Published online Mar 9, 2026. doi: 10.5492/wjccm.v15.i1.114620
Revised: November 3, 2025
Accepted: December 19, 2025
Published online: March 9, 2026
Processing time: 156 Days and 16.3 Hours
Long coronavirus disease (COVID) is a condition characterized by persistent health issues following severe acute respiratory syndrome coronavirus 2 infection. The condition remains poorly understood, especially in terms of long-term impact on health and the quality of life. This study hypothesized that majority of the discharged patients experience long-term post-COVID-19 complications.
To evaluate the long-term post-COVID-19 complications and its impact on the patients’ quality of life.
This retrospective cohort study, with telephonic interview-based follow-up, was conducted at a tertiary care hospital in western India between March and August 2024. The medical records of the patients hospitalized with COVID-19 during the second wave (between March and June 2021) and discharged, were reviewed. The data were collected from the patients via structured telephonic interviews that focused on post-infection sequelae across various bodily systems and was summarized using percentages and proportions.
A total of 1139 patients who met the inclusion criteria, participated in the study with a follow-up period of three years. Amongst the survivors (n = 1052) at the end of three years, 150 (14.25%) developed new or ongoing diseases after recovery from acute COVID-19, while 51 (4.8%) were still under treatment at the time of follow-up. Amongst these 150 long-COVID-19 patients, pulmonary disease (n = 27, 2.57%), body pain (n = 20, 1.90%), coronary artery disease or angioplasty, and diabetes mellitus (n = 17, 1.61% each), hypertension (n = 16, 1.52%), and fatigue (n = 13, 1.24%) were frequently reported. Although statistically insignificant, the patients who received three or more vaccine doses after the second wave of the pandemic reported slightly lower rates of post-COVID-19 morbidity and treatment requirements.
The current study highlights the burdens of long-term complications following COVID-19 infection, with a broad spectrum of post-infection sequelae. However, the impact of vaccination on the course of development and treatment of long COVID could not be ascertained. This finding emphasizes the need for continued research and healthcare planning to address the persistent impact of COVID-19 upon the survivors.
Core Tip: Long coronavirus disease (COVID) can persist even after three years. In this study, we found that 14.3% of the discharged patients developed persistent systemic symptoms, and about 4.8% (n = 51) of patients are still undergoing treatment for post-COVID-19-related illnesses. Patients report a wide variety of symptoms ranging from pulmonary (2.57%), chronic body aches (1.9%), cardiovascular (1.61%), and diabetes (1.61%) to general fatigue (1.24%). Vaccination status was not associated with the development, course, or treatment status of post-COVID-19 disease (P value: 0.82, 0.21, and 0.12, respectively).
- Citation: Jain A, Saraswat P, Sharma A, Sharma V, Jain R. Persistent health complications in COVID-19 hospitalized patients at tertiary care hospital in Western India. World J Crit Care Med 2026; 15(1): 114620
- URL: https://www.wjgnet.com/2220-3141/full/v15/i1/114620.htm
- DOI: https://dx.doi.org/10.5492/wjccm.v15.i1.114620
The Centers for Disease Control and Prevention defines long coronavirus disease (COVID) as a condition characterized by signs, symptoms, and health issues that persist or emerge after the initial infection with COVID-19 or severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)[1].
Long COVID is not a single condition, rather a group of overlapping systemic conditions that possibly exhibit distinct biological mechanisms, risk factors, and outcomes that cannot be attributed to an alternative diagnosis[2]. The clinical presentation can be highly heterogeneous, with over 200 different symptoms reported for the condition[3]. These symptoms can appear after four weeks of the initial infection or even later. It can affect multiple organ systems, may relapse or worsen over time. Sometimes, it can also lead to serious long-term complications. The duration of the sym
Despite the proliferation of patient support groups, clinical surveys, patient interviews, author comments, and scientific literature that aimed at elucidating the chronic nature of COVID-19, the need for a comprehensive overview of potential long-term effects still exists. Thus, the present retrospective observational study was conducted with an aim at evaluating the long-term effects of COVID-19, which continue to manifest more than three years after the initial infection.
This retrospective cohort study, with telephonic interview-based follow-up, was carried out at Mahatma Gandhi Medical College and Hospital, Rajasthan, India, a tertiary care hospital, from March 2024 to August 2024, spanning a period of six months. The study was planned and conducted according to the standards prescribed in STROBE guidelines. The data regarding admissions and hospitalizations were gathered from the medical records of all the patients aged 18 years and above and diagnosed with COVID-19 during the second wave of the pandemic, specifically between March and June 2021. The diagnosis of the patients was confirmed via real-time reverse transcription polymerase chain reaction using nasopharyngeal or upper respiratory tract samples. Patients, who were discharged within 24 hours of their hospitalization, were excluded from the study. Based on the inclusion criteria, the patients discharged and surviving at the time of study were identified. After obtaining a formal telephonic consent for voluntary participation and data sharing for academic purpose, the participants were involved in the study. A structured telephonic interview was conducted to assess the post-COVID sequelae across all the organ system domains (neurological, cardiovascular, musculoskeletal, pulmonary, metabolic, and psychological). The telephonic interview also covered the details of their demographics, symptoms during and after COVID-19, hospitalization status, vaccination after infection, any newly diagnosed conditions, and outpatient treatment for persistent COVID-related symptoms. The study was reviewed and approved by the institutional ethics committee (Mahatma Gandhi University of medical science & technology, Jaipur) (approval No. /MGMC&H/IEC/JPR/2023/1705, date: November 2, 2023) before the commencement of the study.
The final tabulation and documentation of the interviewed subjective data were done using Microsoft Office 365 (Microsoft Corp., 2024). The foundation data tables were prepared after formal tabulation. The results are expressed in frequencies and proportions for categorical variables. Continuous variables were tested for normality using the Shapiro-Wilk test and reported as mean ± SD or median, as appropriate. Additionally, the vaccination history of the patients was evaluated to assess its association with post-COVID-19 syndrome. All the statistical tests were two-tailed, with a P value < 0.05 considered to be statistically significant. All the statistical analyses were conducted utilizing SPSS software (version 25.0, IBM SPSS Inc., Chicago, IL, United States).
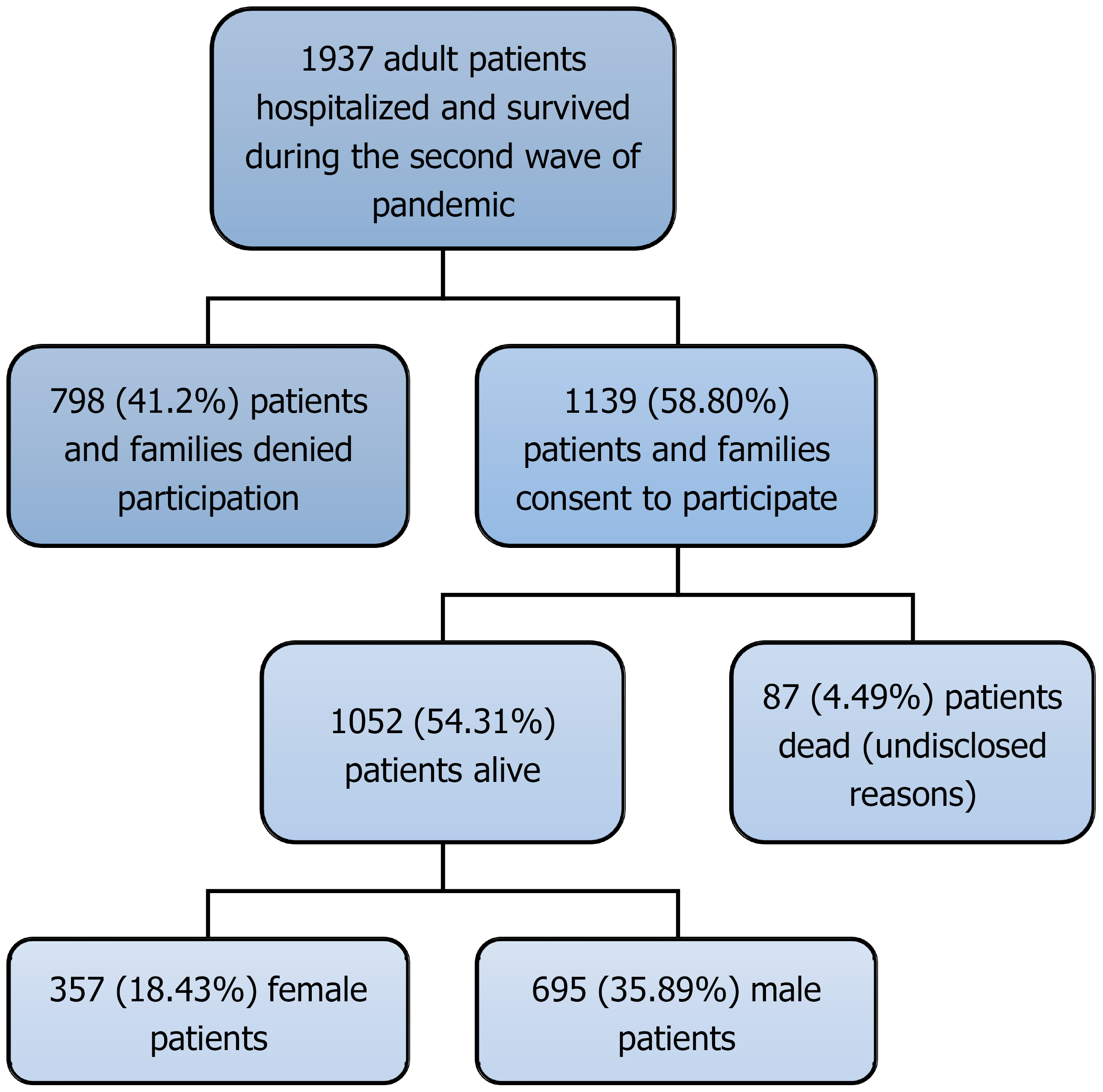
A total of 1937 adult patients were hospitalized and discharged from the study hospital during the second wave of the pandemic, out of which 1139 (58.8%) patients and their families agreed to participate in the study. Males comprised the majority of this cohort (n = 746, 65.5%) compared to their counterparts (females; n = 393, 34.5%). Mortality appeared slightly higher among females (n = 36, 9.16%) than males (n = 51, 6.84%); however, this difference was not statistically significant (P = 0.16). Out of the total 1139 respondents, 1052 (92.36%) were alive at the time of follow-up, i.e., approximately three years after acute COVID-19 infection, while 87 (7.64%) had died. The families of these 87 patients were unable to provide the exact details of the patient’s death scenarios; hence, they were excluded from further analysis (Figure 1).
Among the study population (n = 1052), 150 individuals (14.25%) reported developing a new disease after initial COVID-19 infection. Additionally, 51 patients (4.85%) were under ongoing treatment during the follow-up period. Further, 33 individuals (3.14%) were under treatment for complications that arose during the post-COVID period, and 18 (1.71%) were receiving care for pre-existing conditions. The primary reasons behind pre-COVID treatment were endocrinology disorders (n = 8, 0.76%), cardiac conditions (n = 6, 0.57%), and pulmonary or musculoskeletal issues (n = 2, 0.19% each). The treatments related to post-COVID complications were largely attributed to endocrinology complications (n = 18, 1.71%), followed by cardiac (n = 14, 1.33%), pulmonary (n = 10, 0.95%) and musculoskeletal (n = 6, 0.57%), neurological/psychological (n = 2, 0.19%), and sensory issues such as vision or hearing problems (n = 1, 0.09%).
Out of the 150 patients reporting new diseases post-COVID, the most frequently observed conditions included lung disease (n = 27, 2.57%), body pain (n = 20, 1.90%), coronary artery disease or angioplasty and diabetes mellitus (n = 17, 1.61% each), hypertension (n = 16, 1.52%), and fatigue (n = 13, 1.24%). The least observed conditions (n = 50, 4.75%) in
The vast majority of the study participants (n = 1041, 98.95%) were vaccinated against COVID-19 while only 11 in
| Alive | Expired | Total | P value | ||
| Count among the discharged moderate to severe COVID-19 patients | n | 1052 | 87 | 1139 | |
| Percentage | 92.36% | 7.64% | 100% | ||
| Gender distribution | Female | 357 | 36 | 393 | 0.16 |
| 90.84% | 9.16% | 100% | |||
| Male | 695 | 51 | 746 | ||
| 93.16% | 6.84% | 100% |
| Among the alive patients (n = 1052) | Count | Percentage (%) | |
| Complications developed after COVID-19 | Yes | 150 | 14.25 |
| Complications requiring treatment | Yes | 51 | 4.85 |
| Post COVID-19 complications management | Post-COVID | 33 | 3.14 |
| Post-COVID management of pre-existing conditions | Pre-COVID | 18 | 1.71 |
| Reason for management of pre-existing conditions post-COVID | Endocrinology | 8 | 0.76 |
| Heart | 6 | 0.57 | |
| Lung | 2 | 0.19 | |
| Muscle/bone | 2 | 0.19 | |
| Reason for treatment of post COVID-19 complications | Endocrinology | 18 | 1.71 |
| Heart | 14 | 1.33 | |
| Lung | 10 | 0.95 | |
| Muscle/bone | 6 | 0.57 | |
| Neurological psychology | 2 | 0.19 | |
| Vision/hearing | 1 | 0.09 | |
| Complications developed after COVID-19 | Yes | 150 | 14.25 |
| Lung disease | Yes | 27 | 2.57 |
| Body pain | Yes | 20 | 1.90 |
| Coronary artery disease/angioplasty | Yes | 17 | 1.61 |
| Diabetes mellitus | Yes | 17 | 1.61 |
| Hypertension | Yes | 16 | 1.52 |
| Fatigue | Yes | 13 | 1.24 |
| Arthritis | Yes | 7 | 0.66 |
| Stroke | Yes | 7 | 0.66 |
| Kidney disease | Yes | 5 | 0.47 |
| Asthma | Yes | 4 | 0.38 |
| Thyroid disease | Yes | 4 | 0.38 |
| Headache/dizziness | Yes | 4 | 0.38 |
| Cancer/chemotherapy | Yes | 3 | 0.28 |
| Haemorrhoids | Yes | 2 | 0.19 |
| Alopecia | Yes | 2 | 0.19 |
| Allergy rhinitis | Yes | 2 | 0.19 |
| Gastroenteritis | Yes | 2 | 0.19 |
| Polycystic ovarian disease | Yes | 1 | 0.09 |
| Tuberculosis | Yes | 1 | 0.09 |
| Allergy | Yes | 1 | 0.09 |
| Anxiety | Yes | 1 | 0.09 |
| Clotting | Yes | 1 | 0.09 |
| Liver disease | Yes | 1 | 0.09 |
| Fungemia | Yes | 1 | 0.09 |
| backbone surgery | Yes | 1 | 0.09 |
| Vaccination status | Yes | 1041 | 98.95 |
| Booster dose of vaccine taken | Yes | 816 | 77.57 |
| Type/name of vaccine | COVAXIN | 243 | 22.24 |
| COVISHIELD | 771 | 73.29 | |
| SPUTNIK | 7 | 0.66 | |
| Total doses of vaccination | 0 | 11 | 1.04 |
| 1 | 103 | 9.79 | |
| 2 | 122 | 11.59 | |
| 3 | 816 | 77.57 | |
| Follow up hospital visit | Yes | 179 | 17.01 |
Analysis of the vaccination status, in relation to post-COVID-19 morbidity, revealed that the incidence of new post-COVID-19 disease was slightly lower (14.3%) in individuals who had received three or more doses compared to those with a lesser number of doses (ranging from 11.7% to 18.2%). However, this difference was not statistically significant
| Total number of vaccines | Total | P value | |||||
| 0 | 1 | 2 | 3 and above | ||||
| 11 (1.04) | 103 (9.79) | 122 (11.59) | 816 (77.57) | 1052 (100) | |||
| Ongoing treatment for post COVID-19 complication | Yes | 2 (18.2) | 3 (2.9) | 8 (6.6) | 38 (4.7) | 51 (4.8) | 0.116 |
| Ongoing management for type of complications | Complications developed after COVID-19 | 1 (9.1) | 1 (1.0) | 7 (5.7) | 24 (2.9) | 33 (3.1) | 0.21 |
| Post-COVID management of pre-existing conditions | 1 (9.1) | 2 (1.9) | 1 (0.8) | 14 (1.7) | 18 (1.7) | ||
| Disease developed after COVID-19 | Yes | 2 (18.2) | 12 (11.7) | 19 (15.6) | 117 (14.3) | 150 (14.3) | 0.81 |
Finally, 179 participants (17.0%) reported at least one hospital follow-up visit during their post-COVID recovery period, indicating a modest level of healthcare utilization amongst this group of participants.
The clinical spectrum of long COVID is extensive and affects multiple organ systems. In this study, 150 respondents (14.25% of COVID-19 survivors) reported post-COVID-19 illnesses after three years of acute infection, and 51 individuals (4.85%) were still undergoing treatment. Amongst the survivors, the most commonly reported post-COVID-19 complications included lung disease (n = 27, 2.57%), chronic body pain (n = 20, 1.90%), coronary artery disease (n = 17, 1.61%), diabetes (n = 17, 1.61%), hypertension (n = 16, 1.52%), general fatigue (n = 13, 1.24%), and arthritis (n = 7, 0.66%).
In this study, 14.25% of the survivors reported post-COVID-19 illness. Similar to this finding, a systematic review by Rahmati et al[8] pooled data from 11 relevant studies published internationally. The authors found that 20% (95%CI: 8-43) patients experienced at least one persistent symptom after three years of acute infection. The study reported dyspnea (12%, 95%CI: 10-15), fatigue (11%, 95%CI: 6-20), insomnia (11%, 95%CI: 2-37), and anxiety (6%, 95%CI: 1-32) as other common persisting symptoms. The current study also found that ‘impaired respiratory function’ was the most frequently reported complication (n = 27, 2.57%) amongst the survivors, despite its low frequency. Another review by Davis et al[9] reported a similar finding, i.e., at least 10% overall incidence of long COVID, with pulmonary and cardiovascular sym
Cardiovascular complications were identified among 17 (1.61%) participants in the current study. Cai et al[10] reported a significant cardiovascular risk burden even after three years post-COVID-19 amongst the survivors. The authors reported that the risk of death declined over the years, yet it remained high even after three years amongst the hospitalized COVID-19 patients (incidence rate ratio: 1.29, 95%CI: 1.19-1.40). Another study by Roca-Fernandez et al[11] reported that every 1 in 5 patients with long COVID suffer from cardiac impairment at 6 months, while this impairment persists among half of the patients over a 12-month period. The authors further added that the biomarkers are unable to identify this impairment[11].
In the current study, the prevalence of general body complications, like chronic body pain (n = 20, 1.90%), general fatigue (n = 13, 1.24%), arthritis (n = 7, 0.66%) and neuropsychiatric complications (cerebrovascular stroke, central nervous system disease, and anxiety) was very low among the survivors after three years. While a study by Berentschot et al[12] reported worsening of memory [odds ratio: 1.4, (1.1-1.7), P < 0.001], high fatigue score [mean difference (MD) + 1.0, (0.4-1.6), P = 0.002] and cognitive failure [MD + 2.2 (0.9-3.4), P < 0.001] between second and third year of follow-up. In their study, the patients experienced fatigue (66%), impaired fitness (63%) and memory problems (59%) at the end of three years. The authors also reported 42% intensive care unit treated survivors in their study, which may be a contributory factor for this exceptional prevalence of post-COVID-19 symptoms[12].
Kim et al[13] reported that 59.8% of the patients experienced at least one long COVID symptom at six months. Cognitive dysfunction (26.5%), amnesia (25.8%), depression (25.0%), anxiety (24.2%), and concentration difficulty (23.5%) were prevalent, with many symptoms persisting at 12 months and even 24 months. Symptoms like alopecia, dizziness, and paresthesia were still present among 15%-30% of the patients after two years[13].
A study by Mahmud et al[7] highlighted the following risk factors for long COVID, such as female gender, hospitalization during the acute phase, and pre-existing chronic illness. Another study reported that the quality of life following COVID-19 remained poor in the range of 23% to 67%[14].
While the impact of vaccination on long COVID differs across the studies, vaccination has been proposed largely as a protective factor against long COVID. This difference can be attributed to different methods followed by the studies, different types, duration, and the number of vaccines and their doses used across the globe. In current study, 98.95% of the 1052 respondents received at least one dose of a COVID-19 vaccine, with 77.6% receiving booster doses (three doses or above). COVISHIELD (n = 771, 74.7%) and COVAXIN (n = 243, 23.54%) were the most common vaccines used. In the state of Rajasthan, India, the vaccination drive against COVID-19 started after the second wave of pandemic (i.e., after June 2021). Hence, the study population got vaccinated post-infection. Despite wide vaccination coverage in the state, va
Trinh et al[17] also reported a lower risk of post-COVID arterial thrombosis amongst the vaccinated Norwegian cohorts, except those with existing comorbidities.
Maier et al[18] found that the vaccinated patients had milder symptoms and lower severity scores at 90 days post-infection. During the pre-Omicron phase, 28% of the unvaccinated individuals vs 18% of the vaccinated ones reported long COVID, with vaccinated individuals too experiencing fewer respiratory symptoms (P = 0.01). Al-Aly et al[19] similarly concluded that prior vaccination significantly reduced the incidence and severity of long COVID symptoms.
Strength and limitations: This study has several advantages. The study included a large regional sample of 1139 participants, probably the only study from this region of the country reporting such extensive results. The authors conducted interviews with survivors and focused on those individuals who had received COVID-19 vaccination. So, this study provides insights into the relationship between vaccination and long COVID symptoms in the current context. The study also included a wide range of long COVID symptoms with a follow-up period of three years, a first-of-its-kind report from this region in the country. Thus, the study offers valuable data on the prolonged effects of the infection pertaining to the geographical setting.
However, the study also has a few limitations. As the interviews were taken telephonically, the consent was obtained only through oral confirmation. The data is based on patient self-reports through surveys, which may lead to recall bias or inaccuracies in symptom reporting, and injudicious attribution to the COVID-19 for unrelated diseases. The authors were unable to attribute any long COVID symptoms to a particular patient variable, as the study was conducted based on the interviews and limited available information. Being a cross-sectional study, it captures information at a single point in time, and so it cannot further establish the causal relationships. Additionally, there was no clinical or laboratory veri
In summary, this study highlights that long COVID can lead to persistent multisystem involvement, with a wide range of physical and psychological symptoms. The lasting effects of long COVID that were notable among the survivors include respiratory issues, cardiovascular complications, fatigue, and neurocognitive symptoms. The prevalence of long COVID was comparable between the vaccinated and unvaccinated populations. These findings emphasize the need for continued research to better understand long COVID and develop effective preventive and therapeutic strategies to support the affected individuals and improve long-term outcomes.
The authors gratefully acknowledge the guidance and mentorship of Dr. Vinay K Kapoor, Professor of Surgical Gastroenterology, Mahatma Gandhi Medical College and Hospital, Jaipur, & Pro Vice Chancellor, Mahatma Gandhi University of Medical Sciences and Technology, Jaipur, whose initial motivation and support were instrumental in the conception and initiation of this study.
| 1. | A Long COVID Definition: A Chronic, Systemic Disease State with Profound Consequences. Washington (DC): National Academies Press (US); 2024-Jul-9 . [PubMed] |
| 2. | Liu Y, Gu X, Li H, Zhang H, Xu J. Mechanisms of long COVID: An updated review. Chin Med J Pulm Crit Care Med. 2023;1:231-240. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 3. | Shaffer L. Lots of long COVID treatment leads, but few are proven. Proc Natl Acad Sci USA. 2022;119:e2213524119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 4. | Ballering AV, van Zon SKR, Olde Hartman TC, Rosmalen JGM; Lifelines Corona Research Initiative. Persistence of somatic symptoms after COVID-19 in the Netherlands: an observational cohort study. Lancet. 2022;400:452-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 730] [Cited by in RCA: 588] [Article Influence: 147.0] [Reference Citation Analysis (3)] |
| 5. | Lopez-Leon S, Wegman-Ostrosky T, Perelman C, Sepulveda R, Rebolledo PA, Cuapio A, Villapol S. More than 50 long-term effects of COVID-19: a systematic review and meta-analysis. Sci Rep. 2021;11:16144. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1798] [Cited by in RCA: 1373] [Article Influence: 274.6] [Reference Citation Analysis (2)] |
| 6. | Gang J, Wang H, Xue X, Zhang S. Microbiota and COVID-19: Long-term and complex influencing factors. Front Microbiol. 2022;13:963488. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 37] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 7. | Mahmud R, Rahman MM, Rassel MA, Monayem FB, Sayeed SKJB, Islam MS, Islam MM. Post-COVID-19 syndrome among symptomatic COVID-19 patients: A prospective cohort study in a tertiary care center of Bangladesh. PLoS One. 2021;16:e0249644. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 511] [Cited by in RCA: 143] [Article Influence: 28.6] [Reference Citation Analysis (1)] |
| 8. | Rahmati M, Udeh R, Kang J, Dolja-Gore X, McEvoy M, Kazemi A, Soysal P, Smith L, Kenna T, Fond G, Boussat B, Nguyen DC, Do H, Tran BX, Veronese N, Yon DK, Boyer L. Long-Term Sequelae of COVID-19: A Systematic Review and Meta-Analysis of Symptoms 3 Years Post-SARS-CoV-2 Infection. J Med Virol. 2025;97:e70429. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 20] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 9. | Davis HE, McCorkell L, Vogel JM, Topol EJ. Long COVID: major findings, mechanisms and recommendations. Nat Rev Microbiol. 2023;21:133-146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3506] [Cited by in RCA: 2708] [Article Influence: 902.7] [Reference Citation Analysis (5)] |
| 10. | Cai M, Xie Y, Topol EJ, Al-Aly Z. Three-year outcomes of post-acute sequelae of COVID-19. Nat Med. 2024;30:1564-1573. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 208] [Cited by in RCA: 163] [Article Influence: 81.5] [Reference Citation Analysis (0)] |
| 11. | Roca-Fernandez A, Wamil M, Telford A, Carapella V, Borlotti A, Monteiro D, Thomaides-Brears H, Dennis A, Banerjee R, Robson MD, Lip GYH, Bull S, Heightman M, Ntusi N, Banerjee A. Cardiac impairment in Long Covid 1-year post SARS-CoV-2 infection. Eur Heart J. 2022;43:ehac544.219. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 12. | Berentschot JC, Bek LM, Drost M, van den Berg-Emons RJG, Braunstahl GJ, Ribbers GM, Aerts JGJV, Hellemons ME, Heijenbrok-Kal MH; CO-FLOW collaboration Group. Health outcomes up to 3 years and post-exertional malaise in patients after hospitalization for COVID-19: a multicentre prospective cohort study (CO-FLOW). Lancet Reg Health Eur. 2025;53:101290. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 13. | Kim Y, Bae S, Chang HH, Kim SW. Long COVID prevalence and impact on quality of life 2 years after acute COVID-19. Sci Rep. 2023;13:11207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 93] [Article Influence: 31.0] [Reference Citation Analysis (1)] |
| 14. | Malik P, Patel K, Pinto C, Jaiswal R, Tirupathi R, Pillai S, Patel U. Post-acute COVID-19 syndrome (PCS) and health-related quality of life (HRQoL)-A systematic review and meta-analysis. J Med Virol. 2022;94:253-262. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 565] [Cited by in RCA: 423] [Article Influence: 105.8] [Reference Citation Analysis (1)] |
| 15. | Brunvoll SH, Nygaard AB, Fagerland MW, Holland P, Ellingjord-Dale M, Dahl JA, Søraas A. Post-acute symptoms 3-15 months after COVID-19 among unvaccinated and vaccinated individuals with a breakthrough infection. Int J Infect Dis. 2023;126:10-13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 16. | Ayoubkhani D, Bosworth ML, King S, Pouwels KB, Glickman M, Nafilyan V, Zaccardi F, Khunti K, Alwan NA, Walker AS. Risk of Long COVID in People Infected With Severe Acute Respiratory Syndrome Coronavirus 2 After 2 Doses of a Coronavirus Disease 2019 Vaccine: Community-Based, Matched Cohort Study. Open Forum Infect Dis. 2022;9:ofac464. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 120] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 17. | Trinh NT, Jödicke AM, Català M, Mercadé-Besora N, Hayati S, Lupattelli A, Prieto-Alhambra D, Nordeng HM. Effectiveness of COVID-19 vaccines to prevent long COVID: data from Norway. Lancet Respir Med. 2024;12:e33-e34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 43] [Article Influence: 21.5] [Reference Citation Analysis (1)] |
| 18. | Maier HE, Kowalski-Dobson T, Eckard A, Gherasim C, Manthei D, Meyers A, Davis D, Bakker K, Lindsey K, Chu Z, Warsinske L, Arnold M, Buswinka A, Stoneman E, Valdez R, Gordon A. Reduction in Long COVID Symptoms and Symptom Severity in Vaccinated Compared to Unvaccinated Adults. Open Forum Infect Dis. 2024;11:ofae039. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 19. | Al-Aly Z, Bowe B, Xie Y. Long COVID after breakthrough SARS-CoV-2 infection. Nat Med. 2022;28:1461-1467. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 763] [Cited by in RCA: 601] [Article Influence: 150.3] [Reference Citation Analysis (1)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/