©Author(s) (or their employer(s)) 2026.
World J Clin Pediatr. Mar 9, 2026; 15(1): 109301
Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.109301
Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.109301
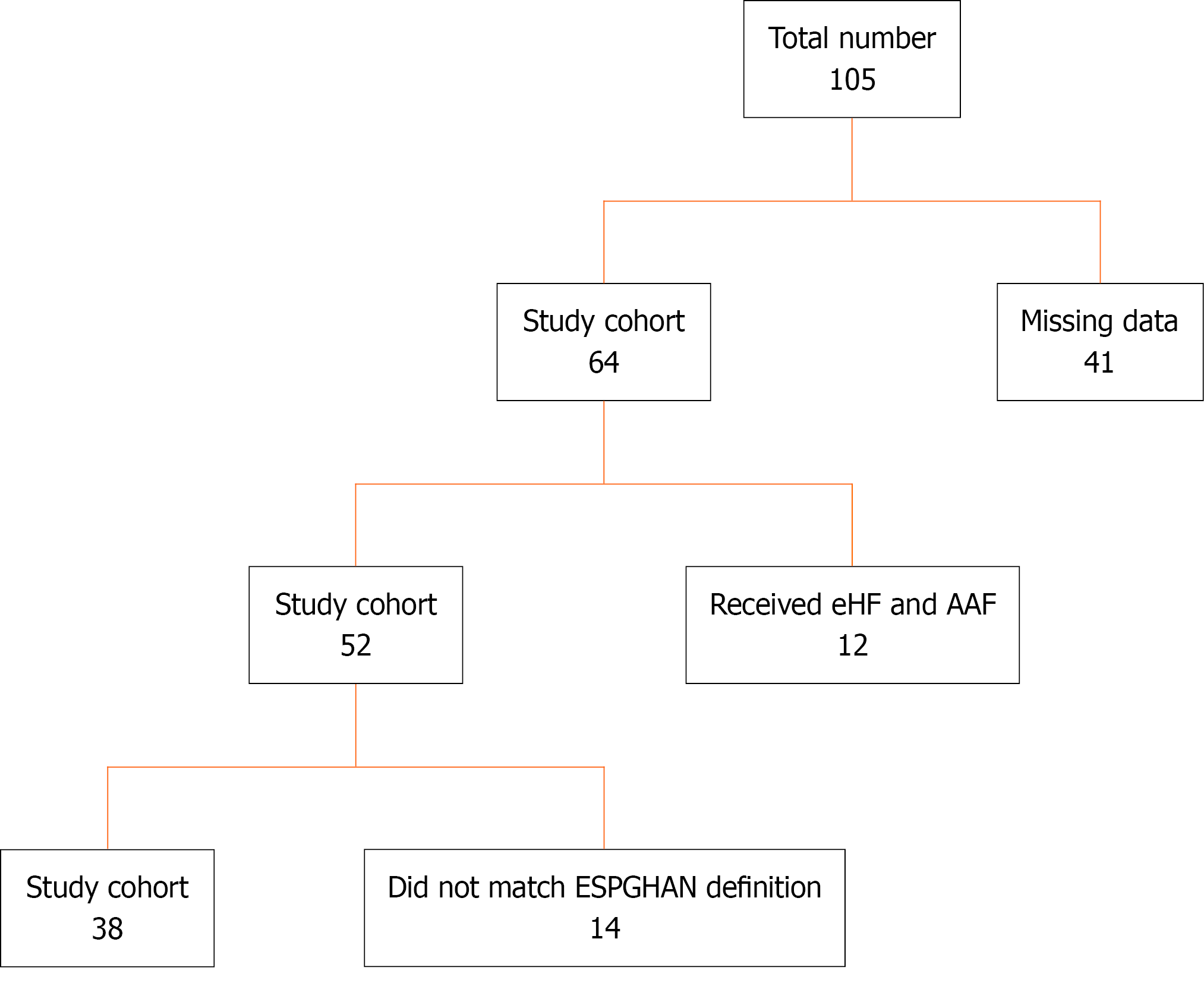
Figure 1 Flow diagram of participant selection and inclusion in the study cohort.
Of the initial 105 patients evaluated, 41 were excluded due to missing data. Among the remaining 64 patients, 12 received both extensively hydrolyzed formula (eHF) and amino acid-based formula (AAF), and 14 did not meet the European Society of Pediatric Gastroenterology, Hepatology, and Nutrition diagnostic criteria for Cow’s milk protein allergy. The final study cohort included 38 patients. AAF: Amino acid-based formula; eHF: Extensively hydrolyzed formula; ESPGHAN: European Society of Pediatric Gastroenterology, Hepatology, and Nutrition.
- Citation: Khdair Ahmad F, Alkayid MR, Ahmad TM, Hadidi RH, Rahhal RM. Cow’s milk protein allergy in infants: Clinical presentations and outcomes. World J Clin Pediatr 2026; 15(1): 109301
- URL: https://www.wjgnet.com/2219-2808/full/v15/i1/109301.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v15.i1.109301