Published online Mar 18, 2026. doi: 10.5312/wjo.v17.i3.113708
Revised: October 19, 2025
Accepted: December 25, 2025
Published online: March 18, 2026
Processing time: 195 Days and 12.3 Hours
According to recent systematic reviews and meta-analyses, the long-term results of total ankle replacement (TAR) are equivalent or superior to ankle arthrodesis with an average 10-year survival approaching 90%. Almost all fourth-generation TAR implants are inserted from the anterior approach except for Zimmer™, which is implanted laterally and with lateral malleolus osteotomy.
To evaluate the clinical outcomes and complication rates and to define a mu
This single-center retrospective cohort study included 85 Zimmer™ lateral-approach TARs performed by one surgeon from 2019 to 2024. Patient-reported outcome measures were evaluated in 65 patients using the Manchester-Oxford Foot Questionnaire (MOXFQ) and European Foot and Ankle Society (EFAS) scores. Underlying diagnoses, functional outcomes, operative time trends, secondary procedures, and complications rates were analyzed to define a learning curve considering the first 60 consecutive cases.
The mean follow-up was 30.8 months. Post-traumatic arthritis was the most common underlying diagnosis accounting for 53.5% of cases. Mean EFAS improved by 13.0 points (P < 0.001), and MOXFQ decreased by 58.5 points (P < 0.001). Quartile analysis showed no significant change in ΔEFAS or ΔMOXFQ (P > 0.4), indicating that proficiency is readily achieved due to the short learning curve of this technique. Operative time decreased from approximately 175 min to 125 min after about 25-30 cases. Complication rates declined with experience. In the first 30 cases, there were 19 reoperations in 13 patients (43%) with an additional 5 being managed conservatively compared with 10 reoperations in 7 patients (23%) with 2 conservatively treated complications in the subsequent 30 cases (relative risk = 1.86, P > 0.05). Infection-related revisions also decreased markedly from 5 (16.7%) to 1 (3.3%) (relative risk = 5.00, P > 0.05) between these cohort. Two cases of deep infection occurred in patients with rheumatoid arthritis. One was successfully managed with debridement, and the other required implant removal with cement spacer implantation.
Lateral-approach TAR achieved early patient-reported outcome measure benefits with efficiency and safety improving with experience, requiring 30 cases. This rapid learning curve integrates functional outcomes, operative timing, and complication rates.
Core Tip: This study provided a multidimensional evaluation of the learning curve in lateral-approach total ankle replacement using the Zimmer™ system. Significant improvements in pain and function were observed as measured by patient-reported outcome measures and remained consistent from the earliest cases, indicating early stabilization of outcomes. Operative time and complication rates improved with surgical experience, plateauing after approximately 25-30 cases. A simultaneous anteromedial approach during the primary procedure may help reduce the need for secondary interventions. Most infection-related complications occurred in patients with rheumatoid arthritis, underscoring the importance of careful preoperative optimization.
- Citation: Liszka H, Gądek A, Zięba AM, Surowiecka K, Kwiatkowski J, Kozioł T, Bochenek MS, Kwolek K. Learning curve of total ankle replacement from the lateral approach. World J Orthop 2026; 17(3): 113708
- URL: https://www.wjgnet.com/2218-5836/full/v17/i3/113708.htm
- DOI: https://dx.doi.org/10.5312/wjo.v17.i3.113708
Arthrodesis and total ankle replacement (TAR) are the two most common surgical options for advanced ankle osteoarthritis when conservative management has failed. Arthrodesis, widely referred to as ankle fusion, has traditionally been the gold standard for end-stage degenerative ankle disease. Although it provides reliable and long-lasting pain relief, the major limitation lies in the negative impact on adjacent joints. By eliminating ankle motion, arthrodesis transfers mechanical stress to neighboring articulations, particularly the subtalar and Chopart joints. Over time this altered load distribution can accelerate degenerative changes, ultimately leading to secondary arthritis and functional impairment[1].
TAR has gained increasing attention as a modern implant design and an advancement in surgical technique. Contemporary prostheses aim to preserve physiological motion at the tibiotalar joint, thereby maintaining more natural biomechanics of the ankle and hindfoot. Unlike arthrodesis, TAR does not impose additional stress on the surrounding joints, reducing the risk of accelerated degenerative changes in the subtalar and Chopart articulations. As a result TAR has become an attractive alternative for select patients with advanced ankle osteoarthritis[2].
The concept of TAR is not new as degenerative and post-traumatic conditions of the ankle joint have long presented a challenge to both patients and physicians. On post-mortem examination Huch et al[3] reported that full-thickness ankle cartilage defects (Collins’ grades III and IV) were present in 6% of young individuals with no established history of joint disease. Another study found degenerative changes in 25% of talocrural joints among individuals aged 30 to 50 years[4]. Despite this prevalence, TAR remained a theoretical option for decades, largely due to the lack of both a defined surgical technique and implant materials capable of withstanding the mechanical stresses experienced by the ankle. Unlike osteoarthritis of the knee and hip, which is often idiopathic or primary in origin, ankle osteoarthritis is predominantly post-traumatic, accounting for more than 75% of cases. Idiopathic arthritis represents less than 9% while secondary causes, including rheumatoid arthritis (RA) and hemophilia, account for approximately 13%[5-7].
Early attempts at TAR dating back to the 1970s were undermined by high complication rates and poor implant longevity, prolonging arthrodesis as the treatment of choice for decades[8]. However, with substantial improvements in implant design and surgical technique, TAR has gradually re-emerged as a viable and effective treatment option. Today, fourth-generation implants with a fixed-bearing design have largely replaced the third-generation mobile-bearing systems[9,10]. Recent publications, including systematic reviews and meta-analyses, suggest that the long-term results of TAR are comparable or superior to those of ankle arthrodesis with reported average 10-year survival rates ranging from 80% to 90%[9-11]. Most TAR systems use an anterior approach, whereas the Zimmer™ implant is unique in being implanted laterally.
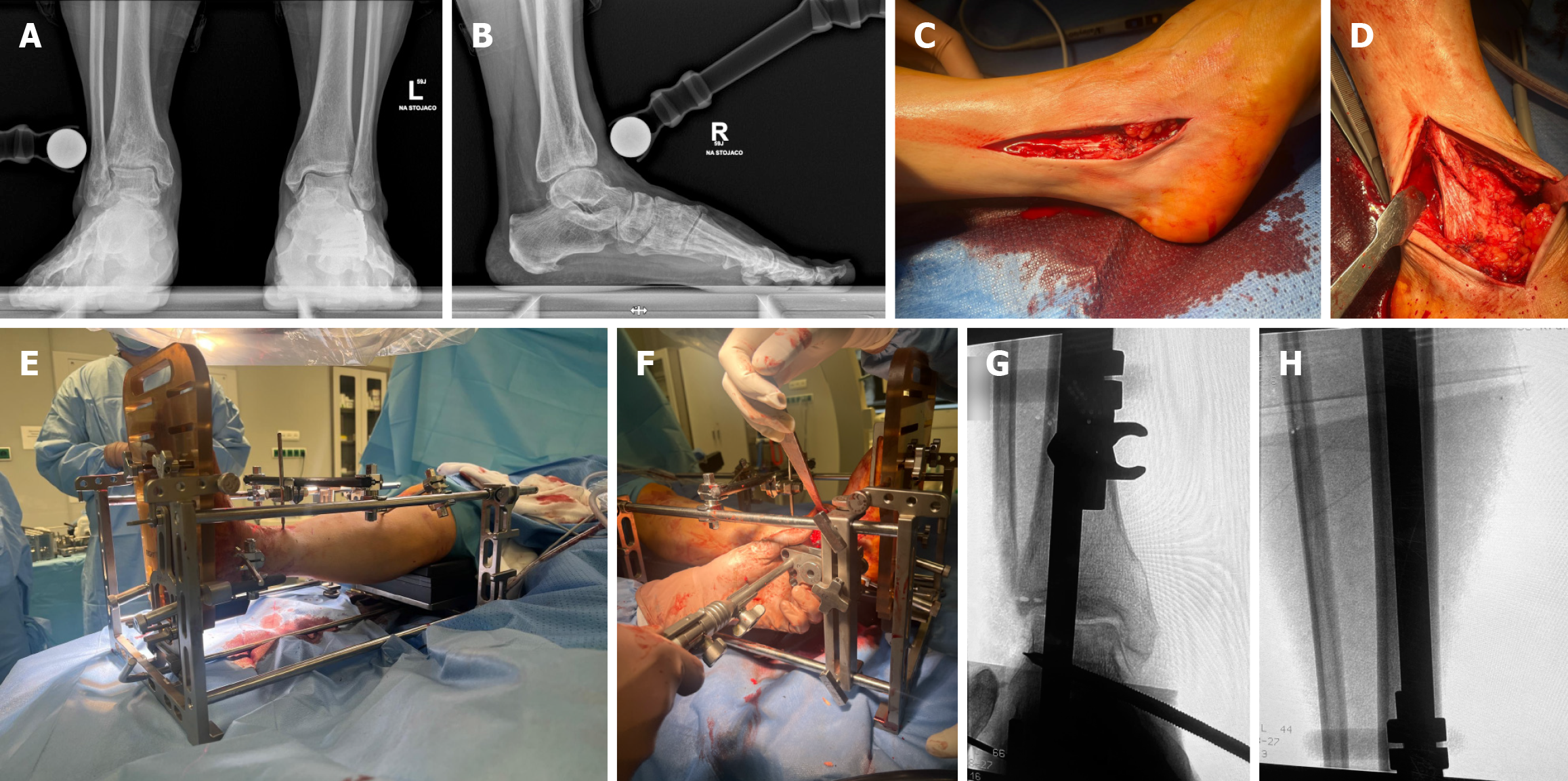
This investigation was designed as a retrospective cohort study. A total of 85 patients underwent TAR via the lateral approach at the Department of Orthopedics and Traumatology from 2019 to 2024. Of these, 65 patients were included in the final analysis. Overall, 10 patients did not complete the final follow-up, 2 died from unrelated causes, 3 could not be contacted, and 5 were excluded due to a short follow-up period at the time of writing this manuscript (< 12 months). All surgeries were performed by the first author using the lateral approach (Figure 1).
Inclusion criteria were symptomatic severe ankle arthritis (grade IV according to the Kellgren and Lawrence radiological scale). Additional indications for surgery included advanced degenerative changes affecting multiple joints of the foot and ankle. Exclusion criteria included: (1) Ankle joint infection; (2) Insufficient soft tissue coverage at the surgical site; (3) Advanced ischemic foot disease; (4) Charcot neuroarthropathy and other neurogenic conditions; (5) Age < 30 years; (6) Strenuous manual labor; (7) Extreme obesity (body mass index ≥ 40 kg/m2); (8) Poorly controlled diabetes mellitus (hemoglobin A1c > 7.0%); (9) Nicotine dependence; and (10) Follow-up shorter than 12 months.
It is important to note that some studies have reported favorable outcomes of TAR even in patients presenting with certain contraindications, such as obesity[12]. Similarly, in our cohort, no significant correlation was found between body mass index and postoperative functional outcomes [European Foot and Ankle Society (EFAS), Manchester-Oxford Foot Questionnaire (MOXFQ); all P > 0.05], indicating that excess body weight did not adversely affect recovery or functional outcomes.
Prior to surgery all patients underwent bilateral weight-bearing anteroposterior and lateral radiographs of the feet and ankle joints (Figure 1A and B). Implant sizing was performed both preoperatively and intraoperatively. Surgical planning was conducted using a dedicated marker and MediCAD software. In patients with prior ankle surgery, all retained fixation hardware, such as plates, screws, or other metallic implants, was removed either during the preoperative period or intraoperatively. Any additional surgical interventions and complications were carefully recorded. The study protocol was approved by the Institutional Ethics Committee (approval No. 118.0043.1.515.2024).
The operative limb was disinfected in a standard sterile manner and draped slightly below the knee. Patients were positioned supine on a radiolucent table with the operated extremity resting on a firm support to facilitate stable frame fixation. Tourniquets were not used routinely. Surgery commenced with a 10-12 cm lateral incision beginning along the distal fibula and extending distally toward the sinus tarsi (Figure 1C). After the anterolateral recess of the ankle was reached, osteophytes from the anterior aspect of the joint were excised. An oblique fibular osteotomy was then performed with special care being given to preserve the integrity of the syndesmosis (Figure 1D). The lateral malleolus was mobilized anteriorly with a broad osteotome and temporarily stabilized to the calcaneus with Kirschner wires.
When posterior capsular contracture or osteophytic impingement was identified, management included capsular release, osteophyte excision, and when indicated Achilles tendon lengthening. In some cases after excision of anterior osteophytes, the tibia was shifted forward to address talar subluxation. For patients with marked anteromedial osteophytes, an accessory anteromedial approach was used to clear the tibiotalar joint.
Neutral foot positioning was achieved, and Schanz pins were introduced into the calcaneus, talus, and tibia to secure the extremity within the external frame (Figure 1E and F). Pins in the talus and calcaneus enabled correction in the coronal plane while the tibial pin allowed for sagittal alignment adjustments (Figure 1G and H). Their precise placement was crucial as the pins functioned as a jig during valgus/varus and rotational corrections. Once alignment and balance were satisfactory, the tibial pins were definitively locked into the frame. Fluoroscopic guidance was employed at every stage.
Implant size was then assessed, and the joint center of rotation was identified with frame-mounted instruments. Both the tibial and talar articular surfaces were reamed spherically. Following insertion of trial components and confirmation of balanced motion, the definitive tibial and talar components were implanted. A fixed-bearing polyethylene insert was secured within the tibial component.
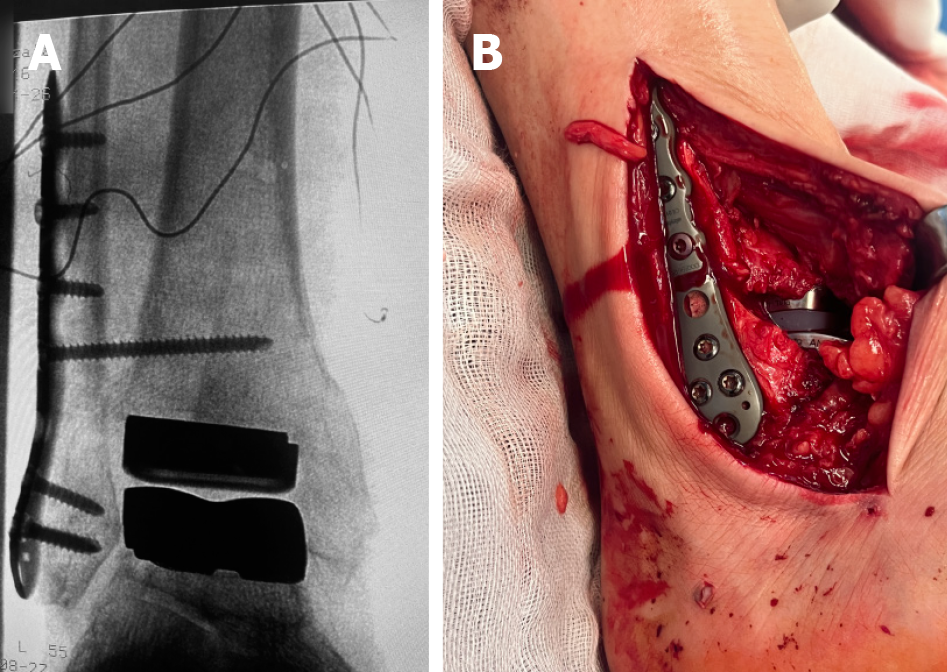
After dismantling the frame the fibula was repositioned and stabilized with either three screws or a plate (Figure 2A). If syndesmotic laxity was present, an additional trans-syndesmotic screw was introduced. In cases requiring enhanced lateral stability, ligament repair was carried out using a modified Broström-Duquennoy technique. When necessary Achilles tendon lengthening was performed using the Hoke technique. Medial gutter debridement and deltoid ligament release were performed in select cases while combined deltoid ligament reconstruction was undertaken in one case. Posterior release was also performed in cases of limited dorsiflexion, which was a relatively frequent finding. One patient underwent a medial displacement calcaneal osteotomy combined with anterotalofibular ligament repair while 3 patients required subtalar or double arthrodesis. Removal of prior fixation material was performed in selected cases. The distribution of these secondary, concomitant procedures is summarized in Table 1. Final intraoperative radiographs were obtained to confirm implant positioning and alignment (Figure 2B). Wound closure was performed in layers-capsule, subcutaneous tissue, and skin. Finally, a sterile dressing was applied, and the limb was immobilized in a below-knee plaster cast.
| Procedure | Early phase (cases 1-33) | Late phase (cases 34-65) | Total (n) | Percentage of total (n = 65) |
| Modified Broström/ATFL repair | 6 | 11 | 17 | 26.2 |
| ATFL + DL repair | 0 | 1 | 1 | 1.5 |
| Subtalar distraction arthrodesis | 1 | 0 | 1 | 1.5 |
| Double/triple arthrodesis (hindfoot) | 1 | 0 | 1 | 1.5 |
| Medial displacement calcaneal osteotomy + ATFL repair | 0 | 1 | 1 | 1.5 |
| Percutaneous Achilles tendon lengthening (Hoke) | 0 | 4 | 4 | 6.2 |
| Deltoid ligament release | 1 | 0 | 1 | 1.5 |
| Medial (anteromedial) gutter debridement | 6 | 4 | 10 | 15.4 |
| Removal of previous fixation (fibular plate/screws) | 1 | 3 | 4 | 6.2 |
| Internal fixation of intraoperative medial malleolar fracture | 0 | 1 | 1 | 1.5 |
| Total number of concomitant procedures | 16/33 (48.5%) | 26/32 (81.3%) | 42 | 64.6 |
Postoperatively, patients remained non-weight-bearing in the initial cast for 2-3 weeks after which a synthetic cast was applied and maintained until week 6. Partial weight-bearing was typically initiated around week 4 and gradually progressed to full weight-bearing by week 6; however, weight-bearing was delayed until week 6 in cases with additional procedures such as medial displacement calcaneal osteotomy or double/triple fusion. The postoperative care protocol was individualized on a patient-by-patient basis, depending on the condition of the surgical site and radiographic findings at 6 weeks and 12 weeks. No standardized physiotherapy or rehabilitation program was applied.
This study retrospectively analyzed patients who underwent TAR via the lateral approach. Data regarding the procedure, indications, demographics, and clinical parameters (sex, age, operated limb, and time elapsed since surgery) were extracted from the hospital electronic medical records database. Following the TAR procedures, patients were contacted by telephone to evaluate their perceived pain levels before and after surgery using two validated questionnaires: The MOXFQ and the EFAS[13].
The MOXFQ is a validated instrument designed to assess the impact of foot disorders on quality of life. It consists of 16 items divided into three domains: Walking/standing; pain; and social interaction. Responses are rated on a five-point Likert scale ranging from 0 (never) to 4 (always). The total score is calculated by summing the item responses and converting the result to a 0-100 scale with higher scores indicating greater impairment.
The EFAS score is another patient-reported outcome measure validated for foot and ankle conditions. It comprises six questions, each scored on a Likert scale from 0 to 4 with higher scores corresponding to better function. The total EFAS score ranges from 0 to 24 with higher values reflecting improved functionality. An additional subsection consisting of four sport-specific questions was excluded from the analysis due to the low response rate. Complete data from 65 patients were included in the final analysis. The characteristics of the study cohort are summarized in Table 2.
| Variable | Category | Number of patients (n = 65) | Percentage |
| Operated leg | Left | 36 | 55.4 |
| Right | 29 | 44.6 | |
| Cause | Post-traumatic | 35 | 53.8 |
| Chronic osteoarthritis | 27 | 41.5 | |
| Rheumatoid arthritis | 3 | 4.6 | |
| BMI (kg/m2) | mean ± SD (range) | 29.4 ± 4.0 (20.8-35.6) | |
| ≥ 25 (overweight + obese) | 51/59 | 86.4 | |
| ≥ 30 (obese) | 27/59 | 45.8 | |
| Diabetes mellitus/prediabetes | Diabetes mellitus (insulin) | 2 | 3.1 |
| Diabetes mellitus (oral therapy) | 1 | 1.5 | |
| Prediabetes | 1 | 1.5 | |
| No | 61 | 93.9 | |
| Smoking status | Occasional smoker | 7 | 10.8 |
| Former smoker | 5 | 7.7 | |
| Never smoker | 53 | 81.5 |
Comprehensive analysis: Patient-reported outcome measures (PROMs) were assessed using the MOXFQ and the EFAS score in 65 patients (Tables 3, 4, 5, and 6). In this group operative time, additional procedures, and postoperative complications were also evaluated to provide an integrated overview of clinical and technical performance (Tables 1, 7, and 8).
| Cause of total ankle replacement procedure | Score before procedure | SD (before) | Score after procedure | SD (after) | Difference before and after surgery (P value) |
| Post-traumatic | 4.42 | 4.00 | 17.35 | 2.46 | 12.93 (< 0.001) |
| Chronic osteoarthritis | 2.27 | 3.04 | 14.23 | 5.83 | 11.96 (< 0.001) |
| Rheumatoid arthritis | 7.67 | 3.79 | 20.33 | 2.08 | 12.66 (0.100) |
| Cause of total ankle replacement procedure | Score before procedure | SD (before) | Score after procedure | SD (after) | Difference before and after surgery (P value) |
| Post-traumatic | 77.23 | 15.42 | 25.31 | 19.15 | 51.92 (< 0.001) |
| Chronic osteoarthritis | 79.58 | 12.41 | 35.38 | 31.02 | 44.20 (< 0.001) |
| Rheumatoid arthritis | 71.33 | 10.97 | 9.00 | 1.73 | 62.33 (0.077) |
| Total | 78.02 | 13.78 | 21.57 | 25.76 | 58.50 (< 0.001) |
| Parameter | Pre-operative (mean ± SD) | Post-operative (mean ± SD) | Δ (mean ± SD) | Median | Range |
| EFAS score | 3.6 ± 2.9 | 17.2 ± 3.1 | + 13.0 ± 4.6 | 14 | -2 to 20 |
| MOXFQ score | 82.5 ± 10.7 | 21.3 ± 22.3 | + 58.5 ± 21.8 | 64 | -6 to 86 |
| Quartile (case order) | n | mean ΔEFAS ± SD | mean ΔMOXFQ ± SD | Interpretation |
| Q1 (cases 1-16) | 16 | 14.6 ± 2.5 | 63.8 ± 20.8 | Stable early improvement |
| Q2 (cases 17-32) | 16 | 12.7 ± 5.3 | 60.9 ± 24.7 | Slight variability, consistent trend |
| Q3 (cases 33-48) | 16 | 13.5 ± 5.2 | 57.1 ± 25.9 | Plateau maintained |
| Q4 (cases 49-65) | 17 | 13.8 ± 5.5 | 62.7 ± 18.2 | Sustained functional gain |
| Case range | Mean operative time (minute) | SD (minute) |
| 1-10 | 175.5 | 25.8 |
| 11-20 | 158.5 | 39.7 |
| 21-30 | 136.0 | 22.4 |
| 31-40 | 122.5 | 16.8 |
| 41-50 | 118.1 | 19.5 |
| 75-85 | 109.6 | 22.4 |
| Type of complication/reoperation | Early phase (cases 1-30) | Late phase (cases 31-60) |
| Operative (reoperations) | ||
| Infection-related procedures (washout, liner exchange, explant/spacer, sinus/peroneal tendon revisions) | 5 | 1 |
| Lateral malleolus stabilization/nonunion revision | 3 | 0 |
| Tarsal tunnel release | 1 | 2 |
| Total talus replacement | 1 | 0 |
| Medial/Lateral recess release ± syndesmosis screw removal/ATFL repair | 8 | 6 |
| Hardware removal (isolated) | 1 | 1 |
| Calcaneal osteotomy (MDCO) | 0 | 1 |
| Total operative procedures (patients) | 19 (13) | 10 (7) |
| Conservatively managed complications | ||
| Superficial infection (occasional smoker) | 1 | 0 |
| Lateral malleolus fixation instability (treated nonoperatively) | 1 | 0 |
| Syndesmotic screw loosening/destabilization | 2 | 1 |
| Equinus contracture (treated with physiotherapy) | 1 | 0 |
| Intraoperative medial malleolar fracture without fixation | 1 | 0 |
| Intraoperative medial malleolar fracture with screw fixation with delayed union | 0 | 1 |
| Total conservative complications (patients) | 6 (6) | 2 (2) |
Functional learning curve analysis: This cohort was analyzed to assess functional trends along the learning curve. The magnitude of improvement (ΔEFAS and ΔMOXFQ) was compared across four chronological quartiles (Q1-Q4; 16-17 cases each) to determine whether surgeon experience influenced patient-reported outcomes (Tables 5 and 6).
Technical and safety learning curve analysis: To assess how increasing surgical experience influenced operative efficiency and perioperative safety, we analyzed the initial 60 consecutive TAR procedures. Operative time stabilization served as the basis for defining two experience phases: Early (cases 1-30) and late (cases 31-60). Complications were categorized as: (1) Reoperations: Any unplanned surgical revision, including washout, liner exchange, implant removal or spacer placement, sinus tarsi or fibular revision, lateral malleolus restabilization or nonunion revision, tarsal tunnel release, medial or lateral recess or posterior capsule release with or without syndesmosis screw removal or anterotalofibular ligament repair, and medial displacement calcaneal osteotomy (Table 8); and (2) Conservatively managed complications: Wound-healing problems, superficial infections (one occurred in an occasional smoker during the early phase), postoperative contracture, and one intraoperative medial malleolar fracture in the late phase that achieved delayed union with conservative treatment (Table 8). Infections were classified as superficial or deep according to the consensus diagnostic criteria proposed by the European Bone and Joint Infection Society and the International Consensus Meeting (2018) on musculoskeletal infection[14,15].
Statistical analyses were performed using R software (version 4.2.2) within the RStudio environment (version 2023.6.0.421). Descriptive statistics were calculated for all variables and expressed as mean ± SD or median (range). The Kruskal-Wallis test and Spearman’s rank correlation coefficient (ρ) were applied to assess functional trends (ΔEFAS, ΔMOXFQ) along the learning curve. The Mann-Whitney U test was used for comparisons between two independent groups, and Fisher’s exact test for categorical variables, including complication rates. Results were reported as risk ratios (RR) with 95% confidence intervals. Statistical significance was set at P < 0.05.
The median time following TAR for all patients was 30.77 months. Secondary, concomitant procedures performed during primary TAR included modified Broström ligament repair in 19 cases (29.2%), medial gutter debridement in 10 cases, percutaneous Achilles tendon lengthening (Hoke technique) in 4 cases, medial displacement calcaneal osteotomy in 1 case, and subtalar or double arthrodesis in 3 cases. Additional procedures included removal of previous fixation material in 4 cases, deltoid ligament release in 1 case, and combined deltoid ligament repair in 1 case. The overall distribution of these procedures is summarized in Table 1. Across the entire group the mean EFAS score improved significantly by 13.0 points postoperatively (P < 0.001) while the MOXFQ score decreased significantly by 58.5 points (P < 0.001). Patients were divided into three subgroups based on the direct cause of the TAR procedure: Post-traumatic [post-traumatic osteoarthritis (PTOA)] (n = 35); chronic osteoarthritis (n = 27); and RA (n = 3). The chronic osteoarthritis group had the lowest preoperative EFAS score (2.27) compared with the post-traumatic group (4.42) and the RA group (7.67). After surgery the post-traumatic group showed the greatest improvement in the EFAS score: An increase of 12.93 points compared with 12.66 in the RA group and 11.96 in the osteoarthritis group (Table 3). Patients in the osteoarthritis group had the highest preoperative MOXFQ score (79.58 points) compared with the PTOA group (77.23) and the RA group (71.33). However, the largest postoperative decrease in MOXFQ score was observed in the RA group (62.33 points) (Table 4).
The quartile-based analysis (Q1-Q4; 16-17 cases each) showed stable and consistently high functional gains (ΔEFAS: 12.7-14.6; ΔMOXFQ: 57.1-63.8; both P > 0.4) with no correlation between functional improvement and case order (Spearman’s ρ = 0.09 for ΔEFAS, ρ = 0.12 for ΔMOXFQ; both P > 0.4) (Tables 5 and 6).
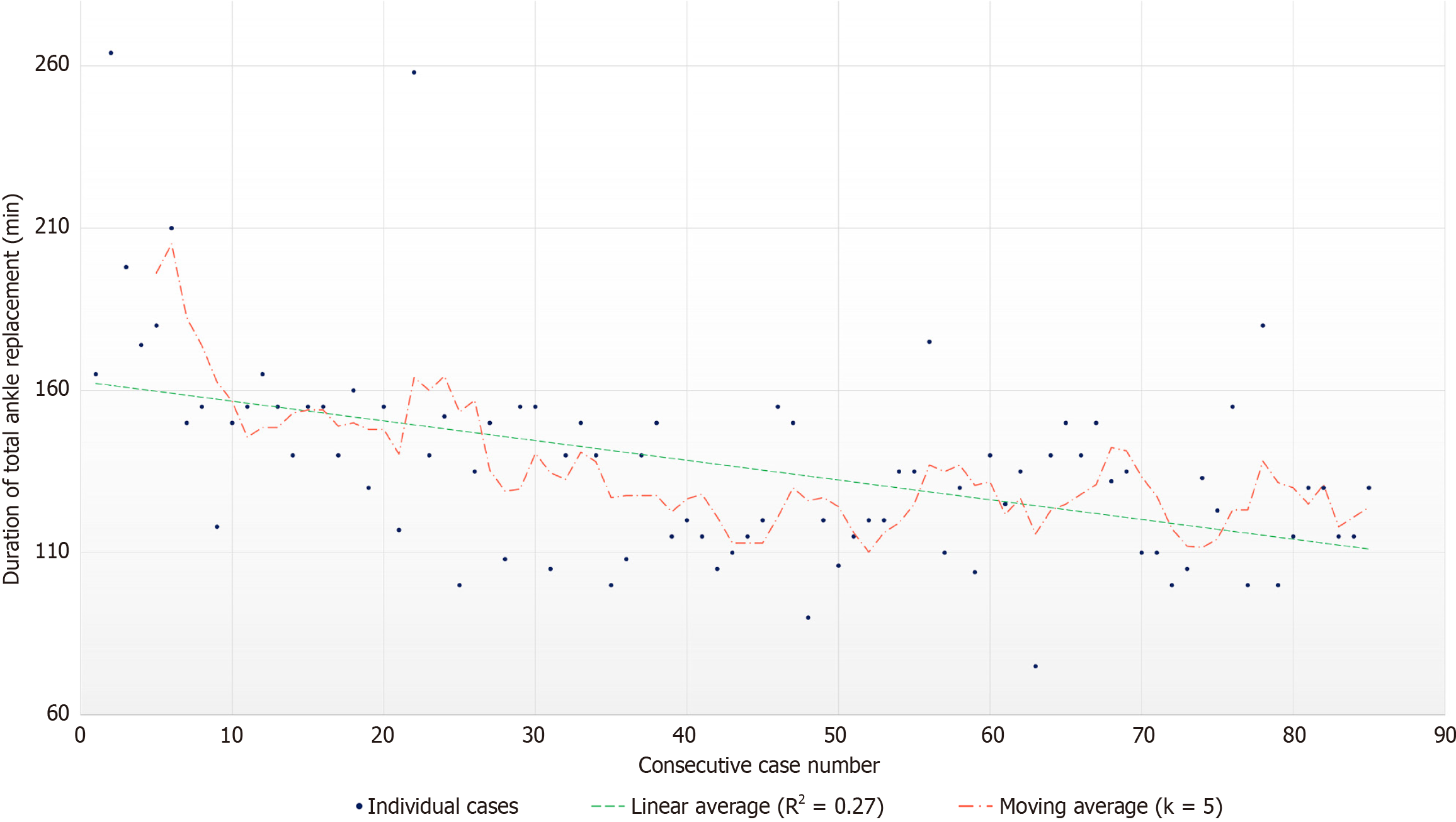
Operative time decreased from approximately 175 min to 125 min after 25-30 procedures, demonstrating a clear technical learning curve (Figure 3, Table 7). A statistically significant reduction in mean operative time was observed when comparing the first 25 cases (cases 1-25) to the subsequent 25 cases (cases 26-50) (160.6 ± 37.6 min vs 126.3 ± 21.0 min; P < 0.0001). In the most recent cases (75-85), the mean operative time further decreased to 109.6 ± 22.4 min, suggesting sustained improvement and consolidation of the surgical technique (Figure 3).
A total of 29 reoperations were performed in 20 patients within the first 60 procedures (Table 8). Reoperations were more frequent in the early phase (19 in 13 patients, 43%) than in the late phase (10 in 7 patients, 23%) (RR = 1.86, 95% confidence interval: 0.83-4.18; P > 0.05). Infection-related revisions declined from five (16.7%) to one (3.3%) (RR = 5.00, P > 0.05). Conservatively treated complications were infrequent: 5 in the early phase and 2 in the late phase. Complications included mild fixation instability, screw loosening, superficial infection, equinus contracture, and delayed union of a medial malleolar fracture. The most common indication for reoperation was anteromedial recess debridement for symptomatic impingement.
Of 3 patients (4.6%) who developed deep infections, 1 patient with RA developed lateral wound dehiscence with purulent peroneal tendovaginitis and a draining fistula 2 months after resuming biologic therapy. Imaging confirmed implant osseointegration without loosening, and revision surgery with lateral hardware removal, tendon debridement, and lavage achieved full recovery with implant retention. Another patient with RA developed early deep infection 1 week after restarting biologic therapy, resulting in septic loosening of the prosthesis that required implant removal with antibiotic-loaded spacer placement. Both patients had controlled systemic inflammation at the time of surgery, with biologic therapy paused preoperatively and resumed postoperatively. The third case, a patient without RA, presented with a sinus tract over the peroneal tendons and was successfully managed by washout, debridement and sinus excision. Two intraoperative medial malleolar fractures occurred, both of which healed successfully.
This study confirmed that TAR performed through a lateral approach provides significant pain relief and functional improvement in patients with advanced ankle arthritis. EFAS and MOXFQ scores improved significantly postoperatively, indicating meaningful functional and quality-of-life benefits. These findings are consistent with previous reports demonstrating the high clinical efficacy of TAR and its advantage over ankle arthrodesis in preserving joint motion[16-18].
Patients with post-traumatic and primary osteoarthritis achieved comparable functional benefits, whereas those with RA experienced the greatest pain reduction as reflected by the largest postoperative decrease in MOXFQ scores. This subgroup was small in our population, and any statistical conclusions herein offer limited external validity However, this trend aligns with the results of van der Heide et al[19], who demonstrated that TAR can substantially improve pain and function in inflammatory arthritis.
RA comprised 5% of the study population with PTOA and primary osteoarthritis representing 54% and 41%, respectively. This distribution differs from earlier registry data, such as the Swedish Ankle Register in which RA represented 36% of TAR indications[11]. Despite favorable functional outcomes these results should be interpreted with caution. RA remains associated with an increased risk of surgical site and infection-related complications following TAR[20], likely due to systemic immunosuppression and compromised soft tissue quality. In contrast, a recent meta-analysis that compared outcomes in 5508 patients who underwent TAR found that 1565 patients with inflammatory arthritis (including RA) and 3943 with non-inflammatory etiologies had no significant differences in functional outcomes, complication rates, or implant survival between the groups[21]. These data together with our findings support the view that TAR can provide comparable functional benefits across diverse etiologies when performed in appropriately selected patients.
EFAS and MOXFQ score changes did not differ by sex, indicating that TAR outcomes and functional recovery are comparable between male and female patients. However, implant component sizes (tibial and talar) were consistently larger in males, reflecting anatomical rather than functional variation. In addition, the use of computer-assisted surgery and navigation systems has been shown to further improve the accuracy of implant positioning[22].
In many cases TAR must be complemented by additional procedures aimed at correcting biomechanical misalignment or coexisting pathology. The most common procedures include ligament reconstruction, Achilles tendon lengthening, and corrective osteotomies to address varus or valgus deformities. Criswell et al[23] reported that 28% of patients undergoing TAR required such additional procedures (excluding percutaneous Achilles tendon lengthening), involving extra incisions but did not significantly increase the rate of short-term complications. The need for adjunctive procedures depends primarily on the underlying pathology and the patient’s anatomy. Frequently, patients requiring such interventions present with more advanced deformities and a lower baseline. Peri et al[24] found that additional procedures are associated with slightly prolonged hospital stays (1.76 days vs 1.52 days in isolated TAR cases) although they do not significantly increase the risk of adverse events.
As with any surgical procedure, TAR carries the risk of complications. A systematic review by Zunarelli et al[25] identified a 3.8% incidence of periprosthetic infection. Nerve injuries may occur in up to 15% of patients although most are transient[26]. Aseptic loosening (the failure of the implant to remain stable in the absence of infection) is among the most serious long-term complications following TAR. Implant design may also play a role in the risk of loosening.
Current evidence suggests that complication rates and revision risk after TAR decrease once the surgeon has performed a sufficient number of procedures. A recent systematic review of learning curves in TAR by Arshad et al[27] reported that complication and revision rates as well as operative efficiency improved markedly after approximately 9 to 39 cases, reflecting a critical threshold beyond which outcomes stabilize. Our findings support these observations, showing a clear learning curve with a significant reduction in operative time and complication rates after approximately 25-30 cases (Figure 3). The early optimization of patient-reported outcomes (EFAS and MOXFQ) indicates that satisfactory functional results were achieved from the outset, likely reflecting the surgeon’s prior experience in foot and ankle reconstruction and understanding of the biomechanical objectives of the procedure. Although technical efficiency and perioperative safety continued to improve with experience, functional outcomes remained consistently high throughout the quartiles, suggesting that patient-reported outcome satisfaction plateaued earlier than technical proficiency. This may also be partly explained by the inclusion of less complex cases in the initial phase.
This observed multidimensional pattern underscores a comprehensive learning curve concept, incorporating operative efficiency, safety, and patient-reported outcomes with the predominant improvements realized during the initial 25-30 procedures. Notably, four of the five infection-related revisions in the early phase occurred in patients with RA receiving biologic therapy. These infections likely reflected the immunosuppressive nature and timing of biologic therapy rather than surgical factors as patients had controlled systemic inflammation at the time of surgery. This observation highlights the importance of optimizing inflammatory status and coordinating biologic management in collaboration with rheumatologists to minimize postoperative infection risk.
Despite promising results this study has several limitations. This study was limited by its retrospective, single-surgeon design, relatively small cohort size, and lack of a control group, restricting direct comparative conclusions (e.g., with anterior-approach TAR). In addition, radiographic outcome measures beyond weightbearing CT in complicated cases were not available, and PROM assessment relied on telephone-based surveys, which may introduce recall bias. Moreover, recent literature indicates that patient-related factors, such as prior hindfoot fusion[28] or complex underlying pathology (e.g., RA), may significantly influence outcomes and revision risk following TAR. These variables were not analyzed in our series, but we acknowledge their importance and believe that future multicenter prospective studies with larger and more heterogeneous populations should incorporate them to provide a more complete understanding of TAR outcomes.
This study confirmed that TAR is an effective treatment option for patients with advanced ankle joint pathology. At a median follow-up of 30.8 months, a statistically significant improvement was observed in both PROMs: The EFAS score increased by 13.0 points (P < 0.001); and the MOXFQ score decreased by 58.5 points (P < 0.001). Subgroup analysis demonstrated that patients with PTOA achieved the greatest functional improvement (EFAS) while those with RA reported the largest reduction in pain (MOXFQ). The significant postoperative reduction in pain and improvement in function as reflected by EFAS and MOXFQ scores suggest that TAR potentially offers advantages over ankle arthrodesis. By preserving joint mobility TAR may reduce the risk of overload and secondary degenerative changes in adjacent joints of the kinetic chain, making it a favorable alternative in select patients. Operative efficiency and perioperative safety improved progressively with experience with complication rates decreasing after approximately 25-30 procedures. Most infection-related events occurred in patients with RA receiving biologic therapy, emphasizing the importance of careful preoperative optimization in this subgroup.
The lateral approach remains a reliable and reproducible technique when performed by experienced surgeons. In selected cases with significant medial impingement or gutter pathology, a simultaneous anteromedial approach may be considered during the primary procedure to minimize the risk of secondary revision. Longer-term follow-up is warranted to assess implant survival and the robustness of clinical outcomes.
| 1. | Krause FG, Schmid T. Ankle arthrodesis versus total ankle replacement: how do I decide? Foot Ankle Clin. 2012;17:529-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 36] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 2. | Valderrabano V, Hintermann B, Nigg BM, Stefanyshyn D, Stergiou P. Kinematic changes after fusion and total replacement of the ankle: part 2: Movement transfer. Foot Ankle Int. 2003;24:888-896. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 82] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 3. | Huch K, Kuettner KE, Dieppe P. Osteoarthritis in ankle and knee joints. Semin Arthritis Rheum. 1997;26:667-674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 123] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 4. | Cengiz M, Ozturk S, Arican RY, Bacanli CC, Gurer IE, Gurer G, Tuncer T, Sindel T, Sindel M. The Localizations of Osteoarthritis in the Knee, Ankle and Foot Joints of Cadaver: Comparison in Radiological, Morphological and Histopathological Aspects. Eur J Ther. 2022;28:270-278. [DOI] [Full Text] |
| 5. | Maia CR, Annichino RF, de Azevedo E Souza Munhoz M, Machado EG, Marchi E, Castano-Betancourt MC. Post-traumatic osteoarthritis: the worst associated injuries and differences in patients' profile when compared with primary osteoarthritis. BMC Musculoskelet Disord. 2023;24:568. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 23] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 6. | Herrera-Pérez M, González-Martín D, Vallejo-Márquez M, Godoy-Santos AL, Valderrabano V, Tejero S. Ankle Osteoarthritis Aetiology. J Clin Med. 2021;10:4489. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 7. | Saltzman CL, Salamon ML, Blanchard GM, Huff T, Hayes A, Buckwalter JA, Amendola A. Epidemiology of ankle arthritis: report of a consecutive series of 639 patients from a tertiary orthopaedic center. Iowa Orthop J. 2005;25:44-46. [PubMed] |
| 8. | Bonasia DE, Dettoni F, Femino JE, Phisitkul P, Germano M, Amendola A. Total ankle replacement: why, when and how? Iowa Orthop J. 2010;30:119-130. [PubMed] |
| 9. | Kvarda P, Susdorf R, Horn-Lang T, Peterhans U, Ruiz R, Hintermann B. Long-Term Survival of Hintegra Revision Ankle Arthroplasty in 116 Patients. Foot Ankle Orthop. 2022;7:2473011421S00737. [DOI] [Full Text] |
| 10. | Zaidi R, Cro S, Gurusamy K, Siva N, Macgregor A, Henricson A, Goldberg A. The outcome of total ankle replacement: a systematic review and meta-analysis. Bone Joint J. 2013;95-B:1500-1507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 267] [Cited by in RCA: 231] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 11. | Henricson A, Nilsson JÅ, Carlsson A. 10-year survival of total ankle arthroplasties: a report on 780 cases from the Swedish Ankle Register. Acta Orthop. 2011;82:655-659. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 165] [Cited by in RCA: 135] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 12. | Barg A, Knupp M, Anderson AE, Hintermann B. Total ankle replacement in obese patients: component stability, weight change, and functional outcome in 118 consecutive patients. Foot Ankle Int. 2011;32:925-932. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 59] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 13. | Richter M, Agren PH, Besse JL, Cöster M, Kofoed H, Maffulli N, Rosenbaum D, Steultjens M, Alvarez F, Boszczyk A, Buedts K, Guelfi M, Liszka H, Louwerens JW, Repo JP, Samaila E, Stephens M, Witteveen AGH. EFAS Score - Multilingual development and validation of a patient-reported outcome measure (PROM) by the score committee of the European Foot and Ankle Society (EFAS). Foot Ankle Surg. 2018;24:185-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 92] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 14. | McNally M, Sousa R, Wouthuyzen-Bakker M, Chen AF, Soriano A, Vogely HC, Clauss M, Higuera CA, Trebše R. The EBJIS definition of periprosthetic joint infection. Bone Joint J. 2021;103-B:18-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 658] [Cited by in RCA: 569] [Article Influence: 113.8] [Reference Citation Analysis (4)] |
| 15. | Parvizi J, Gehrke T, Chen AF. Proceedings of the International Consensus on Periprosthetic Joint Infection. Bone Joint J. 2013;95-B:1450-1452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 891] [Cited by in RCA: 763] [Article Influence: 58.7] [Reference Citation Analysis (1)] |
| 16. | Haddad SL, Coetzee JC, Estok R, Fahrbach K, Banel D, Nalysnyk L. Intermediate and long-term outcomes of total ankle arthroplasty and ankle arthrodesis. A systematic review of the literature. J Bone Joint Surg Am. 2007;89:1899-1905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 205] [Cited by in RCA: 255] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 17. | Daniels TR, Younger AS, Penner M, Wing K, Dryden PJ, Wong H, Glazebrook M. Intermediate-term results of total ankle replacement and ankle arthrodesis: a COFAS multicenter study. J Bone Joint Surg Am. 2014;96:135-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 278] [Cited by in RCA: 228] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 18. | Saltzman CL, Kadoko RG, Suh JS. Treatment of isolated ankle osteoarthritis with arthrodesis or the total ankle replacement: a comparison of early outcomes. Clin Orthop Surg. 2010;2:1-7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 137] [Cited by in RCA: 117] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 19. | van der Heide HJ, Schutte B, Louwerens JW, van den Hoogen FH, Malefijt MC. Total ankle prostheses in rheumatoid arthropathy: Outcome in 52 patients followed for 1-9 years. Acta Orthop. 2009;80:440-444. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 44] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 20. | Robert SK, Chryssa N, Jun ML, Hisham S, John M. Comparison of patient reported outcomes using the Manchester-Oxford Foot Questionnaire and surgical complications following total ankle replacement in rheumatoid arthritis versus osteoarthritis. J Foot Ankle Surg. 2025;S1067-2516(25)00192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 21. | Mousavian A, Baradaran A, Schon LC, Daniel J, Pedowitz D, Kachooei AR. Total Ankle Replacement Outcome in Patients With Inflammatory Versus Noninflammatory Arthritis: A Systematic Review and Meta-analysis. Foot Ankle Spec. 2023;16:314-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 22. | Stauffer TP, Kim BI, Grant C, Adams SB, Anastasio AT. Robotic Technology in Foot and Ankle Surgery: A Comprehensive Review. Sensors (Basel). 2023;23:686. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 23. | Criswell B, Hunt K, Kim T, Chou L, Haskell A. Association of Short-term Complications With Procedures Through Separate Incisions During Total Ankle Replacement. Foot Ankle Int. 2016;37:1060-1064. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 24. | Peri MI, Whitaker S, Cole S, Anastasio A, Satalich JR, O'Neill CN, Patel TT, Nunley JA, Easley ME, Schweitzer KM. Additional Procedures at the Time of Total Ankle Replacement Do Not Increase Risk of Short-term Complications: A Matched Cohort Analysis. Foot Ankle Orthop. 2024;9:24730114241268150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 25. | Zunarelli R, Fiore M, Lonardo G, Pace A, Persiani V, De Paolis M, Sambri A. Total Ankle Replacement Infections: A Systematic Review of the Literature. J Clin Med. 2023;12:7711. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 26. | Primadi A, Xu HX, Yoon TR, Ryu JH, Lee KB. Neurologic injuries after primary total ankle arthroplasty: prevalence and effect on outcomes. J Foot Ankle Res. 2015;8:55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 27] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 27. | Arshad Z, Haq II, Bhatia M. Learning curve of total ankle arthroplasty: a systematic review. Arch Orthop Trauma Surg. 2024;144:591-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 28. | Teehan E, Boden AL, Jones A, Henry J, Demetracopoulos C. Patient-Reported Outcomes and Revision Risk Following Total Ankle Replacement With and Without Ipsilateral Hindfoot Fusion. Foot Ankle Int. 2025;46:845-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |