Copyright: ©Author(s) 2026.
World J Clin Oncol. Apr 24, 2026; 17(4): 117705
Published online Apr 24, 2026. doi: 10.5306/wjco.v17.i4.117705
Published online Apr 24, 2026. doi: 10.5306/wjco.v17.i4.117705
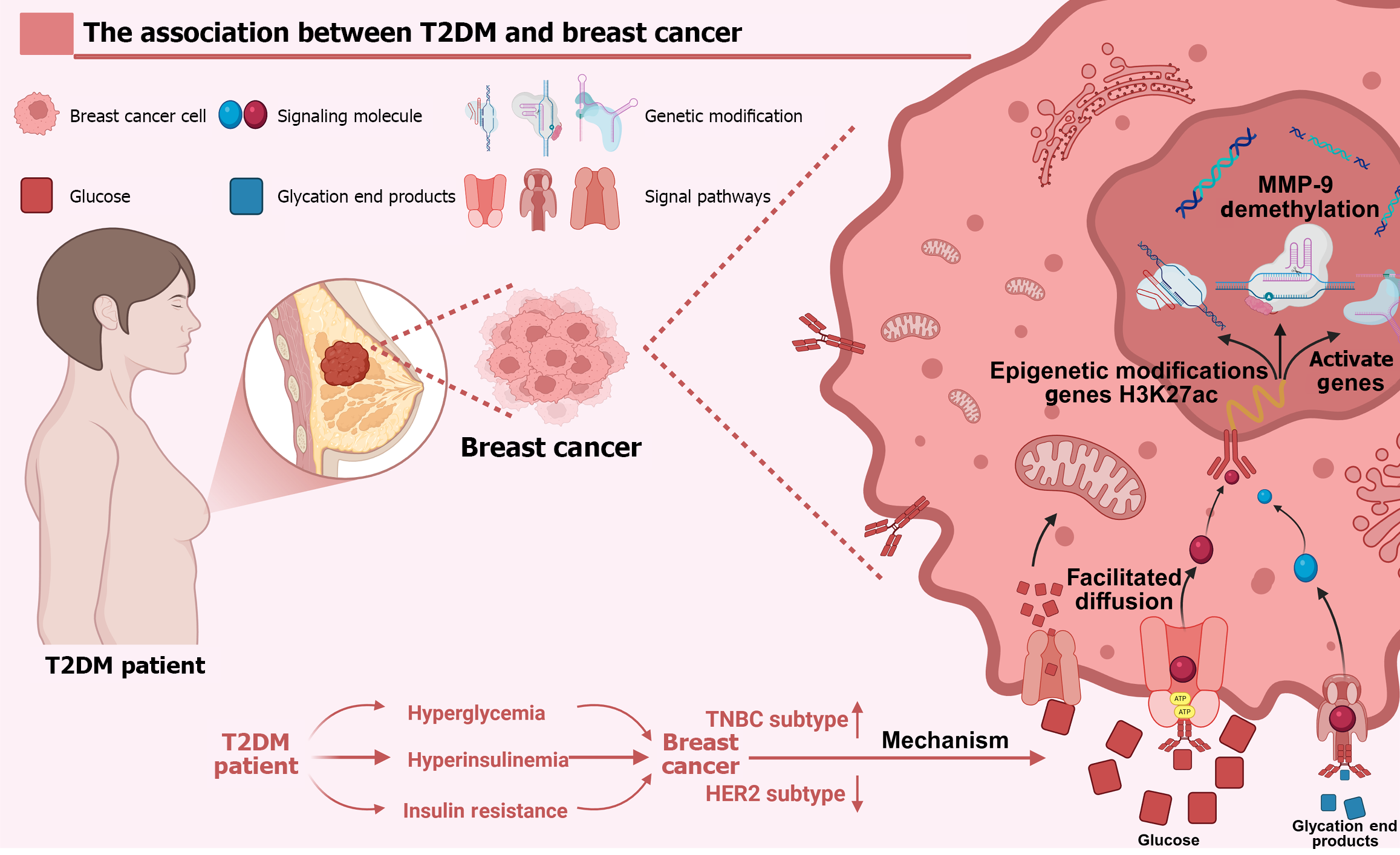
Figure 1 Type 2 diabetes exacerbates breast cancer progression through hyperglycaemia, impaired glucose tolerance and insulin resistance, driving a shift toward triple-negative subtypes while reducing human epidermal growth factor receptor 2-positive cases.
Created in BioRender. Mechanistically, elevated glucose and advanced glycation end products activate fibrinogen C domain containing 1 signalling and induce epigenetic reprogramming - including histone H3 lysine 27 acetylation and matrix metalloproteinase-9 promoter demethylation - that collectively drive metastatic progression. T2DM: Type 2 diabetes mellitus; HER2: Human epidermal growth factor receptor 2; TNBC: Triple-negative breast cancer; MMP-9: Matrix metalloproteinase-9; H3K27ac: Histone H3 lysine 27.
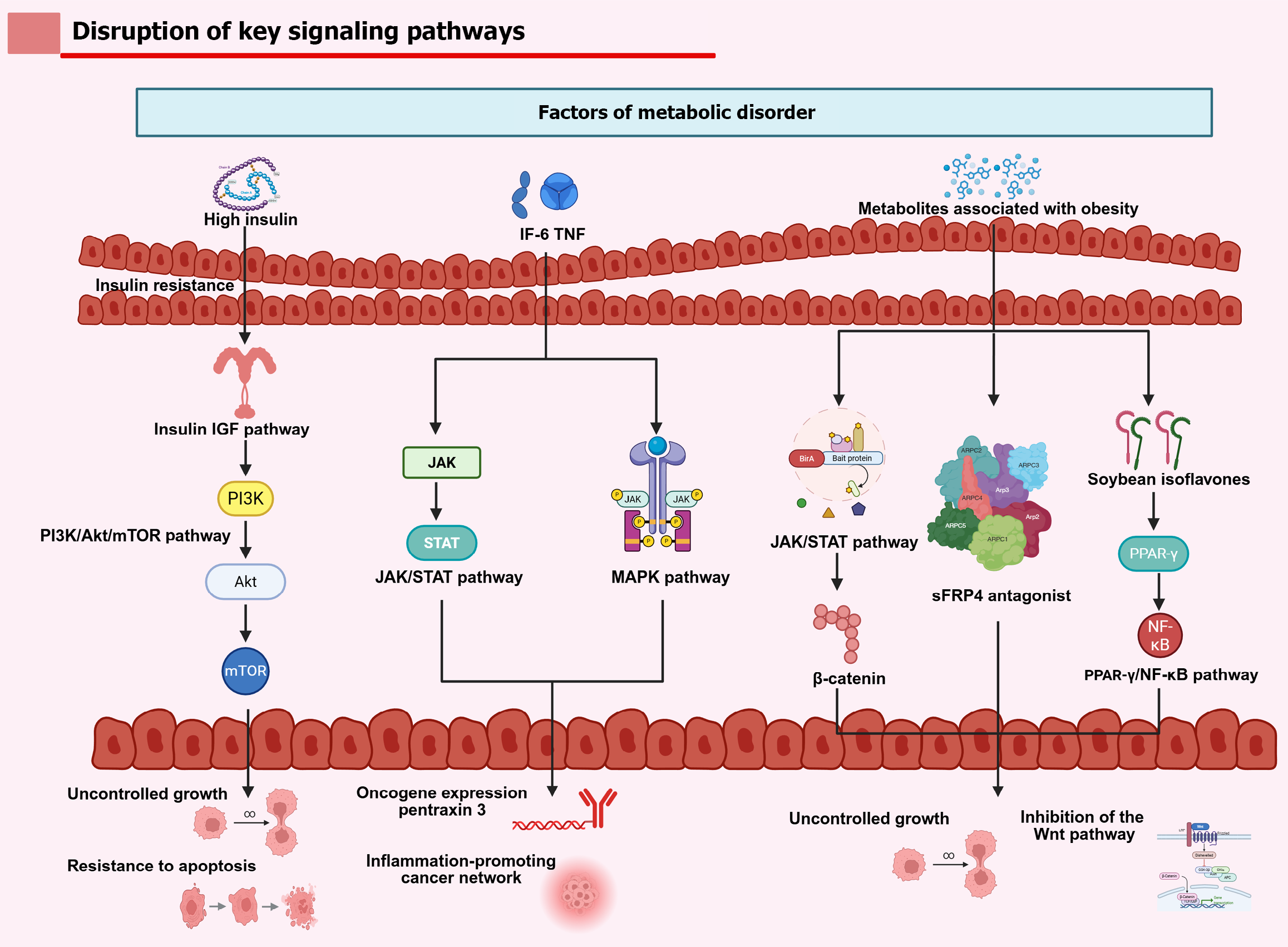
Figure 2 Systemic metabolic disturbances - including hyperinsulinemia, obesity-related inflammatory cytokines (interleukin-6, tumor necrosis factor), and metabolic metabolites - converge to activate key oncogenic pathways.
Created in BioRender. Insulin resistance hyper
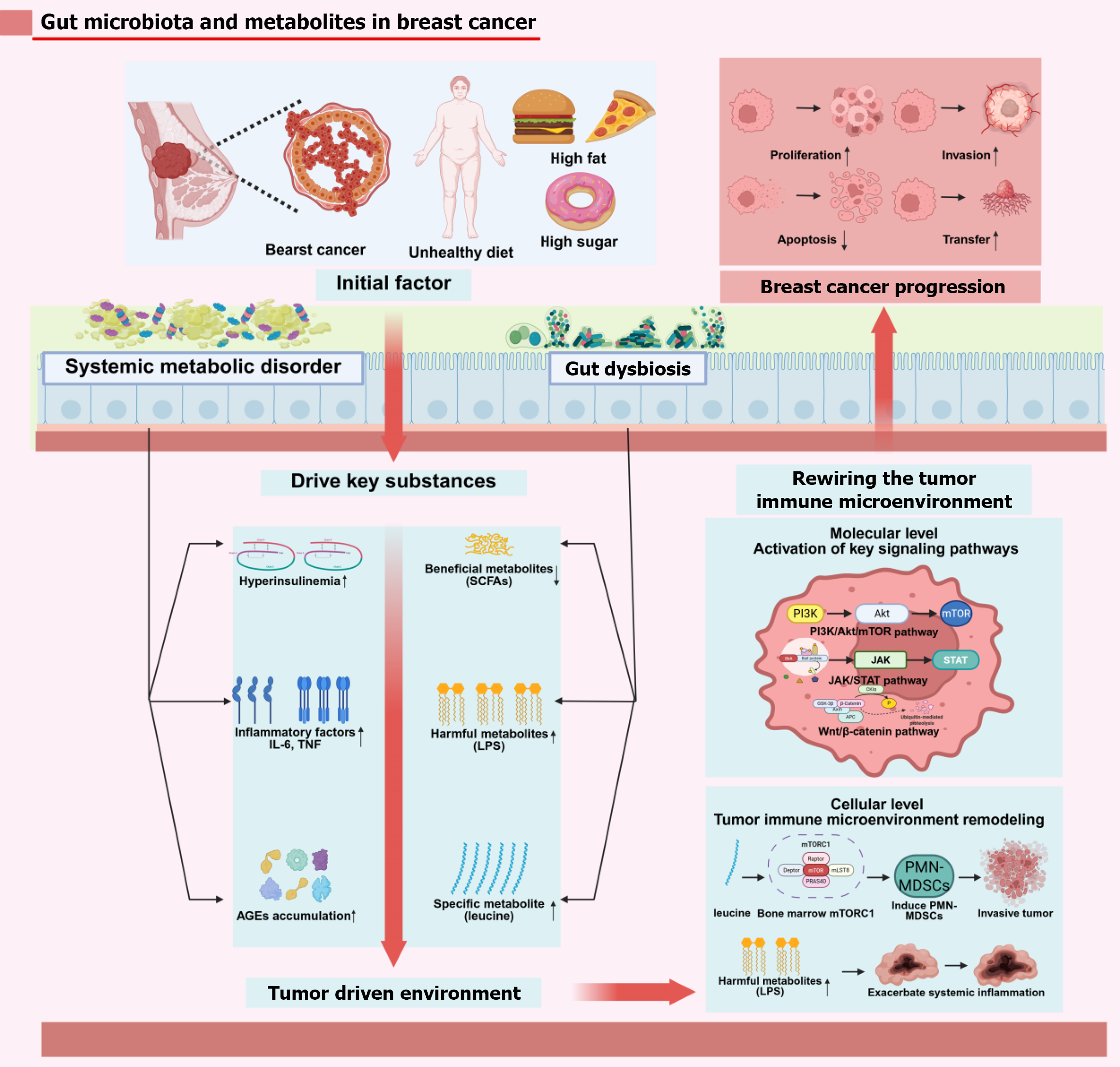
Figure 3 Unhealthy diets and obesity converge to drive breast carcinogenesis through two interconnected axes.
Created in BioRender. Systemic metabolic dysfunction - characterized by hyperinsulinaemia, elevated pro-inflammatory cytokines (interleukin-6, tumor necrosis factor), and accumulation of advanced glycation end products - and gut microbiota dysbiosis, which reduces beneficial short-chain fatty acids while increasing harmful metabolites such as lipopolysaccharides and leucine. These systemic and microbial effectors collectively activate key oncogenic pathways including phosphatidylinositol 3-kinase/protein kinase B/mammalian/mechanistic target of rapamycin, Janus kinase/signal transducer and activator of transcription, and Wnt/β-catenin, and remodel the tumour immune microenvironment via leucine-mediated expansion of polymorphonuclear myeloid-derived suppressor cells and lipopolysaccharides-triggered chronic inflammation, ultimately fostering tumour proliferation, invasion, metastasis, and therapy resistance. IL-6: Interleukin-6; TNF: Tumor necrosis factor; PI3K: Phosphatidylinositol 3-kinase; Akt: Protein kinase B; mTOR: Mammalian/mechanistic target of rapamycin; JAK: Janus kinase; STAT: Signal transducer and activator of transcription; LPS: Lipopolysaccharide; SCFAs: Short-chain fatty acids; AGEs: Advanced glycation end products; PMN-MDSCs: Polymorphonuclear myeloid-derived suppressor cells.
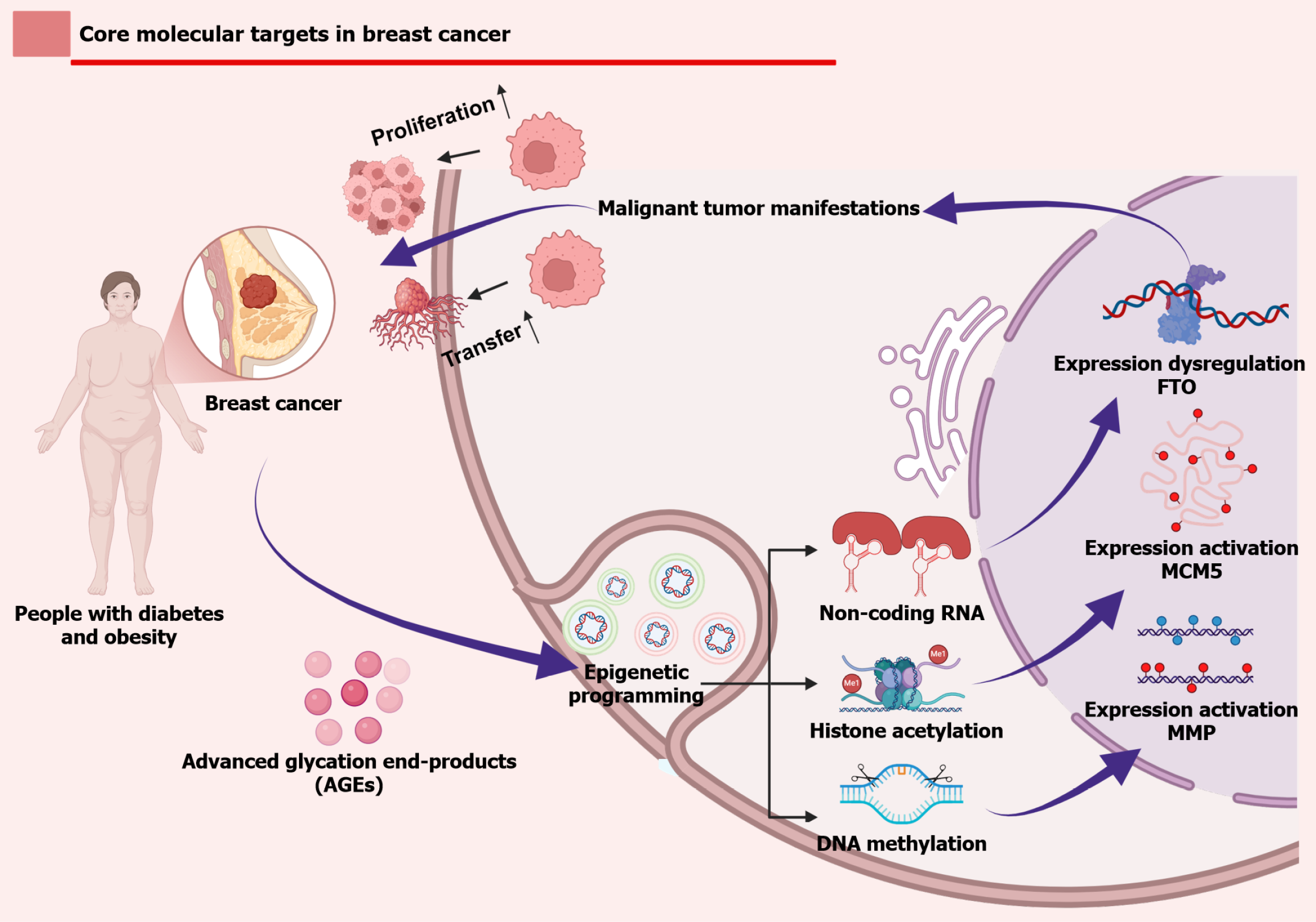
Figure 4 Systemic metabolic perturbations.
Created in BioRender. Including hyperglycaemia, advanced glycation end products, and obesity - remodel the epigenetic landscape through DNA demethylation, histone acetylation, and non-coding RNA regulation, leading to activation of metastasis-associated matrix metalloproteinase-9 and cell cycle gene MCM5, along with dysregulation of obesity-associated genes such as FTO. These changes collectively drive oncogenic phenotypes including proliferation, invasion, and therapy resistance, while molecular interventions - via small molecules or biologics - target key nodes to suppress these epigenetically-driven malignant programmes. MMP: Matrix metalloproteinase; AGEs: Advanced glycation end products.
- Citation: Zhang SH, Yang Y, Zhang Y. Breast cancer and metabolic comorbidities: From epidemiology and molecular mechanisms to precision interventions. World J Clin Oncol 2026; 17(4): 117705
- URL: https://www.wjgnet.com/2218-4333/full/v17/i4/117705.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i4.117705