Published online Mar 26, 2026. doi: 10.4330/wjc.v18.i3.116299
Revised: December 9, 2025
Accepted: February 9, 2026
Published online: March 26, 2026
Processing time: 136 Days and 13.3 Hours
Electrocardiographic (ECG) artifacts are anomalous signals unrelated to cardiac electrical activity that interfere with our interpretation and management. They can mimic serious arrhythmias or ischemic changes and often lead to unnecessary interventions. Understanding why such artifacts occur requires a grasp of how ECG signals are recorded, from tiny millivolt-level cardiac signals traveling from the heart through skin electrodes, cables, amplifiers, filters, and finally the display on the monitors or ECG strip. At each step, artifacts may enter, whether from patient movement, tremor, electrode-skin interface problems, broken leads, en
Core Tip: Electrocardiographic artifacts are non-cardiac signals that corrupt monitoring, often mimicking serious arrhythmias or ischemia and leading to unnecessary interventions. They are introduced at various points in the recording chain - from patient movement and electrode issues to environmental interference - due to the tiny cardiac signal and frequency overlap with noise. Recognizing these artifacts is vital; it requires clinical correlation, checking physiological plausibility, and assessing lead distribution to avoid misdiagnosis and safely manage the patient. This review summarizes the physics of electrocardiographic recording in simple terms, classifies common artifacts and outlines practical bedside clues and pre
- Citation: Mondal S, Raja DP, Muslim NA, Prabhu MA. Electrocardiographic artifacts in clinical practice: A logical approach to recognition and prevention. World J Cardiol 2026; 18(3): 116299
- URL: https://www.wjgnet.com/1949-8462/full/v18/i3/116299.htm
- DOI: https://dx.doi.org/10.4330/wjc.v18.i3.116299
Electrocardiograms constitute the cornerstone of cardiovascular diagnosis. In modern clinical practice, electrocardiographic (ECG) interpretation guides decisions in the emergency department, intensive care unit (ICU), operating theatre, and outpatient clinic and, increasingly, in patients’ homes through wearable devices. Its advantages include technical simplicity, cost-effectiveness, and the ability to deliver instant information about the heart’s rhythm and ischemic status. However, its greatest weakness lies in its fragility: The signal captured at the skin surface is relatively small and vulne
Artifacts, which are signals not generated by the heart, are common in daily practice and are often overlooked. They can be obvious and inconsequential at best and misleading and capable of mimicking life-threatening arrhythmias, inc
Despite their frequency and clinical consequences, artifacts are not routinely emphasized in medical training. As a result, it is common for physicians to recognize artifacts in a pattern-recognizing manner without un
The ECG records very small voltages, typically in the range of 0.5-5 mV. For comparison, a standard household light bulb operates at 220 Volts in India or 110 Volts in the United States. The ECG signal is therefore a million times smaller and can be easily overshadowed by competing signals from skeletal muscle or electrical interference in the environment. The body itself is electrically noisy. Skeletal muscle contraction produces signals that are up to 10 times greater than cardiac potentials. Respiration alters thoracic impedance, causing slow drifts at baseline. Even the act of speaking introduces movement that can distort signals.
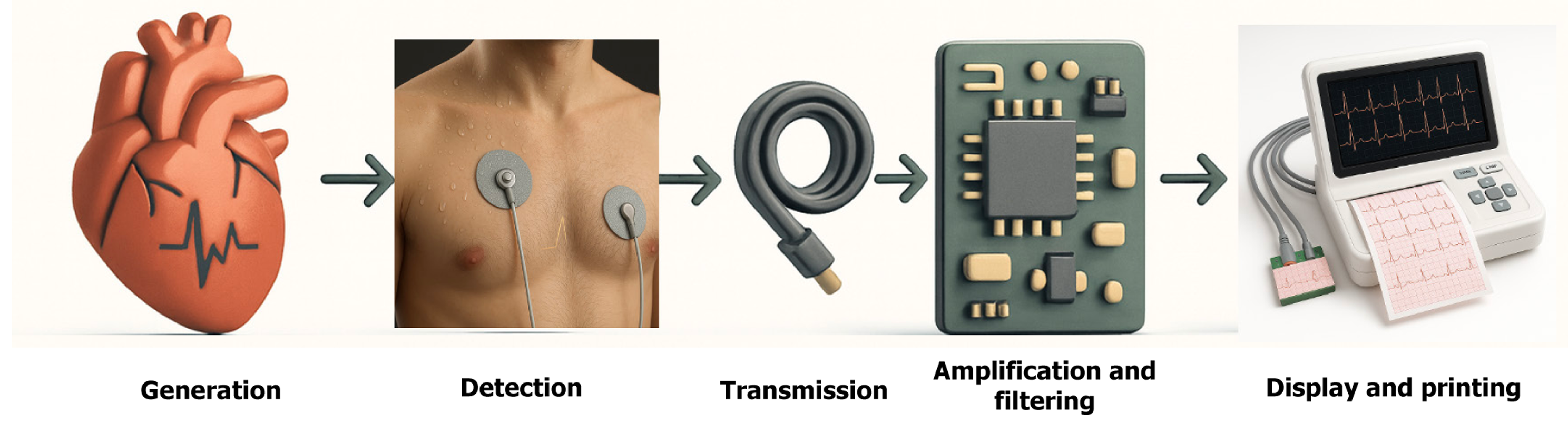
To understand artifacts, tracing the journey of the ECG signal is essential (Figure 1, Table 1).
| Step | Function | Common artifact sources | Clinical example |
| Generation | The heart produces approximately 1 mV currents | Competing signals from the muscle | Myopotential artifacts mimicking atrial fibrillation |
| Detection | Electrodes capture surface signals | Sweat, hair, poor adhesion, movement | Wandering baseline or false ST elevation (baseline drift) |
| Transmission | Leads carry signals | Cable fracture, patient motion, EMI | Flatline due to broken lead; saw-tooth interference from cautery |
| Amplification and filtering | Boosts and cleans signals | Amplifies both signal and noise; filter distortion | QRS smoothed, or ST segments flattened, spurious ST shifts |
| Display and printing | Trace presented | All upstream noise is now visible | Artifact interpreted as ventricular tachycardia, acceleration or deceleration artifacts |
Generation: Myocardial depolarization generates action potentials that summate into measurable electrical fields.
Detection: Surface electrodes act as receivers, picking up these potentials. Their performance depends on skin pre
Transmission: Leads and cables transmit the signal to the ECG machine. Mechanical motion, cable fracture, or electromagnetic interference can corrupt transmission.
Amplification and filtering: The amplifiers enhance the small signal but also amplify unwanted noise. In contrast, the filtering removes specific frequencies, thus retaining only the spectrum of frequencies containing the cardiac signals. However, filtering is not perfect, as some of the noises overlap with the frequency spectrum of the signals.
Display and printing: The final waveform is shown on screen or paper. By this point, artifacts are indistinguishable from true cardiac activity unless identified through logic. The final printing is subsequently performed on calibrated ECG paper.
Amplification - the indiscriminate booster: Amplifiers are crucial for making tiny ECG signals visible. However, they are nonselective and amplify every signal they receive, whether it is a true QRS complex, skeletal muscle tremor, or ele
Filters - sieves of the ECG: Filters aim to retain useful cardiac signals and remove unwanted noise by filtering off the fre
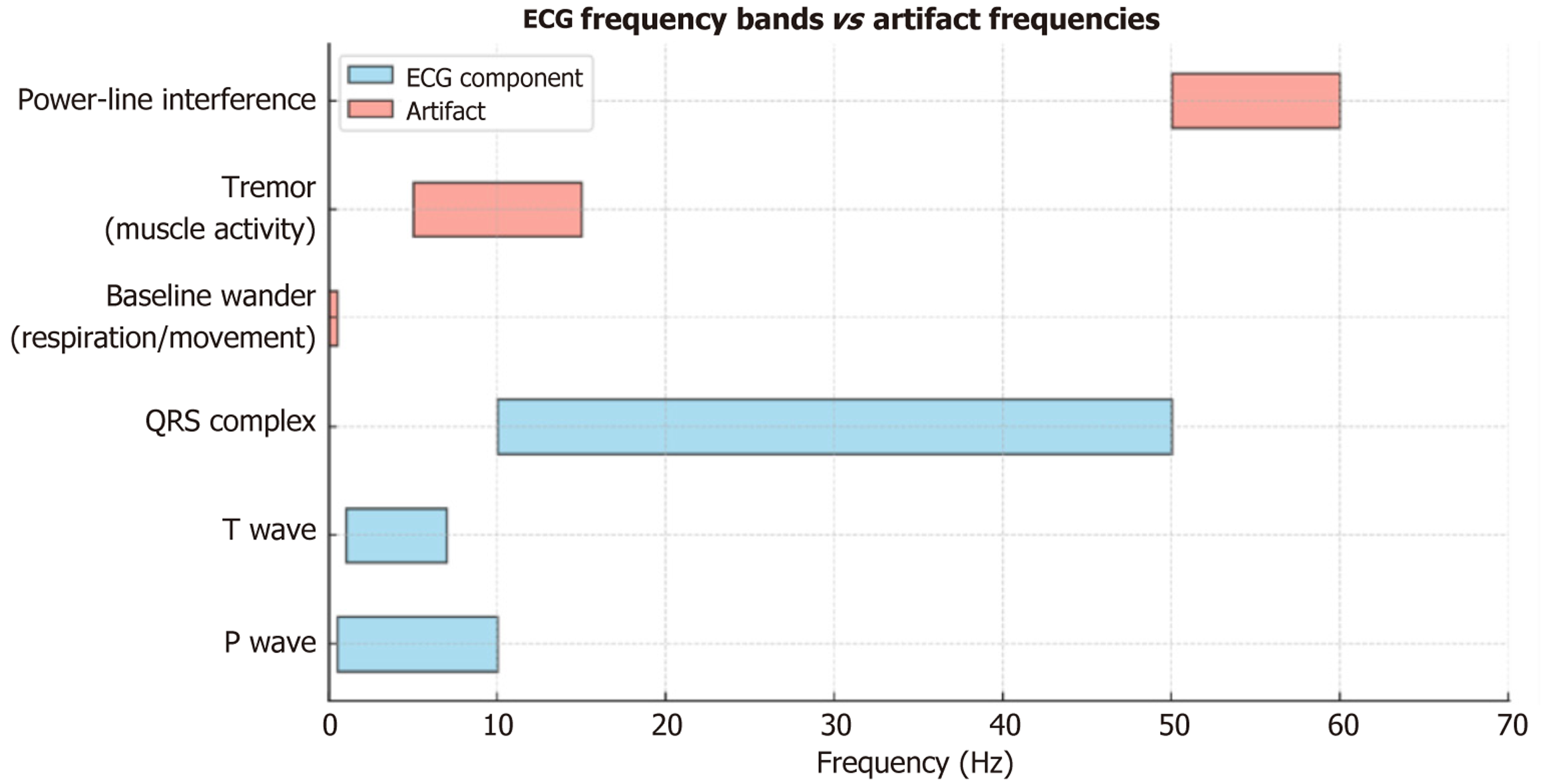
High-pass filters (cutoff approximately 0.05-0.5 Hz): Allow any signal of higher frequency than their cutoff value. Remove very slow drifts, such as baseline wander caused by respiration or electrode movement.
Low-pass filters (cutoff approximately 100-150 Hz for diagnostic ECGs): Allow frequencies lower than their cutoff and remove signals of higher frequency, including tremors or electrical buzzes.
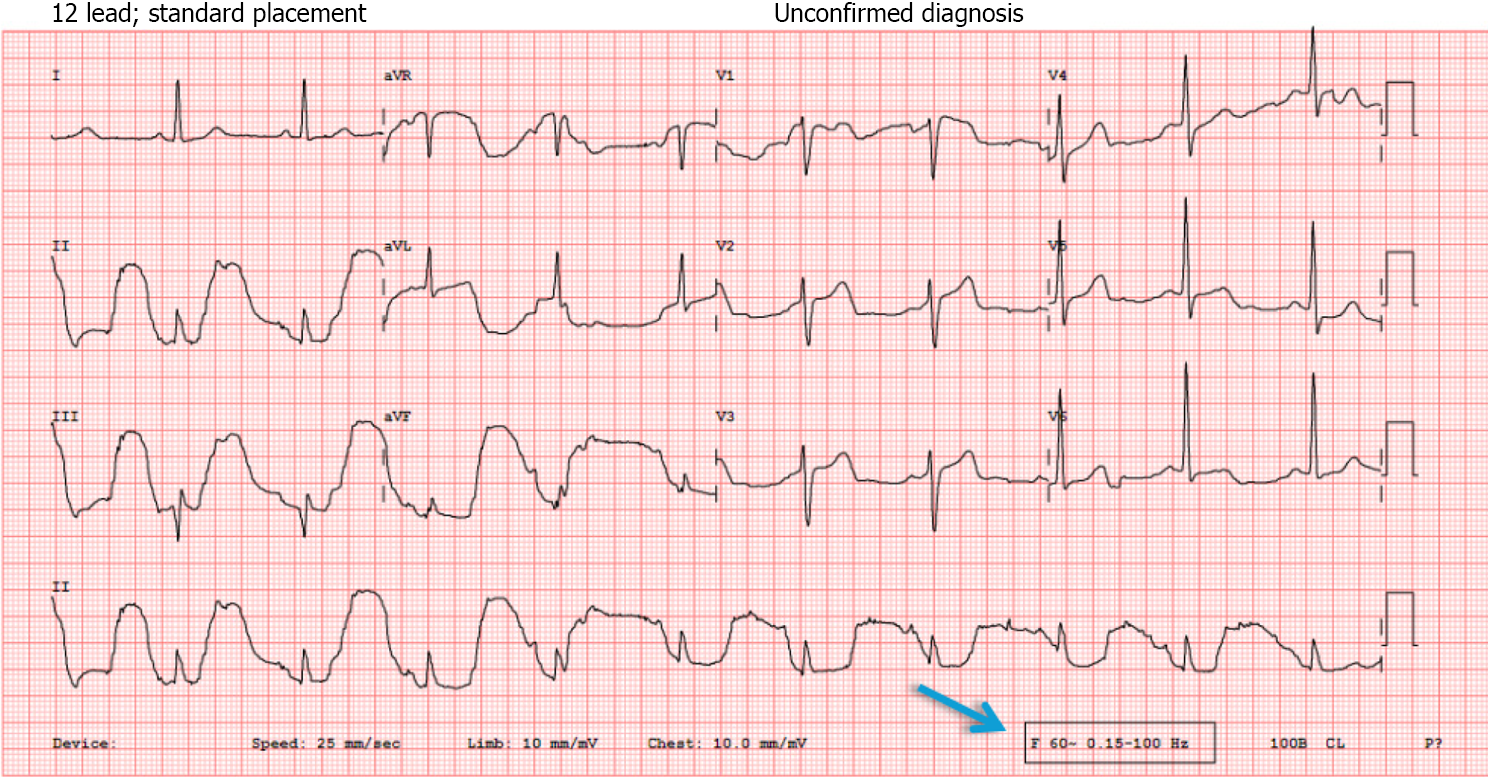
Notch filters (50 Hz in India, 60 Hz in the United States): Remove specific frequency interference from the power supply lines.
Bandpass filters (0.05-150 Hz): Allow only the typical frequency range of cardiac signals. Analogy: Filters work like sieves. They can block very large particles (slow baseline wander) or very fine dust (high-frequency tremors). However, when unwanted particles are the same size as the intended grains, separation becomes impossible.
Despite sophisticated filtering, artifacts persist (Figure 3). The reasons include the following.
Frequency overlap: Tremor (4-10 Hz) overlaps with P- and T-wave frequencies. Removing tremors risks removing true signals.
Risk of distortion: Excessive filtering can smooth sharp deflections, flatten ST segments, or widen QRS complexes. This may create false pathology.
Unpredictable noise: Motion artifacts are irregular and cannot be predicted or filtered, such as steady 50 Hz interference.
Non-electrical faults: Loose electrodes or cable breaks produce sudden baseline shifts. These are not frequency-based or bypass filters.
No clinical reasoning: Machines cannot judge physiologic plausibility. A monitor may display “ventricular fibrillation” while the patient is talking comfortably.
Filters can lessen but not completely eliminate artifacts in ECG recordings, and excessive filtering may also obscure important diagnostic information by distorting true waveforms. Modern ECG devices often provide both filtered and unfiltered traces, enabling clinicians to compare and better differentiate artifacts from genuine signals. Ultimately, the ECG should never be interpreted in isolation, and correlation with the patient’s clinical context remains essential.
Artifacts can be classified in several ways, but for practical bedside teaching, a clinical classification is most effective. We categorize them into four main groups: Patient-related, equipment-related, environmental, and device-related (Table 2).
| Category | Source | ECG appearance | Clinical mimic | Solution |
| Patient-related | Tremor, shivering, respiration, movement | Irregular baseline, wandering drift | AF, flutter, VT, ST changes | Patient cooperation |
| Equipment-related | Loose electrodes, fractured cables, faulty machine | Baseline jumps, flatline, spikes | Asystole, VF | Change cables, better skin preparation, electrode contact |
| Environmental | Power-line hum, cautery, ventilators | Saw-tooth oscillations, rhythmic noise | Flutter, VT | Avoidance of equipment, notch filters |
| Device-related | Pacemaker, LVAD, neurostimulators, IVL | Sharp spikes, continuous noise | VF, tachycardia | Not always possible |
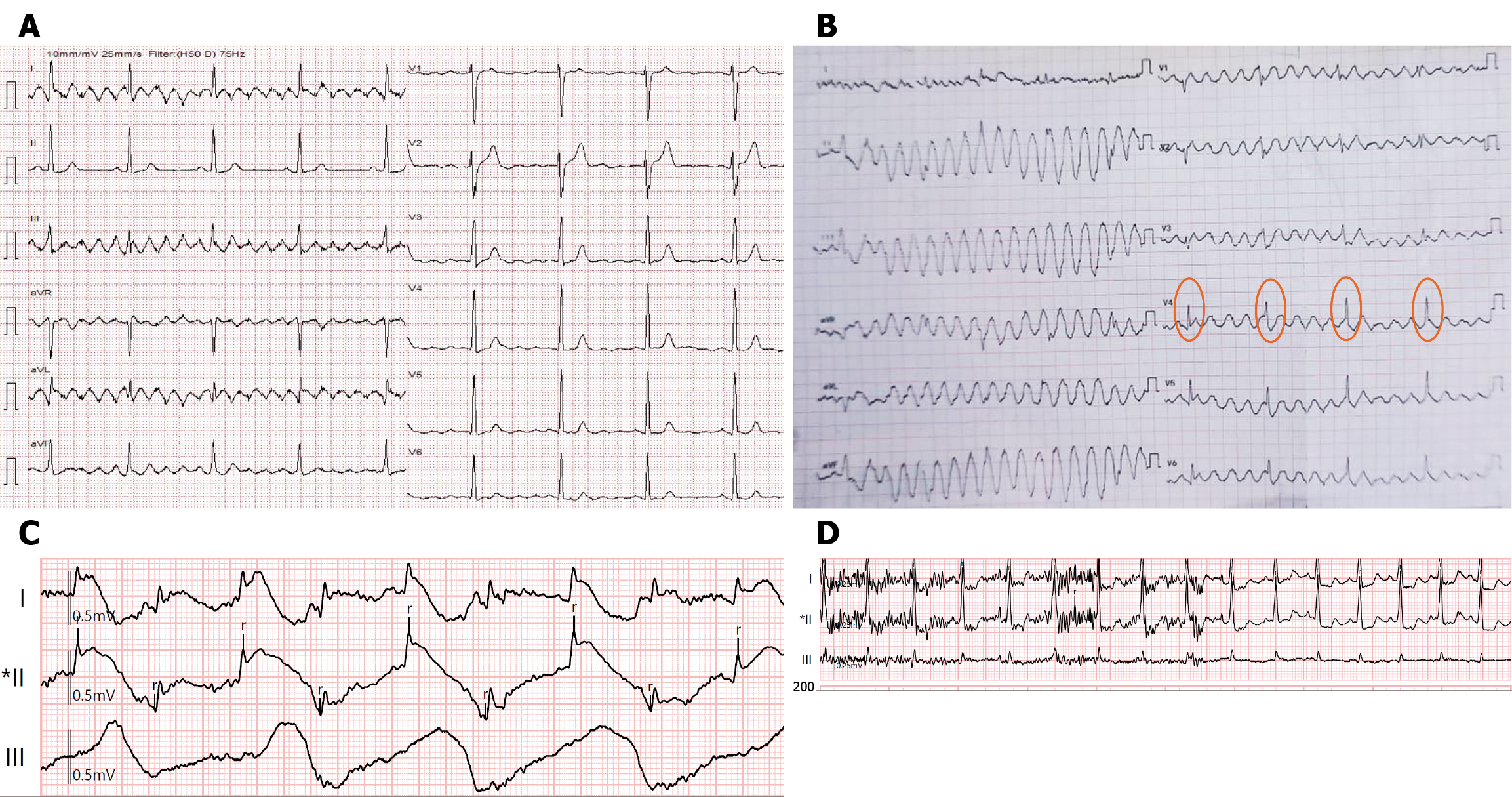
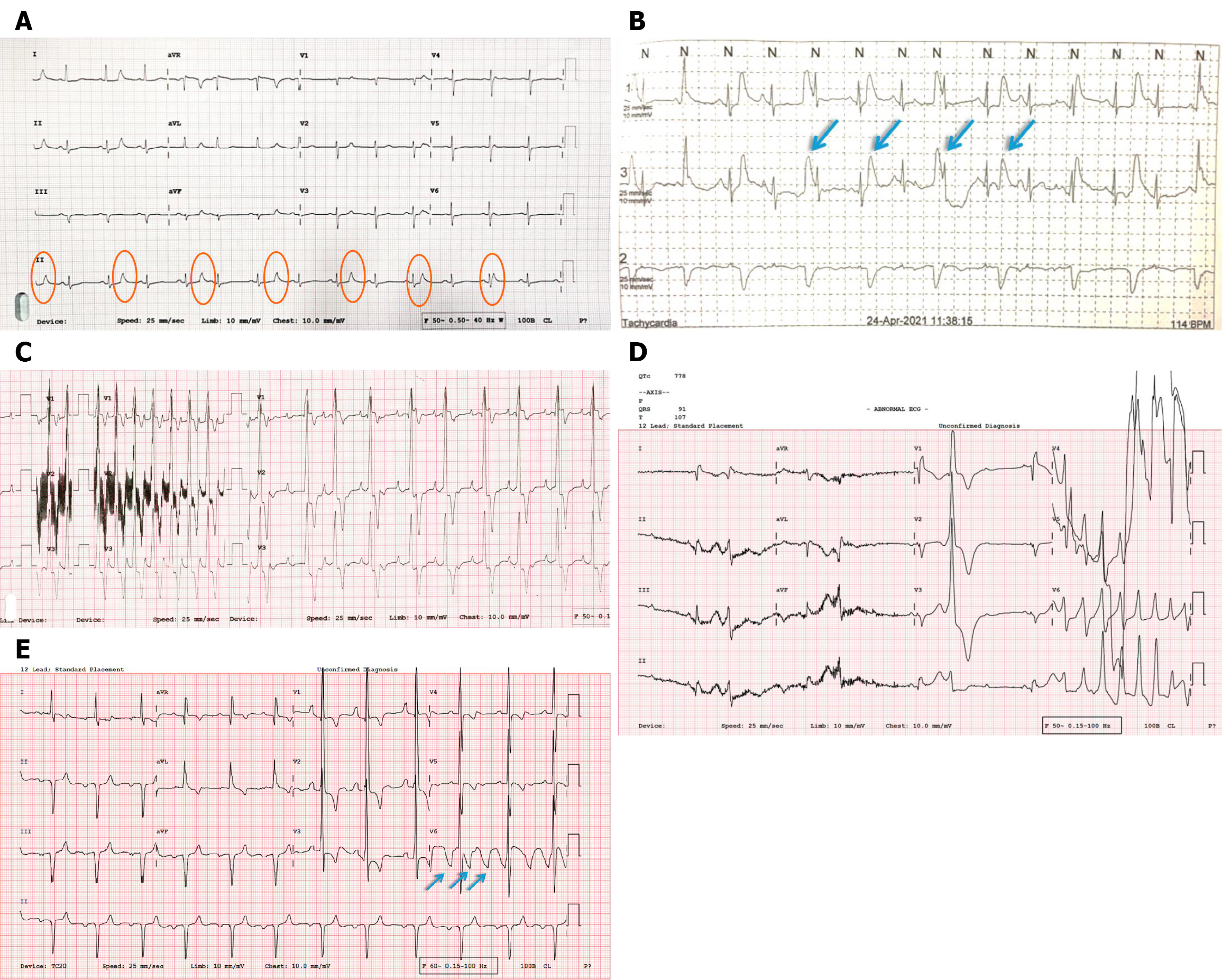
These are the most common sources of spurious ECG signals caused by the patient’s physiological or mechanical movements. Skeletal muscle potentials, which are much greater than cardiac potentials, are easily recorded when muscles contract near the electrodes. This causes quick, irregular baseline oscillations that can resemble atrial fibrillation or coarse undulations from shivering that might be mistaken for atrial flutter or VT (Figure 4A). Gross body movements can also disrupt electrode contact or stretch cables, producing large deflections that may mimic wide-complex tachycardia, sometimes confused as “polymorphic VT” until proven otherwise (Figure 4B). Even breathing affects thoracic impedance and electrode position, leading to slow baseline drift that can resemble ST elevation or depression (Figure 4C). A key point is that when the patient moves, shivers, or trembles, the clinician should strongly suspect artifacts, and inter
Another key source of interference comes from faults in electrodes, cables, or the ECG machine itself. Poor electrode contact, often caused by dried gel, sweat, or insufficient adhesion, weakens electrical connectivity and results in inter
Baseline drift is a low-frequency artifact where the isoelectric line on an electrocardiogram tracing slowly moves up and down instead of remaining flat. It is one of the most common artifacts encountered and is usually due to poor contact between the skin and the electrode. This can be mitigated by improving contact, better skin preparation, the use of app
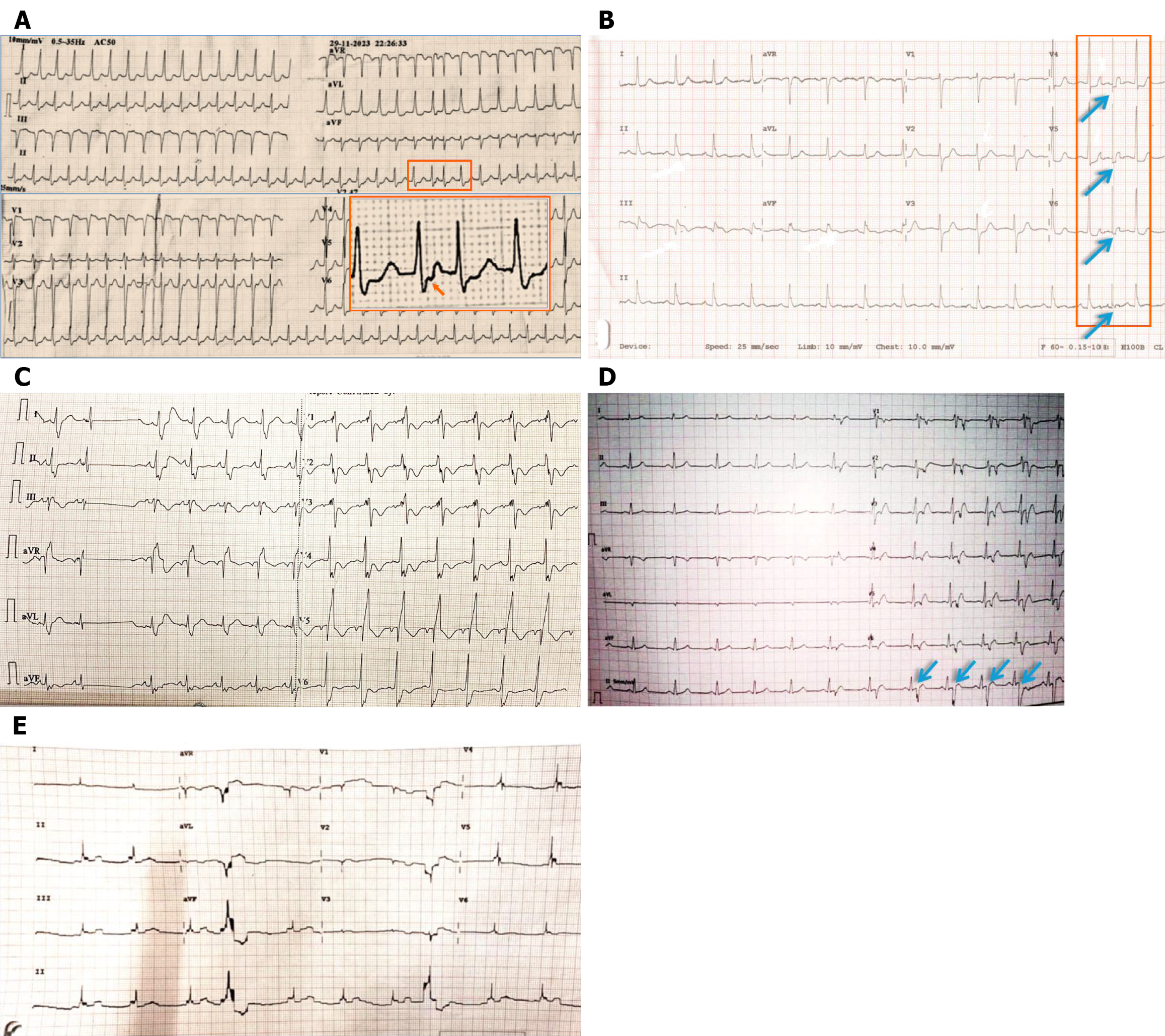
The arterial pulse-tap artifact, also known as Aslanger’s sign or electromechanical association artifact, is caused by an electrode being placed over a pulsating artery (e.g., radial, brachial, axillary, or even the apical impulse on the chest, an arteriovenous fistula created for hemodialysis)[12,13]. The mechanical vibrations of the arterial pulsation are picked up by the electrode and superimposed on the electrical signal of the heart. The key characteristic is that the artifact is regular and occurs every cardiac cycle, usually at a constant time delay after the QRS complex, corresponding to the pulse transit time to that location, mimicking an ST elevation or prolonged QT interval (Figure 7B and C). A repeat ECG with repositioning of the electrode easily removes the artifact.
The hospital environment is filled with electrical devices that can cause interference in ECG recordings. Power-line interference is the most common type of interference, occurring as regular sawtooth waves across all leads, corresponding to the local alternating current frequency (50 Hz in India and 60 Hz in the United States). Interestingly, wires connected from the patient’s body to cardiac monitors may produce square wave artifacts (Figure 7D)[3]. In operating theatres and ICUs, electrocautery produces high-frequency interference that may resemble flutter waves or VT (Figure 8A), whereas infusion pumps, ventilators, and dialysis machines generate rhythmic oscillations aligned with device activity. Even static electricity from blankets, clothing, or staff-handling electrodes can cause sharp baseline jumps, making interpretation more difficult.
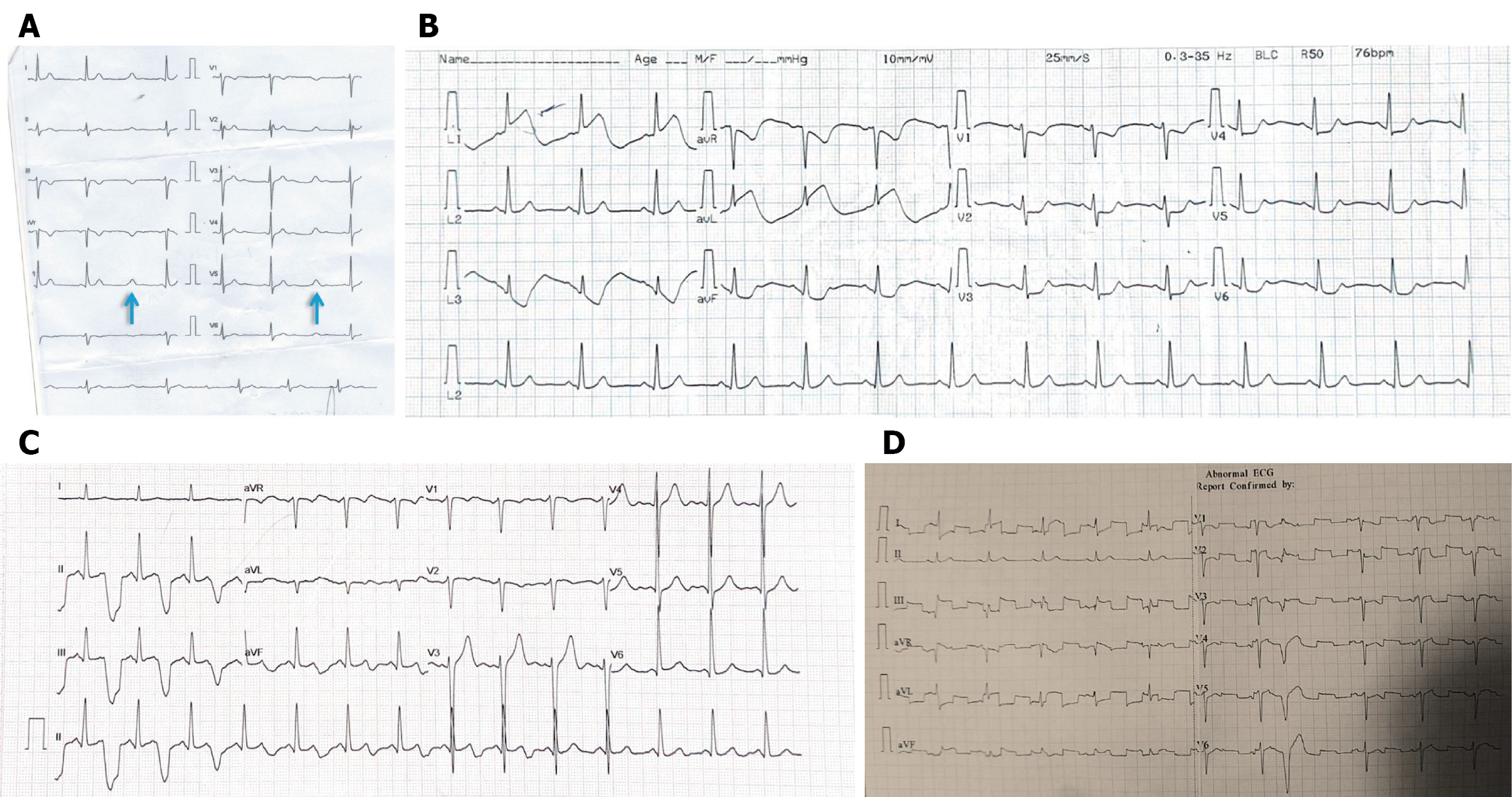
Finally, modern implanted or external devices have introduced new sources of artifacts. Pacemakers are the most well-known example, with pacing spikes often exaggerated or distorted by filtering, sometimes leading to misinterpretation as ventricular fibrillation. Many pseudopacing artifacts can also be observed, which cannot be explained by any pacing algorithm (Figure 8B and C). Owing to their continuous mechanical rotation, left ventricular assist devices produce persistent high-frequency noise that can completely obscure the underlying cardiac rhythm. Neurostimulators - including deep brain, sacral nerve, and gastric stimulators - generate periodic pulses that may overlap with ECG frequencies, adding another layer of complexity (Figure 8D). During intravascular lithotripsy, shock pulses can be detected via ECG. It can even capture, which is called shocktopics (Figure 8E). Even the watch dial can create an artifact at a 1 Hz frequency corresponding to its second dial. As these devices become more common, clinicians must remain aware of their potential to create misleading ECG patterns.
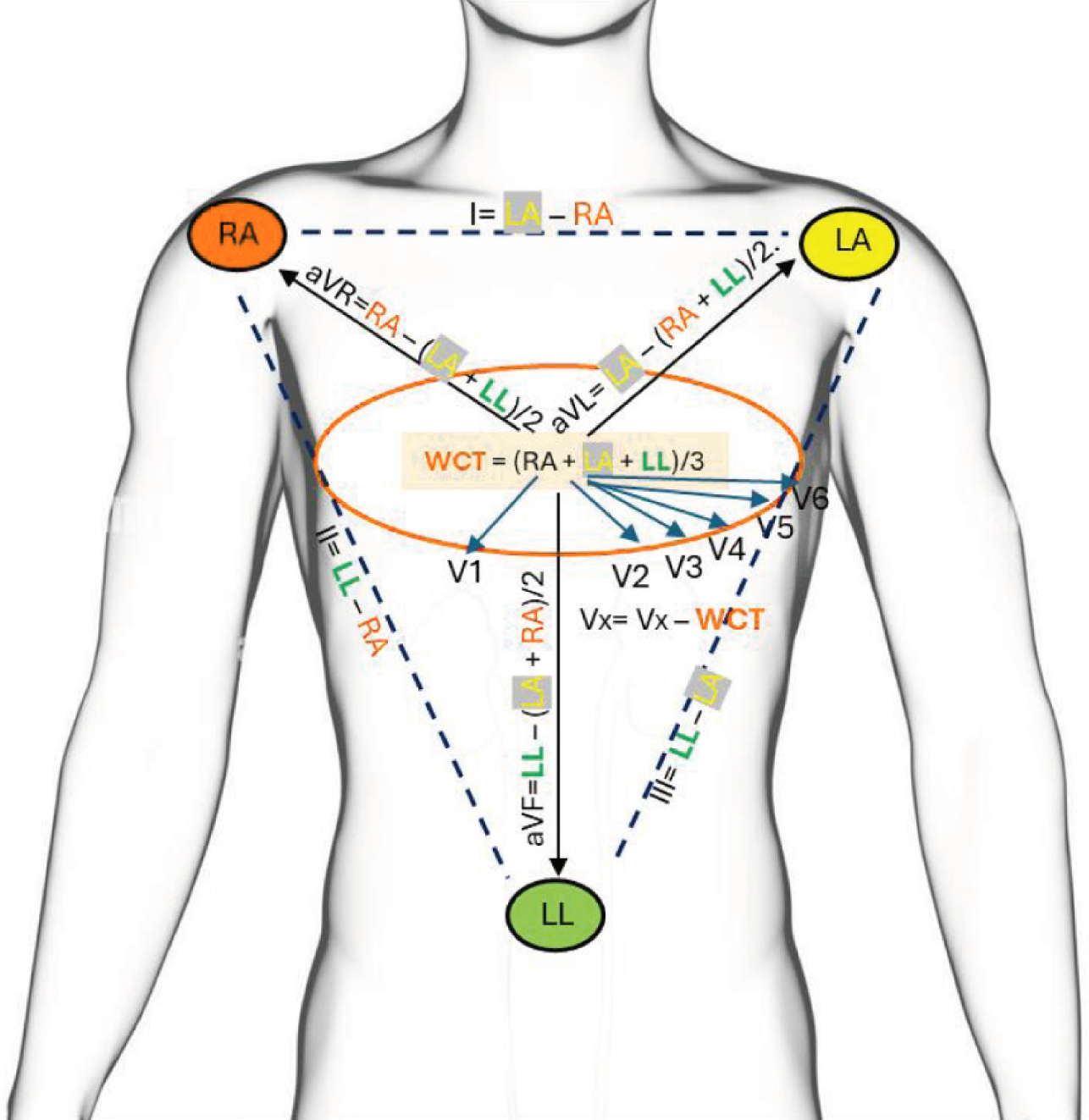
A distinctive but clinically significant aspect of artifact recognition involves understanding why disturbances impact certain leads while sparing others. This is best explained by the role of the Wilson Central Terminal (WCT), which serves as the reference point for all precordial (chest) leads. The WCT is derived by averaging the electrical potentials from the three limb electrodes - right arm, left arm, and left leg. Consequently, when a limb electrode produces noise or becomes unstable, the WCT becomes contaminated, and this disturbance propagates to all chest leads while also distorting the limb leads themselves. Conversely, if a single chest electrode is faulty, the artifact remains confined to that specific lead - for example, noise limited to V3. Since this mechanism is localized to the electrode over the artery, the artifact usually spares one lead completely and affects different leads in a graded manner (Figure 9). Finally, when all leads are affected simultaneously, the source is almost always equipment-related or environmental, such as electrocautery interference or power-line noise. A useful teaching point is that by analyzing the distribution of artifacts across different leads, clinicians can often pinpoint the faulty electrode or differentiate between patient-, equipment-, and environment-related noise.
Artifacts become clinically significant not because they exist but because they closely resemble disease (Table 3). The greatest danger is when an artifact mimics a life-threatening arrhythmia or an ischemic event, potentially leading to mis
| Artifact source | ECG appearance | Clinical mimic | Recognition clues |
| Tremor (Parkinson’s, shivering) | Irregular baseline, absent P waves | Atrial fibrillation | P waves marching through; stable pulse |
| Electrocautery, ventilators | Saw-tooth baseline | Atrial flutter | Localized, no AV conduction ratio |
| Movement, loose cables | Wide irregular complexes | Ventricular tachycardia | Too fast; QRS visible through noise; stable patient |
| Loose electrode, EMI | Chaotic baseline | Ventricular fibrillation | Conscious patient; stable SpO2 |
| Respiration, poor skin contact | Wandering baseline, pseudo ST shift | Myocardial infarction | Inconsistent, resolves with repeat |
| Holter/wearable motion | Drop-outs, spikes | AV block, tachycardia | Correlate with activity; not reproducible |
Atrial fibrillation mimics are often caused by tremors or shivering, which introduce irregular, high-frequency oscilla
Atrial flutter (Figure 4A) mimics often results from rhythmic external oscillations such as electrocautery or ventilators. These produce saw-tooth baseline deflections, especially in the inferior leads, and can be mistaken for flutter waves. In genuine flutter, atrial activity remains consistent across all leads and shows a consistent atrioventricular conduction pattern (for example, 2:1, 3:2 atrioventricular conduction, etc.). Conversely, artifacts tend to be localized and lack physio
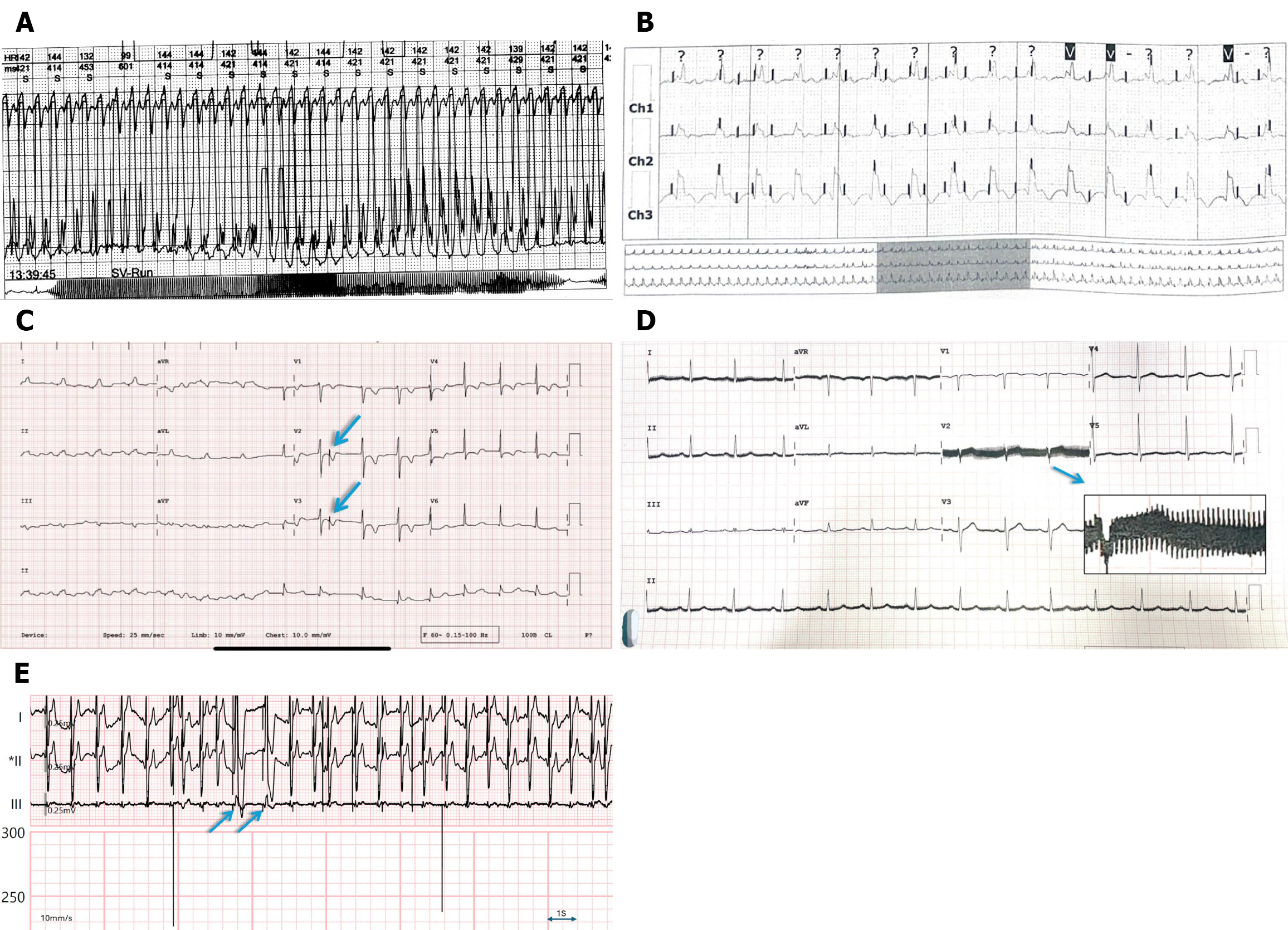
VT (Figures 4B and 8A) mimics are among the most concerning pitfalls. Large, rapid oscillations caused by gross body movement, shivering, or loose lead cables can resemble wide-complex tachycardia. These may appear chaotic or polymorphic, raising concerns of malignant arrhythmia. Clues to identification include the apparent “rate”, which often exceeds 400-600 beats per minute - much faster than true VT, which rarely exceeds 250-300 bpm. Furthermore, the patient’s true rhythm may show faintly visible QRS complexes “marching through” the artifact. Most importantly, patients are often alert and hemodynamically stable, a scenario incompatible with genuine sustained VT. Case reports describe near-interventions caused by such artifacts, highlighting the need for vigilance.
Ventricular fibrillation mimics are another critical danger, as VFs require immediate defibrillation (Figure 4B). Artifacts caused by electrode detachment, tremor, or electrical interference can create a chaotic baseline that looks like fibrillation. Recognizing this involves checking the patient: A conscious, talking patient is not in VF. Simultaneous monitoring of SpO2 or arterial pressure usually reveals an organized rhythm. Additionally, VF artifacts often spare some leads, unlike true ventricular fibrillation, which affects all leads.
ST-segment elevation can mimic true changes due to baseline drift, pulse tapping of the electrodes, poor electrode-skin contact, or respiratory motion (Figures 4C and 7B). These artifacts cause apparent ST elevation or depression and may be mistaken for acute myocardial infarction. The inconsistency of ST changes between beats and across leads is a key diagnostic clue, whereas genuine infarction tends to produce stable and contiguous lead alterations. Simply repreparing the skin and repeating the ECG can eliminate artifacts and avoid unnecessary interventions such as thrombolysis or catheterization. Pacemaker artifact misinterpretation occurs because pacing spikes are often exaggerated or distorted by filters. Tall or broad spikes may be mistaken for fibrillation or electrical noise. Recognition requires awareness of the pacing mode: Spikes occurring at predictable intervals in relation to atrial or ventricular events are almost always pacing artifacts rather than pathology (Figure 8B and C).
Wearable and Holter monitors add another layer of complexity. Movement, sweat, and poor adhesion often cause baseline drift, false pauses, or spurious tachyarrhythmias. For example, electrode detachment can imitate high-grade atrioventricular block, whereas exercise-related noise may be mistaken for tachycardia. These artifacts can lead to unnecessary referrals for pacemaker implantation if tracings are not reviewed carefully and in context.
Finally, pediatric ECGs are especially prone to artifacts (Figure 5C). Children’s frequent movements, crying, or shi
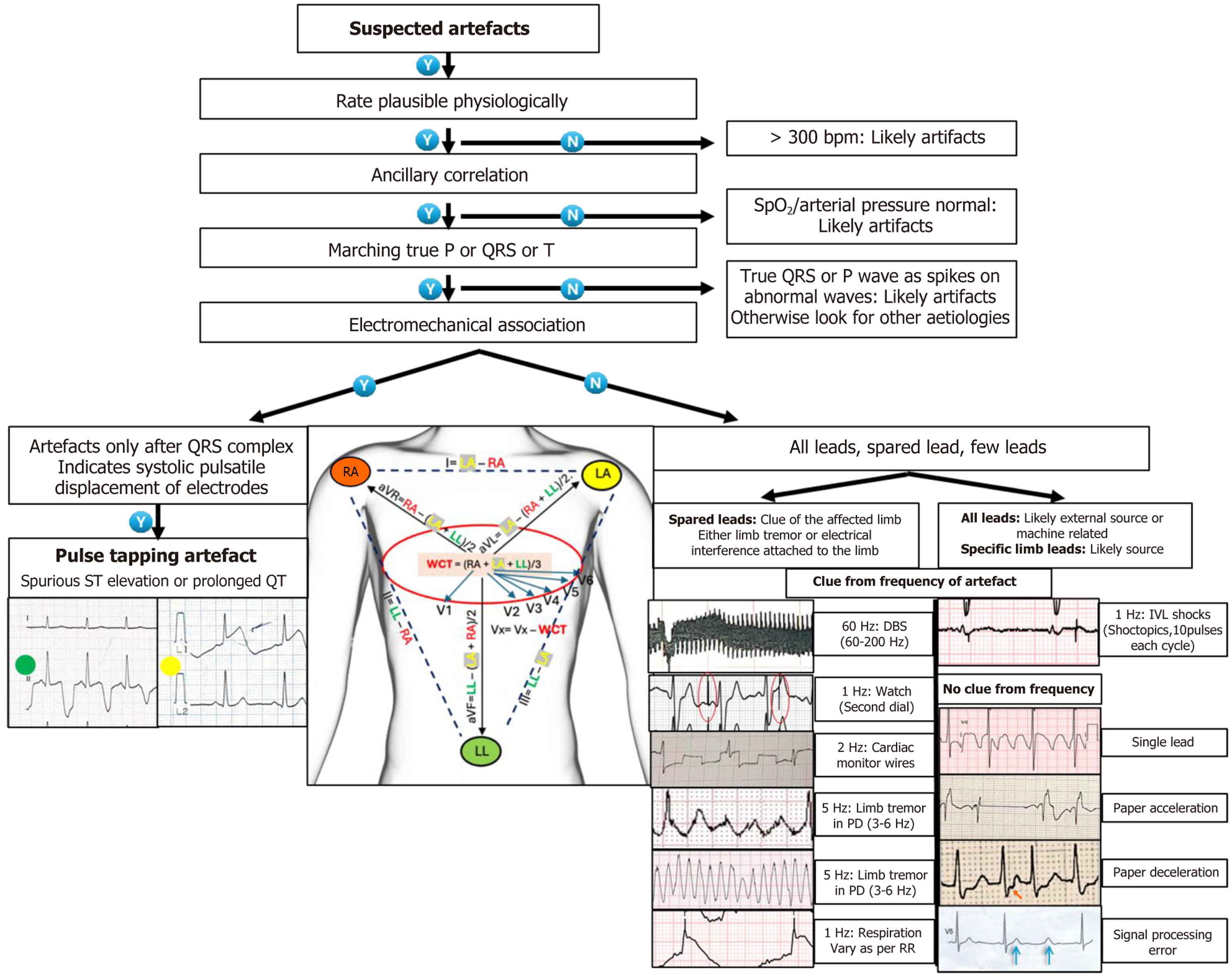
Recognizing ECG artifacts requires clinical judgment rather than simple pattern detection. Machines and algorithms cannot reliably differentiate artifacts from true arrhythmias, especially when they overlap, so the clinician bears responsibility. A structured, step-by-step method ensures consistency, whether at the bedside, in the ICU, or when reviewing Holter and wearable tracings (Figure 10). The initial and most vital step is to assess the patient before analyzing the ECG. A tracing that seems to indicate ventricular fibrillation might actually be an artifact if the patient is awake, talking, and has a normal pulse and blood pressure. The principle is to assess physiologic plausibility. Human conduction has specific limits: Sinus rates rarely exceed 220 beats per minute in adults, atrioventricular conduction usually does not exceed 250-300 beats per minute, and VT above 300 beats per minute is practically impossible. Apparent ventricular “rates” of 400-600 beats per minute, which are often caused by artifacts, are outside human physiological capabilities and therefore cannot reflect a true rhythm. Bedside correlation with clinical status remains the quickest and most dependable method to avoid disastrous misinterpretation.
The next step involves carefully examining the baseline, which often reveals artifacts before the QRS complexes do. A wandering baseline suggests respiration or gradual electrode drift. Sudden baseline jumps indicate poor electrode contact or static discharge. A regular saw-tooth oscillation strongly points to power-line interference, whereas coarse, rhythmic oscillations synchronized with patient shivering indicate tremor artifacts. The next step is to identify true P/QRS/T complexes. In an artifact, genuine P/QRS/T complexes often “march through” the noise with regular intervals (irregular in concomitant atrial fibrillation or other irregular atrial arrhythmias), whereas in true arrhythmia, the baseline and QRS morphology change together. In true monomorphic VT, the interval between successive QRS complexes (the R-R interval) is typically regular or nearly regular. In movement artifacts mimicking VT, the “R-R” intervals of artifactual deflections are often grossly irregular. Recognition of underlying QRS complexes has saved many patients from unnecessary interventions; for example, apparent wide-complex tachycardia was later recognized as a tremor artifact once regular QRS complexes were detected beneath the oscillations.
The next step is to evaluate for electromechanical associations. Arterial pulse tapping artifacts, also known as elec
Finally, the artifacts can be seen as additional findings in the ECG, as a distortion of the existing and expected wave forms or, rarely, may appear as a deletion of the expected wave forms. The initial step would be timing the suspected artifact with the cardiac cycle in the case of repetitive artifacts. Noting whether the artifact is high-frequency vs low-frequency helps, since the causes differ. Examples of high-frequency artifacts include attenuation correction artifacts, myopotential artifacts, electromagnetic interference, and phantom pacing spikes. Examples of low-frequency artifacts are baseline drift, shaking, and arterial pulse tap artifacts. Artifactual deletion of expected waveforms or pacing spikes is more often observed in analog-type monitors than in usual ECGs. Artifactual distortion of waves is also common in such systems and may affect ST analysis. This is the major reason that ICU monitors are not preferred for ST-segment analysis. In analog systems, artifacts are a more significant challenge because filters are hardware-based and less sophisticated than modern digital signal processing algorithms. Correcting an artifact often requires retaking the entire ECG physically, as post-processing options are limited or nonexistent. Accurate interpretation relies heavily on the skill of the technician in acquiring a clean signal and the clinician in visually distinguishing artifacts from genuine cardiac pathology.
Artifacts are not merely nuisances but also preventable sources of error in ECG interpretation. A structured prevention strategy not only saves time and avoids misdiagnosis but also reduces the risk of unnecessary interventions. The prin
At the patient level, several strategies are important. Skin preparation should be meticulous: The skin must be clean and dry to reduce impedance, excessive chest hair should be shaved, and electrodes should be placed away from bony prominences or areas prone to sweating. Patient comfort also plays a role; warming a shivering patient, encouraging muscle relaxation, and minimizing talking or movement during a standard 12-lead recording all reduce noise. Proper positioning can further stabilize the tracing - supporting tremulous limbs - and using pediatric-sized electrodes in chi
At the equipment level, the focus is on electrodes, cables, and machines. Fresh electrodes should always be used, as dried gel increases impedance and noise, and firm adhesion must be ensured. In the ICU and operating theatre, patients undergoing continuous monitoring, the electrodes should be replaced regularly. Cables and leads should be inspected for cracks, fractures, or stretching and secured to prevent tugging or accidental dislodgement. ECG machines must be maintained with appropriate calibration of filters; overfiltering should be avoided, and “unfiltered” displays should be reviewed when necessary to preserve diagnostic clarity. Electrical grounding and routine service are equally important. Environmental measures also contribute to artifact prevention. ECG cables should be kept separate from power cords and electrocautery units, and unnecessary electrical devices should be switched off during recording. In the operating theatre, tracings are best obtained during pauses in electrocautery, and shielded cables may be employed in environments with high electromagnetic interference.
When artifacts do occur, a simple troubleshooting algorithm provides the following structure: First, check the patient and determine whether they are stable or unstable; second, check the electrodes, reposition them and reprepare the skin if needed; third, inspect leads and cables, replace them if suspicious; fourth, check for environmental interference; fifth, record them after corrections; and sixth, crosscheck with ancillary signals such as SpO2 or arterial line tracings. Staff education is the cornerstone of prevention. A formalized “prerecording checklist”, encompassing verification of skin preparation, proper electrode placement, cable integrity, and patient immobility, should be implemented to prevent the formation of ECG artifacts, thereby significantly abating the chain of diagnostic tests. Nurses, technicians, and residents must be trained to recognize artifact patterns and apply simple but critical rules: Never shock a talking patient, always check electrode contact before diagnosing arrhythmia, and always correlate the ECG with pulse oximetry or arterial wav
Despite significant technological advances, distinguishing artifacts from genuine arrhythmia remains a major challenge for automated systems. While digital filtering and AI provide promising solutions, human clinical judgment remains essential. Digital filtering techniques have advanced significantly. Adaptive filters now adjust automatically to changes in the baseline, reducing interference without the excessive smoothing typical of fixed filters. Signal averaging methods can also diminish repetitive noise patterns, improving the clarity of tracings while maintaining clinically relevant infor
AI approaches represent the most exciting frontier. Large datasets, such as those from the United Kingdom Biobank and Chronic Renal Insufficiency Cohort projects, comprising more than 100000 ECGs, have been used to train models specifically for artifact detection. These systems have achieved sensitivity and specificity rates exceeding 90% in distinguishing physiologic from nonphysiologic signals[14]. The major advantage of AI is its ability to rapidly flag poor-quality recordings, thereby saving clinicians valuable time. However, limitations remain: Current models cannot reliably detect electrode misplacement, and they cannot fully replace human interpretation when arrhythmia and artifacts overlap. Thus, AI can assist but not yet supplant the clinician. In the future, AI integration could revolutionize routine monitoring. Bedside monitors might soon provide real-time alerts for suspected artifacts, Holter recorders may automatically mark noisy segments for later inspection, and wearable devices might even prompt users to reposition electrodes when signal quality declines. These advancements have the potential to increase diagnostic accuracy and minimize misinterpretation, especially in busy or resource-constrained environments.
Although artifacts can never be completely eliminated, future developments aim to make their management easier and more reliable. Advances in smarter electrodes, such as self-adhesive patches that continuously monitor skin impedance and generate alerts when contact is poor, could significantly reduce electrode-related noise. Improvements in wearable technology, including sweat-resistant materials and stronger adhesives, may improve the reliability of long-term ambulatory recordings. At the system level, integrating machine learning into clinical monitors will likely become standard, providing real-time artifact detection and prompting corrective actions. In addition to technology, simulation-based training is crucial. Virtual ECG simulators that expose trainees to common artifact patterns alongside genuine arrhythmias can enhance diagnostic skills and reduce misinterpretation in clinical practice. Indeed, such simulators closely align with current educational projects, which aim to bridge the gap between engineering and clinical cardiology.
ECG artifacts are common challenges in clinical practice and are capable of mimicking arrhythmias and ischemia, leading to unnecessary interventions. They occur because the cardiac signal is fragile and easily disrupted during recording. Recognition relies on logic: Always check the patient, assess physiological plausibility, analyze lead distribution and baseline behavior, and confirm with ancillary signals. Prevention depends on careful electrode preparation, equipment maintenance, environmental control, and staff training. While digital filtering and AI offer new tools, clinical reasoning remains essential. The key message is simple: Never treat the ECG until you have checked the patient. Always remember, “The brain filters what machines cannot: Patient safety demands it”.
All authors contributed equally to this manuscript. We thank Dr. P Hema Krishna (neurologist) and Dr. Stalin for providing the deep-brain stimulator artifact and the square wave artifact electrocardiograms, respectively. We thank our Cath-lab staff for their unwavering technical support while collecting the best possible images. We thank Mr. Salehin Anjum for making the audio clip for the Core Tip.
| 1. | Littmann L. Electrocardiographic artifact. J Electrocardiol. 2021;64:23-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 2. | Pérez-Riera AR, Barbosa-Barros R, Daminello-Raimundo R, de Abreu LC. Main artifacts in electrocardiography. Ann Noninvasive Electrocardiol. 2018;23:e12494. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 35] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 3. | Imhoff B, Casey W. An Unusual ECG Artifact Caused by Faulty Cardiac Monitor Leads. Clin Pract Cases Emerg Med. 2021;5:267-269. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 4. | DeLeon AM, Weeks JJ, Su L, Garcia Tomas V. Two Cases of Intraoperative ECG Artifact Mimicking Ventricular Tachycardia and How to Know When It Is Real. Cureus. 2023;15:e38773. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 5. | Avidan Y, Ali J, Aker A, Sliman H. Gone with the lead: the arterial pulse-tapping artifact-a systematic review of case reports. Intern Emerg Med. 2025;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Shanmugam S, Mondal S, Vijay J. Question: Syncope and chest discomfort on flecainide: a diagnostic dilemma. Eur Heart J Acute Cardiovasc Care. 2024;13:810-811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 7. | Shanmugam S, Mondal S, Vijay J. Answer: Syncope and chest discomfort on flecainide: a diagnostic dilemma. Eur Heart J Acute Cardiovasc Care. 2024;13:812-813. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 8. | Matthias AT, Indrakumar J. Electrocardiogram artifact caused by rigors mimicking narrow complex tachycardia: a case report. BMC Res Notes. 2014;7:80. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 9. | Y-Hassan S, Sylvén C. Electrocardiographic artefacts mimicking atrial tachycardia resulted in unnecessary diagnostic and therapeutic measures. Korean J Intern Med. 2013;28:224-230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 10. | Tinati MA, Mozaffary B. A wavelet packets approach to electrocardiograph baseline drift cancellation. Int J Biomed Imaging. 2006;2006:97157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 11. | Luo Y, Hargraves RH, Belle A, Bai O, Qi X, Ward KR, Pfaffenberger MP, Najarian K. A hierarchical method for removal of baseline drift from biomedical signals: application in ECG analysis. ScientificWorldJournal. 2013;2013:896056. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 20] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 12. | Aslanger E. An unusual electrocardiogram artifact in a patient with near syncope. J Electrocardiol. 2010;43:686-688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 13. | Mondal S, Raja DP, Vijay J. Bizarre Electrocardiogram in an Elderly Man. Indian J Clin Cardiol. 2024;5:75-77. [DOI] [Full Text] |
| 14. | Bukhari HA, Kewalramani S, Witzigreuter L, Pourbemany J, Barbato NA, Daw J, Dhar R, Rincon-Choles H, Rao P, Bhat Z, Soliman EZ, Tereshchenko LG; CRIC Study Investigators. Automated detection of non-physiological artifacts on ECG signal: UK Biobank and CRIC. Comput Biol Med. 2025;196:110787. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |