Published online Aug 27, 2023. doi: 10.4240/wjgs.v15.i8.1629
Peer-review started: April 26, 2023
First decision: May 25, 2023
Revised: June 6, 2023
Accepted: July 4, 2023
Article in press: July 4, 2023
Published online: August 27, 2023
Processing time: 121 Days and 3.3 Hours
Minimally invasive surgery had been tailored to individual cases of gastric subepithelial tumors (SETs) after comparing the clinical outcomes of endoscopic resection (ER), laparoscopic resection (LR), and hybrid methods.
To study the use of Goldilocks principle to determine the best form of minimally invasive surgery for gastric SETs.
In this retrospective study, 194 patients of gastric SETs with high probability of surgical intervention were included. All patients underwent tumor resection in the operating theater between January 2013 and December 2021. The patients were divided into two groups, ER or LR, according to the tumor characteristics and the initial intent of intervention. Few patients in the ER group required further backup laparoscopic surgery after an incomplete ER. The patients who had converted open surgery were excluded. A logistic regression model was used to assess the associations between patient characteristics and the likelihood of a treatment strategy. The area under the curve was used to assess the discriminative ability of tumor size and Youden’s index to determine the optimal cut-off tumor size.
One-hundred ninety-four patients (100 in the ER group and 94 in the LR group) underwent tumor resection in the operating theater. In the ER group, 27 patients required backup laparoscopic surgery after an incomplete ER. The patients in the ER group had small tumor sizes and shorter procedure durations while the patients in the LR group had large tumor sizes, exophytic growth, malignancy, and tumors that were more often located in the middle or lower third of the stomach. Both groups had similar durations of hospital stays and a similar rate of major postoperative complications. The patients in the ER group who underwent backup surgery required longer procedures (56.4 min) and prolonged stays (2 d) compared to the patients in the LR group without the increased rate of major postoperative complications. The optimal cut-off point for the tumor size for laparoscopic surgery was 2.15 cm.
Multidisciplinary teamwork leads to the adoption of different strategies to yield efficient clinical outcomes according to the tumor characteristics.
Core Tip: Multidisciplinary teamwork leads to the adoption of different strategies for resection of gastric subepithelial tumors: Endoscopic resection (ER) was indicated for a smaller tumor and intraluminal growth, whereas laparoscopic resection was indicated for a larger tumor (optimal cut-off point: 2.15 cm), tumors located in the middle or lower third of the stomach, exophytic growth, and more aggressive malignancy behavior. Backup surgery is preserved for incomplete ER to effectively reduce associated morbidities.
- Citation: Chang WJ, Tsao LC, Yen HH, Yang CW, Chang HC, Kor CT, Wu SC, Lin KH. Goldilocks principle of minimally invasive surgery for gastric subepithelial tumors. World J Gastrointest Surg 2023; 15(8): 1629-1640
- URL: https://www.wjgnet.com/1948-9366/full/v15/i8/1629.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v15.i8.1629
Gastric subepithelial tumors (SETs) include a broad spectrum of benign and malignant lesions, of which gastrointestinal stromal tumors (GISTs) are the most common. Current guidelines recommend the complete resection of gastric SETs if the size is > 2 cm, malignant features are present, or the patient is symptomatic and would prefer surgical management[1-5]. As small GISTs pose a risk for malignancy, endoscopic resection (ER) could be a good alternative method for obtaining a histological diagnosis and therapeutic resection compared with periodic surveillance[6]. With recent advancements in endoscopic and laparoscopic management, different approaches to minimally invasive surgery have been adopted and tailored to individual cases.
Among the minimally invasive approaches, laparoscopic surgery has proven to be feasible with faster recovery, shorter hospital stays, and equivalent oncological safety compared to open surgery. Initially, open surgery was considered the main treatment for extensive tumors, and laparoscopic surgery was reserved for small tumors (< 5 cm). However, with advanced laparoscopic techniques, tumor size is no longer a restricting factor; even large tumors can be successfully removed via laparoscopic surgery[7-9]. For certain tumors in unfavorable locations[10], Hiki et al[11] introduced laparoscopic and endoscopic cooperative surgery (LECS), with the combined advantages of endoscopy and laparoscopy[12], which helps to achieve precise localization, minimal resection, and functional preservation. Endoscopic submucosal dissection (ESD) techniques have advanced in the resection of tumors located deeper than the submucosal layer[13]. The ER of small gastric SETs (< 5 cm) involves a shorter surgery and less intraoperative blood loss in selected cases of intraluminal tumors[14,15]. By considering complications, such as perforation or bleeding, we modified the LECS procedure as a backup laparoscopic surgery to provide timely management, which required more operative time but reduced postoperative morbidity[16].
Although different minimally invasive approaches can be applied to gastric SETs, the effectiveness and safety of ER, laparoscopic resection (LR), or hybrid methods have not been well established. Thus, in this study, we aimed to use the Goldilocks principle to determine the best type of treatment for gastric SETs by comparing the clinical outcomes of ER, LR, and our hybrid method, as this information can be crucial in improving options for minimally invasive surgery considering the risks and potential benefits in this setting.
We conducted a retrospective study of patients with gastric SETs who underwent ER or LR at the operating theater in our institution between January 2013 and December 2021. Medical records were retrospectively reviewed to define the patient/tumor characteristics and operative outcomes. Based on pathologic diagnosis, the tumor was further divided into two groups: benign disease and malignant or malignant potential disease. All patients underwent endoscopic ultrasonography (EUS) or abdominal computed tomography (CT) to evaluate the tumor size, invasion depth, and characteristics before resection.
The study was approved by the Institutional Review Board of Changhua Christian Hospital (approval No. 220117) and registered at ClinicalTrials.gov (NCT05452265). This work has been reported in line with the “Strengthening the Reporting of Observational studies in Epidemiology (STROBE)” criteria[17]. All relevant data are included in the paper and its Supporting Information files.
Complete resection of gastric SETs is recommended if the tumor size is > 2 cm, malignant features are present, or if the patient is symptomatic, declined periodical surveillance, and preferred to undergo diagnostic and therapeutic resection. Patients with gastric SETs in the superficial layer underwent ER in the endoscopic room and those with a high probability of surgical intervention were evaluated both by endoscopists and general surgeons preoperatively[16,18]. ER with backup surgery was indicated for patients with endoscopic intent, and a small tumor size tolerated the endoscopic retrieval. On the other hand, LR was indicated for patients with surgical intent and those with the following conditions, which were not suitable for ER: (1) Large tumor size with difficult endoscopic retrieval; (2) Symptoms of gastrointestinal tract bleeding with difficulty in endoscopic visualization; (3) Suspicion of tumor rupture that required intra-abdominal exploration; and (4) Histologic diagnosis of GIST with initial treatment of target therapy. Open surgery was performed in patients who were not amenable to laparoscopy due to pulmonary compliance and cardiovascular disease, had large tumors that eventually needed a large incision wound for specimen extraction, and had tumors with suspected multivisceral involvement.
In total, we included 194 patients who underwent tumor resection under general anesthesia at the operating theater, with 100 and 94 patients in the ER and LR groups, respectively. We excluded three patients in the ER group due to anatomic changes in the stomach following previous surgery and four patients in the LR group due to converted open surgery. Among the four patients who underwent converted open surgery, one had splenic metastasis with difficult dissection plain intraoperatively (tumor size 12.5 cm, posterior aspect of the upper third stomach), two had difficulty in tumor localization of the upper third stomach with one receiving preoperative endoscopic tattoo (size 4 cm, posterior side and size 1 cm, lesser curvature side with tattooing), and one was concerned with post-gastrectomy stenosis with further gastro-gastrostomy (size 7 cm, posterior aspect of the lower third stomach).
ER only, ER with backup surgery, and LR were performed according to the protocols in our previous studies[16,18]. In the ER group, most patients (92, 92%) underwent endoscopic submucosal dissection (ESD) and endoscopic mucosal resection if necessary for an R0 attempt, except for eight cases of submucosal tunneling ER (STER). Some cases with iatrogenic perforation could be successfully repaired by endoscope (Video 1). In the LR group, most patients (92, 97.9%) underwent wedge gastrectomy, except for two cases who underwent distal gastrectomy with Roux-en-Y reconstruction (2, 2.1%), given the risk for postoperative stenosis. In addition, 12 patients (12.8%) underwent intraoperative endoscope-assisted LR to localize the tumor more precisely.
Categorical and continuous variables are expressed as number (proportion) and median and interquartile range (IQR), respectively. Chi-squared test was used to compare categorical variables, and Kruskal-Wallis H test was used to compare continuous variables. A logistic regression model was used to assess the association between patient characteristics and likelihood of a treatment strategy. Odds ratios were calculated using a crude multivariate analysis and a 1:1 propensity-matched dataset. Forest plots provide a data visualization method to present multivariate adjustment factors for the likelihood of undergoing treatment. Linear regression models were used to assess the impact of the three treatment strategies on clinical outcomes (procedure time, length of hospital stay, and Clavien grade ≥ III complications). Furthermore, we used the area under the curve (AUC) to assess the discriminative ability of tumor size and Youden’s index to determine the optimal cut-off tumor size. Kaplan–Meier curves and log-rank tests were used to compare the disease-free survival rates and overall survival between the ER and LR groups during long-term surveillance. Statistical analyses were performed using SAS (version 9.4; SAS Institute Inc., Cary, NC, United States), and a visualization plot was constructed using the R software (version 4.1.0; Comprehensive R Archive Network: http://cran.r-project.org). All two-sided P values less than 0.05 were considered statistically significant.
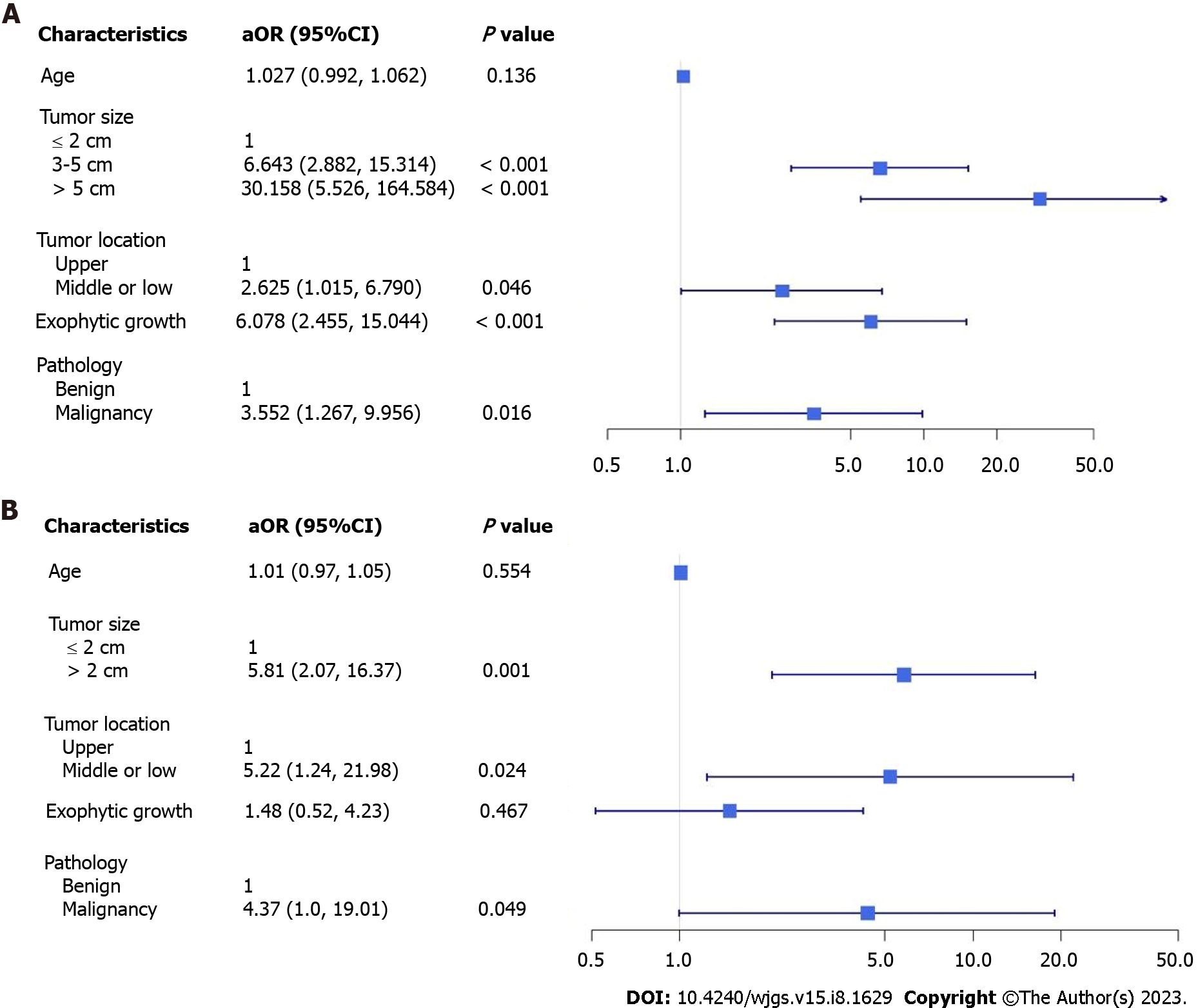
A total of 194 patients were included: 100 in the ER group and 94 in the LR group. In the ER group, 73 patients underwent ER only, and 27 underwent further backup laparoscopic surgery due to uncontrolled bleeding or incidental perforation. There were no significant differences in sex, the layer of tumor origin, length of hospitalization, or major postoperative complications between the ER and LR groups (Table 1). In the ER group, patients who were slightly younger (56 vs 62 years) had a significantly higher percentage of small tumor sizes of ≤ 2 cm (72% vs 16%) and shorter procedure times (75 vs 130 min). In the LR group, significant differences were observed in the percentage of tumors > 3 cm in size (84% vs 28%), tumors in the middle third of the stomach (14.9% vs 6%), exophytic tumor growth (56.4% vs 11%), and pathology of malignancy or malignant potential (85.1% vs 45%). Multivariable analysis results showed that patients tended to undergo laparoscopic surgery rather than ER when the following factors were present: tumor size of > 2 cm (3-5 cm adjusted odds ratio (aOR) 6.643, > 5 cm aOR 30.158), tumor in the middle or lower third of the stomach (aOR 2.625), exophytic growth (aOR 6.0782), or pathology of malignancy (aOR 3.552). However, it is possible that these factors resulted from preoperative selection bias (Figure 1A).
| ESD with/without backup surgery (n = 100) | Laparoscopic surgery (n = 94) | OR (95%CI) | P value | |
| Age, yr, median (range) | 56 (49-62) | 62 (52-70) | 1.04 (1.02-1.07) | 0.001a |
| Male gender, n (%) | 46 (46) | 47 (50) | 1.17 (0.67-2.06) | 0.577 |
| Tumor size, n (%) | ||||
| ≤ 2 cm | 72 (72) | 15 (16) | 1 | |
| 3-5 cm | 26 (26) | 54 (57.4) | 9.97 (4.82-20.62) | < 0.001a |
| > 5 cm | 2 (2) | 25 (26.6) | 60 (12.81-281) | < 0.001a |
| Tumor location, n (%) | ||||
| Upper | 81 (81) | 61 (64.9) | 1 | |
| Middle | 6 (6) | 14 (14.9) | 3.1 (1.13-8.53) | 0.029a |
| Low | 13 (13) | 19 (20.2) | 1.94 (0.89-4.23) | 0.096 |
| Layer of tumor origin, n (%) | ||||
| Submucosa | 6 (6) | 3 (3.2) | 1 | |
| Muscularis propria | 94 (94) | 91 (96.8) | 1.94 (0.47-7.97) | 0.36 |
| Exophytic growth, n (%) | 11 (11) | 53 (56.4) | 10.46 (4.95-22.08) | < 0.001a |
| Pathology, n (%) | ||||
| Benign | 55 (55) | 14 (14.9) | 1 | |
| Malignant or malignant potential | 45 (45) | 80 (85.1) | 6.98 (3.5-13.94) | < 0.001a |
| Procedure time, min, median (IQR) | 75 (40-138) | 130 (100-169) | - | < 0.001a |
| Length of stay, d, median (IQR) | 5 (4-7) | 6 (4-6) | - | 0.923 |
| Clavien ≥ III complication, n (%) | 5 (5) | 3 (3.2) | - | 0.527 |
A total of 27 patients who underwent backup surgery after incomplete ER were compared with those in the LR group (Table 2). No significant differences were observed in age, sex, tumor location, the layer of tumor origin, exophytic tumor growth, or major postoperative complications; however, a higher percentage of tumors of size > 2 cm (84% vs 40.7%), pathology of malignancy or malignant potential (85.1% vs 59.3%), prolonged procedure duration (130 vs 185 min), and a shorter length of hospital stay (6 vs 7 d) were observed in the LR group. Multivariable analysis showed that patients tended to undergo laparoscopic surgery rather than an initial ER attempt if the tumor size was > 2 cm (aOR 5.81), if the tumor was in the middle or lower third of the stomach (aOR 5.22), and if there was pathology of malignancy (aOR 4.37); these results were statistically significantly different (Figure 1B).
| ESD with backup surgery (n = 27) | Laparoscopic surgery (n = 94) | OR (95%CI) | P value | |
| Age, yr, median (range) | 56 (50-64) | 62 (52-70) | 1.03 (1.00-1.06) | 0.053 |
| Male gender, n (%) | 9 (33.3) | 47 (50) | 2.00 (0.82-4.90) | 0.13 |
| Tumor size, n (%) | ||||
| ≤ 2 cm | 16 (59.3) | 15 (16) | 1 | |
| 3-5 cm | 11 (40.7) | 54 (57.4) | 7.66 (2.98-19.72) | < 0.001a |
| > 5 cm | 0 (0) | 25 (26.6) | ||
| Tumor location, n (%) | ||||
| Upper | 23 (85.2) | 61 (64.9) | 1 | |
| Middle | 1 (3.7) | 14 (14.9) | 3.11 (0.99-9.76) | 0.052 |
| Low | 3 (11.1) | 19 (3.2) | ||
| Layer of tumor origin, n (%) | ||||
| Submucosa | 0 (0) | 3 (3.2) | - | 1 |
| Muscularis propria | 27 (100) | 91 (96.8) | - | |
| Exophytic growth, n (%) | 10 (37) | 53 (56.4) | 2.20 (0.91-5.3) | 0.08 |
| Pathology, n (%) | ||||
| Benign | 11 (40.7) | 14 (14.9) | 1 | |
| Malignant or malignant potential | 16 (59.3) | 80 (85.1) | 3.93 (1.51-10.21) | 0.005a |
| Procedure time, min, median (IQR) | 185 (150-245) | 130 (100-169) | - | < 0.001a |
| Length of stay, d, median (IQR) | 7 (7-8) | 6 (4-6) | - | < 0.001a |
| Clavien ≥ III complication, n (%) | 0 (0) | 3 (3.2) | - | 0.347 |
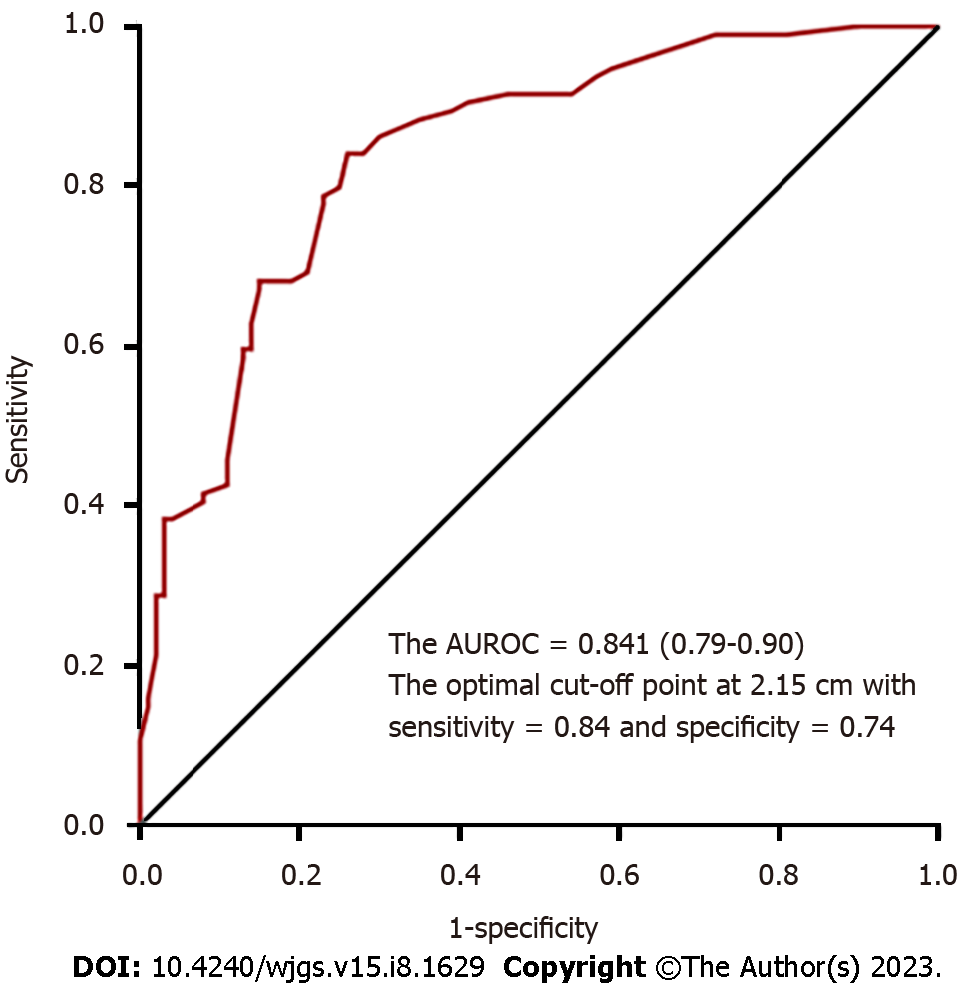
To compare the operative outcomes between ER with backup surgery and LR, the predictor of a prolonged procedure was tumor location in the middle or lower third of the stomach, and the predictor of a prolonged stay was advanced age. However, these factors were not significant after propensity-score matching. More importantly, patients who underwent ER with backup surgery had longer procedures (56.4 min) and prolonged hospital stays (2 d) on average (Table 3). Furthermore, the optimal cut-off point for the tumor size for laparoscopic surgery was 2.15 cm, with an AUC of 0.841, sensitivity of 84%, and specificity of 74% (Figure 2).
| Factors | Without matched data | Propensity score matching | ||||
| Adjusted mean difference | SE | P value | Adjusted mean difference | SE | P value | |
| Procedure time | ||||||
| Surgery type | ||||||
| ESD with backup surgery | Reference | Reference | ||||
| Laparoscopic surgery | -63.42 | 14.70 | < 0.001a | -56.40 | 18.60 | 0.005a |
| Age | -0.43 | 0.42 | 0.304 | - | - | - |
| Tumor size 2 | 22.04 | 13.35 | 0.101 | - | - | - |
| Tumor location: middle or low | 33.58 | 12.54 | 0.008a | - | - | - |
| Exophytic growth | -4.54 | 10.98 | 0.680 | - | - | - |
| Pathology with malignancy | -13.81 | 15.59 | 0.378 | - | - | - |
| Length of stay | ||||||
| Surgery type | ||||||
| ESD with backup surgery | ||||||
| Laparoscopic surgery | -1.215 | 1.089 | 0.267 | -2.00 | 0.516 | 0.001a |
| Age | 0.081 | 0.031a | 0.009a | - | - | - |
| Tumor size 2 | -0.348 | 0.988 | 0.726 | - | - | - |
| Tumor location: middle or low | 0.457 | 0.928 | 0.624 | - | - | - |
| Exophytic growth | -0.218 | 0.813 | 0.789 | - | - | - |
| Pathology with malignancy | -0.268 | 1.154 | 0.817 | - | - | - |
Among patients with major complications (Clavien grade ≥ III), there were five patients (5.3%) in the ER group (three graded IIIa and two graded IIIb), none after backup surgery, and three patients (3.2%) in the LR group (two graded IIIa and one graded IVa). In the ER group, three patients were graded IIIa, two patients had gastric ulcer bleeding and received endoscopic hemostasis, and one patient had massive pneumoperitoneum without peritonitis and received sonography-guided air tapping. Two patients with grade-IIIb perforation had delayed perforation and underwent laparoscopic surgery[15]. In the LR group, two patients with grade IIIa had delayed gastric emptying after laparoscopic wedge gastrectomy and received endoscopic duodenal tube insertion for enteral feeding on postoperative days (POD) 18 and 19, respectively. One patient with grade IVa developed pneumonia with acute respiratory failure on POD 3 and received intensive critical care thereafter. All patients recovered from the complications and were discharged.
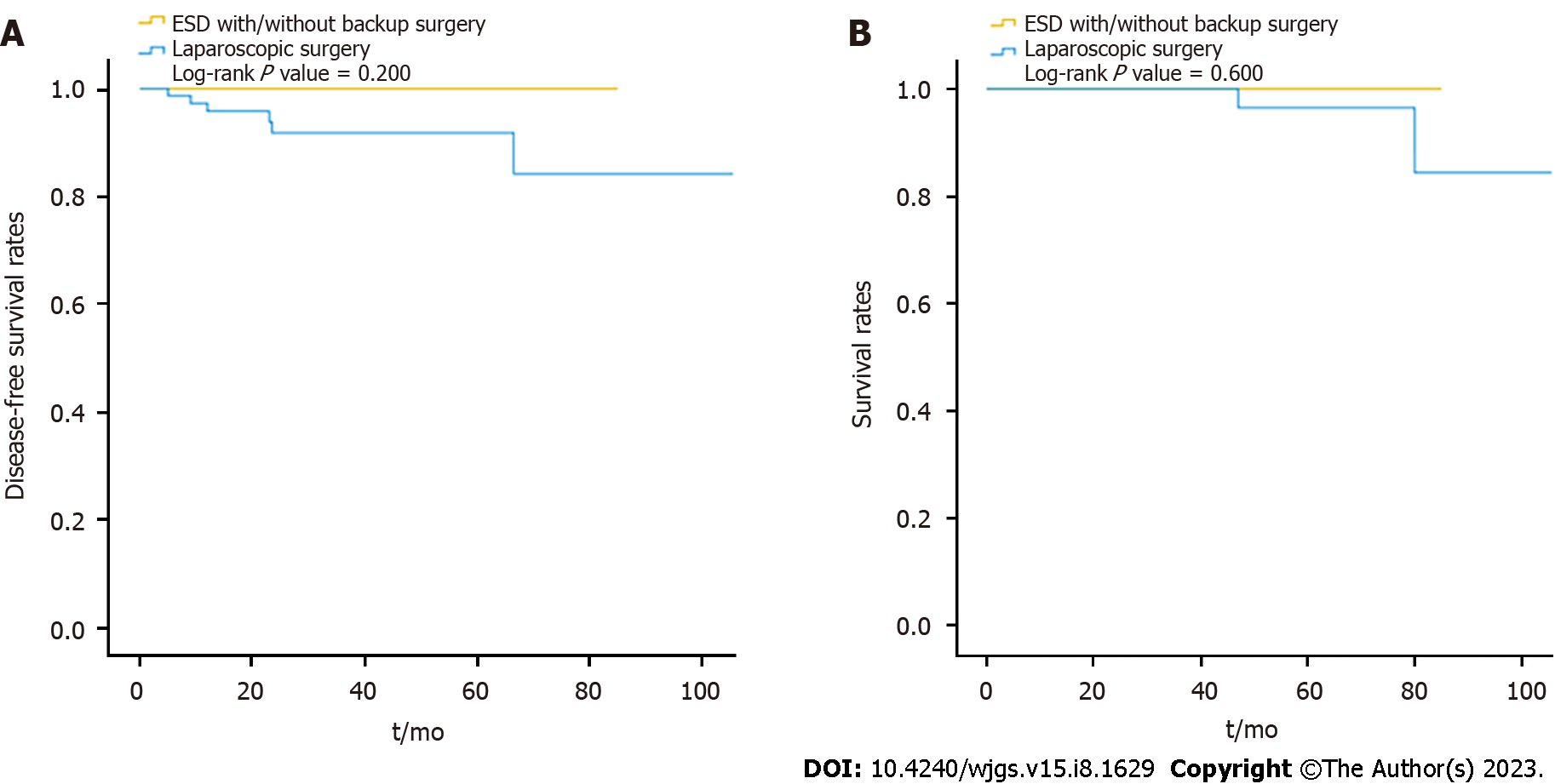
The pathology of the gastric SETs is shown in Table 4. Before laparoscopic surgery, the rate of endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) was 17% (16/94), and the diagnostic accuracy was 10.6% (10/94), with one complication and one perforation. Nine patients had cytopathological diagnoses of GIST, and two patients received neoadjuvant target therapy with imatinib at a dose of 400 mg/day for one year preoperatively. Overall, the percentage of malignant pathology (85.1% vs 45%, P < 0.001), particularly the composition of GIST with intermediate and high risk (39.7% vs. 11.6%), and the recurrence rate (6.4% vs 0, P = 0.012) were significantly higher in the LR group, except for disease-related mortality (Table 4). In the ER group, the majority of the patients diagnosed with GIST were categorized into the very low-risk group (60.5%), followed by the low-risk (27.9%), and the intermediate-risk (11.6%) groups, according to the NIH classification[18]. They underwent long-term surveillance for a mean duration of 27.5 mo, and no recurrence was detected. One patient, diagnosed with neuroendocrine tumor grade 1, received further gastrectomy and adjuvant chemotherapy without recurrence during a 64-month follow-up period. In the LR group, 78 patients were diagnosed with GISTs, with five cases of complicated bleeding and two ruptures, and were categorized into the very low-risk (11, 14.1%), low-risk (36, 46.2%), intermediate-risk (20, 25.6%), and high-risk (11, 14.1%) groups. A total of 12 patients (15.4%) received post-operative adjuvant imatinib therapy. They had long-term surveillance for a mean duration of 37.3 mo; four cases were reported to have recurrences (two local recurrences, one liver metastasis, and one mesenteric metastasis) with a mean disease-free survival of 12.3 mo. Four patients who were still alive during follow-up received target therapy, and one patient with mesenteric metastasis underwent further surgery for tissue proof and occult obstruction. One patient diagnosed with lipoleiomyosarcoma had liver metastasis, with a disease-free survival time of 23.5 mo. The liver metastasis progressed, and the patient died of inferior vena cava syndrome, with an overall survival of 47 mo. Another patient with a neuroendocrine tumor also had liver metastasis, with a disease-free survival time of 66.5 mo. She received octreotide treatment but died due to multiple distant metastases, with an overall survival of 80 mo. The remaining five patients died from other disorders, with an overall mortality rate of 7.4% and a disease-related mortality rate of 2.1%. Overall, although the disease-free survival and survival rates were slightly decreased in the LR group during long-term surveillance, the difference was not significant (log-rank P = 0.600, 0.200) (Figure 3).
| ESD with/without backup surgery (n = 100) | Laparoscopic surgery (n = 94) | P value | |
| Malignant pathology in the group, n (%) | 45 (45) | 80 (85.1) | < 0.001a |
| Malignant pathology | |||
| GIST, n (%) | 43 (43) | 78 (83.1) | < 0.001a |
| High risk | 0 (0) | 11 (14.1) | 0.001a |
| Intermediate risk | 5 (11.6) | 20 (25.6) | 0.002a |
| Low risk | 12 (27.9) | 36 (46.2) | < 0.001a |
| Very low risk | 26 (60.5) | 11 (14.1) | 0.019a |
| Neuroendocrine tumor, n (%) | 1 (1) | 1 (1) | 1.000 |
| Lipoleiomyosarcoma, n (%) | 0 | 1 (1) | 0.485 |
| Recurrence, n (%) | 0 | 6 (6.4) | 0.012a |
| Disease related mortality, n (%) | 0 | 2 (2.1) | 0.233 |
For gastric SETs, complete tumor removal with a free margin, minimal resection of the normal stomach, and avoiding pseudocapsule rupture are the goals of the present treatment. With recent advances in endoscopic techniques, ER has become an alternative option because the endoscopic approach maintains the integrity and anatomical function of the stomach without damaging the abdominal wall. More studies have compared clinical outcomes between ER and LR for gastric SETs of size ≤ 5 cm, and ER has the advantages of lower invasiveness, a shorter procedure duration[14,15,19-23], faster recovery[20,21,23,24], and a shorter hospital stay[20,21,24,25] in selected cases with smaller tumor sizes[14,15,19,20,24,25] and intraluminal growth[14,15,20]. However, ER-related complications are still a greater concern for prolonged hospital stays than those for LR[22]. From our experience, we also found that ER had the advantage of a shorter procedure duration than LR, while similar outcomes for hospital stays and major postoperative complications were observed. Where backup surgery was required to address incomplete ER, which could effectively reduce post-ER morbidity[16], prolonged procedures and hospital stays were observed compared with LR. Thus, ER is considered an effective and less invasive approach in selected cases if the tumor can be successfully resected.
Although ER tends to be used to manage smaller tumors, debates regarding the usual size for minimally invasive surgery for gastric SETs continue. For ER, a systematic review has recorded the average diameter of gastric GISTs, which ranges from 1.1 to 3.8 cm[14]. Recently, the European Society of Gastrointestinal Endoscopy (ESGE) guidelines have suggested that ER for gastric GISTs of size < 3.5 cm with intraluminal growth is an alternative to laparoscopic surgery[6]. With advances in endoscopic techniques for the closure of large gastric wall defects, a Chinese study found that it is feasible to treat giant gastric SETs of size ≥ 6 cm by ER with favorable long-term outcomes, although the minimum diameter of the tumor was associated with en bloc resection[26]. In our early period of ER, there were two cases of tumors > 5 cm in size, and we found it difficult to perform endoscopic retrieval of the entire tumor. Although piecemeal ER was feasible for complete resection, we tended to use en bloc resection for smaller tumors, with a rate of 90.4% based on our data.
On the other hand, laparoscopic surgery was initially suggested for tumors < 5 cm in size with a risk for tumor rupture and concern for oncological safety[27]. However, with the development of techniques and energy devices, laparoscopic surgery is no longer limited by the tumor size. Laparoscopic surgery provides a clear and broad field of vision that facilitates more sophisticated dissection and timely treatment of intraoperative bleeding, thereby realizing equivalent oncological safety and even better 5-year disease-free survival for large tumors (> 5 cm) compared to open surgery[7-9]. In our study, 26.6% of the tumors in the LR group were large tumors, and we also used this for two cases with preoperative target therapy for one year. We continued adjuvant therapy with imatinib at a dose of 400 mg/d postoperatively, and the patients were still disease-free at 25 and 39.5 mo during follow-up. Nevertheless, laparoscopy was limited by its decreased tactile feedback and difficulty in tumor localization; therefore, endoscopic assistance is suggested for small tumors (≤ 1.8 cm) and intraluminal growth types[28]. In our study, intraoperative endoscopy was required to precisely localize the tumors in 12 patients (12.8%) in the LR group. Overall, from our practical experience, we found that the optimal cut-off point for the tumor size for laparoscopic surgery is 2.15 cm.
In our study, patients with tumors in the middle or lower third of the stomach tended to undergo laparoscopic surgery rather than ER or backup surgery, which might have resulted from the initial selection bias of endoscopists and surgeons considering the tumor size. However, the location in the upper third of the stomach has been reported as a risk factor for perforation due to the relatively thin gastric wall and difficultly in endoscopic angulation[18,29]. With the technical expertise of experienced endoscopists, gastric SETs in the upper third of the stomach could be successfully removed with ER. In addition, we considered that ER for gastric SETs in the anterior wall of the stomach body had a high probability of surgical intervention because intragastric gas leakage into the peritoneal cavity without a soft tissue boundary made endoscopic repair more difficult, owing to a poor visual field and a gradually distended abdominal wall[16]. A Japanese study reported that surgeons found it difficult to endoscopically repair large defects on the anterior gastric wall and resorted to laparoscopic surgery[15]. These reasons may explain why tumors in the middle or lower third of the stomach tend to be treated via laparoscopic surgery.
Tumors with exophytic growth have a high risk for perforation[16], and their size may be larger than those without exophytic growth. In the present study, exophytic growth was significantly different between the ER and LR groups; however, no significant difference was observed between the backup surgery and LR groups. Patients with exophytic tumor growth eventually underwent laparoscopic surgery because of the high risk for incidental perforation after ER and easy localization during laparoscopic exploration. Based on the above findings, we assumed that exophytic tumor growth was undoubtedly indicative of LR.
Furthermore, patients with malignant tumors or malignant potential tended to undergo LR (85.1% vs 45%; OR, 6.98; P < 0.001). For malignant pathology, oncological safety is still a concern for ER because the complete resection rate has been found to be lower in ER than that in surgery[23,25]. Nevertheless, ER for small tumor sizes has still achieved R0 resection rates of up to 97%, according to a systematic review and meta-analysis[14,30,31], and no significant differences in long-term oncological outcomes for GISTs have been observed between ER and LR [23]. For gastric GISTs, tumor size is a key factor for recurrence risk rather than resection status, if no macroscopic residual tumor exists[24,29,32] and should be cautiously considered for different strategies of minimally invasive surgery. A previous study suggested that gastric GISTs that were completely resected endoscopically carry a lower stratified risk for aggressive clinical outcomes[24,32,33], and we found similar results in our study. In the ER group, the majority (60.5%) of the GISTs had a very low risk due to their small sizes and lack of disease recurrence during follow-up. In the LR group, 39.7% of the GISTs were categorized as intermediate- or high-risk. Overall, a higher recurrence rate (6.4%) and disease-related mortality rate (2.1%) were observed in the LR group; however, no significant difference was observed during long-term surveillance. Although all the patients underwent complete resection, the en bloc resection rate after ER was only 90.4%, which is not of high concern for LR. For tumors with suspected aggressive behavior of malignant pathology preoperatively, we preferred laparoscopic surgery to achieve similar oncological outcomes.
This study has some limitations. First, it was conducted at a single center with a relatively small sample size. Second, selection bias existed between the ER and LR groups because endoscopists and surgeons evaluated the patients preoperatively to make collaborative decisions regarding minimally invasive surgical methods. A few patients with gastric SETs in the superficial submucosal layer who underwent ER in the endoscopic room were not included in this database. Third, although GISTs are the most common type of malignant pathology, we focused on gastric SETs and other malignant pathologies. Thus, we focused on perioperative clinical outcomes after ER and LR, whereas the long-term outcomes of malignant pathologies require further analysis. Fourth, different ER and LR methods were chosen by endoscopists and surgeons according to tumor characteristics and location. However, we were unable to perform a detailed comparison of the different methods because of the small sample size.
There are different approaches to minimally invasive surgery for gastric SETs with the objective of achieving better perioperative clinical outcomes. ER was indicated for smaller tumor sizes and intraluminal growth, whereas LR was indicated for larger tumor sizes, with an optimal tumor size cut-off point of 2.15 cm, tumors located in the middle or lower third of the stomach, exophytic tumor growth, and more aggressive malignant behavior. Multidisciplinary teamwork is an effective strategy for selecting suitable treatments, leading to better clinical outcomes.
With recent advancements in endoscopic and laparoscopic management of gastric subepithelial tumors (SETs), different approaches to minimally invasive surgery have been adopted to improve the clinical outcomes.
To treat gastric SETs, the effectiveness and safety of endoscopic resection (ER), laparoscopic resection (LR), or our hybrid method were compared in terms of procedure duration, duration of hospital stay, and major complications.
This retrospective study compared the differences between ER and LR, and between ER with backup surgery and LR, in terms of demographic data, tumor characteristics, and perioperative outcomes. Thus, Goldilocks principle was used to determine the best type of minimally invasive surgery for gastric SETs.
This retrospective review of records was performed on all patients of gastric SETs with high probability of surgical intervention undergoing tumor resection in the operating theater between January 2013 and December 2021. All patients were divided into two groups, either group of ER or group of LR.
Totally, 194 patients were divided into the ER group (n = 100) and LR group (n = 94). In the ER group, 27 patients required backup laparoscopic surgery after an incomplete ER. The patients in the ER group had small tumor sizes and shorter procedure durations while the patient in the LR group had large tumor sizes, exophytic growth, malignancy, and tumors that were more often located in the middle or lower third of the stomach. Both groups had similar durations of hospital stays and a similar rate of major postoperative complications. For the patients in the ER group who underwent backup surgery required longer procedures (56.4 min) and prolonged stays (2 d) compared to the patients in the LR group without the increased rate of major postoperative complications. The optimal cut-off point for the tumor size for laparoscopic surgery was 2.15 cm.
ER was indicated for a smaller tumor and intraluminal growth, whereas LR was indicated for a larger tumor (optimal cut-off point: 2.15 cm), tumors located in the middle or lower third of the stomach, exophytic growth, and more aggressive malignancy behavior. Backup surgery is preserved for incomplete ER to effectively reduce associated morbidities.
Multidisciplinary teamwork adopts different strategies to yield the efficient clinical outcome according to the tumor characteristics.
| 1. | Hwang JH, Rulyak SD, Kimmey MB; American Gastroenterological Association Institute. American Gastroenterological Association Institute technical review on the management of gastric subepithelial masses. Gastroenterology. 2006;130:2217-2228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 210] [Cited by in RCA: 192] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 2. | Cho JW; Korean ESD Study Group. Current Guidelines in the Management of Upper Gastrointestinal Subepithelial Tumors. Clin Endosc. 2016;49:235-240. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 83] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 3. | Casali PG, Abecassis N, Aro HT, Bauer S, Biagini R, Bielack S, Bonvalot S, Boukovinas I, Bovee JVMG, Brodowicz T, Broto JM, Buonadonna A, De Álava E, Dei Tos AP, Del Muro XG, Dileo P, Eriksson M, Fedenko A, Ferraresi V, Ferrari A, Ferrari S, Frezza AM, Gasperoni S, Gelderblom H, Gil T, Grignani G, Gronchi A, Haas RL, Hassan B, Hohenberger P, Issels R, Joensuu H, Jones RL, Judson I, Jutte P, Kaal S, Kasper B, Kopeckova K, Krákorová DA, Le Cesne A, Lugowska I, Merimsky O, Montemurro M, Pantaleo MA, Piana R, Picci P, Piperno-Neumann S, Pousa AL, Reichardt P, Robinson MH, Rutkowski P, Safwat AA, Schöffski P, Sleijfer S, Stacchiotti S, Sundby Hall K, Unk M, Van Coevorden F, van der Graaf WTA, Whelan J, Wardelmann E, Zaikova O, Blay JY; ESMO Guidelines Committee and EURACAN. Gastrointestinal stromal tumours: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29:iv68-iv78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 318] [Cited by in RCA: 303] [Article Influence: 37.9] [Reference Citation Analysis (2)] |
| 4. | von Mehren M, Kane JM, Bui MM, Choy E, Connelly M, Dry S, Ganjoo KN, George S, Gonzalez RJ, Heslin MJ, Homsi J, Keedy V, Kelly CM, Kim E, Liebner D, McCarter M, McGarry SV, Meyer C, Pappo AS, Parkes AM, Paz IB, Petersen IA, Poppe M, Riedel RF, Rubin B, Schuetze S, Shabason J, Sicklick JK, Spraker MB, Zimel M, Bergman MA, George GV. NCCN Guidelines Insights: Soft Tissue Sarcoma, Version 1.2021. J Natl Compr Canc Netw. 2020;18:1604-1612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 211] [Article Influence: 35.2] [Reference Citation Analysis (0)] |
| 5. | Koo DH, Ryu MH, Kim KM, Yang HK, Sawaki A, Hirota S, Zheng J, Zhang B, Tzen CY, Yeh CN, Nishida T, Shen L, Chen LT, Kang YK. Asian Consensus Guidelines for the Diagnosis and Management of Gastrointestinal Stromal Tumor. Cancer Res Treat. 2016;48:1155-1166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 168] [Cited by in RCA: 160] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 6. | Deprez PH, Moons LMG, OʼToole D, Gincul R, Seicean A, Pimentel-Nunes P, Fernández-Esparrach G, Polkowski M, Vieth M, Borbath I, Moreels TG, Nieveen van Dijkum E, Blay JY, van Hooft JE. Endoscopic management of subepithelial lesions including neuroendocrine neoplasms: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy. 2022;54:412-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 325] [Cited by in RCA: 258] [Article Influence: 64.5] [Reference Citation Analysis (2)] |
| 7. | Koh YX, Chok AY, Zheng HL, Tan CS, Chow PK, Wong WK, Goh BK. A systematic review and meta-analysis comparing laparoscopic versus open gastric resections for gastrointestinal stromal tumors of the stomach. Ann Surg Oncol. 2013;20:3549-3560. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 96] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 8. | Pelletier JS, Gill RS, Gazala S, Karmali S. A Systematic Review and Meta-Analysis of Open vs. Laparoscopic Resection of Gastric Gastrointestinal Stromal Tumors. J Clin Med Res. 2015;7:289-296. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 9. | Yu M, Wang DC, Wei J, Lei YH, Fu ZJ, Yang YH. Meta-Analysis on the Efficacy and Safety of Laparoscopic Surgery for Large Gastric Gastrointestinal Stromal Tumors. Am Surg. 2021;87:450-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 10. | Liao GQ, Chen T, Qi XL, Hu YF, Liu H, Yu J, Li GX. Laparoscopic management of gastric gastrointestinal stromal tumors: A retrospective 10-year single-center experience. World J Gastroenterol. 2017;23:3522-3529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 22] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 11. | Hiki N, Yamamoto Y, Fukunaga T, Yamaguchi T, Nunobe S, Tokunaga M, Miki A, Ohyama S, Seto Y. Laparoscopic and endoscopic cooperative surgery for gastrointestinal stromal tumor dissection. Surg Endosc. 2008;22:1729-1735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 391] [Cited by in RCA: 355] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 12. | Ntourakis D, Mavrogenis G. Cooperative laparoscopic endoscopic and hybrid laparoscopic surgery for upper gastrointestinal tumors: Current status. World J Gastroenterol. 2015;21:12482-12497. [PubMed] [DOI] [Full Text] |
| 13. | Abe N, Takeuchi H, Ooki A, Nagao G, Masaki T, Mori T, Sugiyama M. Recent developments in gastric endoscopic submucosal dissection: towards the era of endoscopic resection of layers deeper than the submucosa. Dig Endosc. 2013;25 Suppl 1:64-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 51] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 14. | Zhang Q, Gao LQ, Han ZL, Li XF, Wang LH, Liu SD. Effectiveness and safety of endoscopic resection for gastric GISTs: a systematic review. Minim Invasive Ther Allied Technol. 2018;27:127-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 15. | Abe N, Takeuchi H, Ohki A, Hashimoto Y, Mori T, Sugiyama M. Comparison between endoscopic and laparoscopic removal of gastric submucosal tumor. Dig Endosc. 2018;30 Suppl 1:7-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 59] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 16. | Chang WJ, Tsao LC, Yen HH, Yang CW, Lin J, Lin KH. Endoscopic Resection for Gastric Subepithelial Tumor with Backup Laparoscopic Surgery: Description of a Single-Center Experience. J Clin Med. 2021;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (1)] |
| 17. | Cuschieri S. The STROBE guidelines. Saudi J Anaesth. 2019;13:S31-S34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3418] [Cited by in RCA: 2847] [Article Influence: 406.7] [Reference Citation Analysis (0)] |
| 18. | Hsiao SW, Chen MW, Yang CW, Lin KH, Chen YY, Kor CT, Huang SP, Yen HH. A Nomogram for Predicting Laparoscopic and Endoscopic Cooperative Surgery during the Endoscopic Resection of Subepithelial Tumors of the Upper Gastrointestinal Tract. Diagnostics (Basel). 2021;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 19. | Yin X, Yin Y, Chen H, Shen C, Tang S, Cai Z, Zhang B, Chen Z. Comparison Analysis of Three Different Types of Minimally Invasive Procedures for Gastrointestinal Stromal Tumors ≤5 cm. J Laparoendosc Adv Surg Tech A. 2018;28:58-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 20. | Zhang H, Huang X, Qu C, Bian C, Xue H. Comparison between laparoscopic and endoscopic resections for gastric submucosal tumors. Saudi J Gastroenterol. 2019;25:245-250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 21. | Zhu H, Zhao S, Jiao R, Zhou J, Zhang C, Miao L. Comparison of endoscopic versus laparoscopic resection for gastric gastrointestinal stromal tumors: A preliminary meta-analysis. J Gastroenterol Hepatol. 2020;35:1858-1868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 22. | Dong X, Chen W, Cui Z, Chen T, Liu X, Chen D, Jiang W, Li K, Dong S, Feng M, Zheng J, Li Z, Fu M, Lin Y, Liao J, Le H, Yan J. Laparoscopic resection is better than endoscopic dissection for gastric gastrointestinal stromal tumor between 2 and 5 cm in size: a case-matched study in a gastrointestinal center. Surg Endosc. 2020;34:5098-5106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (3)] |
| 23. | Wang C, Gao Z, Shen K, Cao J, Shen Z, Jiang K, Wang S, Ye Y. Safety and efficiency of endoscopic resection versus laparoscopic resection in gastric gastrointestinal stromal tumours: A systematic review and meta-analysis. Eur J Surg Oncol. 2020;46:667-674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 60] [Article Influence: 8.6] [Reference Citation Analysis (1)] |
| 24. | Pang T, Zhao Y, Fan T, Hu Q, Raymond D, Cao S, Zhang W, Wang Y, Zhang B, Lv Y, Zhang X, Ling T, Zhuge Y, Wang L, Zou X, Huang Q, Xu G. Comparison of Safety and Outcomes between Endoscopic and Surgical Resections of Small (≤ 5 cm) Primary Gastric Gastrointestinal Stromal Tumors. J Cancer. 2019;10:4132-4141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (4)] |
| 25. | Soh JS, Kim JK, Lim H, Kang HS, Park JW, Kim SE, Moon SH, Kim JH, Park CK, Cho JW, Lim MS, Kim KO. Comparison of endoscopic submucosal dissection and surgical resection for treating gastric subepithelial tumours. Scand J Gastroenterol. 2016;51:633-638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (2)] |
| 26. | Zhang Y, Meng Q, Zhou XB, Chen G, Zhu LH, Mao XL, Ye LP. Feasibility of endoscopic resection without laparoscopic assistance for giant gastric subepithelial tumors originating from the muscularis propria layer (with video). Surg Endosc. 2022;36:3619-3628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 27. | Demetri GD, von Mehren M, Antonescu CR, DeMatteo RP, Ganjoo KN, Maki RG, Pisters PW, Raut CP, Riedel RF, Schuetze S, Sundar HM, Trent JC, Wayne JD. NCCN Task Force report: update on the management of patients with gastrointestinal stromal tumors. J Natl Compr Canc Netw. 2010;8 Suppl 2:S1-41; quiz S42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 910] [Cited by in RCA: 830] [Article Influence: 51.9] [Reference Citation Analysis (0)] |
| 28. | Dávila JS, Momblán D, Ginès À, Sánchez-Montes C, Araujo I, Saavedra-Pérez D, Lacy AM, Fernández-Esparrach G. Endoscopic-assisted laparoscopic resection for gastric subepithelial tumors. Surg Endosc. 2016;30:199-203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 29. | Pantuso G, Macaione I, Taverna A, Guercio G, Incorvaia L, Di Piazza M, Di Grado F, Cilluffo G, Badalamenti G, Cipolla C. Surgical treatment of primary gastrointestinal stromal tumors (GISTs): Management and prognostic role of R1 resections. Am J Surg. 2020;220:359-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 30. | Cao B, Lu J, Tan Y, Liu D. Efficacy and safety of submucosal tunneling endoscopic resection for gastric submucosal tumors: a systematic review and meta-analysis. Rev Esp Enferm Dig. 2021;113:52-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 31. | Antonino G, Alberto M, Michele A, Dario L, Fabio T, Mario T. Efficacy and safety of gastric exposed endoscopic full-thickness resection without laparoscopic assistance: a systematic review. Endosc Int Open. 2020;8:E1173-E1182. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 32. | Zhu Y, Xu MD, Xu C, Zhang XC, Chen SY, Zhong YS, Zhang YQ, Chen WF, Chen TY, Xu JX, Yao LQ, Li QL, Zhou PH. Microscopic positive tumor margin does not increase the rate of recurrence in endoscopic resected gastric mesenchymal tumors compared to negative tumor margin. Surg Endosc. 2020;34:159-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 33. | McCarter MD, Antonescu CR, Ballman KV, Maki RG, Pisters PW, Demetri GD, Blanke CD, von Mehren M, Brennan MF, McCall L, Ota DM, DeMatteo RP; American College of Surgeons Oncology Group (ACOSOG) Intergroup Adjuvant Gist Study Team. Microscopically positive margins for primary gastrointestinal stromal tumors: analysis of risk factors and tumor recurrence. J Am Coll Surg. 2012;215:53-9; discussion 59. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 136] [Cited by in RCA: 137] [Article Influence: 9.8] [Reference Citation Analysis (2)] |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Surgery
Country/Territory of origin: Taiwan
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Arigami T, Japan; Osera S, Japan S-Editor: Yan JP L-Editor: A P-Editor: Wu RR