CASE PRESENTATION
Chief complaints
A 61-year-old male was admitted to the hospital with intermittent right upper abdominal pain for 1 week on August 16, 2024.
History of present illness
A week ago, the patient presented with right upper abdominal pain with low-grade fever without any obvious cause.
History of past illness
The patient had no previous history of abdominal surgery.
Personal and family history
The patient denied any history of smoking, drinking, or exposure to toxic substances. He also denied any family history of genetic disease.
Physical examination
Physical examination revealed that his temperature was 37.5 °C. Superficial lymph nodes were not palpable. There was no jaundice or ascites. Abdominal examination showed liver tenderness with non-palpable liver margins. Vital signs were stable, and the body mass index was 24.24 kg/m2.
Laboratory examinations
Laboratory data were remarkable for: White blood cell count 15.56 × 109/L, red blood cell count 4.92 × 1012/L, hemoglobin 12.8 g/dL, platelet count 444 × 109/L, aspartate aminotransferase 15 U/L, alkaline phosphatase 18 U/L, total bilirubin 0.4 mg/dL, and total protein 6.4 g/dL. C-reactive protein was 142.25 mg/L (reference range: < 8.0 mg/L). Tumor markers showed: Cancer antigen (CA) 19-9 12 000 U/mL (reference range: < 34 U/mL), CA12-5 115 U/mL (reference range: < 35 U/mL), and carcinoembryonic antigen 13.60 U/mL (reference range: < 5 U/mL).
Imaging examinations
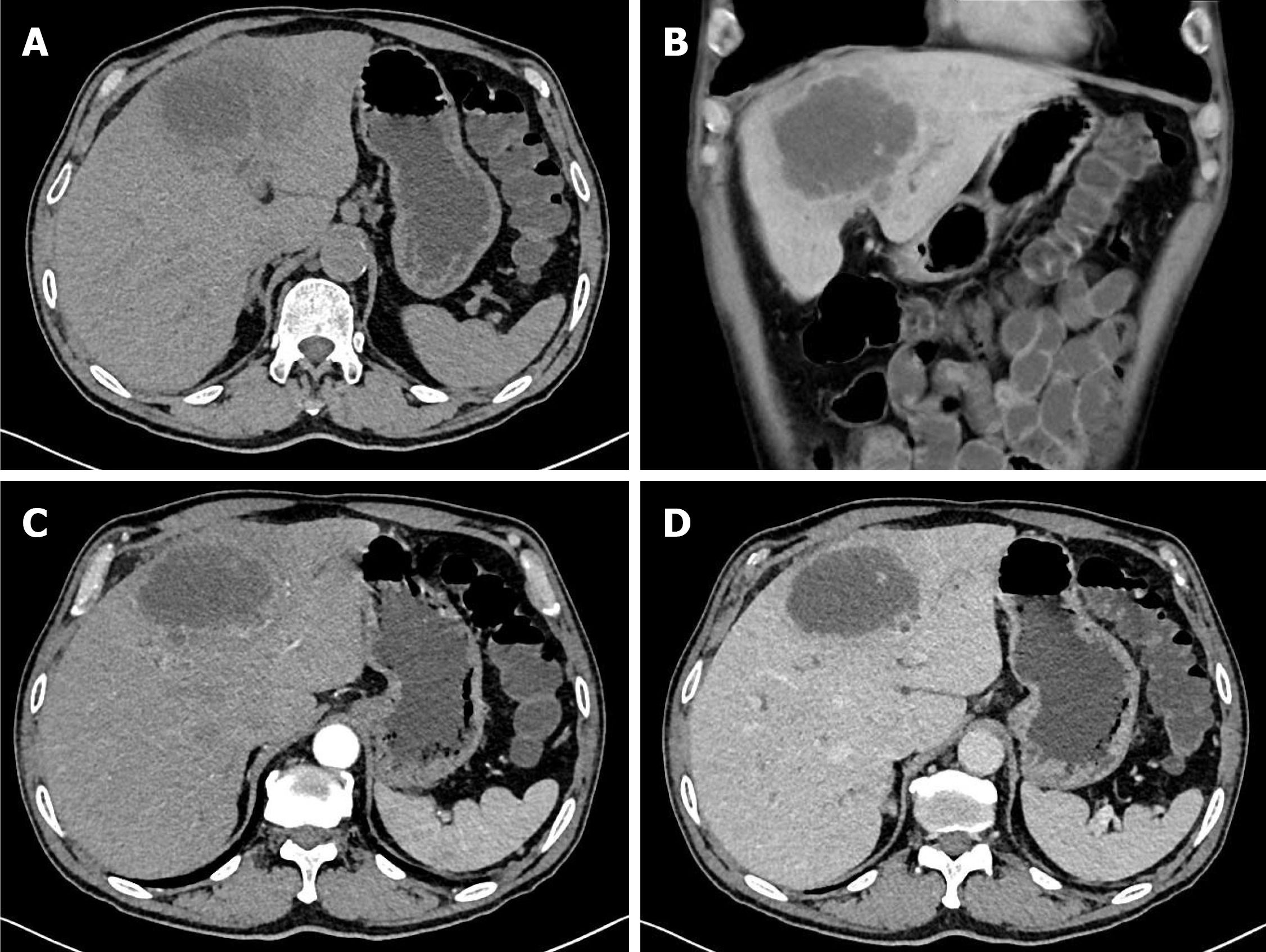
Abdominal computed tomography (CT) disclosed a mass 9 cm in diameter in in the left hepatic lobe (segment II/III). Enhanced abdominal CT showed a peripheral irregular rim-enhanced mass in the left lobe of the liver without lymph node enlargement. Loss of enhancement of the peripheral irregular rim of the mass in the portal phase was considered indicative of a liver abscess in the imaging report (Figure 1).
Figure 1 Computed tomography findings.
A and B: Abdominal computed tomography disclosed a low-density mass, 8 cm in diameter, with central necrosis in the left lobe of the liver; C: Enhanced abdominal computed tomography showed a peripheral irregular rim-enhanced mass in the left lobe of the liver without lymph node enlargement; D: Loss of enhancement of the peripheral irregular rim of the mass in the portal phase.
TREATMENT
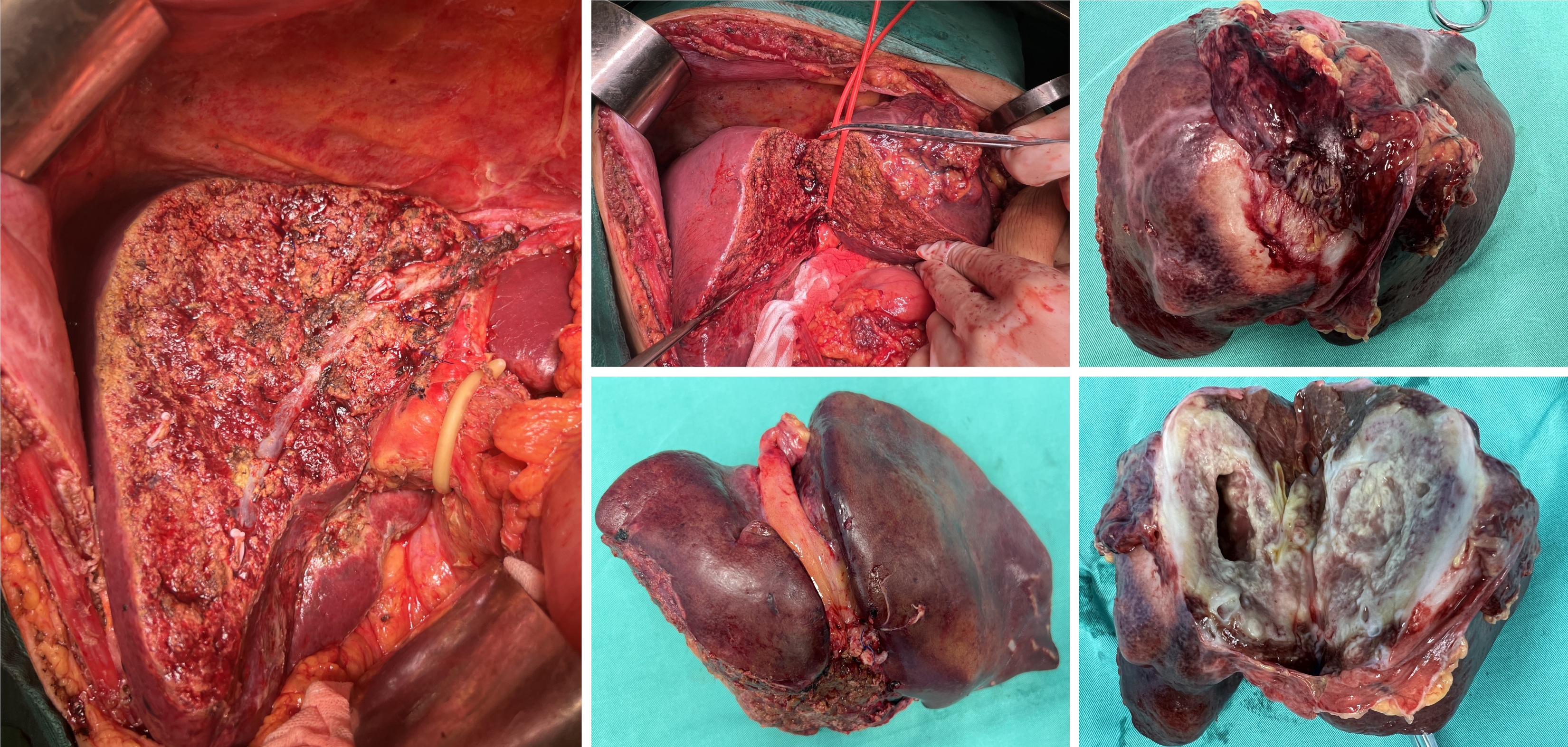
After a multidisciplinary discussion, the patient was diagnosed with ICC and underwent laparotomy 1 week after hospitalization. The patient underwent radical resection for hepatocellular carcinoma under general anesthesia. The procedure was completed in approximately 4 hours with an estimated blood loss of 400 mL, and a peritoneal drain was placed prior to closure. There was a small amount of ascites in the abdominal cavity and a huge multinodular tumor, mainly in the left half of the liver. The lower part of the tumor compressed the upper edge of the pancreas. The cut surface of the resection specimen showed a grayish white solid tumor measuring 9 cm × 7 cm × 6.5 cm with peripheral necrosis (Figure 2). Microscopically, it was observed that the tumor contained two cellular components (30% AC and 70% SCC; Figure 3). The tumor was accompanied by lymph node metastasis, mainly composed of SCC (Figure 4). The patient recovered smoothly without any postoperative complications and was subsequently discharged one week after surgery.
Figure 2
Section of the excised specimen showed a yellowish-white solid tumor measuring 9 cm × 8 cm × 8 cm, with central necrosis.
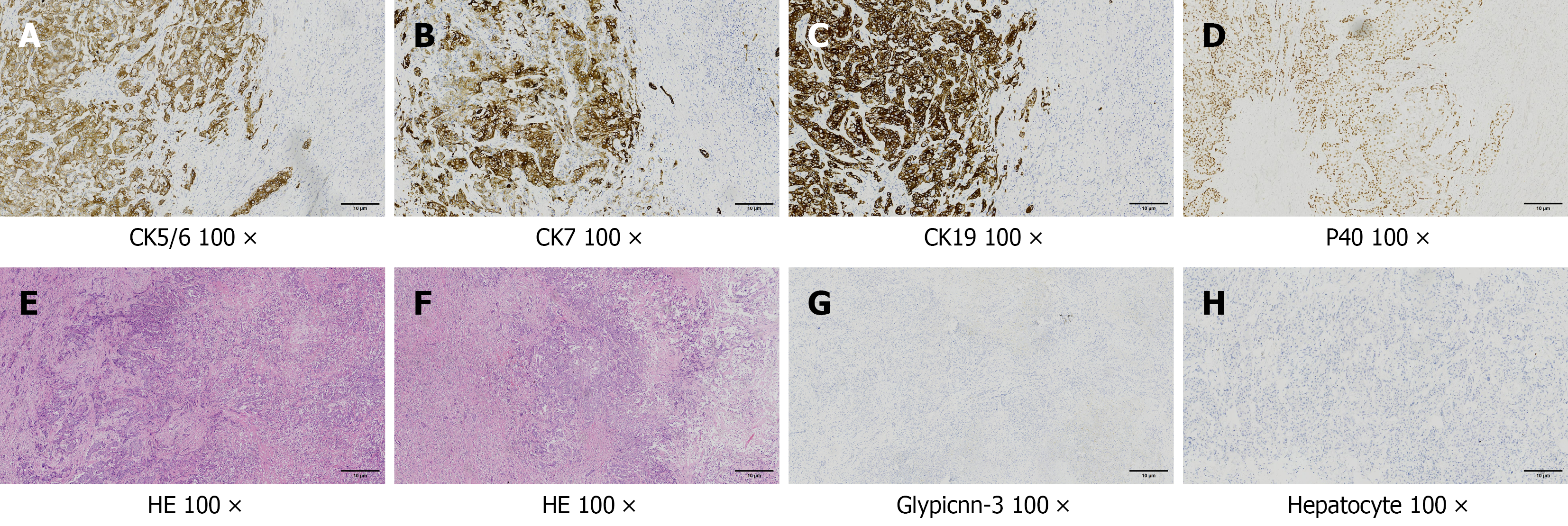
Figure 3 Pathological images of adenosquamous carcinoma in the liver.
A-C: The adenocarcinoma component of the tumor has tubular structures, and the cells show a nest-like arrangement (squamous cell carcinoma component). Positive staining of cytokeratin (CK) 5/6, CK7, and CK19 shows brown and granular deposits in the cytoplasm (× 100); D: Positive staining of protein 40 shows brown and granular deposits in the nucleus (× 100); E and F: The tumor showed atypia, mitosis, solid nests, and tubular structures (hematoxylin and eosin stain, × 100); G and H: Negative staining of glypicnn-3 and hepatocytes in tumor cells (× 100). CK: Cytokeratin; P40: Protein 40; HE: Hematoxylin and eosin.
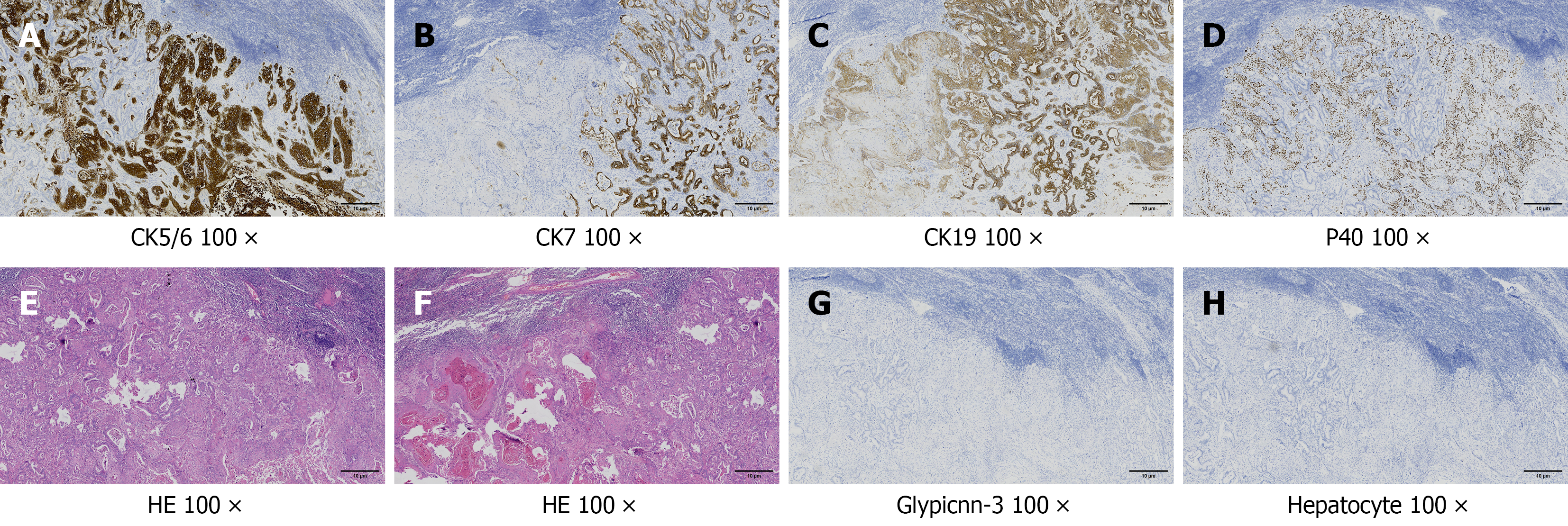
Figure 4 Pathological images of adenosquamous carcinoma in lymph node metastasis.
The adenocarcinoma component of the tumor has tubular structures, and the cells show a nest-like arrangement (squamous cell carcinoma component). A-C: Positive staining of cytokeratin (CK) 5/6, CK7 and CK19 shows brown and granular deposits in the cytoplasm (× 100); D: Positive staining of protein 40 shows brown and granular deposits in the nucleus (× 100); E and F: The tumor showed atypia, mitosis, solid nests, and tubular structures (hematoxylin and eosin stain, × 100); G and H: Negative staining of glypicnn-3 and hepatocytes in tumor cells (× 100). CK: Cytokeratin; P40: Protein 40; HE: Hematoxylin and eosin.
DISCUSSION
Since the first report of ASC of the liver by Pianzola and Durt[3], nearly 100 cases have been reported worldwide[4,8,9]. Although ASC occurs more frequently in organs such as the stomach, pancreas, gallbladder, and thyroid, primary manifestation in the liver is rare[10,11]. Consequently, significant opportunities remain for advancing the diagnostic and therapeutic strategies for hepatic ASC.
According to available reports, ASC contains both AC and SCC components, and the ratio of the two components is not fixed[12]. Squamous differentiation is evidenced by individual cell keratinization, intercellular bridges, keratin pearl formation, and/or dyskeratosis and glandular differentiation by various-sized gland formations and intracellular and intraluminal mucin[13]. Squamous components, which are associated with higher metastatic potential, as reported in previous studies, may contribute to the aggressive behavior observed here[14,15].
ASC is often misdiagnosed due to its rarity and overlapping imaging features with other liver pathologies[16]. In this case, the initial CT interpretation suggested a liver abscess, highlighting the limitations of imaging alone. Although elevated tumor markers, notably CA19-9, prompted consideration of malignancy, definitive diagnosis remained contingent upon histopathological confirmation. The etiology of liver abscesses is predominantly bacterial or parasitic in origin[17]. For the detection of liver abscess, CT imaging demonstrates superior sensitivity to ultrasonography (97% vs 85%)[18]. Given that the incidence of liver fluke infection is high in the area where the patient is located[19], combined with the imaging report, the patient was initially highly suspected of having liver abscess. However, liver abscesses caused by parasitic infections usually do not lead to a significant increase in CA19-9[20].
ICC is a rare and aggressive malignancy, representing the second most common primary liver cancer worldwide and accounting for up to 20% of all hepatic malignancies and 3% of all gastrointestinal cancers[21,22]. Established risk factors for ICC include fibropolycystic liver diseases (such as choledochal cysts), primary sclerosing cholangitis, hepatolithiasis, cirrhosis, viral hepatitis, parasitic infestations (e.g., liver fluke), and exposure to carcinogens (including nitrosamines and thorotrast). ICC typically manifests as a desmoplastic parenchymal liver lesion that is predominantly supplied by the portal venous system and demonstrates delayed phase enhancement on cross-sectional imaging[23]. On contrast-enhanced CT imaging, ICC characteristically demonstrates heterogeneous enhancement, often exhibiting peripheral rim enhancement during the arterial or portal venous phase, with progressive central contrast retention and delayed-phase hyperattenuation typically seen 3-10 minutes after contrast administration[24]. These imaging characteristics are consistent with the patient's radiological findings. Consequently, following multidisciplinary evaluation, a provisional diagnosis of ICC was established. ASC primarily affects older adults and typically manifests clinically with symptoms such as right upper quadrant pain, abdominal distension, and jaundice[15], which overlap with those seen in hepatocellular carcinoma or ICC. However, elevated serum levels of carcinoembryonic antigen and CA19-9, as observed in this patient, help distinguish ASC from hepatocellular carcinoma or ICC. So, the diagnosis of primary hepatic ASC depends on the pathological findings The postoperative pathological diagnosis in this case was ASC. The immunohistochemical results showed cytokeratin (CK) 5/6 (+), CK7 (+), CK19 (+), and protein 40 (+).
Currently, the primary treatment for hepatic ASC is surgical resection. However, most patients are diagnosed at an advanced stage and are not candidates for curative surgery. In this case, the patient received adjuvant targeted therapy (lenvatinib) in combination with immunotherapy (envolizumab). A review 4 months after surgery, no signs of recurrence were found in the liver. However, surveillance imaging performed ten months postoperatively revealed multiple intrahepatic recurrent lesions. Based on the finding of recurrent disease, the decision was made to proceed with transarterial chemoembolization. Follow-up imaging at one year postoperatively demonstrated progressive recurrent disease. As of the last follow-up at 14 months post-diagnosis, the patient remains alive. This approach has demonstrated limited efficacy, highlighting the necessity for innovative treatment modalities. A retrospective analysis of 15 patients with hepatocellular AC treated at this center over the past decade revealed a median overall survival of 6 months. Notably, the 11 patients who underwent surgical resection demonstrated significantly prolonged survival compared to the non-surgical group, with those receiving both surgery and adjuvant chemotherapy exhibiting the most favorable outcomes. In contrast, patients with lymph node metastasis had the poorest prognosis[4]. Therapeutic interventions such as transarterial chemoembolization and systemic chemotherapy have been employed in the management of advanced ASC[4]. Zhang et al[25] described a case of hepatic ASC in which the patient experienced postoperative tumor recurrence and subsequently achieved a survival period of two years following chemotherapy. Nevertheless, due to the rarity of cases, limited data are available, and further clinical studies are needed to validate its efficacy. While this regimen has not been specifically tested in ASC, it provides a potential therapeutic framework for future investigations.
The pathogenesis of ASC in the liver remains unknown, although there are two major hypotheses[13]. Chronic inflammation may cause the metaplastic changes in the biliary epithelium that lead to neoplasia[26], but an alternative theory suggests that ASC of the liver arises from the squamous metaplasia of AC[27-29]. Future efforts to define the molecular landscape of hepatic ASC should integrate multi-omics analyses, including genomics, transcriptomics, and methylomics, to establish a foundation for precision therapy. Parallel advances in liquid biopsy, particularly cfDNA methylation profiling for early cancer detection, hold the potential to revolutionize the early diagnosis of this malignancy through the identification of specific biomarkers[30].