Copyright: ©Author(s) 2026.
World J Gastrointest Surg. Mar 27, 2026; 18(3): 116913
Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.116913
Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.116913
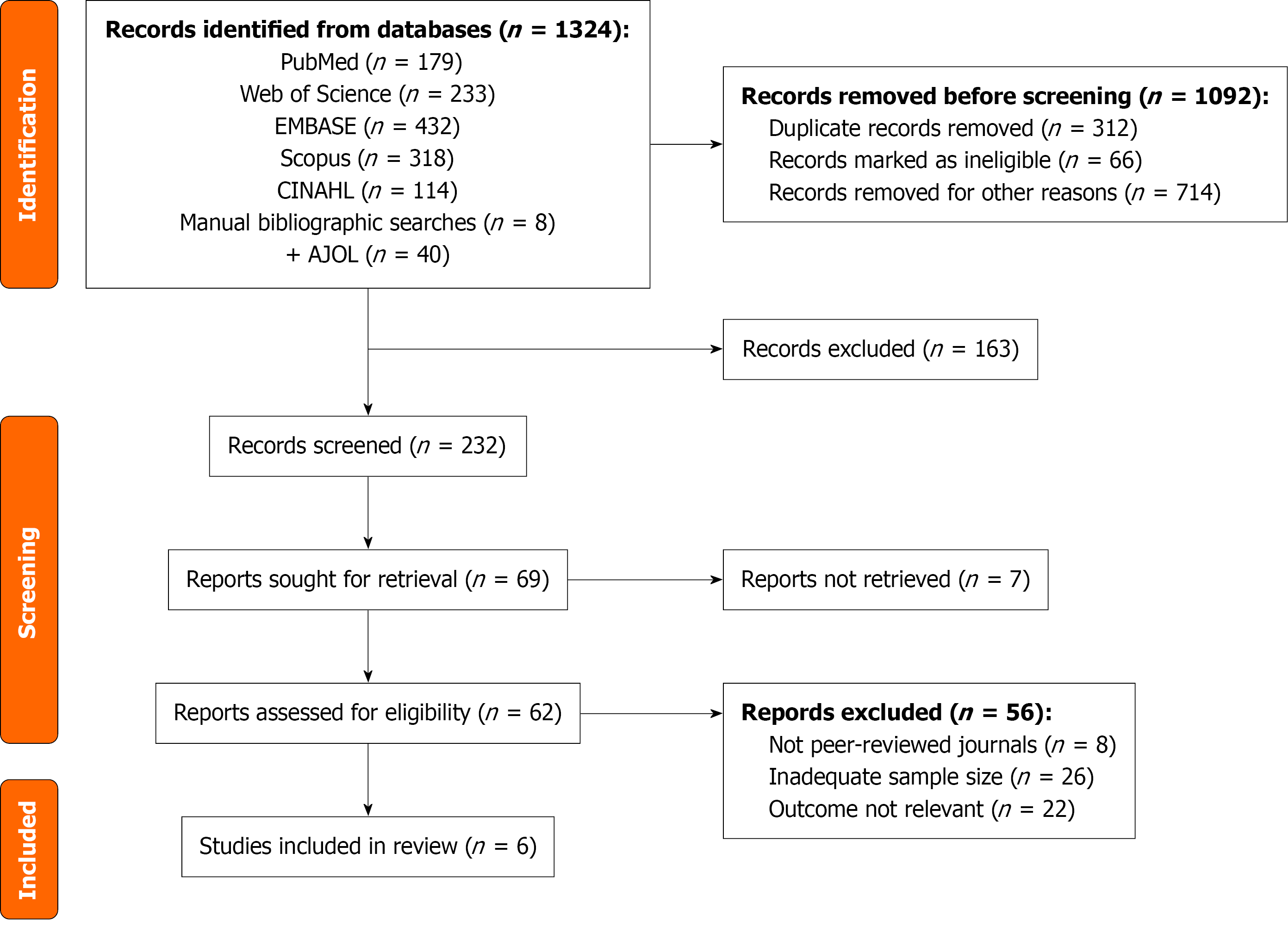
Figure 1 Preferred Reporting Items for Systematic reviews and Meta-Analyses flow diagram of study selection.
Flowchart showing the identification, screening, eligibility assessment, and final inclusion of studies in the systematic review and meta-analysis according to Preferred Reporting Items for Systematic reviews and Meta-Analyses guidelines. AJOL: African Journals Online.
Figure 2 Availability map of chronic proliferative cholangitis reporting and related outcomes across included studies.
CPC: Chronic proliferative cholangitis.
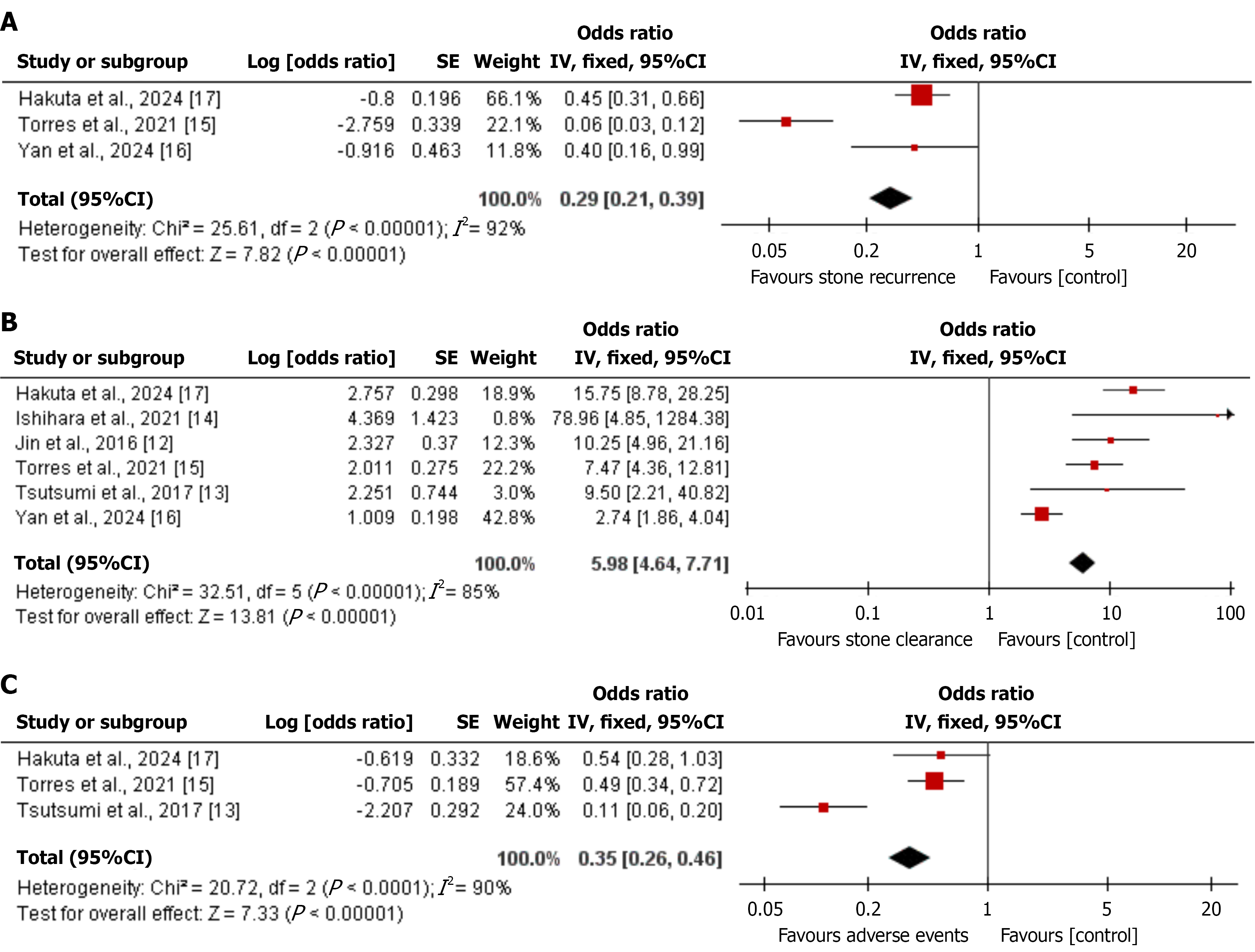
Figure 3 Forest plots of pooled single-arm outcomes after treatment of intrahepatic bile duct stones.
Each forest plot shows the study-specific event rate and the pooled estimate using a random-effects model. Study weights are indicated by square size, and horizontal lines show 95% confidence intervals; the diamond shows the pooled estimate and its 95% confidence interval. The X-axis represents the event rate (proportion). Because these are single-arm meta-analyses, there is no true control group. In these plots, the “control” column shown by Review Manager is not used and should be interpreted as a placeholder required by the software; only the treated study cohort contributes data. A: Stone recurrence: Events are patients with imaging-confirmed recurrent stones during follow-up; B: Stone clearance: Events are patients with complete stone clearance (stone-free status) after the index procedure; C: Adverse events: Events are patients with procedure-related complications as reported in each study. CI: Confidence interval.
- Citation: Muhammad T, He TT, Lin ZY, Shi JL, Zhang TG, Yang Y, Yang XL, Chattha HN, Li J. Impact of chronic proliferative cholangitis on stone recurrence and biliary strictures after intrahepatic stone treatment: A meta-analysis. World J Gastrointest Surg 2026; 18(3): 116913
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/116913.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.116913