©Author(s) (or their employer(s)) 2026.
World J Crit Care Med. Mar 9, 2026; 15(1): 114318
Published online Mar 9, 2026. doi: 10.5492/wjccm.v15.i1.114318
Published online Mar 9, 2026. doi: 10.5492/wjccm.v15.i1.114318
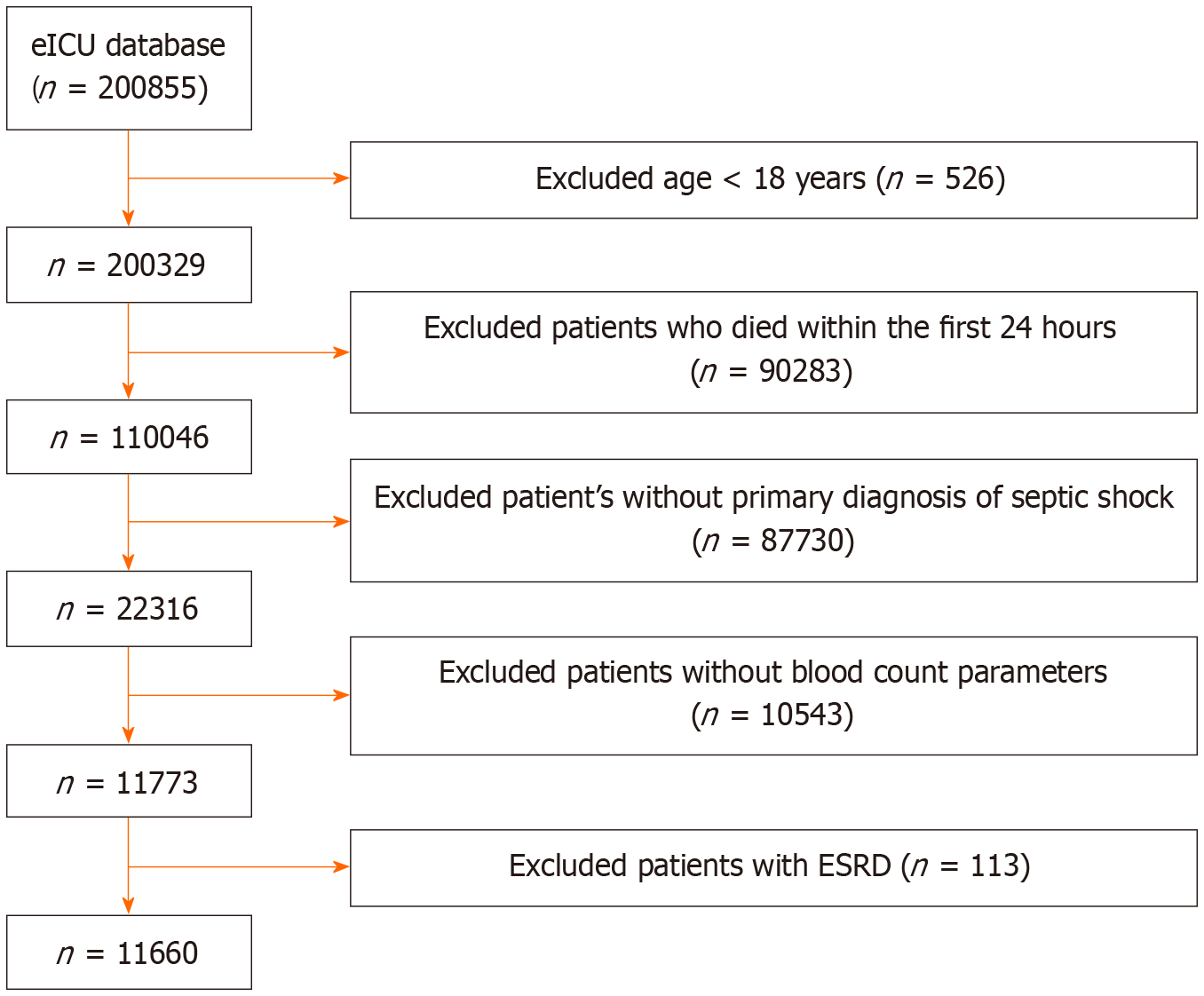
Figure 1 Flowchart of selection of participants.
ESRD: End stage renal disease.
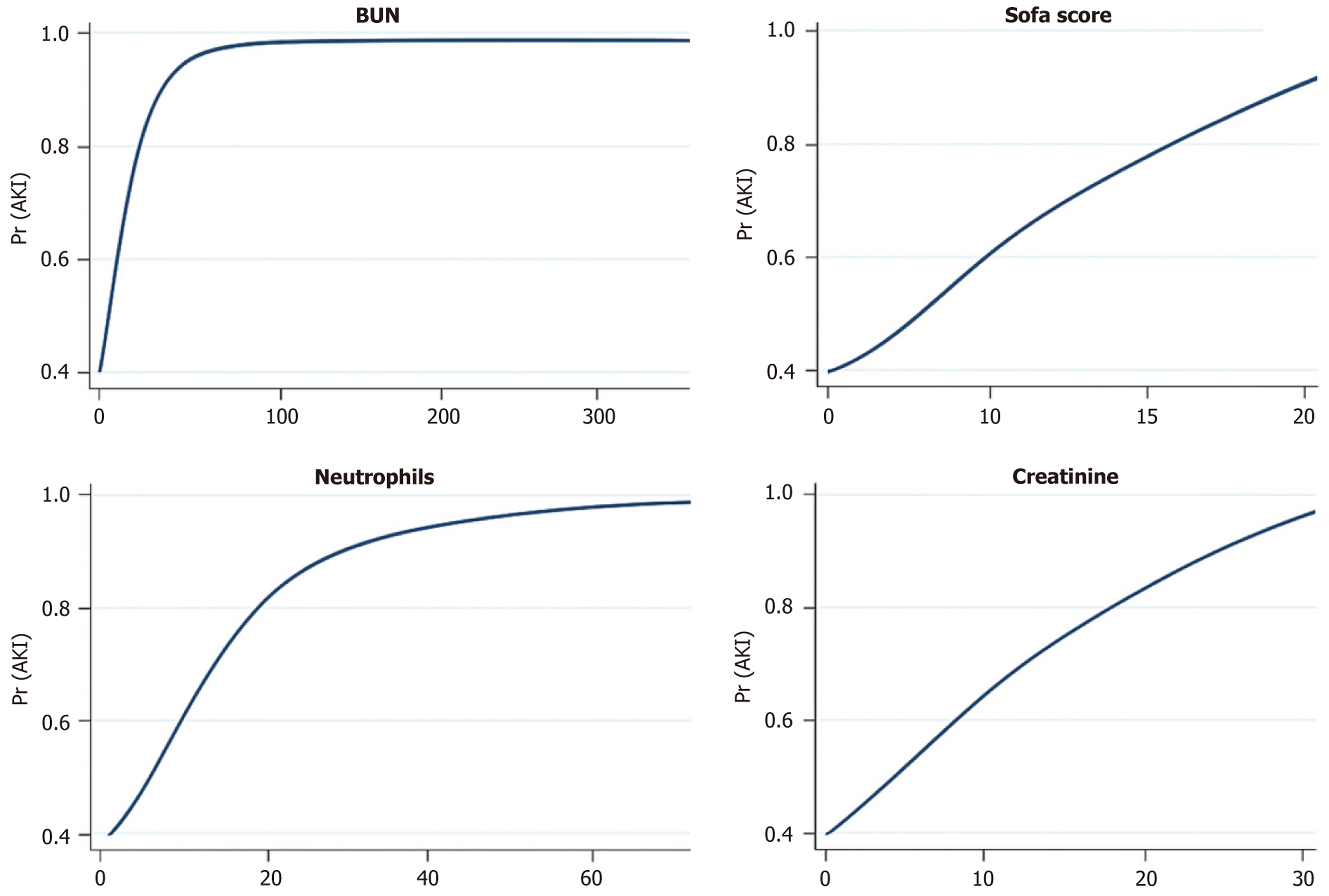
Figure 2 Graph of restricted cubic splines and predictive probabilities of acute kidney injury.
Graphs of restricted cubic splines marginal predicted probabilities for the development of acute kidney injury across four continuous biomarkers: (1) Sequential Organ Failure Assessment score; (2) Serum creatinine; (3) Blood urea nitrogen; and (4) Neutrophils. The fitted curves for the Sequential Organ Failure Assessment score and serum creatinine are near linear. Spline terms were kept in order to improve calibration. AKI: Acute kidney injury; Pr: Predicted probabilities; SOFA: Sequential Organ Failure Assessment.
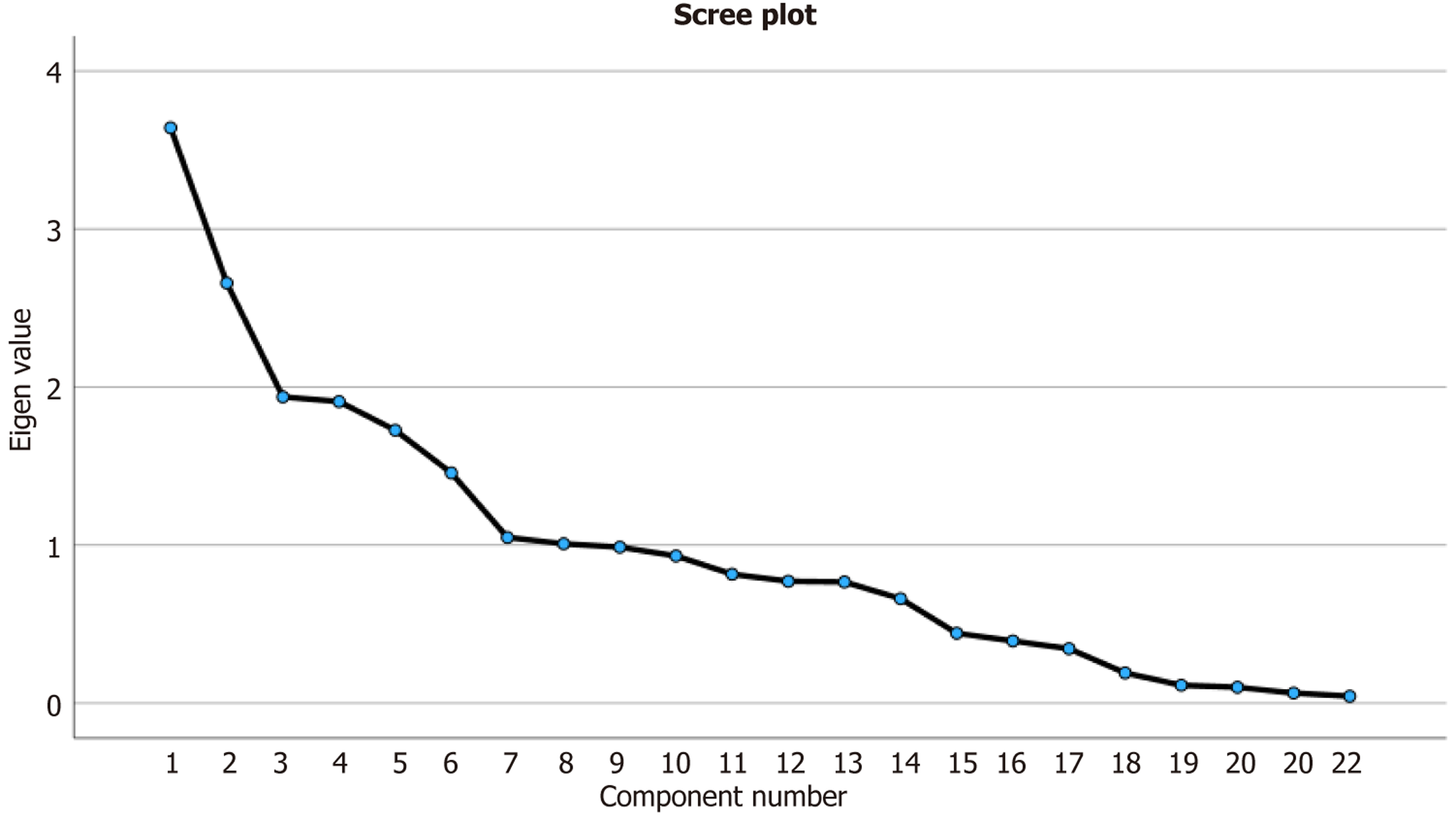
Figure 3 The scree plot.
The number of components for factor analysis was determined by identifying the “elbow” of the plot.
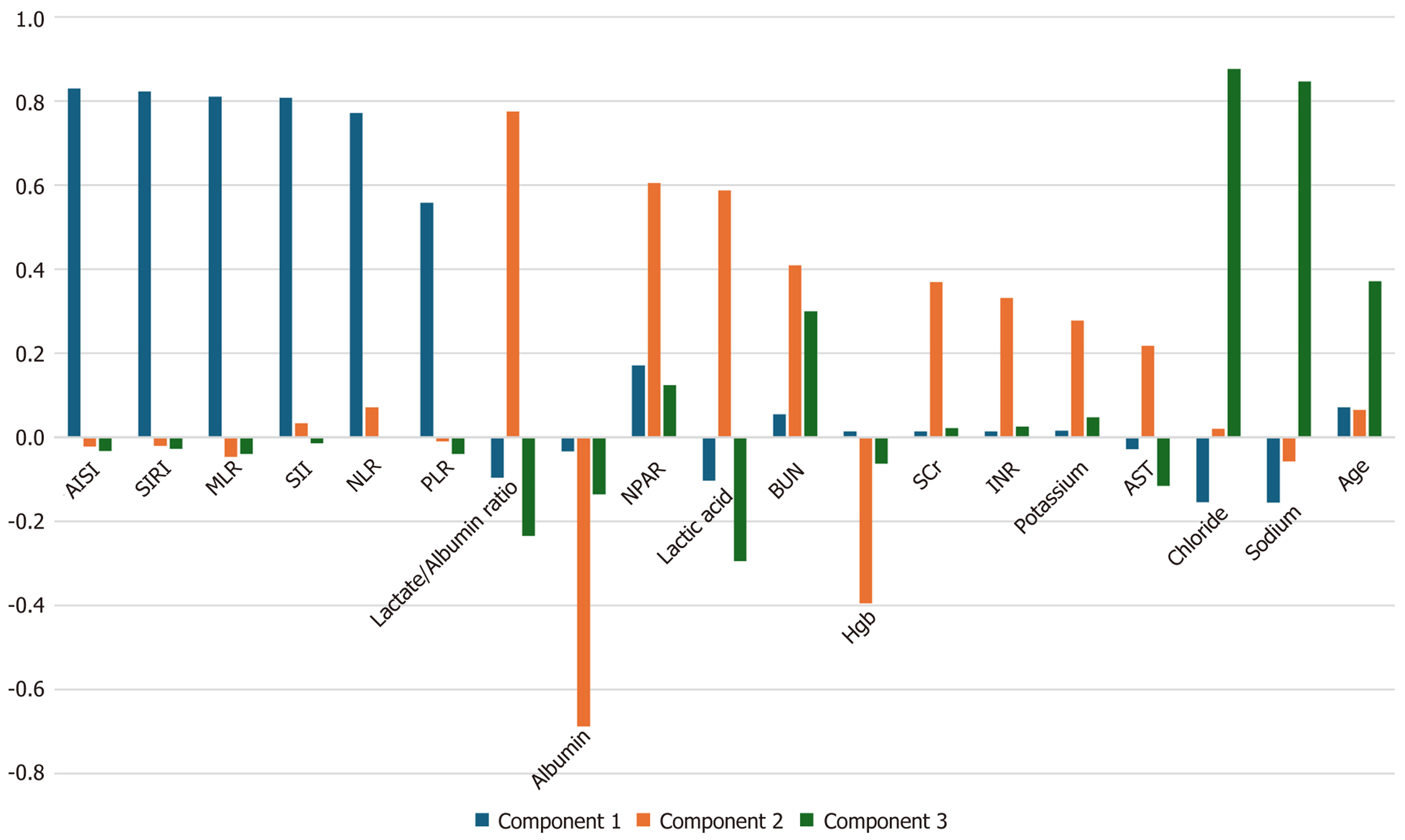
Figure 4 Principle component analysis matrix loading by components.
Graphic representation of pattern matrix of principal component analysis into three components/factors (factor 1: Inflammatory/hematological components; factor 2: Metabolic/renal/inflammatory components; factor 3: Electrolyte/age). AISI: Aggregate index of systemic inflammation; AST: Aspartate aminotransferase; BUN: Blood urea nitrogen; Hgb: Hemoglobin; INR: International normalized ratio; MLR: Monocyte-to-lymphocyte ratio; NLR: Neutrophil-to-lymphocyte ratio; NPAR: Neutrophil-percentage-to-albumin ratio; PLR: Platelet-to-lymphocyte ratio; SCr: Serum creatinine; SII: Systemic immune-inflammation index; SIRI: Systemic inflammation response index.
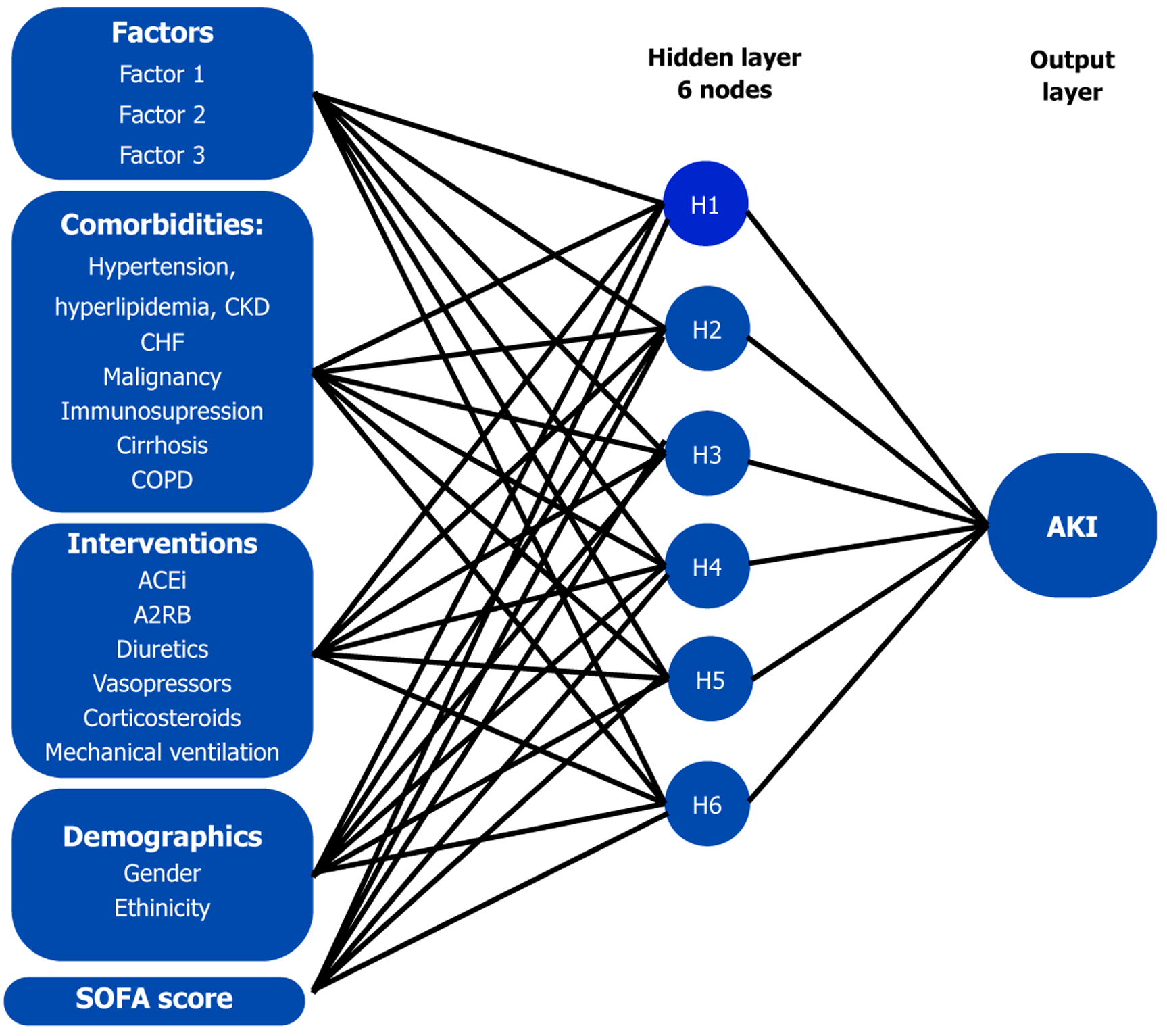
Figure 5 Simplified neural network.
ACEi: Angiotensin converting enzyme inhibitor; AKI: Acute kidney injury; A2RB: Angiotensin 2 receptor antagonist; CHF: Congestive heart failure; CKD: Chronic kidney disease; COPD: Chronic obstructive pulmonary disease; SOFA: Sequential Organ Failure Assessment.
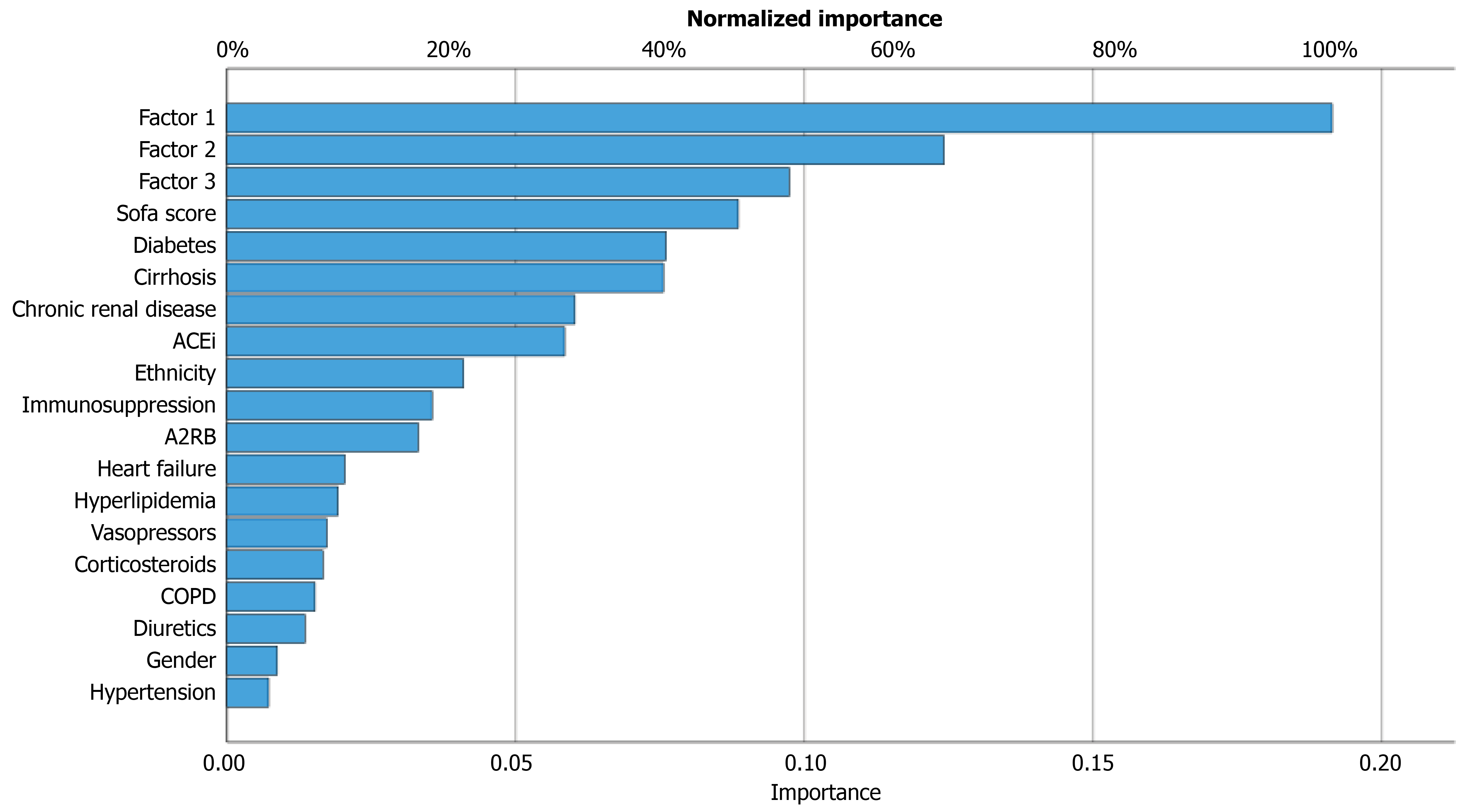
Figure 6 Normalized importance of factors contributing to acute kidney injury.
Multilayer perceptron summary of the level of importance of variables contributing to the development of acute kidney injury. ACEi: Angiotensin converting enzyme inhibitor; A2RB: Angiotensin 2 receptor antagonist; COPD: Chronic obstructive pulmonary disease.
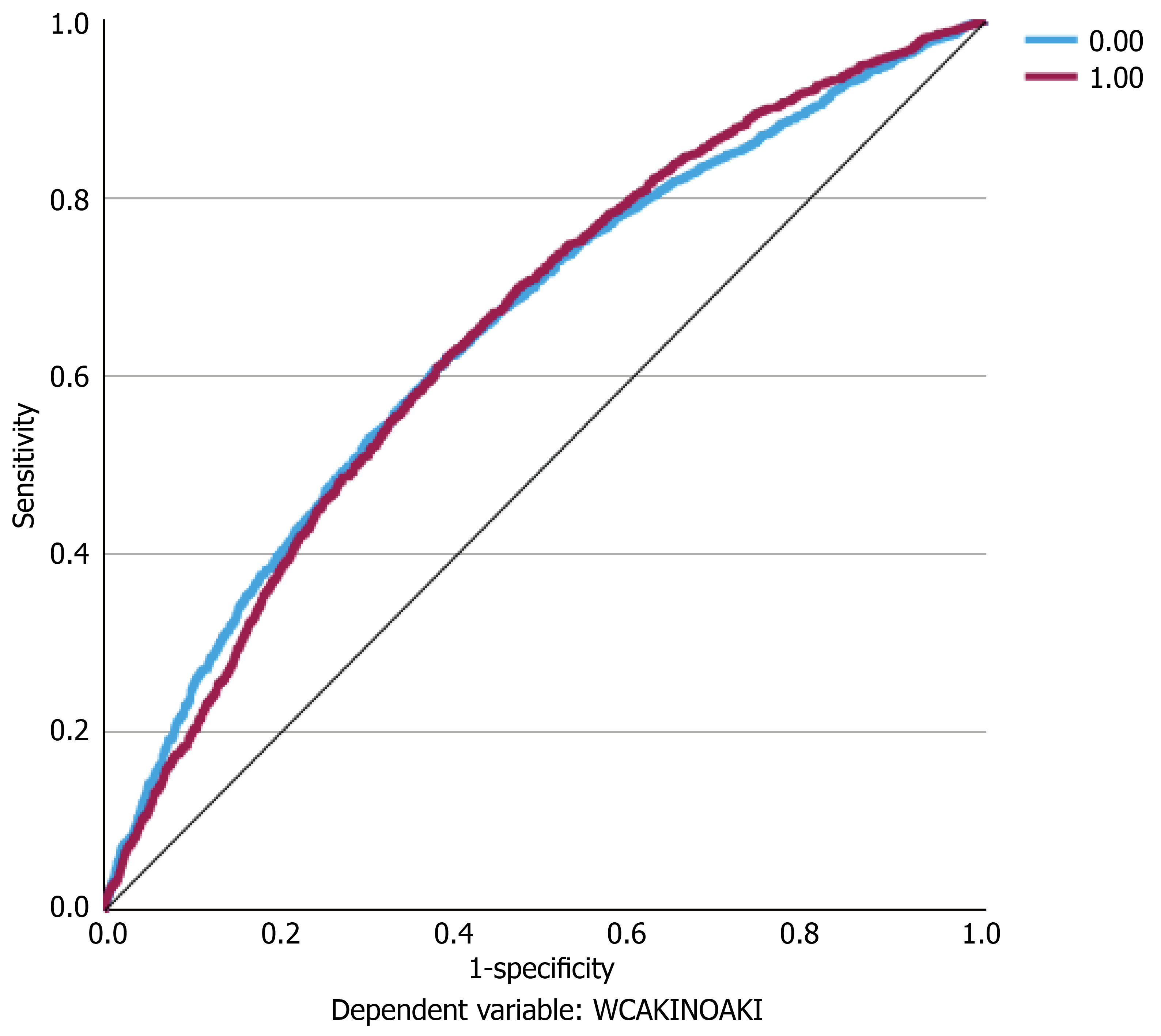
Figure 7 Receiver operating curve for multilayer perceptron.
Receiver operating curve for multilayer perceptron blue line indicates no acute kidney injury, red line indicates acute kidney injury.
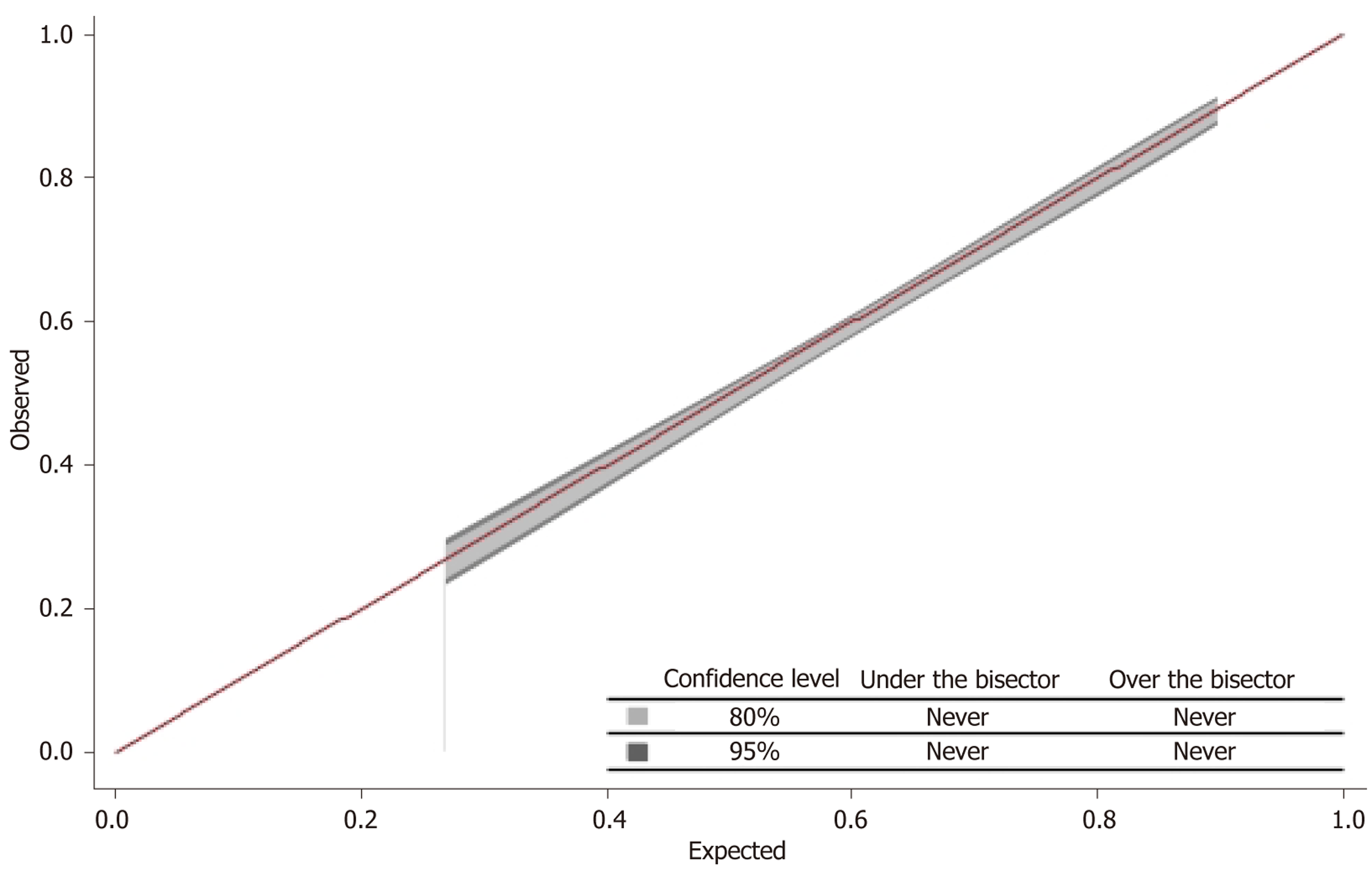
Figure 8 Calibration performance of the multilayer perceptron neural network.
Calibration plot of the multilayer perceptron model showing the expected vs observed probabilities of the outcome. The bisector line aligns closely with the 45-degree reference, indicating good calibration. The 80%CI (light gray) and 95%CI (dark gray) remain within the bisector margins, suggesting stable model performance across risk strata. The Brier score was 0.22, and the Hosmer-Lemeshow goodness-of-fit P-value was 0.67, consistent with adequate calibration.
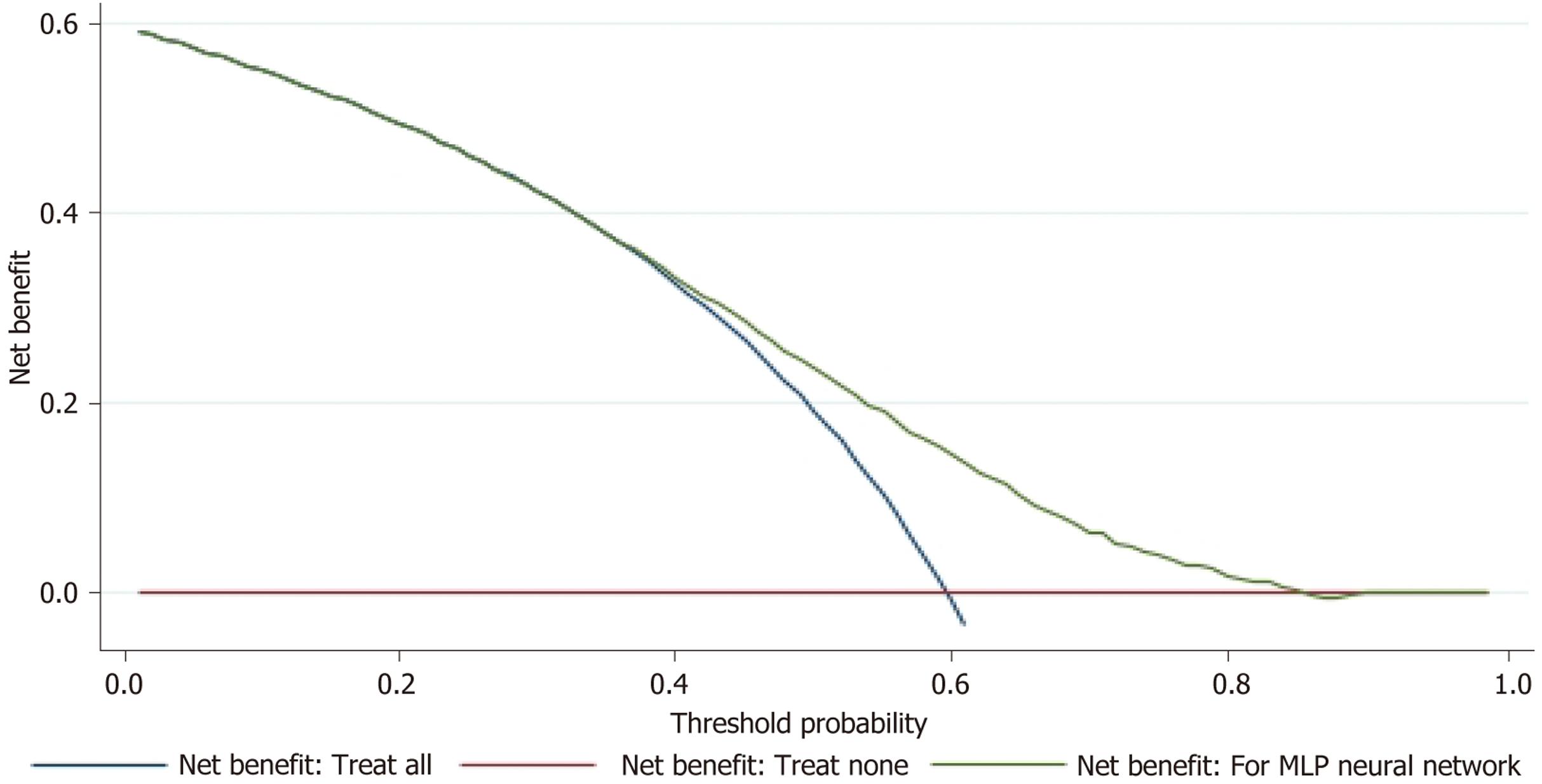
Figure 9 Decision curve analysis of the multilayer perceptron neural network.
Decision curve analysis showing net benefit vs threshold probability. The blue curve (treat-all) decreases from 0.6 at threshold 0 to 0 at 0.6. The red line (treat-none) remains at 0 across thresholds 0-0.8. The green curve (multilayer perceptron) starts near 0.6 and declines toward 0 as the threshold increases to 0.8, while maintaining higher net benefit than both reference strategies across most of the range. This indicates superior clinical utility of the multilayer perceptron model across commonly used decision thresholds.
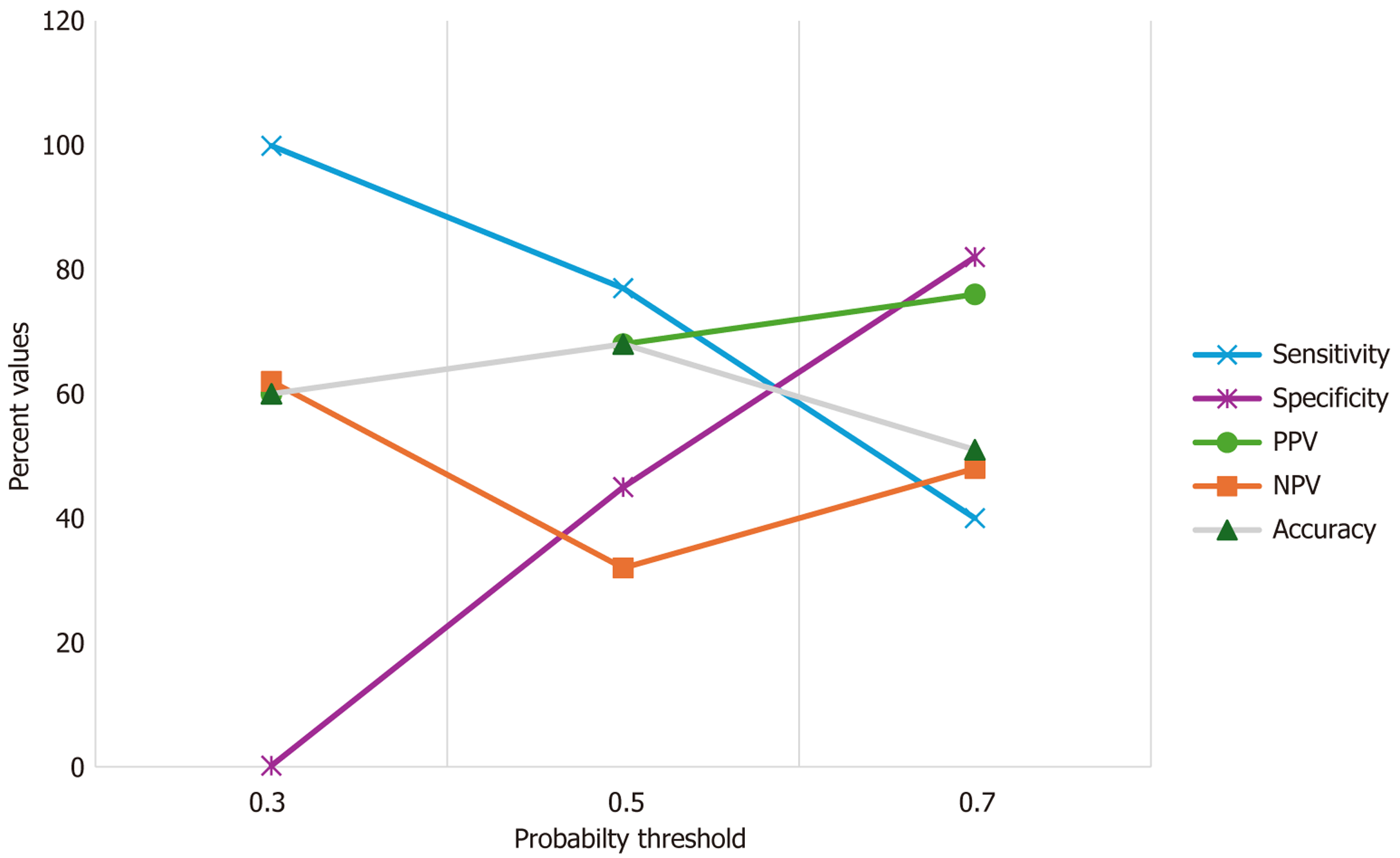
Figure 10 Diagnostic performance across probability thresholds threshold-dependent performance metrics for the multilayer perceptron model.
At a 0.3 threshold, the model achieved high sensitivity (99.9%) but low specificity (0.2). Increasing the threshold to 0.5 balanced sensitivity (77%) and specificity (45%), while a 0.7 threshold improved specificity (82%) at the expense of sensitivity (40%). The optimal threshold (0.5) yielded the best overall accuracy (68%), representing the preferred trade-off for clinical application.
- Citation: Rajendran J, Ang SP, Lorenzo-Capps MJ, Valladares C, Lee E, Bommu VJL, Altarcha G, Pominov S, Gregory B, Chia JE, Iglesias J. Predicting acute kidney injury in septic shock patients using inflammatory indices in the intensive care unit. World J Crit Care Med 2026; 15(1): 114318
- URL: https://www.wjgnet.com/2220-3141/full/v15/i1/114318.htm
- DOI: https://dx.doi.org/10.5492/wjccm.v15.i1.114318