Published online Mar 9, 2026. doi: 10.5492/wjccm.v15.i1.114318
Revised: November 17, 2025
Accepted: January 28, 2026
Published online: March 9, 2026
Processing time: 165 Days and 11.8 Hours
Acute kidney injury (AKI) is a prevalent and common complication in critically ill patients with septic shock, associated with increased morbidity, mortality, and healthcare resource utilization in the intensive care unit (ICU). While inflammato
To evaluate the predictive value of inflammatory indices derived from standard laboratory tests as predictors of AKI in ICU patients with septic shock.
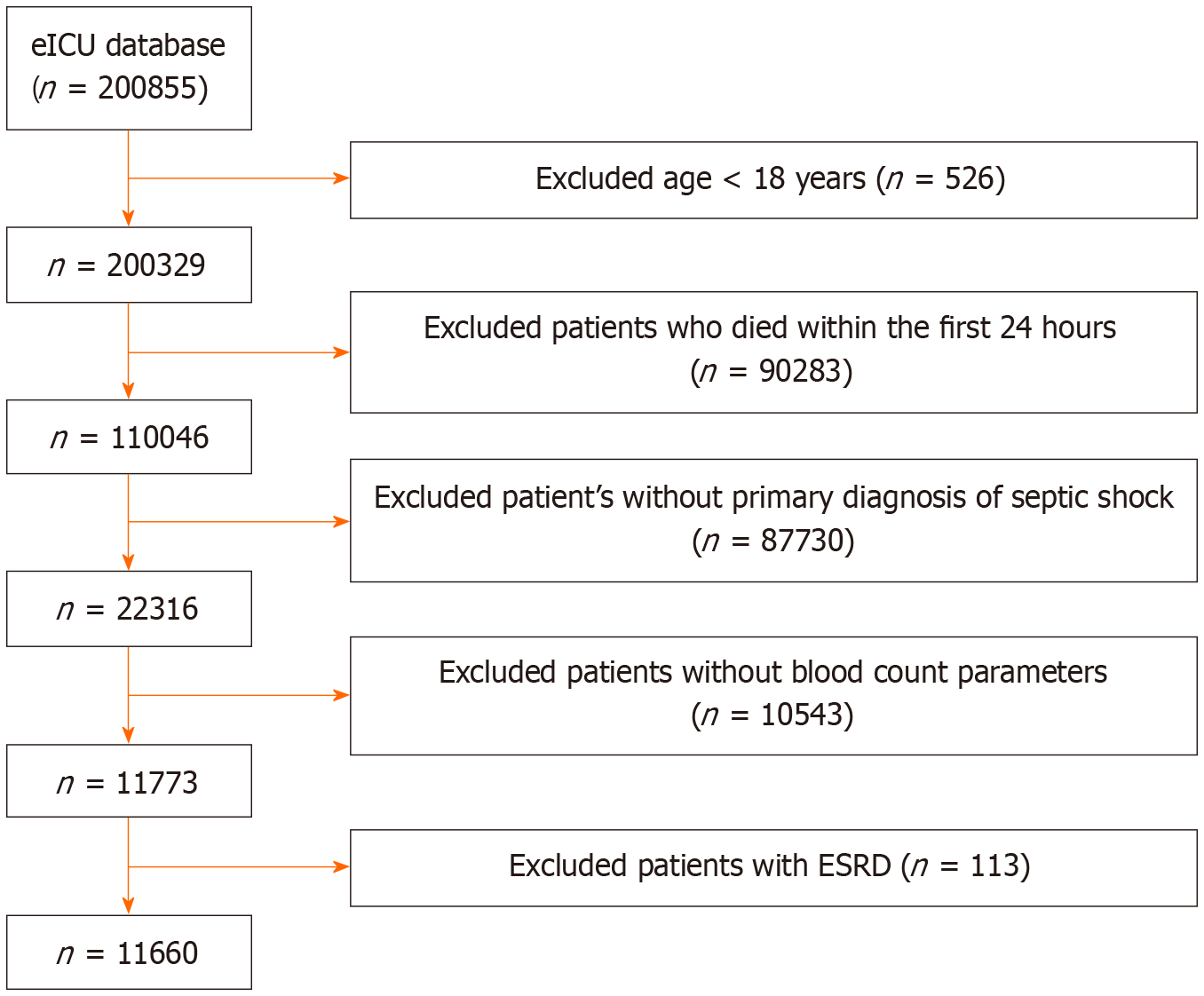
This retrospective cohort study utilized the eICU Collaborative Research Database, including adult patients with septic shock admitted to over 200 ICUs across the United States from 2014 to 2015. Patients with pre-existing end-stage renal disease, death within 24 hours, or insufficient data for inflammatory indices were excluded. Inflammatory markers (NLR, PLR, MLR, NPAR, SII, SIRI, AISI) and clinical variables were analyzed. Multivariable logi
Among 12660 septic shock patients, 6552 (51.7%) developed AKI during their ICU stay. Patients with AKI were older, had higher body mass index and Sequential Organ Failure Assessment scores, and a greater burden of comorbidities such as chronic kidney disease and diabetes. Univariate analysis showed significantly higher levels of NLR, MLR, SII, NPAR, SIRI, and AISI in the AKI group, suggesting an association between systemic inflammation and kidney injury. However, these indices displayed strong multicollinearity with other clinical and laboratory variables. In logistic regression, traditional predictors such as baseline serum creatinine, blood urea nitrogen, Sequential Organ Failure Assessment score, chronic kidney disease, vasopressor use, and selected como
In ICU patients with septic shock, composite inflammatory indices are elevated in those who develop AKI and may serve as important markers of risk. However, after accounting for multicollinearity and confounding, these mar
Core Tip: Composite inflammatory markers are elevated in patients who develop acute kidney injury. However, due to heterogeneity of septic shock, multicollinearity and nonlinear relationships, these markers alone offer limited incremental predictive value. Neural network models further expounded the contribution of both clinical factors and the combined inflammatory/metabolic dimension to accurate acute kidney injury prediction, capturing complex interactions and non-linear relationships not evident in traditional regression models. Implementation of supervised and unsupervised machine learning together may offer further insights.
- Citation: Rajendran J, Ang SP, Lorenzo-Capps MJ, Valladares C, Lee E, Bommu VJL, Altarcha G, Pominov S, Gregory B, Chia JE, Iglesias J. Predicting acute kidney injury in septic shock patients using inflammatory indices in the intensive care unit. World J Crit Care Med 2026; 15(1): 114318
- URL: https://www.wjgnet.com/2220-3141/full/v15/i1/114318.htm
- DOI: https://dx.doi.org/10.5492/wjccm.v15.i1.114318
Acute kidney injury (AKI) remains a frequent and common complication in critically ill patients and is associated with extended intensive care unit (ICU) stays, need for renal replacement therapy (RRT), and increased mortality[1-3]. The underlying mechanisms of AKI in this population are complex, often involving a combination of hemodynamic ins
Timely identification of patients at risk for AKI and its severe forms – especially those who might require hemodialy
In recent years, inflammatory indices derived from routine laboratory parameters – including the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), monocyte-to-lymphocyte ratio (MLR), neutrophil-percen
To determine the role of admission inflammatory markers obtained from routine laboratory testing, we evaluated comorbidities, demographic data, medications, and other clinical laboratory values in the development of septic shock-associated AKI. We performed a retrospective cohort study employing the eICU Collaborative Research Database. The eICU Collaborative Research Database collects de-identified patient data from over 200 ICUs across the United States between 2014 and 2015[15]. The eICU database is a rigorously curated, multicenter dataset developed by the Mass
The eICU database is publicly available after registration, users complete a training course in research with human subjects, and sign a data use agreement that mandates responsible handling of the data and collaborative research[15].
Due to its retrospective design, patient de-identification and the security schema, for which the re-identification risk was certified as meeting safe harbor standards by an independent privacy expert (Privacert, Cambridge, MA, United States) (Health Insurance Portability and Accountability Act Certification, No. 1031219-2). The use of the eICU database is exempt from institutional review board approval[15]. IBM SPSS (IBM Chicago, IL, United States) version 27 and STATA version 16 (STATACORP LLC, College Station, TX, United States) programs were used to analyze data and generate images.
Patients admitted to the ICU with septic shock were defined on admission according to the International Classification of Diseases, 10th Revision, Clinical Modification codes. Individuals greater than 17 years of age were included in the ana
Demographic variables (gender, age, ethnicity), clinical information, comorbidities, the need for RRT, requirement for mechanical ventilation, and initial laboratory parameters were extracted from electronic health records. Comorbidities included in analysis were extracted from the database were based on the International Classification of Diseases, 10th Revision, Clinical Modification coding and included chronic kidney disease (CKD), diabetes mellitus (DM), hypertension (HTN), malignancy, congestive heart failure, cirrhosis and chronic obstructive pulmonary disease (COPD). Included in the current analysis were medications known to impact renal hemodynamics such as angiotensin converting enzyme inhibitor (ACEi), angiotensin 2 receptor antagonist (A2RB), diuretics and vasopressors/inotropic agents. Inflammatory markers easily obtained from routine laboratory studies were calculated for the analysis, NLR, PLR, MLR, NPAR, SII, SIRI, and AISI. The formulas utilized for the computation of these ratios are as follows: (1) NLR = neutrophil count (NC)/Lymphocyte count (LC); (2) PLR = platelet count (PC)/LC; (3) MLR = monocyte count (MC)/LC; (4) SII = PC × NC/LC; (5) NPAR = neutrophil percentage of total white blood cell count (%)× 100/albumin (g/dL); (6) SIRI = NC × MC/LC; and (7) AISI = NC × PC × MC/LC. Additionally, admission lab values and clinical information evaluated included alanine aminotransferase, aspartate aminotransferase, total bilirubin, serum sodium, serum potassium, serum chloride, hem
AKI was defined according to Kidney Disease Global Outcomes guidelines, briefly a rise in serum creatinine (SCr) of ≥ 0.3 mg/dL within 48 hours, a rise in SCr of ≥ 1.5 mg/dL above baseline within 7 days, and or the need for RRT[16]. The baseline SCr was taken as the lowest SCr obtainable within the first 7 days of admission.
The primary outcome of concern is the development of AKI in ICU subjects with septic shock.
In order to determine risk factors for the development of AKI, we performed univariate analysis, supervised (mul
To determine risk factors that were independently associated with the outcome of AKI, we performed multivariate analysis by logistic regression with stepwise forward variable selection. We employed the Omnibus test of model coefficients to determine the statistical significance of the model as a whole, and the Goodness of fit was determined by the Hosmer-Lemeshow test. For continuous variables, the odds ratio (OR) represents the relative amount by which the OR for the outcome variable increases or decreases when the independent variable is changed by exactly one unit. ORs and their 95%CI were determined by exponentiation of the beta coefficient and its upper and lower confidence interval, respectively. Assessment of multicollinearity between predictors was performed by employing a linear regression using the previous independent variables entered into the logistic regression and calculating variance inflation factors (VIF). Problematic multicollinearity was defined as a VIF greater than 5. Additionally, multicollinearity was established if the condition index was found to be greater than 30 and the variance proportions were greater than 0.5, respectively.
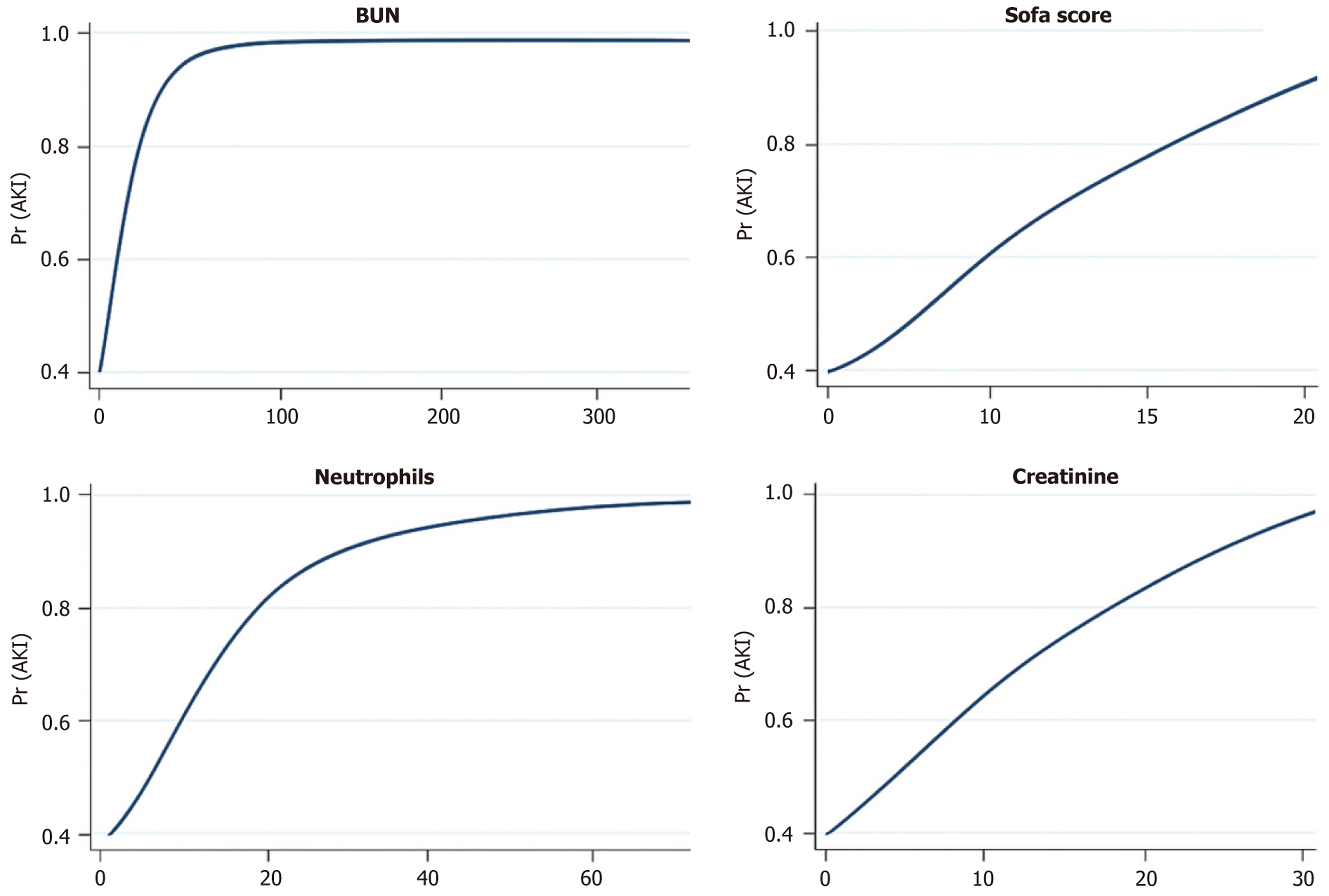
A two-step approach was used to investigate the presence of non-linear relationships. The presence of non-linear relationships between continuous variables and the development of AKI was explored by creating quadratic terms of the continuous variables and entering them into the logistic model. Non-linear relationships were demonstrated if both the continuous variable and the quadratic expression were statistically significant. Predicted probability plots were gene
As there were a large number of variables which were found to be statistically significant on univariate analysis, we next performed PCA in order to reduce the dimensionality of the data and to allow a more focused evaluation of patterns of risk factors in the development of AKI. PCA was performed using the following standardized variables obtained on admission: (1) NLR; (2) PLR; (3) MLR; (4) NPAR; (5) SII; (6) SIRI; and (7) AISI. Additionally, standardized admission lab values included blood urea nitrogen (BUN), SCr, aspartate aminotransferase, total bilirubin, serum sodium, serum pot
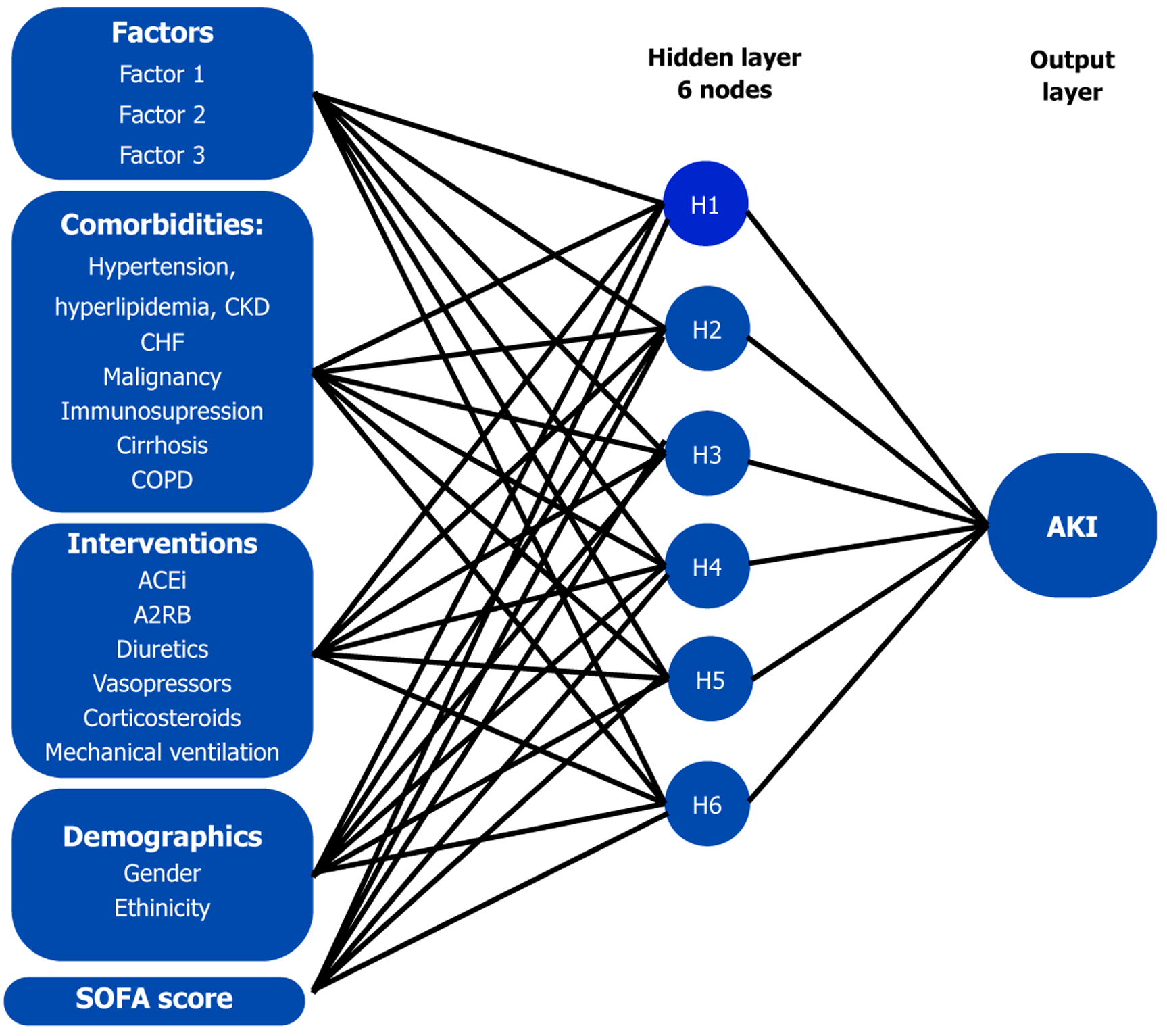
To explore complex and potentially non-linear relationships among risk factors for AKI, we employed a multilayer perceptron (MLP) neural network analysis. Inputs to the network included demographic characteristics (such as ethnicity), clinical comorbidities (including diabetes, cirrhosis, heart failure, COPD, malignancy, hyperlipidemia, HTN, and immunosuppression), use of medications (vasopressors, corticosteroids, diuretics, ACEi, angiotensin receptor blockers), and PCA-derived composite scores (factors 1-3) for inflammatory and renal/metabolic and age/electrolyte dimensions, in addition to SOFA score. The dataset was randomly partitioned, with 70% of cases used for training and 30% for testing. Network architecture and the number of hidden units were optimized automatically. Model performance was assessed by classification accuracy and area under the receiver operating characteristic curve, calibration plot and quantified by the Brier score and Hosmer-Lemeshow goodness of fit[18]. The Brier score measures the performance accuracy of probabilistic predictions across decile grouping in the calibration plot[18]. In addition to employing AUC for the importance of the neural network, a decision curve analysis (DCA) was constructed comparing the advantage of the following strategies: (1) Treating all; (2) Not treating; and (3) Employing the neural network model. A performance me
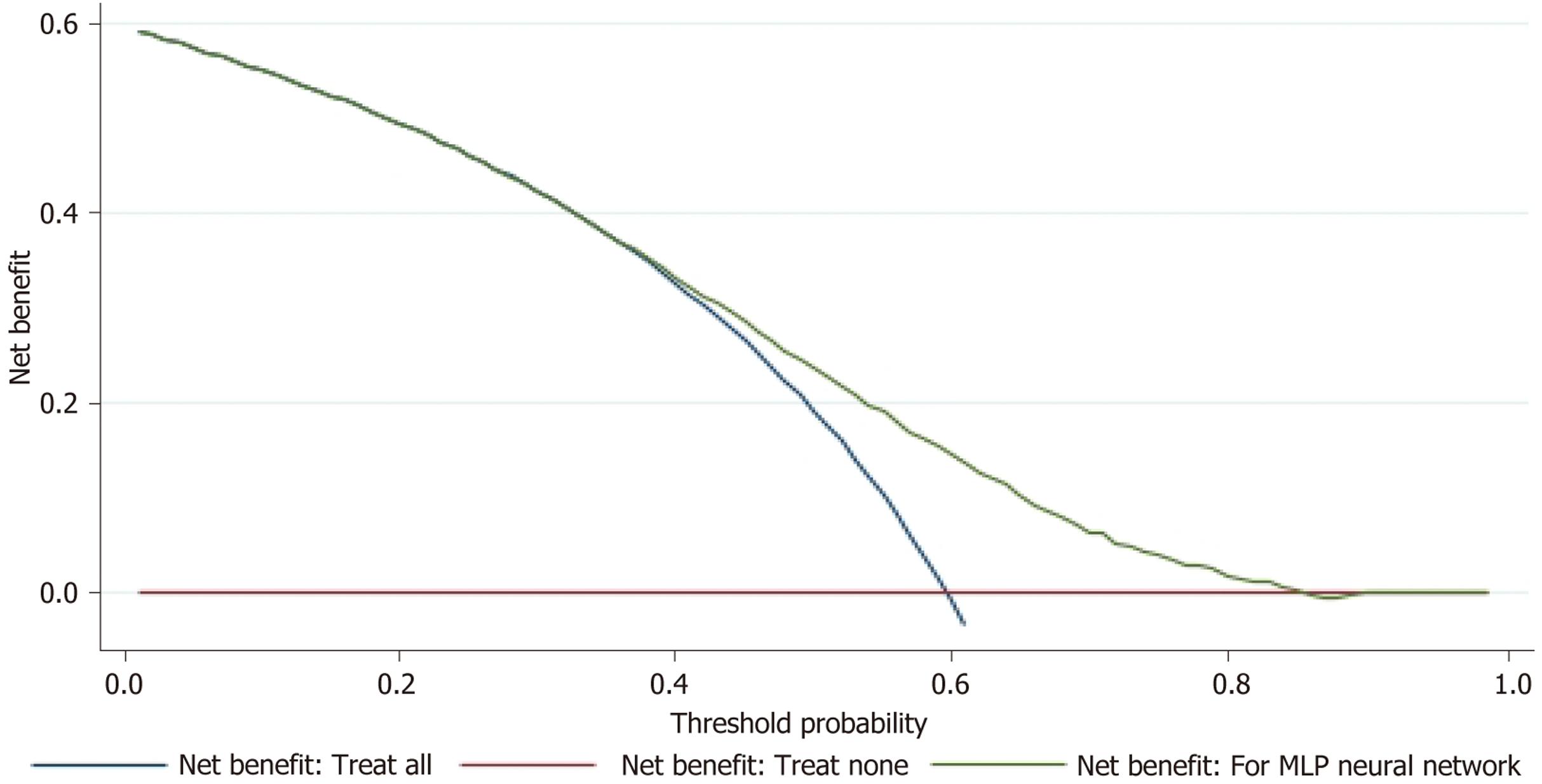
To assess the predictive utility of the MLP neural network, a DCA was performed using the DCA in STATA. The neural network generated predictive probabilities, and the observed outcome status was analyzed employing DCA. DCA evaluated the net benefit over the range of relevant threshold probabilities, comparing the neural network-guided decision and intervention to two reference strategies: (1) Treating all subjects; and (2) Treating none.
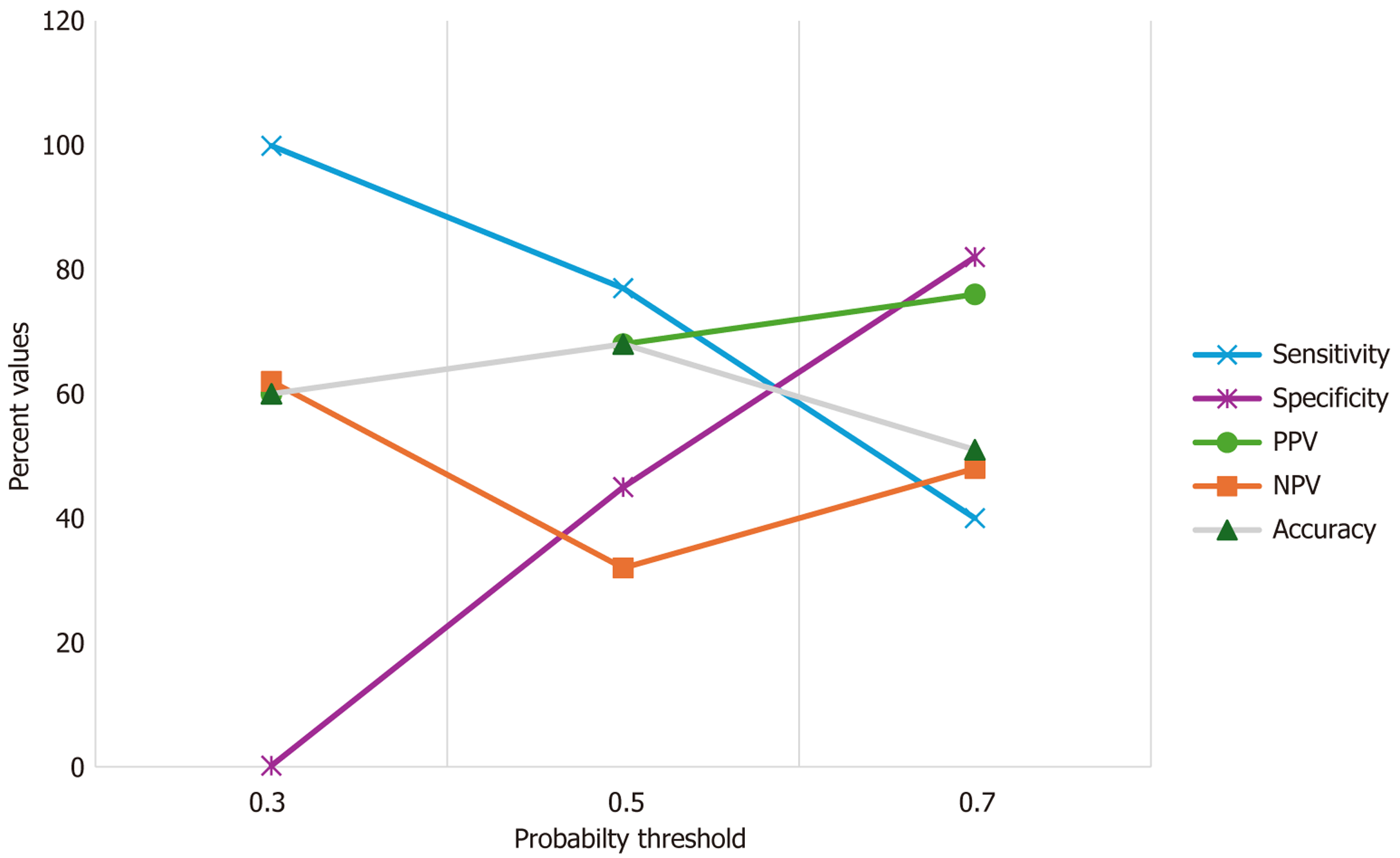
The performance metrics of the MLP neural network were evaluated by calculating sensitivity, specificity, PPV, NPV, and accuracy across three probability thresholds: (1) 0.3; (2) 0.5; and (3) 0.7. The percentage of true positives and true negatives was calculated across the three threshold probabilities. Accuracy was determined by whether the predicted probability of each case exceeded the specified threshold. To demonstrate the changes in performance of the MLP neural network across different thresholds, performance metrics were graphically depicted for varying probability thresholds.
In this retrospective analysis of the eICU database, a total of 12660 patients with sepsis and septic shock were included, of whom 6552 developed AKI and 5108 did not. The demographic and clinical characteristics of the two groups revealed several significant differences. Patients who developed AKI were older, with a median age of 69 years compared to 67 years among those without AKI (P < 0.001), and had a higher BMI (27.3 vs 26.3, P < 0.001). Illness severity was greater in the AKI group, as reflected by higher SOFA scores on admission (7 vs 6, P < 0.001). Male gender was more prevalent among AKI patients (52.3% vs 49.4%, OR = 1.12, 95%CI: 1.05-1.21, P = 0.002), and African American (11.6% vs 8.5%, P < 0.001) and Hispanic (5.7% vs 5.1%, P < 0.001) ethnicities were also associated with increased risk (Tables 1 and 2).
| Variables | Without AKI (n = 5108) | With AKI (n = 6552) | P value |
| Age (years) | 67 (55-79) | 69 (58-79) | < 0.001 |
| Body mass index (kg/m2) | 26.3 (22.3-31.6) | 27.3 (23.0-33.2) | < 0.001 |
| Sequential Organ Failure Assessment on admission | 6 (4-9) | 7 (5-10) | < 0.001 |
| White blood counts (× 103 cells/L) | 13.0 (8.6-18.4) | 14.0 (9.4-19.7) | < 0.001 |
| Platelet count (× 103 cells/L) | 220 (156-298) | 211 (149-294) | < 0.001 |
| Lymphocyte count (× 103 cells/L) | 962 (557-1578) | 930 (534-1560) | 0.023 |
| Neutrophil count (× 103 cells/L) | 10.3 (6.3-15.1) | 11.3 (7.0-16.4) | < 0.001 |
| Monocytes (× 103 cells/L) | 7.4 (4.1-11.5) | 7.7 (4.3-12.1) | 0.003 |
| Neutrophil-to-lymphocyte ratio | 10.1 (5.2-18.2) | 11.4 (6.1-22.0) | < 0.001 |
| Platelet-to-lymphocyte ratio | 226 (132-378) | 222 (130-391) | 0.949 |
| Monocyte-to-lymphocyte ratio | 0.7 (0.4-1.2) | 0.8 (0.4-1.3) | < 0.001 |
| Sodium (mmol/L) | 136 (133-139) | 136 (132-140) | 0.008 |
| Potassium (mmol/L) | 4.0 (3.7-4.5) | 4.2 (3.7-4.8) | < 0.001 |
| Magnesium (mg/dL) | 1.8 (1.5-2.0) | 1.8 (1.5-2.1) | < 0.001 |
| Albumin (g/dL) | 3.1 (2.6-3.6) | 3.1 (2.6-3.6) | < 0.001 |
| Initial lactate (mmol/L) | 2.1 (1.3-3.4) | 2.4 (1.5-3.9) | < 0.001 |
| Initial international normalized ratio | 1.2 (1.1-1.5) | 1.3 (1.1-1.6) | < 0.001 |
| Initial alanine aminotransferase (U/L) | 25 (22-28) | 25 (16-45) | 0.751 |
| Initial aspartate aminotransferase (U/L) | 29 (19-51) | 30 (20-57) | < 0.001 |
| Bicarbonate (mmol/L) | 25 (22-28) | 23 (20-27) | < 0.001 |
| Chloride (mmol/L) | 101 (97-104) | 100 (96-105) | < 0.001 |
| Blood urea nitrogen (mg/dL) | 20 (14-31) | 34 (22-53) | < 0.001 |
| Lactate-albumin ratio | 0.7 (0.4-1.1) | 0.8 (0.5-1.4) | < 0.001 |
| Creatinine (mg/dL) | 0.91 (0.70-1.30) | 1.8 (1.2-3.0) | < 0.001 |
| Total bilirubin (mg/dL) | 0.6 (0.4-1.1) | 0.7 (0.4-1.2) | 0.004 |
| Hemoglobin (g/dL) | 11.9 (10.0-13.5) | 11.5 (9.8-13.2) | < 0.001 |
| Systemic immune-inflammation index | 2185 (995-4393) | 2413 (1131-4838) | < 0.001 |
| Neutrophil-percentage-to-albumin ratio | 25.5 (21.1-31.3) | 26.0 (21.8-32.2) | < 0.001 |
| Systemic inflammation response index | 7072 (2760-15160) | 8216 (3390-18048) | < 0.001 |
| Aggregate index of systemic inflammation (× 109/L) | 1.53 (0.50-3.83) | 1.72 (0.59-4.32) | < 0.001 |
| Without AKI | With AKI | P value | OR | 95%CI for OR | ||
| Male gender | 2523 (49.4) | 3428 (52.3) | 0.002 | 1.124 | 1.045 | 1.210 |
| Ethnicity | ||||||
| African American | 436 (8.5) | 762 (11.6) | < 0.001 | |||
| Asian | 96 (1.9) | 123 (1.9) | ||||
| Caucasian | 3989 (78.1) | 4891 (74.6) | ||||
| Hispanic | 259 (5.1) | 376 (5.7) | ||||
| Native American | 33 (0.6) | 32 (0.5) | ||||
| Other | 294 (5.8) | 368 (5.6) | ||||
| Hypertension | 473 (9.8) | 619 (9.4) | 0.730 | 1.022 | 0.902 | 1.159 |
| Diabetes | 88 (1.7) | 203 (3.1) | < 0.001 | 1.824 | 1.416 | 2.349 |
| Chronic obstructive pulmonary disease | 512 (10) | 586 (8.9) | 0.048 | 0.882 | 0.778 | 0.999 |
| Hyperlipidemia | 94 (1.8) | 193 (2.9) | < 0.001 | 1.619 | 1.262 | 2.077 |
| Cerebrovascular disease | 84 (1.6) | 135 (2.1) | 0.101 | 1.258 | 0.956 | 1.656 |
| Malignancy | 342 (6.7) | 304 (4.6) | < 0.001 | 0.678 | 0.578 | 0.795 |
| Chronic kidney disease | 259 (5.1) | 1154 (17.6) | < 0.001 | 4.002 | 3.479 | 4.605 |
| Cardiomyopathy | 10 (0.2) | 19 (0.3) | 0.311 | 1.483 | 0.689 | 3.191 |
| Heart failure | 460 (9.0) | 679 (10.4) | 0.014 | 1.168 | 1.031 | 1.323 |
| Immunosuppression | 315(6) | 271(4) | 0.0001 | 0.65 | 0.55 | 0.77 |
| Cirrhosis | 128 (2.6) | 144 (2.2) | 0.245 | 0.867 | 0.681 | 1.103 |
| Corticosteroids | 656 (12.8) | 729 (11.1) | 0.004 | 0.850 | 0.759 | 0.951 |
| Ventilation | 2155 (42.2) | 2997 (45.7) | < 0.001 | 1.155 | 1.073 | 1.244 |
| Diuretics | 712 (13.9) | 1185 (18.1) | < 0.001 | 1.363 | 1.232 | 1.508 |
| Angiotensin converting enzyme inhibitor | 345 (6.8) | 602 (9.2) | < 0.001 | 1.397 | 1.217 | 1.603 |
| Angiotensin 2 receptor antagonist | 119 (2.3) | 199 (3.0) | 0.020 | 1.313 | 1.043 | 1.653 |
| Vasopressors | 1576 (30.9) | 2975 (45.4) | < 0.001 | 1.864 | 1.726 | 2.013 |
Comorbidities were important contributors to AKI risk. CKD emerged as the strongest predictor, with an OR of 4.00 (95%CI: 3.48-4.61, P < 0.001). Diabetes (OR = 1.82, 95%CI: 1.42-2.35, P < 0.001) and hyperlipidemia (OR = 1.62, 95%CI: 1.26-2.08, P < 0.001) were also more common in the AKI group, while malignancy was less prevalent (OR = 0.68, 95%CI: 0.58-0.80, P < 0.001). Heart failure was associated with a modestly increased risk of AKI (OR = 1.17, 95%CI: 1.03-1.32, P = 0.014). Clinical interventions such as mechanical ventilation (45.7% vs 42.2%, OR = 1.16, 95%CI: 1.07-1.24, P < 0.001), diuretic use (18.1% vs 13.9%, OR = 1.36, 95%CI: 1.23-1.51, P < 0.001), and vasopressor administration (45.4% vs 30.9%,
Laboratory findings further distinguished the AKI group. These patients had higher median white blood cell counts (14 × 103/μL vs 13 × 103/μL, P < 0.001), NCs (11.3 × 103/μL vs 10.3 × 103/μL, P < 0.001), and LCs (7.7 × 103/μL vs 7.4 × 103/μL, P < 0.001), but lower PCs (211 × 103/μL vs 220 × 103/μL, P < 0.001) and hemoglobin levels (11.5 g/dL vs 11.9 g/dL,
Inflammatory indices derived from routine laboratory tests were significantly elevated in the AKI group. The NLR was higher in AKI patients (11.4 vs 10.1, P < 0.001), as were the NPAR (26.0 vs 25.5, P < 0.001), SII (2413 vs 2185, P < 0.001), SIRI (8216 vs 7072, P < 0.001), and AISI (1.72 × 109 vs 1.53 × 109, P < 0.001). The MLR (0.7 vs 0.8, P < 0.001) was lower in the AKI group. The PLR did not significantly differ between groups (222 vs 226, P = 0.949; Tables 1 and 2).
To identify risk factors for AKI development, we performed a stepwise logistic regression analysis with forward selection. In model 1, which included clinical and lab parameters, Higher SOFA score (OR = 1.04, 95%CI: 1.01-1.06, P = 0.005), BUN (OR = 1.01, 95%CI: 1.004-1.01, P ≤ 0.001), SCr (OR = 3.46, 95%CI: 3.00-4.00, P ≤ 0.001), use of ACEi (OR = 1.56, 95%CI: 1.16-2.08, P = 0.003) were all found to be significant predictors of AKI. Elevated total bilirubin and potassium revealed a modest protective effect. Although the omnibus test for the model was statistically significant at 0.00001, the Hosmer-Lemeshow test demonstrated a poor fit, suggesting a deviation between observed and predicted values, perhaps impacted by model complexity and nonlinear relationships. Individual VIF of several variables such as albumin (3.67), sodium (4.65), chloride (4.78), neutrophil/Lymphocyte ratio (3.10), neutrophil/Lymphocyte ratio (5.66), initial lactic acid (7.81), lactic acid/albumin ratio (8.56), SII (7.55), SIRI (6.69), and AISI (7.71). In addition, among the variables, there was a high condition index of 111.3, and multiple variables demonstrated high variance proportions. These findings suggest problematic multicollinearity.
To detect nonlinear relationships between predictor variables and the risk of AKI in model 1, we performed a mul
Model 2 expanded upon the first by incorporating composite laboratory indices derived from PCA, excluding the SOFA score. Caucasian ethnicity, along with malignancy and immunosuppression, was associated with lower odds of AKI. The presence of CKD remained the strongest clinical risk factor. Vasopressor, A2RB, and ACEi use, and DM were also associated with AKI. Notably, factor 1 (inflammatory indices and hematology related factor score), OR = 1.10, 95%CI: 1.04-1.17, P < 0.001, and factor 2 (metabolic and renal function related factor score), OR = 1.10, 95%CI: 1.04-1.17, substantially improved model discrimination for AKI. The Hosmer-Lemeshow goodness of fit was 0.24. The addition of the PCA improved the fit of the model.
Model 3 included all the above variables plus the SOFA score. Here, Caucasian ethnicity shifted to confer higher odds of AKI, while malignancy and immunosuppression remained protective. CKD, use of Vasopressor, A2RB, and ACEi, along with DM, were significantly associated with increased risk. Factors 1 and 2 continued to predict AKI inde
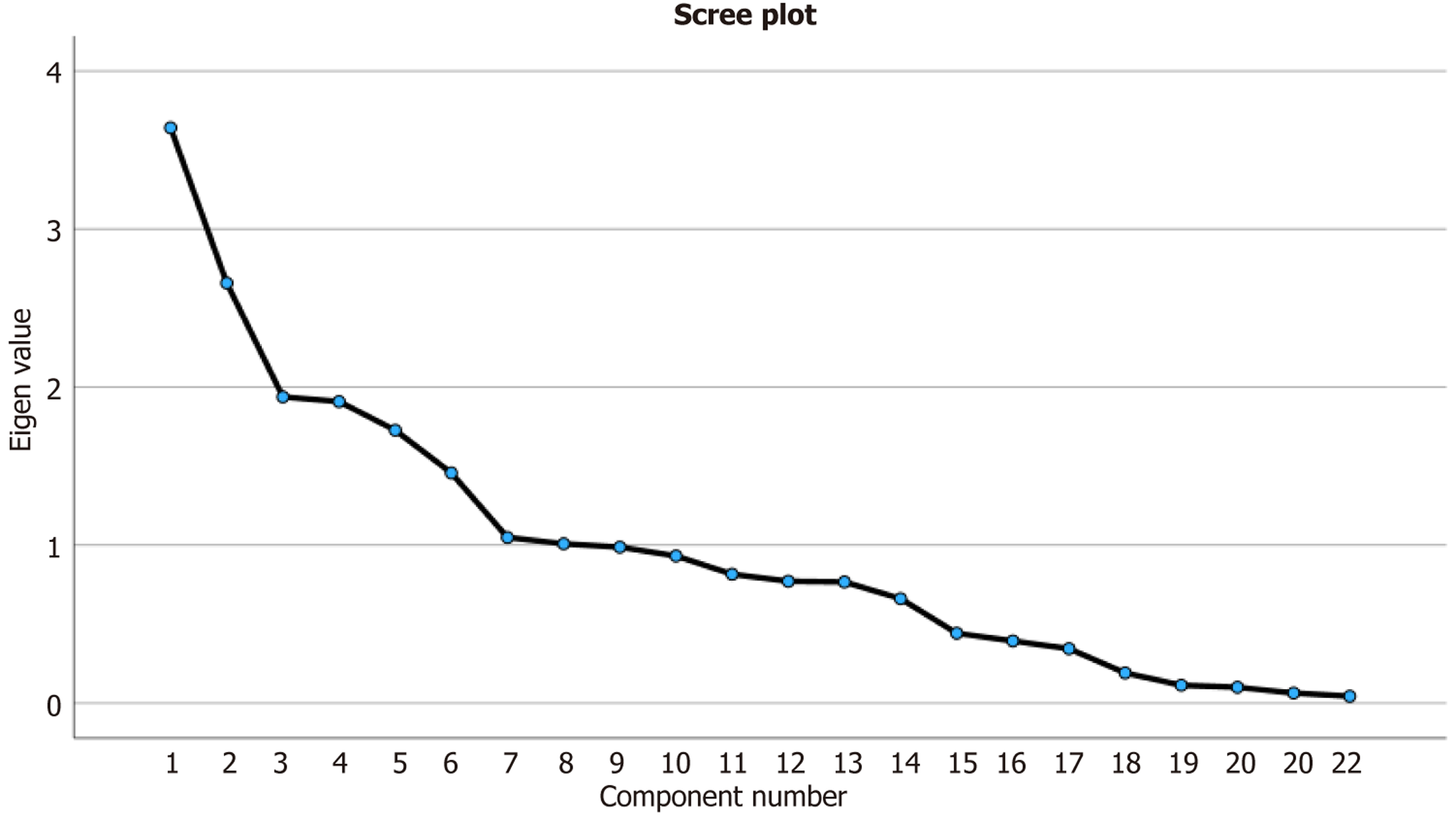
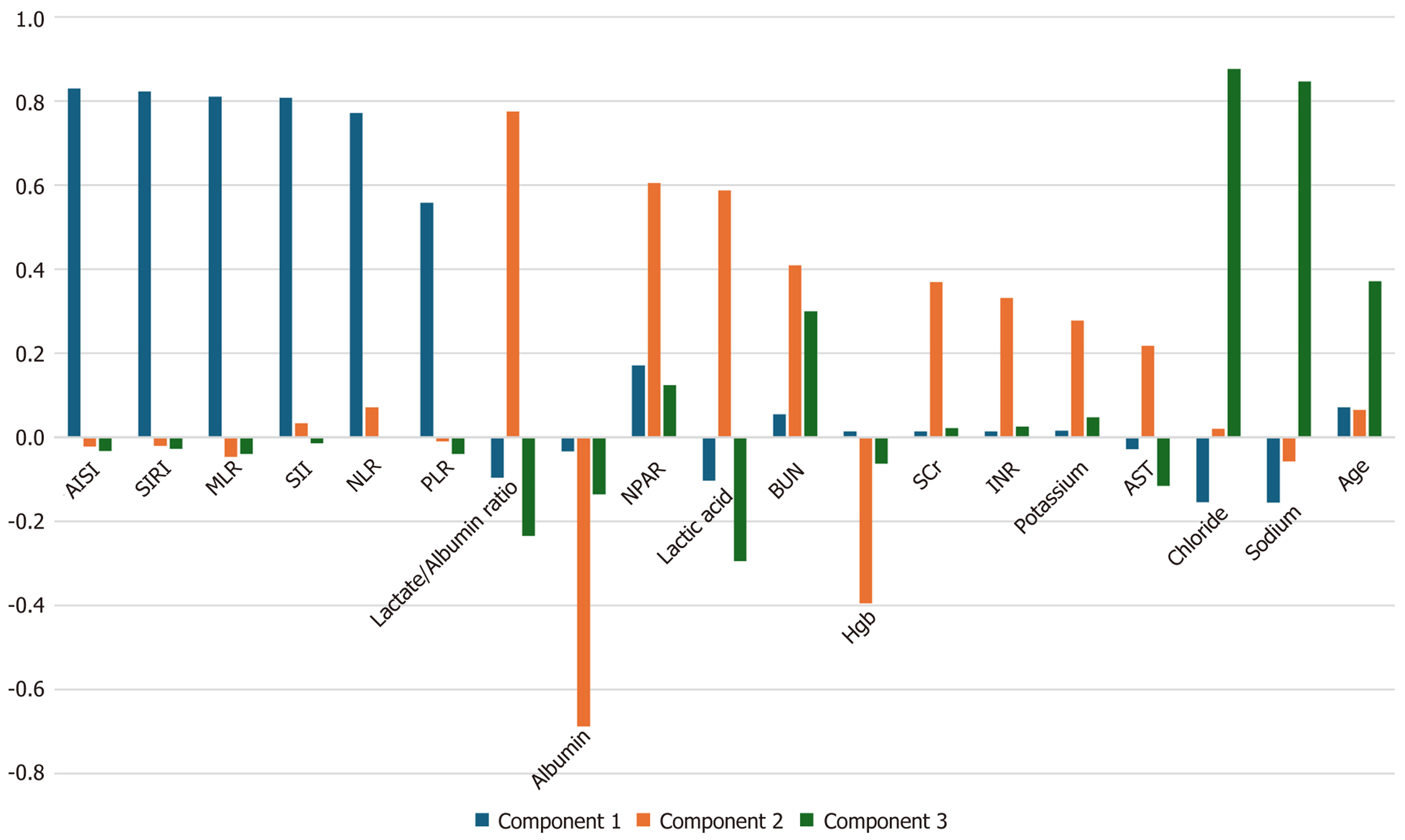
Visual analysis (elbow) of the scree plot yielded three orthogonal dimensions (Figure 3), with a Kaiser-Meyer-Olkin score of 0.6 and the Bartlett’s test of Sphericity result with a P-value of 0.0001. These together accounted for 42% of the total variance in the current septic shock cohort. Component 1 demonstrated high factor loading for inflammatory biomarkers AISI (0.83), SIRI (0.82), MLR (0.81), SII (0.87), NLR (0.77), and PLR (0.56), which accounted for 19.5% of the total variance. Component 2, accounting for approximately 13% of the total variance, demonstrated high factor loading for inflammation-metabolic and renal parameters, lactate/albumin (0.775), NPAR (0.60), lactic acid (0.6), BUN (0.5), and albumin (0.5). Component 3 accounting for 10% of the total variance revealed high factor loading for electrolytes and age varia
The scree plot displays the variance explained by each principal component in the analysis of clinical laboratory markers. The PCA identified three major factors representing distinct clinical domains. Factor 1 (inflammatory/hem
In an attempt to improve model fit, we performed PCA and then created a logistic model (model 2, Table 3, Supple
| β | SE | P value | Odds ratio | 95%CI for EXP(β) | |||
| Lower | Upper | ||||||
| Model 1 | SOFA score | 0.04 | 0.01 | 0.005 | 1.04 | 1.01 | 1.06 |
| Blood urea nitrogen | 0.01 | 0.003 | < 0.001 | 1.01 | 1.00 | 1.01 | |
| Serum creatinine | 1.24 | 0.07 | < 0.001 | 3.46 | 3.00 | 4.00 | |
| Neutrophils | 0.001 | 0.0001 | < 0.001 | 1.00 | 1.00 | 1.00 | |
| Monocytes | 0.001 | 0.001 | 0.09 | 1.00 | 1.00 | 1.00 | |
| Potassium | -0.164 | 0.05 | 0.001 | 0.85 | 0.76 | 0.94 | |
| Total bilirubin | -0.05 | 0.016 | 0.002 | 0.95 | 0.92 | 0.98 | |
| ACEi | 0.44 | 0.15 | 0.003 | 1.56 | 1.16 | 2.08 | |
| Vasopressors | 0.16 | 0.01 | 0.09 | 1.17 | 0.98 | 1.41 | |
| Model 2 | Caucasian | -0.075 | 0.027 | 0.005 | 0.93 | 0.88 | 0.98 |
| Malignancy | -0.41 | 0.12 | < 0.001 | 0.66 | 0.52 | 0.83 | |
| Immunosuppression | -0.26 | 0.12 | 0.034 | 0.76 | 0.60 | 0.98 | |
| CKD | 1.23 | 0.10 | < 0.001 | 3.40 | 2.82 | 4.19 | |
| Vasopressors | 0.44 | 0.06 | < 0.001 | 1.55 | 1.39 | 1.73 | |
| Diabetes | 0.55 | 0.20 | 0.006 | 1.74 | 1.17 | 2.58 | |
| ACEi | 0.45 | 0.10 | < 0.001 | 1.58 | 1.29 | 1.93 | |
| A2RB | 0.51 | 0.18 | 0.005 | 1.68 | 1.16 | 2.41 | |
| Factor 1 (inflammatory/hematologic) | 0.10 | 0.03 | < 0.001 | 1.10 | 1.04 | 1.17 | |
| Factor 2 (metabolic/renal/inflammatory) | 0.26 | 0.03 | < 0.001 | 1.30 | 1.23 | 1.39 | |
| Model 3 | Caucasian | 0.37 | 0.10 | < 0.001 | 1.44 | 1.19 | 1.74 |
| Malignancy | -0.39 | 0.12 | 0.001 | 0.68 | 0.54 | 0.85 | |
| Immunosuppression | -0.27 | 0.13 | 0.034 | 0.77 | 0.60 | 0.98 | |
| CKD | 1.19 | 0.10 | < 0.001 | 3.29 | 2.70 | 4.01 | |
| Vasopressors | 0.30 | 0.07 | < 0.001 | 1.35 | 1.19 | 1.54 | |
| Diabetes | 0.56 | 0.20 | 0.006 | 1.75 | 1.18 | 2.59 | |
| ACEi | 0.47 | 0.10 | < 0.001 | 1.60 | 1.30 | 1.96 | |
| A2RB | 0.52 | 0.19 | 0.005 | 1.69 | 1.17 | 2.43 | |
| Factor 1 (Inflammatory/hematologic) | 0.12 | 0.03 | < 0.001 | 1.13 | 1.06 | 1.20 | |
| Factor 2 (metabolic/renal/inflammatory) | 0.24 | 0.03 | < 0.001 | 1.27 | 1.19 | 1.34 | |
| SOFA score | 0.04 | 0.01 | < 0.001 | 1.04 | 1.01 | 1.05 | |
We developed an MLP to predict AKI using demographic, clinical, and factor score inputs (Figure 5 and Supplementary Figure 1). The cases were randomly split into a training set of 70.8% and a testing set of 29.2% of the population. The final network consisted of one hidden layer with seven units, using a hyperbolic tangent activation function, and an output layer optimized with a cross-entropy error function. On the training sample, the model had 63.5% overall classification accuracy, with a correct classification rate of 80.6% for patients with AKI and 38.6% for those without AKI. Performance was consistent in the testing sample, yielding an overall accuracy of 64.9% (81.8% for AKI vs 38.9% for no AKI).
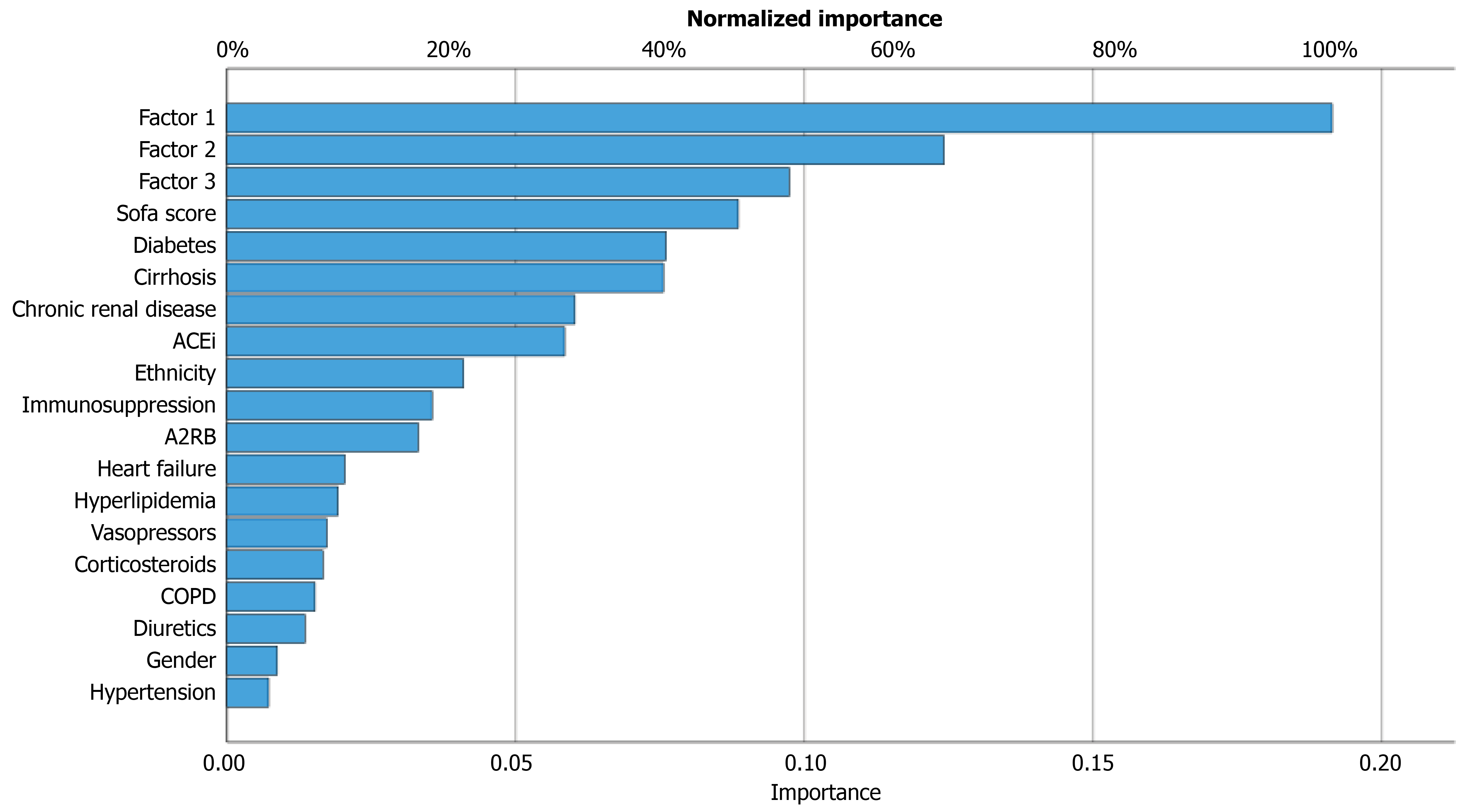
Assessment of predictor importance demonstrated that the latent factor scores and illness severity had the greatest predictive weight in the neural network. Variable-importance profiling ranked factor 1 (Inflammatory and hematological indices) as the most influential variable (normalized importance = 100%), followed by factor 2 (metabolic, renal, and inflammatory indices) of 64.9% and factor 3 (electrolytes and age) of 50.9%. Among clinical covariates SOFA score, dia
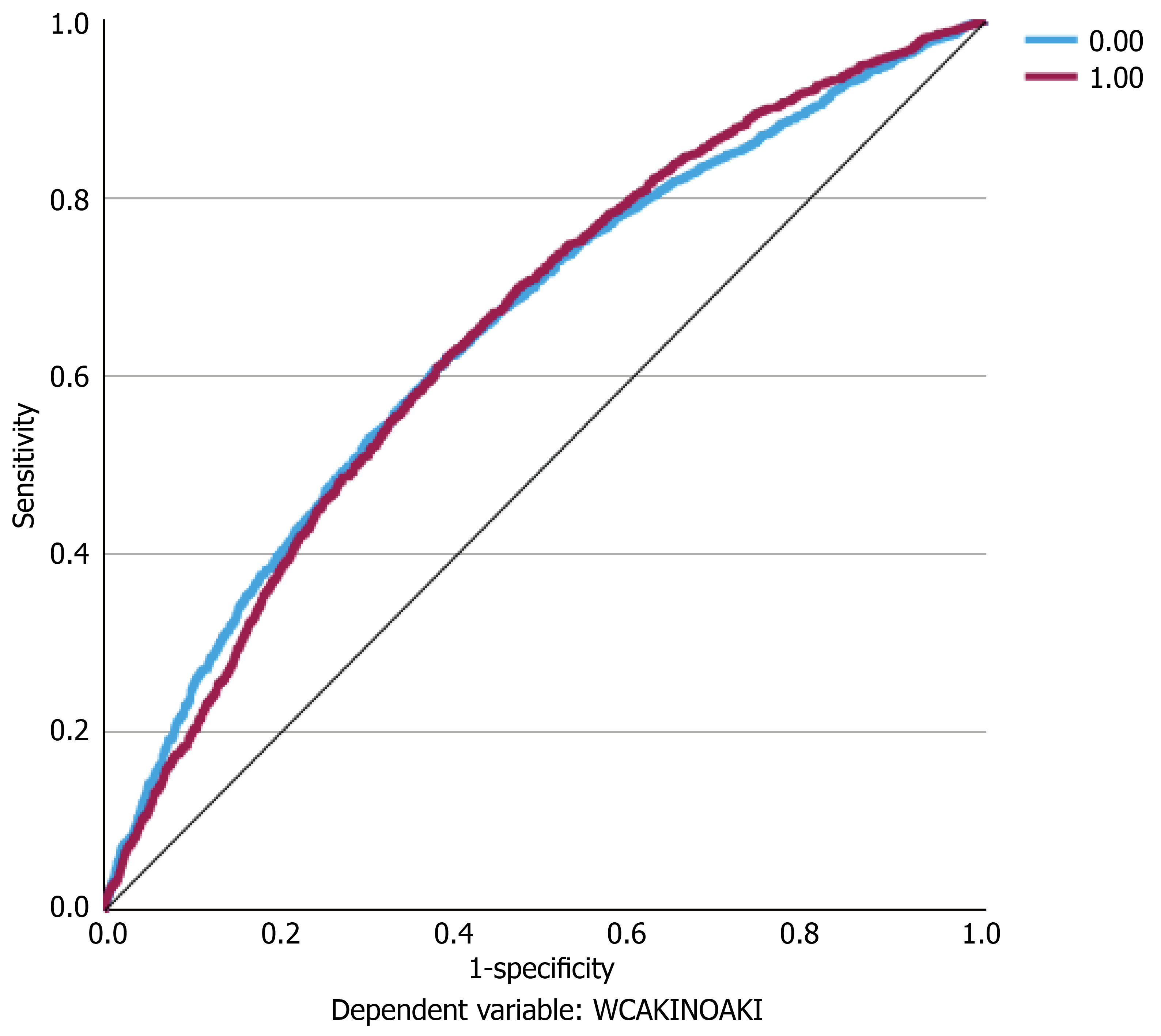
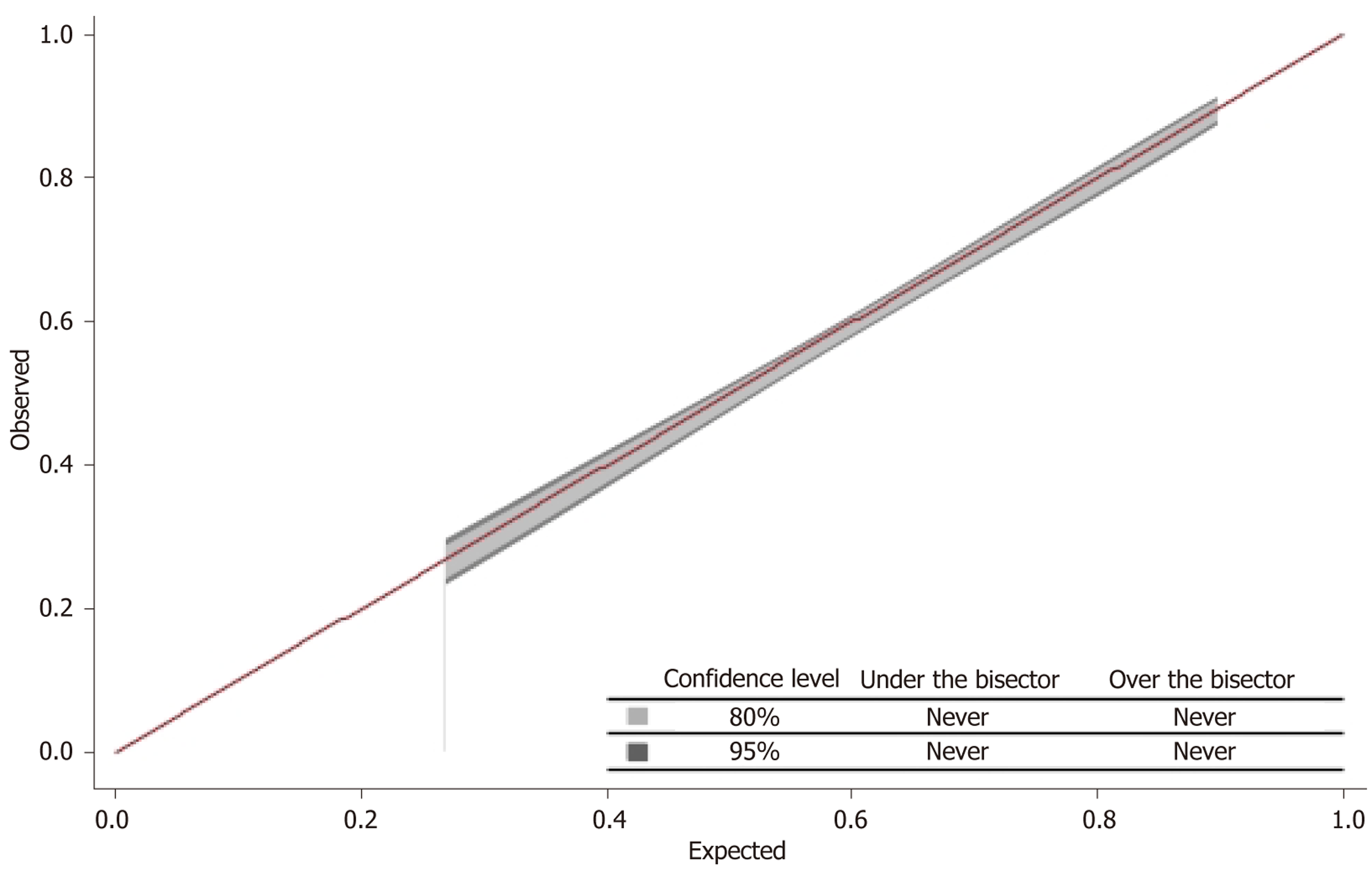
The performance of the MLP as demonstrated by the AUC of 0.67 exhibits only modest discrimination (Figure 7). Factors affecting discrimination include the biological complexity and heterogeneity of septic shock, multifactorial pathophysiology and non-inflammatory factors which influence outcomes in septic shock patients. Therefore, a cali
A DCA (Figure 9) was constructed which demonstrate that when the MLP neural network is compared to “treat all“ the following differences were noted, at a predictive threshold of 0.3-0.7, the neural network model curves remain above the treat-all curve. Thus, a treatment-based strategy is advantageous compared to a treat-all strategy across all predictive probabilities. In the context of the current study, a treatment-based strategy refers to interventions based on predicted risk, such as initiating diagnostic procedures, implementing preventative measures, and intensifying clinical monitoring.
We finally constructed a performance-based metrics graph across the probability thresholds of 0.3, 0.5, and 0.7 (Figure 10). A graphic representation demonstrates that at the lowest probability threshold of 0.3, the MLP neural network showed 100% sensitivity with almost no specificity, yielding modest PPV and NPV values of approximately 60%. This suggests the model could function well as a screening method. At mid-level threshold performance, the thre
Our study confirmed that several established risk factors, such as older age, higher BMI, male sex, African American race, pre-existing CKD, diabetes, and elevated SOFA scores at admission, as well as the use of vasopressors and mechanical ventilation, were all associated with the development of AKI in patients with septic shock in univariate analysis.
The primary goal of our study was to assess whether inflammatory markers provided additional prognostic value for AKI in this high-risk setting. When comparing patients with and without AKI, univariate analysis showed that the infla
Although the role of inflammatory markers NLR, SIRI, SII, AISI, and NPAR showed statistical significance in asso
Moreover, the performance of supervised machine learning was limited by marked multicollinearity and the presence of non-linear relationships between variables. To address these limitations, we employed unsupervised machine learning strategies, including neural network analysis combined with dimension reduction via PCA. This allowed for more flexible modeling of complex and interacting risk factors, providing a deeper understanding of the underlying relationships between inflammatory markers and AKI risk when simple univariate analysis fell short.
PCA and latent-factor approaches aggregate shared variance among inflammatory biomarkers, enabling multidimensional risk phenotyping and biologically coherent patient stratification. Prior studies have shown that PCA and clustering methods applied to circulating biomarkers in sepsis and AKI can identify patient subgroups with distinct immune pro
The neural network’s modest discrimination should be interpreted alongside its concordance with our regression and PCA findings. Consistent with models 2 and 3, the network assigned the greatest importance to latent factors, particularly the composite of metabolic, renal and inflammatory indices (factor 2), and to SOFA, while most stand-alone comorbidities carried smaller incremental weight. This pattern supports our dimension-reduction approach and shows that, in septic shock, acute systemic derangement and aggregated physiologic signatures outweigh traditional chronic risk factors for early AKI prediction. From a practical standpoint, models should emphasize high-signal severity composites such as factor scores and SOFA, incorporate careful calibration and threshold selection to account for asymmetric class per
In this large, multicenter retrospective cohort of ICU patients with septic shock, composite inflammatory indices such as SIRI, NLR, MLR, NPAR, SII, and AISI were significantly elevated in those who developed AKI. While these parameters demonstrated strong univariate associations with AKI, their independent prognostic value was diminished in multi
| 1. | Prowle J, Kolic I, Purdell-Lewis J, Kirwan C. Acute kidney injury of all severity is associated with extended hospitalization after critical illness. Crit Care. 2014;18:368. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 2. | Hoste EA, Bagshaw SM, Bellomo R, Cely CM, Colman R, Cruz DN, Edipidis K, Forni LG, Gomersall CD, Govil D, Honoré PM, Joannes-Boyau O, Joannidis M, Korhonen AM, Lavrentieva A, Mehta RL, Palevsky P, Roessler E, Ronco C, Uchino S, Vazquez JA, Vidal Andrade E, Webb S, Kellum JA. Epidemiology of acute kidney injury in critically ill patients: the multinational AKI-EPI study. Intensive Care Med. 2015;41:1411-1423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2415] [Cited by in RCA: 2034] [Article Influence: 184.9] [Reference Citation Analysis (1)] |
| 3. | Mo S, Bjelland TW, Nilsen TIL, Klepstad P. Acute kidney injury in intensive care patients: Incidence, time course, and risk factors. Acta Anaesthesiol Scand. 2022;66:961-968. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 30] [Reference Citation Analysis (1)] |
| 4. | Ronco C, Bellomo R, Kellum JA. Acute kidney injury. Lancet. 2019;394:1949-1964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1608] [Cited by in RCA: 1375] [Article Influence: 196.4] [Reference Citation Analysis (1)] |
| 5. | Pickkers P, Ostermann M, Joannidis M, Zarbock A, Hoste E, Bellomo R, Prowle J, Darmon M, Bonventre JV, Forni L, Bagshaw SM, Schetz M. The intensive care medicine agenda on acute kidney injury. Intensive Care Med. 2017;43:1198-1209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 87] [Article Influence: 9.7] [Reference Citation Analysis (3)] |
| 6. | Erratum Regarding "Prediction of Mortality and Major Adverse Kidney Events in Critically Ill Patients With Acute Kidney Injury" [Am J Kidney Dis. 2023;81(1):36-47]. Am J Kidney Dis. 2023;82:377-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 7. | Koyner JL, Carey KA, Edelson DP, Churpek MM. The Development of a Machine Learning Inpatient Acute Kidney Injury Prediction Model. Crit Care Med. 2018;46:1070-1077. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 217] [Article Influence: 27.1] [Reference Citation Analysis (2)] |
| 8. | Malhotra R, Kashani KB, Macedo E, Kim J, Bouchard J, Wynn S, Li G, Ohno-Machado L, Mehta R. A risk prediction score for acute kidney injury in the intensive care unit. Nephrol Dial Transplant. 2017;32:814-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 140] [Article Influence: 15.6] [Reference Citation Analysis (1)] |
| 9. | Wan J, Zou G, He B, Zhang C, Zhu Y, Yin L, Lu Z. Development and External Validation a Novel Inflammation-Based Score for Acute Kidney Injury and Prognosis in Intensive Care Unit Patients. Int J Gen Med. 2021;14:2215-2226. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 10. | Hong H, Jin J, Li M, Wang J. WCN23-0711 The relationship between the ratio of neutrophil to lymphocyte and platelet to acute kidney injury associated with sepsis. Kidney Int Rep. 2023;8:S34-S35. [DOI] [Full Text] |
| 11. | Wei W, Huang X, Yang L, Li J, Liu C, Pu Y, Yu W, Wang B, Ma L, Zhang L, Fu P, Zhao Y. Neutrophil-to-Lymphocyte ratio as a prognostic marker of mortality and disease severity in septic Acute kidney injury Patients: A retrospective study. Int Immunopharmacol. 2023;116:109778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 35] [Reference Citation Analysis (1)] |
| 12. | Kim WH, Park JY, Ok SH, Shin IW, Sohn JT. Association Between the Neutrophil/Lymphocyte Ratio and Acute Kidney Injury After Cardiovascular Surgery: A Retrospective Observational Study. Medicine (Baltimore). 2015;94:e1867. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 65] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 13. | Sun J, Qi Y, Wang W, Meng P, Han C, Chen B. Systemic Immune-Inflammation Index (SII) as a Predictor of Short-Term Mortality Risk in Sepsis-Associated Acute Kidney Injury: A Retrospective Cohort Study. Med Sci Monit. 2024;30:e943414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (1)] |
| 14. | Xiao W, Lu Z, Liu Y, Hua T, Zhang J, Hu J, Li H, Xu Y, Yang M. Influence of the Initial Neutrophils to Lymphocytes and Platelets Ratio on the Incidence and Severity of Sepsis-Associated Acute Kidney Injury: A Double Robust Estimation Based on a Large Public Database. Front Immunol. 2022;13:925494. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 15. | Pollard TJ, Johnson AEW, Raffa JD, Celi LA, Mark RG, Badawi O. The eICU Collaborative Research Database, a freely available multi-center database for critical care research. Sci Data. 2018;5:180178. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 657] [Cited by in RCA: 1008] [Article Influence: 126.0] [Reference Citation Analysis (5)] |
| 16. | Khwaja A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract. 2012;120:c179-c184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4761] [Cited by in RCA: 3863] [Article Influence: 275.9] [Reference Citation Analysis (2)] |
| 17. | Harrell FE. Regression Modeling Strategies. In: Bühlmann P, Diggle P, Gather U, editors. Springer Series in Statistics. Cham: Springer, 2015. [DOI] [Full Text] |
| 18. | Zhu K, Zheng Y, Chan KCG. Weighted Brier Score - an Overall Summary Measure for Risk Prediction Models with Clinical Utility Consideration. Stat Biosci. 2025. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 19. | Gershengorn HB, Patel S, Shukla B, Warde PR, Soorus SM, Holt GE, Kett DH, Parekh DJ, Ferreira T. Predictive Value of Sequential Organ Failure Assessment Score across Patients with and without COVID-19 Infection. Ann Am Thorac Soc. 2022;19:790-798. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 20. | Miller WD, Han X, Peek ME, Charan Ashana D, Parker WF. Accuracy of the Sequential Organ Failure Assessment Score for In-Hospital Mortality by Race and Relevance to Crisis Standards of Care. JAMA Netw Open. 2021;4:e2113891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 46] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 21. | Rabb H, Griffin MD, McKay DB, Swaminathan S, Pickkers P, Rosner MH, Kellum JA, Ronco C; Acute Dialysis Quality Initiative Consensus XIII Work Group. Inflammation in AKI: Current Understanding, Key Questions, and Knowledge Gaps. J Am Soc Nephrol. 2016;27:371-379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 307] [Cited by in RCA: 475] [Article Influence: 43.2] [Reference Citation Analysis (1)] |
| 22. | Wang H, Peng J, Wang B, Lu X, Zheng JZ, Wang K, Tu XM, Feng C. Inconsistency Between Univariate and Multiple Logistic Regressions. Shanghai Arch Psychiatry. 2017;29:124-128. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 42] [Reference Citation Analysis (1)] |
| 23. | Misset B, Philippart F, Fitting C, Bedos JP, Diehl JL, Hamzaoui O, Annane D, Journois D, Parlato M, Moucadel V, Cavaillon JM, Coste J; CAPTAIN Study Group. Clustering ICU patients with sepsis based on the patterns of their circulating biomarkers: A secondary analysis of the CAPTAIN prospective multicenter cohort study. PLoS One. 2022;17:e0267517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 24. | Star BS, Boahen CK, van der Slikke EC, Quinten VM, Ter Maaten JC, Henning RH, Kumar V, Bouma HR. Plasma proteomic characterization of the development of acute kidney injury in early sepsis patients. Sci Rep. 2022;12:19705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 25. | Bhatraju PK, Prince DK, Mansour S, Ikizler TA, Siew ED, Chinchilli VM, Garg AX, Go AS, Kaufman JS, Kimmel PL, Coca SG, Parikh CR, Wurfel MM, Himmelfarb J. Integrated Analysis of Blood and Urine Biomarkers to Identify Acute Kidney Injury Subphenotypes and Associations With Long-term Outcomes. Am J Kidney Dis. 2023;82:311-321.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (1)] |
| 26. | Smith LE, Smith DK, Blume JD, Siew ED, Billings FT 4th. Latent variable modeling improves AKI risk factor identification and AKI prediction compared to traditional methods. BMC Nephrol. 2017;18:55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 27. | Ghazi L, Farhat K, Hoenig MP, Durant TJS, El-Khoury JM. Biomarkers vs Machines: The Race to Predict Acute Kidney Injury. Clin Chem. 2024;70:805-819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 28. | Mascle O, Dupuis C, Brailova M, Bonnet B, Mirand A, De Beauchene RC, Philipponnet C, Adda M, Calvet L, Cassagnes L, Henquell C, Sapin V, Evrard B, Souweine B. Clustering based on renal and inflammatory admission parameters in critically ill patients admitted to the ICU. PLoS One. 2024;19:e0307938. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 29. | Ruinelli L, Cippà P, Sieber C, Di Serio C, Ferrari P, Bellasi A. Usability of machine learning algorithms based on electronic health records for the prediction of acute kidney injury and transition to acute kidney disease: A proof of concept study. PLoS One. 2025;20:e0326124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 30. | Tavris BS, Morath C, Rupp C, Szudarek R, Uhle F, Sweeney TE, Liesenfeld O, Fiedler-Kalenka MO, Dubler S, Zeier M, Schmitt FCF, Weigand MA, Brenner T, Nusshag C. Complementary role of transcriptomic endotyping and protein-based biomarkers for risk stratification in sepsis-associated acute kidney injury. Crit Care. 2025;29:136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 31. | Kiernan E, Zelnick LR, Khader A, Coston TD, Bailey ZA, Speckmaier S, Lo JJ, Siew ED, Sathe NA, Kestenbaum BR, Himmelfarb J, Johnson NJ, Shapiro NI, Douglas IS, Hough CL, Bhatraju PK. Molecular Phenotyping of Sepsis and Differential Response to Fluid Resuscitation. Am J Respir Crit Care Med. 2025;211:1681-1688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 32. | Neyra JA, Chen J, Bagshaw SM, Koyner JL. Risk Classification and Subphenotyping of Acute Kidney Injury: Concepts and Methodologies. Semin Nephrol. 2022;42:151285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 33. | Gaderparnah R, van Beuningen F, Lin Y, Sadrian SH, Reijneveld HM, Krabbe H, Jonkheijm P, Bouma HR, Mankowski RT. Sepsis as a complex syndrome: Are combined biomarkers the future of diagnosis and prognosis? Clinical perspective. Immunol Lett. 2026;277:107072. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (4)] |
| 34. | Zarbock A, Nadim MK, Pickkers P, Gomez H, Bell S, Joannidis M, Kashani K, Koyner JL, Pannu N, Meersch M, Reis T, Rimmelé T, Bagshaw SM, Bellomo R, Cantaluppi V, Deep A, De Rosa S, Perez-Fernandez X, Husain-Syed F, Kane-Gill SL, Kelly Y, Mehta RL, Murray PT, Ostermann M, Prowle J, Ricci Z, See EJ, Schneider A, Soranno DE, Tolwani A, Villa G, Ronco C, Forni LG. Sepsis-associated acute kidney injury: consensus report of the 28th Acute Disease Quality Initiative workgroup. Nat Rev Nephrol. 2023;19:401-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 458] [Reference Citation Analysis (1)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/