Published online Nov 6, 2015. doi: 10.4292/wjgpt.v6.i4.183
Peer-review started: April 27, 2015
First decision: July 25, 2015
Revised: September 7, 2015
Accepted: September 25, 2015
Article in press: September 28, 2015
Published online: November 6, 2015
Processing time: 200 Days and 4.2 Hours
Infection with the Gram-negative pathogen Helicobacter pylori (H. pylori) has been associated with gastro-duodenal disease and the importance of H. pylori eradication is underscored by its designation as a group I carcinogen. The standard triple therapy consists of a proton pump inhibitor, amoxicillin and clarithromycin, although many other regimens are used, including quadruple, sequential and concomitant therapy regimens supplemented with metronidazole, clarithromycin and levofloxacin. Despite these efforts, current therapeutic regimens lack efficacy in eradication due to antibiotic resistance, drug compliance and antibiotic degradation by the acidic stomach environment. Antibiotic resistance to clarithromycin and metronidazole is particularly problematic and several approaches have been proposed to overcome this issue, such as complementary probiotic therapy with Lactobacillus. Other studies have identified novel molecules with an anti-H. pylori effect, as well as tailored therapy and nanotechnology as viable alternative eradication strategies. This review discusses current antibiotic therapy for H. pylori infections, limitations of this type of therapy and predicts the availability of newly developed therapies for H. pylori eradication.
Core tip: This article reviews the recent literature describing antibiotic resistance and trends in Helicobacter pylori (H. pylori) treatment. As there is no effective conventional therapy, new treatments are being developed and bismuth quadruple, sequential, concomitant therapies are recommended as a first-line regimen in regions with high levels of clarithromycin resistance. Quinolones have also been used for H. pylori treatment, although the cure rate has gradually reduced with this approach. New therapeutic directions include probiotic supplementation, tailored therapy, novel agents, and nanotechnology.
-
Citation: Kim SY, Choi DJ, Chung JW. Antibiotic treatment for
Helicobacter pylori : Is the end coming? World J Gastrointest Pharmacol Ther 2015; 6(4): 183-198 - URL: https://www.wjgnet.com/2150-5349/full/v6/i4/183.htm
- DOI: https://dx.doi.org/10.4292/wjgpt.v6.i4.183
Core tip: This article reviews the recent literature describing antibiotic resistance and trends in Helicobacter pylori (H. pylori) treatment. As there is no effective conventional therapy, new treatments are being developed and bismuth quadruple, sequential, concomitant therapies are recommended as a first-line regimen in regions with high levels of clarithromycin resistance. Quinolones have also been used for H. pylori treatment, although the cure rate has gradually reduced with this approach. New therapeutic directions include probiotic supplementation, tailored therapy, novel agents, and nanotechnology.
Helicobacter pylori (H. pylori) is a Gram-negative, flagellated, spiral shaped microaerophilic bacterium first identified by Marshall and Warren[1-3]. These bacteria have morphological characteristics penetrate the mucosa and colonize the stomach and duodenum[4]. H. pylori are responsible for the pathogenesis that leads to gastritis, peptic ulcer disease (PUD), gastric adenocarcinoma and gastric mucosa-associated lymphoid tissue (MALT) lymphoma[3,5,6]. The World Health Organization has classified H. pylori as a group I carcinogen with a risk of stomach cancer[7,8]. H. pylori-related stomach cancer represents 5.5% of all cancers worldwide and 25% of all infection-associated malignancies. Socioeconomically, H. pylori infection increases the risk of malignancy and the expense of H. pylori-associated morbidity[9]. H. pylori infection has also been related to non-digestive conditions such as ischemic heart disease, stroke, Alzheimer’s disease, Parkinson’s disease, and iron deficient anemia[4,10]. In other report, some patients with gastritis resolved H. pylori infection without using antibiotic treatment[11]. Although the prevalence of H. pylori infection has been reduced in developed countries, it has remained prevalent in developing countries[12,13] with rates of infection varying according to nation, patient age, and socioeconomic states[14]. Eradication of H. pylori is an effective treatment for PUD, gastric MALT lymphoma, and preventing the recurrence of stomach cancer after endoscopic treatment[15-17].
A standard triple therapy (STT), consisting of a proton pump inhibitor (PPI), clarithromycin and amoxicillin, was established in clinical practice for the eradication of H. pylori infection[18,19]. However, in recent years, the efficacy of STT has been critically altered in many regions of the world as eradication rates have diminished to inadequately low levels[18,20]. The causes for this decline may involve patient compliance, bacterial factors, obesity, smoking, reinfection, and genetic polymorphisms in CYP2C19. However, antibiotic resistance may be the primary reason for reduced eradication of H. pylori infection worldwide[20-22]. In addition, the eradication rates differ by region, even in the same country. In South Korea, one study reported that the eradication rates of first-line therapy decreased from 81.3% to 77.5% from 2001-2007[23], while another showed that no definite evidence of a significant change in the eradication rate during 2000-2010[24]. This may be due to geographical differences in antibiotic resistance and the methods used to confirm eradication. In a region with high rates of clarithromycin resistance, sequential or concomitant therapy is recommended as the first-line H. pylori eradication treatment[25]. The primary reason for the growth in antibiotic resistance is the emergence of point mutations in the H. pylori genome[26]. Thus, the development of novel treatment methods to increase eradication rates and reduce antibiotic resistance is needed. The focus of this review will be on current H. pylori therapies and limitations, as well as alternative anti-H. pylori regimens.
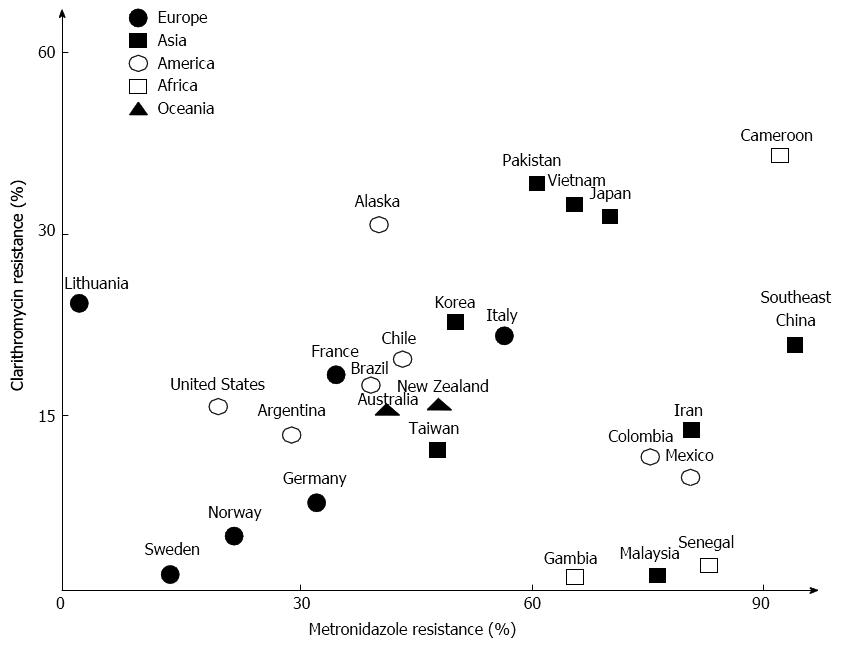
The most important antibiotics in H. pylori treatment are clarithromycin, metronidazole, and amoxicillin. Figure 1 illustrates recently reported clarithromycin and metronidazole resistance rates worldwide. Resistance to these antibiotics is thought to be the main cause of eradication failure[27-29]. Antibiotic resistance is discovered by bacterial culture-based techniques (E-test, modified disk diffusion, agar dilution method, and breakpoint susceptibility test) and molecular methods [polymerase chain reaction (PCR), real-time PCR, allele-specific PCR, sequencing, and fluorescent in situ hybridization][30]. Although these methods are useful for examining antibiotic resistance, their implementation at the early stages of H. pylori remains impractical due to the time required to obtain results and the high cost of the tests.
Clarithromycin is a macrolide antibiotic that inhibits protein synthesis by binding to and slowing the actions of the bacterial ribosome[30]. Clarithromycin resistance is due to three point mutations at A2142C, A2142G, and A2143G in the 23s rRNA gene[31]. In particular, the A2143G mutation has been related to a very low eradication rate[32]. In contrast, the A2143G mutation occurs in only 23% of resistant strains in Eastern countries[31]. This suggests that clarithromycin point mutations may be geographically distinct between Eastern and Western countries and new point mutations have appeared in South America[33]. Clarithromycin resistance is also different depending on the area. In Brazil, stomach biopsy specimens positive for H. pylori were analyzed by PCR to detect the point mutation associated with clarithromycin resistance[34]. The results uncovered primary clarithromycin resistance in 16.5% patients. Recently, the clarithromycin resistance rate in South Korea was reported to range from 17.2% to 23.7%[35]. In a study published in Japan, the clarithromycin resistance rate in 2002 was 18.9%; however, the clarithromycin resistance rate in 2006 increased to 27.2%[36]. Even with third-line eradication therapy, clarithromycin resistance rates in Japan were reported as 86.4%[37]. Several studies in China have reported increased resistance rates Shanghai[38], 21.5% resistance in the southeast coastal region[39], and a relatively high rate of 33% in Vietnam, which is near Southeast China[40]. In Western Asia, resistance to clarithromycin has been reported to be > 10% in Iran and > 20% in Turkey[13]. In one study, clarithromycin resistance was reported in 47.5% of patients with dyspepsia in Turkey[41]. In sharp contrast to other Asian countries, no resistance to clarithromycin has been reported in Malaysia[42] and the prevalence of resistance to clarithromycin in Gambia and Senegal also remains very low[43,44]. Resistance to clarithromycin has also risen by > 20% in Southern Europe, although in Northern Europe the resistance rate is less than 10%[45] compared to 1.5% in a random adult Swedish population[46] and 7.5% in central Germany[47]. During the last 15 years, a twofold increase in clarithromycin resistance was reported in Italy[48] and in Spain, where the mean clarithromycin resistance rate was 18.3% in 1709 patients[49], and 34.7% in Portuguese children[50]. In contrast to the general trend, the rate of H. pylori strains resistant to clarithromycin decreased from 34% to 22% during 6 years in Southern Poland[51]. Despite these variations, the overall frequency of clarithromycin resistance has risen from 10.2% to 21.3% worldwide, and A2143G is the most frequently reported point mutation. Present European guidelines recommend 7 d of STT in regions in which the rate of clarithromycin resistance is < 20%, and 14 d in regions with clarithromycin resistance rates of > 20%[25,45].
The mechanism mediating resistance to metronidazole is complex. Modifications in the rdxA gene, assumed to be point mutations, are considered a primary cause[30]. Metronidazole resistance may also influence the treatment outcome, although it is generally considered less clinically important than clarithromycin resistance[52,53]. Overall, the Eastern Asian region has higher metronidazole resistance rates with 95.4% in the southeast coastal region of China[39] and 71.3% in Japan[37]. In Vietnam, the resistance rate was 69.9% among 103 strains[40]. Unlike the clarithromycin resistance rate, there was a high prevalence of resistance to metronidazole (75.5%) in Malaysia[42], a 76.8% rate in Iran[54] and a high resistance rate in Africa[43,44]. Another study showed that 80% of strains in Mexico were resistant to metronidazole[55]. Overall, metronidazole resistance is > 50% in much of the world but there are reports that metronidazole resistance has declined in Northern Europe[9,30], while in the United States and Europe, the metronidazole resistance rate was reported to be < 40%[30,56], and 22.5% in 102 isolates from Norway[57]. However, in Central and Southern Europe, resistance rates remain markedly higher-34.9% in France and 32.7% in Germany[47,58].
Amoxicillin is a beta-lactam antibiotic that was first used for H. pylori therapy[25]. Unlike clarithromycin and metronidazole, amoxicillin resistance rates are low worldwide[30]: 0% or < 1% in Europe[30]. However, other studies revealed high amoxicillin resistance rates in Iran, Japan, and Cameroon[37,45,54].
Fluoroquinolones are the sole class of antibiotics for treatment of H. pylori that directly inhibit bacterial DNA synthesis. Resistance to fluoroquinolones occurs primarily by mutation in the genes for topoisomerase IV and gyrase[59]. Levofloxacin is currently recommended as a second-line H. pylori treatment when first-line therapy containing clarithromycin has failed, although levofloxacin resistance has been predicted to increase in the near future[25]. Levofloxacin resistance rates in Asia differ from region to region with rates of 20.6% in the southeast coastal region of China and 18.4% in Vietnam[39,40]. Fluoroquinolone resistance was noted as 62.3% in Pakistan[60], while Japan and Malaysia had low resistance rates of 8.2% and 0%, respectively[37,42]. Primary H. pylori resistance to ciprofloxacin occurred at a high frequency (15.7%) in South Korea[61]. A study by Mégraud[62] of more than 2000 patients with H. pylori infection showed resistance rates of 14.1% for levofloxacin, with significantly higher fluoroquinolone resistance in Western/Southern Europe than in Northern Europe[62]. O’Connor et al[63] reported that 11.7% of patients had strains resistant to levofloxacin in Ireland and there was a 29.1% resistance rate in 2011 in Germany[64], 15% in Senegal[44] and 23% in Brazil[65].
The first-line regimen for the eradication of H. pylori infection consists of STT using a PPI, amoxicillin and clarithromycin and was first introduced by Dr. Bazzoli. In studies conducted during the 1990s, STT yielded > 80% treatment success with reports of > 90% possible[66,67]. However, the increased prevalence of clarithromycin resistance has accounted for the diminished efficacy of STT. Table 1 shows eradication rates from recent studies using STT. Generally, STT is not recommended as a first-line regimen when the clarithromycin resistance rate is > 15%-20%, and other therapies such as quadruple therapy or sequential therapy are suggested[25]. Thus, a steady increase in H. pylori resistance to amoxicillin and metronidazole has also resulted in reduced treatment success of STT[27,68,69]. The ideal outcome of H. pylori eradication is > 80% by intention to treat (ITT) analysis and > 90% by per protocol (PP) analysis. According to a recent study, the eradication rate was unacceptably low for treatment success, with only 18% exceeding 85% and approximately 60% failing to attain 80% eradication by ITT analysis[20]. Over the past 20 years, the efficacy of STT has decreased, with eradication rates < 80% by ITT analysis[41]. According to the present formula by Dr. Graham[70], if clarithromycin resistance rate of 20%, the outcome of clarithromycin containing triple therapy is reduced to 77.2% by PP analysis. Already in some countries the eradication rates have been reported to be < 50% and if this trend continues for another 20 years, the efficacy of STT will be negligible.
| Country | Ref. | Publication | Treatment duration | Patients | Therapy regimen | Eradication rate (ITT) | Eradication rate (PP) |
| South Korea | Na et al[177] | 2007 | 7 d | 3267 | Standard PPI | NA | 84.3% |
| Cla 500 mg bid | |||||||
| Amo 1 g bid | |||||||
| Chung et al[178] | 2012 | 10 d | 80 | Lan 30 mg bid | 58.7% | 67.6% | |
| Cla 500 mg bid | |||||||
| Amo 1 g bid | |||||||
| Japan | Asaka et al[179] | 2001 | 7 d | 96 | Lan 30 mg bid | NA | 90.7% |
| Cla 200 mg bid | |||||||
| Amo 750 mg bid | |||||||
| Fujioka et al[180] | 2012 | 7 d | 3162 | Rab 10 mg bid | 80.7% | NA | |
| Amo 750 mg bid | |||||||
| Cla 200 mg bid | |||||||
| Nishizawa et al[27] | 2012 | 7 d | 55 | Lan 30 mg bid | 74.5% | 80.4% | |
| Cla 400 mg bid | |||||||
| Amo 750 mg bid | |||||||
| Nishida et al[181] | 2014 | 7 d | 134/134 | Eso 20 mg bid | 69.4%/73.9% | 76.9%/79.8% | |
| Cla 400 mg bid | |||||||
| Amo 750 mg bid | |||||||
| Lan 30 mg bid | |||||||
| Cla 400 mg bid | |||||||
| Amo 750 mg bid | |||||||
| Taiwan | Sheu et al[182] | 2000 | 7 d or 2 wk | 286 | Ome 20 mg bid | NA | 87.8% |
| Amo 1 g bid | |||||||
| Cla or Met bid | |||||||
| Chen et al[117] | 2014 | 7 d | 73 | Rab 20 mg bid | 57.5% | 61.8% | |
| Cla 500 mg bid | |||||||
| Amo 1 g bid | |||||||
| Turkey | Ozçay et al[183] | 2004 | 4 wk: PPI 2 wk:Cla, Amo | 102 | Ome or Lan | NA | 75.7% |
| Cla 7.5 mg/kg bid | |||||||
| Amo 20 mg/kg bid | |||||||
| Kutluk et al[184] | 2014 | 10 d | 74 | Lan 1 mg/kg per day | 52.7% | 55.7% | |
| Cla 20 mg/kg per day | |||||||
| Amo 50 mg/kg per day | |||||||
| Italy | Catalano et al[185] | 1999 | 10 d | 84 | Ome 20 mg bid | NA | 94.0% |
| Cla 500 mg bid | |||||||
| Amo 1 g bid | |||||||
| Paoluzi et al[186] | 2010 | 7 d | 90 | Eso 20 mg bid | 66.0% | 75.0% | |
| Cla 500 mg bid | |||||||
| Amo 1 g bid | |||||||
| Latin America | Greenberg et al[106] | 2011 | 14 d | 488 | Lan 30 mg bid | 82.2% | 87.1% |
| Cla 500 mg bid | |||||||
| Amo 1 g bid |
Various methods have been considered to circumvent the STT eradication rate decrease. The first method suggested that increasing the STT duration would improve treatment efficacy. In an early meta-analysis, a 14-d STT regimen raised the eradication rate compared to a 7-d regimen[71]. Another meta-analysis supported this result by showing that extending STT over 7 d improved the eradication rate[72]. However, other reports determined that extending STT was not cost-effective and increased adverse events and decreased compliance, resulting in no significant difference between the eradication rate and extended treatment duration[73]. Another means of addressing the decrease in STT eradication rate is to increase the dose of PPI, which has a positive effect on treatment success. PPIs delay gastric emptying and increase gastric pH, which improves the effect of antibiotics by preventing acid-related degradation[74]. A meta-analysis reported increased eradication rates from STT involving PPI administration twice per day compared with once per day[75]. Another systematic review reported that utilizing a high dose of PPI increased the H. pylori treatment rate[76] and the use of high-dose PPI increased the effectiveness of STT compared with a single does PPI[77]. In spite of these positive outcomes, STT is now regarded as an outdated therapy.
Bismuth quadruple therapy (bismuth subcitrate potassium, metronidazole, tetracycline, PPI) has been suggested as a first-line treatment option for regions with a high (> 20%) incidence of clarithromycin resistance[53]. In a meta-analysis of nine randomized controlled trials (RCTs), bismuth quadruple therapy and STT resulted in similar compliance rates, side effects, and eradication rates as a primary therapy for H. pylori infection[78]. For example, the ITT eradication rate with modified bismuth quadruple therapy was 92.7% in a recent randomized study in Chinese patients[79]. A pilot study in United States Hispanics showed that 14-d bismuth quadruple anti-H. pylori therapy achieved a > 95% eradication rate[80]. However, in some studies the eradication rate of bismuth quadruple therapy was < 80%[81-83]. A decrease in the bismuth quadruple therapy eradication rate was highly associated with metronidazole resistance[20].
Sequential therapy was introduced by Zullo et al[84] in Italy in 2000. This regimen includes a PPI and amoxicillin for 5 d, followed by a PPI, clarithromycin, and tinidazole triple therapy for another 5 d. Several studies have indicated that the eradication rate of sequential therapy was significantly higher than that of STT[85-87]. The reason that sequential therapy has a higher eradication rate than STT is that amoxicillin and PPI administered during the first 5 d decreases H. pylori density in the stomach, which increases clarithromycin and metronidazole efficacy[32,88-90]. In addition, amoxicillin damages the bacterial cell wall and limits production of an efflux channel underlying drug resistance. However, it is uncertain whether improvement in the eradication rate is due to sequential therapy or additional use of antibiotics such as tinidazole. Recent data from South Korea, showed a lower H. pylori eradication rate with sequential therapy with eradication rates by ITT analysis of 79.0% and by PP analysis of 84.9%[91]. Another study showed that the eradication rates by ITT were 72.1% and 80.2% in 10-d and 15-d sequential groups, respectively[92]. Although the 15-d sequential therapy group cure rate was higher than that of the 10-d sequential therapy group, the eradication rate remains low. In the study by Zhou et al[93], there was no significant difference between the eradication rates achieved with STT (66.4%) and sequential therapy (72.1%) by ITT analysis. Moreover, the sequential therapy group with dual clarithromycin resistance and metronidazole resistance had a lower eradication rate (43.9%) compared to the rate seen with only clarithromycin resistance (88.9%)[93]. In a 2015 study from India that compared sequential therapy to ciprofloxacin-containing sequential therapy, the ITT cure rate in the sequential therapy group was 66% and only 73.5% in the ciprofloxacin group[94]. Thus, the sequential therapy efficacy in Asia was lower than reported by earlier European studies. Another meta-analysis showed that the overall eradication rate of sequential therapy was 84.3% (95%CI: 82.1%-86.4%), although this was not superior to 14-d STT[86]. However, sequential therapy was able to eradicate 72.8% of the H. pylori resistant to clarithromycin[86]. In addition to the problem of sequential therapy eradication rate reduction, treatment compliance can be reduced due to medication changes during treatment. Furthermore, if eradication fails, no second-line treatment regimen has been established[95].
Concomitant therapy, also known as non-bismuth quadruple therapy, consists of PPI and all three antibiotics (clarithromycin, amoxicillin, metronidazole) administered concomitantly to provide a simpler treatment regimen compared to sequential therapy[96]. Recently, several studies have compared concomitant therapy to STT and sequential therapy. In one study, 10-d concomitant therapy resulted in a better eradication rate in settings with antibiotic-resistant H. pylori strains[97]. Eradication rates for concomitant and sequential therapies were 100% vs 75% for clarithromycin-resistant strains and 75% vs 60% for clarithromycin-resistant/metronidazole-resistant strains[97]. A meta-analysis of 15 studies showed a mean H. pylori eradication rate of 90% by ITT analysis for concomitant therapy and reported that longer treatment improved the outcomes compared to STT[98]. Another meta-analysis showed that concomitant therapy was superior to STT[99]. In studies published in South Korea, the eradication rate for concomitant therapy was considerably higher than that for sequential therapy[100,101]. However, several other studies have reported no difference in eradication rates between sequential and the concomitant therapy[102-104]. In a randomized open-label study, ITT eradication rates were 75.6% (95%CI: 66.3%-84.9%) in the sequential therapy group and 80.8% (95%CI: 71.8%-88.5%) in the concomitant therapy group[104]. In both groups, there was no difference in eradication rates and the treatment rate was lower than expected[104]. Furthermore, in some studies, concomitant therapy had a lower eradication rate than other regimens[83,105,106]. A total of 200 patients were randomized and the ITT eradication rates were 79% (95%CI: 71.0%-87.0%) in the bismuth group and 74% (95%CI: 68%-81%) in the concomitant group, although this was not statistically significant[83]. Another study compared the eradication rate between 10-d sequential therapy, 5-d concomitant therapy, 14-d concomitant therapy and 14-d hybrid therapy[105]. In ITT analysis, sequential therapy showed the highest eradication rate, which was higher than even 5-d concomitant therapy[105]. This is supported by an RCT of 1463 patients in seven Latin American sites (Chile, Colombia, Costa Rica, Honduras, Nicaragua, Mexico) that reported the eradication rate with 14-d standard therapy was 82.2%, compared to 73.6% with 5-d concomitant therapy and 76.5% with 10-d sequential therapy[106]. Currently, concomitant therapy has several limitations. First, side effects were reported to occur more frequently than with sequential therapy[107]. Second, there are few data describing the effect of metronidazole resistance in concomitant therapy. Moreover if dual-resistance to clarithromycin and metronidazole was > 15%, the eradication rate decreased[108,109]. Finally, as with sequential therapy, when first-line treatment fails no second-line treatment for concomitant therapy has been established. Tables 2 and 3 indicate that current trends of H. pylori eradication for sequential and concomitant therapy.
| Country | Ref. | Publication | Treatment duration | Patients | Therapy regimen | Eradication rate (ITT) | Eradication rate (PP) |
| South Korea | Lee et al[92] | 2014 | 10 d | 111 | 1st 5 d: Eso + Amo | 72.1% | 78.4% |
| 2nd 5 d: Eso + Cla + Met | |||||||
| Lee et al[91] | 2015 | 10 d | 100 | 1st 5 d: Rab + Amo | 79.0% | 84.9% | |
| 2nd 5 d: Rab + Cla + Met | |||||||
| China | Zhou et al[93] | 2014 | 10 d | 140 | 1st 5 d: Eso + Amo | 72.1% | 76.5% |
| 2nd 5 d: Eso + Cla + Tin | |||||||
| Qatar | Ben Chaabane et al[94] | 2015 | 14 d | 106 | 1st 7 d: Rab + Amo | 66.0% | 76.0% |
| 2nd 7 d: Rab + Cla + Met | |||||||
| Italy | Pontone et al[187] | 2010 | 10 d | 84 | 1st 5 d: Lan + Amo | 83.3% | 90.9% |
| 2nd 5 d: Lan + Cla + Met | |||||||
| Spain | Molina-Infante et al[114] | 2010 | 10 d | 115 | 1st 5 d: Ome + Amo | 76.5% | 80.8% |
| 2nd 5 d: Ome + Cla + Met |
| Country | Ref. | Publication | Treatment duration | Patients | Therapy regimen | Eradication rate (ITT) | Eradication rate (PP) |
| South Korea | Lim et al[104] | 2013 | 14 d | 78 | Rab 20 mg bid | 80.8% | 81.3% |
| Amo 1 g bid | |||||||
| Cla 500 mg bid | |||||||
| Met 500 mg bid | |||||||
| Lee et al[100] | 2015 | 7 d | 170 | Rab 20 mg bid | 79.4% | 94.4% | |
| Amo 1 g bid | |||||||
| Cla 500 mg bid | |||||||
| Met 500 mg tid | |||||||
| Thailand | Kongchayanun et al[188] | 2012 | 5 d/10 d | 55/55 | Rab 20 mg bid | 89.1%/96.4% | NA |
| Amo 1 g bid | |||||||
| Met 400 mg tid | |||||||
| Cla 1 g qd | |||||||
| Singapore | Ang et al[102] | 2015 | 10 d | 153 | PPI standard does | 81.7% | 95.4% |
| Amo 1 g bid | |||||||
| Cla 500 mg bid | |||||||
| Met 400 mg bid | |||||||
| Spain | Molina-Infante et al[97] | 2012 | 10 d | 209 | PPI standard does | 87.0% | 89.0% |
| Amo 1 g bid | |||||||
| Cla 500 mg bid | |||||||
| Met 500 mg bid | |||||||
| McNicholl et al[103] | 2014 | 10 d | 168 | Ome 20 mg bid | 87.0% | 91.0% | |
| Amo 1 g bid | |||||||
| Cla 500 mg bid | |||||||
| Met 500 mg bid | |||||||
| Latin America | Greenberg et al[106] | 2011 | 5 d | 489 | Lan 30 mg bid | 73.6% | NA |
| Amo 1 g bid | |||||||
| Cla 500 mg bid | |||||||
| Met 500 mg bid |
Levofloxacin has a large spectrum of activity against diverse Gram-positive and -negative bacteria[110] though inhibition of bacterial topoisomeraseII[111]. There have been several studies of levofloxacin use as a first-line treatment[112]. To overcome increasing clarithromycin resistance, levofloxacin has been used as a alternative to clarithromycin in either STT or sequential therapy[53]. Table 4 shows H. pylori eradication rates following levofloxacin-containing therapy. According to a meta-analysis, 10-d of levofloxacin triple therapy is more efficacious than 7-d bismuth-based quadruple therapy (RR = 1.41, 95%CI: 1.25-1.59) in the eradication of H. pylori infection[113]. In another study, levofloxacin-based triple therapy (ITT, 80.8%; 95%CI: 73%-88%) was more effective than STT (ITT: 64%, 95%CI: 55%-73%) and there were no differences in compliance or side effects[114]. However, other studies reported that levofloxacin-containing regimens did not have superior eradication rates compared to other treatments. Meta-analyses and a recent study in 2014 have shown that the outcome of levofloxacin-based first-line therapy was similar to STT[115,116] with an overall crude eradication rate of 79.1% in the levofloxacin group compared to 81.4% in the STT group[116]. A recent RCT in Taiwan with over 153 patients determined there was an advantage to levofloxacin-amoxicillin/clavulanate-PPI therapy over STT, although there was a low eradication rate (ITT analysis: 78.1% vs 57.5%)[117]. Unsatisfactory results were reported in an Asian meta-analysis, which showed that 7-d STT was more effective than 7-d levofloxacin-based therapy[118]. However, in European countries, levofloxacin-based therapy was more effective than STT[118]. Regional differences in H. pylori resistance to antibiotics might account for these results. Although levofloxacin has been suggested as a replacement for clarithromycin in H. pylori treatment, increasing quinolone resistance is a larger problem. According to Graham et al[119], in the presence of fluoroquinolone resistance treatment success with quinolone-containing therapy decreases and these results can be predicted using a formula. A report published in United States in 2015 determined that the prevalence of levofloxacin resistance was 31.3% (95%CI: 23.1%-39.4%)[120] and another study showed a high rate of quinolone resistance (50%) in Congo[121]. We calculated the effect of quinolone resistance on treatment success using the proposed formula, which indicated success rates of 87.6% and 73.5%, respectively[119,122]. The levofloxacin resistance rate is also relatively high in East Asia, where there is also a higher prevalence of stomach cancer compared to other regions[122,123]. Sitafloxacin, which has lower minimum inhibitory concentration for H. pylori, and levofloxacin triple therapy combined with bismuth quadruple therapy have been suggested as solutions to this problem, although further evidence is required to establish this approach[122,124].
| Country | Ref. | Publication | Treatment duration | Patients | Therapy regimen | Eradication rate (ITT) | Eradication rate (PP) |
| South Korea | Choi et al[189] | 2011 | 7 d | 98 | Ome 20 mg bid | 65.3% | 73.6% |
| Lev 200 mg bid | |||||||
| Amo 1 g bid | |||||||
| China | Liao et al[122] | 2013 | 14 d | 81 | Lan 30 mg bid | 82.7% | 85.9% |
| Lev 500 mg qd | |||||||
| Amo 1 g bid | |||||||
| Taiwan | Liou et al[190] | 2010 | 7 d | 217 | Lan 30 mg bid | 74.2% | 80.1% |
| Lev 750 mg qd | |||||||
| Amo 1 g bid | |||||||
| Chen et al[117] | 2014 | 7 d | 73 | Rab 20 mg bid | 78.1% | 80.9% | |
| Lev 500 mg bid | |||||||
| Amo 1 g bid | |||||||
| Spain | Molina-Infante et al[114] | 2010 | 10 d | 115 | Ome 20 mg bid | 80.8% | 82.6% |
| Lev 500 mg bid | |||||||
| Amo 1 g bid |
Many studies have demonstrated that probiotics have an inhibitory effect on H. pylori. Although some studies have reported that probiotics alone have limited efficacy[125,126], they can be useful when used as a supplemental drug. In a study by Lv et al[127], the cure rates in the probiotic supplementation group were superior to those in the group that did not receive probiotics (RR, 1.12; 95%CI: 1.06-1.19), and probiotics reduced the risk of H. pylori therapy related side effects (RR, 0.60; 95%CI: 0.40-0.91). In another meta-analysis, the pooled odd ratios (ORs) for the eradication rate were higher in the probiotic group than the control group (OR = 1.67; 95%CI: 1.38-2.02) by ITT, and adverse effects were lower in the probiotic group (OR = 0.49, 95%CI: 0.26-0.94)[128]. This study also showed racial differences in the sensitivity to probiotics, with greater differences in Asian populations compared to Caucasians. Adults, as well as children, have reported that probiotics positively affect eradication rates[129]. Saccharomyces boulardii is a type of probiotic gaining attention as a supplement for H. pylori. Recent reports have suggested that STT combined with S. boulardii could be effective for enhancing H. pylori eradication rates[130]. Compared with no intervention, S. boulardii-including regimens significantly increased treatment success (RR, 1.13; 95%CI: 1.05-1.21) and reduced H. pylori therapy-related adverse effects (RR, 0.46; 95%CI: 0.3-0.7)[131]. Furthermore, Lactobacillus and Bifidobacterium species also have an anti-H. pylori effect. A meta-analysis of 10 studies on Lactobacillus-containing and Bifidobacterium-containing probiotics use as a supplementation to H. pylori eradication therapy found that the pooled ORs by ITT analysis and PP analysis were 2.066 (95%CI: 1.398-3.055) and 2.321 (95%CI: 1.715-3.142), respectively[132]. In addition to the above references, Lactobacillus acidophilus and Bifidobacterium bifidum supplementation to STT is effective for H. pylori eradication and dynamic changes in intestinal flora[133]. In recent RCT study, Lactobacillus reuteri was identified as a new probiotic proposed for the treatment of H. pylori infection. A combination that includes L. reuteri was able to reduce antibiotic-associated adverse events and to increase the H. pylori eradication rate[134]. Although more research into these probiotics is needed, it is important to note that there are reduced drug complications and treatment is comparatively free from resistance. Therefore, probiotics will be considered important future therapeutics for H. pylori eradication.
It is well known that clarithromycin sensitivity of the H. pylori infection contributes to the success of the STT eradication rate[52,135]. Generally in infectious disease treatment, bacterial culture is carried out prior to determine the antibiotic selection of the organism. However, H. pylori bacterial culture is difficult and time-consuming, with various protocols for evaluating resistance. Thus, H. pylori treatment has depended on empirical antibiotic treatment[109]. In their study, Gerrits et al[136] determined that the A2142G, A2143G mutations were highly related to resistance using PCR, which was partially used to identify resistance to clarithromycin in H. pylori[137]. There are several advantages to this method because it is relatively simple and efficient with a cost similar to a rapid urease test[135]. In a recent study of 1232 patients, the eradication rate by selective treatment in the tailored group was 91.2%, which was significantly higher than control groups (amoxicillin, rabeprazole, clarithromycin; 75.9% and amoxicillin, rabeprazole, metronidazole; 79.1%)[138].
Appropriate stomach acid suppression, as well as resistant strains of H. pylori, remains a problem for successful eradication. PPI plays an important role in H. pylori eradication and the main enzyme involved in PPI metabolism is CYP2C19[139] and CYP2C19 genotypes can influence PPI efficacy[140]. Homozygous extensive metabolizer (HomEM) results in the highest rates of PPI metabolism, heterozygous extensive metabolizer (HetEM) results in moderate rates of PPI metabolism, while poor metabolizers (PM) exhibit the lowest rates of PPI metabolism[139]. The frequency of CYP2C19 polymorphism differs depending on ethnicity. Asians have a higher proportion of PM compared with Western populations, particularly Caucasians and African-Americans[139,141]. In contrast, Caucasians have a higher prevalence rate of HomEM compared with Asians[142]. Accordingly, geographic differences should be considered in selecting doses or types of PPIs for H. pylori treatment since there is a significant difference between HetEM and HomEM (OR = 1.90; 95%CI: 1.38-2.60) in H. pylori eradication rate[143]. In additional subanalysis of individual PPIs revealed that omeprazole was influenced by the CYP2C19 genotype[143]. In another meta-analysis, successful eradication rates differed considerably between PM and HetEM (OR = 1.73, P = 0.002) and between PM and HomEM (OR = 2.79, P < 0.0001) and even between HetEM and HomEM (OR = 2.00, P < 0.0001)[144]. This study showed that a regimen including rabeprazole was not affected by CYP2C19 genotype status[144]. According to a meta-analysis of a RCT in 2013, regardless of the PPI being taken, the eradication rates of PM were higher than HetEM and HomEM[145]. In addition, results of the sub-analysis of the PPI type, omeprazole and lansoprazole were affected by CYP2C19 genotype. Unlike above, esomeprazole and rabeprazole were not affected by CYP2C19 genotype[145]. In studies published in Japan, esomeprazole and rabeprazole are less influenced by CYP2C19 genotype compared with another PPIs[146,147]. The efficacy of tailored H. pylori eradication treatment was demonstrated by Sugimoto et al[148]. In a tailored regimen, H. pylori patients with clarithromycin-sensitivity were treated with clarithromycin, amoxicillin, rabeprazole, while clarithromycin-resistant patients were treated with metronidazole, amoxicillin, rabeprazole for 1 wk. As a result, the overall eradication rate was 96.7% (95%CI: 92.5%-98.9%) by ITT analysis and 97.4% (95%CI: 93.4%-99.3%) by PP analysis[148]. The method achieved high eradication rates of 94.3% in CYP2C19 rapid metabolizers[148]. Although CYP2C19 genotyping remains difficult clinically, tailored therapy may be useful in overcoming decreased eradication rates.
In the last decade, many researchers have argued that new classes of antimicrobials with novel mechanisms of action are necessary to overcome increasing drug resistance. Some agents have shown an antibacterial effect against H. pylori in vitro regardless of drug resistance and are effective even at low pH. Among them, pyloricidin A, B, and C have a strong and selective anti-H. pylori effect which an MIC90 value of 0.013 mg/L[149]. Benzimidazole derivatives (MIC90 = 0.025), polycyclic compound (MIC90 = 0.2-0.39), arylthiazole derivative 44 (MIC90 = 0.0065) also were highly effective against H. pylori[149].
Cathelicidins and defensins are examples of human antimicrobial peptides (AMPs) native to the innate immune system of many eukaryotes that have activity against H. pylori[150]. LL-37 is a cathelicidin with an anti-H. pylori effect[151], and a recent study demonstrated that cathelicidin limited H. pylori colonization and related gastritis in mouse models[152]. Defensin peptides have also been indicated to impede H. pylori[153]. Human beta defensin 2 and 3 are differentially expressed in gastric mucosa during H. pylori infection[154]. Oligo-acyl-lysyl (OAK) peptides, which have a structure and function similar to those of natural AMPs, have broad-spectrum antibacterial activity and anti-H. pylori effect in vivo[155]. Unlike the natural AMPs, OAK peptides are without known proteolytic cleavage sites and thus, resistant to enzymatic cleavage.
SQ109 was developed as a tuberculosis treatment and known to be safe and tolerated in human trials[150]. In an in vitro study, SQ109 had anti-H. pylori activity and a low H. pylori resistance rate[156]. Pyridodiazepines are potent and selective molecules that target the H. pylori MurI inhibitor not effective against other bacteria[157]. Sulfonamides and sulfamates were potent anti-beta-carbonic anhydrase molecules[150]. H. pylori beta-carbonic anhydrase catalyzes the hydration of carbon dioxide to proton and bicarbonate to facilitate H. pylori metabolism of urea and bicarbonate and survive in low pH. Sulfonamides and sulfamates, inhibit the enzyme and are effective against H. pylori[158].
Phytotherapy is expected to be another promising therapy for H. pylori eradication. Ginger rhizome extract has been demonstrated to have defensive activity in the stomach, increase stomach mucin regeneration, reinforce antioxidant enzymes, and suppress H. pylori growth[159]. Capsaicin has an anti-inflammatory effect and inhibited H. pylori-induced interleukin (IL)-8 production by gastric epithelial cells[160]. Sulphoraphane has also been indicated to suppress colonization and inhibit gastritis in H. pylori-infected mice and humans[161]. Red ginseng extract has inhibitory 5-LOX enzyme activity and LOX-inhibiting action that suppresses inflammation of H. pylori-infected gastric epithelial cells[162]. Epigallocatechin gallate, one of the green tea catechins, showed significant cytoprotective effects against H. pylori associated gastric cytotoxicity[163]. Red wine and resveratrol have also been shown to inhibit the growth of H. pylori cagA+ strains in vitro[164]. In an open-label RCT, adding vitamin C and E to antibiotic regimens showed excellent H. pylori eradication rates. Compared to the group that did not contain vitamin, the group that combined vitamin C and E to lansoprazole, amoxicillin, clarithromycin, and bismuth citrate treatment had significantly higher eradication rates of 91.3% by ITT analysis and 93.5% by PP analysis[165]. Thus, vitamin supplementation may be a future treatment option for H. pylori-related disease.
Recently, several studies have determined the antibacterial activity of micro- and nano-technology against H. pylori. Liposomes are spherical vesicles that contain amphiphilic lipids in a bi- or multi-layer with an aqueous core used to encapsulate several compounds[4]. This material contains biocompatible and biodegradable constituents without significant toxicity[4]. According to Obonyo et al[166], a liposomal nanoformulation of linolenic acid is a favorable nanotherapeutic with bactericidal activity against resistant strains of H. pylori. Another study suggested that an epitope-based therapeutic H. pylori vaccine may be beneficial in eradicating H. pylori[167] and a double liposome-based dual drug system may be helpful for treatment of H. pylori infection[168].
Polymeric particles have a number of advantages for use as an antibiotic delivery factor with an anti-H. pylori effect. It is possible to manipulate their shape to affect biodistribution to increase interactions with the target cell. They also have mucoadhesive properties and protects drugs from proteolytic enzyme. Importantly polymeric particles possess several mechanisms to overpower microbes[4]. Encapsulation of clarithromycin and omeprazole using gliadin nanoparticles as a mucoadhesive component has been reported for the treatment of H. pylori[169]. Another study also showed that positively charged gelatin microspheres could be a feasible applicant delivery system for eradication of H. pylori[170]. The amoxicillin-loaded chitosan mucoadhesive microspheres could increase gastrointestinal residence time and enhance amoxicillin stability to contribute to H. pylori treatment[171]. In addition, chitosan nanoparticles improved the anti-H. pylori effect of chitosan[172]. Genipin-cross-linked fucose-chitosan/heparin nanoparticles diminished drug release in stomach acid and then released amoxicillin in an H. pylori survival situation to inhibit H. pylori proliferation. In addition, amoxicillin-loaded nanoparticles increased H. pylori eradication and decreased H. pylori-associated gastric inflammation in an animal model[173]. The metronidazole-loaded porous microparticles that exhibit sustained release of metronidazole could assist H. pylori eradication and healing from mucosal damage[174]. Silver nanoparticles may also be safer bactericidal agents for the treatment of H. pylori-induced gastritis[175]. Berberine-loaded targeted nanoparticles stimulated H. pylori clearance and suppressed stomach inflammation in H. pylori infection[176].
Many studies have determined that novel agents and treatment regimens can improve eradication of H. pylori. With STT, high doses of PPI and prolonged therapy duration can increase eradication rates; indeed, in Europe and some regions of Asia these results are improved further with concomitant therapy. Concomitant therapy is less affected by antibiotic resistance, which adds value as an alternative treatment. Nevertheless, the eradication rates following concomitant therapy will gradually decrease due to the rapidly emerging antibiotic resistance of H. pylori worldwide.
In this review, we highlighted new and promising directions in H. pylori eradication. Although there are some practical limitations in applying probiotics and tailored therapy, they could of assistance in fighting H. pylori. Newer agents, nontraditional therapy, and microtechnology are also expected to play a major role in H. pylori eradication. However, several issues need to be solved to apply these treatments to the clinic. First, novel agents must be devoid of known proteolytic cleavage sites and thus, resistant to human digestive enzymatic cleavage. Second, these agents must be effective in an acidic environment. Third, these novel agents should be free from antibiotic resistance such as OAK. OAK have multiple nonspecific actions, so it would be hard to occur antibiotic resistance of H. pylori. Fourth, further studies are necessary to assess micro- and nano-toxicity, in vitro as well as in vivo. The safety and pharmacokinetic properties of novel treatments for H. pylori in humans also need to be evaluated. Finally, although novel treatments have many advantages, clinical studies are required to determine whether these findings can be applied to humans. In order to improve the eradication rate for H. pylori infection, further studies must be required.
| 1. | Marshall BJ, Warren JR. Unidentified curved bacilli in the stomach of patients with gastritis and peptic ulceration. Lancet. 1984;1:1311-1315. [PubMed] |
| 2. | Kandulski A, Selgrad M, Malfertheiner P. Helicobacter pylori infection: a clinical overview. Dig Liver Dis. 2008;40:619-626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 77] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 3. | McColl KE. Clinical practice. Helicobacter pylori infection. N Engl J Med. 2010;362:1597-1604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 630] [Cited by in RCA: 551] [Article Influence: 34.4] [Reference Citation Analysis (0)] |
| 4. | Lopes D, Nunes C, Martins MC, Sarmento B, Reis S. Eradication of Helicobacter pylori: Past, present and future. J Control Release. 2014;189:169-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 80] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 5. | Wang Y, Wang B, Lv ZF, Yang Y, Wang F, Wang H, Chen S, Xie Y, Zhou X. Efficacy and safety of ecabet sodium as an adjuvant therapy for Helicobacter pylori eradication: a systematic review and meta-analysis. Helicobacter. 2014;19:372-381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 6. | NIH Consensus Conference. Helicobacter pylori in peptic ulcer disease. NIH Consensus Development Panel on Helicobacter pylori in Peptic Ulcer Disease. JAMA. 1994;272:65-69. [PubMed] |
| 7. | Bytzer P, Dahlerup JF, Eriksen JR, Jarbøl DE, Rosenstock S, Wildt S. Diagnosis and treatment of Helicobacter pylori infection. Dan Med Bull. 2011;58:C4271. [PubMed] |
| 8. | Tytgat GN. Etiopathogenetic principles and peptic ulcer disease classification. Dig Dis. 2011;29:454-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 37] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 9. | Testerman TL, Morris J. Beyond the stomach: an updated view of Helicobacter pylori pathogenesis, diagnosis, and treatment. World J Gastroenterol. 2014;20:12781-12808. [PubMed] [DOI] [Full Text] |
| 10. | Georgopoulos SD, Papastergiou V, Karatapanis S. Current options for the treatment of Helicobacter pylori. Expert Opin Pharmacother. 2013;14:211-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 11. | Freeman HJ. Disappearance of Helicobacter without antibiotics in 12 patients with gastritis. Can J Gastroenterol. 1997;11:167-172. [PubMed] |
| 12. | Go MF. Review article: natural history and epidemiology of Helicobacter pylori infection. Aliment Pharmacol Ther. 2002;16 Suppl 1:3-15. [PubMed] |
| 13. | Fakheri H, Bari Z, Aarabi M, Malekzadeh R. Helicobacter pylori eradication in West Asia: a review. World J Gastroenterol. 2014;20:10355-10367. [PubMed] [DOI] [Full Text] |
| 14. | Bouvard V, Baan R, Straif K, Grosse Y, Secretan B, El Ghissassi F, Benbrahim-Tallaa L, Guha N, Freeman C, Galichet L. A review of human carcinogens--Part B: biological agents. Lancet Oncol. 2009;10:321-322. [PubMed] |
| 15. | Fukase K, Kato M, Kikuchi S, Inoue K, Uemura N, Okamoto S, Terao S, Amagai K, Hayashi S, Asaka M. Effect of eradication of Helicobacter pylori on incidence of metachronous gastric carcinoma after endoscopic resection of early gastric cancer: an open-label, randomised controlled trial. Lancet. 2008;372:392-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1023] [Cited by in RCA: 926] [Article Influence: 51.4] [Reference Citation Analysis (1)] |
| 16. | Suzuki H, Nishizawa T, Hibi T. Helicobacter pylori eradication therapy. Future Microbiol. 2010;5:639-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 82] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 17. | Nishizawa T, Nishizawa Y, Yahagi N, Kanai T, Takahashi M, Suzuki H. Effect of supplementation with rebamipide for Helicobacter pylori eradication therapy: a systematic review and meta-analysis. J Gastroenterol Hepatol. 2014;29 Suppl 4:20-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 18. | Molina-Infante J, Gisbert JP. Optimizing clarithromycin-containing therapy for Helicobacter pylori in the era of antibiotic resistance. World J Gastroenterol. 2014;20:10338-10347. [PubMed] [DOI] [Full Text] |
| 19. | Gisbert JP, González L, Calvet X, García N, López T, Roqué M, Gabriel R, Pajares JM. Proton pump inhibitor, clarithromycin and either amoxycillin or nitroimidazole: a meta-analysis of eradication of Helicobacter pylori. Aliment Pharmacol Ther. 2000;14:1319-1328. [PubMed] |
| 20. | Graham DY, Fischbach L. Helicobacter pylori treatment in the era of increasing antibiotic resistance. Gut. 2010;59:1143-1153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 789] [Cited by in RCA: 728] [Article Influence: 45.5] [Reference Citation Analysis (0)] |
| 21. | Gasparetto M, Pescarin M, Guariso G. Helicobacter pylori Eradication Therapy: Current Availabilities. ISRN Gastroenterol. 2012;2012:186734. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 22. | Xie C, Lu NH. Review: clinical management of Helicobacter pylori infection in China. Helicobacter. 2015;20:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 80] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 23. | Chung JW, Lee GH, Han JH, Jeong JY, Choi KS, Kim do H, Jung KW, Choi KD, Song HJ, Jung HY. The trends of one-week first-line and second-line eradication therapy for Helicobacter pylori infection in Korea. Hepatogastroenterology. 2011;58:246-250. [PubMed] |
| 24. | Yoon JH, Baik GH, Sohn KM, Kim DY, Kim YS, Suk KT, Kim JB, Kim DJ, Kim JB, Shin WG. Trends in the eradication rates of Helicobacter pylori infection for eleven years. World J Gastroenterol. 2012;18:6628-6634. [PubMed] [DOI] [Full Text] |
| 25. | Malfertheiner P, Megraud F, O’Morain CA, Atherton J, Axon AT, Bazzoli F, Gensini GF, Gisbert JP, Graham DY, Rokkas T. Management of Helicobacter pylori infection--the Maastricht IV/ Florence Consensus Report. Gut. 2012;61:646-664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1785] [Cited by in RCA: 1570] [Article Influence: 112.1] [Reference Citation Analysis (2)] |
| 26. | Boyanova L. Prevalence of multidrug-resistant Helicobacter pylori in Bulgaria. J Med Microbiol. 2009;58:930-935. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 27. | Nishizawa T, Suzuki H, Suzuki M, Takahashi M, Hibi T. Proton pump inhibitor-amoxicillin-clarithromycin versus proton pump inhibitor-amoxicillin-metronidazole as first-line Helicobacter pylori eradication therapy. J Clin Biochem Nutr. 2012;51:114-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 28. | Oh HS, Lee DH, Seo JY, Cho YR, Kim N, Jeoung SH, Kim JW, Hwang JH, Park YS, Lee SH. Ten-day sequential therapy is more effective than proton pump inhibitor-based therapy in Korea: a prospective, randomized study. J Gastroenterol Hepatol. 2012;27:504-509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 50] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 29. | Perri F, Villani MR, Festa V, Quitadamo M, Andriulli A. Predictors of failure of Helicobacter pylori eradication with the standard ‘Maastricht triple therapy’. Aliment Pharmacol Ther. 2001;15:1023-1029. [PubMed] |
| 30. | Ierardi E, Giorgio F, Losurdo G, Di Leo A, Principi M. How antibiotic resistances could change Helicobacter pylori treatment: A matter of geography. World J Gastroenterol. 2013;19:8168-8180. [PubMed] [DOI] [Full Text] |
| 31. | Oleastro M, Ménard A, Santos A, Lamouliatte H, Monteiro L, Barthélémy P, Mégraud F. Real-time PCR assay for rapid and accurate detection of point mutations conferring resistance to clarithromycin in Helicobacter pylori. J Clin Microbiol. 2003;41:397-402. [PubMed] |
| 32. | De Francesco V, Margiotta M, Zullo A, Hassan C, Troiani L, Burattini O, Stella F, Di Leo A, Russo F, Marangi S. Clarithromycin-resistant genotypes and eradication of Helicobacter pylori. Ann Intern Med. 2006;144:94-100. [PubMed] |
| 33. | Barile KA, Silva AL, Xavier JN, Assumpção MB, Corvelo TC. Characterization of 23S rRNA domain V mutations in gastric biopsy patients from the eastern Amazon. Mem Inst Oswaldo Cruz. 2010;105:314-317. [PubMed] |
| 34. | Lins AK, Lima RA, Magalhães M. Clarithromycin-resistant Helicobacter pylori in Recife, Brazil, directly identified from gastric biopsies by polymerase chain reaction. Arq Gastroenterol. 2010;47:379-382. [PubMed] |
| 35. | Lee JW, Kim N, Kim JM, Nam RH, Chang H, Kim JY, Shin CM, Park YS, Lee DH, Jung HC. Prevalence of primary and secondary antimicrobial resistance of Helicobacter pylori in Korea from 2003 through 2012. Helicobacter. 2013;18:206-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 174] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 36. | Kobayashi I, Murakami K, Kato M, Kato S, Azuma T, Takahashi S, Uemura N, Katsuyama T, Fukuda Y, Haruma K. Changing antimicrobial susceptibility epidemiology of Helicobacter pylori strains in Japan between 2002 and 2005. J Clin Microbiol. 2007;45:4006-4010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 125] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 37. | Murakami K, Furuta T, Ando T, Nakajima T, Inui Y, Oshima T, Tomita T, Mabe K, Sasaki M, Suganuma T. Multi-center randomized controlled study to establish the standard third-line regimen for Helicobacter pylori eradication in Japan. J Gastroenterol. 2013;48:1128-1135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 68] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 38. | Sun QJ, Liang X, Zheng Q, Gu WQ, Liu WZ, Xiao SD, Lu H. Resistance of Helicobacter pylori to antibiotics from 2000 to 2009 in Shanghai. World J Gastroenterol. 2010;16:5118-5121. [PubMed] |
| 39. | Su P, Li Y, Li H, Zhang J, Lin L, Wang Q, Guo F, Ji Z, Mao J, Tang W. Antibiotic resistance of Helicobacter pylori isolated in the Southeast Coastal Region of China. Helicobacter. 2013;18:274-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 133] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 40. | Binh TT, Shiota S, Nguyen LT, Ho DD, Hoang HH, Ta L, Trinh DT, Fujioka T, Yamaoka Y. The incidence of primary antibiotic resistance of Helicobacter pylori in Vietnam. J Clin Gastroenterol. 2013;47:233-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 46] [Article Influence: 3.5] [Reference Citation Analysis (2)] |
| 41. | Dos Santos AA, Carvalho AA. Pharmacological therapy used in the elimination of Helicobacter pylori infection: a review. World J Gastroenterol. 2015;21:139-154. [PubMed] [DOI] [Full Text] |
| 42. | Goh KL, Navaratnam P. High Helicobacter pylori resistance to metronidazole but zero or low resistance to clarithromycin, levofloxacin, and other antibiotics in Malaysia. Helicobacter. 2011;16:241-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 38] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 43. | Secka O, Berg DE, Antonio M, Corrah T, Tapgun M, Walton R, Thomas V, Galano JJ, Sancho J, Adegbola RA. Antimicrobial susceptibility and resistance patterns among Helicobacter pylori strains from The Gambia, West Africa. Antimicrob Agents Chemother. 2013;57:1231-1237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 35] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 44. | Seck A, Burucoa C, Dia D, Mbengue M, Onambele M, Raymond J, Breurec S. Primary antibiotic resistance and associated mechanisms in Helicobacter pylori isolates from Senegalese patients. Ann Clin Microbiol Antimicrob. 2013;12:3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 41] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 45. | De Francesco V, Giorgio F, Hassan C, Manes G, Vannella L, Panella C, Ierardi E, Zullo A. Worldwide H. pylori antibiotic resistance: a systematic review. J Gastrointestin Liver Dis. 2010;19:409-414. [PubMed] |
| 46. | Storskrubb T, Aro P, Ronkainen J, Wreiber K, Nyhlin H, Bolling-Sternevald E, Talley NJ, Engstrand L, Agréus L. Antimicrobial susceptibility of Helicobacter pylori strains in a random adult Swedish population. Helicobacter. 2006;11:224-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 34] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 47. | Selgrad M, Meissle J, Bornschein J, Kandulski A, Langner C, Varbanova M, Wex T, Tammer I, Schlüter D, Malfertheiner P. Antibiotic susceptibility of Helicobacter pylori in central Germany and its relationship with the number of eradication therapies. Eur J Gastroenterol Hepatol. 2013;25:1257-1260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 48] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 48. | De Francesco V, Margiotta M, Zullo A, Hassan C, Giorgio F, Burattini O, Stoppino G, Cea U, Pace A, Zotti M. Prevalence of primary clarithromycin resistance in Helicobacter pylori strains over a 15 year period in Italy. J Antimicrob Chemother. 2007;59:783-785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 77] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 49. | Molina-Infante J, Gisbert JP. [Update on the efficacy of triple therapy for Helicobacter pylori infection and clarithromycin resistance rates in Spain (2007-2012)]. Gastroenterol Hepatol. 2013;36:375-381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 50. | Oleastro M, Cabral J, Ramalho PM, Lemos PS, Paixão E, Benoliel J, Santos A, Lopes AI. Primary antibiotic resistance of Helicobacter pylori strains isolated from Portuguese children: a prospective multicentre study over a 10 year period. J Antimicrob Chemother. 2011;66:2308-2311. [PubMed] [DOI] [Full Text] |
| 51. | Karczewska E, Klesiewicz K, Skiba I, Wojtas-Bonior I, Sito E, Czajecki K, Zwolińska-Wcisło M, Budak A. Variability in Prevalence of Helicobacter pylori Strains Resistant to Clarithromycin and Levofloxacin in Southern Poland. Gastroenterol Res Pract. 2012;2012:418010. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 52. | Houben MH, van de Beek D, Hensen EF, de Craen AJ, Rauws EA, Tytgat GN. A systematic review of Helicobacter pylori eradication therapy--the impact of antimicrobial resistance on eradication rates. Aliment Pharmacol Ther. 1999;13:1047-1055. [PubMed] |
| 53. | Papastergiou V, Georgopoulos SD, Karatapanis S. Treatment of Helicobacter pylori infection: Past, present and future. World J Gastrointest Pathophysiol. 2014;5:392-399. [PubMed] [DOI] [Full Text] |
| 54. | Milani M, Ghotaslou R, Akhi MT, Nahaei MR, Hasani A, Somi MH, Rafeey M, Sharifi Y. The status of antimicrobial resistance of Helicobacter pylori in Eastern Azerbaijan, Iran: comparative study according to demographics. J Infect Chemother. 2012;18:848-852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 40] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 55. | Torres J, Camorlinga-Ponce M, Pérez-Pérez G, Madrazo-De la Garza A, Dehesa M, González-Valencia G, Muñoz O. Increasing multidrug resistance in Helicobacter pylori strains isolated from children and adults in Mexico. J Clin Microbiol. 2001;39:2677-2680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 84] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 56. | Duck WM, Sobel J, Pruckler JM, Song Q, Swerdlow D, Friedman C, Sulka A, Swaminathan B, Taylor T, Hoekstra M. Antimicrobial resistance incidence and risk factors among Helicobacter pylori-infected persons, United States. Emerg Infect Dis. 2004;10:1088-1094. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 163] [Cited by in RCA: 149] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 57. | Larsen AL, Ragnhildstveit E, Moayeri B, Eliassen L, Melby KK. Resistance rates of metronidazole and other antibacterials in Helicobacter pylori from previously untreated patients in Norway. APMIS. 2013;121:353-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 58. | Mégraud F. Current recommendations for Helicobacter pylori therapies in a world of evolving resistance. Gut Microbes. 2013;4:541-548. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 30] [Article Influence: 2.3] [Reference Citation Analysis (2)] |
| 59. | Rispo A, Capone P, Castiglione F, Pasquale L, Rea M, Caporaso N. Fluoroquinolone-based protocols for eradication of Helicobacter pylori. World J Gastroenterol. 2014;20:8947-8956. [PubMed] [DOI] [Full Text] |
| 60. | Rajper S, Khan E, Ahmad Z, Alam SM, Akbar A, Hasan R. Macrolide and fluoroquinolone resistance in Helicobacter pylori isolates: an experience at a tertiary care centre in Pakistan. J Pak Med Assoc. 2012;62:1140-1144. [PubMed] |
| 61. | Chung JW, Lee GH, Jeong JY, Lee SM, Jung JH, Choi KD, Song HJ, Jung HY, Kim JH. Resistance of Helicobacter pylori strains to antibiotics in Korea with a focus on fluoroquinolone resistance. J Gastroenterol Hepatol. 2012;27:493-497. [PubMed] [DOI] [Full Text] |
| 62. | Mégraud F. H pylori antibiotic resistance: prevalence, importance, and advances in testing. Gut. 2004;53:1374-1384. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 715] [Cited by in RCA: 678] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 63. | O’Connor A, Taneike I, Nami A, Fitzgerald N, Ryan B, Breslin N, O’Connor H, McNamara D, Murphy P, O’Morain C. Helicobacter pylori resistance rates for levofloxacin, tetracycline and rifabutin among Irish isolates at a reference centre. Ir J Med Sci. 2013;182:693-695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 22] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 64. | Wueppenhorst N, Stueger HP, Kist M, Glocker EO. High secondary resistance to quinolones in German Helicobacter pylori clinical isolates. J Antimicrob Chemother. 2013;68:1562-1566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 65. | Eisig JN, Silva FM, Barbuti RC, Navarro-Rodriguez T, Moraes-Filho JP, Pedrazzoli Jr J. Helicobacter pylori antibiotic resistance in Brazil: clarithromycin is still a good option. Arq Gastroenterol. 2011;48:261-264. [PubMed] |
| 66. | Current European concepts in the management of Helicobacter pylori infection. The Maastricht Consensus Report. European Helicobacter Pylori Study Group. Gut. 1997;41:8-13. [PubMed] |
| 67. | Lind T, Veldhuyzen van Zanten S, Unge P, Spiller R, Bayerdörffer E, O’Morain C, Bardhan KD, Bradette M, Chiba N, Wrangstadh M. Eradication of Helicobacter pylori using one-week triple therapies combining omeprazole with two antimicrobials: the MACH I Study. Helicobacter. 1996;1:138-144. [PubMed] |
| 68. | Romano M, Cuomo A. Eradication of Helicobacter pylori: a clinical update. MedGenMed. 2004;6:19. [PubMed] |
| 69. | Nishizawa T, Suzuki H, Takahashi M, Suzuki M, Hibi T. Delay of second-line eradication therapy for Helicobacter pylori can increase eradication failure. J Gastroenterol Hepatol. 2013;28:1608-1610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 70. | Graham DY. Helicobacter pylori update: gastric cancer, reliable therapy, and possible benefits. Gastroenterology. 2015;148:719-731.e3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 373] [Cited by in RCA: 338] [Article Influence: 30.7] [Reference Citation Analysis (2)] |
| 71. | Calvet X, García N, López T, Gisbert JP, Gené E, Roque M. A meta-analysis of short versus long therapy with a proton pump inhibitor, clarithromycin and either metronidazole or amoxycillin for treating Helicobacter pylori infection. Aliment Pharmacol Ther. 2000;14:603-609. [PubMed] |
| 72. | Yuan Y, Ford AC, Khan KJ, Gisbert JP, Forman D, Leontiadis GI, Tse F, Calvet X, Fallone C, Fischbach L. Optimum duration of regimens for Helicobacter pylori eradication. Cochrane Database Syst Rev. 2013;12:CD008337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 103] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 73. | Fuccio L, Minardi ME, Zagari RM, Grilli D, Magrini N, Bazzoli F. Meta-analysis: duration of first-line proton-pump inhibitor based triple therapy for Helicobacter pylori eradication. Ann Intern Med. 2007;147:553-562. [PubMed] |
| 74. | Smith SM, Haider RB, O’Connor H, McNamara D, O’Morain C. Practical treatment of Helicobacter pylori: a balanced view in changing times. Eur J Gastroenterol Hepatol. 2014;26:819-825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 75. | Vallve M, Vergara M, Gisbert JP, Calvet X. Single vs. double dose of a proton pump inhibitor in triple therapy for Helicobacter pylori eradication: a meta-analysis. Aliment Pharmacol Ther. 2002;16:1149-1156. [PubMed] |
| 76. | Villoria A, Garcia P, Calvet X, Gisbert JP, Vergara M. Meta-analysis: high-dose proton pump inhibitors vs. standard dose in triple therapy for Helicobacter pylori eradication. Aliment Pharmacol Ther. 2008;28:868-877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 78] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 77. | Nagaraja V, Eslick GD. Evidence-based assessment of proton-pump inhibitors in Helicobacter pylori eradication: a systematic review. World J Gastroenterol. 2014;20:14527-14536. [PubMed] [DOI] [Full Text] |
| 78. | Luther J, Higgins PD, Schoenfeld PS, Moayyedi P, Vakil N, Chey WD. Empiric quadruple vs. triple therapy for primary treatment of Helicobacter pylori infection: Systematic review and meta-analysis of efficacy and tolerability. Am J Gastroenterol. 2010;105:65-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 150] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 79. | Liu KS, Hung IF, Seto WK, Tong T, Hsu AS, Lam FY, But DY, Wong SY, Leung WK. Ten day sequential versus 10 day modified bismuth quadruple therapy as empirical firstline and secondline treatment for Helicobacter pylori in Chinese patients: an open label, randomised, crossover trial. Gut. 2014;63:1410-1415. [PubMed] [DOI] [Full Text] |
| 80. | Salazar CO, Cardenas VM, Reddy RK, Dominguez DC, Snyder LK, Graham DY. Greater than 95% success with 14-day bismuth quadruple anti- Helicobacter pylori therapy: a pilot study in US Hispanics. Helicobacter. 2012;17:382-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 37] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 81. | Sapmaz F, Kalkan IH, Güliter S, Atasoy P. Comparison of Helicobacter pylori eradication rates of standard 14-day quadruple treatment and novel modified 10-day, 12-day and 14-day sequential treatments. Eur J Intern Med. 2014;25:224-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 82. | Uygun A, Kadayifci A, Safali M, Ilgan S, Bagci S. The efficacy of bismuth containing quadruple therapy as a first-line treatment option for Helicobacter pylori. J Dig Dis. 2007;8:211-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 83. | Kadayifci A, Uygun A, Polat Z, Kantarcioğlu M, Kılcıler G, Başer O, Ozcan A, Emer O. Comparison of bismuth-containing quadruple and concomitant therapies as a first-line treatment option for Helicobacter pylori. Turk J Gastroenterol. 2012;23:8-13. [PubMed] |
| 84. | Zullo A, Rinaldi V, Winn S, Meddi P, Lionetti R, Hassan C, Ripani C, Tomaselli G, Attili AF. A new highly effective short-term therapy schedule for Helicobacter pylori eradication. Aliment Pharmacol Ther. 2000;14:715-718. [PubMed] |
| 85. | Nasa M, Choksey A, Phadke A, Sawant P. Sequential therapy versus standard triple-drug therapy for Helicobacter pylori eradication: a randomized study. Indian J Gastroenterol. 2013;32:392-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 86. | Gatta L, Vakil N, Vaira D, Scarpignato C. Global eradication rates for Helicobacter pylori infection: systematic review and meta-analysis of sequential therapy. BMJ. 2013;347:f4587. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 207] [Cited by in RCA: 193] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 87. | Chung JW, Ha M, Yun SC, Kim JH, Lee JJ, Kim YJ, Kim KO, Kwon KA, Park DK, Lee DH. Meta-analysis: Sequential therapy is superior to conventional therapy for Helicobacter pylori infection in Korea. Korean J Gastroenterol. 2013;62:267-271. [PubMed] |
| 88. | Murakami K, Fujioka T, Okimoto T, Sato R, Kodama M, Nasu M. Drug combinations with amoxycillin reduce selection of clarithromycin resistance during Helicobacter pylori eradication therapy. Int J Antimicrob Agents. 2002;19:67-70. [PubMed] |
| 89. | Webber MA, Piddock LJ. The importance of efflux pumps in bacterial antibiotic resistance. J Antimicrob Chemother. 2003;51:9-11. [PubMed] |
| 90. | Maconi G, Parente F, Russo A, Vago L, Imbesi V, Bianchi Porro G. Do some patients with Helicobacter pylori infection benefit from an extension to 2 weeks of a proton pump inhibitor-based triple eradication therapy. Am J Gastroenterol. 2001;96:359-366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 41] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 91. | Lee H, Hong SN, Min BH, Lee JH, Rhee PL, Lee YC, Kim JJ. Comparison of efficacy and safety of levofloxacin-containing versus standard sequential therapy in eradication of Helicobacter pylori infection in Korea. Dig Liver Dis. 2015;47:114-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 92. | Lee JW, Kim N, Kim JM, Nam RH, Kim JY, Lee JY, Lee DH, Jung HC. A comparison between 15-day sequential, 10-day sequential and proton pump inhibitor-based triple therapy for Helicobacter pylori infection in Korea. Scand J Gastroenterol. 2014;49:917-924. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 93. | Zhou L, Zhang J, Chen M, Hou X, Li Z, Song Z, He L, Lin S. A comparative study of sequential therapy and standard triple therapy for Helicobacter pylori infection: a randomized multicenter trial. Am J Gastroenterol. 2014;109:535-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 74] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 94. | Ben Chaabane N, Al-Adhba HS. Ciprofloxacin-containing versus clarithromycin-containing sequential therapy for Helicobacter pylori eradication: A randomized trial. Indian J Gastroenterol. 2015;34:68-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 95. | Kang BK, Park SM, Kim BW. [New therapeutic strategies against Helicobacter pylori]. Korean J Gastroenterol. 2014;63:146-150. [PubMed] |
| 96. | Gisbert JP, Calvet X. Update on non-bismuth quadruple (concomitant) therapy for eradication of Helicobacter pylori. Clin Exp Gastroenterol. 2012;5:23-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 102] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 97. | Molina-Infante J, Pazos-Pacheco C, Vinagre-Rodriguez G, Perez-Gallardo B, Dueñas-Sadornil C, Hernandez-Alonso M, Gonzalez-Garcia G, Mateos-Rodriguez JM, Fernandez-Bermejo M, Gisbert JP. Nonbismuth quadruple (concomitant) therapy: empirical and tailored efficacy versus standard triple therapy for clarithromycin-susceptible Helicobacter pylori and versus sequential therapy for clarithromycin-resistant strains. Helicobacter. 2012;17:269-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 67] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 98. | Gisbert JP, Calvet X. Review article: non-bismuth quadruple (concomitant) therapy for eradication of Helicobater pylori. Aliment Pharmacol Ther. 2011;34:604-617. [PubMed] [DOI] [Full Text] |
| 99. | Essa AS, Kramer JR, Graham DY, Treiber G. Meta-analysis: four-drug, three-antibiotic, non-bismuth-containing “concomitant therapy” versus triple therapy for Helicobacter pylori eradication. Helicobacter. 2009;14:109-118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 170] [Cited by in RCA: 170] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 100. | Lee HJ, Kim JI, Lee JS, Jun EJ, Oh JH, Cheung DY, Chung WC, Kim BW, Kim SS. Concomitant therapy achieved the best eradication rate for Helicobacter pylori among various treatment strategies. World J Gastroenterol. 2015;21:351-359. [PubMed] [DOI] [Full Text] |
| 101. | Kim SY, Park DK, Kwon KA, Kim KO, Kim YJ, Chung JW. [Ten day concomitant therapy is superior to ten day sequential therapy for Helicobacter pylori eradication]. Korean J Gastroenterol. 2014;64:260-267. [PubMed] |
| 102. | Ang TL, Fock KM, Song M, Ang D, Kwek AB, Ong J, Tan J, Teo EK, Dhamodaran S. Ten-day triple therapy versus sequential therapy versus concomitant therapy as first-line treatment for Helicobacter pylori infection. J Gastroenterol Hepatol. 2015;30:1134-1139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 103. | McNicholl AG, Marin AC, Molina-Infante J, Castro M, Barrio J, Ducons J, Calvet X, de la Coba C, Montoro M, Bory F. Randomised clinical trial comparing sequential and concomitant therapies for Helicobacter pylori eradication in routine clinical practice. Gut. 2014;63:244-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 96] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 104. | Lim JH, Lee DH, Choi C, Lee ST, Kim N, Jeong SH, Kim JW, Hwang JH, Park YS, Lee SH. Clinical outcomes of two-week sequential and concomitant therapies for Helicobacter pylori eradication: a randomized pilot study. Helicobacter. 2013;18:180-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 65] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 105. | De Francesco V, Hassan C, Ridola L, Giorgio F, Ierardi E, Zullo A. Sequential, concomitant and hybrid first-line therapies for Helicobacter pylori eradication: a prospective randomized study. J Med Microbiol. 2014;63:748-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 70] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 106. | Greenberg ER, Anderson GL, Morgan DR, Torres J, Chey WD, Bravo LE, Dominguez RL, Ferreccio C, Herrero R, Lazcano-Ponce EC. 14-day triple, 5-day concomitant, and 10-day sequential therapies for Helicobacter pylori infection in seven Latin American sites: a randomised trial. Lancet. 2011;378:507-514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 204] [Article Influence: 13.6] [Reference Citation Analysis (1)] |
| 107. | Zullo A, Scaccianoce G, De Francesco V, Ruggiero V, D’Ambrosio P, Castorani L, Bonfrate L, Vannella L, Hassan C, Portincasa P. Concomitant, sequential, and hybrid therapy for H. pylori eradication: a pilot study. Clin Res Hepatol Gastroenterol. 2013;37:647-650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 68] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 108. | Graham DY, Lee YC, Wu MS. Rational Helicobacter pylori therapy: evidence-based medicine rather than medicine-based evidence. Clin Gastroenterol Hepatol. 2014;12:177-186.e3; Discussion e12-13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 280] [Cited by in RCA: 264] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 109. | Lee JY, Kim N. [Future trends of Helicobacter pylori eradication therapy in Korea]. Korean J Gastroenterol. 2014;63:158-170. [PubMed] |
| 110. | Schito AM, Schito GC. Levofloxacin, a broad spectrum anti-infective: from Streptococcus pneumoniae to Pseudomonas aeruginosa. J Chemother. 2004;16 Suppl 2:3-7. [PubMed] |
| 111. | Just PM. Overview of the fluoroquinolone antibiotics. Pharmacotherapy. 1993;13:4S-17S. [PubMed] |
| 112. | O’Connor A, Gisbert JP, McNamara D, O’Morain C. Treatment of Helicobacter pylori infection 2011. Helicobacter. 2011;16 Suppl 1:53-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 113. | Saad RJ, Schoenfeld P, Kim HM, Chey WD. Levofloxacin-based triple therapy versus bismuth-based quadruple therapy for persistent Helicobacter pylori infection: a meta-analysis. Am J Gastroenterol. 2006;101:488-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 72] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 114. | Molina-Infante J, Perez-Gallardo B, Fernandez-Bermejo M, Hernandez-Alonso M, Vinagre G, Dueñas C, Mateos-Rodriguez JM, Gonzalez-Garcia G, Abadia EG, Gisbert JP. Clinical trial: clarithromycin vs. levofloxacin in first-line triple and sequential regimens for Helicobacter pylori eradication. Aliment Pharmacol Ther. 2010;31:1077-1084. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 51] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 115. | Ye CL, Liao GP, He S, Pan YN, Kang YB, Zhang ZY. Levofloxacin and proton pump inhibitor-based triple therapy versus standard triple first-line therapy for Helicobacter pylori eradication. Pharmacoepidemiol Drug Saf. 2014;23:443-455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 116. | Peedikayil MC, Alsohaibani FI, Alkhenizan AH. Levofloxacin-based first-line therapy versus standard first-line therapy for Helicobacter pylori eradication: meta-analysis of randomized controlled trials. PLoS One. 2014;9:e85620. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 117. | Chen MC, Lei WY, Lin JS, Yi CH, Wu DC, Hu CT. Levofloxacin-amoxicillin/clavulanate-rabeprazole versus a standard seven-day triple therapy for eradication of Helicobacter pylori infection. Biomed Res Int. 2014;2014:158520. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 118. | Xiao SP, Gu M, Zhang GX. Is levofloxacin-based triple therapy an alternative for first-line eradication of Helicobacter pylori A systematic review and meta-analysis. Scand J Gastroenterol. 2014;49:528-538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 119. | Graham DY, Shiotani A. Which Therapy for Helicobacter pylori Infection. Gastroenterology. 2012;143:10-12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 27] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 120. | Shiota S, Reddy R, Alsarraj A, El-Serag HB, Graham DY. Antibiotic Resistance of Helicobacter pylori Among Male United States Veterans. Clin Gastroenterol Hepatol. 2015;13:1616-1624. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 124] [Cited by in RCA: 121] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 121. | Ontsira Ngoyi EN, Atipo Ibara BI, Moyen R, Ahoui Apendi PC, Ibara JR, Obengui O, Ossibi Ibara RB, Nguimbi E, Niama RF, Ouamba JM. Molecular Detection of Helicobacter pylori and its Antimicrobial Resistance in Brazzaville, Congo. Helicobacter. 2015;20:316-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 122. | Liao J, Zheng Q, Liang X, Zhang W, Sun Q, Liu W, Xiao S, Graham DY, Lu H. Effect of fluoroquinolone resistance on 14-day levofloxacin triple and triple plus bismuth quadruple therapy. Helicobacter. 2013;18:373-377. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 93] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 123. | Kim JY, Kim NY, Kim SJ, Baik GH, Kim GH, Kim JM, Nam RH, Kim HB, Lee DH, Jung HC. [Regional difference of antibiotic resistance of helicobacter pylori strains in Korea]. Korean J Gastroenterol. 2011;57:221-229. [PubMed] [DOI] [Full Text] |
| 124. | Furuta T, Sugimoto M, Kodaira C, Nishino M, Yamade M, Uotani T, Sahara S, Ichikawa H, Yamada T, Osawa S. Sitafloxacin-based third-line rescue regimens for Helicobacter pylori infection in Japan. J Gastroenterol Hepatol. 2014;29:487-493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 125. | Akcam M, Koca T, Salman H, Karahan N. The effects of probiotics on treatment of Helicobacter pylori eradication in children. Saudi Med J. 2015;36:286-290. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 126. | Navarro-Rodriguez T, Silva FM, Barbuti RC, Mattar R, Moraes-Filho JP, de Oliveira MN, Bogsan CS, Chinzon D, Eisig JN. Association of a probiotic to a Helicobacter pylori eradication regimen does not increase efficacy or decreases the adverse effects of the treatment: a prospective, randomized, double-blind, placebo-controlled study. BMC Gastroenterol. 2013;13:56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 50] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 127. | Lv Z, Wang B, Zhou X, Wang F, Xie Y, Zheng H, Lv N. Efficacy and safety of probiotics as adjuvant agents for Helicobacter pylori infection: A meta-analysis. Exp Ther Med. 2015;9:707-716. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 81] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 128. | Zhu R, Chen K, Zheng YY, Zhang HW, Wang JS, Xia YJ, Dai WQ, Wang F, Shen M, Cheng P. Meta-analysis of the efficacy of probiotics in Helicobacter pylori eradication therapy. World J Gastroenterol. 2014;20:18013-18021. [PubMed] [DOI] [Full Text] |
| 129. | Li S, Huang XL, Sui JZ, Chen SY, Xie YT, Deng Y, Wang J, Xie L, Li TJ, He Y. Meta-analysis of randomized controlled trials on the efficacy of probiotics in Helicobacter pylori eradication therapy in children. Eur J Pediatr. 2014;173:153-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 60] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 130. | Song MJ, Park DI, Park JH, Kim HJ, Cho YK, Sohn CI, Jeon WK, Kim BI. The effect of probiotics and mucoprotective agents on PPI-based triple therapy for eradication of Helicobacter pylori. Helicobacter. 2010;15:206-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 62] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 131. | Szajewska H, Horvath A, Piwowarczyk A. Meta-analysis: the effects of Saccharomyces boulardii supplementation on Helicobacter pylori eradication rates and side effects during treatment. Aliment Pharmacol Ther. 2010;32:1069-1079. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 150] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 132. | Wang ZH, Gao QY, Fang JY. Meta-analysis of the efficacy and safety of Lactobacillus-containing and Bifidobacterium-containing probiotic compound preparation in Helicobacter pylori eradication therapy. J Clin Gastroenterol. 2013;47:25-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 114] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 133. | Wang YH, Huang Y. Effect of Lactobacillus acidophilus and Bifidobacterium bifidum supplementation to standard triple therapy on Helicobacter pylori eradication and dynamic changes in intestinal flora. World J Microbiol Biotechnol. 2014;30:847-853. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 57] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 134. | Francavilla R, Polimeno L, Demichina A, Maurogiovanni G, Principi B, Scaccianoce G, Ierardi E, Russo F, Riezzo G, Di Leo A. Lactobacillus reuteri strain combination in Helicobacter pylori infection: a randomized, double-blind, placebo-controlled study. J Clin Gastroenterol. 2014;48:407-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 88] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 135. | Hwang TJ, Kim N, Kim HB, Lee BH, Nam RH, Park JH, Lee MK, Park YS, Lee DH, Jung HC. Change in antibiotic resistance of Helicobacter pylori strains and the effect of A2143G point mutation of 23S rRNA on the eradication of H. pylori in a single center of Korea. J Clin Gastroenterol. 2010;44:536-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 105] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 136. | Gerrits MM, van Vliet AH, Kuipers EJ, Kusters JG. Helicobacter pylori and antimicrobial resistance: molecular mechanisms and clinical implications. Lancet Infect Dis. 2006;6:699-709. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 223] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 137. | Woo HY, Park DI, Park H, Kim MK, Kim DH, Kim IS, Kim YJ. Dual-priming oligonucleotide-based multiplex PCR for the detection of Helicobacter pylori and determination of clarithromycin resistance with gastric biopsy specimens. Helicobacter. 2009;14:22-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 70] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 138. | Lee HJ, Kim JI, Cheung DY, Kim TH, Jun EJ, Oh JH, Chung WC, Kim BW, Kim SS, Park SH. Eradication of Helicobacter pylori according to 23S ribosomal RNA point mutations associated with clarithromycin resistance. J Infect Dis. 2013;208:1123-1130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 83] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 139. | Kuo CH, Lu CY, Shih HY, Liu CJ, Wu MC, Hu HM, Hsu WH, Yu FJ, Wu DC, Kuo FC. CYP2C19 polymorphism influences Helicobacter pylori eradication. World J Gastroenterol. 2014;20:16029-16036. [PubMed] [DOI] [Full Text] |
| 140. | Ishizaki T, Horai Y. Review article: cytochrome P450 and the metabolism of proton pump inhibitors--emphasis on rabeprazole. Aliment Pharmacol Ther. 1999;13 Suppl 3:27-36. [PubMed] |
| 141. | Chong E, Ensom MH. Pharmacogenetics of the proton pump inhibitors: a systematic review. Pharmacotherapy. 2003;23:460-471. [PubMed] |
| 142. | Ishizaki T, Sohn DR, Kobayashi K, Chiba K, Lee KH, Shin SG, Andersson T, Regårdh CG, Lou YC, Zhang Y. Interethnic differences in omeprazole metabolism in the two S-mephenytoin hydroxylation phenotypes studied in Caucasians and Orientals. Ther Drug Monit. 1994;16:214-215. [PubMed] |
| 143. | Padol S, Yuan Y, Thabane M, Padol IT, Hunt RH. The effect of CYP2C19 polymorphisms on H. pylori eradication rate in dual and triple first-line PPI therapies: a meta-analysis. Am J Gastroenterol. 2006;101:1467-1475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 113] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 144. | Zhao F, Wang J, Yang Y, Wang X, Shi R, Xu Z, Huang Z, Zhang G. Effect of CYP2C19 genetic polymorphisms on the efficacy of proton pump inhibitor-based triple therapy for Helicobacter pylori eradication: a meta-analysis. Helicobacter. 2008;13:532-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 81] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 145. | Tang HL, Li Y, Hu YF, Xie HG, Zhai SD. Effects of CYP2C19 loss-of-function variants on the eradication of H. pylori infection in patients treated with proton pump inhibitor-based triple therapy regimens: a meta-analysis of randomized clinical trials. PLoS One. 2013;8:e62162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 75] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 146. | Sugimoto M, Shirai N, Nishino M, Kodaira C, Uotani T, Sahara S, Ichikawa H, Kagami T, Sugimoto K, Furuta T. Comparison of acid inhibition with standard dosages of proton pump inhibitors in relation to CYP2C19 genotype in Japanese. Eur J Clin Pharmacol. 2014;70:1073-1078. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 147. | Sahara S, Sugimoto M, Uotani T, Ichikawa H, Yamade M, Iwaizumi M, Yamada T, Osawa S, Sugimoto K, Umemura K. Twice-daily dosing of esomeprazole effectively inhibits acid secretion in CYP2C19 rapid metabolisers compared with twice-daily omeprazole, rabeprazole or lansoprazole. Aliment Pharmacol Ther. 2013;38:1129-1137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 55] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 148. | Sugimoto M, Uotani T, Sahara S, Ichikawa H, Yamade M, Sugimoto K, Furuta T. Efficacy of tailored Helicobacter pylori eradication treatment based on clarithromycin susceptibility and maintenance of acid secretion. Helicobacter. 2014;19:312-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 51] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 149. | Fiorini G, Zullo A, Gatta L, Castelli V, Ricci C, Cassol F, Vaira D. Newer agents for Helicobacter pylori eradication. Clin Exp Gastroenterol. 2012;5:109-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 150. | Makobongo MO, Gilbreath JJ, Merrell DS. Nontraditional therapies to treat Helicobacter pylori infection. J Microbiol. 2014;52:259-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 151. | Leszczyńska K, Namiot A, Fein DE, Wen Q, Namiot Z, Savage PB, Diamond S, Janmey PA, Bucki R. Bactericidal activities of the cationic steroid CSA-13 and the cathelicidin peptide LL-37 against Helicobacter pylori in simulated gastric juice. BMC Microbiol. 2009;9:187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 42] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 152. | Zhang L, Yu J, Wong CC, Ling TK, Li ZJ, Chan KM, Ren SX, Shen J, Chan RL, Lee CC. Cathelicidin protects against Helicobacter pylori colonization and the associated gastritis in mice. Gene Ther. 2013;20:751-760. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 39] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 153. | Uehara N, Yagihashi A, Kondoh K, Tsuji N, Fujita T, Hamada H, Watanabe N. Human beta-defensin-2 induction in Helicobacter pylori-infected gastric mucosal tissues: antimicrobial effect of overexpression. J Med Microbiol. 2003;52:41-45. [PubMed] |
| 154. | Bauer B, Wex T, Kuester D, Meyer T, Malfertheiner P. Differential expression of human beta defensin 2 and 3 in gastric mucosa of Helicobacter pylori-infected individuals. Helicobacter. 2013;18:6-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 42] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 155. | Makobongo MO, Gancz H, Carpenter BM, McDaniel DP, Merrell DS. The oligo-acyl lysyl antimicrobial peptide C12K-2β12 exhibits a dual mechanism of action and demonstrates strong in vivo efficacy against Helicobacter pylori. Antimicrob Agents Chemother. 2012;56:378-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 27] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 156. | Makobongo MO, Einck L, Peek RM, Merrell DS. In vitro characterization of the anti-bacterial activity of SQ109 against Helicobacter pylori. PLoS One. 2013;8:e68917. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 37] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 157. | Geng B, Basarab G, Comita-Prevoir J, Gowravaram M, Hill P, Kiely A, Loch J, MacPherson L, Morningstar M, Mullen G. Potent and selective inhibitors of Helicobacter pylori glutamate racemase (MurI): pyridodiazepine amines. Bioorg Med Chem Lett. 2009;19:930-936. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 37] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 158. | Nishimori I, Minakuchi T, Kohsaki T, Onishi S, Takeuchi H, Vullo D, Scozzafava A, Supuran CT. Carbonic anhydrase inhibitors: the beta-carbonic anhydrase from Helicobacter pylori is a new target for sulfonamide and sulfamate inhibitors. Bioorg Med Chem Lett. 2007;17:3585-3594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 138] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 159. | Haniadka R, Saldanha E, Sunita V, Palatty PL, Fayad R, Baliga MS. A review of the gastroprotective effects of ginger (Zingiber officinale Roscoe). Food Funct. 2013;4:845-855. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 133] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 160. | Lee IO, Lee KH, Pyo JH, Kim JH, Choi YJ, Lee YC. Anti-inflammatory effect of capsaicin in Helicobacter pylori-infected gastric epithelial cells. Helicobacter. 2007;12:510-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 58] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 161. | Yanaka A, Fahey JW, Fukumoto A, Nakayama M, Inoue S, Zhang S, Tauchi M, Suzuki H, Hyodo I, Yamamoto M. Dietary sulforaphane-rich broccoli sprouts reduce colonization and attenuate gastritis in Helicobacter pylori-infected mice and humans. Cancer Prev Res (Phila). 2009;2:353-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 195] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 162. | Park S, Yeo M, Jin JH, Lee KM, Kim SS, Choi SY, Hahm KB. Inhibitory activities and attenuated expressions of 5-LOX with red ginseng in Helicobacter pylori-infected gastric epithelial cells. Dig Dis Sci. 2007;52:973-982. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 163. | Lee KM, Yeo M, Choue JS, Jin JH, Park SJ, Cheong JY, Lee KJ, Kim JH, Hahm KB. Protective mechanism of epigallocatechin-3-gallate against Helicobacter pylori-induced gastric epithelial cytotoxicity via the blockage of TLR-4 signaling. Helicobacter. 2004;9:632-642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 60] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 164. | Mahady GB, Pendland SL, Chadwick LR. Resveratrol and red wine extracts inhibit the growth of CagA+ strains of Helicobacter pylori in vitro. Am J Gastroenterol. 2003;98:1440-1441. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 58] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 165. | Sezikli M, Cetinkaya ZA, Sezikli H, Güzelbulut F, Tiftikçi A, Ince AT, Gökden Y, Yaşar B, Atalay S, Kurdaş OO. Oxidative stress in Helicobacter pylori infection: does supplementation with vitamins C and E increase the eradication rate. Helicobacter. 2009;14:280-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 34] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 166. | Obonyo M, Zhang L, Thamphiwatana S, Pornpattananangkul D, Fu V, Zhang L. Antibacterial activities of liposomal linolenic acids against antibiotic-resistant Helicobacter pylori. Mol Pharm. 2012;9:2677-2685. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 83] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 167. | Moss SF, Moise L, Lee DS, Kim W, Zhang S, Lee J, Rogers AB, Martin W, De Groot AS. HelicoVax: epitope-based therapeutic Helicobacter pylori vaccination in a mouse model. Vaccine. 2011;29:2085-2091. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 68] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 168. | Singh DY, Prasad NK. Double liposomes mediated dual drug targeting for treatment of Helicobacter pylori infections. Pharmazie. 2011;66:368-373. [PubMed] |
| 169. | Ramteke S, Jain NK. Clarithromycin- and omeprazole-containing gliadin nanoparticles for the treatment of Helicobacter pylori. J Drug Target. 2008;16:65-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 44] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 170. | Wang J, Tauchi Y, Deguchi Y, Morimoto K, Tabata Y, Ikada Y. Positively charged gelatin microspheres as gastric mucoadhesive drug delivery system for eradication of H. pylori. Drug Deliv. 2000;7:237-243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 79] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 171. | Patel JK, Patel MM. Stomach specific anti-helicobacter pylori therapy: preparation and evaluation of amoxicillin-loaded chitosan mucoadhesive microspheres. Curr Drug Deliv. 2007;4:41-50. [PubMed] |
| 172. | Luo D, Guo J, Wang F, Sun J, Li G, Cheng X, Chang M, Yan X. Preparation and evaluation of anti-Helicobacter pylori efficacy of chitosan nanoparticles in vitro and in vivo. J Biomater Sci Polym Ed. 2009;20:1587-1596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 20] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 173. | Lin YH, Tsai SC, Lai CH, Lee CH, He ZS, Tseng GC. Genipin-cross-linked fucose-chitosan/heparin nanoparticles for the eradication of Helicobacter pylori. Biomaterials. 2013;34:4466-4479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 90] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 174. | Hao S, Wang Y, Wang B, Zou Q, Zeng H, Chen X, Liu X, Liu J, Yu S. A novel gastroretentive porous microparticle for anti-Helicobacter pylori therapy: preparation, in vitro and in vivo evaluation. Int J Pharm. 2014;463:10-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 175. | Amin M, Hameed S, Ali A, Anwar F, Shahid SA, Shakir I, Yaqoob A, Hasan S, Khan SA. Green Synthesis of Silver Nanoparticles: Structural Features and In Vivo and In Vitro Therapeutic Effects against Helicobacter pylori Induced Gastritis. Bioinorg Chem Appl. 2014;2014:135824. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 176. | Lin YH, Lin JH, Chou SC, Chang SJ, Chung CC, Chen YS, Chang CH. Berberine-loaded targeted nanoparticles as specific Helicobacter pylori eradication therapy: in vitro and in vivo study. Nanomedicine (Lond). 2015;10:57-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 68] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 177. | Na HS, Hong SJ, Yoon HJ, Maeng JH, Ko BM, Jung IS, Ryu CB, Kim JO, Cho JY, Lee JS. [Eradication rate of first-line and second-line therapy for Helicobacter pylori infection, and reinfection rate after successful eradication]. Korean J Gastroenterol. 2007;50:170-175. [PubMed] |
| 178. | Chung JW, Jung YK, Kim YJ, Kwon KA, Kim JH, Lee JJ, Lee SM, Hahm KB, Lee SM, Jeong JY. Ten-day sequential versus triple therapy for Helicobacter pylori eradication: a prospective, open-label, randomized trial. J Gastroenterol Hepatol. 2012;27:1675-1680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 51] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 179. | Asaka M, Sugiyama T, Kato M, Satoh K, Kuwayama H, Fukuda Y, Fujioka T, Takemoto T, Kimura K, Shimoyama T. A multicenter, double-blind study on triple therapy with lansoprazole, amoxicillin and clarithromycin for eradication of Helicobacter pylori in Japanese peptic ulcer patients. Helicobacter. 2001;6:254-261. [PubMed] |
| 180. | Fujioka T, Aoyama N, Sakai K, Miwa Y, Kudo M, Kawashima J, Matsubara Y, Miwa J, Yakabi K. A large-scale nationwide multicenter prospective observational study of triple therapy using rabeprazole, amoxicillin, and clarithromycin for Helicobacter pylori eradication in Japan. J Gastroenterol. 2012;47:276-283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 181. | Nishida T, Tsujii M, Tanimura H, Tsutsui S, Tsuji S, Takeda A, Inoue A, Fukui H, Yoshio T, Kishida O. Comparative study of esomeprazole and lansoprazole in triple therapy for eradication of Helicobacter pylori in Japan. World J Gastroenterol. 2014;20:4362-4369. [PubMed] [DOI] [Full Text] |
| 182. | Sheu BS, Lee SC, Yang HB, Kuo AW, Wang YL, Shiesh SC, Wu JJ, Lin XZ. Selection of lower cutoff point of [13C]urea breath test is helpful to monitor H. pylori eradication after proton pump inhibitor-based triple therapy. Dig Dis Sci. 2000;45:1330-1336. [PubMed] |
| 183. | Ozçay F, Koçak N, Temizel IN, Demir H, Ozen H, Yüce A, Gürakan F. Helicobacter pylori infection in Turkish children: comparison of diagnostic tests, evaluation of eradication rate, and changes in symptoms after eradication. Helicobacter. 2004;9:242-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 39] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 184. | Kutluk G, Tutar E, Bayrak A, Volkan B, Akyon Y, Celikel C, Ertem D. Sequential therapy versus standard triple therapy for Helicobacter pylori eradication in children: any advantage in clarithromycin-resistant strains. Eur J Gastroenterol Hepatol. 2014;26:1202-1208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 185. | Catalano F, Branciforte G, Catanzaro R, Bentivegna C, Cipolla R, Nuciforo G, Brogna A. Comparative treatment of Helicobacter pylori-positive duodenal ulcer using pantoprazole at low and high doses versus omeprazole in triple therapy. Helicobacter. 1999;4:178-184. [PubMed] |
| 186. | Paoluzi OA, Visconti E, Andrei F, Tosti C, Lionetti R, Grasso E, Ranaldi R, Stroppa I, Pallone F. Ten and eight-day sequential therapy in comparison to standard triple therapy for eradicating Helicobacter pylori infection: a randomized controlled study on efficacy and tolerability. J Clin Gastroenterol. 2010;44:261-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 49] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 187. | Pontone S, Standoli M, Angelini R, Pontone P. Efficacy of H. pylori eradication with a sequential regimen followed by rescue therapy in clinical practice. Dig Liver Dis. 2010;42:541-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 188. | Kongchayanun C, Vilaichone RK, Pornthisarn B, Amornsawadwattana S, Mahachai V. Pilot studies to identify the optimum duration of concomitant Helicobacter pylori eradication therapy in Thailand. Helicobacter. 2012;17:282-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 32] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 189. | Choi KH, Chung WC, Lee KM, Paik CN, Kim EJ, Kang BK, Oak JH, Jung SH. Efficacy of levofloxacin and rifaximin based quadruple therapy in Helicobacter pylori associated gastroduodenal disease: a double-blind, randomized controlled trial. J Korean Med Sci. 2011;26:785-790. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 190. | Liou JM, Lin JT, Chang CY, Chen MJ, Cheng TY, Lee YC, Chen CC, Sheng WH, Wang HP, Wu MS. Levofloxacin-based and clarithromycin-based triple therapies as first-line and second-line treatments for Helicobacter pylori infection: a randomised comparative trial with crossover design. Gut. 2010;59:572-578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 83] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
P- Reviewer: Castillo A, Sharara A S- Editor: Ji FF
L- Editor: A E- Editor: Li D