Published online Feb 28, 2026. doi: 10.4329/wjr.v18.i2.115249
Revised: December 24, 2025
Accepted: January 16, 2026
Published online: February 28, 2026
Processing time: 136 Days and 0.4 Hours
Metallic artifacts from coaxial needles can severely interfere with the precision of cone-beam computed tomography (CBCT)-guided percutaneous transthoracic needle biopsy (PTNB), particularly in assessing vital anatomical structures around small lesions.
To evaluate the clinical application of a streak metal artifact reduction technique (SMART) in CBCT-PTNB procedures.
We retrospectively analyzed data from 68 patients (73 CBCT scans) undergoing CBCT-guided PTNB between March 2023 and December 2024. Image quality was compared among original reconstructed CBCT images, those iteratively recon
SMART-Full reconstruction significantly reduced radial, streak, and dark stripe artifacts (P < 0.001) compared to original CBCT, with superior performance in puncture needle diameter measurement and ROI minimum and average density indicators (P < 0.001). The incidence of dark streak artifacts decreased from 71 cases in original CBCT to 26 cases in SMART-Full. Additionally, SMART-Full was more effective than SMART-Small in artifact elimination (P < 0.001).
SMART technology effectively reduces metal artifacts, enabling clearer visualization of hidden anatomical structures. Through quantitative analysis, this study confirms the clinical value of SMART in CBCT-guided PTNB, providing a tech
Core Tip: This retrospective study evaluates the application of streak metal artifact reduction technique (SMART) in cone-beam computed tomography (CBCT)-guided percutaneous transthoracic needle biopsy. SMART iterative reconstruction, particularly in full volume-of-interest mode, significantly reduces radial, streak, and dark stripe arti
- Citation: Wang ZL, Wu G. Application of streak metal artifact reduction technique in cone-beam computed tomography guided percutaneous transthoracic needle biopsy. World J Radiol 2026; 18(2): 115249
- URL: https://www.wjgnet.com/1949-8470/full/v18/i2/115249.htm
- DOI: https://dx.doi.org/10.4329/wjr.v18.i2.115249
Percutaneous transthoracic needle biopsy (PTNB) plays a crucial role in the diagnosis and management of lung nodules, particularly for early detection and characterization of suspicious lesions[1,2]. Cone-beam computed tomography (CBCT) has emerged as a favored imaging modality for guiding PTNB procedures due to its inherent advan
Despite the advancements offered by CBCT, a significant challenge in its application for PTNB is the presence of metal artifacts, predominantly originating from the coaxial biopsy needles. These artifacts manifest as radial streaks, dark bands, and localized signal thickening, which can severely obscure the visualization of lung nodules, especially smaller ones (e.g., those less than 1 cm in diameter)[8,9]. Such image degra
To mitigate metal artifacts in CT and CBCT, various metal artifact reduction (MAR) techniques have been developed, broadly categorized into traditional acquisition modifications, projection-based methods, iterative reconstructions, dual-energy approaches, and emerging deep learning algorithms[10-15]. Traditional methods adjust parameters like higher tube voltage or current to reduce beam hardening and photon starvation, offering simplicity but increasing dose and providing incomplete suppression for severe artifacts[10]. Projection-based techniques, such as linear sinogram interpolation or advanced normalized MAR (NMAR), replace corrupted data with estimates from adjacent projections, preserving edges but potentially introducing secondary streaks[10,14]. Direct sinogram correction, often with metal path-length weighting, directly adjusts attenuation values, proving effective in dental CBCT for implants with faster computation[11]. Iterative methods model noise and artifacts for refined images, excelling in interventional settings like CBCT but requiring more processing time[10]. Dual-energy CT generates virtual monoenergetic images to minimize hardening, versatile for mild artifacts yet hardware-dependent[15]. Deep learning approaches use neural networks to predict artifact-free images, showing promise in complex cases but needing large datasets[10,15]. Despite these options, their efficacy in real-time lung CBCT remains variable, with limited data for PTNB. In this study, we selected the streak MAR technique (SMART), a proprietary iterative projection-based algorithm from Siemens Healthineers, due to its seamless integration with the C-arm system, demonstrated effectiveness in reducing streaks in interventional radiology (e.g., embolization)[13,16], and the need to address its underexplored role in CBCT-guided PTNB for pulmonary lesions. While MAR algorithms have demonstrated considerable success and widespread application in other medical imaging fields, such as orthopedics and neuroradiology, their specific role and efficacy in lung interventions, particularly in the context of CBCT-guided PTNB, remain less extensively investigated[11,17].
This study aims to evaluate the impact of applying CBCT with the SMART algorithm in percutaneous lung nodule biopsy procedures. We hypothesize that the integration of SMART will lead to significant improvements in image quality, thereby enhancing the visualization of lung nodules and surrounding anatomy.
This retrospective study was conducted in compliance with the principles of the Declaration of Helsinki. The study protocol was approved by the Institutional Review Board of our hospital (No. KY-2025-293-01), which waived the requirement for individual patient consent due to the retrospective nature of the analysis.
Data were retrospectively collected from 68 patients (73 CBCT scans) who underwent CBCT-guided PTNB at our institution between March 2023 and December 2024. The patient cohort comprised 40 males and 28 females, with a mean age of 62 ± 13 years (range: 17-81 years). The inclusion and exclusion criteria for patient selection were as follows.
Inclusion criteria: (1) Patients undergoing CBCT-guided PTNB for diagnosis of a pulmonary nodule; (2) Presence of a target lung lesion identifiable on CBCT; (3) Use of a metallic coaxial puncture needle during the procedure, followed by a CBCT scan; and (4) Availability of complete pre-procedural and intra-procedural imaging data for review.
Exclusion criteria: (1) Severe cardiopulmonary dysfunction that would contraindicate the procedure; (2) Uncorrectable coagulopathy; (3) Inability of the patient to cooperate, such as being unable to hold their breath or maintain the required position; (4) Absence of a safe percutaneous access route to the target lesion; and (5) Loss of original DICOM scanning data or data that was corrupted.
All PTNB procedures were performed by an experienced interventional radiologist in a dedicated commercial angiography suite (Artis Q ceiling, Siemens Healthineers, Forchheim, Germany). Prior to the procedure, patients underwent a comprehensive clinical evaluation, including review of prior imaging (e.g., diagnostic CT scans) to determine the optimal approach for biopsy. Patients were placed in the supine, prone, or lateral decubitus position to provide the safest and most direct needle path to the target lesion, as determined from prior diagnostic CT scans and were instructed to hold still during the whole procedure. Then CBCT scanning was performed with the following parameters: Acquisition time 6 seconds, tube voltage 90 kV, flat-panel detector size 30 cm × 40 cm, field of view 48 cm, rotational angle 200°, 0.5°/frame, 397 frames in total, and dose 0.36 mGy/frame. After the rotational scan, the raw data were automatically transferred to a dedicated workstation (syngo workstation, Siemens Healthineers, Forchheim, Germany) and reconstructed there. Original reconstructions used a modified Feldkamp-Davis-Kress algorithm standard for Siemens DynaCT. The CBCT images were then used for precise three-dimensional path planning on a syngo needle guidance software (Siemens Healthineers, Forchheim, Germany), allowing fine adjustments to the needle trajectory to accurately target the lesion while avoiding critical structures such as major blood vessels, bronchi, and fissures. Then the planned path was set to the C-arm to guide puncture with a laser cross on the body surface to indicate the puncture site and angulation. The puncture site was then sterilized, and local anesthesia was administered using 1% lidocaine along the planned needle trajectory from the skin to the pleura. A coaxial biopsy needle system was advanced towards the target lung nodule under real-time fluoroscopic guidance with the planned needle path overlayed. Subsequent CBCT scans were performed intermittently as the needle was advanced to monitor its position relative to the target and surrounding anatomy. Once the needle tip was confirmed to be within the target lesion, tissue samples were obtained using an automated biopsy gun. After adequate samples were acquired, the biopsy needle was carefully withdrawn. Following needle removal, a final CBCT scan was performed to assess for immediate complications, such as pneumothorax or hemorrhage. Patients were then monitored in the recovery area according to standard institutional protocols.
For metal artifacts reduction, an iterative reconstruction was applied following the initial reconstruction. During the second iterative reconstruction, the SMART reconstruction type was applied on the syngo workstation during routine clinical workflow. SMART (syngo DynaCT SMART, Siemens Healthineers, Forchheim, Germany) is a proprietary iterative projection-based MAR algorithm designed to mitigate streak and radial artifacts from metallic objects[9,18]. The process involves: (1) Initial reconstruction of CBCT images; (2) Automated segmentation of metallic structures (e.g., the coaxial needle); (3) Forward projection to identify affected sinograms; (4) Iterative correction of inconsistencies caused by beam hardening and photon starvation through data interpolation and normalization; and (5) Final reconstruction. The kernel parameter for SMART was set to HU, and reconstruction was performed in either Full or Small modes based on the volume of interest (VOI) size. In FULL mode, the entire VOI was processed, while in SMALL mode, the segmented region primarily included the metal object (coaxial needle).
Artifact evaluation was performed independently by two experienced interventional radiologists (each with more than 10 years of experience in thoracic interventions and CBCT-guided procedures), who were blinded to the reconstruction method [original CBCT, SMART with a full VOI (SMART-Full), or with a small VOI containing only the coaxial needle (SMART-Small)]. Assessments were conducted on a dedicated workstation using anonymized images presented in random order. Discrepancies were resolved by consensus discussion. Types of metal artifacts (radial, streak, thickening, dark streak) were classified based on their visual appearance, and quantitative metrics [puncture needle diameter, region of interest (ROI) densities] were measured using standardized tools in the syngo software. We systematically compared the types of metal artifacts (radial, streak, thickening, dark streak), their cross-sectional appearance, the puncture needle diameter (measured in mm), and the CT value (in HU) and standard deviation of the tissue immediately ahead of the needle tip across the three reconstruction methods to evaluate the effectiveness of SMART technology in mitigating metal artifacts during lung nodule puncture.
Statistical analysis was performed using SPSS version 16.0 (IBM Corp., Armonk, NY, United States). The prevalence of artifact types was compared using paired χ2 tests, while quantitative metrics (needle diameter, ROI densities) were analyzed with paired sample t-tests. The significance level was set at α = 0.05. No adjustment for multiple comparisons was applied, as this is an exploratory imaging study focused on hypothesis generation rather than confirmatory testing; however, this may increase the risk of false positives.
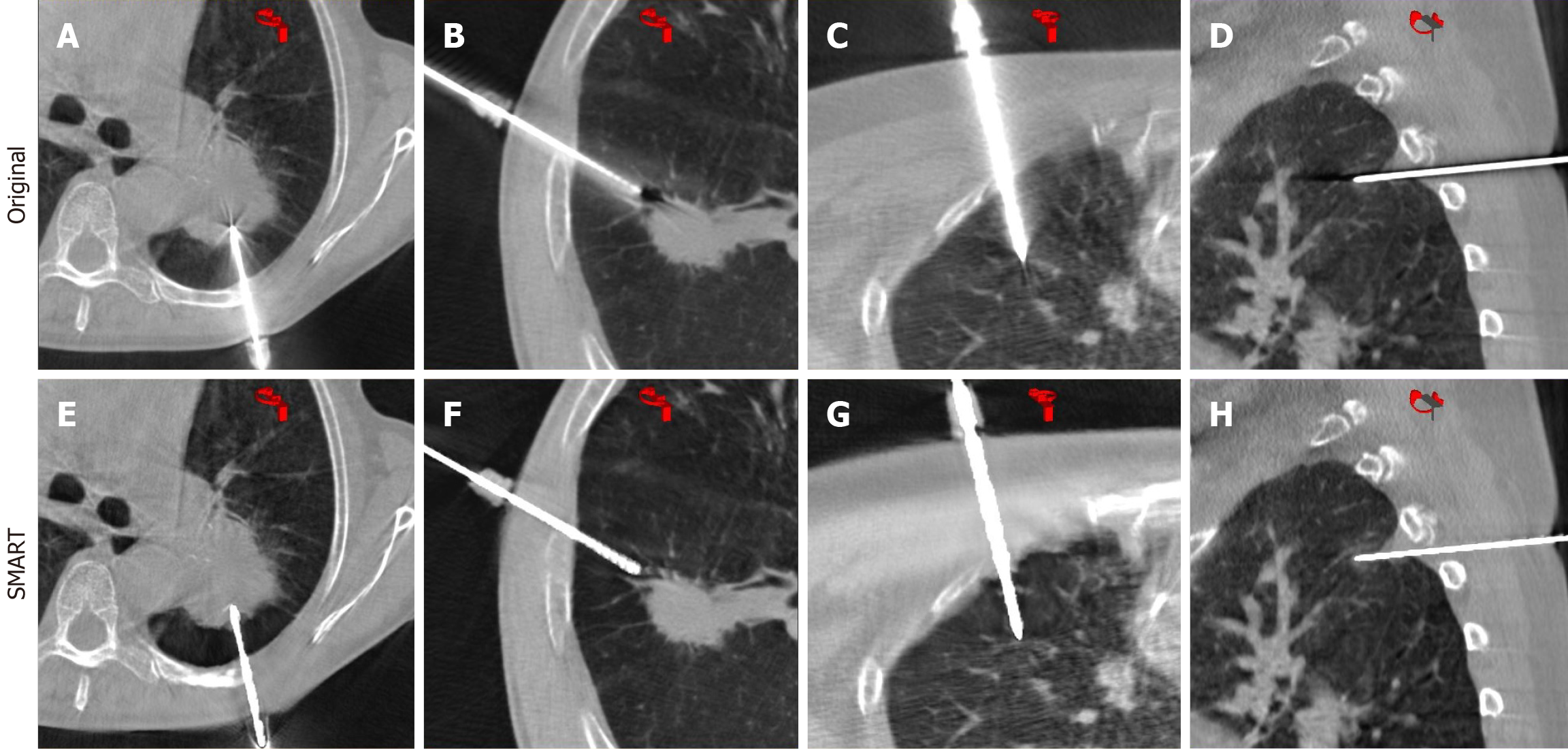
In this study, the application of SMART reconstruction significantly improved image quality by reducing various types of metal artifacts. Figure 1 visually demonstrates the substantial impact of SMART reconstruction on mitigating metal artifacts. The original CBCT images (Figure 1A-D) clearly exhibit four primary types of artifacts: Radial, streak, thickening, and dark streak artifacts, which are commonly caused by the metallic coaxial needle. Following the application of SMART iterative reconstruction, these artifacts were visibly suppressed across all categories (Figure 1E-H), leading to a clearer and more interpretable image. This qualitative improvement suggests that SMART technology effectively addresses the visual degradation caused by metallic objects.
The quantitative analysis of artifact reduction, detailed in Table 1, revealed statistically significant improvements with SMART-Full reconstruction compared to original CBCT images. Radial artifacts, which were present in 21 cases in the original CBCT, were reduced to only 3 cases with SMART-Full reconstruction (P < 0.001). Similarly, streak artifacts were completely eliminated, decreasing from 4 cases in the original CBCT to 0 cases with SMART-Full (P < 0.001). Dark streak artifacts also showed a significant reduction, from 71 cases in the original CBCT to 26 cases after SMART-Full reconstruction (P < 0.001). However, thickening artifacts did not demonstrate a statistically significant reduction (P = 0.118), indicating that SMART-Full may have limited efficacy against this specific artifact type.
| Artifact type | Original | SMART-Full | χ² | P value | ||
| N | Y | Total | ||||
| Radial | N | 51 | 1 | 52 | ||
| Y | 19 | 2 | 21 | |||
| Total | 70 | 3 | 73 | 14.45 | < 0.001 | |
| Streak | N | 69 | 0 | 69 | ||
| Y | 4 | 0 | 4 | |||
| Total | 73 | 0 | 73 | 2.25 | < 0.001 | |
| Thickening | N | 42 | 6 | 48 | ||
| Y | 14 | 11 | 25 | |||
| Total | 56 | 17 | 73 | 2.450 | 0.118 | |
| Dark streak | N | 2 | 0 | 2 | ||
| Y | 45 | 26 | 71 | |||
| Total | 47 | 26 | 73 | 43.022 | < 0.001 | |
Further analysis compared the effectiveness of SMART-Full and SMART-Small reconstruction methods in artifact elimination, as presented in Table 2. SMART-Full reconstruction proved to be significantly more effective than SMART-Small across most artifact types. For radial artifacts, SMART-Full resulted in 0 cases with artifacts, whereas SMART-Small showed 23 cases (P < 0.001). Streak artifacts were also completely absent in SMART-Full reconstructions, while SMART-Small still presented 4 cases (P = 0.008). Dark streak artifacts were eliminated in SMART-Full (0 cases) but persisted in 26 cases with SMART-Small (P < 0.001). Consistent with the previous comparison, thickening artifacts did not show a significant difference between SMART-Full and SMART-Small reconstructions (P = 0.453).
| Artifact type | SMART-Full | SMART-Small | χ² | P value | ||
| N | Y | Total | ||||
| Radial | N | 6 | 44 | 50 | ||
| Y | 0 | 23 | 23 | |||
| Total | 6 | 67 | 73 | 42.023 | < 0.001 | |
| Streak | N | 61 | 8 | 69 | ||
| Y | 0 | 4 | 4 | |||
| Total | 61 | 12 | 73 | 6.125 | 0.008 | |
| Thickening | N | 51 | 5 | 56 | ||
| Y | 2 | 15 | 17 | |||
| Total | 53 | 20 | 73 | 0.571 | 0.453 | |
| Dark streak | N | 36 | 11 | 47 | ||
| Y | 0 | 26 | 26 | |||
| Total | 36 | 37 | 73 | 9.091 | < 0.001 | |
Beyond artifact reduction, SMART-Full reconstruction also demonstrated superior performance in critical quantitative metrics. As shown in Table 3, a paired sample t-test revealed significant differences in puncture needle diameter measurement between original CBCT and SMART-Full reconstructed CBCT [original: 2.05 ± 0.59 mm; SMART-Full: 2.16 ± 0.52 mm; mean difference: -0.12; t (72) = -4.48; P < 0.001; Cohen’s d = -0.53]. Furthermore, significant improvements were observed in ROI minimum density [mean difference: -137.78; t (72) = -5.93; P < 0.001; Cohen’s d = -0.69] and ROI average density [mean difference: -57.01; t (72) = -5.40; P < 0.001; Cohen’s d = -0.63]. The ROI maximum density, however, did not show a statistically significant difference (P = 0.414).
| Variables | Original | SMART-Full | Mean difference | t (df) | P value | Cohen’s d |
| Puncture needle diameter | 2.05 ± 0.59 | 2.16 ± 0.52 | -0.12 | -4.48 (72) | < 0.001 | -0.53 |
| ROI minimum density | -926.12 ± 206.22 | -788.34 ± 299.63 | -137.78 | -5.93 (72) | < 0.001 | -0.69 |
| ROI maximum density | 18.88 ± 287.83 | 2.52 ± 260.45 | 16.36 | 0.82 (72) | 0.414 | 0.1 |
| ROI average density | -483.77 ± 269.29 | -426.76 ± 302.13 | -57.01 | -5.40 (72) | < 0.001 | -0.63 |
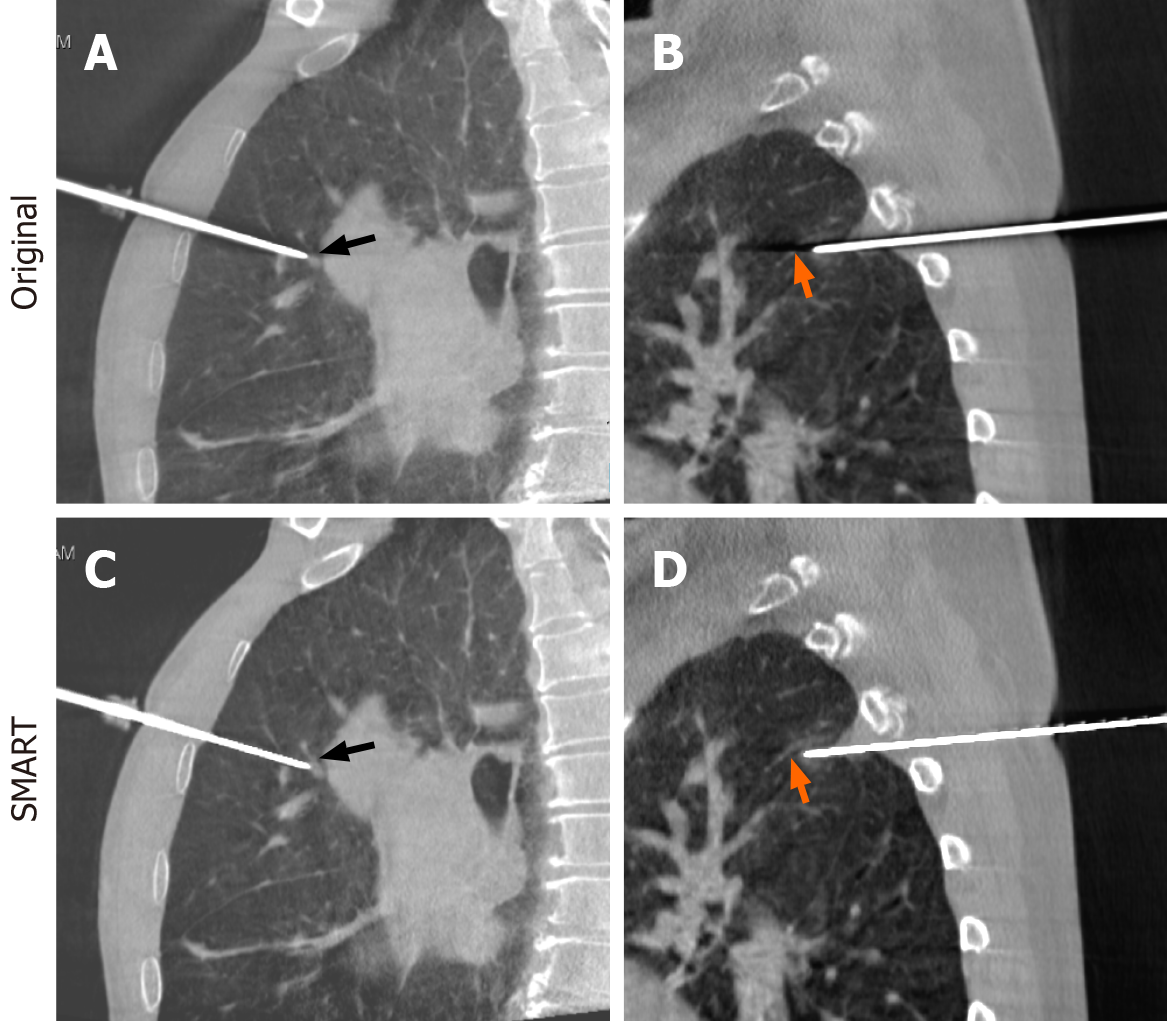
The enhanced image quality facilitated by SMART technology also led to clearer visualization of anatomical structures that were previously obscured by artifacts. Figure 2 illustrates two representative cases where metal artifacts in the original CBCT images (Figure 2A and B) hindered the observation of vessels adjacent to the lung nodules. After SMART-Full reconstruction (Figure 2C and D), these critical vessels became distinctly visible, as indicated by the arrows, which is crucial for precise puncture path planning and avoiding complications during PTNB procedures.
Our study demonstrates that the application of the SMART in CBCT-guided PTNB significantly reduces metal artifacts from coaxial needles, leading to improved image quality and clearer visualization of lung nodules and surrounding anatomical structures.
These findings indicate that SMART iterative reconstruction, particularly in full ROI mode, effectively mitigates radial, streak, and dark streak artifacts by addressing beam hardening and photon starvation effects inherent to metallic objects[19-21]. The observed reductions in artifact prevalence and improvements in quantitative metrics, such as needle diameter measurements and ROI densities, suggest that the algorithm enhances image fidelity without introducing substantial noise or distortion. This is likely due to its iterative projection-based approach, which corrects artifact-induced inconsistencies in the projection data, thereby revealing obscured details critical for procedural accuracy[12,22,23]. However, the limited impact on thickening artifacts implies that SMART may not fully compensate for all types of metal-induced distortions, possibly because these arise from partial volume effects less amenable to iterative correction[24,25].
Our results align with prior research on MAR techniques in interventional imaging. For instance, similar to Estler et al[16] findings on SMART’s efficacy in reducing streak artifacts from catheter tips during bronchial artery embolization, we observed complete elimination of streak artifacts in SMART-full reconstructions, underscoring the algorithm’s robustness against high-density materials[16]. This consistency extends to broader MAR literature, where iterative algorithms and sinogram correction methods have been shown to improve image quality in fields like orthopedics and neuroradiology[10,14]. However, unlike studies focused on qualitative assessments in neurosurgery or dental CBCT, our quantitative evaluation provides more objective evidence of SMART’s benefits in lung interventions, where small lesion targeting demands precise anatomical delineation[26,27]. Discrepancies, such as the variable success against thickening artifacts noted in our work and in phantom studies of biopsy needles, may stem from differences in metallic object size and imaging parameters, highlighting the need for context-specific optimizations in MAR applications[28,29].
The implications of these findings are substantial for interventional radiology, as enhanced image clarity with SMART may potentially optimize puncture path planning, potentially reduce procedural risks like pneumothorax or hemorrhage, and potentially improve diagnostic yield for small lung nodules[15,30]. Clinically, this may translate to shorter operation times, lower infection risks, and greater procedural efficiency, particularly when integrated with CBCT’s real-time guidance capabilities. Theoretically, our work advances the understanding of MAR in dynamic, low-contrast envi
Despite these strengths, our study has several limitations to consider when interpreting results. The retrospective, single-center design, involving only 73 CBCT scans from 68 patients, may introduce selection bias, as only cases with complete and uncorrupted imaging data were included. Additionally, no a priori sample size calculation was performed, which could limit power to detect subtler differences in artifact reduction or quantitative metrics. These factors may restrict external validity and generalizability to other institutions or CBCT systems. Furthermore, artifact evaluation was performed by consensus of two blinded readers without formal inter-observer agreement statistics (e.g., kappa coefficient), which could be addressed in future studies. The statistical analysis relied on multiple paired tests without correction for multiplicity, which is appropriate for exploratory purposes but should be considered when interpreting results. Evaluation focused on imaging endpoints; potential clinical benefits (e.g., reduced procedure time, complications) require prospective validation. SMART is vendor-specific; comparisons to other MAR methods (e.g., NMAR[15], direct sinogram correction[11]) were not performed due to proprietary implementation. Future multi-vendor prospective studies with clinical outcomes are warranted. Alternative explanations for residual artifacts, such as patient motion or suboptimal ROI selection in small-mode reconstructions, were minimized through standardized protocols but warrant further investigation.
In summary, SMART technology enhances CBCT-guided PTNB by reducing metal artifacts and improving anatomical visualization, offering a valuable tool for precise lung nodule interventions. Future research should prospectively evaluate SMART’s impact on clinical outcomes, such as diagnostic yield, complication rates, and procedure times, in larger, multi-vendor cohorts.
Dr. Nan Chang (Siemens Healthineers) kindly supported this work.
| 1. | Veltri A, Bargellini I, Giorgi L, Almeida PAMS, Akhan O. CIRSE Guidelines on Percutaneous Needle Biopsy (PNB). Cardiovasc Intervent Radiol. 2017;40:1501-1513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 135] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 2. | Guo Z, Shi H, Li W, Lin D, Wang C, Liu C, Yuan M, Wu X, Xiong B, He X, Duan F, Han J, Yang X, Yu H, Si T, Xu L, Xing W, Jinhua H, Wang Y, Xie H, Cui L, Gao W, He D, Liu C, Liu Z, Ma C, Pan J, Shao H, Tu Q, Yong L, Xu Y, Weihao Z, Qiang Z, Wang S. Chinese multidisciplinary expert consensus: Guidelines on percutaneous transthoracic needle biopsy. Thorac Cancer. 2018;9:1530-1543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 3. | Ren Q, Zhou Y, Yan M, Zheng C, Zhou G, Xia X. Imaging-guided percutaneous transthoracic needle biopsy of nodules in the lung base: fluoroscopy CT versus cone-beam CT. Clin Radiol. 2022;77:e394-e399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 4. | Fior D, Vacirca F, Leni D, Pagni F, Ippolito D, Riva L, Sironi S, Corso R. Virtual Guidance of Percutaneous Transthoracic Needle Biopsy with C-Arm Cone-Beam CT: Diagnostic Accuracy, Risk Factors and Effective Radiation Dose. Cardiovasc Intervent Radiol. 2019;42:712-719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 5. | Jiao D, Yuan H, Zhang Q, Han X. Flat detector C-arm CT-guided transthoracic needle biopsy of small (≤2.0 cm) pulmonary nodules: diagnostic accuracy and complication in 100 patients. Radiol Med. 2016;121:268-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 6. | Yang L, Wang Y, Li L, Liu D, Wu X, Zhang W, Pan F, Liang H, Zheng C. C-Arm Cone-Beam CT Virtual Navigation versus Conventional CT Guidance in the Transthoracic Lung Biopsy: A Case-Control Study. Diagnostics (Basel). 2022;12:115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 7. | Fang HY, Chen KA, Wen YW, Wen CT, Pan KT, Chiu CH, Hsieh MJ, Chao YK. Efficacy and Safety of Preoperative vs. Intraoperative Computed Tomography-Guided Lung Tumor Localization: A Randomized Controlled Trial. Front Surg. 2021;8:809908. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 8. | Prell D, Kyriakou Y, Struffert T, Dörfler A, Kalender WA. Metal artifact reduction for clipping and coiling in interventional C-arm CT. AJNR Am J Neuroradiol. 2010;31:634-639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 75] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 9. | Prell D, Kalender WA, Kyriakou Y. Development, implementation and evaluation of a dedicated metal artefact reduction method for interventional flat-detector CT. Br J Radiol. 2010;83:1052-1062. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 30] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 10. | Selles M, van Osch JAC, Maas M, Boomsma MF, Wellenberg RHH. Advances in metal artifact reduction in CT images: A review of traditional and novel metal artifact reduction techniques. Eur J Radiol. 2024;170:111276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 56] [Reference Citation Analysis (0)] |
| 11. | Hegazy MAA, Cho MH, Cho MH, Lee SY. Metal Artifact Reduction in Dental CBCT Images Using Direct Sinogram Correction Combined with Metal Path-Length Weighting. Sensors (Basel). 2023;23:1288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 12. | Washio H, Ohira S, Funama Y, Morimoto M, Wada K, Yagi M, Shimamoto H, Koike Y, Ueda Y, Karino T, Inui S, Nitta Y, Miyazaki M, Teshima T. Metal artifact reduction using iterative CBCT reconstruction algorithm for head and neck radiation therapy: A phantom and clinical study. Eur J Radiol. 2020;132:109293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 13. | Shellikeri S, Girard E, Setser R, Bao J, Cahill AM. Metal artefact reduction algorithm for correction of bone biopsy needle artefact in paediatric C-arm CT images: a qualitative and quantitative assessment. Clin Radiol. 2016;71:925-931. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 14. | Meyer E, Raupach R, Lell M, Schmidt B, Kachelriess M. Normalized metal artifact reduction (NMAR) in computed tomography. Med Phys. 2010;37:5482-5493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 345] [Cited by in RCA: 335] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 15. | Katsura M, Sato J, Akahane M, Kunimatsu A, Abe O. Current and Novel Techniques for Metal Artifact Reduction at CT: Practical Guide for Radiologists. Radiographics. 2018;38:450-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 286] [Cited by in RCA: 224] [Article Influence: 28.0] [Reference Citation Analysis (1)] |
| 16. | Estler A, Bongers M, Thomas C, Hefferman G, Hofmann J, Hoffmann R, Nikolaou K, Grosse U, Grözinger G. Application of a Metal Artifact Reduction Algorithm for C-Arm Cone-Beam CT: Impact on Image Quality and Diagnostic Confidence for Bronchial Artery Embolization. Cardiovasc Intervent Radiol. 2019;42:1449-1458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 17. | Otsuka T, Nishihori M, Izumi T, Uemura T, Sakai T, Nakano M, Kato N, Kanamori F, Tsukada T, Uda K, Yokoyama K, Araki Y, Saito R. Streak Metal Artifact Reduction Technique in Cone Beam Computed Tomography Images after Endovascular Neurosurgery. Neurol Med Chir (Tokyo). 2021;61:468-474. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 18. | Prell D, Kyriakou Y, Beister M, Kalender WA. A novel forward projection-based metal artifact reduction method for flat-detector computed tomography. Phys Med Biol. 2009;54:6575-6591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 82] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 19. | Wellenberg RHH, Hakvoort ET, Slump CH, Boomsma MF, Maas M, Streekstra GJ. Metal artifact reduction techniques in musculoskeletal CT-imaging. Eur J Radiol. 2018;107:60-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 123] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 20. | Schmidt TG, Sammut BA, Barber RF, Pan X, Sidky EY. Addressing CT metal artifacts using photon-counting detectors and one-step spectral CT image reconstruction. Med Phys. 2022;49:3021-3040. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 21. | Midthun P, Kirkhus E, Østerås BH, Høiness PR, England A, Johansen S. Metal artifact reduction on musculoskeletal CT: a phantom and clinical study. Eur Radiol Exp. 2023;7:46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 22. | Zeng GL. A projection-domain iterative algorithm for metal artifact reduction by minimizing the total-variation norm and the negative-pixel energy. Vis Comput Ind Biomed Art. 2022;5:1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 23. | Wuest W, May MS, Brand M, Bayerl N, Krauss A, Uder M, Lell M. Improved Image Quality in Head and Neck CT Using a 3D Iterative Approach to Reduce Metal Artifact. AJNR Am J Neuroradiol. 2015;36:1988-1993. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 40] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 24. | Hatamikia S, Biguri A, Kronreif G, Russ T, Kettenbach J, Birkfellner W. Source-detector trajectory optimization for CBCT metal artifact reduction based on PICCS reconstruction. Z Med Phys. 2024;34:565-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 25. | Hakim A, Pastore-Wapp M, Vulcu S, Dobrocky T, Z'Graggen WJ, Wagner F. Efficiency of Iterative Metal Artifact Reduction Algorithm (iMAR) Applied to Brain Volume Perfusion CT in the Follow-up of Patients after Coiling or Clipping of Ruptured Brain Aneurysms. Sci Rep. 2019;9:19423. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 26. | Jo GD, Park CW, Jeon KJ, Han SS. Quantitative evaluation of metal artifact reduction in CBCT under varying exposure modes and rod orientations. Sci Rep. 2025;15:20645. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 27. | Watanabe S, Urikura A, Ohashi K, Kitera N, Tsuchiya T, Kasai H, Kawai T, Hiwatashi A. Artifact reduction in low and ultra-low dose chest computed tomography for patients with pacemaker: A phantom study. Radiography (Lond). 2024;30:770-775. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 28. | Reynoso-Mejia CA, Couillard AB, Kisting MA, Lee FT Jr, Szczykutowicz TP, Wagner MG. Needle artifact redistribution technique (Needle-ART): A method for metal artifact reduction during CT interventionismbased on gantry tilt. Phys Med. 2024;127:104848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 29. | Zhang J, Liu M, Liu D, Li X, Lin M, Tan Y, Luo Y, Zeng X, Yu H, Shen H, Wang X, Liu L, Tan Y, Zhang J. Low-dose CT with tin filter combined with iterative metal artefact reduction for guiding lung biopsy. Quant Imaging Med Surg. 2022;12:1359-1371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 30. | Can E, Böning G, Lüdemann WM, Hosse C, Kolck J, Paparoditis S, Nguyen T, Piper SK, Geisel D, Wieners G, Gebauer B, Elkilany A, Jonczyk M. Evaluation of a prototype metal artifact reduction algorithm for cone beam CT in patients undergoing radioembolization. Sci Rep. 2024;14:16399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/