Published online Mar 9, 2026. doi: 10.5492/wjccm.v15.i1.113426
Revised: September 17, 2025
Accepted: January 7, 2026
Published online: March 9, 2026
Processing time: 187 Days and 6.4 Hours
Acute dyspnoea is a common yet diagnostically complex presentation in emer
To evaluate the diagnostic accuracy and clinical effectiveness of POCUS vs con
A comprehensive literature search was conducted across PubMed, EBSCO Host, MAG Online Library, Elsevier, and ProQuest without date restrictions. Studies were included if they involved adult ED patients undergoing POCUS for dys
Out of 581 identified records, 44 studies met the inclusion criteria, with 19 in
POCUS offers accurate, rapid, and cost-effective evaluation of acute dyspnoea in EDs. Standardised training and further research on outcomes, protocols, and underrepresented populations are essential for consistent, effective implementation.
Core Tip: This systematic review and meta-analysis highlight the diagnostic accuracy of point-of-care ultrasound (POCUS) in evaluating adults presenting with acute dyspnoea in the emergency department. With a pooled sensitivity of 85.6% and specificity of 80.8%, POCUS demonstrates strong potential as a rapid, non-invasive, and radiation-free diagnostic tool. Its bedside applicability and ability to expedite clinical decision-making make it particularly valuable in both high-resource and resource-limited settings. Standardized training and structured protocols are essential to optimize its integration into emergency care and ensure consistent diagnostic performance.
- Citation: Seyala I, Soldera J. Point-of-care ultrasound for evaluating acute dyspnoea in emergency departments: Systematic review and meta-analysis. World J Crit Care Med 2026; 15(1): 113426
- URL: https://www.wjgnet.com/2220-3141/full/v15/i1/113426.htm
- DOI: https://dx.doi.org/10.5492/wjccm.v15.i1.113426
Dyspnoea refers to the subjective experience of shortness of breath. According to the American Thoracic Society, it arises from a “mismatch between central respiratory motor activity and afferent signals received from the airways, lungs, and chest wall structures”[1]. As a significant and frequent presentation in emergency departments (EDs), dyspnoea accounts for approximately 2.4% of all visits[2]. The causes of dyspnoea are highly diverse, encompassing both non-life-threa
In ED, the conventional assessment of dyspnoea typically begins with a detailed clinical history, physical examination, and initial imaging modalities. While these methods remain essential to early evaluation, they frequently lack the precision required for diagnosing complex or atypical cases[5]. Baid et al[3] highlight that although many patients can be accurately diagnosed through clinical assessment alone, approximately 30%-50% still require further diagnostic testing like chest X-ray (CXR) and chest computed tomography (CCT) to confirm or clarify findings. However, both modalities present notable limitations as they expose patients to ionising radiation, raising concerns when used in certain popu
Over recent years, point-of-care ultrasound (POCUS) has gained recognition for its diagnostic accuracy in identifying critical causes of acute dyspnoea[7]. Initially used in trauma settings to detect free intraperitoneal fluid, through the focused assessment sonography in trauma protocol, POCUS has evolved significantly. Today, it is acknowledged as an essential diagnostic tool across numerous medical specialties, including emergency medicine, critical care, acute medicine and respiratory care. The versatility of POCUS allows for its use in a broad range of clinical scenarios, from diagnostic evaluation and resuscitation to surgical intervention and postoperative care[8]. The early incorporation of POCUS into the diagnostic workflow has been shown to accelerate clinical decision-making and improve patient outcomes. As the body of evidence supporting its efficacy continues to grow, there has been a notable shift towards advocating for its routine integration into ED protocols[2].
Applications of POCUS in different clinical fields: Beyond ED, POCUS has shown significant utility in pre-hospital and resource-limited settings. Ünlüer et al[9] underscore the value of early ultrasound application in non-hospital environ
Over the past decade, thoracic ultrasound has become increasingly popular, largely due to its widespread availability in emergency and trauma settings, as well as the growing use of POCUS in medical training programs. Lung ultrasound (LUS) is now an essential competency for the safe and assured performance of procedures such as pleural aspiration and chest drain insertion[12]. Among the characteristic sonographic patterns, A-lines represent horizontal artefacts arising from the pleural line and typically indicate normal, aerated lung tissue[12]. When A-lines are seen together with pre
Other important sonographic features include B-lines, which are vertically oriented hyperechoic artefacts arising from the pleural line, extending to the far field without fading and maintaining a narrow base. The presence of B-lines indicates interstitial oedema, infection, or inflammation[14]. C-lines are hypoechoic, subpleural focal images that appear cone- or dome-shaped and arise from condensed lung tissue without a visible interruption in the visceral pleural line. These are commonly observed in pneumonia, particularly on anterior lung scans, and often reflect adjacent consolidation. Associated signs such as air bronchograms or the shred sign-resulting from ultrasound reflection within air-filled bronchi in consolidated areas-provide additional diagnostic confidence[15]. Pleural effusion is usually recognised by the presence of an anechoic space between the parietal and visceral pleura. A characteristic supportive feature is the sinusoid sign on M-mode, where rhythmic wave-like movements of the lung line towards the pleural line during inspiration reflect the floating lung within the effusion[13].
Despite the widespread enthusiasm for POCUS and its increasingly central role in acute care, important gaps in the evidence base remain. While it is acknowledged as a rapidly evolving modality with expanding applications, further high-quality studies are needed to confirm its impact on clinical outcomes. Current evidence directly linking bedside ultrasound to improved decision-making or measurable patient benefits is still limited[16].
In this context, the present review seeks to provide a comprehensive synthesis of recent and relevant evidence re
This systematic review was prospectively registered with the International Prospective Register of Systematic Reviews (PROSPERO), under registration number CRD42025649145[17]. The review was conducted in strict adherence to the PRISMA 2020 guidelines[18-20].
A detailed literature search was executed across five major electronic databases to identify all potentially relevant studies. These databases included PubMed, MAG Online Library, EBSCO Host, PROQUEST, and Elsevier. The searches were conducted over a three-week period from 13 February to 3 March 2025. The inclusion of multiple databases in
The search strategy combined Medical Subject Headings with relevant free-text terms[21]: PubMed: 13 February 2025: (“Ultrasonography, Point of Care”[tiab] OR “Bedside Ultrasound”[tiab] OR “Lung Ultrasound”[tiab] OR “POCUS”[tiab]) AND (“Dyspnea”[Mesh] OR “Shortness of Breath”[tiab] OR “Respiratory Distress”[tiab] OR “Acute Dyspnea”[tiab] OR “acute respiratory distress syndrome”[tiab] OR “Severe Acute Respiratory Syndrome”[tiab]) AND (“Emergency Service, Hospital”[Mesh] OR “Emergency Department”[tiab] OR “Acute Care”[tiab] OR “Critical Care”[tiab]). MAG Online Library: 25 February 2025: (“Point of Care Ultrasound” OR “Lung Ultrasound” OR “POCUS”) AND (“Dyspnea” OR “Respiratory Distress” OR “ARDS”) AND (“Emergency” OR “Critical Care”). EBSCO Host: 25 February 2025: TI (“Point of Care Ultrasound” OR “Bedside Ultrasound” OR “Lung Ultrasound”) AND TI (“Dyspnea” OR “Respiratory Distress” OR “ARDS”) AND TI (“Emergency Department” OR “Critical Care”). ProQuest: 26 February 2025: TIAB (“Point of Care Ultrasound” OR “Lung Ultrasound” OR “POCUS”) AND TIAB (“Dyspnea” OR “Respiratory Distress” OR “ARDS”) AND TIAB (“Emergency Department” OR “Critical Care” OR “Acute Care”). Elsevier: 3 March 2025: TITLE-ABS-KEY (“Point of Care Ultrasound” OR “Lung Ultrasound”) AND TITLE-ABS-KEY (“Dyspnea” OR “Respiratory Distress”) AND TITLE-ABS-KEY (“Emergency” OR “Critical Care”) (Supplementary material).
Eligibility screening was structured using the Patient, Intervention, Comparison, Outcome framework, which offers a systematic approach to defining inclusion and exclusion criteria:
Patient/population: Adults presenting with dyspnoea or acute shortness of breath in the ED.
Intervention: POCUS used for diagnostic evaluation.
Comparison: Traditional diagnostic modalities, including CXR, computed tomography (CT) scan, or clinical evaluation.
Outcomes: Diagnostic accuracy, time to diagnosis, clinical outcomes, and healthcare resource utilization.
Studies were eligible for inclusion if they were randomized controlled trials (RCT) or observational cohort studies, conducted in ED settings, and focused on adult populations. Studies excluded from the review included those involving paediatric subjects, animal models, non-ED settings, case reports, editorials, review articles, conference abstracts, and grey literature that had not undergone peer review.
Following the initial database search, all duplicates were removed. The remaining studies underwent screening. First, titles and abstracts were independently reviewed for relevance. Subsequently, full-text articles were retrieved and asse
A structured data extraction form was developed and pilot-tested to ensure consistency and completeness. This form collected detailed study information, including first author, publication year, country, study design, clinical setting, sam
The extracted data were compiled using Microsoft Excel, enabling both tabular representation and ease of synthesis. Care was taken to eliminate duplicate data entries or overlapping populations from multiple publications of the same study.
To enhance the reliability of findings and reduce systematic error, a dual-reviewer model was adopted for the assessment of methodological quality and risk of bias. The research supervisor acted as the second reviewer to ensure inter-rater reliability.
The included studies were thoroughly examined to evaluate the relevance and quality of the data and information they provided. The quality assessment focused on ensuring that the studies offered reliable and valid insights pertinent to the research questions. Publication bias was assessed using Deeks' funnel plot asymmetry test, while the methodological quality of the studies was evaluated with QUADAS-2 to ensure the robustness and reliability of the results.
This evaluation process ensured that only studies of sufficient methodological quality were included in the final synthesis, enhancing the reliability and credibility of the review’s conclusions. Using multiple reviewers is a recognized strategy to minimize subjective interpretation and enhance reproducibility in systematic reviews[22].
Data analysis integrated descriptive, qualitative, and quantitative methods, with diagnostic accuracy studies underwent a meta-analysis using MetaDisc software. The software calculated pooled sensitivity, specificity, likelihood ratios, and diagnostic odds ratios (DORs). The overall performance of the test was evaluated using a summary receiver operating characteristic (SROC) curve, along with its area under the curve (AUC), providing a thorough assessment of the sen
Heterogeneity between studies was assessed using the I² statistic and Cochran’s Q test. These tests were essential for determining the variability across studies, allowing us to understand whether the included studies were measuring similar or different phenomena. The I² statistic quantifies the percentage of total variation across studies due to heterogeneity, while Cochran’s Q test assesses whether the observed differences are statistically significant[23,24]. Forest plots were generated to visually assess variation in study estimates, and studies were grouped by design type where appro
A comprehensive search across five databases yielded a total of 581 records: PubMed (n = 231), MAG Online Library (n = 15), EBSCO Host (n = 13), ProQuest (n = 290), and Elsevier (n = 32). Following the removal of six duplicate records, 575 articles were screened. Of these, 455 were excluded after title and abstract review based on predefined exclusion criteria, which included irrelevance to the review topic, case reports, editorials, studies targeting paediatric populations or non-emergency settings, and non-peer-reviewed literature. This step ensured the integrity and specificity of the review to adult patients in acute ED contexts.
This process left 120 records for further screening. Abstract screening performed using the Population, Intervention, Comparator, Outcome, Study design framework, resulted in the exclusion of an additional six studies. A total of 114 articles were sought for full-text review; however, eight could not be retrieved despite attempts to contact authors and institutions, raising concerns about potential availability bias.
The remaining 106 full-text articles were assessed for eligibility. Sixty-two were excluded for the following reasons: Review papers (n = 12), abstract-only publications (n = 2), duplicated datasets (n = 28), irrelevant study focus (n = 13), non-English language (n = 2), and research conducted in non-ED settings (n = 5).
Ultimately, 44 studies met the inclusion criteria and were incorporated into the qualitative synthesis. Of these, 19 studies provided sufficient data for inclusion in the quantitative meta-analysis evaluating diagnostic performance metrics. The included studies represented a spectrum of methodologies, comprising six RCTs and 38 observational studies. This breadth reflects the real-world heterogeneity of POCUS implementation and evaluation across diverse healthcare settings.
The included studies displayed considerable variation in their design, populations, methodologies, and diagnostic contexts. These differences are summarised in Tables 1 and 2[25-55], which categorise the studies based on whether they employed a comparator group.
| Ref. | Year of publication | Country of study | Study design and setting | Number of patients enrolled | Summary of the patient population | Intervention used | Comparison used | Definition of outcome measured | Number of patients who achieved the outcome in the intervention group | Number of patients who achieved the outcome in the comparison group |
| Özkan et al[25] | 2015 | Turkey | RCT | 63 | Patients presenting with dyspnoea in the emergency department | POCUS performed by emergency medicine specialists | Traditional stethoscope examination | Diagnostic performance in identifying causes of dyspnoea | First diagnosis is compatible with final diagnosis: 18 | First diagnosis is compatible with final diagnosis: 19 |
| Pontis et al[26] | 2018 | France | RCT | 107 | Acute dyspnoeic patients | Lung ultrasound | Clinical data without LUS | Diagnostic accuracy and the number of uncertain diagnoses when using LUS compared to clinical data | Number of uncertain diagnoses mean: 2.2 ± 1.7 | Number of uncertain diagnoses mean: 2.9 ± 1.8 |
| Zare et al[2] | 2022 | Iran | RCT | 103 | Patients with undifferentiated acute dyspnoea | POCUS performed by emergency specialists | Standard diagnostic approach without POCUS | Impact of POCUS on early patient disposition decisions in the ED | Mean time of diagnosis 42.61 minutes | Mean time of diagnosis 79.33 minutes |
| Baker et al[27] | 2020 | Australia | RCT | 442 | Patients aged over 60 years presenting with dyspnoea | POCUS performed by non-expert clinicians after brief training | Standard diagnostic assessment without the use of LUS | (1) Accurate diagnosis of pulmonary oedem; and (2) Length of ED stay | Correct diagnosis 60 out of 68 | Correct diagnosis 53 out of 64 |
| Beyer et al[7] | 2021 | United States | Multicentre, prospective, cohort study | 156 | Patients with undifferentiated dyspnoea | POCUS by trained ED | POCUS performed by USS team | Effectiveness of POCUS in narrowing diagnostic uncertainty in dyspnoeic patient | Most likely diagnosis changed in 40% | Diagnosis changed in 32% |
| Baid et al[3] | 2022 | India | Prospective observational study | 237 | Patients presenting with acute dyspnoea | POCUS performed by emergency physicians | Final composite diagnoses made by two emergency medicine consultants | To determine the diagnostic accuracy of POCUS in various causes of acute onset dyspnoea | Median time for diagnosis 16 minutes | Median time for diagnosis 170 minutes |
| Pivett et al[28] | 2015 | Italy | Multicenter, prospective cohort study | 1005 | Patients presenting to the ED with acute dyspnoea | LUS | Initial assessment, CXR and BNP | Diagnostic accuracy of LUS compared to CXR | Sensitivity 97%; specificity 97.4% | CXR: Sensitivity 69.5%; specificity 82.1%; BNP sensitivity 75% specificity 61.7% |
| Nakao et al[29] | 2021 | Canada | Prospective diagnostic accuracy study | 324 | Patients with SOB and cough suspected to be COVID or COPD | POCUS | Patients not evaluated by POCUS | ED length of stay and time to treatment | Time to treatment 61 minutes | Time to treatment 92 minutes |
| Mantuan et al[30] | 2016 | United States | Prospective observational study | 57 | Patients presenting with acute dyspnoea | Triple scan (LUS, Echo and IVC) | Clinical assessment | Compare diagnostic accuracy before and after TS | Diagnostic accuracy 77% | Diagnostic accuracy 53% |
| Papanagnou et al[4] | 2017 | United States | Prospective observational study | 115 | Adult patients with undifferentiated dyspnoea | Bedside USS | Diagnosis before USS | (1) Post-ultrasound diagnoses matched final diagnoses; (2) Frequency ultrasound altered providers’ leading diagnoses; and (3) Change in physicians’ diagnostic confidence | Matched final diagnosis 63% of the time | Matched final diagnosis 69% of the time |
| Buhumaid et al[16] | 2019 | United States | Prospective observational study | 128 | Patients presenting with CP and SOB to ED | POCUS | CXR | (1) How use POCUS in ED differentials; and (2) Evaluate accuracy of POCUS vs CXR | POCUS sensitivity 89%. Specificity 74% | CXR sensitivity 38%. Specificity 96% |
| Núñez-Ramos et al[31] | 2024 | Colombia | Analytical cross-sectional study | 209 | Patients with dyspnoea, suspected ACS, HF, shock and/or HTN | POCUS | Final diagnosis | Evaluate initial POCUS diagnosis with the diagnosis on discharge | Diagnostic accuracy: HF 99, ACS 98, Shock 98 | |
| Pirozzi et al[6] | 2014 | Italy | Single center randomized prospective study | 168 | Patients presenting with undifferentiated dyspnoea | POCUS and routine laboratory | USS in 1 hour | Diagnostic accuracy by using POCUS | (1) Diagnostic concordance 0.94; and (2) Wrong initial diagnosis 5% | (1) Diagnostic concordance 0.22; and (2) Wrong initial diagnosis 50% |
| Martindale et al[32] | 2013 | United States | Prospective, blinded, observational study | 20 | Resident physicians in ED, IM and radiology | LUS | CXR | To evaluate the ability of residents to recognize pulmonary oedema on LUS | Sensitivity 99%. Specificity 61% | Sensitivity 97%. Specificity 32% |
| Sharma et al[1] | 2025 | India | Prospective cohort study | 104 | Patients presenting with acute shortness of breath | Bedside LUS, NT-pro BNP, and clinical parameters | Clinical assessment using modified Boston criteria and NT-pro BNP levels alone | Accuracy of LUS/Pro BNP in differentiating pulmonary from cardiac causes of SOB | LUS sensitivity of 100%, specificity of 62.3% | NT-pro BNP 100% sensitivity, 91.8% specificity 100% |
| Bitar et al[33] | 2015 | Kuwait | A prospective observational study | 61 | Patients presenting with symptoms and signs of pulmonary oedema | LUS and Echo | BNP levels | Diagnostic accuracy of LUS and Echo in pulmonary oedema | Sensitivity 92%. Specificity 90.9% | Sensitivity 97.9%. Specificity 71.4% |
| Zare et al[2] | 2022 | Iran | Prospective multicentre study | 103 | Patients with acute undifferentiated dyspnoea | Early POCUS in ED | Routine USS | Evaluate the role of bedside USS to identify the cause of undifferentiated dyspnoea and faster disposition in ED | Mean randomization-to-diagnosis time 4261 minutes | 79.33 minutes |
| Umuhire et al[5] | 2019 | Rwanda | Prospective observational study | 99 | Patients presenting with dyspnoea | Multi-organ POCUS scans | Pre- and post-ultrasound clinical assessments | Changes in leading diagnosis and diagnostic accuracy | Leading diagnosis matched the final discharge diagnosis in 89% of cases | Leading diagnosis matched the final discharge diagnosis in 34.3% of cases |
| Sartini et al[34] | 2017 | Italy | Prospective study | 236 | Patients presenting with acute dyspnoea | LUS performed to diagnose acute HF | Diagnostic accuracy of LUS compared with CXR and NT-pro BNP levels | Accuracy of LUS, CXR, and NT-pro BNP in the early diagnosis of AHF | Sensitivity 57.73%. Specificity 87.97% | CXR: 74.49%, 86.26. BNP 97.59%, 27.56 |
| Yahia et al[35] | 2022 | Egypt | Prospective observational study | 47 | Patients presenting with dyspnoea and symptoms suggestive of AHF | LUS | Plasma Pro BNP | Accuracy and usefulness of LUS in assessment | Sensitivity of 91.9%. Specificity of 100% | Sensitivity of 100%, specificity of 60% |
| Secko et al[36] | 2022 | United States | Prospective observational study | 102 | Patients with undifferentiated dyspnoea | Bedside LUS | Physical examination | Diagnostic accuracy of LUS vs physical examination | Accuracy is the same for CPD and asthma. LUS more accurate in pneumonia, pulmonary oedema and effusion | Accuracy is the same for CPD and asthma. LUS more accurate in pneumonia, pulmonary oedema and effusion |
| Pivetta et al[28] | 2019 | Italy | RCT | 518 | Patients with acute dyspnoea | Lung ultrasound was integrated with clinical assessment | CXR and BNP | Accuracy and utility of combining LUS/clinical assessment vs CXR/BNP | Diagnostic accuracy AUC 0.95 | AUC 0.78 |
| Arvig et al[37] | 2023 | Denmark | RCT | 206 | Patients presented with dyspnoea | POCUS within 1 hour of arrival plus two additional POCUS | A single POCUS within 1 hour of arrival | A reduction of dyspnoea measured on a verbal dyspnoea scale | The mean difference in VDS -1.09 | The mean difference in VDS -1.66 |
| Sorlini et al[38] | 2021 | Italy | Single retrospective study | 384 | Patients with symptoms and signs of respiratory infection | POCUS performed at the time of admission or shortly afterwards | CXR | Assess diagnostic accuracy of POCUS | Sensitivity 92% CXR 64.9% | Sensitivity 73.9%. Specificity 55.9% |
| Gibbons et al[39] | 2021 | United States | Prospective cohort study | 110 | Patients presenting with symptoms of COVID19 | Portable LUS, CXR and CT (if abnormal) | CXR and CT | Compare test characteristics of LUS and CXR in COVID19 | LUS sensitivity 97.6%. Specificity 33.3% | CXR sensitivity 96.9%. Specificity 44.4% |
| Bianchi et al[40] | 2022 | Italy | Prospective cohort study | 360 | Patients with symptoms suggestive of COVID-19 | POCUS by emergency physician | CT and PCR | Analyse diagnostic power of POCUS for COVID19 | Sensitivity 86%, specificity 70%, accuracy 77% | CXR (sensitivity 64.9%, specificity 88.5% and accuracy 79.6%) BNP (sensitivity 80%, specificity 69.7% and accuracy 75%) |
| Carlino et al[41] | 2009 | Italy | Prospective cohort study | 100 | Patients presenting with acute dyspnoea | Integrated ultrasound examination | CXR and BNP | To identify accuracy of an integrated ultrasound approach for diagnosis of acute heart failure | Sensitivity 100%, specificity 82% and accuracy 89% | CXR (sensitivity 64.9%, specificity 88.5% and accuracy 79.6%) BNP (sensitivity 80%, specificity 69.7% and accuracy 75%) |
| Sforza et al[42] | 2017 | Italy | Diagnostic accuracy study | 68 | Patients presenting with acute dyspnoea to ED | Multi-organ ultrasound using a pocket-sized ultrasound device | CXR | To test the usefulness and accuracy of multi-organ USS | Sensitivity 92.6, specificity 80.5% and accuracy 85.3% | CXR (sensitivity 75%, specificity 85.4% and accuracy 82%) |
| Wooten et al[43] | 2019 | United States | Prospective observational cohort study | 99 | Patients presenting with dyspnoea to ED | Bedside LUS | CXR | Assess accuracy of LUS and CXR in pulmonary oedema | Sensitivity 96%, specificity 90% | Sensitivity 65%. Specificity 95% |
| Ref. | Year of publication | Country of study | Study design and setting | Number of patients enrolled | summary of the patient population | Intervention used | Comparison used | Definition of outcome measured | TP | FP | TN | FN |
| Özkan et al[25] | 2014 | Turkey | Prospective cohort study | 96 | Patients presented with dyspnoea | Bedside LUS | NA | Accuracy of bedside USS to diagnose accuracy of dyspnoea | 41 | 2 | 45 | 4 |
| Sorlini et al[38] | 2020 | Italy | Single retrospective study | 384 | Patients with symptoms and signs of respiratory infection | POCUS performed at the time of admission or shortly afterwards | NA | Assess diagnostic accuracy of POCUS | 264 | 34 | 63 | 23 |
| Markarian et al[54] | 2020 | France | Multicenter prospective observational cohort study | 100 | Patients with acute dyspnoea | LUS | NA | Accuracy of LUS score to detect respiratory support need | ||||
| Sanjan et al[45] | 2019 | India | Prospective observational study | 73 | Patients diagnosed with ARDS according to Berlin criteria | POCUS | No comparison group: The study focused on analysing lung ultrasound findings within the cohort | The utility of POCUS in the initial assessment of ARDS | ||||
| Gibbons et al[39] | 2021 | United States | Prospective cohort study | 110 | patients presenting with symptoms of COVID-19 | Portable LUS, CXR and CT (if abnormal) | NA | Compare test characteristics of LUS and CXR in COVID-19 | 79 | 19 | 10 | 2 |
| Bianchi et al[40] | 2021 | Italy | Prospective cohort study | 360 | Patients with symptoms suggestive of COVID-19 | POCUS by emergency physicians | NA | Analyse diagnostic power of POCUS for COVID-19 | 120 | 64 | 156 | 20 |
| Msolli et al[14] | 2021 | Tunisia | Prospective observational study | 700 | Patients admitted for acute dyspnoea as chief complaint | LUS performed by a pair of ED residents | NA | To assess the accuracy and reproducibility of USS in CHF | 325 | 82 | 247 | 82 |
| Vauthier et al[46] | 2021 | France | Prospective diagnostic accuracy study | 103 | Patients presenting to the ED with acute dyspnoea | POCUS algorithm (LUS and Echo) | No comparison group | Evaluate diagnostic performance of pocus in patients with acute SOB | 40 | 4 | 57 | 2 |
| Altuğ et al[47] | 2024 | Turkey | A single-center prospective study | 100 | Patients with ARDS as per Berlin criteria | LUS score to investigate prognosis | No comparison group | Prognostic value of the LUS Score in ARDS patients | 48 | 4 | 44 | 4 |
| Patel et al[13] | 2018 | Pakistan | observational study | 50 | patients with ARDS | Bedside LUS in emergency protocol | No control group | Determine the accuracy of BLUE protocol in ARDS | 45 | 6 | 194 | 5 |
| Glöckne et al[48] | 2020 | Germany | Prospective mono-center study | 102 | Adult patients presenting with undifferentiated acute dyspnoea | POCUS in diagnosing AHF and monitoring the therapeutic success | Not stated | Evaluates the accuracy of POCUS in diagnosing AHF and monitoring the therapeutic success | 27 | 1 | 39 | 22 |
| Cibinel et al[49] | 2011 | Italy | Prospective study | 56 | Patients presented with acute dyspnoea | PLUS | Not stated | Evaluate diagnostic accuracy of PLUS to differentiate cardiogenic and non-cardiogenic causes of acute dyspnoea | 25 | 5 | 24 | 2 |
| Carlino et al[41] | 2018 | Italy | Prospective observational study | 100 | Patients presenting with acute dyspnoea | Integrated ultrasound examination | Not stated | To identify accuracy of an integrated ultrasound approach for diagnosis of acute heart failure | 37 | 6 | 55 | 2 |
| Liu et al[50] | 2016 | China | A prospective, single-blind trial | 98 (27 with pneumonia HF) | Patients with acute dyspnoea in the ED | LUS and TTE were performed within 30 minutes | Not stated | Identify cardiogenic from infection cause of acute SOB | 26 | 1 | 49 | 22 |
| Ahn et al[51] | 2017 | South Korea | Single-center prospective observational study | 308 | Patients with dyspnoea, CP and symptomatic hypotension | Implementation of the SEARCH 8Es POCUS protocol | NA | POCUS ability to narrow differential diagnoses and increase physicians’ diagnostic confidence in patients with SOB, CP and hypotension | 281 | 2 | 306 | 27 |
| House et al[52] | 2020 | Nepal | Prospective cross-sectional study | 280 | Patients presenting with dyspnoea to the ED | LUS with pre and post scan diagnosis | NA | Changes in diagnosis and management following LUS | 110 | 45 | 57 | 68 |
| Farahmand et al[53] | 2020 | Iran | Diagnostic accuracy study | 120 | Patients were referred to ED with acute dyspnoea | Heart, lung and IVC USS | NA | Accuracy of USS to diagnose decompensated HF | 88 | 89 | 31 | 32 |
| Sforza et al[42] | 2017 | Italy | Diagnostic accuracy study | 68 | Patients presenting with acute dyspnoea to ED | Multi-organ ultrasound using a pocket-sized ultrasound device | NA | To test the usefulness and accuracy of multi-organ USS | 25 | 8 | 33 | 2 |
| Markarian et al[44] | 2019 | France | Observational single-centre study | 137 | Patients over age 64 admitted to the ED for acute dyspnoea with hypoxia | Early LUS by ED doctors followed by usual care | NA | Accuracy of LUS to evaluate severity of acute dyspnoea in elderly patients | 40 | 1 | 93 | 3 |
| Öhman et al[55] | 2019 | Finland | Prospective diagnostic study | 100 | Patients with undifferentiated dyspnoea | Cardiothoracic and LUS | NA | Accuracy of cardiothoracic and LUS in diagnosing HF | 50 | 2 | 48 | 0 |
| Wooten et al[43] | 2019 | United States | prospective observational cohort study | 99 | Patients presenting with dyspnoea to ED | Bedside LUS and CXR | NA | Assess accuracy of LUS and CXR in pulmonary oedema | 31 | 7 | 60 | 1 |
Table 1 presents 29 studies that benchmarked the diagnostic performance of POCUS against established modalities such as CXR, CT, or clinical assessment protocols. These studies varied in sample size, from relatively small cohort studies (n = 20) to large-scale multicenter trials with over 1000 participants and were conducted between 2013 and 2024. Most were situated in academic or tertiary care EDs in North America, Europe, and Asia. The high-resource setting of these studies implies greater access to advanced imaging tools and trained personnel, potentially enhancing diagnostic accuracy.
Table 2 presents a synthesis of 21 studies investigating the use of POCUS as an independent diagnostic modality without comparison to alternative imaging techniques. Due to insufficiently reported or ambiguous data, two studies were excluded from the meta-analysis. Consequently, 19 studies conducted between 2014 and 2024, encompassing sample sizes ranging from 50 to 700 participants, were incorporated into the quantitative analysis.
Notably, six studies appeared in both tables due to their use of multiple diagnostic benchmarks. This dual categorisation highlights the evolving use of POCUS as both a supplementary and primary diagnostic tool in emergency medicine.
Most studies targeted adult patients presenting with acute respiratory distress or undifferentiated dyspnoea. The spectrum of investigated conditions included pneumonia, pleural effusion, pulmonary oedema, pneumothorax, and COPD exacerbations. While many studies focused on a single diagnostic category, others incorporated POCUS into com
A notable source of variation was operator expertise. Some studies relied exclusively on highly experienced radiolo
The geographic distribution revealed a research skew towards high-income countries, including the United States, Italy and France. The absence of robust data from low- and middle-income countries represents a key gap in the literature, especially considering the potential of POCUS to serve as a low-cost, high-impact diagnostic tool in resource-limited ED settings.
The primary aim of the quantitative synthesis was to assess the diagnostic efficacy of POCUS in the evaluation of acute dyspnoea, relative to established diagnostic modalities. The 19 studies included in the meta-analysis provided sufficient data to extract true positive (TP), false positive (FP), true negative (TN), and false negative (FN) values, enabling calcu
Although 29 studies included comparators, substantial heterogeneity in design, reporting standards, and diagnostic endpoints rendered a formal meta-analysis of these studies methodologically unsound. Consequently, only those studies with comparable methodology and raw accuracy data were synthesised quantitatively.
The 29 comparator studies could not be meta-analysed due to multiple layers of heterogeneity:
Comparator variation: Studies benchmarked POCUS against different reference standards [chest radiography, CT, clinical examination, or biomarkers such as brain natriuretic peptide (BNP)], precluding a unified quantitative synthesis.
Outcome diversity: While some reported diagnostic accuracy, others focused on time to diagnosis, prognostic perfor
Study design and operator variability: The included studies ranged from RCTs to prospective and retrospective cohorts, with POCUS performed by experts in some cases and by non-expert or minimally trained clinicians in others.
Population differences: Studies enrolled patients with diverse underlying pathologies [pneumonia, acute respiratory distress syndrome (ARDS), COVID-19, acute heart failure, pleural effusion], leading to highly variable diagnostic targets.
The included studies used a variety of POCUS applications, ranging from focused lung ultrasonography to broader protocols that integrated cardiac, pleural, and venous evaluations. This methodological diversity underscores the adaptability of POCUS in emergency care but also complicates attempts at direct standardized comparisons.
The flexibility of POCUS, while a clinical strength, introduces variability that poses challenges to meta-analytic modelling. Nevertheless, the consistency of core findings, namely, the high diagnostic value of POCUS in identifying or excluding pathology in acutely dyspnoeic patients, supports its clinical utility, particularly when used as part of a structured diagnostic approach.
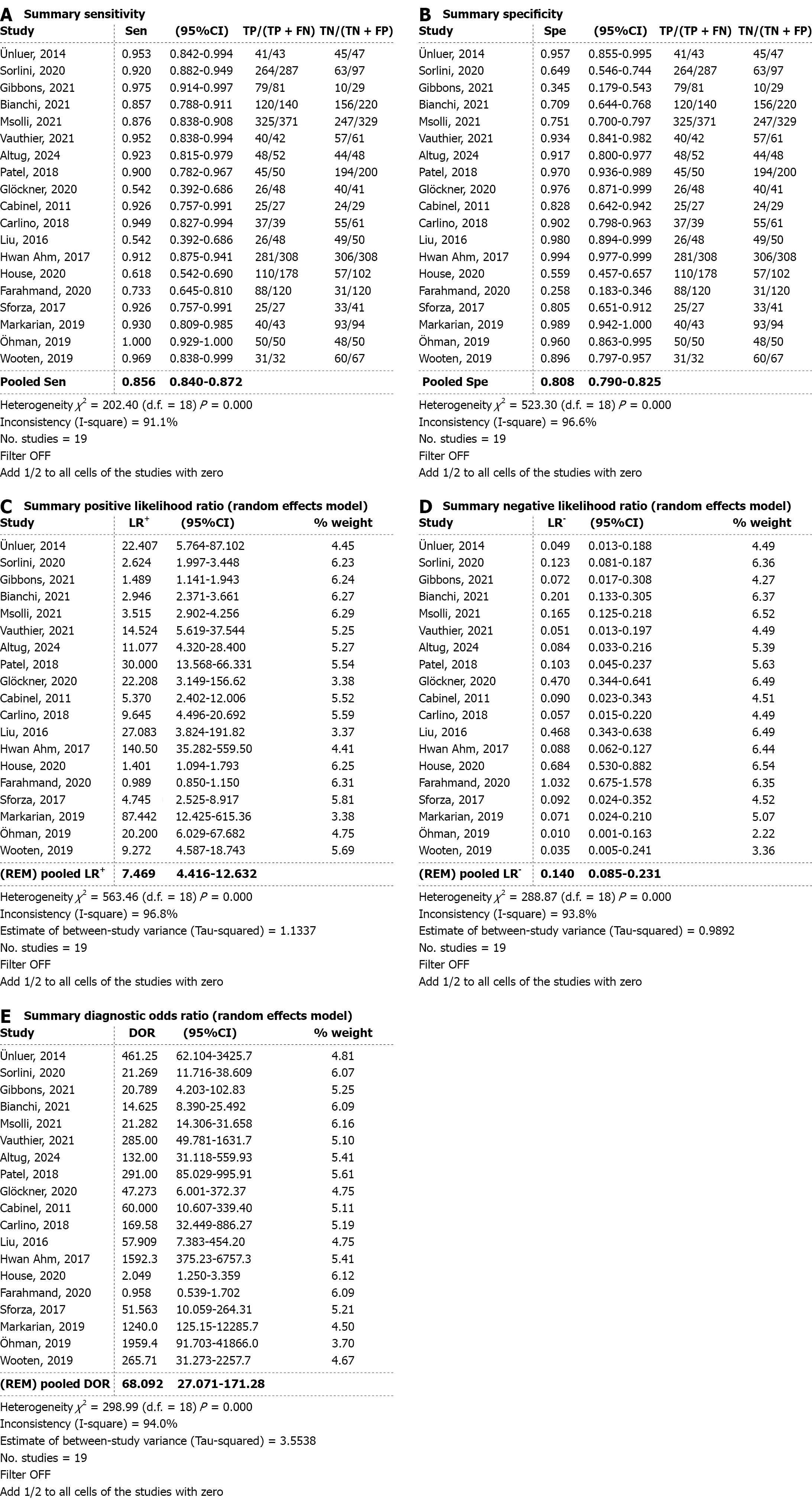
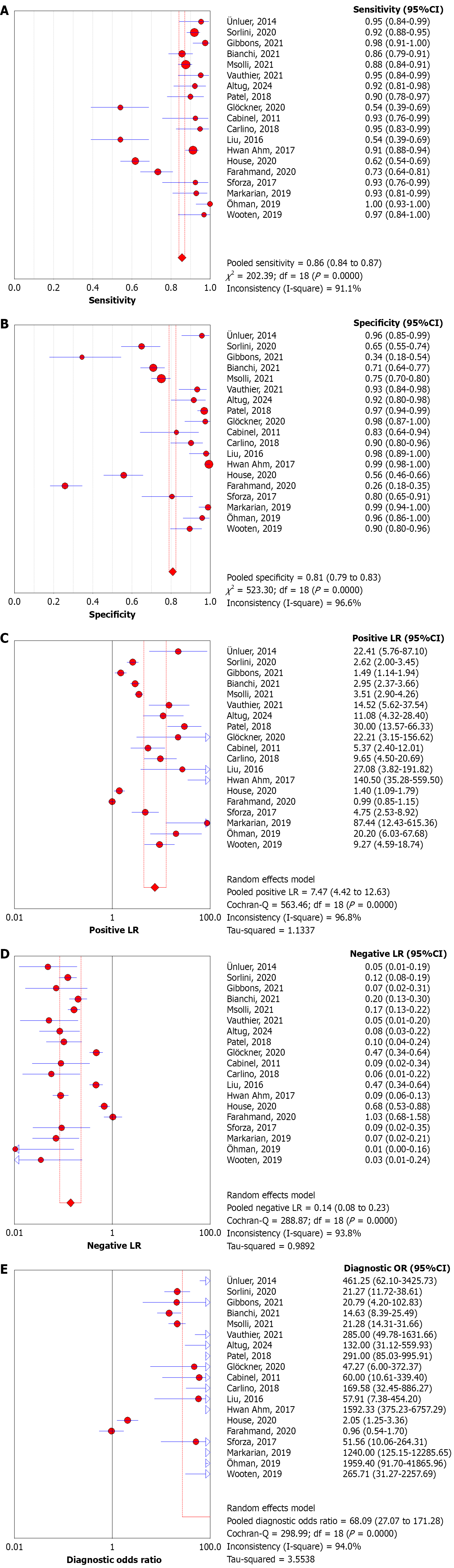
Sensitivity: As shown in both Figures 1A and 2A, the pooled sensitivity for POCUS across all included studies is 0.856 (95%CI: 0.840-0.872), indicating a strong ability of the modality to detect TP cases. Sensitivity values are highly favourable in the majority of studies, such as: Öhman et al[55]: 1.000, Markarian et al[44]: 0.930, Gibbons et al[39]: 0.975. These results underscore POCUS’s effectiveness in not missing cases of disease, a vital consideration in high-risk or acute care settings. However, certain studies report markedly reduced sensitivity: Glöckner et al[48]: 0.542, Liu et al[50]: 0.542. This range suggests variability in operator proficiency, ultrasound protocols, and disease spectrum. Such heterogeneity raises con
From a clinical perspective, high sensitivity supports the utility of POCUS as a “rule-out” tool, especially in emergency situations where timely exclusion of life-threatening conditions (e.g., pneumothorax, intra-abdominal bleeding) is essential.
Specificity: The pooled specificity is 0.808 (95%CI: 0.790-0.825), suggesting that POCUS accurately identifies around 81% of patients who do not have the disease (Figures 1B and 2B). However, specificity values vary considerably between studies: Patel et al[13]: 0.970, Ahn et al[51]: 0.994, Gibbons et al[39]: 0.345, Farahmand et al[53]: 0.258. This wide distribution implies inconsistent ability to rule out disease across studies. Lower specificities might result from FP due to over-interpretation of equivocal findings or image artifacts, especially when less experienced operators are involved.
In practice, while POCUS may be sensitive, its specificity may be insufficient in some settings to confirm diagnoses definitively, highlighting the need for follow-up with more comprehensive imaging modalities such as CT or MRI when diagnostic certainty is needed.
The positive likelihood ratios: The pooled likelihood ratios (LR+) is 7.469 (95%CI: 4.416-12.632) as shown in Figures 1C and 2C. This implies that a patient with a positive POCUS result is approximately 7.5 times more likely to actually have the disease compared to a patient with a negative test.
Some studies, such as Ahn et al[51] and Markarian et al[54], report particularly high LR+ values (140.5 and 87.442, respectively), indicative of POCUS's strong confirmatory power in optimized clinical environments.
Negative LR: The pooled LR- is 0.140 (95%CI: 0.085-0.231), indicating that a negative POCUS result decreases the likelihood of disease by approximately 86% (Figures 1D and 2D). This value approaches the < 0.1 threshold typically associated with strong evidence to rule out disease.
In practice, a negative result substantially lowers the post-test probability and can support safer patient triage, although it may not be definitive.
DOR: The DOR is 68.092 (95%CI: 27.071-171.28), as shown in Figures 1E and 2E, a composite metric that combines both sensitivity and specificity. Öhman et al[55]: DOR = 1959.4 (perfect test performance). Farahmand et al[53]: DOR = 0.958 (barely better than chance).
This wide disparity again points to substantial methodological variability across studies. While the high pooled DOR supports the conclusion that POCUS is highly effective in experienced hands, the inconsistency underscores the necessity for context-specific interpretation.
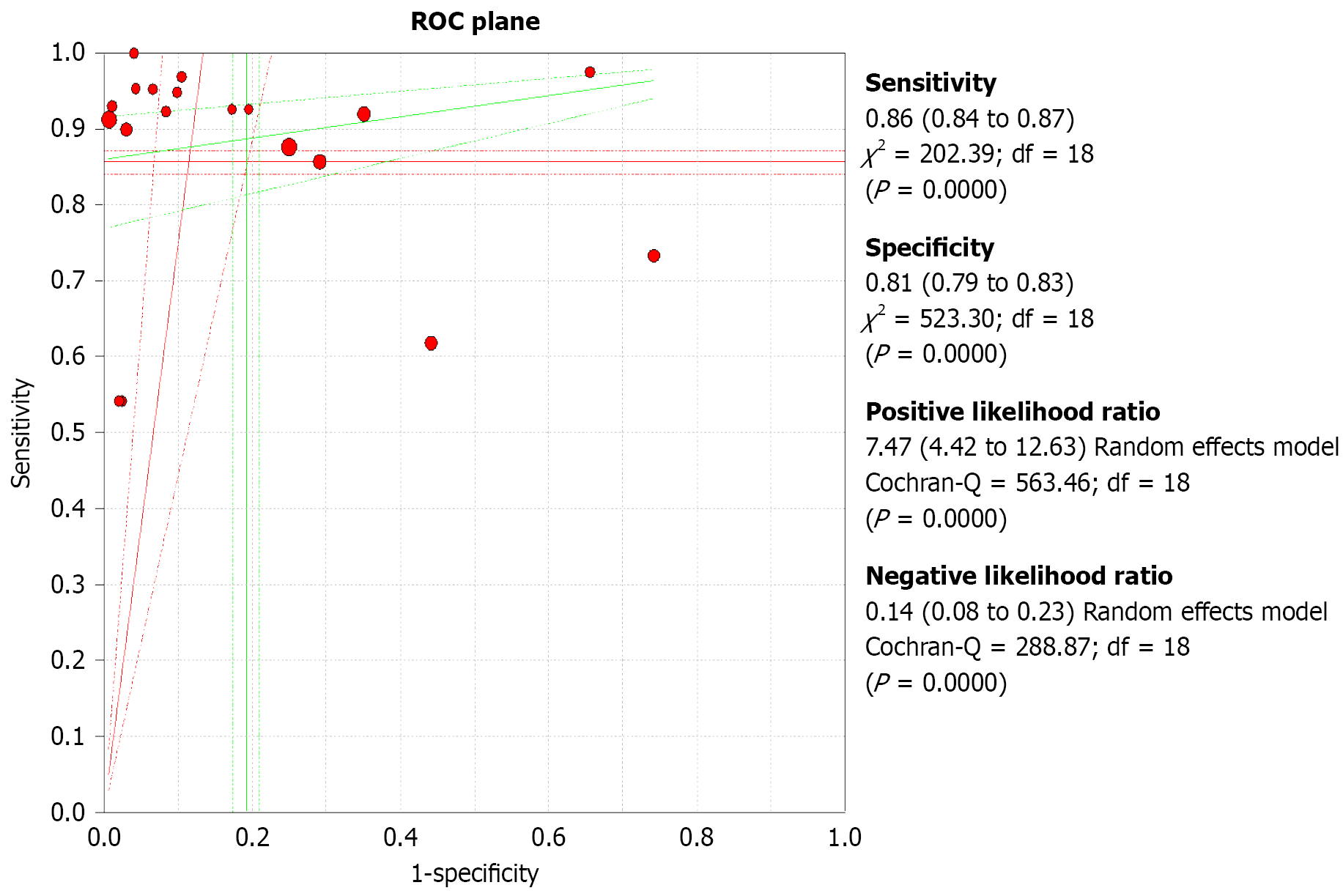
ROC curve analysis: As shown in Figure 3, the ROC curve in this dataset lies close to the upper left corner of the graph, indicating high diagnostic accuracy across most studies. A curve in this position implies that the test is capable of identifying most TP while minimizing FP, a desirable trait in clinical diagnostics.
The AUC, although not explicitly stated in the data, would likely be greater than 0.9, based on the pooled sensitivity (0.856) and specificity (0.808). An AUC > 0.9 is considered excellent, indicating that the diagnostic test performs well across different thresholds and populations.
Each of the included studies underwent a comprehensive and meticulous examination to assess the relevance, reliability, and validity of the data and findings. This quality assessment not only ensured the inclusion of studies that directly addressed the research questions but also evaluated the methodological rigor of each study to determine its robustness and applicability. To assess the presence of publication bias, Deeks' funnel plot asymmetry test was used, providing a visual and statistical method for identifying potential bias in the studies included in the analysis. Furthermore, the methodological quality of the studies was evaluated using QUADAS-2, a tool specifically designed for assessing the risk of bias in diagnostic accuracy studies[56]. This multi-step process aimed to enhance the reliability and credibility of the review's findings by ensuring that only high-quality, relevant studies contributed to the final conclusions. However, despite these measures to ensure methodological quality and reduce bias, several common limitations were observed across the included studies:
Lack of blinding: Many studies did not blind POCUS operators to the patients’ clinical presentations or outcomes, which could inflate diagnostic accuracy due to observer bias.
Non-independent reference standards: Some studies used imperfect comparators, such as clinical diagnosis without imaging confirmation, introducing potential verification bias.
Selection bias: A number of studies excluded unstable or critically ill patients, potentially underestimating the utility of POCUS in the most vulnerable groups.
Challenges in data presentation and extraction: In some studies, key diagnostic metrics were presented graphically rather than in tabulated form, making data extraction more challenging and potentially introducing minor interpretation errors.
This systematic review and meta-analysis offer a thorough assessment of the diagnostic accuracy of POCUS across a range of ED contexts. A key observation from the literature is the variability in how POCUS was assessed in relation to other diagnostic methods, including CXR, CCT, and physical examination. Importantly, POCUS was not typically evaluated in isolation; instead, its diagnostic utility was often measured alongside or against these more traditional approaches. These comparisons were typically embedded within real-time ED clinical workflows, where clinicians faced decisions between immediate bedside imaging and more delayed but potentially more definitive diagnostic tests.
Across multiple studies, POCUS was commonly employed as an initial diagnostic modality, with its findings sub
In assessing comparative diagnostic accuracy, several studies, particularly those using multimodal imaging approaches or expert adjudication panels, found that combining POCUS with adjunctive techniques such as echocardiography or inferior vena cava (IVC) assessment produced diagnostic yields comparable to, and in certain contexts surpassing, those of conventional imaging pathways, as illustrated by Sforza et al[42]. The diagnostic advantage of POCUS in these settings stemmed not solely from image acquisition, but from its capacity to synthesise real-time functional data, such as intravascular volume status and cardiac performance, with structural findings, thereby enhancing its overall clinical applicability.
In addition to imaging-based comparisons, POCUS was frequently assessed against conventional physical examination techniques. Özkan et al[25] directly compared LUS to auscultation using a stethoscope and comprehensive clinical examination. This study consistently demonstrated the superiority of POCUS in detecting the underlying causes of dyspnoea, particularly when identifying conditions like pneumonia and pulmonary oedema that may be clinically subtle or masked by comorbidities.
Several investigations also assessed POCUS's role in diagnosing heart failure relative to biomarkers like NT-proBNP and to chest radiographs. For example, Sartini et al[34] and Yahia et al[35] found that LUS outperformed CXR and approached the diagnostic reliability of NT-proBNP, particularly when integrated with clinical assessment. This multi-modal strength was echoed in studies by Pivetta et al[28] and Carlino et al[41], where POCUS was especially effective when combined with echocardiographic indicators such as left atrial size and ejection fraction.
Furthermore, in the context of ARDS, Sanjan et al[45] and Altuğ et al[47] demonstrated that POCUS reliably identified sonographic features such as coalescent B-lines and lung consolidations. These findings correlated well with clinical severity and supported the role of LUS in both diagnosis and monitoring.
Therefore, POCUS was not simply appraised in binary terms (i.e., superior or inferior to other modalities); instead, its evaluation encompassed a broader spectrum of clinical relevance, operational efficiency, and diagnostic versatility. The collective findings indicate that although POCUS may not consistently replace gold-standard imaging, its ease of deploy
Thoracic ultrasound and pneumonia: Gibbons et al[39] demonstrated that LUS offers high diagnostic accuracy for pneumonia across diverse clinical settings and in both adult and paediatric populations. In their study, LUS exhibited sensitivity comparable to that of non-contrast CT, with subsequent expert review reclassifying both initially missed LUS cases as TP. Although CXR remains more widely available, its sensitivity reached only 70%, even when employing a generous threshold for abnormal findings. Furthermore, Abid et al[15], in their systematic review of 1045 articles, concluded that LUS is a highly effective tool for the early detection of pneumonia, demonstrating strong sensitivity, specificity, and diagnostic accuracy across all age groups, including children, adults, and the elderly. They highlighted LUS as a safe, accessible, and cost-effective alternative to traditional imaging modalities like CXR, particularly valuable in diverse clinical settings such as ED, ITUs, hospital wards, and outpatient clinics when performed by trained professionals.
Thoracic ultrasound and COVID-19 pneumonia: Sorlini et al[38] reported notably high diagnostic accuracy of POCUS during the COVID-19 pandemic, with a sensitivity of 92% for identifying SARS-CoV-2 pneumonia. This elevated performance, relative to aggregated values from broader meta-analyses, likely reflects the consistent pathological and sonographic features characteristic of COVID-19 pneumonia, which render it particularly amenable to ultrasound-based detection. Their results highlight the critical role of POCUS in emergency contexts, where rapid, bedside, radiation-free diagnostics are indispensable and where diagnostic targets tend to be more clearly defined. Similarly, in Bianchi et al[40] prospective observational study carried out during the initial phase of the COVID-19 pandemic (March–May 2020) at the Santa Maria Nuova ED in Italy, LUS exhibited robust diagnostic accuracy, achieving an overall sensitivity of 86%, outperforming chest CT, which demonstrated a sensitivity of 67%.
LUS in ARDS: In observational study conducted over a two-month period by Patel et al[13] in the Emergency Medicine Department of a tertiary care hospital located in Ahmedabad (Pakistan), LUS achieved a diagnostic accuracy of 90.31% in patients presenting with acute respiratory distress, facilitating timely initiation of appropriate management in the ED. By applying the BLUE protocol, clinicians were able to avoid the immediate transfer of hemodynamically unstable patients for advanced imaging such as CT, thereby enhancing resuscitation efforts on-site. These findings support the incor
Furthermore, Sanjan et al[45] in their prospective study found that Coalescent B-lines were observed in approximately 70.4% of patients with moderate ARDS and 92.3% of those with severe ARDS, while lung consolidations were identified in nearly all cases, 100% of moderate and 92.3% of severe ARDS, highlighting their prevalence as key sonographic features in advanced disease stages.
Thoracic ultrasound and heart failure: Glöckner et al[48] utilized an eight-zone LUS approach to evaluate acute heart failure in dyspnoeic patients, reporting high specificity (97.6%) but relatively low sensitivity (54.2%). These findings suggest that while LUS is reliable for confirming heart failure, it may be less effective as a screening tool. Moreover, their analysis of serial LUS over a 72-hour period revealed no significant reduction in B-lines, raising concerns about its sensitivity in monitoring short-term therapeutic response, an important aspect absents from current meta-analyses but vital for ongoing patient management. In contrast, Wooten et al[43] demonstrated the superior diagnostic utility of LUS over chest radiography in identifying pulmonary oedema. Among 99 patients, LUS showed a sensitivity of 96% and specificity of 90%, significantly outperforming CXR, which yielded 65% sensitivity and 96% specificity. Notably, 89% of patients with normal CXR but confirmed pulmonary oedema at discharge had positive LUS findings, underscoring the modality's value in detecting cases overlooked by conventional imaging. Similarly, Carlino et al[41] confirmed LUS as a highly effective diagnostic tool for acute heart failure, achieving 100% sensitivity, 82% specificity, and 89% overall accuracy, figures that rose to 96% accuracy when LUS was combined with either left atrial dilation, reduced ejection fraction (≤ 40%), or both, demonstrating its enhanced diagnostic potential when integrated with cardiac parameters.
POCUS in resource-limited settings: The adaptability of POCUS across different healthcare settings is further illustrated in the work by House et al[52], who studied its impact in a resource-limited ED in Nepal. Their findings showed that LUS changed diagnoses in 44.3% of patients and influenced management decisions in 53.6% of cases. This effect was most pronounced in therapeutic choices (83.3%) but also extended to decisions regarding patient disposition and specialist referrals. While specific sensitivity and specificity metrics were not reported, the study emphasizes the real-world clinical utility of POCUS, particularly in environments where access to advanced imaging is constrained.
POCUS combined with echocardiography and venous ultrasound: Sforza et al[42] explored the diagnostic performance of a multi-organ ultrasound protocol using a pocket-sized device. In diagnosing cardiogenic dyspnoea, LUS alone achieved a sensitivity of 92.6% and specificity of 80.5%. When combined with cardiac or IVC ultrasound, diagnostic accuracy rose to 90%.
Additionally, Liu et al[50] highlighted that LUS serves as a valuable tool for rapidly distinguishing between isolated pulmonary infection and pulmonary infection complicated by acute heart failure. The study suggests that combining LUS with echocardiography may enhance diagnostic accuracy, making this integrated approach particularly useful in emergency and critical care settings.
Enhanced ultrasound accuracy post training: Msolli et al[14] demonstrated that emergency medicine residents can effectively enhance their diagnostic accuracy for congestive heart failure following a brief, focused training in ultrasonographic B-lines assessment. All participants underwent a 2-hour instructional session complemented by a minimum of 10 supervised clinical examinations under the guidance of certified emergency physicians who had completed a comprehensive “Ultra-Sound Life Support” mentorship program. The study reported excellent inter-rater reliability in eva
Moreover, a multicenter, prospective, non-inferiority cohort study conducted by Beyer et al[7] demonstrated that POCUS, when performed by the primary treating team, significantly altered the principal diagnosis in patients presenting with undifferentiated dyspnoea in the ED. In addition to improving diagnostic precision by narrowing differential diagnoses, the study found that the diagnostic utility of POCUS conducted by frontline clinicians was non-inferior to that performed by a dedicated ultrasound team. These findings underscore the value of POCUS as a tool that not only facilitates timely and accurate clinical decision-making but also alleviates the cognitive demands associated with the initial evaluation of acutely dyspnoeic patients.
Notably, the level of ultrasound training among operators varied widely, ranging from fellowship-trained attending physicians to junior residents with less than one month of dedicated ultrasound training. Nevertheless, the observed effectiveness of POCUS in this context, even when performed by clinicians with minimal training, highlights its potential as a scalable and accessible diagnostic aid across varying levels of clinical expertise.
POCUS and early triage: In a randomized study conducted by Zare et al[2] involving 103 patients with undifferentiated acute dyspnoea, participants were assigned to either an “early ultrasound” group, where emergency physicians conducted bedside heart and LUS promptly after triage, or a “routine assessment” group, in which ultrasound was used only when ordered during standard clinical care. The early ultrasound group had a significantly shorter mean time to diagnosis (42.61 ± 19.20 minutes) compared to the routine assessment group (79.33 ± 38.90 minutes), with a P value < 0.01.
This section provides an evaluation of the study’s key strengths and limitations, highlighting its methodological robustness while acknowledging factors that may affect the interpretation and generalisability of the findings.
Strengths of the study: This systematic review and meta-analysis represent one of the most expansive and methodologically rigorous evaluations of POCUS in the context of emergency dyspnoea to date. A key strength is the prospective registration of the review protocol on the PROSPERO database, which ensures adherence to predefined inclusion criteria and enhances transparency. Moreover, the study followed the PRISMA 2020 reporting guidelines, promoting reproducibility and comprehensive reporting.
The scope of included studies is another significant strength. By encompassing a variety of study designs, from pro
Importantly, this review restricted inclusion to studies conducted specifically in EDs, thereby enhancing clinical relevance. It also excluded paediatric populations, thereby controlling for age-related variability in pathology and ultra
Limitations of the study: Despite these strengths, several limitations must be acknowledged. First, while all studies included patients presenting with dyspnoea, some also encompassed other symptoms such as chest pain or cough. This overlap introduces a degree of clinical heterogeneity that could impact the diagnostic accuracy metrics reported. Second, high statistical heterogeneity was observed in virtually all diagnostic indices. This variability likely reflects differences in POCUS implementation, including the ultrasound protocols employed, the anatomical regions scanned, and the skill level of the operators. For example, POCUS protocols themselves contributed to variation: Studies employing structured, comprehensive approaches such as the BLUE protocol achieved higher diagnostic accuracy. Patel et al[13] applied the BLUE protocol in patients with acute respiratory distress and reported 90.3% accuracy. In contrast, Glöckner et al[48] used an eight-zone limited scan for acute heart failure and found sensitivity of only 54.2%, highlighting the limitations of restricted protocols. Diagnostic reproducibility also varied by disease type. Interstitial pathologies (e.g., pulmonary oedema) yielded more consistent results, Wooten et al[43] reported 96% sensitivity for POCUS compared to only 65% for CXR. By contrast, pleural effusion and pneumothorax showed greater variability depending on operator interpretation as revealed by the study conducted by Farahmand et al[53]. In ARDS, Sanjan et al[45] demonstrated that features such as coalescent B-lines and consolidations correlated with severity but required careful operator interpretation, further contributing to variability. Operator training was also a key determinant of accuracy. Pivetta et al[28], using trained sonographers across multiple centers, reported sensitivity and specificity of about 97% for acute dyspnoea. By contrast, Baker et al[27] found that briefly trained non-experts achieved lower accuracy in diagnosing pulmonary oedema. Similarly, Msolli et al[14] showed that emergency residents with only a 2-hour training and 10 supervised scans could apply POCUS effectively, but with more variability than expert users. These observations emphasize that, much like the challenges faced in optimizing dispatcher-assisted telemedicine protocols for cardiac arrest management[56-58], stan
Operator training and experience are especially pertinent issues. Several studies failed to clearly report training levels, and where data were available, they revealed significant variability, from newly trained medical residents to expert sonographers. The absence of standardised training and credentialing protocols undermines consistency and comparability across studies.
Another limitation relates to the heterogeneity in reference standards used to confirm diagnoses. While some studies employed gold-standard imaging such as CT, others relied on clinical judgment, discharge diagnoses, or composite diagnostic endpoints. This lack of uniformity can introduce verification bias and reduce the internal validity of pooled estimates.
Methodological inconsistencies in reporting POCUS results also presented challenges. Some studies used graphical rather than tabulated data, complicating the extraction of key metrics like TP and FN. This may have introduced minor interpretation errors despite rigorous data extraction protocols. Furthermore, a lack of blinding in several studies raised the possibility of performance and detection bias.
This systematic review and meta-analysis demonstrate that POCUS has a significant role in the evaluation of acute dyspnoea in ED settings, with high diagnostic accuracy that supports its use as a frontline tool for conditions such as acute respiratory failure, pulmonary oedema, and pneumothorax. Its advantages, portability, cost-effectiveness, bedside availability, and safety, make it especially valuable in both high- and low-resource environments, enabling timely diagnosis, efficient triage, early intervention, and even safe discharge when negative findings effectively rule out serious pathology. By reducing dependence on radiology, POCUS can decrease ED length of stay, improve patient flow, and enhance overall resource utilisation, though its consistent application depends on structured clinician training, accreditation, and governance. Despite these strengths, heterogeneity across studies highlights variability in operator profi
We extend our appreciation to the Faculty of Life Sciences and Education at the University of South Wales in association with Learna Ltd. for the Acute Medicine MSc program and their invaluable support in our work. We sincerely acknow
| 1. | Sharma A, Kaeley N, Aravindan N, Datta SS, Vasisht S. Using Lung Ultrasound Combined With N-terminal Pro-brain Natriuretic Peptide to Differentiate Acute Heart Failure From Chronic Obstructive Pulmonary Disease and Asthma in Emergency Department Patients Experiencing Acute Shortness of Breath. Cureus. 2025;17:e77171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 2. | Zare MA, Bahmani A, Fathi M, Arefi M, Hossein Sarbazi A, Teimoori M. Role of point-of-care ultrasound study in early disposition of patients with undifferentiated acute dyspnea in emergency department: a multi-center prospective study. J Ultrasound. 2022;25:443-449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 3. | Baid H, Vempalli N, Kumar S, Arora P, Walia R, Chauhan U, Shukla K, Verma A, Chawang H, Agarwal D. Point of care ultrasound as initial diagnostic tool in acute dyspnea patients in the emergency department of a tertiary care center: diagnostic accuracy study. Int J Emerg Med. 2022;15:27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 26] [Reference Citation Analysis (1)] |
| 4. | Papanagnou D, Secko M, Gullett J, Stone M, Zehtabchi S. Clinician-Performed Bedside Ultrasound in Improving Diagnostic Accuracy in Patients Presenting to the ED with Acute Dyspnea. West J Emerg Med. 2017;18:382-389. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 5. | Umuhire OF, Henry MB, Levine AC, Cattermole GN, Henwood P. Impact of ultrasound on management for dyspnea presentations in a Rwandan emergency department. Ultrasound J. 2019;11:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 6. | Pirozzi C, Numis FG, Pagano A, Melillo P, Copetti R, Schiraldi F. Immediate versus delayed integrated point-of-care-ultrasonography to manage acute dyspnea in the emergency department. Crit Ultrasound J. 2014;6:5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 40] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 7. | Beyer A, Lam V, Fagel B, Dong S, Hebert C, Wallace C, Theyyunni N, Tucker R, Cover M, Kessler R, Cranford JA, Huang R, Majkrzak AA, Seleno NR, Fung CM. Undifferentiated Dyspnea with Point-of-Care Ultrasound, Primary Emergency Physician Compared with a Dedicated Emergency Department Ultrasound Team. J Emerg Med. 2021;61:278-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 8. | Chelikam N, Vyas A, Desai R, Khan N, Raol K, Kavarthapu A, Kamani P, Ibrahim G, Madireddy S, Pothuru S, Shah P, Patel UK. Past and Present of Point-of-Care Ultrasound (PoCUS): A Narrative Review. Cureus. 2023;15:e50155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (1)] |
| 9. | Ünlüer EE, Karagöz A, Oyar O, Vandenberk N, Kiyançiçek S, Budak F. Lung ultrasound by emergency nursing as an aid for rapid triage of dyspneic patients: a pilot study. Int Emerg Nurs. 2014;22:226-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 17] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 10. | Leidi A, Saudan A, Soret G, Rouyer F, Marti C, Stirnemann J, Reny JL, Grosgurin O. Confidence and use of physical examination and point-of-care ultrasonography for detection of abdominal or pleural free fluid. A cross-sectional survey. Intern Emerg Med. 2022;17:113-122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 11. | Biart S, Stanley F, Rahman L, Jones H, Smallwood N. Point of care ultrasound: Current and future directions for Acute Medicine. Acute Med. 2023;22:83-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 12. | Clare S, Duncan C. Ultrasound for the Generalist: A Guide to Point of Care Imaging. Cambridge University Press, 2021. [DOI] [Full Text] |
| 13. | Patel CJ, Bhatt HB, Parikh SN, Jhaveri BN, Puranik JH. Bedside Lung Ultrasound in Emergency Protocol as a Diagnostic Tool in Patients of Acute Respiratory Distress Presenting to Emergency Department. J Emerg Trauma Shock. 2018;11:125-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 14. | Msolli MA, Sekma A, Marzouk MB, Chaabane W, Bel Haj Ali K, Boukadida L, Bzeouich N, Gannoun I, Trabelssi I, Laaouiti K, Grissa MH, Beltaief K, Dridi Z, Belguith A, Methamem M, Bouida W, Boukef R, Boubaker H, Nouira S; GREAT Network. Bedside lung ultrasonography by emergency department residents as an aid for identifying heart failure in patients with acute dyspnea after a 2-h training course. Ultrasound J. 2021;13:5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 15. | Abid I, Qureshi N, Lategan N, Williams S, Shahid S. Point-of-care lung ultrasound in detecting pneumonia: A systematic review. Can J Respir Ther. 2024;60:37-48. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 16. | Buhumaid RE, St-Cyr Bourque J, Shokoohi H, Ma IWY, Longacre M, Liteplo AS. Integrating point-of-care ultrasound in the ED evaluation of patients presenting with chest pain and shortness of breath. Am J Emerg Med. 2019;37:298-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 17. | Booth A, Mitchell AS, Mott A, James S, Cockayne S, Gascoyne S, McDaid C. An assessment of the extent to which the contents of PROSPERO records meet the systematic review protocol reporting items in PRISMA-P. F1000Res. 2020;9:773. [PubMed] [DOI] [Full Text] |
| 18. | Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg. 2010;8:336-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10317] [Cited by in RCA: 8242] [Article Influence: 515.1] [Reference Citation Analysis (5)] |
| 19. | Shaheen N, Shaheen A, Ramadan A, Hefnawy MT, Ramadan A, Ibrahim IA, Hassanein ME, Ashour ME, Flouty O. Appraising systematic reviews: a comprehensive guide to ensuring validity and reliability. Front Res Metr Anal. 2023;8:1268045. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 68] [Reference Citation Analysis (1)] |
| 20. | DeMars MM, Perruso C. MeSH and text-word search strategies: precision, recall, and their implications for library instruction. J Med Libr Assoc. 2022;110:23-33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 42] [Article Influence: 10.5] [Reference Citation Analysis (1)] |
| 21. | Stoll CRT, Izadi S, Fowler S, Green P, Suls J, Colditz GA. The value of a second reviewer for study selection in systematic reviews. Res Synth Methods. 2019;10:539-545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 161] [Article Influence: 23.0] [Reference Citation Analysis (1)] |
| 22. | West SL, Gartlehner G, Mansfield AJ, Poole C, Tant E, Lenfestey N, Lux LJ, Amoozegar J, Morton SC, Carey TC, Viswanathan M, Lohr KN. Table 7, Summary of common statistical approaches to test for heterogeneity. In: Lohr KN, editor. Methods Guide for Effectiveness and Comparative Effectiveness Reviews. Rockville (MD): Agency for Healthcare Research and Quality, 2010. |
| 23. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9803] [Reference Citation Analysis (0)] |
| 24. | PRISMA. PRISMA 2020 flow diagram. [cited 26 November 2025]. Available from: https://www.prisma-statement.org/prisma-2020-flow-diagram. |
| 25. | Özkan B, Ünlüer EE, Akyol PY, Karagöz A, Bayata MS, Akoğlu H, Oyar O, Dalli A, Topal FE. Stethoscope versus point-of-care ultrasound in the differential diagnosis of dyspnea: a randomized trial. Eur J Emerg Med. 2015;22:440-443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 26. | Pontis E, Claret PG, Markarian T, Javaudin F, Flacher A, Roger C, Muller L, de La Coussaye JE, Bobbia X. Integration of lung ultrasound in the diagnostic reasoning in acute dyspneic patients: A prospective randomized study. Am J Emerg Med. 2018;36:1597-1602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 27. | Baker K, Brierley S, Kinnear F, Isoardi K, Livesay G, Stieler G, Mitchell G. Implementation study reporting diagnostic accuracy, outcomes and costs in a multicentre randomised controlled trial of non-expert lung ultrasound to detect pulmonary oedema. Emerg Med Australas. 2020;32:45-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 28. | Pivetta E, Goffi A, Lupia E, Tizzani M, Porrino G, Ferreri E, Volpicelli G, Balzaretti P, Banderali A, Iacobucci A, Locatelli S, Casoli G, Stone MB, Maule MM, Baldi I, Merletti F, Cibinel GA, Baron P, Battista S, Buonafede G, Busso V, Conterno A, Del Rizzo P, Ferrera P, Pecetto PF, Moiraghi C, Morello F, Steri F, Ciccone G, Calasso C, Caserta MA, Civita M, Condo' C, D'Alessandro V, Del Colle S, Ferrero S, Griot G, Laurita E, Lazzero A, Lo Curto F, Michelazzo M, Nicosia V, Palmari N, Ricchiardi A, Rolfo A, Rostagno R, Bar F, Boero E, Frascisco M, Micossi I, Mussa A, Stefanone V, Agricola R, Cordero G, Corradi F, Runzo C, Soragna A, Sciullo D, Vercillo D, Allione A, Artana N, Corsini F, Dutto L, Lauria G, Morgillo T, Tartaglino B, Bergandi D, Cassetta I, Masera C, Garrone M, Ghiselli G, Ausiello L, Barutta L, Bernardi E, Bono A, Forno D, Lamorte A, Lison D, Lorenzati B, Maggio E, Masi I, Maggiorotto M, Novelli G, Panero F, Perotto M, Ravazzoli M, Saglio E, Soardo F, Tizzani A, Tizzani P, Tullio M, Ulla M, Romagnoli E; SIMEU Group for Lung Ultrasound in the Emergency Department in Piedmont. Lung Ultrasound-Implemented Diagnosis of Acute Decompensated Heart Failure in the ED: A SIMEU Multicenter Study. Chest. 2015;148:202-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 344] [Cited by in RCA: 286] [Article Influence: 26.0] [Reference Citation Analysis (1)] |
| 29. | Nakao S, Vaillancourt C, Taljaard M, Nemnom MJ, Woo MY, Stiell IG. Diagnostic Accuracy of Lung Point-Of-Care Ultrasonography for Acute Heart Failure Compared With Chest X-Ray Study Among Dyspneic Older Patients in the Emergency Department. J Emerg Med. 2021;61:161-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 30. | Mantuani D, Frazee BW, Fahimi J, Nagdev A. Point-of-Care Multi-Organ Ultrasound Improves Diagnostic Accuracy in Adults Presenting to the Emergency Department with Acute Dyspnea. West J Emerg Med. 2016;17:46-53. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 76] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 31. | Núñez-Ramos JA, Duarte-Misol D, Petro MAB, Pérez KJS, Echeverry VPG, Malagón SV. Agreement of point of care ultrasound and final clinical diagnosis in patients with acute heart failure, acute coronary syndrome, and shock: POCUS not missing the target. Intern Emerg Med. 2024;19:1585-1592. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 32. | Martindale JL, Noble VE, Liteplo A. Diagnosing pulmonary edema: lung ultrasound versus chest radiography. Eur J Emerg Med. 2013;20:356-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 52] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 33. | Bitar Z, Maadarani O, Almerri K. Sonographic chest B-lines anticipate elevated B-type natriuretic peptide level, irrespective of ejection fraction. Ann Intensive Care. 2015;5:56. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 34. | Sartini S, Frizzi J, Borselli M, Sarcoli E, Granai C, Gialli V, Cevenini G, Guazzi G, Bruni F, Gonnelli S, Pastorelli M. Which method is best for an early accurate diagnosis of acute heart failure? Comparison between lung ultrasound, chest X-ray and NT pro-BNP performance: a prospective study. Intern Emerg Med. 2017;12:861-869. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 35. | Yahia M, Soliman M, Fawzy M, Sultan H. Diagnostic accuracy of lung ultrasound in acute heart failure. Res Opin Anesth Intens Care. 2022;9:87-93. [DOI] [Full Text] |
| 36. | Secko M, Cheng Y, Raj S, Goradia E, Reardon L, Thode HC, Singer AJ. Comprehensive Physical Exam versus Lung Ultrasound for Dyspneic Patients in the Emergency Department. 2022 preprint. Available from: medRxiv: 2022.10.08.22280828. [DOI] [Full Text] |
| 37. | Arvig MD, Lassen AT, Gæde PH, Gärtner SW, Falster C, Skov IR, Petersen HØ, Posth S, Laursen CB. Impact of serial cardiopulmonary point-of-care ultrasound exams in patients with acute dyspnoea: a randomised, controlled trial. Emerg Med J. 2023;40:700-707. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 38. | Sorlini C, Femia M, Nattino G, Bellone P, Gesu E, Francione P, Paternò M, Grillo P, Ruffino A, Bertolini G, Cariati M, Cortellaro F; Fenice Network (Italian Group for Clinical Research in Emergency Medicine). The role of lung ultrasound as a frontline diagnostic tool in the era of COVID-19 outbreak. Intern Emerg Med. 2021;16:749-756. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (2)] |
| 39. | Gibbons RC, Magee M, Goett H, Murrett J, Genninger J, Mendez K, Tripod M, Tyner N, Costantino TG. Lung Ultrasound vs. Chest X-Ray Study for the Radiographic Diagnosis of COVID-19 Pneumonia in a High-Prevalence Population. J Emerg Med. 2021;60:615-625. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 40. | Bianchi S, Savinelli C, Paolucci E, Pelagatti L, Sibona E, Fersini N, Buggea M, Tozzi C, Allescia G, Paolini D, Lanigra M. Point-of-care ultrasound (PoCUS) in the early diagnosis of novel coronavirus 2019 disease (COVID-19) in a first-level emergency department during a SARS-CoV-2 outbreak in Italy: a real-life analysis. Intern Emerg Med. 2022;17:193-204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 41. | Carlino MV, Paladino F, Sforza A, Serra C, Liccardi F, de Simone G, Mancusi C. Assessment of left atrial size in addition to focused cardiopulmonary ultrasound improves diagnostic accuracy of acute heart failure in the Emergency Department. Echocardiography. 2018;35:785-791. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 42. | Sforza A, Mancusi C, Carlino MV, Buonauro A, Barozzi M, Romano G, Serra S, de Simone G. Diagnostic performance of multi-organ ultrasound with pocket-sized device in the management of acute dyspnea. Cardiovasc Ultrasound. 2017;15:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 43] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 43. | Wooten WM, Shaffer LET, Hamilton LA. Bedside Ultrasound Versus Chest Radiography for Detection of Pulmonary Edema: A Prospective Cohort Study. J Ultrasound Med. 2019;38:967-973. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 44. | Markarian T, Zieleskiewicz L, Perrin G, Claret PG, Loundou A, Michelet P, Bobbia X. A lung ultrasound score for early triage of elderly patients with acute dyspnea. CJEM. 2019;21:399-405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 45. | Sanjan A, Krishnan SV, Abraham SV, Palatty BU. Utility of Point-of-Care Lung Ultrasound for Initial Assessment of Acute Respiratory Distress Syndrome Patients in the Emergency Department. J Emerg Trauma Shock. 2019;12:248-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 46. | Vauthier C, Chabannon M, Markarian T, Taillandy Y, Guillemet K, Krebs H, Bazalgette F, Muller L, Claret PG, Bobbia X. Point-of-care chest ultrasound to diagnose acute heart failure in emergency department patients with acute dyspnea: diagnostic performance of an ultrasound-based algorithm. Emergencias. 2021;33:441-446. |
| 47. | Altuğ E, Toksul İH, Çakir A, Şener K, Korkut S, Kapci M, Güven R. Evaluation of the Prognosis of Patients With Acute Respiratory Distress Syndrome at the Emergency Department Based on the Lung Ultrasound Score. J Ultrasound Med. 2024;43:1235-1243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 48. | Glöckner E, Wening F, Christ M, Dechêne A, Singler K. Lung Ultrasound Eight-Point Method in Diagnosing Acute Heart Failure in Emergency Patients with Acute Dyspnea: Diagnostic Accuracy and 72 h Monitoring. Medicina (Kaunas). 2020;56:379. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 49. | Cibinel GA, Casoli G, Elia F, Padoan M, Pivetta E, Lupia E, Goffi A. Diagnostic accuracy and reproducibility of pleural and lung ultrasound in discriminating cardiogenic causes of acute dyspnea in the emergency department. Intern Emerg Med. 2012;7:65-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 93] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 50. | Liu ZP, Zhang Y, Bian H, He XR, Zhou YJ, Wang LJ, Ding N. Clinical application of rapid B-line score with lung ultrasonography in differentiating between pulmonary infection and pulmonary infection with acute left ventricular heart failure. Am J Emerg Med. 2016;34:278-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 51. | Ahn JH, Jeon J, Toh HC, Noble VE, Kim JS, Kim YS, Do HH, Ha YR. SEARCH 8Es: A novel point of care ultrasound protocol for patients with chest pain, dyspnea or symptomatic hypotension in the emergency department. PLoS One. 2017;12:e0174581. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 52. | House DR, Amatya Y, Nti B, Russell FM. Impact of bedside lung ultrasound on physician clinical decision-making in an emergency department in Nepal. Int J Emerg Med. 2020;13:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 53. | Farahmand S, Abdolhoseini A, Aliniagerdroudbari E, Babaniamansour S, Baratloo A, Bagheri-Hariri S. Point-of-care ultrasound modalities in terms of diagnosing acute decompensated heart failure in emergency department; a diagnostic accuracy study. Intern Emerg Med. 2020;15:491-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 54. | Markarian T, Persico N, Roch A, Ahriz D, Taguet C, Birman G, Mahboubi A, Ducassou J, Bourenne J, Zieleskiewicz L, Bobbia X, Michelet P. Early assessment of patients with COVID-19 and dyspnea using lung ultrasound scoring. Emergencias. 2021;33:354-360. |
| 55. | Öhman J, Harjola VP, Karjalainen P, Lassus J. Rapid cardiothoracic ultrasound protocol for diagnosis of acute heart failure in the emergency department. Eur J Emerg Med. 2019;26:112-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 32] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 56. | Whiting PF, Rutjes AW, Westwood ME, Mallett S, Deeks JJ, Reitsma JB, Leeflang MM, Sterne JA, Bossuyt PM; QUADAS-2 Group. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med. 2011;155:529-536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11012] [Cited by in RCA: 10534] [Article Influence: 702.3] [Reference Citation Analysis (5)] |
| 57. | Focused Acute Medicine Ultrasound. [cited 26 November 2025]. Available from: https://www.acutemedicine.org.uk/famus/. |
| 58. | Hussain S, Soldera J. Telemedicine in cardiac arrest protocols: Comparative impact of video and audio dispatcher assistance. World J Crit Care Med. 2025;14:103402. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (2)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/