Copyright: ©Author(s) 2026.
World J Cardiol. Mar 26, 2026; 18(3): 116299
Published online Mar 26, 2026. doi: 10.4330/wjc.v18.i3.116299
Published online Mar 26, 2026. doi: 10.4330/wjc.v18.i3.116299
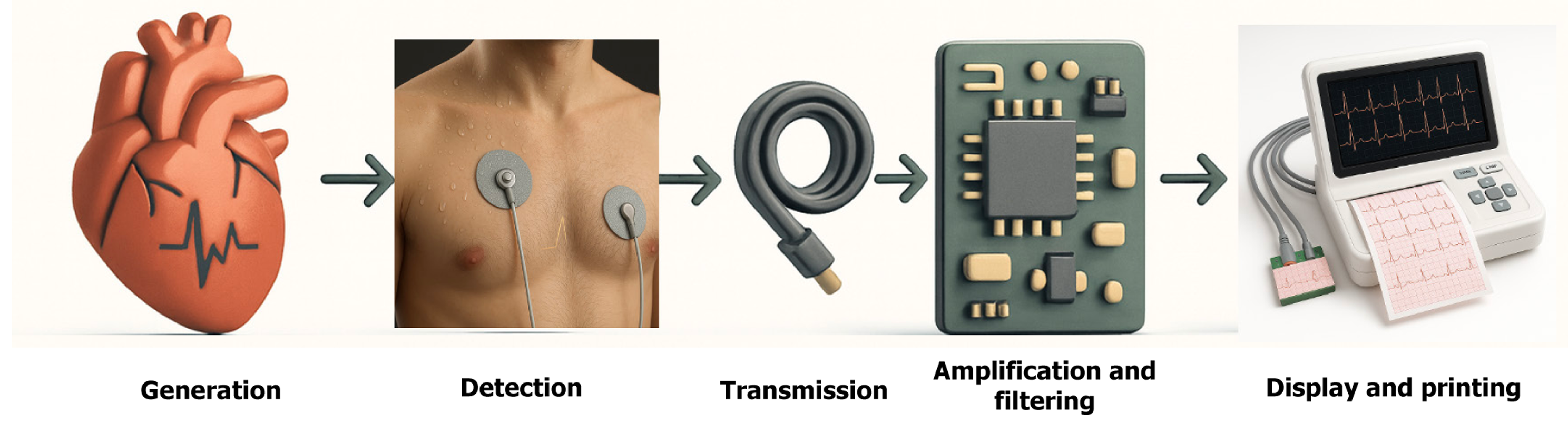
Figure 1 Journey of an electrocardiogram (from signal generation to printed output).
Possible source of artifacts in each step.
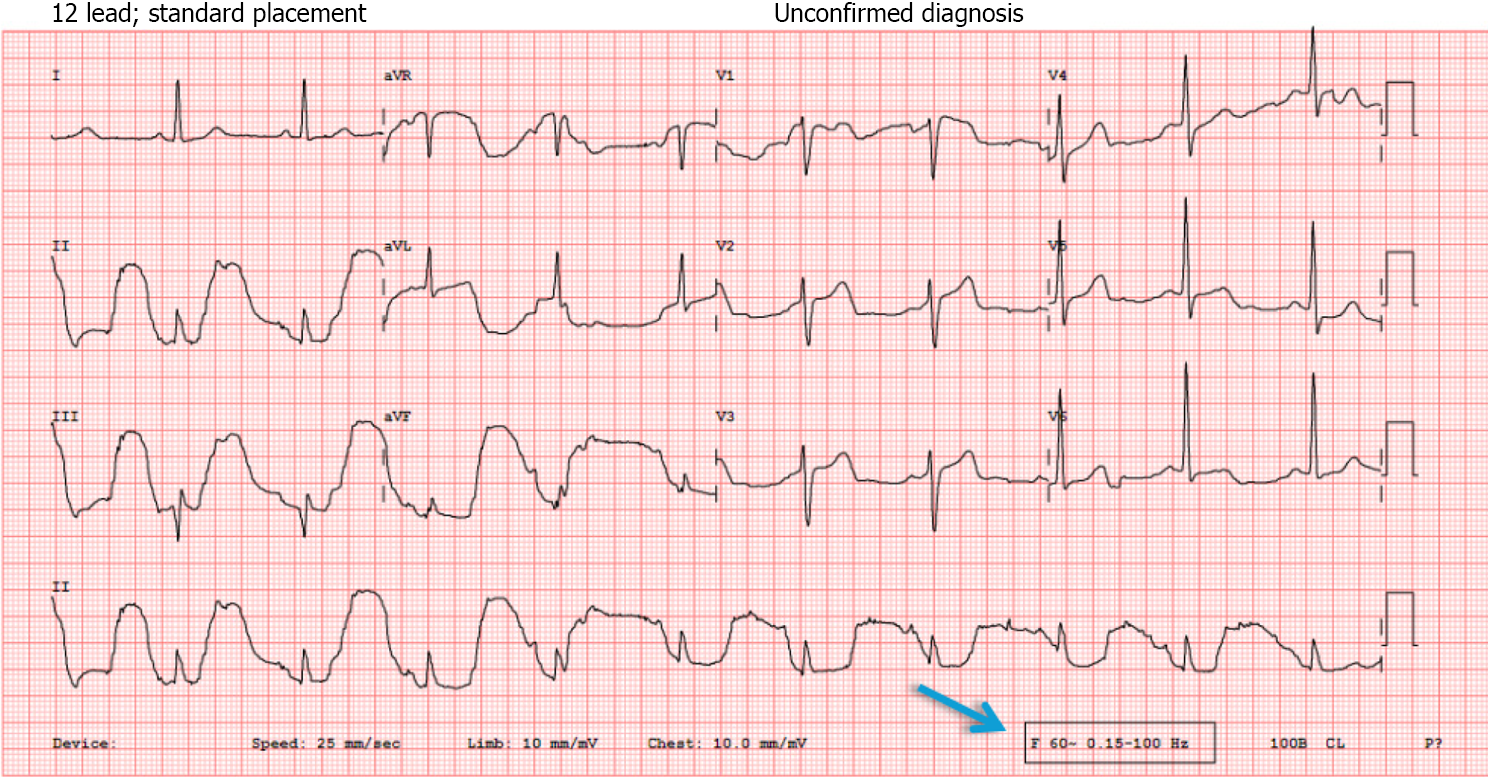
Figure 2 All the electrocardiograms have a notch filter (60 Hz in this example) and the low- to high-pass filter mentioned in the footnote (arrow).
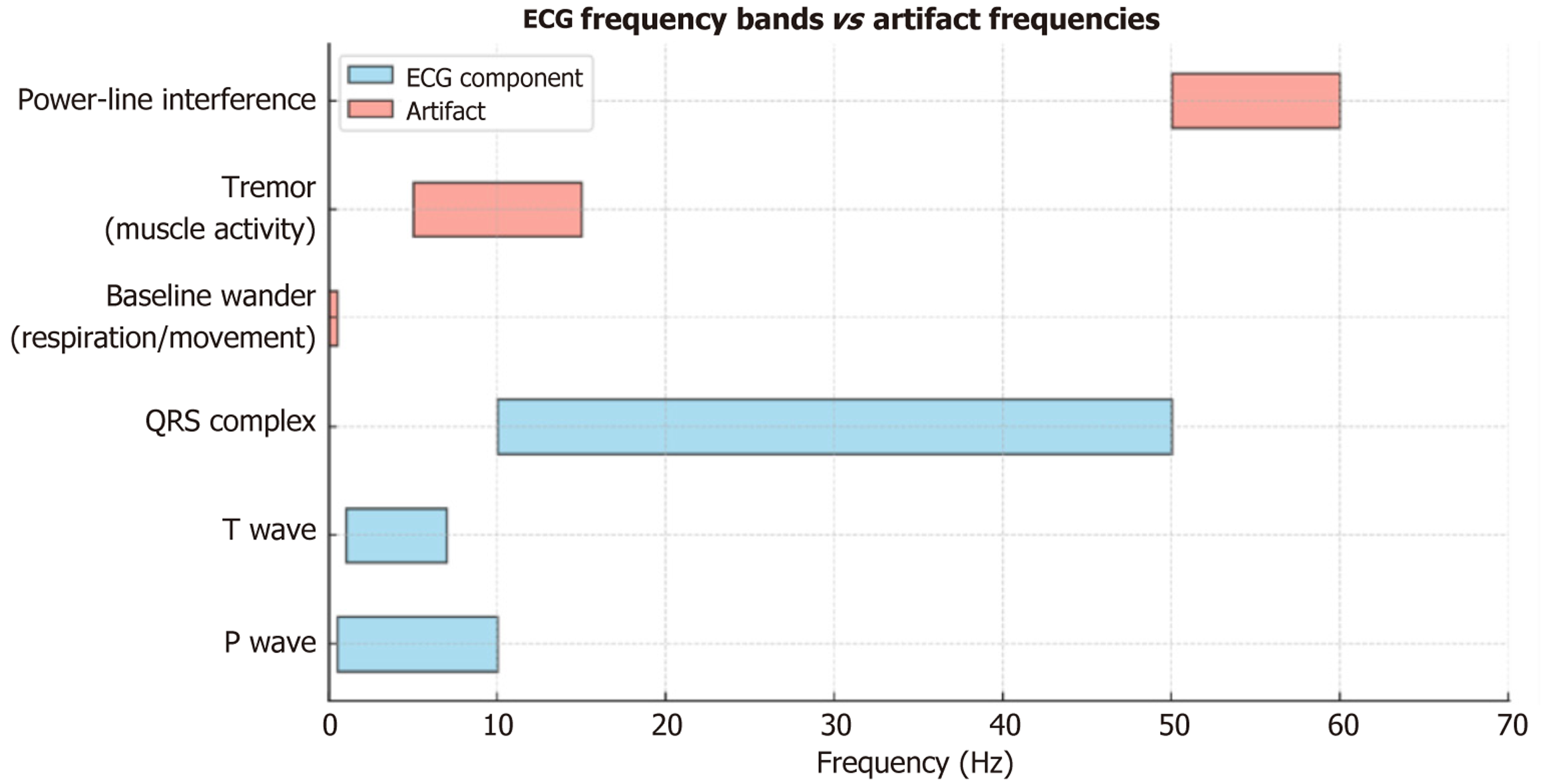
Figure 3 Frequencies of cardiac signals and artifacts showing significant overlap.
P and T waves overlap with tremors; ST shifts overlap with baseline wander; QRS overlaps with 50 Hz interference. ECG: Electrocardiogram.
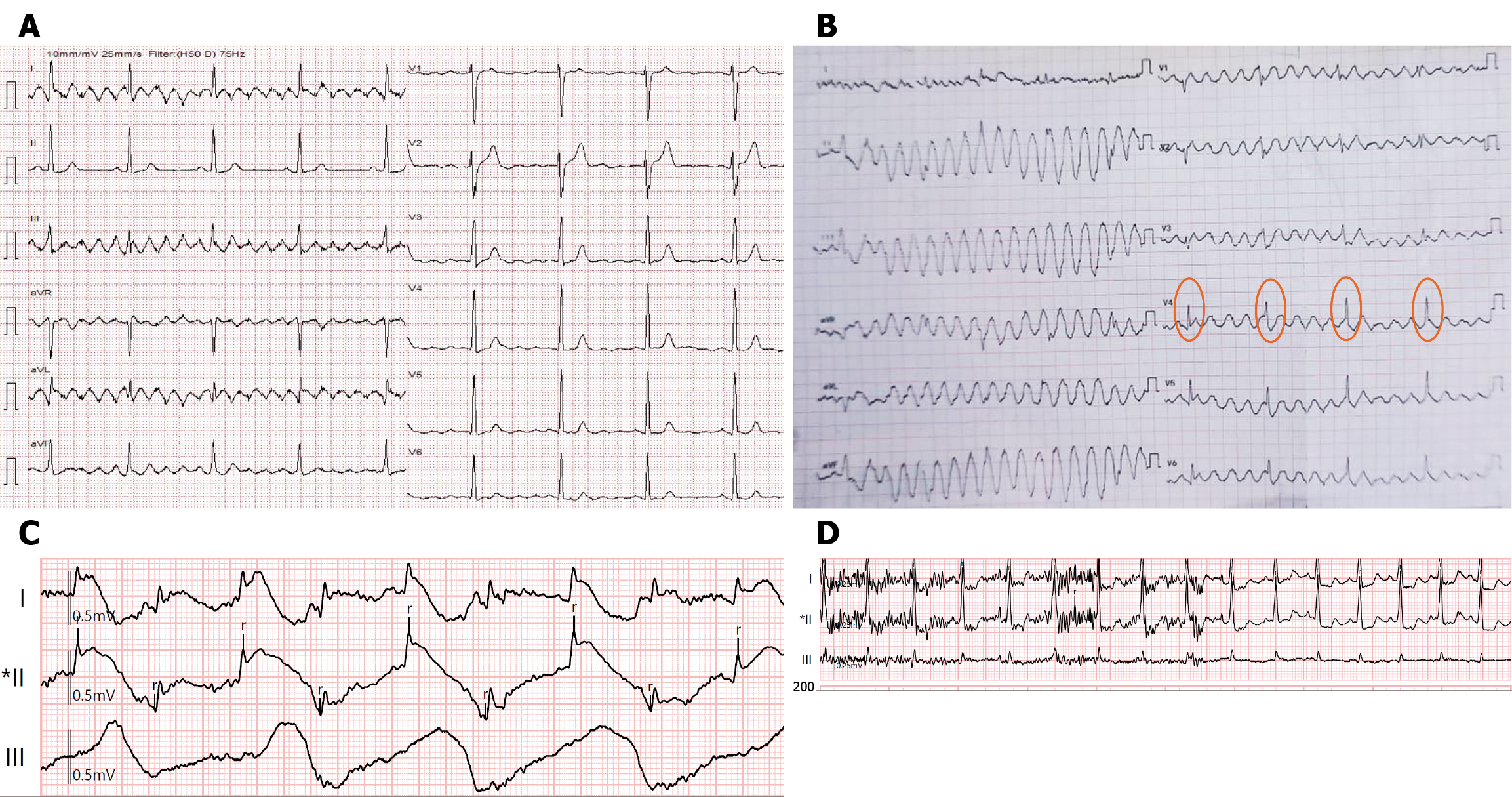
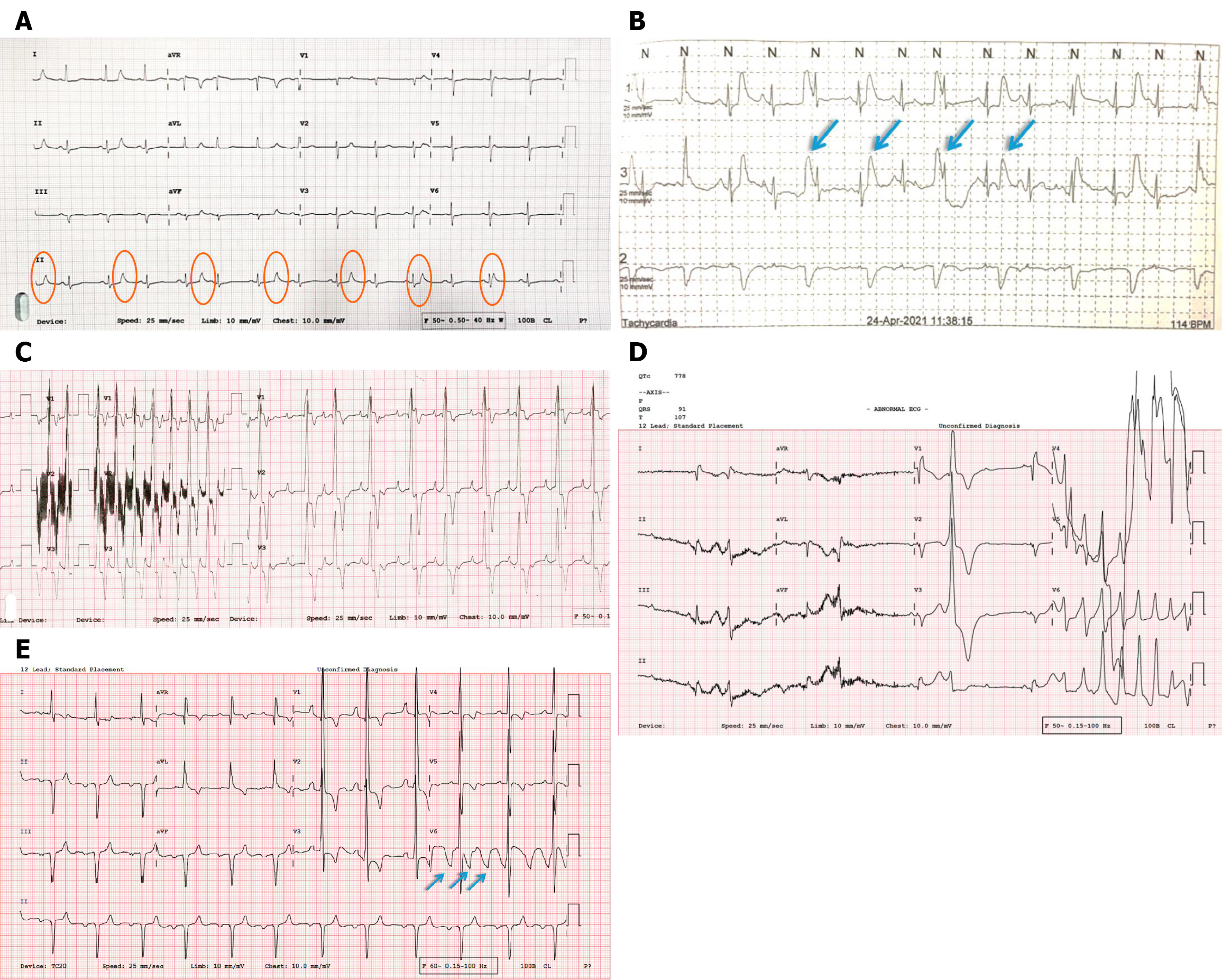
Figure 4 Patient-related motion artifacts.
A: Classical Parkinson’s rest tremor. Note that the frequency of the artifact is approximately 5 Hz (200 microseconds interval). Note that lead 2 does not have an artifact, indicating that the artifact is coming from the left arm; B: Limb movement artifacts resembling polymorphic ventricular tachycardia. Note that the normal QRS complexes (orange circles) are marching through the artifacts; C: Intraprocedural spurious ST elevation due to lead movement (attached to the subcostal region) during respiration; D: Trembling during micturition, causing baseline high-frequency irregular artifacts; note that the stoppage of the act of micturition resolves the baseline artifact.
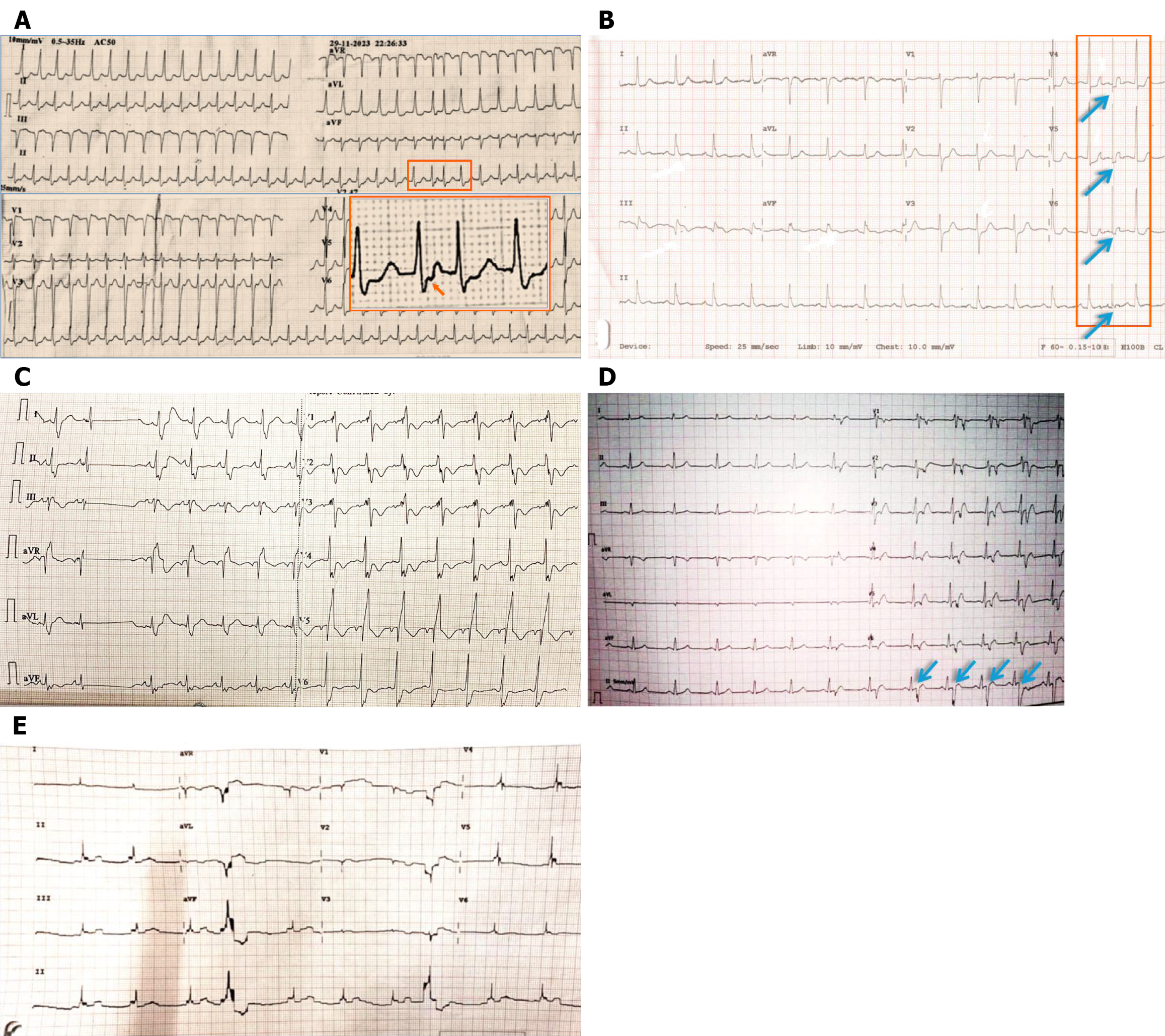
Figure 5 Patient-related and equipment-related artifacts.
A: Low-frequency artifacts (0.7 Hz) can be seen marching through without any PQRST association (orange circles). While the source of the artifact is unclear, they are too slow to be physiological tremors, which fall in the range of 3-12 Hz (or even higher). Respiratory movements can typically cause artifacts in this range (0.7 Hz), but the odd point corresponds to a respiratory rate of approximately 42/minute. The heart rate is too slow for that type of tachypnea; B: Similar artifacts can be seen on a Holter tracing from a young child, likely a respiration artifact (arrows); C: Attenuation correction artifacts showing low-amplitude high-frequency noise picked up by V2 lead to a crying newborn (D/D intercostal muscle fasciculation); D: Poor electrode contact causes intermittent baseline jumps in V4 and V5. Notably, artifacts may coexist with life-threatening arrhythmias. In this example, the patient had bradycardia-related Torsades de Pointes with simultaneous artifacts, emphasizing clinical evaluation while interpreting or suspecting artifacts on electrocardiogram; E: Poor electrode contact causes irregular baseline jumps in V6 (arrows).
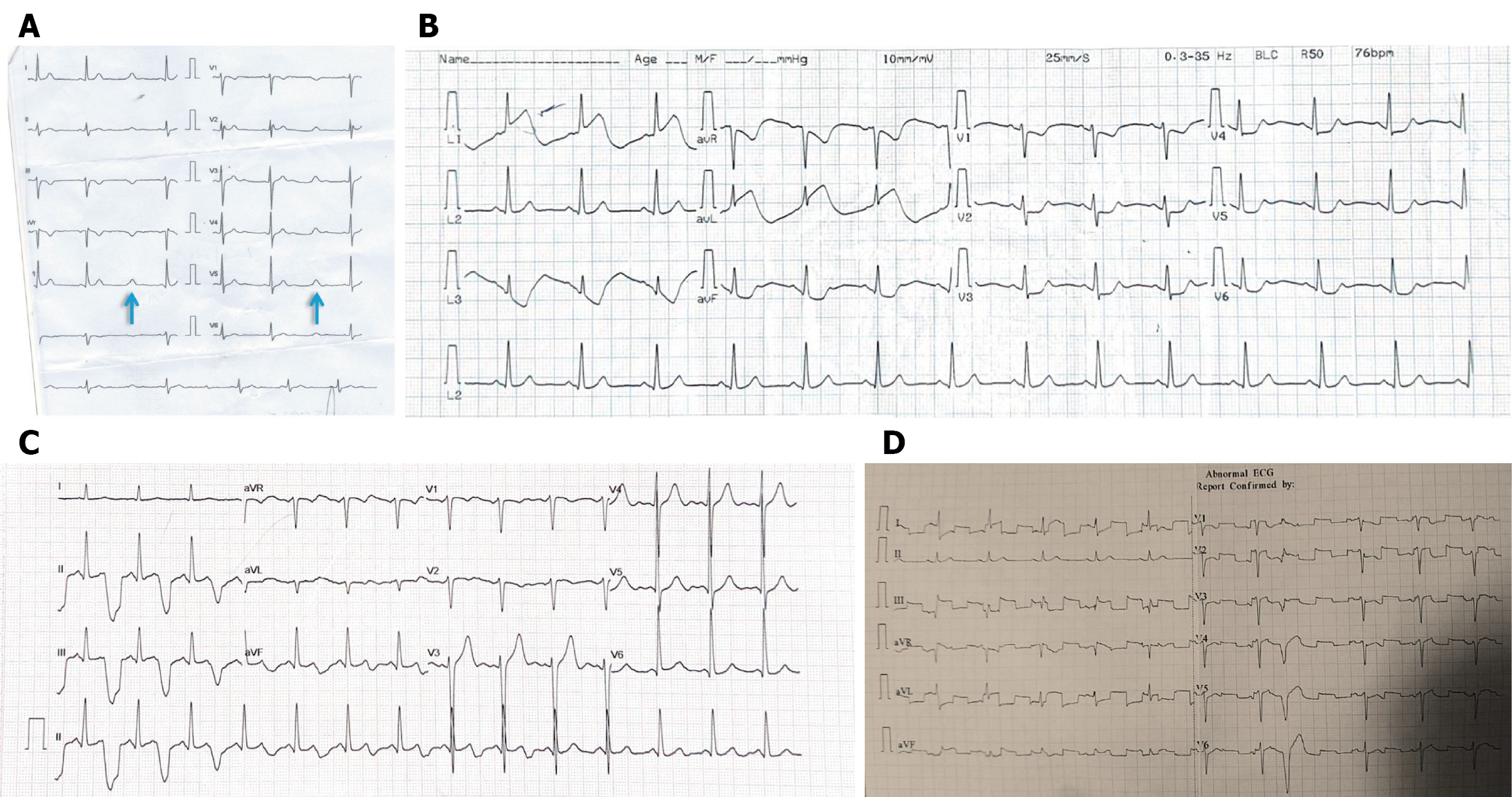
Figure 6 Equipment-related artifacts.
A: Deceleration artifact (orange box) causing narrowing of the QRS complex and giving a false impression of a spurious atrial premature contraction (arrow) advancing the next QRS complex (note the further narrowing of the QRS complex due to paper deceleration) in an ongoing narrow complex tachycardia; B: Deceleration artifact (orange box) causing narrowing of the QRS complex (nearly a straight vertical line - arrows) (note the narrowing of the preceding and the next T waves due to paper deceleration); C: Acceleration artifacts cause stretching of the ST segment, resulting in a false impression of sinus slowing. Prolonged stretching may lead to false impressions of significant sinus pauses or atrioventricular blocks. Note that the initial paper deceleration caused narrowing of the QRS complex, followed by paper acceleration; D: Filtering error causing a sharp artifact after the QRS complex, resulting in a false impression of double-QRS (arrows); E: Similar error in filtering causing significant distortion of waveforms with a baseline shift, making the electrocardiogram incomprehensible.
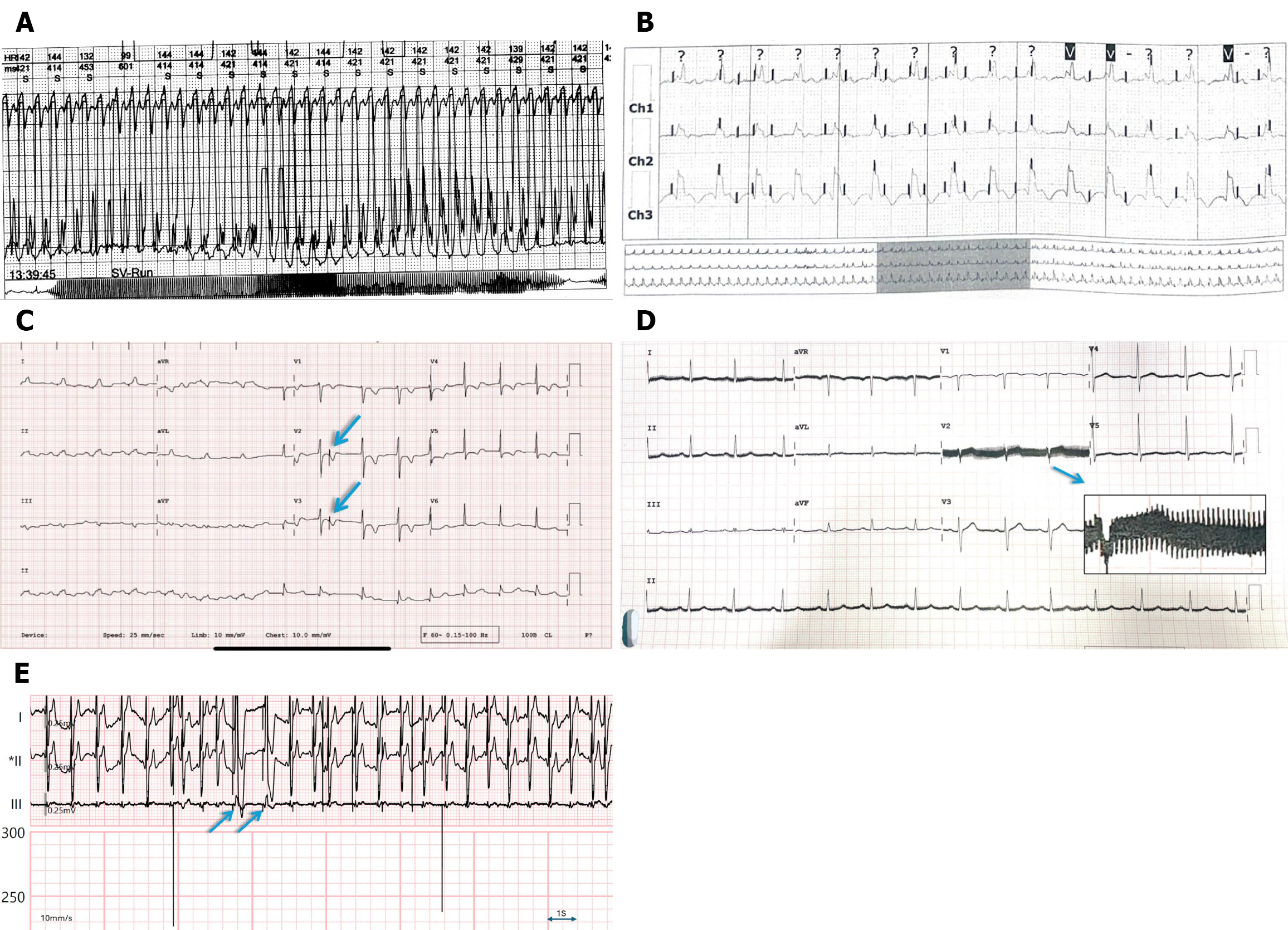
Figure 7 Equipment-related and environmental artifacts.
A: Missing PQRSs due to processing failure with residual T waves give rise to double T waves or “orphan T” waves (arrows); B: Arterial pulse tapping artifact mimicking ST elevation. All QRS complexes are followed by artifacts with electromechanical associations. Note that lead 2 is spared, meaning that the artifact comes from the left arm electrode; C: Similarly, arterial pulse tapping artifacts mimic bizarre T waves with prolonged QT intervals. Note that lead 1 is spared, meaning that the artifact comes from the left lower limb electrode; D: Electrocardiogram showing fixed-frequency square wave artifacts. ECG: Electrocardiogram.
Figure 8 Environmental and device-related artifacts.
A: Electromagnetic interference causes sharp deflections, mimicking monomorphic ventricular tachycardia; B: Holter strip showing pseudopacing artifacts that cannot be explained by any device algorithm; C: Electrocardiogram in a patient with a dual-chamber pacemaker showing pseudopacing artifacts (arrows); D: Deep brain stimulators causing high-frequency (usually 60-200 Hz) artifacts (12 pulses in 200 microseconds = 60 Hz) (arrow - zoomed snippet of V2); E: During intravascular lithotripsy, shock pulses can be detected via electrocardiogram. It can even capture the ventricles, which are called shocktopics (arrows). For each cycle of intravascular lithotripsy shock, there are 10 pulses at 1-second intervals (1 Hz).
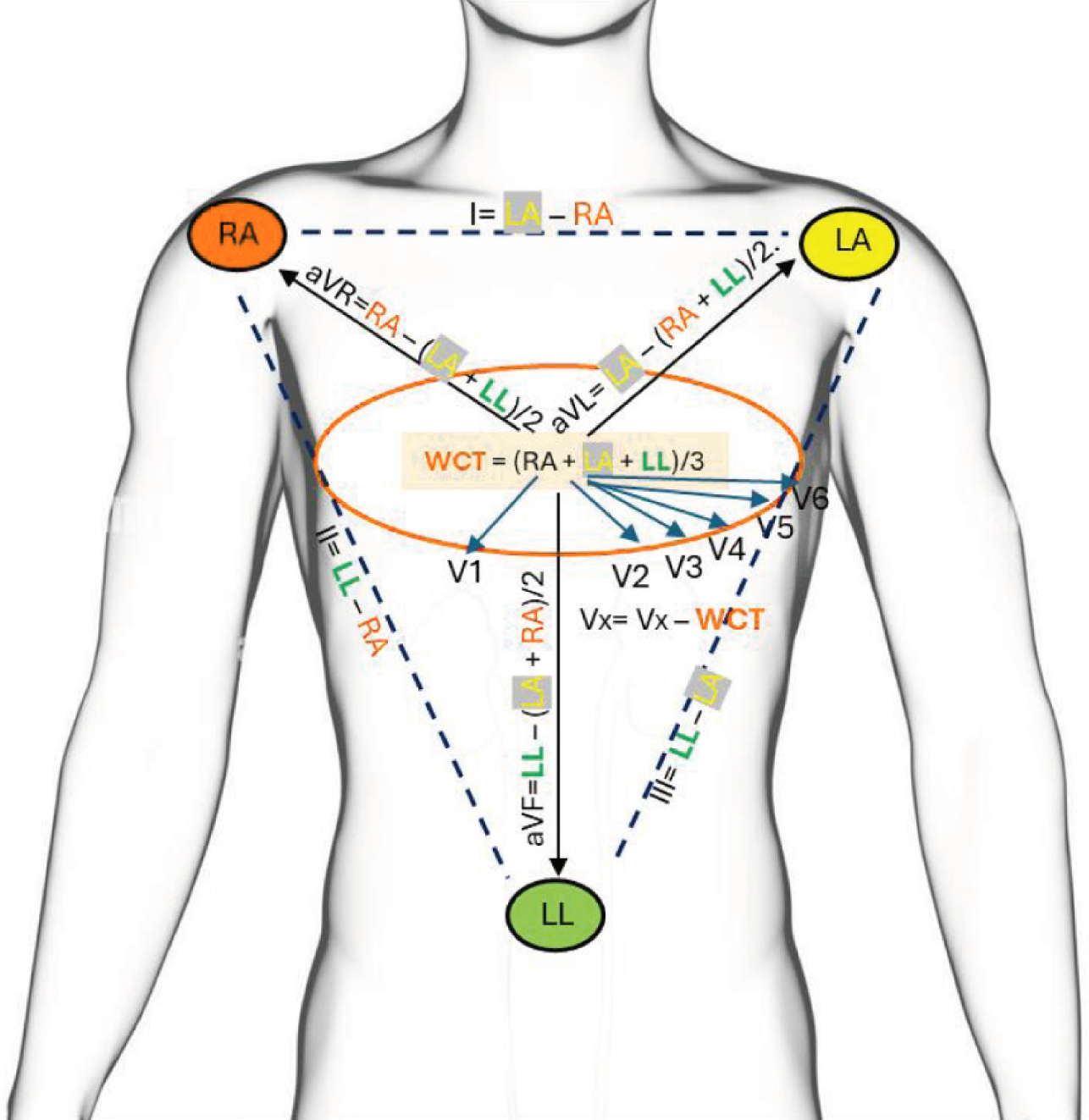
Figure 9 Approach to artifact detection.
Schematic depiction of affected leads when an artifact is generated from a specific limb; color facilitates easy interpretation. LA: Left arm; RA: Right arm; LL: Left leg; Vx: Precordial lead; WCT: Wilson’s central terminal.
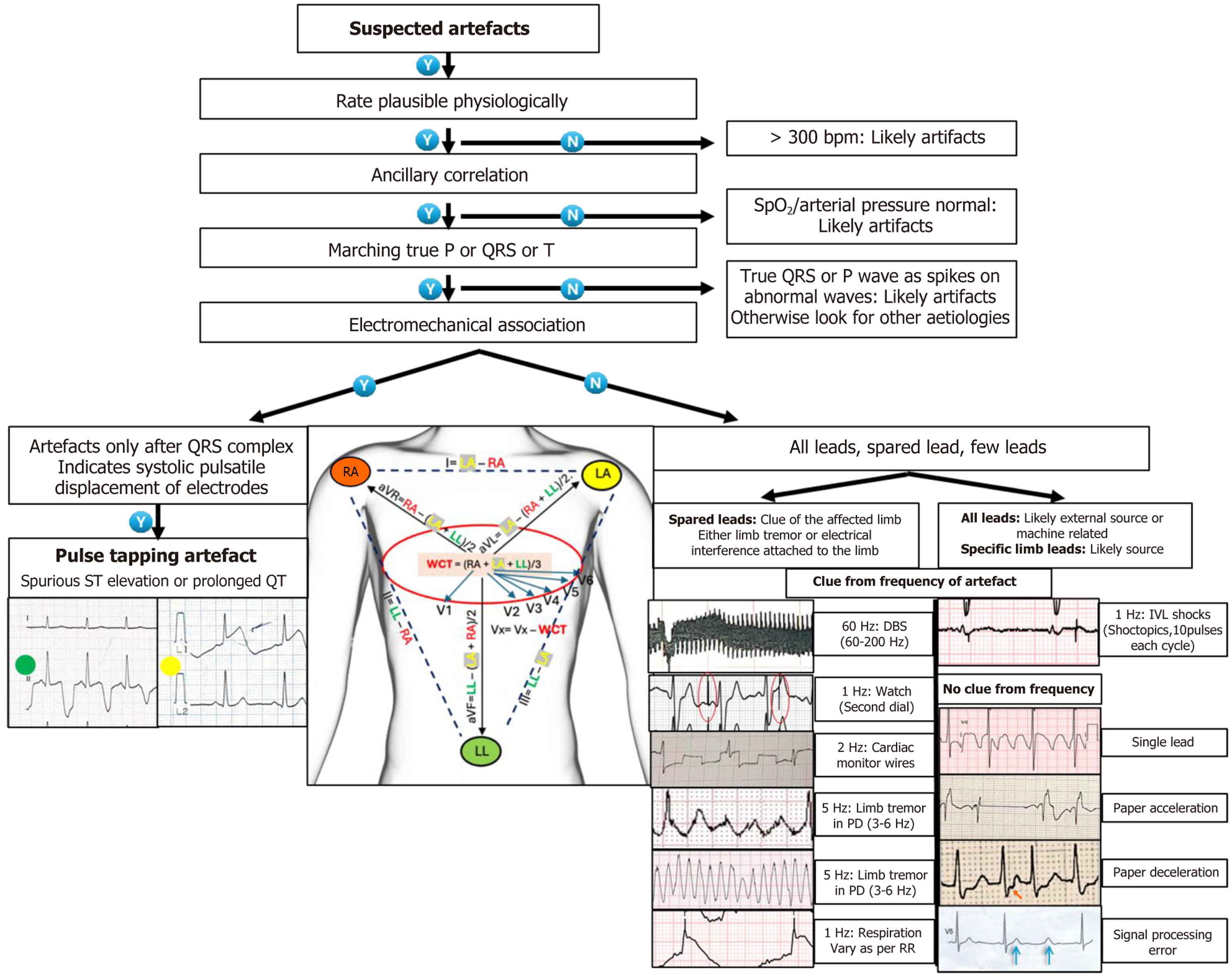
Figure 10 A stepwise approach to electrocardiographic artifacts.
Orange arrow - spurious atrial premature complex, see Figure 6A; blue arrow - orphan T wave of the corresponding missing QRS complex along with normal T wave, color facilitates easy interpretation. LA: Left arm; RA: Right arm; LL: Left leg; Vx: Precordial lead; WCT: Wilson’s central terminal.
- Citation: Mondal S, Raja DP, Muslim NA, Prabhu MA. Electrocardiographic artifacts in clinical practice: A logical approach to recognition and prevention. World J Cardiol 2026; 18(3): 116299
- URL: https://www.wjgnet.com/1949-8462/full/v18/i3/116299.htm
- DOI: https://dx.doi.org/10.4330/wjc.v18.i3.116299