Published online Mar 20, 2026. doi: 10.5493/wjem.v16.i1.116584
Revised: January 20, 2026
Accepted: February 24, 2026
Published online: March 20, 2026
Processing time: 121 Days and 0.1 Hours
Olopatadine hydrochloride (olopatadine), a histamine H1 receptor antagonist, inhibits release of substance P (SP) from peripheral nerve endings. It has anti
To investigate effects of olopatadine on SP concentrations in nasal secretions of patients with cedar pollinosis and their relationship with symptoms.
Nasal secretions were collected before and after administration of olopatadine to 12 Japanese patients with cedar pollinosis. SP concentrations in nasal secretions were measured and their relationship with scores for nasal symptoms evaluated.
After administration of olopatadine, sneezing, rhinorrhea and severity of symptoms improved significantly (all P < 0.05) and SP concentrations in nasal discharge were significantly lower than before treatment. Our findings suggest that symptoms improve when SP concentrations in nasal secretions decrease. It has been reported that patients with allergic rhinitis have significantly higher concentrations of SP in nasal secretions than healthy individuals, suggesting the importance of controlling SP concentrations when treating allergies.
Olopatadine suppresses release of SP, which is involved in itching and inflammation. Our findings suggest that its administration may prove to be a useful new strategy for treating allergies.
Core Tip: Olopatadine hydrochloride (olopatadine), a histamine H1 receptor antagonist, is used to treat various allergic diseases. It has been reported that the concentration of substance P (SP) in the nasal secretions of patients with pollinosis is higher than that of healthy individuals. Furthermore, it has been reported that olopatadine inhibits release of SP. In this study, we found that olopatadine reduces the concentration of SP in the nasal secretions of patients with pollinosis, suggesting that olopatadine reduces SP concentrations and may therefore contribute to improvement in symptoms.
- Citation: Katagiri F, Hatoyama-Tanaka S, Watanabe T, Takayanagi R, Suzuki M, Yamada Y. Effects of olopatadine hydrochloride on substance P concentrations in nasal secretions and nasal symptoms. World J Exp Med 2026; 16(1): 116584
- URL: https://www.wjgnet.com/2220-315x/full/v16/i1/116584.htm
- DOI: https://dx.doi.org/10.5493/wjem.v16.i1.116584
The prevalence of seasonal allergic rhinitis (pollinosis) in Japan increased from 19.6% to 29.8% to 42.5% in 1998, 2008, and 2019, respectively[1]. In particular, the prevalence of Japanese cedar pollinosis increased significantly, from 16.2% to 26.5% to 38.8%, respectively, compared with other pollinoses. It is expected that the prevalence of cedar pollinosis, which rarely resolves spontaneously[2], will continue to increase. Additionally, the amount of scattered cedar pollen is increasing in some areas.
Pollinosis is characterized by repeated paroxysmal sneezing, watery rhinorrhea, and nasal blockage. These manifestations are caused by inhalation of antigens and release of various chemical mediators, such as histamine and leukotriene, from mast cells distributed on the surface of the nasal mucosa. The released histamine binds to histamine H1 receptors on the terminals of sensory nerves, activating the nerves and causing the release of substance P (SP) via the axon reflex[3].
SP, a neuropeptide tachykinin, is widely distributed in the sensory nerves of nasal mucosal epithelium and causes nasal blockage by enhancing vascular permeability[3]. Additionally, SP is released into the nasal secretions by induction of nasal mucosal antigen in patients with pollinosis[4]. It has been reported that, under pollen scattering conditions, SP concentrations in nasal secretions are higher in patients with pollinosis than in healthy persons[5].
Olopatadine hydrochloride (olopatadine), a histamine H1 receptor antagonist, suppresses release of tachykinins, including SP[6,7]. Yuta et al[8] reported that SP concentrations in nasal secretions decrease after administering saline as an antigenic stimulus to healthy volunteers and then administering olopatadine. It has been reported that individuals with atopic dermatitis have higher plasma SP concentrations than do healthy individuals, and that these concentrations correlate with symptoms[9]. Olopatadine significantly decreases SP concentrations in individuals with atopic dermatitis, whereas other histamine H1 receptor antagonists either increase SP concentrations or do not change them[10]. Further
In this study, we investigated the relationship between nasal symptoms and changes in SP concentrations in nasal secretions after olopatadine administration to patients with cedar pollinosis.
The participants had visited our institution during the cedar pollen dispersal period (February to April) for management of cedar pollinosis, and had not started treatment with olopatadine. The subjects were patients with cedar pollinosis who had symptoms of allergic rhinitis and who had positive results in two or more of the following tests: Allergy skin test (or IgE antibody test), nasal provocation test, and nasal eosinophil test. This study was approved by the Ethics Committee of our institution and all participants provided written informed consent for inclusion.
Five mg olopatadine was administered orally twice daily (after breakfast and before bedtime). The participants evaluated their nasal symptoms before and after administration of olopatadine and recorded in a nasal allergy diary their classification of the severity of those symptoms in accordance with the classification provided by the Japanese Guidelines for Allergic Rhinitis 2020[11] (Tables 1 and 2). Test nasal secretions were collected from the participants before and 1 to 8 weeks (average 4.75 weeks) after administration of olopatadine. SP concentrations in nasal secretions were measured by the Pharmacy Department of our institution and changes in those concentrations before and after administration of olopatadine evaluated. The differences between SP concentrations in nasal secretions and scores for each nasal symptom before and after administration of olopatadine were also assessed.
| Severity | Paroxysmal sneezing or rhinorrhea1 | |||||
| 4 (++++) | 3 (+++) | 2 (++) | 1 (+) | 0 (-) | ||
| Nasal blockage | 4 (++++) | Most severe | Most severe | Most severe | Most severe | Most severe |
| 3 (+++) | Most severe | Severe | Severe | Severe | Severe | |
| 2 (++) | Most severe | Severe | Moderate | Moderate | Moderate | |
| 1 (+) | Most severe | Severe | Moderate | Mild | Mild | |
| 0 (-) | Most severe | Severe | Moderate | Mild | No symptoms | |
| Severity | Types | ||
| Sneezing1 | Rhinorrhea2 | Nasal blockage | |
| 4 (++++) | ≥ 21 times | ≥ 21 times | Nose completely obstructed all day |
| 3 (+++) | 11-20 times | 11-20 times | Severe nasal blockage causing oral breathing during a day |
| 2 (++) | 6-10 times | 60-10 times | Severe nasal blockage causing occasional oral breathing during a day |
| 1 (+) | 1-5 times | 1-5 times | Nasal blockage without oral breathing |
| 0 (-) | Below + | Below + | Below + |
Samples of nasal secretions were aspirated and collected using a Juhn Tym-Tap tube, aprotinin (500 KIU units/mL) and ethylenediaminetetraacetic acid (1.2 mg/mL) being added immediately. After centrifugation, the nasal secretions were diluted 20-fold with 4% acetic acid (pH 4.0) and loaded onto a C18 reversed-phase cartridge (Sep-Pak C18; Millipore, Milford, MA, United States). After washing with 4% acetic acid, the peptides in the nasal secretions were eluted with 70% acetonitrile in 0.5% acetic acid (pH 4.0). The eluted samples were concentrated by spin-vacuum evaporation, lyophilized, and stored at -40 °C until assayed.
SP concentrations in nasal secretions were measured using a highly sensitive enzyme immunoassay, as previously described[12]. This assay was performed using a delayed-addition method. Bound and free antigens were separated on anti-rabbit IgG-coated immunoplates (Thermo Scientific MaxiSorp immunoplate; Thermo Fisher Scientific, MA, United States). Human substance P (Peptide Institute, Osaka, Japan) were conjugated with β-D-galactosidase (Bayer, Leverkusen, Germany) using N-(ε-maleimidocaproyl oxy-succinimide (Sigma, St. Louis, MO, USA), as reported by Kitagawa et al[13]. Next, 100 μL of anti-SP rabbit anti-serum (Cambridge Research Biochemicals, Cambridge, United Kingdom) was added to an extracted sample of nasal secretion reconstituted to 100 μL. After reacting at 4 °C for 24 hours, these antigen-antibody mixed solutions were reacted with 50 μL β-D-galactosidase-labeled SP solution at 4 °C for 24 hours. Next, 100 μL of the resultant mixtures were added to microplates coated with anti-rabbit IgG goat anti-sera, reacted overnight at 4 °C and washed four times with 0.01 M phosphate buffered saline containing 0.05% Tween 20. Thereafter 200 μL of 0.1 mmol/L 4-methylumbel-liferyl β-D-galactopyranoside (Sigma) aqueous solution was reacted at 37 °C for 3 hours. The fluorescence intensity was then measured (Ex 360 nm, Em 450 nm) and the SP concentrations in the samples calculated from a standard curve. The enzyme immunoassay for SP is specific and highly sensitive (the detection limit is 0.1 fmol/well).
After analysis of variance for various data, paired t-tests were performed to analyze SP concentrations in nasal secretions and Wilcoxon signed-rank tests for other data. For statistical analysis, JMP pro14 (jmp Statistical Discovery™ from SAS) was used. P values of less than 5% were considered to denote significant differences.
The study cohort comprised 12 patients with cedar pollinosis (four men and eight women) of mean age 41 years (range 24-59 years).
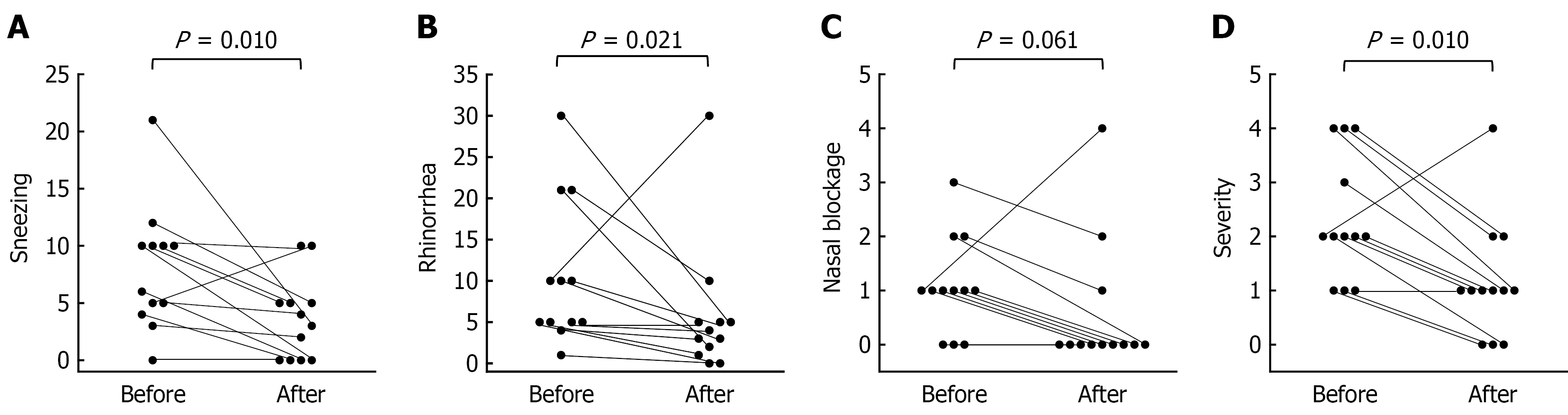
The severity of sneezing before treatment was “4” in 1 patient, “3” in 1, “2” in 5, “1” in 4, and “0” in 1. After treatment, the scores were “2” in 2 patients, “1” in 6, and “0” in 4. The severity of rhinorrhea before treatment was “4” in 3 patients, “2” in 3, and “1” in 6. After treatment, the scores were “4” in 1 patient, “2” in 1, “1” in 8, and “0” in 2. The severity of nasal blockage before treatment was “3” in 1 patient, “2” in 2, “1” in 6, and “0” in 3. After treatment, the scores were “4” in 1 patient, “2” in 1, “1” in 8, and “0” in 2. The severity of symptoms before treatment was “Most severe” in 3 patients, “Severe” in 1, “Moderate” in 4, and “Mild” in 3. After treatment, the scores were “Most severe” in 1 patient, “Moderate” in 2, “Mild” in 6, and “No symptoms” in 3.
After treatment with olopatadine, there were significantly fewer episodes of sneezing and rhinorrhea and they were significantly less severe than before treatment (Figure 1). Nasal blockage tended to improve; however, the difference was not statistically significant (P = 0.061) (Figure 1C). After administration of olopatadine, the number of sneezes did not change or increased in three patients, the number of episodes of rhinorrhea did not change or increased in two, the degree of nasal blockage did not change or increased in four, and the severity of nasal blockage did not change or worsened in two.
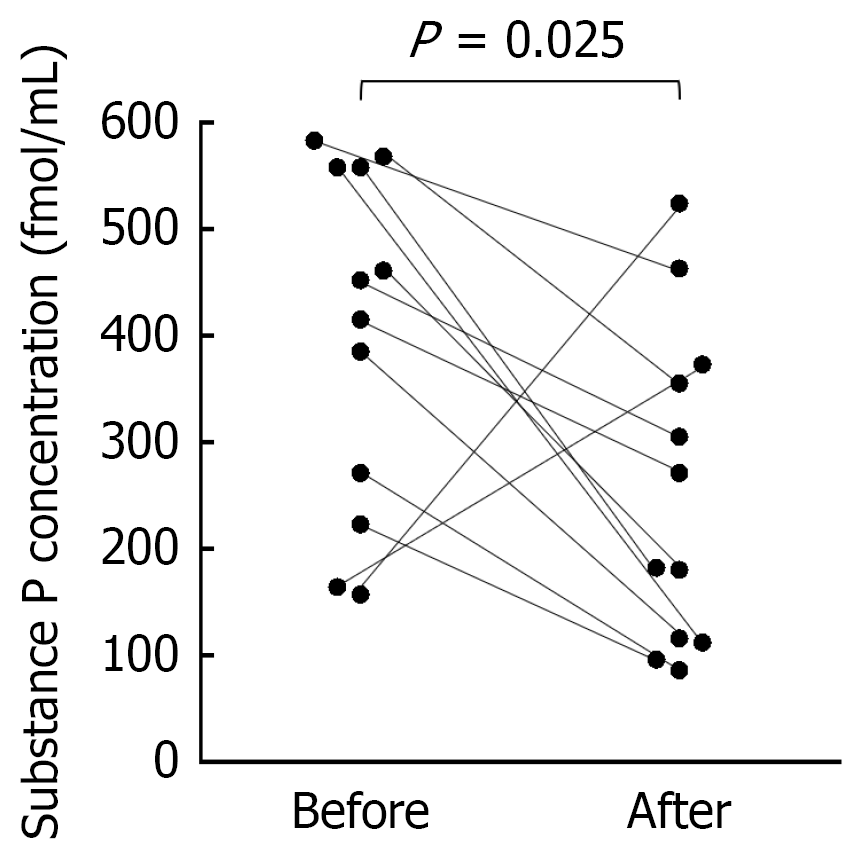
SP concentrations in nasal secretions decreased significantly after administration of olopatadine (Figure 2). Only two patients had high concentrations of SP in their nasal secretions after administration of olopatadine.
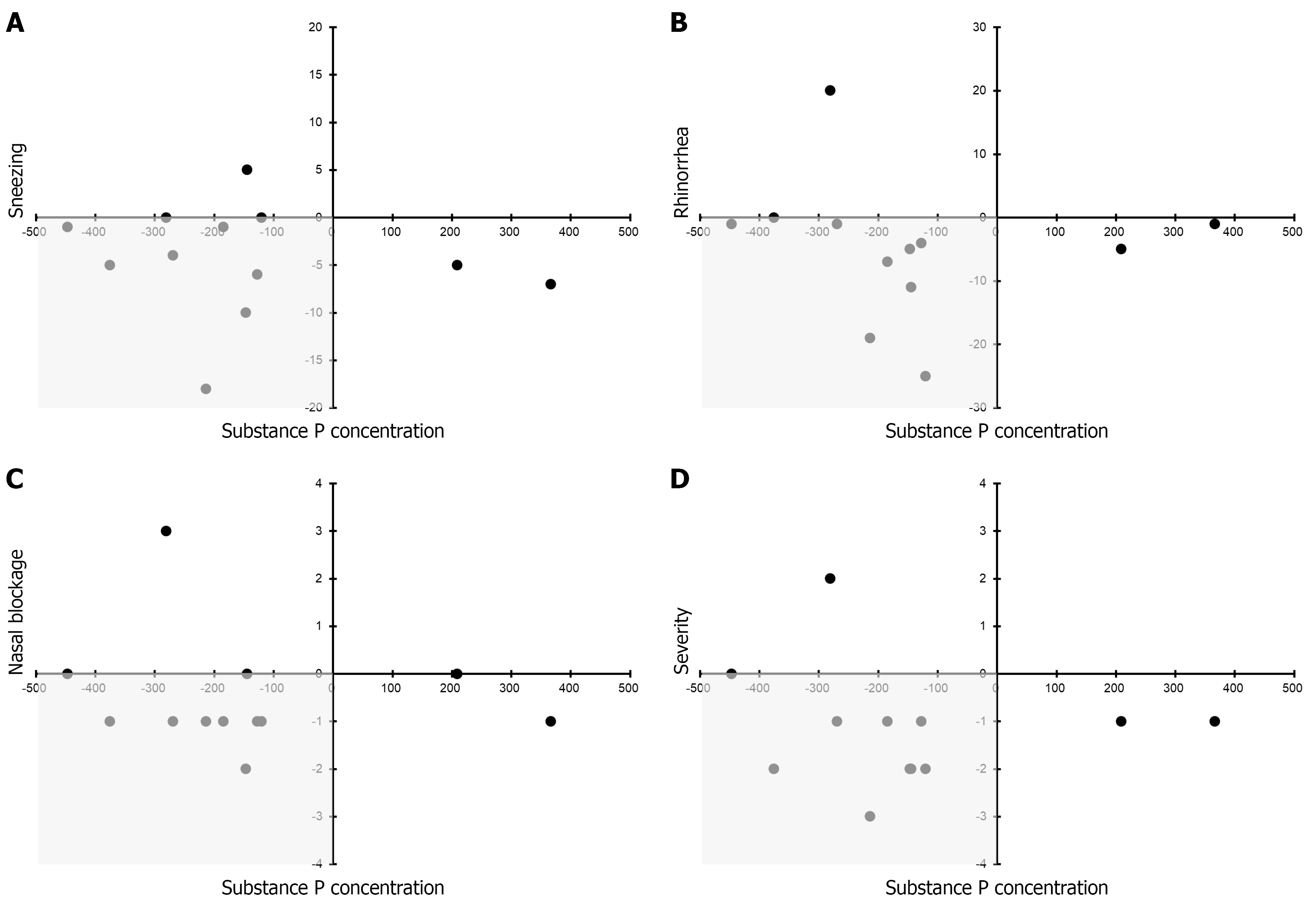
Figure 3 shows the differences between SP concentrations in nasal discharges and scores for each nasal symptom before and after administration of olopatadine. Differences in SP concentrations in nasal secretions before and after treatment are shown on the horizontal axis, whereas differences between scores for each nasal symptom are shown on the vertical axis. Compared with before treatment, both SP concentrations in nasal secretions and scores for each nasal symptom decreased in many patients. However, despite a decrease in SP concentrations in the nasal secretions, nasal symptoms worsened in one patient. Conversely, nasal symptoms improved in two patients despite increases in SP concentrations in their nasal secretions.
The patients were divided into four groups, increases in SP concentration in nasal secretions and exacerbation of nasal symptoms after treatment being denoted by “+”, and decreases in SP concentration in nasal secretions and improvement in nasal symptoms being denoted by “-” (Table 3). Patients whose symptoms did not change after treatment were classified as improved. Nasal secretions decreased and symptoms improved after administration of SP in 67%, 67%, 75%, and 67% of patients for sneezing, rhinorrhea, nasal blockage, and overall severity of symptoms, respectively, these all being high percentages (Table 3).
| Substance P concentration, nasal symptoms | ||||
| (+, +) | (+, -) | (-, +) | (-, -) | |
| Sneezing | 0 | 2 | 2 | 8 |
| Rhinorrhea | 0 | 2 | 2 | 8 |
| Nasal blockage | 0 | 2 | 1 | 9 |
| Severity | 0 | 2 | 2 | 8 |
Similar to the findings of Yuta et al[8], we found that SP concentrations in nasal secretions decreased significantly after administration of olopatadine, suggesting that olopatadine reduces SP concentrations in nasal secretions of patients with cedar pollinosis during pollen dispersal. In this study, SP concentrations in nasal secretions decreased and nasal symptoms improved in eight of the 12 participants. However, one patient’s nasal symptoms worsened despite a decrease in SP concentration in nasal secretions. Furthermore, two patients’ nasal symptoms improved despite increases in SP concentrations in their nasal secretions. Other chemical mediators besides SP, such as leukotrienes and thromboxane A2, may exacerbate the symptoms of cedar pollinosis. In this study, we did not have enough nasal secretions to measure the concentrations in them of any factors other than SP, including histamine.
Nasal administration of SP to histamine H1 receptor-knockout mice has been found to significantly increase sneezing and nasal scraping behavior. Of note, SP’s effects on nasal allergy symptoms are not mediated exclusively by histamine H1 receptors[14]. In addition, another research group stimulated both nasal cavities of histamine H1 receptor-deficient mice with capsaicin, followed by administration of olopatadine and evaluated their nasal symptoms[15]. They found that nose picking behavior was less pronounced in the olopatadine than in the control group. These findings suggest that SP concentrations in nasal secretions are associated with nasal symptoms in individuals with pollinosis and that olopatadine improves their nasal symptoms by both suppressing SP release and antagonizing histamine H1 receptors. SP is released by binding released histamine to the histamine H1 receptors of sensory nerve terminals. However, it has been reported that histamine H1 receptor antagonists other than olopatadine do not reduce SP concentrations in patients with atopic dermatitis[10]. Thus, SP release may not be stimulated exclusively by histamine H1 receptors.
There are four subtypes of histamine receptors: H1 to H4[16] JNJ7777120, a histamine H4 receptor antagonist, reportedly improves pruritus in a mouse model of atopic dermatitis[17]. This effect has not been found with fexofenadine, which is a histamine H1 receptor antagonist, indicating that the reported improvement in the mouse model was histamine H4 receptor-specific. Given that fexofenadine does not reduce serum SP concentrations in individuals with atopic dermatitis[10], it is possible that SP release involves histamine H4 receptors. If SP release does indeed involve histamine H4 re
In conclusion, our findings suggest that olopatadine improves the symptoms of patients with cedar pollinosis by both suppressing SP release and antagonizing histamine H1 receptors; however, the detailed mechanisms are unknown. Both histamine H4 and histamine H1 receptors may be involved in the effects of olopatadine. This study is considered to be a pilot study due to its small sample size, lack of a placebo, and open-label nature. It is believed that no definitive results can be obtained. We believe that a larger, randomized, double-blind study using a placebo or another histamine H1 receptor antagonist as a control is necessary.
We thank Mr. Koji Kimura (Department of Clinical Evaluation of Drug Efficacy, School of Pharmacy, Tokyo University of Pharmacy and Life Sciences) for comments on the manuscript.
| 1. | Matsubara A, Sakashita M, Gotoh M, Kawashima K, Matsuoka T, Kondo S, Yamada T, Takeno S, Takeuchi K, Urashima M, Fujieda S, Okubo K. Epidemiological Survey of Allergic Rhinitis in Japan 2019. Nippon Jibiinkoka Gakkai Kaiho. 2020;123:485-490. [DOI] [Full Text] |
| 2. | Baba K, Tanigaito Y, Tekeda T, Rin S. Natural “Growing Out” in patients with cedar pollinosis. Jibi Rinsho. 1991;37:1187-1191. |
| 3. | Sarin S, Undem B, Sanico A, Togias A. The role of the nervous system in rhinitis. J Allergy Clin Immunol. 2006;118:999-1016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 147] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 4. | Baraniuk JN, Ali M, Yuta A, Fang SY, Naranch K. Hypertonic saline nasal provocation stimulates nociceptive nerves, substance P release, and glandular mucous exocytosis in normal humans. Am J Respir Crit Care Med. 1999;160:655-662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 99] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 5. | Mori K, Asakura S, Morikawa N, Chaen T, Watanabe N, Mogi G, Takeyama M. Seasonal fluctuations of substance P and vasoactive intestinal peptide concentrations in nasal secretions of patients with nasal allergy to Japanese cedar pollen. Arerugi. 1992;41:1651-1657. [PubMed] |
| 6. | Ikemura T, Okarmura K, Sasaki Y, Ishi H, Ohmori K. KW-4679-induced inhibition of tachykininergic contraction in the guinea-pig bronchi by prejunctional inhibition of peripheral sensory nerves. Br J Pharmacol. 1996;117:967-973. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 16] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 7. | Mosimann BL, White MV, Hohman RJ, Goldrich MS, Kaulbach HC, Kaliner MA. Substance P, calcitonin gene-related peptide, and vasoactive intestinal peptide increase in nasal secretions after allergen challenge in atopic patients. J Allergy Clin Immunol. 1993;92:95-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 123] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 8. | Yuta A, Majima Y. Suppressive effect of olopatadine hydrochloride on substance P release during hypertonic saline nasal induction. Arerugi. 2002;51:999. |
| 9. | Toyoda M, Nakamura M, Makino T, Hino T, Kagoura M, Morohashi M. Nerve growth factor and substance P are useful plasma markers of disease activity in atopic dermatitis. Br J Dermatol. 2002;147:71-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 237] [Cited by in RCA: 208] [Article Influence: 8.7] [Reference Citation Analysis (1)] |
| 10. | Izu K, Tokura Y. The various effects of four H1-antagonists on serum substance P levels in patients with atopic dermatitis. J Dermatol. 2005;32:776-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 11. | Okubo K, Kurono Y, Ichimura K, Enomoto T, Okamoto Y, Kawauchi H, Suzaki H, Fujieda S, Masuyama K; Japanese Society of Allergology. Japanese guidelines for allergic rhinitis 2020. Allergol Int. 2020;69:331-345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 175] [Article Influence: 29.2] [Reference Citation Analysis (1)] |
| 12. | Takeyama M, Mori K, Takayama F, Kondo K, Kitagawa K, Fujii N. Enzyme immunoassay of a substance P-like immunoreactive substance in human plasma and saliva. Chem Pharm Bull (Tokyo). 1990;38:3494-3496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 29] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 13. | Kitagawa T, Shimozono T, Aikawa T, Yoshida T, Nishimura H. Preparation and characterization of hetero-bifunctional cross-linking reagents for protein modifications. Chem Pharm Bull. 1981;29:1130-1135. [DOI] [Full Text] |
| 14. | Sugimoto Y. Analysis of allergic rhinitis using histamine H1 receptor-deficient mice. 119th Annual meeting of the Pharmaceutical Society of Japan, Tokushima, 1999. |
| 15. | Sugimoto Y. Inhibitory effect of olopatadine on nasal rubbing induced by capsaicin in histamine H1 receptor-deficient and mast cell-deficient mice. 77th Annual Meeting of the Japanese Pharmacologocal Society, Osaka, 2004. |
| 16. | Kamei C, Nakano Y, Kagawa Y, Izawa K, Yano H. Role of histamine H(4)-receptor in allergic conjunctivitis. Nihon Yakurigaku Zasshi. 2010;135:134-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 17. | Suwa E, Yamaura K, Oda M, Namiki T, Ueno K. Histamine H(4) receptor antagonist reduces dermal inflammation and pruritus in a hapten-induced experimental model. Eur J Pharmacol. 2011;667:383-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 40] [Article Influence: 2.7] [Reference Citation Analysis (1)] |