Published online Mar 20, 2026. doi: 10.5493/wjem.v16.i1.116252
Revised: January 6, 2026
Accepted: February 14, 2026
Published online: March 20, 2026
Processing time: 129 Days and 14.3 Hours
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disorder characterized by insulin resistance and hyperglycemia. Metformin is the standard therapy, however plant-based adjuncts such as Gymnema sylvestre (GS) are gaining interest for their anti-diabetic potential.
To evaluate the anti-diabetic effects of GS, alone and with metformin, on glycemic control, lipid profile, renal function, and body weight in high-fat diet and streptozotocin (STZ)-induced T2DM rats.
Thirty male Sprague Dawley rats (150 ± 20 g) were divided into five groups: Normal control, diabetic control (DC), metformin-treated, GS-treated, and com
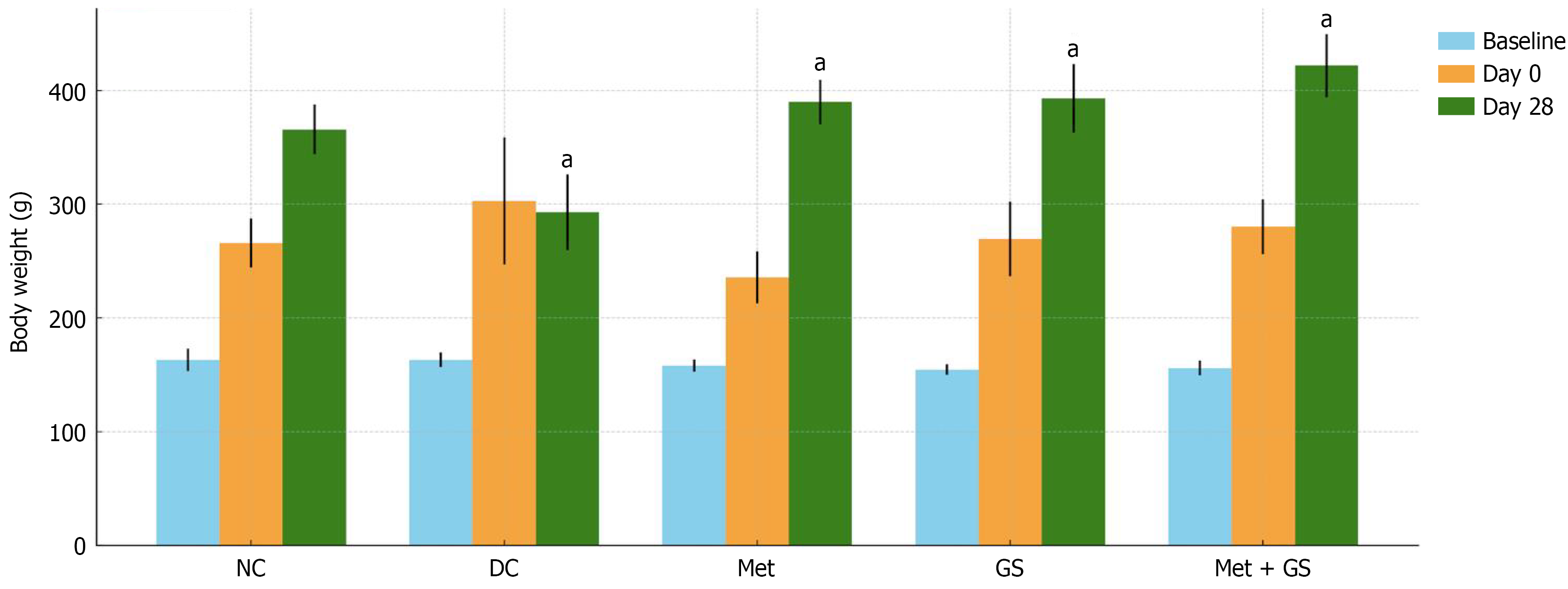
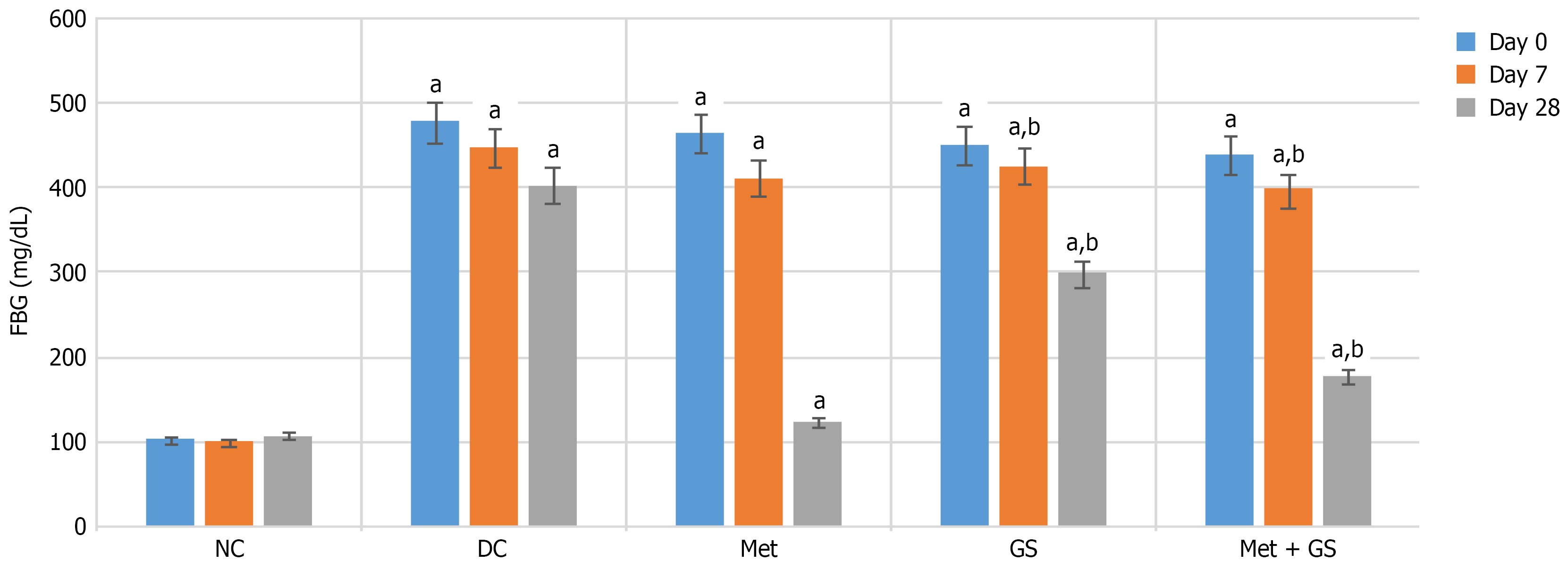
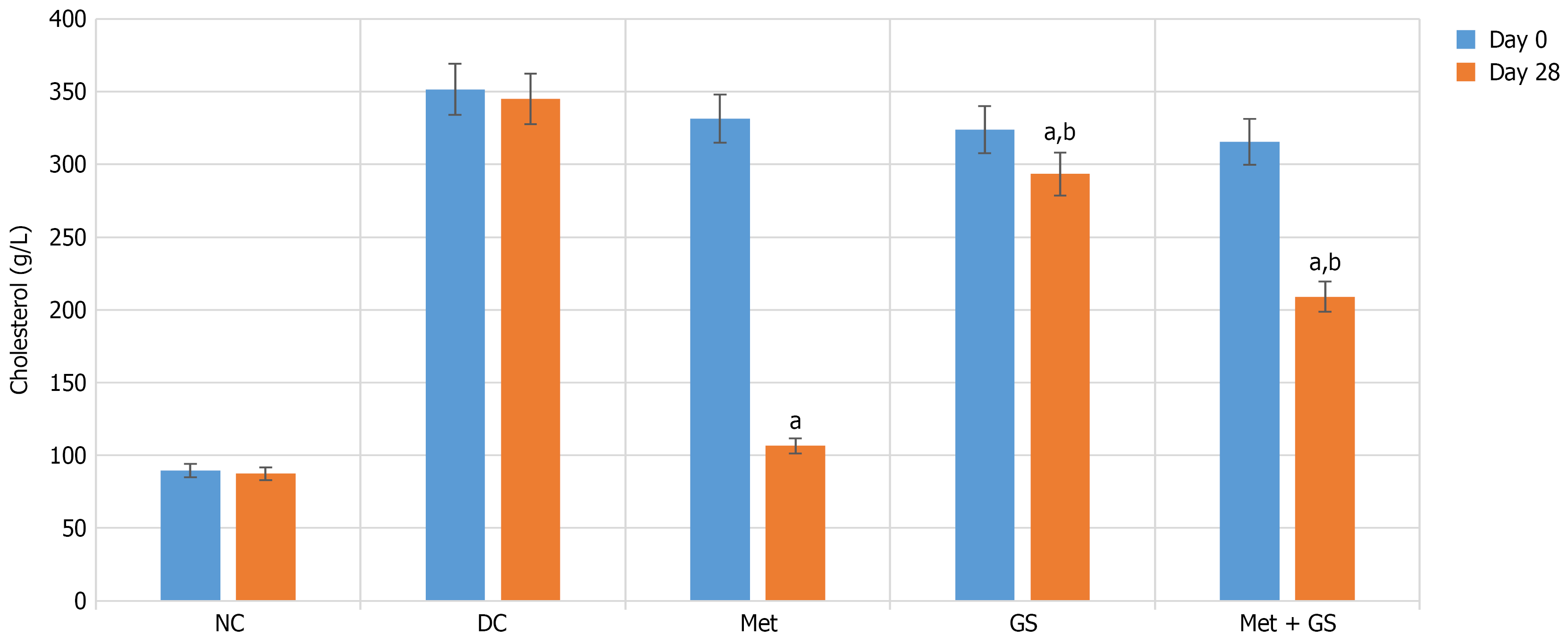
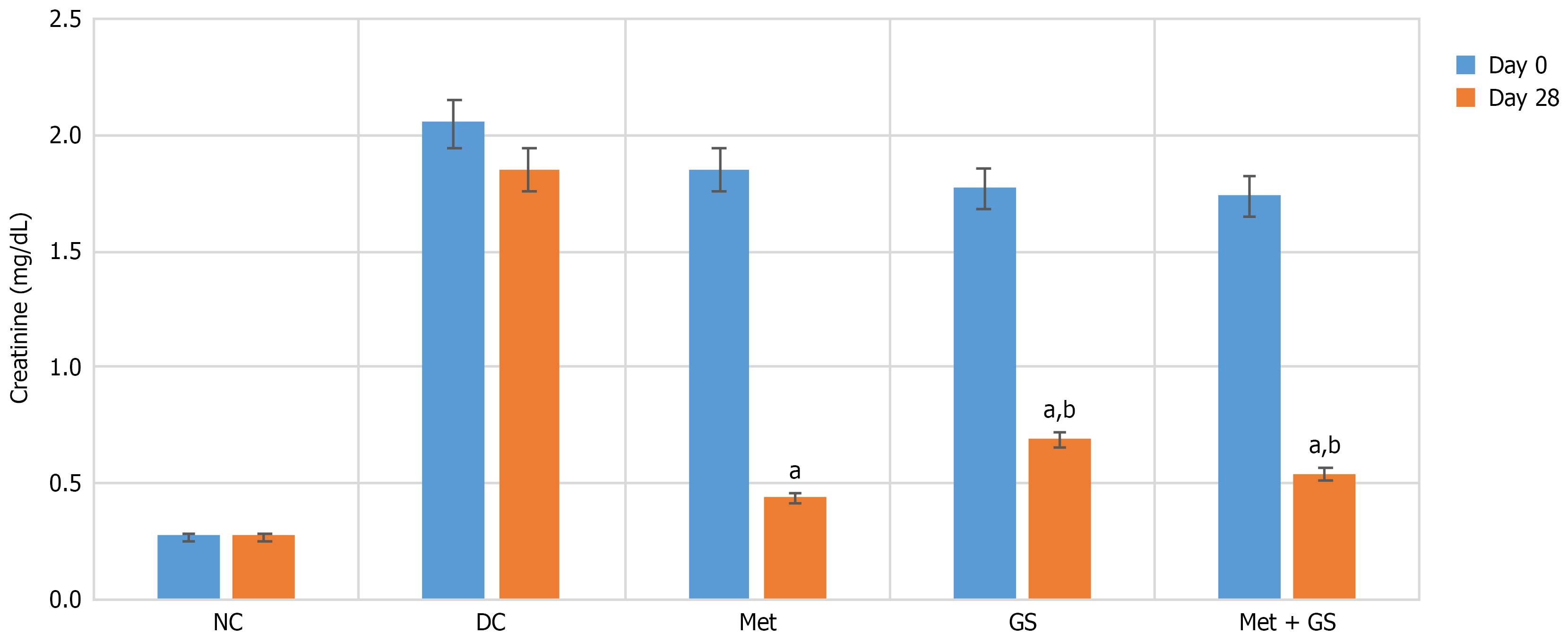
By day 28, all treatment groups showed significant improvements compared to DC. Metformin produced the greatest reductions in FBG, cholesterol, creatinine, and glycated hemoglobin. GS monotherapy had modest but significant effects, while metformin + GS improved glycemic control and lipid profile more effectively than GS alone. Metformin remained superior in renal protection. All treated groups showed significantly higher body weights than DC, indicating protection against STZ-induced weight loss.
This study demonstrated that metformin and GS, individually and in combination, improved glycemic control and metabolic outcomes in T2DM rats. While metformin showed the greater efficacy, combination therapy provided additive benefits, supporting GS as a potential adjunct in T2DM management.
Core Tip: This study evaluates the antidiabetic effects of Gymnema sylvestre (GS) alone and in combination with metformin in a high-fat diet and streptozotocin-induced type 2 diabetic rat model. The combination therapy improved glycemic control, lipid profile, renal parameters, and body weight compared with GS alone, although metformin remained superior. These findings support the potential role of GS as an adjunct to standard metformin therapy in type 2 diabetes mellitus.
- Citation: Kodiyatar A, Mishra A, Dhakad MS, Roy V, Beg MMA, Rai N. Antidiabetic effects of Gymnema sylvestre and metformin co-therapy in a type 2 diabetic rat model. World J Exp Med 2026; 16(1): 116252
- URL: https://www.wjgnet.com/2220-315x/full/v16/i1/116252.htm
- DOI: https://dx.doi.org/10.5493/wjem.v16.i1.116252
Diabetes mellitus is a chronic metabolic disorder characterized by persistent hyperglycemia resulting from impaired insulin secretion, insulin action, or both. It poses a major global health burden due to its increasing prevalence and associated complications such as cardiovascular disease, neuropathy, and nephropathy[1].
Diabetes mellitus is primarily classified into two types: Type 1 and type 2. Type 1 diabetes is an autoimmune condition marked by the destruction of pancreatic β-cells, leading to absolute insulin deficiency. Insulin therapy remains the cornerstone for its management to maintain glycemic control and prevent complications. Type 2 diabetes mellitus (T2DM), the more prevalent form, is characterized by insulin resistance and relative insulin deficiency[2].
Metformin, a biguanide compound, is the first-line oral antidiabetic drug for T2DM. It exerts its effects by reducing hepatic glucose production, enhancing peripheral glucose uptake, and improving insulin sensitivity[3]. While effective in glycemic control and in reducing the risk of diabetic complications, long-term metformin use may lead to adverse effects such as gastrointestinal disturbances and, rarely, lactic acidosis[4]. These limitations highlight the need for complementary or alternative therapies that are effective and safer.
In this context, Gymnema sylvestre (GS)-commonly known as “Gurmar” in Hindi, a woody climber from the Asclepiadaceae (milkweed) family, has garnered attention for its antidiabetic potential. Used for over 2000 years in Ayurvedic medicine, GS contains a range of bioactive phyto-constituents such as gymnemic acids, saponins, flavonoids, triterpenoids, and gurmarin a polypeptide with hypoglycemic properties[5,6]. Among these, gymnemic acids are oleanane-type triterpenoid saponins that competitively inhibit glucose absorption in the intestines by occupying glucose receptor sites on the gut mucosa[6]. Furthermore, they promote insulin secretion and enhance peripheral glucose utilization. The flavonoids and saponins also exhibit potent antioxidant activity, reducing oxidative stress and improving insulin sensitivity[6,7]. These multifaceted mechanisms support the plant’s hypoglycemic, insulinotropic, and antioxidative effects, making it a promising adjunct in the management of T2DM[7].
Experimental models are crucial for studying the pathophysiology of T2DM and evaluating potential therapeutic agents. Streptozotocin (STZ), a nitrosourea derivative, is widely used in experimental diabetes due to its selective cytotoxicity towards pancreatic β-cells, resulting in sustained hyperglycemia[8]. However, to better replicate the complexity of human T2DM, a combination model involving a high-fat diet (HFD) followed by low-dose STZ is often used. The HFD induces insulin resistance through lipid metabolism disruption and impairment of insulin signaling, while STZ partially damages β-cells, mimicking their gradual decline[9]. This HFD/STZ model is widely accepted for its physiological relevance and reproducibility in preclinical diabetes research.
While both metformin and GS have independently demonstrated significant antidiabetic effects, few studies have investigated their combined therapeutic potential. Whether this combination produces additive or synergistic benefits remains uncertain, highlighting a critical gap in current diabetes research. Evaluating the co-administration of metformin and GS in an HFD/STZ-induced T2DM rat model may offer valuable insights into integrated therapeutic strategies for better diabetes management.
Accordingly, the present study aimed to assess the efficacy of GS in combination with metformin in reducing fasting blood glucose (FBG) levels and body weight in STZ-induced diabetic rats. It further compared the therapeutic outcomes of the combination therapy with metformin monotherapy in achieving glycemic control and weight reduction. Ad
In this study, the sample size was determined using the resource equation method[10]. According to this method, “E” represents the degrees of freedom for analysis of variance (ANOVA), and its value should lie between 10 and 20.
The formula is: E = total number of animals - total number of groups, E = (N × n) - N. Where: N = number of groups, n = number of animals per group. For this study: Number of groups (N) = 5, animals per group (n) = 6. E = (5 × 6) - 5 = 30 - 5 = 25.
Since the calculated value of E exceeded the recommended range (10-20), corrections were applied considering biological factors. The efficacy of STZ in inducing T2DM is approximately 80%[11], and with an anticipated attrition rate of 20%, the effective sample size was adjusted to six animals per group. Thus, a total of 30 rats (six per group across five groups) were included in the study to ensure adequate statistical power and account for potential variability[12].
This study was conducted at the Central Animal Facility and Department of Pharmacology, Maulana Azad Medical College (New Delhi). This retrospective study was approved by the Institutional Animal Ethics Committee of Maulana Azad Medical College and Associated Hospitals (Approval No. IAEC/MAMC/CAF/2023/03).
STZ: Obtained from Sisco Research Laboratories Pvt. Ltd. (Mumbai, India) in 500 mg vials.
Preparation of plant extract: Hydroalcoholic extraction was performed on the powdered leaves of GS. GS leaf Extract (100 g powder) sourced from Arjuna Natural Pvt. Ltd. (Kerala, India).
Metformin: Administered as OKAMET 500 mg tablets (Cipla Ltd., Haridwar, India).
HFD: The HFD consisted of 60% kcal from fat (primarily lard), 20% protein (casein), and 20% carbohydrates (sucrose and corn-starch), sourced from Karyome Pvt. Ltd. (Mysuru, India)
Other reagents: Citric acid, sodium citrate, and sodium carboxymethyl cellulose were purchased from Sigma-Aldrich (MO, United States) and used to prepare the citrate buffer.
Thirty male Sprague Dawley rats (6-8 weeks old, 150 ± 20 g) were obtained from the Central Animal Facility, Maulana Azad Medical College (New Delhi, India). Rats were housed three per cage under standard laboratory conditions (25 ±
Phase 1 - induction of T2DM: The HFD/STZ rat model for T2DM induction was based on a HFD and low-dose STZ injections. Rats were fed a HFD ad libitum (approximately 20 g/rat/day), containing 60% fat, 20% protein, and 20% carbohydrates for 21 days to induce insulin resistance. Body weight was monitored weekly during this period. After 21 days, rats were given two intra-peritoneal injections of STZ (25 mg/kg body weight) prepared fresh in cold 0.1 mmol/L citrate buffer (pH 4.5) to preserve STZ stability and enhance beta-cell toxicity with a 5-day break between injections. FBG was measured three days after the second injection using a tail vein blood sample and glucometer. Rats with FBG ≥ 270 mg/dL were considered diabetic and enrolled for treatment[7]. The HFD combined with low-dose STZ model was used to induce T2DM because it closely resembles the pathophysiology of human T2DM. HFD feeding induces insulin resistance, while low-dose STZ causes partial pancreatic β-cell dysfunction rather than complete β-cell destruction. This results in stable hyperglycemia with residual insulin secretion, closely mimicking human T2DM and making the model suitable for evaluating antidiabetic agents[8,9].
Phase 2 - drug administration: Diabetic rats were randomly assigned to four groups (n = 6 per group), while non-diabetic rats were assigned to the normal control (NC) group: Group 1 (NC): Normal rats received normal saline (10 mL/kg) via oral gavage for 4 weeks. Group 2 [diabetic control (DC)]: Diabetic rats received normal saline (10 mL/kg) for 4 weeks. Group 3 (metformin): Diabetic rats received metformin (200 mg/kg/day) via oral gavage for 4 weeks. Group 4 (GS): Diabetic rats received GS (600 mg/kg/day) via oral gavage for 4 weeks. Group 5 (metformin + GS): Diabetic rats received both metformin (200 mg/kg/day) and GS (600 mg/kg/day) via oral gavage for 4 weeks. Dosages were calculated based on previously published studies and the human equivalent dose conversion method[9,10]. Specifically, the dose of GS (600 mg/kg) was selected based on Mall et al[13], which demonstrated significant antidiabetic activity in alloxan-induced diabetic rats.
Blood samples were collected via the retro-orbital plexus under ketamine (87 mg/kg) and xylazine (13 mg/kg) anesthesia. Approximately 0.4-0.5 mL of blood was collected into microcentrifuge tubes on day 0, day 7, and day 28 for FBG analysis, and on day 0 and day 28 for glycated hemoglobin (HbA1c), serum creatinine, and total cholesterol. Samples were centrifuged at 4000 rpm for 15 minutes at 4 °C, and plasma was stored at -80 °C until analysis using a Semi-Automated Biochemistry Analyzer (Rapid, serial No. 5ACL364; Rapid Holding Systems Ltd., Canada). Fasting insulin levels were determined using a commercially available rat insulin enzyme-linked immunosorbent assay kit (Invitrogen, Catalog No.: ERINS; Thermo Fisher, India) as per the manufacturer’s protocol by LisaScan Em, serial No. S180923 (Erba Diagnostics Mannheim GmbH, Germany).
All values were expressed as mean ± SD. Data were analyzed using two-way analysis of variance (ANOVA) followed by Tukey’s post hoc test to assess differences between multiple treatment groups across time points. This method was chosen due to its robustness in detecting group differences in repeated measures and factorial designs. P < 0.05 was considered statistically significant. Statistical analyses were performed using SPSS version 20.0 (Chicago, IL, United States).
At baseline, no significant differences were observed in body weight or FBG levels among the experimental groups. Following 21 days of HFD feeding and subsequent STZ administration, all diabetic groups exhibited significantly elevated FBG levels compared to the NC group (P < 0.05), confirming successful induction of T2DM (Table 1).
After 28 days of treatment, all intervention groups - metformin, GS, and their combination (metformin + GS) - showed significant improvements in key biochemical and physiological parameters compared to the DC group (P < 0.05). Among them, metformin produced the most pronounced improvements across all parameters. GS monotherapy resulted in moderate but significant effects, while the metformin + GS combination demonstrated greater efficacy than GS alone but remained slightly less effective than metformin in most outcomes.
By day 28, body weights in all treatment groups were significantly higher compared to the DC group (P < 0.001), indicating a protective effect against STZ-induced weight loss. The NC group, maintained on a standard pellet diet, exhibited significantly lower body weight than the HFD/STZ groups (Figure 1).
FBG levels were significantly reduced in the metformin and metformin + GS groups as early as day 7 compared to DC (P < 0.001). On day 7, reductions were significant in metformin (P < 0.05) and metformin + GS (P < 0.05) vs DC. By day 28, all treatment groups exhibited significantly lower FBG levels compared to DC (P < 0.001). Pairwise comparisons indicated that metformin + GS achieved superior glycemic control over GS (P < 0.05), although metformin remained more effective (P < 0.05 vs metformin + GS and GS). GS monotherapy produced only a modest reduction by day 28, with no significant effect at day 7 (Figure 2; Table 2).
| Group | Weight (gm) | FBG (mg/dL) | Cholesterol (g/L) | Creatinine (mg/dL) | Hb1Ac (g/L) | ||||
| Day 28 | Day 7 | Day 28 | Day 0 | Day 28 | Day 0 | Day 28 | Day 0 | Day 28 | |
| DC | 293.0 ± 33.41 | 445.66 ± 51.74 | 401.33 ± 24.548 | 351.6 ± 75.99 | 345.14 ± 70.77 | 2.05 ± 0.47 | 1.85 ± 0.04 | 10.55 ± 0.57 | 9.95 ± 0.49 |
| Met | 389.8 ± 19.53a | 409.66 ± 19.53a | 122.50 ± 6.15a | 331.6 ± 10.06 | 106.49 ± 2.73a | 1.85 ± 0.76 | 0.44 ± 0.03a | 10.65 ± 0.11 | 5.82 ± 0.15a |
| GS | 393.0 ± 30.21a | 423.83 ± 13.61 | 296.33 ± 8.26a,b | 323.9 ± 16.91 | 293.43 ± 3.29a,b | 1.77 ± 0.89 | 0.69 ± 0.04a,b | 10.62 ± 0.27 | 8.24 ± 0.13a,b |
| Met + GS | 421.8 ± 27.96a,b | 395.83 ± 10.88a,b | 176.50 ± 9.20a,b | 315.7 ± 14.53 | 208.99 ± 1.18b | 1.74 ± 0.12 | 0.54 ± 0.02a,b | 10.65 ± 0.70 | 7.26 ± 0.09a,b |
On day 28, all treatment groups showed significantly reduced cholesterol levels compared to DC (P < 0.001). Two-way repeated measures ANOVA confirmed that metformin, GS, and metformin + GS groups had substantially lower levels than DC. Pairwise comparisons revealed significant differences between metformin and metformin + GS (P < 0.05) and between metformin and GS (P < 0.05; Figure 3; Table 2).
On day 28, all treatment groups demonstrated significantly reduced creatinine levels compared to DC (P < 0.001). Two-way repeated measures ANOVA confirmed lower values in metformin, GS, and metformin + GS groups vs DC. Pairwise analysis showed significant differences between metformin and metformin + GS (P < 0.05) and between metformin and GS (P < 0.05; Figure 4; Table 2).
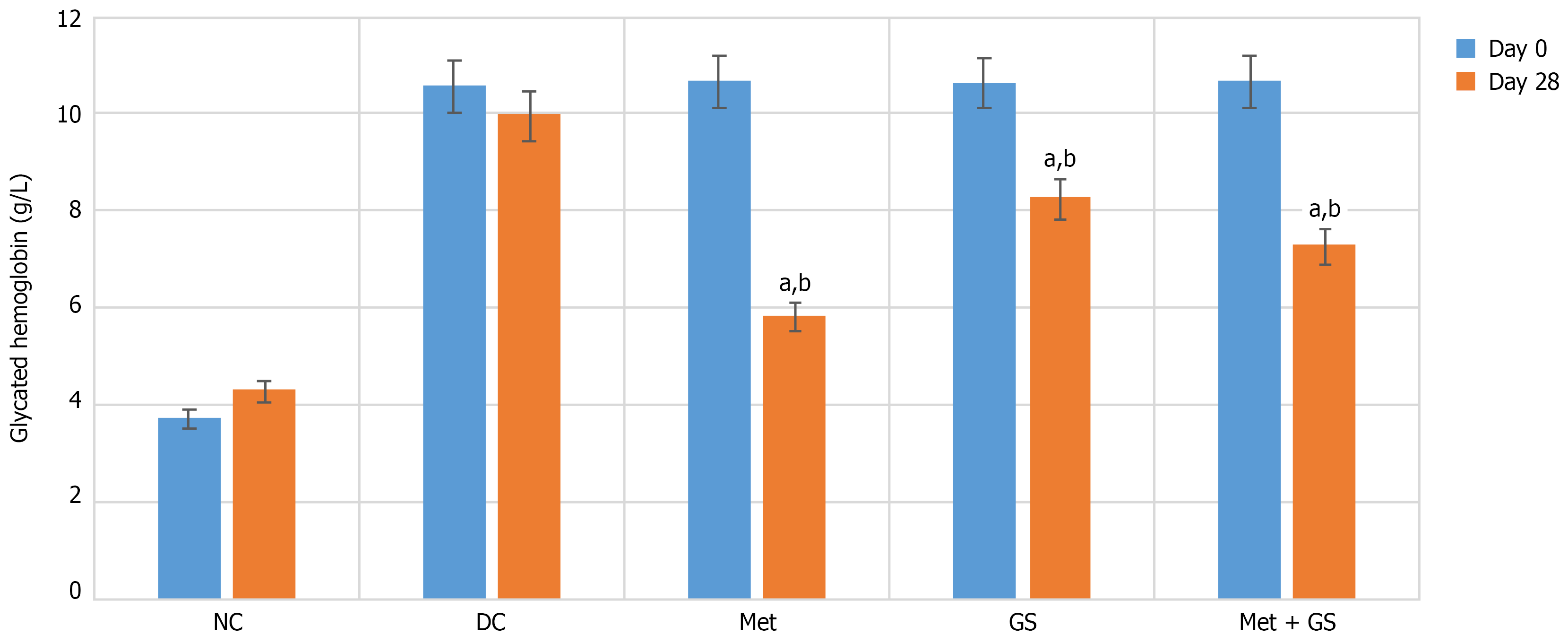
On day 28, HbA1c levels were significantly lower in all treatment groups compared to DC (P < 0.001). Two-way repeated measures ANOVA confirmed substantial reductions in metformin, GS, and metformin + GS groups relative to DC. Pairwise comparisons revealed significant differences between metformin and metformin + GS (P < 0.05) and between metformin and GS (P < 0.05; Figure 5; Table 2).
T2DM is a progressive metabolic disorder characterized by chronic hyperglycemia, insulin resistance, and impaired insulin secretion. Its global prevalence is rising rapidly, contributing to significant health and economic burdens worldwide. Although standard treatments like metformin remain the mainstay of therapy due to their proven efficacy and safety profile, there is increasing interest in adjunctive therapies, especially those derived from traditional medicinal plants, to enhance therapeutic outcomes and minimize side effects[14].
In the present study, T2DM was successfully induced in rats using a combination of a HFD followed by low-dose STZ, effectively mimicking the dual pathophysiological mechanisms of insulin resistance and pancreatic β-cell dysfunction seen in human T2DM[11]. This model is widely recognized for evaluating the efficacy of antidiabetic agents due to its translational relevance and reproducibility.
Metformin, used as the standard comparator, significantly improved glycemic control and metabolic markers, including FBG, serum cholesterol, creatinine, and HbA1c. These findings are consistent with extensive literature highlighting metformin’s ability to suppress hepatic gluconeogenesis, enhance insulin sensitivity, and improve lipid and renal profiles[15]. The observed nephroprotective effect may be attributed to metformin’s activation of AMP-activated protein kinase, a mechanism known to protect against diabetic nephropathy and oxidative stress-related tissue damage[16].
GS, a traditional Ayurvedic herb, demonstrated moderate anti-diabetic activity. Its bioactive constituents, particularly gymnemic acids, are known to reduce intestinal glucose absorption, stimulate insulin secretion, and protect pancreatic β-cells from oxidative damage[17,18]. The modest improvements observed in the GS-treated group support its traditional use and pharmacological potential as a glucose-lowering agent. The improved reductions in cholesterol and creatinine imply beneficial effects on lipid homeostasis and kidney function. These findings are clinically relevant given the complexities of diabetic dyslipidemia and nephropathy.
Combination therapy with GS and metformin resulted in better outcomes than GS monotherapy across all measured parameters, suggesting additive effects. However, it did not significantly surpass metformin alone in most biochemical markers. This indicates that while GS may complement metformin through additional mechanisms, the interaction is likely additive rather than synergistic[11,18]. Such herbal-drug combinations have been reported to provide multi-targeted benefits by modulating glycemic control, lipid metabolism, oxidative stress, and β-cell protection[19].
Notably, the combination therapy also improved cholesterol and creatinine levels, suggesting potential lipid-lowering and renoprotective benefits. This is of clinical importance given the high prevalence of dyslipidemia and nephropathy among diabetic patients[15,20]. These effects may be due to the antioxidant and anti-inflammatory properties of GS compounds acting in concert with metformin’s metabolic regulation. Despite these promising findings, the relatively lower efficacy of GS alone emphasizes the necessity of standard pharmacotherapy, particularly in cases of moderate to severe hyperglycemia. However, its role as an adjunct especially in patients preferring integrative medicine or those intolerant to higher doses of metformin warrants further exploration[21].
This study has certain limitations. The short duration of treatment (28 days) and small sample size (n = 6 per group) limit the assessment of long-term metabolic effects, disease progression, and chronic safety. Advanced metabolic assessments such as oral glucose tolerance test and insulin tolerance test were not performed. Histopathological evaluation of vital organs, including the pancreas, kidneys, and liver, was not included. In addition, molecular and mechanistic studies were not conducted, and the phytochemical composition of the GS extract was not characterized.
This study demonstrated that both metformin and GS, alone and in combination, exert significant anti-diabetic effects in STZ-induced T2DM rats. Metformin exhibited superior efficacy in lowering FBG, HbA1c, serum cholesterol, and creatinine levels. GS provided modest yet significant benefits, and its combination with metformin yielded additive improvements. These findings support the potential utility of GS as an adjunct therapy, though further research is necessary to validate its clinical applicability and long-term safety.
| 1. | American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2014;37 Suppl 1:S81-S90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4149] [Cited by in RCA: 3548] [Article Influence: 295.7] [Reference Citation Analysis (2)] |
| 2. | DeFronzo RA. Pharmacologic therapy for type 2 diabetes mellitus. Ann Intern Med. 1999;131:281-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 840] [Cited by in RCA: 727] [Article Influence: 26.9] [Reference Citation Analysis (1)] |
| 3. | Rena G, Hardie DG, Pearson ER. The mechanisms of action of metformin. Diabetologia. 2017;60:1577-1585. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1875] [Cited by in RCA: 1582] [Article Influence: 175.8] [Reference Citation Analysis (2)] |
| 4. | Bailey CJ, Turner RC. Metformin. N Engl J Med. 1996;334:574-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1624] [Cited by in RCA: 1364] [Article Influence: 45.5] [Reference Citation Analysis (1)] |
| 5. | Tiwari P, Mishra BN, Sangwan NS. Phytochemical and pharmacological properties of Gymnema sylvestre: an important medicinal plant. Biomed Res Int. 2014;2014:830285. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 163] [Cited by in RCA: 93] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 6. | Shanmugasundaram ER, Rajeswari G, Baskaran K, Rajesh Kumar BR, Radha Shanmugasundaram K, Kizar Ahmath B. Use of Gymnema sylvestre leaf extract in the control of blood glucose in insulin-dependent diabetes mellitus. J Ethnopharmacol. 1990;30:281-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 103] [Article Influence: 2.9] [Reference Citation Analysis (2)] |
| 7. | Baskaran K, Kizar Ahamath B, Radha Shanmugasundaram K, Shanmugasundaram ER. Antidiabetic effect of a leaf extract from Gymnema sylvestre in non-insulin-dependent diabetes mellitus patients. J Ethnopharmacol. 1990;30:295-300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 141] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 8. | Lenzen S. The mechanisms of alloxan- and streptozotocin-induced diabetes. Diabetologia. 2008;51:216-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1553] [Cited by in RCA: 1233] [Article Influence: 68.5] [Reference Citation Analysis (0)] |
| 9. | Srinivasan K, Viswanad B, Asrat L, Kaul CL, Ramarao P. Combination of high-fat diet-fed and low-dose streptozotocin-treated rat: a model for type 2 diabetes and pharmacological screening. Pharmacol Res. 2005;52:313-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1453] [Cited by in RCA: 1309] [Article Influence: 62.3] [Reference Citation Analysis (1)] |
| 10. | Arifin WN, Zahiruddin WM. Sample Size Calculation in Animal Studies Using Resource Equation Approach. Malays J Med Sci. 2017;24:101-105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 631] [Cited by in RCA: 490] [Article Influence: 54.4] [Reference Citation Analysis (0)] |
| 11. | Furman BL. Streptozotocin-Induced Diabetic Models in Mice and Rats. Curr Protoc Pharmacol. 2015;70:5.47.1-5.47.20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 825] [Cited by in RCA: 745] [Article Influence: 67.7] [Reference Citation Analysis (1)] |
| 12. | Charan J, Kantharia ND. How to calculate sample size in animal studies? J Pharmacol Pharmacother. 2013;4:303-306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1468] [Cited by in RCA: 1348] [Article Influence: 103.7] [Reference Citation Analysis (1)] |
| 13. | Mall GK, Mishra PK, Prakash V. Antidiabetic and hypolipidemic activity of Gymnemasylvestre in alloxan induced diabetic rats. Global J Biotech Biochem. 2009;4:37-42. |
| 14. | Galicia-Garcia U, Benito-Vicente A, Jebari S, Larrea-Sebal A, Siddiqi H, Uribe KB, Ostolaza H, Martín C. Pathophysiology of Type 2 Diabetes Mellitus. Int J Mol Sci. 2020;21:6275. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2494] [Cited by in RCA: 1756] [Article Influence: 292.7] [Reference Citation Analysis (15)] |
| 15. | Miura T, Itoh C, Iwamoto N, Kato M, Kawai M, Park SR, Suzuki I. Hypoglycemic activity of the fruit of the Momordica charantia in type 2 diabetic mice. J Nutr Sci Vitaminol (Tokyo). 2001;47:340-344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 73] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 16. | Chakrabarti S, Biswas TK, Rokeya B, Ali L, Mosihuzzaman M, Nahar N, Khan AK, Mukherjee B. Advanced studies on the hypoglycemic effect of Caesalpinia bonducella F. in type 1 and 2 diabetes in Long Evans rats. J Ethnopharmacol. 2003;84:41-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 43] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 17. | Kim MJ, Ryu GR, Chung JS, Sim SS, Min DS, Rhie DJ, Yoon SH, Hahn SJ, Kim MS, Jo YH. Protective effects of epicatechin against the toxic effects of streptozotocin on rat pancreatic islets: in vivo and in vitro. Pancreas. 2003;26:292-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 87] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 18. | Eddouks M, Maghrani M, Lemhadri A, Ouahidi ML, Jouad H. Ethnopharmacological survey of medicinal plants used for the treatment of diabetes mellitus, hypertension and cardiac diseases in the south-east region of Morocco (Tafilalet). J Ethnopharmacol. 2002;82:97-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 425] [Cited by in RCA: 300] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 19. | Hasani-Ranjbar S, Larijani B, Abdollahi M. A systematic review of the potential herbal sources of future drugs effective in oxidant-related diseases. Inflamm Allergy Drug Targets. 2009;8:2-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 108] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 20. | Akinlade OM, Owoyele BV, Soladoye AO. Streptozotocin-induced type 1 and 2 diabetes in rodents: a model for studying diabetic cardiac autonomic neuropathy. Afr Health Sci. 2021;21:719-727. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 21. | Zhang R, Cheng K, Xu S, Li S, Zhou Y, Zhou S, Kong R, Li L, Li J, Feng J, Wu L, Liu T, Xia Y, Lu J, Guo C, Zhou Y. Metformin and Diammonium Glycyrrhizinate Enteric-Coated Capsule versus Metformin Alone versus Diammonium Glycyrrhizinate Enteric-Coated Capsule Alone in Patients with Nonalcoholic Fatty Liver Disease and Type 2 Diabetes Mellitus. Gastroenterol Res Pract. 2017;2017:8491742. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 2.1] [Reference Citation Analysis (1)] |