Published online Apr 27, 2025. doi: 10.4240/wjgs.v17.i4.104187
Revised: January 23, 2025
Accepted: March 7, 2025
Published online: April 27, 2025
Processing time: 98 Days and 23.1 Hours
Transcatheter arterial chemoembolization (TACE) is a key treatment approach for advanced invasive liver cancer (infiltrative hepatocellular carcinoma). However, its therapeutic response can be difficult to evaluate accurately using conventional two-dimensional imaging criteria due to the tumor’s diffuse and multifocal grow
To investigate whether MRI bias field correction enhances the accuracy of volumetric assessment of infiltrative hepatocellular carcinoma treated with TACE, and to analyze how this improved measurement impacts prognostic prediction.
We retrospectively collected data from 105 patients with invasive liver cancer who underwent TACE treatment at the Affiliated Hospital of Xuzhou Medical Uni
Bias field correction significantly affected ETV measurements, with the corrected baseline ETV mean (505.235 cm³) being significantly lower than before correction (825.632 cm³, P < 0.001). Cox analysis showed that the hazard ratio (HR) for corrected baseline ETV (HR = 1.165, 95%CI: 1.069-1.268) was higher than before correction (HR = 1.063, 95%CI: 1.031-1.095). Using 412 cm³ as the cutoff, the group with baseline ETV < 415 cm³ had a longer median survival time compared to the ≥ 415 cm³ group (18.523 months vs 8.926 months, P < 0.001). The group with an ETV reduction rate ≥ 41% had better prognosis than the < 41% group (17.862 months vs 9.235 months, P = 0.006). Multivariate analysis confirmed that ETV reduction rate (HR = 0.412, P < 0.001), Child-Pugh classification (HR = 0.298, P < 0.001), and Barcelona Clinic Liver Cancer stage (HR = 0.578, P = 0.045) were independent prognostic factors.
Volume imaging based on MRI bias field correction can improve the accuracy of evaluating the efficacy of TACE treatment for invasive liver cancer. The corrected ETV and its reduction rate can serve as independent indicators for predicting patient prognosis, providing important reference for developing individualized treatment strategies.
Core Tip: This study highlights the value of magnetic resonance imaging bias field correction in improving enhanced tumor volume measurements for evaluating transcatheter arterial chemoembolization efficacy in invasive liver cancer. Corrected enhanced tumor volume and its reduction rate were identified as independent prognostic factors, enhancing accuracy in assessing tumor burden and outcomes. These findings support integrating bias field correction into imaging protocols to optimize treatment strategies and improve prognostic evaluations in interventional oncology.
- Citation: Liu K, Li JB, Wang Y, Li Y. Magnetic resonance imaging bias field correction improves tumor prognostic evaluation after transcatheter arterial chemoembolization for liver cancer. World J Gastrointest Surg 2025; 17(4): 104187
- URL: https://www.wjgnet.com/1948-9366/full/v17/i4/104187.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v17.i4.104187
Infiltrative hepatocellular carcinoma (iHCC) is a distinctive type of primary liver cancer characterized by diffuse infiltrative growth, unclear boundaries, and diagnostic challenges[1]. Compared to nodular hepatocellular carcinoma, iHCC has a poorer prognosis and significantly shorter median survival[2]. Transcatheter arterial chemoembolization (TACE) is a crucial treatment for advanced iHCC, but accurately assessing treatment efficacy is challenging due to the tumor's infiltrative nature[3].
Traditional criteria for evaluating solid tumor response, such as the World Health Organization criteria and the Modified Response Evaluation Criteria in Solid Tumors, primarily rely on changes in the maximum tumor diameter[4]. However, these standards have limitations in assessing iHCC. The tumor's unclear boundaries and multifocal fusion make it difficult to measure the maximum diameter accurately, and two-dimensional measurements cannot fully capture the spatial distribution and overall tumor burden[5]. Recently, advancements in volumetric imaging have provided new approaches to overcoming these challenges[6]. Magnetic resonance imaging (MRI), with its high soft tissue resolution and multiparametric imaging capabilities, plays a significant role in evaluating iHCC treatment response. However, MRI images are susceptible to bias field inhomogeneity, potentially leading to signal intensity anomalies and volumetric measurement errors[7].
Bias field correction is a crucial technique for improving MRI image quality. By compensating for factors like radiofrequency coil sensitivity and static field inhomogeneity, it achieves more accurate signal intensity distribution[8]. Nonetheless, research on the application of bias field correction in iHCC volumetric measurement is limited, and its impact on TACE efficacy evaluation has not been systematically validated. Enhanced tumor volume (ETV), a novel quantitative assessment metric that considers both tumor volume and enhancement characteristics, may provide a more objective basis for evaluating iHCC treatment response[9]. However, the accuracy of ETV measurement largely depends on image quality, and bias field inhomogeneity may affect its prognostic value[10].
Therefore, this study aims to explore the application value of volumetric imaging based on MRI bias field correction in assessing the efficacy of TACE treatment for iHCC. By comparing ETV measurements before and after correction, we analyze their relationship with patient prognosis, providing new technical methods and clinical evidence to enhance the accuracy of iHCC treatment response evaluation. This has significant clinical implications for optimizing individualized treatment strategies and improving patient outcomes. Additionally, this study will serve as a reference for establishing a more scientific efficacy evaluation system for iHCC.
A retrospective collection of patients diagnosed with primary iHCC and treated with TACE surgery from January 2020 to January 2024 at our hospital was conducted. The study included a total of 150 patients, with a mean age of 58.7 ± 10.4 years (range: 35-78 years). The majority of patients were male (72.0%, n = 108), reflecting the higher prevalence of liver cancer in males.
Inclusion criteria: (1) Age between 18-70 years; (2) Confirmed by pathological biopsy or imaging examinations (meeting the following characteristics: Significant hyposignal on T1-weighted images, homogeneous light to moderate high signal on T2-weighted images; (3) Significant pseudo-capsule enhancement in the arterial phase, and significant contrast agent washout in the delayed phase) consistent with the diagnosis of iHCC[11]; (4) Eastern Cooperative Oncology Group performance status ≤ 3[12]; (5) Child-Pugh classification A or B; (6) Barcelona Clinic Liver Cancer (BCLC) staging B, C, or D[13]; (7) Expected survival > 3 months; (8) Complete clinical data and follow-up information; and (9) Informed consent obtained and informed consent form signed.
Exclusion criteria: (1) Concurrent other malignant tumors; (2) Severe cardiac, pulmonary, or renal insufficiency; (3) Severe coagulation abnormalities (prothrombin time > 18 seconds or platelet count < 50 × 109/L); (4) Severe hepatic or renal damage (total bilirubin > 51.3 μmol/L); (5) Pregnant or lactating women; (6) Allergy to iodinated contrast media; (7) Contraindications for MRI examination (such as cardiac pacemaker, metallic implants, etc.); and (8) Inability to tolerate TACE surgery or presence of surgical contraindications.
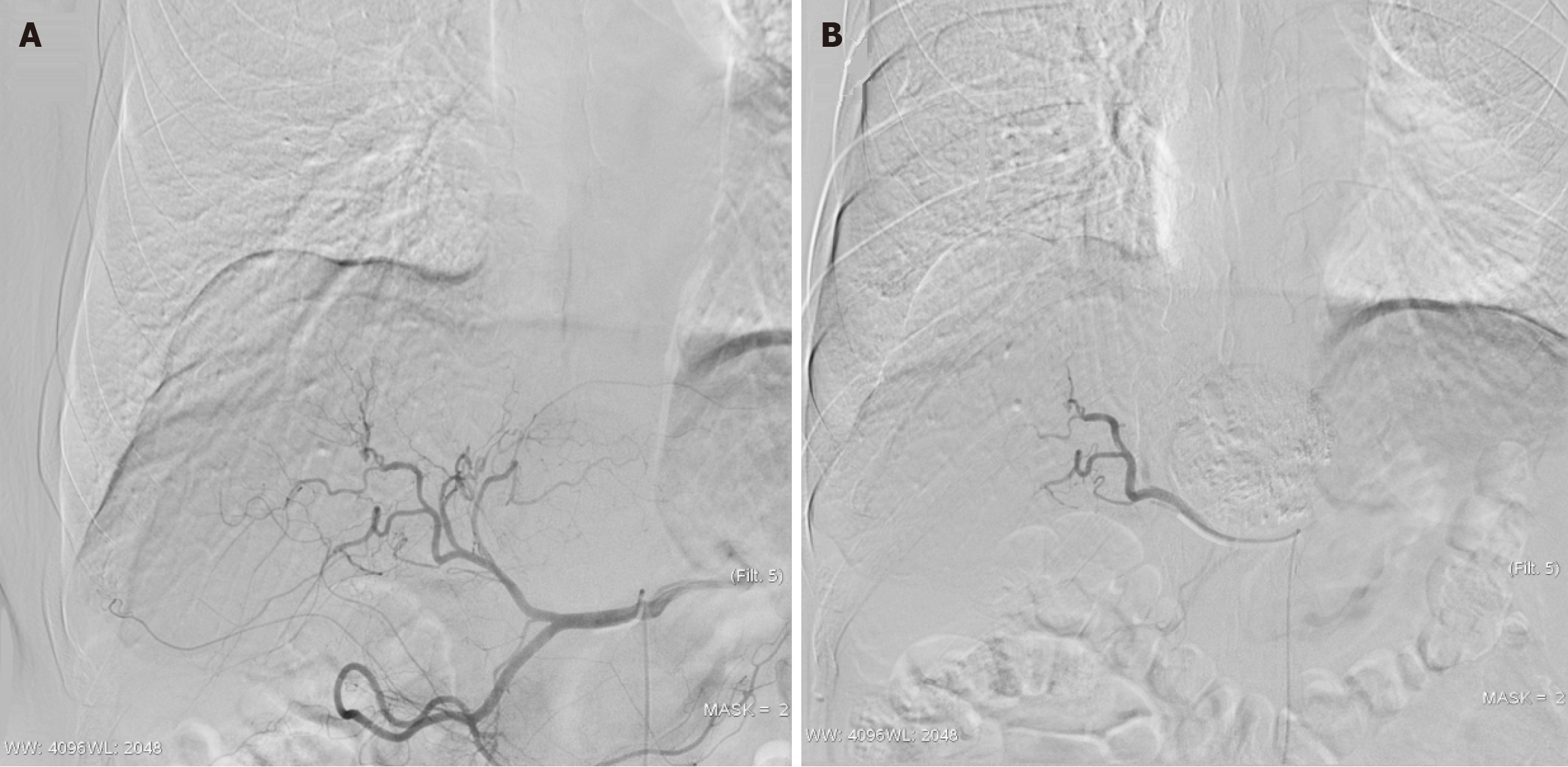
All patients underwent TACE treatment under the guidance of Digital Subtraction Angiography. Preoperative laboratory tests were conducted, including complete blood count, coagulation function, liver and kidney function, and tumor markers. The surgery was performed under local anesthesia, using the modified Seldinger technique through puncture of the right femoral artery. First, celiac trunk, common hepatic artery, and superior mesenteric artery angiography were performed to comprehensively assess the arterial blood supply of the liver, tumor vascular characteristics, and the presence of abnormal feeding arteries. Based on the vascular anatomy, selective catheterization to the proper hepatic artery or left/right hepatic artery was performed, followed by super-selective catheterization to the tumor-feeding artery. For the conventional lipiodol TACE group (69 cases), lipiodol was emulsified with idarubicin (dose 10 mg) and lobaplatin (dose 30-50 mg) and slowly injected until significant reduction in tumor-feeding artery blood flow. For the drug-eluting bead group (36 cases), CalliSpheres® drug-loaded microspheres were used (Jiangsu Hengrui Medicine Co., Ltd.), mixing 100-300 μm or 300-500 μm microspheres with epirubicin (dose 50-75 mg), diluted with non-ionic contrast medium, and slowly transfused (Figure 1). Intraoperatively, special attention was paid to prevent non-target embolization, and angiography was closely monitored to avoid reflux. For patients with extrahepatic feeding arteries, additional em
All patients were examined using 1.5T or 3.0T MRI scanners (Magnetom Avanto or Skyra, Siemens Healthcare Systems, Germany). Standardized scanning sequences included: Transverse T1-weighted dual echo sequence (TR/TE1/TE2 = 200/2.2/4.4 milliseconds, matrix 256 × 192, slice thickness 5 mm, interslice gap 1 mm), respiratory-triggered T2-weighted fast spin echo sequence (TR/TE = 4500/84 milliseconds, matrix 320 × 240, slice thickness 5 mm, interslice gap 1 mm), diffusion-weighted imaging sequence (TR/TE = 7500/65 milliseconds, b-values = 0, 500, 800 seconds/mm², matrix 128 × 128, slice thickness 5 mm, interslice gap 1 mm). Dynamic contrast-enhanced scans were performed using the volumetric interpolated breath-hold examination sequence (TR/TE = 4.4/2.2 milliseconds, matrix 256 × 192, slice thickness 3 mm, no gap). Contrast agents used were gadopentetate dimeglumine (Magnevist, Bayer Healthcare) or gadobutrol (Gadovist, Bayer Healthcare), with a dose of 0.1 mmol/kg, injected via the antecubital vein at a rate of 3 mL/s, followed by a 20 mL saline flush. Fluorescence-triggered multi-phase dynamic contrast-enhanced scans were performed, including arterial phase (20-35 seconds after contrast injection), portal venous phase (60-75 seconds), delayed phase (180 seconds), and hepatobiliary phase (20 minutes). To ensure image quality, all scans were completed during breath-hold, with each breath-hold time controlled within 20 seconds. The scan range included the entire liver, and the field of view was adjusted to 350-400 mm according to the patient’s body size.
All patient MRI images were registered using a deformable field-based non-rigid registration algorithm. Initially, T1-weighted sequence images before and after TACE treatment were imported into a medical image processing workstation (Syngo.via VB20A, Siemens Healthcare Systems). The registration processed a multi-resolution strategy, optimizing from low resolution to the original resolution to improve registration efficiency and accuracy. The registration algorithm included two stages: Global affine transformation and local non-rigid deformation. In the global affine transformation stage, overall rotation, translation, and scaling parameters were determined by minimizing mutual information. In the local non-rigid deformation stage, a B-spline free-form deformation model and gradient-based similarity metric were used to fine-tune local deformations. To ensure registration quality, control points were set at a distance of 5 mm, with a deformation regularization parameter λ = 0.01 and a maximum of 200 iterations. Registration results were assessed by two radiologists with over 5 years of experience using a 5-point scale (1 point - very poor, 5 points - very good), and cases with scores below 3 required re-registration. Additionally, the quality of registration was quantitatively evaluated by calculating the normalized cross-correlation coefficient and structural similarity index of the images before and after registration, with requirements of normalized cross-correlation coefficient > 0.85 and structural similarity index > 0.80. The resulting deformation fields were used to establish spatial correspondence between different sequences and phases in subsequent analyses.
This study employed a semi-automatic segmentation method combining deep learning with manual correction for three-dimensional segmentation of the liver and tumors. Initially, a deep convolutional neural network based on the 3D U-Net architecture was used for rough segmentation of the liver, with an encoder-decoder structure, including 4 down-sampling and 4 up-sampling layers, each using two 3 × 3 × 3 convolutional kernels and ReLU activation functions. To improve segmentation accuracy, data augmentation techniques were used during the training phase, including random rotation (± 15°), scaling (0.8-1.2), and elastic deformation. The network training used a combined loss function, including Dice loss and weighted cross-entropy loss, with a weight ratio of 1:1. After obtaining preliminary segmentation results, manual corrections were made by two radiologists with over 8 years of experience using three-dimensional segmentation software (Syngo.via MM Oncology, Siemens Healthcare Systems). The segmentation process was synchronized in the axial, sagittal, and coronal directions to ensure continuity and accuracy. For tumor regions, based on multi-phase contrast-enhanced scan features, a semi-automatic watershed algorithm was used for preliminary segmentation on arterial phase images, combined with portal venous and delayed phase image features for boundary optimization. Segmentation results were quality-controlled by calculating the Dice coefficient and volume overlap rate, requiring a Dice coefficient > 0.90 and a volume overlap rate > 0.85. The final segmentation results were used for subsequent quantitative analysis and evaluation.
To eliminate bias field inhomogeneity in MRI images, this study employed an improved N4 bias field correction algorithm for whole-liver volume correction. The algorithm is based on the N4 Intensity Transformation Kit framework, approximating the bias field distribution with B-spline basis functions. The correction process first applies a logarithmic transformation to the input image, converting multiplicative bias fields to additive bias fields. Subsequently, a multi-scale iterative strategy is used to estimate the bias field at different resolution levels. Each scale level is set with a maximum of 50 iterations, a convergence threshold of 0.001, and an initial B-spline control point distance of 30 mm, gradually reduced to 10 mm during iteration. To improve the correction effect, a tissue segmentation-based constraint term is introduced, using the aforementioned liver segmentation results as a mask to correct only the liver region. The correction process uses the Otsu thresholding method to adaptively estimate different tissue categories, optimizing bias field estimation by minimizing intra-class variance. The correction effect is evaluated by calculating the coefficient of variation of the image intensity, requiring a reduction of over 30% in the coefficient of variation value of the liver parenchymal region after correction. Additionally, the correction effect is verified by comparing the uniformity of the gray-level histogram and the signal intensity profile before and after correction. The final correction results were used for subsequent quantitative feature extraction and analysis.
This study used enhanced volume as the main quantitative assessment indicator. Based on the aforementioned segmentation results and bias field-corrected images, a voxel-based analysis method was used for tumor volume calculation. Initially, on arterial phase images, the region of interest (ROI) was determined according to World Health Organization standards to include all visible tumor tissue. By comparing with portal venous and delayed phase images and combining tumor enhancement features, the ROI boundary was further optimized to exclude necrotic areas and non-tumor tissue. Tumor volume was calculated using three-dimensional reconstruction software (Syngo.via VB20A, Siemens Healthcare Systems) by accumulating voxels within the ROI on each axial slice and correcting the volume considering slice thickness and interslice gap. To ensure measurement accuracy, measurements were made by two radiologists with over 8 years of experience, and their average values were taken as the final results. Inter-measurer consistency was assessed using the Intraclass Correlation Coefficient, requiring an Intraclass Correlation Coefficient > 0.85. For the evaluation of TACE treatment effects, the absolute change in ETV before and after treatment (ΔETV = post-treatment ETV - pre-treatment ETV) and the relative change percentage (ETV% = ΔETV/pre-treatment ETV × 100%) were calculated. Additionally, to verify the impact of MRI bias field correction on measurement results, the aforementioned measurements were performed on both original and bias field-corrected images and compared. In patients with multiple lesions, the maximum lesion method was used, i.e., the lesion with the largest diameter was selected for measurement and follow-up assessment. Furthermore, for cases with portal vein invasion, the tumor thrombus in the portal vein was included in the measurement range. To reduce the impact of partial volume effects, for lesions with blurred edges, a semi-automatic threshold segmentation method was used, combined with morphological operations for boundary optimization.
The study employed a rigorous set of statistical analysis methods. First, the data was processed and analyzed using SPSS 26.0 software and R software version 4.1.0. Continuous variables with normal distribution were represented as mean ± SD, and paired t-tests were used to compare differences in ETV before and after TACE treatment. Non-normally distributed variables were represented as median (interquartile range), and the Wilcoxon rank-sum test was used for group comparisons. Categorical variables were represented as number (percentage) and analyzed using χ2 tests or Fisher’s exact tests. For survival analysis, the study used the Kaplan-Meier method to plot survival curves and the log-rank test for group comparisons. Additionally, the Cox proportional hazards regression model was applied to analyze risk factors affecting patient survival, calculating the hazard ratio (HR) and its 95% confidence interval (CI). To evaluate the non-linear relationships between continuous variables and prognosis, the study employed the local weighted scatterplot smoothing (LOWESS) method and the Q statistic method maximizing the standardized log-rank statistic.
A total of 105 patients with iHCC were included in this study, consisting of 78 males (74.29%) and 27 females (25.71%). In terms of age distribution, 74 patients (70.48%) were under 65 years old, and 31 patients (29.52%) were 65 years old or above. In etiological analysis, hepatitis B virus infection was the most common, accounting for 89 cases (84.76%), followed by hepatitis C virus infection with 9 cases (8.57%), alcoholic liver disease with 7 cases (6.66%), and cryptogenic liver cancer with 1 case (0.95%). A total of 71 patients (67.62%) had concurrent liver cirrhosis.
Regarding disease staging and scoring, according to the BCLC, there were 31 patients (29.52%) in stage B, 71 patients (67.62%) in stage C, and 3 patients (2.86%) in stage D. In the Child-Pugh classification, there were 70 patients (66.67%) in grade A, 34 patients (32.38%) in grade B, and 1 patient (0.95%) in grade C. Eastern Cooperative Oncology Group per
In terms of treatment regimens, 69 patients (65.71%) underwent conventional lipiodol TACE, and 36 patients (34.29%) received drug-eluting bead treatment. The distribution of treatment sessions was as follows: 50 patients (47.62%) had a single treatment, 19 patients (18.10%) had two treatments, 14 patients (13.33%) had three treatments, and 22 patients (20.95%) had four or more treatments. During the follow-up period, 81 patients (77.14%) died, and 24 patients (22.86%) were alive. The average follow-up time was 17.32 months (range 0.21-142.55 months), with a median follow-up time of 10.95 months (interquartile range 4.23-21.32 months) (Table 1).
| Item | Statistic |
| Total number of patients, baseline | 105 |
| Age | |
| < 65 years old | 74 (70.48) |
| ≥ 65 years old | 31 (29.52) |
| Gender | |
| Male | 78 (74.29) |
| Female | 27 (25.71) |
| Survival status at analysis | |
| Alive | 24 (22.86) |
| Deceased | 81 (77.14) |
| Follow-up time (months) | |
| Mean (range) | 17.32 (0.21-142.55) |
| Median (interquartile range) | 10.95 (4.23-21.32) |
| Etiology | |
| Hepatitis B virus | 89 (84.76) |
| Hepatitis C virus | 9 (8.57) |
| Alcoholic liver disease | 7 (6.66) |
| Cryptogenic liver cancer | 1(0.95) |
| Cirrhosis | 71 (67.62) |
| Barcelona Clinic Liver Cancer staging | |
| B | 31 (29.52) |
| C | 71 (67.62) |
| D | 3 (2.86) |
| Child-Pugh classification | |
| A | 70 (66.67) |
| B | 34 (32.38) |
| C | 1 (0.95) |
| ECOG performance status | |
| 0 | 58 (55.24) |
| 1 | 41 (39.05) |
| 2 | 4 (3.81) |
| 3 | 2 (1.90) |
| Number of tumors | |
| 1 | 19 (18.10) |
| 2 | 20 (19.05) |
| 3 | 12 (11.43) |
| More than 4 | 54 (51.43) |
| Tumor size (cm) | |
| Mean (range) | 8.72 (1.6-19.8) |
| Median (interquartile range) | 8.15 (5.2-11.8) |
| Portal vein invasion | |
| Yes | 44 (41.90) |
| No | 61 (58.10) |
| Extrahepatic metastasis | |
| Yes | 9 (8.57) |
| No | 96 (91.43) |
| TACE Type | |
| Lipiodol | 69 (65.71) |
| Drug-eluting beads | 36 (34.29) |
| Number of TACE treatments | |
| 1 | 50 (47.62) |
| 2 | 19 (18.10) |
| 3 | 14 (13.33) |
| 4 or more than 4 | 22 (20.95) |
Comparative analysis showed (Table 2), MRI bias field correction significantly affected the measurement results of tumor volume before and after TACE treatment. At baseline, the mean ETV without bias field correction was 825.632 cm³ (range: 63.523-3563.126 cm³), while the mean ETV after bias field correction dropped to 505.235 cm³ (range: 142.352-1292.223 cm³), with a statistically significant difference between the two groups (P < 0.001). Similarly, after TACE treatment, the mean ETV without bias field correction was 632.523 cm³ (range: 47.235-3382.532 cm³), and it dropped to 325.236 cm³ (range: 110.135-1018.516 cm³) after bias field correction, with a statistically significant difference (P < 0.001).
| Variables baseline ETV | Mean (interval) (cm3) | P value | |
| No MR bias field correction | With MR bias field correction | ||
| After TACE treatment | 825.632 (63.523-3563.126) | 505.235 (142.352-1292.223) | < 0.001 |
| ETV | 632.523 (47.235-3382.532) | 325.236 (110.135-1018.516) | < 0.001 |
Cox proportional hazards model analysis indicated (Table 3), in measurements not using bias field correction, the HR for baseline ETV was 1.063 (95%CI: 1.031-1.095, P < 0.001), while the HR for baseline ETV using bias field correction increased to 1.165 (95%CI: 1.069-1.268, P < 0.001). For absolute ETV change, the HR for the uncorrected group was 1.012 (95%CI: 0.985-1.042, P = 0.481), and for the corrected group, it was 1.040 (95%CI: 0.935-1.160, P = 0.512), with neither group showing statistical significance. However, for the percentage change in ETV, the HR for the uncorrected group was 1.005 (95%CI: 0.981-1.030, P = 0.793), and it significantly decreased to 0.665 (95%CI: 0.538-0.825, P < 0.001) for the corrected group.
| Variable | Not using bias field correction | Use bias field correction | ||
| Hazard ratio | P value | Hazard ratio | P value | |
| Baseline ETV | 1.063 (1.031-1.095) | < 0.001 | 1.165 (1.069-1.268) | < 0.001 |
| Absolute ETV change | 1.012 (0.985-1.042) | 0.481 | 1.040 (0.935-1.160) | 0.512 |
| ETV% | 1.005 (0.981-1.030) | 0.793 | 0.665 (0.538-0.825) | < 0.001 |
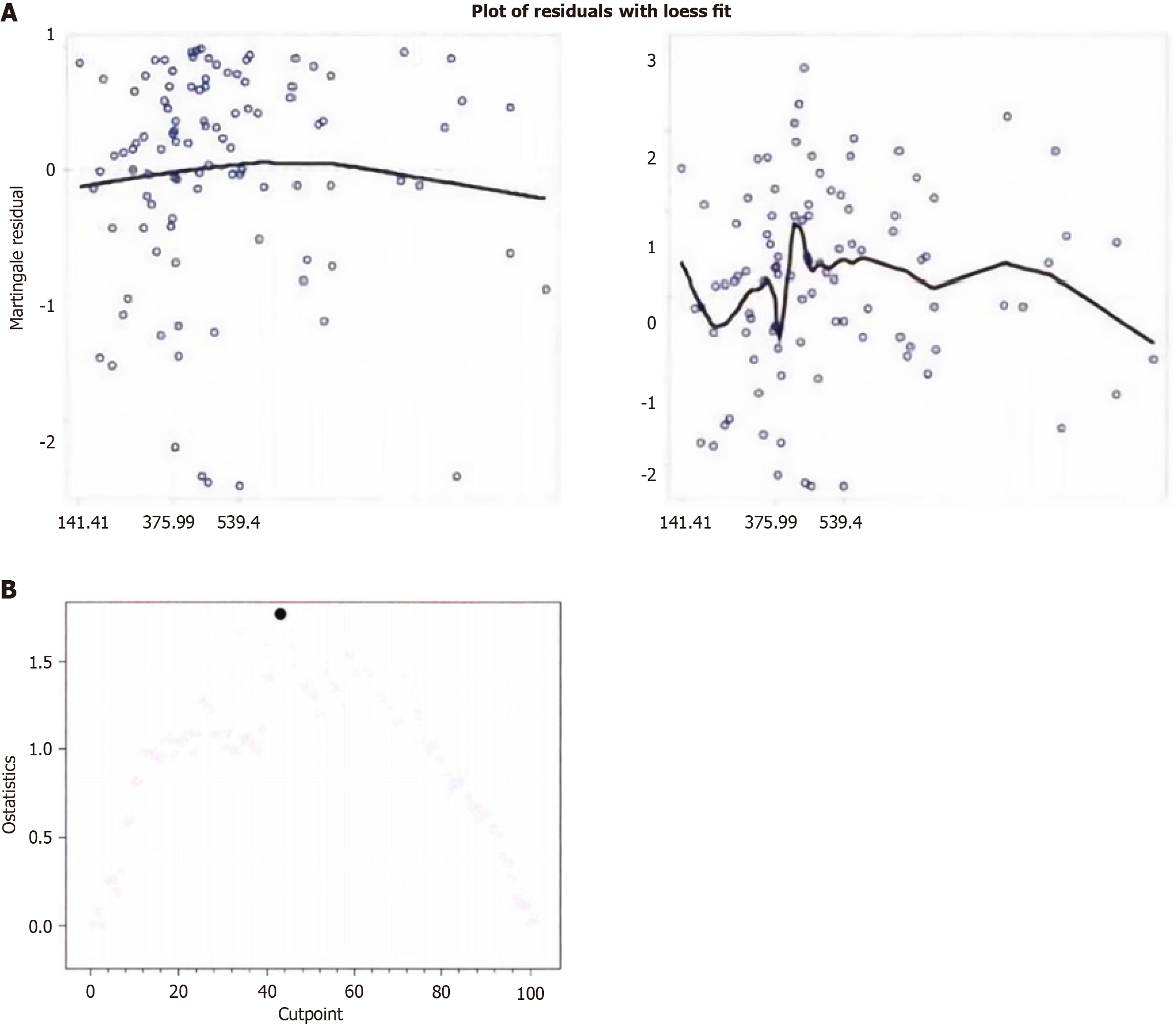
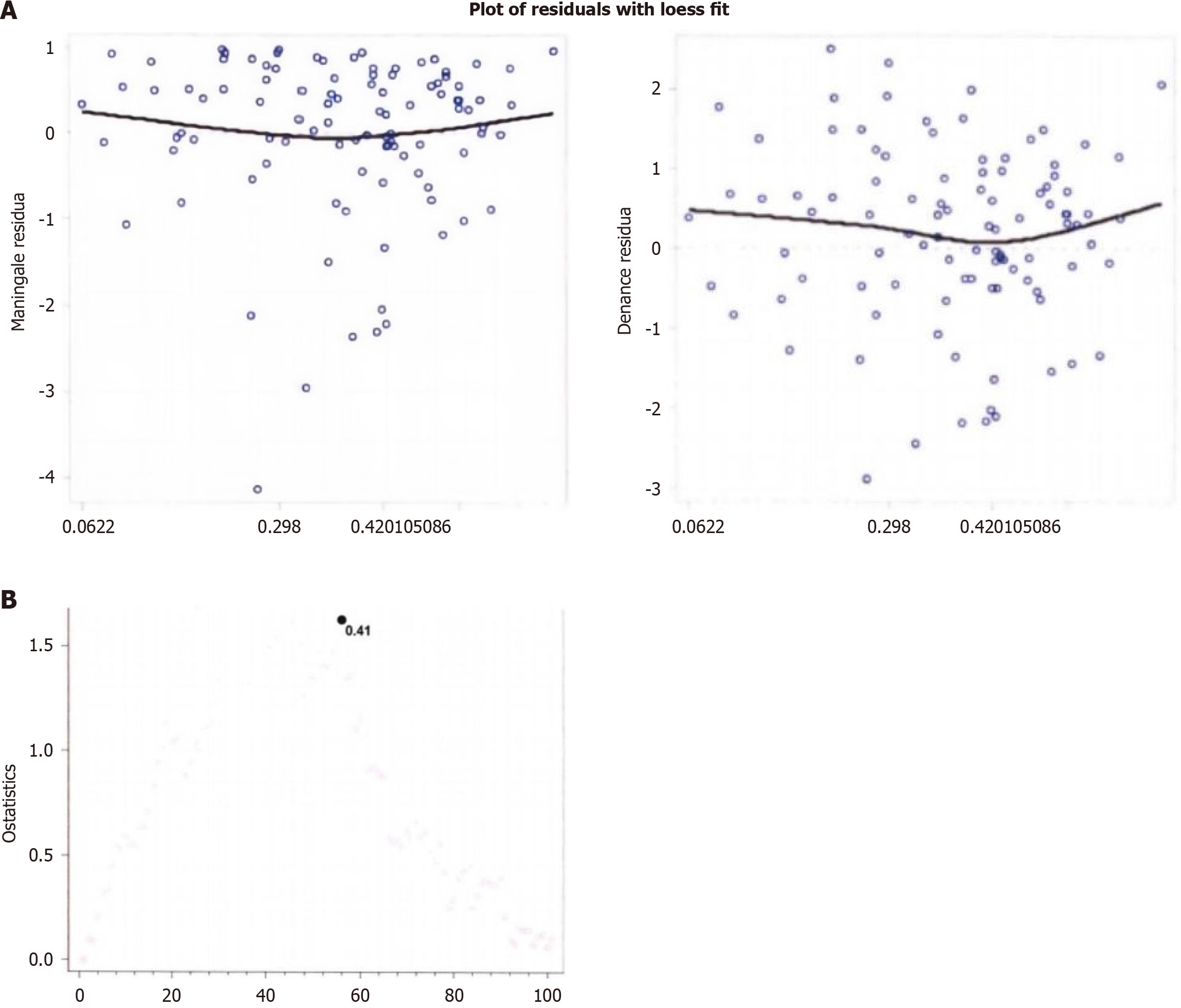
Analysis using Martingale residual LOWESS smoothing curves (Figure 2A) showed a clear turning point in the curve of ETV before the first TACE treatment, indicating that the critical value using the Cox model is appropriate as an indicator variable for baseline ETV. According to the Q statistic method (Figure 2B), the critical value for baseline ETV using bias field correction was determined to be 412 cm³ (P = 0.003). Similarly, for the analysis of the percentage change in ETV (Figure 3A), the Martingale residual LOWESS smoothing curve showed a trend of decreasing and then increasing, suggesting that using the reduction rate of ETV before and after treatment as a linear variable in the Cox model may not be suitable and should be transformed into an indicator with an appropriate critical value. After Q statistic analysis (Figure 3B), the critical value for the percentage change in ETV before and after TACE treatment using magnetic re
Based on the analysis results of the critical values, we grouped patients and assessed their survival status. In the pre-treatment ETV threshold grouping, 45 patients (42.86%) had ETV < 415 cm³, and 60 patients (57.14%) had ETV ≥ 415 cm³. Kaplan-Meier survival analysis showed that the median survival time for patients with ETV < 415 cm³ was 18.523 months (95%CI: 14.862-29.635 months), while for patients with ETV ≥ 415 cm³, the median survival time was 8.926 months (95%CI: 5.923-10.832 months). The quartile analysis revealed that the 25th, 50th, and 75th survival time points for the ETV < 415 cm³ group were 10.523 months (95%CI: 4.862-15.235 months), 18.523 months (95%CI: 14.862-29.635 months), and 35.862 months (95%CI: 22.523-45.236 months), respectively; for the ETV ≥ 415 cm³ group, the corresponding time points were 3.862 months (95%CI: 1.523-5.862 months), 8.926 months (95%CI: 5.923-10.832 months), and 15.235 months (95%CI: 10.523-19.862 months). The comparison of survival curves between the two groups showed a statistically significant difference (log-rank test, P < 0.001) (Table 4).
| Percentage (%) | < 415 cm3 | ≥ 415 cm3 | ||
| Survival time (months) | 95%CI | Survival time (months) | 95%CI | |
| 25 | 10.523 | 4.862-15.235 | 3.862 | 1.523-5.862 |
| 50 | 18.523 | 14.862-29.635 | 8.926 | 5.923-10.832 |
| 75 | 35.862 | 22.523-45.236 | 15.235 | 10.523-19.862 |
In the ETV reduction rate threshold grouping before and after treatment, 58 patients (55.24%) had an ETV reduction rate < 41%, and 47 patients (44.76%) had an ETV reduction rate ≥ 41%. The median survival time for patients with an ETV reduction rate < 41% was 9.235 months (95%CI: 5.862-12.523 months), while for patients with an ETV reduction rate ≥ 41%, the median survival time was 17.862 months (95%CI: 13.926-20.635 months). The quartile analysis showed that the 25th, 50th, and 75th survival time points for the ETV reduction rate < 41% group were 3.235 months (95%CI: 1.523-5.862 months), 9.235 months (95%CI: 5.862-12.523 months), and 15.235 months (95%CI: 10.235-31.523 months), respectively; for the ETV reduction rate ≥ 41% group, the corresponding time points were 10.523 months (95%CI: 6.523-14.862 months), 17.862 months (95%CI: 13.926-20.635 months), and 29.635 months (95%CI: 19.862-72.523 months). The comparison of survival curves between the two groups showed a statistically significant difference (log-rank test, P = 0.006) (Table 5).
| Percent (%) | < 0.414 | ≥ 0.414 | ||
| Survival time (months) | 95%CI | Survival time (months) | 95%CI | |
| 25 | 3.235 | 1.523-5.862 | 10.523 | 6.523-14.862 |
| 50 | 9.235 | 5.862-12.523 | 17.862 | 13.926-20.635 |
| 75 | 15.235 | 10.235-31.523 | 29.635 | 19.862-72.523 |
To comprehensively assess the factors affecting patient prognosis, this study conducted univariate and multivariate Cox regression analyses on baseline ETV and the rate of ETV reduction. In the analysis of baseline ETV (Table 6), univariate analysis showed that the following factors were significantly associated with patient prognosis: Pre-treatment ETV (≥ 415 cm³ vs < 415 cm³, HR = 1.982, 95%CI: 1.212-3.236, P = 0.012), Child-Pugh classification (Class A vs class B or C, HR = 0.423, 95%CI: 0.252-0.668, P < 0.001), and BCLC staging (Stage B vs stage C or D, HR = 0.523, 95%CI: 0.318-0.852, P = 0.013). The results of the multivariate analysis indicated that Child-Pugh classification (HR = 0.432, 95%CI: 0.256-0.702, P < 0.001) and BCLC staging (HR = 0.538, 95%CI: 0.318-0.916, P = 0.023) remained independent prognostic factors. Notably, age (per one-year increase, HR = 1.008, 95%CI: 0.964-1.032, P = 0.832), gender (female vs male, HR = 0.728, 95%CI: 0.416-1.282, P = 0.273), and type of TACE (lipiodol vs drug-eluting bead-TACE, HR = 1.016, 95%CI: 0.612-1.682, P = 0.976) did not show statistical significance in the multivariate analysis.
| Variables | Control | Single factor | Multi-factor | ||
| Age | Increase by 1 | Hazard ratio | P value | Hazard ratio | P value |
| Pre-treatment ETV (cm³) | ≥ 415 vs < 415 | 1.018 (0.982-1.036) | 0.453 | 1.008 (0.964-1.032) | 0.832 |
| Gender | Female vs male | 1.982 (1.212-3.236) | 0.012 | 1.442 (0.823-2.558) | 0.178 |
| Child-Pugh classification | Class A vs (class B or C) | 0.736 (0.438-1.264) | 0.234 | 0.728 (0.416-1.282) | 0.273 |
| BCLC staging | Stage B vs (stage C or D) | 0.423 (0.252-0.668) | < 0.001 | 0.432 (0.256-0.702) | < 0.001 |
| Type of TACE | Control | 0.523 (0.318-0.852) | 0.013 | 0.538 (0.318-0.916) | 0.023 |
| Variables | Lipiodol vs DEB-TACE | 1.178 (0.723-1.916) | 0.548 | 1.016 (0.612-1.682) | 0.976 |
In the analysis of the rate of ETV reduction (Table 7), univariate analysis showed that the rate of ETV reduction (≥ 41% vs < 41%, HR = 0.523, 95%CI: 0.346-0.918, P = 0.018), Child-Pugh classification (Class A vs class B or C, HR = 0.423, 95%CI: 0.252-0.668, P < 0.001), and BCLC staging (Stage B vs stage C or D, HR = 0.523, 95%CI: 0.318-0.852, P = 0.013) were significantly associated with patient prognosis. Multivariate analysis further confirmed that the rate of ETV reduction (HR = 0.412, 95%CI: 0.238-0.678, P < 0.001), Child-Pugh classification (HR = 0.298, 95%CI: 0.162-0.502, P < 0.001), and BCLC staging (HR = 0.578, 95%CI: 0.332-0.976, P = 0.045) were all independent prognostic factors. Similarly, age (HR = 1.008, 95%CI: 0.964-1.032, P = 0.812), gender (HR = 0.748, 95%CI: 0.432-1.328, P = 0.324), and type of TACE (HR = 0.832, 95%CI: 0.482-1.396, P = 0.464) did not show statistical significance in the multivariate analysis.
| Variable | Control | Single factor | Multi-factor | ||
| Hazard ratio | P value | Hazard ratio | P value | ||
| Age | Increase by 1 | 1.018 (0.982-1.036) | 0.453 | 1.008 (0.964-1.032) | 0.812 |
| ETV reduction rate | ≥ 41% vs < 41% | 0.523 (0.346-0.918) | 0.018 | 0.412 (0.238-0.678) | < 0.001 |
| Gender | Female vs male | 0.736 (0.438-1.264) | 0.234 | 0.748 (0.432-1.328) | 0.324 |
| Child-Pugh | Class A vs (class B or C) | 0.423 (0.252-0.668) | < 0.001 | 0.298 (0.162-0.502) | < 0.001 |
| BCLC staging | Stage B vs (stage C or D) | 0.523 (0.318-0.852) | 0.013 | 0.578 (0.332-0.976) | 0.045 |
| TACE type | Lipiodol vs DEB-TACE | 1.178 (0.723-1.916) | 0.548 | 0.832 (0.482-1.396) | 0.464 |
This study prospectively analyzed the application value of MRI bias field correction in volumetric imaging for assessing the therapeutic effects of TACE in iHCC, finding that bias field correction technology significantly improves the accuracy of volumetric measurements and provides more reliable quantitative indicators for predicting patient prognosis. This difference may stem from signal intensity variations caused by factors such as RF coil sensitivity non-uniformity, static field non-uniformity, and tissue magnetization rate differences during MRI imaging[14,15]. The improved N4 bias field correction algorithm can effectively compensate for these influences, providing volume data closer to reality[16]. This finding is consistent with previous studies reporting the impact of MRI bias fields on tumor volume measurements, but this study is the first to systematically validate the value of this technology in the assessment of TACE treatment for iHCC[17]. The study results showed that MRI bias field correction significantly affected the tumor volume measurements before and after TACE treatment, with the corrected ETV measurements being significantly lower than the uncorrected group (baseline ETV: 505.235 cm³ vs 825.632 cm³, P < 0.001; post-treatment ETV: 325.236 cm³ vs 632.523 cm³, P < 0.001), which is similar to the study by Koska et al[17].
Cox proportional hazards model analysis revealed that bias field correction significantly enhanced the predictive power of ETV as a prognostic indicator. The HR for the corrected baseline ETV (HR = 1.165) was significantly higher than the uncorrected group (HR = 1.063), indicating a stronger correlation between the corrected measurements and patient prognosis, similar to the study by Liu et al[18]. Notably, in terms of the percentage change in ETV, only the corrected group showed statistical significance (HR = 0.665, P < 0.001), while the uncorrected group failed to reflect this prognostic correlation (HR = 1.005, P = 0.793). This result indicates that bias field correction not only improves the accuracy of single measurements but, more importantly, enhances the reliability of longitudinal follow-up assessments[19,20]. The critical values determined by Martingale residual analysis (baseline ETV 412 cm³ and ETV reduction rate 40.253%) further support this conclusion, providing practical prognostic stratification standards for clinical practice[21].
Survival analysis results indicated that patient grouping based on corrected ETV had significant prognostic discrimination. The group with baseline ETV < 415 cm³ showed a longer median survival time compared to the ≥ 415 cm³ group (18.523 months vs 8.926 months), and similarly, the group with an ETV reduction rate ≥ 41% also demonstrated better prognosis compared to the < 41% group (17.862 months vs 9.235 months). This survival difference was consistent at different percentile time points, reflecting good temporal stability of prognosis stratification based on corrected ETV[22,23]. Multivariate analysis further confirmed that the ETV reduction rate (HR = 0.412, P < 0.001), along with Child-Pugh classification (HR = 0.298, P < 0.001) and BCLC staging (HR = 0.578, P = 0.045), constitutes independent prognostic factors. This finding highlights the supplementary value of volumetric response assessment based on the existing staging system[24,25].
The clinical significance of the study results is mainly reflected in three aspects: First, the application of bias field correction technology provides a more reliable tool for accurately assessing TACE treatment responses, helping to overcome technical limitations in traditional volumetric measurements[26]; second, the prognostic stratification standards based on corrected ETV have good clinical practicality and can be used to guide the formulation and adjustment of treatment strategies[27]; third, the discovery of ETV reduction rate as an independent prognostic factor provides a new quantitative indicator for assessing TACE treatment effects[28]. Despite significant strides in hepatocellular carcinoma imaging assessment, our study confronts numerous challenges and limitations. As a single-center retrospective research, we must candidly acknowledge its inherent methodological deficiencies: Potential selection bias may restrict the generalizability of our findings, while the relatively brief follow-up period fails to comprehensively capture long-term prognostic changes, necessitating more sustained and in-depth observations. By focusing primarily on tumor volume measurements, we risk overlooking complex imaging features such as tumor heterogeneity, potentially underestimating the multifaceted value of imaging indicators in disease progression.
To transcend current research limitations, future studies should pursue improvements across multiple dimensions. The primary task is to construct a multicenter prospective research paradigm, expanding sample size, enhancing research representativeness, and mitigating selection bias risks to provide more robust evidence. Simultaneously, we must actively integrate emerging imaging technologies like radiomics, moving beyond traditional volume measurements to comprehensively analyze tumor morphological characteristics and develop more dynamic and comprehensive prognostic assessment models. Research should not remain confined to technical dimensions but should explore the complex associations between imaging features and clinical prognosis, seeking pathways to personalized precision medicine. At the same time, it is recommended to incorporate bias field correction technology into routine imaging assessment processes to improve the accuracy of clinical decision-making.
Specifically, future research should prioritize four key strategic areas: Future investigations must transcend the limitations of single-center, retrospective studies by implementing robust, multicenter prospective research frameworks. This approach will enhance sample diversity, reduce potential selection biases, and improve the generalizability of research findings. Expanding sample size and incorporating diverse patient populations will provide more comprehensive and representative insights into hepatocellular carcinoma imaging assessment. The convergence of advanced imaging technologies, computational approaches, and clinical insights promises to revolutionize our understanding and management of hepatocellular carcinoma, transforming how we conceptualize and treat this complex disease.
In summary, this study confirms the important value of volumetric imaging based on MRI bias field correction in assessing the TACE treatment effects of iHCC, providing new technical means and clinical evidence to improve the accuracy of treatment response assessment and the reliability of prognostic prediction. These findings are of significant guidance for optimizing personalized treatment strategies for patients with invasive liver cancer.
| 1. | Wang S, Wang W, Zeng J. Role of CALCR expression in liver cancer: Implications for the immunotherapy response. Mol Med Rep. 2025;31:41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 2. | Ekanayaka SPN, Luke N, Thilakarathne SB, Dassanayake A, Gunetilleke MB, Niriella MA, Siriwardana RC. Characteristics and survival of advanced untreated hepatocellular carcinoma of non-viral etiology. Indian J Gastroenterol. 2024;43:1176-1183. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 3. | Shi J, Wei X, Jiang F, Zhu J, Shen J, Sun Y. Construction and validation of transcriptionfactorbased prognostic signature for TACE nonresponse and characterization of tumor microenvironment infiltration in hepatocellular carcinoma. Oncol Lett. 2025;29:42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Reference Citation Analysis (0)] |
| 4. | Sun R, Gou Y, Pan L, He Q, Zhou Y, Luo Y, Wu C, Zhao Y, Fu Z, Huang P. Hepatic arterial infusion chemotherapy (HAIC) combined with Tislelizumab and Lenvatinib for unresectable hepatocellular carcinoma: a retrospective single-arm study. Cell Oncol (Dordr). 2024;47:2265-2276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 5. | Zhu X, Zhang Z, Zhang J, Xiao Y, Wang H, Wang M, Jiang M, Xu Y. Single-cell and Bulk Transcriptomic Analyses Reveal a Stemness and Circadian Rhythm Disturbance-related Signature Predicting Clinical Outcome and Immunotherapy Response in Hepatocellular Carcinoma. Curr Gene Ther. 2025;25:178-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 6. | Hood S, Newall M, Butler P, O'Brien R, Petasecca M, Dillon O, Rosenfeld A, Hardcastle N, Jackson M, Metcalfe P, Alnaghy S. First linac-mounted photon counting detector for image guided radiotherapy: Planar image quality characterization. Med Phys. 2025;52:1159-1171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 7. | Sokmen BK, Inan N. 18 F-FDG PET/MRI of Primary Hepatic Malignancies: Differential Diagnosis and Histologic Grading. Curr Med Imaging. 2024;20:e080523216636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 8. | Singh R, Singh N, Kaur L. Deep learning methods for 3D magnetic resonance image denoising, bias field and motion artifact correction: a comprehensive review. Phys Med Biol. 2024;69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 9. | Gong S, Liu H, Gou H, Sun W. METTL5: A Potential Biomarker for Nonsmall Cell Lung Cancer That Promotes Cancer Cell Proliferation by Interacting with IGF2BP3. Genet Test Mol Biomarkers. 2024;28:311-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 10. | Yang S, Zhang Z, Su T, Chen Q, Wang H, Jin L. Comparison of quantitative volumetric analysis and linear measurement for predicting the survival of Barcelona Clinic Liver Cancer 0- and A stage hepatocellular carcinoma after radiofrequency ablation. Diagn Interv Radiol. 2023;29:450-459. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 11. | Ugonabo O, Mohamed M, Frandah W, Jones P, Joshi T. Imaging Negative Hepatic Lesions: A Rare Case of Infiltrative Hepatocellular Carcinoma Diagnosed With Endoscopic Ultrasound. ACG Case Rep J. 2022;9:e00945. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Reference Citation Analysis (0)] |
| 12. | Ellison C, Martens M, Alvarez Argote J, Benz S, Currey A, Johnstone C, Klawikowski S, Livingston K, Longo JM, Menon S, Ortiz de Choudens S, Puckett L, Retseck J, Shukla M, Thompson J, Gore E. High-Grade Pneumonitis Events in Patients With Unresectable, Locally Advanced NSCLC Treated With Definitive Chemoradiation Followed by Adjuvant Durvalumab. JTO Clin Res Rep. 2024;5:100537. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 13. | Diamond BH, Banson K, Ayash J, Lee P, Shukla UC, Jones G, Rava P, Fitzgerald TJ, Sioshansi S. Outcomes After Stereotactic Body Radiation for Hepatocellular Carcinoma in Patients With Child-Pugh A Versus Child-Pugh B/C Cirrhosis. Adv Radiat Oncol. 2024;9:101631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 14. | Jaiswal SK, Ojha BK, Jaiswal S, Bajaj A. Clinical Outcome and Predictors of Traumatic Cervical Injury: A Prospective Observational Study. Asian J Neurosurg. 2024;19:641-649. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Reference Citation Analysis (0)] |
| 15. | Zarfati A, Guérin F, Dioguardi Burgio M, Fuchs J, Sarnacki S, Losty PD, Pio L. Preoperative Identification of Adamkiewicz Artery in Pediatric Posterior Thoracic Tumors: Fact or Fiction? A Systematic Review from the International Society of Pediatric Surgical Oncology (IPSO). J Pediatr Surg. 2024;59:161985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 16. | Avard E, Shiri I, Hajianfar G, Abdollahi H, Kalantari KR, Houshmand G, Kasani K, Bitarafan-Rajabi A, Deevband MR, Oveisi M, Zaidi H. Non-contrast Cine Cardiac Magnetic Resonance image radiomics features and machine learning algorithms for myocardial infarction detection. Comput Biol Med. 2022;141:105145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 47] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 17. | Koska IO, Selver MA, Gelal F, Uluc ME, Çetinoğlu YK, Yurttutan N, Serindere M, Dicle O. Voxel level dense prediction of acute stroke territory in DWI using deep learning segmentation models and image enhancement strategies. Jpn J Radiol. 2024;42:962-972. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 18. | Liu C, Smolka S, Papademetris X, Minh DD, Gan G, Deng Y, Lin M, Chapiro J, Wang X, Georgiades C, Hong K. Predicting Infiltrative Hepatocellular Carcinoma Patient Outcome Post-TACE: MR Bias Field Correction Effect on 3D-quantitative Image Analysis. J Clin Transl Hepatol. 2020;8:292-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 19. | Kumar S S, S P S, R S. Parallel-way: Multi-modality-based brain tumor segmentation using parallel capsule network. Electromagn Biol Med. 2024;43:267-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 20. | Veiga-Canuto D, Fernández-Patón M, Cerdà Alberich L, Jiménez Pastor A, Gomis Maya A, Carot Sierra JM, Sangüesa Nebot C, Martínez de Las Heras B, Pötschger U, Taschner-Mandl S, Neri E, Cañete A, Ladenstein R, Hero B, Alberich-Bayarri Á, Martí-Bonmatí L. Reproducibility Analysis of Radiomic Features on T2-weighted MR Images after Processing and Segmentation Alterations in Neuroblastoma Tumors. Radiol Artif Intell. 2024;6:e230208. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 21. | Aydin S, Schmitz J, Dellacasa CM, Dogliotti I, Giaccone L, Busca A. WT1 Expression Is Associated with Poor Overall Survival after Azacytidine and DLI in a Cohort of Adult AML and MDS Patients. Cancers (Basel). 2024;16:3070. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 22. | Izah J, Haizel-Cobbina J, Zhao S, Vance EH, Dunlap M, Gannon SR, Liles C, Yengo-Kahn AM, Pontell ME, Naftel RP, Wellons JC, Dewan MC. Treatment of hydrocephalus following fetal repair of myelomeningocele: comparing endoscopic third ventriculostomy with choroid plexus cauterization to ventricular shunting. J Neurosurg Pediatr. 2024;34:207-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 23. | Bogaczyk V, Fleck S, Berneiser J, Opolka M, Vollmer M, Baldauf J, Gasch CM, Lemke EM, Refaee EE, Matthes M, Hirschfeld H, Lauffer H, Gaab M, Schroeder HWS, Marx S. Correction to: Longterm quality of life after ETV or ETV with consecutive VP shunt placement in hydrocephalic pediatric patients. Childs Nerv Syst. 2022;38:1895. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 24. | Peyraut A, Genet M. A model of mechanical loading of the lungs including gravity and a balancing heterogeneous pleural pressure. Biomech Model Mechanobiol. 2024;23:1933-1962. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 25. | Hubbeling H, Leithner D, Silverman EA, Flynn J, Devlin S, Shah G, Fregonese B, Wills B, Bedmutha A, Alarcon Tomas A, Parascondola A, Saldia A, Landego I, Hajj C, Boardman AP, Dahi PB, Ghosh A, Giralt S, Lin RJ, Park J, Scordo M, Salles G, Yahalom J, Palomba ML, Schöder H, Perales MA, Shouval R, Imber BS. Metabolic Tumor Volume Response after Bridging Therapy Determines Chimeric Antigen Receptor T-Cell Outcomes in Large B-Cell Lymphoma. Clin Cancer Res. 2024;30:5083-5093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 26. | Ott K, Cepeda S, Hartmann D, Kramer F, Müller D. Predicting Overall Survival of Glioblastoma Patients Using Deep Learning Classification Based on MRIs. Stud Health Technol Inform. 2024;317:356-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 27. | Gupta R, Bilgin C, Jabal MS, Kandemirli S, Ghozy S, Kobeissi H, Kallmes DF. Quality Assessment of Radiomics Studies on Functional Outcomes After Acute Ischemic Stroke-A Systematic Review. World Neurosurg. 2024;183:164-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 28. | Li MR, Xi HL, Wang QH, Hou FQ, Huo N, Zhang XX, Li F, Xu XY. Kinetics and prediction of HBsAg loss during long-term therapy with nucleos(t)ide analogues of different potency in patients with chronic hepatitis B. PLoS One. 2014;9:e98476. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/