INTRODUCTION
Behavioral disorders are amongst the leading causes of disability and their characteristic features including early age of onset as well as high prevalence among the working-age population are an influential determinant of their negative socio-economic impact[1,2]. The severe mental illnesses, such as schizophrenia and mood disorders demonstrate a high comorbidity rate with somatic diseases as well as with other psychiatric conditions, which in addition to the significant risk for self-inflicted death, marks a high mortality rate[3].
Unlike other branches of medicine, where the clinician can rely on objective measures (blood tests, imaging methods, etc.), in psychiatry, diagnosis is mediated by subjectivism, which affects the reliability of the assessment methods used. Consequently, there is a frequent change in psychotropic drugs which leads to increased risk of drug-resistant disorders and relapse[4]. Тhe integration of neuroimaging evidence with comprehension of the role of brain networks and their abnormal interactions as a pathogenic mechanism for the development of certain behavioral deviance is an important step toward a systematic biological understanding of mental disorders.
Establishing a diagnosis and treating these disorders is challenging. Numerous molecules are assumed to have the appropriate characteristics to induce improvement; however, therapeutic outcome and adverse side effects differ significantly among patients[5]. With conventional treatments (psychopharmacology and psychotherapy), a large percentage of patients report inadequate symptom resolution, and relapse rates remain high[6,7]. A growing amount of interest has been centered on focal neuromodulation in the search for better treatments. The interest in this field is driven by the latest data considering neuroanatomical models of modulation of mood, thought, perception, and behavior, as well as more sophisticated techniques for direct and focal modification of the neural function.
In the current review we will discuss new perspectives of the translational approach in the diagnosis and treatment of severe mental disorders, namely schizophrenia and mood disorders. Although genetic risk factors and pharmacogenomics in the field of psychiatry are promising strategies to predict or mitigate the therapeutic response of medication, and thus to inform medication selection and dosing decisions, the focus in this review will be on the adaptation of new transdisciplinary diagnostic methods such as neuroimaging and concurrently administered psychopathological questionnaires and the implementation of the results into treatment using various advanced biomedical treatment methods like electro-convulsive therapy (ECT), transcranial magnetic stimulation (TMS), transcranial direct current stimulation (tDCS) and deep brain stimulation (DBS).
PSYCHIATRIC DIAGNOSTICS IN THE 21ST CENTURY
One of the most widely discussed problems in psychiatry seems to be the fact that the diagnostic process is primarily focused on patients’ reports, behavioral observation and the capacity to draw conclusions about the real inner nature of patients’ experiences, rather than on accurate objective biomarkers[8].
The primary diagnostic tools in contemporary psychiatry are the International Classification of Diseases[9] and the Diagnostic and Statistical Manual of Mental Disorders[10], yet their foundational criterial categorization lacks biological validity[11,12]. This phenomenon is illustrated by existing barriers, such as heterogeneity, comorbidity, and ambiguous distinctions between healthy and abnormal behavior, which impede the diagnostic process. Another significant concern is that mental illnesses are categorized purely on clinical manifestations, without consideration for their etiology. When the conventional diagnostic labels themselves are not absolutely accurate, it is difficult to recognize a biological marker for a specific disorder. For this purpose, the National Institute of Mental Health has introduced the Research Domain Criteria Project[13,14], which seeks to extend the understanding of brain-behavior correlations and eventually incorporate this knowledge into clinical practice and enhance the development of more successful treatments. Innovative brain imaging technology provides the possibility to discover disease-specific anatomical and functional neural impairments.
Neuroimaging techniques such as magnetic resonance imaging (MRI), positron emission tomography, single-photon emission computed tomography, magnetoencephalography and electroencephalography provide resources for a non-invasive analysis of the brain structure and function associated with mental diseases with excellent precision[15]. Understanding the complex mechanisms of communication between different brain regions at rest and in the conditional performance of tasks is a promising field for studying the etiological mechanisms of severe mental illnesses that lead to personal and social decline. For example, functional MRI (fMRI) used in schizophrenia research was able to detect anatomical and functional aberrations of brain plasticity and connectivity, which clinically manifest as cognitive disturbances[16]. Another example of an accessible translational method is pharmacological fMRI[17], which facilitates the study of the effects of different pharmacological agents on the central nervous system (CNS) and the search for accurate neuroimaging biomarkers[18].
TRANSLATIONAL APPROACH
The concept of translation can be defined as a mechanism for improving health status, thus reducing mortality and morbidity rates among patients by incorporating scientific findings from research into clinical applications[19]. The translational approach in psychiatry encompasses a wide range of scientific disciplines and innovative techniques. MRI techniques such as quantitative structural imaging, voxel-based neuromorphometry, functional neuroimaging and spectroscopy are widely used in psychiatric research. These imaging tools, together with the knowledge gained regarding the underlying neurochemical dysfunctions have the potential to change the current state of "crisis" in psychiatry[20,21] through the use of evidence-based biomarkers and insight into the etiology of mental disorders.
Disruptions in communication within and between brain networks and the pathological processes involving their nodes are a promising discovery of translational neuroscience towards gaining a biological understanding of mental disorders and defining them as impaired connectivity disorders. Low-frequency variations in random brain functions can be localized by resting state fMRI (rs-fMRI), representing a common instrument for macro-scale functional connectomics to characterize inter-individual discrepancies in brain activity and mind-brain interactions observed in various mental disorders[22]. These metrics have a tremendous potential for accelerating the discovery of biomarkers for different mental disorders, which need first and foremost reliability and reproducibility to be addressed.
In this sense, critical questions are whether the progress in this field has succeeded in contributing to the development of more effective therapeutic methods, and whether biomarkers could be incorporated into everyday practice. However, despite years of efforts in this area, the results remain inconsistent[23]. Over the past decades, substantial advancement has been made in recognizing and understanding the fundamental neurobiological correlations of psychiatric disorders, such as schizophrenia spectrum disorders and mood disorders, but the effects of these advancements have not yet been established in clinical practice[24-26]. This may be partly due to the specificity of the design of the methods used. A common practice in fMRI studies is to conduct a clinical assessment before and after the scanning. Yet, in cases such as examining patients with bipolar disorder with rapid cycling, which presents with mood shifts from depression to mania within hours for example, this could affect the accuracy of the findings. To address this issue, fMRI gathered neuroimaging data, together with the parallel implementation of a self-assessment scale[27], could construct a scientifically valid instrument that can be used by clinicians in daily practice with trust in the reliability of the method. This initiative might positively affect the diagnostics, prevention, treatment, as well as facilitate the decision regarding the choice of the appropriate therapeutic method.
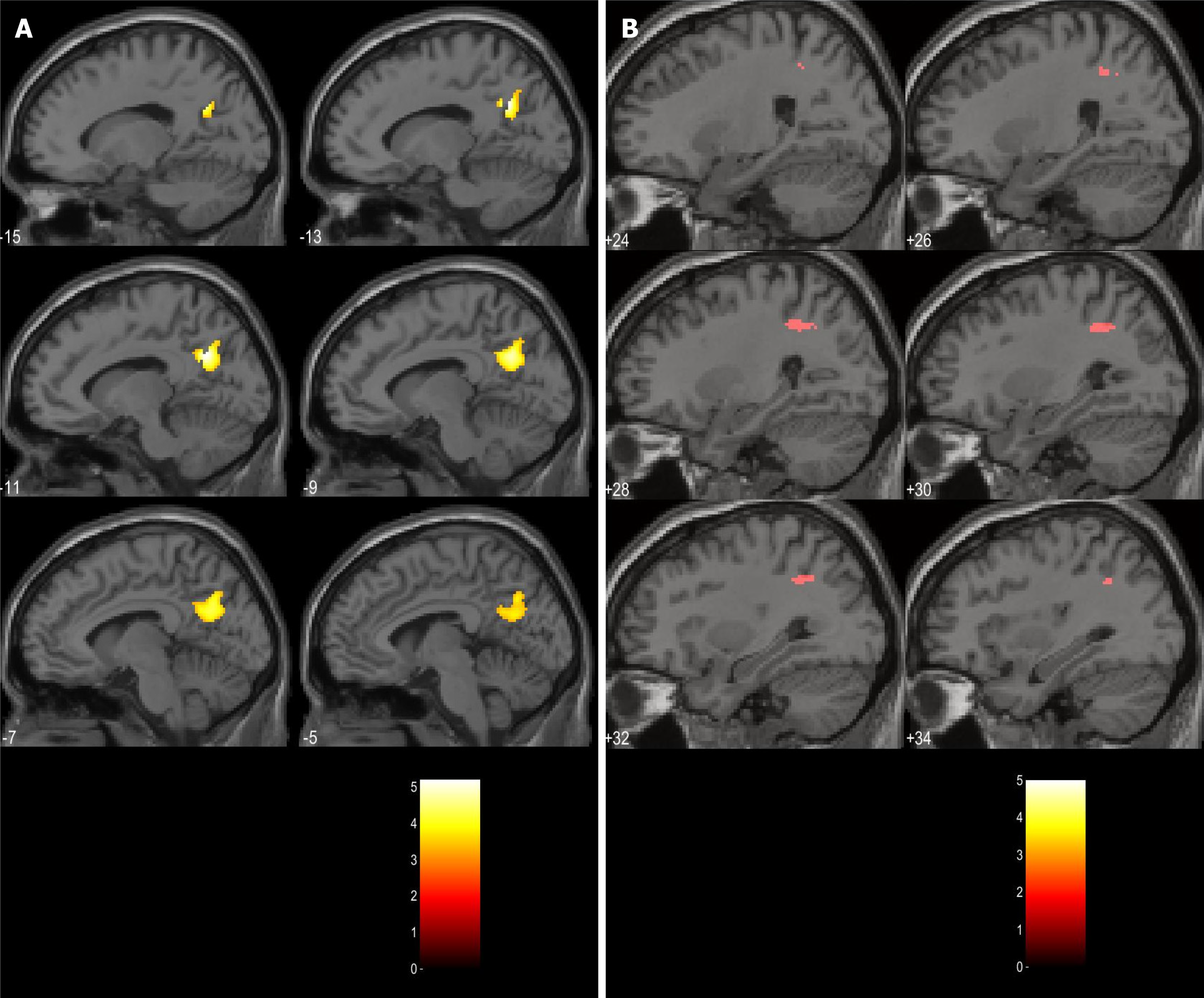
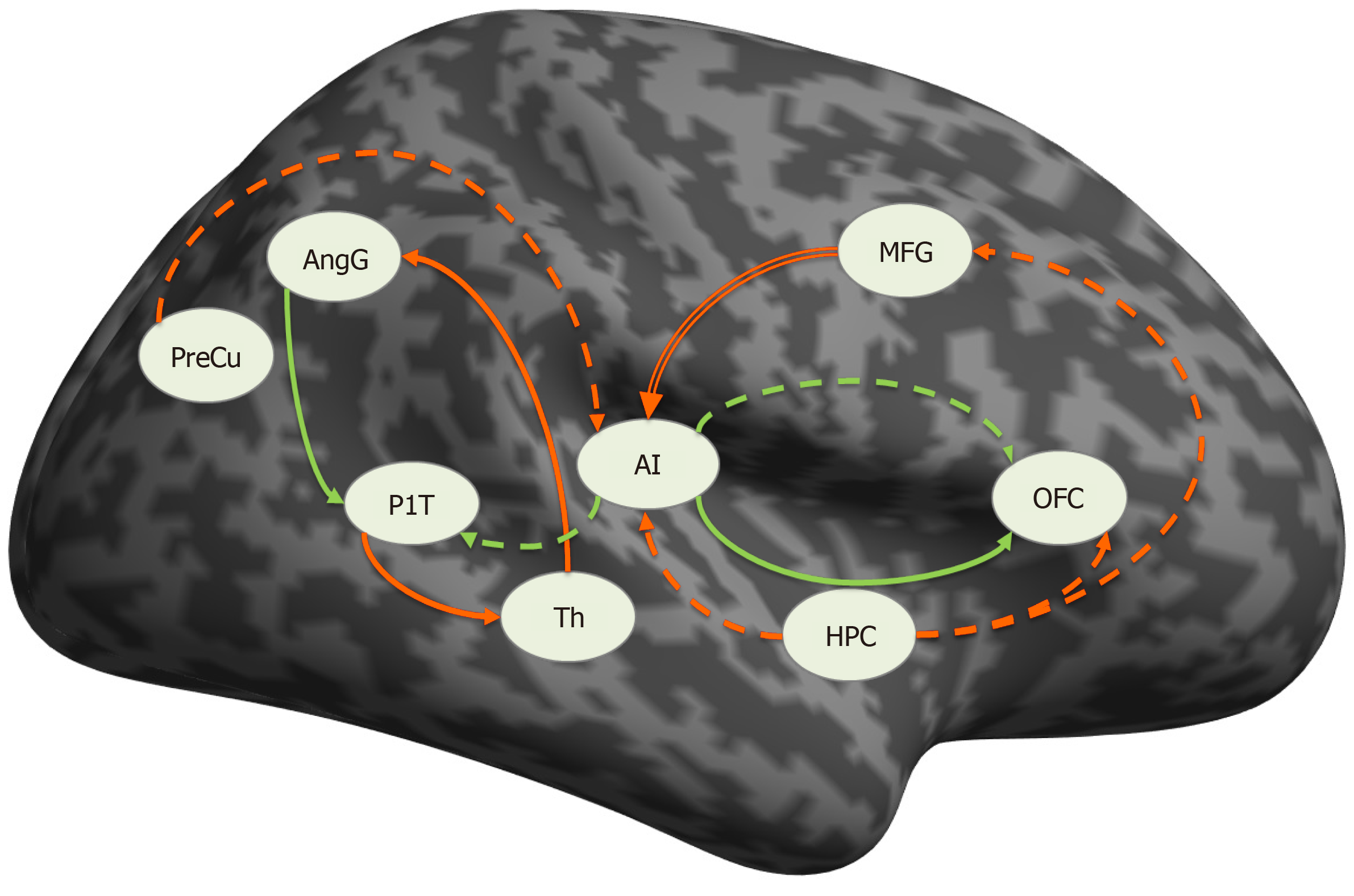
With the aim of implementing the translational model in psychiatry, our research group is concentrating on developing a new paradigm that combines a clinical self-assessment scale (von Zerssen and Koeller[28]'s paranoid-depressive scale) applied simultaneously with the acquisition of fMRI in two groups of patients—with a paranoid syndrome in the context of schizophrenia and with a depressive syndrome in the context of recurrent depressive disorder or bipolar disorder[29-33]. The goal is to compare the differences in brain activation during the task-related fMRI (tr-fMRI) with structural and functional changes in connectivity at rest with the hypothesis that the zones which demonstrate differential activation may have aberrant effective connectivity patterns and structural abnormalities. The results of the study can be summarized as follows: (1) In terms of grey matter (GM) volume alterations, the comparison at a whole brain level, voxel by voxel, yielded no significant structural differences between the two groups; (2) The paranoid group was associated with significantly more robust activations in the default mode network (DMN) structures—left precuneus (PreCu), left posterior cingulate cortex (PCC), and angular gyrus (AngG) during the processing of paranoid items from the scale (Figure 1); and (3) There was an aberrant connectivity pattern in the paranoid group—abnormal inhibitory connection from the prefrontal cortex (PFC) to the anterior insula (aI) (Figure 2).
Figure 1 Clusters of activations significantly higher in schizophrenia.
A: Left precuneus; B: Right posterior parietal lobule. Citation: Stoyanov D, Aryutova K, Kandilarova S, Paunova R, Arabadzhiev Z, Todeva-Radneva A, Kostianev S, Borgwardt S. Diagnostic Task Specific Activations in Functional MRI and Aberrant Connectivity of Insula with Middle Frontal Gyrus Can Inform the Differential Diagnosis of Psychosis. Diagnostics (Basel) 2021; 11(1): 95. Copyright ©The Author(s) 2021. Published by MDPI[32].
Figure 2 Connections significantly different from zero.
Solid line: schizophrenia, dashed line: in depression, green: excitatory, red: inhibitory, double red line: significantly different between the groups. PreCu: Precuneus; HPC: Hippocampus; AI: Anterior insula; AngG: Angular gyrus; OFC: Orbitofrontal cortex; PlT: Planum temporale; Th: Thalamus; MFG: Middle frontal gyrus. Citation: Stoyanov D, Aryutova K, Kandilarova S, Paunova R, Arabadzhiev Z, Todeva-Radneva A, Kostianev S, Borgwardt S. Diagnostic Task Specific Activations in Functional MRI and Aberrant Connectivity of Insula with Middle Frontal Gyrus Can Inform the Differential Diagnosis of Psychosis. Diagnostics (Basel) 2021; 11(1): 95. Copyright ©The Author(s) 2021. Published by MDPI[32].
We suggest that the observed activations of the PreCu, PCC, and AngG (DMN components) may be indirect evidence of the inhibitory influence from PFC⇒aI, which interferes with the balancing function of the insula as a dynamic switch in the DMN and fronto-parietal network (FPN)[32]. Impaired aI⇒DMN and aI⇒FPN connections lead to hyperactivity of DMN[34], which could explain social withdrawal and autistic behavior observed in patients with schizophrenia. In addition, the identified inhibitory effective connectivity from PFC⇒aI offers new insights into how the PFC dysfunction may contribute to cognitive impairment[35-37], behavioral disorganization[38], and functional decline observed in patients with schizophrenia. We propose a pathophysiological model in which cognitive deficits are due to the inability of the CNS to initiate and maintain an organized fronto-insular network[36].
On the other hand, we raise the question of whether and to what extent the prediction of clinical diagnostic classes can be improved by superimposing different dimensions of MRI (structural, tr-fMRI and rs-fMRI) compared to a single modality by application of a multivariate linear model (MLM)[36]. Using MLM and principal component analysis, we have made progress towards distinguishing the two psychiatric populations. The dopamine-ergic and noradrenaline-ergic pathways with DMN nodes were reported to be significant. The regions involved [planum polare (PP), inferior frontal gyrus (IFG), insula, nucleus accumbens, etc.] have both structural and functional characteristics that can distinguish the two groups. When focusing on the discriminatory power of the functional modalities, the predominant regions are a part of the effort mode network, DMN, and subcortical areas with mainly glutamate-ergic and GABA-ergic neurotransmission. The most significant structure in our MLM study is PP, which is а part of the superior temporal gyrus (STG) and is not only involved in auditory and linguistic processing but is also a critical construct for cognitive processes. Neuroimaging studies show that subjects with schizophrenia have structural abnormalities in the STG[39], suggesting that it plays a key role in the etiology of auditory perceptual disturbances and disorganization of thoughts[40]. There is evidence that auditory hallucinations are associated with a functional network of brain areas, namely the auditory and linguistic areas of the STG and IPG, as well as speech motor areas in the IFG[41-43], which are also significant regions in our study. Those findings support the abovementioned aim to develop a translationally valid instrument in the diagnostic process of schizophrenia and depression.
TREATMENT IMPLICATION OF THE TRANSLATIONAL APPROACH
Considering the controversial efficacy of the conventional treatments (psychopharmacology and psychotherapy), a large percentage of patients do not achieve complete remission (i.e., become asymptomatic) or retain symptomatic improvement. This lack of efficacy has led to the quest for alternative solutions, which involve the utilization of more invasive interventions for treatment-resistant patients with psychiatric illnesses[44]. Therefore, the focus has been on neuromodulation, i.e., modulation of the connections between various pathways and neurotransmitter systems in the brain. This emphasis has been driven by enhanced neuroanatomical concepts of mood, thought, and behavioral control, as well as more techniques to modify neural activity directly and focally.
ECT
ECT was first performed by a team of physicians led by Ugo Cerletti and Lucio Bini in 1938[45,46], along with Accornero[47] who contributed to the development of what they called electroshock[48] leading to the replacement of less effective biological interventions such as deep sleep therapy[49] and insulin shock therapy[50]. ECT has come a long way in refining the method and is still used today in cases of severe major depressive disorder (MDD), mania, and catatonia as a safe and effective treatment, despite the numerous critiques. ECT is an ideal example of translational neuroscience, involving the application of an instrumental method that generates electricity acting at a neurobiological level and modulating neuronal plasticity and brain connectivity. In the macro- and micro-environment of the brain, ECT induces numerous neurophysiological as well as neurochemical changes. It has been suggested that changes in gene expression, functional connectivity, neurotransmission, membrane permeability, and immune system function are responsible for the therapeutic effects of ECT[51]. Neuroimaging studies show that the GM volume of key regions like the amygdala, hippocampus, and anterior cingulate cortex (ACC) is decreased in patients with depression and increases following administration of ECT[52]. It remains an unresolved issue as to whether ECT efficacy in various psychiatric conditions (depression, mania, catatonia, delirium, and psychosis) is due to common neurobiological features shared by those disorders or to a single mechanism of action[53]. A mechanism that has recently been suggested is that the seizures triggered by ECT may induce a significant alteration in the sleep structure[54].
We suggest that this and other hypotheses concerning neuroplasticity can be tested by our paradigm as it can be used as a tool to monitor the therapeutic effect of ECT by comparing the results before and after the procedure, considering the fact that the von Zerssen and Koeller[28]’s self-assessment scale was primarily designed for monitoring the treatment response.
TMS
TMS was developed in 1985 as a non-invasive method to stimulate the cerebral cortex[55]. A strong magnetic field is produced by a TMS device, which induces an electrical current in a specific area. This is a method based on the principle of electromagnetic induction, which can have a different effect on the targeted areas depending on the stimulation parameters and the physiological activity of the areas. Low frequencies around and below 1 Hz (low frequency TMS) have been found to suppress the excitability of the stimulated cortical zone and functionally related structures, while those above 1-3 Hz (high frequency TMS) lead to its activation[56].
Unlike ECT, the administration of TMS requires no anesthesia and therefore patients are awake and alert during the procedure and it is often performed in an out-patient setting[57]. The short-term effect of the procedure is prolonged by applying a series of stimuli with a certain frequency, the so-called repetitive TMS (rTMS). TMS affects the underlying brain structures by altering the regional activity in the underlying cortical structures. Such changes have also been found in brain areas distant from the site of stimulation, proving that the effect of rTMS propagates in other remote parts of the brain, and these changes are functionally significant and take place on existing neural networks[58].
Depressive disorders are the most studied area of therapeutic use of TMS. Recently there has been an increasing interest in the management of schizophrenia as research results indicate that the negative symptoms of schizophrenia appear to be attributed to PFC hypoactivity, whereas positive symptoms appear to be correlated with left temporo-parietal cortex hyperactivity[59]. It has also been hypothesized that auditory hallucinations may be attributed to aberrant activation of language perception zones (i.e., Wernicke's area) in the left temporo-parietal cortex[60]. It is therefore proposed that the use of high frequency TMS in the left PFC may reduce negative symptoms of schizophrenia, whereas the application of low-frequency TMS to the left temporo-parietal cortex may reduce the positive symptoms[60,61].
Considering the fact that all of those structures appear to be significant in our MLM study, we support the abovementioned model for applying rTMS in individuals suffering from schizophrenia. Moreover, our finding of the effective connectivity aberration between the PFC and the insula provides further insight on the etiopathophysiology of schizophrenia and the role of the disruption of the salience and executive systems processing due to the insular dysfunction because of its failure as a dynamic switch between the DMN, FPN and salience network (SN). Due to its deep location, the insula is considered to be beyond the reach of rTMS. However, we suggest that the insula may still be indirectly modulated by the identified abnormal connectivity from PFC to aI by focusing on PFC, the localization of which makes it accessible for stimulation.
Despite the promising applications of TMS, in about 30% of cases the standard procedure misses the target area, as it only allows stimulation of focal areas just below the skull[62,63]. Deep TMS (dTMS) has been implemented to overcome these drawbacks. dTMS offers all the advantages of conventional TMS with the benefit of stimulating deeper brain zones with lower focal distribution of the electric field[64]. Some studies have been conducted on the use of conventional TMS for the treatment of auditory hallucinations in drug-resistant patients with schizophrenia, which have shown that the method is moderately effective[65,66]. In this sense, we propose inhibitory dTMS as a therapeutic intervention targeting temporal, frontal, and parietal areas, taking into account data from functional neuroimaging which established that hallucinations are accompanied by transient hyperactivity in specific zones—STG, PFC, ACC, inferior parietal cortex, etc.[67] and dTMS may be even more effective than conventional TMS due to greater stimulus depth. In addition, dTMS can even be used as a method to modulate the function of the insula directly, unlike standard TMS, in which this area is inaccessible and can only be reached indirectly. Restoring the function of the insula and the networks in which it participates will further improve the function of the SN as a dynamic switch between DMN and FPN. Finally, our paradigm in tr-fMRI can also be applied as a tool for monitoring TMS, with good enough accuracy and reliability of the method.
tDCS
TDCS is a neuromodulatory procedure that facilitates or inhibits spontaneous neuronal activity by delivering low-intensity direct current to cortical areas via scalp electrodes. Over the past decade, the physiological mechanisms of action of tDCS have been studied to support the investigation of its clinical neuropsychiatry applications. TDCS was developed as a treatment method for brain injuries and neuropsychiatric disorders, especially MDD. Given the absence of a triggered seizure, it is hypothesized that this technique operates by plastic effects exerted by endogenous firing potentiation[68].
A variety of studies have found that in healthy individuals, anodal tDCS to dorsolateral PFC (DLPFC) leads to increased cognitive performance[69]. In the treatment of MDD, the anode is located over the left DLPFC, which is hypoactive, helping to improve local activation and regain regular functioning[70]. The cathode is located either over the right DLPFC or the right supraorbital or frontotemporal area, leading to distinct current distributions. In addition, after anodal DLPFC tDCS in patients with schizophrenia, enhanced results on working memory tasks[71], probabilistic learning tasks[72], adaptive control tasks[73], and attention-vigilance tasks[74] have been demonstrated. A consistent feature of these diverse tasks is that they all depend, in part, on cognitive control mechanisms such as goal maintenance mediated by DLPFC.
We suggest a model in which the tDCS, instead of focusing on a single cortical area, could simultaneously target several zones that are part of a common network. Considering the results of our imaging studies, they point to the hypothesis that in schizophrenic patients the DMN is incorrectly activated in conditions that are not typical for such activation (tr-fMRI), while based on the study of connectivity we note that the DMN is deactivated in rest conditions, which is the normal condition of its activation.
Our views are that tDCS can be useful for restoring the normal connectivity of the different components of the DMN if the electrodes are placed simultaneously in several areas of the parietal cortex, namely the PreCu, PCC and AngG, which can potentially regulate the disrupted connection between the individual components of DMN and thus lead to improved symptoms such as cognitive impairment in schizophrenia.
DBS
DBS is the most invasive focal neuromodulation tool for mental disorders available. This technique allows us to deliver electrical stimulation of GM or white matter in a therapeutic attempt to improve the dysfunctional activity of the brain. DBS is probably the most elegant and complex perfect example of the translational approach – integration of instrumental, neurobiological, neurosurgical, and psychopathological knowledge in performing this precise and innovative psychosurgical procedure. It involves a high-frequency deep-brain stimulation by stereotactic inserted electrodes. It is known to act primarily by functional inhibition around the electrode, but it may also have excitatory effects on local axons and far-reaching excitatory effects[75].
DBS has proven to be beneficial in the treatment of severe mental illnesses like major depression[76], obsessive-compulsive disorder (OCD)[77], Tourette’s syndrome[78] and addiction[79]. The use of DBS in patients suffering from schizophrenia is still in its early stages and at present has not been officially approved. There seems to be no consensus regarding the particular brain structures, networks, and circuits which could be clinically beneficial for DBS treatment of schizophrenia. Studies have reported DBS of the habenula[80], nucleus accumbens and ACC[81]. These early experiments indicate the capacity of DBS to have beneficial effects in schizophrenic patients who do not respond to any other treatment. In order to assess the magnitude of the advantages and whether they can be accomplished without psychiatric side-effects, further studies would be needed with thoughtful consideration.
We suggest that the future research on the therapeutic effect of DBS in schizophrenia should focus extensively on two important areas, namely the insula and the PreCu, as they are the key nodes of the SN and DMN — the two brain networks that appear to be disconnected in the paranoid group in our study. We speculate that the enhancement of the altered function of the DMN and SN could ultimately restore the cognitive deficits and the disorganized thought and behavior observed in schizophrenic patients.
CONCLUSION
Psychotic and mood disorders affect a large number of people all over the world and have significant effects for individuals, their loved ones and society. To improve the prognosis and outcome of these disorders, it is crucial to make changes in their management. Unconventional instrumental biomedical methods have proven effectiveness for different psychiatric conditions (e.g., ECT for depression, mania, and catatonia; TMS for drug-resistant depression; tDCS for cognitive enhancement in schizophrenia; DBS for OCD, addiction and severe MDD). However, their usage is still limited, and these tools remain in the shadow of psychopharmacology and psychotherapy.
Results from our studies on cross-validation of tr-fMRI, effective connectivity at rest and the MLM point to some interesting and new therapeutic applications that can modulate the SN and thereby the DMN and FPN function that are impaired in people suffering from schizophrenia. Moreover, these findings could be translated in the protocols of methods like ECT, TMS, tDCS and DBS that have the potential to modulate the network plasticity of the brain. The comprehensive approach towards modifying dysfunctional SN and DMN found in patients with schizophrenia is likely to require a combination of pharmacogenomic testing to inform pharmacotherapy selection and dosing decisions in order to restore the aberrant plasticity of brain networks, together with targeted neurostimulation to trigger network reorganization. However, this approach and the specific mechanisms that influence the complex abnormal interactions between the SN and the DMN are not fully understood and need further investigation.
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/Licenses/by-nc/4.0/
Manuscript source: Invited manuscript
Specialty type: Psychiatry
Country/Territory of origin: Bulgaria
Peer-review report’s scientific quality classification
Grade A (Excellent): A, A
Grade B (Very good): 0
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Kotzalidis GD S-Editor: Gao CC L-Editor: Webster JR P-Editor: Li JH