Published online Feb 28, 2026. doi: 10.4329/wjr.v18.i2.116973
Revised: December 8, 2025
Accepted: January 13, 2026
Published online: February 28, 2026
Processing time: 92 Days and 0.3 Hours
Hepatic encephalopathy (HE) is the most common serious complication after transjugular intrahepatic portosystemic shunts (TIPS) surgery, the pathogenesis of which is not well understood.
To explore the mechanisms of HE after TIPS from a cerebral hemodynamic per
Patients who meet the inclusion criteria were selected as the case group, and 18 healthy volunteers were assigned as the control group. The differences in am
Compared with the healthy control group, the CBF values of patients with cirrhosis and portal hypertension who were treated with TIPS were reduced in the area centered on the right orbitofrontal gyrus and the left superior temporal gyrus. ALFF scores decreased in the area centered on the left superior temporal gyrus, the left inferior frontal gyrus of the operculum and the right precuneus. Compared with preoperatively, CBF in the TIPS group increased in the area centered on the left fusiform gyrus at 1 month after surgery and the difference in CBF in this area was negatively correlated with the difference in the Child-Pugh liver function score. ALFF values increased in the area centered on the left superior temporal gyrus and the ALFF difference in this area was positively correlated with the portal vein pressure difference. There was no significant difference in CBF 3 months after TIPS in com
TIPS surgery has a certain effect on spontaneous brain activity in patients with portal hypertension and cirrhosis for the increase in plasma ammonia as well as postoperative hemodynamic changes increasing CBF and may be factors causing HE. Resting-state functional magnetic resonance imaging is a sensitive diagnostic tool for HE, especially mild HE.
Core Tip: Using three-dimensional arterial spin labeling and resting-state functional magnetic resonance imaging, we dynamically evaluated cerebral blood flow and spontaneous brain activity in cirrhotic patients undergoing transjugular intrahepatic portosystemic shunts (TIPS). We demonstrated region-specific cerebral blood flow and amplitude of low-frequency fluctuation alterations after TIPS that correlated with changes in Child-Pugh score, portal pressure gradient and neuropsychological performance. These findings indicate that postoperative cerebral hemodynamic changes, together with hyperammonemia, may contribute to hepatic encephalopathy and that resting-state functional magnetic resonance imaging is a sensitive tool for detecting early or mild cognitive impairment after TIPS.
- Citation: Zhuang WL, Xu SW, Fang J, Zhang ZQ, Zhang SA, Wang JL, Zheng JL, Wang XY, Li DL. Role of cerebral blood flow changes in post-transjugular intrahepatic portosystemic shunts hepatic encephalopathy. World J Radiol 2026; 18(2): 116973
- URL: https://www.wjgnet.com/1949-8470/full/v18/i2/116973.htm
- DOI: https://dx.doi.org/10.4329/wjr.v18.i2.116973
Transjugular intrahepatic portosystemic shunts (TIPS) is an effective treatment for complications of severe portal hypertension and cirrhosis, such as esophageal and gastric variceal bleeding and refractory ascites[1]. Despite the continuous development and improvement of TIPS technology, the incidence of overt hepatic encephalopathy (HE) after TIPS surgery in patients with cirrhosis is as high as 25% to 50%[2]. This not only seriously affects the quality of life of patients, but also limits the promotion and application of TIPS surgery.
How to reduce the occurrence and recurrence of HE after TIPS? First of all, we need to fully understand and recognize the mechanism of HE after TIPS surgery in cirrhotic portal hypertension. However, the pathogenesis of HE after TIPS has not been fully elucidated in this regard. The currently widely accepted pathogenesis is that after TIPS surgery, a portion of the portal vein blood passes through the stent directly into the systemic circulation without being detoxified by the liver, leading to increased levels of blood ammonia and other enterogenic toxins, resulting in varying degrees of brain dysfunction. In patients at high risk of postoperative HE on preoperative evaluation, despite the selection of small-diameter stents to reduce portosystemic shunt flow during surgery and active prevention and treatment of postoperative HE, 8% of patients still developed HE that was difficult to control with medical therapy[2]. In addition, in our clinical work, we have found that the incidence of HE is highest within the first 3 months after TIPS surgery. After 3 months, the incidence and frequency of HE is significantly reduced, but at this time, many patients’ blood ammonia levels have not significantly decreased compared to the early postoperative period. Nolte et al[3] also had similar findings. Therefore, we believe that the “ammonia toxicity” theory is not the only factor that causes the high incidence of early HE after TIPS surgery.
TIPS is a procedure that creates a shunt between the hepatic vein and the portal vein system in the liver. The reduction in portal venous pressure and the sudden change in hemodynamics will inevitably disrupt the patient’s original pathophysiological balance to which the body has long adapted. In particular, the increase in venous return and the changes in right atrial and right ventricular pressure and flow will affect cerebral blood flow (CBF). There are few research reports on whether this affects certain brain functions and becomes a trigger or cause of HE. Most related studies focus on the fact that changes in liver hemodynamics after TIPS surgery led to changes in liver function, which is one of the causes of HE after TIPS surgery[4].
Three-dimensional arterial spin labeling (3D-ASL) detects changes in CBF from the perspective of energy metabolism[5]. Zheng et al[6,7] used 3D-ASL technology and found that patients with cirrhosis and such patients after TIPS surgery had different degrees of changes in CBF. Resting-state functional magnetic resonance imaging (rs-fMRI) indirectly reflects the activity state of neurons by detecting changes in the concentration of deoxyhemoglobin in the brain at rest, which can provide us with information about brain activity at rest[8]. Studies have shown that a variety of neurocognitive disorders, including HE, are associated with changes in spontaneous brain activity detected by rs-fMRI[9,10]. Among them, the fluctuation of the blood oxygen level-dependent signal observed by the amplitude of low frequency fluctuation (ALFF) method is related to local neuronal activity[11]. With the development of imaging technology and clinical application of TIPS technology, some reports using rs-fMRI have shown the effects of TIPS on CBF changes and brain function[12,13]. However, there are few reports on the effects of CBF changes on brain function.
In this study, 3D-ASL technology was used to analyze the dynamic changes in CBF after TIPS in patients with liver cirrhosis and portal hypertension, and rs-fMRI was used to analyze the changes in spontaneous activity of the brain after TIPS. The aim is to demonstrate the effects of changes in CBF after TIPS on brain function and its role in the development of HE after TIPS, to elucidate the novel mechanism of HE after TIPS, and to provide a theoretical basis and guidance for clinical management.
This study was approved by the Institutional Review Board of the 900th Hospital of the Joint Logistics Support Force (Approval No. 2023-001). Patients who met the diagnostic criteria for portal hypertension and cirrhosis and were scheduled to undergo TIPS surgery between June 2022 and December 2023 were selected[14].
Include criteria: (1) The patients’ age ranges from 18 years old to 65 years old; (2) They meet the diagnostic criteria for portal hypertension associated with liver cirrhosis; (3) They have suffered from rupture of esophageal and gastric varices bleeding or recurrent and refractory ascites, and the conservative treatment in the department has been ineffective, and they are scheduled to undergo TIPS surgery; and (4) Patients who have fully understood the purpose and significance of the study and are willing to sign the informed consent form.
Exclusion criteria: (1) Age < 18 years or > 65 years; (2) Contraindications to TIPS surgery; (3) Patients with active esopha
The case group underwent MRI one week before surgery, one month, and three months after surgery, respectively, and the control group underwent MRI at the time of enrollment. Blood was drawn during the MRI examination for laboratory indicators such as plasma ammonia, plasma albumin, bilirubin, and prothrombin time. Neuropsychological tests [Number Connection Test Part A (NCT-A), Digit Symbol Substitution Test (DSST)] and liver function assessments were also performed.
Functional MRI was performed in the case group one week before surgery, one month after surgery, three months after surgery, and at the time of enrollment in the control group. Image data were acquired on a 3T magnetic resonance scanner (Discovery MR 750, GE Healthcare, Milwaukee, WI, United States) using an 8-channel phased-array head coil. First, a positioning image scan was performed to determine the best scan field of view (FOV). A high-resolution 3D T1-weighted imaging structural image scan was then performed [repetition time (TR): 8.2 millisecond, echo time (TE): 3.2 millisecond, FOV: 256 mm × 256 mm, image matrix: 256 × 256, inversion angle: 12°, slice thickness: 1.0 mm, slice interval: 0 mm, number of slices: 168 slices]. A single-shot planar gradient echo sequence was then used for the rs-fMRI scan (TR: 2000 milliseconds, TE: 35 milliseconds, FOV: 240 mm × 240 mm, matrix: 64 × 64, inversion angle: 90°, slice thickness: 4.0 mm, slice spacing: 0.8 mm, number of slices: 33 slices). Finally, a 3D-ASL scan was performed, using the pseudo-continuous arterial spin labeling method. The tagging slice was usually placed approximately 2 centimeters below the lower edge of the imaging block (TR: 5029 milliseconds, TE: 14.6 milliseconds, post label delay: 2025 milliseconds, FOV: 240 mm × 240 mm, matrix: 512 × 512, inversion angle: 12°, slice thickness: 4.0 mm, slice spacing: 0 mm, number of slices: 36 slices).
The raw imaging data were post-processed and analyzed using the Data Processing & Analysis for (Resting-State) Brain Imaging (DPABI) software on the Matlab platform (MathWorks Inc., Natick, MA, United States). To eliminate the instability of the initial scanning magnetic field, the first 10 time points were eliminated. To eliminate the influence of time difference and head motion on the functional information, time slice correction and head motion correction were performed. The images were then registered to the high-resolution structural images and resampled to the Montreal Neurological Institute standard space with a voxel size of 3 mm × 3 mm × 3 mm. The functional MRI were spatially smoothed and linear drift removed with a full width at half maximum of 6 mm × 6 mm × 6 mm, and the time series of each voxel was filtered (bandpass 0.01-0.08 Hz). The time series power spectrum of the whole-brain voxel signal was calculated by Fourier transform, and the power spectrum of 0.01-0.08 Hz was squared to obtain the square root of the obtained ALFF value.
The CBF maps (labeled images) of each subject generated by the FuncTool post-processing software were first registered to the high-resolution three-dimensional T1-weighted structural images (unlabeled images), spatially normalized, and resampled to a voxel size of 3 mm × 3 mm × 3 mm. Finally, a Gaussian kernel with a full width at half maximum of 6 mm × 6 mm × 6 mm was used to spatially smooth the CBF maps. The viewer module of the DPABI software is used to display the brain regions with differences. The brain regions with differences are reported using the automated anatomical labeling partitioning method.
SPSS version 25.0 software was used for statistical analysis of demographic and clinical parameters, and MATLAB was used for statistical analysis of postprocessed functional MRI data. To explore the whole-brain spontaneous activity and CBF patterns in patients with portal hypertension and cirrhosis, two independent samples t-tests were performed on the CBF data and ALFF data of the case and control groups before TIPS on a voxel-by-voxel basis. To investigate the effect of TIPS on CBF and brain function, paired t-tests were performed on CBF data and ALFF data before and after TIPS on a voxel-by-voxel manner. The mask uses the gray matter template provided with DPABI, and age, sex, and education level are used as covariates. Gaussian random field correction was applied, and a P < 0.05 after correction was considered to be statistically significant.
The ALFF and CBF values of the brain regions with significant differences were extracted, and the difference between the ALFF, CBF, laboratory indicators, and neuropsychological test scores of the brain regions of each subject with significant differences at the detection time points was calculated using the formula (Δvalue = one month after surgery - preoperative, three months after surgery-preoperative, one month after surgery-three months after surgery), expressed as ΔALFF, ΔCBF, ΔNCT-A, ΔDSST, ΔCP, Δammonia, and Δpressure, respectively. Correlation analysis was then performed using SPSS version 25.0 software. A P < 0.05 was considered to indicate a statistically significant result.
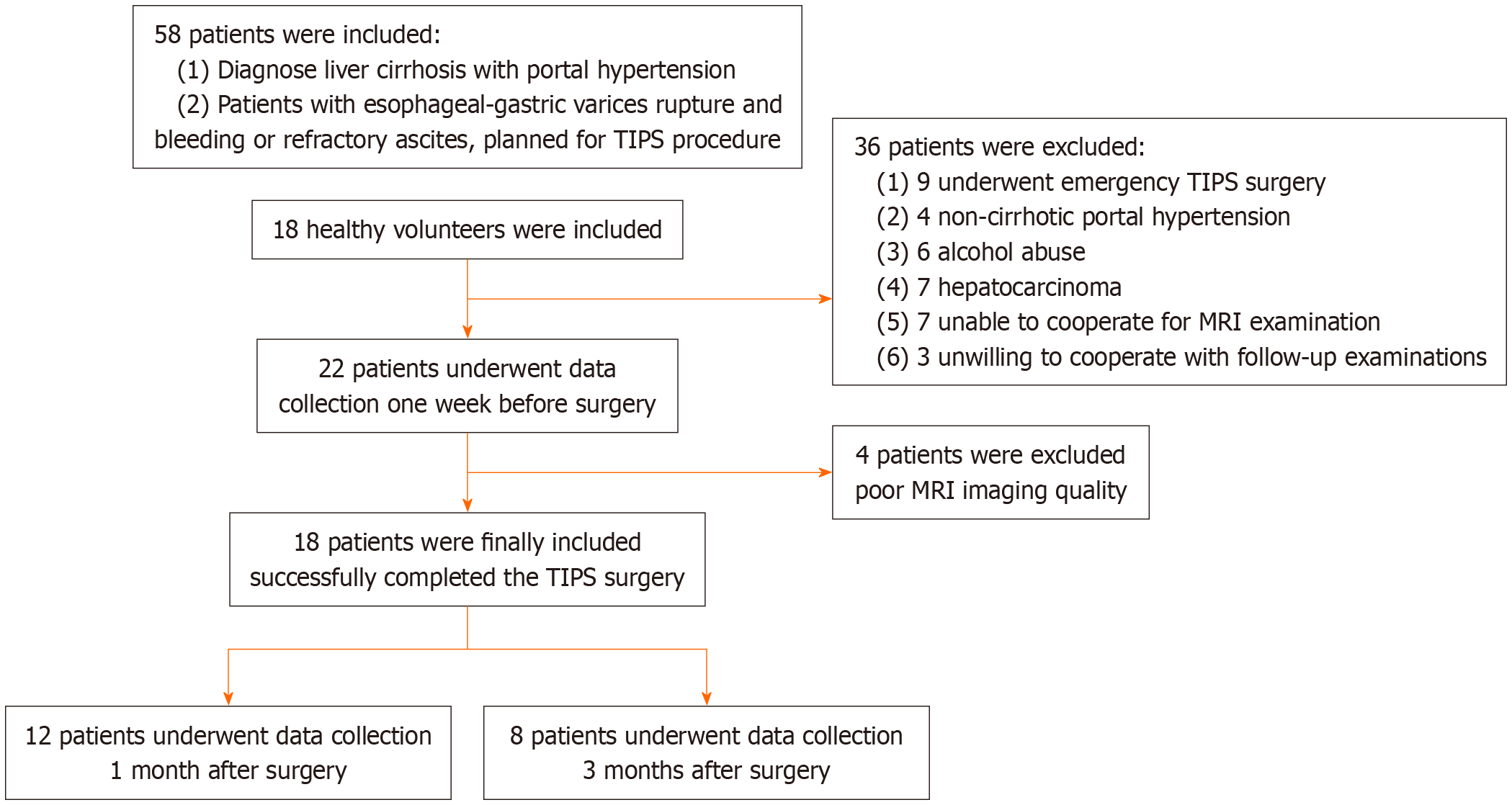
According to the inclusion and exclusion criteria, 18 patients with cirrhosis and portal hypertension who were scheduled to undergo TIPS treatment were selected as the case group, including 15 males and 3 females with a mean age of 57.61 ± 1.68 years. There were 9 cases (50%) of hepatitis B-related cirrhosis and 9 cases (50%) of other causes such as alcoholic liver disease and autoimmune liver disease. The reasons for TIPS in the case group were gastrointestinal bleeding in 16 cases (88.89%) and refractory ascites in 2 cases (11.11%).
All 18 patients successfully completed the TIPS procedure. During the TIPS procedure, portal venous pressure was measured before and after stent implantation and portal venous pressure gradient was calculated. 12 patients completed early postoperative follow-up (one month after surgery) and data collection, and eight patients completed short-term postoperative follow-up (three months after surgery) and data collection. One patient developed grade II HE one week after surgery and two patients developed grade I HE four weeks after surgery. None of the patients experienced recurrence after medical treatment. Eighteen sex-matched and age-matched volunteers with no health problems were recruited as a control group, including 12 males and six females with a mean age of 59.56 ± 1.97 years. The screening procedure is shown in Figure 1.
There were no statistical differences in sex and age between the case and control groups (P > 0.05). There were statistical differences in plasma ammonia, Child-Pugh score, Model for End-Stage Liver Disease score, NCT-A score, and DSST score between the case and control groups before and after TIPS (all P < 0.001). There was no statistical difference in plasma ammonia, Child-Pugh score, Model for End-Stage Liver Disease score, NCT-A score, and DSST score between the case group and the control group before and after TIPS (P > 0.05). The preoperative portal vein pressure was 34.93 ± 2.49 cmH2O, which decreased to 23.87 ± 1.99 cmH2O after surgery, and the difference was statistically significant (t = 7.95, P < 0.001; Table 1).
| Protocols | HC | Before TIPS (n = 18) | After TIPS | P value | |
| 1 month (n = 12) | 3 months (n = 8) | ||||
| Sex (male/female) | 12/6 | 15/3 | 11/1 | 7/1 | > 0.05a |
| Age (years) | 59.56 ± 1.97 | 57.61 ± 1.68 | 56.58 ± 2.01 | 58.25 ± 2.33 | > 0.05b |
| Child-Pugh scores | 5.00 ± 0.00 | 7.61 ± 1.58 | 7.42 ± 1.38 | 8.38 ± 1.69 | < 0.001c |
| Child-Pugh class | 5A/11B/2C | 2A/8B/2C | 2A/3B/3C | ||
| Ammonia level (μmol/L) | 17.20 ± 2.11 | 31.08 ± 10.61 | 37.18 ± 12.78 | 32.96 ± 8.19 | < 0.001c |
| NCT-A (seconds) | 35.94 ± 5.65 | 51.22 ± 4.34 | 53.33 ± 5.28 | 58.75 ± 7.93 | < 0.001c |
| DSST (score) | 53.94 ± 1.72 | 33.89 ± 8.76 | 32.58 ± 7.19 | 31.88 ± 5.33 | < 0.001c |
| Portal pressure (cmH2O) | 34.93 ± 2.49 | 23.87 ± 1.99 | < 0.001c | ||
| HE or MHE | 3 HE | ||||
| Etiology | 9 hepatitis cirrhosis/2 alcoholic cirrhosis/7 other causes including autoimmune liver disease | ||||
| TIPS indication | 16 bleeding/2 refractory ascites | ||||
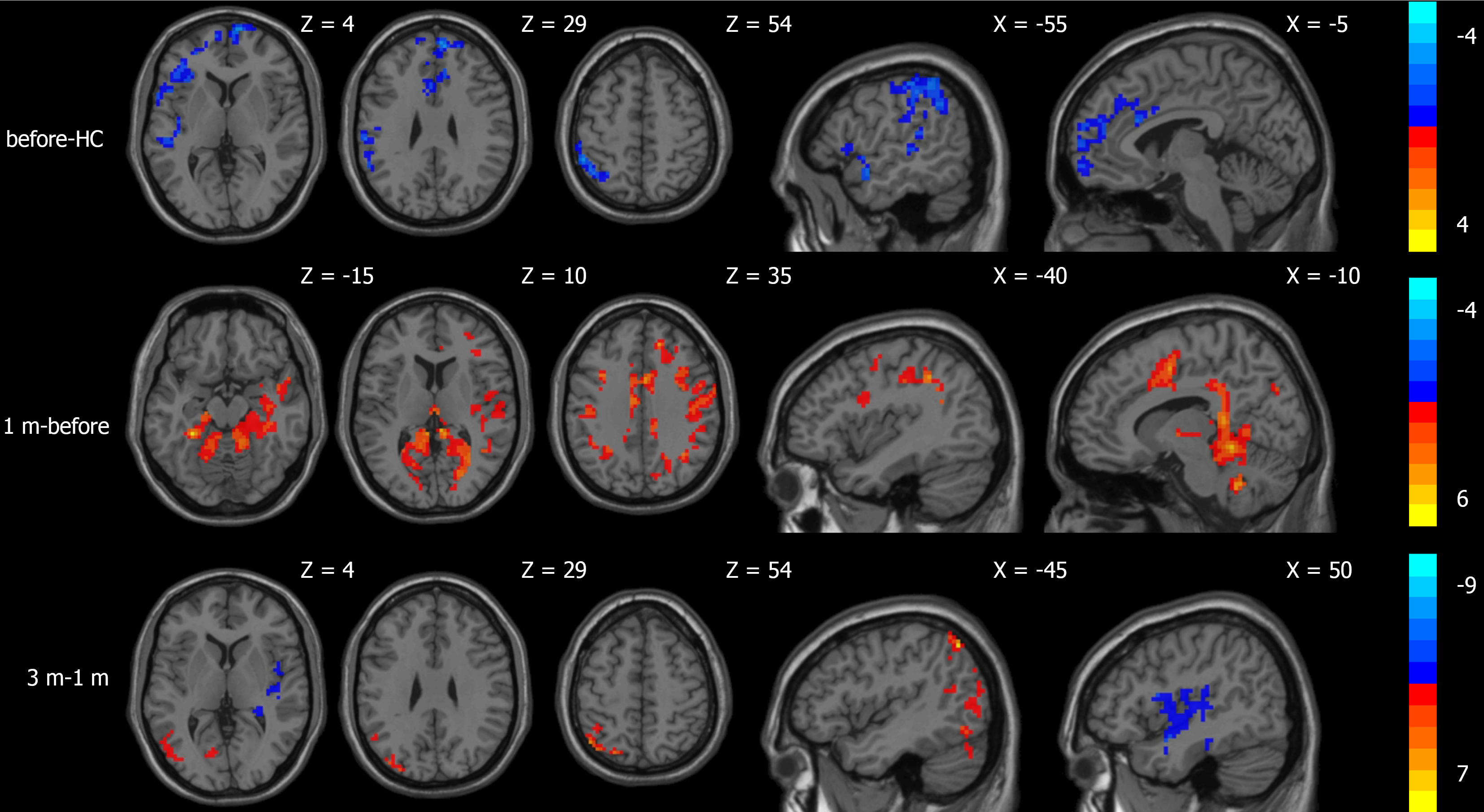
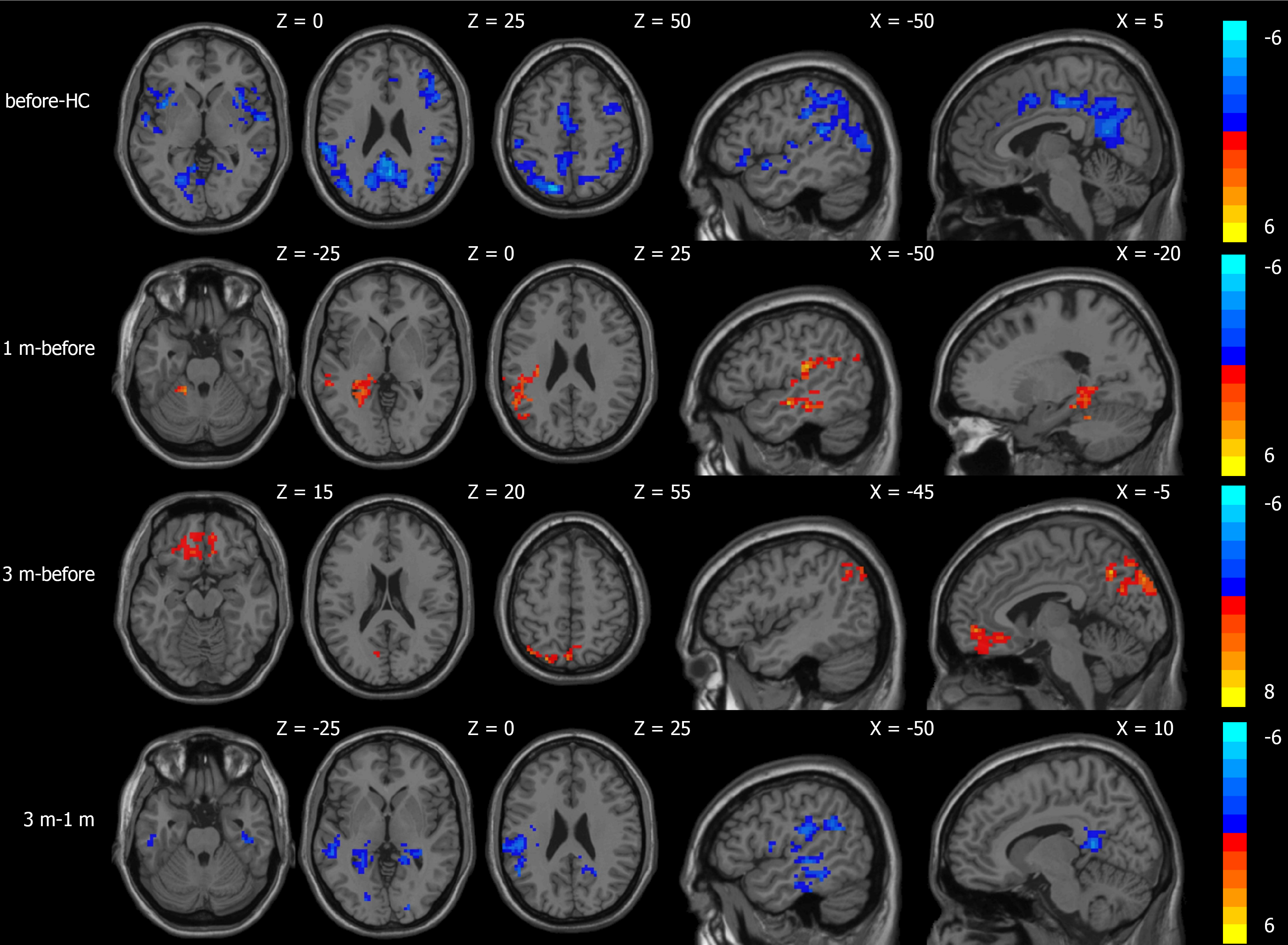
Compared to the normal control group, the CBF values of patients with portal hypertension and cirrhosis decreased in the area centered on the right orbitofrontal gyrus and the left superior temporal gyrus. The ALFF values decreased in the area centered on the left superior temporal gyrus, the left inferior frontal gyrus of the operculum, and the right precuneus. Compared to before TIPS, the CBF values increased in the area centered on the left fusiform gyrus 1 month after TIPS. The ALFF values increased in the area centered on the left superior temporal gyrus; there were no areas with significant CBF differences 3 months after TIPS. The ALFF values increased in the area centered on the left orbitofrontal gyrus and the left precuneus 3 months after TIPS. In comparison to 1 month after TIPS, CBF values decreased in the area centered on the right fusiform gyrus and increased in the area centered on the left angular gyrus 3 months after TIPS. ALFF values decreased in the area centered on the right insula and the cerebellum (Table 2; Figures 2 and 3).
| Group | Brain regions | MNI coordinates (mm) | Volume (mm3) | Peak (t-value) | ||
| X | Y | Z | ||||
| Comparison results of CBF differences | ||||||
| Before-HC | R. orbitofrontal superior gyrus | 9 | 66 | -12 | 1056 | -4.62 |
| L. superior temporal gyrus | -60 | -27 | 18 | 537 | -3.96 | |
| 1 m-before | L. fusiform gyrus | -30 | -39 | -18 | 5280 | +6.56 |
| 3 m-1 m | R. fusiform gyrus | 39 | -33 | -21 | 512 | -4.39 |
| L. angular gyrus | -45 | -63 | 51 | 587 | +7.93 | |
| Comparison results of ALFF differences | ||||||
| Before-HC | L. superior temporal gyrus | -39 | -33 | 12 | 1502 | -6.68 |
| L. opercular part of inferior frontal gyrus | -48 | 9 | 21 | 213 | -6.36 | |
| R. precuneus | 15 | -60 | 21 | 3765 | -6.04 | |
| 1 m-before | L. superior temporal gyrus | -48 | -30 | 21 | 687 | +4.68 |
| 3 m-before | L. orbitofrontal superior gyrus | -6 | 51 | -6 | 298 | +5.95 |
| L. precuneus | -3 | -75 | 51 | 458 | +7.69 | |
| 3 m-1 m | R. insula | 36 | -27 | 18 | 513 | -6.43 |
| Cerebelum_Crus1_L | -24 | -72 | -33 | 851 | -6.64 | |
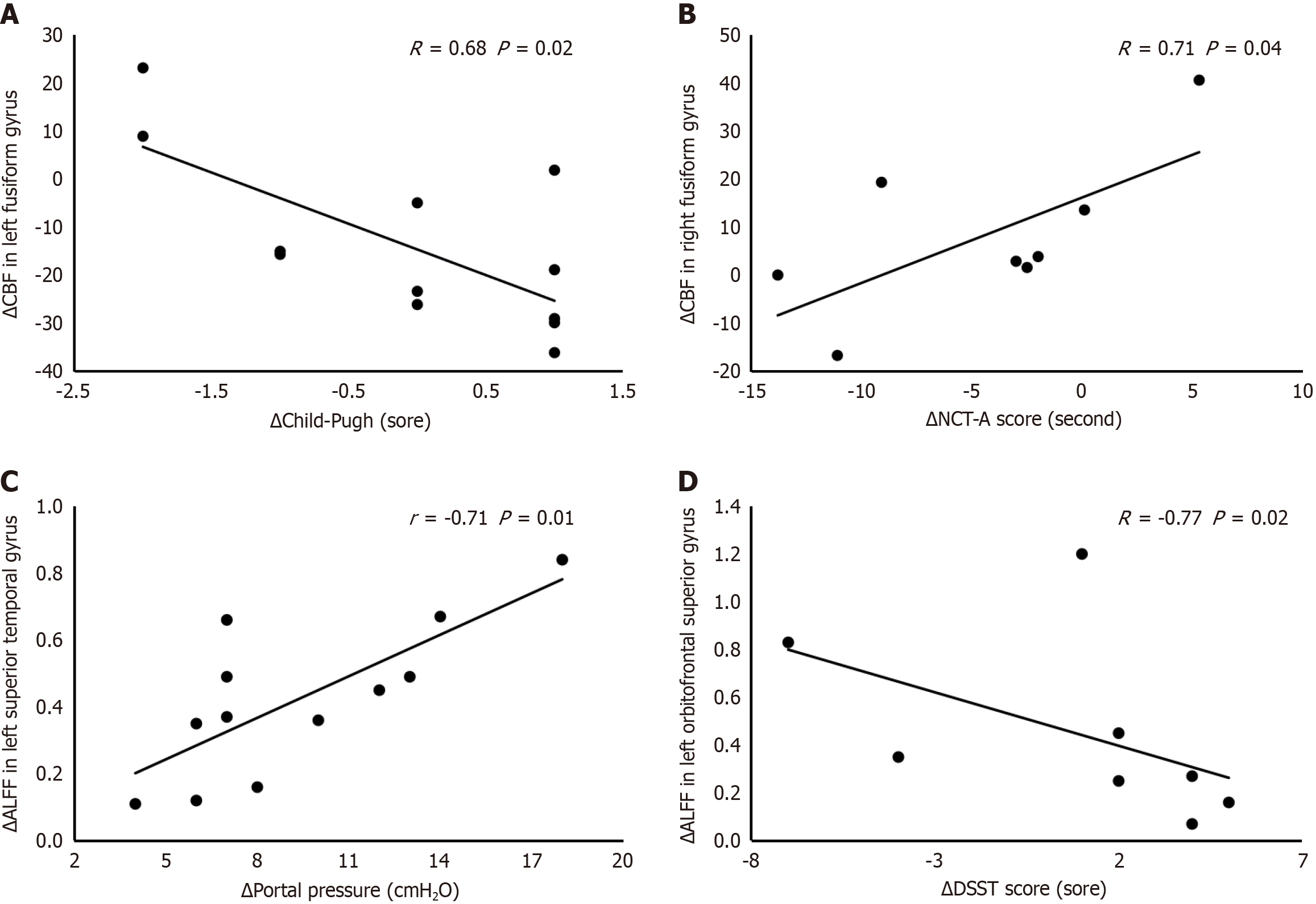
The ΔCBF of the left fusiform gyrus, which had a significant difference in CBF values 1 month after TIPS surgery compared to before surgery, was negatively correlated with the ΔChild-Pugh score (R = 0.68, P = 0.02). The ΔCBF of the right fusiform gyrus, which showed a significant difference in CBF values 3 months after TIPS surgery compared to before surgery, was positively correlated with the ΔNCT-A score (R = 0.71, P = 0.04). The ΔALFF of the left superior temporal gyrus, which presented with significant differences in ALFF values 1 month after TIPS surgery compared to before surgery, was positively correlated with the ΔPVP (r = -0.71, P = 0.01). The ΔALFF of the left orbitofrontal superior gyrus, which exhibited significant differences in ALFF values 3 months after TIPS surgery compared to before surgery, was negatively correlated with the ΔDSST score (R = -0.77, P = 0.02; Figure 4).
TIPS surgery had a dynamic effect on CBF in patients with portal hypertension and cirrhosis. CBF increased in the early postoperative period and gradually returned to the preoperative state 3 months after surgery. We believe that the change in cerebral hemodynamics may be one of the main reasons for the susceptibility to HE in the early postoperative period. The trend of CBF changes after TIPS was similar to the 3D-ASL results of Zheng et al[7] and consistent with the trend of abnormal EEG changes reported by Nolte et al[3].
The increase in CBF in the early period after TIPS surgery may be due to the fact that blood diverted by the stent after TIPS surgery entered the right atrium directly through the inferior vena cava, which reduces portal vein pressure and increases the velocity of blood flow and blood volume of the right atrium and right ventricle[15,16]. The preload of the right heart was increased, which can affect the return velocity and flow of blood from the superior vena cava, causing changes in CBF. As CBF increases, a large quantity of neurotoxins enters the brain tissue, and the accumulation of glutamine caused by blood ammonia overload induces cytotoxic cerebral edema[17]. Other vasodilator factors, such as the accumulation of extracellular potassium and an increase of nitric oxide generation, can also lead to excessive cerebral perfusion[7]. To reduce damage, cerebral hemodynamics will make adaptive adjustments, and there is often a degree of adaptability to neurotoxins[3]. Therefore, the blood ammonia level did not decrease significantly 3 months after TIPS surgery compared with the preoperative and early postoperative periods, but the onset of HE was significantly reduced and alleviated.
It was also found that the ΔCBF of the left fusiform gyrus was correlated with the ΔChild-Pugh score 1 month after TIPS surgery, suggesting that liver function deterioration or extrahepatic complications are likely to lead to CBF changes. In addition to CBF changes, the systemic hemodynamic changes caused by TIPS will also lead to a short-term decrease in hepatic blood flow, resulting in varying degrees of liver function impairment[18]. The ΔCBF difference of the right fusiform gyrus was correlated with the ΔNCT-A score 3 months after TIPS surgery, suggesting that changes in neurocognitive functions may be related to CBF changes after TIPS surgery. The fusiform gyrus is closely related in structure and function to several brain regions and is critical for cognitive and emotional processing[19].
The rs-fMRI results of the present study showed that the spontaneous activity of the brain increased in a compensatory manner in the early stage after TIPS surgery and then gradually recovered. Previous studies have shown that a decrease in ALFF indicates a decrease in spontaneous activity in the brain region associated with damage to the functional area; an increase in ALFF means an increase in spontaneous activity in the brain region, which is related to compensatory relocalization of the brain region functions. Compensatory brain region relocalization means that after brain damage, other undamaged brain regions take over or enhance their functions to compensate for the lack of functions of the damaged brain region[20]. In our study, the spontaneous activity of some brain regions increased after TIPS surgery, which may be a functional compensation caused by postoperative CBF changes. The changes in spontaneous activity recovered with the progression of disease, and the early changes in CBF gradually decreased. The above results are entirely consistent with the research conclusions of Qi et al[12] and Ni et al[21], both of which reflected that the resting activity pattern of the brain in patients with cirrhosis after TIPS surgery changed with the progression of the disease.
In theory, portal venous pressure should be reduced as much as possible during surgery to reduce the harm of portal hypertension to patients. However, during actual surgery, physicians should try to balance the reduction of portal vein pressure with the maintenance of hepatic blood flow to avoid deterioration of liver functions or increased risk of postoperative HE due to excessive shunting[22]. Our study found that the ΔALFF of the left superior temporal gyrus was correlated with the Δportal vein pressure 1 month after TIPS surgery, indicating that the hemodynamic changes after TIPS surgery and the rapid decrease in portal vein pressure may be one of the factors causing abnormal brain functions and HE after TIPS surgery. The ΔALFF of the left orbitofrontal gyrus was correlated with the ΔDSST score 3 months after TIPS surgery, indicating that the neurocognitive dysfunction 3 months after TIPS surgery is closely related to the changes in spontaneous activity of the brain region. The orbitofrontal lobe can integrate external stimuli with internal states and regulate high-level cognitive functions such as reward prediction, emotion regulation and working memory[23].
Contrary to the conclusions of previous research, which mainly used positron emission tomography and single-photon emission computed tomography[24], the present study showed that patients with cirrhotic portal hypertension had decreased CBF in the cerebral cortex rather than increased CBF in the subcortical area before surgery. Apart from the differences in measurement methods and subjects, Jalan et al[25] believed that cirrhosis and HE were multifocal cortical dysfunctions, and due to brain damage, CBF is compensatory redistributed from the cerebral cortex to the subcortical area. Combined with clinical analysis, we believe that acute esophageal variceal bleeding may reduce blood pressure and cerebral blood perfusion, while large amounts of fluid and blood transfusion may cause blood dilution, which will affect the oxygen-carrying capacity of blood and further reduce the blood oxygen level of brain tissue. Some studies have reported that the cerebral blood oxygen level is related to CBF[26]. In addition, most of these patients had certain cerebrovascular changes before surgery, which lead to a widespread decrease in CBF in the cerebral cortex[27].
This study is limited by its small sample size, mainly due to time constraints and the fact that some participants were unable to cooperate with follow-up examinations. This may limit the generalization of our results. However, this study is the first to evaluate the effect of CBF changes after TIPS surgery on brain function, the results have certain guiding significance. Future studies will expand the sample size to further explore this issue.
This study is the first to evaluate the effect of CBF changes after TIPS surgery on brain function. The results showed that in addition to increased blood ammonia levels, postoperative hemodynamic changes, especially changes in CBF, may be an important cause of HE after TIPS. Rs-fMRI may be a sensitive diagnostic method for HE, especially mild HE.
We sincerely thank every member of our team for their invaluable contributions, dedication, and support throughout the conduct of this study and the preparation of the manuscript.
| 1. | Boike JR, Thornburg BG, Asrani SK, Fallon MB, Fortune BE, Izzy MJ, Verna EC, Abraldes JG, Allegretti AS, Bajaj JS, Biggins SW, Darcy MD, Farr MA, Farsad K, Garcia-Tsao G, Hall SA, Jadlowiec CC, Krowka MJ, Laberge J, Lee EW, Mulligan DC, Nadim MK, Northup PG, Salem R, Shatzel JJ, Shaw CJ, Simonetto DA, Susman J, Kolli KP, VanWagner LB; Advancing Liver Therapeutic Approaches (ALTA) Consortium. North American Practice-Based Recommendations for Transjugular Intrahepatic Portosystemic Shunts in Portal Hypertension. Clin Gastroenterol Hepatol. 2022;20:1636-1662.e36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 196] [Cited by in RCA: 179] [Article Influence: 44.8] [Reference Citation Analysis (2)] |
| 2. | Wang LJ, Yao X, Qi Q, Qin JP. Prevention and treatment of hepatic encephalopathy during the perioperative period of transjugular intrahepatic portosystemic shunt. World J Gastrointest Surg. 2023;15:1564-1573. [PubMed] [DOI] [Full Text] |
| 3. | Nolte W, Wiltfang J, Schindler C, Münke H, Unterberg K, Zumhasch U, Figulla HR, Werner G, Hartmann H, Ramadori G. Portosystemic hepatic encephalopathy after transjugular intrahepatic portosystemic shunt in patients with cirrhosis: clinical, laboratory, psychometric, and electroencephalographic investigations. Hepatology. 1998;28:1215-1225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 117] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 4. | Yang M, Qin F, Zhou Y, Yao Y, Lu Z, Chen W. Evaluation and clinical significance of contrast-enhanced ultrasound on changes in liver blood flow perfusion after TIPS surgery. Medicine (Baltimore). 2024;103:e37899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 5. | Wang K, Guo H, Tian X, Miao Y, Han P, Jin F. Efficacy of three-dimensional arterial spin labeling and how it compares against that of contrast enhanced magnetic resonance imaging in preoperative grading of brain gliomas. Environ Toxicol. 2023;38:1723-1731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 6. | Zheng G, Zhang LJ, Zhong J, Wang Z, Qi R, Shi D, Lu GM. Cerebral blood flow measured by arterial-spin labeling MRI: a useful biomarker for characterization of minimal hepatic encephalopathy in patients with cirrhosis. Eur J Radiol. 2013;82:1981-1988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 27] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 7. | Zheng G, Zhang LJ, Cao Y, Pan Z, Qi RF, Ni L, Shi D, Fan X, Lu GM. Transjugular intrahepatic portosystemic shunt induced short- and long-term cerebral blood flow variations in cirrhotic patients: an arterial spin labeling MRI study. Metab Brain Dis. 2013;28:463-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 8. | Chen K, Azeez A, Chen DY, Biswal BB. Resting-State Functional Connectivity: Signal Origins and Analytic Methods. Neuroimaging Clin N Am. 2020;30:15-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 9. | Hacker CD, Roland JL, Kim AH, Shimony JS, Leuthardt EC. Resting-state network mapping in neurosurgical practice: a review. Neurosurg Focus. 2019;47:E15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 33] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 10. | Qin B, Liang S, Tang S, Liang H, Zhang Y, Liang Z. Altered Spontaneous Brain Activity in Cirrhotic Patients with Minimal Hepatic Encephalopathy: A Meta-Analysis of Resting-State Functional Imaging. Brain Sci. 2023;13:960. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 11. | Yang J, Gohel S, Vachha B. Current methods and new directions in resting state fMRI. Clin Imaging. 2020;65:47-53. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 77] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 12. | Qi R, Zhang LJ, Zhong J, Wu S, Zhang Z, Zhong Y, Ni L, Zheng G, Jiao Q, Wu X, Fan X, Liu Y, Lu G. Dynamic changes of intrinsic brain activity in cirrhotic patients after transjugular intrahepatic portosystemic shunt: a resting-state FMRI study. PLoS One. 2012;7:e46681. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 13. | Liu C, Wang HB, Yu YQ, Wang MQ, Zhang GB, Xu LY, Wu JM. [Resting-state functional magnetic resonance study of brain function changes after TIPS operation in patients with liver cirrhosis]. Zhonghua Yi Xue Za Zhi. 2016;96:3787-3792. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 14. | de Franchis R, Bosch J, Garcia-Tsao G, Reiberger T, Ripoll C; Baveno VII Faculty. Baveno VII - Renewing consensus in portal hypertension. J Hepatol. 2022;76:959-974. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2244] [Cited by in RCA: 2054] [Article Influence: 513.5] [Reference Citation Analysis (15)] |
| 15. | Manguso G, Vignone A, Merli M, Miotti C, Caputo A, Vizza CD, Badagliacca R. Hemodynamic Evaluation of the Right Heart-Pulmonary Circulation Unit in Patients Candidate to Transjugular Intrahepatic Portosystemic Shunt. J Clin Med. 2022;11:461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 16. | Busk TM, Bendtsen F, Henriksen JH, Fuglsang S, Clemmesen JO, Larsen FS, Møller S. Effects of transjugular intrahepatic portosystemic shunt (TIPS) on blood volume distribution in patients with cirrhosis. Dig Liver Dis. 2017;49:1353-1359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 17. | Zielińska M, Albrecht J, Popek M. Dysregulation of Astrocytic Glutamine Transport in Acute Hyperammonemic Brain Edema. Front Neurosci. 2022;16:874750. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 18. | Luo SH, Zhou MM, Cai MJ, Han SL, Zhang XQ, Chu JG. Reduction of portosystemic gradient during transjugular intrahepatic portosystemic shunt achieves good outcome and reduces complications. World J Gastroenterol. 2023;29:2336-2348. [PubMed] [DOI] [Full Text] |
| 19. | Edmiston EK, Chase HW, Jones N, Nhan TJ, Phillips ML, Fournier JC. Differential role of fusiform gyrus coupling in depressive and anxiety symptoms during emotion perception. Soc Cogn Affect Neurosci. 2024;19:nsae009. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 20. | Li B, Li WG, Guo Y, Wang Y, Xu LY, Yang Y, Xu SG, Tan ZL, Mei YR, Wang KY. Integrating fractional amplitude of low-frequency fluctuation and functional connectivity to investigate the mechanism and prognosis of severe traumatic brain injury. Front Neurol. 2023;14:1266167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 21. | Ni L, Qi R, Zhang LJ, Zhong J, Zheng G, Wu X, Fan X, Lu GM. Brain regional homogeneity changes following transjugular intrahepatic portosystemic shunt in cirrhotic patients support cerebral adaptability theory--a resting-state functional MRI study. Eur J Radiol. 2014;83:578-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 22. | Trivedi S, Lam K, Ganesh A, Hasnain Y, Hassan W, Herren J, Gaba RC. Hepatic Encephalopathy after Transjugular Intrahepatic Portosystemic Shunt Creation. Semin Intervent Radiol. 2023;40:9-14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 23. | Rolls ET, Cheng W, Feng J. The orbitofrontal cortex: reward, emotion and depression. Brain Commun. 2020;2:fcaa196. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 399] [Cited by in RCA: 330] [Article Influence: 55.0] [Reference Citation Analysis (0)] |
| 24. | Zhang W, Ning N, Li X, Li M, Duan X, Guo Y, Dang Y, Li Y, Gao J, Ye J, Yang J. Impaired brain glucose metabolism in cirrhosis without overt hepatic encephalopathy: a retrospective 18F-FDG PET/CT study. Neuroreport. 2019;30:776-782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 25. | Jalan R, Olde Damink SW, Lui HF, Glabus M, Deutz NE, Hayes PC, Ebmeier K. Oral amino acid load mimicking hemoglobin results in reduced regional cerebral perfusion and deterioration in memory tests in patients with cirrhosis of the liver. Metab Brain Dis. 2003;18:37-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 25] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 26. | Liu J, Li S, Qian L, Xu X, Zhang Y, Cheng J, Zhang W. Effects of acute mild hypoxia on cerebral blood flow in pilots. Neurol Sci. 2021;42:673-680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 27. | Häussinger D, Dhiman RK, Felipo V, Görg B, Jalan R, Kircheis G, Merli M, Montagnese S, Romero-Gomez M, Schnitzler A, Taylor-Robinson SD, Vilstrup H. Hepatic encephalopathy. Nat Rev Dis Primers. 2022;8:43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 146] [Article Influence: 36.5] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/