INTRODUCTION
With the aging population on the rise, disorders of muscle function and obstructive sleep apnea (OSA) syndrome[1-5] have garnered increasing attention from the medical community. Muscle dysfunction, characterized by muscle weakness and atrophy, primarily contributes to the decline in physical function among the elderly. OSA, a sleep-related respiratory disorder characterized by recurrent episodes of airway obstruction and reduced ventilation during sleep, significantly impairs sleep quality and daily functioning[6,7]. In recent years, a growing body of evidence suggests a close association between muscle disorders and OSA, potentially sharing common pathogenic mechanisms and influencing each other to form a complex pathological network[8-10].
The correlation between muscle disorders and OSA holds significant practical and theoretical implications[11]. Firstly, both conditions are highly prevalent in the elderly population, with incidence rates rising with age[12]. Therefore, exploring their relationship in depth can enhance our understanding of the mechanisms underlying age-related decline in physical function and inform the development of effective intervention strategies[13,14]. Secondly, muscle disorders and OSA exhibit similarities in clinical manifestations and pathophysiological mechanisms, such as abnormalities in neuromuscular transmission and central nervous system (CNS) regulation[15,16]. This similarity provides a basis for investigating potential therapeutic targets and approaches. Finally, studying the correlation between muscle disorders and OSA promotes interdisciplinary collaboration and innovation in medical research. Currently, research on the relationship between muscle dysfunction and OSA has become a focal point both domestically and internationally[6,11,15]. Significant progress has been made at various levels, including neuroimaging, CNS regulation, and respiratory control. For instance, abnormal neuromuscular transmission is considered a common pathogenic mechanism underlying both muscle disorders and OSA. Studies have also indicated that reduced sensitivity of the respiratory center in OSA patients may be linked to decreased muscle function. Moreover, pathophysiological processes such as oxidative stress and inflammation have been identified as key contributors to the development of these diseases. However, despite these advancements, the correlation between muscle disorders and OSA still faces numerous challenges and unresolved questions. Is the relationship between muscular disorders and OSA causal or coincidental, and to what extent do confounding factors such as lifestyle play a role? What specific pathomechanisms drive the vicious cycle between these two conditions? How can these conditions be effectively intervened upon and treated to enhance patients’ quality of life?
To integrate research findings from the past five years and provide a systematic explanation of the pathogenesis of muscle disorders and OSA syndrome, as well as their interconnectedness. This study distinguishes itself from previous single-level investigations by innovatively analysing interactions across multiple levels, including neuromuscular transmission, CNS regulation, and respiratory control. It aims to elucidate the reciprocal relationship between muscular disorders and OSA, thereby providing valuable support for the prevention and management of these conditions.
RESEARCH PROGRESS ON THE PATHOGENESIS OF SARCOPENIA
Sarcopenia, a muscle disorder closely associated with aging, involves multiple levels of pathogenesis, including abnormalities in neuromuscular transmission and disorders in CNS regulation[15-18]. In recent years, as research has deepened, these mechanisms have gradually been elucidated, offering new insights for the treatment and prevention of muscle disorders[19-21].
Neuromuscular transmission abnormalities
Neuromuscular transmission refers to the process by which nerve impulses are transmitted to skeletal muscle fibers, leading to muscle contraction[22,23]. During this process, acetylcholine (ACh) is released at the synapse and binds to acetylcholine receptors (AChRs) on the synaptic membrane, initiating muscle contraction. Abnormalities in neuromuscular transmission in patients with sarcopenia primarily manifest in two aspects: Synaptic dysfunction and muscle neurotransmitter disorders[24,25]. Synaptic dysfunction refers to obstacles encountered when nerve impulses are transmitted at the neuromuscular junction. Studies have revealed a decrease in the number of synapses at neuromuscular junctions in patients with muscle disorders, along with abnormalities in synaptic structure, leading to impaired nerve impulse transmission[26]. Furthermore, ACh release from the synaptic membrane is also affected, exacerbating abnormalities in neuromuscular transmission[27]. This synaptic dysfunction may be associated with age-related neurodegenerative changes or may serve as one of the significant causes of sarcopenia[28].
Muscle neurotransmitter disorders involve imbalances in neurotransmitters within muscle fibers. Among patients with sarcopenia, there is a decrease in the number of AChRs in muscle fibers, resulting in reduced sensitivity and weakened responses to muscle impulses[29]. Additionally, other neurotransmitters such as glutamic acid and glycine may also play a role in the onset of sarcopenia[30]. Dysregulation of these neurotransmitters may interfere with signal conduction within muscle fibers, affecting muscle growth, differentiation, and metabolism[31].
Recent research has not only deepened our understanding of AChR receptor damage but also highlighted the complex involvement of other immune and cellular factors in the pathogenesis of sarcopenia[16,32]. Some studies focused on identifying new therapeutic targets to restore normal neuromuscular transmission function[33]. For example, strategies targeting T cells and B cells, as well as methods to regulate immune system balance, have become areas of research focus. Moreover, research on the neuroimmune regulatory network has been strengthened to gain a more comprehensive understanding of sarcopenia pathogenesis. This includes investigating interactions between neurons and immune cells, as well as regulatory mechanisms governing neurotransmission and immune cell activation[34]. In summary, the latest research findings reveal the multi-level complexity of sarcopenia pathogenesis and provide deeper insights into the development of more effective treatment strategies. These studies offer new avenues for personalized therapy and drug design targeting various aspects of pathogenesis.
Imbalance in the CNS regulation
The CNS plays a crucial role in regulating muscle growth, differentiation, and metabolic processes[35,36]. Patients with sarcopenia often exhibit disorders in CNS regulation, characterized by an imbalance between neuronal degeneration and nerve growth factor (NGF) levels, affecting muscle quality maintenance. Neuronal degeneration refers to age-related changes in CNS neurons, resulting in diminished regulatory capacity over muscle function. Studies have reported decreased neuron counts in the CNS of sarcopenia patients, along with synaptic abnormalities and disruptions in neurotransmitter synthesis and release. These alterations may lead to reduced nerve impulses reaching muscles, thereby impairing muscle growth and metabolism[36]. The imbalance between NGF and muscle quality further contributes to sarcopenia pathogenesis. NGF is a bioactive substance crucial for promoting the growth, differentiation, and survival of neurons and muscle fibers. Among sarcopenia patients, reduced NGF levels impair muscle fiber growth and survival. Additionally, disruptions in the balance between NGF and muscle quality exacerbate the progression of muscle laxity. In summary, the pathogenesis of sarcopenia involves multiple aspects, including abnormalities in neuromuscular transmission and CNS regulation. The interplay between these mechanisms likely plays a significant role in sarcopenia development. Future research should delve into the specific details and interrelationships of these mechanisms to devise more effective prevention and treatment strategies for sarcopenia. Furthermore, deeper investigations may unveil novel therapeutic targets and drugs, ultimately enhancing the quality of life for sarcopenia patients.
DISCUSSION ON THE PATHOGENESIS OF OSA AND RESPIRATORY SUSPENSION SYNDROME
OSA is a sleep-related respiratory disorder characterized by recurrent episodes of respiratory suspension and reduced ventilation during sleep. Its pathogenesis encompasses multiple levels, with anatomical abnormalities in the upper airway and dysregulation of the respiratory center being two central elements[37,38]. In recent years, as research has advanced, the pathogenesis of OSA has gradually become better understood, offering novel insights into disease treatment and prevention[39,40].
Abnormal anatomy of the upper airway and OSA
The abnormal anatomy of the upper airway is a significant factor contributing to OSA. During sleep, muscle relaxation and gravity can lead to the collapse of the upper airway, resulting in airway narrowing or complete obstruction, leading to respiratory pauses[41]. This vicious cycle of airway collapse and respiratory suspension further exacerbates the structural abnormalities of the upper airway. Additionally, airway remodeling plays a crucial role in the progression of OSA[42,43]. Remodeling involves structural and functional changes in the airway due to chronic hypoxia and inflammation. These changes include thickening of the airway wall, hyperplasia of smooth muscle, and increased mucus secretion, all of which increase airway resistance and worsen OSA. In recent years, researchers have extensively studied the relationship between upper airway anatomy and OSA using high-resolution imaging technology and molecular biology methods. They have identified abnormalities in the soft tissue structure, bone structure, and neuroanatomy control of OSA patients[44]. These abnormalities may be influenced by factors such as age, gender, genetics, and obesity, collectively contributing to the onset and progression of OSA.
Abnormal regulation of the respiratory center and OSA
Another significant aspect of OSA pathogenesis is the abnormal regulation of the respiratory center. Studies have revealed decreased sensitivity of the respiratory center in OSA patients, resulting in weakened responses to hypoxia and hypercapnia, leading to respiratory regulatory dysfunction[45]. Although clinical studies have demonstrated an association between the pathogenesis of OSA and impaired respiratory regulation, the causal relationship remains controversial and warrants further investigation. This regulatory dysfunction may be attributed to the sleep-wake mechanism in OSA patients. The sleep-wake mechanism involves the transition between sleep and wakefulness in the body. In OSA patients, repeated episodes of respiratory pauses and reduced ventilation lead to hypoxia and carbon dioxide retention, triggering an arousal response. Although this arousal response may temporarily alleviate OSA symptoms, it disrupts sleep architecture and reduces sleep quality over time. Additionally, the arousal response itself may further disrupt respiratory center regulation and exacerbate OSA. In recent years, research on abnormalities in respiratory center regulation and OSA has made significant advancements. Through animal experiments and clinical studies, researchers have identified genes and molecular mechanisms associated with abnormal respiratory center control. These findings offer new insights into OSA pathogenesis and provide novel avenues for disease treatment and prevention.
The physiological mechanism of the development of OSA
In the development of OSA, changes and oxidation stress of oxygen state are the key physiological mechanisms. During periods of apnoea, patients experience hypoxaemia and hypertonia, triggering adaptive responses to hypoxic conditions. These adaptive responses encompass sympathetic nervous system activation, renin-angiotensin system activation, and heightened inflammatory responses. Recent research indicates that alterations in hypoxic states correlate with the activation of oxidative stress pathways[35]. In the environment of hypoxemia, cells produce more oxygen-free radicals, leading to an increase in oxidation stress. Oxidation stimulation can cause cell membrane lipid peroxidation, protein oxidation, and nucleic acid oxidation, thereby damaging the cell structure and function[46]. The changes in the state of oxygen and oxidation should have a variety of effects on the pathological level of pathophysiology. First of all, changes in oxygen syndrome cause the sympathetic nerve activation, increasing heart rate, blood pressure, and blood sugar levels[47]. Secondly, oxidation stimulation can lead to an increase in inflammatory reactions, which are closely related to the development of cardiovascular disease and diabetes[48]. In addition, the changes in oxygen condensation and oxidation stress are also related to the decline in sleep quality, cognitive dysfunction, depression, and other nervous system problems[49]. These effects may be manifested in daytime drowsiness, decline in learning and memory ability, and emotional instability. In summary, the latest research emphasizes the key role of changes in oxygen-oxygen and oxidation stimulation in the OSA pathogenesis. Understanding these mechanisms will help us better understand OSA’s pathology and provide strong support for the development of new therapeutic strategies.
The pathogenesis of obstructive sleep respiratory suspension syndrome involves multiple aspects, such as anatomical structure abnormalities in the upper airway and abnormal respiratory center regulation. The interaction and interaction between these mechanisms may be an important reason for the development of OSA. In the future, research should further explore the specific details and interrelated correlations of these mechanisms in order to provide more effective strategies and methods for the prevention and treatment of OSA. At the same time, with the deepening of research, we are also expected to discover new therapeutic targets and drugs, bringing a better quality of life to OSA patients.
THE CORRELATION ANALYSIS OF MUSCLE DISORDER AND OSA SYNDROME
The correlation between muscle disorders and OSA syndrome has garnered significant attention in recent years. Existing cross-sectional clinical studies indicate that these two conditions are not only prevalent among the elderly but also exhibit a mutually reinforcing relationship, leading to the exacerbation of both disorders. This study aims to examine the correlation between them by investigating the impact of muscle hypotonia on OSA and how OSA exacerbates muscle hypotonia. The present research provides a detailed and in-depth analysis of this association, drawing upon current research advances reported in the literature[45-49].
The impact of muscle laxity on OSA
The influence of muscle disorder on OSA primarily manifests in two aspects: Decreased muscle function and airway stability, and respiratory muscle fatigue leading to worsening of OSA. Decrease in muscle function and airway stability: Patients with muscle disorders experience a reduction in muscle quality and strength, resulting in weakened support of the muscles surrounding the upper airway. This renders the upper airway more susceptible to collapse during sleep, leading to respiratory pauses and reduced ventilation. Additionally, decreased muscle function may also disrupt the normal structure and function of the airway, further compromising airway stability and exacerbating OSA symptoms[44,45]. Respiratory muscle fatigue and worsening of OSA: Respiratory muscles in patients with muscle disorders are also affected, leading to respiratory muscle fatigue[46]. This increases the burden on respiratory muscles to maintain normal breathing function, which can result in weakness and fatigue. During sleep, respiratory muscle fatigue may escalate, causing respiratory regulatory dysfunction and worsening OSA. Furthermore, respiratory muscle fatigue may lead to atrophy and fibrosis of respiratory muscles, further impairing respiratory function and perpetuating a vicious cycle.
OSA’s role in promoting muscle laxity
OSA’s promotion of muscle disorder is evident in three main aspects: Chronic hypoxia and muscle metabolism abnormalities, sleep fragmentation and deterioration of muscle quality, and the induction of oxidative stress contributing to muscle laxity. Chronic hypoxia and muscle metabolism abnormalities: OSA patients experience recurrent episodes of breathing cessation and reduced ventilation during sleep, resulting in chronic hypoxia. Experimental data indicate prolonged hypoxia not only impacts muscle energy metabolism and protein synthesis but may also lead to alterations in muscle fiber type, diminishing muscle quality and strength. Additionally, chronic hypoxia can impair mitochondrial function in muscle cells, exacerbating metabolic abnormalities[47]. Sleep fragmentation and decline in muscle quality: OSA significantly disrupts sleep quality, leading to sleep fragmentation. Longitudinal studies suggest prolonged sleep fragmentation not only interferes with the body’s recovery and repair processes but also impedes muscle growth and development. Studies suggest that sleep fragmentation may hinder the activation and proliferation of muscle satellite cells, affecting muscle growth and repair. Moreover, sleep fragmentation can disrupt the balance between muscle protein synthesis and degradation, resulting in a decline in muscle quality[48]. Induction of oxidative stress and muscle laxity: OSA patients experience chronic hypoxia and sleep fragmentation, leading to increased oxidative stress in the body[49,50]. Mechanistically, oxidative stress not only compromises the survival and function of muscle cells but also exacerbates degeneration and apoptosis of muscle fibers[51]. Furthermore, oxidative stress may impair the activity and function of muscle satellite cells, further hindering muscle growth and repair[52,53]. These combined factors contribute to the onset and progression of muscle disorders. In summary, the interplay between muscle laxity and OSA involves complex mechanisms, with each condition exacerbating the other. Understanding these interactions is crucial for developing effective therapeutic strategies for both conditions.
COMMON PATHOLOGICAL CHANGES
Change of oxygen-syndrome state and oxidation should be stimulated in the two diseases
The pathophysiological changes observed in both muscle laxity (sarcopenia) and OSA involve alterations in oxygenation status and oxidative stress[54-57]. Oxygen and carbon dioxide retention are shared challenges faced by both conditions, exacerbating disease progression and affecting patient quality of life. In sarcopenia, the immune system’s attack on AChR impedes neuromuscular transmission. Changes in oxygenation status can worsen sarcopenia symptoms by further impairing neuromuscular transmission, leading to increased muscle fatigue and weakness. Moreover, oxidative stress may hasten AChR damage and intensify immune system attacks on AChR.
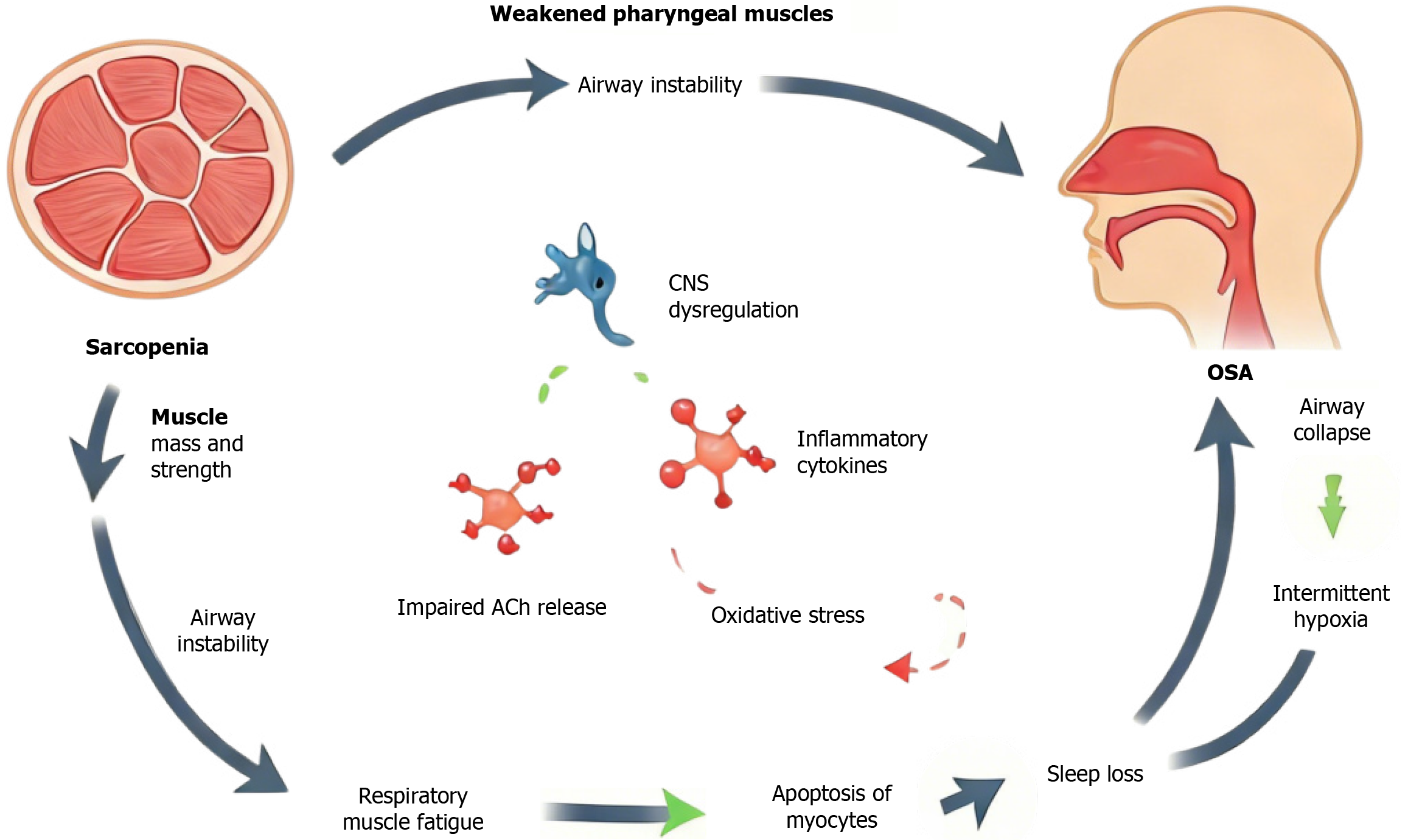
In OSA, repeated episodes of respiratory pauses result in oxygenation changes. Patients experience hypoxia and carbon dioxide retention, triggering various physiological and metabolic responses[55-59]. Instability in oxygenation status may activate the immune system through oxidative stress mechanisms, promoting inflammation. This inflammation not only affects upper respiratory tract tissues but also impacts systemic immune function (Figure 1): (1) Potential interaction mechanisms: Recent research suggests that changes in oxygenation status and oxidative stress may influence the development of sarcopenia and OSA through multiple interaction mechanisms in patients with both conditions; (2) Immune response regulation: Hypoxia and oxidative stress may aberrantly activate the immune system, leading to the generation of inflammatory factors. In sarcopenia, this may exacerbate immune attacks on AChR, while in OSA, it may induce systemic inflammation affecting respiratory and other tissues; (3) Effects on neuromuscular transmission: Hypoxia and oxidative stress may impact both diseases by directly affecting neuromuscular transmission. In sarcopenia, oxidative stress may worsen AChR damage, hindering neuromuscular transmission. In OSA, hypoxia may negatively affect CNS regulation, leading to respiratory pauses that further disrupt oxygenation status; and (4) Cell injury and repair: Changes and oxidative stress in oxygenation status may directly damage muscle cells and neurons, leading to cell injury and death, which accelerates tissue degeneration, worsening both conditions.
Figure 1 Potential interaction mechanisms in sarcopenia and obstructive sleep apnea.
The diagram elucidates the intricate interplay between sarcopenia, a condition of age-related loss of muscle mass and function, and obstructive sleep apnea, a common sleep disorder characterized by recurrent upper airway obstruction during sleep. The mechanisms portrayed in the diagram suggest that there may be direct physiological and pathological links between sarcopenia and obstructive sleep apnea, which can exacerbate each other through their impacts on neuromuscular transmission, immune responses, and cellular injury and repair processes. CNS: Central nervous system; OSA: Obstructive sleep apnea; Ach: Acetylcholine.
Understanding the changes in oxygenation status and the common mechanisms stimulated in sarcopenia and OSA can facilitate the formulation of more comprehensive and personalized treatment strategies. Interventions targeting oxygenation status and oxidative stress may offer more effective treatment options for patients with both conditions, enhancing their quality of life and slowing disease progression. The latest research progress provides valuable insights into this field, offering new avenues for future treatment directions.
FUTURE RESEARCH DIRECTION AND CHALLENGE
With the continuous deepening of muscle disorder and OSA syndrome pathogenesis, future research is facing unprecedented opportunities and challenges. The common role of neuromuscular transmission and OSA, the potential role of the CNS regulation in muscle laxity and OSA, and the development of cross-disciplinary cooperation and comprehensive intervention strategies have become an important direction for future research.
Common treatment targets of neuromuscular transmission and OSA
Neuromuscular transmission is a common key link in muscle dysfunction and OSA pathogenesis. Therefore, finding a common treatment target in the process of neuromuscular transmission is of great significance for treating muscle laxation and OSA. Future research can find possible therapeutic targets by in-depth discussion of the specific molecular mechanism of neurotomaging. For example, in response to problems such as synaptic dysfunction and muscle neurotransmitter disorders, researchers can develop new types of neurotransmitter receptor agonists or antagonists to improve the efficiency and quality of neurotransmitter transmission. In addition, with the development of gene editing technology, future research can also fundamentally improve the abnormalities of neurotomer transmission through editing related genes. For example, through gene editing tools such as clustered regularly interspaced short palindromic repeats (CRISPR-Cas9), editing a specific gene that causes abnormalities that cause neuromuscular transmission, thereby restoring the normal function of neurotomer transmission.
The potential role of the CNS regulates in muscle liberation and OSA
The CNS plays an important regulatory role in the course of muscle disorders and OSA. Future research can further explore the specific mechanism of CNS regulation and how to treat muscle disorders and OSA by regulating the CNS. For example, for the imbalances of neuronal degeneration and neurological growth factor and muscle quality maintenance, researchers can develop new types of neuroplasma or growth factors to promote the survival and regeneration of neurons, while improving muscle quality. In addition, with the development of neuroscience, future research can also improve the regulation of muscle and breathing by regulating the activity of neurotransmitters or neuropathy to improve the CNS. For example, by regulating the levels of neurotransmitters such as dopamine and 5-hydroxytryptamine, it can improve the regulation of muscle growth and respiratory function in the CNS, thereby alleviating the symptoms of muscle symptoms and OSA.
Development of cross-disciplinary cooperation and comprehensive intervention strategies
The research of muscle disorders and OSA requires a variety of disciplines such as biology, medicine, and biological engineering. In the future, research needs to strengthen cross-disciplinary cooperation, integrate knowledge and technical means of different disciplines, and jointly develop comprehensive intervention strategies. For example, combined with the technology of biomedical engineering, development can monitor and evaluate equipment and systems that can monitor and evaluate muscular disorders and OSA conditions in real time; combine the knowledge of nutrition and sports medicine, formulate personalized diet and sports plans to improve patients’ muscle quality and breathing function. In addition, future research needs to pay attention to clinical transformation and application. Through clinical trials and practical applications, the effectiveness and safety of comprehensive intervention strategies provide new methods and means for the treatment of muscle laxity and OSA.
In short, future research on muscles and OSA will face huge challenges and opportunities. Through in-depth discussion of the common treatment targets of neuromuscular transmission and OSA, the potential role of the CNS regulation in muscle disorders and OSA, and the development of cross-disciplinary cooperation and comprehensive intervention strategies, it is expected that the treatment and prevention of muscle disorders and OSA will be addressed. Provide new ideas and methods. At the same time, it is also necessary to strengthen cross-disciplinary cooperation and clinical transformation applications to promote the practical application and promotion of research results.
CONCLUSION
The correlation between muscle hypotonia and OSA syndrome is significant, particularly among the elderly population. Findings from this study indicate that muscle hypotonia exacerbates OSA by impairing muscular function and increasing airway instability, whilst OSA worsens muscle hypotonia through mechanisms such as chronic hypoxia and oxidative stress. However, confounding factors are likely to exert a substantial influence on the observed comorbidity, necessitating further investigation. Future research should focus on identifying shared therapeutic targets, elucidating the bidirectional effects of both conditions, and fostering interdisciplinary collaboration to develop comprehensive intervention strategies. Rigorous longitudinal and interventional studies are essential to validate these proposed mechanisms, addressing the limitations of existing observational data. This approach will enhance patients' quality of life, delay disease progression, and improve the efficacy of clinical interventions.
ACKNOWLEDGEMENTS
We would like to express our gratitude to the Department of Orthopedics of the First People’s Hospital of Yunnan Province and the Department of Otolaryngology of Yunnan Medical University Affiliated Yan’an Hospital for providing the academic platform.
Peer review: Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Orthopedics
Country of origin: China
Peer-review report’s classification
Scientific quality: Grade C
Novelty: Grade D
Creativity or innovation: Grade D
Scientific significance: Grade C
P-Reviewer: Kumar S, Consultant, Full Professor, Head, Senior Researcher, India S-Editor: Bai SR L-Editor: A P-Editor: Xu J