Published online Mar 26, 2026. doi: 10.4330/wjc.v18.i3.112189
Revised: November 6, 2025
Accepted: January 19, 2026
Published online: March 26, 2026
Processing time: 246 Days and 13.9 Hours
Obesity-related heart failure in patients is often associated with a high symptom burden. However, no treatments have been proven to specifically target obesity-related heart failure with preserved ejection fraction (HFpEF).
To evaluate the efficacy of semaglutide in patients with obesity and HFpEF.
Per the PRISMA guidelines, studies reporting clinical outcomes of semaglutide in patients with obesity and HFpEF were included. The outcomes included percen
In total, three studies with 1463 patients with obesity and HFpEF were included in the study. The mean age of the patients was 68.8 ± 3.47 years. 50.7% of the patients were females. Patients who received semaglutide had statistically higher odds of achieving 10% (OR = 6.35; 95%CI: 1.54-26.21; P < 0.00001) and 15% (OR = 9.44; 95%CI: 2.91-30.60; P < 0.0001) weight reductions compared to those who received placebo. Additionally, patients who received semaglutide had lower odds of adju
Our study demonstrates that semaglutide is significantly effective in reducing weight and potentially lowering the risk of heart failure events. This suggests that semaglutide could be a promising therapeutic option for managing obesity-related HFpEF.
Core Tip: This meta-analysis demonstrates that semaglutide is effective in reducing weight and potentially lowering the risk of heart failure events in patients with obesity and heart failure with preserved ejection fraction (HFpEF). This suggests semaglutide as a promising therapeutic option for managing obesity-related HFpEF, a condition with limited current treatment options. The findings showed statistically higher odds of 10% and 15% weight reduction, as well as lower odds of adjudicated heart failure events, in patients receiving semaglutide compared to those receiving placebo. Further large-scale trials are needed to confirm these benefits and explore long-term outcomes.
- Citation: Mylavarapu M, Obi O, Abarca Y, Fatima H, Roshni P, Huda NU, Lysak Y, Gandapur A, Vazquez SC, Siddiqui MA, Mowo-Wale A. Semaglutide in patients with obesity and heart failure with preserved ejection fraction: A systematic review and meta-analysis. World J Cardiol 2026; 18(3): 112189
- URL: https://www.wjgnet.com/1949-8462/full/v18/i3/112189.htm
- DOI: https://dx.doi.org/10.4330/wjc.v18.i3.112189
Heart failure with preserved ejection fraction (HFpEF) represents a significant and growing clinical challenge, characterized by its substantial prevalence and complex diagnostic criteria, leading to delayed identification and notable difficulties in effective management[1-3]. Unlike heart failure with reduced ejection fraction (HFrEF), mana
Obesity enables and fosters chronic systemic inflammation and, in turn, dysregulates the secretion of adipokines, ultimately contributing to cardiovascular dysfunction[8,9]. Epicardial adipose tissue releases paracrine factors that can directly impact cardiac cells[10]. Obesity is also implicated in the development of left ventricular diastolic dysfunction and adverse cardiac remodeling, increases both preload and afterload on the heart, and promotes endothelial dysfunction and microvascular disease[7,8,11]. Furthermore, obesity significantly increases the risk of obstructive sleep apnea, chronic kidney disease, metabolic syndrome, hypertension, diabetes, and dyslipidemia[12-17], all of which are known risk factors for the development of coronary artery disease, which in turn is a predisposing factor for heart failure[8]. Studies have reported obesity-related HFpEF as a clinically relevant phenotype, a genuine form of cardiac failure[18,19].
However, despite the interplay between obesity and HFpEF, there exists a significant gap in management, with a lack of evidence-based therapies specifically designed to target this complex interplay and improve outcomes in this vul
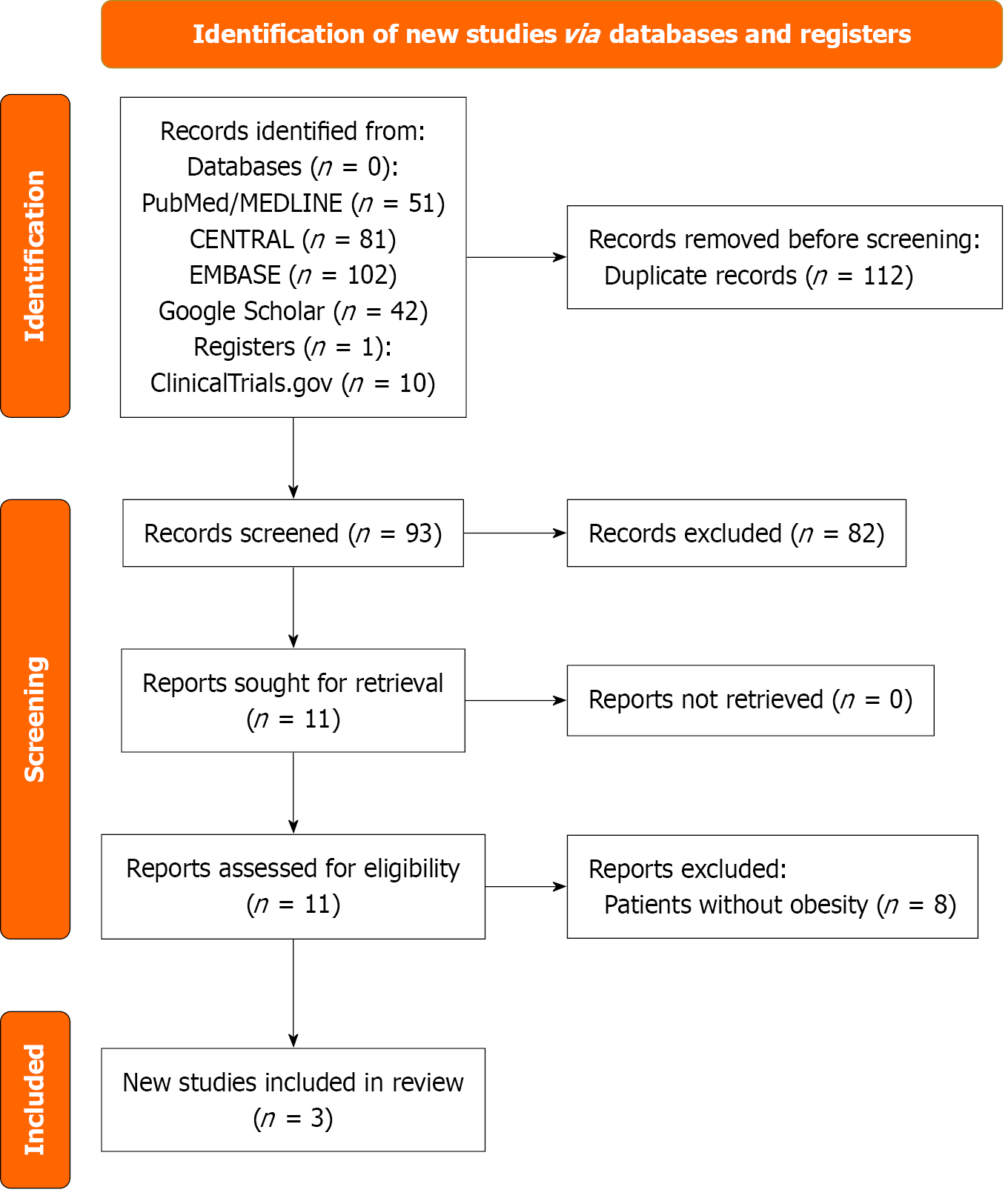
Per the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines[39], a comprehensive literature search was conducted in several prominent, reliable databases, including PubMed/MEDLINE, Cochrane Central Register of Controlled Trials, EMBASE, Google Scholar, and ClinicalTrials.gov. Subject headings and keywords for “semaglutide”, “obesity”, “heart failure”, and “heart failure with preserved ejection fraction” were used along with appropriate Boolean operators. The references of the selected studies were also examined to verify the comprehensiveness of the search. The search strategy utilized for the study is outlined in Supplementary Table 1. Studies reporting clinical outcomes of semaglutide in patients with obesity and HFpEF were included. A detailed list of inclusion and exclusion criteria was outlined in Supplementary Table 2. Screening of the title and abstract was done independently by two reviewers, Vazquez SC and Obi O, and conflicts were resolved by a third reviewer, heart failure. Full-text screening was done independently by two reviewers, Huda NU and Lysak Y, and conflicts were resolved by a third reviewer, Mylavarapu M. Figure 1 depicts the PRISMA flow chart outlining the study selection process[40].
The risk of bias assessment was conducted using the Cochrane Risk of Bias tool[41] for randomized control trials and the adaptation of the New Castle Ottawa Scale[42] for observational study. Primary endpoints include weight reduction and adjudicated heart failure events (suspected clinical outcome due or because of heart failure). Secondary endpoints include adverse events, namely all-cause mortality (ACM), cardiac arrhythmias, and infections. Both random and common effects models were used for the data analysis. A random-effects model (DerSimonian-Laird) was applied for outcomes with anticipated significant heterogeneity, such as weight reduction. The Peto method, a fixed-effect model, was utilized for analyzing rare dichotomous outcomes, specifically adjudicated heart failure events, as it minimizes bias when event rates are low. Heterogeneity was assessed using the χ2 test and I2 statistics. The I2 statistic was interpreted as follows: 25% (low heterogeneity), 50% (moderate heterogeneity), and 75% (high heterogeneity). Visualization interpretation of funnel plots was utilized to evaluate for publication bias. All the statistical analyses were performed using the Review Manager (RevMan) version 5.4.1[43]. A P value < 0.05 was considered to be statistically significant. The protocol of the study was registered in the open science framework registry (Id: OSF.IO/K6HZV).
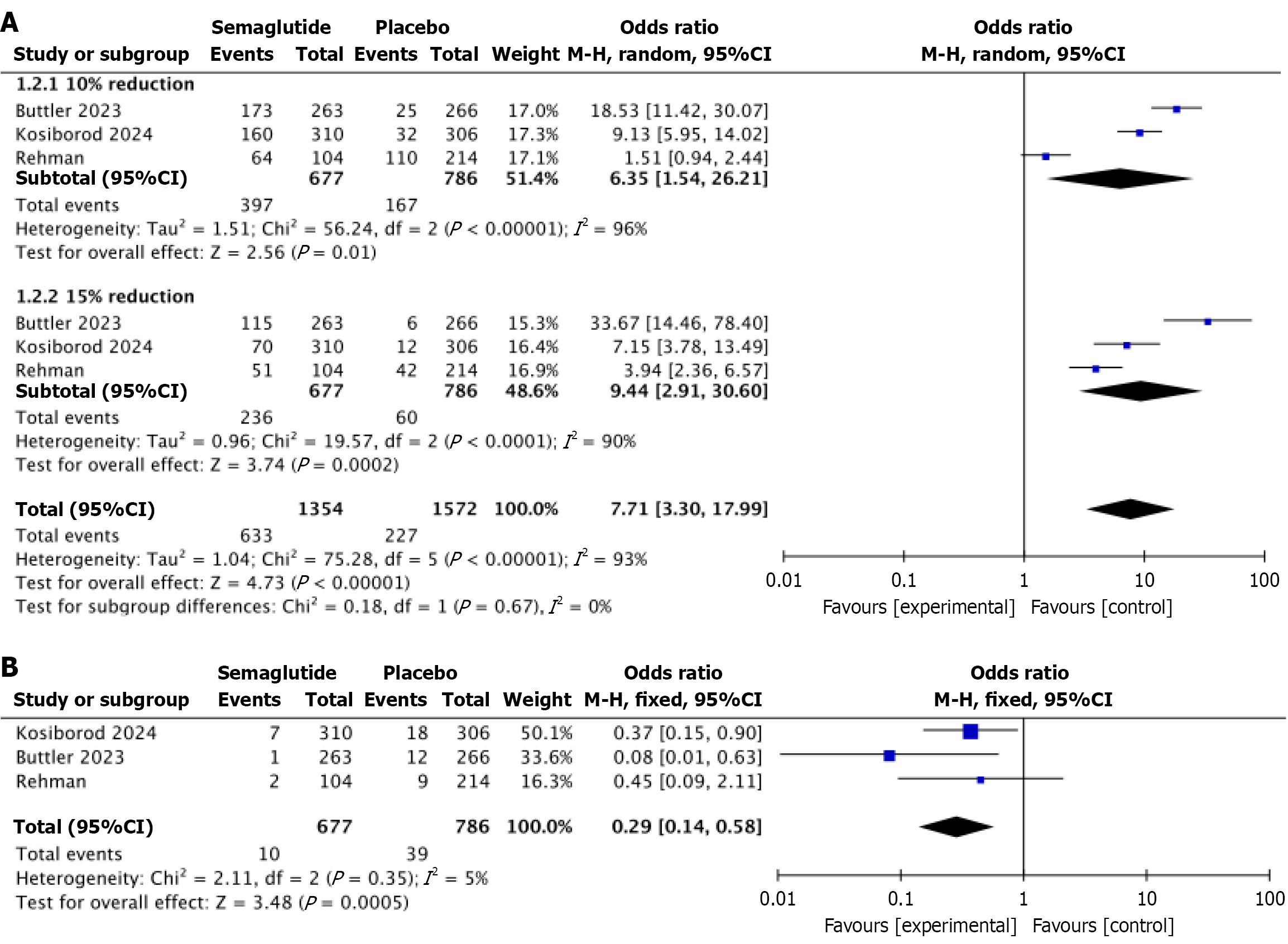
A total of three studies with 1463 patients with obesity and HFpEF[44-46]. Table 1 outlines the key characteristics of the included studies. The mean age of the patients was 68.8 ± 3.47 years, with 50.7% being female. Table 2 outlines the baseline characteristics of the included population. Regarding primary endpoints, patients who received semaglutide had significantly higher odds of weight reduction [odds ratio (OR) = 7.71; 95%CI: 3.30-17.99; P < 0.00001] compared to those who received placebo (Figure 2A). When sub-grouped, patients who received semaglutide had significantly higher odds in both 10% weight reduction (OR = 6.35; 95%CI: 1.54-26.21; P = 0.01) and 15% weight reduction (OR = 9.44; 95%CI: 2.91-30.60; P = 0.0002). Furthermore, patients who received semaglutide had significantly lower odds of adjudicated heart failure events (OR = 0.29; 95%CI: 0.14-0.58; P = 0.0005) (Figure 2B).
| Ref. | Study design | Population characteristics | Type of HF | Intervention | Control |
| Kosiborod et al[44], 2024 | RCT (STEP-HFpEF DM) | O with HF and DM-II | HFpEF | Semaglutide (2.4 mg) | Placebo |
| Butler et al[45], 2023 | RCT (STEP-HFpEF) | O with HF | HFpEF | Semaglutide (2.4 mg) | Placebo |
| Rehman et al[46], 2024 | Retrospective | O with HF | HFpEF | Semaglutide (2.4 mg) | Placebo |
| Ref. | mean ± SD | Total patients | Number of females | BMI | LVEF | Hypertension | CAD | ||||
| Semaglutide | Placebo | Semaglutide | Placebo | Semaglutide | Placebo | Semaglutide | Placebo | ||||
| Kosiborod et al[44] | 69.0 ± 3.49 | 616 | 273 | 36.9 (33.6-41.5) | 36.9 (33.5-41.1) | 57.0 (50.0-61.0) | 55.0 (50.0-60.0) | 255 (82.3) | 271 (88.6) | 79 (25.5) | 69 (22.5) |
| Butler et al[45] | 68.5 ± 3.45 | 529 | 297 | 37.2 (33.9-41.1) | 36.9 (33.3-41.6) | 57.0 (50.0-60.0) | 57.0 (50.0-60.0) | 216 (82.1) | 217 (81.6) | 53 (20.2) | 45 (16.9) |
| Rehman et al[46] | 69.0 ± 3.47 | 318 | 172 | 37.2 (33.9-41.1) | 36.9 (33.3-41.6) | 57.0 (50.0-60.0) | 57.0 (50.0-60.0) | 82 (78.8) | 163 (76.2) | 23 (22.1) | 47 (22.0) |
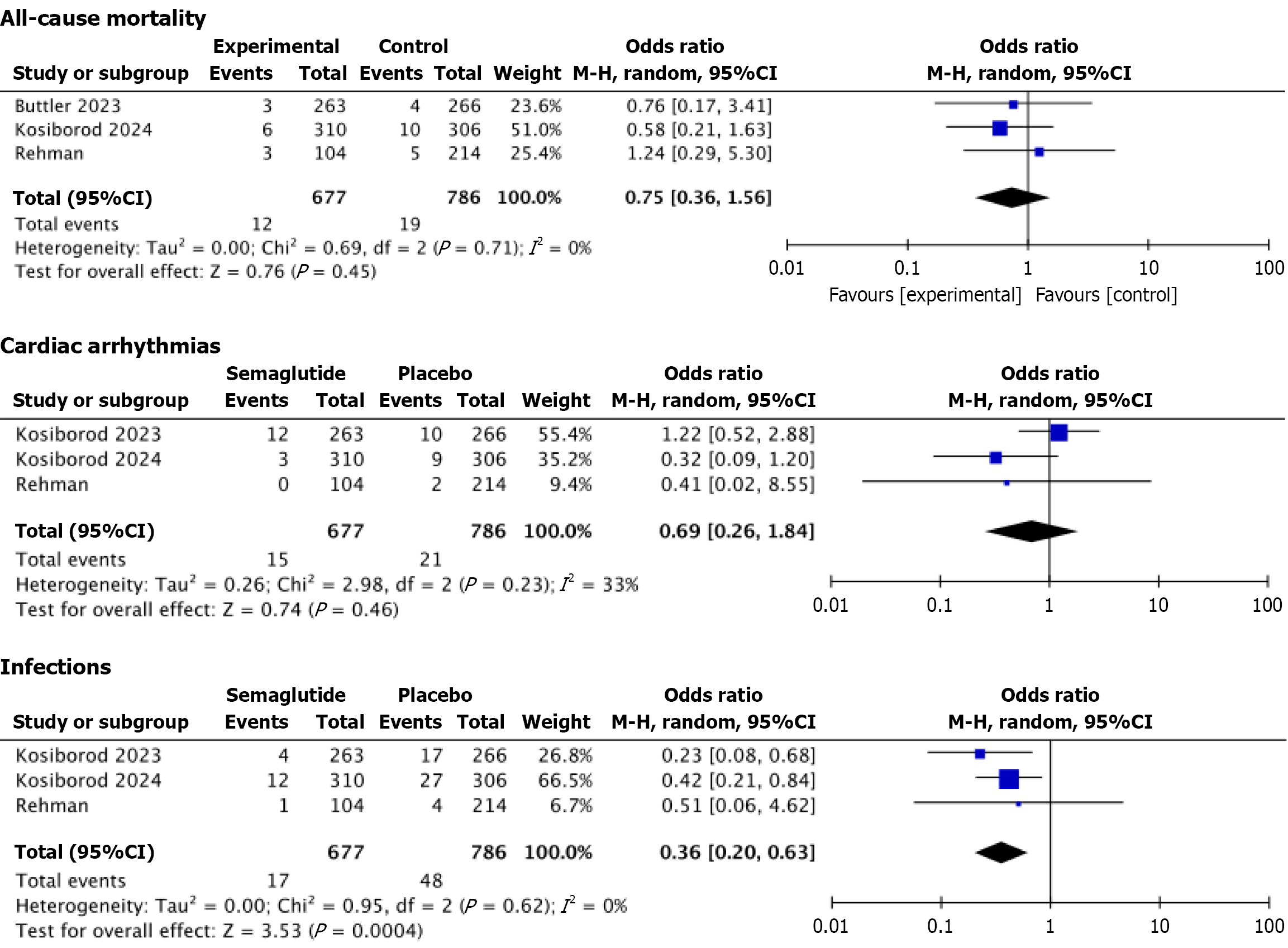
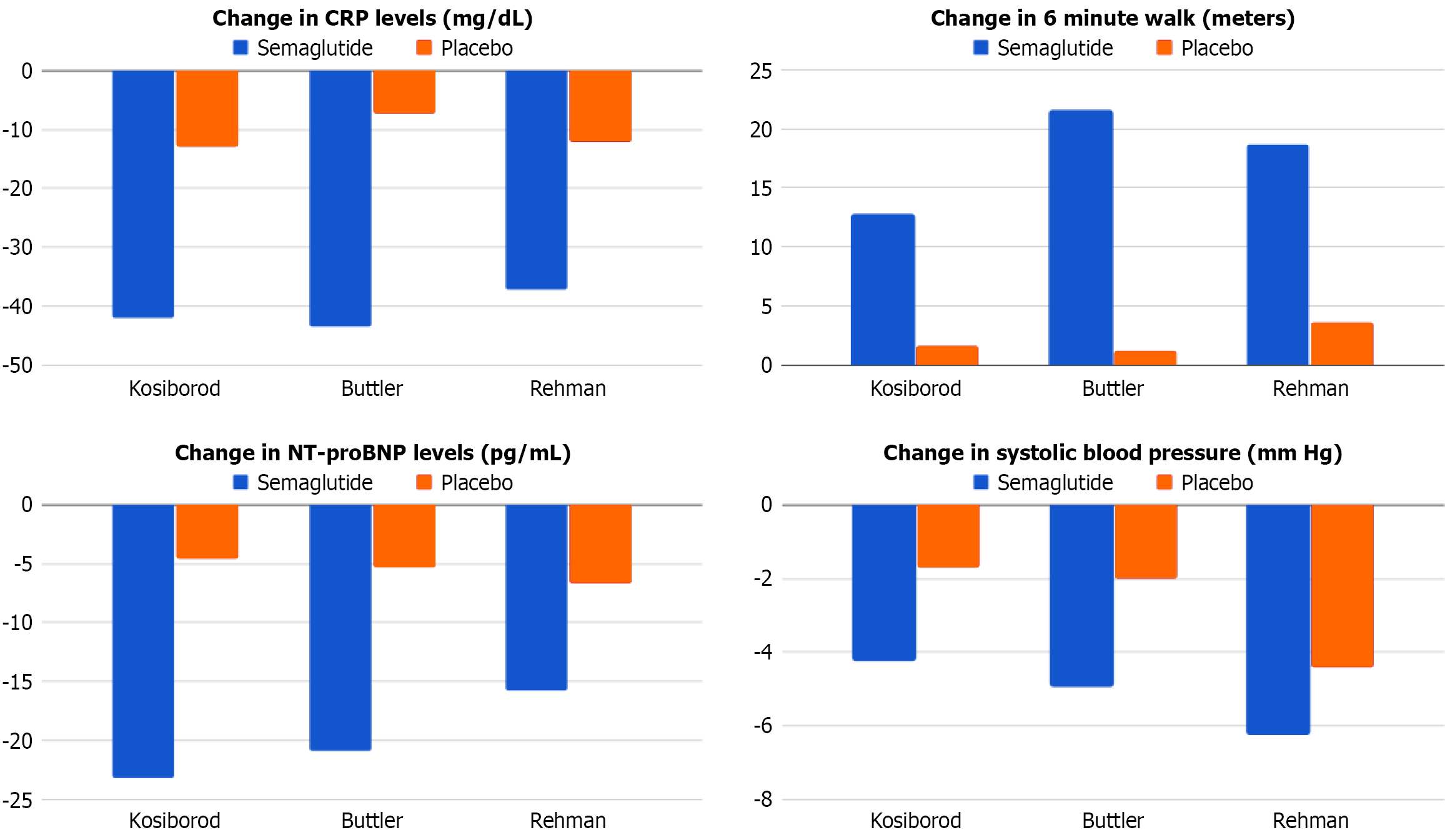
Regarding adverse events, the semaglutide group had significantly lower odds of infections (OR = 0.36; 95%CI: 0.20-0.63; P = 0.0004). Although ACM (OR = 0.75; 95%CI: 0.36-1.56; P = 0.45) and cardiac arrhythmias (OR = 0.69; 95%CI: 0.26-1.84; P = 0.46) were also lower, they weren’t significant (Figure 3). Additionally, patients who received semaglutide had a higher frequency of other clinical and laboratory outcomes, including change in systolic blood pressure, change in 6-minute walking distance, and change in C-reactive protein (CRP) (Figure 4).
Overall, the risk of bias assessment indicates that while one randomised controlled trial (RCT) was of high quality, the other RCT and the observational study had some methodological limitations (Supplementary Table 3). Regarding heterogeneity, the primary endpoints showed significant heterogeneity (Supplementary Table 4). Leave-one-out sensitivity analysis, performed to investigate high heterogeneity in weight reduction, revealed that removing the Rehman et al[46] study reduced I2 from 96% to 70% for the 10% weight reduction outcome, while removing the Butler et al[45] study reduced I2 from 90% to 52% for the 15% weight reduction outcome (Supplementary Table 5). Publication bias is insignificant (Supplementary Figure 1).
This systematic review and meta-analysis, the first-ever meta-analysis to evaluate the efficacy of semaglutide in patients with obesity and HFpEF, demonstrates that semaglutide is associated with a significant increase in the odds of weight reduction, including both 10% and 15% weight reduction thresholds, when compared to placebo. The observed weight reduction with semaglutide is consistent with findings from previous studies that have established its efficacy in pro
The finding that semaglutide is associated with reducing adjudicated heart failure events is particularly noteworthy. HFpEF is a complex and challenging condition with limited effective treatment options[51]. As shown by previous research, while medications like angiotensin-converting enzyme inhibitors[52], ARBs[53,54], and beta-blockers have shown significant benefits in treating HFrEF, their impact on HFpEF has been limited[55]. The Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist trial demonstrated that spironolactone reduced hospitalizations in HFpEF patients but did not significantly improve mortality or morbidity[55,56], highlighting the need for effective interventions to reduce heart failure events in this population. The reduction in adjudicated heart failure events is clinically significant, as these events often lead to hospitalization, reduced functional capacity, and a decline in overall quality of life, which could potentially lead to increased mortality[57-60].
The impact of GLP-1RA on heart failure has always been debated. Ferreira et al[61], in their meta-analysis of RCTs, reported that although GLP-1RAs did not reduce the mortality due to heart failure and heart failure-based hospitalizations in patients with a history of heart failure, they significantly prevented new-onset heart failure in patients with type 2 diabetes without heart failure. Notably, the meta-analysis did not discuss HFpEF and HFrEF separately, nor did it assess the effects in obese patients. However, new clinical trials report evidence supporting the use of GLP-1RAs in patients with heart failure, especially HFpEF. The functional impact of GLP-1 RA for Heart Failure Treatment trial[62] reports that patients with HFpEF have improved exercise capabilities and quality of life in the liraglutide group com
Several limitations of our meta-analysis should be acknowledged. First, the number of included studies was small (n = 3), limiting the statistical power of our analysis and the generalizability of the findings. Second, while one RCT was of high quality, another RCT and the observational study had some methodological limitations, which may introduce bias. Third, significant heterogeneity was observed for the primary endpoints related to weight reduction. This variability, particularly in terms of study populations, intervention protocols, and outcome definitions, resulting in wider confidence intervals, restricts our ability to synthesize the data more conclusively and limits the generalizability of the findings. Fourth, the included trials did not provide stratified data regarding background therapies, such as sodium-glucose cotransporter 2 (SGLT2) inhibitors, which prevented a subgroup analysis to assess for potential confounding or additive effects.
While these findings are promising, it is important to situate them within current clinical practice. SGLT2 inhibitors remain the first-line, guideline-directed therapy for HFpEF, regardless of diabetes or obesity status. Semaglutide is not yet guideline-approved specifically for HFpEF management, and its use in this context (outside of type 2 diabetes or weight loss indications) would be considered off-label.
Current guidelines[64] recommend SGLT2 inhibitors as the first line of management in patients with HFpEF due to a significant reduction in cardiovascular death or hospitalizations in patients with heart failure[65]. However, this meta-analysis provides preliminary evidence suggesting that semaglutide may be a promising therapeutic option for managing obesity-related HFpEF. The observed benefits of weight reduction and heart failure events warrant further investigation in larger, well-designed clinical trials. Future research should also explore the long-term effects of semaglutide on cardiovascular outcomes, quality of life, and mortality in this patient population. Furthermore, research is needed to elucidate the precise mechanisms by which semaglutide affects the heart in patients with HFpEF. Studies should investigate the potential role of inflammation, endothelial function, and cardiac remodeling in mediating the beneficial effects of semaglutide. Additionally, studies should also assess the cost-effectiveness of semaglutide in managing obesity-related HFpEF, considering both the direct costs of the medication and the potential cost savings associated with reduced hospitalizations and improved long-term outcomes.
This meta-analysis provides evidence that semaglutide is effective in promoting significant weight reduction and poten
| 1. | NHFA CSANZ Heart Failure Guidelines Working Group; Atherton JJ, Sindone A, De Pasquale CG, Driscoll A, MacDonald PS, Hopper I, Kistler PM, Briffa T, Wong J, Abhayaratna W, Thomas L, Audehm R, Newton P, O'Loughlin J, Branagan M, Connell C. National Heart Foundation of Australia and Cardiac Society of Australia and New Zealand: Guidelines for the Prevention, Detection, and Management of Heart Failure in Australia 2018. Heart Lung Circ. 2018;27:1123-1208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 316] [Article Influence: 39.5] [Reference Citation Analysis (0)] |
| 2. | Naing P, Forrester D, Kangaharan N, Muthumala A, Mon Myint S, Playford D. Heart failure with preserved ejection fraction: A growing global epidemic. Aust J Gen Pract. 2019;48:465-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 3. | Ma C, Luo H, Fan L, Liu X, Gao C. Heart failure with preserved ejection fraction: an update on pathophysiology, diagnosis, treatment, and prognosis. Braz J Med Biol Res. 2020;53:e9646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 52] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 4. | Reddy YNV, Melenovsky V, Redfield MM, Nishimura RA, Borlaug BA. High-Output Heart Failure: A 15-Year Experience. J Am Coll Cardiol. 2016;68:473-482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 266] [Cited by in RCA: 247] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 5. | Alpert MA. Obesity cardiomyopathy: pathophysiology and evolution of the clinical syndrome. Am J Med Sci. 2001;321:225-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 521] [Cited by in RCA: 506] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 6. | Avelar E, Cloward TV, Walker JM, Farney RJ, Strong M, Pendleton RC, Segerson N, Adams TD, Gress RE, Hunt SC, Litwin SE. Left ventricular hypertrophy in severe obesity: interactions among blood pressure, nocturnal hypoxemia, and body mass. Hypertension. 2007;49:34-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 158] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 7. | Kenchaiah S, Gaziano JM, Vasan RS. Impact of obesity on the risk of heart failure and survival after the onset of heart failure. Med Clin North Am. 2004;88:1273-1294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 82] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 8. | Ebong IA, Goff DC Jr, Rodriguez CJ, Chen H, Bertoni AG. Mechanisms of heart failure in obesity. Obes Res Clin Pract. 2014;8:e540-e548. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 205] [Cited by in RCA: 173] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 9. | Zorena K, Jachimowicz-Duda O, Ślęzak D, Robakowska M, Mrugacz M. Adipokines and Obesity. Potential Link to Metabolic Disorders and Chronic Complications. Int J Mol Sci. 2020;21:3570. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 280] [Article Influence: 46.7] [Reference Citation Analysis (0)] |
| 10. | Cho DH, Park SM. Epicardial Adipose Tissue and Heart Failure, Friend or Foe? Diabetes Metab J. 2024;48:373-384. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 23] [Article Influence: 11.5] [Reference Citation Analysis (2)] |
| 11. | Wong CY, O'Moore-Sullivan T, Leano R, Byrne N, Beller E, Marwick TH. Alterations of left ventricular myocardial characteristics associated with obesity. Circulation. 2004;110:3081-3087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 474] [Cited by in RCA: 493] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 12. | Poirier P, Després JP. Waist circumference, visceral obesity, and cardiovascular risk. J Cardiopulm Rehabil. 2003;23:161-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 96] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 13. | Poirier P, Giles TD, Bray GA, Hong Y, Stern JS, Pi-Sunyer FX, Eckel RH; American Heart Association; Obesity Committee of the Council on Nutrition, Physical Activity, and Metabolism. Obesity and cardiovascular disease: pathophysiology, evaluation, and effect of weight loss: an update of the 1997 American Heart Association Scientific Statement on Obesity and Heart Disease from the Obesity Committee of the Council on Nutrition, Physical Activity, and Metabolism. Circulation. 2006;113:898-918. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2280] [Cited by in RCA: 1908] [Article Influence: 95.4] [Reference Citation Analysis (3)] |
| 14. | Lavie CJ, Milani RV, Ventura HO. Obesity and cardiovascular disease: risk factor, paradox, and impact of weight loss. J Am Coll Cardiol. 2009;53:1925-1932. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1626] [Cited by in RCA: 1522] [Article Influence: 89.5] [Reference Citation Analysis (4)] |
| 15. | Russell GV, Pierce CW, Nunley L. Financial implications of obesity. Orthop Clin North Am. 2011;42:123-127, vii. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 16. | Hammond RA, Levine R. The economic impact of obesity in the United States. Diabetes Metab Syndr Obes. 2010;3:285-295. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 202] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 17. | Wang Y, Chen X, Song Y, Caballero B, Cheskin LJ. Association between obesity and kidney disease: a systematic review and meta-analysis. Kidney Int. 2008;73:19-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 543] [Cited by in RCA: 484] [Article Influence: 26.9] [Reference Citation Analysis (3)] |
| 18. | Obokata M, Reddy YNV, Pislaru SV, Melenovsky V, Borlaug BA. Evidence Supporting the Existence of a Distinct Obese Phenotype of Heart Failure With Preserved Ejection Fraction. Circulation. 2017;136:6-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1002] [Cited by in RCA: 903] [Article Influence: 100.3] [Reference Citation Analysis (0)] |
| 19. | Kitzman DW, Lam CSP. Obese Heart Failure With Preserved Ejection Fraction Phenotype: From Pariah to Central Player. Circulation. 2017;136:20-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 68] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 20. | Mahapatra MK, Karuppasamy M, Sahoo BM. Semaglutide, a glucagon like peptide-1 receptor agonist with cardiovascular benefits for management of type 2 diabetes. Rev Endocr Metab Disord. 2022;23:521-539. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 92] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 21. | Knudsen LB, Lau J. The Discovery and Development of Liraglutide and Semaglutide. Front Endocrinol (Lausanne). 2019;10:155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 804] [Cited by in RCA: 663] [Article Influence: 94.7] [Reference Citation Analysis (5)] |
| 22. | Christou GA, Katsiki N, Blundell J, Fruhbeck G, Kiortsis DN. Semaglutide as a promising antiobesity drug. Obes Rev. 2019;20:805-815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 113] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 23. | Kalas MA, Galura GM, McCallum RW. Medication-Induced Gastroparesis: A Case Report. J Investig Med High Impact Case Rep. 2021;9:23247096211051919. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 24. | Fonseca VA, Capehorn MS, Garg SK, Jódar Gimeno E, Hansen OH, Holst AG, Nayak G, Seufert J. Reductions in Insulin Resistance are Mediated Primarily via Weight Loss in Subjects With Type 2 Diabetes on Semaglutide. J Clin Endocrinol Metab. 2019;104:4078-4086. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 25. | Nauck MA, Meier JJ, Cavender MA, Abd El Aziz M, Drucker DJ. Cardiovascular Actions and Clinical Outcomes With Glucagon-Like Peptide-1 Receptor Agonists and Dipeptidyl Peptidase-4 Inhibitors. Circulation. 2017;136:849-870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 509] [Cited by in RCA: 438] [Article Influence: 48.7] [Reference Citation Analysis (5)] |
| 26. | Nauck MA, Kleine N, Orskov C, Holst JJ, Willms B, Creutzfeldt W. Normalization of fasting hyperglycaemia by exogenous glucagon-like peptide 1 (7-36 amide) in type 2 (non-insulin-dependent) diabetic patients. Diabetologia. 1993;36:741-744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 877] [Cited by in RCA: 794] [Article Influence: 24.1] [Reference Citation Analysis (0)] |
| 27. | Meier JJ, Gethmann A, Götze O, Gallwitz B, Holst JJ, Schmidt WE, Nauck MA. Glucagon-like peptide 1 abolishes the postprandial rise in triglyceride concentrations and lowers levels of non-esterified fatty acids in humans. Diabetologia. 2006;49:452-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 210] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 28. | Asmar A, Simonsen L, Asmar M, Madsbad S, Holst JJ, Frandsen E, Moro C, Jonassen T, Bülow J. Renal extraction and acute effects of glucagon-like peptide-1 on central and renal hemodynamics in healthy men. Am J Physiol Endocrinol Metab. 2015;308:E641-E649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 78] [Article Influence: 7.1] [Reference Citation Analysis (4)] |
| 29. | Karstoft K, Mortensen SP, Knudsen SH, Solomon TP. Direct effect of incretin hormones on glucose and glycerol metabolism and hemodynamics. Am J Physiol Endocrinol Metab. 2015;308:E426-E433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 25] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 30. | Skov J, Holst JJ, Gøtze JP, Frøkiær J, Christiansen JS. Glucagon-like peptide-1: effect on pro-atrial natriuretic peptide in healthy males. Endocr Connect. 2014;3:11-16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 52] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 31. | Aronis KN, Chamberland JP, Mantzoros CS. GLP-1 promotes angiogenesis in human endothelial cells in a dose-dependent manner, through the Akt, Src and PKC pathways. Metabolism. 2013;62:1279-1286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 82] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 32. | Ding L, Zhang J. Glucagon-like peptide-1 activates endothelial nitric oxide synthase in human umbilical vein endothelial cells. Acta Pharmacol Sin. 2012;33:75-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 122] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 33. | Basu A, Charkoudian N, Schrage W, Rizza RA, Basu R, Joyner MJ. Beneficial effects of GLP-1 on endothelial function in humans: dampening by glyburide but not by glimepiride. Am J Physiol Endocrinol Metab. 2007;293:E1289-E1295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 178] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 34. | Pfeffer MA, Claggett B, Diaz R, Dickstein K, Gerstein HC, Køber LV, Lawson FC, Ping L, Wei X, Lewis EF, Maggioni AP, McMurray JJ, Probstfield JL, Riddle MC, Solomon SD, Tardif JC; ELIXA Investigators. Lixisenatide in Patients with Type 2 Diabetes and Acute Coronary Syndrome. N Engl J Med. 2015;373:2247-2257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2059] [Cited by in RCA: 1815] [Article Influence: 165.0] [Reference Citation Analysis (4)] |
| 35. | Green JB, Bethel MA, Armstrong PW, Buse JB, Engel SS, Garg J, Josse R, Kaufman KD, Koglin J, Korn S, Lachin JM, McGuire DK, Pencina MJ, Standl E, Stein PP, Suryawanshi S, Van de Werf F, Peterson ED, Holman RR; TECOS Study Group. Effect of Sitagliptin on Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med. 2015;373:232-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2174] [Cited by in RCA: 1906] [Article Influence: 173.3] [Reference Citation Analysis (4)] |
| 36. | Marso SP, Daniels GH, Brown-Frandsen K, Kristensen P, Mann JF, Nauck MA, Nissen SE, Pocock S, Poulter NR, Ravn LS, Steinberg WM, Stockner M, Zinman B, Bergenstal RM, Buse JB; LEADER Steering Committee; LEADER Trial Investigators. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med. 2016;375:311-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6216] [Cited by in RCA: 5430] [Article Influence: 543.0] [Reference Citation Analysis (3)] |
| 37. | Marso SP, Bain SC, Consoli A, Eliaschewitz FG, Jódar E, Leiter LA, Lingvay I, Rosenstock J, Seufert J, Warren ML, Woo V, Hansen O, Holst AG, Pettersson J, Vilsbøll T; SUSTAIN-6 Investigators. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N Engl J Med. 2016;375:1834-1844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5404] [Cited by in RCA: 4723] [Article Influence: 472.3] [Reference Citation Analysis (3)] |
| 38. | Mylavarapu M, Gandapur A, Abarca Y, Roshni P, Fatima H, Contreras Vazquez S, Obi O, Siddiqui MA, Huda NU, Lysak Y, Mowo-wale A. Abstract 4143622: Efficacy of Semaglutide in Patients with Obesity and Heart Failure With Preserved Ejection Fraction (HFpEF): A Systematic Review&Meta-Analysis. Circulation. 2024;150 Suppl 1:A4143622. [DOI] [Full Text] |
| 39. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9803] [Reference Citation Analysis (0)] |
| 40. | Haddaway NR, Page MJ, Pritchard CC, McGuinness LA. PRISMA2020: An R package and Shiny app for producing PRISMA 2020-compliant flow diagrams, with interactivity for optimised digital transparency and Open Synthesis. Campbell Syst Rev. 2022;18:e1230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 649] [Cited by in RCA: 1480] [Article Influence: 370.0] [Reference Citation Analysis (5)] |
| 41. | Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, Cates CJ, Cheng HY, Corbett MS, Eldridge SM, Emberson JR, Hernán MA, Hopewell S, Hróbjartsson A, Junqueira DR, Jüni P, Kirkham JJ, Lasserson T, Li T, McAleenan A, Reeves BC, Shepperd S, Shrier I, Stewart LA, Tilling K, White IR, Whiting PF, Higgins JPT. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22257] [Cited by in RCA: 20409] [Article Influence: 2915.6] [Reference Citation Analysis (7)] |
| 42. | Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25:603-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14281] [Cited by in RCA: 13915] [Article Influence: 869.7] [Reference Citation Analysis (8)] |
| 43. | Review Manager 5. [cited 3 January 2026]. Available from: https://www.cochrane.org/authors/handbooks-and-manuals/style-manual/references/reference-types/software#revman. |
| 44. | Kosiborod MN, Petrie MC, Borlaug BA, Butler J, Davies MJ, Hovingh GK, Kitzman DW, Møller DV, Treppendahl MB, Verma S, Jensen TJ, Liisberg K, Lindegaard ML, Abhayaratna W, Ahmed FZ, Ben-Gal T, Chopra V, Ezekowitz JA, Fu M, Ito H, Lelonek M, Melenovský V, Merkely B, Núñez J, Perna E, Schou M, Senni M, Sharma K, van der Meer P, Von Lewinski D, Wolf D, Shah SJ; STEP-HFpEF DM Trial Committees and Investigators. Semaglutide in Patients with Obesity-Related Heart Failure and Type 2 Diabetes. N Engl J Med. 2024;390:1394-1407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 533] [Cited by in RCA: 481] [Article Influence: 240.5] [Reference Citation Analysis (0)] |
| 45. | Butler J, Abildstrøm SZ, Borlaug BA, Davies MJ, Kitzman DW, Petrie MC, Shah SJ, Verma S, Abhayaratna WP, Chopra V, Ezekowitz JA, Fu M, Ito H, Lelonek M, Núñez J, Perna E, Schou M, Senni M, van der Meer P, von Lewinski D, Wolf D, Altschul RL, Rasmussen S, Kosiborod MN. Semaglutide in Patients With Obesity and Heart Failure Across Mildly Reduced or Preserved Ejection Fraction. J Am Coll Cardiol. 2023;82:2087-2096. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 62] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 46. | Rehman A, Saidullah S, Asad M, Gondal UR, Ashraf A, Khan MF, Akhtar W, Mehmoodi A, Malik J. Efficacy and safety of semaglutide in patients with heart failure with preserved ejection fraction and obesity. Clin Cardiol. 2024;47:e24283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 47. | Ryan DH, Lingvay I, Deanfield J, Kahn SE, Barros E, Burguera B, Colhoun HM, Cercato C, Dicker D, Horn DB, Hovingh GK, Jeppesen OK, Kokkinos A, Lincoff AM, Meyhöfer SM, Oral TK, Plutzky J, van Beek AP, Wilding JPH, Kushner RF. Long-term weight loss effects of semaglutide in obesity without diabetes in the SELECT trial. Nat Med. 2024;30:2049-2057. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 193] [Article Influence: 96.5] [Reference Citation Analysis (0)] |
| 48. | Gao X, Hua X, Wang X, Xu W, Zhang Y, Shi C, Gu M. Efficacy and safety of semaglutide on weight loss in obese or overweight patients without diabetes: A systematic review and meta-analysis of randomized controlled trials. Front Pharmacol. 2022;13:935823. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 53] [Reference Citation Analysis (0)] |
| 49. | Garg SK, Kaur G, Haider Z, Rodriquez E, Beatson C, Snell-Bergeon J. Efficacy of Semaglutide in Overweight and Obese Patients with Type 1 Diabetes. Diabetes Technol Ther. 2024;26:184-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 57] [Article Influence: 28.5] [Reference Citation Analysis (0)] |
| 50. | Ahrén B, Atkin SL, Charpentier G, Warren ML, Wilding JPH, Birch S, Holst AG, Leiter LA. Semaglutide induces weight loss in subjects with type 2 diabetes regardless of baseline BMI or gastrointestinal adverse events in the SUSTAIN 1 to 5 trials. Diabetes Obes Metab. 2018;20:2210-2219. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 114] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 51. | Roh J, Houstis N, Rosenzweig A. Why Don't We Have Proven Treatments for HFpEF? Circ Res. 2017;120:1243-1245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 91] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 52. | Cleland JG, Tendera M, Adamus J, Freemantle N, Polonski L, Taylor J; PEP-CHF Investigators. The perindopril in elderly people with chronic heart failure (PEP-CHF) study. Eur Heart J. 2006;27:2338-2345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1232] [Cited by in RCA: 1029] [Article Influence: 51.5] [Reference Citation Analysis (3)] |
| 53. | Yusuf S, Pfeffer MA, Swedberg K, Granger CB, Held P, McMurray JJ, Michelson EL, Olofsson B, Ostergren J; CHARM Investigators and Committees. Effects of candesartan in patients with chronic heart failure and preserved left-ventricular ejection fraction: the CHARM-Preserved Trial. Lancet. 2003;362:777-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2481] [Cited by in RCA: 2030] [Article Influence: 88.3] [Reference Citation Analysis (3)] |
| 54. | Nielsen OW, Køber L, Torp-Pedersen C. Heart failure with preserved ejection fraction: dangerous, elusive, and difficult. Eur Heart J. 2008;29:285-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 55. | Chiorescu RM, Lazar RD, Ruda A, Buda AP, Chiorescu S, Mocan M, Blendea D. Current Insights and Future Directions in the Treatment of Heart Failure with Preserved Ejection Fraction. Int J Mol Sci. 2023;25:440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 56. | Pitt B, Pfeffer MA, Assmann SF, Boineau R, Anand IS, Claggett B, Clausell N, Desai AS, Diaz R, Fleg JL, Gordeev I, Harty B, Heitner JF, Kenwood CT, Lewis EF, O'Meara E, Probstfield JL, Shaburishvili T, Shah SJ, Solomon SD, Sweitzer NK, Yang S, McKinlay SM; TOPCAT Investigators. Spironolactone for heart failure with preserved ejection fraction. N Engl J Med. 2014;370:1383-1392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2427] [Cited by in RCA: 2092] [Article Influence: 174.3] [Reference Citation Analysis (4)] |
| 57. | Riedinger MS, Dracup KA, Brecht ML. Predictors of quality of life in women with heart failure. SOLVD Investigators. Studies of Left Ventricular Dysfunction. J Heart Lung Transplant. 2000;19:598-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 42] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 58. | Boyd KJ, Murray SA, Kendall M, Worth A, Frederick Benton T, Clausen H. Living with advanced heart failure: a prospective, community based study of patients and their carers. Eur J Heart Fail. 2004;6:585-591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 127] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 59. | Rector TS, Anand IS, Cohn JN. Relationships between clinical assessments and patients' perceptions of the effects of heart failure on their quality of life. J Card Fail. 2006;12:87-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 89] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 60. | Heo S, Lennie TA, Okoli C, Moser DK. Quality of life in patients with heart failure: ask the patients. Heart Lung. 2009;38:100-108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 179] [Cited by in RCA: 180] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 61. | Ferreira JP, Saraiva F, Sharma A, Vasques-Nóvoa F, Angélico-Gonçalves A, Leite AR, Borges-Canha M, Carvalho D, Packer M, Zannad F, Leite-Moreira A, Neves JS. Glucagon-like peptide 1 receptor agonists in patients with type 2 diabetes with and without chronic heart failure: A meta-analysis of randomized placebo-controlled outcome trials. Diabetes Obes Metab. 2023;25:1495-1502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 48] [Reference Citation Analysis (0)] |
| 62. | Ussher JR, Drucker DJ. Glucagon-like peptide 1 receptor agonists: cardiovascular benefits and mechanisms of action. Nat Rev Cardiol. 2023;20:463-474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 378] [Article Influence: 126.0] [Reference Citation Analysis (1)] |
| 63. | Sobral MVS, Rodrigues LK, Barbosa AMP, da Rocha NC, Moulaz IR, Dos Santos JPP, Oliveira BHC, Moreira JLML, Pacagnelli FL, Guida CM. Cardiovascular Effects of Semaglutide in Patients with Heart Failure with Preserved Ejection Fraction: A Systematic Review and Meta-Analysis. Am J Cardiovasc Drugs. 2025;25:461-467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 64. | Voors AA. Novel Recommendations for the Treatment of Patients With Heart Failure: 2023 Focused Update of the 2021 ESC Heart Failure Guidelines. J Card Fail. 2023;29:1667-1671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 65. | Anker SD, Butler J, Filippatos G, Ferreira JP, Bocchi E, Böhm M, Brunner-La Rocca HP, Choi DJ, Chopra V, Chuquiure-Valenzuela E, Giannetti N, Gomez-Mesa JE, Janssens S, Januzzi JL, Gonzalez-Juanatey JR, Merkely B, Nicholls SJ, Perrone SV, Piña IL, Ponikowski P, Senni M, Sim D, Spinar J, Squire I, Taddei S, Tsutsui H, Verma S, Vinereanu D, Zhang J, Carson P, Lam CSP, Marx N, Zeller C, Sattar N, Jamal W, Schnaidt S, Schnee JM, Brueckmann M, Pocock SJ, Zannad F, Packer M; EMPEROR-Preserved Trial Investigators. Empagliflozin in Heart Failure with a Preserved Ejection Fraction. N Engl J Med. 2021;385:1451-1461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3998] [Cited by in RCA: 3426] [Article Influence: 685.2] [Reference Citation Analysis (6)] |