formulations or novel peptides that selectively activate OXTR, could overcome some of these limitations but require further development and testing[165,166]. Overcoming these delivery challenges may involve exploring alternative systems, such as slow-release formulations or the development of novel blood-brain barrier permeable oxytocin analogs.
Addressing these unresolved questions demands a concerted effort towards large-scale, longitudinal, and well-controlled studies that are specifically designed to characterize dose-response relationships, systematically examine sex differences, and monitor long-term safety. Until these fundamental issues are clarified, the transition of oxytocin from a promising research tool to a reliable therapeutic agent will remain incomplete.
The promising yet complex role of oxytocin in reducing the effects of ELS underscores a critical need to address several substantial research gaps before its therapeutic potential can be fully realized. A primary deficiency lies in the lack of standardized, validated biomarkers predictive of treatment response[153]. Current intervention studies exhibit significant interindividual variability in outcomes, highlighting an urgent need to identify who is most likely to benefit from oxytocin administration[36,66,140,141]. Integrating multi-level biomarkers, such as epigenetic markers (e.g., OXTR methylation status), neuroendocrine profiles (e.g., baseline cortisol levels), neuroimaging phenotypes (e.g., amygdala reactivity), and genetic variants, could form the basis for predictive algorithms[43,61,63,144]. This precision medicine approach would move the field beyond a one-size-fits-all model and enable the stratification of individuals with ELS histories into biologically defined subgroups most amenable to oxytocin-based interventions[150].
Furthermore, the optimization of treatment parameters remains largely empirical. Fundamental questions regarding the optimal dosing, timing, frequency, and duration of oxytocin administration for specific clinical endpoints are unanswered[36,141,153]. Rigorous dose-finding studies that establish clear dose-response relationships across different demographic groups (e.g., stratified by sex, type of ELS, and developmental stage) are essential[152,167,168]. Similarly, the ideal timing for intervention and the long-term consequences of chronic oxytocin exposure require systematic longitudinal investigation[122,169]. The development of more efficient and reliable methods for central nervous system delivery, potentially including novel intranasal devices or blood-brain barrier permeable analogs, also represents a crucial area for pharmaceutical development[36].
Another significant gap is the underexplored potential for synergistic effects between oxytocin and other neuromodulatory techniques. Combining intranasal oxytocin with interventions such as transcranial magnetic stimulation or real-time functional magnetic resonance imaging (fMRI) neurofeedback presents a novel strategy to enhance treatment efficacy[114-116,170,171]. The premise is that oxytocin may prime or “open” neural circuits for plasticity, thereby increasing the brain’s receptivity to these subsequent neuromodulatory techniques that directly target maladaptive neural patterns[170-173]. Research is needed to determine the most effective sequencing and timing of these combined interventions and to elucidate the underlying mechanisms of synergy.
Finally, a broader translational gap exists between the controlled environments of laboratory research and the complex realities of clinical practice[172]. Most studies administer oxytocin in highly standardized settings, which do not reflect the variable and often stressful contexts of real-world therapy[36,140]. Effectiveness trials conducted in community mental health settings, with clinically diverse populations and involving therapists in the administration protocol, are necessary to evaluate the feasibility, acceptability, and generalizability of oxytocin-augmented therapy[28,108,128]. Concurrently, a greater focus on the development of oxytocin-integrated psychotherapeutic protocols is needed to provide a structured and safe context that maximizes the peptide’s beneficial effects on social engagement and learning[140,150,153].
Addressing these research gaps will require a concerted shift toward larger, longitudinal, and collaborative studies that integrate methods from molecular biology, neuroscience, and clinical psychology. This multi-disciplinary effort is paramount to translating the compelling neurobiological promise of oxytocin into safe, effective, and personalized clinical applications for survivors of early adversity.
The heterogeneous nature of both ELS exposures and individual biological responses necessitates a shift away from uniform treatment models toward precision medicine approaches for oxytocin-based interventions[4]. The goal is to move beyond asking if oxytocin is effective, and instead develop the tools to determine for whom, at what dose, and in which context it will be most beneficial. This paradigm relies on identifying a suite of measurable biomarkers that can predict treatment response and guide personalized therapeutic strategies[153].
A cornerstone of this approach is the integration of genetic and epigenetic profiling. Specific single-nucleotide polymorphisms of the OXTR gene, such as rs53576 and rs2254298, have been linked to differences in social processing and stress reactivity[174-177]. Similarly, the degree of methylation in the OXTR promoter region provides a quantifiable measure of the gene’s environmental silencing, which may predict the need for and response to exogenous oxytocin[178,179]. Pre-treatment assessment of this epigenetic landscape could help identify individuals with a “hypofunctioning” oxytocin system who might benefit most from supplementation[153]. Furthermore, extending this profiling to other stress-related genes, such as the GR gene NR3C1 and FKBP5, could provide a more comprehensive biological signature of ELS-related vulnerability and resilience[180-184].
Beyond molecular markers, neurophenotyping using functional neuroimaging offers a powerful tool for target engagement and personalization. Individuals with ELS often exhibit distinct neural signatures, such as heightened amygdala reactivity to threat or reduced prefrontal-amygdala connectivity[21,22]. Baseline fMRI could be used to identify these specific neural circuit deficits, allowing clinicians to tailor oxytocin administration to the individual’s neurobiological profile[43,49]. For instance, a person with severe amygdala hyperreactivity might receive a different dosing regimen than someone whose primary deficit is in ventral striatal reward processing[21,185]. Furthermore, imaging can serve as a biomarker to confirm that oxytocin is engaging its intended target neural circuit before a full clinical trial is undertaken[115,116].
The timing and context of intervention are also critical pillars of a precision approach. Developmental stage is a key variable; administering oxytocin during windows of heightened plasticity, such as adolescence, may yield more enduring effects on neural circuit reorganization compared to administration in adulthood[36,150,164]. Moreover, as research underscores the context-dependent effects of oxytocin, precision medicine must account for the concurrent psychosocial environment. This involves systematically integrating oxytocin administration with structured, evidence-based psychotherapy that provides a safe and supportive framework[39,40]. Oxytocin is not seen as a standalone treatment but as a biological catalyst that enhances neural plasticity and receptivity to the therapeutic process, thereby potentiating the benefits of psychotherapy for individuals who have been resistant to treatment alone[31,99].
Finally, the development of novel oxytocin analogs and delivery systems is intrinsic to advancing precision medicine. Research into small-molecule agonists that can selectively activate central OXTRs, or that have improved blood-brain barrier penetration, could offer alternatives to intranasal delivery[186-189]. These innovations could provide more consistent central bioavailability and allow for more precise dosing control.
Implementing these precision medicine approaches will require large-scale, longitudinal studies that collect deep phenotypic data combining genetic, epigenetic, endocrine, neural, and behavioral measures to build predictive models of oxytocin response. By embracing this multifaceted and individualized framework, oxytocin therapy can evolve into a targeted and effective strategy to promote resilience in survivors of early adversity.
Accumulating evidence from preclinical and clinical research underscores the significant potential of oxytocin as a therapeutic agent for mitigating the long-term neuropsychiatric consequences of ELS. The neuropeptide exerts its protective effects through a multi-level mechanism, rescuing ELS-induced deficits in amygdala-prefrontal connectivity, normalizing HPA axis hyperactivity, promoting hippocampal neurogenesis, and attenuating neuroinflammatory processes. Furthermore, emerging data suggest that oxytocin may contribute to the reversal of maladaptive epigenetic modifications imposed by early adversity, thereby opening unique windows of plasticity for therapeutic intervention.
However, the translation of these promising findings into clinical practice is fraught with complexity. The effects of oxytocin are profoundly context-dependent, modulated by factors such as the individual’s environment, sex, genetic and epigenetic background, and the nature of their early adverse experiences. It is increasingly clear that oxytocin is not a simple anxiolytic or social lubricant, but rather a modulator of social salience that amplifies the processing of the surrounding environment. This underscores the critical importance of administering oxytocin within a safe, supportive, and structured therapeutic context, ideally as an adjunct to evidence-based psychosocial interventions.
Substantial challenges remain. Key unanswered questions regarding optimal dosing, long-term safety, sex-specific responses, and reliable delivery methods necessitate further rigorous investigation. The future of oxytocin-based interventions lies in a precision medicine approach that moves beyond a one-size-fits-all model. This will require the identification of biomarkers predictive of treatment response and the development of novel therapeutic strategies that combine oxytocin with other neuromodulatory techniques to enhance neural target engagement.
In conclusion, while oxytocin is not a panacea, it represents a profoundly promising biological tool for fostering resilience. By continuing to elucidate its complex mechanisms and thoughtfully integrating it into a holistic treatment framework, oxytocin-based strategies offer a compelling pathway toward alleviating the enduring burden of ELS and improving mental health outcomes across the lifespan.
Shu Wang is a senior collaborator of the Global Burden of Disease Collaborator Network, which is supported by the Institute for Health Metrics and Evaluation, the University of Washington School of Medicine.
| 1. | Nakama N, Usui N, Doi M, Shimada S. Early life stress impairs brain and mental development during childhood increasing the risk of developing psychiatric disorders. Prog Neuropsychopharmacol Biol Psychiatry. 2023;126:110783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 29] [Article Influence: 9.7] [Reference Citation Analysis (1)] |
| 2. | Bi H, Jin J, Sun M, Chen M, Li X, Wang Y. Epigenetic changes caused by early life stress in the pathogenesis of depression. Eur J Pharmacol. 2025;999:177671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 3. | Zhang Y, Wang S, Hei M. Maternal separation as early-life stress: Mechanisms of neuropsychiatric disorders and inspiration for neonatal care. Brain Res Bull. 2024;217:111058. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 4. | LeMoult J, Humphreys KL, Tracy A, Hoffmeister JA, Ip E, Gotlib IH. Meta-analysis: Exposure to Early Life Stress and Risk for Depression in Childhood and Adolescence. J Am Acad Child Adolesc Psychiatry. 2020;59:842-855. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 521] [Cited by in RCA: 414] [Article Influence: 69.0] [Reference Citation Analysis (0)] |
| 5. | Lin L, Wang HH, Lu C, Chen W, Guo VY. Adverse Childhood Experiences and Subsequent Chronic Diseases Among Middle-aged or Older Adults in China and Associations With Demographic and Socioeconomic Characteristics. JAMA Netw Open. 2021;4:e2130143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 250] [Cited by in RCA: 200] [Article Influence: 40.0] [Reference Citation Analysis (0)] |
| 6. | Loewy RL, Corey S, Amirfathi F, Dabit S, Fulford D, Pearson R, Hua JPY, Schlosser D, Stuart BK, Mathalon DH, Vinogradov S. Childhood trauma and clinical high risk for psychosis. Schizophr Res. 2019;205:10-14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 95] [Article Influence: 13.6] [Reference Citation Analysis (1)] |
| 7. | Madigan S, Thiemann R, Deneault AA, Fearon RMP, Racine N, Park J, Lunney CA, Dimitropoulos G, Jenkins S, Williamson T, Neville RD. Prevalence of Adverse Childhood Experiences in Child Population Samples: A Systematic Review and Meta-Analysis. JAMA Pediatr. 2025;179:19-33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 55] [Article Influence: 55.0] [Reference Citation Analysis (1)] |
| 8. | Morgart K, Harrison JN, Hoon AH Jr, Wilms Floet AM. Adverse childhood experiences and developmental disabilities: risks, resiliency, and policy. Dev Med Child Neurol. 2021;63:1149-1154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 9. | Petruccelli K, Davis J, Berman T. Adverse childhood experiences and associated health outcomes: A systematic review and meta-analysis. Child Abuse Negl. 2019;97:104127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 893] [Cited by in RCA: 640] [Article Influence: 91.4] [Reference Citation Analysis (0)] |
| 10. | Hustedde C. Adverse Childhood Experiences. Prim Care. 2021;48:493-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 11. | Senaratne DNS, Thakkar B, Smith BH, Hales TG, Marryat L, Colvin LA. The impact of adverse childhood experiences on multimorbidity: a systematic review and meta-analysis. BMC Med. 2024;22:315. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 46] [Article Influence: 23.0] [Reference Citation Analysis (1)] |
| 12. | Abate BB, Sendekie AK, Merchaw A, Abebe GK, Azmeraw M, Alamaw AW, Zemariam AB, Kitaw TA, Kassaw A, Wodaynew T, Kassie AM, Yilak G, Kassa MA. Adverse Childhood Experiences Are Associated with Mental Health Problems Later in Life: An Umbrella Review of Systematic Review and Meta-Analysis. Neuropsychobiology. 2025;84:48-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 30] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 13. | Mousikou M, Kyriakou A, Skordis N. Stress and Growth in Children and Adolescents. Horm Res Paediatr. 2023;96:25-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 46] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 14. | Smith KE, Pollak SD. Early life stress and development: potential mechanisms for adverse outcomes. J Neurodev Disord. 2020;12:34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 406] [Cited by in RCA: 318] [Article Influence: 53.0] [Reference Citation Analysis (1)] |
| 15. | Featherstone RE, Gifford RL, Crown LM, Amirfathi F, Alaniz JP, Yi J, Tran A, Adomian D, Schwenk A, Melnychenko O, Duval C, Parekh K, Lee DJ, Siegel SJ. Early life social instability stress causes lasting cognitive decrement and elevated hippocampal stress-related gene expression. Exp Neurol. 2022;354:114099. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 16. | Nicolaides NC, Kanaka-Gantenbein C, Pervanidou P. Developmental Neuroendocrinology of Early-Life Stress: Impact on Child Development and Behavior. Curr Neuropharmacol. 2024;22:461-474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 17. | Lopatina OL, Panina YA, Malinovskaya NA, Salmina AB. Early life stress and brain plasticity: from molecular alterations to aberrant memory and behavior. Rev Neurosci. 2021;32:131-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 18. | Lesuis SL, Lucassen PJ, Krugers HJ. Early life stress amplifies fear responses and hippocampal synaptic potentiation in the APPswe/PS1dE9 Alzheimer mouse model. Neuroscience. 2021;454:151-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 19. | González-Pardo H, Arias JL, Gómez-Lázaro E, López Taboada I, Conejo NM. Sex-Specific Effects of Early Life Stress on Brain Mitochondrial Function, Monoamine Levels and Neuroinflammation. Brain Sci. 2020;10:447. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 20. | Catale C, Carola V, Viscomi MT. Early life stress-induced neuroinflammation and neurological disorders: a novel perspective for research. Neural Regen Res. 2022;17:1971-1972. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 21. | McEwen BS, Nasca C, Gray JD. Stress Effects on Neuronal Structure: Hippocampus, Amygdala, and Prefrontal Cortex. Neuropsychopharmacology. 2016;41:3-23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1326] [Cited by in RCA: 1108] [Article Influence: 110.8] [Reference Citation Analysis (1)] |
| 22. | Johnson FK, Delpech JC, Thompson GJ, Wei L, Hao J, Herman P, Hyder F, Kaffman A. Amygdala hyper-connectivity in a mouse model of unpredictable early life stress. Transl Psychiatry. 2018;8:49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 99] [Article Influence: 12.4] [Reference Citation Analysis (1)] |
| 23. | Bhutta ZA, Bhavnani S, Betancourt TS, Tomlinson M, Patel V. Adverse childhood experiences and lifelong health. Nat Med. 2023;29:1639-1648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 99] [Article Influence: 33.0] [Reference Citation Analysis (1)] |
| 24. | Makris G, Eleftheriades A, Pervanidou P. Early Life Stress, Hormones, and Neurodevelopmental Disorders. Horm Res Paediatr. 2023;96:17-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 25. | Matsushita H, Latt HM, Koga Y, Nishiki T, Matsui H. Oxytocin and Stress: Neural Mechanisms, Stress-Related Disorders, and Therapeutic Approaches. Neuroscience. 2019;417:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 68] [Article Influence: 9.7] [Reference Citation Analysis (1)] |
| 26. | Walter MH, Abele H, Plappert CF. The Role of Oxytocin and the Effect of Stress During Childbirth: Neurobiological Basics and Implications for Mother and Child. Front Endocrinol (Lausanne). 2021;12:742236. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 92] [Article Influence: 18.4] [Reference Citation Analysis (1)] |
| 27. | Marsh N, Marsh AA, Lee MR, Hurlemann R. Oxytocin and the Neurobiology of Prosocial Behavior. Neuroscientist. 2021;27:604-619. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 83] [Article Influence: 16.6] [Reference Citation Analysis (1)] |
| 28. | Carter CS, Kenkel WM, MacLean EL, Wilson SR, Perkeybile AM, Yee JR, Ferris CF, Nazarloo HP, Porges SW, Davis JM, Connelly JJ, Kingsbury MA. Is Oxytocin "Nature's Medicine"? Pharmacol Rev. 2020;72:829-861. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 354] [Cited by in RCA: 288] [Article Influence: 48.0] [Reference Citation Analysis (5)] |
| 29. | Onaka T, Takayanagi Y. Role of oxytocin in the control of stress and food intake. J Neuroendocrinol. 2019;31:e12700. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 64] [Article Influence: 9.1] [Reference Citation Analysis (1)] |
| 30. | Kingsbury MA, Bilbo SD. The inflammatory event of birth: How oxytocin signaling may guide the development of the brain and gastrointestinal system. Front Neuroendocrinol. 2019;55:100794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 50] [Article Influence: 7.1] [Reference Citation Analysis (1)] |
| 31. | Froemke RC, Young LJ. Oxytocin, Neural Plasticity, and Social Behavior. Annu Rev Neurosci. 2021;44:359-381. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 411] [Cited by in RCA: 326] [Article Influence: 65.2] [Reference Citation Analysis (0)] |
| 32. | Rault JL, Carter CS, Garner JP, Marchant JN, Richert BT, Lay DC Jr. Repeated intranasal oxytocin administration in early life dysregulates the HPA axis and alters social behavior. Physiol Behav. 2013;112-113:40-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 80] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 33. | Ochedalski T, Subburaju S, Wynn PC, Aguilera G. Interaction between oestrogen and oxytocin on hypothalamic-pituitary-adrenal axis activity. J Neuroendocrinol. 2007;19:189-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 103] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 34. | Park HY, Lee S, Koo SJ, Li Z, Seo E, Lee E, An SK. Epigenetic modulation of social cognition: exploring the impact of methylation in brain-derived neurotrophic factor and oxytocin receptor genes across sex. Sci Rep. 2025;15:3412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 35. | Unternaehrer E, Luers P, Mill J, Dempster E, Meyer AH, Staehli S, Lieb R, Hellhammer DH, Meinlschmidt G. Dynamic changes in DNA methylation of stress-associated genes (OXTR, BDNF ) after acute psychosocial stress. Transl Psychiatry. 2012;2:e150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 220] [Cited by in RCA: 182] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 36. | Keech B, Crowe S, Hocking DR. Intranasal oxytocin, social cognition and neurodevelopmental disorders: A meta-analysis. Psychoneuroendocrinology. 2018;87:9-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 94] [Article Influence: 11.8] [Reference Citation Analysis (2)] |
| 37. | Evans SL, Dal Monte O, Noble P, Averbeck BB. Intranasal oxytocin effects on social cognition: a critique. Brain Res. 2014;1580:69-77. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 70] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 38. | Browne J, Harvey PD, Buchanan RW, Kelly DL, Strauss GP, Gold JM, Holden JL, Granholm E. A Longitudinal Examination of Real-World Sedentary Behavior in Adults with Schizophrenia-Spectrum Disorders in a Clinical Trial of Combined Oxytocin and Cognitive Behavioral Social Skills Training. Behav Sci (Basel). 2022;12:60. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 39. | Strauss GP, Granholm E, Holden JL, Ruiz I, Gold JM, Kelly DL, Buchanan RW. The effects of combined oxytocin and cognitive behavioral social skills training on social cognition in schizophrenia. Psychol Med. 2019;49:1731-1739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 40. | Buchanan RW, Kelly DL, Strauss GP, Gold JM, Weiner E, Zaranski J, Chen S, Blatt F, Holden J, Granholm E. Combined Oxytocin and Cognitive Behavioral Social Skills Training for Social Function in People With Schizophrenia. J Clin Psychopharmacol. 2021;41:236-243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 41. | Zhang Y, Jiang M, Wang S, Xiang X, He W, Du J, Hei M. Effect of family integrated care on stress in mothers of preterm infants: A multicenter cluster randomized controlled trial. J Affect Disord. 2024;350:304-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 42. | Zhang Y, Jiang M, Xiang X, He W, Du J, Hei M. Effect of Family Integrated Care on maternal stress in preterm infants in the Neonatal Intensive Care Unit: A bi-center experience. Asia Pac Psychiatry. 2023;15:e12521. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 43. | Fan Y, Herrera-Melendez AL, Pestke K, Feeser M, Aust S, Otte C, Pruessner JC, Böker H, Bajbouj M, Grimm S. Early life stress modulates amygdala-prefrontal functional connectivity: implications for oxytocin effects. Hum Brain Mapp. 2014;35:5328-5339. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 97] [Article Influence: 8.1] [Reference Citation Analysis (1)] |
| 44. | Yuan R, Nechvatal JM, Buckmaster CL, Ayash S, Parker KJ, Schatzberg AF, Lyons DM, Menon V. Long-term effects of intermittent early life stress on primate prefrontal-subcortical functional connectivity. Neuropsychopharmacology. 2021;46:1348-1356. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 45. | Zhu J, Zhong Z, Shi L, Huang L, Lin C, He Y, Xia X, Zhang T, Ding W, Yang Y. Gut microbiota mediate early life stress-induced social dysfunction and anxiety-like behaviors by impairing amino acid transport at the gut. Gut Microbes. 2024;16:2401939. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 46. | Li D, Li YC, Zhu ZY, Zhang FC, Zhao QY, Jiang JH, Shen B, Tang Y, Xu GY. The paraventricular thalamus mediates visceral pain and anxiety-like behaviors via two distinct pathways. Neuron. 2025;113:2310-2324.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 20] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 47. | Grant MM, White D, Hadley J, Hutcheson N, Shelton R, Sreenivasan K, Deshpande G. Early life trauma and directional brain connectivity within major depression. Hum Brain Mapp. 2014;35:4815-4826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 55] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 48. | Purcell JB, Goodman AM, Harnett NG, Davis ES, Wheelock MD, Mrug S, Elliott MN, Emery ST, Schuster MA, Knight DC. Stress-elicited neural activity in young adults varies with childhood sexual abuse. Cortex. 2021;137:108-123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 49. | Hakamata Y, Mizukami S, Izawa S, Moriguchi Y, Hori H, Matsumoto N, Hanakawa T, Inoue Y, Tagaya H. Childhood trauma affects autobiographical memory deficits through basal cortisol and prefrontal-extrastriate functional connectivity. Psychoneuroendocrinology. 2021;127:105172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 50. | van Bodegom M, Homberg JR, Henckens MJAG. Modulation of the Hypothalamic-Pituitary-Adrenal Axis by Early Life Stress Exposure. Front Cell Neurosci. 2017;11:87. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 408] [Cited by in RCA: 373] [Article Influence: 41.4] [Reference Citation Analysis (5)] |
| 51. | Jiang L, Xue L, Juruena MF. The impact of early life stress on the hypothalamic-pituitary-adrenal axis in unipolar major depression: A systematic review. Psychoneuroendocrinology. 2025;181:107607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 52. | Di Iorio CR, Carey CE, Michalski LJ, Corral-Frias NS, Conley ED, Hariri AR, Bogdan R. Hypothalamic-pituitary-adrenal axis genetic variation and early stress moderates amygdala function. Psychoneuroendocrinology. 2017;80:170-178. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 53] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 53. | Shea A, Walsh C, Macmillan H, Steiner M. Child maltreatment and HPA axis dysregulation: relationship to major depressive disorder and post traumatic stress disorder in females. Psychoneuroendocrinology. 2005;30:162-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 291] [Cited by in RCA: 240] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 54. | Ceruso A, Martínez-Cengotitabengoa M, Peters-Corbett A, Diaz-Gutierrez MJ, Martínez-Cengotitabengoa M. Alterations of the HPA Axis Observed in Patients with Major Depressive Disorder and Their Relation to Early Life Stress: A Systematic Review. Neuropsychobiology. 2020;79:417-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 44] [Article Influence: 7.3] [Reference Citation Analysis (1)] |
| 55. | Hartmann J, Bajaj T, Klengel C, Chatzinakos C, Ebert T, Dedic N, McCullough KM, Lardenoije R, Joëls M, Meijer OC, McCann KE, Dudek SM, Sarabdjitsingh RA, Daskalakis NP, Klengel T, Gassen NC, Schmidt MV, Ressler KJ. Mineralocorticoid receptors dampen glucocorticoid receptor sensitivity to stress via regulation of FKBP5. Cell Rep. 2021;35:109185. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 79] [Article Influence: 15.8] [Reference Citation Analysis (1)] |
| 56. | Pariante CM, Lightman SL. The HPA axis in major depression: classical theories and new developments. Trends Neurosci. 2008;31:464-468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1622] [Cited by in RCA: 1435] [Article Influence: 79.7] [Reference Citation Analysis (6)] |
| 57. | Duman RS, Sanacora G, Krystal JH. Altered Connectivity in Depression: GABA and Glutamate Neurotransmitter Deficits and Reversal by Novel Treatments. Neuron. 2019;102:75-90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 925] [Cited by in RCA: 757] [Article Influence: 108.1] [Reference Citation Analysis (5)] |
| 58. | McEwen BS. Glucocorticoids, depression, and mood disorders: structural remodeling in the brain. Metabolism. 2005;54:20-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 563] [Cited by in RCA: 496] [Article Influence: 23.6] [Reference Citation Analysis (4)] |
| 59. | Sanchez CM, Titus DJ, Wilson NM, Freund JE, Atkins CM. Early Life Stress Exacerbates Outcome after Traumatic Brain Injury. J Neurotrauma. 2021;38:555-565. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 60. | Peña CJ, Kronman HG, Walker DM, Cates HM, Bagot RC, Purushothaman I, Issler O, Loh YE, Leong T, Kiraly DD, Goodman E, Neve RL, Shen L, Nestler EJ. Early life stress confers lifelong stress susceptibility in mice via ventral tegmental area OTX2. Science. 2017;356:1185-1188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 378] [Cited by in RCA: 310] [Article Influence: 34.4] [Reference Citation Analysis (1)] |
| 61. | Kocamaz D, Franzke C, Gröger N, Braun K, Bock J. Early Life Stress-Induced Epigenetic Programming of Hippocampal NPY-Y2 Receptor Gene Expression Changes in Response to Adult Stress. Front Cell Neurosci. 2022;16:936979. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 62. | Watkeys OJ, Kremerskothen K, Quidé Y, Fullerton JM, Green MJ. Glucocorticoid receptor gene (NR3C1) DNA methylation in association with trauma, psychopathology, transcript expression, or genotypic variation: A systematic review. Neurosci Biobehav Rev. 2018;95:85-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 75] [Article Influence: 9.4] [Reference Citation Analysis (1)] |
| 63. | Berretta E, Guida E, Forni D, Provenzi L. Glucocorticoid receptor gene (NR3C1) methylation during the first thousand days: Environmental exposures and developmental outcomes. Neurosci Biobehav Rev. 2021;125:493-502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 34] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 64. | Chubar V, Vaessen T, Noortgate WVD, Lutin E, Bosmans G, Bekaert B, Van Leeuwen K, Calders F, Weyn S, Bijttebier P, Goossens L, Claes S. Mild daily stress, in interaction with NR3C1 DNA methylation levels, is linked to alterations in the HPA axis and ANS response to acute stress in early adolescents. Psychoneuroendocrinology. 2023;150:106045. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 65. | Bakusic J, Ghosh M, Polli A, Bekaert B, Schaufeli W, Claes S, Godderis L. Role of NR3C1 and SLC6A4 methylation in the HPA axis regulation in burnout. J Affect Disord. 2021;295:505-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 66. | Ellis BJ, Horn AJ, Carter CS, van IJzendoorn MH, Bakermans-Kranenburg MJ. Developmental programming of oxytocin through variation in early-life stress: Four meta-analyses and a theoretical reinterpretation. Clin Psychol Rev. 2021;86:101985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 68] [Article Influence: 13.6] [Reference Citation Analysis (1)] |
| 67. | Danoff JS, Wroblewski KL, Graves AJ, Quinn GC, Perkeybile AM, Kenkel WM, Lillard TS, Parikh HI, Golino HF, Gregory SG, Carter CS, Bales KL, Connelly JJ. Genetic, epigenetic, and environmental factors controlling oxytocin receptor gene expression. Clin Epigenetics. 2021;13:23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 47] [Article Influence: 9.4] [Reference Citation Analysis (1)] |
| 68. | Myers AJ, Williams L, Gatt JM, McAuley-Clark EZ, Dobson-Stone C, Schofield PR, Nemeroff CB. Variation in the oxytocin receptor gene is associated with increased risk for anxiety, stress and depression in individuals with a history of exposure to early life stress. J Psychiatr Res. 2014;59:93-100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 73] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 69. | Zheng S, Masuda T, Matsunaga M, Noguchi Y, Ohtsubo Y, Yamasue H, Ishii K. Oxytocin Receptor Gene (OXTR) and Childhood Adversity Influence Trust. Psychoneuroendocrinology. 2020;121:104840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 70. | Andersen SL. Neuroinflammation, Early-Life Adversity, and Brain Development. Harv Rev Psychiatry. 2022;30:24-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 52] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 71. | Balouek JA, Mclain CA, Minerva AR, Rashford RL, Bennett SN, Rogers FD, Peña CJ. Reactivation of Early-Life Stress-Sensitive Neuronal Ensembles Contributes to Lifelong Stress Hypersensitivity. J Neurosci. 2023;43:5996-6009. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 27] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 72. | Ng H, Ohmura N, Miyazawa E, Yoshihara C, Okuma L, Kuroda KO. Effects of oxytocin ablation on pup rescue, nursing behaviors and response to pup separation in early-to-mid postpartum mice. J Neuroendocrinol. 2023;35:e13247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 73. | Amini-Khoei H, Mohammadi-Asl A, Amiri S, Hosseini MJ, Momeny M, Hassanipour M, Rastegar M, Haj-Mirzaian A, Mirzaian AH, Sanjarimoghaddam H, Mehr SE, Dehpour AR. Oxytocin mitigated the depressive-like behaviors of maternal separation stress through modulating mitochondrial function and neuroinflammation. Prog Neuropsychopharmacol Biol Psychiatry. 2017;76:169-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 128] [Article Influence: 14.2] [Reference Citation Analysis (1)] |
| 74. | Zhang FR, Liu J, Wen J, Zhang ZY, Li Y, Song E, Hu L, Chen ZF. Distinct oxytocin signaling pathways synergistically mediate rescue-like behavior in mice. Proc Natl Acad Sci U S A. 2025;122:e2423374122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 75. | Knobloch HS, Charlet A, Hoffmann LC, Eliava M, Khrulev S, Cetin AH, Osten P, Schwarz MK, Seeburg PH, Stoop R, Grinevich V. Evoked axonal oxytocin release in the central amygdala attenuates fear response. Neuron. 2012;73:553-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 956] [Cited by in RCA: 821] [Article Influence: 58.6] [Reference Citation Analysis (1)] |
| 76. | Mitchell IJ, Gillespie SM, Abu-Akel A. Similar effects of intranasal oxytocin administration and acute alcohol consumption on socio-cognitions, emotions and behaviour: Implications for the mechanisms of action. Neurosci Biobehav Rev. 2015;55:98-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 77. | Shi DD, Zhang YD, Ren YY, Peng SY, Yuan TF, Wang Z. Predictable maternal separation confers adult stress resilience via the medial prefrontal cortex oxytocin signaling pathway in rats. Mol Psychiatry. 2021;26:7296-7307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 78. | Stamatakis A, Kalpachidou T, Raftogianni A, Zografou E, Tzanou A, Pondiki S, Stylianopoulou F. Rat dams exposed repeatedly to a daily brief separation from the pups exhibit increased maternal behavior, decreased anxiety and altered levels of receptors for estrogens (ERα, ERβ), oxytocin and serotonin (5-HT1A) in their brain. Psychoneuroendocrinology. 2015;52:212-228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 46] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 79. | Baracz SJ, Everett NA, Robinson KJ, Campbell GR, Cornish JL. Maternal separation changes maternal care, anxiety-like behaviour and expression of paraventricular oxytocin and corticotrophin-releasing factor immunoreactivity in lactating rats. J Neuroendocrinol. 2020;32:e12861. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 80. | Rich ME, deCárdenas EJ, Lee HJ, Caldwell HK. Impairments in the initiation of maternal behavior in oxytocin receptor knockout mice. PLoS One. 2014;9:e98839. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 80] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 81. | Silva RC, Maffioletti E, Gennarelli M, Baune BT, Minelli A. Biological correlates of early life stressful events in major depressive disorder. Psychoneuroendocrinology. 2021;125:105103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (1)] |
| 82. | Weisman O, Zagoory-Sharon O, Feldman R. Oxytocin administration alters HPA reactivity in the context of parent-infant interaction. Eur Neuropsychopharmacol. 2013;23:1724-1731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 52] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 83. | Tost H, Kolachana B, Hakimi S, Lemaitre H, Verchinski BA, Mattay VS, Weinberger DR, Meyer-Lindenberg A. A common allele in the oxytocin receptor gene (OXTR) impacts prosocial temperament and human hypothalamic-limbic structure and function. Proc Natl Acad Sci U S A. 2010;107:13936-13941. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 469] [Cited by in RCA: 404] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 84. | Ménard S, Gelez H, Coria-Avila GA, Pfaus JG. Sexual experience increases oxytocin, but not vasopressin, receptor densities in the medial preoptic area, ventromedial hypothalamus, and central amygdala of male rats. Psychoneuroendocrinology. 2022;146:105900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 85. | Talpo F, Spaiardi P, Castagno AN, Maniezzi C, Raffin F, Terribile G, Sancini G, Pisani A, Biella GR. Neuromodulatory functions exerted by oxytocin on different populations of hippocampal neurons in rodents. Front Cell Neurosci. 2023;17:1082010. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 86. | Lin YT, Hsu KS. Oxytocin receptor signaling in the hippocampus: Role in regulating neuronal excitability, network oscillatory activity, synaptic plasticity and social memory. Prog Neurobiol. 2018;171:1-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 68] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 87. | Bukatova S, Reichova A, Bacova Z, Bakos J. Neonatal oxytocin treatment alters levels of precursor and mature BDNF forms and modifies the expression of neuronal markers in the male rat hippocampus. Neuropeptides. 2023;102:102384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 88. | Liu R, Sun D, Xing X, Chen Q, Lu B, Meng B, Yuan H, Mo L, Sheng L, Zheng J, Wang Q, Chen J, Chen X. Intranasal oxytocin alleviates comorbid depressive symptoms in neuropathic pain via elevating hippocampal BDNF production in both female and male mice. Neuropharmacology. 2024;242:109769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 9.5] [Reference Citation Analysis (1)] |
| 89. | Baracz SJ, Robinson KJ, Wright AL, Turner AJ, McGregor IS, Cornish JL, Everett NA. Oxytocin as an adolescent treatment for methamphetamine addiction after early life stress in male and female rats. Neuropsychopharmacology. 2022;47:1561-1573. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 90. | Lauby SC, Agarwal I, Lapp HE, Salazar M, Semyrenko S, Chauhan D, Margolis AE, Champagne FA. Interplay between prenatal bisphenol exposure, postnatal maternal care, and offspring sex in predicting DNA methylation relevant to anxiety-like behavior in rats. Horm Behav. 2025;172:105745. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 91. | Harony-Nicolas H, Mamrut S, Brodsky L, Shahar-Gold H, Barki-Harrington L, Wagner S. Brain region-specific methylation in the promoter of the murine oxytocin receptor gene is involved in its expression regulation. Psychoneuroendocrinology. 2014;39:121-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 48] [Article Influence: 4.0] [Reference Citation Analysis (2)] |
| 92. | Colleluori G, Galli C, Severi I, Perugini J, Giordano A. Early Life Stress, Brain Development, and Obesity Risk: Is Oxytocin the Missing Link? Cells. 2022;11:623. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 93. | Yao S, Kendrick KM. How does oxytocin modulate human behavior? Mol Psychiatry. 2025;30:1639-1651. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 26] [Article Influence: 26.0] [Reference Citation Analysis (1)] |
| 94. | Lapp HE, Bartlett AA, Zup SL, Hunter RG, Moore CL. Early experience alters developmental trajectory of central oxytocin systems involved in hypothalamic-pituitary-adrenal axis regulation in Long-Evans rats. Horm Behav. 2020;126:104822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 95. | Demarchi L, Sanson A, Bosch OJ. Brief versus long maternal separation in lactating rats: Consequences on maternal behavior, emotionality, and brain oxytocin receptor binding. J Neuroendocrinol. 2023;35:e13252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 96. | Morais LH, Golubeva AV, Casey S, Scott KA, Ramos Costa AP, Moloney GM, Dinan TG, Cryan JF. Early-life oxytocin attenuates the social deficits induced by caesarean-section delivery in the mouse. Neuropsychopharmacology. 2021;46:1958-1968. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 97. | Dayi A, Kiray M, Sisman A, Ozbal S, Baykara B, Aksu I, Uysal N. Dose dependent effects of oxytocin on cognitive defects and anxiety disorders in adult rats following acute infantile maternal deprivation stress. Biotech Histochem. 2019;94:469-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 98. | Zoratto F, Sbriccoli M, Martinelli A, Glennon JC, Macrì S, Laviola G. Intranasal oxytocin administration promotes emotional contagion and reduces aggression in a mouse model of callousness. Neuropharmacology. 2018;143:250-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 99. | Lee SY, Park SH, Chung C, Kim JJ, Choi SY, Han JS. Oxytocin Protects Hippocampal Memory and Plasticity from Uncontrollable Stress. Sci Rep. 2015;5:18540. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 86] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 100. | Takahashi J, Yamada D, Ueta Y, Iwai T, Koga E, Tanabe M, Oka JI, Saitoh A. Oxytocin reverses Aβ-induced impairment of hippocampal synaptic plasticity in mice. Biochem Biophys Res Commun. 2020;528:174-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 101. | Liu Y, Shan L, Liu T, Li J, Chen Y, Sun C, Yang C, Bian X, Niu Y, Zhang C, Xi J, Rao Y. Molecular and cellular mechanisms of the first social relationship: A conserved role of 5-HT from mice to monkeys, upstream of oxytocin. Neuron. 2023;111:1468-1485.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 31] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 102. | Ramos L, Hicks C, Caminer A, Couto K, Narlawar R, Kassiou M, McGregor IS. MDMA ('Ecstasy'), oxytocin and vasopressin modulate social preference in rats: A role for handling and oxytocin receptors. Pharmacol Biochem Behav. 2016;150-151:115-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 103. | Djerdjaj A, Rieger NS, Brady BH, Carey BN, Ng AJ, Christianson JP. Social affective behaviors among female rats involve the basolateral amygdala and insular cortex. PLoS One. 2023;18:e0281794. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 104. | Rea JJ, Liu CM, Hayes AMR, Bashaw AG, Schwartz GM, Ohan R, Décarie-Spain L, Kao AE, Klug ME, Phung KJ, Waldow AI, Wood RI, Kanoski SE. Hippocampus Oxytocin Signaling Promotes Prosocial Eating in Rats. Biol Psychiatry. 2025;97:540-549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 105. | Logan CN, Rojas G, Wilkinson CS, Polo Escorcia AK, Reichel CM, Peris J, Knackstedt LA. Systemic oxytocin increases glutamate efflux in the nucleus accumbens core of cocaine-experienced male and female rats but only increases dopamine efflux in males. Behav Brain Res. 2022;417:113590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 106. | Georgiou P, Zanos P, Garcia-Carmona JA, Hourani S, Kitchen I, Kieffer BL, Laorden ML, Bailey A. The oxytocin analogue carbetocin prevents priming-induced reinstatement of morphine-seeking: Involvement of dopaminergic, noradrenergic and MOPr systems. Eur Neuropsychopharmacol. 2015;25:2459-2464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 41] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 107. | Toepfer P, Heim C, Entringer S, Binder E, Wadhwa P, Buss C. Oxytocin pathways in the intergenerational transmission of maternal early life stress. Neurosci Biobehav Rev. 2017;73:293-308. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 70] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 108. | Wang M, Hou S, Tian C, Fu Z, Jie J. Effects of Intranasal Oxytocin Across Various Depressive Disorders. Psychiatry Investig. 2025;22:964-978. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 109. | Riem MME, Loheide-Niesmann L, Beijers R, Tendolkar I, Mulders PC. Boosting oxytocin in postpartum depression: Intranasal oxytocin enhances maternal positive affect and regard for the infant. Psychoneuroendocrinology. 2025;179:107530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 110. | Koch SB, van Zuiden M, Nawijn L, Frijling JL, Veltman DJ, Olff M. Intranasal oxytocin as strategy for medication-enhanced psychotherapy of PTSD: salience processing and fear inhibition processes. Psychoneuroendocrinology. 2014;40:242-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 92] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 111. | Chen X, Hackett PD, DeMarco AC, Feng C, Stair S, Haroon E, Ditzen B, Pagnoni G, Rilling JK. Effects of oxytocin and vasopressin on the neural response to unreciprocated cooperation within brain regions involved in stress and anxiety in men and women. Brain Imaging Behav. 2016;10:581-593. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 64] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 112. | Begemann MJH, Schutte MJL, van Dellen E, Abramovic L, Boks MP, van Haren NEM, Mandl RCW, Vinkers CH, Bohlken MM, Sommer IEC. Childhood trauma is associated with reduced frontal gray matter volume: a large transdiagnostic structural MRI study. Psychol Med. 2023;53:741-749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 44] [Article Influence: 14.7] [Reference Citation Analysis (1)] |
| 113. | Harb F, Liuzzi MT, Huggins AA, Webb EK, Fitzgerald JM, Krukowski JL, deRoon-Cassini TA, Larson CL. Childhood Maltreatment and Amygdala-Mediated Anxiety and Posttraumatic Stress Following Adult Trauma. Biol Psychiatry Glob Open Sci. 2024;4:100312. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 114. | Quintana DS, Westlye LT, Alnæs D, Kaufmann T, Mahmoud RA, Smerud KT, Djupesland PG, Andreassen OA. Low-dose intranasal oxytocin delivered with Breath Powered device modulates pupil diameter and amygdala activity: a randomized controlled pupillometry and fMRI study. Neuropsychopharmacology. 2019;44:306-313. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 115. | Frijling JL, van Zuiden M, Koch SB, Nawijn L, Veltman DJ, Olff M. Intranasal Oxytocin Affects Amygdala Functional Connectivity after Trauma Script-Driven Imagery in Distressed Recently Trauma-Exposed Individuals. Neuropsychopharmacology. 2016;41:1286-1296. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 52] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 116. | Grace SA, Labuschagne I, Castle DJ, Rossell SL. Intranasal oxytocin alters amygdala-temporal resting-state functional connectivity in body dysmorphic disorder: A double-blind placebo-controlled randomized trial. Psychoneuroendocrinology. 2019;107:179-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 117. | Koch SB, van Zuiden M, Nawijn L, Frijling JL, Veltman DJ, Olff M. Intranasal Oxytocin Normalizes Amygdala Functional Connectivity in Posttraumatic Stress Disorder. Neuropsychopharmacology. 2016;41:2041-2051. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 121] [Cited by in RCA: 111] [Article Influence: 11.1] [Reference Citation Analysis (1)] |
| 118. | Dodhia S, Hosanagar A, Fitzgerald DA, Labuschagne I, Wood AG, Nathan PJ, Phan KL. Modulation of resting-state amygdala-frontal functional connectivity by oxytocin in generalized social anxiety disorder. Neuropsychopharmacology. 2014;39:2061-2069. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 182] [Cited by in RCA: 159] [Article Influence: 13.3] [Reference Citation Analysis (1)] |
| 119. | Gorka SM, Fitzgerald DA, Labuschagne I, Hosanagar A, Wood AG, Nathan PJ, Phan KL. Oxytocin modulation of amygdala functional connectivity to fearful faces in generalized social anxiety disorder. Neuropsychopharmacology. 2015;40:278-286. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 108] [Article Influence: 9.8] [Reference Citation Analysis (1)] |
| 120. | Jurek B, Neumann ID. The Oxytocin Receptor: From Intracellular Signaling to Behavior. Physiol Rev. 2018;98:1805-1908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 887] [Cited by in RCA: 721] [Article Influence: 90.1] [Reference Citation Analysis (1)] |
| 121. | Marazziti D, Diep PT, Carter S, Carbone MG. Oxytocin: An Old Hormone, a Novel Psychotropic Drug and its Possible Use in Treating Psychiatric Disorders. Curr Med Chem. 2022;29:5615-5687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 122. | Moerkerke M, Daniels N, Tibermont L, Tang T, Evenepoel M, Van der Donck S, Debbaut E, Prinsen J, Chubar V, Claes S, Vanaudenaerde B, Willems L, Steyaert J, Boets B, Alaerts K. Chronic oxytocin administration stimulates the oxytocinergic system in children with autism. Nat Commun. 2024;15:58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 30] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 123. | Giel KE, Schag K, Leehr EJ, Mack I, Schuster LS, Wiegand A, Zipfel S, Hallschmid M, Nieratschker V. OXTR DNA methylation differentiates men on the obesity spectrum with and without binge eating disorder. Clin Epigenetics. 2022;14:108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 124. | Kenkel WM, Perkeybile AM, Yee JR, Pournajafi-Nazarloo H, Lillard TS, Ferguson EF, Wroblewski KL, Ferris CF, Carter CS, Connelly JJ. Behavioral and epigenetic consequences of oxytocin treatment at birth. Sci Adv. 2019;5:eaav2244. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 53] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 125. | Boyle A, Johnson A, Ellenbogen M. Intranasal oxytocin alters attention to emotional facial expressions, particularly for males and those with depressive symptoms. Psychoneuroendocrinology. 2022;142:105796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 126. | Jones C, Barrera I, Brothers S, Ring R, Wahlestedt C. Oxytocin and social functioning. Dialogues Clin Neurosci. 2017;19:193-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 85] [Article Influence: 9.4] [Reference Citation Analysis (1)] |
| 127. | di Giacomo E, Andreini E, Santambrogio J, Arcara A, Clerici M. The interplay between borderline personality disorder and oxytocin: a systematic narrative review on possible contribution and treatment options. Front Psychiatry. 2024;15:1439615. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 128. | Ricchiuti G, Tuerlinckx E, Taillieu A, Prinsen J, Steyaert J, Boets B, Alaerts K. Toward effective oxytocin interventions in autism: Overcoming challenges and harnessing opportunities. J Psychopharmacol. 2025;39:179-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 129. | Anagnostou E, Soorya L, Brian J, Dupuis A, Mankad D, Smile S, Jacob S. Intranasal oxytocin in the treatment of autism spectrum disorders: a review of literature and early safety and efficacy data in youth. Brain Res. 2014;1580:188-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 113] [Article Influence: 9.4] [Reference Citation Analysis (1)] |
| 130. | Horta M, Pehlivanoglu D, Ebner NC. The Role of Intranasal Oxytocin on Social Cognition: An Integrative Human Lifespan Approach. Curr Behav Neurosci Rep. 2020;7:175-192. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 131. | Shamay-Tsoory SG, Abu-Akel A. The Social Salience Hypothesis of Oxytocin. Biol Psychiatry. 2016;79:194-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 799] [Cited by in RCA: 646] [Article Influence: 64.6] [Reference Citation Analysis (1)] |
| 132. | Chaulagain RP, Shrestha Y, Shrestha H, Bhandari R, Gurung P. The neurobiological impact of oxytocin in mental health disorders: a comprehensive review. Ann Med Surg (Lond). 2025;87:1479-1486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 133. | Jin Y, Song D, Yan Y, Quan Z, Qing H. The Role of Oxytocin in Early-Life-Stress-Related Neuropsychiatric Disorders. Int J Mol Sci. 2023;24:10430. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (1)] |
| 134. | Florea T, Palimariciuc M, Cristofor AC, Dobrin I, Chiriță R, Bîrsan M, Dobrin RP, Pădurariu M. Oxytocin: Narrative Expert Review of Current Perspectives on the Relationship with Other Neurotransmitters and the Impact on the Main Psychiatric Disorders. Medicina (Kaunas). 2022;58:923. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 135. | Sikich L, Kolevzon A, King BH, McDougle CJ, Sanders KB, Kim SJ, Spanos M, Chandrasekhar T, Trelles MDP, Rockhill CM, Palumbo ML, Witters Cundiff A, Montgomery A, Siper P, Minjarez M, Nowinski LA, Marler S, Shuffrey LC, Alderman C, Weissman J, Zappone B, Mullett JE, Crosson H, Hong N, Siecinski SK, Giamberardino SN, Luo S, She L, Bhapkar M, Dean R, Scheer A, Johnson JL, Gregory SG, Veenstra-VanderWeele J. Intranasal Oxytocin in Children and Adolescents with Autism Spectrum Disorder. N Engl J Med. 2021;385:1462-1473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 246] [Cited by in RCA: 202] [Article Influence: 40.4] [Reference Citation Analysis (1)] |
| 136. | Josselsohn A, Zhao Y, Espinoza D, Hollander E. Oxytocin in neurodevelopmental disorders: Autism spectrum disorder and Prader-Willi syndrome. Pharmacol Ther. 2024;264:108734. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 137. | Olff M, Frijling JL, Kubzansky LD, Bradley B, Ellenbogen MA, Cardoso C, Bartz JA, Yee JR, van Zuiden M. The role of oxytocin in social bonding, stress regulation and mental health: an update on the moderating effects of context and interindividual differences. Psychoneuroendocrinology. 2013;38:1883-1894. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 530] [Cited by in RCA: 447] [Article Influence: 34.4] [Reference Citation Analysis (1)] |
| 138. | Gozzi M, Dashow EM, Thurm A, Swedo SE, Zink CF. Effects of Oxytocin and Vasopressin on Preferential Brain Responses to Negative Social Feedback. Neuropsychopharmacology. 2017;42:1409-1419. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 139. | Flechsenhar A, Levine SM, Müller LE, Herpertz SC, Bertsch K. Oxytocin and social learning in socially anxious men and women. Neuropharmacology. 2024;251:109930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 140. | Pérez-Arqueros V, Soler J, Schmidt C, Vega D, Pascual JC. Could intranasal oxytocin enhance the effects of psychotherapy in individuals with mental disorders? A systematic review and meta-analysis. Psychoneuroendocrinology. 2025;171:107206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 141. | Kang H, Glaser BD, Sartorius AI, Audunsdottir K, Kildal ES, Nærland T, Andreassen OA, Westlye LT, Quintana DS. Effects of oxytocin administration on non-social executive functions in humans: a preregistered systematic review and meta-analysis. Mol Psychiatry. 2025;30:2239-2251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 142. | Bhat US, Shahi N, Surendran S, Babu K. Neuropeptides and Behaviors: How Small Peptides Regulate Nervous System Function and Behavioral Outputs. Front Mol Neurosci. 2021;14:786471. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 34] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 143. | Zhou H, Zhu R, Xia Y, Zhang X, Wang Z, Lorimer GH, Ghiladi RA, Bayram H, Wang J. Neuropeptides affecting social behavior in mammals: Oxytocin. Peptides. 2024;177:171223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 144. | Lawson EA. Understanding oxytocin in human physiology and pathophysiology: A path towards therapeutics. Compr Psychoneuroendocrinol. 2024;19:100242. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 145. | Cui X, Xiao L. Complexity of the Hypothalamic Oxytocin System and its Involvement in Brain Functions and Diseases. Neurosci Bull. 2025;41:1267-1288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 146. | Feldman R. Oxytocin and social affiliation in humans. Horm Behav. 2012;61:380-391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 572] [Cited by in RCA: 424] [Article Influence: 30.3] [Reference Citation Analysis (1)] |
| 147. | De Dreu CK, Kret ME. Oxytocin Conditions Intergroup Relations Through Upregulated In-Group Empathy, Cooperation, Conformity, and Defense. Biol Psychiatry. 2016;79:165-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 237] [Cited by in RCA: 179] [Article Influence: 17.9] [Reference Citation Analysis (1)] |
| 148. | Zhou M, Zhu S, Xu T, Wang J, Zhuang Q, Zhang Y, Becker B, Kendrick KM, Yao S. Neural and behavioral evidence for oxytocin's facilitatory effects on learning in volatile and stable environments. Commun Biol. 2024;7:109. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 149. | Zik JB, Roberts DL. The many faces of oxytocin: implications for psychiatry. Psychiatry Res. 2015;226:31-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 57] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 150. | Quintana DS, Lischke A, Grace S, Scheele D, Ma Y, Becker B. Advances in the field of intranasal oxytocin research: lessons learned and future directions for clinical research. Mol Psychiatry. 2021;26:80-91. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 226] [Cited by in RCA: 182] [Article Influence: 36.4] [Reference Citation Analysis (0)] |
| 151. | Shafer SL, Ririe DG, Miller S, Curry RS, Hsu DT, Sullivan GM, Eisenach JC. Plasma pharmacokinetics of intravenous and intranasal oxytocin in nonpregnant adults. Br J Anaesth. 2025;134:1513-1522. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 152. | Martins D, Brodmann K, Veronese M, Dipasquale O, Mazibuko N, Schuschnig U, Zelaya F, Fotopoulou A, Paloyelis Y. "Less is more": A dose-response account of intranasal oxytocin pharmacodynamics in the human brain. Prog Neurobiol. 2022;211:102239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 153. | Winterton A, Westlye LT, Steen NE, Andreassen OA, Quintana DS. Improving the precision of intranasal oxytocin research. Nat Hum Behav. 2021;5:9-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 154. | Mitre M, Kranz TM, Marlin BJ, Schiavo JK, Erdjument-Bromage H, Zhang X, Minder J, Neubert TA, Hackett TA, Chao MV, Froemke RC. Sex-Specific Differences in Oxytocin Receptor Expression and Function for Parental Behavior. Gend Genome. 2017;1:142-166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 10] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 155. | Proverbio AM. Sex differences in the social brain and in social cognition. J Neurosci Res. 2023;101:730-738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 52] [Article Influence: 17.3] [Reference Citation Analysis (1)] |
| 156. | Lu Q, Hu S. Sex differences of oxytocin and vasopressin in social behaviors. Handb Clin Neurol. 2021;180:65-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 157. | Marazziti D, Carter CS, Carmassi C, Della Vecchia A, Mucci F, Pagni G, Carbone MG, Baroni S, Giannaccini G, Palego L, Dell'Osso L. Sex matters: The impact of oxytocin on healthy conditions and psychiatric disorders. Compr Psychoneuroendocrinol. 2023;13:100165. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 158. | Grimm S, Pestke K, Feeser M, Aust S, Weigand A, Wang J, Wingenfeld K, Pruessner JC, La Marca R, Böker H, Bajbouj M. Early life stress modulates oxytocin effects on limbic system during acute psychosocial stress. Soc Cogn Affect Neurosci. 2014;9:1828-1835. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 77] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 159. | Thornton JL, Everett NA, Webb P, Turner AJ, Cornish JL, Baracz SJ. Adolescent oxytocin administration reduces depression-like behaviour induced by early life stress in adult male and female rats. Prog Neuropsychopharmacol Biol Psychiatry. 2021;110:110279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 160. | Maoz H, Grossman-Giron A, Sedoff O, Nitzan U, Kashua H, Yarmishin M, Arad O, Tzur Bitan D. Intranasal oxytocin as an adjunct treatment among patients with severe major depression with and without comorbid borderline personality disorder. J Affect Disord. 2024;347:39-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 161. | Rung JM, Horta M, Tammi EM, Perez E, Ojeda MC, Lin T, Harris G, Somerville J, Salmeron D, Beltz SE, Sandesara B, Feifel D, Ebner NC. Safety and tolerability of chronic intranasal oxytocin in older men: results from a randomized controlled trial. Psychopharmacology (Berl). 2021;238:2405-2418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 162. | Leffa DD, Daumann F, Damiani AP, Afonso AC, Santos MA, Pedro TH, Souza RP, Andrade VM. DNA damage after chronic oxytocin administration in rats: a safety yellow light? Metab Brain Dis. 2017;32:51-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 163. | Bales KL, Solomon M, Jacob S, Crawley JN, Silverman JL, Larke RH, Sahagun E, Puhger KR, Pride MC, Mendoza SP. Long-term exposure to intranasal oxytocin in a mouse autism model. Transl Psychiatry. 2014;4:e480. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 103] [Article Influence: 8.6] [Reference Citation Analysis (1)] |
| 164. | Veening JG, Olivier B. Intranasal administration of oxytocin: behavioral and clinical effects, a review. Neurosci Biobehav Rev. 2013;37:1445-1465. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 136] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 165. | Sarnecky BA, Vanderwall DK, Mason HM, Kirschner SM, Ambrose B, Parker TL. Evaluation of a Proprietary Slow-Release Oxytocin Formulation on Corpus Luteum Function in Mares. J Equine Vet Sci. 2019;77:28-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 166. | Brackley AD, Toney GM. Oxytocin Receptor Activation Rescues Opioid-Induced Respiratory Depression by Systemic Fentanyl in the Rat. J Pharmacol Exp Ther. 2021;378:96-107. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 167. | Cardoso C, Ellenbogen MA, Orlando MA, Bacon SL, Joober R. Intranasal oxytocin attenuates the cortisol response to physical stress: a dose-response study. Psychoneuroendocrinology. 2013;38:399-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 155] [Article Influence: 11.9] [Reference Citation Analysis (1)] |
| 168. | Sabe M, Zhao N, Crippa A, Strauss GP, Kaiser S. Intranasal Oxytocin for Negative Symptoms of Schizophrenia: Systematic Review, Meta-Analysis, and Dose-Response Meta-Analysis of Randomized Controlled Trials. Int J Neuropsychopharmacol. 2021;24:601-614. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 169. | Horta M, Kaylor K, Feifel D, Ebner NC. Chronic oxytocin administration as a tool for investigation and treatment: A cross-disciplinary systematic review. Neurosci Biobehav Rev. 2020;108:1-23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 54] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 170. | Amin N, Hussein AB, Ye Q, Chen S, Wu F, Yuan X, Abbasi IN, Sundus J, Hu Z, Fang M. Combination of rTMS and oxytocin agonist attenuate depression-like behavior after postpartum depression in mice. Brain Res. 2025;1851:149459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 171. | Ren J, Zhang Y, Song H, Gou H, Zhao Q, Hong W, Piao Y, Chen Y, Chen Y, Wen S, Du Z, Li C, Qiu B, Ma Y, Zhang X, Wei Z. The interaction of oxytocin and nicotine addiction on psychosocial stress: an fMRI study. Transl Psychiatry. 2024;14:348. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 172. | Rigney N, de Vries GJ, Petrulis A, Young LJ. Oxytocin, Vasopressin, and Social Behavior: From Neural Circuits to Clinical Opportunities. Endocrinology. 2022;163:bqac111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 165] [Cited by in RCA: 128] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 173. | Marlin BJ, Froemke RC. Oxytocin modulation of neural circuits for social behavior. Dev Neurobiol. 2017;77:169-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 98] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 174. | Cataldo I, Azhari A, Lepri B, Esposito G. Oxytocin receptors (OXTR) and early parental care: An interaction that modulates psychiatric disorders. Res Dev Disabil. 2018;82:27-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 50] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 175. | Yang S, Dong X, Guo X, Han Y, Song H, Gao L, Dai W, Su Y, Zhang X. Serum Oxytocin Levels and an Oxytocin Receptor Gene Polymorphism (rs2254298) Indicate Social Deficits in Children and Adolescents with Autism Spectrum Disorders. Front Neurosci. 2017;11:221. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 176. | Conner TS, McFarlane KG, Choukri M, Riordan BC, Flett JAM, Phipps-Green AJ, Topless RK, Merriman ME, Merriman TR. The Oxytocin Receptor Gene (OXTR) Variant rs53576 Is Not Related to Emotional Traits or States in Young Adults. Front Psychol. 2018;9:2548. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 177. | Chander RJ, Mather KA, Cleary R, Grainger SA, Thalamuthu A, Numbers K, Kochan NA, Armstrong NJ, Brodaty H, Henry JD, Sachdev PS. The influence of rs53576 polymorphism in the oxytocin receptor (OXTR) gene on empathy in healthy adults by subtype and ethnicity: a systematic review and meta-analysis. Rev Neurosci. 2022;33:43-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 178. | Erickson EN, Myatt L, Danoff JS, Krol KM, Connelly JJ. Oxytocin receptor DNA methylation is associated with exogenous oxytocin needs during parturition and postpartum hemorrhage. Commun Med (Lond). 2023;3:11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 179. | Siu MT, Goodman SJ, Yellan I, Butcher DT, Jangjoo M, Grafodatskaya D, Rajendram R, Lou Y, Zhang R, Zhao C, Nicolson R, Georgiades S, Szatmari P, Scherer SW, Roberts W, Anagnostou E, Weksberg R. DNA Methylation of the Oxytocin Receptor Across Neurodevelopmental Disorders. J Autism Dev Disord. 2021;51:3610-3623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 180. | Navailles S, Zimnisky R, Schmauss C. Expression of glucocorticoid receptor and early growth response gene 1 during postnatal development of two inbred strains of mice exposed to early life stress. Dev Neurosci. 2010;32:139-148. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 34] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 181. | Forum DMK, Bjerregaard C, Thomsen PH. The significance of DNA methylation of the NR3C1 gene encoding the glucocorticoid receptor for developing resilience in individuals exposed to early life stress. Nord J Psychiatry. 2025;79:1-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 182. | Park C, Rosenblat JD, Brietzke E, Pan Z, Lee Y, Cao B, Zuckerman H, Kalantarova A, McIntyre RS. Stress, epigenetics and depression: A systematic review. Neurosci Biobehav Rev. 2019;102:139-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 388] [Cited by in RCA: 306] [Article Influence: 43.7] [Reference Citation Analysis (2)] |
| 183. | Wang Q, Shelton RC, Dwivedi Y. Interaction between early-life stress and FKBP5 gene variants in major depressive disorder and post-traumatic stress disorder: A systematic review and meta-analysis. J Affect Disord. 2018;225:422-428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 177] [Cited by in RCA: 156] [Article Influence: 19.5] [Reference Citation Analysis (1)] |
| 184. | Criado-Marrero M, Smith TM, Gould LA, Kim S, Penny HJ, Sun Z, Gulick D, Dickey CA, Blair LJ. FKBP5 and early life stress affect the hippocampus by an age-dependent mechanism. Brain Behav Immun Health. 2020;9:100143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 185. | Yamamoto T, Toki S, Siegle GJ, Takamura M, Takaishi Y, Yoshimura S, Okada G, Matsumoto T, Nakao T, Muranaka H, Kaseda Y, Murakami T, Okamoto Y, Yamawaki S. Increased amygdala reactivity following early life stress: a potential resilience enhancer role. BMC Psychiatry. 2017;17:27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 44] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 186. | Pati D, Harden SW, Sheng W, Kelly KB, de Kloet AD, Krause EG, Frazier CJ. Endogenous oxytocin inhibits hypothalamic corticotrophin-releasing hormone neurones following acute hypernatraemia. J Neuroendocrinol. 2020;32:e12839. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 187. | Yao S, Kendrick KM. Effects of Intranasal Administration of Oxytocin and Vasopressin on Social Cognition and Potential Routes and Mechanisms of Action. Pharmaceutics. 2022;14:323. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 38] [Article Influence: 9.5] [Reference Citation Analysis (1)] |
| 188. | Lee MR, Jayant RD. Penetration of the blood-brain barrier by peripheral neuropeptides: new approaches to enhancing transport and endogenous expression. Cell Tissue Res. 2019;375:287-293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 189. | Wiśniewski K, Alagarsamy S, Galyean R, Tariga H, Thompson D, Ly B, Wiśniewska H, Qi S, Croston G, Laporte R, Rivière PJ, Schteingart CD. New, potent, and selective peptidic oxytocin receptor agonists. J Med Chem. 2014;57:5306-5317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (1)] |