Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.111021
Revised: July 10, 2025
Accepted: November 7, 2025
Published online: March 9, 2026
Processing time: 258 Days and 20.5 Hours
Post-infectious bronchiolitis obliterans (PIBO) is a rare chronic obstructive pul
Core Tip: Post-infectious bronchiolitis obliterans leads to permanent small-airway obstruction and significant spirometric deficits in children, notably reduced forced expiratory volume in one second and forced expiratory flow at 25%-75% with poor bronchodilator response, while diffusing capacity of the lung for carbon monoxide often remains normal. This narrative review links these pulmonary function parameters with therapeutic interventions, showing that early use of inhaled or systemic corticosteroids and macrolides can stabilize or modestly improve lung function. Emerging combination regimens, such as fluticasone-azithromycin-montelukast, show promise but need further validation. Integrating regular spirometric monitoring with tailored anti-inflammatory strategies may optimize clinical outcomes and underlines the need for prospective studies.
- Citation: Chiellino S. Therapeutic interventions and pulmonary function in pediatric patients with post-infectious bronchiolitis obliterans. World J Clin Pediatr 2026; 15(1): 111021
- URL: https://www.wjgnet.com/2219-2808/full/v15/i1/111021.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v15.i1.111021
The term bronchiolitis obliterans (BO) includes a group of diseases with different characteristics. There is no universally accepted definition for this condition. BO refers to a heterogeneous group of diseases characterized by persistent airway obstruction due to small airway involvement, often showing poor response to bronchodilator treatment[1]. Among its forms, post-infectious bronchiolitis obliterans (PIBO) is a rare but severe chronic lung disease that can develop after a lower respiratory tract infection in childhood, commonly caused by adenovirus, respiratory syncytial virus (RSV), or measles, especially in children under two years of age. BO is characterized by persistent airway obstruction with radiological and functional evidence of small airway involvement and poor response to bronchodilator treatment[1]. The pathogenesis involves bronchiolar epithelial injury, followed by an inflammatory reaction and progressive fibrotic remodeling that leads to irreversible luminal obliteration[1,2]. Clinically, PIBO presents with persistent cough, wheezing, dyspnea, and progressive decline in pulmonary function parameters such as forced expiratory volume in one second (FEV1) and forced vital capacity (FVC). Due to the chronic and progressive nature of PIBO, therapies focus on managing inflammation, reducing symptoms, and preserving lung function. Treatment approaches range from corticosteroids, macrolides, bronchodilators and combined therapies. The efficacy of these therapies is often assessed through pulmonary function tests (PFTs), particularly spirometry, which provides objective measures of airway obstruction and lung capacity. Previous studies on the pulmonary function parameters in children affected by PIBO demonstrated a variable range of impairment due to differences in the characteristics of the study population and the degree and age of respi
Although corticosteroids, macrolides, bronchodilators, and combination regimens are commonly used, current the
This review aims to critically evaluate the existing evidence on therapeutic interventions for children with PIBO and their impact on pulmonary function outcomes, highlighting the need for standardized diagnostic criteria, larger multicenter studies, and clear clinical guidelines to improve care for this condition.
PIBO is a rare but severe chronic lung disease in children, with its true incidence likely underestimated due to diagnostic challenges and variability in clinical presentation. It most commonly affects previously healthy children under the age of three. Epidemiological studies suggest that PIBO is more prevalent in developing countries, where access to timely and advanced medical care for severe respiratory infections may be limited. It is also more frequent among Argentinians, Native Americans, and native Koreans, high
The pathogenesis of PIBO in children is thought to result from an exaggerated and prolonged inflammatory response after a severe lower respiratory tract infection, which leads to chronic and often irreversible damage to the small airways[2]. The bronchiolar epithelial damage following lower respiratory tract infections leads to progressive epithelial dys
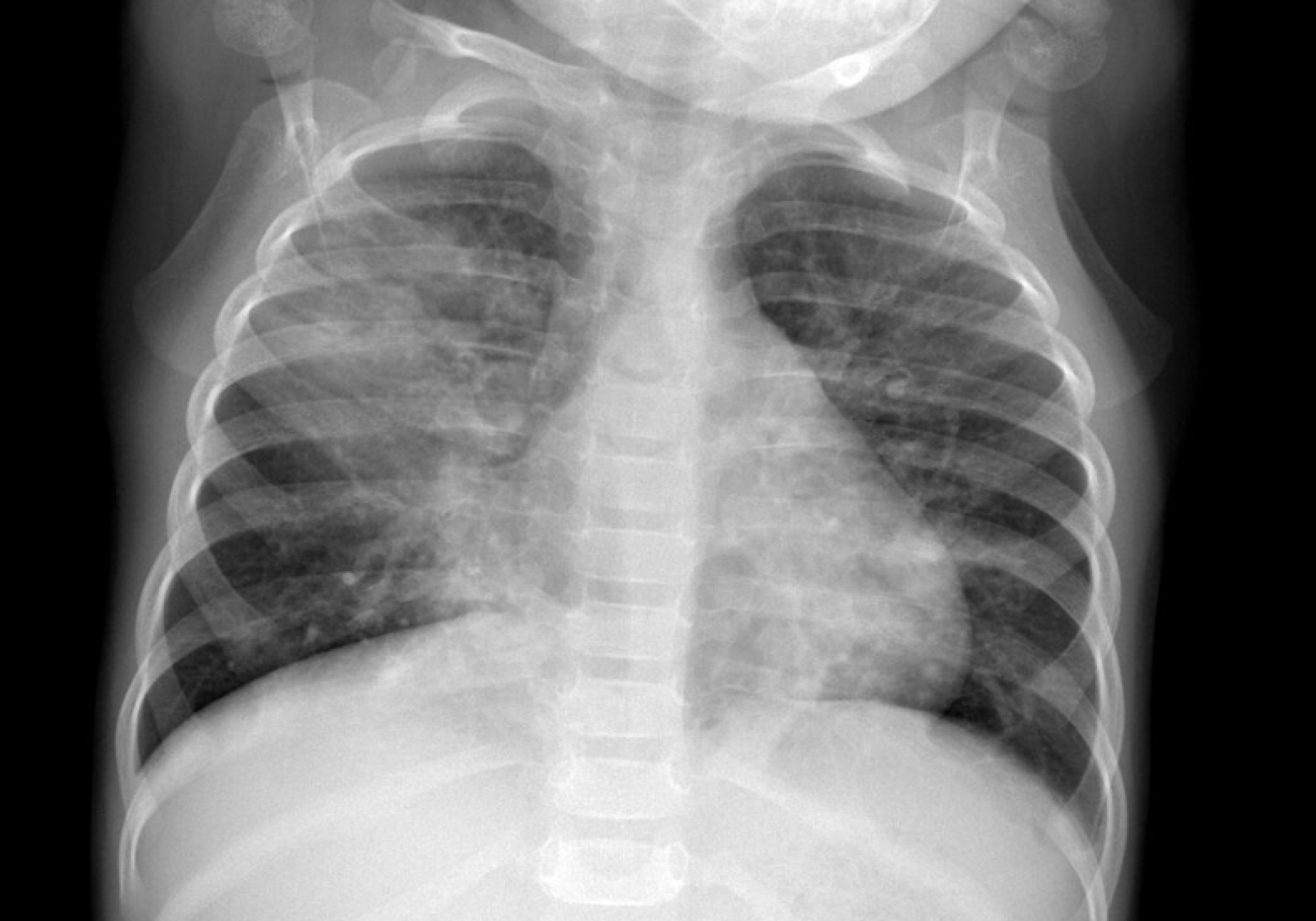
Children with PIBO typically present with persistent respiratory symptoms that do not fully resolve following an acute lower respiratory tract infection, often of viral origin (Figure 1). The hallmark symptoms include chronic cough, exer
The diagnosis of PIBO in children is primarily clinical, based on persistent respiratory symptoms, and is supported by complementary findings from imaging and pulmonary function tests[2]. Histopathology through lung biopsy is still considered the gold standard for definitive diagnosis, as it provides direct evidence of bronchiolar obliteration and fibrosis. However, its invasive nature, risk of complications, and limited feasibility in young children make it rarely per
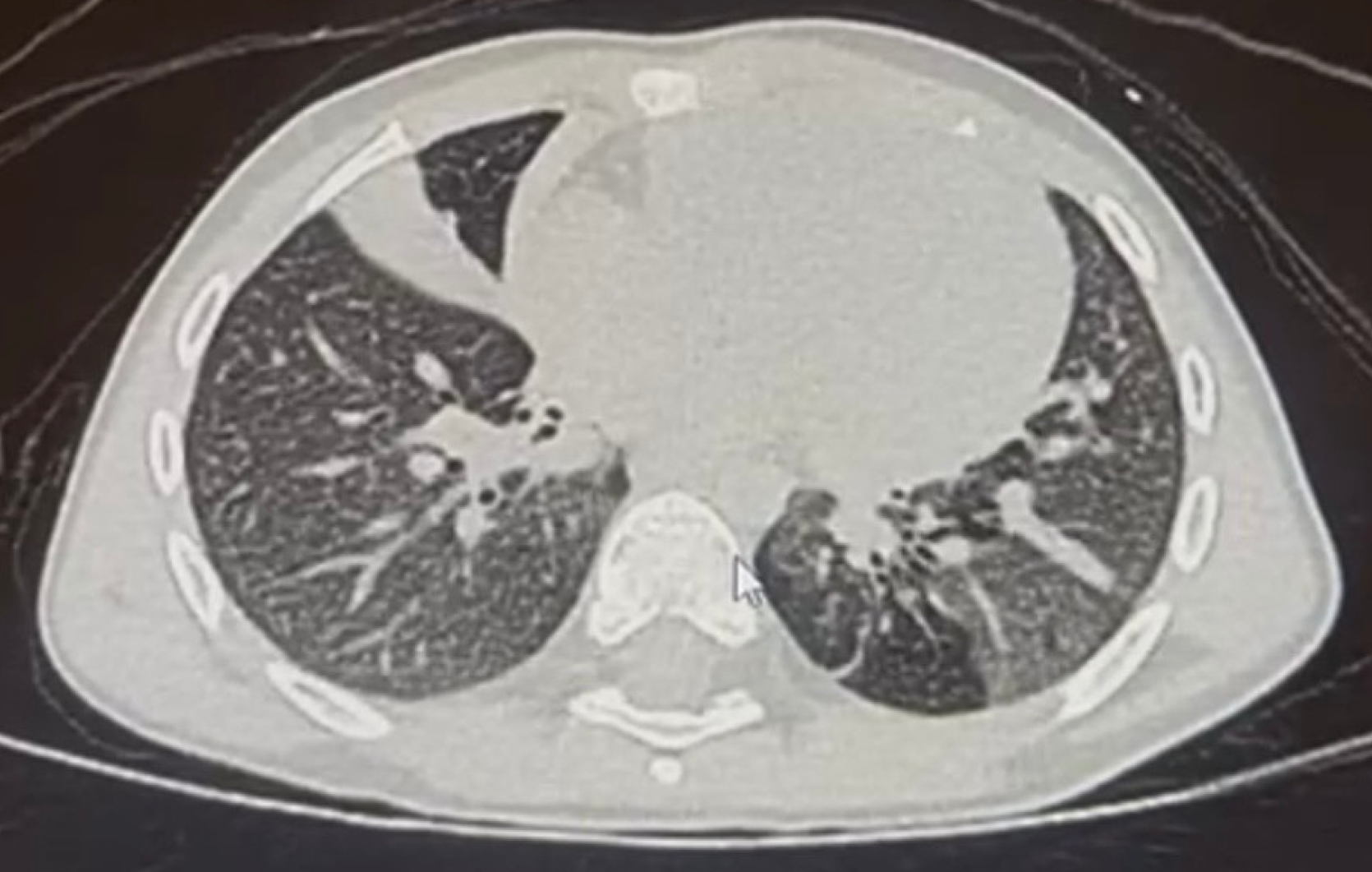
High-resolution computed tomography (HRCT) of the chest is currently the most informative non-invasive tool, revealing typical features such as mosaic attenuation, air trapping, bronchial wall thickening, and hyperinflation, es
Chest X-ray, while easily accessible, has limited sensitivity and specificity for PIBO and may only show non-specific signs such as hyperinflation or atelectasis (Figure 1). Therefore, it should not be relied upon alone to exclude or confirm the diagnosis.
Laboratory tests are non-specific and mainly serve to rule out other conditions, as there are no validated biomarkers for PIBO.
Overall, each diagnostic tool has significant limitations: Lung biopsy is invasive and impractical; PFTs are age-restricted and may be normal early on; HRCT, while informative, carries radiation risks and requires cooperation or se
Comment on imaging: The combination of chest X-ray (Figure 1) and HRCT (Figure 2) illustrates how initial pneumonia-related consolidations can evolve into structural changes typical of PIBO, such as mosaic perfusion and bronchiectasis. However, radiologic patterns alone should always be interpreted in the context of clinical history and pulmonary function results, given their limited specificity.
Pulmonary function assessment is essential in the follow-up of children with PIBO, but its role in the initial diagnosis is limited by practical and technical challenges. Spirometry, when feasible — usually in cooperative children older than 5-6 years — typically shows small airway involvement[3]. However, it is important to recognize that spirometry may be normal in early stages or in mild cases, and its sensitivity depends heavily on the child’s ability to perform reliable ma
Body plethysmography, where available, may reveal lung hyperinflation and increased residual volume (RV) and total lung capacity (TLC), suggesting air trapping and small airway involvement[1,6]. However, the application of pulmonary function testing in young children remains challenging due to cooperation issues, and age-appropriate normative data are limited[1,4] (Table 1).
| Technique | Recommended age | Advantages | Limitations | Role in PIBO |
| Spirometry | > 5-6 years (cooperative) | Widely available; standard obstructive pattern; non-invasive | Not feasible in very young children; variable sensitivity in early/mild disease | Useful to detect irreversible obstruction and monitor over time |
| Impulse oscillometry | 3-6 years (or older) | Minimal cooperation; measures small airway resistance | Limited availability; lack of standard pediatric reference ranges; interpretation may vary | Helpful in uncooperative children but not diagnostic alone |
| Body plethysmography | > 5-6 years (cooperative) | Detects air trapping; measures lung volumes (RV, TLC) | Requires full cooperation; not always available; sometimes sedation needed | Supports diagnosis of air trapping; complements spirometry |
| DLCO | > 7-8 years (good technique needed) | Assesses alveolar-capillary integrity; typically preserved in PIBO | Technically demanding; requires good breath-hold and cooperation; limited use in young children | Helps distinguish PIBO from interstitial diseases |
Measurement of the diffusing capacity of the lung for carbon monoxide (DLCO) can be informative: In PIBO, DLCO is generally preserved because the primary injury involves the conducting airways rather than the alveolar-capillary interface[2]. This helps differentiate PIBO from interstitial lung diseases where DLCO is typically reduced. Still, DLCO testing also relies on good patient technique and is feasible only in older, cooperative children.
Overall, pulmonary function tests provide supportive but not definitive information. They have limited sensitivity in early disease stages, require age-appropriate cooperation or specialized equipment under sedation, and cannot distin
Therefore, while abnormal spirometry and lung volumes can strengthen the suspicion of PIBO, they must always be interpreted together with clinical history and imaging results to avoid misdiagnosis. Over time, lung function may remain stable, deteriorate, or show minor improvements depending on the severity of the initial insult and ongoing inflammation[3,4]. Regular longitudinal pulmonary function monitoring remains essential to assess disease progression, evaluate response to therapy, and identify candidates who may benefit from advanced interventions.
Spirometry generally shows an obstructive pattern involving the small airways. However, in the early stages of the disease, pulmonary function tests may appear normal, while in later stages, a typical pattern can be observed. The spirometric pattern usually reveals a fixed or irreversible obstruction on the flow- volume curve, with a reduction in FEV1, the Tiffeneau index (FEV1/FVC) and end-expiratory flow (MEF25) and elevated RV/TLC[1]. There is usually a poor response to bronchodilators, reflecting the fixed nature of the small airways narrowing[2]. Additionally, forced expiratory flow between 25% and 75% of the pulmonary volume (FEF25%-75%) is frequently decreased, further highlighting small airway involvement[3]. Nevertheless, reliable spirometry requires patient cooperation, so it is usually feasible only in children over five or six years of age[1]. In younger patients, this can limit its diagnostic utility, and early disease stages may yield normal results, potentially delaying diagnosis[3,7]. Longitudinal cohort studies confirm that severe impairment can persist for years, underscoring the importance of repeated testing to monitor functional decline and guide therapy[3]. Despite these strengths, spirometry remains effort-dependent and less sensitive for detecting early small airway changes[6].
In children with (PIBO), the preserved DLCO represents a key functional parameter. Cazzato et al[2] reported that while FEV1 and FEF25%-75% decline progressively over time due to airway fibrosis, DLCO values frequently result within normal ranges, suggesting that alveolar-capillary gas exchange remains unchanged. This finding aligns with the work
Body plethysmography can provide additional insight into lung volumes such as RV and TLC. In PIBO, these often reveal hyperinflation and air trapping, particularly when spirometry is inconclusive[1,2,6]. This technique may be useful to detect peripheral airway involvement, which is typical in PIBO but harder to assess with spirometry alone. However, like other functional tests, it requires good cooperation and is usually feasible only in children older than 6-7 years. Younger or non-cooperative children may need sedation, which limits its routine application. Furthermore, there are few stan
Overall, while these functional assessments remain fundamental tools in the diagnostic work-up of PIBO, the available evidence suffers from significant heterogeneity between studies, small sample sizes, and a lack of standardized long-term follow-up. This limits the strength of recommendations and underscores the need for collaborative research to better define diagnostic thresholds and prognostic trajectories[1,4,8].
The treatment of PIBO is empirical. Therapy for this condition includes pharmacological and supportive treatments combined with non-pharmacological interventions[11,12] (Table 2). The goal of treatment is to limit inflammation by inhibiting lymphocytic proliferation. Generally, before initiating systemic anti-inflammatory therapy, it is advisable to perform a bronchoscopy with BAL to investigate any persistent infections.
| Treatment option | Description | Comments/evidence |
| Corticosteroids | Systemic or inhaled steroids used to reduce inflammation | Often used during acute exacerbations; long-term benefits uncertain; some improvement in symptoms reported |
| Bronchodilators | Inhaled β2-agonists and anticholinergics to relieve airway obstruction | Symptomatic relief, but variable response due to fixed airway obstruction |
| Macrolide antibiotics | Anti-inflammatory and immunomodulatory properties (e.g., azithromycin) | May reduce inflammation; some evidence in other chronic airway diseases; limited data in PIBO |
| Immunosuppressants | Agents like azathioprine, cyclophosphamide, or mycophenolate mofetil in severe cases | Used in refractory disease; evidence limited; risks of immunosuppression must be balanced |
| Oxygen therapy | Supplemental oxygen for hypoxemia | Supportive care in advanced disease with chronic hypoxia |
| Pulmonary rehabilitation | Exercise training, airway clearance, and breathing techniques | Improves quality of life and functional status; standard supportive care |
| Lung transplantation | Considered in end-stage PIBO with respiratory failure | Rare; only for selected severe cases; long-term outcomes variable |
| Other therapies | Experimental or adjunctive therapies, including antivirals, mucolytics, or novel agents | Limited evidence; research ongoing; no standard recommendations |
Therapeutic interventions for PIBO in children aim to reduce inflammation, alleviate symptoms, and preserve lung function, although no universally effective treatments exist due to the irreversible nature of airway damage. Corticosteroids, both systemic and inhaled, are commonly used in the early stages or during disease exacerbations to suppress ongoing inflammation. Oral prednisolone or intravenous methylprednisolone pulse therapy may be considered in moderate to severe cases, especially if active inflammation is suspected. Immunosuppressive agents such as azithromycin (used for its anti-inflammatory properties), hydroxychloroquine, or methotrexate have been explored in selected cases with variable success, primarily in children with a progressive clinical course. Bronchodilators, including beta-agonists and anticholinergics, may provide symptomatic relief, although their effect on lung function is often limited due to fixed airway obstruction.
Previous studies[4,5,13] described the bronchodilator response as an increase in the FEV1 by at least 12% following sal
Steroids: Based on the clinical course of the disease, inhaled and systemic steroids are used to contrast the inflammatory component. Corticosteroids should be administered as early as possible after diagnosis, before airway fibrosis develops[16].
There is general consensus that the treatment of choice is intravenous steroid pulse therapy, using methylprednisolone at a dose of 10-30 mg/kg for three consecutive days, repeated monthly for a period of 3 to 6 months. This treatment regimen has proven effective in pediatric patients with interstitial lung disease[1]. Long-term oral systemic corticosteroid therapy should be avoided, as it is associated with undesirable side effects and serious complications such as infection-related mortality and bone fractures.
Previous studies have shown that children affected by PIBO may benefit from methylprednisolone pulse therapy if bronchial wall thinning is observed on pre-treatment chest CT[17].
Given the unclear long-term effects of corticosteroids, their toxicity, and the high risk of severe infections, it would be advisable to use a non-steroidal treatment for the long-term management of this disease.
Azithromycin: In contrast to steroid therapy, azithromycin treatment is known to be effective in controlling neutrophilic inflammation and in promoting the improvement of lung function in various diseases such as diffuse panbronchiolitis, cystic fibrosis, and post-transplant bronchiolitis obliterans syndrome (BOS)[18-20]. The exact mechanism by which azithromycin modulates the inflammatory response is still unclear. Different mechanisms have been described such as a reduction in neutrophil count in the airways, suppression of interleukin-8 and other neutrophil-chemotactic cytokines and interference with neutrophil function. Regarding the use of azithromycin in PIBO, the data in the literature are scarce. In any case, even though no randomized controlled trials exist in children with PIBO, oral azithromycin at a dose of 10 mg/kg three times per week is recommended, having demonstrated efficacy in other obstructive diseases[16].
Fluticasone, azithromycin, and montelukast: In initial trials, montelukast slowed FEV1 decline in fibroproliferative BOS following lung transplantation[21], although a subsequent study by Ruttens et al. found no survival advantage over placebo[22]. However, several reports have indicated that combining inhaled fluticasone, azithromycin, and montelukast (FAM) may offer clinical and functional benefits in BOS patients[23]. A recent phase II, open-label, multicenter trial assessed FAM together with an initial corticosteroid pulse in new-onset BOS post-hematopoietic stem cell trans
| Ref. | Therapies | Pulmonary function outcomes | Key points/notes |
| Zheng et al[25] retrospective (2022) — 34 children, age > 5 years (n = 20); | Continuous vs intermittent ICS (budesonide ± terbutaline) | After 1-year, continuous ICS showed improvements in FVC, FEV1, MMEF 25%-75%, tidal flow ratios; intermittent ICS did not | Continuous ICS significantly improved airway obstruction; > 50% had positive bronchodilator tests |
| Zhang et al[26] (2018) Clinical cohort, China (2014-2017) — 30 children, median age 17 months | Long-term nebulized budesonide + terbutaline + ipratropium | Significant increase in TPEF%/TE and VPEF%/VE; HRCT improved in 82% of patients; symptoms greatly improved | Triple nebulization well tolerated, effective in young children, with both functional and radiologic improvements |
| Li et al[31] workshop | Oral prednisone (1.5 mg/kg/day taper over 6-9 months) + azithromycin (5-10 mg/kg, 3 days/week × 6 months) | Defined “effective”: Stable lung function (as < 10% decline); > 50% responded with reduced wheezing; effective in 86% of cases. No HRCT improvement | Combined steroids + azithromycin frequently effective; no control groups, but high subjective + functional response rates |
| Jerkic et al[1] Workshop report (BOS studies) | FAM regimen: Fluticasone + azithromycin + montelukast + steroid pulse | In BOS, poor pulmonary decline halted: Treatment failure (≥ 10% FEV1 drop) was only 6% at 3 months vs 40% historical controls | While untested in PIBO, single-center use suggested safety; efficacy needs formal trials |
| Yilmaz et al[27] (2023) IVIG study (2010-2021) — 11 severe PIBO patients | Regular IVIG infusions (weekly/monthly) | Reduced infections and hospital visits; all patients weaned off oxygen; radiological scores improved; BMI increased | Retrospective uncontrolled but showed clinical and radiologic gains in severe PIBO |
| Teixeira et al[28] (2013) randomized control trial — 30 patients | Tiotropium (LAMA) ± short-acting β2 agonists | Improvement in acute FEV1, FVC and FEF25%-75%; bronchodilator reversibility seen in approximately 25% of PIBO patients in related studies | Suggests LAMAs may be useful in PIBO—especially those with some reversibility |
Inhaled corticosteroids: A retrospective study that evaluates the effects of continuous inhaled corticosteroids (ICSs) on lung function in patients with PIBO in remission found that ICSs can effectively improve lung function and relieve airway obstruction in patients with PIBO > 5 years of age in remission, especially continuous ICSs (Table 3)[25]. After one year of ICSs therapy, patients over the age of 5 showed significant improvement in FVC and FEV1 compared to the beginning of follow-up. Also triple nebulized therapy (budesonide + bronchodilators) in toddlers significantly improves small airway tidal flow and imaging (Table 3)[26].
Intravenous immunoglobulin: Intravenous immunoglobulin (IVIG) offers notable benefits in severe PIBO when first-line measures fail, improving oxygenation and reducing infections and hospital visits. As demonstrated in the study by Yilmaz et al[27], PIBO patients exhibited favorable clinical and radiological responses to regular IVIG therapy, potentially due to steroid-induced or underlying immune dysfunction in severe PIBO (Table 3).
Long-acting muscarinic antagonists: Long-acting muscarinic antagonists (LAMAs) show potential benefit, particularly in partially reversible PIBO, improving FEV1. The trial conducted by Teixeira demonstrated a significant acute broncho
Supportive management of pediatric PIBO focuses on optimizing respiratory function, nutrition, immunity, and quality of life. Non-pharmacological interventions play a crucial role in the comprehensive management of PIBO. Supplemental oxygen is administered in case of hypoxemia especially during the first years of disease, with pulsed oximetry value below 92%[29]. Long-term oxygen therapy may be required in patients with chronic hypoxemia to support adequate tissue oxygenation and prevent complications such as pulmonary hypertension. Adequate nutritional support is essential to promote growth and respiratory muscle strength. Immunizations, specifically seasonal influenza and pneumococcal vaccines, are routinely recommended to prevent respiratory infections that could exacerbate disease. In patients with bronchiectasis, airway clearance via inhalation of hypertonic saline or physiotherapy can help mobilize secretions from the small airways. Finally, physical exercise and pulmonary rehabilitation are recommended components of chronic care. Pulmonary rehabilitation programs, including physical therapy and breathing exercises, help improve exercise tolerance, reduce symptoms, and enhance quality of life, but data are available only for patients with BOS. In end-stage or refractory cases with severely impaired lung function and poor quality of life, lung transplantation may be considered, although it remains a last resort due to its complexity and associated risks[29] (Table 4). The main purpose of treatment is tailored control for each patient according to lung damage and clinical response.
| Intervention | Benefit | Limitations |
| Oxygen therapy | Corrects hypoxemia; prevents pulmonary hypertension[29] | Does not modify disease; burden of long-term use |
| Airway clearance | May reduce secretions in bronchiectasis | Extrapolated from BOS data; no PIBO-specific trials |
| Pulmonary rehabilitation | Improves exercise tolerance and quality of life in BOS | No pediatric PIBO data; resource-intensive |
| Nutrition vaccination | Supports growth and immunity | Standard of care, not disease-specific |
| Lung transplantation | Life-saving in end-stage disease | Early graft dysfunction, higher perioperative complications, low mortality rate; limited donor supply[32] |
Because no randomized, PIBO-specific trials exist, all current treatments for children remain empirical. The following hierarchy ranks interventions by pediatric evidence strength:
Critical summary: Strongest pediatric data support systemic steroid pulses, but evidence is limited to small (n < 50), uncontrolled series with short follow-up. ICS and azithromycin show promising safety and moderate efficacy in cohorts of 30-42 children, yet lack prospective randomization. Tiotropium and IVIG remain hypothesis-generating in small pediatric samples. Non-pharmacological measures are biologically plausible but untested in PIBO children.
Overall, establishment of a true therapeutic hierarchy in pediatric PIBO mandates multicenter, controlled trials, direct head-to-head comparisons, and extended follow-up to balance efficacy against toxicity and to develop evidence-based management guidelines.
This review evaluated the pulmonary function outcomes in pediatric PIBO and offered a descriptive overview of various therapeutic interventions. Our findings underline the difficulties in managing PIBO in children. Despite increasing awareness of pediatric PIBO, its management remains driven more by expert opinion than by strong evidence. Due to the rarity of the disease, the studies available are mostly retrospective, small-scale, and methodologically heterogeneous, and many are based on adult patients, which severely limits the strength of their conclusions. Functional impairments are well documented but based on small, retrospective cohorts. Cazzato et al[2] analyzed 10 children in a single-region cohort, confirming a predominantly obstructive pattern with significantly reduced FEV1 and FEF25%-75% in the majority. However, these studies lack control groups, and follow-up duration varies widely, limiting conclusions about natural history or treatment effects. Similarly, Colom et al[3] followed 46 children for up to 12 years, noting that some patients demonstrate mild improvements in FEV1 and FVC over time, but the lack of standardized interventions and possible survival bias constrain generalizability.
Radiological correlates and quantitative CT analysis have shown promise, as highlighted by Jung et al[4] and Kim et al[5] with cohorts of 47 and 23 children, respectively. Both studies emphasize the association between air trapping, mosaic perfusion, and worse pulmonary function. However, small sample sizes, retrospective designs, and lack of external validation reduce the reliability of proposed imaging biomarkers.
Several studies assessing bronchodilator responsiveness, such as Zheng et al[25] and Zhang et al[26], suggest that a variable percentage of patients may show partial reversibility. However, these reports combine patients with varying disease severity, stages, and treatment histories, confounding the interpretation. Moreover, the small sample sizes (e.g., Zhang et al[26], n = 30) and retrospective design heighten the risk of overestimating treatment effects due to regression to the mean or measurement variability. Longitudinal data show that pulmonary function may remain stable, decline, or slightly improve depending on the severity of initial damage and response to therapy[30] (Table 5).
| Ref. | Sample size (n) | Design | Main findings | Key limitations/biases |
| Jerkic et al[1] | (Multiple studies) | Retrospective, multicenter workshop summary | Provided diagnostic framework based on expert consensus; highlighted frequent severe obstructive patterns on PFTs and HRCT | Mainly expert opinion, heterogeneous cases, lack of standardized treatment comparisons |
| Cazzato et al[2] | 10 | Case series | Confirmed persistent airway obstruction; FEV1 and FEF25%-75% significantly reduced in most patients | Small cohort, no control group, variable follow-up, single-region recruitment |
| Colom et al[3] | 46 | Cross sectional (12-year follow-up) | Some children showed mild improvements in FEV1 and FVC over 12 years; severity depends on initial damage | Limited generalizability, possible survival bias, lack of treatment standardization |
| Jung et al[4] | 47 | Cross sectional | Identified predictors of poor prognosis (e.g., mosaic perfusion, air trapping on CT) | Short term follow-up, single-center, no external validation |
| Kim et al[5] | 23 | Cross-sectional with quantitative CT | Suggested quantitative CT correlates well with lung function | Variability in CT technique, no standard thresholds |
| Li et al[31] | 42 | Prospective observational | Azithromycin + corticosteroids improved symptoms in 86% of children | Non-randomized, no control group, subjective symptom assessment |
| Zheng et al[25] | 34 | Retrospective ICS comparison | Continuous inhaled corticosteroids improved FEV1 more than intermittent use in children > 5 years | Lack of randomization, possible adherence bias, short follow-up |
| Zhang et al[26] | 30 | Case series nebulization therapy | Long term budesonide + bronchodilator nebulization in toddlers improved small airway tidal flows and CT findings | Small cohort, no control group; subjective imaging interpretation; variable treatment duration |
| Teixeira et al[28] | 30 | Randomized control trial | Tiotropium showed acute bronchodilator response in some PIBO patients | Single center, short follow-up |
| Yilmaz et al[27] | 11 | Retrospective, single center | IVIG treatment showed clinical stability in severe PIBO | Small sample, no comparative arm, retrospective bias |
| Colom and Teper[30] | 125 | Retrospective observational study | Proposed criteria to diagnose PIBO early | Single center, retrospective design, limited generalizability, Needs validation in prospective cohorts |
| Rosewich et al[12] | 20 (+ 22 controls) | Cross-sectional | Highlighted persistent neutrophilic airway inflammation | No intervention tested, only descriptive, single time-point, age variability |
Corticosteroid treatment, the mainstay of therapy, is supported by limited evidence of variable quality. Lee et al[15] conducted a meta-analysis pooling small observational studies but acknowledged high heterogeneity (I2 > 70%) and moderate to high risk of bias in most included studies. Similarly, Yoon et al[17] found that pulse methylprednisolone may improve lung function in selected patients, however their study had only 17 subjects and lacked randomization or placebo control. These design limitations weaken the validity of conclusions about optimal dosing, timing, or duration. Regarding macrolides, Li et al[31] described clinical benefit from azithromycin plus corticosteroids in 42 children but did not include a comparator arm, making it impossible to isolate the drug effect from the natural disease course (Table 6). While randomized controlled trials are lacking for pediatric patients, azithromycin is currently recommended at 10 mg/kg three times per week, based on its efficacy in related chronic airway diseases[16]. The FAM regimen (fluticasone, azithromycin, montelukast), initially trialed in BOS post-transplant, demonstrated a marked reduction in FEV1 decline and treatment failure rates (6% vs 40% in historical controls) and improvement in HRCT findings of PIBO patients[29,31]; however, data in PIBO are limited to small case series[22]. The immunological milieu and airway remodeling in post-transplant BOS may differ significantly from PIBO in previously healthy children, making direct application of these findings questionable. Emerging therapies like IVIG (27, n = 11) or tiotropium (28, n = 30) have been tested only in small, single-center cohorts without controls. Such anecdotal evidence is useful for hypothesis generation but cannot support routine clinical use. Similarly, the role of non-pharmacological interventions, such as pulmonary rehabilitation, remains hypothetical in PIBO due to the lack of robust pediatric trials[29]. Oxygen supplementation, while helpful in improving saturation value, does not appear to significantly improve spirometric parameters, supporting its role as supportive care rather than a disease-modifying treatment[1,29]. Lung transplantation remains the ultimate intervention for children with end-stage PIBO, but it is associated with severe complications, including early graft dysfunction and high rates of postoperative extracorporeal membrane oxygenation[32]; these inherent risks and complexities restrict its widespread applicability.
| Ref. | Efficacy | Limitations |
| Systemic corticosteroid pulses (IPMT) (Yoon et al[17], 2015) | Short-term FEV1 gains; better IPMT response with bronchial wall thickening on pre-treatment CT | Small, uncontrolled series; no symptom scores, no pulmonary function test, growth suppression; adverse effects |
| ICS (Zheng et al[25], 2022) (Zhang et al[26], 2018) | ↑ in FEV1, FVC of continuous ICS group over 12 months; improved small-airway flows in toddlers | Retrospective; modest cohorts; adherence bias; durability beyond 1 year unknown |
| Azithromycin (macrolide) (Li et al[31], 2014) | Clinical stability in 86% when combined with steroids | Uncontrolled; symptom-based outcomes; risk of resistance |
| Tiotropium (LAMA) (Teixeira et al[28], 2013) | Good acute FEV1 increase | Small cohort; no placebo; unknown long-term impact |
| Intravenous immunoglobulin (IVIG) (Yilmaz et al[27], 2023) | Improved oxygenation; fewer infections in severe cases | Retrospective; no comparator; high cost; limited availability |
Another recurring limitation is the absence of standardized diagnostic criteria and objective outcome measures. Studies differ in how they define PIBO, measure lung function, and report response to therapy, making meta-analyses unreliable[15]. Only a few studies, like Jung et al[4] and Kim et al[5], used quantitative CT or structured follow-up to correlate imaging with functional prognosis; however, the second study had a cohort of only 23 children and both studies lacked external validation.
Moreover, the vast majority of studies fail to account for potential confounders such as co-existing conditions, adherence to therapy, or socioeconomic factors that could influence outcomes. Very few include blinded assessment of spirometry, leading to possible measurement bias. Finally, the heterogeneity in reporting adverse effects of prolonged corticosteroid or macrolide therapy means that risk-benefit analyses remain incomplete.
The overall picture that emerges is clear: The current evidence base is too fragmented and low-quality to inform robust, standardized treatment algorithms for PIBO. Clinicians must remain cautious in interpreting apparent benefits from interventions that lack adequately powered, controlled trials. The field urgently needs prospective multicenter studies with larger, well-characterized cohorts, uniform diagnostic definitions, longer follow-up and standardized spirometric endpoints.
Additionally, future trials should include stratified analyses to identify which subgroups — based on age, pathogen, severity, or inflammatory phenotype — benefit from specific regimens. It is crucial to move beyond descriptive series and develop hypothesis-driven research, including randomized controlled trials of immunomodulators, antifibrotics, or biologic agents targeting airway remodeling.
Until then, current practice should emphasize timely diagnosis, close lung function monitoring, and individualized therapy while balancing the limited efficacy and potential harms of repeated systemic corticosteroid use. Careful mo
In summary, the management of PIBO must evolve from empirical, fragmented care to evidence-informed practice. This transition will only be possible if the pediatric pulmonology community invests in methodologically rigorous studies that can deliver answers relevant to everyday clinical decision-making. Importantly, the broader impact of PIBO on children’s daily activities and quality of life must not be overlooked. Many pediatric patients experience chronic sym
Table 5 provides a structured summary of the key studies, highlighting the methodological limitations and the overall low level of evidence.
Overall evidence is based on small, single-center pediatric cohorts (n often < 50) with heterogeneous designs and short or variable follow-up, limiting external validity.
Quantitative CT (Jung et al[4], Kim et al[5]) and inhaled steroids (Zheng et al[25], Zhang et al[26]) offer the most consistent pediatric-specific data, yet remain retrospective and uncontrolled.
Imaging studies highlight potential predictive markers (air trapping, mosaic attenuation), but there is no standardized quantitative cut-off.
Pulmonary function data are frequently heterogeneous, with variable definitions of “improvement” and inconsistent use of bronchodilator testing.
Systemic steroid pulse efficacy is inferred from case series and expert consensus[1], but lacks prospective pediatric trials.
Interventions such as corticosteroids, azithromycin, and FAM show promise in limited case series, but robust randomized trials are lacking, increasing the risk of selection bias and publication bias.
Novel therapies (IVIG, tiotropium, azithromycin combination) are hypothesis-generating only, without randomized evaluation in children.
Non-pharmacological approaches, like pulmonary rehabilitation, remain under-investigated, with evidence often extrapolated from BOS post-transplant populations rather than PIBO specifically.
Managing PIBO in children can be challenging due to the lack of standardized treatments. Crucially, this field suffers from a deficit of multicenter, randomized controlled trials with sufficiently large cohorts to generate evidence capable of guiding clinical decision-making. Without such data, treatment remains fragmented and largely guided by individual clinician experience rather than scientific consensus. The complexity of PIBO demands a multidisciplinary, individualized treatment approach, but these are often not systematically implemented due to the lack of clear, evidence-based protocols. Moreover, it is imperative that future research focus on the identification of biomarkers, genetic factors, and individual risk profiles to develop targeted therapies. However, until these approaches transition from theory to clinical reality, meaningful improvements in long-term outcomes will remain elusive. Future research must address these gaps through prospective multicenter studies, standardized diagnostic criteria, and longer follow-up with robust pulmonary function endpoints, in order to establish evidence-based guidelines for pediatric PIBO management.
| 1. | Jerkic SP, Brinkmann F, Calder A, Casey A, Dishop M, Griese M, Kurland G, Niemitz M, Nyilas S, Schramm D, Schubert R, Tamm M, Zielen S, Rosewich M. Postinfectious Bronchiolitis Obliterans in Children: Diagnostic Workup and Therapeutic Options: A Workshop Report. Can Respir J. 2020;2020:5852827. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 61] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 2. | Cazzato S, Poletti V, Bernardi F, Loroni L, Bertelli L, Colonna S, Zappulla F, Timoncini G, Cicognani A. Airway inflammation and lung function decline in childhood post-infectious bronchiolitis obliterans. Pediatr Pulmonol. 2008;43:381-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 59] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 3. | Colom AJ, Maffey A, Garcia Bournissen F, Teper A. Pulmonary function of a paediatric cohort of patients with postinfectious bronchiolitis obliterans. A long term follow-up. Thorax. 2015;70:169-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 57] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 4. | Jung JH, Kim GE, Min IK, Jang H, Kim SY, Kim MJ, Kim YH, Shin HJ, Yoon H, Sohn MH, Lee MJ, Kim KW. Prediction of postinfectious bronchiolitis obliterans prognosis in children. Pediatr Pulmonol. 2021;56:1069-1076. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 5. | Kim J, Kim MJ, Sol IS, Sohn MH, Yoon H, Shin HJ, Kim KW, Lee MJ. Quantitative CT and pulmonary function in children with post-infectious bronchiolitis obliterans. PLoS One. 2019;14:e0214647. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 6. | Smith KJ, Fan LL. Insights into post-infectious bronchiolitis obliterans in children. Thorax. 2006;61:462-463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 7. | Kim CK, Kim SW, Kim JS, Koh YY, Cohen AH, Deterding RR, White CW. Bronchiolitis obliterans in the 1990s in Korea and the United States. Chest. 2001;120:1101-1106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 83] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 8. | Liu D, Liu J, Zhang L, Chen Y, Zhang Q. Risk Factors for Post-infectious Bronchiolitis Obliterans in Children: A Systematic Review and Meta-Analysis. Front Pediatr. 2022;10:881908. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 9. | Arakawa H, Webb WR. Air trapping on expiratory high-resolution CT scans in the absence of inspiratory scan abnormalities: correlation with pulmonary function tests and differential diagnosis. AJR Am J Roentgenol. 1998;170:1349-1353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 112] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 10. | Siegel MJ, Bhalla S, Gutierrez FR, Hildebolt C, Sweet S. Post-lung transplantation bronchiolitis obliterans syndrome: usefulness of expiratory thin-section CT for diagnosis. Radiology. 2001;220:455-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 52] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 11. | Kavaliunaite E, Aurora P. Diagnosing and managing bronchiolitis obliterans in children. Expert Rev Respir Med. 2019;13:481-488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 68] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 12. | Rosewich M, Zissler UM, Kheiri T, Voss S, Eickmeier O, Schulze J, Herrmann E, Dücker RP, Schubert R, Zielen S. Airway inflammation in children and adolescents with bronchiolitis obliterans. Cytokine. 2015;73:156-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 46] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 13. | Zhang L, Irion K, Kozakewich H, Reid L, Camargo JJ, da Silva Porto N, Abreu e Silva FA. Clinical course of postinfectious bronchiolitis obliterans. Pediatr Pulmonol. 2000;29:341-350. [PubMed] [DOI] [Full Text] |
| 14. | Yu X, Wei J, Li Y, Zhang L, Che H, Liu L. Longitudinal Assessment of Pulmonary Function and Bronchodilator Response in Pediatric Patients With Post-infectious Bronchiolitis Obliterans. Front Pediatr. 2021;9:674310. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 15. | Lee E, Park S, Yang HJ. Pulmonary Function in Post-Infectious Bronchiolitis Obliterans in Children: A Systematic Review and Meta-Analysis. Pathogens. 2022;11:1538. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 16. | Moonnumakal SP, Fan LL. Bronchiolitis obliterans in children. Curr Opin Pediatr. 2008;20:272-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 94] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 17. | Yoon HM, Lee JS, Hwang JY, Cho YA, Yoon HK, Yu J, Hong SJ, Yoon CH. Post-infectious bronchiolitis obliterans in children: CT features that predict responsiveness to pulse methylprednisolone. Br J Radiol. 2015;88:20140478. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 18. | Steinkamp G, Schmitt-Grohe S, Döring G, Staab D, Pfründer D, Beck G, Schubert R, Zielen S. Once-weekly azithromycin in cystic fibrosis with chronic Pseudomonas aeruginosa infection. Respir Med. 2008;102:1643-1653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 40] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 19. | Simpson JL, Powell H, Baines KJ, Milne D, Coxson HO, Hansbro PM, Gibson PG. The effect of azithromycin in adults with stable neutrophilic COPD: a double blind randomised, placebo controlled trial. PLoS One. 2014;9:e105609. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 74] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 20. | Verleden GM, Vanaudenaerde BM, Dupont LJ, Van Raemdonck DE. Azithromycin reduces airway neutrophilia and interleukin-8 in patients with bronchiolitis obliterans syndrome. Am J Respir Crit Care Med. 2006;174:566-570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 219] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 21. | Verleden GM, Verleden SE, Vos R, De Vleeschauwer SI, Dupont LJ, Van Raemdonck DE, Vanaudenaerde BM. Montelukast for bronchiolitis obliterans syndrome after lung transplantation: a pilot study. Transpl Int. 2011;24:651-656. [PubMed] [DOI] [Full Text] |
| 22. | Ruttens D, Verleden SE, Demeyer H, Van Raemdonck DE, Yserbyt J, Dupont LJ, Vanaudenaerde BM, Vos R, Verleden GM. Montelukast for bronchiolitis obliterans syndrome after lung transplantation: A randomized controlled trial. PLoS One. 2018;13:e0193564. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 42] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 23. | Norman BC, Jacobsohn DA, Williams KM, Au BK, Au MA, Lee SJ, Moravec CK, Chien JW. Fluticasone, azithromycin and montelukast therapy in reducing corticosteroid exposure in bronchiolitis obliterans syndrome after allogeneic hematopoietic SCT: a case series of eight patients. Bone Marrow Transplant. 2011;46:1369-1373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 57] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 24. | Williams KM, Cheng GS, Pusic I, Jagasia M, Burns L, Ho VT, Pidala J, Palmer J, Johnston L, Mayer S, Chien JW, Jacobsohn DA, Pavletic SZ, Martin PJ, Storer BE, Inamoto Y, Chai X, Flowers MED, Lee SJ. Fluticasone, Azithromycin, and Montelukast Treatment for New-Onset Bronchiolitis Obliterans Syndrome after Hematopoietic Cell Transplantation. Biol Blood Marrow Transplant. 2016;22:710-716. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 187] [Cited by in RCA: 161] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 25. | Zheng H, Yu X, Chen Y, Lin W, Liu L. Effects of Inhaled Corticosteroids on Lung Function in Children With Post-infectious Bronchiolitis Obliterans in Remission. Front Pediatr. 2022;10:827508. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 26. | Zhang XM, Lu AZ, Yang HW, Qian LL, Wang LB, Zhang XB. Clinical features of postinfectious bronchiolitis obliterans in children undergoing long-term nebulization treatment. World J Pediatr. 2018;14:498-503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 27. | Yilmaz Aİ, Gül Y, Kapaklı H, Ünal G, Çağlar HT, Ercan F, Reisli İ, Keleş S, Poyraz N, Pekcan S. Successful treatment of postinfectious bronchiolitis obliterans with gamma globulin in a tertiary center: 10 years of experience. Pediatr Pulmonol. 2023;58:2769-2776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 28. | Teixeira MFC, Rodrigues JC, Leone C, Adde FV. Acute bronchodilator responsiveness to tiotropium in postinfectious bronchiolitis obliterans in children. Chest. 2013;144:974-980. [PubMed] [DOI] [Full Text] |
| 29. | Mazenq J, Dubus JC, Chanez P, Gras D. Post viral bronchiolitis obliterans in children: A rare and potentially devastating disease. Paediatr Respir Rev. 2024;52:58-65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 30. | Colom AJ, Teper AM. Clinical prediction rule to diagnose post-infectious bronchiolitis obliterans in children. Pediatr Pulmonol. 2009;44:1065-1069. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 50] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 31. | Li YN, Liu L, Qiao HM, Cheng H, Cheng HJ. Post-infectious bronchiolitis obliterans in children: a review of 42 cases. BMC Pediatr. 2014;14:238. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 65] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 32. | Iablonskii P, Carlens J, Mueller C, Aburahma K, Niehaus A, Boethig D, Franz M, Floethmann K, Sommer W, Optenhoefel J, Tudorache I, Greer M, Koeditz H, Jack T, Hansmann G, Kuehn C, Horke A, Hansen G, Haverich A, Warnecke G, Avsar M, Salman J, Bobylev D, Ius F, Schwerk N. Indications and outcome after lung transplantation in children under 12 years of age: A 16-year single center experience. J Heart Lung Transplant. 2022;41:226-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/