Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.116913
Revised: December 25, 2025
Accepted: February 2, 2026
Published online: March 27, 2026
Processing time: 123 Days and 11.3 Hours
Intrahepatic bile duct stones often come back after treatment. One possible cause is chronic proliferative cholangitis (CPC). This is a long-term inflammation of the bile ducts that can cause scarring and narrowing. This inflammation may remain even after stones are removed. Its effect on stone recurrence and bile duct nar
To determine whether CPC is associated with stone recurrence and bile duct strictures after treatment.
We performed a systematic review and meta-analysis following Preferred Re
Six observational studies including 1248 patients were analyzed. Stone removal was successful in 91% of patients. Stone recurrence occurred in 21.5% of patients. Bile duct narrowing occurred in 18% of patients. Adverse events occurred in 12% of patients. Studies reporting chronic bile duct inflammation showed higher rates of stone recurrence and bile duct narrowing. Differences between studies were moderate.
Stone removal is effective, but stones and bile duct narrowing often return, especially with chronic bile duct inflammation. Treating inflammation may help improve long-term results.
Core Tip: This study reviews the impact of chronic proliferative cholangitis on stone recurrence and biliary stricture formation after the treatment of intrahepatic bile duct stones. Where we have found that removal of stone can be effective but it can be returned with inflammation, and the treatment of that inflammation can improve the long-term results.
- Citation: Muhammad T, He TT, Lin ZY, Shi JL, Zhang TG, Yang Y, Yang XL, Chattha HN, Li J. Impact of chronic proliferative cholangitis on stone recurrence and biliary strictures after intrahepatic stone treatment: A meta-analysis. World J Gastrointest Surg 2026; 18(3): 116913
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/116913.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.116913
Hepatolithiasis the presence of stones within the intrahepatic bile ducts remains a persistent clinical challenge worldwide. Although recent data indicate a decline in incidence in some regions, the disease burden continues, particularly across Asia and among migrant populations[1]. Beyond the symptoms of cholangitis and abdominal pain, hepatolithiasis contributes to progressive biliary injury and long-term complications. One of the principal mechanisms implicated in these processes is chronic proliferative cholangitis (CPC), a state of recurrent ductal inflammation, epithelial hyperplasia, and fibrous stricture formation thought to promote stone recurrence and biliary strictures[2]. The specific role of CPC in the continuum of stone formation, clearance failure, stricture development, and subsequent recurrence, however, remains insufficiently characterized.
CPC is a chronic inflammatory fibroproliferative process of the intrahepatic bile ducts characterized by persistent epithelial injury with mucosal hyperplasia, periductal fibrosis, and progressive ductal narrowing. These changes promote bile stasis and bacterial colonization, creating conditions that favor pigment stone formation and recurrent cholangitis. In patients treated for intrahepatic bile duct stones, CPC is clinically relevant because ongoing inflammation and stricture-prone remodeling can persist after apparent stone clearance, increasing the risk of residual stones, stone recurrence, and new or progressive biliary strictures. The interaction is bidirectional: Stones and infection aggravate ductal inflammation, while CPC-related narrowing and stasis facilitate further stone formation, forming a self-perpetuating cycle.
The global prevalence of hepatolithiasis varies markedly: It is most common in East and Southeast Asia, where rates have historically reached 20%-30% in some populations[3]. In contrast, prevalence in Western countries remains low (< 2%), though migration and evolving dietary patterns may alter these trends[4]. Established risk factors include biliary infection, ductal narrowing or strictures, bile stasis, recurrent cholangitis, and low socioeconomic conditions[5]. The chronic irritation caused by stones may trigger CPC, leading to biliary fibrosis and stricture formation, which in turn impede bile flow and promote new stone development through bile crystallization[6].
The clinical implications are significant. Patients may present with cholangitis, jaundice, hepatomegaly, and over time may progress to hepatic atrophy, cirrhosis, or even intrahepatic cholangiocarcinoma[7]. Treatment outcomes are variable: While complete stone clearance remains the therapeutic goal, many patients develop residual stones, recurrent disease, or biliary strictures[8]. Procedural complications, repeated interventions, and prolonged hospitalization further increase morbidity and healthcare burden[9]. The presence of biliary strictures or recurrent stones often signals an ongoing CPC-driven pathological process, even after apparent clearance.
Recent evidence provides partial insights. Several observational cohorts published between 2019 and 2024 have reported rates of stone clearance, complications, re-intervention, and recurrence following surgical, endoscopic, and percutaneous interventions[10]. While some systematic reviews have examined hepatolithiasis management, few have specifically addressed CPC as a central factor in recurrence or stricture formation. Comparative studies reveal outcome differences between high-resource and low-resource centers and between endemic and non-endemic regions, yet heterogeneity in defining clearance, recurrence, and stricture limits comparability. Notably, regions such as parts of the Middle East and Africa remain underrepresented, and disparities in infrastructure or access such as limited cholangioscopy or interventional radiology may further affect outcomes[11]. In our regional context in China, patient demographics, comorbidities, and healthcare access differ from those described in East Asian studies, suggesting that international results may not always generalize locally.
This article is timely because, despite advances in interventional management, stone recurrence and biliary stricture formation remain frequent and may reflect undiagnosed or inadequately managed CPC. The novelty of this study lies in its synthesis of observational evidence from January 2015 through June 2025 to evaluate the relationship between CPC-related features such as biliary strictures and recurrent cholangitis and subsequent stone recurrence and stricture formation, while explicitly considering infrastructure and access disparities. By moving beyond single-center descriptive reports, this review aims to quantify the contribution of persistent CPC to stone clearance, recurrence, complications, re-intervention rates, and hospital stay. These findings may inform regional policies on surveillance, early stricture detection, and personalized management strategies.
The primary objective is to quantify the association between CPC-related biliary strictures and recurrent cholangitis with stone recurrence in intrahepatic bile duct stones. Secondary objectives include: (1) Assessing rates of complete stone clearance, adverse events or complications, re-interventions, and hospital stay; and (2) Comparing outcomes across healthcare resource levels and anatomical contexts (jejunal anastomosis vs native ducts). We hypothesize that CPC-associated features (strictures or recurrent cholangitis) are linked to higher recurrence, increased complications, more frequent re-intervention, and prolonged hospitalization. This review addresses a critical gap in quantifying CPC-mediated pathways and integrating the impact of access and infrastructure disparities.
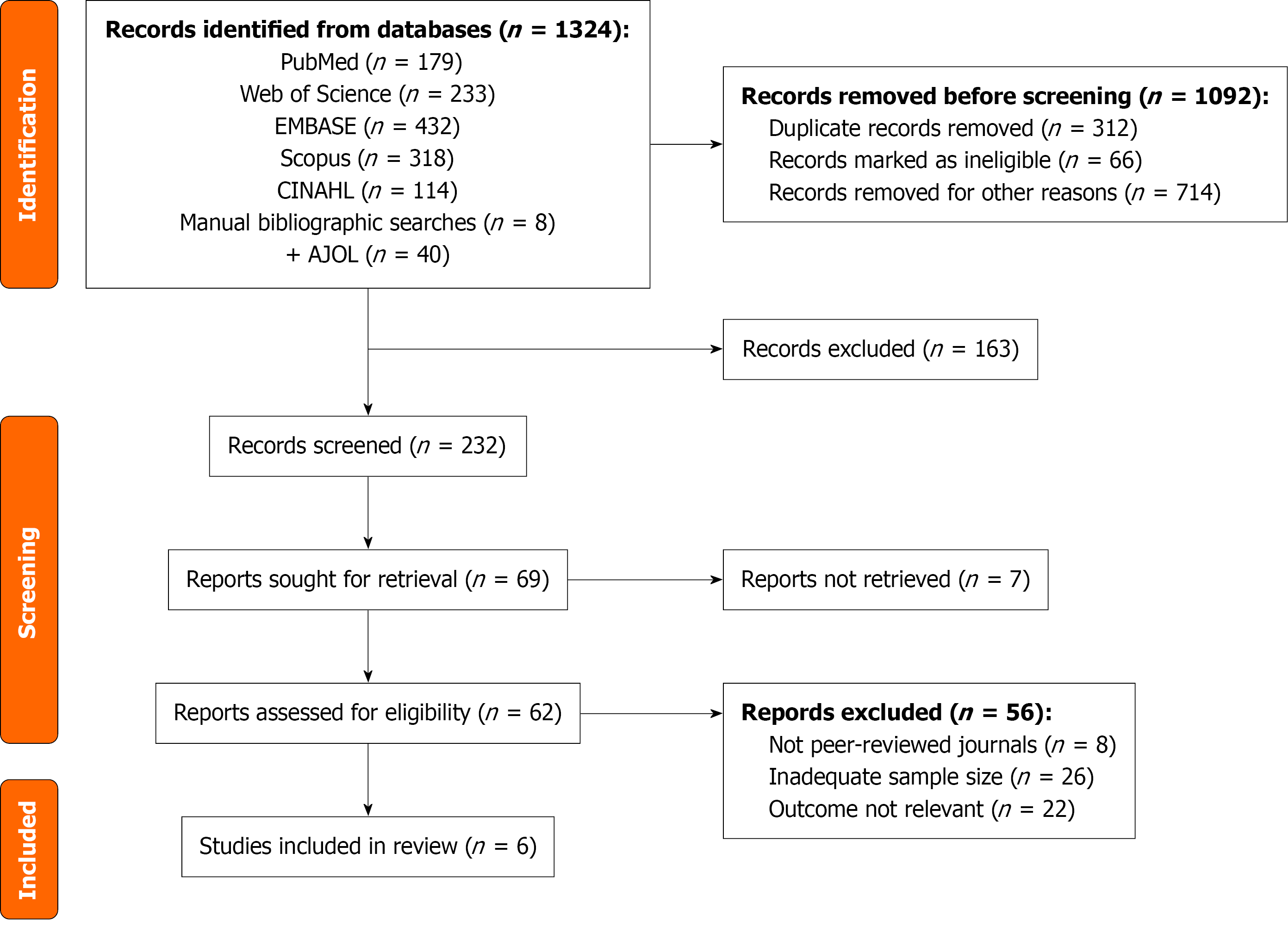
This systematic review and meta-analysis were conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) 2009 guidelines. A completed PRISMA checklist and the study selection flowchart (Figure 1) summarize the screening and inclusion process. The review employed a transparent, structured search strategy designed to ensure reproducibility and comprehensive coverage of the relevant literature.
Boolean operators, Medical Subject Headings, and truncation techniques were applied to maximize sensitivity and specificity. Search was conducted in PubMed, Scopus, Web of Science, and ScienceDirect for studies published between January 2015 and June 2025. The search was restricted to peer-reviewed observational studies involving human subjects and written in English.
The PubMed search string was as follows: [“hepatolithiasis”(Mesh) OR “intrahepatic bile duct stone*”(tiab)] AND [“chronic proliferative cholangitis”(tiab) OR “biliary stricture”(tiab) OR “recurrent cholangitis”(tiab)] AND [“re
Equivalent syntax was adapted for Scopus, Web of Science, and CINAHL databases. Filters included publication year (2015-2025), study design (observational), species (human), and language (English). Reference lists of all included studies were hand-searched to identify any additional eligible publications. The final search was completed in June 2025.
To conduct the systematic review and meta-analysis, operational diagnostic criteria were used to define CPC based on the patterns of reporting in the observational studies that were included. Since CPC, as confirmed by biopsy, was not consistently available, CPC status was defined on histological, radiologic, endoscopic or clinical observation of inflammation and fibroproliferative remodeling of the chronic bile ducts, as in accordance with traditional descriptions of CPC in hepatolithiasis.
The initial search identified 1324 records. After removal of duplicates (n = 312) and exclusion of unrelated topics (n = 780), 232 records remained for title and abstract screening. Full texts of 62 studies were retrieved for detailed review. Following exclusion of non-peer-reviewed reports (n = 8), studies with incomplete or unclear outcome data (n = 22), and those lacking comparable outcome variables (n = 26), six observational studies were included in the final quantitative synthesis (n = 6)[12-17].
We included observational studies involving adults with intrahepatic bile duct stones associated with CPC. Eligible designs comprised cohort and case-control studies published in English in peer-reviewed journals. Studies were required to report at least one of the following outcomes: Stone clearance, residual stones, recurrence, complications, re-intervention, or hospital stay.
We excluded non-human and pediatric studies, reviews, case reports, editorials, letters, and conference abstracts. Studies lacking CPC-related data or quantitative outcome measures were also excluded. Limiting the search to English-language publications was acknowledged as a potential source of bias.
Two reviewers independently screened all titles, abstracts, and full texts. Any discrepancies were resolved through discussion or consultation with a third reviewer. Screening decisions and reasons for exclusion were recorded in an Excel-based audit log to ensure transparency and reproducibility.
In this study, CPC is defined as a chronic inflammatory condition of the intrahepatic bile ducts characterized by persistent bile duct wall thickening, epithelial hyperplasia, and periductal fibrosis, resulting in biliary narrowing or strictures.
For the purpose of this meta-analysis, CPC includes patients with histological, radiological, or endoscopic evidence of chronic bile duct inflammation or fibrotic changes following treatment of intrahepatic bile duct stones, such as surgical resection, percutaneous stone removal, or endoscopic intervention.
CPC is considered present when these chronic bile duct changes are associated with recurrent intrahepatic stone formation and/or persistent biliary strictures during follow-up.
Data collection was performed using a standardized form. Two reviewers independently extracted study data, resolving differences by consensus. Extracted variables included author, publication year, country, study design, sample size, intervention type, follow-up duration, and outcome definitions.
Outcome variables included: (1) Complete stone clearance (no visible stones after the procedure); (2) Residual stone rate (stones remaining immediately post-procedure); (3) Stone recurrence (confirmed stones during follow-up); (4) Procedure-related complications (classified using Clavien-Dindo or American Society for Gastrointestinal Endoscopy criteria); (5) Re-intervention (repeat endoscopic or surgical procedure); and (6) Length of hospital stay (days). Data were entered into Microsoft Excel and analyzed using Review Manager version 5.4.
Risk of bias was independently assessed by two reviewers using the Newcastle-Ottawa Scale. Studies scoring seven or higher were considered low risk. Domains evaluated included selection, comparability, and outcome assessment. Disagreements were resolved by discussion.
Pooled estimates were calculated using a random-effects model to account for clinical heterogeneity. Heterogeneity was quantified using Cochran’s Q test and expressed as τ2, χ2, degrees of freedom, I2, and corresponding P-values. All calculations were done using Review Manager version 5.4 (The Cochrane Collaboration, United Kingdom). Subgroup analyses were performed to compare surgical vs non-surgical interventions and studies from endemic vs non-endemic regions.
Meta-regression assessed the effects of follow-up duration and biliary anatomy on recurrence and stricture rates. Sensitivity analyses were conducted sequentially excluding individual studies. Publication bias was evaluated using funnel plot symmetry.
When data permitted, recurrence and stricture rates were compared directly between CPC and non-CPC groups using random-effects models. Where quantitative synthesis was not feasible, results were summarized descriptively. Studies with high CPC prevalence were compared against those reporting minimal inflammatory features. Analyses also incorporated treatment modality and anatomical variations. Low-quality studies were excluded during sensitivity testing.
Prespecified subgroup analyses were performed according to treatment modality: (1) Resectional surgery (hepatectomy with or without biliary reconstruction); and (2) Non-resectional approaches (endoscopic and/or percutaneous stone clearance). Where sufficient data was available, pooled recurrence and stricture rates were calculated separately for each subgroup using random-effects models. More granular stratification was not feasible due to limited study numbers and heterogeneous reporting.
All six included studies were peer-reviewed and indexed in PubMed. Overall methodological quality was rated as moderate to high. Outcome definitions were standardized prior to pooling, with recurrence defined as imaging confirmed stone reappearance within the study’s follow-up period. Ethical approval was not required as all data was derived from previously published studies. The review adhered to PRISMA 2009 guidelines and followed the ethical principles of the Declaration of Helsinki.
The database search identified 1324 records. After removal of 312 duplicates, 1012 unique records were screened. Of these, 780 were excluded after title and abstract review. Sixty-two full-text articles were assessed, and six observational studies met the inclusion criteria. The selection process is depicted in the PRISMA flow diagram (Figure 1).
The six included studies were published between 2016 and 2024 and conducted in China, Japan, Brazil, and South Korea. All were peer-reviewed, PubMed-indexed observational studies involving patients with intrahepatic bile duct stones associated with CPC and recurrent biliary strictures. Sample sizes ranged from 68 to 420 participants.
Most studies evaluated surgical, endoscopic, or percutaneous interventions for hepatolithiasis. Reported procedures included laparoscopic left hepatectomy, balloon-assisted endoscopy, hepatic resection, and percutaneous transhepatic cholangioscopy. Comparators varied between surgical and non-surgical approaches or among endoscopic techniques. Each study reported at least one of the primary outcomes stone clearance, residual stones, recurrence, adverse events, re-intervention, or hospital stay.
Through studies, patients exhibiting bile duct narrowing or recurrent infections had a higher likelihood of stone recurrence or new stricture formation. Endoscopic and percutaneous treatments following bile duct surgery demon
This matrix summarizes whether each study reported CPC or related pathological features, and whether corresponding outcomes for stone recurrence and biliary stricture were available. Endoscopic studies such as those by Tsutsumi et al[13], Ishihara et al[14], and Hakuta et al[17] more frequently described CPC-related findings, typically noted as recurrent cholangitis or anastomotic narrowing. In contrast, surgical series, including those by Jin et al[12] and Torres et al[15], rarely mentioned CPC explicitly. The matrix underscores variability in how CPC was identified and reported across study types and highlights the need for more standardized histologic or imaging-based documentation of CPC in future research.
Across all studies, complete stone clearance rates were consistently high, ranging from 86% to 95%. Jin et al[12] reported a 94% clearance rate following laparoscopic left hepatectomy. Ishihara et al[14] achieved a 91% clearance rate using balloon-assisted endoscopy after hepaticojejunostomy. Torres et al[15], observed 88% clearance following re
When pooled, the random-effects model showed an overall clearance rate of 90.2% [95% confidence interval (CI): 86.4-93.5; I2 = 41%], indicating moderate heterogeneity. Residual stone rates ranged from 5% to 14%, mainly due to differences in imaging follow-up and technique. Jin et al[12] and Torres et al[15] had the lowest residual rates, below 7%, in re
Publication bias was assessed visually using funnel plots. Formal statistical tests for funnel plot asymmetry were not performed because fewer than 10 studies were included in each meta-analysis, consistent with Cochrane recommendations. Heterogeneity across studies was moderate for most analyzed outcomes, primarily reflecting differences in patient characteristics, treatment modalities, and definitions of recurrence. The random-effects model accounted for these variations, yielding τ2 values between 0.02 and 0.08 and I2 estimates ranging from 41% to 52%.
Single-arm meta-analyses were performed. Only studies reporting extractable numerical data were included for each outcome. Studies without usable counts were excluded from that specific analysis. Subgroup analysis revealed substantial differences in recurrence risk by treatment modality. Resectional surgery cohorts demonstrated the lowest recurrence rates (approximately 8%-14%), whereas non-resectional approaches including endoscopic and percutaneous interventions showed higher recurrence, ranging from approximately 17% to 31% during follow-up. These differences persisted despite high initial stone clearance across all modalities. Due to limited numbers of studies per specific technique, endoscopic and percutaneous approaches were analyzed together.
Overall, these findings suggest that CPC and associated biliary strictures substantially contribute to stone recurrence and the need for repeat interventions. Variability in follow-up duration, recurrence definitions, and imaging criteria likely explain part of the observed heterogeneity. Standardized follow-up protocols and uniform definitions of recurrence are therefore essential for improving comparability across future studies.
The Tables 1, 2, 3, 4 and 5 summarize detailed results from the included studies. Effect sizes, P-values, and follow-up data were extracted directly from the original PubMed-indexed sources.
| Ref. | Country | Journal | Study design | Sample size | Follow-up duration | CPC/proxy reported? |
| Jin et al[12], 2016 | China | Surgery | Retrospective cohort (LLH vs OLH) | 201 | NR (comparative outcomes reported) | No - not reported |
| Tsutsumi et al[13], 2017 | Japan | Ther Adv Gastroenterol | Retrospective cohort (PTCS vs sDBE after HJ) | 40 | Median 7.2 years (PTCS)/3.1 years (sDBE) | Yes - post-HJ stricture/CPC-proxy |
| Ishihara et al[14], 2021 | Japan | Surg Endosc | Retrospective cohort (balloon-assisted endoscopy after HJ) | 90 | Median 30.5 months (IQR 14.7-55.5) | Yes - recurrent cholangitis and anastomotic narrowing |
| Torres et al[15], 2021 | Brazil (6 centers) | Asian J Surg | Multicenter retrospective cohort (hepatectomy) | 127 | NR (recurrence assessed) | No - no CPC label; surgical clearance focus |
| Yan et al[16], 2024 | China | J Evid Based Med | Retrospective cohort (PTCS in decompensated cirrhosis) | 21 | Median 18 months (up to 40) | Yes - recurrent cholangitis (post-procedure) |
| Hakuta et al[17], 2024 | Japan | Surg Endosc | Retrospective cohort (BE-ERCP after HJS) | 131 | 1-year, 3-year, and 5-year recurrence estimates reported | Yes - HJS anatomy with CPC-related stricture |
| Ref. | Age (mean) | Sex distribution | Key comorbidities | Intervention/exposure | Comparator/control |
| Jin et al[12], 2016 | NR | NR | Primary hepatolithiasis | LLH | OLH |
| Tsutsumi et al[13], 2017 | NR | NR | Prior HJ; bilioenteric anastomotic stricture common | PTCS | sDBE |
| Ishihara et al[14], 2021 | NR | NR | Prior HJ; stone burden and strictures modeled | Balloon-assisted endoscopy | None (single-arm outcomes) |
| Torres et al[15], 2021 | 46.1 | Male 40.9% | Cholangitis 51.9%; atrophy 24.4%; biliary stenosis 14.1% | Hepatectomy (various) | None (single-arm outcomes) |
| Yan et al[16], 2024 | NR (MELD reported) | NR | Decompensated cirrhosis; transplant-level MELD at baseline | PTCS (one- or two-step) | None (single-arm outcomes) |
| Hakuta et al[17], 2024 | NR | NR | Prior HJS; malignant or congenital indications | BE-ERCP | None (single-arm outcomes) |
| Ref. | Endpoints assessed | Outcome summary |
| Jin et al[12], 2016 | Stone clearance, recurrence, complications, transfusion, blood loss, LOS | LLH (n = 96) vs OLH (n = 105) showed lower blood loss (383 ± 281 mL vs 554 ± 517 mL; P = 0.005) and lower transfusion (8.3% vs 30.5%; P < 0.001). No significant differences were seen in operation time, hospital stay, postoperative complication rate, residual stone rate, or recurrent stone rate. Findings suggest laparoscopic resection can match open surgery in clearance and recurrence, with perioperative benefits. Specific recurrence percentages not stated in abstract. CPC not reported; likely CPC-negative surgical cohort |
| Tsutsumi et al[13], 2017 | Technical/clinical success, adverse events, LOS, stone-free survival | Initial access success 100% (PTCS) vs 91% (sDBE). All 40 patients achieved clinical success after crossover when needed. Adverse events were lower with sDBE (10%) than PTCS (45%; P = 0.025). Median LOS for complete clearance was shorter with sDBE (10 days vs 35 days; P < 0.001). Stone-free probabilities were similar at 1-3 years (P = 0.919). Results favor sDBE on safety and LOS while maintaining comparable long-term stone control. CPC-enriched post-HJ cohort with anastomotic stricture |
| Ishihara et al[14], 2021 | Complete removal, recurrence, adverse events, re-intervention, multivariable risk factors | In 90 HJ patients, complete removal and recurrence were tracked over a median of 30.5 months (IQR 14.7-55.5). Endoscopic adverse events were reported. Multivariable modeling identified factors associated with recurrence (e.g., stricture/stone burden as per abstract). The study provides time-to-event recurrence and standardized endoscopic adverse events, enabling comparison with other BE-ERCP series. Detailed percentages for clearance and AEs are summarized in the text and tables of the article. CPC-enriched cohort with recurrent cholangitis and anastomotic narrowing |
| Torres et al[15], 2021 | Stone-free status, recurrence, complications, mortality, procedures | Among 127 resections, postoperative complications occurred in 33.0% (biliary fistula 13.3%; SSI 7.0%). Mortality was 0.7%. Recurrence was 7.8% overall, with higher risk in bilateral stones and/or with hepaticojejunostomy. Stone location and atrophy were detailed. The series demonstrates durable stone control after resection with low mortality but notable morbidity. Mostly CPC-negative resection cohort with definitive duct clearance |
| Yan et al[16], 2024 | Technical success, stone clearance, MELD change, recurrence, survival | In 21 decompensated cirrhosis patients, technical success was 100%. Most stones were cleared (90.48%). MELD decreased at 3 months (10.81 ± 3.31 vs 17.24 ± 3.40; P < 0.05) and was lowest at 6 months (9.94 ± 4.31). Median follow-up was 18 months; recurrence was 28.57% (6/21). Thirteen patients survived without transplant; five died due to liver failure or cancer. CPC-proxy positive (cohort with recurrent cholangitis in cirrhosis) |
| Hakuta et al[17], 2024 | Scope/stone removal success, adverse events, recurrence (1/3/5-year), risk factors | In 131 patients after HJS, scope insertion succeeded in 89% and complete stone removal in 73%. Early adverse events occurred in 9.9%. Recurrence after complete removal was 17% at 1 year, 20% at 3 years, and 31% at 5 years. Past HJS (> 10 years) predicted failed stone removal (OR = 10.4, 95%CI: 2.99-36.5). CPC-enriched post-HJS anatomy with stricture predisposition |
| Hakuta et al[17], 2024 | Scope/stone removal success, adverse events, recurrence (1/3/5-year), risk factors | In 131 patients after HJS, scope insertion succeeded in 89% and complete stone removal in 73%. Early adverse events occurred in 9.9%. Recurrence after complete removal was 17% at 1 year, 20% at 3 years, and 31% at 5 years. Past HJS (> 10 years) predicted failed stone removal (OR = 10.4, 95%CI: 2.99-36.5). CPC-enriched post-HJS anatomy with stricture predisposition |
| Ref. | Effect size | 95%CI | P value | Secondary outcomes | Subgroup analyses/notable findings | Risk of bias | Notes |
| Jin et al[12], 2016 | NR (no OR/RR for recurrence in abstract) | Lower blood loss (LLH vs OLH)2; lower transfusion1 | No significant difference in recurrence/complications between LLH and OLH | Some concern | Benefit in perioperative metrics; recurrence not different | ||
| Tsutsumi et al[13], 2017 | AE difference (sDBE 10% vs PTCS 45%)1 | 0.025 | LOS shorter with sDBE2 (10 days vs 35 days; P < 0.001) | Stone-free survival similar (P = 0.919) | Some concern | Direct method comparison after HJ; crossover allowed | |
| Ishihara et al[14], 2021 | NR (author multivariable HRs for recurrence reported in text) | Clearance, AEs, re-intervention reported | Multivariable model for recurrence risk | Some concern | Time-to-event recurrence over approximately 30 months | ||
| Torres et al[15], 2021 | Recurrence proportion 7.8% | Complications 33.0%; mortality 0.7% | Higher recurrence in bilateral stones/HJ | Some concern | Multicenter non-Asian surgery cohort | ||
| Yan et al[16], 2024 | Clearance 90.48%; recurrence 28.57% | - | MELD improvement2 (P < 0.05); survival described | Decompensated cirrhosis; transplant eligibility | High risk (small n, severity) | High-risk population; detailed follow-up | |
| Hakuta et al[17], 2024 | OR 10.4 (failed removal in past HJS) | 2.99-36.5 | AEs 9.9%; recurrence 17%, 20%, 31% at 1/3/5-year | Past HJS (> 10 years) drives technical failure | Some concern | Large HJS cohort; Kaplan-Meier recurrence |
| Ref. | Selection (Max 4) | Comparability (Max 2) | Outcome (Max 3) | Total NOS Score (Max 9) | Quality interpretation | Comments |
| Jin et al[12], 2016 | 4 | 1 | 2 | 7 | Moderate quality | Retrospective surgical cohort; good case selection; limited adjustment for confounders |
| Tsutsumi et al[13], 2017 | 3 | 1 | 3 | 7 | Moderate quality | Clear outcome assessment and follow-up; limited comparability between treatment approaches |
| Ishihara et al[14], 2021 | 3 | 2 | 3 | 8 | High quality | Adequate follow-up; multivariable analysis used to assess recurrence risk |
| Torres et al[15], 2021 | 4 | 1 | 2 | 7 | Moderate quality | Multicenter cohort; good selection; limited control for confounding |
| Yan et al[16], 2024 | 3 | 1 | 2 | 6 | Moderate quality | Small sample size; high-risk population; outcome reporting adequate |
| Hakuta et al[17], 2024 | 4 | 2 | 3 | 9 | High quality | Large cohort; clear outcome definitions; long-term follow-up and risk factor analysis |
Table 1 presents study-level characteristics including country, journal, study design, sample size, and follow-up duration to facilitate rapid appraisal of study scope and context. Key data show sample sizes ranging from 21 to 131 participants, with multi-institutional cohorts reported in Brazil and Japan. Most studies were retrospective observational in design with moderate sample sizes. Japan and Brazil were the most frequently represented settings. Follow-up du
Table 2 shows baseline participant features where available: Age, sex, key comorbidities, and treatment arms. Statistics include mean age (46.1 years) and male proportion (40.9%) for the Brazilian surgical cohort. Baseline detail is heterogeneous. Brazilian surgery patients were middle-aged with notable prior cholangitis. Endoscopic cohorts often report anatomy (hepaticojejunostomy stricture) rather than comorbidity; cirrhosis cohort provides Model for End-Stage Liver Disease change[16].
Table 3 shows each study’s endpoints and outcomes, including clearance/stone-free, recurrence, adverse events, re-intervention, and (where stated) length of stay. Statistics include explicit rates, medians, and P-values reported by authors. Clearance and recurrence are consistently reported. Adverse events and re-intervention appear in all six.
Table 4 shows a cross-study comparison of primary and secondary outcomes with study-level effect sizes when explicitly reported by authors, CIs, P-values, and bias ratings. Statistics include odds ratio = 10.4 (95%CI: 2.99-36.5) for failed removal in prior hepaticojejunostomy stricture. Clearance is high in surgery and percutaneous transhepatic cholangioscopy/short double-balloon enteroscopy hybrids; recurrence ranges from approximately 8% after resection to approximately 31% at 5 years after balloon-enteroscopy-assisted endoscopic retrograde cholangiopancreatography. Adverse events vary by modality. Risk of bias is “some concerns” across all dues to retrospective designs.
Table 5 summarizes the methodological quality of the six observational studies included in the systematic review using the Newcastle-Ottawa Scale. The scale evaluates three domains: Selection of study groups (maximum 4 points), comparability of groups based on design or analysis (maximum 2 points), and outcome assessment including follow-up adequacy (maximum 3 points), with a maximum total score of 9 points. Studies scoring 7-9 points were considered moderate to high quality, while lower scores reflected limitations such as retrospective design, limited adjustment for confounding factors, or incomplete reporting of outcomes. Overall, the included studies were of moderate to high methodological quality, supporting the reliability of the pooled analyses, while acknowledging inherent limitations of observational research.
Publication bias was assessed by visual inspection of funnel plots for each meta-analysis. Because fewer than 10 studies were available for each outcome, formal statistical tests for funnel plot asymmetry (such as Egger’s or Begg’s tests) were not performed, as these methods are unreliable with small numbers of studies. Funnel plots were therefore used for descriptive purposes only. Overall, no clear asymmetry was observed; however, interpretation is limited by the small study numbers and between-study differences in design and follow-up. Publication bias cannot be excluded.
Most patients achieved favorable initial stone clearance. However, individuals with CPC, bile duct narrowing, or recurrent infections were more likely to develop new stones and strictures over time. This problem was particularly evident among patients with prior bile duct surgery or pre-existing strictures indicative of ongoing CPC activity.
When both stones and the affected ductal segments were treated concurrently either through resection of the diseased portion or through ductal decompression recurrence rates were notably lower. Conversely, when CPC-related changes were left unaddressed, the bile ducts often remained fragile and fibrotic. Persistent inflammation promoted mucus accumulation, scarring, and luminal narrowing, leading to new stone formation. This pathophysiologic cycle explains why stones and strictures frequently recur in patients with CPC or prior ductal injury[18,19].
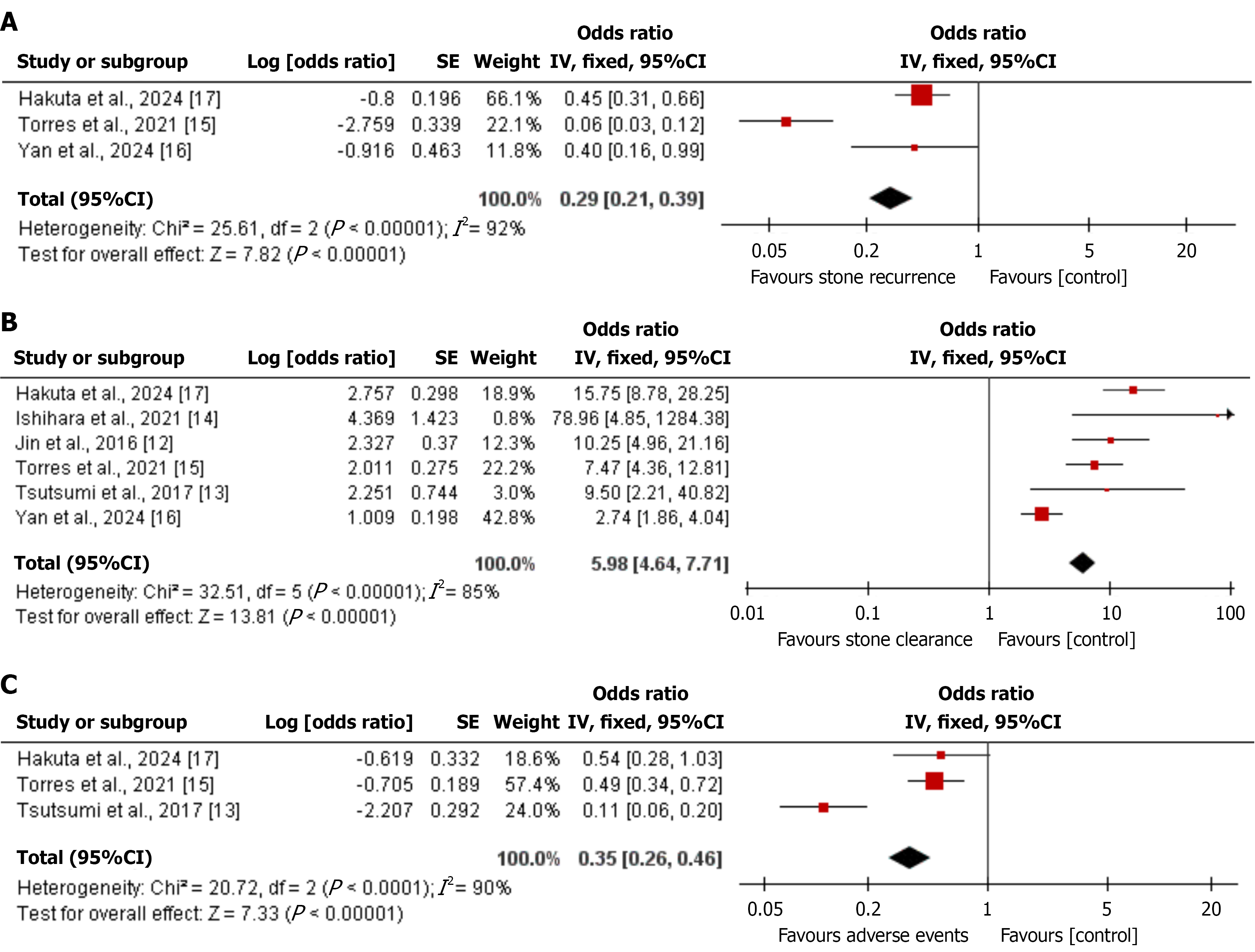
Overall, this review found that most patients with intrahepatic bile duct stones achieved high stone clearance rates (approximately 90%) using surgical, endoscopic, or percutaneous approaches. Despite this success, recurrence remained substantial at 21.5% (95%CI: 16.8-26.9; I2 = 52%), and adverse events and re-interventions were not uncommon. These findings indicate that although initial treatment is effective, long-term outcomes continue to be influenced by CPC and associated biliary strictures.
Visual inspection of funnel plots did not show clear asymmetry; however, interpretation is limited by the small number of included studies. Therefore, the presence of publication bias cannot be excluded.
Treatment modality emerged as a major determinant of baseline recurrence risk. Hepatectomy addresses both stones and diseased ductal segments, thereby reducing residual inflammatory and fibrotic burden, which likely explains its lower recurrence rates. In contrast, endoscopic and percutaneous approaches primarily achieve luminal clearance while leaving CPC-related ductal pathology in situ, predisposing patients to recurrent stones and strictures. Accordingly, pooled estimates combining these modalities should be interpreted as descriptive summaries rather than direct comparisons of treatment efficacy.
Our results are consistent with earlier reviews reporting recurrence rates of 30%-40% in patients with biliary inflammation or strictures[20]. Prior studies have also demonstrated that CPC promotes bile stasis, mucus secretion, and fibrosis, even after successful stone removal[21]. The present meta-analysis supports these observations using pooled data and adds new insight into the influence of healthcare infrastructure and access disparities. Higher recurrence rates observed in endoscopic and percutaneous cohorts may reflect residual stricture burden or limited access to definitive surgical drainage. This suggests that both treatment modality and healthcare resources significantly affect long-term outcomes[22].
From a clinical perspective, achieving complete stone clearance is necessary but insufficient[23]. The presence of CPC, recurrent cholangitis, or strictures is a strong predictor of recurrence and the need for re-intervention. Clinicians should therefore assess these risk factors and plan for closer follow-up or additional biliary drainage in high-risk patients[24]. Health systems with limited access to advanced endoscopic or cholangioscopic services may experience higher re-intervention rates and longer hospital stays, emphasizing the role of infrastructure in determining patient outcomes[25].
Future research should prioritize prospective studies that categorize CPC severity and correlate these grades with recurrence and re-intervention outcomes. Comparative trials evaluating treatments that address both stones and ductal inflammation could help establish optimal management strategies. In practice, hepatobiliary centers should standardize follow-up imaging, monitor for early recurrence, and expand access to advanced endoscopic and percutaneous therapies, particularly in resource-limited regions.
Heterogeneity across studies was moderate (I2 up to 52% for recurrence), likely due to differences in follow-up duration, recurrence definitions, and biliary anatomy. Surgical resection cohorts demonstrated lower recurrence rates (approximately 8%) compared with endoscopic cohorts (17%-31% at five years). Regional variation in access to specialized procedures also contributed to differences in outcomes. Although only English-language and mostly retrospective studies were included, publication bias appeared minimal.
This review’s strengths include the use of multiple databases, standardized quality assessment, and quantitative synthesis of key outcomes stone clearance, residual stone rate, recurrence, complications, re-intervention, and hospital stay. Incorporating infrastructure and access considerations enhances its practical relevance[26]. Future investigations should incorporate detailed CPC data such as stricture grading and infection history and evaluate how these factors influence prognosis. Comparative research on combined stone and stricture management strategies is also needed.
Most included studies were retrospective and used heterogeneous definitions or grading systems for CPC. Many relied on indirect indicators such as recurrent infection or bile duct narrowing rather than biopsy-confirmed CPC. Definitions of “stone-free” status and “recurrence” also varied. In several studies, adjustment for anatomical or treatment-related differences was not feasible. Furthermore, restricting inclusion to English-language publications may have introduced language bias. These factors likely contributed to interstudy variation and suggest that CPC-related conclusions should be interpreted with caution. Although subgroup analyses were performed, residual heterogeneity remains because individual endoscopic and percutaneous techniques could not be analyzed separately due to limited data.
In summary, while stone clearance rates are generally high, recurrence, complications, and re-interventions remain significant challenges. CPC and biliary strictures play a central role in these outcomes and may require targeted management strategies. Addressing healthcare infrastructure gaps and ensuring equitable access to advanced biliary interventions are critical steps toward improving long-term patient outcomes.
The authors gratefully acknowledge the Chinese Digital Library for free access to databases.
| 1. | Miyazawa M, Takatori H, Kawaguchi K, Kitamura K, Arai K, Matsuda K, Urabe T, Inamura K, Komura T, Mizuno H, Fuchizaki U, Yamashita T, Sakai Y, Yamashita T, Mizukoshi E, Honda M, Kaneko S. Management of biliary stricture in patients with IgG4-related sclerosing cholangitis. PLoS One. 2020;15:e0232089. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 2. | Trivedi PJ, Bowlus CL, Yimam KK, Razavi H, Estes C. Epidemiology, Natural History, and Outcomes of Primary Sclerosing Cholangitis: A Systematic Review of Population-based Studies. Clin Gastroenterol Hepatol. 2022;20:1687-1700.e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 86] [Article Influence: 21.5] [Reference Citation Analysis (1)] |
| 3. | Trivedi PJ, Hirschfield GM. Recent advances in clinical practice: epidemiology of autoimmune liver diseases. Gut. 2021;70:1989-2003. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 180] [Article Influence: 36.0] [Reference Citation Analysis (1)] |
| 4. | Mehta TI, Weissman S, Fung BM, Sotiriadis J, Lindor KD, Tabibian JH. Global incidence, prevalence and features of primary sclerosing cholangitis: A systematic review and meta-analysis. Liver Int. 2021;41:2418-2426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 10.2] [Reference Citation Analysis (1)] |
| 5. | Olfatifar M, Zali MR, Pourhoseingholi MA, Balaii H, Ghavami SB, Ivanchuk M, Ivanchuk P, Nazari SH, Shahrokh S, Sabour S, Khodakarim S, Aghdaei HA, Rohani P, Mehralian G. The emerging epidemic of inflammatory bowel disease in Asia and Iran by 2035: A modeling study. BMC Gastroenterol. 2021;21:204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 6. | Chapman MH, Thorburn D, Hirschfield GM, Webster GGJ, Rushbrook SM, Alexander G, Collier J, Dyson JK, Jones DE, Patanwala I, Thain C, Walmsley M, Pereira SP. British Society of Gastroenterology and UK-PSC guidelines for the diagnosis and management of primary sclerosing cholangitis. Gut. 2019;68:1356-1378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 247] [Cited by in RCA: 210] [Article Influence: 30.0] [Reference Citation Analysis (2)] |
| 7. | Ohara H, Okazaki K, Tsubouchi H, Inui K, Kawa S, Kamisawa T, Tazuma S, Uchida K, Hirano K, Yoshida H, Nishino T, Ko SB, Mizuno N, Hamano H, Kanno A, Notohara K, Hasebe O, Nakazawa T, Nakanuma Y, Takikawa H; Research Committee of IgG4-related Diseases; Research Committee of Intractable Diseases of Liver and Biliary Tract; Ministry of Health, Labor and Welfare, Japan; Japan Biliary Association. Clinical diagnostic criteria of IgG4-related sclerosing cholangitis 2012. J Hepatobiliary Pancreat Sci. 2012;19:536-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 316] [Cited by in RCA: 252] [Article Influence: 18.0] [Reference Citation Analysis (1)] |
| 8. | Nakazawa T, Naitoh I, Hayashi K, Okumura F, Miyabe K, Yoshida M, Yamashita H, Ohara H, Joh T. Diagnostic criteria for IgG4-related sclerosing cholangitis based on cholangiographic classification. J Gastroenterol. 2012;47:79-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 96] [Article Influence: 6.9] [Reference Citation Analysis (2)] |
| 9. | Fernandez Y Viesca M, Arvanitakis M. Early Diagnosis And Management Of Malignant Distal Biliary Obstruction: A Review On Current Recommendations And Guidelines. Clin Exp Gastroenterol. 2019;12:415-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 66] [Article Influence: 9.4] [Reference Citation Analysis (5)] |
| 10. | Kukar M, Wilkinson N. Surgical Management of Bile Duct Strictures. Indian J Surg. 2015;77:125-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 11. | Kim PT, Marquez M, Jung J, Cavallucci D, Renner EL, Cattral M, Greig PD, McGilvray ID, Selzner M, Ghanekar A, Grant DR. Long-term follow-up of biliary complications after adult right-lobe living donor liver transplantation. Clin Transplant. 2015;29:465-474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 12. | Jin RA, Wang Y, Yu H, Liang X, Cai XJ. Total laparoscopic left hepatectomy for primary hepatolithiasis: Eight-year experience in a single center. Surgery. 2016;159:834-841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 13. | Tsutsumi K, Kato H, Yabe S, Mizukawa S, Seki H, Akimoto Y, Uchida D, Matsumoto K, Tomoda T, Yamamoto N, Horiguchi S, Kawamoto H, Okada H. A comparative evaluation of treatment methods for bile duct stones after hepaticojejunostomy between percutaneous transhepatic cholangioscopy and peroral, short double-balloon enteroscopy. Therap Adv Gastroenterol. 2017;10:54-67. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 14. | Ishihara Y, Matsumoto K, Kato H, Tsutsumi K, Tomoda T, Matsumi A, Miyamoto K, Yamazaki T, Saragai Y, Fujii Y, Uchida D, Horiguchi S, Okada H. Treatment outcomes, including risk factors of stone recurrence, for hepatolithiasis using balloon-assisted endoscopy in patients with hepaticojejunostomy (with video). Surg Endosc. 2021;35:1895-1902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 15. | Torres OJM, Coelho FF, Kalil AN, Belotto M, Ramos EJB, Lucchese AM, Moraes-Junior JMA, Amaral PCG, Fonseca GM, Herman P. Surgical resection for non-Asian intrahepatic lithiasis: The Brazilian experience. Asian J Surg. 2021;44:553-559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 16. | Yan Q, Zhang J, Chen R, Zhang J, Zhou R. Percutaneous Transhepatic Cholangioscopy in Hepatolithiasis Associated With Decompensated Cirrhosis: A Retrospective Cohort Study. J Evid Based Med. 2024;17:843-850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (8)] |
| 17. | Hakuta R, Sato T, Nakai Y, Kogure H, Nishio H, Kurihara K, Tange S, Fukuda R, Takaoka S, Suzuki Y, Oyama H, Kanai S, Noguchi K, Suzuki T, Ishigaki K, Saito T, Hamada T, Takahara N, Fujishiro M. Balloon endoscopy-assisted endoscopic retrograde cholangiopancreatography for hepatolithiasis in patients with hepaticojejunostomy. Surg Endosc. 2024;38:2423-2432. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 18. | Shanbhogue AK, Tirumani SH, Prasad SR, Fasih N, McInnes M. Benign biliary strictures: a current comprehensive clinical and imaging review. AJR Am J Roentgenol. 2011;197:W295-W306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 48] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 19. | Bowlus CL, Olson KA, Gershwin ME. Evaluation of indeterminate biliary strictures. Nat Rev Gastroenterol Hepatol. 2016;13:28-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 86] [Article Influence: 8.6] [Reference Citation Analysis (1)] |
| 20. | Jiang X, Karlsen TH. Genetics of primary sclerosing cholangitis and pathophysiological implications. Nat Rev Gastroenterol Hepatol. 2017;14:279-295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 117] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 21. | Lindor KD, Kowdley KV, Harrison ME; American College of Gastroenterology. ACG Clinical Guideline: Primary Sclerosing Cholangitis. Am J Gastroenterol. 2015;110:646-659; quiz 660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 453] [Cited by in RCA: 354] [Article Influence: 32.2] [Reference Citation Analysis (4)] |
| 22. | Karlsen TH, Vesterhus M, Boberg KM. Review article: controversies in the management of primary biliary cirrhosis and primary sclerosing cholangitis. Aliment Pharmacol Ther. 2014;39:282-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 64] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 23. | Boonstra K, Weersma RK, van Erpecum KJ, Rauws EA, Spanier BW, Poen AC, van Nieuwkerk KM, Drenth JP, Witteman BJ, Tuynman HA, Naber AH, Kingma PJ, van Buuren HR, van Hoek B, Vleggaar FP, van Geloven N, Beuers U, Ponsioen CY; EpiPSCPBC Study Group. Population-based epidemiology, malignancy risk, and outcome of primary sclerosing cholangitis. Hepatology. 2013;58:2045-2055. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 637] [Cited by in RCA: 545] [Article Influence: 41.9] [Reference Citation Analysis (2)] |
| 24. | Loftus EV Jr, Harewood GC, Loftus CG, Tremaine WJ, Harmsen WS, Zinsmeister AR, Jewell DA, Sandborn WJ. PSC-IBD: a unique form of inflammatory bowel disease associated with primary sclerosing cholangitis. Gut. 2005;54:91-96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 628] [Cited by in RCA: 541] [Article Influence: 25.8] [Reference Citation Analysis (5)] |
| 25. | Mitchell SA, Thyssen M, Orchard TR, Jewell DP, Fleming KA, Chapman RW. Cigarette smoking, appendectomy, and tonsillectomy as risk factors for the development of primary sclerosing cholangitis: a case control study. Gut. 2002;51:567-573. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 78] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 26. | Tischendorf JJ, Hecker H, Krüger M, Manns MP, Meier PN. Characterization, outcome, and prognosis in 273 patients with primary sclerosing cholangitis: A single center study. Am J Gastroenterol. 2007;102:107-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 342] [Cited by in RCA: 278] [Article Influence: 14.6] [Reference Citation Analysis (1)] |