Published online Mar 19, 2026. doi: 10.5498/wjp.v16.i3.114446
Revised: November 6, 2025
Accepted: December 9, 2025
Published online: March 19, 2026
Processing time: 162 Days and 16.8 Hours
Anhedonia in major depressive disorder remains therapeutically challenging. Building on our prior randomized evidence of clinical benefit with Pediococcus acidilactici CCFM6432, we further investigated candidate peripheral immune and central reward correlates of treatment response.
To evaluate whether add-on CCFM6432 alleviates anhedonia through immune-inflammatory modulation and changes in reward-related electrophysiological measures.
Adults with major depressive disorder and anhedonia received standard antidepressant therapy plus CCFM6432 or plus placebo for 30 days. Assessments com
CCFM6432 produced greater reductions in lipopolysaccharide, C-reactive protein, and interleukin-6 vs placebo (P < 0.05). Decreases in these markers were aligned with improvements in the Temporal Experience of Pleasure Scale (total and anticipatory subscales) and with increased stimulus-preceding negativity amp
These findings provide preliminary clinical support that immune-inflammatory modulation may contribute to the anhedonia-relieving effects of CCFM6432, par
Core Tip: This study investigated whether Pediococcus acidilactici CCFM6432 all
- Citation: Li DX, Gu Y, Xia WJ, Sun YF, Hou L, Zhu WX, Wang J. Pediococcus acidilactici CCFM6432 alleviates anhedonia in major depression through immune-inflammatory modulation: An extended trial analysis. World J Psychiatry 2026; 16(3): 114446
- URL: https://www.wjgnet.com/2220-3206/full/v16/i3/114446.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i3.114446
Major depressive disorder (MDD) is a highly prevalent and disabling psychiatric condition, with core symptoms including depressed mood and anhedonia[1]. Anhe
In recent years, the inflammation hypothesis has gained traction as a key exp
The gut-brain axis, recognized as an important pathway for immune regulation and the pathogenesis of mood dis
Against this background, the present study conducted an extended analysis based on our prior RCT. We systematically assessed peripheral inflammatory markers [IL-6, IL-1β, TNF-α, CRP, LPS, toll-like receptor-4 (TLR-4), nuclear factor kappaB (NF-κB), nucleotide-binding oligomerization domain 2 (NOD-2)], neurotrophic factors and related proteins [BDNF, Pro-BDNF, tropomyosin related kinase B (TrkB)], as well as neurotransmitter-related indicators (Trp, DA), and integrated these biomarkers with subjective scales [Temporal Experience of Pleasure Scale (TEPS)] and objective electrophysiological indices [SPN, feedback related negativity (FRN)] to evaluate their associations with improvements in anhedonia. The novelty of this study lies in three aspects: (1) For the first time within the RCT framework, multiple categories of peripheral molecular markers were simultaneously measured to comprehensively assess the potential pathways of CCFM6432; (2) By comparing results across pathways, we identified that inflammatory factors were most closely associated with improvements in anhedonia; and (3) By integrating clinical symptomatology, electrophysiological measures, and peripheral blood molecular markers, we established a multimodal chain of evidence, strengthening the proposed role of gut-brain axis-mediated inflammatory mechanisms in anhedonia and providing new clinical and translational insights for precision interventions in MDD.
This study was an extended analysis of our previous randomized, double-blind, placebo-controlled clinical trial[29]. In that trial, we reported the beneficial effects of CCFM6432 on anhedonia symptoms and event-related potentials in patients with MDD. The present study further evaluated hematological indicators and their associations with im
In the previous RCT[29], 71 patients completed baseline assessments and were randomly allocated to the CCFM6432 group (n = 36) or the placebo group (n = 35). During follow-up, 16 patients dropped out due to medication adjustments, modified electroconvulsive therapy, antibiotic use, or poor compliance, resulting in incomplete clinical and electroencephalography (EEG) data. Ultimately, 55 patients (27 in the CCFM6432 group and 28 in the placebo group) completed follow-up and provided full clinical and EEG datasets. Although blood samples were collected from all enrolled par
In the original RCT, participants were randomly assigned in a 1:1 ratio to two groups. The CCFM6432 group received standard antidepressant treatment plus daily supplementation with CCFM6432 (≥ 109 colony-forming units/sachet; developed by the School of Food Science and Technology, Jiangnan University, and manufactured by the Yangzhou Institute of Food Biotechnology, Jiangnan University) for 30 consecutive days. The placebo group received standard antidepressant treatment plus a maltodextrin-based placebo identical to CCFM6432 in appearance, taste, color, and weight. No changes were made to the participants’ prescribed antidepressant regimens during the intervention. Treat
Depressive symptoms were assessed using the 17-item Hamilton Depression Rating Scale (HAMD-17), anxiety was assessed with the Hamilton Anxiety Rating Scale (HAMA), and anhedonia was evaluated with the TEPS, which includes ANT and CON subscales. EEG recordings during the “Doors Guessing Task” were used to extract SPN and FRN components, reflecting reward anticipation and feedback processing, respectively. Detailed procedures have been described in our previous report[29].
Peripheral venous blood (5 mL) was collected from each participant after an overnight fast at baseline and after 30 days of intervention. Samples were processed immediately by centrifugation at 3000 rpm for 10 minutes at 4 °C; sera were aliquoted and stored at -80 °C until analysis. To avoid batch effects, all samples were tested in a single batch at a third-party laboratory (Nanjing Lapuda Biotechnology Co., Ltd., Nanjing, China). Serum biomarkers were quantified using sandwich Enzyme-linked immunosorbent assay (ELISA) kits (brand: LAPUDA®; manufacturer: Nanjing Lapuda Biotechnology Co., Ltd., Nanjing, China; catalog and lot numbers listed in Supplementary Table 1) according to the manufac
All analyses were performed using SPSS version 27.0 (IBM Corp., Armonk, NY, United States). The normality of continuous variables was assessed with the Shapiro-Wilk test. For normally distributed data, independent-samples t tests were used for between-group comparisons; for non-normally distributed data, the Mann-Whitney U test was applied. Categorical variables were analyzed using the χ2 test. For hematological indicators, analysis of covariance (ANCOVA) was adopted as the primary analytical approach, with baseline values entered as covariates and follow-up values as dependent variables; partial eta squared (ηp2) was reported as the effect size. Change scores (Δ = baseline - follow-up) were calculated and compared between groups as supplementary analyses. To examine concurrent changes in clinical and biological measures, Pearson or partial correlation analyses were conducted using Δ values. Partial correlations were applied to control for changes in depressive and anxiety symptom severity (ΔHAMD and ΔHAMA), thereby accounting for potential confounding by overall symptom improvement. This approach allowed us to explore associations between reductions in inflammatory markers (CRP, IL-6, LPS) and improvements in anhedonia (TEPS total and subscale scores) as well as EEG indices (SPN and FRN). A two-tailed P value < 0.05 was considered statistically significant.
In the initial screening of the previous RCT, 92 patients were assessed, of whom 10 were excluded for not meeting the inclusion criteria, and 11 declined participation. The remaining 71 patients completed baseline assessments and were randomly allocated to the CCFM6432 group (n = 36) or the placebo group (n = 35). During follow-up, 16 patients dropped out due to medication adjustments, receipt of modified electroconvulsive therapy, antibiotic use, or poor compliance. Ultimately, 55 patients (27 in the CCFM6432 group and 28 in the placebo group) completed follow-up and were included in the present analysis. At baseline, there were no significant between-group differences in demographic or clinical characteristics, including sex, age, body mass index, years of education, illness duration, number of episodes, or antidepressant dosage (all P > 0.05), indicating comparability between groups. The complete baseline characteristics were reported in the previous RCT[29].
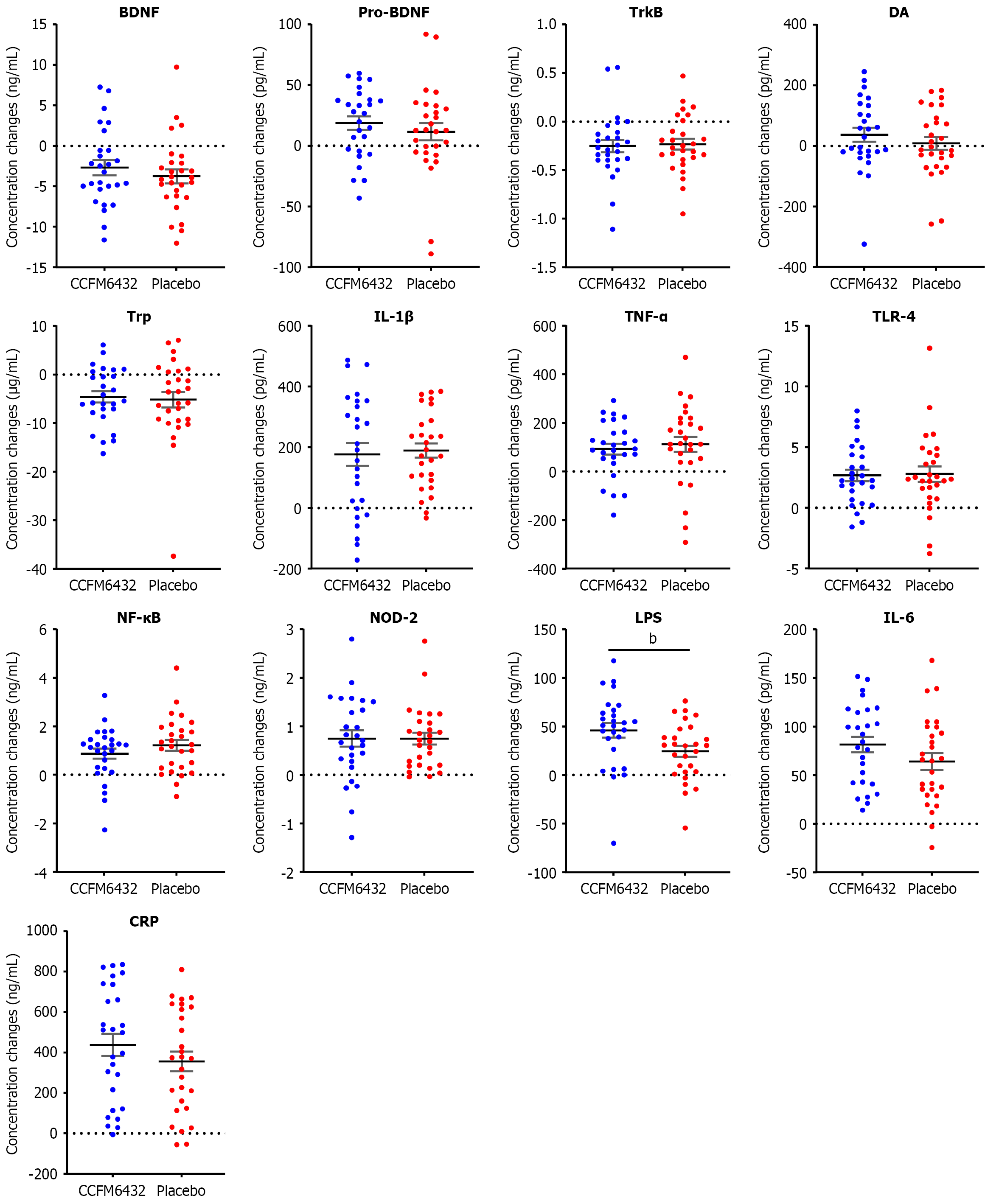
With respect to hematological indicators, ANCOVA revealed that after 30 days of intervention, peripheral blood levels of LPS, CRP, and IL-6 were significantly reduced in the CCFM6432 group, with greater decreases than in the placebo group (LPS: F = 6.705, P = 0.012, ηp2 = 0.114; IL-6: F = 8.672, P = 0.005, ηp2 = 0.143; CRP: F = 11.162, P = 0.002, ηp2 = 0.177). No significant between-group differences were observed for the other inflammatory markers (IL-1β, TNF-α, TLR-4, NF-κB, NOD-2). Similarly, no significant group differences were detected for neurotrophic factors and related proteins (BDNF, pro-BDNF, TrkB) or neurotransmitter-related markers (DA, Trp) (Table 1). As a supplementary exploratory sensitivity analysis, we also compared unadjusted change scores. Consistent with the ANCOVA results, LPS showed a significantly greater reduction in the CCFM6432 group, whereas CRP and IL-6 did not reach significance under this less statistically efficient approach (Figure 1). Importantly, the direction of effects remained consistent across both analytical strategies, indicating that the inflammatory findings are robust in their overall pattern despite differences in statistical power.
| Variable | CCFM6432 baseline | CCFM6432 follow-up | Placebo baseline | Placebo follow-up | F value | P value | ηp2 |
| BDNF (ng/mL) | 7.82 ± 3.48 | 10.53 ± 2.45 | 7.12 ± 3.49 | 10.87 ± 3.25 | 0.163 | 0.688 | 0.003 |
| Pro-BDNF (pg/mL) | 70.39 ± 22.98 | 51.56 ± 19.68 | 69.34 ± 24.66 | 57.70 ± 27.10 | 0.900 | 0.347 | 0.017 |
| TrkB (ng/mL) | 0.42 ± 0.27 | 0.67 ± 0.24 | 0.43 ± 0.19 | 0.66 ± 0.22 | 0.015 | 0.902 | 0.000 |
| DA (pg/mL) | 258.55 ± 72.95 | 221.03 ± 113.67 | 271.09 ± 86.52 | 261.96 ± 92.65 | 1.829 | 0.182 | 0.034 |
| Trp (μg/mL) | 12.99 ± 3.20 | 17.56 ± 5.46 | 12.17 ± 4.47 | 17.34 ± 8.07 | 0.000 | 0.999 | 0.000 |
| IL-1β (pg/mL) | 345.27 ± 177.58 | 169.06 ± 92.36 | 339.67 ± 138.30 | 150.20 ± 75.87 | 0.658 | 0.421 | 0.012 |
| TNF-α (pg/mL) | 289.41 ± 97.85 | 196.58 ± 65.27 | 327.21 ± 123.55 | 214.90 ± 115.88 | 0.404 | 0.528 | 0.008 |
| TLR-4 (ng/mL) | 5.44 ± 2.31 | 2.77 ± 1.28 | 6.03 ± 3.35 | 3.25 ± 1.71 | 1.019 | 0.317 | 0.019 |
| NF-κB (ng/mL) | 2.18 ± 0.75 | 1.31 ± 0.67 | 2.47 ± 0.85 | 1.25 ± 0.88 | 0.058 | 0.810 | 0.001 |
| NOD-2 (ng/mL) | 1.68 ± 0.62 | 0.93 ± 0.45 | 1.65 ± 0.52 | 0.90 ± 0.33 | 0.121 | 0.730 | 0.002 |
| LPS (ng/mL) | 74.05 ± 30.69 | 27.89 ± 18.15 | 68.67 ± 21.99 | 44.11 ± 27.44 | 6.705 | 0.012a | 0.114 |
| IL-6 (pg/mL) | 148.66 ± 55.29 | 67.18 ± 18.32 | 144.03 ± 47.59 | 80.02 ± 23.09 | 8.672 | 0.005a | 0.143 |
| CRP (ng/mL) | 597.79 ± 314.75 | 160.08 ± 42.55 | 543.70 ± 277.00 | 187.12 ± 47.45 | 11.162 | 0.002a | 0.177 |
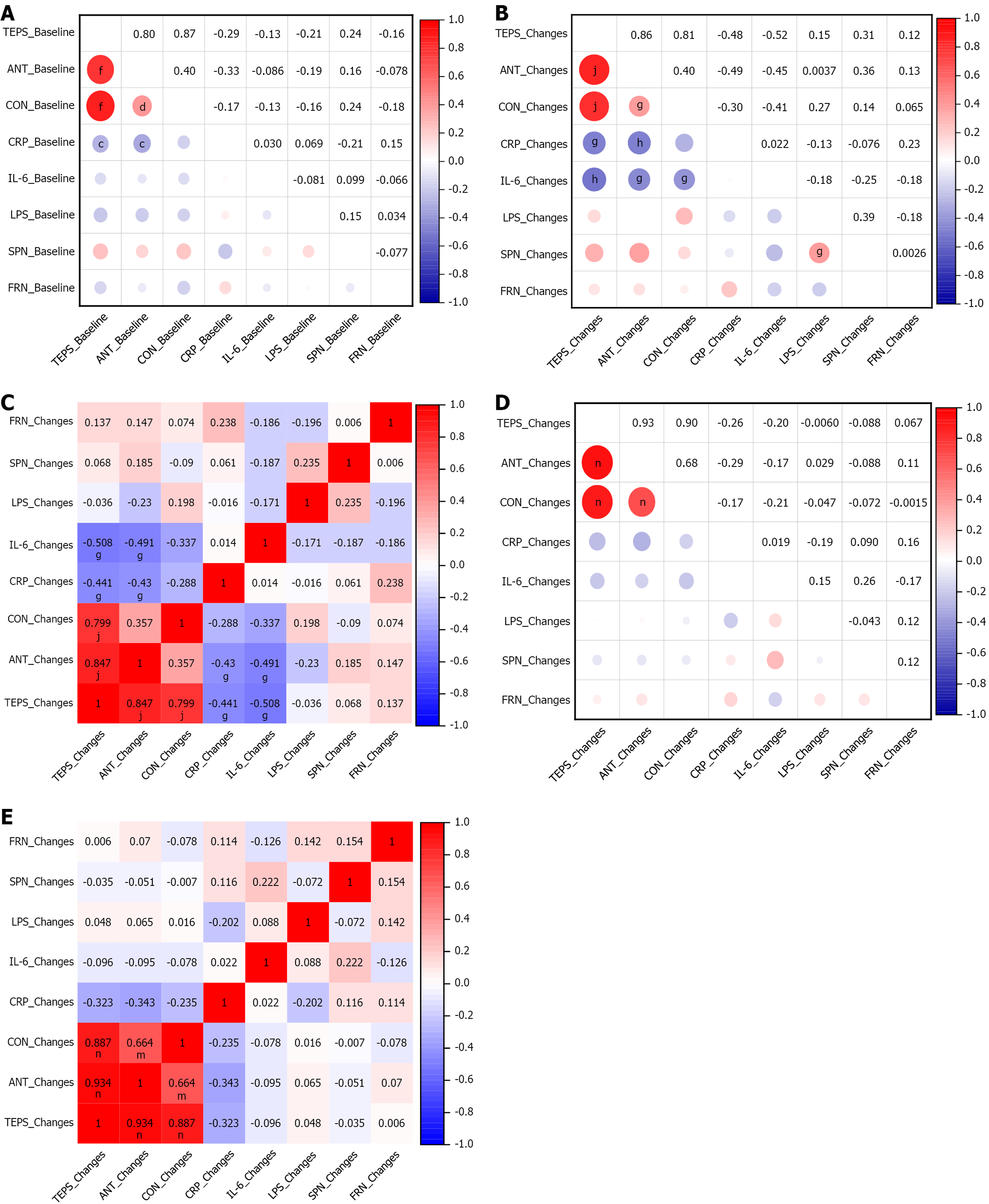
At baseline, correlation analyses showed that TEPS total scores and ANT subscale scores in MDD patients were negatively correlated with CRP levels (r approximately -0.29 to -0.33), indicating a close relationship between inflammatory status and anhedonia symptoms (Figure 2A). In post-intervention correlation analyses (calculated as baseline minus follow-up values), improvements in TEPS total and ANT subscale scores in the CCFM6432 group were signifi
Regarding EEG indices, the primary focus was the relationship between SPN and hematological markers. In the CCFM6432 group, the reduction in LPS was significantly positively correlated with improvements in SPN amplitude (r = 0.39, P < 0.05), indicating that greater reductions in LPS were associated with enhanced reward anticipation–related neural activity. Although this correlation attenuated and was no longer statistically significant after adjusting for changes in depressive and anxiety symptoms (r = 0.235), the directional trend remained. No such relationship was observed in the placebo group. No significant associations were found between FRN and inflammatory markers. Taken together, the CCFM6432 intervention significantly reduced peripheral blood levels of LPS, CRP, and IL-6 in patients with MDD. Furthermore, reductions in inflammatory markers were closely associated with improvements in anhedonia - particularly ANT pleasure - as well as with enhanced SPN amplitude. These findings suggest that among the multiple pathways assessed, inflammatory markers showed the strongest associations with improvements in anhedonia, supporting a potentially central role of inflammatory mechanisms.
As an extension of our previous RCT, this study is the first to simultaneously examine inflammatory factors, neurotrophic factors, and neurotransmitter-related markers in order to explore the peripheral molecular correlates underlying im
Previous research has shown that exogenous inflammatory stimuli (e.g., LPS or cytokine administration) can induce depression-like behaviors and anhedonic experiences in animals and healthy individuals[13,16]. In human studies, elevated inflammatory levels in patients with MDD have been closely linked to the severity of anhedonia[12,14,17,18]. Importantly, a recent RCT in MDD patients demonstrated that an acute LPS challenge induces rapid worsening of anhedonia, particularly among individuals with elevated baseline CRP levels[31]. This provides clinical evidence supporting a potential causal link between systemic inflammation and anhedonic symptoms in depression, complementing findings from animal and endotoxin-challenge studies. Mechanistically, inflammation-induced dopaminergic inhibition, kynurenine pathway activation, and reductions in BDNF signaling may converge to disrupt reward circuitry and synaptic plasticity[12,15,23]. Consistent with this framework, our findings demonstrate that CCFM6432 significantly reduced CRP and IL-6 levels, and these reductions were associated with improvements in ANT anhedonia, suggesting that immune-inflammatory modulation may represent a key pathway through which this probiotic exerts its therapeutic effects. This aligns with accumulating evidence indicating that anti-inflammatory interventions can improve depressive symptoms, including anhedonia[32].
Notably, the association between inflammation reduction and improvements in ANT pleasure was more pronounced than that for CON pleasure. This aligns with recent findings that elevated inflammation is more strongly related to ANT deficits than to CON[11,12,17]. For example, patients with higher CRP or IL-6 levels are more likely to show impairments in ANT pleasure rather than CON pleasure[17]. This may be attributable to the greater impact of inflammation on neural circuits involved in reward anticipation, such as the anterior cingulate cortex and ventral striatum, compared to circuits subserving immediate hedonic responses[12,17]. Thus, our study highlights the importance of differentiating anhedonia subtypes in future clinical and mechanistic research to identify specific pathological substrates and treatment targets.
This study also demonstrated that reductions in LPS were associated with enhanced SPN amplitudes, suggesting a potential cross-level association between peripheral inflammation and central reward processing. SPN, an event-related potential component reflecting reward anticipation, is consistently reported to be blunted in patients with MDD[33] and correlates with symptom severity. While our findings align with a “peripheral inflammation-central reward processing” framework, they remain correlational and cannot establish causality. Nevertheless, these results indicate that peripheral inflammatory markers may serve as external correlates of reward-related neural dysfunction, underscoring the need for future longitudinal and mechanistic studies. Moreover, associations between inflammatory improvements and reward anticipation (SPN and TEPS-ANT) were more prominent than those with CON reward processing (FRN and TEPS-CON). This pattern is consistent with evidence that inflammation preferentially disrupts motivational aspects of reward processing[12]. Future studies with larger samples and paradigms optimized for both anticipation and consummation will be essential to delineate subtype-specific neural and immune mechanisms.
Meanwhile, some indicators (e.g., IL-1β, TNF-α, BDNF, TrkB, DA, and Trp) did not show significant changes after intervention. This suggests that the effects of CCFM6432 may be primarily concentrated on specific aspects of immune-inflammatory responses (CRP, IL-6, LPS), rather than generalized inflammatory changes or the neurotransmitter/neurotrophic factors measured here. Previous animal studies have demonstrated that CCFM6432 downregulated hippo
From a clinical perspective, this study has several important implications. First, baseline analyses confirmed that inflammatory levels were closely correlated with the severity of anhedonia, suggesting that peripheral inflammatory markers may serve as potential biomarkers. Second, the improvements in anhedonia with CCFM6432 were partly inde
Several limitations should be acknowledged. First, the sample size was modest and derived from a single center, which may limit generalizability and constrain the feasibility of advanced multivariate modeling. Second, hematological biomarkers were collected only at baseline and day 30, limiting the ability to characterize inflammatory trajectories; future studies with denser sampling and longitudinal modeling are warranted. Third, this extension analysis was not originally powered for biomarker outcomes; therefore, multiple comparison correction was not applied, and these findings should be interpreted as preliminary and hypothesis-generating. Fourth, peripheral inflammatory markers cannot fully index central neuroimmune processes (e.g., functional magnetic resonance imaging), and EEG-biomarker associations were based solely on scalp recordings. Future multimodal investigations incorporating neuroimaging, cerebrospinal fluid cytokines, and advanced EEG analyses (e.g., source localization, topographical characterization) are needed to more definitively evaluate the inflammation-reward circuitry. Fifth, the study did not collect fecal or metabo
Taken together, this study demonstrates that Pediococcus acidilactici CCFM6432 effectively alleviates anhedonia in patients with MDD by downregulating peripheral inflammatory markers, particularly LPS, CRP, and IL-6. The alignment between decreases in inflammation and enhancements in SPN suggests that gut-brain axis inflammation regulation may represent a key mechanism. These findings not only provide new evidence for the pathophysiology of anhedonia but also highlight probiotics as a potential clinical strategy for precision interventions, warranting replication and extension in larger, multicenter, and multimodal studies.
As an extended analysis of a previous RCT, this study demonstrated that adjunctive administration of Pediococcus acidilactici CCFM6432 significantly reduced peripheral inflammatory markers (LPS, CRP, IL-6) in patients with MDD, and these reductions were closely associated with improvements in anhedonia, particularly ANT anhedonia. The observed link between decreases in inflammation and enhancements in SPN suggests that gut–brain axis inflammation regulation may represent a key mechanism. These findings provide both clinical and mechanistic support for probiotic interventions in anhedonia, while emphasizing the need for future validation in larger, multicenter, and multimodal studies.
We sincerely thank all participants who generously contributed their time and effort to this study.
| 1. | Marx W, Penninx BWJH, Solmi M, Furukawa TA, Firth J, Carvalho AF, Berk M. Major depressive disorder. Nat Rev Dis Primers. 2023;9:44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 543] [Cited by in RCA: 428] [Article Influence: 142.7] [Reference Citation Analysis (1)] |
| 2. | American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, 2013: 779. [RCA] [DOI] [Full Text] [Cited by in Crossref: 98503] [Cited by in RCA: 60284] [Article Influence: 3546.1] [Reference Citation Analysis (11)] |
| 3. | Wu C, Mu Q, Gao W, Lu S. The characteristics of anhedonia in depression: a review from a clinically oriented perspective. Transl Psychiatry. 2025;15:90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 29.0] [Reference Citation Analysis (0)] |
| 4. | Kasahara-Kiritani M, Kato T, Wakamatsu A, Webb T, Herr K, Vandervoort L, Li N. Understanding anhedonia in major depressive disorder in Japan: epidemiology and unmet needs from patients' and physicians' perspectives. BMC Psychiatry. 2025;25:631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 5. | Kale H, Cambron-Mellott MJ, Drissen T, Perkowski K, King-Concialdi K, Jha MK. Association between anhedonia severity and clinical, humanistic, and economic outcomes among US adults with major depressive disorder. Int J Neuropsychopharmacol. 2025;28:pyaf048. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 6. | Trøstheim M, Eikemo M, Meir R, Hansen I, Paul E, Kroll SL, Garland EL, Leknes S. Assessment of Anhedonia in Adults With and Without Mental Illness: A Systematic Review and Meta-analysis. JAMA Netw Open. 2020;3:e2013233. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 131] [Cited by in RCA: 124] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 7. | Nierenberg AA. Residual symptoms in depression: prevalence and impact. J Clin Psychiatry. 2015;76:e1480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 50] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 8. | Wong S, Le GH, Phan L, Rhee TG, Ho R, Meshkat S, Teopiz KM, Kwan ATH, Mansur RB, Rosenblat JD, McIntyre RS. Effects of anhedonia on health-related quality of life and functional outcomes in major depressive disorder: A systematic review and meta-analysis. J Affect Disord. 2024;356:684-698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 57] [Article Influence: 28.5] [Reference Citation Analysis (0)] |
| 9. | Cao B, Zhu J, Zuckerman H, Rosenblat JD, Brietzke E, Pan Z, Subramanieapillai M, Park C, Lee Y, McIntyre RS. Pharmacological interventions targeting anhedonia in patients with major depressive disorder: A systematic review. Prog Neuropsychopharmacol Biol Psychiatry. 2019;92:109-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 136] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 10. | Ducasse D, Loas G, Dassa D, Gramaglia C, Zeppegno P, Guillaume S, Olié E, Courtet P. Anhedonia is associated with suicidal ideation independently of depression: A meta-analysis. Depress Anxiety. 2018;35:382-392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 234] [Cited by in RCA: 199] [Article Influence: 24.9] [Reference Citation Analysis (0)] |
| 11. | Costi S, Morris LS, Collins A, Fernandez NF, Patel M, Xie H, Kim-Schulze S, Stern ER, Collins KA, Cathomas F, Parides MK, Whitton AE, Pizzagalli DA, Russo SJ, Murrough JW. Peripheral immune cell reactivity and neural response to reward in patients with depression and anhedonia. Transl Psychiatry. 2021;11:565. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 62] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 12. | Lucido MJ, Bekhbat M, Goldsmith DR, Treadway MT, Haroon E, Felger JC, Miller AH. Aiding and Abetting Anhedonia: Impact of Inflammation on the Brain and Pharmacological Implications. Pharmacol Rev. 2021;73:1084-1117. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 92] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 13. | Eisenberger NI, Berkman ET, Inagaki TK, Rameson LT, Mashal NM, Irwin MR. Inflammation-induced anhedonia: endotoxin reduces ventral striatum responses to reward. Biol Psychiatry. 2010;68:748-754. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 508] [Cited by in RCA: 466] [Article Influence: 29.1] [Reference Citation Analysis (0)] |
| 14. | Felger JC, Haroon E, Patel TA, Goldsmith DR, Wommack EC, Woolwine BJ, Le NA, Feinberg R, Tansey MG, Miller AH. What does plasma CRP tell us about peripheral and central inflammation in depression? Mol Psychiatry. 2020;25:1301-1311. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 360] [Cited by in RCA: 344] [Article Influence: 57.3] [Reference Citation Analysis (0)] |
| 15. | Felger JC, Treadway MT. Inflammation Effects on Motivation and Motor Activity: Role of Dopamine. Neuropsychopharmacology. 2017;42:216-241. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 385] [Cited by in RCA: 348] [Article Influence: 38.7] [Reference Citation Analysis (0)] |
| 16. | Brebner K, Hayley S, Zacharko R, Merali Z, Anisman H. Synergistic effects of interleukin-1beta, interleukin-6, and tumor necrosis factor-alpha: central monoamine, corticosterone, and behavioral variations. Neuropsychopharmacology. 2000;22:566-580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 164] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 17. | Burrows K, Stewart JL, Kuplicki R, Figueroa-Hall L, Spechler PA, Zheng H, Guinjoan SM; Tulsa 1000 Investigators, Savitz JB, Kent Teague T, Paulus MP. Elevated peripheral inflammation is associated with attenuated striatal reward anticipation in major depressive disorder. Brain Behav Immun. 2021;93:214-225. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 70] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 18. | Treadway MT. Anhedonia and Inflammation: Hiding in Plain Sight. Am J Psychiatry. 2025;182:509-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 19. | Haroon E, Daguanno AW, Woolwine BJ, Goldsmith DR, Baer WM, Wommack EC, Felger JC, Miller AH. Antidepressant treatment resistance is associated with increased inflammatory markers in patients with major depressive disorder. Psychoneuroendocrinology. 2018;95:43-49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 254] [Cited by in RCA: 225] [Article Influence: 28.1] [Reference Citation Analysis (0)] |
| 20. | Yang C, Wardenaar KJ, Bosker FJ, Li J, Schoevers RA. Inflammatory markers and treatment outcome in treatment resistant depression: A systematic review. J Affect Disord. 2019;257:640-649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 103] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 21. | Wu C, Lu J, Lu S, Huang M, Xu Y. Increased ratio of mature BDNF to precursor-BDNF in patients with major depressive disorder with severe anhedonia. J Psychiatr Res. 2020;126:92-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 22. | Fang X, Yang C, Li S, Zhan G, Zhang J, Huang N, Du X, Xu H, Hashimoto K, Luo A. Brain-derived neurotrophic factor-TrkB signaling in the medial prefrontal cortex plays a role in the anhedonia-like phenotype after spared nerve injury. Eur Arch Psychiatry Clin Neurosci. 2020;270:195-205. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 23. | Boyle CC, Bower JE, Eisenberger NI, Irwin MR. Stress to inflammation and anhedonia: Mechanistic insights from preclinical and clinical models. Neurosci Biobehav Rev. 2023;152:105307. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 34] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 24. | Zeng QB, Huang XB, Xu R, Shang DW, Huang SQ, Huang X, Yang XH, Balbuena L, Xiang YT, Zheng W. Kynurenine pathway metabolites predict antianhedonic effects of electroconvulsive therapy in patients with treatment-resistant depression. J Affect Disord. 2025;379:764-771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 25. | Liu P, Liu Z, Wang J, Wang J, Gao M, Zhang Y, Yang C, Zhang A, Li G, Li X, Liu S, Liu L, Sun N, Zhang K. Immunoregulatory role of the gut microbiota in inflammatory depression. Nat Commun. 2024;15:3003. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 179] [Cited by in RCA: 148] [Article Influence: 74.0] [Reference Citation Analysis (0)] |
| 26. | Cao Y, Cheng Y, Pan W, Diao J, Sun L, Meng M. Gut microbiota variations in depression and anxiety: a systematic review. BMC Psychiatry. 2025;25:443. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 41] [Article Influence: 41.0] [Reference Citation Analysis (0)] |
| 27. | Cryan JF, O'Riordan KJ, Cowan CSM, Sandhu KV, Bastiaanssen TFS, Boehme M, Codagnone MG, Cussotto S, Fulling C, Golubeva AV, Guzzetta KE, Jaggar M, Long-Smith CM, Lyte JM, Martin JA, Molinero-Perez A, Moloney G, Morelli E, Morillas E, O'Connor R, Cruz-Pereira JS, Peterson VL, Rea K, Ritz NL, Sherwin E, Spichak S, Teichman EM, van de Wouw M, Ventura-Silva AP, Wallace-Fitzsimons SE, Hyland N, Clarke G, Dinan TG. The Microbiota-Gut-Brain Axis. Physiol Rev. 2019;99:1877-2013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4131] [Cited by in RCA: 3445] [Article Influence: 492.1] [Reference Citation Analysis (4)] |
| 28. | Gao J, Zhao L, Cheng Y, Lei W, Wang Y, Liu X, Zheng N, Shao L, Chen X, Sun Y, Ling Z, Xu W. Probiotics for the treatment of depression and its comorbidities: A systemic review. Front Cell Infect Microbiol. 2023;13:1167116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 42] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 29. | Li DX, Hu QM, Xu CC, Yang HY, Liu JK, Sun YF, Wang G, Wang J, Zhou ZH. Efficacy of Pediococcus acidilactici CCFM6432 in alleviating anhedonia in major depressive disorder: A randomized controlled trial. World J Psychiatry. 2025;15:105249. [PubMed] [DOI] [Full Text] |
| 30. | Yang HY, Jiang JH, Pei YX, Qian X, Tian PJ, Wang G, Zhao JX. [Pediococcus acidilactici CCFM6432 improves depressive behavior in mice by alleviating brain inflammation]. Shipin Yu Fajiao Gongye. 2024;50:25-32. [DOI] [Full Text] |
| 31. | Savitz J, Figueroa-Hall LK, Teague TK, Yeh HW, Zheng H, Kuplicki R, Burrows K, El-Sabbagh N, Thomas M, Ewers I, Cha YH, Guinjoan S, Khalsa SS, Paulus MP, Irwin MR. Systemic Inflammation and Anhedonic Responses to an Inflammatory Challenge in Adults With Major Depressive Disorder: A Randomized Controlled Trial. Am J Psychiatry. 2025;182:560-568. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 32. | Raison CL, Rutherford RE, Woolwine BJ, Shuo C, Schettler P, Drake DF, Haroon E, Miller AH. A randomized controlled trial of the tumor necrosis factor antagonist infliximab for treatment-resistant depression: the role of baseline inflammatory biomarkers. JAMA Psychiatry. 2013;70:31-41. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1384] [Cited by in RCA: 1254] [Article Influence: 96.5] [Reference Citation Analysis (0)] |
| 33. | Ren X, White EJ, Nacke M, Mayeli A, Touthang J, Al Zoubi O, Kuplicki R, Victor TA, Paulus MP, Aupperle RL, Stewart JL. Blunted stimulus-preceding negativity during reward anticipation in major depressive disorder. J Affect Disord. 2024;362:779-787. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/