Published online Mar 19, 2026. doi: 10.5498/wjp.v16.i3.113765
Revised: October 22, 2025
Accepted: December 16, 2025
Published online: March 19, 2026
Processing time: 164 Days and 23.6 Hours
Schizophrenia presents complex challenges in older patients due to cognitive and social decline. Existing drug treatments offer limited benefits and pose risks, while aerobic exercise shows promise as a noninvasive intervention whose impact re
To investigate the effects of aerobic exercise on cognitive and social function in older patients with schizophrenia.
A retrospective study was conducted in 158 older patients with schizophrenia treated at The First Affiliated Hospital of Chongqing Medical and Pharmaceutical College (June 2023 and December 2024). The patients were divided into an ob
Post-treatment, Positive and Negative Syndrome Scale total scores were lower in the observation group than in the controls (P < 0.001), with significant improvements in positive, negative, and general symptoms. Measurement and Treatment Research to Improve Cognition in Schizophrenia Consensus Cognitive Battery domain scores were improved in processing speed, working memory, verbal/visual learning, and executive function (all P < 0.05). IPROS total scores were decreased (P < 0.001), indicating better social functioning. Longer exercise duration correlated with greater cognitive gains and lower social function deficits. Body mass index declined (P < 0.001), and adverse events were mild and transient (9.3%). Multivariate linear regression confirmed that aerobic exercise was independently associated with a significant reduction in IPROS scores (Adjusted β = -4.57, 95% confidence intervals: -6.08 to -3.05, P < 0.001).
Aerobic exercise effectively improves cognitive function, social function, and psychotic symptoms in older patients with schizophrenia, is safe, and serves as a valuable adjunctive intervention.
Core Tip: This study investigated the impact of aerobic exercise on the cognitive and social functions of older patients with schizophrenia. In a 12-week retrospective analysis, significant improvements were observed in cognitive domains (pro
- Citation: Ao C, Zhan L, Mou YL, Hu S, Huang YH. Influence of aerobic exercise on the cognitive and social functions of elderly patients with schizophrenia. World J Psychiatry 2026; 16(3): 113765
- URL: https://www.wjgnet.com/2220-3206/full/v16/i3/113765.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i3.113765
Schizophrenia is a severe chronic mental disorder with core symptoms including positive symptoms (e.g., hallucinations and delusions), negative symptoms (e.g., emotional blunting and social withdrawal), and cognitive impairment[1,2]. As the global population ages, the number of older patients with schizophrenia is increasing annually, presenting unique challenges for clinical management[3]. Older patients must cope with the symptoms caused by the disease itself and face age-related physiological decline, particularly the deterioration of cognitive and social functions[4]. A study on a Chinese population showed that cognitive dysfunction is widespread and relatively severe among older patients with schizophrenia[5]. Cognitive impairment encompasses multiple dimensions, including attention, working memory, and executive function, whereas social functioning includes activities of daily living, interpersonal relationships, and social participation[6]. The decline in these functions not only impairs patients’ quality of life but also increases the burden of care for families and society. Therefore, exploring effective interventions to improve the cognitive and social functioning of older patients with schizophrenia is clinically and socially significant.
Current schizophrenia treatment mainly relies on antipsychotic drugs, but their effects on cognitive and social functioning are limited[7]. Additionally, older patients are more prone to adverse reactions due to factors such as metabolic changes, polypharmacy, and increased drug sensitivity, further limiting the applicability of drug therapy[8]. Aerobic exercise, as a noninvasive therapeutic approach, has demonstrated unique advantages in neuropsychiatric disease research in recent years[9]. Its potential benefits are mediated through a confluence of neurobiological mechanisms. These include the upregulation of brain-derived neurotrophic factor, which is pivotal for synaptic plasticity and neuronal survival in key brain regions such as the hippocampus and prefrontal cortex[10,11]. Furthermore, exercise-induced enhancements in cerebral blood flow and oxygenation optimizes neural efficiency, whereas the modulation of systemic and neuroinflammation may contribute to symptom alleviation and cognitive improvement[12,13]. Collectively, these mechanisms may underpin the observed positive effects of regular aerobic exercise on cognitive function in schizophrenia, including improvements in memory and executive function linked to hippocampal and prefrontal integrity[14,15]. However, systematic research investigating the impact of aerobic exercise on cognitive and social functions is still lacking for the specific and growing population of older patients with schizophrenia.
Current research suggests that the potential mechanisms encompass multiple pathways, including enhanced neuroplasticity, reduced inflammatory responses, and improved cardiovascular function[10]. Multiple studies have shown that regular aerobic exercise may positively impact the cognitive function in patients with schizophrenia by promoting an increase in hippocampal volume, optimizing prefrontal cortex function, and enhancing white matter[11,12]. However, systematic research on the impact of aerobic exercise on cognitive and social functions is still lacking for older patients with schizophrenia.
Therefore, this study systematically explored the effects of aerobic exercise on cognitive and social functions in older patients with schizophrenia through a retrospective analysis of clinical data. This study utilized existing clinical data to answer specific scientific questions within a relatively short period, making it suitable for exploring the long-term effects of aerobic exercise in real-world clinical settings. This study aimed to clarify the effects of exercise intervention and possible influencing factors, thereby providing clinical evidence for individualized exercise prescriptions. This not only helps to expand intervention strategies for functional rehabilitation in older schizophrenia patients, but also has significant implications for promoting the application of the “biological-psychological-social” integrated model in the field of geriatric mental health.
This study had a retrospective design. We collected data from all older patients with schizophrenia treated at The First Affiliated Hospital of Chongqing Medical and Pharmaceutical College between June 2023 and December 2024. Overall, 192 patients were initially identified via a hospital information system, and 158 were included in the study after applying the inclusion and exclusion criteria. These patients were divided into two groups based on the actual treatment received: The observation group (n = 86), which underwent a 3-month aerobic rehabilitation training in addition to conventional treatment. The control group (n = 72) received conventional treatment alone, which comprised stable doses of second-generation antipsychotics (risperidone, olanzapine, or aripiprazole, adjusted to ≤ ± 25% chlorpromazine-equivalent change), routine nursing care, and weekly psychiatric consultations without any structured exercise or non-pharmacological cognitive interventions. The study was reviewed and approved by the Ethics Committee of The First Affiliated Hospital of Chongqing Medical and Pharmaceutical College.
Inclusion criteria: (1) Age ≥ 18-years-old; (2) Patients who meet diagnostic criteria for schizophrenia and have been jointly diagnosed by two psychiatrists with the title of associate chief physician or above[16]; (3) At least baseline and last evaluations were completed during the follow-up period; (4) Patients whose condition has been stable in the past 6 months and who can cooperate to complete the assessment of cognitive and social functions; and (5) The clinical data is complete.
Exclusion criteria: (1) Patients with other severe mental disorders (e.g., major depressive disorder, bipolar disorder, and Alzheimer’s disease); (2) Patients with severe physical diseases, such as acute myocardial infarction or malignant tumors; (3) Patients with contraindications to exercise or limited mobility; and (4) Patients who participated in other cognitive training or non-pharmaceutical intervention studies.
Considering the unique characteristics of older patients with schizophrenia, aerobic exercise programs were designed and implemented to fully consider physiological decline, high comorbidity rates, weak exercise endurance, and reduced balance abilities. The main types of exercise included: (1) Indoor stationary bicycles (padded seats, low step design); (2) Brisk walking on flat surfaces in corridors or rehabilitation halls (non-slip flooring, handrails on both sides); (3) Upper limb fluid resistance rowing machines (for patients with lower limb joint disorders); and (4) Outdoor Tai Chi, aerobics, and jogging. Exercise intensity was set at moderate intensity, with the target heart rate calculated as (220 - age - resting heart rate) × (50%-70%) + resting heart rate. Following the principle of individualization, exercise intensity was appropriately adjusted based on subjective exercise perception. Prior to each training session, a rehabilitation therapist conducts a 5-minute joint warm-up and 5-minute stretching routine, followed by a 5-minute cool-down after training. Initial phase (weeks 1-4): Twice weekly for 20 minutes per session; after adaptation (weeks 5-8), increase to three times weekly for 30 minutes per session; maintenance phase (week 9 onwards): Maintain three-four times weekly for 30-40 minutes per session, with a maximum of 45 minutes to prevent fatigue-induced fluctuations in mental symptoms. If patients experience symptoms, such as palpitations, chest tightness, dizziness, or joint pain during exercise, they should immediately stop exercising and take appropriate measures. The treatment cycle for this aerobic exercise rehabilitation program was 3 months.
Baseline patient data was collected through hospital information system, including age, sex, educational level, disease duration, medication use, marital status, family history of schizophrenia, body mass index (BMI), and fasting blood glucose. The mental symptoms and cognitive and social functions of the patients were evaluated at baseline and 3 months post-treatment.
The Positive and Negative Syndrome Scale (PANSS) was developed by Kay as a standardized tool for assessing the severity of psychotic symptoms in patients with schizophrenia[17]. It has been demonstrated to have good reliability and validity in the Chinese population[18]. The PANSS consists of three dimensions: Positive symptoms (seven items, reflecting an excess or distortion of normal functions, such as delusions, hallucinations, and conceptual disorganization), negative symptoms (seven items, representing a diminution or loss of normal functions, such as blunted affect, emotional withdrawal, and passive social avoidance), and general psychopathological symptoms (16 items, assessing other aspects of psychopathology such as anxiety, guilt feelings, and poor attention). Each item is rated on a 7-point severity continuum (1 = “absent” to 7 = “extreme”), yielding a total score ranging from 30 to 210, with higher scores indicating greater symptom severity.
The Measurement and Treatment Research to Improve Cognition in Schizophrenia Consensus Cognitive Battery (MCCB) was used to assess cognitive function in patients with schizophrenia[19]. The reliability and validity of the MCCB in a Chinese population have been verified[20]. The MCCB evaluates seven cognitive domains through 10 standardized tests, which are particularly sensitive to the cognitive impairments commonly observed in schizophrenia: Speed of processing (assessed by Trail Making Test-A, symbol coding, and Verbal Fluency Test, reflecting the ability to quickly and fluently perform cognitive tasks), working memory (assessed by spatial span, measuring the capacity to hold and manipulate information online), verbal learning (assessed by Hopkins Verbal Learning Test-Revised), visual learning (assessed by Brief Visuospatial Memory Test-Revised; these learning domains reflect the ability to encode and retrieve new information), and reasoning and problem solving (assessed by Mazes, evaluating executive functions and planning ability). A shorter duration on the Trail Making Test A and higher scores on the other evaluations indicate better cognitive function in the corresponding dimension.
The Inpatient Psychiatric Rehabilitation Outcome Scale (IPROS) is a tool developed for assessing the rehabilitation outcomes of inpatients with psychiatric disorders, with a specific focus on their ability to reintegrate into society and live independently post-treatment[21]. The IPROS includes the following five assessment dimensions: Activities of daily living (e.g., eating, dressing), occupational therapy performance (e.g., engagement in assigned tasks), social interaction (e.g., initiating conversations, cooperating with others), interests/concerns (e.g., participation in recreational activities, showing future plans), and hygiene management (e.g., personal cleanliness). The scale uses a 5-point scoring system (0-4 points) across 36 items, with a higher total score indicating more severe deficits in social and daily living functions.
Statistical analysis was performed using IBM SPSS Statistics for Windows, version 26 (IBM Corp., Armonk, NY, United States). Measurement data were expressed as mean ± SD, whereas categorical data were expressed as n (%). Comparisons between groups before and after the intervention were analyzed using an independent samples t-test or Mann-Whitney U test, whereas intragroup pre- and post-intervention differences were tested with a paired t-test or Wilcoxon signed-rank test. Categorical data were compared using the χ2 test. Pearson’s or Spearman’s correlation analysis was used to explore the relationship between exercise duration and improvements in cognitive and social functions. To further explore the influence of aerobic exercise on rehabilitation outcomes, univariate linear regression analysis was performed with the post-treatment IPROS total score as the dependent variable and group assignment (aerobic exercise yes/no) as the independent variable. Subsequently, a multivariate linear regression model was constructed to adjust for potential confounders, including age, sex, disease duration, and chlorpromazine equivalent dose. The results were presented as β coefficients with their 95% confidence intervals (CI). Statistical significance was set at P < 0.05.
This study included 158 older patients with schizophrenia, of whom 86 and 72 were in the observation and control groups, respectively (Table 1). The results of the baseline characteristic comparisons showed no significant differences between the two groups regarding age, disease duration, medication dosage, sex, educational attainment, marital status, or family history of schizophrenia (P > 0.05), indicating that the two groups were comparable. The control group did not engage in any structured aerobic exercise program as part of their routine treatment.
| Variables | Total (n = 158) | Control group (n = 72) | Observation group (n = 86) | Statistic | P value |
| Age (years) | 66.93 ± 3.66 | 67.11 ± 3.71 | 66.78 ± 3.63 | t = 0.57 | 0.572 |
| Disease duration (years) | 7.39 ± 4.38 | 7.17 ± 4.51 | 7.58 ± 4.29 | t = -0.59 | 0.555 |
| Chlorpromazine equivalent (mg/day) | 309.99 ± 23.89 | 311.74 ± 25.16 | 308.53 ± 22.81 | t = 0.84 | 0.402 |
| Sex | χ2 = 0.02 | 0.887 | |||
| Female | 89 (56.33) | 41 (56.94) | 48 (55.81) | ||
| Male | 69 (43.67) | 31 (43.06) | 38 (44.19) | ||
| Degree of education | χ2 = 0.07 | 0.963 | |||
| Primary school and below | 64 (40.51) | 30 (41.67) | 34 (39.53) | ||
| Junior to senior high school | 74 (46.84) | 33 (45.83) | 41 (47.67) | ||
| Senior high school and above | 20 (12.66) | 9 (12.50) | 11 (12.79) | ||
| Marital status | χ2 = 0.49 | 0.483 | |||
| Married | 121 (76.58) | 57 (79.17) | 64 (74.42) | ||
| Unmarried/divorced/widowed | 37 (23.42) | 15 (20.83) | 22 (25.58) | ||
| Family history | χ2 = 0.42 | 0.518 | |||
| No | 141 (89.24) | 63 (87.50) | 78 (90.70) | ||
| Yes | 17 (10.76) | 9 (12.50) | 8 (9.30) |
All patients in the observation group completed 3 months of aerobic exercise rehabilitation training. Patients in the observation group completed an average of 32.85 ± 3.25 aerobic exercise sessions, with an average total exercise duration of 937.59 ± 77.43 minutes. Among them, 62 (72.09%) participated in indoor stationary bicycle exercises, 78 (90.70%) walked briskly on flat ground in the corridor or rehabilitation hall, 15 (17.44%) used an upper limb fluid resistance rowing machine, and 43 (50.00%) participated in outdoor Tai Chi and yoga.
Pre-treatment, no significant differences were found in the PANSS total score or scores for positive, negative, and general psychopathological symptoms between the two groups (P > 0.05), indicating comparability at baseline (Table 2). Post-treatment, the total PANSS score in the observation group was significantly lower than that in the control group (63.49 vs 68.28, P < 0.001), with significant reductions in positive symptoms (15.83 vs 16.64, P = 0.019), negative symptoms (13.09 vs 15.46, P < 0.001), and general psychopathological symptoms (34.57 vs 36.18, P = 0.024) were significantly improved, indicating that aerobic exercise has a positive alleviating effect on the psychotic symptoms of older patients with schizophrenia (Table 2).
| Variable | Group | Control group (n = 72) | Observation group (n = 86) | t value | P value |
| Total score | Pre-treatment | 81.61 ± 7.29 | 81.29 ± 6.26 | 0.30 | 0.767 |
| Post-treatment | 68.28 ± 5.32c | 63.49 ± 4.84c | 5.92 | < 0.001 | |
| Positive | Pre-treatment | 19.15 ± 2.68 | 19.55 ± 2.17 | -1.02 | 0.309 |
| Post-treatment | 16.64 ± 2.27c | 15.83 ± 2.04c | 2.37 | 0.019 | |
| Negative | Pre-treatment | 18.79 ± 2.58 | 19.02 ± 2.16 | -0.61 | 0.540 |
| Post-treatment | 15.46 ± 2.30c | 13.09 ± 1.86c | 7.14 | < 0.001 | |
| General | Pre-treatment | 43.67 ± 5.80 | 42.72 ± 5.29 | 1.07 | 0.286 |
| Post-treatment | 36.18 ± 4.57c | 34.57 ± 4.29c | 2.28 | 0.024 |
Pre-treatment, no significant differences were observed in the scores of each dimension of the MCCB scale between the two groups (Table 3). The observation group showed significantly better post- treatment scores on the MCCB scale compared to the control group across multiple dimensions: Trail Making Test (54.68 vs 61.94, P < 0.001), symbol coding (33.38 vs 30.33, P = 0.024), Hopkins Verbal Learning Test (19.26 vs 17.12, P = 0.003), spatial span (19.78 vs 18.62, P = 0.024), Mazes (10.53 vs 8.28, P < 0.001), Brief Visuospatial Memory Test-Revised (17.16 vs 15.33, P < 0.001), and verbal fluence test (19.37 vs 17.54, P = 0.013). These findings suggest that aerobic exercise significantly enhanced cognitive function in this patient population.
| Variable | Group | Control group (n = 72) | Observation group (n = 86) | t value | P value |
| Trail Making Test-A | Pre-treatment | 77.53 ± 29.47 | 81.24 ± 25.58 | -0.85 | 0.397 |
| Post-treatment | 61.94 ± 10.08c | 54.68 ± 11.27c | 4.22 | < 0.001 | |
| Symbol coding | Pre-treatment | 29.22 ± 11.44 | 28.59 ± 12.85 | 0.32 | 0.748 |
| Post-treatment | 30.33 ± 9.15 | 33.38 ± 7.37a | -2.28 | 0.024 | |
| Hopkins | Pre-treatment | 15.93 ± 5.71 | 15.38 ± 5.28 | 0.62 | 0.533 |
| Post-treatment | 17.12 ± 4.49 | 19.26 ± 4.29c | -3.04 | 0.003 | |
| Spatial span | Pre-treatment | 17.12 ± 3.68 | 17.56 ± 3.39 | -0.77 | 0.443 |
| Post-treatment | 18.62 ± 3.15a | 19.78 ± 3.20c | -2.27 | 0.024 | |
| Mazes | Pre-treatment | 7.44 ± 3.13 | 7.63 ± 2.82 | -0.39 | 0.699 |
| Post-treatment | 8.28 ± 2.74 | 10.53 ± 2.37c | -5.55 | < 0.001 | |
| Visuospatial Memory | Pre-treatment | 13.92 ± 3.17 | 13.58 ± 3.78 | 0.60 | 0.552 |
| Post-treatment | 15.33 ± 3.03a | 17.16 ± 2.88c | -3.88 | < 0.001 | |
| Verbal fluence | Pre-treatment | 16.90 ± 4.54 | 16.52 ± 5.11 | 0.49 | 0.625 |
| Post-treatment | 17.54 ± 4.70 | 19.37 ± 4.42c | -2.52 | 0.013 |
There was no significant difference in the IPROS scores between the two groups pre-treatment (Table 4). Post-treatment, the observation group exhibited significantly lower total IPROS scores than the control group (29.40 vs 33.89, P < 0.001). Notable improvements were observed in specific domains: Activities of daily living (6.41 vs 7.51, P = 0.003), occupational therapy performance (7.29 vs 8.33, P = 0.018), social interaction (3.85 vs 4.85, P < 0.001), and interests/concerns (8.10 vs 9.11, P = 0.019). These results indicate that aerobic exercise facilitates the recovery of social function in older patients with schizophrenia.
| Variable | Group | Control group (n = 72) | Observation group (n = 86) | t value | P value |
| Activities of daily living | Pre-treatment | 9.36 ± 2.65 | 9.20 ± 3.14 | 0.35 | 0.727 |
| Post-treatment | 7.51 ± 2.30c | 6.41 ± 2.28c | 3.03 | 0.003 | |
| Occupational therapy performance | Pre-treatment | 11.31 ± 3.28 | 11.37 ± 3.43 | -0.12 | 0.902 |
| Post-treatment | 8.33 ± 2.66c | 7.29 ± 2.77c | 2.40 | 0.018 | |
| Social interaction | Pre-treatment | 7.92 ± 2.93 | 8.27 ± 2.62 | -0.79 | 0.428 |
| Post-treatment | 4.85 ± 1.55c | 3.85 ± 1.29c | 4.42 | < 0.001 | |
| Interests/concerns | Pre-treatment | 11.01 ± 3.27 | 10.90 ± 2.88 | 0.24 | 0.809 |
| Post-treatment | 9.11 ± 2.71c | 8.10 ± 2.62c | 2.37 | 0.019 | |
| Hygiene management | Pre-treatment | 6.43 ± 2.37 | 6.73 ± 2.46 | -0.78 | 0.436 |
| Post-treatment | 4.08 ± 1.46c | 3.67 ± 1.61c | 1.66 | 0.100 | |
| Total score | Pre-treatment | 46.03 ± 6.97 | 46.47 ± 6.51 | -0.41 | 0.684 |
| Post-treatment | 33.89 ± 4.66c | 29.40 ± 4.94c | 5.84 | < 0.001 |
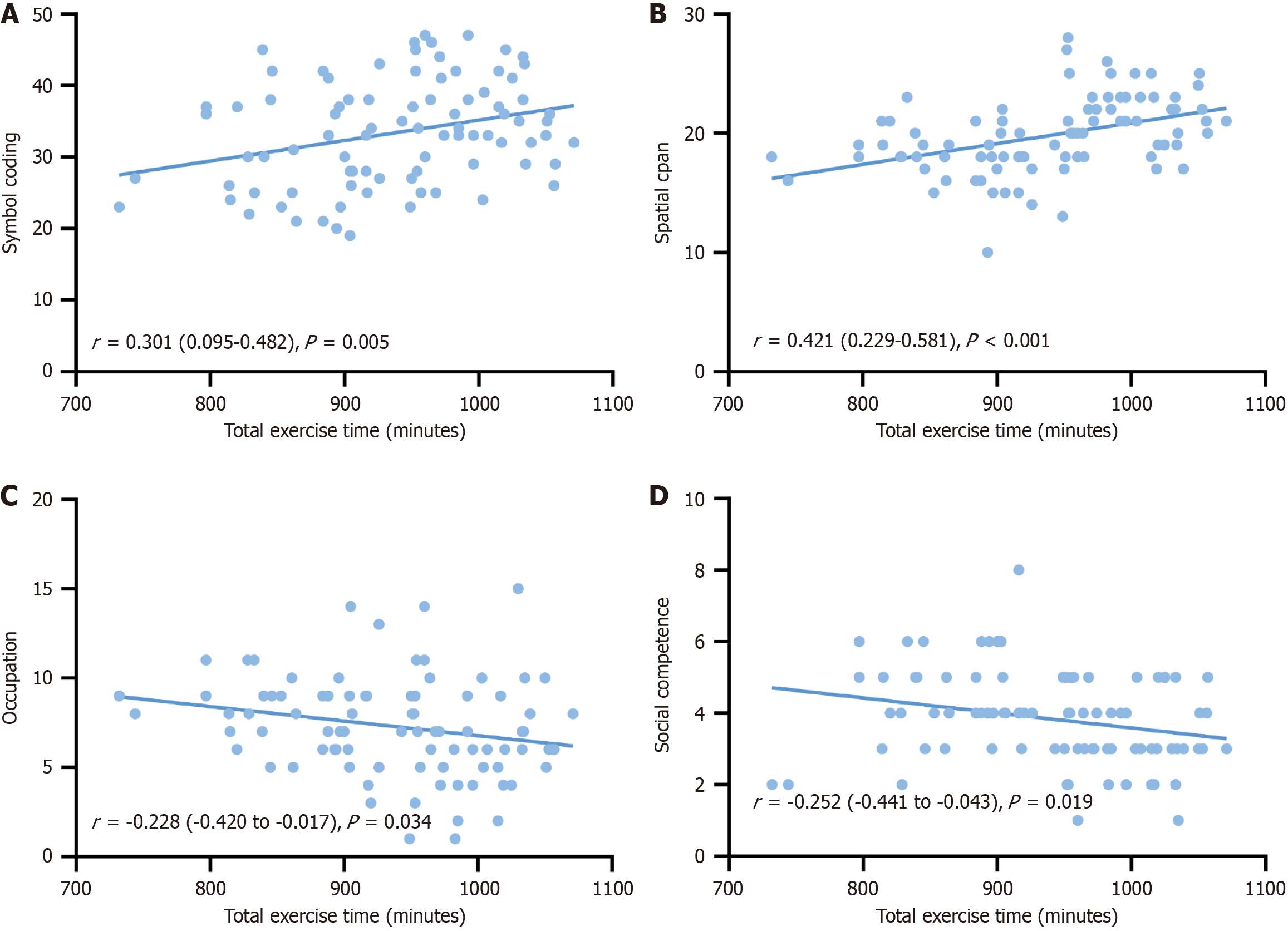
Correlation analysis revealed a positive association between exercise duration and MCCB symbol coding scores (r = 0.301, P = 0.005) and spatial span scores (r = 0.421, P < 0.001; Figure 1). Conversely, exercise duration was negatively correlated with IPROS occupational therapy scores (r = -0.228, P = 0.034) and social interaction scores (r = -0.252, P = 0.019). These findings suggest that longer exercise duration is associated with greater cognitive improvement and enhanced recovery of social function.
Post-treatment, the observation group showed a significant reduction in BMI compared to the control group (25.41 vs 26.82, P < 0.001), with no significant changes in fasting blood glucose levels (4.97 vs 5.09, P = 0.541; Table 5). During the 12-week aerobic exercise rehabilitation period, eight mild adverse events (incidence rate, 9.30%) were reported in the observation group: Four cases of mild joint pain, three cases of transient palpitations, and one case of mild hypotension. All patients experienced complete relief after a short rest period or symptomatic treatment. No severe adverse events were reported. These results demonstrated the safety and metabolic benefits of aerobic exercise in this patient cohort.
| Variable | Group | Control group (n = 72) | Observation group (n = 86) | t value | P value |
| BMI | Pre-treatment | 26.67 ± 2.70 | 26.39 ± 3.04 | 0.61 | 0.542 |
| Post-treatment | 26.82 ± 2.49 | 25.41 ± 2.53a | 3.55 | < 0.001 | |
| FBG | Pre-treatment | 5.24 ± 1.37 | 5.25 ± 1.43 | -0.04 | 0.965 |
| Post-treatment | 5.09 ± 1.16 | 4.97 ± 1.28 | 0.61 | 0.541 |
Univariate linear regression analysis identified a significant negative association between participation in the aerobic exercise program and post-treatment IPROS total score (β = -4.49, 95%CI: -6.00 to -2.99, P < 0.001), indicating better social functioning in the observation group (Table 6). This association remained significant after adjusting for age, sex, disease duration, and chlorpromazine equivalent dose in the multivariate model (adjusted β = -4.57, 95%CI: -6.08 to -3.05, P < 0.001). These results demonstrate that aerobic exercise is an independent factor associated with improved psychosocial rehabilitation outcomes in older patients with schizophrenia.
With the continuous increase in the aging global population, the number of older patients with schizophrenia is growing increasingly large[22]. Antipsychotic drugs have limited efficacy in improving functional outcomes, and the older population is prone to adverse reactions owing to their metabolic characteristics. Therefore, there is an urgent need for safe and effective non-pharmacological interventions. This study systematically investigated improvements in cognitive and social functioning in older patients with schizophrenia through a retrospective analysis of aerobic exercise over three months. Our retrospective study provides real-world evidence that a 12-week aerobic exercise program significantly improved psychotic symptoms, cognitive function, and social functioning in older inpatients with schizophrenia. Furthermore, the total duration of exercise correlated with the degree of improvement in cognitive and social functions. Additionally, aerobic exercise can reduce BMI, and the incidence of adverse events is low (9.30%), suggesting that aerobic exercise is a safe and effective adjunctive intervention for this population.
From a neurobiological perspective, the improvement in cognitive function observed in this study may be related to the neurobiological mechanisms induced by aerobic exercise. Guo et al[23] reported the effectiveness of aerobic exercise in reducing PANSS scores through a review study, particularly in significantly alleviating negative symptoms. They proposed the possibility of using aerobic exercise as an adjunctive treatment in patients with schizophrenia. The observed robust gains across multiple MCCB domains, particularly in processing speed, working memory, and executive function, strongly suggest that aerobic exercise induces beneficial neuroplastic changes[24]. Although our study did not measure neurobiological markers, our findings align with the proposed mechanism that exercise may upregulate brain-derived neurotrophic factor, which is crucial for hippocampal and prefrontal cortex function[10,11]. The prefrontal cortex is closely associated with executive functions (e.g., maze and connection tests), whereas the hippocampus is the core brain region for memory processing (e.g., the Hopkins Word Learning Test)[25,26]. This provides a plausible explanation for why our exercise group showed superior performance in the Hopkins Verbal Learning Test (hippocampus-dependent) and the Mazes test (prefrontal cortex-dependent) compared to that of controls. Furthermore, the concurrent reduction in negative symptoms and cognitive deficits raises the possibility of a shared underlying pathway, perhaps through enhanced dopaminergic and glutamatergic transmission in fronto-temporal circuits, a hypothesis worthy of future investigation.
Furthermore, regular exercise increases cerebral blood flow, improves blood oxygen saturation in the prefrontal cortex and parietal lobe, and enhances information processing efficiency[12,27]. This may explain why symbolic coding and spatial span scores are positively correlated with exercise duration. Additionally, Ventura et al[13] reported that the frequency of aerobic exercise was significantly associated with a reduction in inflammatory factors, suggesting that aerobic exercise alleviates mental symptoms by reducing the release of inflammatory factors and neuroinflammatory damage in the brain. These findings provide new insights into the target sites for exercise interventions.
In terms of social functioning, the observation group showed significant improvement over the control group in multiple dimensions of the IPROS scale, and total exercise time was negatively correlated with occupational therapy and social interaction scores (i.e., the longer the exercise time, the more significant the improvement in social functioning). This result may be related to how aerobic exercise promotes the restoration of social functioning by enhancing the patients’ confidence, emotional regulation abilities, and social skills. Korman et al[28] indicated that aerobic exercise enhances the social and daily living functions of patients with schizophrenia and should be regarded as an important auxiliary care method to routine care. Şenormancı et al[29] also reported that exercise significantly enhanced the patients’ social function and self-efficacy. A possible explanation for this is that exercise enhances patients’ self-efficacy by improving their body image and physical condition, thereby increasing their willingness to participate in daily activities and social interactions.
Furthermore, aerobic exercise promotes the secretion of endorphins and dopamine, improves anxiety and depression, and provides a positive emotional foundation for social interaction[30]. Additionally, group exercise programs provide patients with a platform for social support and skill learning, which further promotes the restoration of social functioning[31,32]. The significant improvement in social functioning observed in this study suggests that aerobic exercise may have a breakthrough effect on social isolation in older patients with schizophrenia, through a combination of physiological, psychological, and social mechanisms.
Based on the results of this study, aerobic exercise can be incorporated into rehabilitation programs for older patients with schizophrenia. Individualized exercise prescriptions should be developed for older patients with schizophrenia, focusing on moderate-intensity aerobic exercise. Exercise programs should have a progressive design, and the application of group exercise formats should be emphasized. Additionally, safety monitoring during exercise should be strengthened, particularly for patients at risk of cardiovascular disease, with regular assessments of heart rate, blood pressure, and other indicators to ensure exercise safety.
Furthermore, it is recommended that aerobic exercise be combined with medication therapy and psychosocial interventions to form an integrated rehabilitation model, thereby maximizing the functional recovery outcomes for patients. Although this study achieved specific results, it has some limitations. First, this was a retrospective analysis, which may have been subject to selection and information bias. Future studies should use prospective randomized controlled trials to validate the effects of aerobic exercise. Second, this study had a limited sample size and did not conduct a detailed analysis of the effects of different exercise types, intensities, and durations. Future studies should expand the sample size and explore strategies to optimize exercise programs. Finally, this study did not assess the effects of aerobic exercise on neurobiological markers. Future studies should integrate neuroimaging and molecular biology techniques to elucidate the underlying mechanisms.
This retrospective study provides real-world evidence that a 12-week moderate-intensity aerobic exercise program significantly improved cognitive, social, and symptomatic outcomes in older inpatients with schizophrenia. Using validated tools (MCCB, IPROS, and PANSS), we demonstrated robust gains in processing speed, working memory, verbal/visual learning, and executive function. We also found a marked reduction in social disability and PANSS total, positive, negative, and general scores. Exercise duration showed linear correlation with cognitive and social improvement. Furthermore, the multivariate regression analysis identified aerobic exercise as an independent predictor of improved psychosocial rehabilitation outcomes after adjusting for key confounders. These findings support the integration of individualized progressive aerobic exercise into multimodal geriatric schizophrenia care and call for larger randomized controlled trials to refine dosing and elucidate neurobiological mechanisms.
| 1. | Javitt DC. Cognitive Impairment Associated with Schizophrenia: From Pathophysiology to Treatment. Annu Rev Pharmacol Toxicol. 2023;63:119-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 104] [Article Influence: 34.7] [Reference Citation Analysis (0)] |
| 2. | Jauhar S, Johnstone M, McKenna PJ. Schizophrenia. Lancet. 2022;399:473-486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 871] [Cited by in RCA: 734] [Article Influence: 183.5] [Reference Citation Analysis (2)] |
| 3. | Solmi M, Seitidis G, Mavridis D, Correll CU, Dragioti E, Guimond S, Tuominen L, Dargél A, Carvalho AF, Fornaro M, Maes M, Monaco F, Song M, Il Shin J, Cortese S. Incidence, prevalence, and global burden of schizophrenia - data, with critical appraisal, from the Global Burden of Disease (GBD) 2019. Mol Psychiatry. 2023;28:5319-5327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 415] [Cited by in RCA: 354] [Article Influence: 118.0] [Reference Citation Analysis (0)] |
| 4. | Baran B, Lee EE. Age-Related Changes in Sleep and Its Implications for Cognitive Decline in Aging Persons With Schizophrenia: A Critical Review. Schizophr Bull. 2025;51:513-521. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 5. | Li W, Lin S, Yue L, Fang Y, Xiao S. Sex Differences in Obesity and Cognitive Function in Chinese Elderly Patients With Chronic Schizophrenia. Front Endocrinol (Lausanne). 2022;13:742474. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 6. | Li X, Zhang J, Hou R, Zheng M, Singh M, Li H, Li C, Zhang X, Yang X, Wu L, Wang Y, Zheng D. Bidirectional associations of intellectual and social activities with cognitive function among middle-aged and elderly adults in China. J Affect Disord. 2022;319:83-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 7. | Tiihonen J, Mittendorfer-Rutz E, Majak M, Mehtälä J, Hoti F, Jedenius E, Enkusson D, Leval A, Sermon J, Tanskanen A, Taipale H. Real-World Effectiveness of Antipsychotic Treatments in a Nationwide Cohort of 29 823 Patients With Schizophrenia. JAMA Psychiatry. 2017;74:686-693. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 567] [Cited by in RCA: 490] [Article Influence: 54.4] [Reference Citation Analysis (0)] |
| 8. | Stuhec M. Antipsychotic treatment in elderly patients on polypharmacy with schizophrenia. Curr Opin Psychiatry. 2022;35:332-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 9. | Smith PJ, Merwin RM. The Role of Exercise in Management of Mental Health Disorders: An Integrative Review. Annu Rev Med. 2021;72:45-62. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 285] [Cited by in RCA: 210] [Article Influence: 42.0] [Reference Citation Analysis (0)] |
| 10. | Liberona A, Jones N, Zúñiga K, Garrido V, Zelada MI, Silva H, Nieto RR. Brain-Derived Neurotrophic Factor (BDNF) as a Predictor of Treatment Response in Schizophrenia and Bipolar Disorder: A Systematic Review. Int J Mol Sci. 2024;25:11204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 11. | Sharma V, Singh TG, Kaur A, Mannan A, Dhiman S. Brain-Derived Neurotrophic Factor: A Novel Dynamically Regulated Therapeutic Modulator in Neurological Disorders. Neurochem Res. 2023;48:317-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 34] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 12. | Roell L, Fischer T, Keeser D, Papazov B, Lembeck M, Papazova I, Greska D, Muenz S, Schneider-Axmann T, Sykorova E, Thieme CE, Vogel BO, Mohnke S, Huppertz C, Roeh A, Keller-Varady K, Malchow B, Stoecklein S, Ertl-Wagner B, Henkel K, Wolfarth B, Tantchik W, Walter H, Hirjak D, Schmitt A, Hasan A, Meyer-Lindenberg A, Falkai P, Maurus I. Effects of aerobic exercise on hippocampal formation volume in people with schizophrenia - a systematic review and meta-analysis with original data from a randomized-controlled trial. Psychol Med. 2024;54:1-12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 13. | Ventura J, McEwen S, Subotnik KL, Hellemann GS, Ghadiali M, Rahimdel A, Seo MJ, Irwin MR, Nuechterlein KH. Changes in inflammation are related to depression and amount of aerobic exercise in first episode schizophrenia. Early Interv Psychiatry. 2021;15:213-216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 14. | Shimada T, Ito S, Makabe A, Yamanushi A, Takenaka A, Kawano K, Kobayashi M. Aerobic exercise and cognitive functioning in schizophrenia: An updated systematic review and meta-analysis. Psychiatry Res. 2022;314:114656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 65] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 15. | Maurus I, Roell L, Lembeck M, Papazova I, Greska D, Muenz S, Wagner E, Campana M, Schwaiger R, Schneider-Axmann T, Rosenberger K, Hellmich M, Sykorova E, Thieme CE, Vogel BO, Harder C, Mohnke S, Huppertz C, Roeh A, Keller-Varady K, Malchow B, Walter H, Wolfarth B, Wölwer W, Henkel K, Hirjak D, Schmitt A, Hasan A, Meyer-Lindenberg A, Falkai P. Exercise as an add-on treatment in individuals with schizophrenia: Results from a large multicenter randomized controlled trial. Psychiatry Res. 2023;328:115480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 29] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 16. | Tandon R, Gaebel W, Barch DM, Bustillo J, Gur RE, Heckers S, Malaspina D, Owen MJ, Schultz S, Tsuang M, Van Os J, Carpenter W. Definition and description of schizophrenia in the DSM-5. Schizophr Res. 2013;150:3-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 629] [Cited by in RCA: 501] [Article Influence: 38.5] [Reference Citation Analysis (0)] |
| 17. | Kay SR, Fiszbein A, Opler LA. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull. 1987;13:261-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17607] [Cited by in RCA: 16192] [Article Influence: 415.2] [Reference Citation Analysis (0)] |
| 18. | Sun J, Yee JY, See YM, Tang C, Zheng S, Ng BT, Lee J. Association between treatment resistance and cognitive function in schizophrenia. Singapore Med J. 2024;65:552-557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 19. | Green MF, Nuechterlein KH. The MATRICS initiative: developing a consensus cognitive battery for clinical trials. Schizophr Res. 2004;72:1-3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 197] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 20. | Lai S, Zhong S, Wang Y, Zhang Y, Xue Y, Zhao H, Ran H, Yan S, Luo Y, He J, Zhu Y, Lv S, Song Z, Miao H, Hu Y, Huang X, Lu X, Zhou J, Jia Y. The prevalence and characteristics of MCCB cognitive impairment in unmedicated patients with bipolar II depression and major depressive disorder. J Affect Disord. 2022;310:369-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 39] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 21. | Li G, Hu X, Jin D, Tian W, Phillips MR. A rating instrument for the evaluation of in-patient rehabilitation programmes in China. Results of reliability and validity testing. Br J Psychiatry Suppl. 1994;58-65. [PubMed] |
| 22. | GBD 2019 Mental Disorders Collaborators. Global, regional, and national burden of 12 mental disorders in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Psychiatry. 2022;9:137-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5012] [Cited by in RCA: 3880] [Article Influence: 970.0] [Reference Citation Analysis (1)] |
| 23. | Guo J, Liu K, Liao Y, Qin Y, Yue W. Efficacy and feasibility of aerobic exercise interventions as an adjunctive treatment for patients with schizophrenia: a meta-Analysis. Schizophrenia (Heidelb). 2024;10:2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 24. | Bang-Kittilsen G, Egeland J, Ueland T, Andersen E, Bigseth TT, Holmen TL, Mordal J, Holst R, Engh JA. The relationship between the brain-derived neurotrophic factor and neurocognitive response to physical exercise in individuals with schizophrenia. Psychoneuroendocrinology. 2023;157:106356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 25. | Friedman NP, Robbins TW. The role of prefrontal cortex in cognitive control and executive function. Neuropsychopharmacology. 2022;47:72-89. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1355] [Cited by in RCA: 1016] [Article Influence: 254.0] [Reference Citation Analysis (8)] |
| 26. | Ikeda N, Yamada S, Yasuda K, Uenishi S, Tamaki A, Ishida T, Tabata M, Tsuji T, Kimoto S, Takahashi S. Structural connectivity between the hippocampus and cortical/subcortical area relates to cognitive impairment in schizophrenia but not in mood disorders. J Neuropsychol. 2023;17:351-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 27. | Oliva HNP, Monteiro-Junior RS, Oliva IO, Powers AR. Effects of exercise intervention on psychotic symptoms: A meta-analysis and hypothetical model of neurobiological mechanisms. Prog Neuropsychopharmacol Biol Psychiatry. 2023;125:110771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 28. | Korman N, Stanton R, Vecchio A, Chapman J, Parker S, Martland R, Siskind D, Firth J. The effect of exercise on global, social, daily living and occupational functioning in people living with schizophrenia: A systematic review and meta-analysis. Schizophr Res. 2023;256:98-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 39] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 29. | Şenormancı G, Korkmaz N, Şenormancı Ö, Uğur S, Topsaç M, Gültekin O. Effects of Exercise on Resilience, Insight and Functionality in Patients with Chronic Schizophrenia in a Psychiatric Nursing Home Setting: A Randomized Controlled Trial. Issues Ment Health Nurs. 2021;42:690-698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 30. | Alizadeh Pahlavani H. Possible role of exercise therapy on depression: Effector neurotransmitters as key players. Behav Brain Res. 2024;459:114791. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 104] [Article Influence: 52.0] [Reference Citation Analysis (0)] |
| 31. | Schuch FB, Vancampfort D. Physical activity, exercise, and mental disorders: it is time to move on. Trends Psychiatry Psychother. 2021;43:177-184. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 101] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 32. | Farina EA, Assaf M, Corbera S, Chen CM. Factors Related to Passive Social Withdrawal and Active Social Avoidance in Schizophrenia. J Nerv Ment Dis. 2022;210:490-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/