Published online Apr 19, 2025. doi: 10.5498/wjp.v15.i4.103092
Revised: January 12, 2025
Accepted: February 13, 2025
Published online: April 19, 2025
Processing time: 102 Days and 0.5 Hours
Hypertension is a common chronic disease in the elderly population, and its association with cognitive impairment has been increasingly recognized. Cogni
To explore the differences between clinical data and cognitive function of elderly hypertensive patients with different nutritional status, analyze the internal relationship between nutritional statuses and cognitive impairment, and build a nomogram model for predicting nutritional status in elderly hypertensive patients.
The present study retrospectively analyzed 200 elderly patients admitted to our hospital for a hypertension during the period July 1, 2024 to September 30, 2024 as study subjects, and the 200 patients were divided into a modeling cohort (140 patients) and a validation cohort (60 patients) according to the ratio of 7:3. The modeling cohort were divided into a malnutrition group (26 cases), a malnutrition risk group (42 cases), and a normal nutritional status group (72 cases) according to the patients’ Mini-Nutritional Assessment Scale (MNA) scores, and the modeling cohort was divided into a hypertension combined with cognitive impairment group (34 cases) and a hypertension cognitively normal group (106 cases) according to the Montreal Cognitive Assessment Scale (MoCA) scores, and the validation cohort was divided into a hypertension combined with cognitive impairment group (14 cases) and hypertension cognitively normal group (46 cases). The study outcome was the occurrence of cognitive impairment in elderly hypertensive patients. Univariate and multivariate logistic regression was used to explore the relationship between the general information of the elderly hypertensive patients and the influence indicators and the occurrence of cognitive impairment, the roadmap prediction model was established and validated, the patient work receiver operating characteristic curve was used to evaluate the predictive efficacy of the model, the calibration curve was used to assess the consistency between the predicted events and the actual events, and the decision curve analysis was used to evaluate the validity of the model. Pearson correlation analysis was used to explore the relationship between nutrition-related indicators and MoCA scores.
In this research, the modeling cohort comprised 140 cases, while the verification cohort consisted of 60 cases, with no notable discrepancy in the data between the two groups. In the modeling cohort, there were significant differences in body mass index (BMI), albumin (ALB), hemoglobin (Hb) and homocysteine levels among the malnourished group, the malnourished risk group and the normal nutritional status group. The results of univariate and multivariate analysis showed that BMI [odds ratio (OR) = 0.830, P = 0.014], ALB (OR = 0.860, P = 0.028), Hb (OR = 0.939, P = 0.035) and MNA score (OR = 0.640, P = 0.000) were independent protective factors for patients without cognitive impairment, and alkaline phosphatase (ALP) (OR = 1.074, P = 0.000) was an independent risk factor for patients with cognitive impairment. In this study, the prediction nomogram tailored for cognitive deterioration in elderly patients with hypertension demonstrated robust predictive power and a close correspondence between predicted and observed outcomes. This model offers significant potential as a means to forestall cognitive decline in hypertensive elderly patients. ALP was negatively correlated with MoCA score, while BMI, MNA score, Hb and ALB were positively correlated with MoCA score.
BMI, MNA score, Hb and ALB were independent protective factors for cognitive impairment in elderly hypertensive patients and were positively correlated with MoCA score. ALP was an independent risk factor for cognitive impairment in elderly hypertensive patients and was negatively correlated with the MoCA score. The column line graph model established in the study has a good predictive value.
Core Tip: In summary, this study investigated the relationship between nutritional status and cognitive function in elderly hypertensive patients. By retrospectively analyzing data from 200 patients admitted for hypertension, the study divided them into modeling and validation cohorts, and further categorized them based on Mini-Nutritional Assessment Scale and Montreal Cognitive Assessment Scale (MoCA) scores. The results indicated that body mass index, Mini-Nutritional Assessment Scale score, hemoglobin, and albumin were independent protective factors for cognitive impairment, positively correlated with MoCA scores, while alkaline phosphatase was an independent risk factor, negatively correlated with MoCA scores. A predictive nomogram model for cognitive impairment was constructed and validated, exhibiting strong predictive accuracy and consistency in aligning predicted with actual events.
- Citation: Xu Q, Lu SR, Shi ZH, Yang Y, Yu J, Wang Z, Zhang BS, Hong K. Nutritional status of elderly hypertensive patients and its relation to the occurrence of cognitive impairment. World J Psychiatry 2025; 15(4): 103092
- URL: https://www.wjgnet.com/2220-3206/full/v15/i4/103092.htm
- DOI: https://dx.doi.org/10.5498/wjp.v15.i4.103092
In recent years, with the aging of society, the incidence of hypertension in the elderly in our country has exceeded 55%[1]. Hypertension, as the main disease to patients with elevated systemic arterial pressure, will further aggravate the damage of large and small arteries, the heart, the kidney, the brain and other organs[2]. At the same time, long-term hypertension will lead to the occurrence and accumulation of cerebrovascular damage such as atherosclerosis, cerebral infarction and leukoencephalopathy in the body, thus increasing the risk of cognitive disorders such as Alzheimer’s disease and vascular dementia[3]. Malnutrition is defined as micronutrient deficiencies such as wasting, obesity or iron-deficiency anemia occurring in the body due to insufficient, excessive or unbalanced energy intake[4]. Some studies have pointed out that[5], nutrition is an important factor affecting the recovery of the cognitive function in elderly stroke patients. The decline of gastrointestinal function and nutritional absorption ability of elderly patients may easily lead to malnutrition and then affect the metabolism of the patients, which may reduce the speed of recovery from neurological damage and lead to cognitive impairment[6]. Although there are studies suggesting that hypertension and nutritional status affect the cognitive function of elderly patients, there are limited studies exploring the correlation between nutritional status and cognitive function in elderly hypertensive patients, and there are large differences in the results. Therefore, this study aims to investigate the effect of nutritional status on cognitive function of elderly hypertensive patients by analyzing the differences and correlations between cognitive function and nutritional status of different elderly hypertensive patients, and to further construct a columnar graphical model of the occurrence of cognitive impairment in elderly hypertensive patients with a view to reducing the occurrence of cognitive impairment and improving the quality of life of elderly hypertensive patients.
A total of 200 elderly patients with hypertension admitted to our hospital from July 1, 2024 to September 30, 2024 were selected as the research subjects. The subjects were divided into a modeling cohort (140 cases) and a validation cohort (60 cases) in a ratio of 7:3. This study was approved by the hospital ethics committee. Inclusion criteria were: (1) Patients were diagnosed with hypertension according to JSH2019 hypertension management guidelines[7]; (2) Age 65 years and above; (3) Blood pressure was measured for 3 days, and the average systolic blood pressure was not less than 140 mmHg, and diastolic blood pressure was not less than 90 mmHg; (4) Patients received normal western medicine to control their blood pressure; and (5) Clinical data were kept intact. Exclusion criteria were: (1) Secondary hypertension, hypertensive crisis; (2) Combination of cerebrovascular diseases such as cerebral infarction and cerebral ischemia; (3) Combination of Alzheimer’s disease and other diseases affecting cognitive function; (4) Combination of psychiatric disorders; and (5) Combination of digestive disorders or diseases affecting nutritional status.
Baseline data were collected from all elderly hypertensive patients, including their general information, body composition and clinical indicators, and all general information and body composition and clinical indicators were collected on the day of admission to the hospital using serum.
General data: Sex, age, body mass index (BMI), family history of hypertension, duration of hypertension, grading of hypertension, history of alcohol consumption and smoking, as well as Mini-Nutritional Assessment (MNA)[8] and Montreal Cognitive Assessment (MoCA) scores[9], were collected and recorded in elderly hypertensive patients. BMI = weight/height2 (kg/m2). The basis for grading hypertension is shown in Table 1[10]. The MNA scale assesses the nutritional status of patients in four dimensions: Subjective rating, composite rating, dietary situation, and anthropometric measurements, with a total of 18 questions in the full scale. Patients with a score of < 17 were assessed as malnourished, those with a score of 17-23 were assessed as being at nutritional risk, and those with a score of ≥ 24 were assessed as being adequately nourished. The Cronbach’s alpha coefficient for the MNA scale was 0.711. The MoCA scale is mainly used for the assessment of cognitive function. The scale covers 8 subscales of language, attention, naming, memory, orientation, visuospatial, executive function, and abstraction, with a total scale score of 30, and the higher the total score, the better the patient’s cognitive function. A score of ≥ 26 was considered to be cognitively normal, and 18-26 was classified as mild cognitive impairment, with a Cronbach’s alpha coefficient of 0.818 for the scale.
| Separate into different kinds | Criteria for judgement |
| Grade 1 hypertension | SBP 140-159 mmHg and/or DBP 90-99 mmHg |
| Grade 2 hypertension | SBP 160 to 179 mmHg and/or DBP 100 to 109 mmHg |
| Grade 3 hypertension | SBP ≥ 180 mmHg and/or DBP ≥ 110 mmHg |
Body composition index: Patients’ fat-free body weight and upper arm muscle dimensions were measured using a body composition analyzer (InbodyS10 model) using bio-impedance analysis.
Clinical indicators: 5 mL of fasting peripheral venous blood was collected early in the morning, and the supernatant was centrifuged at 1500 r/minute and 8 cm for 10 minutes. The absolute value of lymphocytes was detected by fully automatic blood cell analyzer (F800), albumin (ALB) and hemoglobin (Hb) were detected by the immunoturbidimetric method, and aspartate aminotransferase and alanine aminotransferase (ALT) were detected by the rate method using a fully automatic biochemical analyzer (CM-800). ALT, serum creatinine, enzyme-linked immunosorbent assay for homocysteine (Hcy), alkaline phosphatase (ALP), uric acid was measured by radioimmunoassay, total cholesterol by cholesterol oxidase, triglycerides (TG) by glycerol kinase, and low-density lipoprotein cholesterol (LDL-C), and so on. TG were measured by glycerol kinase assay, LDL-C and high-density lipoprotein cholesterol by enzyme colourimetric assay.
Two hundred elderly hypertensive patients were divided into a modeling cohort (140 patients) and a validation cohort (60 patients) in a ratio of 7:3, and the baseline data of the two cohorts were compared. Based on the MNA score, patients in the modeling cohort were divided into a malnutrition group (MNA ≤ 16 points, n = 26), a malnutrition risk group (MNA 17-23 points, n = 42) and a group with normal nutritional status (MNA ≥ 24 points, n = 72). The 140 patients in the modeling cohort were then divided into the hypertension combined with cognitive impairment group (n = 34) and the hypertension cognitively normal group (n = 106) and the 60 patients in the validation cohort were divided into the hypertension combined with cognitive impairment group (n = 14) and the hypertension cognitively normal group (n = 46) by whether the patients developed cognitive impairment. The patients’ cognitive impairment was determined based on the MoCA score, and based on the results of the MoCA score, patients with a MoCA score of ≥ 26 were included in the hypertensive cognitive normal group, and patients with a MoCA score of < 26 were included in the hypertensive combined cognitive impairment group.
The primary observational outcome of this study was the occurrence of cognitive impairment in elderly hypertensive patients as judged by MoCA score. The secondary observational outcomes were the nutritional status of elderly hypertensive patients and baseline information.
Statistical analysis was performed using SPSS 26.0 and R software, and linear correlation analysis was performed using GraphPad Prism 8.0 software. Patients’ age, duration of hypertension, and all other clinical indicators and scale scores were expressed using mean ± SD, and independent samples t-tests were used to compare differences between two groups that conformed to normal distribution, one-way analysis of variance was used for comparisons between multiple groups, and counting information such as the number of cases was expressed as n (%), and the χ2-test was performed. One-way and multifactorial logistic regression analysis models were used to investigate the relationship between baseline information, nutritional status indicators and the occurrence of cognitive impairment in elderly patients, and the results were expressed as odds ratio (OR) and 95% confidence interval (CI). A prediction model of the column line plot was built to assess the accuracy of the column line plot with the discriminative power of the validation set and calibration plots. The area under the patient operating characteristic curve was used to evaluate the discriminative ability of the column-line diagram. Calibration curves for the model were calculated and the consistency of the model was verified with the Hosmer-Lemeshow test. Decision curve analysis was used to evaluate the discriminative ability of the model. Pearson correlation analysis was used to explore the relationship between nutrition-related indicators and MoCA scores. Differences were considered statistically significant at P < 0.05.
A total of 200 elderly hypertensive patients were included, with a male-to-female ratio of 93:107 and a mean age of 71.49 ± 8.01 years. They were divided into 140 cases in the modeling cohort and 60 cases in the validation cohort. There was no significant difference between the data of the modeling cohort and the validation cohort (P > 0.05) (Table 2).
| Norm | Total cases | Modeling queue | Verification queue | t/χ2/F | P value |
| Distinguishing between the sexes | 0.775 | 0.781 | |||
| Male | 93 (46.50) | 66 (47.14) | 27 (45.00) | ||
| Women | 107 (53.50) | 74 (52.86) | 33 (55.00) | ||
| Age, year | 71.49 ± 8.01 | 71.55 ± 8.03 | 71.44 ± 7.23 | 0.091 | 0.927 |
| BMI, kg/m2 | 23.31 ± 2.54 | 23.15 ± 2.42 | 22.52 ± 2.61 | 1.648 | 0.101 |
| Family history of hypertension | 1.046 | 0.307 | |||
| Yes | 91 (45.50) | 67 (47.86) | 24 (40.00) | ||
| No | 109 (54.50) | 73 (52.14) | 36 (60.00) | ||
| Course of hypertension, years | 9.36 ± 2.70 | 9.24 ± 2.81 | 9.71 ± 3.10 | 1.051 | 0.295 |
| High blood pressure classification | 1.200 | 0.549 | |||
| Level 1 | 107 (53.50) | 73 (52.14) | 34 (56.67) | ||
| Level 2 | 64 (32.00) | 48 (34.29) | 16 (26.67) | ||
| Level 3 | 29 (14.50) | 19 (13.57) | 10 (16.67) | ||
| Drinking history | 0.577 | 0.448 | |||
| Yes | 78 (39.00) | 57 (40.71) | 21 (35.00) | ||
| No | 122 (61.00) | 83 (59.29) | 39 (65.00) | ||
| Smoking history | |||||
| Yes | 109 (54.50) | 76 (54.29) | 33 (55.00) | 0.009 | 0.926 |
| No | 91 (45.50) | 64 (45.71) | 27 (45.00) | ||
| defatted body weight, kg | 54.92 ± 8.68 | 55.06 ± 8.92 | 54.46 ± 8.59 | 0.441 | 0.660 |
| Upper arm muscle dimension, cm | 25.01 ± 2.05 | 25.08 ± 2.11 | 24.88 ± 3.11 | 0.529 | 0.598 |
| ALB, g/L | 38.79 ± 5.66 | 38.82 ± 5.72 | 37.76 ± 3.62 | 1.325 | 0.187 |
| Hb, g/L | 133.62 ± 10.57 | 131.81 ± 10.92 | 134.47 ± 11.57 | 1.551 | 0.123 |
| AST, U/L | 20.44 ± 2.46 | 20.59 ± 2.51 | 20.35 ± 3.31 | 0.561 | 0.575 |
| ALT, U/L | 17.91 ± 2.93 | 17.95 ± 3.02 | 17.87 ± 2.81 | 0.175 | 0.861 |
| Cr, mmol/L | 80.23 ± 20.32 | 80.18 ± 20.42 | 80.43 ± 20.22 | 0.080 | 0.937 |
| Hcy, mmol/L | 20.67 ± 6.67 | 20.84 ± 6.83 | 20.42 ± 7.31 | 0.390 | 0.697 |
| UA, μmol/L | 260.66 ± 69.37 | 262.25 ± 70.15 | 259.68 ± 65.97 | 0.242 | 0.809 |
| ALP, U/L | 119.43 ± 19.60 | 119.18 ± 19.52 | 119.57 ± 19.71 | 0.129 | 0.897 |
| Absolute lymphocytes | 1.49 ± 0.60 | 1.49 ± 0.14 | 1.49 ± 0.65 | 0.000 | 1.000 |
| TC, mmol/L | 4.37 ± 1.01 | 4.08 ± 1.05 | 3.94 ± 0.81 | 0.922 | 0.358 |
| TG, mmol/L | 1.37 ± 0.82 | 1.44 ± 0.92 | 1.30 ± 0.62 | 1.078 | 0.283 |
| LDL-C, mmol/L | 1.20 ± 0.27 | 1.23 ± 0.23 | 1.15 ± 0.46 | 1.638 | 0.103 |
| HDL-C, mmol/L | 2.51 ± 0.70 | 2.53 ± 0.69 | 2.45 ± 0.77 | 0.725 | 0.469 |
| MNA score, cent | 21.77 ± 3.16 | 21.81 ± 3.52 | 21.74 ± 3.08 | 0.134 | 0.894 |
When the 140 modelled cohort of elderly hypertensive patients was divided into the malnutrition group (n = 26), the malnutrition risk group (n = 42), and the group with normal nutritional status (n = 72) according to MNA scores, there were significant differences (P < 0.05) between the BMI, ALB, Hb, and Hcy levels of the patients with different nutritional statuses as shown in Table 3.
| Norm | Total cases | Malnutrition group | Malnutrition risk group (n = 42) | Normal nutritional status group (n = 72) | t/χ2/F | P value |
| Distinguishing between the sexes | 0.541 | 0.763 | ||||
| Male | 66 (47.14) | 11 (42.31) | 19 (45.24) | 36 (50.00) | ||
| Women | 74 (52.86) | 15 (57.69) | 23 (54.76) | 36 (50.00) | ||
| Age, year | 71.55 ± 8.03 | 71.42 ± 8.71 | 71.53 ± 8.62 | 71.69 ± 8.51 | 0.011 | 0.989 |
| BMI, kg/m2 | 23.15 ± 2.42 | 20.63 ± 2.71 | 22.14 ± 2.27 | 24.69 ± 2.59 | 29.774 | 0.000 |
| Family history of hypertension | 0.243 | 0.886 | ||||
| Yes | 67 (47.86) | 13 (50.00) | 21 (50.00) | 33 (45.83) | ||
| No | 73 (52.14) | 13 (50.00) | 21 (50.00) | 39 (54.17) | ||
| Course of hypertension, year | 9.24 ± 2.81 | 9.68 ± 3.12 | 9.62 ± 3.14 | 9.71 ± 3.10 | 0.011 | 0.989 |
| High blood pressure classification | 4.486 | 0.344 | ||||
| Level 1 | 73 (52.14) | 12 (54.76) | 23 (54.76) | 38 (52.78) | ||
| Level 2 | 48 (34.29) | 9 (26.19) | 11 (26.19) | 28 (38.89) | ||
| Level 3 | 19 (13.57) | 5 (19.23) | 8 (19.05) | 6 (8.33) | ||
| Drinking history | 1.784 | 0.410 | ||||
| Yes | 57 (40.71) | 10 (38.46) | 14 (33.33) | 33 (45.83) | ||
| No | 83 (59.29) | 16 (61.54) | 28 (66.67) | 39 (54.17) | ||
| Smoking history | 0.006 | 0.997 | ||||
| Yes | 76 (54.29) | 14 (53.85) | 23 (54.76) | 39 (54.17) | ||
| No | 64 (45.71) | 12 (46.15) | 19 (45.24) | 33 (45.83) | ||
| Defatted body weight, kg | 55.06 ± 8.92 | 54.33 ± 8.29 | 54.62 ± 8.36 | 55.71 ± 9.06 | 0.341 | 0.712 |
| Upper arm muscle dimension, cm | 25.08 ± 2.11 | 24.12 ± 3.11 | 24.92 ± 2.22 | 25.18 ± 2.81 | 1.465 | 0.235 |
| ALB, g/L | 38.82 ± 5.72 | 36.59 ± 3.77 | 38.14 ± 3.62 | 39.89 ± 4.92 | 6.047 | 0.003 |
| Hb, g/L | 131.81 ± 10.92 | 140.36 ± 10.07 | 123.58 ± 10.28 | 123.67 ± 12.19 | 23.286 | 0.000 |
| AST, U/L | 20.59 ± 2.51 | 20.33 ± 3.34 | 20.32 ± 3.36 | 20.35 ± 3.31 | 0.058 | 0.954 |
| ALT, U/L | 17.95 ± 3.02 | 17.85 ± 2.82 | 17.82 ± 2.84 | 17.87 ± 2.81 | 0.115 | 0.909 |
| Cr, mmol/L | 80.18 ± 20.42 | 82.09 ± 19.11 | 80.20 ± 19.37 | 79.03 ± 22.21 | 0.210 | 0.811 |
| Hcy, mmol/L | 20.84 ± 6.83 | 22.96 ± 6.85 | 20.86 ± 6.57 | 17.03 ± 11.52 | 4.551 | 0.012 |
| UA, μmol/L | 262.25 ± 70.15 | 250.37 ± 60.14 | 258.34 ± 60.52 | 266.11 ± 77.32 | 0.527 | 0.592 |
| ALP, U/L | 119.18 ± 19.52 | 120.18 ± 19.11 | 119.18 ± 19.31 | 118.18 ± 20.42 | 0.105 | 0.900 |
| Absolute lymphocytes | 1.49 ± 0.14 | 1.45 ± 0.04 | 1.46 ± 0.13 | 1.50 ± 0.12 | 2.684 | 0.072 |
| TC, mmol/L | 4.08 ± 1.05 | 3.88 ± 0.84 | 3.81 ± 0.90 | 4.16 ± 0.96 | 2.199 | 0.115 |
| TG, mmol/L | 1.44 ± 0.92 | 1.30 ± 0.66 | 1.32 ± 0.63 | 1.35 ± 0.58 | 0.075 | 0.928 |
| LDL-C, mmol/L | 1.23 ± 0.23 | 1.13 ± 0.32 | 1.13 ± 0.31 | 1.14 ± 0.31 | 0.018 | 0.982 |
| HDL-C, mmol/L | 2.53 ± 0.69 | 2.44 ± 0.78 | 2.46 ± 0.77 | 2.50 ± 0.73 | 0.076 | 0.927 |
Comparison of the clinical data between the two groups in the modeling cohort in terms of BMI, duration of hypertension, ALB, Hb, ALP and MNA scores showed significant differences, with BMI, ALB, Hb and MNA scores being significantly lower in the hypertension combined with cognitive impairment group than in the control group, and the duration of hypertension as well as ALP being significantly higher (P < 0.05), and the rest of the indices being P > 0.05 (Table 4).
| Norm | Total cases | Hypertension combined with cognitive impairment group | Cognitively normal group with hypertension (n = 106) | t/χ2/F | P value |
| Distinguishing between the sexes | 0.165 | 0.685 | |||
| Male | 66 (47.14) | 15 (44.12) | 51 (48.11) | ||
| Women | 74 (52.86) | 19 (55.88) | 55 (51.89) | ||
| Age, year | 71.55 ± 8.03 | 72.15 ± 8.15 | 70.15 ± 6.15 | 1.518 | 0.131 |
| BMI, kg/m2 | 23.15 ± 2.42 | 22.56 ± 2.18 | 24.58 ± 2.98 | 3.648 | 0.000 |
| Family history of hypertension | 0.465 | 0.495 | |||
| Yes | 67 (47.86) | 18 (52.94) | 49 (46.23) | ||
| No | 73 (52.14) | 16 (47.06) | 57 (53.77) | ||
| Course of hypertension, year | 9.24 ± 2.81 | 10.84 ± 3.01 | 8.84 ± 2.67 | 3.683 | 0.000 |
| High blood pressure classification | 0.097 | 0.953 | |||
| Level 1 | 73 (52.14) | 18 (52.94) | 55 (51.89) | ||
| Level 2 | 48 (34.29) | 11 (32.35) | 37 (34.91) | ||
| Level 3 | 19 (13.57) | 5 (14.71) | 14 (13.21) | ||
| Drinking history | 0.114 | 0.735 | |||
| Yes | 57 (40.71) | 13 (38.24) | 44 (41.51) | ||
| No | 83 (59.29) | 21 (61.76) | 62 (58.49) | ||
| Smoking history | 0.332 | 0.564 | |||
| Yes | 76 (54.29) | 17 (50.00) | 59 (55.66) | ||
| No | 64 (45.71) | 17 (50.00) | 47 (44.34) | ||
| Defatted body weight, kg | 55.06 ± 8.92 | 54.18 ± 8.42 | 55.18 ± 9.43 | 0.552 | 0.582 |
| Upper arm muscle dimension, cm | 25.08 ± 2.11 | 24.81 ± 2.08 | 25.13 ± 2.05 | 0.789 | 0.431 |
| ALB, g/L | 38.82 ± 5.72 | 36.81 ± 4.51 | 39.71 ± 4.82 | 3.099 | 0.002 |
| Hb, g/L | 131.81 ± 10.92 | 128.98 ± 10.99 | 134.17 ± 11.31 | 2.344 | 0.021 |
| AST, U/L | 20.59 ± 2.51 | 20.81 ± 2.81 | 19.88 ± 2.48 | 1.841 | 0.067 |
| ALT, U/L | 17.95 ± 3.02 | 17.51 ± 2.91 | 18.05 ± 3.16 | 0.883 | 0.379 |
| Cr, mmol/L | 80.18 ± 20.42 | 82.18 ± 19.11 | 78.28 ± 19.52 | 1.019 | 0.310 |
| Hcy, mmol/L | 20.84 ± 6.83 | 21.13 ± 6.21 | 19.48 ± 6.28 | 1.337 | 0.184 |
| UA, μmol/L | 262.25 ± 70.15 | 256.81 ± 67.15 | 269.51 ± 50.31 | 1.176 | 0.242 |
| ALP, U/L | 119.18 ± 19.52 | 122.41 ± 20.51 | 102.62 ± 15.48 | 2.048 | 0.042 |
| Absolute lymphocytes | 1.49 ± 0.14 | 1.46 ± 0.13 | 1.53 ± 0.15 | 1.744 | 0.083 |
| TC, mmol/L | 4.08 ± 1.05 | 3.95 ± 1.01 | 4.21 ± 1.02 | 1.296 | 0.197 |
| TG, mmol/L | 1.44 ± 0.92 | 1.26 ± 0.53 | 1.56 ± 1.01 | 1.658 | 0.100 |
| LDL-C, mmol/L | 1.23 ± 0.23 | 1.21 ± 0.13 | 1.25 ± 0.11 | 1.763 | 0.080 |
| HDL-C, mmol/L | 2.53 ± 0.69 | 2.43 ± 0.61 | 2.61 ± 0.72 | 1.314 | 0.191 |
| MNA score, cent | 21.81 ± 3.52 | 19.21 ± 3.41 | 23.15 ± 3.14 | 6.234 | 0.000 |
Whether cognitive impairment occurred in elderly hypertensive patients was assigned (occurrence = 1, not occurred = 0), and gender, age, BMI, family history of hypertension, duration of hypertension, hypertension classification, history of alcohol consumption, history of smoking, defatted body weight, upper arm muscle dimension, ALB, Hb, aspartate aminotransferase, ALT, serum creatinine, Hcy, uric acid, ALP, absolute lymphocyte value, total cholesterol, TG, LDL-C, high-density lipoprotein cholesterol, and MNA scores were used as independent variables and assigned values, and one-way logistic analysis showed that BMI (OR = 0.846, P = 0.021), ALB (OR = 0.874, P = 0.004), Hb (OR = 0.959, P = 0.022), and ALP (OR = 1.070, P = 0.000), MNA score (OR = 0.667, P = 0.000) were significantly associated with the occurrence of cognitive impairment in patients (P < 0.05) (Tables 5 and 6).
| Independent variable | Definitions and assignments |
| Distinguishing between the sexes | Men (1), women (0) |
| Age | Substitute the original value |
| BMI | Substitute the original value |
| Family history of hypertension | Yes (1), no (0) |
| Course of hypertension | Substitute the original value |
| High blood pressure classification | Level 3 (3), level 2 (2), level 1 (1) |
| Drinking history | Yes (1), no (0) |
| Smoking history | Yes (1), no (0) |
| Defatted body weight | Substitute the original value |
| Upper arm muscle dimension | Substitute the original value |
| ALB | Substitute the original value |
| Hb | Substitute the original value |
| AST | Substitute the original value |
| ALT | Substitute the original value |
| Cr | Substitute the original value |
| Hcy | Substitute the original value |
| UA | Substitute the original value |
| ALP | Substitute the original value |
| Absolute lymphocytes | Substitute the original value |
| TC | Substitute the original value |
| TG | Substitute the original value |
| LDL-C | Substitute the original value |
| HDL-C | Substitute the original value |
| MNA score | Substitute the original value |
| Considerations | β | SE | Wald χ2 | P value | OR | 95%CI |
| Distinguishing between the sexes | -0.161 | 0.396 | 0.165 | 0.685 | 0.851 | 0.392-1.851 |
| Age | 0.045 | 0.030 | 2.255 | 0.133 | 1.046 | 0.986-1.108 |
| BMI | -0.167 | 0.072 | 5.350 | 0.021 | 0.846 | 0.734-0.975 |
| Family history of hypertension | 0.269 | 0.395 | 0.464 | 0.496 | 1.309 | 0.603-2.838 |
| Course of hypertension | 0.139 | 0.073 | 3.662 | 0.056 | 1.149 | 0.997-1.325 |
| High blood pressure classification | 0.009 | 0.276 | 0.001 | 0.975 | 1.009 | 0.587-1.733 |
| Drinking history | -0.137 | 0.404 | 0.114 | 0.735 | 0.872 | 0.395-1.926 |
| Smoking history | -0.227 | 0.395 | 0.332 | 0.565 | 0.797 | 0.367-1.727 |
| Defatted body weight | -0.012 | 0.022 | 0.301 | 0.583 | 0.988 | 0.947-1.031 |
| Upper arm muscle dimension | -0.077 | 0.098 | 0.626 | 0.429 | 0.926 | 0.764-1.121 |
| ALB | -0.135 | 0.046 | 8.518 | 0.004 | 0.874 | 0.799-0.957 |
| Hb | -0.042 | 0.018 | 5.264 | 0.022 | 0.959 | 0.925-0.994 |
| AST | 0.145 | 0.080 | 3.321 | 0.068 | 1.157 | 0.989-1.352 |
| ALT | -0.057 | 0.065 | 0.780 | 0.377 | 0.945 | 0.832-1.072 |
| Cr | 0.011 | 0.010 | 1.042 | 0.307 | 1.011 | 0.990-1.031 |
| Hcy | 0.042 | 0.032 | 1.767 | 0.184 | 1.043 | 0.980-1.110 |
| UA | -0.004 | 0.004 | 1.373 | 0.241 | 0.996 | 0.989-1.003 |
| ALP | 0.067 | 0.014 | 22.325 | 0.000 | 1.070 | 1.040-1.100 |
| Absolute lymphocytes | 0.734 | 1.388 | 0.279 | 0.597 | 2.082 | 0.137-31.614 |
| TC | -0.242 | 0.195 | 1.532 | 0.216 | 0.785 | 0.536-1.151 |
| TG | -0.384 | 0.234 | 2.700 | 0.100 | 0.681 | 0.431-1.077 |
| LDL-C | -3.302 | 1.826 | 3.271 | 0.071 | 0.037 | 0.001-1.318 |
| HDL-C | -0.397 | 0.290 | 1.882 | 0.170 | 0.672 | 0.381-1.186 |
| MNA score | -0.405 | 0.086 | 22.357 | 0.000 | 0.667 | 0.564-0.789 |
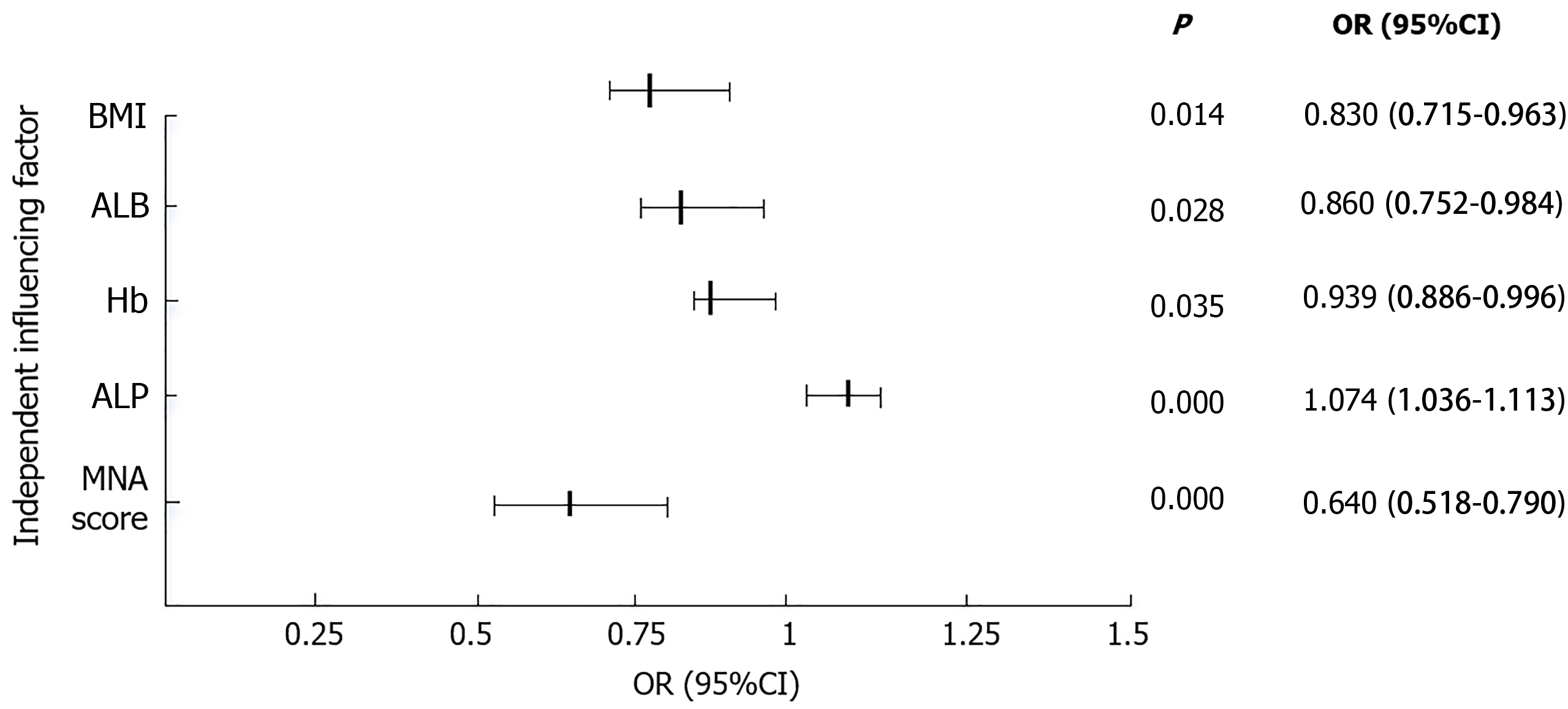
Whether cognitive impairment occurred in elderly hypertensive patients was assigned (occurred = 1, not occurred = 0), and BMI, ALB, Hb, ALP, and MNA scores were included as independent variables and assigned (Table 7), and multifactorial logistic analysis showed that BMI (OR = 0.830, P = 0.014), ALB (OR = 0.860, P = 0.028), Hb (OR = 0.939, P = 0.035), and MNA score (OR = 0.640, P = 0.000) were independent protective factors for the development of cognitive impairment in patients, and ALP (OR = 1.074, P = 0.000) was an independent risk factor for the development of cognitive impairment in patients (Figure 1).
| Considerations | β | SE | Wald χ2 | P value | OR | 95%CI |
| BMI | -0.187 | 0.076 | 6.041 | 0.014 | 0.830 | 0.715-0.963 |
| ALB | -0.151 | 0.069 | 4.805 | 0.028 | 0.860 | 0.752-0.984 |
| Hb | -0.063 | 0.030 | 4.432 | 0.035 | 0.939 | 0.886-0.996 |
| ALP | 0.071 | 0.018 | 14.927 | 0.000 | 1.074 | 1.036-1.113 |
| MNA score | -0.447 | 0.108 | 17.279 | 0.000 | 0.640 | 0.518-0.790 |
| Constant | 19.006 | 6.728 | 7.980 | 0.005 | 179488137.767 |
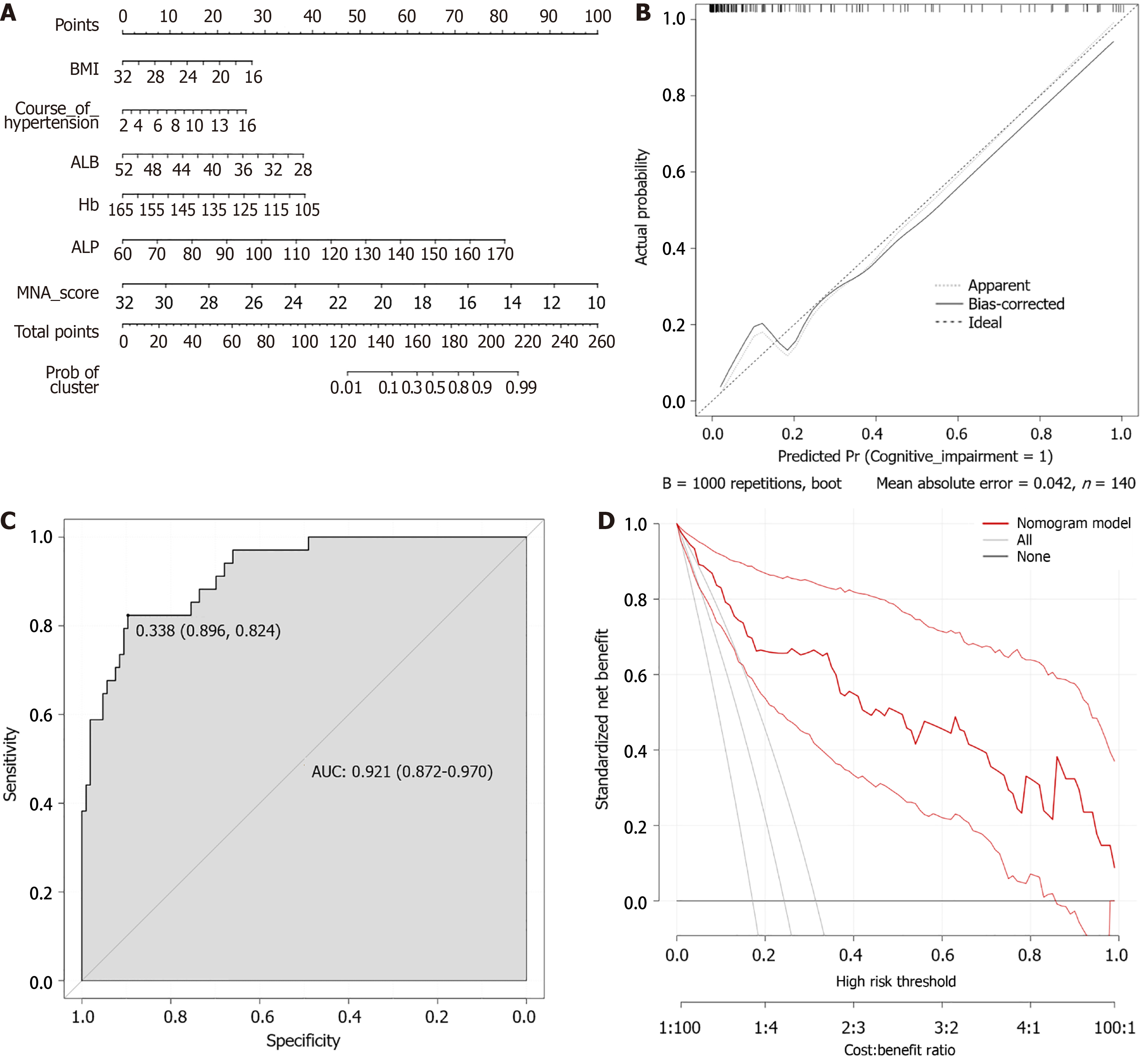
The five independent risk factors obtained (BMI, ALB, Hb, ALP, MNA score) were used to construct a prediction model by R software, and the column-line graph model was established (Figure 2A), and after the generated column-line graph calibration curve (Figure 2B), the consistency between predicted events and actual events was high. The area under the receiver operating characteristic (ROC) curve of the column-line diagram prediction model was 0.921 (95%CI: 0.872-0.970) (Figure 2C). The decision analysis curve is shown in Figure 2D, where the X-axis indicates the threshold probability, the Y-axis indicates the net return, and the black solid line indicates the net return using the column-line diagram prediction model, which shows a higher return and further confirms the effectiveness of the column-line diagram prediction model.
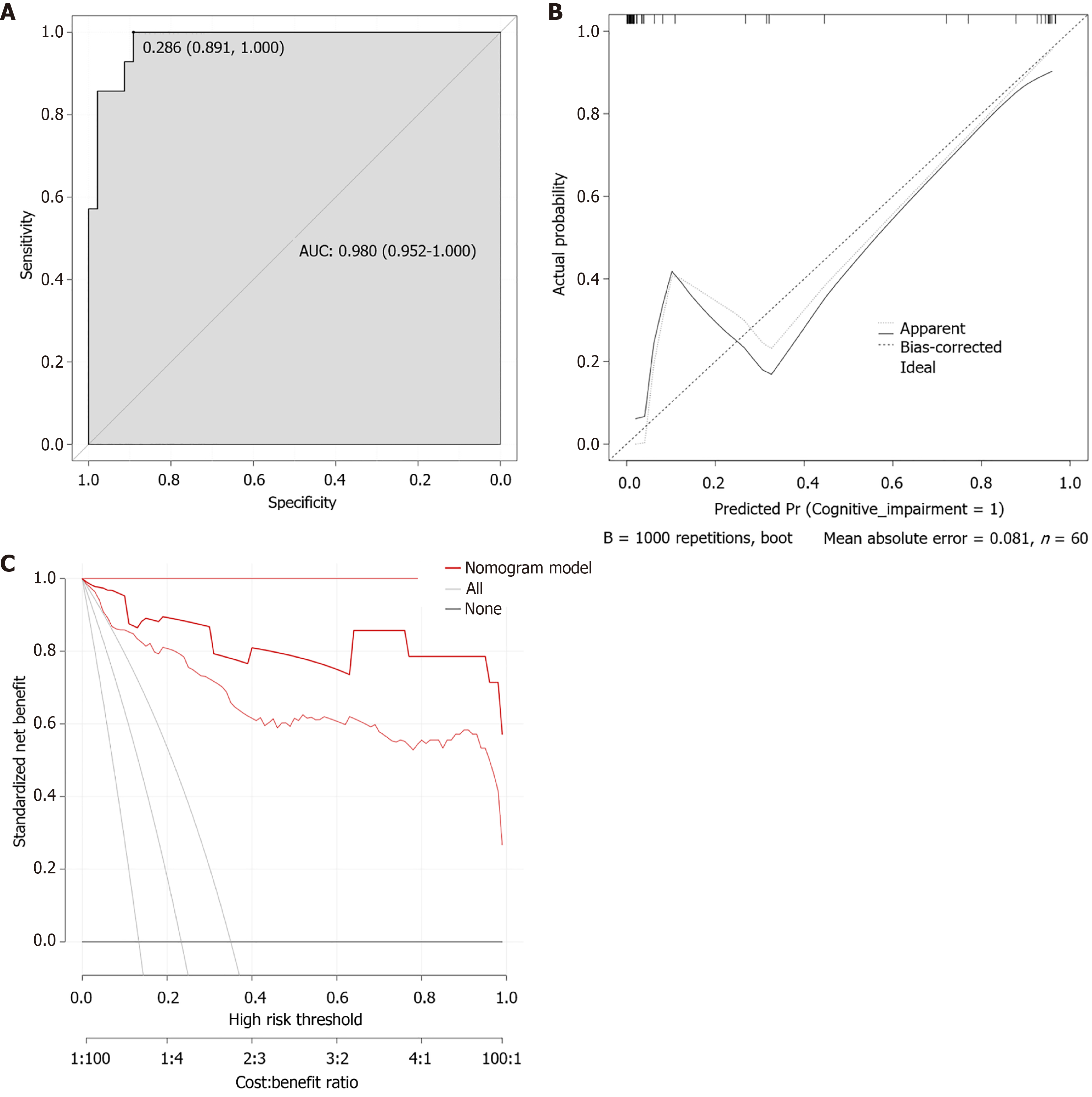
Based on the clinical data of the patients in the validation cohort (n = 36) (Table 8), the results of the Hosmer-Lemeshow test are: χ2= 1.813, and P = 0.986. The external validation of the column-line diagram of the risk of developing cognitive impairment was performed with a ROC curve, and the results showed that the lower product of the ROC curve was 0.980 (95%CI: 0.952-1.000) (Figure 3A). The slope of the generated calibration curve for the column-line diagram was close to 1 (Figure 3B), and the decision curve showed a high net gain for the model (Figure 3C), suggesting that the column-line diagram model was well calibrated in the validation group.
| Norm | Total cases | Hypertension combined with cognitive impairment group | Cognitively normal group with hypertension (n = 46) | t/χ2/F | P value |
| Distinguishing between the sexes | 0.034 | 0.854 | |||
| Male | 27 (45.00) | 6 (42.86) | 21 (45.65) | ||
| Women | 33 (55.00) | 8 (57.14) | 25 (54.35) | ||
| Age, year | 71.44 ± 7.23 | 72.81 ± 8.14 | 70.66 ± 7.15 | 9.540 | 0.344 |
| BMI, kg/m2 | 22.52 ± 2.61 | 20.12 ± 2.94 | 24.68 ± 2.41 | 5.885 | 0.000 |
| Family history of hypertension | 3.375 | 0.066 | |||
| Yes | 24 (40.00) | 7 (50.00) | 17 (36.96) | ||
| No | 36 (60.00) | 7 (50.00) | 29 (63.04) | ||
| Course of hypertension, year | 9.71 ± 3.10 | 10.29 ± 3.66 | 8.23 ± 3.14 | 2.068 | 0.043 |
| High blood pressure classification | 0.059 | 0.953 | |||
| Level 1 | 34 (56.67) | 8 (57.14) | 26 (56.52) | ||
| Level 2 | 16 (26.67) | 3 (18.75) | 13 (28.26) | ||
| Level 3 | 10 (16.67) | 3 (18.75) | 7 (15.22) | ||
| Drinking history | 0.066 | 0.798 | |||
| Yes | 21 (35.00) | 5 (35.71) | 16 (34.78) | ||
| No | 39 (65.00) | 9 (64.29) | 30 (65.22) | ||
| Smoking history | 0.034 | 0.854 | |||
| Yes | 33 (55.00) | 8 (57.14) | 25 (54.35) | ||
| No | 27 (45.00) | 6 (42.86) | 21 (45.65) | ||
| Defatted body weight, kg | 54.46 ± 8.59 | 54.65 ± 8.28 | 55.71 ± 9.12 | 0.389 | 0.699 |
| Upper arm muscle dimension, cm | 24.88 ± 3.11 | 25.01 ± 2.08 | 24.13 ± 2.35 | 1.257 | 0.214 |
| ALB, g/L | 37.76 ± 3.62 | 36.22 ± 3.58 | 39.71 ± 4.68 | 2.565 | 0.013 |
| Hb, g/L | 134.47 ± 11.57 | 123.27 ± 12.32 | 141.39 ± 11.27 | 5.156 | 0.000 |
| AST, U/L | 20.35 ± 3.31 | 20.21 ± 3.25 | 20.42 ± 3.40 | 0.204 | 0.839 |
| ALT, U/L | 17.87 ± 2.81 | 17.74 ± 2.81 | 17.91 ± 2.86 | 0.196 | 0.846 |
| Cr, mmol/L | 80.43 ± 20.22 | 82.11 ± 19.10 | 78.85 ± 22.14 | 0.497 | 0.621 |
| Hcy, mmol/L | 20.42 ± 7.31 | 22.92 ± 6.83 | 19.11 ± 7.38 | 1.719 | 0.091 |
| UA, μmol/L | 259.68 ± 65.97 | 252.63 ± 61.02 | 265.63 ± 75.27 | 0.589 | 0.558 |
| ALP, U/L | 119.57 ± 19.71 | 122.85 ± 18.57 | 102.41 ± 22.05 | 3.141 | 0.003 |
| Absolute lymphocytes | 1.49 ± 0.65 | 1.44 ± 0.62 | 1.53 ± 0.66 | 0.453 | 0.652 |
| TC, mmol/L | 3.94 ± 0.81 | 3.98 ± 0.78 | 4.14 ± 0.80 | 0.659 | 0.513 |
| TG, mmol/L | 1.30 ± 0.62 | 1.41 ± 0.59 | 1.32 ± 0.63 | 0.475 | 0.637 |
| LDL-C, mmol/L | 1.15 ± 0.46 | 1.16 ± 0.33 | 1.12 ± 0.35 | 0.379 | 0.706 |
| HDL-C, mmol/L | 2.45 ± 0.77 | 2.38 ± 0.82 | 2.47 ± 0.76 | 0.381 | 0.705 |
| MNA score, cent | 21.74 ± 3.08 | 17.92 ± 3.41 | 22.88 ± 2.61 | 5.785 | 0.000 |
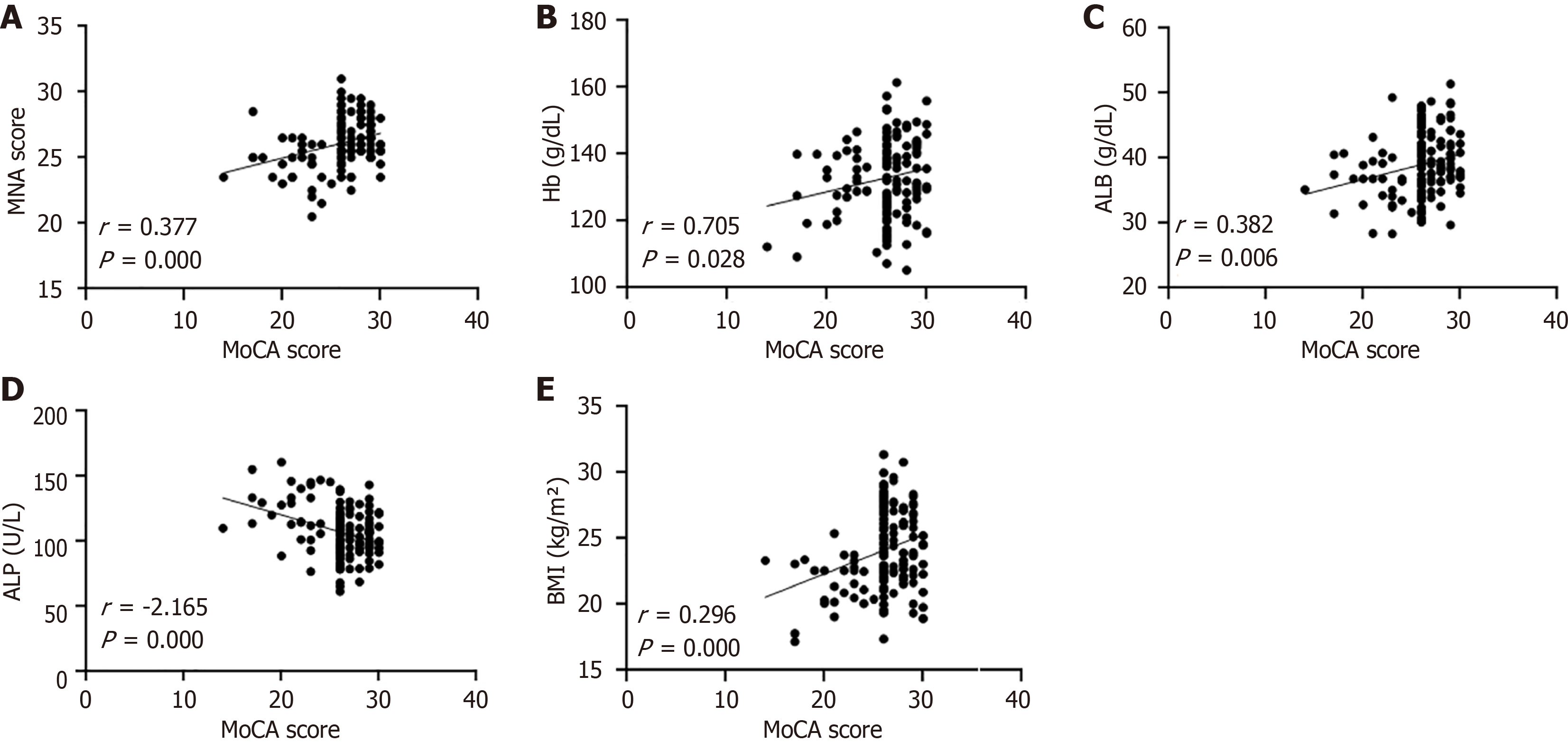
Pearson’s analysis found that ALP was negatively correlated with MoCA scores (r = -2.165, P = 0.000), and that BMI (r = 0.296, P = 0.000), MNA scores (r = 0.377, P = 0.000), Hb (r = 0.705, P = 0.028), ALB (r = 0.382, P = 0.006) were positively correlated with MoCA scores (Figure 4).
In elderly patients with hypertension, long-term elevated blood pressure will lead to chronic inflammation of tissues and organs including blood vessels, and further accelerate the metabolism of nutrients such as lipids and proteins, thus increasing the risk of malnutrition[11]. Malnutrition, on the other hand, will lead to insufficient blood supply to the brain, resulting in chronic ischemia of the brain[12], thus promoting cognitive impairment. There are many types of scales for determining cognitive impairment in clinical practice. For example, studies recommend the use of the Mini-Mental State Examination (MMSE) and MoCA scores to determine mild cognitive impairment. Among them, the MMSE scale is more suitable for screening for dementia, while the MoCA score is more suitable for screening for mild cognitive impairment. This study aims to construct a bar chart model to analyze the risk factors for cognitive decline in elderly hypertensive patients. This article selects the MoCA score as the basis for determining whether a patient has cognitive impairment.
In the present study, when comparing the indicators of patients with different nutritional statuses, it was found that there were significant differences between patients’ BMI, ALB, Hb and Hcy levels, of which BMI, ALB and Hb were related to the nutritional status of the patients, while Hcy levels were significantly higher in malnourished patients, which might be related to the lack of Hcy metabolizing cofactors folic acid and vitamin B12[13]. It has been pointed out in previous studies[14] that Hcy is involved in the process of dopaminergic neuronal transmission in the brain, affecting the synthesis, release, and reuptake of dopamine, which mediates the occurrence of psychiatric disorders such as depression and anxiety, and impairs patients’ cognitive function to a certain extent[15]. However, in this study, no significant difference in Hcy levels was observed in patients with different cognitive functions, which may be related to the small sample size selected for the study and the failure to reflect significant differences.
In the present study, it was found that MNA score and BMI were lower in patients with cognitive impairment, and MNA score and BMI were independent protective factors for the occurrence of cognitive impairment in elderly hypertensive patients, and both of them showed a positive correlation with MoCA score, which suggests that the worse the nutritional state of elderly hypertensive patients, the higher the risk of cognitive impairment, and this result is in line with the results of the previous related studies. The reason may be related to inflammation. In elderly patients with hypertension, interleukin-17 (IL-17) and other inflammatory factors mediated by T helper type 17/regulatory T cells interact with reactive oxygen species[16], which can then recruit macrophages, neutrophils and other inflammatory cells to infiltrate the blood vessel wall, thus leading to increased inflammation in the body. However, malnutrition in the body will reduce the binding level of leptin receptors and leptin on the surface of hematopoietic stem cells, thus affecting the synthesis and development of lymphocytes in the body, further promoting the disorder of the body’s immune system[17], aggravating the body’s inflammatory response, inducing cerebral vascular and brain tissue structure lesions in patients, and even causing cerebral cortex damage. Eventually, cognitive impairment occurs. At the same time, in the study of Nyulas KI et al[18], it was found that BMI was significantly different between hypertensive patients with or without cognitive dysfunction, which was also consistent with the results of this study.
Hb and ALB are commonly used indicators for evaluating the nutritional status of an organism. When the organism is in a state of malnutrition, the lack of serum iron and folic acid reduces the ability of the blood to synthesize Hb and decreases the level of Hb, whereas ALB is the main protein in the peripheral circulation synthesized by the liver, and is a marker of malnutrition in the organism. The reason was analyzed. In the state of low-level Hb, the oxygen transport function, brain tissue blood flow and oxygen supply decreased, leading to local brain hypoxia in the body, further inducing the production of hypoxia inducible factor-1α, and regulating the nuclear factor kappa B/nucleotide-binding domain, leucine-rich repeat, and pyrin domain-containing protein 3 signaling pathway, thereby up-regulating the expression of caspase-1. This will further aggravate brain tissue damage and lead to cognitive impairment[19]. Some studies have used Hb oxygen carrier to intervene in rats and found that Hb oxygen carrier can aggravate oxidative stress level in brain tissue by inhibiting the activity of the phosphatidylinositol 3-kinase/Akt signaling pathway in glial cells, inhibit iron death and alleviate brain injury in rats’ subarachnoid space[20], and this study also suggests the protective effect of Hb oxygen transport function on the body’s cognitive function. ALB is an antioxidant with mercaptan group, which can directly participate in scavenging free oxygen radicals in the body and reduce oxidative stress reactions[21]. At low levels of ALB, the body’s ability to clear oxygen free radicals is reduced, which leads to a large release of inflammatory factors such as tumor necrosis factor α (TNF-α) and IL-6, and further induces the activation of microglia under the action of the toll like receptor 4/nuclear factor kappa B signaling pathway[22], and promotes their transformation into M1 type. As a result, numerous inflammatory factors such as TNF-α, IL-6 and IL-1β are released, further aggravating brain damage[23], thus increasing the risk of cognitive impairment in patients.
ALP is a regulator of vascular calcification and is involved in the process of calcium and phosphorus metabolism in the body, and it rises significantly in the presence of inflammatory malnutrition in the body leading to excessive metabolism in the liver[24]. The results of the present study showed that ALP levels were higher in cognitively impaired patients and ALP was also an independent risk factor for the development of cognitive impairment in elderly hypertensive patients, and all of them were negatively correlated with the MoCA score. This suggests that malnutrition-induced high ALP expression increases brain damage, promoting cognitive impairment. Analyzing the reasons, the high expression of ALP in elderly hypertensive patients is involved in the aldosterone-mediated vascular calcification process. ALP can form atherosclerotic plaques by promoting the deposition of collagen in periventricular blood vessels. This will lead to thickening of the walls of cerebral veins and venules, reduced blood flow in brain tissue[25], and further lead to ischemic lesions such as white matter lesions in brain tissue, thereby affecting cognitive function. Previous studies have also pointed out that ALP is a risk factor for the development of ischemic cerebral white matter demyelination[26]. On the other hand, ALP can promote the metabolism of gamma-aminobutyric acid[27], thus weakening the transformation function of microglia into the M2 phenotype, and increasing the levels of TNF-α and IL-1β in brain tissue, thus aggravating the inflammatory response and apoptosis of brain tissue[28]. Li et al[29] found in their study that elevated serum ALP level is an independent predictor of the occurrence of cognitive dysfunction in patients, and the occurrence of cognitive dysfunction is related to inflammation. This further confirms the conclusion of this study. However, the MMSE scale used in the study by Li et al[29] to assess the cognitive function of patients is different from that used in this study. Therefore, the universality of the conclusion still needs to be further explored.
In order to clarify the predictive value of MNA score, Hb, ALB, and ALP levels in the occurrence of cognitive impairment in elderly hypertensive patients, the present study used a modeling cohort to establish a column line graph model for predicting cognitive impairment. The area under the ROC curve of the column line graph prediction model was larger, and the predictive efficacy was good. Furthermore, the subjects in the validation cohort predicted the probability of cognitive impairment in the elderly hypertensive patients in the validation group through the bar graph, suggesting that there is a certain predictive value. In addition, the factors of the model are all patients’ medical record data, which are easier to obtain and have higher clinical adaptability. In addition, the calibration curve of the validation cohort shows that the deviation between the actual outcome curve and the calibration curve is small, indicating that the consistency between the predicted events and the actual events is high. As can be seen from the validation cohort decision analysis curve, the decision analysis curve is located in the upper right corner, which usually indicates that the model has a high true positive rate and a low false positive rate, which means that this model has a certain degree of accuracy and reliability.
This study revealed the independent influencing factors of cognitive impairment in elderly hypertensive patients and established a bar chart prediction model. It also further explored the relationship between nutrition-related indicators, MNA scores, Hb, ALB, ALP levels and patients’ cognitive levels. However, it still has certain limitations. First, this study is a single-center, small sample study. Due to the differences in people in different places, it is not globally applicable. Second, the study selected the MoCA scale to evaluate the cognitive function of patients, and the patient’s knowledge level may also affect the results of this article. In addition, the patient’s education level may also have a certain impact on the scoring results, thereby interfering with the comprehensive assessment of the occurrence of cognitive impairment in elderly hypertensive patients. Future research needs to further expand the sample size and observation indicators to improve the experiment. In addition, the time span of the patients selected for the study was small, and the cognitive function of the patients was not evaluated, and the patients may have cognitive impairment in their subsequent life, so future studies need to continue to extend the observation time.
In summary, for elderly hypertensive patients, the independent risk factor for cognitive impairment is ALP, and the independent protective factors are BMI, MNA score, Hb, ALB, and ALP is negatively correlated with the MoCA score, and BMI, MNA score, Hb and ALB is positively correlated with the MoCA score, and all of them have good predictive value. This means that nutritional intervention is a potential management strategy to prevent cognitive impairment in elderly hypertensive patients. Therefore, in the prevention and treatment of cognitive impairment in elderly hypertensive patients, nutritional intervention should be combined with the indexes of BMI, Hb, ALB and ALP of patients, to maintain a good nutritional state of patients and reduce the brain damage caused by long-term hypertension to a maximum extent. Avoid the occurrence of cognitive impairment in patients. The successful implementation of this study is expected to provide more scientific basis for the health management of elderly patients with hypertension and provide reference for the formulation of public health policies.
| 1. | Wang JG, Zhang W, Li Y, Liu L. Hypertension in China: epidemiology and treatment initiatives. Nat Rev Cardiol. 2023;20:531-545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 122] [Article Influence: 40.7] [Reference Citation Analysis (0)] |
| 2. | Ott C, Schmieder RE. Diagnosis and treatment of arterial hypertension 2021. Kidney Int. 2022;101:36-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 71] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 3. | Santisteban MM, Iadecola C, Carnevale D. Hypertension, Neurovascular Dysfunction, and Cognitive Impairment. Hypertension. 2023;80:22-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 193] [Article Influence: 64.3] [Reference Citation Analysis (0)] |
| 4. | Norman K, Haß U, Pirlich M. Malnutrition in Older Adults-Recent Advances and Remaining Challenges. Nutrients. 2021;13:2764. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 478] [Article Influence: 95.6] [Reference Citation Analysis (0)] |
| 5. | Di Vincenzo O, Luisi MLE, Alicante P, Ballarin G, Biffi B, Gheri CF, Scalfi L. The Assessment of the Risk of Malnutrition (Undernutrition) in Stroke Patients. Nutrients. 2023;15:683. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 58] [Reference Citation Analysis (0)] |
| 6. | Shestopalov AE, Yakovleva AV, Yadgarov MY, Sergeev IV, Kuzovlev AN. Prevalence and Impact of Malnutrition Risk on Outcomes in Critically Ill Patients with Traumatic Brain Injury and Stroke: A Retrospective Cohort Study Using Electronic Health Records. Nutrients. 2024;16:2396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 7. | Umemura S, Arima H, Arima S, Asayama K, Dohi Y, Hirooka Y, Horio T, Hoshide S, Ikeda S, Ishimitsu T, Ito M, Ito S, Iwashima Y, Kai H, Kamide K, Kanno Y, Kashihara N, Kawano Y, Kikuchi T, Kitamura K, Kitazono T, Kohara K, Kudo M, Kumagai H, Matsumura K, Matsuura H, Miura K, Mukoyama M, Nakamura S, Ohkubo T, Ohya Y, Okura T, Rakugi H, Saitoh S, Shibata H, Shimosawa T, Suzuki H, Takahashi S, Tamura K, Tomiyama H, Tsuchihashi T, Ueda S, Uehara Y, Urata H, Hirawa N. The Japanese Society of Hypertension Guidelines for the Management of Hypertension (JSH 2019). Hypertens Res. 2019;42:1235-1481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1594] [Cited by in RCA: 1445] [Article Influence: 206.4] [Reference Citation Analysis (0)] |
| 8. | Vellas B, Guigoz Y, Garry PJ, Nourhashemi F, Bennahum D, Lauque S, Albarede JL. The Mini Nutritional Assessment (MNA) and its use in grading the nutritional state of elderly patients. Nutrition. 1999;15:116-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1149] [Cited by in RCA: 1397] [Article Influence: 51.7] [Reference Citation Analysis (4)] |
| 9. | Nasreddine ZS, Phillips NA, Bédirian V, Charbonneau S, Whitehead V, Collin I, Cummings JL, Chertkow H. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53:695-699. [PubMed] [DOI] [Full Text] |
| 10. | Angeli F, Verdecchia P, Reboldi G. Prognostic impact of hypertension grading. Eur J Intern Med. 2024;126:83-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 11. | Patterson GT, Manthi D, Osuna F, Muia A, Olack B, Mbuchi M, Saldarriaga OA, Ouma L, Inziani M, Yu X, Otieno P, Melby PC. Environmental, Metabolic, and Inflammatory Factors Converge in the Pathogenesis of Moderate Acute Malnutrition in Children: An Observational Cohort Study. Am J Trop Med Hyg. 2021;104:1877-1888. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 12. | Kanda D, Ikeda Y, Sonoda T, Tokushige A, Kosedo I, Yoshino S, Takumi T, Ohishi M. Malnutrition and Clopidogrel Non-Use Worsen Prognosis of Critical Limb Ischemia Patients After Revascularization. Circ Rep. 2019;2:121-127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 13. | Ulloque-Badaracco JR, Hernandez-Bustamante EA, Alarcon-Braga EA, Al-Kassab-Córdova A, Cabrera-Guzmán JC, Herrera-Añazco P, Benites-Zapata VA. Vitamin B12, folate, and homocysteine in metabolic syndrome: a systematic review and meta-analysis. Front Endocrinol (Lausanne). 2023;14:1221259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 38] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 14. | Wang H, Du YS, Xu WS, Li CJ, Sun H, Hu KR, Hu YZ, Yu TJ, Guo HM, Xie L, Wang GJ, Liang Y. Exogenous glutathione exerts a therapeutic effect in ischemic stroke rats by interacting with intrastriatal dopamine. Acta Pharmacol Sin. 2022;43:541-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 15. | Gerasimova E, Burkhanova G, Chernova K, Zakharov A, Enikeev D, Khaertdinov N, Giniatullin R, Sitdikova G. Hyperhomocysteinemia increases susceptibility to cortical spreading depression associated with photophobia, mechanical allodynia, and anxiety in rats. Behav Brain Res. 2021;409:113324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 16. | Oh S, Shim M, Son M, Jang JT, Son KH, Byun K. Attenuating Effects of Dieckol on Endothelial Cell Dysfunction via Modulation of Th17/Treg Balance in the Intestine and Aorta of Spontaneously Hypertensive Rats. Antioxidants (Basel). 2021;10:298. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 17. | Lemos JP, Tenório LPG, Mouly V, Butler-Browne G, Mendes-da-Cruz DA, Savino W, Smeriglio P. T cell biology in neuromuscular disorders: a focus on Duchenne Muscular Dystrophy and Amyotrophic Lateral Sclerosis. Front Immunol. 2023;14:1202834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 18. | Nyulas KI, Germán-Salló M, Fazakas Z, Preg Z, Pál T, Pál S, Tripon RG, Cseh MJ, Simon-Szabó Z, Arbănași EM, Nemes-Nagy E. Relationship between Nutrition, Lifestyle Habits and Laboratory Parameters in Hypertensive Patients with/without Cognitive Dysfunction. Life (Basel). 2023;13:311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 19. | Teng JF, Mei QB, Zhou XG, Tang Y, Xiong R, Qiu WQ, Pan R, Law BY, Wong VK, Yu CL, Long HA, Xiao XL, Zhang F, Wu JM, Qin DL, Wu AG. Polyphyllin VI Induces Caspase-1-Mediated Pyroptosis via the Induction of ROS/NF-κB/NLRP3/GSDMD Signal Axis in Non-Small Cell Lung Cancer. Cancers (Basel). 2020;12:193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 255] [Cited by in RCA: 276] [Article Influence: 46.0] [Reference Citation Analysis (0)] |
| 20. | Yuan Z, Zhou X, Zou Y, Zhang B, Jian Y, Wu Q, Chen S, Zhang X. Hypoxia Aggravates Neuron Ferroptosis in Early Brain Injury Following Subarachnoid Hemorrhage via NCOA4-Meditated Ferritinophagy. Antioxidants (Basel). 2023;12:2097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 21. | Tabata F, Wada Y, Kawakami S, Miyaji K. Serum Albumin Redox States: More Than Oxidative Stress Biomarker. Antioxidants (Basel). 2021;10:503. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 90] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 22. | Li R, Zhou Y, Zhang S, Li J, Zheng Y, Fan X. The natural (poly)phenols as modulators of microglia polarization via TLR4/NF-κB pathway exert anti-inflammatory activity in ischemic stroke. Eur J Pharmacol. 2022;914:174660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 87] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 23. | Rahimian R, Wakid M, O'Leary LA, Mechawar N. The emerging tale of microglia in psychiatric disorders. Neurosci Biobehav Rev. 2021;131:1-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 24. | Das S, Meena RK, Shah D, Almeida EA, Mehndiratta M, Gupta P. Children with severe acute malnutrition have high rates of bone resorption: A cross-sectional comparative study. Nutr Res. 2023;117:66-72. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 25. | Zhang X, Zhou X, Huang Z, Fan X, Tan X, Lu C, Yang J. Aldosterone is a possible new stimulating factor for promoting vascular calcification. Front Biosci (Landmark Ed). 2021;26:1052-1063. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 26. | van Wageningen TA, Gerrits E, Brouwer N, Brevé JJP, Geurts JJG, Eggen BJL, Boddeke HWGME, van Dam AM. Distinct gene expression in demyelinated white and grey matter areas of patients with multiple sclerosis. Brain Commun. 2022;4:fcac005. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 27. | Li H, Yang M. Ligustrazine activate the PPAR-γ pathway and play a protective role in vascular calcification. Vascular. 2022;30:1224-1231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 28. | Yang G, Liu K, Ma S, Qi P. PPARγ inhibition promotes osteogenic differentiation of bone marrow mesenchymal stem cells and fracture healing. J Cell Biochem. 2024;125:e30568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 29. | Li X, Zhao L, Shi H, Wang H, Wang X, Wang L. Elevated serum alkaline phosphatase correlates with postoperative cognitive dysfunction: A retrospective cohort study based on STROBE statement. Medicine (Baltimore). 2022;101:e31530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/