Published online Apr 24, 2026. doi: 10.5306/wjco.v17.i4.117197
Revised: January 5, 2026
Accepted: February 24, 2026
Published online: April 24, 2026
Processing time: 141 Days and 16.8 Hours
Pancreatic ductal adenocarcinoma has a dismal five-year survival rate of less than 12%, with therapeutic resistance stemming from its distinctive “cold tumor” microenvironment characterized by neural infiltration, stromal remodeling, and immune suppression - elements that urgently require an integrated framework. On the basis of mechanistic, histopathological, and clinical correlative evidence, this narrative review reveals bidirectional coupling among tumor-associated nerve fibers, cancer-associated fibroblasts, and immune cells, forming a “neuro-stromal-immune” network that promotes tumor progression. Here, we propose the “stroma-nerve axis (SNA)” framework, which conceptualizes nerve fibers and stromal cells as functional units of the pancreatic ductal adenocarcinoma microenvironment, and construct a hypothesis of SNA activation stratification: Three hypothetical subtypes (nerve-dominant SNA-I, stroma-dominant SNA-II, and balanced SNA-III) corresponding to distinct invasion patterns, immune ecologies, and therapeutic windows. Furthermore, we propose a conceptual SNA Activity Score centered on neural density, cancer-associated fibroblast activation, and inflammatory signaling as a hypothesis-generating tool for future research. We emphasize that the SNA Activity Score represents solely a theoretical model lacking prospective validation and cannot be applied to clinical decision-making. Finally, we critically analyze the evidence strength and limitations of the SNA framework and recommend testing this hypothesis through spatial omics, bioinformatics, and prospective studies.
Core Tip: We propose the novel “stroma-nerve axis” framework, conceptualizing nerve fibers and stromal components as a functional unit driving pancreatic ductal adenocarcinoma progression. By defining three hypothetical stroma-nerve axis activation subtypes (nerve-dominant, stroma-dominant, and balanced) and a conceptual Stroma-Nerve Axis Activity Score, this review offers a unified theoretical model to explain therapeutic resistance. This perspective shifts the paradigm from single-target interventions to systematic “network dismantling” strategies, providing a roadmap for future stratified clinical trials.
- Citation: Yu YN, Jin SQ, Lu YT, Han LZ. Stroma-nerve axis in pancreatic ductal adenocarcinoma: Bidirectional regulatory mechanisms and clinical translation hypothesis. World J Clin Oncol 2026; 17(4): 117197
- URL: https://www.wjgnet.com/2218-4333/full/v17/i4/117197.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i4.117197
Pancreatic ductal adenocarcinoma (PDAC) has a persistently dismal five-year survival rate of 11%-12%, representing one of the most formidable challenges in solid tumor therapeutics[1,2]. This intractable nature extends beyond delayed diagnosis or surgical limitations - even early-stage resectable cases achieve less than 30% five-year survival, indicating fundamental biological constraints[3]. Strategies that have achieved significant breakthroughs in other malignancies have consistently failed in patients with PDAC, as manifested by low objective response rates to immune checkpoint inhibitors, limited clinical benefit from emerging Kirsten rat sarcoma viral oncogene homolog G12C inhibitors, and plateauing improvements in conventional chemotherapy regimens[4,5]. This systematic therapeutic resistance directly stems from the unique tumor microenvironment of PDAC, which is characterized by dense fibrotic stroma comprising up to 90% of the tumor volume, severe immune-excluded phenotypes, and extensive neural infiltration[6,7]. These three elements synergistically construct a pathological ecosystem that is highly resistant to exogenous interventions.
Over the past two decades, PDAC microenvironment research has predominantly employed reductionist paradigms, dissecting complex systems into independent components: Stromal studies focusing on the pro-fibrotic functions of cancer-associated fibroblasts (CAFs), immunological investigations examining T-cell exhaustion, and neural research primarily viewing perineural invasion as a prognostic marker[8]. This “divide-and-conquer” strategy has repeatedly failed in clinical translation, most notably exemplified by the phase III HALO-301, which targeted hyaluronic acid; despite successful stromal depletion, this strategy failed to translate into a survival benefit and potentially increased metastatic risk by disrupting the stroma’s “mechanical restraint” on tumors[9]. Similarly, nonspecific CAF elimination resulted in compensatory immune suppression enhancement and increased tumor invasiveness[10]. These failures share a common oversight of dynamic reciprocity between microenvironmental components: Neurotransmitters reprogram CAFs toward inflammatory phenotypes, whereas activated CAFs secrete neurotrophic factors, creating “soil” for nerve regeneration and weaving a robust immune-exclusion network. These findings indicate that the PDAC microenvironment represents not only a summation of isolated components but also a highly adaptive integrated regulatory network in which linear single-node interventions readily trigger compensatory system feedback.
With advancements in research, neural infiltration, CAF-dominated stromal responses, and immune-suppressive microenvironments in pancreatic cancer have increasingly demonstrated interconnections through multiple signaling pathways and spatial structures rather than through isolated phenomena[11]. While previous work has separately revealed the roles of neurotransmitters, neurotrophic factors, CAF subpopulations, and immune cells in PDAC progression, a unifying perspective that systematically integrates these findings and explains clinical features such as invasive expansion and therapeutic resistance is lacking. Accordingly, this article formally proposes the “stroma-nerve axis (SNA)” concept, aiming to establish an integrative framework in which nerve fibers and stromal components are viewed as minimal functional units that jointly drive tumor progression. We reconstruct existing fragmented evidence from three dimensions - mechanistic interaction, spatiotemporal dynamics, and microenvironmental heterogeneity - and propose the SNA-I/II/III subtyping hypothesis and SNA Activity Score (SNAAS) concept. This article positions itself beyond traditional literature compilation, constructing a testable theoretical model within a narrative review framework, thereby providing novel perspectives for decoding PDAC heterogeneity and guiding stratified treatment[12].
This article employs a narrative review research paradigm, aiming to construct a theoretical framework through multidimensional evidence integration[13]. The literature search primarily covered the PubMed and Web of Science Core Collection databases, spanning from January 2000 to March 2025. The search strategies combined subject headings and free-text terms, with core search terms including “pancreatic ductal adenocarcinoma”, “stroma”, “cancer-associated fibroblast (CAF)”, “perineural invasion”, and “neuroimmune interaction”[14]. The inclusion criteria prioritized clinical pathological studies involving human PDAC samples, in vivo/in vitro experiments elucidating neuro-stromal interaction mechanisms, and key translational medicine research; purely descriptive reports lacking mechanistic exploration were excluded. Given the theoretical construction nature of this article, quantitative bias risk assessment was not implemented. Instead, following evidence pyramid principles, we conducted qualitative stratified analysis of different evidence tiers, including mechanistic experiments, observational studies, and clinical trials, with objective evaluation of their limitations[15,16].
The cognitive evolution of PDAC neural infiltration reflects a systematic leap from a singular pathomorphological description to a complex microenvironmental ecology. Early research (1980s-2000s) emphasized morphological observation, with Bockman et al[17] establishing perineural invasion as an independent prognostic factor, although neural infiltration was then viewed as a passive result of tumor spread along anatomical structures. Subsequent molecular mechanism studies (2000s-2015) revealed active process characteristics, with Ceyhan et al[18] elucidating how tumor cells induce axonal growth through the secretion of neurotrophic factors [e.g., nerve growth factor (NGF) and brain-derived neurotrophic factor] and the “neurotumor reciprocity” mechanism whereby neurotransmitters reciprocally promote tumor proliferation, although the research scope has remained confined primarily to direct bilateral interactions[18,19]. Since 2015, with the application of single-cell sequencing and spatial omics technologies, research has entered the systems biology phase[20]. Current evidence indicates that neural infiltration has become a core microenvironmental network component, with Schwann cells regulating immune and stromal microenvironments through reprogramming, sympathetic neural signals directly participating in CAF phenotype shaping, and nerve-dense regions spatially coupled with specific stromal remodeling and immune suppression features[21-23]. This evolution establishes neural infiltration during PDAC progression not only as a “pathway” but also as a key “hub” that actively regulates microenvironmental homeostasis[24].
The paradigm shift in stromal biology has undergone fundamental innovation from “static structure” to “dynamic function”[25]. Early perspectives (1990s-2005s) tended to view the fibrotic stroma as a physical barrier impeding drug delivery, leading to therapeutic strategies focused on mechanical stromal clearance. The turning point came from CAF heterogeneity analysis, with Öhlund et al[26] identifying myofibroblast CAF subpopulations that produce extracellular matrix and inflammatory CAFs that secrete inflammatory factors [interleukin (IL)-6, C-X-C motif chemokine 12 (CXCL12)], marking the cognitive transition from a passive scaffold to an active regulatory center. Recent key breakthroughs have revealed that stromal plasticity and upstream regulatory mechanisms by the nervous system, for example, norepinephrine, promote inflammatory CAF polarization, whereas cholinergic signals induce differentiated CAF phenotypes[27,28]. These findings indicate that the stroma represents not a rigid structure but rather an adaptive system capable of responding to neural signals and dynamically adjusting the immune microenvironment accordingly, laying the foundation for understanding its active defensive role in tumor progression and therapeutic resistance[29].
SNA theory emerges from cross-disciplinary integration of neurobiology and stromal biology, aiming to integrate independent discoveries from both fields. Zahalka et al[30] pioneering concept of neural regulation of CAFs was rapidly validated in PDAC, with subsequent research further refining this bidirectional circuit wherein neural signals activate CAFs while activated CAFs reciprocally secrete neurotrophic factors promoting nerve regeneration[31]. Bressy et al[31], Stopczynski et al[32] and spatial omics studies provided crucial experimental evidence for this theory, confirming that sympathetic denervation alters CAF composition and that CAF-derived neurotrophic factors are necessary for neural infiltration, with tight “functional coupling” in spatial distribution and functional states. Accordingly, the SNA framework represents an epistemological shift from the unidirectional “soil” metaphor to the multidirectional “ecosystem” concept. This paradigm reconstruction not only rationally explains systematic failures of single-target interventions due to network compensation but also suggests that future therapeutic strategies must transition from linear “target hitting” to more systematic “network dismantling” - disrupting mutually reinforcing positive feedback loops through synchronized intervention of neural and stromal nodes.
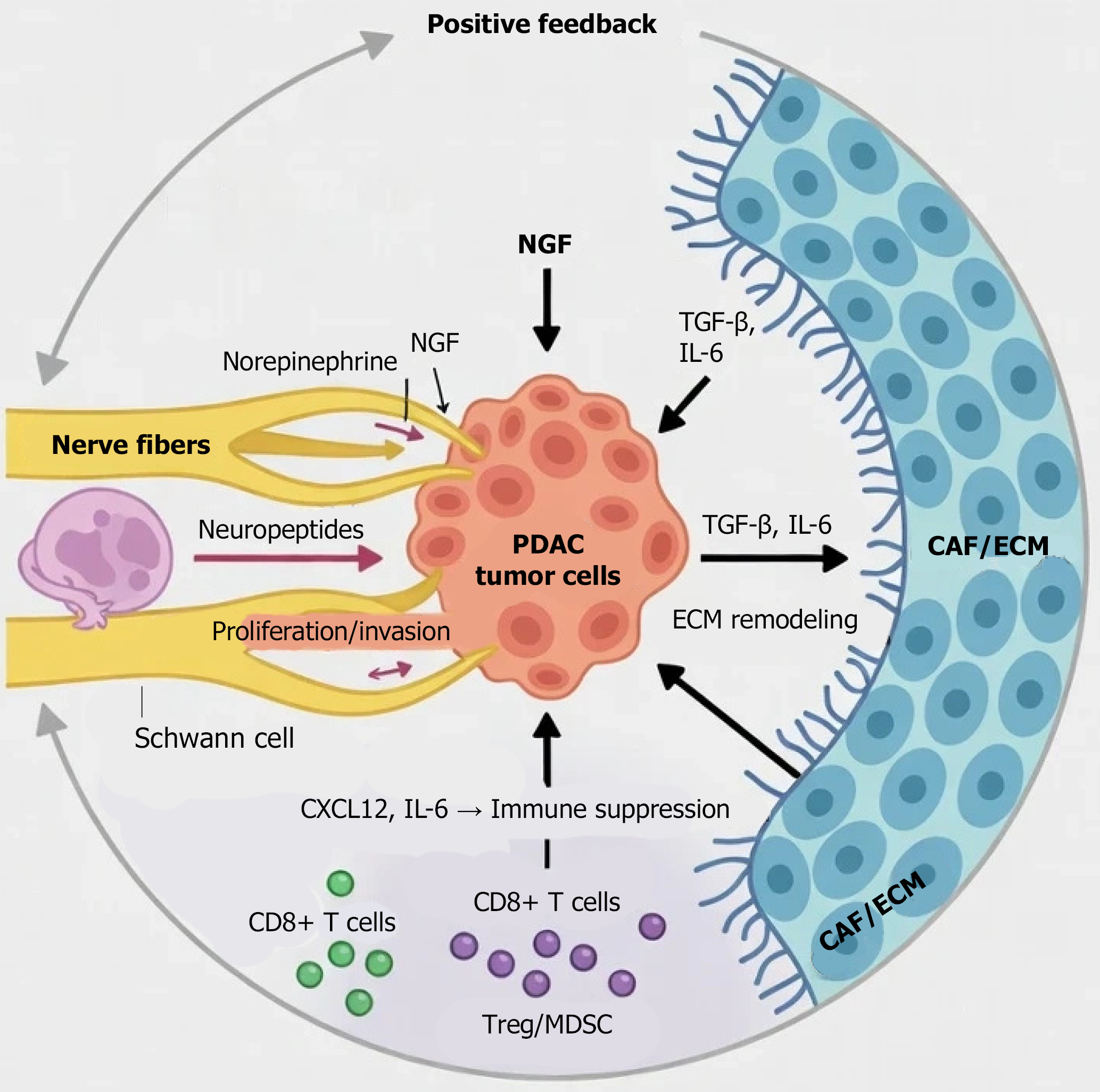
From the SNA perspective, the PDAC microenvironment represents not a simple summation of components but a complex ecological network constructed through bidirectional neural-stromal regulation as the hub[33]. First, tight signaling reciprocity exists among nerve-tumor-CAF triads: Tumor cells induce chemotactic regeneration and rearrangement of nerve fibers through the secretion of NGF and axon guidance molecules[34]; conversely, neurotransmitters released by neurons and Schwann cells (dopamine, norepinephrine) not only directly promote tumor proliferation and stemness maintenance but also more critically regulate CAF activation states and secretory profiles, thereby remodeling stromal density and mechanical properties[35-37].
Second, these physical and biochemical alterations collectively drive immune-suppressive microenvironment formation[38]. Activated CAFs deposit a dense extracellular matrix and increase interstitial fluid pressure, providing low-resistance “physical scaffolds” for tumor invasion along nerve bundles while constructing barriers limiting drug and immune cell infiltration[39-41]. Additionally, SNA network components synergistically secrete high levels of transforming growth factor (TGF)-β, IL-6, and CXCL12, leading to effector T-cell exhaustion and recruitment of Tregs and myeloid-derived suppressor cells[42-44]. This multidimensional interaction forms self-reinforcing positive feedback loops, progressively transforming the microenvironment from a reversible stress state to a highly stable therapeutic resistance state (Figure 1)[45].
SNA activation exhibits “initiation-amplification-lock-in” evolutionary characteristics along the temporal dimension[46]. In early lesions (initiation phase), neurotrophic factors secreted by tumor or precancerous cells induce local nerve fiber chemotaxis and Schwann cell phenotype remodeling, with a minimal stromal response at this stage, presenting as “nerve-dominant” microenvironmental perturbation[47]. As the disease progresses (amplification phase), CAF activation and stromal deposition significantly increase, and nerve fibers and dense stroma form tight spatial accompanying structures, synergistically amplifying proinvasive and immune-suppressive signals[48,49]. In advanced stages (lock-in phase), neural density, CAF activation, and immune exclusion form a stable triangular balance, maintaining high tumor invasiveness and multidrug resistance[50].
Notably, the above spatiotemporal dynamic model is derived primarily from mechanistic experiments and limited clinical observations. The exact temporal boundaries of each phase and correspondence with clinical staging await further validation through longitudinal spatial omics and functional imaging technologies[51].
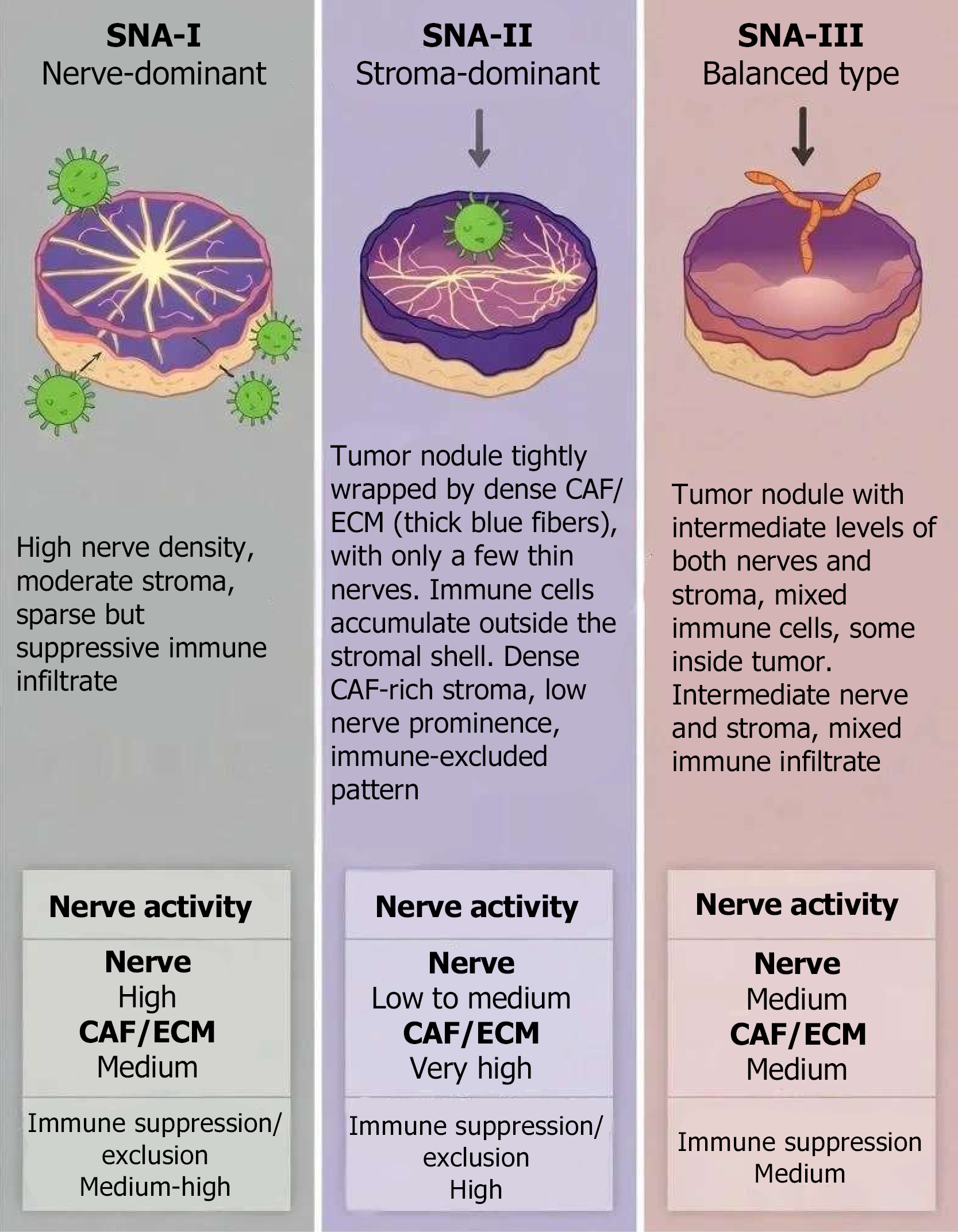
Current evidence suggests that nerve fiber density, CAF activation states, and immune infiltration patterns in the PDAC microenvironment exhibit coordinated variation patterns, suggesting potential molecular lineages underlying “SNA” activation[52]. Accordingly, we theoretically deconstruct SNA activation patterns into three hypothetical subtypes defined by distinct molecular and morphological criteria. First, SNA-I (nerve-dominant) is characterized morphologically by significant nerve fiber enrichment and molecularly by hyperactive neurotrophic and sympathetic signaling. We hypothesize that this subtype exhibits upregulated expression of neurotrophic factors (e.g., NGF, brain-derived neurotrophic factor) and axon guidance molecules (e.g., L1 cell adhesion molecule), alongside enriched adrenergic receptor signaling [e.g., alpha(2A)-adrenergic receptor] in tumor cells[53]. In such microenvironments, stromal responses remain relatively loose, and immune infiltration is sparse and biased toward suppression, with clinical phenotypes favoring perineural invasion and early pain.
In stark contrast, SNA-II (stroma-dominant) features massive proliferation of fibroblast activation protein (FAP)+/α-smooth muscle actin+ CAFs and dense stromal deposition[54]. Biologically, this subtype is likely driven by dominant TGF-β and inflammatory Janus kinase-signal transducer and activator of transcription signaling, enriched with matricellular proteins such as periostin to resemble the “activated stroma” signature described in transcriptomic studies[55]. These features result in high interstitial fluid pressure and construct typical “immune-excluded” barriers, where neural signals are secondary to physical drug resistance[56]. Between these extremes lies SNA-III (balanced type), with concurrent but intermediate expression of both neurogenic and fibrotic markers, jointly shaping diverse local niches. This implies that the invasion behavior and therapeutic responses of SNA-III may be the most heterogeneous (Figure 2).
Importantly, the above SNA-I/II/III subtyping currently remains a theoretical construct derived from mechanistic experiments, pathological observations, and limited clinical clues. While specific biomarkers (e.g., L1 cell adhesion molecule for SNA-I or periostin for SNA-II) provide potential targets for identification, this classification system has not undergone systematic validation through large-scale spatial transcriptomics or prospective cohorts. Therefore, it should not be misinterpreted as mature typing capable of directly guiding clinical decisions at this stage. The core purpose of proposing this hypothesis is to provide a logical framework for developing the SNAAS and to provide testable theoretical targets for future stratified. intervention research aimed at decoding PDAC heterogeneity.
Finally, delineating the intersection between these SNA phenotypes and established consensus molecular subtypes represents a critical frontier. We postulate that SNA-II (stroma-dominant), defined by dense FAP+ fibroblasts and inflammatory signaling, likely overlaps significantly with the “activated stroma” subtype described by Moffitt et al[55]. Conversely, SNA-I (nerve-dominant) may be intrinsically linked to the aggressive “basal-like” tumor phenotype, given that intense adrenergic signaling is known to drive epithelial-mesenchymal transition and dedifferentiation in PDAC cells. Integrating SNA profiling with these molecular classifications could therefore provide a higher-resolution map of PDAC heterogeneity, moving beyond simple “epithelial” or “stromal” dichotomies to capture the complete neuro-stromal ecosystem[57].
From the SNA perspective, perineural invasion is redefined as an active collaborative process among nerves, stroma, and tumors rather than mere morphological accompaniment[58]. Mechanistically, neurotrophic factors and axon guidance molecules secreted by tumor cells induce pathological nerve regeneration[59]; conversely, neurotransmitter (e.g., norepinephrine) and Schwann cell phenotype remodeling directly drive tumor epithelial-mesenchymal transition and migration[60]. Critically, CAFs construct “fiber track rails” rich in laminin and fibronectin through extracellular matrix rearrangement, providing low-resistance physical scaffolds for tumor cells migrating along nerves[61,62]. This neural-stromal structural coupling constitutes the physical foundation for the evolution of local invasion to distant metastasis.
However, heterogeneity in SNA activation patterns may determine differentiated metastatic lineages[63]. Theoretically, SNA-I (nerve-dominant) may favor early perineural invasion and pain occurrence, whereas SNA-II (stroma-dominant) may promote vascular compression and hematogenous metastatic risk through high interstitial pressure[64]. Current systematic evidence regarding the correspondence between different SNA subtypes and specific metastatic organ spectra (liver, lung, and peritoneum) and recurrence patterns is lacking, indicating that validation in prospective cohorts combined with spatial pathology is urgently needed.
PDAC “cold tumor” phenotype development results from dual mechanisms of physical isolation and functional suppression, with SNA playing a core maintenance role[65]. Physically, CAF-mediated dense stromal deposition and high interstitial fluid pressure construct mechanical barriers limiting effector T-cell infiltration, with nerve fibers and accompanying vascular structures further reinforcing this spatial compartmentalization[66]. Molecularly, the SNA network synergistically secretes high levels of TGF-β, IL-6, and CXCL12, inducing T-cell exhaustion and recruiting Tregs and myeloid-derived suppressor cells[67,68]; simultaneously, sympathetic neural signals can directly inhibit lymphocyte activity through β-adrenergic receptors, thereby establishing stable immune-suppressive niches[69].
Notably, these immune evasion mechanisms may exhibit differential weighting across SNA subtypes[70]. In nerve-dominant types, the functional suppression of immune cells by neurogenic stress signaling may predominate, whereas in stroma-dominant types, dense stroma-induced physical immune exclusion represents the primary contradiction[71]. This mechanistic divergence between “functional suppression” and “physical exclusion” suggests that future “cold tumor” reversal strategies require precise strategic adjustments on the basis of SNA subtyping rather than universal approaches.
SNAs constitute the systematic root of PDAC therapeutic resistance through maintaining microenvironmental homeostasis and shaping multilevel adaptability[72]. One aspect involves drug delivery barriers: CAF-induced stromal densification and vascular collapse limit the effective perfusion and distribution of chemotherapeutic agents in tumor core regions[73]. Another involves survival signal compensation: Under radiochemotherapy pressure, neural signals and inflammatory factor networks become activated, conferring enhanced tumor cell survival through the upregulation of DNA damage repair and antiapoptotic pathways[74]. Additionally, this network dynamically remodels the immune microenvironment, weakening immune checkpoint inhibitor efficacy and solidifying tumors into “locked” states that are tolerant to multiple interventions after multiple rounds of treatment[75].
Previous clinical trials targeting single nodes (e.g., pure stromal depletion or neural blockade) yielded contradictory results or worsened prognoses, fundamentally owing to overlooking SNA’s compensatory capacity as a homeostatic system - local removal of one “pillar” may instead disrupt mechanical restraint on highly invasive clones, inducing more aggressive phenotypes. Therefore, the key to truly reversing therapeutic resistance lies in adopting a “network dismantling” perspective, designing temporally rational multinode intervention protocols that disrupt SNA positive feedback loops while avoiding triggering malignant adaptive system evolution.
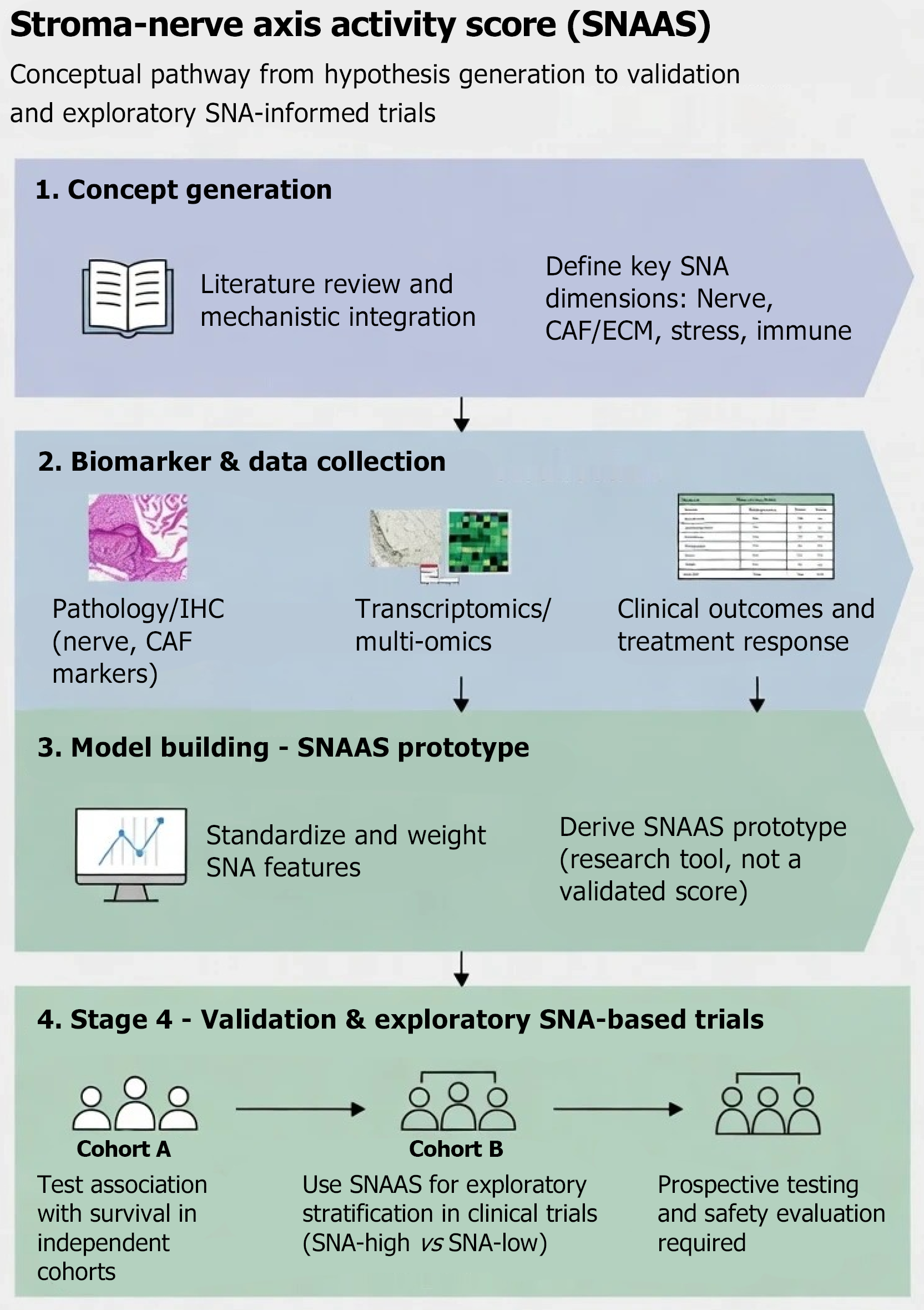
To translate SNA theory into operational research tools, this article proposes the conceptual framework of the SNAAS, aiming to quantitatively assess overall microenvironmental network activity[76]. SNAAS is conceived as a multidimensional weighted integration model. To operationalize this, we propose quantifying specific biomarkers via multiplex immunohistochemistry or digital pathology: (1) Neural activation (e.g., S-100+ or protein gene product 9.5+ nerve density area); (2) Stromal/CAF status (e.g., α-smooth muscle actin+/collagen ratio or FAP intensity); (3) Stress signal intensity [e.g., alpha(2A)-adrenergic receptor expression]; and (4) Immune suppression (e.g., factor forkhead box protein 3+/CD8+ T-cell ratio)[77]. We recommend that subsequent studies utilize machine learning algorithms (e.g., random forest or Cox regression) to determine the prognostic weight of each dimension, thereby transforming qualitative morphological descriptions into a continuous quantitative biological score for future stratification analysis (Figure 3).
On the basis of SNA’s “initiation-amplification-lock-in” evolutionary characteristics, intervention strategy formulation must possess strict temporal specificity[78]. In the prevention and early control phases, for genetically high-risk or chronic pancreatitis populations, lifestyle management or β-blocker intervention of systemic stress levels aims to reduce premature SNA axis activation; for precancerous lesions (pancreatic intraepithelial neoplasias), the combination of mild neural modulation and stromal “normalization” strategies may delay the microenvironmental transition from “perturbation” to “lock-in” states[79]. In locally advanced or neoadjuvant therapy phases, with mature SNA networks, intensified multinode intervention is needed - synchronously disrupting neural-stromal interaction circuits alongside chemotherapy to remodel the microenvironment and improve R0 resection rates[80]. In the postoperative phase, maintenance protocols should be tailored on the basis of residual SNA activity, with a focus on monitoring and blocking early neural-stromal network reconstruction in high-activity subgroups. This phase-specific strategy emphasizes matching intervention targets with microenvironmental maturity, avoiding blind drug stacking.
To address the highly coupled homeostatic system of PDAC, this article proposes SNA-stratified treatment concepts on the basis of “network dismantling”[81]. For SNA-I (nerve-dominant), we recommend the addition of neurostress axis blockers (e.g., nonselective β-blockers) and immune checkpoint inhibitors to standard therapy to weaken the neurogenic drive and relieve functional immune suppression[82]. For SNA-II (stroma dominant), the therapeutic focus should shift to stromal “normalization” strategies (e.g., vitamin D analogs or FAP-targeted modulation), aiming to reduce interstitial fluid pressure and improve drug perfusion rather than simple physical clearance[83]. For SNA-III (balanced type), with both neural and stromal networks active, more complex multitarget combination regimens may be needed but must be premised on strict dose titration and toxicity monitoring[84]. This stratified protocol provides a theoretical framework for future “basket trials” or biomarker-driven clinical research, aiming to replace current “one-size-fits-all” trial-and-error approaches[85].
The clinical translation of SNA-related strategies faces two challenges. The first is benefit/risk ratio uncertainty: Without prospective evidence supporting the predictive value of the SNAAS, implementing high-intensity combination interventions based solely on theoretical subtyping may increase unnecessary toxicity, violating the “do no harm” principle[86]. The second factor is accessibility and resource allocation: Refined SNA subtyping depends on complex pathological assessment or multiomics testing, potentially exacerbating health inequalities in resource-limited regions[87]. Therefore, future translational research should follow evidence-based medicine principles, prioritizing small-sample, exploratory mechanism validation trials to gradually establish biomarker efficacy boundaries, strictly preventing premature concept translation to routine clinical practice during hypothesis validation stages[88].
Surveying the literature, the evidence chain supporting the SNA framework exhibits a pronounced “strong mechanism, weak clinical” gradient decline[89]. At the microscopic mechanistic level, evidence is solid: Multiple in vivo/in vitro experiments have consistently confirmed that neuro-tumor chemotaxis, CAF-nerve bidirectional signaling, and proinvasive Schwann cell remodeling, establishing a stable biological foundation for SNA. At the histopathological level, evidence strength is intermediate: While correlations between high neural density and specific CAF markers with poor prognosis have received support from multiple retrospective studies, limited by inconsistent detection standards and sample heterogeneity, correlations have yet to translate into definitive causal inference[90]. At the intervention level, the evidence is the weakest and most controversial: Previous clinical trials targeting neural blockade or stromal targeting yielded complex or contradictory results, insufficient to prove that SNA state-based stratified intervention improves long-term survival[91]. Therefore, SNA should currently be viewed as an integrative theoretical derivation model, with clinical efficacy urgently requiring evidence gap closure “from mechanism to intervention”.
PDAC research has long reached “contradictory conclusions” regarding stromal clearance (pro-tumor vs anti-tumor), neural signals (sympathetic/parasympathetic effects), and immune phenotypes (exclusion vs exhaustion)[92,93]. The SNA framework provides a unified explanation for these paradoxes by introducing “context dependency”: These differences essentially reflect unrecognized SNA heterogeneity. For example, in stromal clearance controversies, for “stroma-dominant and network-locked” SNA-II types, nonspecific stromal depletion may disrupt “mechanical restraint” on highly invasive clones instead of inducing metastasis, whereas for early stromal remodeling SNA-I types, stromal intervention may be beneficial[94]. In immune phenotype controversies, SNA-II corresponds to physical barrier-induced “immune exclusion”, whereas other subtypes may exhibit more functional “immune exhaustion”[95]. In other words, contradictions in previous research largely stem from overlooking microenvironmental state stratification; reconstructing data with SNA subtypes as coordinates promises to unify fragmented observations within the same theoretical framework (Table 1).
| Controversial point | Supporting evidence (A) | Conflicting evidence (B) | SNA framework explanatory hypothesis (mechanistic detail) |
| Stromal depletion | FAP+ cell depletion or enzymatic degradation (e.g., PEGPH20) enhances drug delivery in select murine models[38] | Phase III HALO-301 trial failed to show survival benefit and paradoxically increased adverse events[9] | Disruption of mechanical restraint: Aggressive depletion in SNA-II (stroma-dominant) tumors may compromise the physical “capsule” that restrains tumor spread, as seen in α-SNA depletion models[91,92]. SNA theory favors “stromal normalization” over ablation to improve perfusion without breaching the barrier |
| Parasympathetic role | Activation of muscarinic receptors (e.g., M3R) promotes tumor cell proliferation and invasion in vitro[37] | Vagotomy (parasympathetic denervation) accelerates pancreatic cancer progression and metastasis in animal models[22] | Pathway context-dependency: The cholinergic anti-inflammatory pathway likely inhibits SNA-II associated inflammation. Thus, systemic vagotomy removes this “brake”, worsening the microenvironment. SNA theory posits that parasympathetic effects depend on the balance between direct tumor stimulation and indirect immune modulation |
| Neural blockade efficacy | Surgical or chemical denervation significantly slows tumor initiation and progression in genetically engineered mouse models[24] | Clinical trials targeting nerve signaling (e.g., neurolytic celiac plexus block) have largely failed to extend survival in unselected patients[95] | Dilution effect in unselected cohorts: Current trials treat PDAC as a uniform entity. We hypothesize that only SNA-I (nerve-dominant) tumors possess the high neurotrophic dependency required for response. In contrast, SNA-II tumors are driven by stromal signaling, rendering them refractory to neural blockade alone |
SNA theory faces fundamental limitations regarding model fidelity and causal inference[96]. Standard mouse models often fail to fully replicate the complex neuro-stromal architecture of human PDAC, and determining whether SNA activation is a driver of progression or merely a passenger phenomenon remains a critical challenge[97-99]. Therefore, the future validation roadmap must prioritize functional causality over simple correlation. We specifically recommend employing optogenetic or chemogenetic platforms to manipulate neural signaling with high spatiotemporal precision, alongside inducible dual-recombinase genetic models (e.g., Cre-loxP/FLP-FRT systems) to selectively deplete specific CAF subsets at defined disease stages. This rigorous progression - from multiomics association to in vivo manipulation - is essential to confirm the hierarchical role of the SNA network before clinical translation.
Building upon comprehensive research on neural infiltration, stromal remodeling, and the immune microenvironment, this article proposes the SNA, with its SNA-I/II/III subtyping and SNAAS conceptual models, to organize and explain key phenomena, including PDAC invasion, immune evasion, and therapeutic resistance. Current evidence supporting this framework remains primarily mechanistic and correlative, lacking systematic validation through large-sample cohorts and interventional trials. Therefore, it currently serves better as a theoretical tool for generating and organizing research hypotheses, with actual prognostic and therapeutic value awaiting further validation and refinement through prospective studies based on multiomics integration and stratified design.
| 1. | Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74:12-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7368] [Cited by in RCA: 6587] [Article Influence: 3293.5] [Reference Citation Analysis (4)] |
| 2. | Park W, Chawla A, O'Reilly EM. Pancreatic Cancer: A Review. JAMA. 2021;326:851-862. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1548] [Cited by in RCA: 1418] [Article Influence: 283.6] [Reference Citation Analysis (4)] |
| 3. | Neoptolemos JP, Kleeff J, Michl P, Costello E, Greenhalf W, Palmer DH. Therapeutic developments in pancreatic cancer: current and future perspectives. Nat Rev Gastroenterol Hepatol. 2018;15:333-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 967] [Cited by in RCA: 870] [Article Influence: 108.8] [Reference Citation Analysis (4)] |
| 4. | Bear AS, Vonderheide RH, O'Hara MH. Challenges and Opportunities for Pancreatic Cancer Immunotherapy. Cancer Cell. 2020;38:788-802. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 554] [Cited by in RCA: 501] [Article Influence: 83.5] [Reference Citation Analysis (4)] |
| 5. | Strickler JH, Satake H, George TJ, Yaeger R, Hollebecque A, Garrido-Laguna I, Schuler M, Burns TF, Coveler AL, Falchook GS, Vincent M, Sunakawa Y, Dahan L, Bajor D, Rha SY, Lemech C, Juric D, Rehn M, Ngarmchamnanrith G, Jafarinasabian P, Tran Q, Hong DS. Sotorasib in KRAS p.G12C-Mutated Advanced Pancreatic Cancer. N Engl J Med. 2023;388:33-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 265] [Cited by in RCA: 384] [Article Influence: 128.0] [Reference Citation Analysis (0)] |
| 6. | Sherman MH, Beatty GL. Tumor Microenvironment in Pancreatic Cancer Pathogenesis and Therapeutic Resistance. Annu Rev Pathol. 2023;18:123-148. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 366] [Cited by in RCA: 319] [Article Influence: 106.3] [Reference Citation Analysis (1)] |
| 7. | Gil Z, Cavel O, Kelly K, Brader P, Rein A, Gao SP, Carlson DL, Shah JP, Fong Y, Wong RJ. Paracrine regulation of pancreatic cancer cell invasion by peripheral nerves. J Natl Cancer Inst. 2010;102:107-118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 191] [Cited by in RCA: 199] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 8. | Hosein AN, Brekken RA, Maitra A. Pancreatic cancer stroma: an update on therapeutic targeting strategies. Nat Rev Gastroenterol Hepatol. 2020;17:487-505. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 800] [Cited by in RCA: 748] [Article Influence: 124.7] [Reference Citation Analysis (5)] |
| 9. | Van Cutsem E, Tempero MA, Sigal D, Oh DY, Fazio N, Macarulla T, Hitre E, Hammel P, Hendifar AE, Bates SE, Li CP, Hingorani SR, de la Fouchardiere C, Kasi A, Heinemann V, Maraveyas A, Bahary N, Layos L, Sahai V, Zheng L, Lacy J, Park JO, Portales F, Oberstein P, Wu W, Chondros D, Bullock AJ; HALO 109-301 Investigators. Randomized Phase III Trial of Pegvorhyaluronidase Alfa With Nab-Paclitaxel Plus Gemcitabine for Patients With Hyaluronan-High Metastatic Pancreatic Adenocarcinoma. J Clin Oncol. 2020;38:3185-3194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 126] [Cited by in RCA: 315] [Article Influence: 52.5] [Reference Citation Analysis (4)] |
| 10. | Chen Y, Kim J, Yang S, Wang H, Wu CJ, Sugimoto H, LeBleu VS, Kalluri R. Type I collagen deletion in αSMA(+) myofibroblasts augments immune suppression and accelerates progression of pancreatic cancer. Cancer Cell. 2021;39:548-565.e6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 494] [Cited by in RCA: 453] [Article Influence: 90.6] [Reference Citation Analysis (4)] |
| 11. | Halbrook CJ, Lyssiotis CA, Pasca di Magliano M, Maitra A. Pancreatic cancer: Advances and challenges. Cell. 2023;186:1729-1754. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1043] [Cited by in RCA: 921] [Article Influence: 307.0] [Reference Citation Analysis (2)] |
| 12. | Ho WJ, Jaffee EM, Zheng L. The tumour microenvironment in pancreatic cancer - clinical challenges and opportunities. Nat Rev Clin Oncol. 2020;17:527-540. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1052] [Cited by in RCA: 978] [Article Influence: 163.0] [Reference Citation Analysis (5)] |
| 13. | Ferrari R. Writing narrative style literature reviews. Med Writ. 2015;24:230-235. [DOI] [Full Text] |
| 14. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9803] [Reference Citation Analysis (0)] |
| 15. | Murad MH, Asi N, Alsawas M, Alahdab F. New evidence pyramid. Evid Based Med. 2016;21:125-127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1033] [Cited by in RCA: 794] [Article Influence: 79.4] [Reference Citation Analysis (0)] |
| 16. | Greenhalgh T, Thorne S, Malterud K. Time to challenge the spurious hierarchy of systematic over narrative reviews? Eur J Clin Invest. 2018;48:e12931. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 290] [Cited by in RCA: 474] [Article Influence: 59.3] [Reference Citation Analysis (0)] |
| 17. | Bockman DE, Büchler M, Beger HG. Interaction of pancreatic ductal carcinoma with nerves leads to nerve damage. Gastroenterology. 1994;107:219-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 125] [Article Influence: 3.9] [Reference Citation Analysis (3)] |
| 18. | Ceyhan GO, Bergmann F, Kadihasanoglu M, Altintas B, Demir IE, Hinz U, Müller MW, Giese T, Büchler MW, Giese NA, Friess H. Pancreatic neuropathy and neuropathic pain--a comprehensive pathomorphological study of 546 cases. Gastroenterology. 2009;136:177-186.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 327] [Cited by in RCA: 295] [Article Influence: 17.4] [Reference Citation Analysis (2)] |
| 19. | Bakst RL, Xiong H, Chen CH, Deborde S, Lyubchik A, Zhou Y, He S, McNamara W, Lee SY, Olson OC, Leiner IM, Marcadis AR, Keith JW, Al-Ahmadie HA, Katabi N, Gil Z, Vakiani E, Joyce JA, Pamer E, Wong RJ. Inflammatory Monocytes Promote Perineural Invasion via CCL2-Mediated Recruitment and Cathepsin B Expression. Cancer Res. 2017;77:6400-6414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 104] [Article Influence: 11.6] [Reference Citation Analysis (3)] |
| 20. | Peng J, Sun BF, Chen CY, Zhou JY, Chen YS, Chen H, Liu L, Huang D, Jiang J, Cui GS, Yang Y, Wang W, Guo D, Dai M, Guo J, Zhang T, Liao Q, Liu Y, Zhao YL, Han DL, Zhao Y, Yang YG, Wu W. Single-cell RNA-seq highlights intra-tumoral heterogeneity and malignant progression in pancreatic ductal adenocarcinoma. Cell Res. 2019;29:725-738. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1114] [Cited by in RCA: 949] [Article Influence: 135.6] [Reference Citation Analysis (4)] |
| 21. | Deborde S, Omelchenko T, Lyubchik A, Zhou Y, He S, McNamara WF, Chernichenko N, Lee SY, Barajas F, Chen CH, Bakst RL, Vakiani E, He S, Hall A, Wong RJ. Schwann cells induce cancer cell dispersion and invasion. J Clin Invest. 2016;126:1538-1554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 223] [Article Influence: 22.3] [Reference Citation Analysis (3)] |
| 22. | Renz BW, Takahashi R, Tanaka T, Macchini M, Hayakawa Y, Dantes Z, Maurer HC, Chen X, Jiang Z, Westphalen CB, Ilmer M, Valenti G, Mohanta SK, Habenicht AJR, Middelhoff M, Chu T, Nagar K, Tailor Y, Casadei R, Di Marco M, Kleespies A, Friedman RA, Remotti H, Reichert M, Worthley DL, Neumann J, Werner J, Iuga AC, Olive KP, Wang TC. β2 Adrenergic-Neurotrophin Feedforward Loop Promotes Pancreatic Cancer. Cancer Cell. 2018;33:75-90.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 242] [Cited by in RCA: 388] [Article Influence: 48.5] [Reference Citation Analysis (0)] |
| 23. | Murakami T, Hiroshima Y, Matsuyama R, Homma Y, Hoffman RM, Endo I. Role of the tumor microenvironment in pancreatic cancer. Ann Gastroenterol Surg. 2019;3:130-137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 146] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 24. | Saloman JL, Albers KM, Li D, Hartman DJ, Crawford HC, Muha EA, Rhim AD, Davis BM. Ablation of sensory neurons in a genetic model of pancreatic ductal adenocarcinoma slows initiation and progression of cancer. Proc Natl Acad Sci U S A. 2016;113:3078-3083. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 355] [Cited by in RCA: 329] [Article Influence: 32.9] [Reference Citation Analysis (4)] |
| 25. | Sahai E, Astsaturov I, Cukierman E, DeNardo DG, Egeblad M, Evans RM, Fearon D, Greten FR, Hingorani SR, Hunter T, Hynes RO, Jain RK, Janowitz T, Jorgensen C, Kimmelman AC, Kolonin MG, Maki RG, Powers RS, Puré E, Ramirez DC, Scherz-Shouval R, Sherman MH, Stewart S, Tlsty TD, Tuveson DA, Watt FM, Weaver V, Weeraratna AT, Werb Z. A framework for advancing our understanding of cancer-associated fibroblasts. Nat Rev Cancer. 2020;20:174-186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3212] [Cited by in RCA: 2865] [Article Influence: 477.5] [Reference Citation Analysis (5)] |
| 26. | Öhlund D, Handly-Santana A, Biffi G, Elyada E, Almeida AS, Ponz-Sarvise M, Corbo V, Oni TE, Hearn SA, Lee EJ, Chio II, Hwang CI, Tiriac H, Baker LA, Engle DD, Feig C, Kultti A, Egeblad M, Fearon DT, Crawford JM, Clevers H, Park Y, Tuveson DA. Distinct populations of inflammatory fibroblasts and myofibroblasts in pancreatic cancer. J Exp Med. 2017;214:579-596. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2219] [Cited by in RCA: 1988] [Article Influence: 220.9] [Reference Citation Analysis (4)] |
| 27. | Huang H, Wang Z, Zhang Y, Pradhan RN, Ganguly D, Chandra R, Murimwa G, Wright S, Gu X, Maddipati R, Müller S, Turley SJ, Brekken RA. Mesothelial cell-derived antigen-presenting cancer-associated fibroblasts induce expansion of regulatory T cells in pancreatic cancer. Cancer Cell. 2022;40:656-673.e7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 459] [Cited by in RCA: 416] [Article Influence: 104.0] [Reference Citation Analysis (4)] |
| 28. | Grünwald BT, Devisme A, Andrieux G, Vyas F, Aliar K, McCloskey CW, Macklin A, Jang GH, Denroche R, Romero JM, Bavi P, Bronsert P, Notta F, O'Kane G, Wilson J, Knox J, Tamblyn L, Udaskin M, Radulovich N, Fischer SE, Boerries M, Gallinger S, Kislinger T, Khokha R. Spatially confined sub-tumor microenvironments in pancreatic cancer. Cell. 2021;184:5577-5592.e18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 319] [Article Influence: 63.8] [Reference Citation Analysis (1)] |
| 29. | Garcia PE, Scales MK, Allen BL, Pasca di Magliano M. Pancreatic Fibroblast Heterogeneity: From Development to Cancer. Cells. 2020;9:2464. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 41] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 30. | Zahalka AH, Arnal-Estapé A, Maryanovich M, Nakahara F, Cruz CD, Finley LWS, Frenette PS. Adrenergic nerves activate an angio-metabolic switch in prostate cancer. Science. 2017;358:321-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 418] [Article Influence: 52.3] [Reference Citation Analysis (2)] |
| 31. | Bressy C, Lac S, Nigri J, Leca J, Roques J, Lavaut MN, Secq V, Guillaumond F, Bui TT, Pietrasz D, Granjeaud S, Bachet JB, Ouaissi M, Iovanna J, Vasseur S, Tomasini R. LIF Drives Neural Remodeling in Pancreatic Cancer and Offers a New Candidate Biomarker. Cancer Res. 2018;78:909-921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 89] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 32. | Stopczynski RE, Normolle DP, Hartman DJ, Ying H, DeBerry JJ, Bielefeldt K, Rhim AD, DePinho RA, Albers KM, Davis BM. Neuroplastic changes occur early in the development of pancreatic ductal adenocarcinoma. Cancer Res. 2014;74:1718-1727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 169] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 33. | Zahalka AH, Frenette PS. Nerves in cancer. Nat Rev Cancer. 2020;20:143-157. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 374] [Cited by in RCA: 338] [Article Influence: 56.3] [Reference Citation Analysis (0)] |
| 34. | Jurcak NR, Rucki AA, Muth S, Thompson E, Sharma R, Ding D, Zhu Q, Eshleman JR, Anders RA, Jaffee EM, Fujiwara K, Zheng L. Axon Guidance Molecules Promote Perineural Invasion and Metastasis of Orthotopic Pancreatic Tumors in Mice. Gastroenterology. 2019;157:838-850.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 160] [Article Influence: 22.9] [Reference Citation Analysis (4)] |
| 35. | Thiel V, Renders S, Panten J, Dross N, Bauer K, Azorin D, Henriques V, Vogel V, Klein C, Leppä AM, Barriuso Ortega I, Schwickert J, Ourailidis I, Mochayedi J, Mallm JP, Müller-Tidow C, Monyer H, Neoptolemos J, Hackert T, Stegle O, Odom DT, Offringa R, Stenzinger A, Winkler F, Sprick M, Trumpp A. Characterization of single neurons reprogrammed by pancreatic cancer. Nature. 2025;640:1042-1051. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 75] [Article Influence: 75.0] [Reference Citation Analysis (0)] |
| 36. | Zhang Y, Chen F, Chandrashekar DS, Varambally S, Creighton CJ. Proteogenomic characterization of 2002 human cancers reveals pan-cancer molecular subtypes and associated pathways. Nat Commun. 2022;13:2669. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 147] [Article Influence: 36.8] [Reference Citation Analysis (4)] |
| 37. | Jiang SH, Hu LP, Wang X, Li J, Zhang ZG. Neurotransmitters: emerging targets in cancer. Oncogene. 2020;39:503-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 169] [Article Influence: 24.1] [Reference Citation Analysis (3)] |
| 38. | Feig C, Jones JO, Kraman M, Wells RJ, Deonarine A, Chan DS, Connell CM, Roberts EW, Zhao Q, Caballero OL, Teichmann SA, Janowitz T, Jodrell DI, Tuveson DA, Fearon DT. Targeting CXCL12 from FAP-expressing carcinoma-associated fibroblasts synergizes with anti-PD-L1 immunotherapy in pancreatic cancer. Proc Natl Acad Sci U S A. 2013;110:20212-20217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1768] [Cited by in RCA: 1666] [Article Influence: 128.2] [Reference Citation Analysis (4)] |
| 39. | DuFort CC, DelGiorno KE, Carlson MA, Osgood RJ, Zhao C, Huang Z, Thompson CB, Connor RJ, Thanos CD, Scott Brockenbrough J, Provenzano PP, Frost GI, Michael Shepard H, Hingorani SR. Interstitial Pressure in Pancreatic Ductal Adenocarcinoma Is Dominated by a Gel-Fluid Phase. Biophys J. 2016;110:2106-2119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 154] [Cited by in RCA: 144] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 40. | Laklai H, Miroshnikova YA, Pickup MW, Collisson EA, Kim GE, Barrett AS, Hill RC, Lakins JN, Schlaepfer DD, Mouw JK, LeBleu VS, Roy N, Novitskiy SV, Johansen JS, Poli V, Kalluri R, Iacobuzio-Donahue CA, Wood LD, Hebrok M, Hansen K, Moses HL, Weaver VM. Genotype tunes pancreatic ductal adenocarcinoma tissue tension to induce matricellular fibrosis and tumor progression. Nat Med. 2016;22:497-505. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 449] [Cited by in RCA: 501] [Article Influence: 50.1] [Reference Citation Analysis (0)] |
| 41. | Jiang H, Torphy RJ, Steiger K, Hongo H, Ritchie AJ, Kriegsmann M, Horst D, Umetsu SE, Joseph NM, McGregor K, Pishvaian MJ, Blais EM, Lu B, Li M, Hollingsworth M, Stashko C, Volmar K, Yeh JJ, Weaver VM, Wang ZJ, Tempero MA, Weichert W, Collisson EA. Pancreatic ductal adenocarcinoma progression is restrained by stromal matrix. J Clin Invest. 2020;130:4704-4709. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 103] [Article Influence: 17.2] [Reference Citation Analysis (5)] |
| 42. | Biffi G, Oni TE, Spielman B, Hao Y, Elyada E, Park Y, Preall J, Tuveson DA. IL1-Induced JAK/STAT Signaling Is Antagonized by TGFβ to Shape CAF Heterogeneity in Pancreatic Ductal Adenocarcinoma. Cancer Discov. 2019;9:282-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1163] [Cited by in RCA: 1060] [Article Influence: 151.4] [Reference Citation Analysis (3)] |
| 43. | Li K, Shi H, Zhang B, Ou X, Ma Q, Chen Y, Shu P, Li D, Wang Y. Myeloid-derived suppressor cells as immunosuppressive regulators and therapeutic targets in cancer. Signal Transduct Target Ther. 2021;6:362. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 736] [Article Influence: 147.2] [Reference Citation Analysis (5)] |
| 44. | Zhang Y, Lazarus J, Steele NG, Yan W, Lee HJ, Nwosu ZC, Halbrook CJ, Menjivar RE, Kemp SB, Sirihorachai VR, Velez-Delgado A, Donahue K, Carpenter ES, Brown KL, Irizarry-Negron V, Nevison AC, Vinta A, Anderson MA, Crawford HC, Lyssiotis CA, Frankel TL, Bednar F, Pasca di Magliano M. Regulatory T-cell Depletion Alters the Tumor Microenvironment and Accelerates Pancreatic Carcinogenesis. Cancer Discov. 2020;10:422-439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 295] [Article Influence: 49.2] [Reference Citation Analysis (0)] |
| 45. | Hutton C, Heider F, Blanco-Gomez A, Banyard A, Kononov A, Zhang X, Karim S, Paulus-Hock V, Watt D, Steele N, Kemp S, Hogg EKJ, Kelly J, Jackstadt RF, Lopes F, Menotti M, Chisholm L, Lamarca A, Valle J, Sansom OJ, Springer C, Malliri A, Marais R, Pasca di Magliano M, Zelenay S, Morton JP, Jørgensen C. Single-cell analysis defines a pancreatic fibroblast lineage that supports anti-tumor immunity. Cancer Cell. 2021;39:1227-1244.e20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 264] [Article Influence: 52.8] [Reference Citation Analysis (0)] |
| 46. | Mace TA, Shakya R, Pitarresi JR, Swanson B, McQuinn CW, Loftus S, Nordquist E, Cruz-Monserrate Z, Yu L, Young G, Zhong X, Zimmers TA, Ostrowski MC, Ludwig T, Bloomston M, Bekaii-Saab T, Lesinski GB. IL-6 and PD-L1 antibody blockade combination therapy reduces tumour progression in murine models of pancreatic cancer. Gut. 2018;67:320-332. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 458] [Cited by in RCA: 442] [Article Influence: 55.3] [Reference Citation Analysis (5)] |
| 47. | Zhu Y, Herndon JM, Sojka DK, Kim KW, Knolhoff BL, Zuo C, Cullinan DR, Luo J, Bearden AR, Lavine KJ, Yokoyama WM, Hawkins WG, Fields RC, Randolph GJ, DeNardo DG. Tissue-Resident Macrophages in Pancreatic Ductal Adenocarcinoma Originate from Embryonic Hematopoiesis and Promote Tumor Progression. Immunity. 2017;47:323-338.e6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 595] [Cited by in RCA: 534] [Article Influence: 59.3] [Reference Citation Analysis (0)] |
| 48. | Elyada E, Bolisetty M, Laise P, Flynn WF, Courtois ET, Burkhart RA, Teinor JA, Belleau P, Biffi G, Lucito MS, Sivajothi S, Armstrong TD, Engle DD, Yu KH, Hao Y, Wolfgang CL, Park Y, Preall J, Jaffee EM, Califano A, Robson P, Tuveson DA. Cross-Species Single-Cell Analysis of Pancreatic Ductal Adenocarcinoma Reveals Antigen-Presenting Cancer-Associated Fibroblasts. Cancer Discov. 2019;9:1102-1123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1841] [Cited by in RCA: 1646] [Article Influence: 235.1] [Reference Citation Analysis (3)] |
| 49. | Krishnamurty AT, Shyer JA, Thai M, Gandham V, Buechler MB, Yang YA, Pradhan RN, Wang AW, Sanchez PL, Qu Y, Breart B, Chalouni C, Dunlap D, Ziai J, Elstrott J, Zacharias N, Mao W, Rowntree RK, Sadowsky J, Lewis GD, Pillow TH, Nabet BY, Banchereau R, Tam L, Caothien R, Bacarro N, Roose-Girma M, Modrusan Z, Mariathasan S, Müller S, Turley SJ. LRRC15(+) myofibroblasts dictate the stromal setpoint to suppress tumour immunity. Nature. 2022;611:148-154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 301] [Article Influence: 75.3] [Reference Citation Analysis (1)] |
| 50. | Dominguez CX, Müller S, Keerthivasan S, Koeppen H, Hung J, Gierke S, Breart B, Foreman O, Bainbridge TW, Castiglioni A, Senbabaoglu Y, Modrusan Z, Liang Y, Junttila MR, Klijn C, Bourgon R, Turley SJ. Single-Cell RNA Sequencing Reveals Stromal Evolution into LRRC15(+) Myofibroblasts as a Determinant of Patient Response to Cancer Immunotherapy. Cancer Discov. 2020;10:232-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 243] [Cited by in RCA: 648] [Article Influence: 92.6] [Reference Citation Analysis (3)] |
| 51. | Hwang WL, Jagadeesh KA, Guo JA, Hoffman HI, Yadollahpour P, Reeves JW, Mohan R, Drokhlyansky E, Van Wittenberghe N, Ashenberg O, Farhi SL, Schapiro D, Divakar P, Miller E, Zollinger DR, Eng G, Schenkel JM, Su J, Shiau C, Yu P, Freed-Pastor WA, Abbondanza D, Mehta A, Gould J, Lambden C, Porter CBM, Tsankov A, Dionne D, Waldman J, Cuoco MS, Nguyen L, Delorey T, Phillips D, Barth JL, Kem M, Rodrigues C, Ciprani D, Roldan J, Zelga P, Jorgji V, Chen JH, Ely Z, Zhao D, Fuhrman K, Fropf R, Beechem JM, Loeffler JS, Ryan DP, Weekes CD, Ferrone CR, Qadan M, Aryee MJ, Jain RK, Neuberg DS, Wo JY, Hong TS, Xavier R, Aguirre AJ, Rozenblatt-Rosen O, Mino-Kenudson M, Castillo CF, Liss AS, Ting DT, Jacks T, Regev A. Single-nucleus and spatial transcriptome profiling of pancreatic cancer identifies multicellular dynamics associated with neoadjuvant treatment. Nat Genet. 2022;54:1178-1191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 311] [Article Influence: 77.8] [Reference Citation Analysis (0)] |
| 52. | Moncada R, Barkley D, Wagner F, Chiodin M, Devlin JC, Baron M, Hajdu CH, Simeone DM, Yanai I. Integrating microarray-based spatial transcriptomics and single-cell RNA-seq reveals tissue architecture in pancreatic ductal adenocarcinomas. Nat Biotechnol. 2020;38:333-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 352] [Cited by in RCA: 736] [Article Influence: 122.7] [Reference Citation Analysis (0)] |
| 53. | Banh RS, Biancur DE, Yamamoto K, Sohn ASW, Walters B, Kuljanin M, Gikandi A, Wang H, Mancias JD, Schneider RJ, Pacold ME, Kimmelman AC. Neurons Release Serine to Support mRNA Translation in Pancreatic Cancer. Cell. 2020;183:1202-1218.e25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 237] [Cited by in RCA: 227] [Article Influence: 37.8] [Reference Citation Analysis (0)] |
| 54. | Zhou B, Gao S. Pan-Cancer Analysis of FURIN as a Potential Prognostic and Immunological Biomarker. Front Mol Biosci. 2021;8:648402. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 55. | Moffitt RA, Marayati R, Flate EL, Volmar KE, Loeza SG, Hoadley KA, Rashid NU, Williams LA, Eaton SC, Chung AH, Smyla JK, Anderson JM, Kim HJ, Bentrem DJ, Talamonti MS, Iacobuzio-Donahue CA, Hollingsworth MA, Yeh JJ. Virtual microdissection identifies distinct tumor- and stroma-specific subtypes of pancreatic ductal adenocarcinoma. Nat Genet. 2015;47:1168-1178. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1898] [Cited by in RCA: 1686] [Article Influence: 153.3] [Reference Citation Analysis (5)] |
| 56. | Pereira BA, Vennin C, Papanicolaou M, Chambers CR, Herrmann D, Morton JP, Cox TR, Timpson P. CAF Subpopulations: A New Reservoir of Stromal Targets in Pancreatic Cancer. Trends Cancer. 2019;5:724-741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 236] [Article Influence: 33.7] [Reference Citation Analysis (3)] |
| 57. | Collisson EA, Sadanandam A, Olson P, Gibb WJ, Truitt M, Gu S, Cooc J, Weinkle J, Kim GE, Jakkula L, Feiler HS, Ko AH, Olshen AB, Danenberg KL, Tempero MA, Spellman PT, Hanahan D, Gray JW. Subtypes of pancreatic ductal adenocarcinoma and their differing responses to therapy. Nat Med. 2011;17:500-503. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1688] [Cited by in RCA: 1481] [Article Influence: 98.7] [Reference Citation Analysis (4)] |
| 58. | Xu Z, Pothula SP, Wilson JS, Apte MV. Pancreatic cancer and its stroma: a conspiracy theory. World J Gastroenterol. 2014;20:11216-11229. [PubMed] [DOI] [Full Text] |
| 59. | Secq V, Leca J, Bressy C, Guillaumond F, Skrobuk P, Nigri J, Lac S, Lavaut MN, Bui TT, Thakur AK, Callizot N, Steinschneider R, Berthezene P, Dusetti N, Ouaissi M, Moutardier V, Calvo E, Bousquet C, Garcia S, Bidaut G, Vasseur S, Iovanna JL, Tomasini R. Stromal SLIT2 impacts on pancreatic cancer-associated neural remodeling. Cell Death Dis. 2015;6:e1592. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 74] [Article Influence: 6.7] [Reference Citation Analysis (4)] |
| 60. | Li J, Kang R, Tang D. Cellular and molecular mechanisms of perineural invasion of pancreatic ductal adenocarcinoma. Cancer Commun (Lond). 2021;41:642-660. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 94] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 61. | Tao J, Yang G, Zhou W, Qiu J, Chen G, Luo W, Zhao F, You L, Zheng L, Zhang T, Zhao Y. Targeting hypoxic tumor microenvironment in pancreatic cancer. J Hematol Oncol. 2021;14:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 344] [Cited by in RCA: 306] [Article Influence: 61.2] [Reference Citation Analysis (4)] |
| 62. | Pothula SP, Xu Z, Goldstein D, Pirola RC, Wilson JS, Apte MV. Targeting HGF/c-MET Axis in Pancreatic Cancer. Int J Mol Sci. 2020;21:9170. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 61] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 63. | Wang S, Li Y, Xing C, Ding C, Zhang H, Chen L, You L, Dai M, Zhao Y. Tumor microenvironment in chemoresistance, metastasis and immunotherapy of pancreatic cancer. Am J Cancer Res. 2020;10:1937-1953. [PubMed] |
| 64. | Aiello NM, Kang Y. Context-dependent EMT programs in cancer metastasis. J Exp Med. 2019;216:1016-1026. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 327] [Cited by in RCA: 449] [Article Influence: 64.1] [Reference Citation Analysis (6)] |
| 65. | Hegde S, Krisnawan VE, Herzog BH, Zuo C, Breden MA, Knolhoff BL, Hogg GD, Tang JP, Baer JM, Mpoy C, Lee KB, Alexander KA, Rogers BE, Murphy KM, Hawkins WG, Fields RC, DeSelm CJ, Schwarz JK, DeNardo DG. Dendritic Cell Paucity Leads to Dysfunctional Immune Surveillance in Pancreatic Cancer. Cancer Cell. 2020;37:289-307.e9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 354] [Article Influence: 59.0] [Reference Citation Analysis (4)] |
| 66. | Jacobetz MA, Chan DS, Neesse A, Bapiro TE, Cook N, Frese KK, Feig C, Nakagawa T, Caldwell ME, Zecchini HI, Lolkema MP, Jiang P, Kultti A, Thompson CB, Maneval DC, Jodrell DI, Frost GI, Shepard HM, Skepper JN, Tuveson DA. Hyaluronan impairs vascular function and drug delivery in a mouse model of pancreatic cancer. Gut. 2013;62:112-120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 927] [Cited by in RCA: 879] [Article Influence: 67.6] [Reference Citation Analysis (4)] |
| 67. | Ligorio M, Sil S, Malagon-Lopez J, Nieman LT, Misale S, Di Pilato M, Ebright RY, Karabacak MN, Kulkarni AS, Liu A, Vincent Jordan N, Franses JW, Philipp J, Kreuzer J, Desai N, Arora KS, Rajurkar M, Horwitz E, Neyaz A, Tai E, Magnus NKC, Vo KD, Yashaswini CN, Marangoni F, Boukhali M, Fatherree JP, Damon LJ, Xega K, Desai R, Choz M, Bersani F, Langenbucher A, Thapar V, Morris R, Wellner UF, Schilling O, Lawrence MS, Liss AS, Rivera MN, Deshpande V, Benes CH, Maheswaran S, Haber DA, Fernandez-Del-Castillo C, Ferrone CR, Haas W, Aryee MJ, Ting DT. Stromal Microenvironment Shapes the Intratumoral Architecture of Pancreatic Cancer. Cell. 2019;178:160-175.e27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 351] [Cited by in RCA: 433] [Article Influence: 61.9] [Reference Citation Analysis (4)] |
| 68. | Freed-Pastor WA, Lambert LJ, Ely ZA, Pattada NB, Bhutkar A, Eng G, Mercer KL, Garcia AP, Lin L, Rideout WM 3rd, Hwang WL, Schenkel JM, Jaeger AM, Bronson RT, Westcott PMK, Hether TD, Divakar P, Reeves JW, Deshpande V, Delorey T, Phillips D, Yilmaz OH, Regev A, Jacks T. The CD155/TIGIT axis promotes and maintains immune evasion in neoantigen-expressing pancreatic cancer. Cancer Cell. 2021;39:1342-1360.e14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 218] [Article Influence: 43.6] [Reference Citation Analysis (0)] |
| 69. | Nywening TM, Belt BA, Cullinan DR, Panni RZ, Han BJ, Sanford DE, Jacobs RC, Ye J, Patel AA, Gillanders WE, Fields RC, DeNardo DG, Hawkins WG, Goedegebuure P, Linehan DC. Targeting both tumour-associated CXCR2(+) neutrophils and CCR2(+) macrophages disrupts myeloid recruitment and improves chemotherapeutic responses in pancreatic ductal adenocarcinoma. Gut. 2018;67:1112-1123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 432] [Cited by in RCA: 404] [Article Influence: 50.5] [Reference Citation Analysis (7)] |
| 70. | Steele NG, Biffi G, Kemp SB, Zhang Y, Drouillard D, Syu L, Hao Y, Oni TE, Brosnan E, Elyada E, Doshi A, Hansma C, Espinoza C, Abbas A, The S, Irizarry-Negron V, Halbrook CJ, Franks NE, Hoffman MT, Brown K, Carpenter ES, Nwosu ZC, Johnson C, Lima F, Anderson MA, Park Y, Crawford HC, Lyssiotis CA, Frankel TL, Rao A, Bednar F, Dlugosz AA, Preall JB, Tuveson DA, Allen BL, Pasca di Magliano M. Inhibition of Hedgehog Signaling Alters Fibroblast Composition in Pancreatic Cancer. Clin Cancer Res. 2021;27:2023-2037. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 237] [Article Influence: 47.4] [Reference Citation Analysis (4)] |
| 71. | Yamamoto K, Venida A, Yano J, Biancur DE, Kakiuchi M, Gupta S, Sohn ASW, Mukhopadhyay S, Lin EY, Parker SJ, Banh RS, Paulo JA, Wen KW, Debnath J, Kim GE, Mancias JD, Fearon DT, Perera RM, Kimmelman AC. Autophagy promotes immune evasion of pancreatic cancer by degrading MHC-I. Nature. 2020;581:100-105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1047] [Cited by in RCA: 987] [Article Influence: 164.5] [Reference Citation Analysis (4)] |
| 72. | Connor AA, Denroche RE, Jang GH, Lemire M, Zhang A, Chan-Seng-Yue M, Wilson G, Grant RC, Merico D, Lungu I, Bartlett JMS, Chadwick D, Liang SB, Eagles J, Mbabaali F, Miller JK, Krzyzanowski P, Armstrong H, Luo X, Jorgensen LGT, Romero JM, Bavi P, Fischer SE, Serra S, Hafezi-Bakhtiari S, Caglar D, Roehrl MHA, Cleary S, Hollingsworth MA, Petersen GM, Thayer S, Law CHL, Nanji S, Golan T, Smith AL, Borgida A, Dodd A, Hedley D, Wouters BG, O'Kane GM, Wilson JM, Zogopoulos G, Notta F, Knox JJ, Gallinger S. Integration of Genomic and Transcriptional Features in Pancreatic Cancer Reveals Increased Cell Cycle Progression in Metastases. Cancer Cell. 2019;35:267-282.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 188] [Article Influence: 26.9] [Reference Citation Analysis (4)] |
| 73. | Olive KP, Jacobetz MA, Davidson CJ, Gopinathan A, McIntyre D, Honess D, Madhu B, Goldgraben MA, Caldwell ME, Allard D, Frese KK, Denicola G, Feig C, Combs C, Winter SP, Ireland-Zecchini H, Reichelt S, Howat WJ, Chang A, Dhara M, Wang L, Rückert F, Grützmann R, Pilarsky C, Izeradjene K, Hingorani SR, Huang P, Davies SE, Plunkett W, Egorin M, Hruban RH, Whitebread N, McGovern K, Adams J, Iacobuzio-Donahue C, Griffiths J, Tuveson DA. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science. 2009;324:1457-1461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2693] [Cited by in RCA: 2570] [Article Influence: 151.2] [Reference Citation Analysis (4)] |
| 74. | Qin C, Yang G, Yang J, Ren B, Wang H, Chen G, Zhao F, You L, Wang W, Zhao Y. Metabolism of pancreatic cancer: paving the way to better anticancer strategies. Mol Cancer. 2020;19:50. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 341] [Cited by in RCA: 308] [Article Influence: 51.3] [Reference Citation Analysis (4)] |
| 75. | Marabelle A, Le DT, Ascierto PA, Di Giacomo AM, De Jesus-Acosta A, Delord JP, Geva R, Gottfried M, Penel N, Hansen AR, Piha-Paul SA, Doi T, Gao B, Chung HC, Lopez-Martin J, Bang YJ, Frommer RS, Shah M, Ghori R, Joe AK, Pruitt SK, Diaz LA Jr. Efficacy of Pembrolizumab in Patients With Noncolorectal High Microsatellite Instability/Mismatch Repair-Deficient Cancer: Results From the Phase II KEYNOTE-158 Study. J Clin Oncol. 2020;38:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2532] [Cited by in RCA: 2280] [Article Influence: 380.0] [Reference Citation Analysis (9)] |
| 76. | Collisson EA, Bailey P, Chang DK, Biankin AV. Molecular subtypes of pancreatic cancer. Nat Rev Gastroenterol Hepatol. 2019;16:207-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 776] [Cited by in RCA: 660] [Article Influence: 94.3] [Reference Citation Analysis (6)] |
| 77. | Bailey P, Chang DK, Nones K, Johns AL, Patch AM, Gingras MC, Miller DK, Christ AN, Bruxner TJ, Quinn MC, Nourse C, Murtaugh LC, Harliwong I, Idrisoglu S, Manning S, Nourbakhsh E, Wani S, Fink L, Holmes O, Chin V, Anderson MJ, Kazakoff S, Leonard C, Newell F, Waddell N, Wood S, Xu Q, Wilson PJ, Cloonan N, Kassahn KS, Taylor D, Quek K, Robertson A, Pantano L, Mincarelli L, Sanchez LN, Evers L, Wu J, Pinese M, Cowley MJ, Jones MD, Colvin EK, Nagrial AM, Humphrey ES, Chantrill LA, Mawson A, Humphris J, Chou A, Pajic M, Scarlett CJ, Pinho AV, Giry-Laterriere M, Rooman I, Samra JS, Kench JG, Lovell JA, Merrett ND, Toon CW, Epari K, Nguyen NQ, Barbour A, Zeps N, Moran-Jones K, Jamieson NB, Graham JS, Duthie F, Oien K, Hair J, Grützmann R, Maitra A, Iacobuzio-Donahue CA, Wolfgang CL, Morgan RA, Lawlor RT, Corbo V, Bassi C, Rusev B, Capelli P, Salvia R, Tortora G, Mukhopadhyay D, Petersen GM; Australian Pancreatic Cancer Genome Initiative, Munzy DM, Fisher WE, Karim SA, Eshleman JR, Hruban RH, Pilarsky C, Morton JP, Sansom OJ, Scarpa A, Musgrove EA, Bailey UM, Hofmann O, Sutherland RL, Wheeler DA, Gill AJ, Gibbs RA, Pearson JV, Waddell N, Biankin AV, Grimmond SM. Genomic analyses identify molecular subtypes of pancreatic cancer. Nature. 2016;531:47-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3189] [Cited by in RCA: 2792] [Article Influence: 279.2] [Reference Citation Analysis (4)] |
| 78. | Springfeld C, Ferrone CR, Katz MHG, Philip PA, Hong TS, Hackert T, Büchler MW, Neoptolemos J. Neoadjuvant therapy for pancreatic cancer. Nat Rev Clin Oncol. 2023;20:318-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 309] [Cited by in RCA: 279] [Article Influence: 93.0] [Reference Citation Analysis (0)] |
| 79. | Versteijne E, van Dam JL, Suker M, Janssen QP, Groothuis K, Akkermans-Vogelaar JM, Besselink MG, Bonsing BA, Buijsen J, Busch OR, Creemers GM, van Dam RM, Eskens FALM, Festen S, de Groot JWB, Groot Koerkamp B, de Hingh IH, Homs MYV, van Hooft JE, Kerver ED, Luelmo SAC, Neelis KJ, Nuyttens J, Paardekooper GMRM, Patijn GA, van der Sangen MJC, de Vos-Geelen J, Wilmink JW, Zwinderman AH, Punt CJ, van Tienhoven G, van Eijck CHJ; Dutch Pancreatic Cancer Group. Neoadjuvant Chemoradiotherapy Versus Upfront Surgery for Resectable and Borderline Resectable Pancreatic Cancer: Long-Term Results of the Dutch Randomized PREOPANC Trial. J Clin Oncol. 2022;40:1220-1230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 648] [Cited by in RCA: 554] [Article Influence: 138.5] [Reference Citation Analysis (1)] |
| 80. | Ghaneh P, Palmer D, Cicconi S, Jackson R, Halloran CM, Rawcliffe C, Sripadam R, Mukherjee S, Soonawalla Z, Wadsley J, Al-Mukhtar A, Dickson E, Graham J, Jiao L, Wasan HS, Tait IS, Prachalias A, Ross P, Valle JW, O'Reilly DA, Al-Sarireh B, Gwynne S, Ahmed I, Connolly K, Yim KL, Cunningham D, Armstrong T, Archer C, Roberts K, Ma YT, Springfeld C, Tjaden C, Hackert T, Büchler MW, Neoptolemos JP; European Study Group for Pancreatic Cancer. Immediate surgery compared with short-course neoadjuvant gemcitabine plus capecitabine, FOLFIRINOX, or chemoradiotherapy in patients with borderline resectable pancreatic cancer (ESPAC5): a four-arm, multicentre, randomised, phase 2 trial. Lancet Gastroenterol Hepatol. 2023;8:157-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 270] [Cited by in RCA: 235] [Article Influence: 78.3] [Reference Citation Analysis (0)] |
| 81. | Murphy JE, Wo JY, Ryan DP, Clark JW, Jiang W, Yeap BY, Drapek LC, Ly L, Baglini CV, Blaszkowsky LS, Ferrone CR, Parikh AR, Weekes CD, Nipp RD, Kwak EL, Allen JN, Corcoran RB, Ting DT, Faris JE, Zhu AX, Goyal L, Berger DL, Qadan M, Lillemoe KD, Talele N, Jain RK, DeLaney TF, Duda DG, Boucher Y, Fernández-Del Castillo C, Hong TS. Total Neoadjuvant Therapy With FOLFIRINOX in Combination With Losartan Followed by Chemoradiotherapy for Locally Advanced Pancreatic Cancer: A Phase 2 Clinical Trial. JAMA Oncol. 2019;5:1020-1027. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 471] [Cited by in RCA: 441] [Article Influence: 63.0] [Reference Citation Analysis (4)] |
| 82. | O'Reilly EM, Oh DY, Dhani N, Renouf DJ, Lee MA, Sun W, Fisher G, Hezel A, Chang SC, Vlahovic G, Takahashi O, Yang Y, Fitts D, Philip PA. Durvalumab With or Without Tremelimumab for Patients With Metastatic Pancreatic Ductal Adenocarcinoma: A Phase 2 Randomized Clinical Trial. JAMA Oncol. 2019;5:1431-1438. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 628] [Cited by in RCA: 601] [Article Influence: 85.9] [Reference Citation Analysis (4)] |
| 83. | Sherman MH, Yu RT, Tseng TW, Sousa CM, Liu S, Truitt ML, He N, Ding N, Liddle C, Atkins AR, Leblanc M, Collisson EA, Asara JM, Kimmelman AC, Downes M, Evans RM. Stromal cues regulate the pancreatic cancer epigenome and metabolome. Proc Natl Acad Sci U S A. 2017;114:1129-1134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 124] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 84. | Padrón LJ, Maurer DM, O'Hara MH, O'Reilly EM, Wolff RA, Wainberg ZA, Ko AH, Fisher G, Rahma O, Lyman JP, Cabanski CR, Yu JX, Pfeiffer SM, Spasic M, Xu J, Gherardini PF, Karakunnel J, Mick R, Alanio C, Byrne KT, Hollmann TJ, Moore JS, Jones DD, Tognetti M, Chen RO, Yang X, Salvador L, Wherry EJ, Dugan U, O'Donnell-Tormey J, Butterfield LH, Hubbard-Lucey VM, Ibrahim R, Fairchild J, Bucktrout S, LaVallee TM, Vonderheide RH. Sotigalimab and/or nivolumab with chemotherapy in first-line metastatic pancreatic cancer: clinical and immunologic analyses from the randomized phase 2 PRINCE trial. Nat Med. 2022;28:1167-1177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 301] [Cited by in RCA: 282] [Article Influence: 70.5] [Reference Citation Analysis (1)] |
| 85. | Byrne KT, Betts CB, Mick R, Sivagnanam S, Bajor DL, Laheru DA, Chiorean EG, O'Hara MH, Liudahl SM, Newcomb C, Alanio C, Ferreira AP, Park BS, Ohtani T, Huffman AP, Väyrynen SA, Dias Costa A, Kaiser JC, Lacroix AM, Redlinger C, Stern M, Nowak JA, Wherry EJ, Cheever MA, Wolpin BM, Furth EE, Jaffee EM, Coussens LM, Vonderheide RH. Neoadjuvant Selicrelumab, an Agonist CD40 Antibody, Induces Changes in the Tumor Microenvironment in Patients with Resectable Pancreatic Cancer. Clin Cancer Res. 2021;27:4574-4586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 146] [Article Influence: 29.2] [Reference Citation Analysis (1)] |
| 86. | Tempero MA, Malafa MP, Al-Hawary M, Behrman SW, Benson AB, Cardin DB, Chiorean EG, Chung V, Czito B, Del Chiaro M, Dillhoff M, Donahue TR, Dotan E, Ferrone CR, Fountzilas C, Hardacre J, Hawkins WG, Klute K, Ko AH, Kunstman JW, LoConte N, Lowy AM, Moravek C, Nakakura EK, Narang AK, Obando J, Polanco PM, Reddy S, Reyngold M, Scaife C, Shen J, Vollmer C, Wolff RA, Wolpin BM, Lynn B, George GV. Pancreatic Adenocarcinoma, Version 2.2021, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2021;19:439-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1026] [Cited by in RCA: 883] [Article Influence: 176.6] [Reference Citation Analysis (3)] |
| 87. | Puleo F, Nicolle R, Blum Y, Cros J, Marisa L, Demetter P, Quertinmont E, Svrcek M, Elarouci N, Iovanna J, Franchimont D, Verset L, Galdon MG, Devière J, de Reyniès A, Laurent-Puig P, Van Laethem JL, Bachet JB, Maréchal R. Stratification of Pancreatic Ductal Adenocarcinomas Based on Tumor and Microenvironment Features. Gastroenterology. 2018;155:1999-2013.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 463] [Cited by in RCA: 413] [Article Influence: 51.6] [Reference Citation Analysis (3)] |
| 88. | Nicolle R, Blum Y, Duconseil P, Vanbrugghe C, Brandone N, Poizat F, Roques J, Bigonnet M, Gayet O, Rubis M, Elarouci N, Armenoult L, Ayadi M, de Reyniès A, Giovannini M, Grandval P, Garcia S, Canivet C, Cros J, Bournet B, Buscail L; BACAP Consortium, Moutardier V, Gilabert M, Iovanna J, Dusetti N. Establishment of a pancreatic adenocarcinoma molecular gradient (PAMG) that predicts the clinical outcome of pancreatic cancer. EBioMedicine. 2020;57:102858. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 66] [Article Influence: 11.0] [Reference Citation Analysis (4)] |
| 89. | Morrison AH, Byrne KT, Vonderheide RH. Immunotherapy and Prevention of Pancreatic Cancer. Trends Cancer. 2018;4:418-428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 397] [Cited by in RCA: 367] [Article Influence: 45.9] [Reference Citation Analysis (4)] |
| 90. | Carstens JL, Correa de Sampaio P, Yang D, Barua S, Wang H, Rao A, Allison JP, LeBleu VS, Kalluri R. Spatial computation of intratumoral T cells correlates with survival of patients with pancreatic cancer. Nat Commun. 2017;8:15095. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 562] [Cited by in RCA: 509] [Article Influence: 56.6] [Reference Citation Analysis (4)] |
| 91. | Özdemir BC, Pentcheva-Hoang T, Carstens JL, Zheng X, Wu CC, Simpson TR, Laklai H, Sugimoto H, Kahlert C, Novitskiy SV, De Jesus-Acosta A, Sharma P, Heidari P, Mahmood U, Chin L, Moses HL, Weaver VM, Maitra A, Allison JP, LeBleu VS, Kalluri R. Depletion of carcinoma-associated fibroblasts and fibrosis induces immunosuppression and accelerates pancreas cancer with reduced survival. Cancer Cell. 2014;25:719-734. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2178] [Cited by in RCA: 2028] [Article Influence: 169.0] [Reference Citation Analysis (6)] |
| 92. | Rhim AD, Oberstein PE, Thomas DH, Mirek ET, Palermo CF, Sastra SA, Dekleva EN, Saunders T, Becerra CP, Tattersall IW, Westphalen CB, Kitajewski J, Fernandez-Barrena MG, Fernandez-Zapico ME, Iacobuzio-Donahue C, Olive KP, Stanger BZ. Stromal elements act to restrain, rather than support, pancreatic ductal adenocarcinoma. Cancer Cell. 2014;25:735-747. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1792] [Cited by in RCA: 1691] [Article Influence: 140.9] [Reference Citation Analysis (5)] |
| 93. | Lu F, Wang X, Tian J, Li X. Early versus delayed computed tomography-guided celiac plexus neurolysis for palliative pain management in patients with advanced pancreatic cancer: a retrospective cohort study. Front Neurol. 2023;14:1292758. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 94. | Lee JJ, Perera RM, Wang H, Wu DC, Liu XS, Han S, Fitamant J, Jones PD, Ghanta KS, Kawano S, Nagle JM, Deshpande V, Boucher Y, Kato T, Chen JK, Willmann JK, Bardeesy N, Beachy PA. Stromal response to Hedgehog signaling restrains pancreatic cancer progression. Proc Natl Acad Sci U S A. 2014;111:E3091-E3100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 429] [Cited by in RCA: 420] [Article Influence: 35.0] [Reference Citation Analysis (4)] |
| 95. | Daley D, Zambirinis CP, Seifert L, Akkad N, Mohan N, Werba G, Barilla R, Torres-Hernandez A, Hundeyin M, Mani VRK, Avanzi A, Tippens D, Narayanan R, Jang JE, Newman E, Pillarisetty VG, Dustin ML, Bar-Sagi D, Hajdu C, Miller G. γδ T Cells Support Pancreatic Oncogenesis by Restraining αβ T Cell Activation. Cell. 2016;166:1485-1499.e15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 292] [Cited by in RCA: 284] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 96. | Kota J, Hancock J, Kwon J, Korc M. Pancreatic cancer: Stroma and its current and emerging targeted therapies. Cancer Lett. 2017;391:38-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 152] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 97. | Nevala-Plagemann C, Hidalgo M, Garrido-Laguna I. From state-of-the-art treatments to novel therapies for advanced-stage pancreatic cancer. Nat Rev Clin Oncol. 2020;17:108-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 315] [Cited by in RCA: 290] [Article Influence: 48.3] [Reference Citation Analysis (5)] |
| 98. | O'Kane GM, Grünwald BT, Jang GH, Masoomian M, Picardo S, Grant RC, Denroche RE, Zhang A, Wang Y, Lam B, Krzyzanowski PM, Lungu IM, Bartlett JMS, Peralta M, Vyas F, Khokha R, Biagi J, Chadwick D, Ramotar S, Hutchinson S, Dodd A, Wilson JM, Notta F, Zogopoulos G, Gallinger S, Knox JJ, Fischer SE. GATA6 Expression Distinguishes Classical and Basal-like Subtypes in Advanced Pancreatic Cancer. Clin Cancer Res. 2020;26:4901-4910. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 270] [Article Influence: 45.0] [Reference Citation Analysis (0)] |
| 99. | Espinet E, Gu Z, Imbusch CD, Giese NA, Büscher M, Safavi M, Weisenburger S, Klein C, Vogel V, Falcone M, Insua-Rodríguez J, Reitberger M, Thiel V, Kossi SO, Muckenhuber A, Sarai K, Lee AYL, Backx E, Zarei S, Gaida MM, Rodríguez-Paredes M, Donato E, Yen HY, Eils R, Schlesner M, Pfarr N, Hackert T, Plass C, Brors B, Steiger K, Weichenhan D, Arda HE, Rooman I, Kopp JL, Strobel O, Weichert W, Sprick MR, Trumpp A. Aggressive PDACs Show Hypomethylation of Repetitive Elements and the Execution of an Intrinsic IFN Program Linked to a Ductal Cell of Origin. Cancer Discov. 2021;11:638-659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 98] [Article Influence: 16.3] [Reference Citation Analysis (0)] |