Published online Apr 24, 2026. doi: 10.5306/wjco.v17.i4.117705
Revised: January 5, 2026
Accepted: February 11, 2026
Published online: April 24, 2026
Processing time: 128 Days and 20.6 Hours
Breast cancer is increasingly recognized as a systemic disease influenced by the metabolic environment. Growing evidence suggests that obesity, type 2 diabetes mellitus, metabolic syndrome, and associated cardiovascular diseases signifi
Core Tip: Reframes metabolic comorbidities as active, modifiable drivers of breast cancer. Integrates epidemiology, tumour microenvironment, signalling and gut microbiota. Maps obesity, type 2 diabetes mellitus and metabolic syndrome pathways to pharmacologic and lifestyle interventions. Highlights glucagon-like peptide-1 receptor agonists, sodium-glucose cotransporter 2 inhibitors and bariatric surgery as precision tools. Proposes a metabolic oncology model with artificial intelligence-enabled multimodal risk prediction.
- Citation: Zhang SH, Yang Y, Zhang Y. Breast cancer and metabolic comorbidities: From epidemiology and molecular mechanisms to precision interventions. World J Clin Oncol 2026; 17(4): 117705
- URL: https://www.wjgnet.com/2218-4333/full/v17/i4/117705.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i4.117705
Breast cancer remains the most prevalent malignancy among women globally, with its incidence and mortality continuing to present significant public health challenges[1-3]. The global rise in metabolic disorders, such as obesity, type 2 diabetes mellitus (T2DM), and metabolic syndrome (MetS), has revealed complex and profound associations between these chronic noncommunicable diseases and breast cancer initiation, progression, treatment response, and prognosis[4-6]. This relationship extends beyond coincidental co-occurrence, driven by shared biological pathways and intricate interaction networks. Obesity, particularly central obesity, has been firmly established as a major risk factor for various cancers, including postmenopausal breast cancer[7-9]. T2DM and its precursor, insulin resistance, contribute to a tumor-promoting microenvironment through mechanisms such as hyperinsulinemia, hyperglycemia, and chronic inflammation[10,11]. The aggregation of these risk factors within MetS amplifies the detrimental impact on breast cancer, significantly increasing recurrence and mortality risks[12].
The connection between breast cancer and metabolic comorbidities extends across the entire care continuum, influencing diagnosis, therapeutic decision-making, and long-term survivorship. Obesity and diabetes may necessitate treatment adjustments, reduce therapeutic efficacy, and increase the risk of postoperative complications and therapy-related toxicities, such as cardiotoxicity[13,14]. Additionally, these comorbidities are strongly linked to an elevated risk of second primary malignancies and reduced overall survival (OS)[15,16]. Consequently, understanding the epidemiological correlations, biological intersections, and clinical implications of the interplay between breast cancer and metabolic comorbidities is essential for optimizing prevention, treatment, and long-term management.
Recent advances in molecular biology, multi-omics, and artificial intelligence have shifted our understanding from broad epidemiological patterns to specific mechanistic insights. Notably, adipose tissue remodeling and alterations in critical signaling pathways, such as phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT)/mammalian target of rapamycin (mTOR) and interleukin (IL)-6/Janus kinase/signal transducer and activator of transcription 3 (STAT3), have been identified as central to linking metabolic imbalance to breast cancer progression[17-21]. These findings have sparked renewed interest in repurposing metabolic agents [e.g., metformin, glucagon-like peptide-1 (GLP-1) receptor agonists (RAs)] and innovating lifestyle interventions as precision strategies[22-24].
This article synthesizes recent advancements to explore the multidimensional connections between breast cancer and major metabolic comorbidities, including obesity, diabetes, MetS, and related cardiovascular diseases (CVD). It begins by mapping the epidemiology and risk relationships, then examines the convergent mechanisms from systemic to cellular and molecular levels, evaluates the clinical impact on management and prognosis, and concludes with precision interventions and integrated care models to inform the development of a “metabo-oncology” paradigm.
Epidemiological research consistently supports associations between clusters of metabolic disorders - such as obesity, diabetes, MetS, and CVD - and breast cancer outcomes.
Obesity, a major global health challenge, is strongly linked to breast cancer, with the association particularly pronounced in postmenopausal women, who experience higher rates of both breast cancer incidence and mortality[7,9,25,26]. In a Brazilian cohort, eliminating overweight and obesity was estimated to prevent approximately 14.3% of breast cancer deaths among women aged ≥ 50 years[27]. This relationship spans racial and ethnic groups but varies in magnitude; for instance, obesity is a significant risk factor in Pakistani and African women[28,29]. In high-risk populations, such as African American women, obesity is notably associated with early-onset breast cancer and more aggressive triple-negative breast cancer (TNBC)[30,31].
In addition to static adiposity, adult weight changes represent a critical risk factor. A weight gain of > 5 kg during adulthood - particularly if it occurs before age 50 - correlates with a significant increase in breast cancer risk[32,33]. Among breast cancer survivors, post-diagnosis weight gain is common and associated with poorer outcomes[34], along with increased long-term risks of CVD and other complications[35]. In postmenopausal hormone receptor-positive early breast cancer treated with aromatase inhibitors, several studies suggest that obesity is linked to a higher recurrence risk, possibly due to diminished endocrine treatment efficacy; however, differences in population characteristics and methodology require careful interpretation[26]. Sarcopenic obesity, characterized by excess fat combined with reduced muscle mass, is another important metabolic comorbidity in breast cancer patients. Unlike typical obesity, sarcopenic obesity is associated with poorer treatment tolerance, increased chemotherapy-related toxicities, and worse overall prognosis[36]. Risk assessment has become more nuanced. While body mass index (BMI) is commonly used, it inadequately captures fat distribution. Central adiposity - specifically abdominal fat accumulation - appears to be a more robust predictor. Measures such as waist circumference (WC) and waist-to-hip ratio indicate that central obesity is independently associated with increased breast cancer risk, regardless of menopausal status[37,38]. Increases in visceral adipose tissue and abdominal subcutaneous adipose tissue are linked to elevated postmenopausal breast cancer risk, with higher visceral adipose tissue/subcutaneous adipose tissue ratios further enhancing this risk[39,40]. Emerging indices like the weight-adjusted waist index show potential as risk predictors, although evidence regarding their independent predictive value is still developing[41,42].
Obesity complicates treatment outcomes. Patients with higher BMI are less likely to achieve pathologic complete response (pCR) after neoadjuvant chemotherapy (NACT)[43], although some multivariable analyses report contradictory findings[44] High BMI is also associated with more frequent chemotherapy dose reductions and delays[13], as well as increased risks of breast cancer-related lymphedema (BCRL) and reconstructive complications[45-47]. From suboptimal screening adherence[48] to altered treatment choices and recovery, obesity adversely impacts nearly every stage of the care continuum.
T2DM and its precursor states, such as prediabetes and insulin resistance, are strongly associated with breast cancer. A large meta-analysis revealed that diabetes increases both the incidence and mortality of breast cancer by approximately 20%[10], with stronger associations observed in some Asian populations[10]. Epidemiologic studies further establish diabetes as an independent risk factor for several cancers, including breast cancer, with a particularly elevated risk shortly after diabetes diagnosis[49]. Among breast cancer survivors, pre-existing or newly diagnosed diabetes is linked to poorer outcomes, higher risks of second primary cancers (SPCs) (e.g., liver, brain), and increased all-cause mortality[16,50,51].
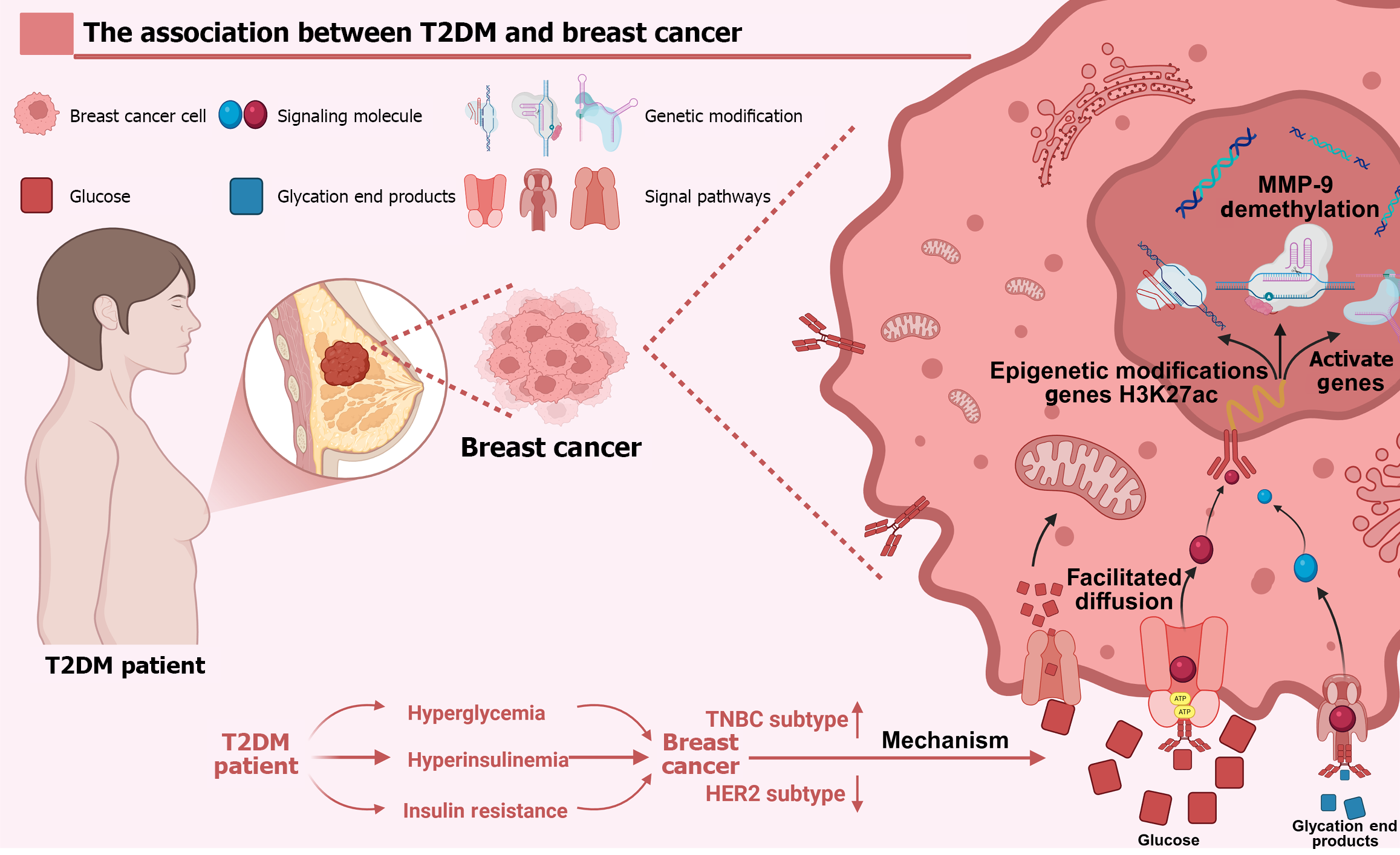
From a mechanistic perspective, hyperglycemia, hyperinsulinemia, and insulin resistance are key pathophysiological pathways connecting T2DM to breast cancer[6,11]. A study utilizing multiple causal-inference methods suggested a direct causal relationship between T2DM and breast cancer[52]. Hyperglycemia not only provides metabolic “fuel” but also promotes tumor progression through gene activation (e.g., fibrinogen C domain containing 1) and epigenetic alterations (e.g., histone H3 lysine 27 acetylation)[52,53]. Advanced glycation end-products (AGEs), central mediators of diabetic complications, can further facilitate metastasis through epigenetic modifications, such as the demethylation of the matrix metalloproteinase-9 (MMP-9) promoter[54]. These mechanistic links between chronic dysglycemia and more aggressive disease are summarised schematically in Figure 1.
In clinical practice, T2DM influences both pathology and treatment management. Women with T2DM are more likely to present with TNBC and less frequently have human epidermal growth factor receptor 2 (HER2)-positive disease, reflecting an epidemiological inverse association rather than an active suppression of HER2 expression. Women with T2DM are more likely to present with TNBC and less likely to have HER2-positive disease[55]. Glycemic control is crucial, as elevated fasting blood glucose correlates with higher risks of TNBC and high-grade tumors[55]. Diabetes also increases the risk of postoperative complications[14,50]. Treatment decisions are often affected by diabetes; patients with diabetes are less likely to receive chemotherapy at certain stages and among specific ethnic groups[56]. Additionally, newer targeted therapies, such as the PI3K inhibitor alpelisib and the AKT inhibitor capivasertib, can induce metabolic disturbances, including hyperglycemia, posing unique safety challenges for diabetic patients[57,58]. For gestational diabetes mellitus, the association with breast cancer risk varies by region. A meta-analysis found no overall association, but indicated a reduced risk in North America and an increased risk in Asia[59], emphasizing the role of genetic, environmental, and lifestyle factors in modifying these associations. In conclusion, dysglycemia is a critical comorbidity throughout breast cancer care, influencing risk assessment, tumor phenotype, treatment decisions, and prognosis.
MetS encompasses a cluster of metabolic abnormalities - central obesity, hypertension, hyperglycemia (or insulin resistance), and dyslipidemia[12] - which collectively represent the interaction of multiple pathophysiological processes, posing a greater risk than any individual component. Epidemiological studies and meta-analyses consistently show that MetS and its components are positively correlated with breast cancer risk, particularly among postmenopausal women[60]. Cumulative metabolic disturbances contribute to tumorigenesis through mechanisms such as insulin resistance, chronic inflammation, and oxidative stress. In patient cohorts, MetS is prevalent and closely associated with poorer clinical outcomes.
Extensive evidence indicates that MetS is an independent adverse prognostic factor. A systematic review and meta-analysis involving over 40000 survivors revealed significantly higher risks of recurrence, breast cancer-specific mortality, and all-cause mortality among those diagnosed with MetS[12,40]. MetS is linked to shorter disease-free survival and OS[61]. The effects of MetS may differ by molecular subtype; its detrimental impact on recurrence and breast cancer-specific mortality appears most pronounced in HER2-overexpressing cases[62]. Among patients undergoing NACT, MetS is associated with lower pCR rates, further emphasizing its negative influence on treatment response[61,63].
The individual components of MetS - central obesity, hypertension, hypertriglyceridemia, and low high-density lipoprotein cholesterol - collectively create a tumor-promoting environment. Central obesity and hyperglycemia, as core features, drive cancer progression through chronic inflammation, hyperinsulinemia, and hormonal dysregulation. AGEs, resulting from hyperglycemia and oxidative stress, are proposed as potential biomarkers linking MetS to breast cancer risk[64]. In TNBC, microRNA-877-5p may serve as a molecular link between MetS and tumor development[65]. Gut dysbiosis is another common pathological connection, with specific microbial shifts observed in both MetS and breast cancer[66].
For survivors, the long-term effects of MetS present significant challenges. Among women with breast cancer, the presence of three or more MetS components substantially increases the risk of developing SPCs, such as endometrial cancer[67]. Therefore, systematic screening and management of MetS and its components are crucial, not only for improving primary breast cancer outcomes but also for preventing other hormone-related cancers. A holistic approach to treating MetS is essential in addressing the comorbidity burden.
CVD is the leading non-cancer cause of death among breast cancer survivors[68,69]. The relationship between breast cancer and CVD is bidirectional, with shared risk factors such as obesity, diabetes, poor lifestyle, and chronic inflammation. Furthermore, cancer treatments can induce or exacerbate cardiac injury[70]. Hypertension, the most common CVD comorbidity, is especially prevalent among patients with breast cancer, often co-occurring with other cardiovascular risks[71-73].
Pre-existing CVD risks complicate both diagnosis and prognosis. For instance, older women residing in greener urban areas have a lower likelihood of CVD comorbidity at the time of breast cancer diagnosis, suggesting that environmental factors influence cardio-oncological health[74]. Mendelian randomization studies indicate a genetic correlation between breast cancer and heart failure, pointing to shared susceptibility or pathways, particularly involving inflammatory signaling[75].
The treatment period represents a critical window for cardiovascular events, a field known as “cardio-oncology”. Certain cancer therapies, including anthracyclines, anti-HER2 agents (e.g., trastuzumab), and CDK4/6 inhibitors, can precipitate cancer therapy-related cardiac dysfunction (CTRCD), leading to declines in left ventricular ejection fraction and heart failure[71,76]. Hypertension severity and higher baseline cardiovascular comorbidity index (CCI), along with prolonged chemotherapy, independently increase the risk of CTRCD[71,77]. CDK4/6 inhibitors are linked to elevated hypertension rates and major adverse cardiovascular events (MACE), with distinct drug-specific risk profiles[78]. These toxicities contribute to increased healthcare costs, reduced quality of life, and diminished survival[78].
In cardio-oncology, the risk is not solely the result of an additive effect of pre-existing cardiovascular conditions and cancer treatments[79]. The “multiple-hit hypothesis” suggests that metabolic dysfunction, such as obesity and insulin resistance, increases the baseline vulnerability of heart cells, making them more susceptible to the cardiotoxic effects of cancer therapies like chemotherapy and radiation. This heightened baseline fragility of cardiomyocytes amplifies the risk of CTRCD, underscoring the importance of metabolic optimization before and during treatment[80].
Long-term cardiometabolic management in survivors remains a challenge. In Australian data, the use of cardiovascular medications (e.g., RAS inhibitors, statins, β-blockers) increases over time, alongside rising polypharmacy, reflecting the growing burden of CVD comorbidity[81]. In obese survivors, weight gain of more than 10% after diagnosis further heightens the risks of myocardial infarction and ischemic stroke[35]. A comprehensive baseline risk assessment, vigilant monitoring during treatment, and sustained post-treatment follow-up are essential, particularly for older patients and those with pre-existing cardiovascular risks[73,82].
The epidemiology of breast cancer and its associated metabolic comorbidities is significantly shaped by social, environmental, and lifestyle factors. These determinants influence cancer risk both directly and indirectly by impacting metabolic health, thereby affecting susceptibility and prognosis. Socioeconomic status (SES), race/ethnicity, residential envi
Racial and social disparities play a central role. Compared to non-Hispanic White women, African American women have higher obesity rates, with stronger associations between obesity, early-onset breast cancer, and TNBC[31,83]. Hypertension is also more prevalent among African American survivors[84,85]. These disparities are rooted in social determinants such as lower SES, greater exposure to adverse life events, and systemic inequities in healthcare access[86-89]. Poverty is particularly linked to an increased risk of SPCs[87]. In certain populations, such as American Indians and Alaska Natives, comorbidity indices are higher compared to White patients, compounded by fragmented healthcare systems that complicate care coordination[90]. The environment plays a pivotal role as well. Exposure to urban green spaces has been associated with lower cardiovascular comorbidity, particularly among older women at breast cancer diagnosis, with effects varying by race and SES[74]. Greater access to green spaces is linked to lower all-cause and breast cancer-specific mortality in survivors[68]. In contrast, “food deserts” and poor food environments elevate the risks of obesity and hypertension, especially in underserved communities[91]. Lifestyle factors serve as the intersection between environment and individual health. Prolonged sedentary behavior is positively correlated with breast cancer prevalence among women with obesity, whereas regular physical activity can mitigate this risk[92]. However, adherence to dietary and exercise guidelines remains suboptimal among survivors, limiting long-term outcomes[93]. Diet patterns also play a critical role: Fruit-rich diets are associated with a lower genetic risk of breast cancer[94], while diets high in ultra-processed foods contribute to overweight and abdominal obesity[95]. Smoking and alcohol use further exacerbate the comorbidity burden[96,97]. These interactions collectively determine an individual’s position on the breast cancer-metabolic comorbidity spectrum. Low physical activity amplifies the negative effects of sedentary behavior in obesity[92], while lower SES and certain racial/ethnic groups are linked to reduced screening participation[98,99] and higher odds of non-guideline-directed de-escalation of treatment[100]. To address these inequities, effective strategies must integrate social, environmental, and lifestyle factors.
The strong epidemiological connection between breast cancer and metabolic comorbidities reflects a profound mechanistic convergence. From organ-level interactions to cellular signaling and gene regulation, metabolic dysregulations remodel the tumor microenvironment (TME), disrupt key signaling pathways, alter gut microbiota, and induce epigenetic reprogramming, thereby driving tumorigenesis, progression, and resistance.
The TME acts as the “soil” that supports tumor cells, and metabolic states such as obesity significantly reshape it to promote tumorigenesis. Peritumoral adipose tissue plays a central role in this process[101]. In obesity, adipocytes undergo hypertrophy, hypoxia, and cell death, initiating chronic low-grade inflammation characterized by abundant cytokines, chemokines, and adipokines - including leptin, IL-6, and tumor necrosis factor (TNF)-α[4,30] - which directly stimulate proliferation, invasion, and angiogenesis in breast cancer cells[102].
Beyond changes in the secretome, adipocytes transform into cancer-associated adipocytes, which release fatty acids as fuel and pro-invasive factors[103]. Obesity also alters the extracellular matrix composition by increasing collagen deposition, stiffening tissue, and creating “highways” for cellular migration[104]. Collagen fibers within the extracellular matrix can undergo linearization, a process driven by tissue stiffness, which transduces mechanical signals via pathways such as es-associated protein/transcriptional coactivator with PDZ-binding motif, further enhancing cell migration and invasion[105]. Immune dysregulation is another hallmark of the obesity-conditioned TME, where antitumor immunity is suppressed. This includes impaired CD8+ T-cell function and an enrichment of M2 tumor-associated macrophages and myeloid-derived suppressor cells (MDSCs)[19,103,106]. High-fat diet-induced changes in gut microbiota increase systemic leucine levels, which promote the differentiation of polymorphonuclear neutrophils-MDSCs via mTOR complex 1 (mTORC1) signaling, further suppressing antitumor immunity[19]. Notably, weight loss has been shown to reverse this immunosuppression and restore antitumor signaling[106]. Interestingly, loss of myeloid triggering receptor expressed on myeloid cells 2 inhibits tumor growth in lean, but not obese, mice, highlighting the context-specific nature of immune regulation in the TME[107]. Obesity also induces DNA double-strand breaks in the mammary epithelium, which may represent an early oncogenic event. This effect is more pronounced in African American women and correlates with higher levels of leptin and insulin-like growth factor (IGF)-1[108]. Additionally, lymphangiogenesis and lymphatic dysfunction in obese mammary tissue may further facilitate metastasis[109].
In adipose tissue, androgens are converted to estrogens through a process called peripheral aromatization, which plays a major role in linking obesity to estrogen receptor-positive breast cancer. Obesity causes inflammation in the adipose tissue, which increases the production of aromatase and local estrogen, contributing to the growth and spread of tumors[110].
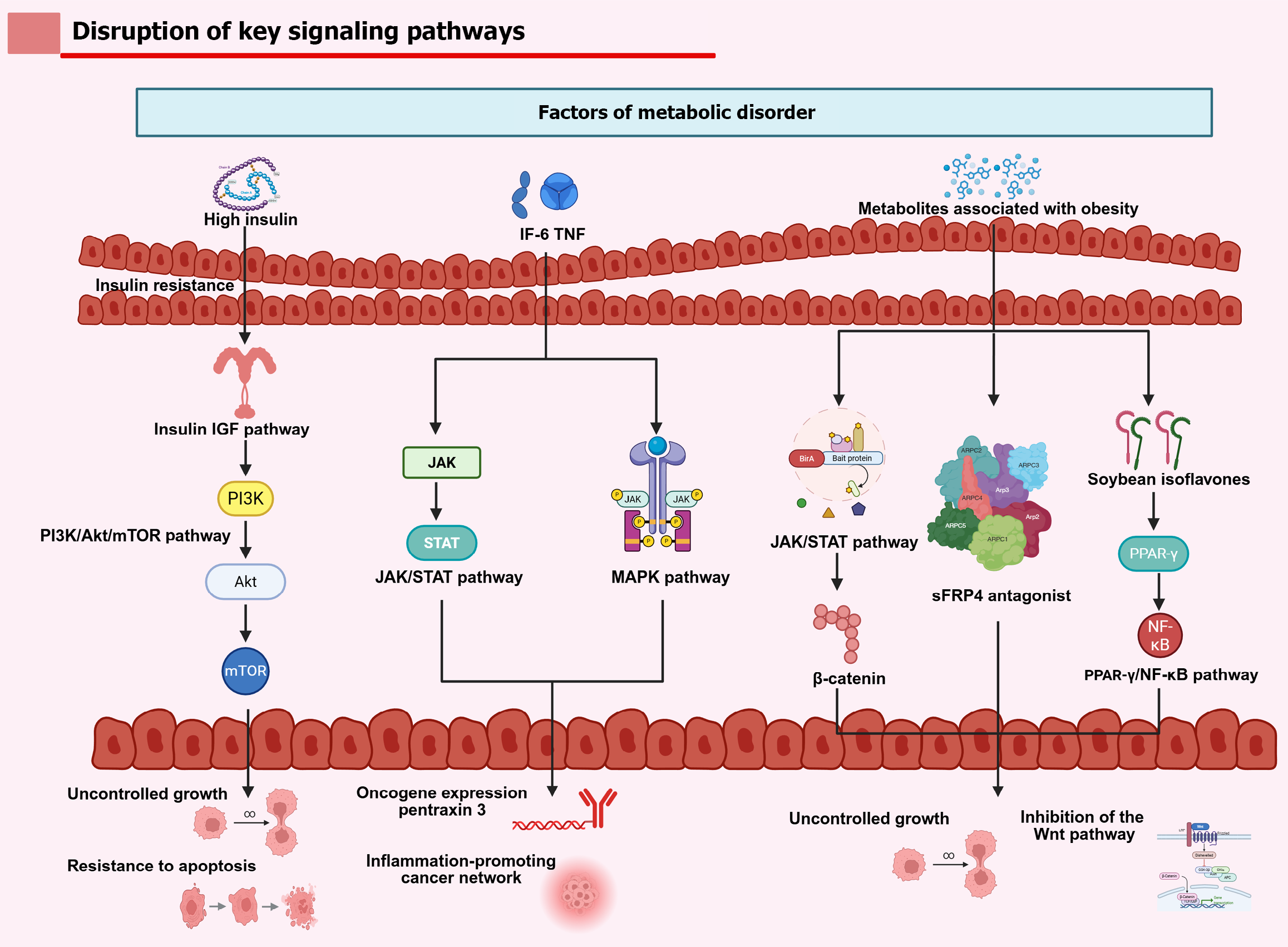
Systemic metabolic imbalances, including hyperinsulinemia, hyperleptinemia, and proinflammatory cytokines, drive oncogenic programs through interconnected signaling networks. The insulin/IGF and PI3K/AKT/mTOR pathways serve as central hubs, with chronic activation in obesity and T2DM promoting uncontrolled cell growth and resistance to apoptosis[111]. IGF binding protein 6, a specific inhibitor of IGF-II, is associated with a favorable prognosis - especially in nondiabetic patients - highlighting the clinical significance of IGF signaling in metabolically driven cancers[6,17,111]. The proinflammatory IL-6/Janus kinase/STAT3 pathway upregulates genes that promote invasion and metastasis, such as pentraxin-3[21,112]. The TNF and mitogen-activated protein kinase pathways cooperate within a broader proinflammatory network[113].
Wnt signaling is commonly dysregulated in cancer. Secreted frizzled-related protein 4 (sFRP4), a natural antagonist of Wnt signaling, links metabolic disease with cancer progression. Pharmacologic upregulation of sFRP4 (e.g., using silibinin or isotretinoin) inhibits breast cancer cell growth, suggesting a potential therapeutic strategy for metabolically associated cancers[114]. In obesity-driven models, soy isoflavones like formononetin can activate peroxisome proliferator-activated receptor gamma, degrade nuclear nuclear factor kappa B, and block Wnt3a/β-catenin signaling to suppress invasion[103]. Additionally, the tissue renin-angiotensin-aldosterone system contributes to the pathophysiology of obesity, T2DM, and breast cancer[115]. AGEs interact with their receptor, receptor for advanced glycation end products, to induce MMP-9 expression and promote metastasis[54]. Obesity-related metabolites, such as glutathione, may directly activate mTORC1 via scavenger receptor class B member 2 at lysosomes, linking metabolic signals to growth regulation[18]. The integrated crosstalk among insulin/IGF, inflammatory and Wnt/β-catenin pathways within this metabolically altered milieu is depicted in Figure 2.
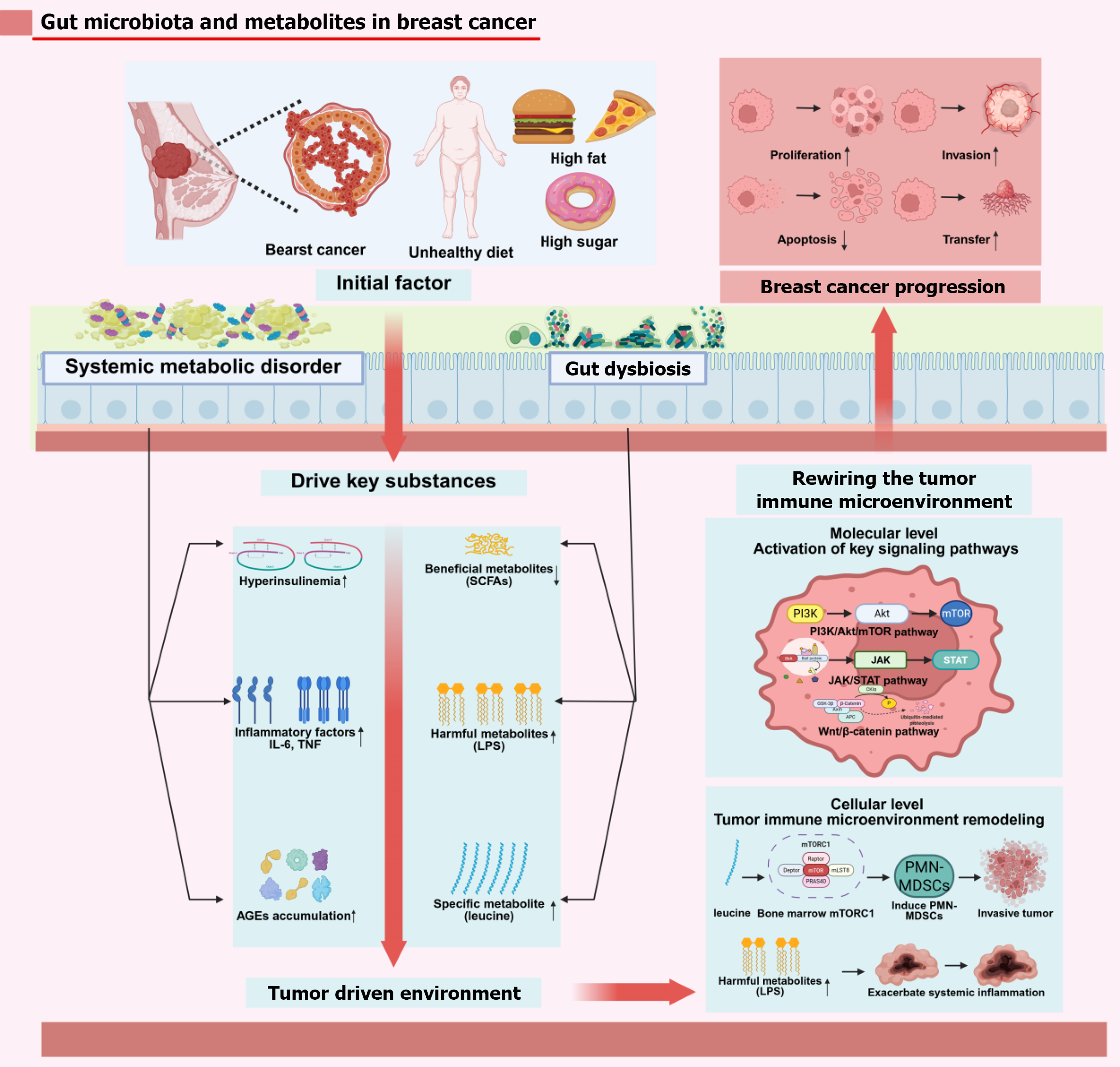
The gut microbiome, often referred to as the “second genome”, is increasingly recognized as a critical mediator linking metabolic health and cancer risk. Dysbiosis is associated with MetS, obesity, diabetes, and several cancers, including breast cancer[1,66,116]. Patients with breast cancer, particularly those with MetS, exhibit reduced abundances of beneficial bacteria such as Bifidobacterium and Lactobacillus, compared to controls, while Streptococcus levels remain unchanged[66]. The microbiota influences systemic immunity, inflammation, and hormone metabolism (e.g., enterohepatic estrogen cycling), thereby affecting risk in hormone-sensitive diseases[1].
Microbial metabolites exert wide-reaching effects on the body. Short-chain fatty acids - butyrate, propionate, and acetate - regulate energy balance, insulin sensitivity, and inflammation[1]. Dysbiosis diminishes beneficial metabolites and elevates harmful ones, such as lipopolysaccharides, which promote systemic inflammation and impact neuropsychological health via the gut-brain axis[1]. A pioneering study identified a high-fat diet-microbiota-leucine-mTORC1-polymorphonuclear neutrophils-MDSC axis that undermines antitumor immunity and accelerates breast cancer progression[19]. Further analyses highlight the involvement of AGE-receptor for advanced glycation end products, TNF, and IL-17 pathways as microbiota-modulated links between diabetes and cancer[113]. Targeting the microbiome - through probiotics, prebiotics, fecal microbiota transplantation, or dietary interventions - offers promising noninvasive prevention and adjunctive therapeutic strategies[116]. Figure 3 integrates these dietary, systemic and microbial disturbances into a unified gut-metabolism-breast axis that underpins tumour initiation and progression.
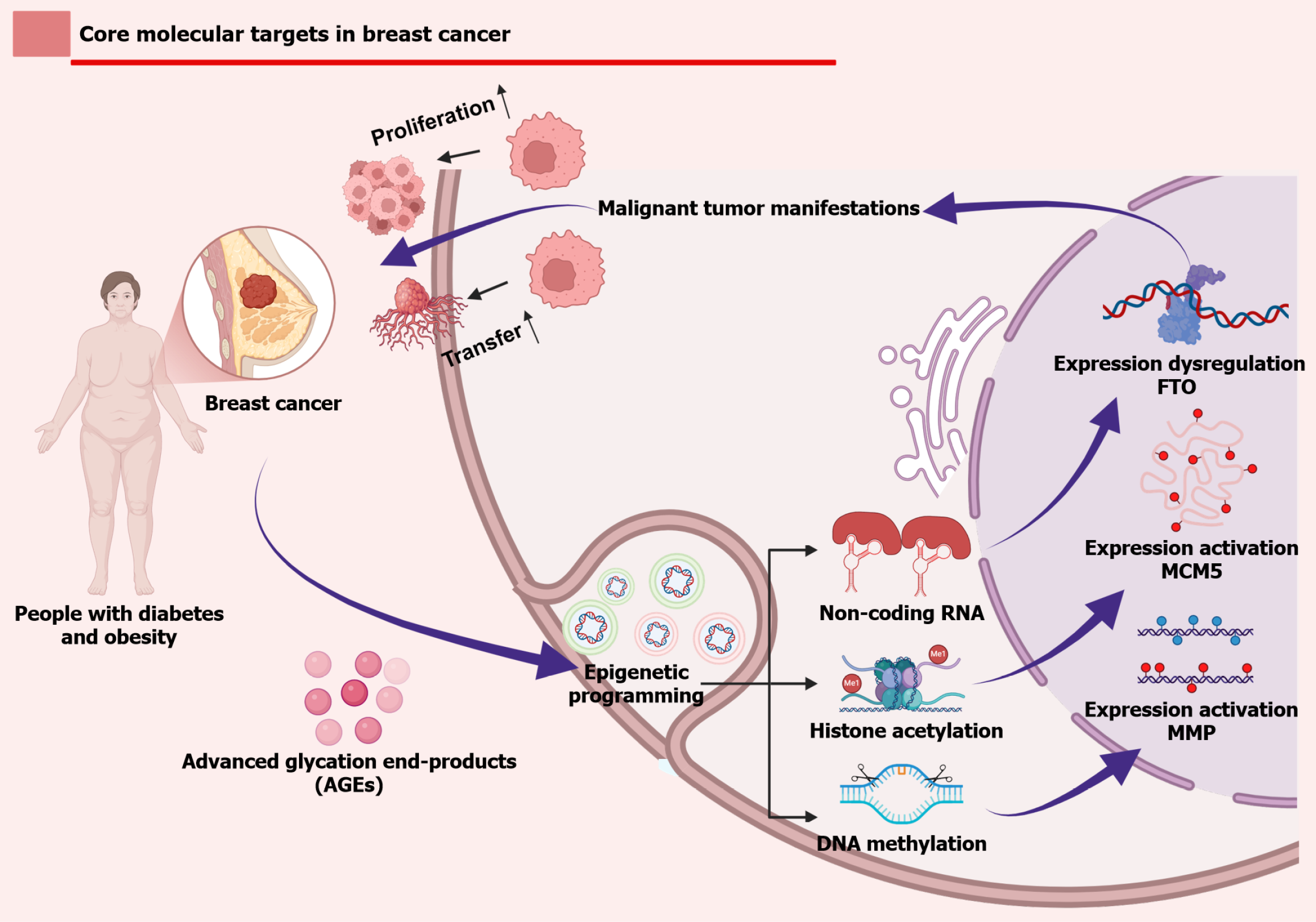
Epigenetic mechanisms transduce environmental signals, such as overnutrition and hyperglycemia, into stable transcriptional programs that drive oncogenesis. Obesity and diabetes broadly reprogram the epigenome in breast tissues, influencing survival, metastasis, and therapeutic responses[20,52,117]. In hyperglycemic conditions, the pyruvate dehydrogenase-acetyl-CoA axis enhances histone H3 lysine 27 acetylation at the minichromosome maintenance complex component 5 promoter, accelerating cell-cycle progression[52]. AGEs trigger growth arrest and DNA damage-inducible 45α-mediated base-excision repair and demethylation of the MMP-9 promoter, thereby promoting metastasis[54].
Beyond metabolite-driven alterations, dysregulated gene expression links metabolic diseases with cancer. FTO, the first obesity-susceptibility gene identified by genome-wide association study, may play a pivotal role in the shared susceptibility between obesity, diabetes, and breast cancer by modulating m6A methylation and energy metabolism pathways[118]. Its mechanisms likely involve DNA repair and inflammatory signaling, highlighting the intersection of genetic predisposition and cellular processes in mediating comorbidity risk. Multi-omics approaches have further revealed shared regulatory modules across comorbid conditions. For instance, joint transcriptomic analyses of depression and breast cancer identified overlapping immune-related differentially expressed genes, such as STAT1, IRF7, and CXCL10[119]. Integrated analyses of thyroid and breast cancer highlighted common hub genes and regulatory miRNAs linked to autophagy and ferroptosis[120]. These convergent metabolic and genetic influences on the breast cancer epigenome are summarized in Figure 4.
Decoding these mechanistic links aims to uncover actionable therapeutic targets, with transcriptional and enzymatic regulators standing out. Sterol regulatory element-binding protein 1, which controls lipogenesis, is aberrantly activated in obesity-driven breast cancer through PI3K/AKT/mTOR and adenosine monophosphate-activated protein kinase signaling pathways, promoting lipid accumulation and tumor growth. Small-molecule inhibitors, such as fatostatin, show promise in targeting this pathway[17]. Protein tyrosine phosphatase 1B, a negative regulator of insulin and leptin signaling, is under clinical investigation for its dual potential in treating both T2DM/obesity and metastatic breast cancer[121]. Aldo-keto reductase family 1 member B10, involved in inflammation and metabolism, is another promising candidate[122].
Cell-surface receptors and their signaling pathways also present therapeutic opportunities. GLP-1R expression is a prognostic marker across various cancers, with higher levels correlating with improved survival in breast cancer, suggesting potential antitumor effects of GLP-1 RAs[123]. The pregnane X receptor, which regulates xenobiotic metabolism, inflammation, and energy homeostasis, is being explored as a target for metabolic diseases and cancer[124]. Leucine-rich repeat-containing leucine-rich repeat-containing G protein-coupled receptor 4, involved in bone metabolism and osseous metastasis, presents a potential target for preventing or treating bone metastases[125]. Among secreted proteins, sFRP4, an antagonist of Wnt signaling, inhibits breast cancer cell growth when upregulated pharmacologically, offering therapeutic potential[114]. IGF binding protein 6, a specific inhibitor of IGF-II, is associated with a favorable prognosis, suggesting opportunities to mimic or enhance its function[111]. In obesity-associated TNBC, astragaloside IV activates forkhead box A1 to upregulate galactose-3-O-sulfotransferase 1, remodel sphingolipid metabolism, and suppress tumor growth[126]. Additionally, miR-877-5p has been proposed as a molecular link between MetS and TNBC, supporting the potential of RNA-based strategies[65].
Metabolic comorbidities extend beyond mere risk factors; they exert widespread effects across diagnosis, decision-making, therapeutic efficacy, toxicity, recurrence, second primaries, and survival, complicating individualized care and prognostication. The cross-cutting impact of major metabolic comorbidities across tumour pathology, treatment delivery, and long-term outcomes is summarized in Table 1.
| Clinical dimension | Obesity | Type 2 diabetes | Metabolic syndrome | Cardiovascular disease |
| Tumour pathology and subtype | Associated with postmenopausal ER+ disease; central obesity linked to luminal B/TNBC in younger[125,127] | Higher incidence of TNBC; lower prevalence of HER2+ disease[54] | Potentially stronger association with poor prognosis in HER2-overexpressing subtypes[61] | - |
| Treatment delivery and efficacy | Therapeutic decisions: Contributes to treatment de-escalation in older adults[98,132] | Therapeutic decisions: Lower likelihood of receiving chemotherapy[55] | Efficacy: Lower pCR rates after NACT[60,62] | Therapeutic decisions: Contributes to undertreatment, particularly in metastatic HER2+ disease[131] |
| Efficacy: Reduced rate of pCR following NACT[42,62] | Safety: Increased risk of postoperative complications[14,49] | Safety: Key predictor of CTRCD; CDK4/6 inhibitors increase risk of hypertension and MACE[71,77,78] | ||
| Long-term survival outcomes | Recurrence: Increased risk, especially in HR+ patients on aromatase inhibitors[35,135] | Second primary cancers: Elevated risk of several SPCs (e.g., liver, brain)[16] | Recurrence: Significantly increased risk[12,60] | Mortality: Leading cause of non-cancer death among survivors[67,68] |
| Mortality: Elevated breast cancer-specific and all-cause mortality[9,26,136] | Mortality: 20% increased risk of breast cancer-related death; causal evidence exists[10,51] | Mortality: Elevated breast cancer-specific and all-cause mortality[12,60] |
Comorbid states exhibit nonrandom associations with molecular subtypes and pathological features, suggesting selective pressures on tumor biology. In postmenopausal women, obesity (high BMI) is strongly linked with estrogen receptor-positive/HER2- breast cancer[127,128]. A large Korean cohort revealed that high BMI correlated with increased progesterone receptor (PR) positivity among postmenopausal estrogen receptor-positive/HER2- patients, with PR negativity predicting poorer prognosis only in obese patients, suggesting that BMI modifies the prognostic significance of PR[127]. In younger women, general obesity (high BMI) was associated with a lower risk of Luminal A subtype, whereas central obesity (larger WC) correlated with increased risks of Luminal B and TNBC[129]. Obesity also correlates with larger tumor size, nodal positivity, and poorer differentiation[130,131].
T2DM and metabolic alterations shape histopathology. In Chinese women with non-metastatic breast cancer, T2DM was linked to a higher incidence of TNBC and lower HER2 positivity compared to non-diabetic women[55]. Severe hyperglycemia (fasting blood glucose ≥ 10 mmol/L) was associated with higher risks of TNBC and grade III tumors and lower ER positivity. Treatment patterns also varied: Metformin use was associated with a higher incidence of TNBC, whereas insulin therapy was linked to a higher prevalence of ductal carcinoma in situ[55].
As a composite condition, MetS may affect subtypes differently. One study found that MetS was associated with poorer outcomes (higher recurrence and breast cancer-specific mortality) specifically in HER2-overexpressing disease, with weaker or absent associations in Luminal or TNBC subtypes[62]. Understanding this heterogeneity is essential for subtype-specific prevention and management based on metabolic status.
Comorbidity burden significantly influences clinical decisions, treatment response, and tolerance, especially in older and metabolically compromised patients[132]. Older age and higher comorbidity indices often result in treatment de-escalation or under-treatment. A substantial proportion of older, metastatic HER2-positive patients did not receive guideline-recommended anti-HER2 therapy[133]. Many older women with high-risk disease opt out of multimodal treatment (surgery and systemic therapy), with decisions shaped by age, CCI, and SES[100,134]. Diabetes has been shown to reduce chemotherapy use in specific populations, such as Pacific Islanders and those with stage II disease[56].
Metabolic comorbidities can diminish therapeutic efficacy. Obesity and MetS are associated with lower pCR rates after NACT[43,61,63]. While some adjusted analyses show different trends[44], the general pattern suggests that inflammation, altered pharmacokinetics, and resistance mechanisms reduce chemosensitivity. For certain agents, such as the antibody-drug conjugate sacituzumab govitecan, obesity does not negate its superiority over chemotherapy but may increase the risk of grade ≥ 3 adverse events[135].
Comorbidities also heighten toxicity risks. Diabetes increases systemic and surgical complications following breast surgery[14,50]. CVD burden predicts CTRCD[71], and a high baseline CCI increases cardiotoxicity risk when using anthracyclines, anti-HER2 agents, or CDK4/6 inhibitors[71,73]. PI3K and AKT inhibitors can induce severe hyperglycemia and even diabetic ketoacidosis, presenting challenges for prediabetic and diabetic patients[57,58]. Vitamin D deficiency, common during chemotherapy, is associated with cachexia[136]. Thus, comprehensive comorbidity assessment and proactive management are critical to optimizing benefit-risk ratios.
Metabolic comorbidities significantly elevate long-term risks, including recurrence, SPCs, and mortality, highlighting the necessity of integrating metabolic care into survivorship strategies.
Obesity, diabetes, and MetS are independent predictors of recurrence. High BMI is associated with increased recurrence, particularly among postmenopausal hormone receptor-positive patients undergoing aromatase inhibitor therapy[26] as well as in NACT cohorts[137]. MetS substantially raises recurrence risk[12,61]. However, while postoperative complications - detrimental to recovery and survival - did not independently influence early or late recurrence in a large Danish cohort, they remain significant in clinical management[50].
Comorbidity burden also predicts SPCs. Higher Elixhauser or Charlson comorbidity indices are linked to an increased risk of non-breast SPCs[15,87]. Diabetes is associated with elevated risks of subsequent liver, brain, and thyroid cancers in breast cancer survivors[16]. Additionally, MetS and its components are correlated with endometrial and other hormone-related cancers[67]. Shared mechanisms, including hyperinsulinemia and chronic inflammation, may contribute to both recurrence and de novo tumorigenesis.
Ultimately, metabolic comorbidities worsen survival outcomes. Obesity and overweight are linked to higher breast cancer-specific and all-cause mortality[9,27,40,138] with the potential for eliminating obesity to prevent a significant proportion of deaths[27]. Diabetes increases breast cancer-related mortality, and causal-inference analyses suggest that T2DM causally raises 5-year mortality rates[52]. Meta-analyses and multicenter cohort studies further indicate that MetS independently predicts poorer outcomes, including higher recurrence and all-cause mortality[9,12,139,140]. Other factors, such as postoperative complications (especially in diabetic patients)[50] and persistent opioid use after treatment[141], are also linked to increased all-cause mortality. Thus, effective metabolic management is crucial for improving long-term survival outcomes.
Over the past decade, accumulating mechanistic and clinical evidence has reframed obesity, T2DM, MetS and related CVD as actionable entry points along the breast cancer continuum rather than mere background comorbidities. Building on the metabo-oncologic pathways summarized above, this section outlines precision interventions that target key pathogenic processes and integrate metabolic management into breast cancer care across pharmacologic, lifestyle, data-driven, and survivorship domains. Table 2 provides an overview of major mechanistic axes, representative molecular pathways and corresponding therapeutic mor preventive strategies across multiple biological scales.
| Biological scale | Core pathophysiological process | Key molecular mediators and pathways | Potential intervention strategies |
| Systemic and microenvironmental | Adipose tissue dysfunction: Promotes chronic inflammation via adipokine/cytokine secretion[4,29] | Leptin, IL-6, TNF-α; TREM2; collagen deposition[4,19,29,103,105] | Lifestyle: Weight loss reverses obesity-associated immunosuppression[104] |
| Immune suppression: Enrichment of M2 macrophages and MDSCs in the TME[19,101,104] | Pharmacological: GLP-1 receptor agonists (e.g., semaglutide)[21,22] | ||
| Extracellular matrix remodelling: Favours tumour invasion and metastasis[103] | Immunotherapy: Combination with checkpoint inhibitors[19,101,104] | ||
| Cellular and signalling | Metabolic pathway hyperactivation: Drives cell proliferation and survival[6,17] | PI3K/Akt/mTOR; JAK/STAT; Wnt/β-catenin[6,17,101,109,110,112] | Drug repurposing: Metformin[11,22] |
| Persistent inflammatory signalling: Fuels tumour progression[109,110] | Targeted therapies: PTP1B inhibitors (dual-target); PI3K/AKT inhibitors (e.g., alpelisib, capivasertib)[56,57,119] | ||
| Developmental pathway dysregulation: Contributes to oncogenesis[101,112] | Natural compounds: Genistein (soy isoflavone)[101] | ||
| Epigenetic and molecular | Metabolite-driven reprogramming: Alters gene expression via histone modifications and DNA methylation[51,53] | H3K27ac at MCM5 promoter; MMP-9 promoter demethylation; miR-877-5p[51,53,64] | Epigenetic drugs (in development): Strict metabolic control RNA-based therapies (potential) |
| Non-coding RNA dysregulation: Connects metabolic stress to cancer phenotypes[64] | |||
| Emerging frontiers | Gut-microbiota-host crosstalk: Microbial dysbiosis and metabolites systemically influence immunity and cancer progression[19,65] | Microbiota-derived leucine; FOXA1/GAL3ST1 axis[19,124] | Microbiome modulation: Pre/probiotics, faecal microbiota transplantation[114] |
| Sphingolipid metabolic reprogramming: Identified in obesity-associated TNBC[124] | Natural compounds: Astragaloside IV[124] |
Mechanistic insights have prompted the pharmacologic modulation of metabolic pathways, with repurposing antidiabetic agents emerging as a key strategy. Metformin, an adenosine monophosphate-activated protein kinase activator that suppresses mTOR and lowers insulin, demonstrates preclinical anticancer activity[11,12,139]. However, epidemiological evidence on its benefits for breast cancer prevention and treatment is mixed: Some large cohorts report no significant reduction in incidence[140], while others suggest context-specific protective effects, particularly for digestive cancers[142].
GLP-1RAs, such as semaglutide and tirzepatide, provide potent weight loss and glycemic control and are increasingly explored for use in obese patients with breast cancer[22,23]. In addition to their indirect effects on insulin sensitivity and appetite suppression, preliminary data and Mendelian randomization/meta-analyses suggest a potential reduction in breast cancer risk[123]. While early safety signals are promising, large prospective trials are necessary to establish long-term oncologic safety and efficacy[22].
However, rapid weight loss induced by GLP-1RAs could potentially lead to muscle mass loss, increasing the risk of sarcopenia, particularly in cancer survivors. Therefore, it is crucial to monitor muscle health in breast cancer patients undergoing treatment with GLP-1RAs. Resistance training and nutritional support should be incorporated into the treatment plan to mitigate the risk of muscle loss and prevent the development of sarcopenia, which could further compromise functional status and quality of life[143].
SGLT2 inhibitors, which reduce glucose via glycosuria, may also influence tumor metabolism[23]. Meta-analyses show no significant association with overall breast cancer incidence, but they suggest potential reductions in breast cancer-specific mortality and improvements in OS[144]. However, signals of increased renal cancer risk require caution[145].
Novel agents are targeting more precise pathways. protein tyrosine phosphatase 1B inhibitors are being clinically evaluated in the context of T2DM/obesity and metastatic breast cancer[121]. Sterol regulatory element-binding protein 1 inhibitors, such as fatostatin, have shown preclinical efficacy in obesity-associated diseases[17]. Other approaches include pharmacologic upregulation of sFRP4[114] and the use of phytochemicals, like astragaloside IV, which target forkhead box A1/galactose-3-O-sulfotransferase 1 and sphingolipid metabolism[126]. Non-traditional agents, such as anti
Lifestyle modification is fundamental to the integrated management of breast cancer survivors with metabolic comorbidities, aiming to disrupt the vicious cycle between metabolic disturbances and tumor progression. Evidence from both structured and community-based programs consistently supports its effectiveness. In a large randomized trial, two years of telephone-delivered counseling combining dietary adjustments and physical activity resulted in sustained weight loss, reduced WC, and improvements in glycemia, blood pressure, and lipid profiles among overweight and obese survivors[24]. Similarly, community-based multimodal programs have been shown to reduce the prevalence of MetS and enhance quality of life, particularly in postmenopausal women[148]. Dietary strategies are central to these interventions. An eight-week whole-food, plant-based program for patients with metastatic breast cancer led to significant weight loss and improvements in insulin resistance, lipid metabolism, and sex hormone levels, with good tolerability[149]. Adherence to Mediterranean dietary patterns further supports metabolic control and weight management[150], while reducing intake of ultra-processed foods helps mitigate adiposity[95]. Moreover, consumption of whole foods, such as dried fruits, has been linked to a reduced genetic risk of breast cancer[94]. Emerging nutritional approaches, such as caloric restriction and fasting-mimicking diets, have shown promising survival advantages in preclinical models and are undergoing clinical evaluation[151].
In addition to the types of food consumed, the timing of meals has gained attention in metabolic oncology. Time-restricted eating, which limits eating to specific hours of the day, may offer benefits in improving metabolic health and restoring circadian rhythms, which are often disrupted in cancer survivors. Studies suggest that time-restricted eating can help regulate blood glucose levels, reduce inflammation, and improve overall metabolic function, providing a potential complementary approach to traditional dietary strategies[152].
Physical activity complements dietary modifications and is essential for holistic metabolic control. Exercise enhances energy expenditure, supports weight management, improves immune function, and reduces chronic inflammation. Regular physical activity can counteract the negative effects of prolonged sedentary behavior[92], while weight loss - whether through diet, exercise, or both - can reverse obesity- and aging-related immunosuppression within tumors[106]. Innovative formats, such as exergaming, may improve participation, though they appear insufficient as standalone strategies, highlighting the need for multimodal, personalized interventions that emphasize motivation and enjoyment[153]. For individuals with severe obesity, bariatric surgery offers a highly effective therapeutic option, significantly reducing the risk of obesity-related malignancies, including breast cancer[154-156], and improving adherence to recommended screening practices[157]. In conclusion, lifestyle modification - ranging from daily behavioral changes to structured interventions and surgical therapies - represents a powerful, multifaceted approach to disrupting the metabo-oncologic cycle and improving long-term outcomes for breast cancer survivors.
With the rapid expansion of health data and computational power, machine learning enables precise risk stratification, prognostication, and decision support by integrating clinical, pathological, genomic, lifestyle, and socioeconomic factors.
Unsupervised clustering techniques, such as k-prototypes, can identify phenotypic subgroups with distinct risk profiles, incorporating comorbidity indices like CCI. In TNBC, four clusters were found to exhibit significantly different recurrence and mortality rates, with the highest-risk group showing poorer ECOG performance and higher breast cancer susceptibility gene 1/2 mutation frequencies[158]. Supervised models, including random forests and least absolute shrinkage and selection operator, demonstrated 80% and 75% accuracy for OS and relapse-free survival, respectively, outperforming traditional clinical and genomic models[159]. In older survivors, random forest models incorporating frailty, comorbidities, geography, and out-of-pocket costs predicted 3- and 5-year all-cause and cancer-specific mortality and recurrence with 85%-97% accuracy[159]. Across these models, comorbidity burden and functional status were consistently among the most significant predictors.
Machine learning also facilitates the identification of emerging biomarkers. While weight-adjusted waist index did not emerge as an independent predictor in conventional regressions, random forest and least absolute shrinkage and selection operator models ranked it highly, improving discrimination (area under the curve 0.79)[41]. Integrative computational analyses have revealed cross-disease genes, such as CCNB2, XRCC2, and CENPI, linking T2DM with breast cancer[53,160]. Multimodal nomograms, which combine clinical histories, imaging, and pathology, can predict costly genomic signatures (e.g., MammaPrint high/Low risk and quartiles) with robust performance in training and validation cohorts, offering cost-effective decision support in resource-limited settings[161].
As outcomes improve and the population of survivors grows, managing overlapping diseases becomes increasingly critical. Patients with metabolic comorbidities require dual-track care: Ongoing surveillance for recurrence and SPCs alongside management of comorbidity progression to maintain quality of life and survival. A patient-centered, multi
However, current survivorship care often fails to meet recommended standards across both primary and specialty settings[86]. The gaps are especially pronounced in resource-limited primary care systems, where patients face higher comorbidity burdens, poorer quality of life, increased chronic pain, and heightened mental health needs[86,162]. Shared-care models that bridge oncology with primary care are urgently needed, particularly for groups facing systemic barriers, such as American Indians navigating the Indian Health Service and external cancer centers[90]. Routine screening and integrated mental health services should be embedded within survivorship care pathways to address these challenges[162].
Clinicians can play a pivotal role in addressing these social barriers by recommending patients seek assistance from patient navigators or social workers. These professionals can help with practical issues such as securing food, accessing exercise facilities, and navigating healthcare systems, ensuring patients receive comprehensive support in managing both their physical and social needs.
Despite substantial progress, significant limitations remain in current research. Much of the existing evidence is associative rather than causal, limiting the ability to establish directionality and specificity between metabolic dysregulation and breast cancer progression. Mechanistic studies often focus on isolated pathways, neglecting the complex crosstalk between adipocytes, immune cells, and stromal components that shape the TME. Additionally, the role of the gut microbiota and its metabolites in tumor immunity and host metabolism is insufficiently understood. Current risk prediction models seldom integrate genetic susceptibility, metabolic biomarkers, and lifestyle factors within large, diverse populations, and real-world cohort data remains scarce. These gaps hinder the translation of epidemiological associations into actionable, precision-based prevention and management strategies.
Future research must shift from correlation to causality and precision, leveraging advanced technologies such as single-cell multi-omics and spatial transcriptomics to define cell-type–specific interactions across various metabolic states (e.g., obesity, diabetes, post-weight loss). Investigating adipocyte–immune–stromal communication will reveal actionable molecular targets. The emerging gut microbiota-host metabolism–tumor immunity axis warrants further exploration to enable the development of microecology-based therapies. Integrating genetic polymorphisms (e.g., FTO variants) with environmental and behavioral exposures in large-scale prospective cohorts could refine individualized risk models. Clinically, a “metabo-oncology” paradigm is essential - embedding routine metabolic assessments (BMI, WC, glucose, lipid profiles, MetS diagnosis) into oncologic care for risk stratification and personalized intervention. High-risk patients should receive coordinated multidisciplinary management to tailor antitumor therapy, mitigate treatment-related metabolic toxicity, and concurrently address metabolic dysfunction through pharmacologic interventions (e.g., GLP-1RAs, metformin) and structured lifestyle strategies. Given their therapeutic potential, GLP-1RAs require rigorous, long-term evaluation to confirm oncologic safety and survival benefits in breast cancer populations.
In conclusion, incorporating metabolic health as a core component of breast cancer management, rather than a peripheral concern, is critical to advancing prevention, optimizing treatment efficacy, and extending healthy survivorship. As mechanistic understanding deepens and precision clinical models evolve, targeted modulation of metabolic dysfunction offers great promise to reduce cancer incidence, enhance outcomes, and ultimately improve the lives of patients with breast cancer and survivors. Looking ahead, future clinical trial designs should consider stratifying patients based on their metabolic phenotypes. This approach would enable more accurate assessments of how metabolic backgrounds influence the efficacy of breast cancer therapies, helping to identify personalized treatment strategies. By considering metabolic factors such as obesity, insulin resistance, and inflammatory markers, trials could better evaluate the differential responses to treatment across various metabolic profiles, paving the way for more tailored and effective therapeutic interventions.
| 1. | Yan X, Shi L, Zhu X, Zhao Y, Luo J, Li Q, Xu Z, Zhao J. From Microbial Homeostasis to Systemic Pathogenesis: A Narrative Review on Gut Flora's Role in Neuropsychiatric, Metabolic, and Cancer Disorders. J Inflamm Res. 2025;18:8851-8873. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (5)] |
| 2. | Parsons K, Montalvo M, Fischbach N, Taylor M, Alfaro S, Lustberg M. The Impact and Safety of GLP-1 Agents and Breast Cancer. Cancer Med. 2025;14:e70932. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 3. | Hansen CL, Viboud C, Simonsen L. Disentangling the relationship between cancer mortality and COVID-19 in the US. Elife. 2024;13:RP93758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 4. | Mallya S, Gangadhar V, Aldrin SE, Acharya M, Kabekkodu SP, Kolathur KK, Chakrabarty S. Insights into the molecular and genetic role of obesity in breast cancer pathogenesis. Cancer Biol Ther. 2025;26:2501345. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 5. | Zhang X, Nguyen MH. Metabolic dysfunction-associated steatotic liver disease: A sexually dimorphic disease and breast and gynecological cancer. Metabolism. 2025;167:156190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 6. | Javed SR, Skolariki A, Zameer MZ, Lord SR. Implications of obesity and insulin resistance for the treatment of oestrogen receptor-positive breast cancer. Br J Cancer. 2024;131:1724-1736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 24] [Reference Citation Analysis (4)] |
| 7. | Feng X, Li R, Yi H, Chen S, Liu M, Wu Y. Global cancer burden attributable to excess body weight, 1990 to 2021, decomposed by population size, aging, and epidemiological change. Obesity (Silver Spring). 2025;33:567-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 8. | Engin A. Obesity-Associated Breast Cancer: Analysis of Risk Factors and Current Clinical Evaluation. Adv Exp Med Biol. 2024;1460:767-819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 9. | Calvo I, González-Rodríguez M, Neria F, Gallegos I, García-Sánchez L, Sánchez-Gómez R, Pérez S, Arenas MF, Estévez LG. An analysis of the association between breast density and body mass index with breast cancer molecular subtypes in early breast cancer: data from a Spanish population. Clin Transl Oncol. 2024;26:2541-2548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 10. | Rashmi R, Singhal R, Misra G. Diabetes driven effects on incidence and mortality of breast cancer: A meta-analysis. Public Health. 2025;247:105914. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 11. | Khan I, Kamal A, Akhtar S. Diabetes Driven Oncogenesis and Anticancer Potential of Repurposed Antidiabetic Drug: A Systemic Review. Cell Biochem Biophys. 2024;82:1907-1929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 12. | Harborg S, Larsen HB, Elsgaard S, Borgquist S. Metabolic syndrome is associated with breast cancer mortality: A systematic review and meta-analysis. J Intern Med. 2025;297:262-275. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 13. | Bhimani J, Wang P, Gallagher GB, O'Connell K, Blinder V, Burganowski R, Ergas IJ, Griggs JJ, Heon N, Kolevska T, Kotsurovskyy Y, Kroenke CH, Laurent CA, Liu R, Nakata KG, Persaud S, Roh JM, Tabatabai S, Valice E, Bandera EV, Bowles EJA, Kushi LH, Kantor ED. Patient factors and modifications to intended chemotherapy for women with Stages I-IIIA breast cancer. Int J Cancer. 2025;157:1342-1353. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 14. | Xu B, Kang B, Li S, Fan S, Zhou J. Sodium-glucose cotransporter 2 inhibitors and cancer: a systematic review and meta-analysis. J Endocrinol Invest. 2024;47:2421-2436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 29] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 15. | Mukherjee A, Gu Z, Chen LH, Chlebowski RT, Potosky AL, Haque R. Comorbidity burden and risk of second primary non-breast cancer in breast cancer survivors. Cancer Epidemiol. 2025;97:102867. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 16. | Jung HI, Kwon SH, Nam JH, Cho JY, Lee EK. Real-world effectiveness of CDK4/6 inhibitors on patients with HR+/HER2- advanced breast cancer in South Korea, focusing on underrepresented patients. Curr Med Res Opin. 2024;1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 17. | Tiwari U, Akhtar S, Mir SS, Khan MKA. SREBP-1 in obesity-induced breast cancer: mechanisms and therapeutic perspectives. Mol Biol Rep. 2025;52:663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 18. | Zhao C, Zhang T, Xue ST, Zhang P, Wang F, Li Y, Liu Y, Zhao L, Wu J, Yan Y, Mao X, Chen Y, Yuan J, Li Z, Li K. Adipocyte-derived glutathione promotes obesity-related breast cancer by regulating the SCARB2-ARF1-mTORC1 complex. Cell Metab. 2025;37:692-707.e9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 28] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 19. | Sun Y, Liu Y, Dian Y, Zeng F, Deng G, Lei S. Association of glucagon-like peptide-1 receptor agonists with risk of cancers-evidence from a drug target Mendelian randomization and clinical trials. Int J Surg. 2024;110:4688-4694. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 53] [Article Influence: 26.5] [Reference Citation Analysis (0)] |
| 20. | Eslami B, Alipour S, Seyyedsalehi MS, Nahvijou A, Omranipour R, Rajabpour MV, Zendehdel K. Feasibility of measuring comorbidity indices based on clinical breast cancer records. East Mediterr Health J. 2023;26:870-876. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 21. | Xie H, Ruan G, Wei L, Zhang H, Shi J, Lin S, Liu C, Liu X, Zheng X, Chen Y, Deng L, Shi H. Obesity-associated metabolic inflammation promotes triple-negative breast cancer progression through the interleukin-6/STAT3/pentraxin 3/matrix metalloproteinase 7 axis. Int Immunopharmacol. 2024;136:112332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 22. | Xande JG, Del Giglio A. GLP-1 Receptor Agonists in Breast Cancer: A New Frontier in Obesity and Prognosis Management. Int J Mol Sci. 2025;26:7744. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 23. | Albini A, Cantelmo AR, Mortara L, Noonan DM, Corso G. Cancer prevention and interception with antidiabetic and anti-obesity drugs: Current and future perspectives. Semin Cancer Biol. 2025;115:40-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 24. | Hauner D, Günther J, Schederecker F, Donik L, Meyer D, Hermsdörfer J, Friedl TWP, Rack B, Beckmann MW, Janni W, Hauner H. Effect of a comprehensive lifestyle intervention program on body weight and health behavior in women with breast cancer: Results from a randomized controlled trial. Clin Nutr. 2024;43:1937-1951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 25. | Fontvieille E, Jansana A, Peruchet-Noray L, Córdova R, Gan Q, Rinaldi S, Dossus L, Mahamat-Saleh Y, Gunter MJ, Heath A, Aune D, Inan-Eroglu E, Schulze MB, Bock N, Dahm CC, Castro-Espin C, Sánchez MJ, Perez-Cornago A, Tin ST, Sieri S, Simeon V, Ricceri F, Tumino R, Onland-Moret NC, Koop Y, Ferrari P, Freisling H. Body mass index and breast cancer risk among postmenopausal women with and without cardiometabolic diseases: Findings from two prospective cohort studies in Europe. Cancer. 2025;131:e35911. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 26. | Harborg S, Cronin-Fenton D, Jensen MR, Ahern TP, Ewertz M, Borgquist S. Obesity and Risk of Recurrence in Patients With Breast Cancer Treated With Aromatase Inhibitors. JAMA Netw Open. 2023;6:e2337780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 34] [Reference Citation Analysis (2)] |
| 27. | Carneiro PL, Aguiar IWO, Bezerra IN, Verde SMML. [Mortality from female breast cancer attributable to overweight and obesity in Brazil]. Cien Saude Colet. 2025;30:e12612023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 28. | Mane N, Fouqani A, Mrah S, Omari M, Bouaddi O, Faure E, El Fahime EM, Lkhoyaali S, Boutayeb S, El Rhazi K, Nejjari C, Huybrechts I, Khalis M. Obesity and Risk of Pre- and Postmenopausal Breast Cancer in Africa: A Systematic Review. Curr Oncol. 2025;32:167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 29. | Lagarde CB, Kavalakatt J, Benz MC, Hawes ML, Arbogast CA, Cullen NM, McConnell EC, Rinderle C, Hebert KL, Khosla M, Belgodere JA, Hoang VT, Collins-Burow BM, Bunnell BA, Burow ME, Alahari SK. Obesity-associated epigenetic alterations and the obesity-breast cancer axis. Oncogene. 2024;43:763-775. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 30. | Newport-Ratiu PA, Hussein KA, Carter T, Panjarian S, Jonnalagadda SC, Pandey MK. Unveiling the intricate dance: Obesity and TNBC connection examined. Life Sci. 2024;357:123082. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 31. | Sudan SK, Sharma A, Vikramdeo KS, Davis W, Deshmukh SK, Poosarla T, Holliday NP, Prodduturvar P, Nelson C, Singh KP, Singh AP, Singh S. Obesity and Early-Onset Breast Cancer and Specific Molecular Subtype Diagnosis in Black and White Women: NIMHD Social Epigenomics Program. JAMA Netw Open. 2024;7:e2421846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 32. | Lin XY, Wang J, Zhang WS, Jiang CQ, Jin YL, Cheng KK, Lam TH, Xu L. Maximal weight change during adulthood and breast cancer risk: A 14-year follow-up of the Guangzhou Biobank Cohort Study. Cancer Epidemiol. 2025;97:102825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 33. | Khalil AA, ElSharkawy OA, Alsharkawy K, Youssif S, Khalil HH. Supra-arcuate free fascial MS-TRAM, a modified technique to reduce donor site morbidity in obese population-comparative study. Microsurgery. 2024;44:e31124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 34. | Larrad-Sáinz A, Hernández Núñez MG, Barabash Bustelo A, Gil Prados I, Valerio J, Espadas Gil JL, Olivares Crespo ME, Herrera de la Muela M, Bernaldo Madrid B, Serrano García I, Cristóbal García I, Rubio-Herrera MÁ, Calle-Pascual AL, Brenes Sánchez JM, Matía-Martín P. Relationship Between Body Composition and Biomarkers in Adult Females with Breast Cancer: 1-Year Follow-Up Prospective Study. Nutrients. 2025;17:2487. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 35. | Jung W, Park SH, Park YM, Song YM, Park JH, Yu J, Cho IY, Kim BS, Han K, Shin DW. Weight change and cardiovascular disease incidence in breast cancer survivors: a nationwide cohort study. Breast Cancer Res Treat. 2025;210:583-593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 36. | Dumas E, Grandal Rejo B, Gougis P, Houzard S, Abécassis J, Jochum F, Marande B, Ballesta A, Del Nery E, Dubois T, Alsafadi S, Asselain B, Latouche A, Espie M, Laas E, Coussy F, Bouchez C, Pierga JY, Le Bihan-Benjamin C, Bousquet PJ, Hotton J, Azencott CA, Reyal F, Hamy AS. Concomitant medication, comorbidity and survival in patients with breast cancer. Nat Commun. 2024;15:2966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 37. | Motuma A, Mussa I, Deressa A, Regassa LD, Birhanu A. Central obesity increases the risk of breast cancer irrespective of menopausal status in women: Systematic review and meta-analysis. Cancer Treat Res Commun. 2025;44:100965. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 38. | Nayan SI, Rahman MH, Hasan MM, Raj SMRH, Almoyad MAA, Liò P, Moni MA. Network based approach to identify interactions between Type 2 diabetes and cancer comorbidities. Life Sci. 2023;335:122244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 39. | Bea JW, Ochs-Balcom HM, Valencia CI, Chen Z, Blew RM, Lind KE, Caan BJ, Roe DJ, Rohan TE, Reeves KW, Manson JE, Ballinger T, Reding KW, Follis S, Ziller SG, Odegaard AO. Abdominal visceral and subcutaneous adipose tissue associations with postmenopausal breast cancer incidence. JNCI Cancer Spectr. 2025;9:pkaf007. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 40. | Patel RK, Frankel L, Cardeiro M, Hansen W, Takabe K, Rashid OM. The Role of Crohn Disease on Breast Cancer Incidence: A Clinical Analysis. World J Oncol. 2023;14:457-463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 41. | Wang W, Wu B, Li J, Shang Y, Liu M, Fang Q, Zhang H, Li X, Wu D. The application and predictive value of the weight-adjusted-waist index in BC prevalence assessment: a comprehensive statistical and machine learning analysis using NHANES data. BMC Cancer. 2025;25:1234. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 42. | Huang X, Cheng H, Deng L, Wang S, Li J, Qin A, Chu C, Du W, Liu X. Weight-adjusted-waist index: an innovative indicator of breast cancer hazard. BMC Womens Health. 2024;24:660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 43. | Guliyev M, Alan Ö, Günaltılı M, Safarov S, Fidan MC, Alkan Şen G, Değerli E, Papila B, Demirci NS, Papila Ç. Obesity Is an Independent Prognostic Factor That Reduced Pathological Complete Response in Operable Breast Cancer Patients. Medicina (Kaunas). 2024;60:1953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 44. | Englisch JF, Englisch A, Dannehl D, Eissler K, Tegeler CM, Matovina S, Volmer LL, Wallwiener D, Brucker SY, Hartkopf A, Engler T. Impact of obesity on pathological complete remission in early stage breast cancer patients after neoadjuvant chemotherapy: a retrospective study from a German University breast center. Arch Gynecol Obstet. 2025;311:437-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 45. | Matsumoto A, Ushio K, Kimura H, Tomioka S, Sasada S, Asaeda M, Nakashima Y, Fukuhara K, Mikami Y. Database study of risk factors for breast cancer-related lymphedema: a statistical analysis of 2359 cases over 10 years. Surg Today. 2025;55:685-692. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 46. | Silverstein ML, Sorice-Virk S, Wan DC, Momeni A. Microsurgical Breast Reconstruction can be Performed Safely in Patients with Obesity. J Reconstr Microsurg. 2024;40:730-742. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 47. | King CA, Shaposhnik G, Sayyed AA, Bartholomew AJ, Bozzuto LM, Sosin M, Greenwalt IT, Fan KL, Song D, Tousimis EA. Expanded Indications for Nipple-Sparing Mastectomy and Immediate Breast Reconstruction in Patients Older Than 60 Years. Ann Plast Surg. 2024;92:279-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 48. | McBride KA, Munasinghe S, Sperandei S, Page AN. Trajectories in mammographic breast screening participation in middle-age overweight and obese women: A retrospective cohort study using linked data. Cancer Epidemiol. 2024;93:102675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 49. | Xiong F, Wang J, Nierenberg JL, Van Blarigan EL, Kenfield SA, Chan JM, Schmajuk G, Huang CY, Graff RE. Diabetes mellitus and risk of breast cancer: a large-scale, prospective, population-based study. Br J Cancer. 2023;129:648-655. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 50. | Kjærgaard KA, Christiansen P, Borgquist S, Cronin-Fenton D. Impact of postoperative complications and type 2 diabetes on breast cancer recurrence and mortality. Br J Surg. 2025;112:znaf176. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 51. | Chlebowski RT, Aragaki AK, Pan K, Simon MS, Neuhouser ML, Haque R, Rohan TE, Wactawski-Wende J, Orchard TS, Mortimer JE, Lane D, Kaunitz AM, Desai P, Wild RA, Barac A, Manson JE. Breast cancer incidence and mortality by metabolic syndrome and obesity: The Women's Health Initiative. Cancer. 2024;130:3147-3156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 35] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 52. | Tan B, Liu Y, Chen Q, Yang W, Yang W, Gao K, Fu L, Zhang T, Chen P, Huang Y, Wang Y, Zhang G, Xiong J, Zhai R. Diabetes is causally associated with increased breast cancer mortality by inducing FIBCD1 to activate MCM5-mediated cell cycle arrest via modulating H3K27ac. Cell Death Dis. 2025;16:546. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 53. | Bao X, Zeng Z, Tang W, Li D, Fan X, Chen K, Wang Y, Ai W, Yang Q, Liu S, Chen T. Bioinformatics Combined With Biological Experiments to Identify the Pathogenetic Link of Type 2 Diabetes for Breast Cancer. Cancer Med. 2025;14:e70759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 54. | Zhou L, Bai G, Song Y, Liu X, Li X, Deng Y, Si Y, Shi Y, Li H. AGEs induce MMP-9 promoter demethylation through the GADD45α-mediated BER pathway to promote breast cancer metastasis in patients with diabetes. Endocr Relat Cancer. 2024;31:e230330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 55. | Liu L, Gao Y, Liu P, Hui R, Zhang J. Association of type 2 diabetes mellitus with histopathological features of Non-metastatic breast cancer in Chinese women: a retrospective Cross-sectional study. Sci Rep. 2025;15:28645. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 56. | Lao C, Gurney J, Stanley J, Teng A, Kuper-Hommel M, Campbell I, Krebs J, Sika-Paotonu D, Koea J, Stairmand J, Lawrenson R. Differences in systemic treatments for breast cancer between patients with and without diabetes. N Z Med J. 2025;138:67-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 57. | Chu A, Hakami-Majd N, Whall JO. Acute hyperglycemic hyperosmolar syndrome associated with capivasertib, a new, oral targeted therapy for advanced breast cancer. Am J Emerg Med. 2025;88:273.e5-273.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 58. | Ng WH, Abu Zaid Z, Mohd Yusof BN, Amin Nordin S, Lim PY. Association between dietary inflammatory index and body fat percentage among newly diagnosed breast cancer patients. Ann Med. 2023;55:2303399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 59. | Li J, Li J, Jin J, Zhang R, Li R, Xu X, Wang Y, Hu X, Wang L, Yu S. Association between gestational diabetes mellitus and risk of breast cancer: a systematic review and meta-analysis. Front Endocrinol (Lausanne). 2025;16:1621932. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 60. | Guo M, Liu T, Li P, Wang T, Zeng C, Yang M, Li G, Han J, Wu W, Zhang R. Association Between Metabolic Syndrome and Breast Cancer Risk: An Updated Meta-Analysis of Follow-Up Studies. Front Oncol. 2019;9:1290. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (40)] |
| 61. | Ma Y, Zhang J, Jiao D, Chen X, Liu Z. Metabolic Syndrome Is Associated With Poor Prognosis in Patients With Breast Cancer Receiving Neoadjuvant Therapy. Cancer Med. 2024;13:e70484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 62. | Loroña NC, Othus M, Malone KE, Linden HM, Tang MC, Li CI. Metabolic Syndrome and Risks of Breast Cancer Outcomes for Luminal, Triple-Negative, and HER2-Overexpressing Subtypes. Cancer Epidemiol Biomarkers Prev. 2025;34:117-124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 63. | Zhao X, Jia C, Ji S, Lu K, Yang P, Wang Y. Development and validation of a metabolic syndrome and its components to predict the efficacy of neoadjuvant chemotherapy in breast cancer: An observational, single-center, cohort study. Medicine (Baltimore). 2025;104:e41221. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 64. | Agnoli C, Perlino F, Guerra G, Quartiroli M, Vener C, Mauri P, de Palma A, Venturelli E, Sieri S. Advanced glycation end products and breast cancer risk in a sample of the ORDET cohort. Int J Biol Markers. 2025;40:75-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 65. | Mubtasim N, Gollahon L. The Effect of Adipocyte-Secreted Factors in Activating Focal Adhesion Kinase-Mediated Cell Signaling Pathway towards Metastasis in Breast Cancer Cells. Int J Mol Sci. 2023;24:16605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 66. | Abdelqader EM, Mahmoud WS, Gebreel HM, Kamel MM, Abu-Elghait M. Correlation between gut microbiota dysbiosis, metabolic syndrome and breast cancer. Sci Rep. 2025;15:6652. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 14] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 67. | Khaled S, Abdelkhalek S, Aljuwaybiri R, Almatrafi J, AlHarbi A, Almarhabi R, Alyamani F, Soliman M, Jubran E, Shalaby G. Cardiac dysfunction and their determinants in patients treated for breast cancer and lymphoma: A cardio-oncology center experience. Curr Probl Cardiol. 2024;49:102187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 68. | Bikomeye JC, McGinley EL, Zhou Y, Tarima S, Kwarteng JL, Beyer AM, Yen TWF, Winn AN, Beyer KMM. Greenspace and Survival Among Older Women With Breast Cancer: Regional Variations Within the U.S. SEER-Medicare-Linked Database. JACC Adv. 2025;4:102069. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 69. | Ferrero-Hernández P, Farías-Valenzuela C, Castillo-Paredes A, Rezende LFM, Cristi-Montero C, Sadarangani KP, Christofaro DGD, Ferrari G. Preventable incidence cases from non-communicable diseases attributable to insufficient physical activity in Chile. Public Health. 2024;226:53-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 70. | Taborda Ribas H, Sogayar MC, Dolga AM, Winnischofer SMB, Trombetta-Lima M. Lipid profile in breast cancer: From signaling pathways to treatment strategies. Biochimie. 2024;219:118-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 71. | Machado BB, Biolo A, Pivatto Júnior F, Schroeder AQ, Saffi MAL. Predictive risk factors associated with cancer therapy-related cardiac dysfunction: A retrospective cohort study. J Oncol Pharm Pract. 2025;10781552251358183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 72. | Perrone V, Leogrande M, Cappuccilli M, Saragoni S, Cinti Luciani A, Degli Esposti L. Real World Evidence on Hormone Receptor Positive and Human Epidermal Growth Factor Receptor 2 Negative Metastatic Breast Cancer in Italy: Insights From 2017 to 2021 Data. Clinicoecon Outcomes Res. 2025;17:147-155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 73. | Dent S, Guha A, Moore H, Makari D, McCaleb R, Arias I, Stergiopoulos S, Li B, Fradley M. CARDIAC-STAR: prevalence of cardiovascular comorbidities in patients with HR + /HER2 - metastatic breast cancer. Cardiooncology. 2025;11:7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 74. | Bikomeye JC, McGinley EL, Zhou Y, Tarima S, Kwarteng JL, Beyer AM, Yen TWF, Winn AN, Beyer KMM. Urban greenspace and cardiovascular disease comorbidity at breast cancer diagnosis in the US: Regional, racial/ethnic, and socioeconomic variations among older women. Cancer Surviv Res Care. 2025;3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 75. | Long Y, Huang R, Li W. Mendelian randomization study of breast cancer-related genes and their association with inflammatory signaling pathways. Discov Oncol. 2025;16:1619. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 76. | Harris CM, Kauffman R, Khaliq W. Cancer Screening Prevalence and Preference among Hospitalized Women With and Without Obesity. South Med J. 2023;116:874-882. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 77. | Safadi H, Balogh Á, Lám J, Nagy A, Belicza É. Associations between diabetes and cancer: A 10-year national population-based retrospective cohort study. Diabetes Res Clin Pract. 2024;211:111665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 78. | Park C, Liu YS, Kenawy AS, Lin YH, Liu Y, Heo JH. Cardiovascular Adverse Events and Associated Costs of CDK4/6 Inhibitors in Patients With Breast Cancer. J Natl Compr Canc Netw. 2025;23:e257001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 79. | Karlstaedt A, Taegtmeyer H. Cardio-Onco-Metabolism - Metabolic vulnerabilities in cancer and the heart. J Mol Cell Cardiol. 2022;171:71-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 80. | Contaldi C, D'Aniello C, Panico D, Zito A, Calabrò P, Di Lorenzo E, Golino P, Montesarchio V. Cancer-Therapy-Related Cardiac Dysfunction: Latest Advances in Prevention and Treatment. Life (Basel). 2025;15:471. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 81. | Ng HS, Johansen C, Li M, Roder D, Beckmann K, Koczwara B. Patterns of medication use following breast cancer diagnosis: an Australian population-based study. Support Care Cancer. 2025;33:668. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 82. | Chen VC, Chuang W, Tsai YH, McIntyre RS, Weng JC. Longitudinal assessment of chemotherapy-induced brain connectivity changes in cerebral white matter and its correlation with cognitive functioning using the GQI. Front Neurol. 2024;15:1332984. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 83. | Cho J, Higgason N, Rothman J, Safford M, Pinheiro LC. "Should I Prioritize My Cancer or My Diabetes?": Patient-Perceived Barriers to Co-Managing Cancer and Diabetes Mellitus. J Cancer Educ. 2024;39:437-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 84. | Sanogo F, Jefferson M, Beard TA, Salhia B, Babatunde OA, Cho J, Hughes Halbert C. Social and clinical drivers of stress responses in African American breast cancer survivors. Sci Rep. 2024;14:19729. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 85. | Pirikahu S, Darcey E, Lund H, Wylie E, Stone J. The impact of height and weight on rescreening rates within a population-based breast screening program. Cancer Med. 2024;13:e6883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 86. | Puschel K, Arancibia V, Rioseco A, Paz S, Soto MG, Martinez J, Faundez M, Acevedo F, Di Biase F, Emery J, León A, Are C, Thompson B. Challenges of cancer survivorship care in Chile: a longitudinal study comparing the quality of care and quality of life for cancer survivors in a primary care network and a cancer centre in Chile. BMJ Open. 2025;15:e097015. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (2)] |
| 87. | Golani R, Kagenaar E, Jégu J, Belot A, Ling S. Socio-economic inequalities in second primary cancer incidence: A competing risks analysis of women with breast cancer in England between 2000 and 2018. Int J Cancer. 2025;156:2283-2293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 88. | Sánchez-Díaz CT, Fejerman L, Peterson C, Basu S, Fitzgibbon M, Rauscher GH. Ethnic enclaves, neighborhood socioeconomic status, and obesity among Hispanic women in Chicago: a latent profile analysis approach. Cancer Causes Control. 2025;36:567-575. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 89. | Stout NL, Dierkes M, Oliveri JM, Rockson S, Paskett ED. The influence of non-cancer-related risk factors on the development of cancer-related lymphedema: a rapid review. Med Oncol. 2024;41:274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 90. | Nash SH, Loeffler BT, Verhage E, Sorensen J, Slater VE, Elenwa F, Erdrich J, Livermont T, Sanderson PR, Blackwater C, Smith BJ, Ulmer KK, McDowell BD. Comorbidities among American Indian and Alaska Native People with Cancer: A Surveillance, Epidemiology, and End Results-Medicare Study. Cancer Epidemiol Biomarkers Prev. 2025;34:1914-1923. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 91. | Tu Y, Ho KL, Dibble KE, Visvanathan K, Connor AE. The food environment and hypertension: A cross-sectional analysis in Black breast cancer survivors in Maryland. Cancer Epidemiol. 2024;92:102634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 92. | Wang L, Lai C, Wu Z, Luo H, Xu T. Association between daily sitting time and breast cancer among obese women: a nationwide population-based study. BMC Womens Health. 2025;25:337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 93. | Phothikul J, Chung J, Faro J, Seven M. Diet and Physical Activity Behaviors of Breast Cancer Survivors: A Scoping Review. Semin Oncol Nurs. 2025;41:151763. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 94. | Zou K, Zheng Y, Ren X, Cui W. Exploring the genetic intersection of dried fruit intake and breast cancer risk: a multi-trait genomic analysis with epidemiological context. J Health Popul Nutr. 2025;44:314. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 95. | Jang S, Hwang SO. The risk factors for premalignant and malignant endometrial polyps in premenopausal and postmenopausal women and trends over the past decade: A retrospective study in a single center, South Korea. Eur J Obstet Gynecol Reprod Biol. 2024;295:118-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 96. | Shadmani FK, Abedi Gheshlaghi L, Jafari-Khounigh A, Rezakhani L, Khazaei M. Burden and mortality of breast cancer attributed to diabetes and smoking in women: a secondary analysis based on Global Burden of Disease 2019 in North Africa and the Middle East. BMJ Open. 2025;15:e087178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 97. | Toss A, Piombino C, Quarello P, Trama A, Mascarin M, Lambertini M, Canesi M, Incorvaia L, Milano GM, Maruzzo M, Perrone F, Peccatori F, Ferrari A. Risk factors behind the increase of early-onset cancer in Italian adolescents and young adults: An investigation from the Italian AYA Working group. Eur J Cancer. 2024;212:115042. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 98. | Decker H, Graham L, Titan A, Hawn M, Kushel M, Kanzaria HK, Wick E. Housing Status and Cancer Screening in US Veterans. J Gen Intern Med. 2025;40:1297-1305. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 99. | von Euler-Chelpin M, Napolitano G, Lynge E, Borstrøm S, Vejborg I. Non-participation in breast screening in Denmark: Sociodemographic determinants. BMC Public Health. 2024;24:2024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 100. | Lorentzen EH, Chen YJ, Jones AL, Kantor O, King TA, Mittendorf EA, Minami CA. Omission of multimodal therapy in older adults with high-risk breast cancer. Breast Cancer Res Treat. 2025;212:337-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 101. | Zhang C, Xiao W, Wang H, Li L, Yang Y, Hao Y, Xu Z, Chen H, Nan W. Exosomes Derived from Mouse Breast Carcinoma Cells Facilitate Diabetic Wound Healing. Tissue Eng Regen Med. 2024;21:571-586. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 102. | Roy R, Man E, Aldakhlallah R, Gonzalez K, Merritt L, Daisy C, Lombardo M, Yordanova V, Sun L, Isaac B, Rockowitz S, Lotz M, Pories S, Moses MA. Mammary adipocytes promote breast tumor cell invasion and angiogenesis in the context of menopause and obesity. Biochim Biophys Acta Mol Basis Dis. 2024;1870:167325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 103. | Jin S, Zheng Y, Li D, Liu X, Zhu T, Wang S, Liu Z, Liu Y. Effect of genistein supplementation on microenvironment regulation of breast tumors in obese mice. Breast Cancer Res. 2024;26:147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 104. | Conner SJ, Borges HB, Guarin JR, Gerton TJ, Yui A, Salhany KJ Jr, Mensah DN, Hamilton GA, Le GH, Lew KC, Zhang C, Oudin MJ. Obesity Induces Temporally Regulated Alterations in the Extracellular Matrix That Drive Breast Tumor Invasion and Metastasis. Cancer Res. 2024;84:2761-2775. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 24] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 105. | Dupont S, Morsut L, Aragona M, Enzo E, Giulitti S, Cordenonsi M, Zanconato F, Le Digabel J, Forcato M, Bicciato S, Elvassore N, Piccolo S. Role of YAP/TAZ in mechanotransduction. Nature. 2011;474:179-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5248] [Cited by in RCA: 4541] [Article Influence: 302.7] [Reference Citation Analysis (3)] |
| 106. | Smith LA, Craven DM, Ho AN, Glenny EM, Rezeli ET, Carson MS, Paules EM, Fay M, Cozzo AJ, Hursting SD, Coleman MF. Weight Loss Reverses the Effects of Aging and Obesity on Mammary Tumor Immunosuppression and Progression. Cancer Prev Res (Phila). 2025;18:453-463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 107. | Pierro EW, Cottam MA, An H, Lehmann BD, Pietenpol JA, Wellen KE, Makowski L, Rathmell JC, Fingleton B, Hasty AH. Comparison of Lean, Obese, and Weight-Loss Models Reveals TREM2 Deficiency Attenuates Breast Cancer Growth Uniquely in Lean Mice and Alters Clonal T-cell Populations. Cancer Res. 2025;85:1219-1235. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 108. | Gaber M, Quentel A, Holmes J, Lepetit C, Triki H, Wilson A, Payne V, Tenvooren I, Dehours C, Peoples A, Duet ML, Katz AJ, Pécot T, Bougras-Cartron G, Cartron PF, Cook KL, Vidi PA. Obesity increases DNA damage in the breast epithelium. Breast Cancer Res. 2025;27:11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 109. | Karaman S, Lehti S, Zhang C, Taskinen MR, Käkelä R, Mardinoglu A, Brorson H, Alitalo K, Kivelä R. Multi-omics characterization of lymphedema-induced adipose tissue resulting from breast cancer-related surgery. FASEB J. 2024;38:e70097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 110. | Kuryłowicz A. Estrogens in Adipose Tissue Physiology and Obesity-Related Dysfunction. Biomedicines. 2023;11:690. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 108] [Reference Citation Analysis (0)] |
| 111. | Lu H, Yu X, Xu Z, Deng J, Zhang MJ, Zhang Y, Sun S. Prognostic Value of IGFBP6 in Breast Cancer: Focus on Glucometabolism. Technol Cancer Res Treat. 2024;23:15330338241271998. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 112. | Kim GE, Ibrahim AR, Shalatouni D, Abouzeid NH, Othman F. Paclitaxel-induced acute myocardial infarction: a case report and literature review. BMC Cardiovasc Disord. 2024;24:167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 113. | Pan B, Xu Y, Yao R, Cao X, Zhou X, Hao Z, Zhang Y, Wang C, Shen S, Luo Y, Zhu Q, Ren X, Kong L, Zhou Y, Sun Q. Nomogram prediction of the 70-gene signature (MammaPrint) binary and quartile categorized risk using medical history, imaging features and clinicopathological data among Chinese breast cancer patients. J Transl Med. 2023;21:798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 114. | Terroba G, Heman A, Saltijeral MT, Martínez P. [Attempted suicide among Mexican adolescents: significant clinical and sociodemographic factors]. Salud Publica Mex. 1986;28:48-55. [PubMed] |
| 115. | Kalupahana NS, Moustaid-Moussa N. Beyond blood pressure, fluid and electrolyte homeostasis - Role of the renin angiotensin aldosterone system in the interplay between metabolic diseases and breast cancer. Acta Physiol (Oxf). 2024;240:e14164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 116. | Mohammed AM, Hamed HB, Noaman MK, Alieldin N. Metabolic syndrome and breast cancer risk. J Egypt Natl Canc Inst. 2023;35:42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 117. | Townsel A, Jaffe M, Wu Y, Henry CJ, Haynes KA. The Epigenetic Landscape of Breast Cancer, Metabolism, and Obesity. Adv Exp Med Biol. 2024;1465:37-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 118. | Niu Y, Lin Z, Wan A, Chen H, Liang H, Sun L, Wang Y, Li X, Xiong XF, Wei B, Wu X, Wan G. RNA N6-methyladenosine demethylase FTO promotes breast tumor progression through inhibiting BNIP3. Mol Cancer. 2019;18:46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 248] [Cited by in RCA: 467] [Article Influence: 66.7] [Reference Citation Analysis (4)] |
| 119. | Ma Y, Ming Y, Hou Z, Yu Y, Liu J, Wang Z. Deciphering the Overlapping Immune Mechanism Between Depression and Breast Cancer. Int J Mol Sci. 2025;26:5229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 120. | Yang R, Li J, Zhang H, Liu H, Lian B, Li J. Exploring the molecular mechanisms of comorbidity between thyroid cancer and breast cancer through multi-omics data. Sci Rep. 2025;15:23309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 121. | Bai Y, Niu L, Song L, Dai G, Zhang W, He B, San W, Li S. Uncovering the effect and mechanism of Jiawei Xiaoyao Wan in treating breast cancer complicated with depression based on network pharmacology and experimental analysis. Phytomedicine. 2024;128:155427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 122. | Guo M, Wang T, Ge W, Ren C, Ko BC, Zeng X, Cao D. Role of AKR1B10 in inflammatory diseases. Scand J Immunol. 2024;100:e13390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 123. | Ungvari Z, Bartha Á, Ungvari A, Fekete M, Bianchini G, Győrffy B. Prognostic impact of glucagon-like peptide-1 receptor (GLP1R) expression on cancer survival and its implications for GLP-1R agonist therapy: an integrative analysis across multiple tumor types. Geroscience. 2025;47:4413-4427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 124. | Bi Y, Liu S, Wang L, Peng D, Chen W, Zhang Y, Wang Y. Mechanisms and Therapeutic Advances of PXR in Metabolic Diseases and Cancer. Int J Mol Sci. 2025;26:8029. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 125. | Huang J, Jin Y, Yi Z. The role of LGR4 in bone metabolism and tumor bone metastasis. Front Endocrinol (Lausanne). 2025;16:1541601. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 126. | Yang Y, Lu J, Zhu Y, Chen D, Tang J, Zhang M, Lu J, Yang Y, Tian S, Zhao H. Astragaloside IV inhibits the growth of obesity-associated triple-negative breast cancer by activating FOXA1 transcription factor to regulate GAL3ST1-GalCer signaling and remodel sphingolipid metabolism. Phytomedicine. 2025;144:156907. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 127. | Lee J, Bae SJ, Kim HK, Nam SJ, Kim HJ, Bae SY, Park HY, Ko BK, Park JH, Kwon Y, Park Y, Baek SH, Kook Y, Kim S, Lim YA, Kang HJ, Kim D, Jeong J, Ahn SG. Body mass index and progesterone receptor in postmenopausal ER-positive/HER2-negative breast cancer: A nation-wide study in Korean breast cancer society and the multi-institutional cohort. Breast. 2025;82:104515. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 128. | Hardell KNL, Schonfeld SJ, Ramin C, Vo JB, Morton LM. Association between diabetes and subsequent malignancy risk among older breast cancer survivors. JNCI Cancer Spectr. 2024;8:pkae036. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (21)] |
| 129. | Marcus Post L, Pathak DR, Hamilton AS, Hirko KA, Houang RT, Guseman EH, Sanfelippo D, Bohme Carnegie N, Olson LK, Rui H, Schwartz AG, Velie EM. Adiposity throughout Adulthood and Risk of Young-Onset Breast Cancer Tumor Subtypes in the Young Women's Health History Study. Cancer Epidemiol Biomarkers Prev. 2024;33:1659-1670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 130. | Proskuriakova E, Aryal BB, Shrestha DB, Valencia S, Kovalenko I, Adams M, Boxwala M, Verda L, Khosla PG. Impact of Obesity on Breast Cancer Clinicopathological Characteristics in Underserved US Community Safety-Net Hospital: A Retrospective Single-Center Study. Clin Breast Cancer. 2024;24:e714-e722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 131. | Vivoni S, Garratón F, Carro M, González-Sepúlveda L, Romaguera J, Umpierre S. Knowledge of Obesity's Health Related Outcomes among Hispanic Women living in Puerto Rico. P R Health Sci J. 2023;42:291-297. [PubMed] |
| 132. | Carmona-Gonzalez CA, Kumar S, Menjak IB. Current approaches to the pharmacological management of metastatic breast cancer in older women. Expert Opin Pharmacother. 2024;25:1785-1794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 133. | Vandraas KF, Hjorth S, Trewin-Nybråten CB, Ursin G, Botteri E, Andreassen BK, Reinertsen KV, Blix ES, Naume B, Støer NC. Anti-Her2 therapy patterns in metastatic breast cancer-Real-world data suggest undertreatment. Int J Cancer. 2026;158:633-641. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 134. | Rodrigues ED, Bento MJ, Calisto R, Rodrigues J, Almeida P, López E, Teixeira L. Utilisation patterns of radiotherapy among older patients: Insights from Portuguese National Cancer Registry data. J Cancer Policy. 2025;43:100522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 135. | García-Estévez L, Bardia A, Rugo HS, Carey LA, Diéras VC, Loibl S, Piccart M, Gianni L, Kalinsky K, O'Shaughnessy J, Hurvitz SA, Harting E, Valdez T, Phan S, Lai C, Cortés J. The association of high body mass index with the safety and efficacy of sacituzumab govitecan in patients with metastatic triple-negative breast cancer from the ASCENT study. ESMO Open. 2025;10:105294. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 136. | Hutajulu SH, Aresy S, Astari YK, Wiranata JA, Puspitaningtyas H, Sulistyoningrum DC, Taroeno-Hariadi KW, Kurnianda J, Purwanto I, Hardianti MS. Severe Deficiency of Vitamin D and Anthracycline-Taxane Regimen are associated with Cachexia Following Breast Cancer Chemotherapy: A Single Center Assessment Using Two Consensus-Based Criteria. Asian Pac J Cancer Prev. 2025;26:189-197. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 137. | Chala Y, Techane T, Bekele B, Girma T, Fekede W, Abdeta C, Mardasa K, Abdeta T, Teshome B, Atinkut G, Wako A, Adem M, Deksisa A. Time to breast cancer recurrence and associated predictors in Public Hospitals of Addis Ababa, Central Ethiopia: a retrospective Cohort Study. J Cancer Res Clin Oncol. 2025;151:187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 138. | Kong YH, Huang JY, Ding Y, Chen SH, Li QS, Xiong Y. The effect of BMI on survival outcome of breast cancer patients: a systematic review and meta-analysis. Clin Transl Oncol. 2025;27:403-416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 139. | Sarathi R, Sarumathy S, Durai Mavalavan VM. EVOLUTION OF METFORMIN IN BREAST CANCER THERAPY IN LAST TWO DECADES: A REVIEW. Exp Oncol. 2024;46:185-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 140. | Delibegović M, Dall'Angelo S, Dekeryte R. Protein tyrosine phosphatase 1B in metabolic diseases and drug development. Nat Rev Endocrinol. 2024;20:366-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 49] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 141. | Hechter RC, Chen LH, Shi J, Gu Z, Brady-Rogers M, Chlebowski RT, Haque R. Persistent prescription opioid use and all-cause mortality following the first-year breast cancer survivorship. JNCI Cancer Spectr. 2025;9:pkaf060. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 142. | Xu B, Kang B, Li S, Chen J, Zhou J. Association between sodium-glucose cotransporter 2 inhibitors and cancer: a systematic review and meta-analysis of randomized active-controlled trials. Int J Clin Pharm. 2025;47:1121-1131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 143. | Ceasovschih A, Asaftei A, Lupo MG, Kotlyarov S, Bartušková H, Balta A, Sorodoc V, Sorodoc L, Banach M. Glucagon-like peptide-1 receptor agonists and muscle mass effects. Pharmacol Res. 2025;220:107927. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 23] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 144. | Karimi MA, Khadem R, Haraj A, Farahani MA, Charehsaz Avari Firouzeh P, Azizinezhad A, Raki HK, Belbasi M, Bafrani MA, Deravi N, Khakpour Y, Habibi AF, Aghsaghloo V. The impact of sodium-glucose cotransporter-2 inhibitors on breast cancer and cancer-related mortality: a systematic review and meta-analysis of randomized controlled trials. BMC Cancer. 2025;25:991. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 145. | Fassio A, Porciello G, Carioli G, Palumbo E, Vitale S, Luongo A, Montagnese C, Prete M, Grimaldi M, Pica R, Rotondo E, Falzone L, Calabrese I, Minopoli A, Grilli B, Cuomo M, Fiorillo PC, Evangelista C, Cavalcanti E, De Laurentiis M, Cianniello D, Pacilio C, Pinto M, Thomas G, Rinaldo M, D'Aiuto M, Serraino D, Massarut S, Steffan A, Ferraù F, Rossello R, Messina F, Catalano F, Adami G, Bertoldo F, Libra M, Crispo A, Celentano E, La Vecchia C, Augustin LSA, Gatti D. Post-diagnosis serum 25-hydroxyvitamin D concentrations in women treated for breast cancer participating in a lifestyle trial in Italy. Reumatismo. 2024;76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 146. | Chai KC, Chou TC, Chen WM, Shia BC, Soong RS, Wu SY. Unlocking the Potential: Antihistamine Use and Breast Cancer Risk in Women With Type 2 Diabetes Mellitus. J Natl Compr Canc Netw. 2025;23:34-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 147. | Anson M, Poon JS, Henney AE, Riley D, Ibarbaru GH, Sieberhagen C, Cuthbertson DJ, Alam U, Hydes T. The chemoprotective effect of anti-platelet agents on cancer incidence in people with non-alcoholic fatty liver disease (NAFLD): a retrospective cohort study. BMC Med. 2024;22:574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 148. | Cespedes P, Martínez-Arnau FM, Torregrosa MD, Cauli O, Buigues C. Impact of a Physical Exercise and Health Education Program on Metabolic Syndrome and Quality of Life in Postmenopausal Breast Cancer Women Undergoing Adjuvant Treatment with Aromatase Inhibitors. Medicina (Kaunas). 2024;60:1893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 149. | Wen H, Deng G, Shi X, Liu Z, Lin A, Cheng Q, Zhang J, Luo P. Body mass index, weight change, and cancer prognosis: a meta-analysis and systematic review of 73 cohort studies. ESMO Open. 2024;9:102241. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 48] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 150. | Yaqoob SM, Haidar L, Allicock MA, Heredia NI. Does the Mediterranean Diet Play a Beneficial Role in Managing the Health of Overweight/Obese Breast Cancer Survivors? Nutrients. 2024;16:4214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 151. | Majumder KR, Shrivastava A, Maitra D, Siddique MI, Rahman MM. Fish-tail Plasty: A Secure Technique to Enhance Cosmesis at the Lateral End of Mastectomy Scar and Prevent Dog Ear. Mymensingh Med J. 2024;33:160-167. [PubMed] |
| 152. | Stringer EJ, Cloke RWG, Van der Meer L, Murphy RA, Macpherson NA, Lum JJ. The Clinical Impact of Time-restricted Eating on Cancer: A Systematic Review. Nutr Rev. 2025;83:e1660-e1676. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 153. | Hicks J, Dash C, Smith D, Hagberg J, Makambi K, Adams-Campbell L. Is exergaming a viable exercise option for obese black women? Contemp Clin Trials. 2025;151:107846. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 154. | Tome J, Khatib M, Nizri E, Grigg LM, Orbach L, Lahat G, Eldar SM, Abu-Abeid A. Breast Cancer Characteristics after Metabolic and Bariatric Surgery: A Matched Comparison to Patients with Severe Obesity. Obes Surg. 2025;35:2847-2853. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 155. | Madar LO, Goldberg N, Netz U, Berenstain IF, Abu Zeid EED, Avital I, Perry ZH. Association between metabolic and bariatric surgery and malignancy: a systematic review, meta-analysis, trends, and conclusions. Surg Obes Relat Dis. 2025;21:434-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 156. | Peila R, Rohan TE. Metabolic Syndrome and the Risk of Ductal Carcinoma In Situ of the Breast in the UK Biobank. Cancer Epidemiol Biomarkers Prev. 2024;33:333-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 157. | Alexander A, Brown N, Horns JJ, Hardikar S, Playdon M, Das R, Driggs N, Paudel N, Matsen C, Ibele A. Screening Mammography Adherence Improves After Bariatric Surgery. J Surg Res. 2024;303:476-481. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 158. | Alzate-Granados JP, Niño LF. Prediction of Overall and Relapse-Free Survival in Triple-Negative Breast Cancer Patients Through Machine Learning-Based Clustering on Clinical Data. Clin Breast Cancer. 2025;25:714-719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 159. | Wheeler SB, Rotter J, Spees LP, Biddell CB, Trogdon JG, Alfano CM, Mayer DK, Dinan MA, Nekhlyudov L, Birken SA. Machine Learning Risk Stratification for Older Breast Cancer Survivors: Clinical Care Implications. Health Serv Res. 2025;e70005. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 160. | Ho C, Ha NT, Youens D, Abhayaratna WP, Bulsara MK, Hughes JD, Mishra G, Pearson SA, Preen DB, Reid CM, Ruiter R, Saunders CM, Stricker BH, van Rooij FJA, Wright C, Moorin R. Association between long-term use of calcium channel blockers (CCB) and the risk of breast cancer: a retrospective longitudinal observational study protocol. BMJ Open. 2024;14:e080982. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 161. | Mukherjee A, Gu Z, Chen LH, Potosky AL, Haque R. Association of metabolic syndrome conditions with risk of second primary uterine cancer in breast cancer survivors. J Cancer Res Clin Oncol. 2023;149:17749-17755. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 162. | Kumaravel SS, Andisamy ND, Appadurai PD, Lakshmanamoorthy T, Raja AM, Kathiah R, Shanmugavinayagam A. A cross-sectional study on psychiatric comorbidity, coping strategies, and quality of life in women diagnosed with breast and cervical cancers attending a tertiary care center in South India. Ind Psychiatry J. 2024;33:312-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |