Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.116918
Revised: December 18, 2025
Accepted: February 2, 2026
Published online: April 27, 2026
Processing time: 151 Days and 5 Hours
Severe acute pancreatitis (SAP), a prevalent and life-threatening abdominal emer
Core Tip: Severe acute pancreatitis (SAP)-associated acute lung injury is a pivotal determinant of mortality. The gut-lung axis plays a central role in its pathogenesis. SAP-induced gut microbiota dysbiosis compromises the intestinal barrier, facilitating bacterial/endotoxin translocation and gut-derived immune cell migration to the lungs. This process culminates in a profound pulmonary inflammatory response, thereby identifying the gut-lung axis as a promising therapeutic target for novel interventions against SAP-associated acute lung injury.
- Citation: Ye XT, Yu CY, Zhao HY, Qian ST, Huang Y, Liu QS. Exploring the mechanism and potential treatment of severe acute pancreatitis-associated lung injury based on the gut-lung axis. World J Gastrointest Surg 2026; 18(4): 116918
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/116918.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.116918
Severe acute pancreatitis (SAP) is a serious inflammatory disease of the pancreas that is often accompanied by systemic inflammatory response syndrome and multiple organ dysfunction syndrome[1,2]. The incidence of SAP has been in
Acute lung injury (ALI)/acute respiratory distress syndrome (ARDS) represents a severe complication of SAP with mortality rates reaching up to 50%[4,5]. However, the pathogenesis of SAP-associated ALI (SAP-ALI) remains incomple
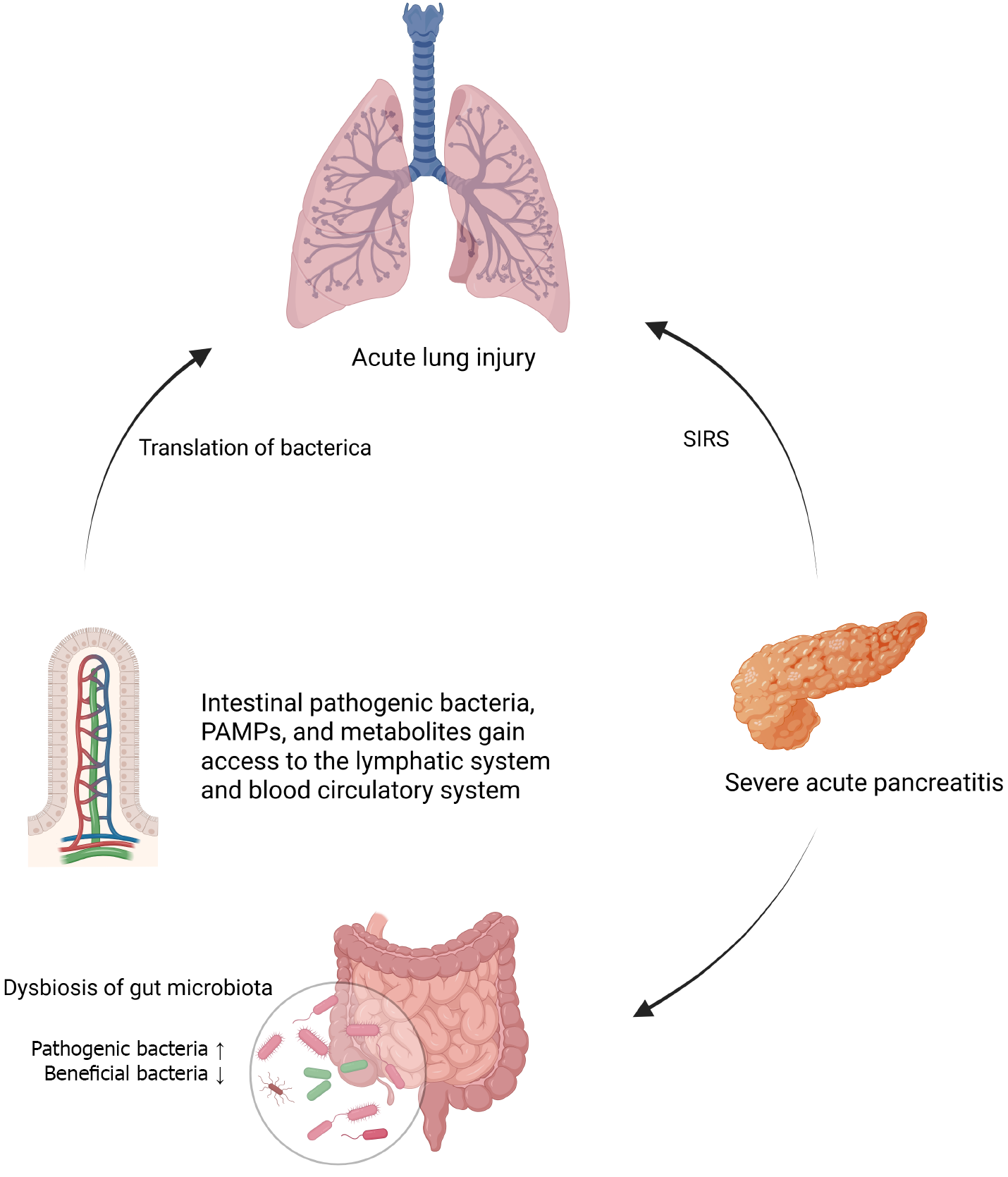
The gut-lung axis is a bidirectional regulatory system between the intestine and the lungs, operating through complex interactive pathways that involve host-microbial relationships, microbial community crosstalk, and immune response regulation[6]. In SAP gut microbiota dysbiosis, characterized by reduced diversity, correlates with disease severity, thus impairing intestinal mucosal immunity, increasing permeability, and facilitating bacterial translocation (BT)[3]. BT to the lungs activates inflammatory pathways, releasing proinflammatory cytokines and exacerbating lung injury. Patients with ARDS exhibit elevated levels of gut-associated bacteria (e.g., Bacteroidaceae and Enterobacteriaceae) in the lower respiratory tract[7]. Short-chain fatty acids (SCFAs) (e.g., butyrate and propionate), the main metabolites of the intestinal flora, act as key regulators that enhance the intestinal barrier function and inhibit the activation of inflammatory vesicles by inhibiting histone deacetylase (HDAC) activity or activating G protein-coupled receptors (GPCRs) and may directly affect lung tissues to reduce ALI[8,9]. In addition, gut-derived immune cells migrate to the lungs, modulating local inflammatory responses.
Systematic research on the role of the gut–lung axis in SAP-ALI pathogenesis remains limited. This mini-review summarized gut microbiota alterations, the bidirectional regulatory mechanisms of the gut-lung axis, and advancements in SAP-targeted interventions, offering insights for future clinical applications.
Growing evidence highlights the pivotal role of the gut microbiota in acute pancreatitis (AP) progression. In healthy individuals Bacillota dominate the intestinal microbiota, followed by Bacteroidota, Actinomycetota, and Pseudomonadota. Compared with healthy controls, patients with AP exhibited increased levels of Bacteroidota and Pseudomonadota (e.g., Escherichia and Streptococcus) and reduced levels of Bacillota and Actinomycetota[10-13]. AP-associated bacteria replaced the host-specific microbial community (Table 1)[10-19]. Previous studies have shown that the microbial composition of fecal samples from patients with early-stage AP is different from that of healthy controls[17-19].
| Ref. | Type of sample | Subjects | Sequencing methods | Phylum | Genus | Species |
| Zhang et al[10] | Fecal | AP (n = 45) vs healthy volunteers (n = 44) | 16S rRNA sequencing | Increase: Bacteroidota, Pseudomonadota. Decrease: Bacillota, Actinomycetota | - | - |
| Gong et al[11] | Fecal | AP (n = 12) vs healthy controls (n = 12) | Metagenomics and culturomics | Increase: Bacillota, Pseudomonadota. Decrease: Bacteroidota | Increase: Enterococcus, Klebsiella. Decrease: Bacteroides, Eubacterium | Increase: Enterococcus faecium, Enterococcus faecalis, unclassified Enterococcus, Escherichia coli. Decrease: Unclassified Bacteroides, Eubacterium rectale, Prevotella copri |
| Xie et al[12] | Fecal | AP (n = 86) vs healthy controls (n = 46) | 16S rRNA sequencing | - | Increase: Enterococcus, Pseudomonas, Escherichia-Shigella. Decrease: Akkermansia, Lachnoclostridium, Ruminococcus | - |
| Hu et al[13] | Fecal | AP (n = 65) vs healthy volunteers (n = 20) | 16S rRNA gene sequencing | Increase: Pseudomonadota, Bacteroidota | Increase: Escherichia-Shigella, Bacteroides, Enterococcus. Decrease: Bifidobacterium | Increase: Clostridium ramosum. Decrease: Bifidobacterium longum |
| He et al[14] | Fecal | SAP (n = 73) vs healthy volunteers (n = 28) | - | Increase: Pseudomonadota. Decrease: Bacillota, Actinomycetota | Increase: Escherichia-Shigella, Enterococcus, Klebsiella. Decrease: Blautia, Bifidobacterium, Romboutsia | - |
| Wang et al[15] | Fecal | SAP vs mild AP | 16S rRNA sequencing | Increase: Pseudomonadota, Planctomycetota | Increase: Stenotrophomonas, Enterobacter | Increase: Cloacibacillus evryensis |
| Ammer-Herrmenau et al[16] | Fecal | SAP (n = 28) vs non-SAP (n = 363) | Metagenomic sequencing | - | - | Increase: Parabacteroides distasonis, Enterocloster bolteae, Flavonifractor plautii, Dysosmobacter welbionis, Anaerobutyricum hallii, Roseburia hominis, Clostridium CCN A10. Decrease: Lachnospiraceae sp., Lachnospira GAM79, Lachnospira eligens, Finegoldia magna, Peptostreptococcus anaerobius, Porphyromonas asaccharolytica, Lawsonella clevelandensis, Streptococcus pyogenes, Anaerococcus mediterraneensis |
| Zhao et al[17] | Ileum tissues | AP mice (n = 15) vs control group (n = 10) | 16S rRNA sequencing | Increase: Verrucomicrobiota. Decrease: Deferribacterota, Actinomycetota, Campylobacterota | Increase: Muribaculum, Akkermansia, Parabacteroides, Oscillospiraceae_unclassified, Bacteroides. Decrease: Helicobacter, Dietzia | - |
| Fu et al[18] | Cecal contents | AP mice vs control group | 16S rRNA sequencing | Increase: Pseudomonadota, Bacillota. Decrease: Bacteroidota | Increase: Escherichia-Shigella, Bacteroides, Helicobacter. Decrease: Blautia | - |
| Zhang et al[19] | Colonic contents | SAP mice vs sham-operated group | RNA and assay for transposase accessible chromatin sequencing | Increase: Pseudomonadota. Decrease: Bacillota | Increase: Escherichia-Shigella, Morganella, Flagellated bacteria. Decrease: Lactobacillus | - |
Compared with that in patients with mild AP, the diversity of the gut microbiome was significantly lower in patients with SAP, particularly with an increase in pathogenic bacteria and a decrease in beneficial bacteria such as Blautia, Enterococcus, Faecalicatena contorta, and Ruminococcaceae. Specifically, at the phylum level Pseudomonadota and Planctomycetota were significantly enriched in the SAP group. At the family level, Enterobacteriaceae, Veillonellaceae and Pseudonocardiaceae were more abundant in the SAP group. At the genus level, the abundance of Stenotrophomonas and Enterobacter was significantly increased in the SAP group. At the species level, the abundance of Cloacibacillus evryensis was significantly increased in the SAP group[15]. Therefore, there is a significant correlation between the composition of the gut microbiome and the severity of pancreatitis. Changes in specific microbial communities may reflect the severity of the disease and the corresponding physiological state.
In a prospective multicenter European study, the rectal and oral microbiomes of 450 patients with AP were collected at the time of admission to the hospital; unexpectedly, the abundance of 10 known SCFA-producing bacteria was greater in the SAP group than in the control group[16]. One explanation is that rectal swabs represent a relatively large mucosa-associated microbiome, and the differences observed in this study may be a result of niche-specific changes in the SAP group. This spatial variation in dysbiosis is a common phenomenon in patients with other diseases associated with an altered intestinal microbiome[20,21]. Evidence suggests that SCFA-producing species and strains may also be harmful[22,23]. However, compared with those in SAP non-survivors, the levels of SCFAs in SAP survivors were found to be in
In healthy individuals the lung flora is dominated by Bacteroidota and Bacillota with Streptococcus, Prevotella and Veillonella being the most common[24]. A study revealed that the proportion of gut-associated bacteria, especially Bacteroidota and Enterobacteriaceae, increased in the lower respiratory tracts of patients with ARDS[25]. Further studies have shown that a predominance of Enterobacteriaceae in the gut and its enrichment in the lungs of patients with ALI/ARDS are major drivers of lung microbiota dysbiosis, are positively correlated with serum interleukin-6 (IL-6) and alveolar tumor necrosis factor alpha levels, and are highly valuable in predicting the development of ALI/ARDS[26-28]. Even transient translocation of such bacteria can alter the lung microbiota[29].
Prior to ARDS onset in patients with SAP, the gut microbiota already exhibits ARDS-associated characteristics with Escherichia-Shigella identified as a key predictive biomarker. Compared with patients with SAP but without ARDS, those with ARDS have increased abundances of Pseudomonadota, Enterobacteriaceae, Escherichia-Shigella, and Enterococcus but reduced abundances of Bifidobacterium. At the species level, potentially pathogenic bacteria such as Klebsiella pneumoniae, Prevotella copri, and Clostridium ramosum are significantly elevated in patients with SAP-associated ARDS[13]. Addi
The impact of BT on SAP is widespread. BT refers to the entry of enteric pathogens into the lungs through a compro
Clinical evidence further links intestinal bacterial load to the severity of lung injury in patients with SAP. A hydrogen breath test study revealed a significant correlation between bacterial counts and the progression from ALI (PaO2/FiO2 ≤ 200) to more severe ARDS (PaO2/FiO2 < 300), indicating that early organ failure is associated with gut-derived bacteria[36]. This pulmonary injury is exacerbated by impaired oxidative stress defense and marked by decreased superoxide dismutase and glutathione peroxidase activities and elevated malondialdehyde levels[37].
Tang et al[38] by constructing a model of antibiotic-induced imbalance of intestinal bacteria found that the dysbiosis would damage the intestinal mucosal barrier and induce lipopolysaccharide (LPS) translocation to the systemic circulation. Hashimoto et al[39] showed that antibiotic-induced microbiota depletion could improve LPS-induced ALI and significantly reduce the increase of IL-6 levels.
SAP promotes an expansion in the abundance of flagellated bacteria and disrupts metabolite homeostasis, leading to the activation of toll-like receptor 5 (TLR5) on lamina propria dendritic cells[27]. This signaling impairs the intestinal barrier and alters helper T cell differentiation, ultimately facilitating BT. Thus, the flagellin-TLR5 axis represents a promising therapeutic target for mitigating gut-derived complications in patients with SAP. Although the mechanisms underlying microbiota dysbiosis require further elucidation, restoring intestinal barrier integrity remains crucial for improving SAP-ALI.
SCFAs, primarily acetate, propionate, and butyrate, are organic fatty acids (≤ 6 carbon atoms) produced by the gut mi
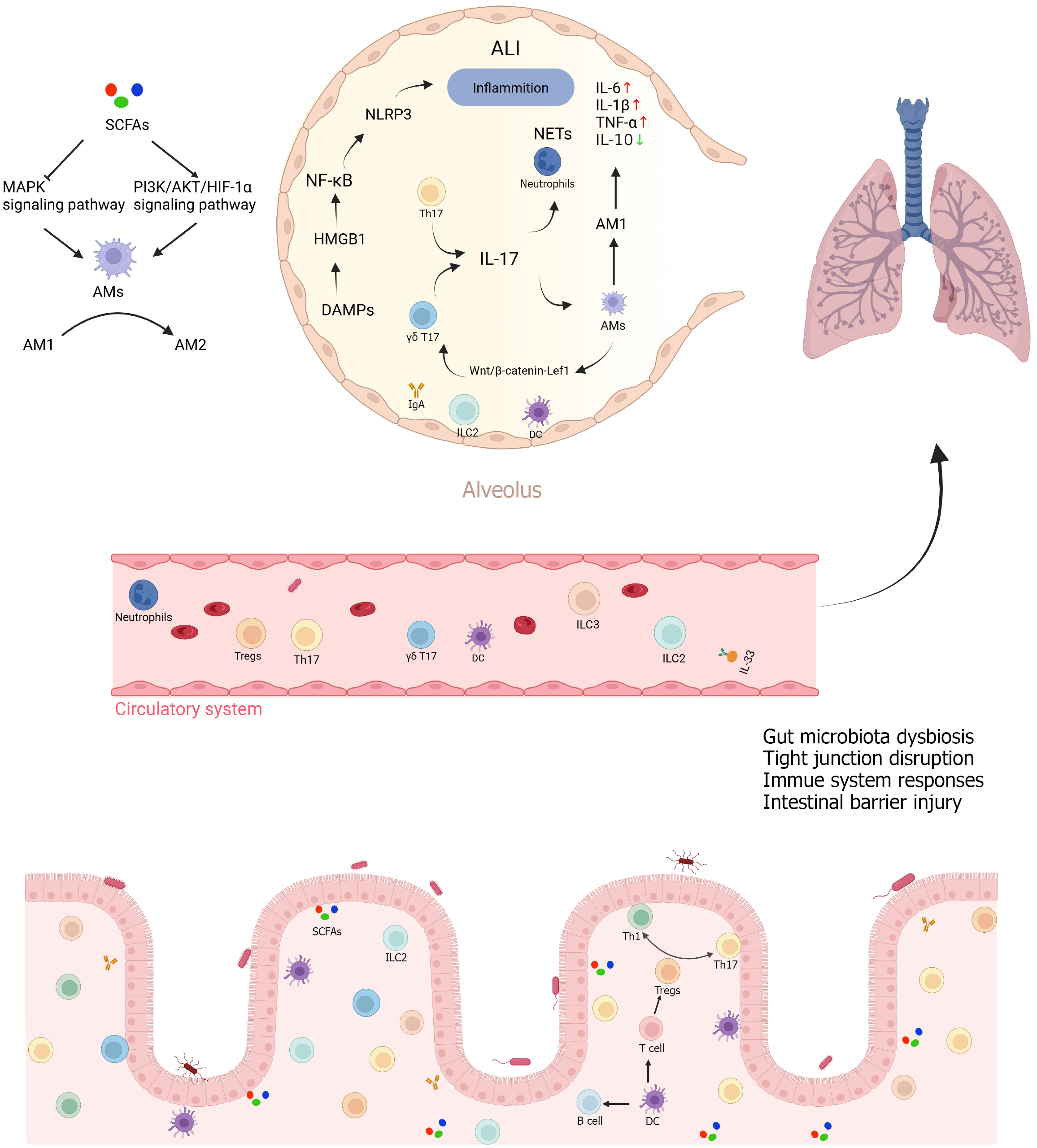
SCFAs function primarily through two pathways: Inhibition of HDAC activity to exert epigenetic effects and activation of GPCRs to modulate immunity. By binding to GPCRs on intestinal and lung cells, SCFAs activate the AMPK pathway while suppressing NF-κB and NLRP3 inflammasome activities. These actions enhance epithelial barrier integrity, attenuate proinflammatory responses, and ultimately ameliorate histopathological damage in SAP-ALI[9,41].
Receptors for SCFAs, FFAR2 and FFAR3, are expressed on alveolar macrophages and type 2 epithelial cells with their levels modulated by LPS exposure[42]. This finding supports a direct role for SCFAs in lung protection as they sig
As a natural HDAC inhibitor, butyrate modulates inflammatory gene expression, suppressing the expression of proinflammatory cytokines (e.g., tumor necrosis factor alpha, IL-1β, and IL-6) while promoting anti-inflammatory IL-10 release[45]. In hypoxic models butyrate attenuated pulmonary vascular edema and leakage, reduced macrophage infiltration, and enhanced lung endothelial barrier integrity by upregulating the expression of tight junction proteins (occludin, cingulin, and claudin-1) and increasing histone H3 acetylation[46]. Similarly, in murine AP models, butyrate supplementation strengthened the intestinal barrier function via tight junction upregulation and HDAC inhibition, thereby reducing BT, systemic inflammation, and secondary infection risk.
Acetate and propionate, which are likely derived from the gut and transferred to the lungs, are present in human lung tissue at varying levels. They exert anti-inflammatory effects by activating GPCR41/43 and inhibiting the NLRP3 inflammasome, thereby reducing the maturation and release of IL-1β and IL-18 and mitigating local inflammation in the intestine and pancreas. Tian et al[47] demonstrated that enrichment of propionate-producing bacteria, particularly Lachnospiraceae, attenuated lung inflammation following pulmonary ischemia-reperfusion injury in vivo.
In SAP mice propionate and butyrate mitigate lung injury by inhibiting macrophage M1 polarization and neutrophil infiltration via suppression of the mitogen-activated protein kinase pathway, thereby modulating inflammation and restoring the intestinal barrier function[8]. Conversely, succinate, a bacterial metabolite, activates the phosphoinositide 3-kinase/protein kinase B/hypoxia-inducible factor 1 alpha pathway by binding to SUCNR1 on pulmonary macrophages, promoting their polarization toward a reparative M2 phenotype to alleviate injury[48].
To overcome the low bioavailability of SCFAs, chitosan-protocatechuic aldehyde/calcium alginate/sodium butyrate microspheres were developed by encapsulating sodium butyrate in a chitosan-based carrier, offering a targeted approach for intestinal barrier repair[49]. This delivery system represents a promising strategy for SCFA application. Although existing studies have suggested that SCFA supplementation has beneficial effects on SAP-ALI, the available evidence remains limited and somewhat controversial, warranting further investigation[50,51].
Immune cells are pivotal for maintaining the proinflammatory and anti-inflammatory balance during SAP-ALI (Figure 2)[52,53]. Immune cells originating from the bone marrow trigger immune responses in the lungs while intestinal immune cells migrate directly from the intestine to the lungs via the bloodstream, influencing pulmonary immune activity[54]. Group 2 innate lymphoid cells (ILC2s) rapidly migrate from the intestine to the lungs following helminth infection, and circulating ILC2s have been observed in the blood in experimental settings. These early responders enhance type 2 immunity and barrier repair[55]. Specific populations, including ILC2s, ILC3s, and T helper 17 cells (Th17) cells, can migrate from the gut to the lungs via circulation, a process in which ILCs depend on sphingosine-1-phosphate signaling, and directly participate in dysregulated immune responses[56,57]. Furthermore, gut Pseudomonadota facilitate ILC2 trafficking along the gut-lung axis. While IL-33 and CXCL16 guide natural ILC2 accumulation in the lungs, the IL-25 and CCL25 axis promotes inflammatory ILC2 migration to the intestine, collectively modulating axis-specific inflammation[58].
In ALI/ARDS regulatory T cells (Treg cells) provide protection by suppressing pulmonary inflammation, whereas Th17 cells exacerbate pulmonary injury. Additionally, gamma delta T cells (γδ T cells), antibody-secreting cells, and ILCs play roles in pulmonary immune responses and are important for ALI/ARDS[59]. Notably, SCFAs entering the peri
The intestinal mucosal immune barrier orchestrates immune cell migration along the gut-lung axis by activating immune cells, delivering microbial metabolites, maintaining barrier integrity, and regulating immune polarization[61]. For instance, during sepsis alveolar macrophages activate the Wnt/β-catenin-Lef1 pathway, upregulate CCL1, and recruit intestinal γδ T17 cells via CCR8. These lung-infiltrating γδ T17 cells exacerbate injury through the secretion of IL-17A, which promotes neutrophil infiltration, M1 polarization, and NETosis[62]. Although gut-derived immune cells are clearly important in AP-ALI, their migration pathways require further elucidation. A deeper understanding of their precise roles will facilitate the development of targeted immunotherapies for SAP-ALI.
Defined as live microorganisms that colonize the gut and maintain intestinal homeostasis[63,64], probiotics such as Bifidobacterium and Lactobacillus species protect against SAP-ALI through multiple mechanisms[65,66]: Remodeling the microbiota to increase SCFA production; enhancing intestinal barrier integrity; inhibiting pathogenic bacteria; and reinforcing mucosal immunity[67-70]. Specific mixtures can also promote anti-inflammatory M2 macrophage polarization via elevation of butyrate levels[71]. Clinical evidence includes a randomized trial showing that a Bacillus subtilis and Enterococcus faecium blend reduced hospitalization duration in patients with AP and reports indicating that strains such as Lactobacillus reuteri and Saccharomyces boulardii lower BT and SAP-ALI incidence[72-75].
Prebiotics, defined as non-digestible food ingredients that selectively stimulate beneficial gut bacteria, confer health advantages to the host[76]. For instance, inulin suppresses pathogenic bacteria such as Shigella and Klebsiella while enriching the probiotic Akkermansia[77]. Shi et al[78] further demonstrated that fritillary polysaccharides modulate the gut–lung immune axis by regulating the differentiation and migration of CCR6+ Th17/CCR6+ Treg cells while also enhancing intestinal barrier integrity and immunoglobulin A production. Although generally safe, the application of probiotics and prebiotics requires careful safety monitoring and should be tailored to disease severity and variations in individual patients. Future studies must focus on optimizing strain selection, dosage, and treatment duration and elucidating the precise mechanisms to validate the efficacy of probiotics and prebiotics in SAP-ALI.
The intestinal lymphatic system serves as a major route for transporting gut-derived bacteria and toxins to distant organs with the lungs being the primary target because of their direct drainage from the mesenteric lymph via the thoracic duct[34]. Animal studies have consistently demonstrated that interrupting mesenteric lymph flow through ligation in models of shock, trauma, burns, or ischemia/reperfusion effectively attenuates lung injury[79,80]. This protective effect is mediated through the inhibition of key inflammatory pathways, including HMGB1/RAGE/NF-κB signaling, leading to reduced proinflammatory cytokine release and the downregulation of NF-κB, iNOS, ICAM-1, and TLR4 expression[81]. These findings suggest that mesenteric lymphatic ligation could represent a novel therapeutic strategy for SAP-ALI.
Several Chinese herbal compounds and monomers, often acting via the gut-lung axis following oral administration, have been shown to have clinical utility in patients with SAP. For instance, multiple studies have shown that rhubarb-derived rhubarbin alleviates SAP (SAP-ALI)[82-84]. Qingyi decoction, a common multi-herb formula, exerts multi-target effects; it has been shown to reduce systemic inflammation and restore the intestinal barrier function via the SCFA-mediated AMPK/NF-κB/NLRP3 pathway[41,85]. Chaihuang Qingfu Pills, a traditional Chinese medicine formula modified from Qingyi decoction, was able to reduce matrix metalloprotease 9 and NLRP3 to inhibit sepsis in alveolar macrophages and protect against SAP-ALI[86]. Other active ingredients, such as those from Zingiber officinale, ameliorate pancreatic and lung injury by reducing oxidative stress and inflammation, and the injectable formulation hemopexin has also been shown to be effective against SAP-ALI/ARDS[87,88].
The increasing recognition of the gut–lung axis in SAP-ALI prompted this review. We summarized how SAP-induced gut microbiota dysbiosis impairs intestinal barrier function, facilitating BT, and how gut immune imbalances drive the migration of immune cells along the gut-lung axis, thereby amplifying pulmonary inflammation. Special attention should be given to SCFAs and their role in preserving barrier integrity and attenuating lung injury. Finally, we discussed potential interventions targeting these pathways, offering new strategies for clinical management.
Despite recent advances, several limitations and unanswered questions remain. The molecular mechanisms governing gut-lung cross talk in SAP-ALI require further elucidation, and current evidence, largely derived from animal studies, necessitates clinical validation. Future research should focus on delineating the migration routes and precise functions of immune cells along the gut-lung axis, revealing novel diagnostic and therapeutic targets. A deeper understanding of these pathways will undoubtedly open new avenues for preventing and treating SAP-ALI.
| 1. | Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG, Tsiotos GG, Vege SS; Acute Pancreatitis Classification Working Group. Classification of acute pancreatitis--2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013;62:102-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5667] [Cited by in RCA: 4802] [Article Influence: 369.4] [Reference Citation Analysis (7)] |
| 2. | Lankisch PG, Apte M, Banks PA. Acute pancreatitis. Lancet. 2015;386:85-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1000] [Cited by in RCA: 893] [Article Influence: 81.2] [Reference Citation Analysis (1)] |
| 3. | Wu L, Hu J, Yi X, Lv J, Yao J, Tang W, Zhang S, Wan M. Gut microbiota interacts with inflammatory responses in acute pancreatitis. Therap Adv Gastroenterol. 2023;16:17562848231202133. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 4. | Lee PJ, Papachristou GI. New insights into acute pancreatitis. Nat Rev Gastroenterol Hepatol. 2019;16:479-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 696] [Cited by in RCA: 625] [Article Influence: 89.3] [Reference Citation Analysis (2)] |
| 5. | Garg PK, Singh VP. Organ Failure Due to Systemic Injury in Acute Pancreatitis. Gastroenterology. 2019;156:2008-2023. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 533] [Cited by in RCA: 466] [Article Influence: 66.6] [Reference Citation Analysis (1)] |
| 6. | Lin X, Yu Z, Liu Y, Li C, Hu H, Hu JC, Liu M, Yang Q, Gu P, Li J, Nandakumar KS, Hu G, Zhang Q, Chen X, Ma H, Huang W, Wang G, Wang Y, Huang L, Wu W, Liu NN, Zhang C, Liu X, Zheng L, Chen P. Gut-X axis. Imeta. 2025;4:e270. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 61] [Article Influence: 61.0] [Reference Citation Analysis (2)] |
| 7. | Dickson RP, Singer BH, Newstead MW, Falkowski NR, Erb-Downward JR, Standiford TJ, Huffnagle GB. Enrichment of the lung microbiome with gut bacteria in sepsis and the acute respiratory distress syndrome. Nat Microbiol. 2016;1:16113. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 610] [Cited by in RCA: 512] [Article Influence: 51.2] [Reference Citation Analysis (0)] |
| 8. | Chen X, Chen X, Yan D, Zhang N, Fu W, Wu M, Ge F, Wang J, Li X, Geng M, Wang J, Tang D, Liu J. GV-971 prevents severe acute pancreatitis by remodeling the microbiota-metabolic-immune axis. Nat Commun. 2024;15:8278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 9. | Dang AT, Marsland BJ. Microbes, metabolites, and the gut-lung axis. Mucosal Immunol. 2019;12:843-850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 854] [Cited by in RCA: 741] [Article Influence: 105.9] [Reference Citation Analysis (1)] |
| 10. | Zhang XM, Zhang ZY, Zhang CH, Wu J, Wang YX, Zhang GX. Intestinal Microbial Community Differs between Acute Pancreatitis Patients and Healthy Volunteers. Biomed Environ Sci. 2018;31:81-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 36] [Reference Citation Analysis (0)] |
| 11. | Gong L, Li X, Ji L, Chen G, Han Z, Su L, Wu D. Characterization and comparison of gut microbiota in patients with acute pancreatitis by metagenomics and culturomics. Heliyon. 2025;11:e42243. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 12. | Xie J, Du L, Lu Y, Guo X, Zhou X, Tong Y, Shen B, Yu X, Guo F, Yu H. Akkermansia Muciniphila Alleviates Severe Acute Pancreatitis via Amuc1409-Ube2k-Foxp3 Axis in Regulatory T Cells. Adv Sci (Weinh). 2025;12:e04214. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 13. | Hu X, Han Z, Zhou R, Su W, Gong L, Yang Z, Song X, Zhang S, Shu H, Wu D. Altered gut microbiota in the early stage of acute pancreatitis were related to the occurrence of acute respiratory distress syndrome. Front Cell Infect Microbiol. 2023;13:1127369. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 36] [Reference Citation Analysis (0)] |
| 14. | He C, Wang J, Ding L, Li X, Hu Y, He W, Xia L, Xiong H, Huang X, Lei Y, Luo L, Lu N, Zhu Y. Longitudinal dysbiosis of gut bacteriome and mycobiome in patients with severe acute pancreatitis and its association with mortality. BMC Microbiol. 2025;25:730. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 15. | Wang H, Chen Y, Han Y, Mu S, Wei W, Lan L, Li X, Xiang H, Tong C, Du S. Comparative study of gut microbiota and metabolite variations between severe and mild acute pancreatitis patients at different stages. Microb Pathog. 2025;198:107030. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 16. | Ammer-Herrmenau C, Antweiler KL, Asendorf T, Beyer G, Buchholz SM, Cameron S, Capurso G, Damm M, Dang L, Frost F, Gomes A, Hamm J, Henker R, Hoffmeister A, Meinhardt C, Nawacki L, Nunes V, Panyko A, Pardo C, Phillip V, Pukitis A, Rasch S, Riekstina D, Rinja E, Ruiz-Rebollo ML, Sirtl S, Weingarten M, Sandru V, Woitalla J, Ellenrieder V, Neesse A. Gut microbiota predicts severity and reveals novel metabolic signatures in acute pancreatitis. Gut. 2024;73:485-495. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 33] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 17. | Zhao M, Cui M, Jiang Q, Wang J, Lu Y. Profile of Pancreatic and Ileal Microbiota in Experimental Acute Pancreatitis. Microorganisms. 2023;11:2707. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 18. | Fu Y, Mei Q, Yin N, Huang Z, Li B, Luo S, Xu B, Fan J, Huang C, Zeng Y. Paneth Cells Protect against Acute Pancreatitis via Modulating Gut Microbiota Dysbiosis. mSystems. 2022;7:e0150721. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 19. | Zhang C, Chen S, Wang Z, Zhang J, Yu W, Wang Y, Si W, Zhang Y, Zhang Y, Liang T. Exploring the mechanism of intestinal bacterial translocation after severe acute pancreatitis: the role of Toll-like receptor 5. Gut Microbes. 2025;17:2489768. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 20. | Yang M, Hong G, Jin Y, Li Y, Li G, Hou X. Mucosal-Associated Microbiota Other Than Luminal Microbiota Has a Close Relationship With Diarrhea-Predominant Irritable Bowel Syndrome. Front Cell Infect Microbiol. 2020;10:515614. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 21. | Lavelle A, Lennon G, O'Sullivan O, Docherty N, Balfe A, Maguire A, Mulcahy HE, Doherty G, O'Donoghue D, Hyland J, Ross RP, Coffey JC, Sheahan K, Cotter PD, Shanahan F, Winter DC, O'Connell PR. Spatial variation of the colonic microbiota in patients with ulcerative colitis and control volunteers. Gut. 2015;64:1553-1561. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 174] [Cited by in RCA: 218] [Article Influence: 19.8] [Reference Citation Analysis (4)] |
| 22. | Png CW, Lindén SK, Gilshenan KS, Zoetendal EG, McSweeney CS, Sly LI, McGuckin MA, Florin TH. Mucolytic bacteria with increased prevalence in IBD mucosa augment in vitro utilization of mucin by other bacteria. Am J Gastroenterol. 2010;105:2420-2428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1213] [Cited by in RCA: 1075] [Article Influence: 67.2] [Reference Citation Analysis (3)] |
| 23. | Sanna S, van Zuydam NR, Mahajan A, Kurilshikov A, Vich Vila A, Võsa U, Mujagic Z, Masclee AAM, Jonkers DMAE, Oosting M, Joosten LAB, Netea MG, Franke L, Zhernakova A, Fu J, Wijmenga C, McCarthy MI. Causal relationships among the gut microbiome, short-chain fatty acids and metabolic diseases. Nat Genet. 2019;51:600-605. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1345] [Cited by in RCA: 1153] [Article Influence: 164.7] [Reference Citation Analysis (4)] |
| 24. | Dickson RP, Erb-Downward JR, Freeman CM, McCloskey L, Falkowski NR, Huffnagle GB, Curtis JL. Bacterial Topography of the Healthy Human Lower Respiratory Tract. mBio. 2017;8:e02287-e02216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 356] [Cited by in RCA: 395] [Article Influence: 43.9] [Reference Citation Analysis (0)] |
| 25. | Zhang P, Liu B, Zheng W, Chen Y, Wu Z, Lu Y, Ma J, Lu W, Zheng M, Wu W, Meng Z, Wu J, Zheng Y, Zhang X, Zhang S, Huang Y. Pulmonary Microbial Composition in Sepsis-Induced Acute Respiratory Distress Syndrome. Front Mol Biosci. 2022;9:862570. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 26. | Dickson RP, Schultz MJ, van der Poll T, Schouten LR, Falkowski NR, Luth JE, Sjoding MW, Brown CA, Chanderraj R, Huffnagle GB, Bos LDJ; Biomarker Analysis in Septic ICU Patients (BASIC) Consortium. Lung Microbiota Predict Clinical Outcomes in Critically Ill Patients. Am J Respir Crit Care Med. 2020;201:555-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 302] [Cited by in RCA: 260] [Article Influence: 43.3] [Reference Citation Analysis (1)] |
| 27. | Dickson RP. The Lung Microbiome and ARDS. It Is Time to Broaden the Model. Am J Respir Crit Care Med. 2018;197:549-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 28. | Kyo M, Nishioka K, Nakaya T, Kida Y, Tanabe Y, Ohshimo S, Shime N. Unique patterns of lower respiratory tract microbiota are associated with inflammation and hospital mortality in acute respiratory distress syndrome. Respir Res. 2019;20:246. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 70] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 29. | Dickson RP. The microbiome and critical illness. Lancet Respir Med. 2016;4:59-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 301] [Cited by in RCA: 330] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 30. | Zhang P, Chen Y, Zheng W, Wu M, Wu Z, Lu Y, Zhang S, Zhang X, Huang Y. [Changes of lung microbiome of acute respiratory distress syndrome before and after treatment under open airway]. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2021;33:1063-1068. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 31. | Liu J, Huang L, Luo M, Xia X. Bacterial translocation in acute pancreatitis. Crit Rev Microbiol. 2019;45:539-547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 74] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 32. | Fromentin M, Ricard JD, Roux D. Lung Microbiome in Critically Ill Patients. Life (Basel). 2021;12:7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 33. | Chen C, Lin J, Wang X, Yang S, Duan X, Deng Y, Zeng S, Zhou T, Li J, Yu J, Liang Y, Shen X, Shi R, Wang Y, Shu Z. Novel insights into immune mechanisms in acute lung injury: Focusing on gut microbiota and its metabolites. Microbiol Res. 2025;300:128279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 34. | Deitch EA. Gut lymph and lymphatics: a source of factors leading to organ injury and dysfunction. Ann N Y Acad Sci. 2010;1207 Suppl 1:E103-E111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 143] [Article Influence: 8.9] [Reference Citation Analysis (3)] |
| 35. | Ge P, Luo Y, Okoye CS, Chen H, Liu J, Zhang G, Xu C, Chen H. Intestinal barrier damage, systemic inflammatory response syndrome, and acute lung injury: A troublesome trio for acute pancreatitis. Biomed Pharmacother. 2020;132:110770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 165] [Article Influence: 27.5] [Reference Citation Analysis (2)] |
| 36. | Liang XY, Jia TX, Zhang M. Intestinal bacterial overgrowth in the early stage of severe acute pancreatitis is associated with acute respiratory distress syndrome. World J Gastroenterol. 2021;27:1643-1654. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 12] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 37. | Li L, Liu S, Zhang X, He W, Zhu P, Shi J, Wang W, Sun X, Shi N, Xia L, Lu N, Philips AR, Singh VK, Sutton R, Zhu Y, Huang W, Windsor JA, Deng L, Jin T, Xia Q. Predicting Persistent Acute Respiratory Failure in Acute Pancreatitis: The Accuracy of Two Lung Injury Indices. Dig Dis Sci. 2023;68:2878-2889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 38. | Tang J, Xu L, Zeng Y, Gong F. Effect of gut microbiota on LPS-induced acute lung injury by regulating the TLR4/NF-kB signaling pathway. Int Immunopharmacol. 2021;91:107272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 419] [Cited by in RCA: 367] [Article Influence: 73.4] [Reference Citation Analysis (1)] |
| 39. | Hashimoto Y, Eguchi A, Wei Y, Shinno-Hashimoto H, Fujita Y, Ishima T, Chang L, Mori C, Suzuki T, Hashimoto K. Antibiotic-induced microbiome depletion improves LPS-induced acute lung injury via gut-lung axis. Life Sci. 2022;307:120885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 40. | Sivaprakasam S, Bhutia YD, Yang S, Ganapathy V. Short-Chain Fatty Acid Transporters: Role in Colonic Homeostasis. Compr Physiol. 2017;8:299-314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 197] [Article Influence: 21.9] [Reference Citation Analysis (4)] |
| 41. | Wang Z, Liu J, Li F, Ma S, Zhao L, Ge P, Wen H, Zhang Y, Liu X, Luo Y, Yao J, Zhang G, Chen H. Mechanisms of Qingyi Decoction in Severe Acute Pancreatitis-Associated Acute Lung Injury via Gut Microbiota: Targeting the Short-Chain Fatty Acids-Mediated AMPK/NF-κB/NLRP3 Pathway. Microbiol Spectr. 2023;11:e0366422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 51] [Reference Citation Analysis (0)] |
| 42. | Liu Q, Tian X, Maruyama D, Arjomandi M, Prakash A. Lung immune tone via gut-lung axis: gut-derived LPS and short-chain fatty acids' immunometabolic regulation of lung IL-1β, FFAR2, and FFAR3 expression. Am J Physiol Lung Cell Mol Physiol. 2021;321:L65-L78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 128] [Article Influence: 25.6] [Reference Citation Analysis (0)] |
| 43. | Li N, Liu XX, Hong M, Huang XZ, Chen H, Xu JH, Wang C, Zhang YX, Zhong JX, Nie H, Gong Q. Sodium butyrate alleviates LPS-induced acute lung injury in mice via inhibiting HMGB1 release. Int Immunopharmacol. 2018;56:242-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 62] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 44. | Liu J, Chang G, Huang J, Wang Y, Ma N, Roy AC, Shen X. Sodium Butyrate Inhibits the Inflammation of Lipopolysaccharide-Induced Acute Lung Injury in Mice by Regulating the Toll-Like Receptor 4/Nuclear Factor κB Signaling Pathway. J Agric Food Chem. 2019;67:1674-1682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 83] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 45. | Xia H, Guo J, Shen J, Jiang S, Han S, Li L. Butyrate ameliorated the intestinal barrier dysfunction and attenuated acute pancreatitis in mice fed with ketogenic diet. Life Sci. 2023;334:122188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 46. | Karoor V, Strassheim D, Sullivan T, Verin A, Umapathy NS, Dempsey EC, Frank DN, Stenmark KR, Gerasimovskaya E. The Short-Chain Fatty Acid Butyrate Attenuates Pulmonary Vascular Remodeling and Inflammation in Hypoxia-Induced Pulmonary Hypertension. Int J Mol Sci. 2021;22:9916. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 72] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 47. | Tian X, Hellman J, Horswill AR, Crosby HA, Francis KP, Prakash A. Corrigendum: Elevated Gut Microbiome-Derived Propionate Levels Are Associated With Reduced Sterile Lung Inflammation and Bacterial Immunity in Mice. Front Microbiol. 2019;10:518. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 48. | Wang YH, Yan ZZ, Luo SD, Hu JJ, Wu M, Zhao J, Liu WF, Li C, Liu KX. Gut microbiota-derived succinate aggravates acute lung injury after intestinal ischaemia/reperfusion in mice. Eur Respir J. 2023;61:2200840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 92] [Article Influence: 30.7] [Reference Citation Analysis (0)] |
| 49. | Lv Y, Jiang M, Ouyang Y, Zheng X, Zhang L, Yao J, Hu L, Zhao J, Li Z, Wang S. Sodium Butyrate-Loaded Microspheres With Enhanced Bioavailability for Targeted Treatment of Intestinal Barrier Injury. Adv Healthc Mater. 2025;14:e2402773. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 50. | Wang Z, Liu J, Li F, Luo Y, Ge P, Zhang Y, Wen H, Yang Q, Ma S, Chen H. The gut-lung axis in severe acute Pancreatitis-associated lung injury: The protection by the gut microbiota through short-chain fatty acids. Pharmacol Res. 2022;182:106321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 70] [Reference Citation Analysis (0)] |
| 51. | Yan X, Li J, Wu D. The Role of Short-Chain Fatty Acids in Acute Pancreatitis. Molecules. 2023;28:4985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 52. | Liu Q, Zhu X, Guo S. From pancreas to lungs: The role of immune cells in severe acute pancreatitis and acute lung injury. Immun Inflamm Dis. 2024;12:e1351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 53. | Mititelu A, Grama A, Colceriu MC, Pop TL. Overview of the cellular and immune mechanisms involved in acute pancreatitis: In search of new prognosis biomarkers. Expert Rev Mol Med. 2025;27:e9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 54. | Ma PJ, Wang MM, Wang Y. Gut microbiota: A new insight into lung diseases. Biomed Pharmacother. 2022;155:113810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 119] [Reference Citation Analysis (0)] |
| 55. | Can UI, Stenske SE, Rosenbaum MD, Reinhardt RL. Rapid group-2 innate lymphoid cell mobilization from the intestine aids in early lung defense and repair. Cell Rep. 2025;44:115868. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 56. | Mjösberg J, Rao A. Lung inflammation originating in the gut. Science. 2018;359:36-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 74] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 57. | Miskolci V, Klemm LC, Huttenlocher A. Cell Migration Guided by Cell-Cell Contacts in Innate Immunity. Trends Cell Biol. 2021;31:86-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 58. | Pu Q, Lin P, Gao P, Wang Z, Guo K, Qin S, Zhou C, Wang B, Wu E, Khan N, Xia Z, Wei X, Wu M. Gut Microbiota Regulate Gut-Lung Axis Inflammatory Responses by Mediating ILC2 Compartmental Migration. J Immunol. 2021;207:257-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 79] [Article Influence: 15.8] [Reference Citation Analysis (4)] |
| 59. | Ziaka M, Exadaktylos A. Gut-derived immune cells and the gut-lung axis in ARDS. Crit Care. 2024;28:220. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 51] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 60. | Zhao M, Chu J, Feng S, Guo C, Xue B, He K, Li L. Immunological mechanisms of inflammatory diseases caused by gut microbiota dysbiosis: A review. Biomed Pharmacother. 2023;164:114985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 347] [Cited by in RCA: 265] [Article Influence: 88.3] [Reference Citation Analysis (1)] |
| 61. | Li F, Wang Z, Cao Y, Pei B, Luo X, Liu J, Ge P, Luo Y, Ma S, Chen H. Intestinal Mucosal Immune Barrier: A Powerful Firewall Against Severe Acute Pancreatitis-Associated Acute Lung Injury via the Gut-Lung Axis. J Inflamm Res. 2024;17:2173-2193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 15] [Reference Citation Analysis (1)] |
| 62. | Xie B, Wang M, Zhang X, Zhang Y, Qi H, Liu H, Wu Y, Wen X, Chen X, Han M, Xu D, Sun X, Zhang X, Zhao X, Shang Y, Yuan S, Zhang J. Gut-derived memory γδ T17 cells exacerbate sepsis-induced acute lung injury in mice. Nat Commun. 2024;15:6737. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 60] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 63. | Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B, Morelli L, Canani RB, Flint HJ, Salminen S, Calder PC, Sanders ME. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014;11:506-514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8109] [Cited by in RCA: 6162] [Article Influence: 513.5] [Reference Citation Analysis (14)] |
| 64. | Meng C, Bai C, Brown TD, Hood LE, Tian Q. Human Gut Microbiota and Gastrointestinal Cancer. Genomics Proteomics Bioinformatics. 2018;16:33-49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 382] [Cited by in RCA: 312] [Article Influence: 39.0] [Reference Citation Analysis (6)] |
| 65. | Gu WJ, Liu JC. Probiotics in patients with severe acute pancreatitis. Crit Care. 2014;18:446. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 66. | Zhu Y, Mei Q, Fu Y, Zeng Y. Alteration of gut microbiota in acute pancreatitis and associated therapeutic strategies. Biomed Pharmacother. 2021;141:111850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 67. | Wang Z, Guo M, Yang S, Chen Y, Cheng J, Huang Z, Wang T, Luo X, He X, Wang D, Xu X. Intestinal microflora and metabolites affect the progression of acute pancreatitis (AP). Gut Pathog. 2024;16:64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 68. | Liu Y, Hoang TK, Taylor CM, Park ES, Freeborn J, Luo M, Roos S, Rhoads JM. Limosilactobacillus reuteri and Lacticaseibacillus rhamnosus GG differentially affect gut microbes and metabolites in mice with Treg deficiency. Am J Physiol Gastrointest Liver Physiol. 2021;320:G969-G981. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 69. | Zhang T, Gao G, Sakandar HA, Kwok LY, Sun Z. Gut Dysbiosis in Pancreatic Diseases: A Causative Factor and a Novel Therapeutic Target. Front Nutr. 2022;9:814269. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 70. | Wang C, Xiao Y, Yu L, Tian F, Zhao J, Zhang H, Chen W, Zhai Q. Protective effects of different Bacteroides vulgatus strains against lipopolysaccharide-induced acute intestinal injury, and their underlying functional genes. J Adv Res. 2022;36:27-37. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 102] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 71. | Li M, Pan T, Yu J, Wang X, Gao R, Wang Y, Zhao D, Zhou X, Li H, Guo J, Wang N, Huang H, Wang C, Yang G. Butyrate-Producing Mixed Probiotics Alleviate MRSA-Induced Acute Lung Injury in Mice by Promoting M2 Macrophage Polarization. Probiotics Antimicrob Proteins. 2025;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 72. | Takauji S, Konishi H, Fujiya M, Ueno N, Tanaka H, Sato H, Isozaki S, Kashima S, Moriichi K, Mizukami Y, Okumura T. Polyphosphate, Derived from Lactobacillus brevis, Modulates the Intestinal Microbiome and Attenuates Acute Pancreatitis. Dig Dis Sci. 2021;66:3872-3884. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 73. | Wu H, Xie S, Miao J, Li Y, Wang Z, Wang M, Yu Q. Lactobacillus reuteri maintains intestinal epithelial regeneration and repairs damaged intestinal mucosa. Gut Microbes. 2020;11:997-1014. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 324] [Cited by in RCA: 295] [Article Influence: 49.2] [Reference Citation Analysis (0)] |
| 74. | Karen M, Yuksel O, Akyürek N, Ofluoğlu E, Cağlar K, Sahin TT, Paşaoğlu H, Memiş L, Akyürek N, Bostanci H. Probiotic agent Saccharomyces boulardii reduces the incidence of lung injury in acute necrotizing pancreatitis induced rats. J Surg Res. 2010;160:139-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 75. | Wan YD, Zhu RX, Bian ZZ, Sun TW. Effect of probiotics on length of hospitalization in mild acute pancreatitis: A randomized, double-blind, placebo-controlled trial. World J Gastroenterol. 2021;27:224-232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 9] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (2)] |
| 76. | Gibson GR, Hutkins R, Sanders ME, Prescott SL, Reimer RA, Salminen SJ, Scott K, Stanton C, Swanson KS, Cani PD, Verbeke K, Reid G. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat Rev Gastroenterol Hepatol. 2017;14:491-502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4684] [Cited by in RCA: 3545] [Article Influence: 393.9] [Reference Citation Analysis (6)] |
| 77. | Li X, Zheng P, Zou Y, Guan L, Li N, Liu J, Lu N, Zhu Y, He C. Dietary inulin ameliorates obesity-induced severe acute pancreatitis via gut-pancreas axis. Gut Microbes. 2024;16:2436949. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 78. | Shi CC, Zhu HY, Li H, Zeng DL, Shi XL, Zhang YY, Lu Y, Ling LJ, Wang CY, Chen DF. Regulating the balance of Th17/Treg cells in gut-lung axis contributed to the therapeutic effect of Houttuynia cordata polysaccharides on H1N1-induced acute lung injury. Int J Biol Macromol. 2020;158:52-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 58] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 79. | Ma Y, Zabell T, Creasy A, Yang X, Chatterjee V, Villalba N, Kistler EB, Wu MH, Yuan SY. Gut Ischemia Reperfusion Injury Induces Lung Inflammation via Mesenteric Lymph-Mediated Neutrophil Activation. Front Immunol. 2020;11:586685. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 42] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 80. | Tong H, Chen R, Yin H, Shi X, Lu J, Zhang M, Yu B, Wu M, Wen Q, Su L. Mesenteric Lymph Duct Ligation Alleviating Lung Injury in Heatstroke. Shock. 2016;46:696-703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 81. | Tang Y, Kong J, Zhou B, Wang X, Liu X, Wang Y, Zhu S. Mesenteric Lymph Duct Ligation Alleviates Acute Lung Injury Caused by Severe Acute Pancreatitis Through Inhibition of High Mobility Group Box 1-Induced Inflammation in Rats. Dig Dis Sci. 2021;66:4344-4353. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 82. | Chen Z, Dong X, Song Y, Lan B, Luo Y, Wen H, Chen H. Emodin: an alveolar macrophage protector in acute pancreatitis induced lung injury. Int J Med Sci. 2025;22:2075-2087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 83. | Shen G, Wen H, Li H, Zhang X, Lan B, Dong X, Ge P, Luo Y, Chen H. Emodin protects against severe acute pancreatitis-associated acute lung injury by activating Nrf2/HO-1/GPX4 signal and inhibiting ferroptosis in vivo and in vitro. BMC Gastroenterol. 2025;25:57. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (3)] |
| 84. | Xu Q, Wang M, Guo H, Liu H, Zhang G, Xu C, Chen H. Emodin Alleviates Severe Acute Pancreatitis-Associated Acute Lung Injury by Inhibiting the Cold-Inducible RNA-Binding Protein (CIRP)-Mediated Activation of the NLRP3/IL-1β/CXCL1 Signaling. Front Pharmacol. 2021;12:655372. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 85. | An Y, Tu Z, Wang A, Gou W, Yu H, Wang X, Xu F, Li Y, Wang C, Li J, Zhang M, Xiao M, Di Y, Hou W, Cui Y. Qingyi decoction and its active ingredients ameliorate acute pancreatitis by regulating acinar cells and macrophages via NF-κB/NLRP3/Caspase-1 pathways. Phytomedicine. 2025;139:156424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 22] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 86. | Xiao W, Shi H, Tian Y, Chen F, Xie Y, Han X, Zhang X, Cao Y, Liu W, Zhu Y, Liu Y, Jiang Y. Chaihuang Qingfu Pills Protect Against Acute Pancreatitis-Associated Acute Lung Injury Through MMP9-NLRP3-Pyroptosis Pathway. J Inflamm Res. 2025;18:2317-2338. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 87. | Bin Y, Peng R, Lee Y, Lee Z, Liu Y. Efficacy of Xuebijing injection on pulmonary ventilation improvement in acute pancreatitis: a systematic review and meta-analysis. Front Pharmacol. 2025;16:1549419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 24] [Reference Citation Analysis (0)] |
| 88. | Soliman GA, Alamri MA, Abdel-Rahman RF, Elbaset MA, Ogaly HA, Abdel-Kader MS. Tephrosia purpurea, with (-)-Pseudosemiglabrin as the Major Constituent, Alleviates Severe Acute Pancreatitis-Mediated Acute Lung Injury by Modulating HMGB1 and IL-22. Int J Mol Sci. 2025;26:2572. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |