Published online Mar 9, 2026. doi: 10.5492/wjccm.v15.i1.113515
Revised: September 12, 2025
Accepted: November 21, 2025
Published online: March 9, 2026
Processing time: 185 Days and 4.7 Hours
Managing pregnant patients in the coronary care unit and the intensive care unit has been a challenge for many clinicians, as they do not encounter those special populations on a routine basis. Peripartum cardiomyopathy (PPCM) is an uncom
Core Tip: Early recognition of peripartum cardiomyopathy is critical, as symptoms mimic heart failure and other acute conditions. Management includes guideline-directed heart failure therapy tailored to pregnancy/lactation, individualized hemodynamic and respiratory support, and mechanical circulatory support if needed. Bromocriptine may be considered in severe cases to improve left ventricular recovery, with concurrent anticoagulation due to thrombotic risk. Long-term care requires serial echocardiography, natriuretic peptide monitoring, and counseling on future pregnancy risks, as residual cardiac dysfunction may persist.
- Citation: Zaidi SF, Prasad A, Gangadhar AM, Khan SA, Zaidi AH, Mushtaq M, Anil G, Surani S. Peripartum cardiomyopathy in an intensive care unit setting. World J Crit Care Med 2026; 15(1): 113515
- URL: https://www.wjgnet.com/2220-3141/full/v15/i1/113515.htm
- DOI: https://dx.doi.org/10.5492/wjccm.v15.i1.113515
Peripartum cardiomyopathy (PPCM) is maternal heart failure with systolic dysfunction and an ejection fraction below 45%. It usually develops in the last month of pregnancy or within five months after delivery. The diagnosis requires exclusion of prior cardiac disease and other common causes of hemodynamic decompensation[1,2].
The incidence of PPCM varies widely worldwide, from 0.03 per 1000 live births in the United States[3] to rates nearly 200 times higher in parts of Haiti and Nigeria[4]. In the United States, Black women represent 43.9% of all PPCM hospitalizations despite comprising a smaller proportion of the population, and some studies report a six-fold higher mortality risk compared to White women. This hints towards the importance of high-quality health care and attention to genetic predispositions, especially in certain ethnic subgroups[5].
The onset of PPCM often coincides with other pregnancy complications that cause similar hemodynamic instability, such as preeclampsia or pulmonary embolism[6]. Pregnancy itself is a major hemodynamic stressor, and women with a familial predisposition to cardiomyopathy may develop the condition during this period[7]. These factors make diagnosis challenging. The disease has an unpredictable course, with sudden deterioration sometimes necessitating urgent intensive care unit (ICU) admission-about 30% of hospitalized patients require intensive care. As most deaths occur within 30 days of diagnosis, maintaining a high index of suspicion and initiating prompt evaluation are critical[8].
Advanced maternal age is a recognized risk factor for PPCM, with the mean presentation age around 30–31 years, but higher rates and worse outcomes are seen in mothers over 40[9]. This is likely due to diminished cardiac reserve, accumulated comorbidities, and reduced adaptation to pregnancy-related changes[10]. Race and ethnicity also influence risk, with African American women experiencing a threefold higher incidence compared to White women[8]. Slower recovery rates and a greater likelihood of clinical deterioration after diagnosis compound this disparity. Hypertensive disorders of pregnancy, particularly severe preeclampsia, are among the strongest predictors, with over a 20-fold increase in risk[11]. Autoimmune factors, such as autoantibodies against cardiac proteins and receptors, are linked to more severe dysfunction and poorer recovery[4]. Nutritional factors, notably selenium deficiency, are associated with a higher incidence, and supplementation may improve symptoms and reduce mortality[12,13].
Additional risk factors include gestational diabetes, obesity, tocolytic therapy, viral myocarditis, and multiple gestation[4].
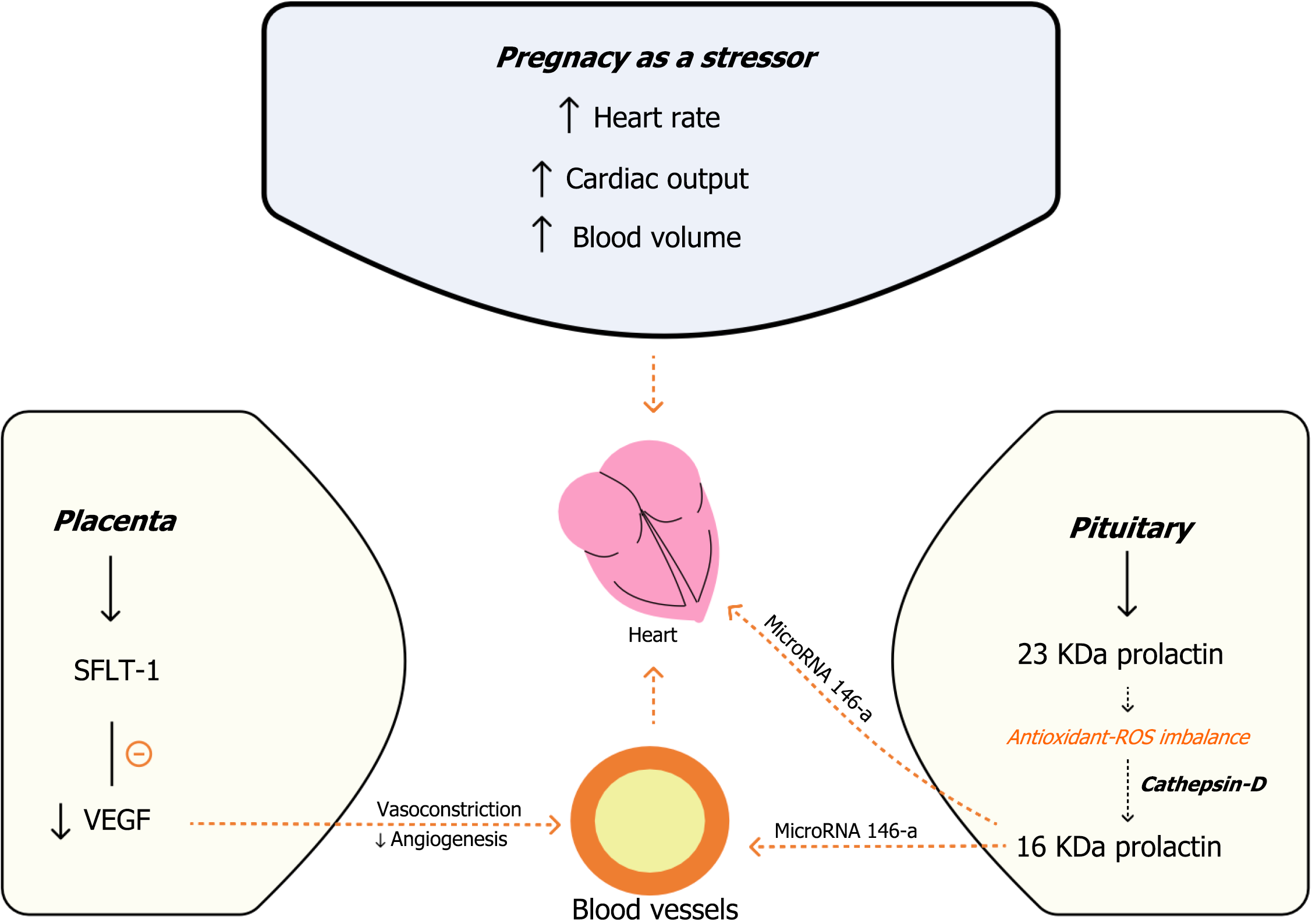
The physiological expansion of maternal blood volume, combined with increased heart rate and cardiac output, raises myocardial workload during pregnancy. These adaptations promote structural and homeostatic remodeling of cardiovascular tissue, which can exacerbate cardiac stress in the presence of other vulnerabilities[14]. Interestingly, the incidence of PPCM peaks in the last trimester. This is a time when hemodynamic stressors begin to decline. Therefore, this suggests that there exist additional pathophysiological mechanisms, such as hormonal influences, that contribute to the disease process[15].
Another contributing factor is elevated soluble fms-like tyrosine kinase-1 (sFlt-1), a decoy receptor that inhibits VEGF’s vasodilatory and angiogenic effects[16,17]. Elevated sFlt-1 levels, also seen in multiple gestations and preeclampsia, may explain the shared vascular injury patterns between PPCM and preeclampsia[18]. A simplified illustration is depicted in Figure 1.
Genetic testing has shown that approximately 15%-20% of PPCM patients carry pathogenic truncating variants in genes linked to familial cardiomyopathy. The most common are TTN (titin) variants, found in 10%-15% of cases, followed by mutations in BAG3, FLNC, DSP, LMNA, MYBPC3, and SCN5A. About two-thirds of these variants are located in the A-band region of the TTN gene, mirroring the distribution seen in familial cardiomyopathy[19].
TTN truncating variants are associated with poorer outcomes, as carriers tend to have significantly lower ejection fractions at one year. This could be important to prognosticate recovery outcomes after discharge[20].
To differentiate between familial cardiomyopathy and PPCM, the timing of symptom onset may help. Women with genetic cardiomyopathy often present earlier, around the second trimester when hemodynamic stress peaks, whereas classic PPCM typically develops in the last month of pregnancy or postpartum[21]. However, current evidence suggests routine genetic testing for all PPCM patients as it could potentially help predict disease severity and manage resources[22].
Clinical features of PPCM resemble typical heart failure symptoms, including dyspnea, orthopnea, peripheral edema, dizziness, and cough, usually presenting late in pregnancy or within months after delivery. However, these symptoms do not reveal the underlying cause of heart failure[22]. Heart failure may result from chronic conditions such as hyper
Careful attention to clinical features is essential, as certain symptoms may suggest alternative diagnoses. For example, chest pain may shift suspicion from PPCM to conditions like pulmonary embolism or pregnancy-associated myocardial infarction[24]. Alongside clinical evaluation, basic imaging and laboratory tests provide valuable insights. Key investigations include chest X-ray and echocardiography[22].
In heart failure, a chest X-ray may reveal signs such as pulmonary edema, cardiomegaly, interstitial or alveolar infiltrates, and pleural effusions. It is also useful for excluding pulmonary conditions that can mimic or contribute to symptoms, such as chronic obstructive pulmonary disease, pneumonia, pulmonary fibrosis, and pneumothorax[22]. Echocardiography helps identify structural cardiac abnormalities such as valvular disease, congenital defects, or regional wall motion abnormalities as seen in myocardial infarction. Additionally, an electrocardiogram (ECG) is important to exclude ischemic causes of heart failure and detect arrhythmias that may contribute to decompensation[25]. Biomarkers such as NT-proBNP (> 300 pg/mL) and BNP (> 100 pg/mL) are useful for ruling out heart failure[26]. Troponins are sensitive markers of acute myocardial injury, such as MI or myocarditis[27].
Cardiac magnetic resonance (CMR) imaging plays a crucial role in the postpartum evaluation of patients presenting with heart failure or suspected PPCM. CMR offers high-resolution, tissue-characterizing capabilities that help differentiate PPCM from Takotsubo syndrome (TTS), myocarditis, and other causes of cardiomyopathy[28]. Maintaining a high index of suspicion is essential until all differential diagnoses are thoroughly ruled out.
In PPCM, CMR typically shows global or regional left ventricular (LV) systolic dysfunction with minimal or absent late gadolinium enhancement (LGE), suggesting non-ischemic, non-inflammatory injury[29].
In TTS, CMR may demonstrate transient apical or mid-ventricular ballooning with myocardial edema on T2-weighted imaging and no LGE, consistent with myocardial stunning rather than necrosis[30].
In myocarditis, CMR often reveals myocardial edema, subepicardial or mid-wall LGE, and regional wall motion abnormalities that follow a non-coronary distribution, helping to distinguish it from PPCM or ischemic injury[31,32].
Additionally, CMR is highly sensitive for detecting LV thrombus, especially when echocardiographic windows are suboptimal. Thrombi are best visualized using contrast-enhanced, delayed imaging[32].
It is worth noting that TTS should be clearly distinguished as a differential diagnosis in the peripartum period. Its presentation often overlaps with PPCM, including symptoms such as chest pain, dyspnea, and transient LV dysfunction. This is important because the treatment approach and prognosis differ between TTS and PPCM.
The RETAKO study (REcapTure of takotsubo cardiomyopathy in obstetric patients)[33] is a recent research effort focused on understanding the features, diagnosis, and management of TTS in the peripartum period. While PPCM and TTS can present with overlapping features, their underlying mechanisms, imaging characteristics, and management differ significantly. Treatment for TTS is primarily supportive, focusing on heart failure management, avoidance of cate
Severe presentations of PPCM often require intensive care for hemodynamic stabilization, respiratory support, careful fluid and thromboembolic risk management, and, in a subset, mechanical circulatory support (MCS).
Acute hemodynamic support in PPCM aims to restore end-organ perfusion while minimizing further myocardial injury.
For cardiogenic shock, shortterm inotropic support is often used to augment contractility while vasoactive therapy stabilizes perfusion pressure. Dobutamine increases cardiac output primarily through beta1–mediated positive inotropy and modest chronotropy, with generally mild but unpredictable reductions in systemic vascular resistance due to beta2 vasodilation. Milrinone provides inotropy with added lusitropy and acts as an inodilator by reducing systemic and pulmonary vascular resistance. This can improve forward flow but also increases the risk of hypotension. Careful titration is therefore required for either drug. Choice and dosing should be individualized according to the patient's blood pressure, filling status, and arrhythmic risk. Regular reassessment with hemodynamics at the bedside is important[34,35].
Invasive monitoring: Arterial line placement is indicated for continuous blood-pressure monitoring in unstable patients and titration of vasoactive agents. Central venous access (and, in centers where available and appropriate, pulmonary artery catheterization or advanced hemodynamic monitoring) can help with fluid and inotropic management in compli
Avoidance of afterload reduction in hypotension. Although afterload reduction (e.g., with ACE inhibitors) benefits chronic systolic dysfunction, these agents are contraindicated in the setting of hypotension or cardiogenic shock. Afterload-reducing therapy can be deferred until the patient is hemodynamically stable and (in the postpartum) maternal/fetal concerns are addressed[34,35] (Table 1).
| Suggested ICU hemodynamics order set |
| Insert an arterial line and central venous catheter for unstable patients |
| Start norepinephrine if MAP < 65 mmHg with signs of hypoperfusion |
| Initiate inotrope for low-output states: |
| Dobutamine 2-5 μg/kg/minute, titrated every 30-60 minutes based on invasive measures |
| OR Milrinone 0.25-0.5 μg/kg/minute, titrate every 30-60 minutes based on invasive measures |
| Avoid nitroprusside or hydralazine in the presence of hypotension |
| Daily ECG |
| Daily electrolytes: |
| Maintain K+ > 4.0 mmol/L |
| Maintain Mg2+ > 2.0 mg/dL |
| Escalation checklist: |
| Persistent hypotension despite inotrope → Add vasopressor |
| Cardiac index < 2.0 L/minute/m2 after 2 hours → Consider mechanical circulatory support (see section E) |
| Lactate > 4 mmol/L or worsening acidosis → Activate advanced heart failure consult |
Respiratory dysfunction caused by pulmonary edema or respiratory muscle fatigue is seen in severe PPCM.
Oxygen and noninvasive ventilation (NIV). Supplemental oxygen and NIV are first lines for hypoxemia and alveolar edema if the patient can protect their airway. NIV can reduce the work of breathing and improve oxygenation while avoiding intubation in many patients.
Endotracheal intubation and mechanical ventilation. Indicated for persistent hypoxemia, inability to protect the airway, progressive respiratory fatigue, or when aggressive invasive hemodynamic support is required. Ventilator strategy should minimize cardiopulmonary interactions that worsen preload/afterload-avoid large tidal volumes and unnecessary positive end-expiratory pressures that may reduce venous return unless needed for oxygenation.
Pulmonary edema management. Optimize preload (see fluid management) and use diuretics and, when indicated, positive pressure ventilation to redistribute fluid and improve oxygenation. Rapid correction of hypoxemia and attention to left-sided filling pressures are priorities (Table 2).
| Suggested ICU respiratory order set |
| Oxygen via nasal cannula or HFNC; titrated to target saturation |
| NIV (CPAP 5-10 cm H2O) if hemodynamically stable and cooperative |
| Early intubation if PaO2/FiO2 < 150 or signs of fatigue |
| Daily chest X-ray and ABG for ventilated patients |
| Avoid high PEEP in preload-dependent patients |
| Escalation checklist |
| SpO2 < 88% despite HFNC/NIV → Prepare for intubation |
| Worsening pulmonary edema → Intensify diuresis and evaluate for MCS |
| Signs of ventilator-induced hypotension → Adjust PEEP and fluids cautiously |
Careful balancing of preload and perfusion is central to optimizing cardiac output without worsening pulmonary congestion.
Diuretics for volume overload. Loop diuretics (e.g., furosemide) are used to relieve pulmonary and systemic con
Avoid over diuresis in preload-dependent patients. Overly aggressive diuresis can precipitate hypotension and reduce cardiac output in patients who are dependent on higher preload; invasive monitoring or dynamic measures of volume responsiveness help guide therapy. Titrate diuretics to clinical response and hemodynamic data[34,35] (Table 3).
| Suggested ICU fluid order set |
| Furosemide IV bolus (20-40 mg), repeat or switch to infusion if inadequate diuresis |
| Strict input/output monitoring; daily weights |
| CVP-guided diuresis in invasive monitoring patients |
| Hold diuretics if MAP < 60 mmHg or rising creatinine |
| Escalation checklist |
| Persistent volume overload despite high-dose loop diuretics → Add thiazide synergy |
| Rising creatinine > 0.3 mg/dL in 48 hours → Reassess fluid goals |
| CVP < 5 cmH2O and hypotension → Stop diuretics and reassess preload |
PPCM is at high risk of intracardiac thrombus and thromboembolism due to severe LV systolic dysfunction and peri
Indications. Anticoagulation is considered if LV ejection fraction (LVEF) is greatly reduced (< 30%) or when an LV thrombus is present. The decision must weigh against the risk of bleeding (including risk of postpartum hemorrhage) and be coordinated with obstetric management if delivery is near[34,35] (Table 4).
| Suggested ICU anticoagulation order set |
| Initiate unfractionated heparin infusion; target PTT 60-80 seconds |
| Daily CBC and coagulation profile |
| Transition to warfarin postpartum if stable and no planned procedures |
| Escalation checklist |
| New embolic event → Evaluate anticoagulation adequacy |
| Active bleeding → Hold anticoagulant and reverse if indicated |
Multiple expert sources recommend heparin prophylaxis-typically low molecular weight heparin (LMWH)-when bromocriptine is administered, due to its pro-thrombotic potential[36].
Need to balance with obstetric bleeding risk. Major reviews emphasize that anticoagulation should be initiated only after careful consideration of the bleeding risk, particularly in the peripartum context.
When pharmacologic therapy cannot maintain perfusion, early MCS consideration is lifesaving and can bridge to recovery or definitive treatment. Refractory cardiogenic shock despite optimized inotropes/vasopressors, progressive end-organ dysfunction, or severe arrhythmias may prompt escalation to MCS. Key triggers include:
Hemodynamic instability: Mean arterial pressure < 65 mmHg accompanied by signs of ongoing hypoperfusion (e.g., cool extremities, altered mentation, oliguria).
Metabolic derangement: Low cardiac output. Escalating support requirements: Increasing doses of inotropes and/or vasopressors to maintain perfusion.
End-organ hypoperfusion: Evidence of organ dysfunction such as elevated creatinine, liver enzymes, rising lactate, or oliguria.
These criteria correspond to the Society for Cardiovascular Angiography and Interventions cardiovascular shock. Stage C (classic) for standard hypoperfusion, D (deteriorating) if escalating despite therapy, and E (extremis) if in refractory shock or arrest-each stage warranting urgent MCS escalation[37].
Devices available in the ICU: A range of devices exist to offer cardiopulmonary support. Intra-aortic balloon pump-provides modest afterload reduction and coronary perfusion augmentation; may be used as a temporizing measure in selected patients. Percutaneous ventricular assist devices (e.g., Impella)-can unload the left ventricle and increase systemic flow.
Venoarterial extracorporeal membrane oxygenation (VA-ECMO)-offers full cardiopulmonary support and is especially useful when combined with respiratory failure or refractory shock. LV unloading during VAECMO is advised when there is an elevated PCWP (commonly > 15-18 mmHg), absence of aortic valve opening or poor pulsatility, increasing LV dimension or pulmonary edema and spontaneous echocardiographic contrast or LV thrombus. LV unloading strategies include conservative approaches (e.g., optimizing ECMO flow, afterload, and preload). These are selected based on the patient's condition and the institution's capability. ECMO can serve as a bridge to recovery, durable mechanical support, or transplantation[34,38] (Table 5).
| Suggested ICU MCS order set |
| Early heart team consult if escalating inotropes/pressors > 24 hours |
| Prepare femoral access for urgent IABP or ECMO |
| Daily echocardiographic monitoring while on support |
| Escalation checklist |
| MAP < 60 mmHg and lactate rising despite inotropes → Initiate MCS |
| Multiorgan failure progression → Reassess goals of care |
The selection of modality depends on local expertise, hemodynamic goals (unloading vs support), and the anticipated duration of support. Early consultation with a tertiary center experienced in MCS is recommended when deteriorating hemodynamics are expected.
Medical therapy in PPCM combines standard heart-failure measures (modified for pregnancy/postpartum) with some disease-specific approaches.
Diuretics (furosemide): First-line for symptomatic congestion. Use with attention to hemodynamics and renal function.
Beta-blockers: Evidence-based guideline-directed beta-blockers are evidence-based in systolic heart failure, but their initiation in PPCM should be guarded and typically deferred until the patient is hemodynamically stabilized. Selective agents such as metoprolol are typically used once tolerated.
ACE inhibitors/ARBs: These agents improve remodeling and outcomes in systolic heart failure but are contraindicated during pregnancy. They are typically initiated postpartum once breastfeeding and maternal considerations are addressed.
Bromocriptine (prolactin-inhibition strategy): An emerging, disease-targeted therapy has been proposed based on the hypothesis that a cleaved 16-kDa prolactin fragment contributes to myocardial injury in PPCM. Small trials and registry data suggest bromocriptine added to standard heart-failure therapy may improve recovery of LVEF in selected patients; because bromocriptine can increase thrombotic risk, most protocols pair it with concurrent anticoagulation. Use of bromocriptine is not yet standard of care within institutions and must be reserved on a case-by-case basis and within multidisciplinary discussion. Breastfeeding and continued use of Bromocriptine should be approached on a case-by-case basis[29,39].
Inotropes and vasopressors: Use the lowest effective dose for the shortest duration. All agents cross the placenta to some degree; maternal stabilization takes priority.
Levosimendan: ESC guidelines mention it as an option where available (non-catecholamine inodilator, calcium sensitizer)-may be advantageous in PPCM with high catecholamine load, though safety data in pregnancy remain limited[40] (Table 6).
| Suggested ICU pharmacotherapy order set |
| Furosemide IV as above |
| Metoprolol succinate 125-25 mg daily once off inotropes for 24 hours |
| Lisinopril 25-5 mg daily postpartum if SBP > 90 mmHg |
| Bromocriptine with concurrent anticoagulation (UFH or LMWH) |
| Levosimendan 6-12 μg/kg over 10 minute (loading) 0.05-0.2 μg/kg/minute for 24 hours (maintenance). Limited data available in pregnancy. Not advisable during lactation |
| Escalation checklist |
| Hypotension after beta-blocker → Hold and reassess |
| Worsening renal function after ACEi → Stop and monitor |
As the pregnancy nears term, patients with PPCM require careful obstetric planning. This includes weighing maternal cardiac stability against fetal development and well-being. A team-based approach involving an obstetrician, maternal-fetal medicine, cardiology, critical-care, anesthesia, and neonatology has been shown to improve outcomes in the intrapartum period and prevent post-partum complications[15].
In women with compensated heart function, delivery is aimed at term (39-40 weeks) with the aim to ensure adequate fetal lung maturity and development. Refractory heart failure, increased mechanical, ionotropic support, and fetal distress are indications for urgent delivery. Thus, in worsening of disease status, maternal safety is prioritized over gestational age[41].
Individualized decisions on the method of delivery depend upon the hemodynamic state, with vaginal delivery and titrated epidural anesthesia being the preferred mode in a stable mother. It mitigates abrupt fluid shifts, and there is generally a lower risk of blood loss[42]. In women with significant heart failure (NYHA Class III-IV), shortening the second stage of labor with assistance from vacuum or forceps alleviates the stress of maternal Valsalva. The Cesarean mode is an option in such instances of heart failure where rapid hemodynamic control becomes necessary. It is also performed in stable mothers with standard obstetric indications like fetal distress, malpresentations, etc.[43]. Several studies, however, have shown that there is increased morbidity associated with sections due to a change in volume status and the use of central neuraxial anesthesia[44]. There must be continuous fetal and maternal monitoring with vigilance in volume status and necessary pain control, irrespective of the delivery mode.
Breastfeeding post-delivery should be approached on a case-by-case basis, considering the caveat of heightened metabolic demand in heart failure in lactation, against the benefits that breast milk confers on the newborn. Lactating mothers have elevated Prolactin levels and increased cytotoxic T-cell activity, but studies have shown that in stable PPCM, breastfeeding is not inferior to non-breastfeeding mothers[45]. Bromocriptine, a D-2 agonist drug, has been shown to play a dual role in treatment by rapidly suppressing lactation and improving LVEF. In the ICU, doses begin at 2.5 mg twice a day, which can be titrated to 10-20 mg based on serum prolactin levels, with prophylactic anticoagulation. Therapy is continued on low-dose maintenance after the acute phase to prevent rebound lactation[35].
The 2025 ESC Guidelines for the Management of Cardiovascular Disease in Pregnancy highlight the importance of recognizing it as a life-threatening condition with significant implications for maternal morbidity and mortality[40]. Key recommendations include:
Counselling on recurrence risk and contraception: Every woman with PPCM should receive structured counselling about the risk of recurrence in future pregnancies, even if LV function normalizes. Guideline emphasize shared decision-making, involvement of multidisciplinary teams, and the importance of reliable contraception. Long-acting reversible contraceptives are often preferred in women with residual LV dysfunction due to its efficacy and reliability. Estrogen-containing methods should be avoided if LV dysfunction persists, as they may pose additional cardiovascular risks.
Bromocriptine as a disease-modifying therapy: Based on the prolactin-cleavage hypothesis, bromocriptine may be considered after delivery to improve LV recovery. However, prothrombotic risk is significant; thus, it should always be co-administered with at least prophylactic LMWH[46].
Continuation of guideline-directed medical therapy: Even in women who demonstrate apparent full recovery of LVEF, heart failure therapies (ACEi/ARB/ARNI, beta-blocker, MRA, and SGLT2i if tolerated) should be continued for at least 12 months. The rationale is the high rate of late relapses and the possibility of subclinical myocardial vulnerability. Tapering may be cautiously considered thereafter under close follow-up with cardiac imaging and biomarkers.
For women with PPCM and an LVEF < 35: Wearable Cardioverter-Defibrillator (WCD) can be considered in the vulne
Some predictors of poor recovery include patients with cardiogenic shock with low EF (< 30%-35%) and LV dilation. Reduced right ventricular fractional area change (RVFAC < 31.4%), a sign of right ventricular dysfunction, and low left atrial volume are strong and independent predictors of mortality and morbidity[51,52]. The Presence of Preeclampsia is interestingly associated with improved LV recovery.
It is important to establish a close follow-up plan post-discharge. This includes serial echocardiograms and natriuretic peptides to track cardiac function during the first 6 months. Surveillance for arrhythmias with electrocardiogram and ambulatory rhythm monitoring is a regular practice[53]. Advanced cardiac imaging, such as magnetic resonance imaging, can be considered at 6 months to monitor signs of recovery or disease progression.
Medical treatment for heart failure should be continued and titrated step-wise for 12-24 months based on functional status, with reinforcement of adherence[54]. Studies determine that an LVEF of > 50%-55% is the cut-off to indicate recovered LV function[48]. Even when the LVEF has normalized, imaging often reveals residual cardiac abnormalities and reduced exercise capacity[50]. Preconception counseling should therefore include discussion of the potential pregnancy risks that remain despite apparent recovery, with strong recommendations to avoid subsequent pregnancies if there is evidence of prior adverse outcomes. A multidisciplinary team and shared decision-making process provide a collaborative framework to guide safe planning for any future pregnancy.
PPCM is a diagnosis of exclusion. It is marked by LV systolic dysfunction developing near term or postpartum. PPCM has a variable course that can deteriorate rapidly and carries an early mortality risk. Early escalation of diagnostics using echocardiography, biomarkers, and targeted imaging can help rule out more common causes of cardiac decompensation. Patients can deteriorate unexpectedly and require ICU-level care. Diligent optimization of hemodynamic, respiratory, and thromboembolic components is central to stabilizing patients and preventing organ injury. Bromocriptine represents a promising disease-targeted adjunct. However, it requires anticoagulation and multidisciplinary governance due to thrombotic risk and lactation effects. Obstetric planning prioritizes maternal stability, favors vaginal delivery only when feasible, and promotes vigilant peripartum monitoring to navigate fluid shifts. Recovery is heterogeneous but can be navigated with a structured followup plan. This includes serial echocardiography, rhythm surveillance, and sustained heartfailure therapy for at least 12-24 months. A coordinated, multidisciplinary approach across cardiology, critical care, and obstetrics is the key to improving maternal and neonatal outcomes.
| 1. | Davis MB, Arany Z, McNamara DM, Goland S, Elkayam U. Peripartum Cardiomyopathy: JACC State-of-the-Art Review. J Am Coll Cardiol. 2020;75:207-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 339] [Article Influence: 56.5] [Reference Citation Analysis (1)] |
| 2. | Sliwa K, Hilfiker-Kleiner D, Petrie MC, Mebazaa A, Pieske B, Buchmann E, Regitz-Zagrosek V, Schaufelberger M, Tavazzi L, van Veldhuisen DJ, Watkins H, Shah AJ, Seferovic PM, Elkayam U, Pankuweit S, Papp Z, Mouquet F, McMurray JJ; Heart Failure Association of the European Society of Cardiology Working Group on Peripartum Cardiomyopathy. Current state of knowledge on aetiology, diagnosis, management, and therapy of peripartum cardiomyopathy: a position statement from the Heart Failure Association of the European Society of Cardiology Working Group on peripartum cardiomyopathy. Eur J Heart Fail. 2010;12:767-778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 616] [Cited by in RCA: 635] [Article Influence: 52.9] [Reference Citation Analysis (1)] |
| 3. | Pathak I, Kuklina EV, Hollier LM, Busacker AA, Vaughan AS, Wright JS, Coronado F. Cardiomyopathy Prevalence and Pregnancy-Related Mortality: United States, 2010 to 2020. JACC Adv. 2025;4:101692. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 4. | Farroni E, Taggiasco A, Santulli G. Peripartum cardiomyopathy: a clinical review. Vessel Plus. 2025;9:2. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 5. | Irizarry OC, Levine LD, Lewey J, Boyer T, Riis V, Elovitz MA, Arany Z. Comparison of Clinical Characteristics and Outcomes of Peripartum Cardiomyopathy Between African American and Non-African American Women. JAMA Cardiol. 2017;2:1256-1260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 119] [Article Influence: 17.0] [Reference Citation Analysis (1)] |
| 6. | Lasinska-Kowara M, Dudziak M, Suchorzewska J. Two cases of postpartum cardiomyopathy initially misdiagnosed for pulmonary embolism. Can J Anaesth. 2001;48:773-777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 7. | Wang M. Peripartum cardiomyopathy: case reports. Perm J. 2009;13:42-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 8. | Jackson AM, Macartney M, Brooksbank K, Brown C, Dawson D, Francis M, Japp A, Lennie V, Leslie SJ, Martin T, Neary P, Venkatasubramanian S, Vickers D, Weir RA, McMurray JJV, Jhund PS, Petrie MC. A 20-year population study of peripartum cardiomyopathy. Eur Heart J. 2023;44:5128-5141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 41] [Article Influence: 13.7] [Reference Citation Analysis (1)] |
| 9. | Rodriguez Ziccardi M, Siddique MS. Peripartum Cardiomyopathy. 2025 Feb 20. In: StatPearls. Treasure Island (FL): StatPearls Publishing, 2025. [PubMed] |
| 10. | Perea Rojas DM, Seni Hernandez CD, Rojas Torres IL, Olivares Olmos M, Garcia Jarava CM, Gaivao Arciniegas DJ, Seni Hernandez SA, Corrales Calderon LM, Perea Vasquez LE, Salva Camano S. Peripartum Cardiomyopathy: A Case Report of Mortality From a Rare and Potentially Fatal Condition. J Med Cases. 2024;15:171-179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 11. | Behrens I, Basit S, Lykke JA, Ranthe MF, Wohlfahrt J, Bundgaard H, Melbye M, Boyd HA. Hypertensive disorders of pregnancy and peripartum cardiomyopathy: A nationwide cohort study. PLoS One. 2019;14:e0211857. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 39] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 12. | Karaye KM, Sa'idu H, Ishaq NA, Balarabe SA, Ahmed BG, Mohammed IY, Habib AG, Henein MY; Peripartum Cardiomyopathy in Nigeria (PEACE) Registry Investigators. Selenium Deficiency as a Risk Factor for Peripartum Cardiomyopathy. West Afr J Med. 2024;41:209-214. [PubMed] |
| 13. | Karaye KM, Sa'idu H, Balarabe SA, Ishaq NA, Sanni B, Abubakar H, Mohammed BL, Abdulsalam T, Tukur J, Mohammed IY. Selenium supplementation in patients with peripartum cardiomyopathy: a proof-of-concept trial. BMC Cardiovasc Disord. 2020;20:457. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 14. | Chung E, Leinwand LA. Pregnancy as a cardiac stress model. Cardiovasc Res. 2014;101:561-570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 163] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 15. | Khan AA, Fayad FH, Kim CW, Ataklte F, Has P, Nebipasagil A, Arany Z, Poppas A, Wu WC, Choudhary G, Imran TF. Right Ventricular Dysfunction and Adverse Clinical Outcomes in Peripartum Cardiomyopathy: A Meta-Analysis. JACC Adv. 2025;4:102047. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 16. | Hilfiker-Kleiner D, Kaminski K, Podewski E, Bonda T, Schaefer A, Sliwa K, Forster O, Quint A, Landmesser U, Doerries C, Luchtefeld M, Poli V, Schneider MD, Balligand JL, Desjardins F, Ansari A, Struman I, Nguyen NQ, Zschemisch NH, Klein G, Heusch G, Schulz R, Hilfiker A, Drexler H. A cathepsin D-cleaved 16 kDa form of prolactin mediates postpartum cardiomyopathy. Cell. 2007;128:589-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 581] [Cited by in RCA: 576] [Article Influence: 30.3] [Reference Citation Analysis (2)] |
| 17. | Patten IS, Rana S, Shahul S, Rowe GC, Jang C, Liu L, Hacker MR, Rhee JS, Mitchell J, Mahmood F, Hess P, Farrell C, Koulisis N, Khankin EV, Burke SD, Tudorache I, Bauersachs J, del Monte F, Hilfiker-Kleiner D, Karumanchi SA, Arany Z. Cardiac angiogenic imbalance leads to peripartum cardiomyopathy. Nature. 2012;485:333-338. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 482] [Cited by in RCA: 404] [Article Influence: 28.9] [Reference Citation Analysis (5)] |
| 18. | Rana S, Lemoine E, Granger JP, Karumanchi SA. Preeclampsia: Pathophysiology, Challenges, and Perspectives. Circ Res. 2019;124:1094-1112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 514] [Cited by in RCA: 1450] [Article Influence: 207.1] [Reference Citation Analysis (5)] |
| 19. | Spracklen TF, Chakafana G, Schwartz PJ, Kotta MC, Shaboodien G, Ntusi NAB, Sliwa K. Genetics of Peripartum Cardiomyopathy: Current Knowledge, Future Directions and Clinical Implications. Genes (Basel). 2021;12:103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 20. | Kryczka KE, Dzielińska Z, Franaszczyk M, Wojtkowska I, Henzel J, Śpiewak M, Stępińska J, Bilińska ZT, Płoski R, Demkow M. Severe Course of Peripartum Cardiomyopathy and Subsequent Recovery in a Patient with a Novel TTN Gene-Truncating Mutation. Am J Case Rep. 2018;19:820-824. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 21. | Morales A, Painter T, Li R, Siegfried JD, Li D, Norton N, Hershberger RE. Rare variant mutations in pregnancy-associated or peripartum cardiomyopathy. Circulation. 2010;121:2176-2182. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 167] [Cited by in RCA: 150] [Article Influence: 9.4] [Reference Citation Analysis (1)] |
| 22. | Bauersachs J, König T, van der Meer P, Petrie MC, Hilfiker-Kleiner D, Mbakwem A, Hamdan R, Jackson AM, Forsyth P, de Boer RA, Mueller C, Lyon AR, Lund LH, Piepoli MF, Heymans S, Chioncel O, Anker SD, Ponikowski P, Seferovic PM, Johnson MR, Mebazaa A, Sliwa K. Pathophysiology, diagnosis and management of peripartum cardiomyopathy: a position statement from the Heart Failure Association of the European Society of Cardiology Study Group on peripartum cardiomyopathy. Eur J Heart Fail. 2019;21:827-843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 332] [Cited by in RCA: 266] [Article Influence: 38.0] [Reference Citation Analysis (0)] |
| 23. | van Spaendonck-Zwarts KY, van Tintelen JP, van Veldhuisen DJ, van der Werf R, Jongbloed JD, Paulus WJ, Dooijes D, van den Berg MP. Peripartum cardiomyopathy as a part of familial dilated cardiomyopathy. Circulation. 2010;121:2169-2175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 152] [Article Influence: 9.5] [Reference Citation Analysis (1)] |
| 24. | Bauersachs J, Arrigo M, Hilfiker-Kleiner D, Veltmann C, Coats AJ, Crespo-Leiro MG, De Boer RA, van der Meer P, Maack C, Mouquet F, Petrie MC, Piepoli MF, Regitz-Zagrosek V, Schaufelberger M, Seferovic P, Tavazzi L, Ruschitzka F, Mebazaa A, Sliwa K. Current management of patients with severe acute peripartum cardiomyopathy: practical guidance from the Heart Failure Association of the European Society of Cardiology Study Group on peripartum cardiomyopathy. Eur J Heart Fail. 2016;18:1096-1105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 161] [Article Influence: 16.1] [Reference Citation Analysis (8)] |
| 25. | Tibazarwa K, Lee G, Mayosi B, Carrington M, Stewart S, Sliwa K. The 12-lead ECG in peripartum cardiomyopathy. Cardiovasc J Afr. 2012;23:322-329. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 26. | Kampman MA, Balci A, van Veldhuisen DJ, van Dijk AP, Roos-Hesselink JW, Sollie-Szarynska KM, Ludwig-Ruitenberg M, van Melle JP, Mulder BJ, Pieper PG; ZAHARA II investigators. N-terminal pro-B-type natriuretic peptide predicts cardiovascular complications in pregnant women with congenital heart disease. Eur Heart J. 2014;35:708-715. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 76] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 27. | Hu CL, Li YB, Zou YG, Zhang JM, Chen JB, Liu J, Tang YH, Tang QZ, Huang CX. Troponin T measurement can predict persistent left ventricular dysfunction in peripartum cardiomyopathy. Heart. 2007;93:488-490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 68] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 28. | Ray JG, Vermeulen MJ, Bharatha A, Montanera WJ, Park AL. Association Between MRI Exposure During Pregnancy and Fetal and Childhood Outcomes. JAMA. 2016;316:952-961. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 475] [Cited by in RCA: 544] [Article Influence: 54.4] [Reference Citation Analysis (0)] |
| 29. | Koczo A, Acharya D, Ozbay B, Alharethi R, Givertz MM, Elkayam U, Schelbert EB, McNamara DM, Wong TC. CMR and adverse clinical outcomes in peripartum cardiomyopathy. Am Heart J Plus. 2024;48:100484. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 30. | Friedrich MG, Sechtem U, Schulz-Menger J, Holmvang G, Alakija P, Cooper LT, White JA, Abdel-Aty H, Gutberlet M, Prasad S, Aletras A, Laissy JP, Paterson I, Filipchuk NG, Kumar A, Pauschinger M, Liu P; International Consensus Group on Cardiovascular Magnetic Resonance in Myocarditis. Cardiovascular magnetic resonance in myocarditis: A JACC White Paper. J Am Coll Cardiol. 2009;53:1475-1487. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2062] [Cited by in RCA: 1767] [Article Influence: 103.9] [Reference Citation Analysis (1)] |
| 31. | Ferreira VM, Schulz-Menger J, Holmvang G, Kramer CM, Carbone I, Sechtem U, Kindermann I, Gutberlet M, Cooper LT, Liu P, Friedrich MG. Cardiovascular Magnetic Resonance in Nonischemic Myocardial Inflammation: Expert Recommendations. J Am Coll Cardiol. 2018;72:3158-3176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1765] [Cited by in RCA: 1497] [Article Influence: 187.1] [Reference Citation Analysis (2)] |
| 32. | Weinsaft JW, Kim HW, Shah DJ, Klem I, Crowley AL, Brosnan R, James OG, Patel MR, Heitner J, Parker M, Velazquez EJ, Steenbergen C, Judd RM, Kim RJ. Detection of left ventricular thrombus by delayed-enhancement cardiovascular magnetic resonance prevalence and markers in patients with systolic dysfunction. J Am Coll Cardiol. 2008;52:148-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 278] [Cited by in RCA: 224] [Article Influence: 12.4] [Reference Citation Analysis (1)] |
| 33. | Almendro-Delia M, López-Flores L, Uribarri A, Vedia O, Blanco-Ponce E, López-Flores MDC, Rivas-García AP, Fernández-Cordón C, Sionis A, Martín-García AC, Vazirani R, Corbí-Pascual M, Salamanca J, Pérez-Castellanos A, Martínez-Sellés M, Becerra VM, Aritza-Conty D, López-País J, Guillén-Marzo M, Lluch-Requerey C, García-Rubira JC, Núñez-Gil IJ; RETAKO Investigators. Recovery of Left Ventricular Function and Long-Term Outcomes in Patients With Takotsubo Syndrome. J Am Coll Cardiol. 2024;84:1163-1174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 27] [Article Influence: 13.5] [Reference Citation Analysis (1)] |
| 34. | Elkayam U, Akhter MW, Singh H, Khan S, Bitar F, Hameed A, Shotan A. Pregnancy-associated cardiomyopathy: clinical characteristics and a comparison between early and late presentation. Circulation. 2005;111:2050-2055. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 368] [Cited by in RCA: 327] [Article Influence: 15.6] [Reference Citation Analysis (1)] |
| 35. | Regitz-Zagrosek V, Roos-Hesselink JW, Bauersachs J, Blomström-Lundqvist C, Cífková R, De Bonis M, Iung B, Johnson MR, Kintscher U, Kranke P, Lang IM, Morais J, Pieper PG, Presbitero P, Price S, Rosano GMC, Seeland U, Simoncini T, Swan L, Warnes CA; ESC Scientific Document Group. 2018 ESC Guidelines for the management of cardiovascular diseases during pregnancy. Eur Heart J. 2018;39:3165-3241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1874] [Cited by in RCA: 1467] [Article Influence: 183.4] [Reference Citation Analysis (5)] |
| 36. | Koenig T, Bauersachs J, Hilfiker-Kleiner D. Bromocriptine for the Treatment of Peripartum Cardiomyopathy. Card Fail Rev. 2018;4:46-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 37. | Baran DA, Grines CL, Bailey S, Burkhoff D, Hall SA, Henry TD, Hollenberg SM, Kapur NK, O'Neill W, Ornato JP, Stelling K, Thiele H, van Diepen S, Naidu SS. SCAI clinical expert consensus statement on the classification of cardiogenic shock: This document was endorsed by the American College of Cardiology (ACC), the American Heart Association (AHA), the Society of Critical Care Medicine (SCCM), and the Society of Thoracic Surgeons (STS) in April 2019. Catheter Cardiovasc Interv. 2019;94:29-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 653] [Cited by in RCA: 536] [Article Influence: 76.6] [Reference Citation Analysis (2)] |
| 38. | John R, Rajasinghe HA, Chen JM, Weinberg AD, Itescu S, Oz MC, Mancini D. Mechanical circulatory support for peripartum cardiomyopathy: outcomes and considerations. J Heart Lung Transplant. 2006;25:67-74. |
| 39. | Sliwa K, Blauwet L, Tibazarwa K, Libhaber E, Smedema JP, Becker A, McMurray J, Hall D, van Spaendonck-Zwarts K, Mebazaa A, Roos-Hesselink J, Walker N, Wessels P, Kruger A, Essop R, Tsabedze N, Kiage J, Hoffmann B, Mbakwem A, Skudicky D. Bromocriptine and peripartum cardiomyopathy: rationale and clinical data. Eur Heart J. 2010;31:648-653. |
| 40. | De Backer J, Haugaa KH, Hasselberg NE, de Hosson M, Brida M, Castelletti S, Cauldwell M, Cerbai E, Crotti L, de Groot NMS, Estensen ME, Goossens ES, Haring B, Kurpas D, McEniery CM, Peters SAE, Rakisheva A, Sambola A, Schlager O, Schoenhoff FS, Simoncini T, Steinbach F, Sudano I, Swan L, Valente AM; ESC Scientific Document Group. 2025 ESC Guidelines for the management of cardiovascular disease and pregnancy. Eur Heart J. 2025;46:4462-4568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 89] [Article Influence: 89.0] [Reference Citation Analysis (1)] |
| 41. | American College of Obstetricians and Gynecologists' Presidential Task Force on Pregnancy and Heart Disease and Committee on Practice Bulletins—Obstetrics. ACOG Practice Bulletin No. 212: Pregnancy and Heart Disease. Obstet Gynecol. 2019;133:e320-e356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 299] [Article Influence: 49.8] [Reference Citation Analysis (2)] |
| 42. | Easter SR, Rouse CE, Duarte V, Hynes JS, Singh MN, Landzberg MJ, Valente AM, Economy KE. Planned vaginal delivery and cardiovascular morbidity in pregnant women with heart disease. Am J Obstet Gynecol. 2020;222:77.e1-77.e11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 51] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 43. | Patel NH, Agusala V, Chuckaree ID, Wang M, Gangavelli A, Padove L, Sperling LS, Mehta PK. Hyperlipidaemia in the peripartum: epidemiology, physiology, and treatment options. Eur J Prev Cardiol. 2025;zwaf383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 44. | Liu S, Liston RM, Joseph KS, Heaman M, Sauve R, Kramer MS; Maternal Health Study Group of the Canadian Perinatal Surveillance System. Maternal mortality and severe morbidity associated with low-risk planned cesarean delivery versus planned vaginal delivery at term. CMAJ. 2007;176:455-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 510] [Cited by in RCA: 566] [Article Influence: 29.8] [Reference Citation Analysis (1)] |
| 45. | Mehta LS, Warnes CA, Bradley E, Burton T, Economy K, Mehran R, Safdar B, Sharma G, Wood M, Valente AM, Volgman AS; American Heart Association Council on Clinical Cardiology; Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Cardiovascular and Stroke Nursing; and Stroke Council. Cardiovascular Considerations in Caring for Pregnant Patients: A Scientific Statement From the American Heart Association. Circulation. 2020;141:e884-e903. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 292] [Article Influence: 48.7] [Reference Citation Analysis (3)] |
| 46. | Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JG, Coats AJ, Falk V, González-Juanatey JR, Harjola VP, Jankowska EA, Jessup M, Linde C, Nihoyannopoulos P, Parissis JT, Pieske B, Riley JP, Rosano GM, Ruilope LM, Ruschitzka F, Rutten FH, van der Meer P; Authors/Task Force Members; Document Reviewers. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2016;18:891-975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5367] [Cited by in RCA: 4908] [Article Influence: 490.8] [Reference Citation Analysis (3)] |
| 47. | Arrigo M, Blet A, Mebazaa A. Bromocriptine for the treatment of peripartum cardiomyopathy: welcome on BOARD. Eur Heart J. 2017;38:2680-2682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 47] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 48. | Jackson AM, Goland S, Farhan HA, Yaseen IF, Prameswari HS, Böhm M, Jhund PS, Maggioni AP, van der Meer P, Sliwa K, Bauersachs J, Petrie MC. A novel score to predict left ventricular recovery in peripartum cardiomyopathy derived from the ESC EORP Peripartum Cardiomyopathy Registry. Eur Heart J. 2024;45:1430-1439. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 27] [Article Influence: 13.5] [Reference Citation Analysis (5)] |
| 49. | Prameswari HS, Dewi TI, Hasan M, Martanto E, Astuti A, Saboe A, Cool CJ. Clinical Presentation and 6-Month Outcomes of Patients with Peripartum Cardiomyopathy in Indonesia. Int J Gen Med. 2024;17:1073-1083. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 50. | Goland S, Modi K, Bitar F, Janmohamed M, Mirocha JM, Czer LS, Illum S, Hatamizadeh P, Elkayam U. Clinical profile and predictors of complications in peripartum cardiomyopathy. J Card Fail. 2009;15:645-650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 232] [Cited by in RCA: 191] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 51. | Ravi Kiran G, RajKumar C, Chandrasekhar P. Clinical and echocardiographic predictors of outcomes in patients with peripartum cardiomyopathy: A single centre, six month follow-up study. Indian Heart J. 2021;73:319-324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 52. | Shahin O, Shahin H, Heineke H, Biswas M. Predictors of ICU admission in patients with peripartum cardiomyopathy. Eur Heart J. 2022;43:ehac544.837. [DOI] [Full Text] |
| 53. | Sigauke FR, Ntsinjana H, Tsabedze N. Peripartum cardiomyopathy: a comprehensive and contemporary review. Heart Fail Rev. 2024;29:1261-1278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (1)] |
| 54. | Hilfiker-Kleiner D, Haghikia A, Masuko D, Nonhoff J, Held D, Libhaber E, Petrie MC, Walker NL, Podewski E, Berliner D, Bauersachs J, Sliwa K. Outcome of subsequent pregnancies in patients with a history of peripartum cardiomyopathy. Eur J Heart Fail. 2017;19:1723-1728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 90] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/