Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.115349
Revised: December 8, 2025
Accepted: January 12, 2026
Published online: March 27, 2026
Processing time: 140 Days and 3.3 Hours
The anesthesia recovery period after gastrointestinal surgery is a critical stage with a high risk of hypothermia, agitation, infusion failure, and other adverse events. Delayed or incomplete transfer of key information during handover may result in missed risks, inappropriate management, and compromised patient sa
To construct and validate a safety handover indicator system for patients under
This observational study included 500 patients recovering from gastrointestinal surgery at a tertiary hospital from June 2024 to June 2025. A preliminary handover indicator system was developed through literature review, semi-structured inter
Expert engagement in both Delphi rounds was 100%, with authority coefficients of 0.87 and 0.89, respectively. Kendall's harmony coefficient increased from 0.216 to 0.331 (both P < 0.01). The final system comprised three first-level, 12 second-level, and 38 third-level indicators. Compared with the control group, the experimental group showed longer handover time but markedly lower omission rate. Rates of hypothermia, agitation, and infusion failure were significantly reduced (all P < 0.05), and staff satisfaction scores were higher across all dimensions (P < 0.001).
The validated safety handover system standardizes postoperative transfer in gastrointestinal surgery, reduces errors and adverse events, and enhances patient safety and workflow efficiency, supporting its suitability for cli
Core Tip: This study developed a structured safety handover system for patients undergoing gastrointestinal surgery using literature review, Delphi consultation, and the analytic hierarchy process. The final system included three first-level, 12 second-level, and 38 third-level indicators. In clinical application, the experimental group showed longer handover time, reduced information omission, lower rates of hypothermia, agitation, and infusion failure, and higher staff satisfaction (all P < 0.05). These findings demonstrate the effectiveness of the system in improving handover quality and patient safety.
- Citation: Hu J, Yang WW, Qin W, Ren TT, Zhang MX, Zhu Q. Developing a standardized safety handover system for post-anesthesia patients in gastrointestinal surgery: Construction and validation. World J Gastrointest Surg 2026; 18(3): 115349
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/115349.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.115349
Patients undergoing gastrointestinal surgery often require multiple tubes, including gastric tubes, abdominal drains, and central venous catheters. They also face several challenges, such as pain, nausea and vomiting, fluid management needs, and temperature instability. Gastrointestinal surgery is among the most common procedures in general surgery, and these patients frequently have complex physiology and multiple comorbidities. Metabolic disturbances, electrolyte im
The postoperative anesthesia recovery period is a critical transition between the operating room and the ward. During this phase, respiratory and circulatory functions have not fully stabilized, and the capacity for physiological self-regu
Existing international guidelines and standards largely provide overarching principles but lack refined, quantifiable, and operable management indicators tailored to specific specialties, clinical scenarios, and patient populations[7,8]. Most available tools do not offer detailed or structured criteria for postoperative handover in gastrointestinal surgery, nor do they incorporate weighting, risk prioritization, or measurable evaluation components. This emphasizes the urgent need to develop a specialty-specific, scenario-based, and standardized safety handover system for patients in the anesthesia recovery period after gastrointestinal surgery.
To address this gap, this study aimed to establish a scientifically rigorous safety handover management indicator system specifically for patients undergoing gastrointestinal surgery during postoperative anesthesia recovery. Using literature review, semi-structured interviews, and Delphi expert consultation, a multi-dimensional indicator framework was developed and refined. The analytic hierarchy process was applied to determine indicator weights, enabling quantification and prioritization of key items. The resulting system was then validated in clinical practice through a controlled study, which demonstrated improved information completeness, fewer postoperative complications, and higher staff satisfaction. This dual approach - system construction combined with empirical verification - represents the primary in
After receiving approval from the Ethics Committee of the hospital, 500 patients who underwent gastrointestinal surgery at the Affiliated Hospital of Nantong University between June 2024 and June 2025 were enrolled. Of these, 250 patients who received the traditional handover method from June 2024 to December 2024 were assigned to the control group, and 250 patients who received handover based on the new indicator system developed in this study from January 2025 to June 2025 were assigned to the experimental group. The inclusion criteria were as follows: Patients aged 18 years or older, those undergoing elective or urgent open or laparoscopic gastrointestinal surgery, those classified as American Society of Anesthesiologists physical status I-III, and those admitted to the post-anesthesia care unit (PACU) for postoperative recovery. The exclusion criteria were as follows: Emergency surgery, death during the operation, or direct transfer to the intensive care unit without recovery in the PACU, and incomplete clinical data.
This study employed a time-series grouping method, assigning patients from June 2024 to December 2024 to the control group and those from January 2025 to June 2025 to the experimental group, without random allocation. The primary rationale was that the newly established handover system required unified implementation within a defined period, as simultaneous use of different handover protocols could disrupt workflow and compromise safety. To minimize bias associated with non-random grouping, the study applied strict inclusion criteria and conducted statistical compa
Traditional handover method: The traditional handover process followed routine clinical practice and consisted of the following steps: (1) Operating room nurses and anesthesiologists jointly transferred the patient to the PACU, connected the electrocardiographic monitor and oxygen delivery device, and ensured that vital signs were stable; (2) The anesthesiologist, serving as the primary handover provider, delivered a free-narrative oral handover. The main content included basic patient information, anesthesia method, current vital signs, key intraoperative events, level of consciousness, respiratory status, tube conditions, and medication status. Operating room nurses supplemented additional information, such as the submission of surgical specimens, intraoperative counts, and skin integrity. The receiving nurse documented key points and asked clarifying questions when necessary; (3) Operating room nurses completed a general, non-specialized operating room patient handover record form, which primarily consisted of checkboxes and a few open fields (e.g., vital signs were recorded as stable or unstable; tube condition was assessed as normal or abnormal); (4) The hand
Construction of the safety handover management indicator system: The construction of the safety handover management indicator system proceeded through the following steps: (1) A research team of 10 medical staff members was formed, including the directors of gastrointestinal surgery, anesthesiology, and the nursing department; the head nurses of the PACU and the gastrointestinal surgery ward; senior operating room nurses; and postgraduate students. The team carried out a literature review, development of interview outlines and expert consultation questionnaires, expert selection, organization of consultations, data collection and analysis, system construction, and implementation of the em
| Primary indicator and weight | Secondary indicator and weight | Composite weight |
| A1: Pre-handover preparation (0.3512) | B1: Completeness of patient information preparation (0.4015) | 0.1411 |
| B2: Preparation of drugs/supplies/equipment (0.3287) | 0.1155 | |
| B3: Environment and personnel readiness (0.2698) | 0.0948 | |
| A2: Handover process execution (0.4635) | B4: Accurate patient identification (0.2150) | 0.0997 |
| B5: Completeness of intra-operative status transfer (0.1832) | 0.0849 | |
| B6: Handover of vital signs and level of consciousness (0.1714) | 0.0794 | |
| B7: Tubing check and secure fixation (0.1555) | 0.0721 | |
| B8: Fluid balance and medication summary (0.1349) | 0.0625 | |
| B9: Skin integrity and belongings transfer (0.0751) | 0.0348 | |
| B10: Special issues and key medical orders (0.0649) | 0.0301 | |
| A3: Post-handover verification (0.1853) | B11: Information recheck and clarification of doubts (0.6328) | 0.1172 |
| B12: Mutual signature and documentation (0.3672) | 0.0681 |
| Level 2 indicator | Level 3 indicator | Importance score (mean ± SD) | Weight |
| B4: Accurate patient identification | Use at least two methods to verify identity (name, hospital ID) | 4.95 ± 0.22 | 0.402 |
| Check wristband information | 4.85 ± 0.37 | 0.318 | |
| Verify operative site and procedure | 4.75 ± 0.44 | 0.280 | |
| B7: Tubing check and secure fixation | Name, insertion depth, and fixation of each tube (drains, NG, CVC, etc.) | 4.90 ± 0.31 | 0.256 |
| Confirm drain patency and characteristics/volume of drainage | 4.95 ± 0.22 | 0.294 | |
| IV access patent; insertion site without leakage or swelling | 4.80 ± 0.41 | 0.235 | |
| Oxygen catheter/mask patent and properly secured | 4.60 ± 0.50 | 0.215 | |
| B11: Information recheck and clarification of doubts | Receiving nurse repeats key data (blood loss, special drugs, allergies) | 4.75 ± 0.44 | 0.421 |
| Handover staff confirm or correct the repeated information | 4.70 ± 0.47 | 0.342 | |
| Both parties immediately clarify any discrepancies and reach agreement | 4.85 ± 0.37 | 0.237 |
Baseline data: Baseline data for the two groups were recorded, including sex, age, surgical type, American Society of Anesthesiologists classification, and operation duration. These variables were compared to determine whether differences existed between the groups.
Results of expert consultation: A descriptive analysis was performed for basic expert information, the expert positive coefficient, and the expert authority coefficient for both rounds of consultation. The degree of coordination among expert opinions was also analyzed.
Handover quality: Differences between the two groups were compared in terms of handover time and the handover information omission rate, defined as the proportion of checklist items not addressed during handover.
Patient outcome indicators: The incidence of outcomes during the anesthesia recovery period was compared between the two groups. Complications included hypothermia (temperature < 36 °C), agitation (Riker Sedation-Agitation Scale score ≥ 5), and nausea and vomiting. Adverse events included unplanned extubation, falls, and medication errors.
Healthcare staff satisfaction: A self-developed satisfaction questionnaire (Cronbach’s α = 0.892) was used for assessment. The questionnaire evaluated clarity of process, completeness of information, work efficiency, and safety. Each dimension was scored out of 100, and the total score was calculated as the average of all dimension scores.
Data analysis was conducted using SPSS 26.0. Categorical data, expressed as n (%), were analyzed using the χ2 test. Continuous data, expressed as mean ± SD, were tested for normality using the Shapiro-Wilk test, and intergroup com
No significant differences were found in sex, age, type of operation, American Society of Anesthesiologists grade, or operation duration between the two groups (P > 0.05; Table 3).
| Group | n | Sex | Age (years) | Surgery time (minutes) | ASA | Types of surgery | ||||
| Male | Female | II | Ⅲ | IV | Open-abdominal surgery | Laparoscopic | ||||
| Experimental | 250 | 142 (56.80) | 108 (43.20) | 58.76 ± 8.23 | 182.54 ± 35.63 | 48 (19.20) | 152 (60.80) | 50 (20.00) | 95 (38.00) | 155 (62.00) |
| Control | 250 | 139 (55.60) | 111 (44.40) | 57.95 ± 9.14 | 178.96 ± 35.26 | 52 (20.80) | 146 (58.40) | 52 (20.80) | 88 (35.20) | (64.80) |
| χ2/t | - | 0.073 | 1.091 | 1.239 | 0.218 | 0.456 | ||||
| P value | - | 0.787 | 0.276 | 0.216 | 0.897 | 0.500 | ||||
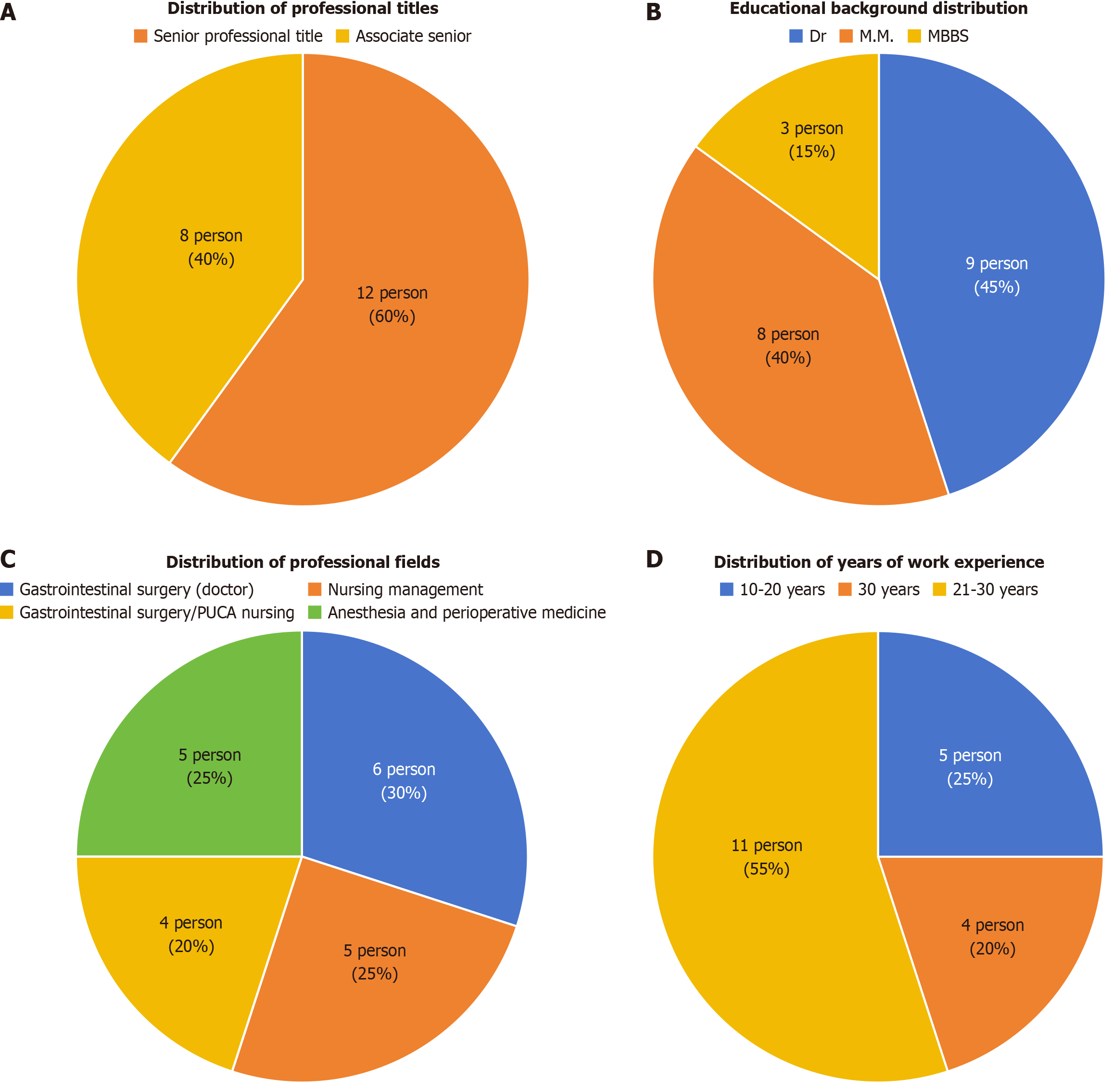
Both rounds of consultation achieved an effective recovery rate of 100%, with an expert positive coefficient of 100%. The expert authority coefficient was 0.87 in the first round and 0.89 in the second round. Kendall’s W was 0.216 (χ2 = 135.24, P < 0.001) in the first round and 0.331 (χ2 = 207.51, P < 0.001) in the second round. Figure 1 and Table 4 present basic expert information and consultation results, respectively.
| Round | Questionnaires distributed | Valid responses | Valid response rate | Expert authority coefficient | Kendall’s W | χ2 value | P value |
| 1 | 20 | 20 | 100 | 0.87 | 0.216 | 135.24 | < 0.001 |
| 2 | 20 | 20 | 100 | 0.89 | 0.331 | 207.51 | < 0.001 |
Compared with the control group, the experimental group showed a longer handover time and a lower information omission rate, with statistically significant differences (P < 0.05; Table 5).
| Group | n | Handover time (minutes) | Information omission rate (%) |
| Experimental | 250 | 9.86 ± 1.83 | 3.28 ± 1.57 |
| Control | 250 | 8.52 ± 2.15 | 15.86 ± 4.34 |
| t-value | - | 8.624 | 45.217 |
| P value | - | < 0.001 | < 0.001 |
The incidence rates of hypothermia, agitation, and infusion failure in the experimental group were lower than those in the control group, with statistically significant differences (P < 0.05; Table 6).
| Group | n | Hypothermia | Agitation | Nausea/vomiting | Respiratory depression | Unplanned extubation | Infusion failure | Pressure ulcer |
| Experimental | 250 | 35 (14.00) | 28 (11.20) | 33 (13.20) | 5 (2.00) | 0 (0.00) | 4 (1.60) | 1 (0.40) |
| Control | 250 | 68 (27.20) | 55 (22.00) | 42 (16.80) | 8 (3.20) | 2 (0.80) | 15 (6.00) | 3 (1.20) |
| χ2 value | - | 15.789 | 11.429 | 1.563 | 0.714 | 2.008 | 7.143 | 1.026 |
| P value | - | < 0.001 | 0.001 | 0.211 | 0.398 | 0.156 | 0.008 | 0.311 |
Scores for process clarity, information completeness, work efficiency perception, overall sense of safety, and total handover satisfaction in the experimental group were all higher than those in the control group, with statistically signi
| Group | n | Process clarity | Information completeness | Work efficiency | Overall security | Total score |
| Experimental | 30 | 90.22 ± 5.35 | 93.66 ± 4.14 | 85.45 ± 6.83 | 92.51 ± 5.02 | 90.43 ± 4.56 |
| Control | 30 | 76.43 ± 8.26 | 72.81 ± 9.58 | 70.69 ± 10.14 | 75.27 ± 8.75 | 73.88 ± 7.64 |
| t-value | - | 7.892 | 11.234 | 6.543 | 9.876 | 10.123 |
| P value | - | < 0.001 | < 0.001 | < 0.001 | < 0.001 | < 0.001 |
In recent years, with the widespread adoption of enhanced recovery after surgery principles and rapid advances in minimally invasive technology, perioperative management in gastrointestinal surgery has become increasingly refined, placing higher demands on medical quality and patient safety[9,10]. During the postoperative anesthesia recovery period, patients are transferred from anesthesiologists and operating room nurses to nurses in the PACU. The long information transmission chain, multiple handover links, and complex content make this phase a high-risk management node for medical errors and adverse events[11,12]. Evidence indicates that up to 70% of serious medical adverse events are as
This study followed rigorous scientific procedures and successfully established a safety handover management indicator system for patients undergoing gastrointestinal surgery during the postoperative anesthesia recovery period using the Delphi method. The system consists of three first-level, 12 second-level, and 38 third-level indicators. Weights were assigned through the analytic hierarchy process, providing an effective management tool to enhance safety during postoperative anesthesia recovery. Extensive literature review and in-depth qualitative interviews ensured comprehensive indicator sources and strong clinical applicability, covering the modern three-dimensional quality management framework of structure, process, and outcome[15,16]. The application of the Delphi expert consultation method was central to system development. The 20 participating experts came from several relevant fields and demonstrated high authority (expert authority coefficient > 0.8) and strong representativeness. The effective recovery rate of 100% across both rounds of consultation reflected high expert engagement. Kendall’s W in the first round was relatively low (0.216), a common finding during early consultation among senior experts, indicating variation in opinion. After revision and feedback, Kendall’s W increased markedly to 0.331 in the second round (P < 0.05), demonstrating stronger consistency and good coordination. The final indicator system, therefore, achieved a high degree of expert consensus and robust content validity. The analytic hierarchy process transformed subjective expert judgments into objective weight assign
The study findings showed that the average handover time in the experimental group was slightly longer than in the control group (P < 0.05). Although this may appear to indicate reduced efficiency, the additional duration (approximately 1.3 minutes) was mainly allocated to item-by-item verification and confirmation using the standardized checklist. This approach eliminated skipped items and selective handover driven by the pursuit of speed, and the direct result was a substantial reduction in the information omission rate - from 15.8% in the control group to 3.2% in the experimental group (P < 0.05). This outcome aligns with the findings of Jacobsen et al[18], which showed that a small increase in time investment produces marked improvement in information completeness and prevents the additional time required for repeated communication and clarification later in the process. Considered from a whole-process perspective, this approach enhances both safety and efficiency.
Regarding patient outcomes, the incidence rates of hypothermia, agitation, and infusion failure in the experimental group were significantly lower than those in the control group (P < 0.05). This improvement reflects the refinement and application of relevant items in the indicator system, such as body temperature monitoring and heat preservation measures, pain assessment and sedation scoring, and tube handover and fixation checks. The standardized handover process allowed medical staff to identify risks in advance and implement preventive measures, thereby reducing the occurrence of complications and adverse events. This result aligns with the findings of Kaltoft et al[19], which reported that structured handover tools can significantly improve safety during the postoperative recovery period.
In addition, the study found that handover satisfaction among medical staff in the experimental group was signi
However, this study was conducted at a single center with a relatively limited sample size and research scope. Long-term effect tracking of the indicators was not performed, which introduces certain limitations. Future studies should include multicenter, large-sample randomized controlled trials and establish long-term follow-up mechanisms to more comprehensively verify the effectiveness and reliability of the indicator system.
In conclusion, the safety handover management system for patients undergoing gastrointestinal surgery during the postoperative anesthesia recovery period developed in this study is scientific, systematic, and reliable. It standardizes the handover process, reduces handover errors and adverse events, and improves both safety assurance and work efficiency. This system is therefore suitable for wider clinical adoption.
| 1. | Birkeli G, Deilkås ECT, Ballangrud R, Lindahl AK. Implementing Learning from Excellence in a postanaesthesia care unit: a qualitative study of healthcare professionals' experiences after six months. BMC Health Serv Res. 2025;25:493. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 2. | Deb P, Das R, Bhattacharyya P. Focal hand warming for post-anaesthesia shivering control: A simple and safe non-pharmacological approach in resource limited-area. J Family Med Prim Care. 2023;12:3434-3436. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 3. | Rattana-Arpa S, Chaikittisilpa N, Srikongrak S, Udomnak S, Aroonpruksakul N, Kiatchai T. Incidences and outcomes of intra-operative vs. postoperative paediatric cardiac arrest: A retrospective cohort study of 42 776 anaesthetics in children who underwent noncardiac surgery in a Thai tertiary care hospital. Eur J Anaesthesiol. 2023;40:483-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 4. | Essa S, Venter S, Jordaan JD. The effect of a post-anaesthesia high-care unit (PAHCU) admission on mobilization, length of stay and in-hospital mortality post-surgery in low energy neck of femur fracture patients. Eur J Orthop Surg Traumatol. 2024;34:1389-1396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 5. | Festa R, Del Vicario M, Antonicelli F, Amato A. Improving anesthesia patient safety in NORA settings: utility of high-flow nasal cannula in the management of superobese patients undergoing deep sedation for gastrointestinal endoscopic procedures. Minerva Anestesiol. 2023;89:104-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 6. | Seo YE, Kim TY, Yoo HS, Chae MS. A postanaesthesia workload instrument can provide objective information promoting appropriate workload distribution between day and evening shift nursing staff in the postanaesthesia care unit: A prospective observational cohort study. Eur J Anaesthesiol. 2022;39:722-724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 7. | Wang Z, Wang S, Liu L, Zhang X, Ren M, Zhang Q, Liu C. Efficacy and safety of ciprofol versus propofol for anesthesia in patients undergoing gastrointestinal endoscope: a systematic review and meta-analysis of randomized controlled trials (RCT). BMC Anesthesiol. 2025;25:354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 8. | Birkeli GH, Ballangrud R, Jacobsen HK, Tveter Deilkas EC, Lindahl AK. Green Cross method in a postanaesthesia care unit: a qualitative study of the healthcare professionals' experiences after 3 years, including the COVID-19 pandemic period. BMJ Open Qual. 2023;12:e002247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 9. | Wu JJ. Anaesthetic Management of Patients With Do-Not-Attempt Resuscitation Orders: A Case for and Against Suspension During Anaesthesia and Surgery. Br J Hosp Med (Lond). 2025;86:1-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 10. | Lee FC, Queliza K, Chumpitazi BP, Rogers AP, Seipel C, Fishman DS. Outcomes of Non-anesthesiologist-Administered Propofol in Pediatric Gastroenterology Procedures. Front Pediatr. 2020;8:619139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 11. | Sugur T, Metinyurt HF, Kavakli AS, Kavrut Ozturk N, Ozmen S. Regional anesthesia in two consecutive surgeries in a patient with mitochondrial neurogastrointestinal encephalomyopathy: a case report. Braz J Anesthesiol. 2021;71:84-86. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 12. | Tassopoulos A, Chalkias A, Papalois A, Karlovasiti P, Zanda JSA, Chatzidakis S, Gazouli M, Iacovidou N, Fanni D, Xanthos T. Assessment of Post-Resuscitation Intestinal Injury and Timing of Bacterial Translocation in Swine Anaesthetized With Propofol-Based Total Intravenous Anaesthesia. Cureus. 2020;12:e10362. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 13. | Jansen G, Latka E, Bernhard M, Deicke M, Fischer D, Hoyer A, Keller Y, Kobiella A, Strickmann B, Strototte LM, Thies KC, Johanning K. Prehospital anesthesia in postcardiac arrest patients: a multicenter retrospective cohort study. Eur J Med Res. 2024;29:263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 14. | Birkeli GH, Thomas OMT, Deilkås ECT, Ballangrud R, Lindahl AK. Effect of the Green Cross method on patient safety culture in a postanaesthesia care unit: a longitudinal quasi-experimental study. BMJ Open Qual. 2024;13:e002964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 15. | Sliman MP, Risselada M, Moore GE, Rahn AP. Longer preoperative fluid resuscitation decreased anesthetic fluid interventions in 297 dogs undergoing surgery for gastrointestinal foreign body obstruction. J Am Vet Med Assoc. 2025;263:351-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 16. | Dahake JS, Verma N. Comparative Analysis of the Modified Aldrete Score and Fast-Track Criteria for Post-general Anaesthesia Recovery: A Narrative Review. Cureus. 2024;16:e64439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 17. | Torrano V, Anastasi S, Balzani E, Barbara E, Behr AU, Bosco M, Buttarelli C, Bruletti S, Bugada D, Cadeddu C, Cappelleri G, Cardia L, Casarano S, Cortegiani A, D'Ambrosio F, Del Vicario M, Fanelli A, Fusco P, Gazzerro G, Ghisi D, Giarratano A, Gori F, Greco M, Grossi PA, Manassero A, Russo G, Sardo S, Savoia C, Tescione M, Tinti G, De Cassai A. Enhancing Safety in Regional Anesthesia: Guidelines from the Italian Society of Anesthesia, Analgesia, Resuscitation and Intensive Care (SIAARTI). J Anesth Analg Crit Care. 2025;5:26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 21] [Reference Citation Analysis (1)] |
| 18. | Jacobsen HK, Ballangrud R, Birkeli GH. Learning from patient safety incidents: The Green Cross method. Nurs Crit Care. 2025;30:e13114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 19. | Kaltoft A, Jacobsen YI, Tangsgaard M, Jensen HI. ISBAR as a Structured Tool for Patient Handover During Postoperative Recovery. J Perianesth Nurs. 2022;37:34-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 20. | Wang G, Zhen B, Li JJ, Jin CN, Jia J, Liu BH, Bai YH. Insights into anesthesia administration for elderly individuals undergoing painless gastroenteroscopy: A bibliometric study. World J Gastrointest Endosc. 2025;17:101382. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 21. | Xiong YY, Chen CY, Li X, Yue XX, Zhao ZY. Efficacy of a self-made tracheostomy oxygen delivery device for oxygen therapy during postoperative anesthesia recovery. Technol Health Care. 2025;33:519-527. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 22. | Deng YF, Jiang XR, Feng ZG. Comparative observation of the effectiveness and safety of remimazolam besylate versus dexmedetomidine in gastrointestinal surgery in obese patients. World J Gastrointest Surg. 2024;16:1320-1327. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 23. | Dorman T, Drever B, Plumridge S, Gregory K, Cooper M, Roderick A, Arruzza E. Radiation dose to staff from scatter radiation in the post-anaesthetic recovery ward. J Med Imaging Radiat Sci. 2023;54:349-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 24. | Skiljic S, Budrovac D, Cicvaric A, Neskovic N, Kvolik S. Advances in Analgosedation and Periprocedural Care for Gastrointestinal Endoscopy. Life (Basel). 2023;13:473. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 25. | Meek T, Clyburn R, Fritz Z, Pitcher D, Ruck Keene A, Young PJ. Implementing advance care plans in the peri-operative period, including plans for cardiopulmonary resuscitation: Association of Anaesthetists clinical practice guideline. Anaesthesia. 2022;77:456-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 26. | Ang TL, Seet E, Goh YC, Ng WK, Koh CJ, Lui HF, Li JW, Oo AM, Lim KBL, Ho KS, Chew MH, Quan WL, Tan DMY, Ng KH, Goh HS, Cheong WK, Tseng P, Ling KL. Academy of Medicine, Singapore clinical guideline on the use of sedation by non-anaesthesiologists during gastrointestinal endoscopy in the hospital setting. Ann Acad Med Singap. 2022;51:24-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 27. | Erondu MU, Mahoney DP. Palliative care, resuscitation status, and end-of-life considerations in pediatric anesthesia. Curr Opin Anaesthesiol. 2020;33:354-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 28. | Kozarek R. Are Gastrointestinal Endoscopic Procedures Performed by Anesthesiologists Safer Than When Sedation is Given by the Endoscopist? Clin Gastroenterol Hepatol. 2020;18:1935-1938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (2)] |