Copyright: ©Author(s) 2026.
World J Clin Oncol. Mar 24, 2026; 17(3): 115774
Published online Mar 24, 2026. doi: 10.5306/wjco.v17.i3.115774
Published online Mar 24, 2026. doi: 10.5306/wjco.v17.i3.115774
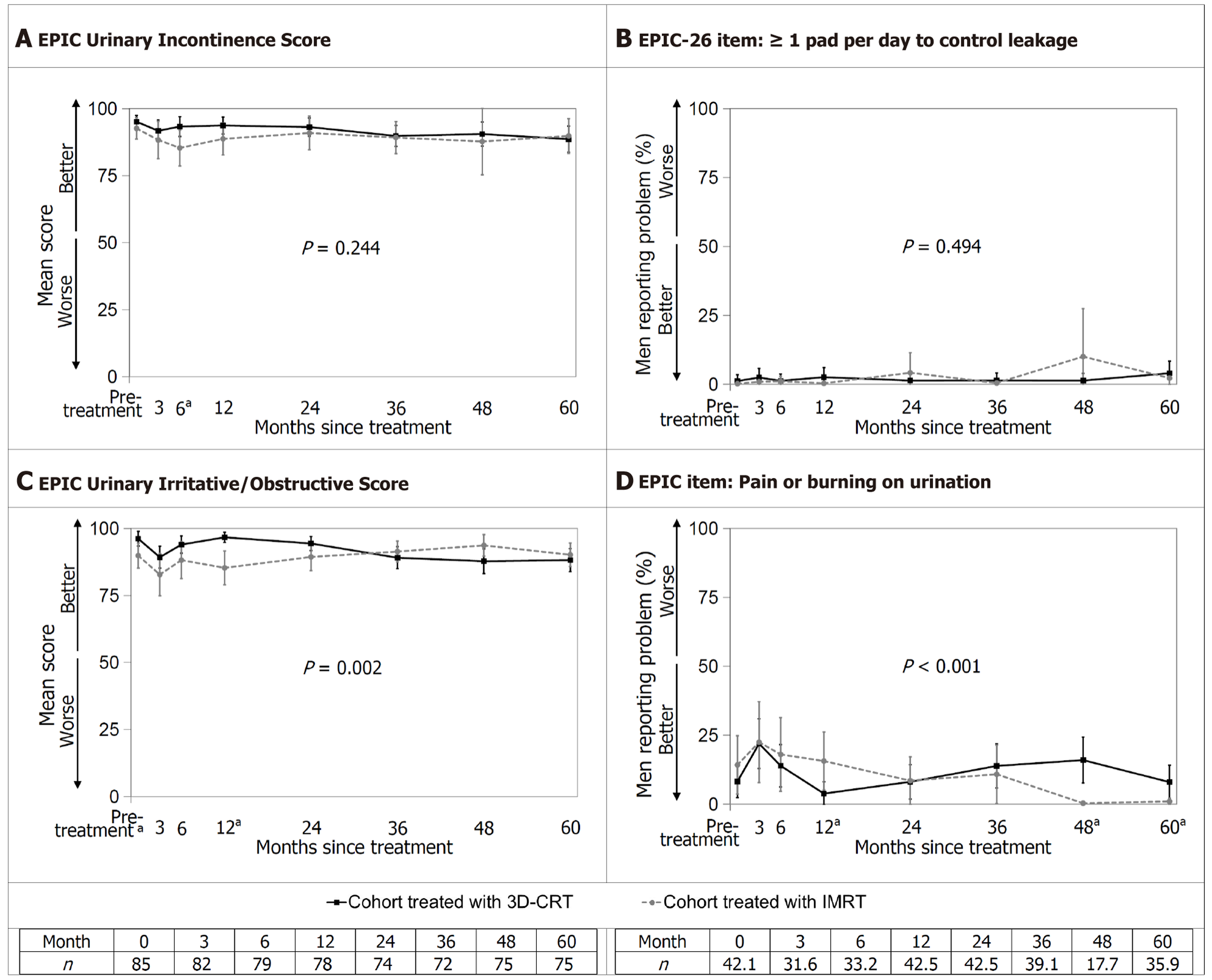
Figure 1 Weighted results of the Urinary domain measured with the expanded prostate cancer index composite.
A: Expanded prostate cancer index composite (EPIC)-26 Urinary Incontinence Score; B: EPIC-26 item “≥ 1 pad per day to control leakage”; C: EPIC-26 Urinary Irritative/Obstructive Score; D: EPIC-26 item “pain or burning on urination”. Urinary outcomes over 60 months. Error bars represent 95%CI. The P value for the interaction between treatment and time is shown; aP < 0.05 for the t-test at that assessment when the treatment-by-time interaction is significant. EPIC: Expanded prostate cancer index composite; IMRT: Intensity-modulated radiotherapy; 3D-CRT: Three-dimensional conformal radiotherapy.
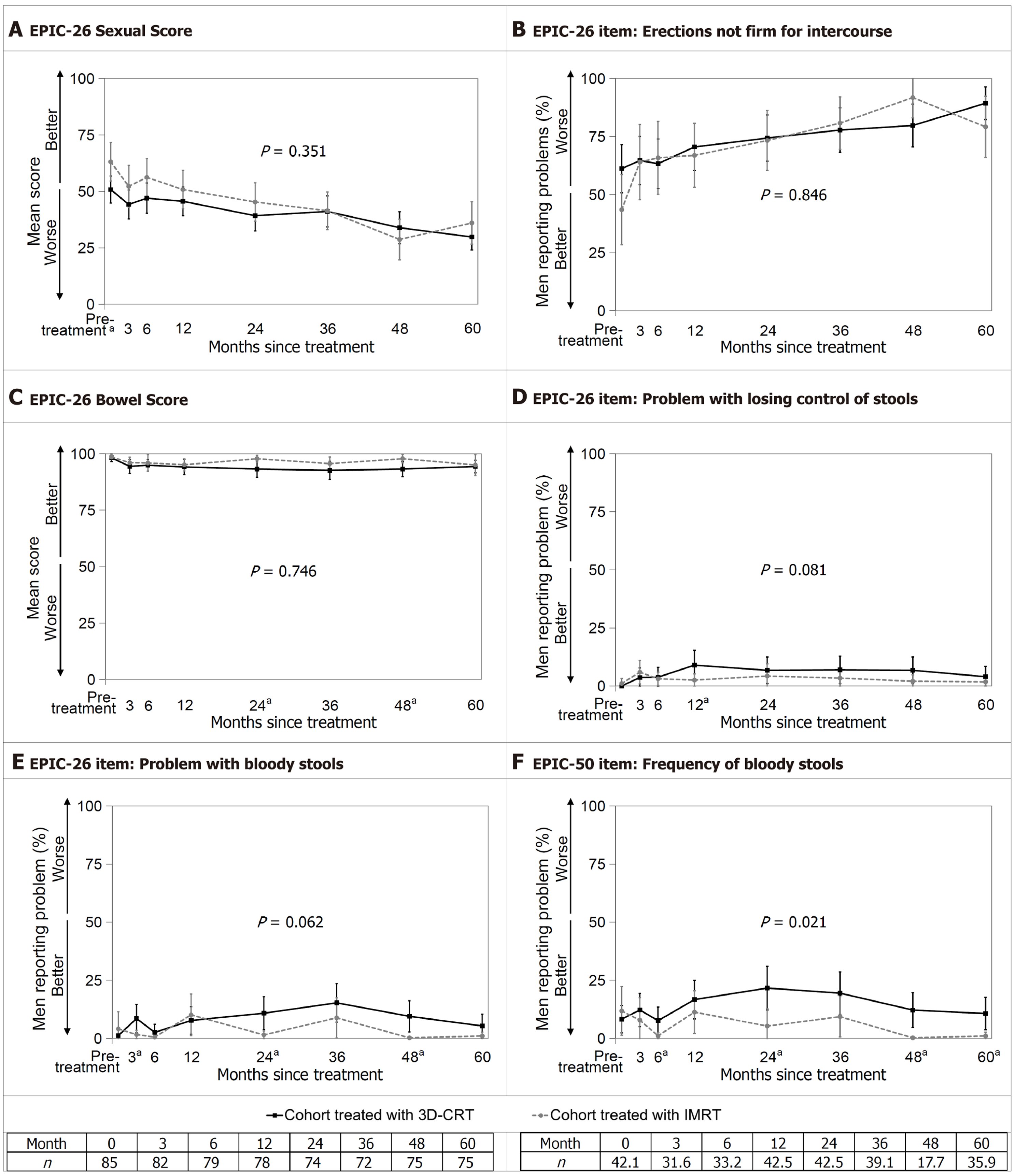
Figure 2 Weighted results of the sexual and bowel domains measured with the expanded prostate cancer index composite.
A: Expanded prostate cancer index composite (EPIC)-26 Sexual Score; B: EPIC-26 item “erections not firm for intercourse”; C: EPIC-26 Bowel Score; D: EPIC-26 item “problem with losing control of stools”; E: EPIC-26 item “problem with bloody stools”; F: EPIC-50 item “frequency of bloody stools”. Sexual and bowel outcomes over 60 months. Error bars represent 95%CI. The P value for the interaction between treatment and time is shown; aP < 0.05 for the t-test at that assessment when the treatment-by-time interaction is significant. EPIC: Expanded prostate cancer index composite; IMRT: Intensity-modulated radiotherapy; 3D-CRT: Three-dimensional conformal radiotherapy.
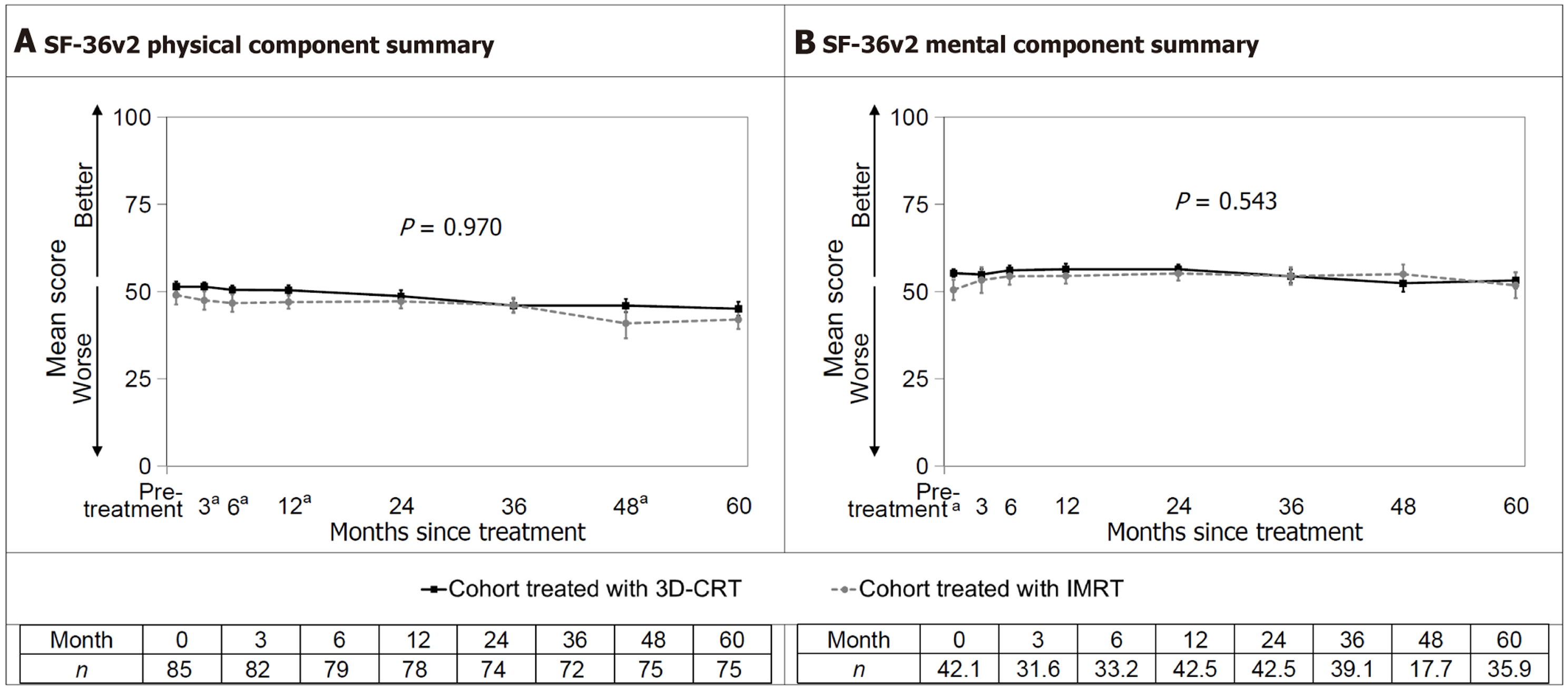
Figure 3 Weighted results of the physical and mental component summaries measured with the short form-36 health survey.
A: Short form-36 health survey version 2 physical component summary; B: Short form-36 health survey version 2 mental component summary. Health-related quality of life over 60 months. Error bars represent 95%CI. The P value for the interaction between treatment and time is shown; aP < 0.05 for the t-test at that assessment when the treatment-by-time interaction is significant. SF-36: Short form-36 health survey; IMRT: Intensity-modulated radiotherapy; 3D-CRT: Three-dimensional conformal radiotherapy.
- Citation: Mera A, Sancho G, Garin O, Pont À, Pardo Y, Macias V, Hervas A, Cabrera P, Ferrer F, Roselló À, Samper Ots P, García ME, Jové-Teixidó J, Ferrer M, The Multicentric Spanish Group of Clinically Localized Prostate Cancer. Patient-reported outcome measures for technology assessment: A comparison between three-dimensional conformal and intensity-modulated radiotherapy in prostate cancer. World J Clin Oncol 2026; 17(3): 115774
- URL: https://www.wjgnet.com/2218-4333/full/v17/i3/115774.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i3.115774