Published online Apr 18, 2026. doi: 10.5312/wjo.v17.i4.114757
Revised: November 17, 2025
Accepted: January 12, 2026
Published online: April 18, 2026
Processing time: 195 Days and 5.3 Hours
Charcot neuroarthropathy of the ankle presents significant surgical challenges with conventional tibiotalocalcaneal (TTC) fusion techniques often prioritizing mechanical stability while neglecting biological deficiencies, resulting in high non-union rates. This study evaluated a refined surgical approach integrating biological augmentation with fibular preservation.
To evaluate trans-malleolar TTC fusion with bone marrow aspirate (BMA)-soaked allograft and fibular preservation for chronic Charcot ankle arthropathy.
We conducted a retrospective case series of 24 adult patients with chronic ankle Charcot arthropathy who underwent TTC fusion with biologic augmentation between 2023 and 2024 at a tertiary care trauma center. The technique involved a transmalleolar approach with fibular preservation, comprehensive joint preparation, and biologic augmentation using BMA-soaked allograft. Outcomes were assessed using the validated American Orthopedic Foot and Ankle Society hindfoot scale and Short Form-36 health survey with comprehensive radio
At a mean follow-up of 1.5 years, 100% (24/24) of patients achieved successful TTC fusion confirmed by robust bony bridging. Mean American Orthopedic Foot and Ankle Society scores demonstrated significant improvement from 31.2 ± 9.1 preoperatively to 76.8 ± 14.2 at the final follow-up (P < 0.001), exceeding the minimal clinically important difference. Short Form-36 scores showed significant improvements across all domains with physical function scores improving from 29.4 ± 10.8 to 72.1 ± 15.3 (P < 0.001). Complications included breakage of both fibular fixation screws managed conservatively without compromising stability, nail protrusion requiring a minor removal procedure, and a periprosthetic tibial fracture treated conservatively with walker boot immobilization. No major infections, wound complications, or amputations were observed.
Trans-malleolar TTC fusion with BMA-soaked allograft and fibular preservation achieved high union and improved outcomes in chronic Charcot ankle arthropathy.
Core Tip: This retrospective case series evaluated a transmalleolar tibiotalocalcaneal fusion with biologic augmentation while preserving the fibula for chronic Charcot neuroarthropathy of the ankle. The technique aims to enhance stability, promote fusion, and reduce wound morbidity by avoiding fibular osteotomy and supplementing fixation with orthobiologics. We reported limb salvage, fusion, alignment, and complication outcomes, demonstrating consistent union and acceptable risks in a high-risk cohort. Transmalleolar tibiotalocalcaneal fusion with fibular preservation is a pragmatic, tissue-sparing option for complex Charcot neuroarthropathy ankles.
- Citation: Elalfy MM, Embaby OM, Abulaban OA, Aladl AN, Abdelrazek M, Youssef K, Abushal MH, Kassab F. Transmalleolar tibiotalocalcaneal fusion with biologic augmentation and fibular preservation for chronic Charcot ankle arthropathy: A retrospective cohort study. World J Orthop 2026; 17(4): 114757
- URL: https://www.wjgnet.com/2218-5836/full/v17/i4/114757.htm
- DOI: https://dx.doi.org/10.5312/wjo.v17.i4.114757
Charcot neuroarthropathy of the ankle represents one of the most challenging conditions in foot and ankle surgery. It is characterized by progressive joint destruction, deformity, and potential limb loss. The condition predominantly affects patients with diabetes and peripheral neuropathy with ankle involvement occurring in approximately 10%-20% of cases[1]. The fundamental therapeutic objective involves achieving a stable, plantigrade, ulcer-free foot capable of with
Tibiotalocalcaneal (TTC) fusion remains the primary surgical intervention for advanced ankle Charcot arthropathy. However, conventional techniques have historically focused on mechanical stabilization through rigid fixation, often overlooking the inherent biological deficiencies that characterize the Charcot process[2]. These biological challenges include compromised bone quality, impaired vascularity, and dysregulated inflammatory responses that significantly impact healing potential. Consequently, traditional approaches report non-union rates ranging from 30%-50%, high
Recent advances in orthobiologic therapies have demonstrated promise in enhancing bone healing and fusion rates in challenging orthopedic contexts. Bone marrow aspirate (BMA) concentrate, rich in mesenchymal stem cells, growth factors, and osteoprogenitor cells, has emerged as a valuable adjunct in promoting osteogenesis[4].
The integration of biological augmentation with mechanical stabilization represents a paradigm shift toward addressing both aspects of the healing process. A study by Sherman et al[5] demonstrated that biologic augmentation of TTC arthrodesis using allogeneic bone block and osteoinductive agents combined with protected weight-bearing yielded high fusion rates (92.9% on plain radiographs and 90.9% on CT scans). This approach also led to favorable patient-reported outcomes, including reduced pain and preserved lower extremity function, suggesting its efficacy in challenging cases[5].
This study presented a technical refinement to the traditional transmalleolar TTC fusion approach that incorporates biological augmentation with BMA-soaked allograft and enhanced fibular preservation techniques. These technical modifications aim to address both mechanical instability and biological deficiencies inherent to Charcot arthropathy, potentially improving fusion rates and clinical outcomes over conventional methods.
This retrospective case series analyzed 24 consecutive patients diagnosed with chronic ankle Charcot arthropathy who underwent TTC fusion with biological augmentation between January 2023 and December 2024 at a tertiary care trauma center. All procedures were performed by a single experienced foot and ankle surgery consultant to ensure technical consistency.
Inclusion criteria comprised adult patients (≥ 18 years) with chronic ankle Charcot arthropathy classified as Eichenholtz stage II (coalescence) or stage III (reconstruction) with anatomical classification as Brodsky type 3A or Sanders and Frykberg type IV. All patients had documented diabetes mellitus with recorded glycated hemoglobin levels and absence of active infection or severe peripheral vascular disease (ankle-brachial index ≥ 0.6).
Exclusion criteria included acute Charcot flares (Eichenholtz stage I), mechanical ulceration (Wagner grade ≥ 2), active osteomyelitis, severe peripheral vascular disease requiring revascularization, and systemic conditions significantly impairing bone healing.
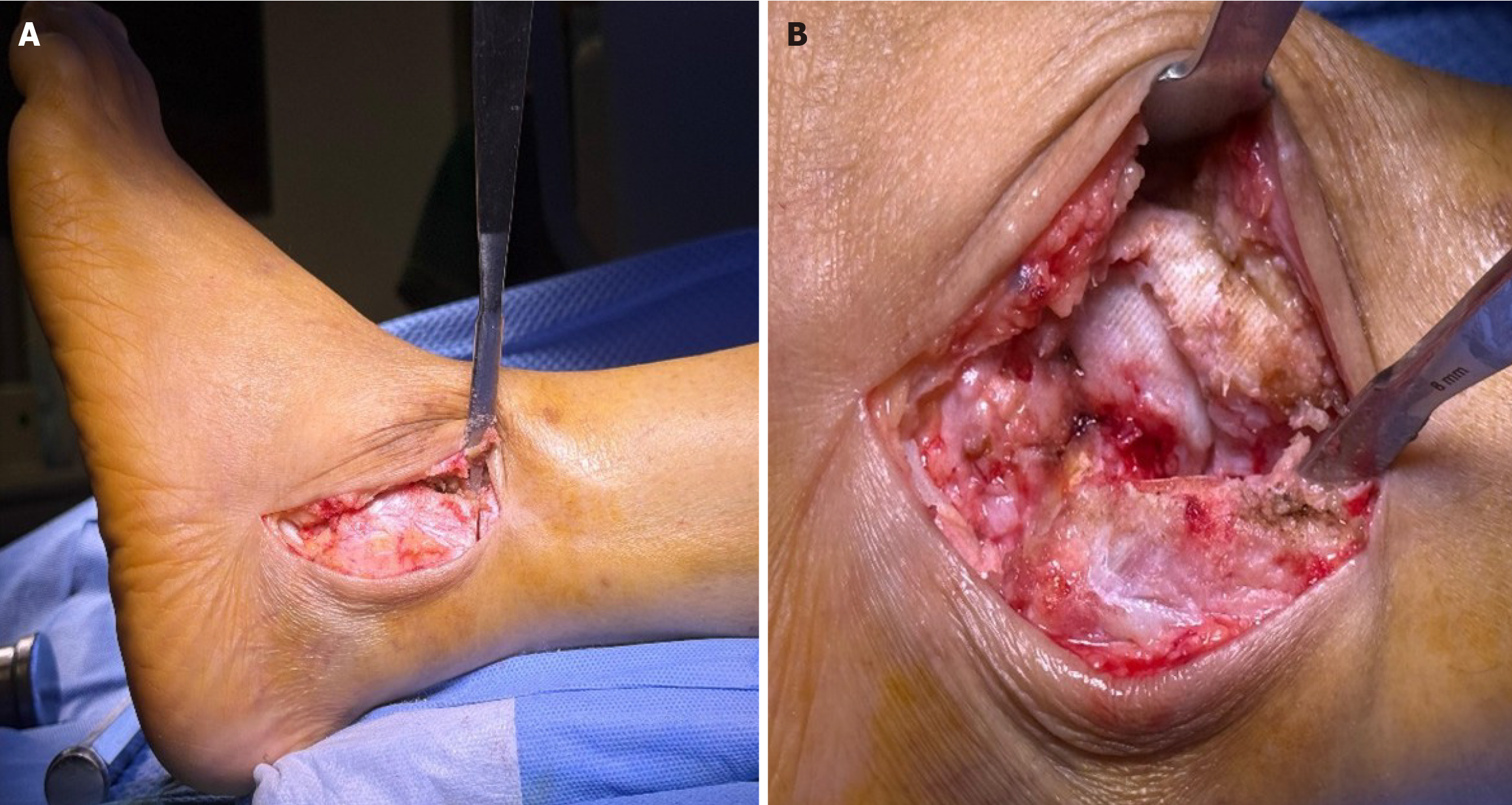
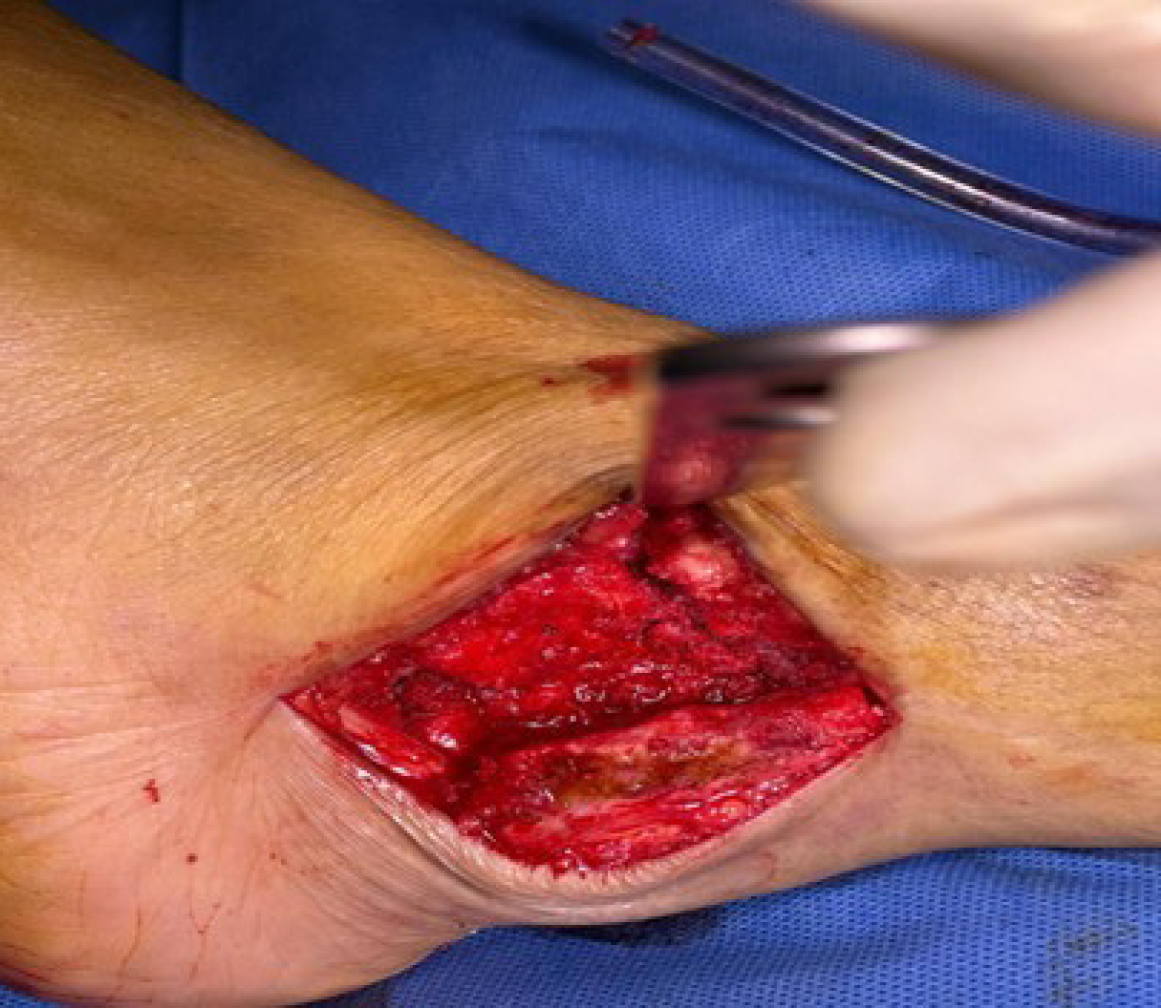
Step 1: Transmalleolar approach with periosteal preservation: The surgical approach was initiated through a lateral transmalleolar incision with a controlled lateral malleolar osteotomy performed approximately 2-4 cm proximal to the ankle joint line using an oscillating saw with the cut made perpendicular to the long axis of the fibula. The critical innovation involves opening the syndesmosis from anterior to facilitate preparation, followed by careful sliding and eversion of the fibula downward while maintaining its attachment to all related anatomical structures. Meticulous attention was paid to preserving the periosteal attachment and vascular supply to the entire distal osteotomized fragment, ensuring the fibular segment remained viable as a vascularized flap. This was achieved by careful subperiosteal dissection and protection of the perforating branches of the peroneal artery. This technique allows the preserved lateral malleolus to be retracted posteriorly using a Hohmann retractor, providing excellent visualization and access to both the ankle and subtalar joint complexes while maintaining the biological viability of the malleolar fragment for subsequent reconstruction and enhanced healing potential (Figures 1 and 2).
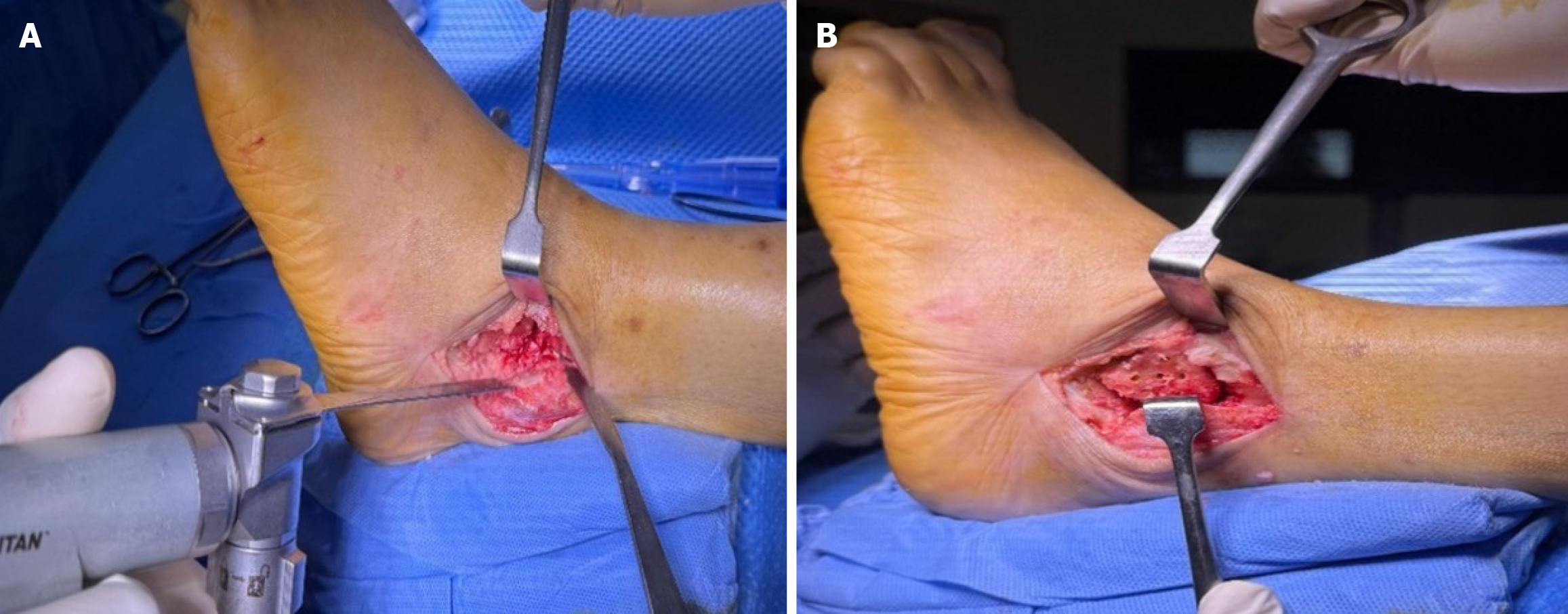
Step 2: Comprehensive joint surface preparation: Following adequate exposure, comprehensive joint surface pre
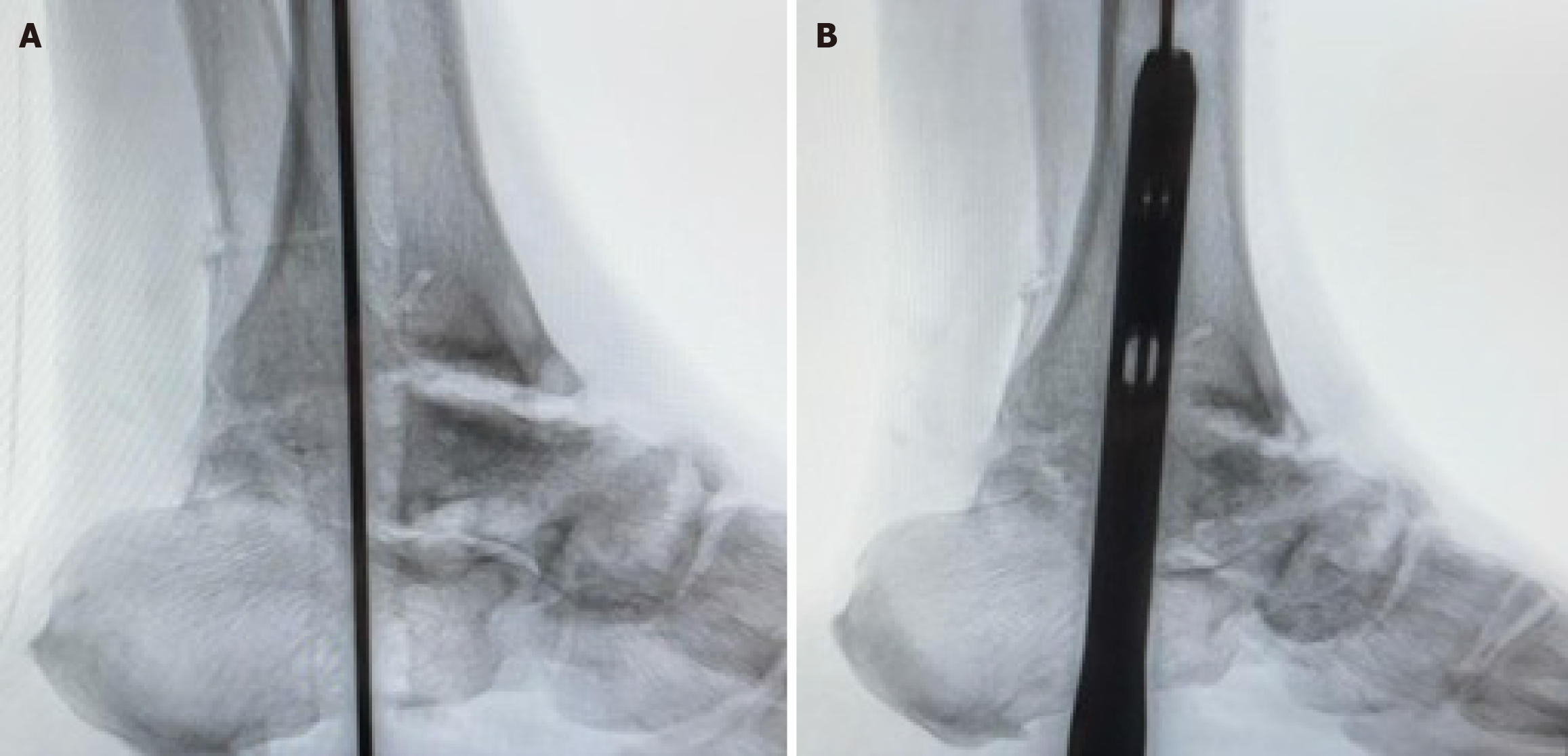
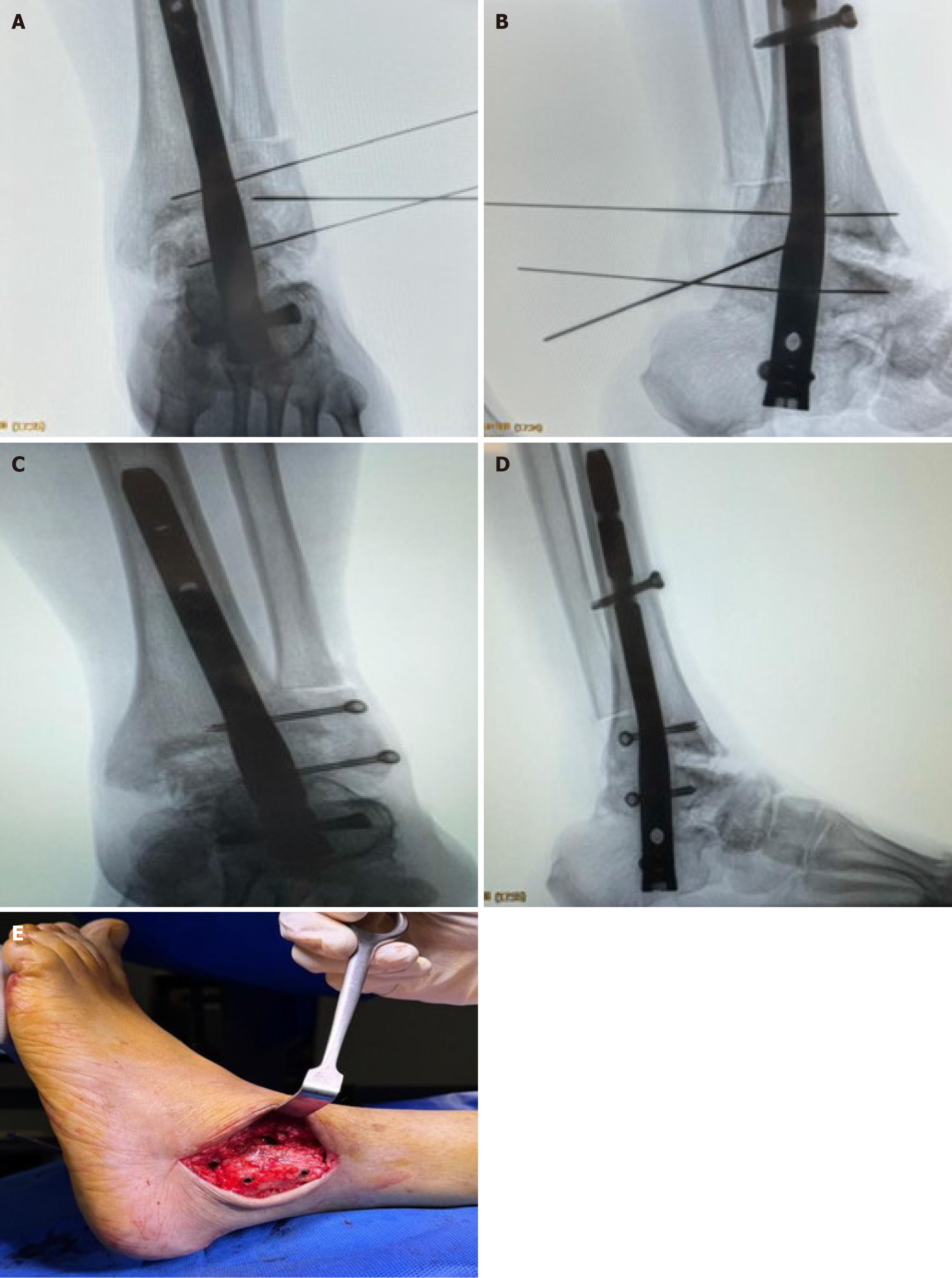
Step 3: Guide wire insertion and retrograde intramedullary nail placement: Under fluoroscopic guidance a guide wire was inserted through the plantar aspect of the calcaneus and advanced across the subtalar and ankle joints into the tibial medullary canal. Following sequential reaming, an appropriately sized retrograde intramedullary nail was inserted and advanced until adequate purchase was achieved in the tibial metaphysis with final positioning confirmed fluoroscopically (Figure 3).
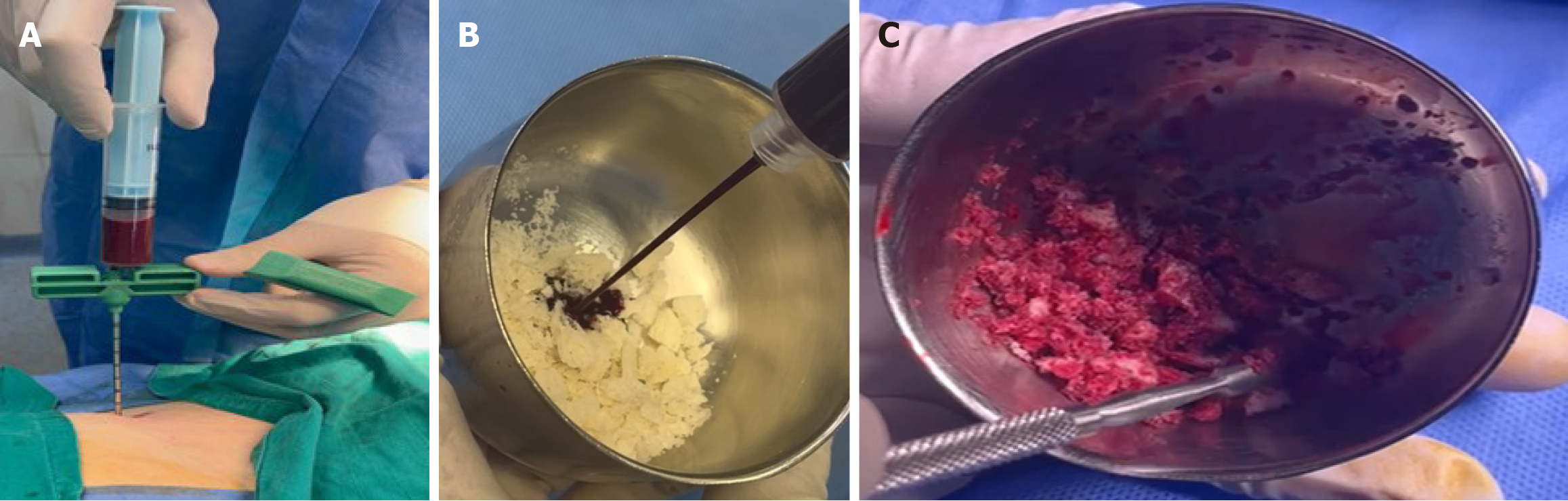
Step 4: Biologic augmentation with BMA and allograft: BMA was performed using a Jamshidi needle inserted into the ipsilateral iliac crest or proximal tibia to harvest autologous BMA concentrate. Processed allograft chips were prepared and subsequently soaked with the concentrated BMA to create a biologically enhanced graft material that provided both osteoconductive scaffolding and osteoinductive properties for enhanced fusion potential (Figure 4).
Step 5: Biologic graft insertion and impaction: The BMA-soaked allograft was carefully inserted into the prepared subtalar and ankle joint spaces, filling all void areas created by debridement. The biologically enhanced graft material was then impacted into the inner aspect of the fibula and all fusion interfaces using bone tamps to ensure optimal contact and integration with the prepared bony surfaces. This integrated biological augmentation strategy provided biological enhancement while preventing post-compression shortening (Figure 5).
Step 6: Fibular reduction and “trap door” fixation: The lateral malleolus was anatomically reduced and temporarily secured with cannulated guide wires under fluoroscopic guidance. Two cortical screws were then inserted in a posteroanterior direction at approximately 18-20 degrees, similar to syndesmotic screw placement but positioned to avoid interference with the intramedullary nail. This innovative “trap door” technique transforms the preserved fibular fragment into a multifunctional construct that provides: (1) Enhanced mechanical stability through tibiofibular compression; (2) Biological augmentation by creating a sealed chamber for the BMA and allograft; and (3) Additional fusion surface area between the fibula, tibia, and talus (Figure 6).
Patients underwent strict non-weight bearing immobilization in below-knee casts for 6 weeks, followed by 6 weeks of partial weight-bearing in range of motion walker boots. Gradual progression to full weight-bearing occurred as tolerated with radiographic confirmation of healing. Protective footwear was maintained for a minimum of 6 months postoperatively.
Clinical evaluation utilized the American Orthopedic Foot and Ankle Society (AOFAS) hindfoot scale, which has been validated as the most widely used outcome measure for hindfoot fusion procedures and demonstrates excellent reliability in assessing pain, function, and alignment parameters in TTC fusion studies[6]. Radiological assessment included serial weight-bearing radiographs and CT scans at 6 months and 12 months to evaluate fusion progression. Radiological union was defined by trabecular bridging across joint interfaces or absence of clearly demarcated joint spaces. All complications were systematically documented.
Continuous variables were summarized as mean ± SD and range and categorical variables as n (%). Preoperative and postoperative AOFAS scores were compared using a paired-samples t test (two-tailed). A P < 0.05 was considered statistically significant. Statistical analyses were performed using SPSS Statistics (IBM Corp., Armonk, NY, United States).
The study cohort comprised 24 patients with a mean age of 57.8 years (range 42-74). There were 14 males (58.3%) and 10 females (41.7%). All patients had diabetes mellitus with a mean glycated hemoglobin of 8.4% (range 6.9%-10.3%). The mean follow-up was 1.5 years (range 0.8-2.2 years).
At the final follow-up successful TTC fusion was achieved in all 24 patients (100%). Fusion was confirmed by robust bony bridging across ankle and subtalar joints on both plain radiographs and CT scans, demonstrating complete osseous consolidation in the entire cohort.
Clinical outcomes demonstrated significant improvements across all measured parameters. Mean AOFAS hindfoot scores improved from 31.2 ± 9.1 preoperatively to 76.8 ± 14.2 at the final follow-up (P < 0.001), representing a mean improve
Complications occurred in 3 patients (12.5%), all of which were managed successfully without compromising the overall treatment outcomes. The first patient developed breakage of both fibular fixation screws at 8 months postoperatively. Clinical and radiographic evaluation revealed that the screws had fractured but fusion was progressing satisfactorily with no evidence of construct instability or fibular displacement. This was managed conservatively with continued protected weight-bearing, and the patient achieved successful fusion without requiring screw removal or revision surgery.
The second patient presented with distal nail protrusion through the plantar heel at 6 months postoperatively, causing discomfort during weight-bearing. Radiographic assessment confirmed solid fusion across both ankle and subtalar joints. The protruding portion of the nail was surgically removed in the operating theater under spinal anesthesia with immediate relief of symptoms and no compromise to the fusion construct.
The third patient sustained a periprosthetic tibial fracture at the proximal extent of the nail at 10 months postoperatively following a minor fall. The fracture was classified as a non-displaced hairline crack without involvement of the fusion site. Management consisted of immediate cessation of weight-bearing and immobilization in a below-knee cast for 8 weeks, followed by progressive weight-bearing in a walker boot. The fracture healed uneventfully with conservative management, and the patient maintained successful TTC fusion.
No major infections, wound healing complications, deep vein thrombosis, or amputations occurred in this series. All patients maintained plantigrade foot positioning with no significant loss of correction at final follow-up.
Mean time to radiographic union was 4.1 months (range 2.8-7.2 months). All patients with successful fusion demon
This case series demonstrated excellent outcomes for a refined transmalleolar TTC fusion technique incorporating biological augmentation and fibular preservation in chronic ankle Charcot arthropathy. The achieved 100% fusion rate represents a substantial improvement over historical reports of conventional techniques, which typically report fusion rates of 50%-70% in similar patient populations[7].
The biological enhancement strategy utilizing a BMA-soaked allograft directly addresses the fundamental healing deficiencies characteristic of Charcot arthropathy. BMA concentrate provides mesenchymal stem cells, growth factors, and osteoprogenitor cells essential for robust osteogenesis[8]. This biological augmentation, combined with comprehensive joint preparation and strategic graft placement, creates an optimal environment for fusion while addressing the compromised healing potential inherent to Charcot bone.
Siddiqui et al[9] investigated a novel approach to enhance arthrodesis rates in Charcot neuroarthropathy by combining distal tibial distraction osteogenesis with TTC or tibiocalcaneal fusion. This technique resulted in a high arthrodesis rate of 93.3% (14 out of 15 patients) with a mean time to fusion of 4.75 months. The authors suggested that distraction osteo
He et al[10] conducted a retrospective study comparing fibular osteotomy, fibular strut, and fibular preservation techniques in TTC arthrodesis using retrograde intramedullary nails. Their findings indicated that the fibular strut group had a significantly shorter fusion time (15.1 ± 2.8 weeks) compared with fibular osteotomy (17.2 ± 1.9 weeks) and fibular preservation (16.8 ± 1.9 weeks). The fibular strut technique also provided superior anti-rotation ability and enhanced stability due to the lateral support of the fibula, suggesting its benefits in accelerating bone healing and improving construct stability. Our technique built upon the principles of fibular preservation but with the added benefits of the “trap door” mechanism, which not only preserves the fibula but actively incorporates it into the fusion mass, creating a contained environment for the biologic graft and enhancing the overall stability of the construct. This differs from simple fibular preservation, which may not provide the same degree of containment or rotational control.
The innovative fibular preservation technique represents a significant departure from traditional approaches that often involve fibular resection. By maintaining the fibula as a viable, vascularized flap and incorporating it into the fusion construct through the “trap door” mechanism, we achieved multiple benefits: Enhanced mechanical stability; additional fusion surface area; and a biological chamber for graft material. This approach also prevented post-compression shor
The complication rate of 12.5% compared favorably with published series of similar complexity. Importantly, all three complications were managed successfully without compromising fusion outcomes or requiring major revision proce
While studies like Love et al[11] have found non-union rates as high as 44% and significant infection rates in patients with high-risk Charcot undergoing tibiocalcaneal arthrodesis, our refined technique demonstrated a substantially lower complication profile. Consistent with findings by Kim et al[12], our study indicates that successful limb salvage through TTC fusion is achievable even in cases with severe deformity, underscoring the paramount importance of infection control and comprehensive surgical planning.
The significant improvement in AOFAS scores with a mean improvement of 45.6 points demonstrated not only statistical significance but also clinically meaningful functional enhancement. This improvement substantially exceeds published minimal clinically important difference values, indicating that patients experienced genuine functional benefit from the procedure.
Recent literature supports the integration of biological and mechanical strategies in challenging orthopedic recon
This study had several inherent limitations that should be acknowledged. The retrospective design, moderate sample size, and absence of a control group limit the strength of the conclusions that can be drawn. The single-surgeon experience ensured technical consistency but may have limited the generalizability of the findings to other surgical practices. Additionally, 1 patient had a follow-up period of only 9 months, which may be insufficient to fully assess long-term fusion durability and functional outcomes. The relatively short overall follow-up period while adequate for as
Future research should focus on prospective, comparative studies with larger cohorts and extended follow-up to validate these findings. Direct comparison with traditional TTC fusion techniques would provide valuable insights into the relative benefits of biological augmentation. Long-term studies examining construct durability and functional preservation would strengthen the evidence base for this approach. Additionally, a cost-effectiveness analysis would be valuable given the additional expense of biological augmentation materials.
The refined transmalleolar TTC fusion technique incorporating biological augmentation with BMA-soaked allograft and fibular preservation demonstrated high fusion rates and favorable clinical outcomes in chronic ankle Charcot arth
The authors thank the surgical and nursing teams who assisted with patient care.
| 1. | Wukich DK, Sung W. Charcot arthropathy of the foot and ankle: modern concepts and management review. J Diabetes Complications. 2009;23:409-426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 143] [Article Influence: 8.4] [Reference Citation Analysis (1)] |
| 2. | Bajuri MY, Ong SL, Das S, Mohamed IN. Charcot Neuroarthropathy: Current Surgical Management and Update. A Systematic Review. Front Surg. 2022;9:820826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 3. | Emara KM, Ahmed Diab R, Amr Hemida M. Tibio-calcaneal fusion by retrograde intramedullary nailing in charcot neuroarthropathy. Foot (Edinb). 2018;34:6-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 4. | Lee JS, Gillinov SM, Siddiq BS, Dowley KS, Martin SD. Surgical Applications for Bone Marrow Aspirate Concentrate. Arthroscopy. 2024;40:2350-2352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 5. | Sherman AE, Mehta MP, Nayak R, Mutawakkil MY, Ko JH, Patel MS, Kadakia AR. Biologic Augmentation of Tibiotalocalcaneal Arthrodesis With Allogeneic Bone Block Is Associated With High Rates of Fusion. Foot Ankle Int. 2022;43:353-362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 6. | Zhang H, Jiang N, Zhao JF, Zeng XT. Effectiveness of intramedullary nails in Tibiotalocalcaneal arthrodesis for Charcot neuroarthropathy: a systematic review and meta-analysis. J Orthop Surg Res. 2025;20:696. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 7. | Shan AK, Binoy S, Biju S, Kumar KA, Shibu R. Tibiotalocalcaneal arthrodesis with retrograde intramedullary nail for Charcot neuroarthropathy of foot - A prospective observational study. J Med Sci Res. 2023;11:131-135. [DOI] [Full Text] |
| 8. | Harford JS, Dekker TJ, Adams SB. Bone Marrow Aspirate Concentrate for Bone Healing in Foot and Ankle Surgery. Foot Ankle Clin. 2016;21:839-845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 9. | Siddiqui NA, Millonig KJ, Mayer BE, Fink JN, McClure PK, Bibbo C. Increased Arthrodesis Rates in Charcot Neuroarthropathy Utilizing Distal Tibial Distraction Osteogenesis Principles. Foot Ankle Spec. 2022;15:394-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 10. | He W, Zhou H, Li Z, Zhao Y, Xia J, Li Y, Chen C, Huang H, Zhang Y, Li B, Yang Y. Comparison of different fibula procedures in tibiotalocalcaneal arthrodesis with a retrograde intramedullary nail: a mid-term retrospective study. BMC Musculoskelet Disord. 2023;24:882. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 11. | Love B, Alexander B, Ray J, Halstrom J, Barranco H, Solar S, Singh M, Shah A. Outcomes of Tibiocalcaneal Arthrodesis in High-Risk Patients: An Institutional Cohort of 18 Patients. Indian J Orthop. 2020;54:14-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 12. | Kim S, Lee HS, Choi Y. Outcomes of Tibiotalocalcaneal Arthrodesis in Hindfoot Charcot Neuroarthropathy According to Coronal-Plane Deformity and Talar Osteolysis. Clin Orthop Surg. 2025;17:331-339. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 13. | Impieri L, Pezzi A, Hadad H, Peretti GM, Mangiavini L, Rossi N. Orthobiologics in delayed union and non-union of adult long bones fractures: A systematic review. Bone Rep. 2024;21:101760. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |