Published online May 15, 2026. doi: 10.4239/wjd.v17.i5.119756
Revised: February 28, 2026
Accepted: March 30, 2026
Published online: May 15, 2026
Processing time: 96 Days and 0.1 Hours
This review examines the role of sex in modulating the relationship between metabolic dysfunction-associated steatotic liver disease (MASLD) and various components of type 2 diabetes (T2D) pathobiology. Data suggest that women with prediabetes and diabetes have a greater risk of MASLD compared to men. Furthermore, postmenopausal women with T2D demonstrate a high prevalence of advanced MASLD, and the female sex appears to interact with T2D to enhance the detrimental effects of the PNPLA3 rs738409 variant on adverse liver outcomes. MASLD serves as a risk factor for incident T2D, and the predictive biomarkers for diabetes onset may vary by sex. Additionally, sex may influence the likelihood of diabetic complications, while MASLD appears to more significantly impact the development of coronary artery disease in women. Finally, sex differences in the management of T2D among individuals with MASLD are analyzed.
Core Tip: Animal models indicate that males are more susceptible to obesity and metabolic disorders. Men, especially those in middle age, exhibit higher rates of type 2 diabetes. Sex also influences risk factors for atherosclerotic cardiovascular disease (ASCVD), chronic kidney disease, and metabolic dysfunction-associated steatotic liver disease (MASLD). Among individuals with diabetes, women’s risk for ASCVD and MASLD increases over time as the protective effects of sex hormones diminish. While estradiol has demonstrated protective properties, further investigation into the roles of sex chromosomes and epigenetic factors is warranted. Recognizing these distinctions is essential for advancing precision medicine and optimizing diabetes management.
- Citation: Lonardo A, Jamalinia M, Weiskirchen R. Type 2 diabetes, sex and metabolic dysfunction-associated steatotic liver disease. World J Diabetes 2026; 17(5): 119756
- URL: https://www.wjgnet.com/1948-9358/full/v17/i5/119756.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i5.119756
Type 2 diabetes (T2D), characterized by chronic hyperglycaemia resulting from insulin resistance, reduced insulin secretion, or both, is associated with multiple metabolic dysfunctions that can eventually result in target organ damage, including the cardiovasculature, kidneys, liver, and eyes[1]. In combination with other noncommunicable diseases, such as obesity, arterial hypertension, dyslipidemia, and metabolic dysfunction-associated steatotic liver disease (MASLD), T2D represents a major global health challenge with high morbidity and mortality[2]. These findings underscore the necessity for a coordinated global health initiative aimed at addressing these debilitating diseases and enhancing population health worldwide[3].
Sex, the biological attribute, and gender, its socio-cultural counterpart, are increasingly identified as major modifiers of disease risk, presentation, and outcomes[4]. In animal models, males are generally more susceptible to obesity and obesity-related metabolic dysfunction. In humans, sex and gender influence the development of T2D, which is more prevalent in men, particularly in middle-aged individuals[5]. Target organ damage, including atherosclerotic cardiovascular disease (ASCVD), chronic kidney disease (CKD), and MASLD, exhibits substantial mutual overlaps and intertwined sex- and gender-specific pathomechanisms[4]. Although younger women benefit from certain ASCVD protection mechanisms, ASCVD remains a leading cause of death among women. Notably, T2D and MASLD seemingly abrogate women’s cardiovascular and hepatic advantages, as diabetic women display risks of ASCVD and MASLD comparable to those of men, owing to incompletely defined mechanisms[6,7]. These likely include sex hormone-related differences in energy storage and glucose metabolism between sexes, with women having certain metabolic advantages during reproductive years, which, however, decline over time. While estradiol is protective, additional factors like sex chromosomes and epigenetics need further investigation. Recognizing these differences is vital for precision medicine and optimal diabetes care[5].
In hepatology, the interaction between T2D and sex differences is reflected in marked disparities across several forms of chronic liver disease (CLD), including alcohol-related liver disease, viral hepatitis, MASLD, cirrhosis, and hepatocellular carcinoma (HCC)[8]. MASLD is the prototypical CLD associated with obesity and T2D[9,10]. It is increasingly recognized as a systemic disorder featuring both liver-related and extra-hepatic consequences. Liver-related outcomes comprise steatohepatitis, fibrosis, cirrhosis, end-stage liver failure, and HCC[9], whereas extra-hepatic outcomes include incident T2D, metabolic syndrome (MetS), accelerated ASCVD and non-hepatic cancers[11-15].
Sex disparities in the MASLD arena, which are now increasingly appreciated, are deemed to result from combined chromosomal and hormonal influences[16]. Fibrosis-driven organ failure affecting the heart, kidneys, liver, and lungs often promoted by diabetes, obesity, and MASLD is an emerging clinical challenge, particularly as severity increases with age[17,18]. Within this intriguing context, this scoping review focuses on published animal and human studies that examine the interconnected pathobiological, epidemiological, and clinical relationships linking T2D, sex, and MASLD.
On January 10, 2026, a bibliographic search was performed in PubMed, EMBASE, Cochrane Library, and Web of Science using the following strategy: [Sex (Title)] AND [diabetes (title/abstract)] AND [liver (title/abstract)]. This search yielded 214 results. Studies were included if they met the following criteria: (1) Original research articles (human or animal studies); (2) Investigation of T2D in relation to liver disease or hepatic outcomes; (3) Explicit reporting or the possibility to assess sex-based differences; and (4) Inclusion of adult human populations or experimental animal models limited to mice or rats. Studies were excluded if they were: (1) Review articles, editorials, commentaries, or letters to the editor; (2) Exclusive focus on paediatric populations; (3) Investigation of toxic- or drug-induced liver disease rather than metabolic liver disease; (4) Use of animal models other than mice or rats; (5) Primary focus on hepatitis C virus infection; (6) Focus on gallbladder disease; or (7) Lack of relevant data or discussion on sex differences. After application of these criteria, 133 publications were excluded, and 81 studies were retained for qualitative review. In addition, relevant articles identified through cross-referencing and other pertinent publications by the authors of the included studies were also considered. Human studies underwent critical appraisal using the Joanna Briggs Institute (JBI) Critical Appraisal Tools, applying the appropriate checklist for each study design (e.g., cohort, case-control, cross-sectional, etc.)[19]. Overall risk of bias was categorized as low, moderate, or high according to the domain-based structure of the JBI tools. Studies were classified as low risk when all domains showed minimal concern, moderate risk when one or more domains showed some concern without serious flaws in critical domains, and high risk when at least one critical domain demonstrated serious methodological limitations likely to bias results.
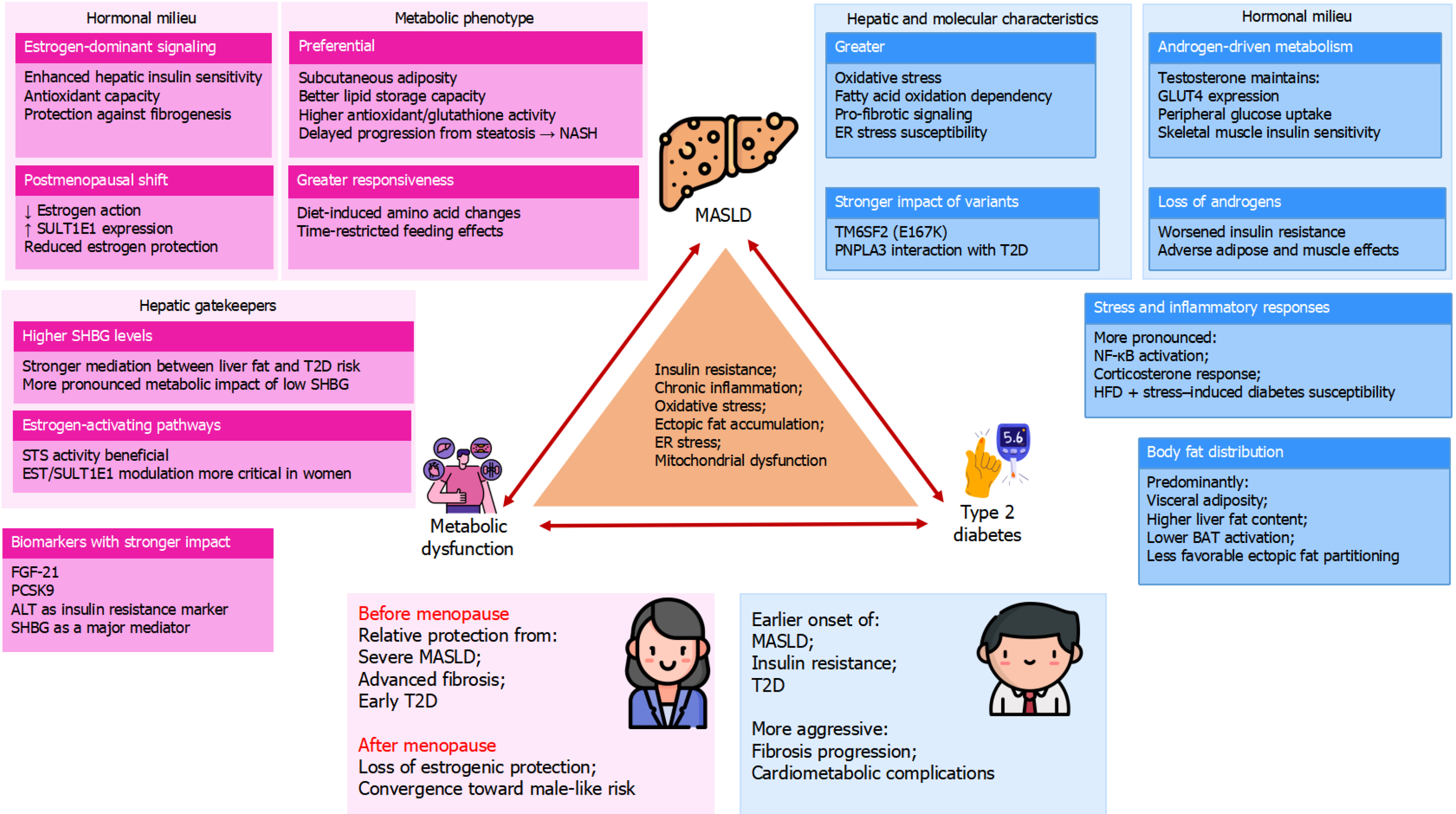
The triangular relationship between sex, MASLD and T2D illustrated in Figure 1 is rooted in fundamental differences in hormone signaling, hepatic metabolism, adipose biology, and stress-response pathways between females and males[20,21]. These biological differences modulate how excess nutrients, obesity, and insulin resistance translate into steatotic liver disease (SLD), systemic metabolic dysfunction, and ultimately, T2D and its complications[22,23].
Sex steroids orchestrate glucose and lipid metabolism in the liver, skeletal muscle, and adipose tissue in a highly tissue- and sex-specific fashion[24,25]. In male rats, orchiectomy leads to hyperglycemia, reduced circulating insulin, impaired protein kinase B (Akt/PKB) phosphorylation, downregulated glucose transporter type 4 (GLUT4) expression, diminished glucose uptake in skeletal muscle and adipose tissue, and reduced hepatic glycogen synthase activity with reciprocal increases in glycogen phosphorylase[26]. Testosterone replacement restores these abnormalities, while estradiol alone increases Akt phosphorylation but fails to normalize GLUT4 expression and glucose disposal, underscoring the dominant role of androgens in maintaining male peripheral insulin sensitivity[26].
At the hepatic level, sex hormones directly regulate hepatokines that link liver fat to systemic insulin resistance[27,28]. Fetuin-A inhibits insulin receptor signaling and promotes low-grade inflammation, showing sexually dimorphic expression in rat liver and are modulated via estrogen and androgen receptors, as confirmed in complementary in vitro experiments, while the role of fetuin-B is more complex[27]. These findings provide a mechanistic underpinning for clinical observations that fetuins associate with MASLD, insulin resistance and cardiometabolic risk in a sex-dependent manner[29,30].
Hepatic enzymes that control estrogen activation and inactivation add a further sex-specific layer[31]. In mouse models of obesity and T2D, estrogen sulfotransferase (EST encoded by SULT1E1) expression is increased in the liver[32]. Genetic ablation of EST improves insulin sensitivity, reduces hepatic steatosis, and lowers inflammation in females, but these benefits disappear after ovariectomy, demonstrating that preserved estrogen action is required for protection[32]. In contrast, EST deletion in males aggravates diabetes due to reduced β-cell mass, impaired insulin secretion, and enhanced adipose inflammation[32], suggesting that the same estrogen-inactivating pathway exerts opposite net metabolic effects according to sex.
Conversely, hepatic overexpression of steroid sulfatase (STS), which regenerates active estrogens from their sulfated forms, improves insulin sensitivity and reduces hepatic fat and inflammation in both sexes, but via partly distinct mechanisms[33]. In females, the benefit is largely estradiol-dependent and abolished by ovariectomy, whereas in males the improvement is mediated by reduced inflammation and higher energy expenditure even after castration[33]. Collectively, these basic studies indicate that modest shifts in sex-steroid activation or inactivation within the liver can profoundly modulate MASLD severity and its diabetogenic potential in a sex-dependent way[32,33].
Human data support these experimental insights[34]. In diabetic women, but not in men, hepatic expression of the estrogen-inactivating enzyme SULT1E1 and the estrogen sulfate exporter ABCG2 is selectively upregulated[34]. This induction is driven by phosphorylated estrogen receptor-α (ERα) and retinoid acid receptor-related orphan receptor-α (RORα) binding to the SULT1E1 promoter and likely represents a hepatic defense mechanism against estrogen toxicity in postmenopausal women with T2D[34]. However, the same process may erode the systemic metabolic advantages of estrogens and contribute to the convergence of MASLD and cardiovascular risk between the sexes after menopause[22,35].
Sex hormone-binding globulin (SHBG) is produced by the liver and regulates the bioavailability of circulating androgens and estrogens, while also reflecting and modulating hepatic metabolic status[25]. Large cohort data have established sex- and age-specific thresholds defining low SHBG and estimated that approximately 3% of adults meet these criteria, with obesity, diabetes, smoking, chronic lung and heart disease as key correlates in both sexes[25]. Epidemiological and mechanistic studies indicate that low SHBG is a sensitive biomarker of insulin resistance and the MetS and predicts incident hyperglycemia and T2D, particularly in women[25,36].
In the Maastricht Study, intrahepatic lipid content was independently linked to T2D in both women and men, but mediation analysis showed that serum SHBG accounted for roughly half of this association in women vs less than one-fifth in men[37]. This suggests that SLD lowers SHBG and thereby increases free sex steroid levels, amplifying sex specific metabolic and vascular effects more strongly in women[37]. Experimental work in hepatocytes shows that exogenous SHBG attenuates palmitate-induced ER stress by reducing inositol-requiring enzyme 1α (IRE1α), activating transcription factor 6, C/EBP homologous protein, and binding immunoglobulin protein expression and modulates lipolytic gene expression, indicating that SHBG is not merely a passive marker but actively shapes hepatocyte stress responses and lipid handling[25].
Consistent with these observations, low SHBG is associated with MASLD and impaired renal function, and the combination of low SHBG with low vitamin D identifies men and postmenopausal women at particularly high risk for MASLD[24]. Meta-analytic and cohort data further indicate that higher SHBG is linked to lower odds of MASLD in both sexes, whereas total testosterone (TT) shows opposite associations in men and women, emphasizing distinct hormonal milieus in male and female MASLD[38].
Animal models of obesity and diabetes consistently show that hepatic lipid metabolism and oxidative stress pathways are sexually dimorphic[31,39]. In Zucker diabetic fatty (ZDF) rats, male animals develop overt diabetes with low insulin and severe insulin resistance, while female ZDF rats remain in a prediabetic state despite comparable obesity[31]. Hepatic transcriptomics reveal that females maintain higher rates of lipogenesis and lipid export coupled with enhanced glutathione turnover and antioxidant capacity, whereas males rely more on fatty acid oxidation and experience higher oxidative stress[31]. This anabolic vs catabolic bias may delay the transition from simple steatosis to steatohepatitis and fibrosis in females, particularly before menopause[22].
In a study investigating spontaneously hypertensive obese mice [Tsumura Suzuki obese diabetes (TSOD)] and db/db mice, both sexes develop hepatic steatosis driven by upregulation of sterol regulatory element-binding protein (SREBP)-1 and SREBP-2 and lipogenic enzymes such as acetyl-CoA carboxylase 1 and fatty acid synthase, but male livers show greater induction of peroxisome proliferator-activated receptor α/γ, PNPLA3 and transport genes like Slc2a4, whereas females display higher activation of c-Jun N-terminal kinase and adenosine 5’-monophosphate-activated protein kinase and increased fatty acid translocase cluster of differentiation 36 expression[40]. These sex-specific patterns in lipid transport, oxidation, and stress signaling align with human data showing that female sex interacts with T2D to enhance the detrimental effect of PNPLA3 variants on adverse liver outcomes[41,42].
Untargeted metabolomics in mice fed different dietary patterns demonstrates that the liver is the most diet-responsive organ and that the metabolic imprint of diet is strongly modified by sex in hepatocytes[43]. Time-restricted feeding induces pronounced alterations in hepatic amino acid metabolism, more marked in females, whereas adipose tissue metabolite profiles remain relatively stable[44]. These findings highlight the liver as a central hub where sex interacts with diet to shape systemic metabolic phenotypes, including susceptibility to MASLD and T2D.
Sex differences also affect early fibrogenesis[22]. In TSOD and db/db mouse models of MASLD, young males exhibit higher aminotransferases and larger steatotic and fibrotic areas than females, but these differences diminish with age, mirroring the human pattern in which premenopausal women are relatively protected from advanced MASLD and fibrosis, with risk converging or surpassing that of men after menopause[22,40].
Multiple genetic and molecular pathways modulate the MASLD-T2D axis in a sex-specific manner[22,42]. The TM6SF2 rs58542926 (E167K) variant, which impairs hepatic triglyceride export, is associated with an increased risk of MASLD and T2D, with stronger effect sizes in men[45]. In E167K knock-in mice, male but not female carriers exhibit impaired glucose tolerance and defective IRE1α-mediated ER-stress signaling in the liver, implicating sex-biased disruption of ER homeostasis as a mechanism linking this variant to glucose dysregulation[45].
Gene expression studies in obese and lean rats identify more than 100 hepatic genes with sex-differential expression, with male-biased genes enriched for xenobiotic metabolism and secretion and female-biased genes enriched for lipid metabolism and glycolysis[31,39]. Such baseline differences likely influence how the liver handles lipids, drugs, and inflammatory stimuli in obesity and T2D. Knockout models further emphasize sex-biased metabolic roles: Id2-deficient male mice show improved glucose tolerance, enhanced skeletal muscle and brown fat glucose uptake, and reduced intramuscular lipid content, whereas female Id2-deficient mice mainly display smaller adipocytes without the same degree of systemic metabolic improvement[46].
Other genetic manipulations illustrate how stress-response pathways intersect with sex to shape metabolic outcomes[45]. Deletion of cytochrome P450 family 1 subfamily A enzymes CYP1A1/CYP1A2 partially protects female mice from high-fat diet (HFD)-induced glucose intolerance and lowers insulin levels on chow, while in males it delays hyperinsulinemia and attenuates islet stress, with limited effects on hepatic steatosis[47]. Similarly, DNAJB3 deficiency precipitates greater weight gain, hepatosteatosis, inflammation, and impaired glucose clearance in high-fat-fed females than in males, pointing to chaperone-mediated stress responses as a key determinant of female metabolic resilience[48].
Sex differences in body composition and fat distribution critically modulate liver fat accumulation and diabetes risk[20]. Women generally have higher total body fat and more subcutaneous adipose tissue, whereas men accumulate more visceral adipose tissue and liver fat, differences that are accentuated in T2D[22]. In women, visceral adipose tissue and liver fat content are most strongly associated with oral glucose tolerance test (OGTT)-derived glucose parameters, while in men, the waist-to-height ratio and overall adiposity show the strongest associations and the relative fat-free mass is inversely related to glycemic measures in both sexes[49], suggesting that the same anthropometric markers do not carry identical pathophysiological meaning in women and men.
Brown adipose tissue (BAT) and thermogenic capacity also differ by sex and early life history[50]. Prepubertal appropriate-for-gestational-age girls exhibit a higher BAT activation index than boys, and higher BAT activity is associated with lower visceral fat and a more favorable metabolic profile[51]. Fluorodeoxyglucose positron emission tomography studies in adult mice confirm sex-dependent partitioning of glucose uptake, with females showing higher BAT uptake and males showing higher skeletal muscle uptake[52]. In line with these findings, high-fat feeding reduces glucose uptake in BAT, muscle, and heart but increases brain uptake in both sexes[52]. These data indicate that sex-specific patterns of substrate utilization and thermogenesis influence the propensity to store excess energy as ectopic liver fat[50].
Maternal nutrition further programs sex-dependent metabolic trajectories[22]. Exposure to a maternal HFD induces glucose intolerance, hepatic steatosis, and adipose inflammation in both male and female offspring, but male offspring develop reduced insulin content, decreased pancreatic and duodenal homeobox 1 expression and heightened islet oxidative stress, whereas female offspring increase β-cell mass and insulin content and maintain higher estradiol levels[22]. These early-life differences may contribute to the greater vulnerability of males to T2D and MASLD across the life course[22].
Low-grade inflammation and organ-specific stress responses are central to both MASLD and T2D and are strongly shaped by sex[22]. In animal models, male rats exposed to a HFD and chronic stress are more prone to develop T2D, with higher fasting blood glucose, homeostasis model assessment of insulin resistance, corticosterone and pancreatic nuclear factor kappa-light-chain-enhancer of activated B cells activation than females; interventions such as linalyl acetate can mitigate these changes, particularly in males, by reducing corticosterone and improving hepatic mitochondrial membrane potential[53]. Time-restricted feeding of a Western diet confers metabolic benefits independent of age but dependent on sex, improving fatty liver and glucose intolerance in both sexes while reducing body weight mainly in males, illustrating that the timing of food intake interacts with biological sex to shape metabolic outcomes[44].
In humans, circulating biomarkers reflect similar sex biases[20]. Higher serum fibroblast growth factor-21 (FGF-21) concentrations predict incident diabetes in women but not in men in prospective data, and stronger associations for incident T2D have also been reported in general cohorts with effect modification by glycemia[35,54]. Elevated proprotein convertase subtilisin/kexin type 9 (PCSK9) is more strongly associated with insulin resistance, MetS, MASLD, and all-cause and cancer mortality in women, consistent with higher circulating PCSK9 levels in females across cohorts[55,56]. Alanine aminotransferase (ALT) and the γ-glutamyl transferase (GGT)/high-density lipoprotein (HDL)-cholesterol ratio show sex-specific relationships with insulin resistance and cardiovascular disease, with ALT serving as a particularly useful marker of insulin resistance in women[22]. These sex-dependent biomarker profiles mirror the differential hepatic and systemic stress responses observed in experimental models and can be leveraged for sex-tailored risk stratification[20].
Taken together, basic and translational science support a model in which sex chromosomes, sex steroids, hepatically derived proteins (SHBG, fetuins, STS/SULT1E1), genetic variants (e.g., in TM6SF2, PNPLA3), adipose distribution, and stress-response pathways interact to shape the trajectory from steatosis to MASLD, metabolic dysfunction and T2D[20,21]. In premenopausal women, estrogens, higher SHBG, more favorable fat distribution and stronger antioxidant capacity confer relative protection from severe MASLD and T2D[22,37]. However, this advantage is attenuated or reversed by menopause, diabetes-induced changes in hepatic estrogen handling, and additive genetic and environmental insults[34,35]. In men, higher visceral and hepatic fat accumulation, different patterns of sex- steroid signaling, greater oxidative stress and distinct genetic susceptibilities promote earlier and often more aggressive MASLD and T2D, with a substantial impact on cardiovascular and liver-related outcomes[42,57]. Understanding these layered mechanisms is crucial for designing sex-specific preventive and therapeutic strategies for MASLD and T2D[20,58].
Large epidemiological studies, summarized in Table 1[37,59-62], support the notion that sex modulates the link associating SLD and altered glucose regulation. Large epidemiological studies have examined how sex modifies the association between SLD and the risk of dysglycemia and T2D. Overall, these studies show clear sex-specific differences in risk patterns, biomarkers, and metabolic features, which help explain differences in T2D pathogenesis between men and women. Sex influences the relationship between hepatic biomarkers and T2D risk, with SLD conferring a higher risk in non-obese Asian men but not in women[59].
| Method | Findings | Conclusion | Risk of bias | Ref. |
| This retrospective cohort study included 13596 men and 6037 women to examine the association between SLD, sex, and T2D in non-obese Japanese adults undergoing health check-ups. Baseline BMI was grouped into six categories, and SLD was diagnosed by abdominal ultrasonography | During the follow-up, 738 men and 138 women developed T2D. Men with a BMI of 20-22.9 kg/m2 and SLD had a higher risk, while women in this group did not. Among men, higher BMI without SLD did not raise the risk of diabetes, but women with a BMI of 23-27.4 kg/m2 had an increased risk regardless of SLD status | Sex influences the relationship between liver biomarkers and the development of T2D. SLD is associated with an increased risk of T2D in non-obese Asian males, but not in females | Low | Narisada et al[59] |
| This analysis used cross-sectional data from 1554 participants in the Maastricht study. T2D was determined by OGTT, and IHL content was measured via 3T Dixon MRI. Mediation analysis evaluated whether serum SHBG mediates the link between IHL content and T2D | IHL content was linked to T2D in both women (OR = 1.08, 95%CI: 1.04-1.14) and men (OR = 1.12, 95%CI: 1.08-1.17), and serum SHBG significantly mediated this association. Serum SHBG contributed more in women (OR = 1.04, 95%CI: 1.02-1.07; 50.9% mediated, 95%CI: 26.7-81.3) than men (OR = 1.02, 95%CI: 1.01-1.03; 17.2% mediated, 95%CI: 9.6-27.6). Repeating analyses with T2D proxies and covariate adjustment yielded similar results | Serum SHBG mediated the association between IHL content and T2D in both sexes, but its contribution was stronger in women than in men | Low | Simons et al[37] |
| Of 1309 screened individuals, 748 without diabetes were enrolled. Over 15 years, correlations between metabolic markers (including APN) and T2D onset were assessed in this group | In multivariate LRA, the independent predictors of incident T2D differed by sex: In men, only eGFR and SLD were significantly related to the onset of T2D. In women, APN was the only significant risk factor | Intervention strategies to prevent T2D vary between men and women | Low | Yoshimoto et al[60] |
| PPG was determined as the AUC for glucose during OGTT using the trapezoidal rule among 794 middle-aged Soweto cohort participants. PC analysis clustered sex hormones, liver enzymes, and cardiometabolic factors by sex. Multivariable linear regression assessed how much variance in PPG was explained by PCs and T2D PRSs, adjusting for relevant covariates in men and women | In men, the PCs’ cluster of sex hormones, liver enzymes, and cardiometabolic factors explained 10.6% of the variance in PPG, with PC1 (peripheral fat), PC2 (liver enzymes and steroid hormones), and PC3 (lipids and peripheral fat) contributing significantly to PPG. In women, PC factors of sex hormones, cardiometabolic factors, and liver enzymes explained a similar amount of the variance in PPG (10.8%), with PC1 (central fat) and PC2 (lipids and liver enzymes) contributing significantly to PPG | Variations in PPG responses to an OGTT among individuals may be influenced by factors such as body fat distribution, serum lipid profiles, liver enzyme levels, and steroid hormone concentrations, with differing effects observed between men and women | Low | Masango et al[61] |
| The PREVADIAB2 study assessed metabolic changes over 5 years in people initially without T2D. Researchers used hierarchical clustering on 953 participants based on IGI, fasting insulin secretion rate, HOMA-IR, and insulin clearance; 488 had their lipid profiles analyzed by LC/MS-QTOF | Four clusters (LS, PGS, ID, IR) with distinct dysglycemia risk were found. Although metabolic features were similar for both sexes, threshold differences led to sex-specific lipid profiles. Women had higher dihydroceramide and SM levels, while men showed a greater ceramide-to-SM ratio. All clusters except LS showed unique lipid signatures linked to metabolic dysfunction, with clear sex-based differences | Distinct insulin-associated metabolic characteristics and sex delineate phenotypes with unique lipidomic profiles, highlighting the importance of considering prediabetes within a comprehensive metabolic framework that extends beyond glycaemic measures | Low | Pina et al[62] |
In addition, serum SHBG appears to mediate the link between hepatic fat and T2D more strongly in women than in men[37]. Sex-specific predictors of incident T2D have also been reported: Only estimated glomerular filtration rate (eGFR) and SLD were significant predictors in men, whereas adiponectin was the only significant predictor in women. These observations suggest that strategies for T2D prevention should be differentiated by sex[60].
Further evidence from Masango et al[61] indicates that body fat distribution, serum lipid profiles, hepatic enzyme concentrations, and steroid hormone levels differentially affect postprandial glucose (PPG) responses during OGTT in a sex-dependent manner. More recently, Pina et al[62] emphasized the necessity of incorporating both sex and insulin-related metabolic traits into prediabetes risk assessment, extending beyond conventional glycaemic parameters.
In summary, epidemiological data consistently demonstrate that the relationship between SLD and the risk of dysglycemia and T2D is substantially influenced by sex. SLD, related metabolic markers, and lipidomic and hormonal mediators exert different effects in men and women. These observations support the need for sex-specific approaches to the assessment, prevention, and management of T2D risk in individuals with SLD.
Declining estrogen levels during menopause may raise T2D risk in women. The liver responds by increasing enzymes and transporters tied to estrogen inactivation and excretion, which may connect estrogen metabolism with liver disease and glucose issues. Research shows diabetic women have higher hepatic expression of SULT1E1 and ABCG2, regulated by phosphorylated ERα and RORα[34]. These findings indicate a sex-specific liver process that helps protect women from estrogen toxicity through increased inactivation and elimination.
A detailed overview of experimental studies investigating sex differences in the pathogenesis, physiology and metabolic response to T2D and related condition in animal models using mice and rats is provided in Table 2[27,31,33,40,47,48,52,53,63-82]. Studies in rats and mice show that protein malnutrition and HFDs have different metabolic effects in males and females. For example, protein malnutrition leads to a greater deterioration of glucose tolerance in male rats compared with females[63,64]. In contrast, obesity reduces hepatic insulin clearance and insulin receptor binding in both sexes, with additive effects that appear largely independent of sex[31,65].
| Method | Findings | Comment | Ref. |
| This study examined protein malnutrition in rats fed a LP or NP diet from weeks 3 to 6, after which all rats received the NP diet for the remainder of the experiment | Fasting glucose levels and GTT results significantly deteriorated from 12 weeks to 48 weeks in all rats, with the most pronounced changes observed in LP males | Male rats are particularly susceptible to impaired glucose tolerance due to protein malnutrition | Crace et al[63] |
| The effects of obesity and sex on hepatic insulin metabolism were evaluated in the SHR/Mcc-cp rat model | CLR decreased by 58% in obese females and 68% in obese males vs lean controls. In lean animals, males had 46% lower CLR; in obese animals, 59% lower than in females. Obesity led to a 50% drop in insulin-receptor binding in hepatocytes. Male sex reduced insulin binding by about 34% in both groups, mainly due to fewer cell surface receptors. Receptor-mediated insulin degradation was 40% less in obese animals compared to lean animals, and 27% lower in males than in females | Male sex and obesity are independently and additively linked to reduced hepatic insulin clearance and fewer cell surface insulin receptors, resulting in proportionally lower insulin compartmentalization and degradation | Hennes et al[64] |
| Adult male Wistar albino rats were orchidectomized and given daily doses of testosterone, estradiol, or both for 15 days starting 11 days after surgery. Plasma levels of glucose, insulin, testosterone, and estradiol were measured after treatment. The gastrocnemius muscle, adipose tissue, and liver were isolated to assess Akt phosphorylation, GLUT2/4 expression, glucose uptake, and the activities of glycogenic and glycogenolytic enzymes | Castration raised blood glucose and inhibited serum insulin, Akt phosphorylation, GLUT4 expression, glucose uptake, glycogen content and synthase activity, while increasing GLUT2 expression and glycogen phosphorylase activity. Testosterone alone or plus estradiol restored normal levels for all parameters. Estradiol alone increased Akt phosphorylation but did not affect other measures | Deficiency of sex steroids results in impaired glucose uptake in skeletal muscle and adipose tissue, associated with reduced Akt phosphorylation and decreased expression of GLUT4 on the plasma membrane | Muthusamy et al[65] |
| To identify protective factors in females, hepatic molecular profiles of non-diabetic obese fZDF rats were compared to mZDF or HF-fZDF rats | HFDs in fZDF rats caused greater weight gain, elevated glucose and insulin, lower insulin sensitivity, and prediabetes. mZDF rats were diabetic, with low insulin, high glucose, poor insulin sensitivity, and impaired glucose tolerance. Diet changes shifted female transcript and metabolite profiles toward a male pattern with reduced lipogenesis, increased FA oxidation, and higher oxidative stress. Males showed reduced GSH production capacity, while females had increased GSH turnover | Overall, the data support the hypothesis that anabolic pathways such as lipogenesis and lipid export may restrict FA oxidation and mitigate oxidative stress in female rats. Along with an enhanced capacity for GSH production, these hepatic sex differences could play a role in the sex-specific progression of T2D in ZDF rats | Gustavsson et al[31] |
| Obesity was induced, and food intake and weights were monitored weekly in age-matched mice | Hepatic EST induction is common in T2D. In female mice, loss of EST improved metabolic function in various T2D models. However, this benefit disappeared in ovariectomized mice. In males, EST ablation worsened diabetes due to reduced β-cell mass and impaired insulin secretion, with increased inflammation in WAT | The findings indicate that EST plays a critical, sex-specific role in energy metabolism and T2D pathogenesis | Gao et al[66] |
| A replacement vector was generated to delete two exons covering the entire Id2 coding region, with successful deletion confirmed by qRT-PCR. The resulting 2.2 kb mutant protein from neo-exon3 lacks functional domains, as exon 3 consists only of 3’ UTR sequence. Id2 WT and Id2-/- mice were bred on mixed backgrounds and received standard chow and antibiotic-treated water ad libitum. Wild-type controls were always Id2+/+ littermates | Male Id2-/- mice ate more food per body mass but gained less weight, while female Id2-/- mice had smaller adipocytes, indicating sex-specific effects on adipogenesis. Male Id2-/- mice also showed improved glucose tolerance and insulin sensitivity, especially with age. FDG-PET scans showed higher glucose uptake in their skeletal muscle and brown fat, suggesting increased metabolism and thermogenesis. These mice also had reduced intramuscular triacylglycerol and diacylglycerol, which may explain their enhanced insulin sensitivity | The findings enhance current knowledge regarding the development of sex-specific obesity and diabetes, with relevance to shift work personnel at risk of metabolic disorders | Mathew et al[67] |
| To evaluate the impact of hepatic STS overexpression on metabolic functions in mouse models of obesity and T2D through sex-specific mechanisms, STS transgenic mice were exposed to an HFD | STS liver expression increased in HFD and ob/ob models of obesity and T2D, as well as during fasting. Overexpressing STS in transgenic mice reduced weight, improved insulin sensitivity, and lowered liver fat and inflammation. In females, benefits were linked to the conversion of estrogen sulfates to active estrogens, which was lost after ovariectomy. In males, advantages were due to decreased inflammation and higher energy expenditure, even after castration. Treatment with estrone sulfate also improved metabolic health in both models | These findings reveal a previously unidentified role of STS in energy metabolism and T2D | Jiang et al[33] |
| C57/BL6J mice were bred and housed on either a CD or HFD from conception through weaning. The offspring then received either CD or HFD between 6 and 20 weeks of age. At 20 weeks, those exposed to maternal HFD showed glucose intolerance and insulin resistance | At 20 weeks, maternal HFD induced glucose intolerance and insulin resistance in offspring. Hepatic triacylglycerol levels, adipose tissue mass, and inflammatory markers were elevated following maternal HFD exposure. In male offspring, insulin secretion, islet area, insulin content, and PDX-1 mRNA expression decreased; conversely, these parameters increased in females. Islet oxidative stress was heightened in males, while remaining unchanged in females. Plasma estradiol concentrations were lower in males and further diminished by HFD, indicating that female offspring may be protected against insulin deficiency due to reduced oxidative stress | Maternal HFD caused insulin resistance and impaired pancreatic β-cell function in adult offspring, with notable sex-specific differences. These effects included adipose inflammation and liver steatosis. The differing β-cell outcomes may relate to increased oxidative stress and lower estradiol levels in males | Yokomizo et al[68] |
| An experimental model of streptozotocin-induced GD was used to assess the protein expression of LXRα (NR1H3) and LXRβ (NR1H2) | LXR expression in CO varied by tissue and receptor, with sex differences only seen for hypothalamic LXRβ at 35 days and 9 months. Most groups showed differences between CO and DO at 1 day, but these mostly disappeared except for male hypothalamic LXRβ. Glucose tolerance correlated negatively with LXRβ in CO, not DO; however, in male DO animals, this correlation was positive, as seen in intolerant subjects | Data indicate that GD affects hypothalamic LXR expression differently in male and female offspring | Kruse et al[69] |
| The effects of sex hormones on hepatic fetuin expression were evaluated in a rat model subjected to sex hormone administration. Results were corroborated by complementary in vitro investigations | E2 and DHT exerted contrasting influences on body weight among male and female rats. The expression of Ft-A and Ft-B in rat livers demonstrated sex-specific patterns and was subject to regulation by hormone receptors. In vitro experiments utilizing E2 or DHT corroborated the in vivo results, while antagonist assays confirmed that these effects were mediated through sex hormone receptors | Sex hormones modulate the sex-specific expression of hepatic fetuins via direct engagement with their respective hormone receptors | Kim et al[27] |
| Selected candidate genes were validated using real-time PCR analysis performed on hepatic tissues from both obese and lean rats | A total of 103 sex-different genes were found, mainly linked to chemical response, lipid metabolism, and organic substance response. Male-specific genes are related to liver metabolism, detoxification, and secretion, while most female-specific genes are involved in lipid metabolism or glycolysis | The data underscore the importance of considering sex- and diabetes-related variations during pre-clinical evaluation of drugs metabolized and secreted by the liver | Babelova et al[70] |
| This study used untargeted metabolomics to assess water-soluble metabolites in C57BL/6J male and female mice fed five different diets (Japanese, ketogenic, Mediterranean, American, and standard chow) for 7 months. Metabolite levels were measured in the liver, muscle, and fat, focusing on sex, diet, and their interaction | ANOVA shows liver tissue is most metabolically adaptable to diet changes, compared to adipose and skeletal muscle. The ketogenic diet was clearly distinct for both sexes by partial least-squares discriminant analysis. Most affected pathways, identified through pathway analysis, involve liver amino acid metabolism. Dietary patterns also influenced skeletal muscle amino acid profiles. Adipose tissue showed minimal metabolite changes, indicating stability despite dietary shifts | The ketogenic diet had the strongest physiological impact, especially in female mice. Metabolomics showed that diet alters metabolites differently by tissue, with the liver being the most responsive | Wells et al[71] |
| Hepatic pathology was compared between male and female SHROB rats, with key biochemical and molecular signaling pathways related to hyperinsulinemia and hyperlipidaemia assessed. The expression of 45 lipid biosynthesis and metabolism genes in rat livers was quantified using qPCR and Western blot | All SHROB rats exhibited hepatic steatosis, accompanied by increased expression of SREBP1, SREBP2, ACC1, and FASN proteins. In male rats, liver tissue showed greater induction of Pparg, Ppara, Slc2a4, Atox1, Skp1, Angptl3, and Pnpla3 mRNA levels. Conversely, female SHROB rat livers demonstrated consistently higher concentrations of phosphorylated JNK and AMPK, along with elevated CD36 expression | In SHROB rats, increased de novo lipogenesis primarily caused hepatic steatosis, with males and females showing different severity due to sex-specific differences in fatty acid transport and esterification | Dong et al[72] |
| Metabolic changes in the heart, liver, and kidneys of male and female mice, from healthy to diabetic states, were analyzed using 1H NMR metabolomics to identify sex-specific mechanisms in diabetes and its complications | Male mice experienced more pronounced metabolic disorders during diabetes progression than females. The kidneys were most affected, followed by the liver and heart. The altered metabolites chiefly involved energy, amino acid, choline, and nucleotide metabolism | The findings of this study indicate that the progression of DKD varies according to sex | Zhang et al[73] |
| Rats developed T2D after 3 weeks of a high-fat diet and daily 2-hour immobilization stress followed by streptozotocin treatment on day 15, and a 3-week course of LA | FBG increased in HFD-fed male rats, but not females. CS further raised FBS in HFD-fed rats; CS alone had no effect. HOMA-IR followed the same pattern as FBS. Serum corticosterone rose notably only in HFD-fed males exposed to CS. Pancreatic NF-κB levels were higher in HFD-fed males with CS exposure compared to controls, with no sex difference. LA at 10 mg/kg significantly lowered FBS, serum insulin, HOMA-IR, and corticosterone in HFD-fed males with CS, tended to reduce pancreatic NF-κB, and significantly improved liver MMP | Male rats are vulnerable to CS- and HFD-induced T2D, and LA can prevent T2D by reducing insulin resistance and corticosterone levels and also by increasing MMP in the liver | Shin et al[53] |
| The effect of diets on glycerol metabolism in mice and the influence of sex and GLP-1RA agonist treatment were investigated in female and male C57BL/6JRj mice fed either a CD or HFD for 12 or 24 weeks, with liraglutide administered to a subset of female mice | After 12 weeks of HFD, females gained less weight than males. Only females showed increased AQP7 in adipose tissue, while only males had higher glycerol kinase and larger adipocytes. At 24 weeks, weight gain and adipose changes became similar for both sexes. HFD caused notable hepatic steatosis in males but did not affect AQP9 levels in the liver. Liraglutide generally reduced HFD effects on glycerol metabolism. Overall, there was no coordinated increase in glycerol channels in adipose and liver tissues following HFD | The impact of a HFD on glycerol metabolism exhibits sex-specific differences in mice. An elevated abundance of AQP7 in female adipose tissue may underlie their comparatively attenuated response to HFD exposure | Iena et al[74] |
| This study examines the effects of exogenous SHBG on metabolically impaired hepatocytes, focusing on ER stress and lipid metabolism, using palmitate-treated HepG2 cells and post-mortem liver tissue samples cultured with 50 nM and 100 nM SHBG | SHBG protects against the development and progression of endoplasmic reticulum stress. It leads to reduced expression of IRE1α, ATF6, CHOP, and BIP. Additionally, SHBG regulates lipolytic gene expression in ex vivo settings | This research elucidates the cellular and molecular processes through which SHBG influences hepatocyte metabolism | Kornicka-Garbowska et al[75] |
| Young 3-month-old and middle-aged 12-month-old male and female mice were subjected to ad libitum or TRF of a Western diet to assess the metabolic impact | TRF confers metabolic benefits that are independent of age but dependent on sex. TRF protects against fatty liver and glucose intolerance in both males and females, whereas reductions in body weight are observed exclusively in males | These findings emphasize the notion that both timing and biological sex can significantly influence the effectiveness of treatments in preventing diet-induced metabolic disorders | Chaix et al[76] |
| This research analyzed the effects of a HFD on glucose regulation and T2D risk in Swiss Webster male and female subjects across the preweaning, peripubertal, and post pubertal developmental stages | In males, lifelong HFDs resulted in the highest T2D rates, but switching to chow after puberty lowered risk more than switching at weaning. The timing of HFD affected liver steatosis more than duration. Females avoided hyperglycemia with any HFD pattern, though postpubertal HFD caused notable liver and fat changes without impacting glucose tolerance. Most females had peri-insulitis, which did not affect glucose regulation | The data indicate distinct phases of glucose dysregulation induced by a high-fat diet in Swiss Webster mice, with notable sex disparities | Glavas et al[77] |
| The study investigated the role of Negr1 in preserving systemic metabolism, including glucose homeostasis, using male and female Negr1-/- mice receiving a standard or HFD | After 6 weeks of HFD Negr1-/- mice had higher glycaemic levels. HFD induced glucose tolerance variations only in male mice; Negr1-/- male mice displayed altered glucose tolerance, accompanied with upregulated BCAA circulating levels. Negr1-/- mice are biased towards gluconeogenesis, FA synthesis, and higher protein catabolism, all of which are amplified by HFD. Negr1 deficiency impairs the efficiency of energy storage, and reduced food intake could attempt to compensate for the metabolic challenge present in the Negr1-/- males during HFD feeding | Male mice exposed to HFD are protected from developing glucose intolerance, liver steatosis, and excessive weight gain by the presence of functional Negr1 | Kaare et al[78] |
| For 8 weeks, weight, food and water intake, and blood glucose were measured in mice to assess sex differences in T2D | Water intake and fasting blood glucose did not differ between db/db female and male mice, but glomerular injury and hepatic fibrosis showed sex-specific differences | Experimental design should account for sex differences in both male and female animals | Gao et al[79] |
| Ay mice underwent a seven-day course of FGF-21 administration, followed by an assessment of metabolic parameters and gene expression profiles in multiple tissues | Placebo-treated females vs males exhibited more obesity, reduced insulin levels and liver fat content, higher expression of insulin signaling and inflammatory genes. FGF-21 increased food intake without affecting body weight and the expression of genes for fat catabolism and insulin action in both sexes. Only in males, FGF-21 reduced high insulin and liver fat, while boosting muscle Cpt1 and Irs1 expression | FGF-21 demonstrates a more pronounced beneficial effect on metabolic disorders associated with melanocortin obesity in male animals | Makarova et al[80] |
| 18FDG-PET was used to evaluate baseline differences in whole-body glucose uptake between young male and female mice maintained on standard chow and HFDs | Sex and diet each affect glucose uptake in organs. Brown fat and heart showed the most FDG, with young females having 47% more brown fat uptake than males, and males having 49% more skeletal muscle uptake. HFDs lowered FDG uptake in brown fat, muscle, and heart, but increased it in the brain for both sexes | Glucose homeostasis regulation differs based on contextual factors and organ systems, highlighting the necessity to examine sex-specific outcomes and mechanisms with respect to T2D, obesity, and MetS | Gandhi et al[52] |
| Sex differences in hepatic histology were evaluated in TSOD and db/db mice, both being genetic models of NAFLD. Male and female mice from each strain received a standard diet and water, and groups of six were sacrificed at 3 months and 9 months for serum, pathology, and molecular analyses | At 3 months, male mice of both strains showed significantly higher serum AST and ALT levels (in TSOD mice), as well as greater steatotic and fibrotic areas (in db/db and both strains, respectively) compared to females; these sex differences were not observed at 9 months | Age-related sex differences in serum liver enzymes, as well as hepatic steatosis and fibrosis in TSOD and db/db mice, were consistent with patterns observed in human NAFLD | Dungubat et al[40] |
| Streptozotocin-induced diabetic rats received eugenol treatment at doses of 12 mg/kg and 24 mg/kg for four weeks | Streptozotocin raised serum glucose, cholesterol, triglycerides, LDL, liver enzymes, oxidative stress markers, pancreas damage, COX-2 expression, ovarian cysts, and anovulation, while lowering insulin, HDL, antioxidant activity, sex hormones, reproductive hormones, and PPAR-α. Eugenol improved diabetes indicators by enhancing lipid profile, antioxidant status, hormone levels, liver function, COX-2 and PPAR-α expression, and pancreas health, but did not affect ovarian cysts or follicle development | Eugenol may be useful for ameliorating some adverse features of diabetes regardless of sex | Kokabiyan et al[81] |
| Elderly Sprague-Dawley rats were divided into four groups: Control (STD diet), HFHSD diet, HFHSD plus metformin (HFHSD + M), and HFHSD plus liraglutide (HFHSD + L). Antidiabetic treatment began 5 weeks after diet initiation and continued for 13 weeks | Contrary to expectations, animals fed a HFHSD did not exhibit weight gain; however, they experienced notable metabolic alterations. Both antidiabetic interventions demonstrated sex-specific effects, yet neither was effective in preventing the development of prediabetes or diabetes | Liraglutide conferred beneficial effects on hepatic and skeletal muscle tissue in males, while in females it was associated with insulin resistance | Ivić et al[82] |
| This study started from the assumption that chronic CYP1A1/CYP1A2 activation may play a role in HFD-induced metabolic dysfunction in mice, and deletion of these enzymes could be protective. Male and female global CYP1A1/CYP1A2 KO and Cyp WT mice aged 29 weeks to 31 weeks were fed either a 45% HFD or standard chow for 14 weeks | Cyp KO females demonstrated partial protection against HFD-induced glucose intolerance, and chow-fed Cyp KO females exhibited reduced plasma insulin levels as well as diminished insulin secretion from isolated islets compared to Cyp WT females. Additionally, HFD-induced hyperinsulinemia developed later in Cyp KO males relative to Cyp WT males. Elevated expression of CYP1A1 and other stress-related genes was observed in Cyp WT male islets following HFD feeding, but not in Cyp KO islets, suggesting that CYP1A1 plays a role in mediating islet stress responses. Across both sexes, liver pathology, adiposity, and adipose tissue inflammation were predominantly influenced by dietary factors rather than genotype | This study reveals a sex-specific role for CYP1A1/CYP1A2 in regulating systemic metabolism under HFD conditions. Deleting CYP1A1/CYP1A2 partially protects female mice from HFD-induced glucose intolerance and lowers insulin on a chow diet, while in males, it delays HFD-induced hyperinsulinemia and reduces islet stress. The finding that these genotype effects are limited to islets, indicates a distinct function for islet CYP1A1/CYP1A2 in metabolic stress response | Ching et al[47] |
| Both male and female DNAJB3 KO and WT mice were administered high-sucrose, HFDs, or LFDs for 12 weeks. Body weight, food intake, glucose tolerance, and energy expenditure were evaluated; blood, adipose, and liver tissues were collected for histological examination and gene expression analyses | HF-fed KO females had greater body and fat mass, reduced glucose clearance, and lower energy expenditure than other groups. In males, both genotypes were affected by the HFD. HF-KO females had increased leptin, IL-6, and insulin, while HF-KO males showed higher leptin and resistin. DNAJB3 deficiency altered inflammatory and glucose transporter gene expression in adipose tissue and pancreas, indicating impaired glucose metabolism | This study highlights the important role of DNAJB3 in the regulation of glucose and metabolism, with a notable emphasis on its impact among female individuals | Nejat et al[48] |
Sex hormone deficiency further disrupts glucose uptake in skeletal muscle and adipose tissue, as demonstrated in castrated rats. These effects are associated with reduced Akt phosphorylation and lower GLUT4 expression. Testosterone effectively restores these metabolic pathways, whereas estradiol shows only partial effects[66,67]. In addition, sex hormones regulate hepatic expression of key proteins, such as fetuins, through hormone receptor-mediated mechanisms[33,68].
Sex-specific differences also shape hepatic and metabolic responses in animal models of obesity and diabetes. Female animals exhibit a greater capacity for glutathione synthesis and a more anabolic response, which helps limit oxidative stress. In contrast, males tend to show increased fatty acid oxidization and higher oxidative stress[27,69]. Metabolomic studies further indicate that the liver is the most metabolically adaptable tissue across various dietary conditions, with more pronounced changes observed in females, particularly under ketogenic diets[70,71].
Knockout and transgenic models provide additional evidence of sex disparities in body weight regulation, insulin sensitivity, and lipid metabolism. For example, in Id2-/- mice, males display improved glucose tolerance and enhanced muscle metabolism, while females develop smaller adipocytes[72,73]. The liver enzyme STS also plays a sex-specific role: In females, it promotes estrogen activation and improves metabolic regulation, whereas in males it reduces inflammation and boosts energy expenditure[53,74]. Maternal exposure to a HFD induces sex-specific metabolic and inflammatory reactions in offspring. Females’ offspring are relatively protected against pancreatic beta-cell dysfunction, likely due to higher estradiol levels and reduced oxidative stress[75,76]. Conversely, males’ offspring are more susceptible to insulin resistance and hyperglycaemia, particularly following chronic stress and high-fat feeding[77,78].
In mouse models of MASLD, males generally show higher liver enzyme levels, more severe steatosis, and greater liver fibrosis than females, especially at younger ages; however, these sex differences tend to diminish with aging[79,80]. Similarly, genetic models reveal sex-specific responses in body weight, glucose tolerance, and inflammatory pathways[40,52].
Collectively, these studies emphasize the importance of accounting for biological sex in the experimental design and in the evaluation of therapeutic strategies for T2D and metabolic disorders. Males and females differ substantially in glucose regulation, insulin sensitivity, and the pathobiology of inflammatory response[81,82]. Interventions like time-restricted feeding also demonstrate sex-specific metabolic benefits, reinforcing the necessity for personalized strategies[47,48].
Overall, sex differences play a central role in the pathophysiology and management of T2D and related metabolic disorders. These differences involve hormonal, metabolic, genetic, and environmental factors that differentially affect key organs, including the liver, muscles, adipose tissue, and pancreas. A better understanding of these variations may support the development of more effective, sex-informed strategies to prevent and manage T2D and its complications.
Pivotal studies that investigate sex-specific associations between metabolic dysfunction, liver health, and related cardiometabolic risk factors are summarized in Table 3[56,83-92]. The findings highlight the importance of considering sex differences in the diagnosis, management, and risk assessment of metabolic dysfunction, and MASLD.
| Method | Findings | Conclusion | Risk of bias | Ref. |
| Of 158 women (47 normal and 111 with IGT) and 148 men (74 normal and 74 with IGT) were enrolled. They underwent a hyperinsulinemic normoglycemic clamp to determine M3, besides liver enzymes (ALT, AST, and GGT), metabolic and anthropometric parameters | Significant bivariate correlations were found between clamp measured M3 and all three liver enzymes in both sexes. After adjustment for possible metabolic confounders, correlations ceased in the male population alone. Feature selection analysis showed that ALT is an important attribute for M3 among women alone. MRA confirmed that BMI (P < 0.0001) and ALT (P = 0.00991) significantly and independently predicted clamp measured muscle glucose uptake in women [R (2) = 0.5259], while in men serum fasting insulin (P = 0.0210) and leptin levels (P = 0.0294) but none of the liver enzymes were confirmed as significant independent predictors of M3 [R (2) = 0.4989] | The existing sex-specific association between insulin sensitivity, MRFs, and ALT might explain the sex difference in the predictive role of ALT elevation for CVD. Accordingly, ALT may be used as a simple biomarker of IR in women | Moderate | Buday et al[83] |
| A cohort with T2D (n = 64, 30 male/34 female) and a sample of healthy subjects (n = 25, 13 male/12 female) were enrolled. Intraorgan and visceral fat were quantified by MR and VLDL1-TG export by intralipid infusion techniques | Intrahepatic and intrapancreatic TG content was elevated among those with T2D, irrespective of sex. In non-diabetic subjects, fat levels in both organs were significantly lower in women than men [1.0% (0.9%-1.7%) vs 4.5% (1.9%-8.0%), P = 0.005, and 4.7% ± 0.4% vs 7.6% ± 0.5%, P < 0.0001, respectively]. T2D women had higher hepatic VLDL1-TG production rate and plasma VLDL1-TG than healthy women [559.3 ± 32.9 mg/kg/day vs 403.2 ± 45.7 mg/kg/day, P = 0.01, and 0.45 (0.26-0.77) mmol/L vs 0.25 (0.13-0.33) mmol/L, P = 0.02], whereas there were no differences in men [548.8 ± 39.8 mg/kg/day vs 506.7 ± 29.2 mg/kg/day, P = 0.34, and 0.72 (0.53-1.15) mmol/L vs 0.50 (0.32-0.68) mmol/L, P = 0.26]. Weight loss decreased intraorgan fat content and VLDL1-TG production rates regardless of sex, and these changes were accompanied by similar rates of T2D remission (65.4% vs 71.0%) and CVD risk reduction (59.8% vs 41.5%) in women and men, respectively | In T2D, women have liver and pancreas fat accumulation like men, associated with raised hepatic VLDL1-TG production rates. Dynamics of TG turnover differ between sexes in T2D and after weight loss. Collectively, these changes may explain the raised CVR among women with T2D | High | Jesuthasan et al[84] |
| This cross-sectional observational study enrolled n = 16126 adult participants from the KNHANES. Absolute and body size-adjusted HGS indices were evaluated | Prevalence of T2D in all, younger, and older groups were 131%, 4.2%, and 21.4%, respectively. Proportions of cardiometabolic diseases were all higher in those with than without T2D in sex-stratified age groups, whereas obesity and MASLD were higher in younger T2D, and HTN was higher in older T2D in both sexes. Adjusted HGS significantly correlated with cardiometabolic parameters, and thus, high ORs for T2D in low tertiles of adjusted HGS were shown in all groups, whereas high ORs for T2D in low tertiles of absolute HGS were observed only in older men | Obesity and MASLD were more prevalent in younger T2D, while HTN was more prevalent in older T2D in both sexes. Low adjusted HGS was associated with higher T2D risk in all groups, whereas low absolute HGS was associated with higher T2D risk in older men, indicating that adjusted HGS might be useful in screening, especially for younger or obese individuals with T2D | Low | Bae et al[85] |
| The study comprised 332 hospitalized patients. The following data on leading CVD and risks related to CV drug administration were collected: Age, hyperlipidemia, T2D, CKD, CLD, HF, HTN, MI, and S. The amount of the CV drugs administered during hospitalization was expressed as the sex-specific DDD/100 BD | During hospitalization in the ICU, women were less likely to be treated with statins than male patients (30.1 DDD/100 BD vs 57.5 DDD/100 BD, P < 0.05). There was no difference between sexes regarding the use of antihypertensive drugs. Women were less likely to be treated with ASA (30.4 DDD/100 BD vs 36.9 DDD/100 BD, P < 0.05) | This study identifies sex differences in CV drug administration, and it is possible that these discrepancies mirror differences in primary care | Moderate | Drakul et al[86] |
| HTG was prospectively assessed among 2331 individuals using 1H MRS magnetic resonance spectroscopy, and plasma concentrations of TG, T-chol, LDL-chol, HDL-chol, and UA | The 95th percentile for HTG in lean non-AI individuals was 1.85%. Plasma insulin, TG, T-chol, LDL-chol, and UA concentrations were increased, and HDL-chol was decreased in individuals with HTG content > 1.85% and ≤ 5.56% compared to those individuals with HTG content ≤ 1.85%, and these altered parameters were associated with increased IR. Lean non-AI women had lower mean HTG than lean non-AI men, but both lean AI men and women showed a 40%-100% increase in HTG compared to their non-AI counterparts, with this rise linked to higher CMRFs | HTG concentrations > 1.85% (the 95th percentile of HTG in lean non-AI individuals) were associated with IR and CVR factors. Premenopausal women were protected from these changes, while young, lean AI men and women showed increased HTG levels and related CMRFs | Low | Petersen et al[87] |
| Of 5027 men were enrolled in this population-based study conducted in China. Low eGFR was defined as 60 mL/minute/1.73 m2 | After adjusting for age, smoking, metabolic factors, and testosterone, through increasing quartiles of SHBG, a significantly positive association between SHBG quartiles and eGFR was detected in men (Q1 vs Q4, β = -2.53, 95%CI: -3.89 to -1.17, P < 0.001). Compared to the highest quartile of SHBG, SHBG in the lowest quartile was associated with 96% higher odds of low eGFR (OR = 1.96, 95%CI: 1.10 to 3.48) in the fully adjusted model. At stratified analyses, the associations between a 1-SD increase in serum SHBG and the prevalence of low eGFR were significant in men aged ≥ 60 years old, WC, diabetes, HTN, dyslipidemia, and MASLD | Reduced serum SHBG levels were independently associated with decreased eGFR and an increased prevalence of low eGFR among Chinese men, after adjusting for demographic characteristics, lifestyle factors, MRFs, and testosteronemia | Low | Zhang et al[88] |
| This cross-sectional study included data from 33216 randomly selected Spanish adult workers submitted to occupational medical check-ups. Sociodemographic, anthropometric, and clinical parameters were recorded, and MASLD was identified with FLI ≥ 60. MetS was diagnosed according to the IDF criteria, and CVR was determined with the REGICOR-Framingham equation | The global prevalence of MASLD was 19.1%, 27.9% (95%CI: 23.3%-28.5%) for men and 6.8% (95%CI: 6.4%-7.3%) for women, and increasing across age intervals. Compared to women, men had worse cardiometabolic and anthropometric profiles. At MVA, SLD was strongly associated with age, HDL-chol, social class, prediabetes, diabetes, preHTN, HTN, and smoking in both sexes. The association between diabetes, HTN, and MASLD was stronger in women than in men | Compared to women, men had a higher prevalence of MASLD, a worse cardiometabolic profile and a higher CVR. Nevertheless, MASLD was more strongly associated with T2D or HTN in women than in men, suggesting that metabolic dysfunction poses a more severe threat to liver health in women than in men | Low | Fresneda et al[89] |
| This retrospective longitudinal study of 92997 patients seen between January 2017 to January 2019 used electronic medical records to abstract data back to April 1997 | Globally, 59323 individual without known CLD received an ALT test. Age, ethnicity, and metabolic factors were associated with higher odds of ALT testing (P < 0.01). At MVA, female sex (OR = 2.7), Latinx ethnicity (OR = 2.6), API race (OR = 1.3), overweight/obesity (OR = 1.8, OR = 2.6), and dyslipidemia (OR = 1.3) were associated with abnormal ALT (P ≤ 0.001) | Among individuals without any known CLD, women, Latinx, API, and persons with excess BMI had greater risks of abnormal ALT | Low | Kim and Khalili[90] |
| Two-hundred and twelve T2D patients, equally assigned to oral or subcutaneous semaglutide (n = 106 per group), were enrolled. The primary endpoint was the change in HbA1c. Secondary endpoints included variations in anthropometric and metabolic parameters. | A total of 208 patients completed the study. In men, sc semaglutide significantly reduced Δ weight (P = 0.010), Δ HbA1c (P = 0.037), and Δ LDL-chol (P = 0.038) compared to oral semaglutide. In women, sc semaglutide caused a significant decrease in Δ HSI (P = 0.024) and also led to a greater Δ GOT reduction (P = 0.035) than in men | This real-world study indicates that sc semaglutide offers superior metabolic benefits compared to the oral formulation, especially among male patients | High | Piccione et al[91] |
| This retrospective study included 6107 subjects submitted to annual health check-ups. CMI was calculated by multiplying the ratio of TG and HDL-chol (TG/HDL-C) by WHtR | MAFLD prevalence rose with higher CMI quartiles in both sexes. Higher CMI independently increased MAFLD risk (per SD: OR = 2.72 for males, OR = 3.26 for females). Those in the highest CMI quartile had the greatest odds of MAFLD (males: OR = 15.82; females: OR = 22.60), with significant trends (P < 0.001). CMI showed a non-linear association with MAFLD and had the largest AUC among obesity indexes for identifying MAFLD (males: 0.796; females: 0.853). FLD in males (AUC = 0.796, 95%CI: 0.782-0.810) and females (AUC = 0.853, 95%CI: 0.834-0.872) | CMI served as an effective marker for MAFLD screening in Chinese adults. The diagnostic value of CMI for MAFLD was higher in women than in men | Low | Gu et al[92] |
| A total of 7950 Taiwan Biobank participants were enrolled; 6478 had anthropometric, biochemical, and hematologic data, and 4185 underwent abdominal sonography for MASLD assessment | High PCSK9 levels were linked to older age, female sex, adverse cardiometabolic factors, and changes in blood markers. In women, associations with platelet count were stronger. Increasing PCSK9 quartiles correlated with higher odds of IR, MetS, DM, and MASLD, particularly for women. Elevated PCSK9 predicted higher risks of all-cause, non-cardiovascular, and cancer mortality, mainly affecting women | Elevated PCSK9 levels are linked to higher risks of IR, MetS, DM, MASLD, and mortality, reflecting poorer metabolic health among Taiwanese individuals. These effects are more pronounced in women, emphasizing the need for sex-specific risk assessment in metabolic disorders | Low | Yeh et al[56] |
Sex differences in liver health and cardiometabolic risk are increasingly recognized as important factors in both clinical practice and public health research. Recent studies have systematically explored these distinctions using diverse methodologies and large population-based cohorts. Table 3[56,83-92] summarizes key findings from several pivotal studies, each contributing unique insights into how sex modifies the relationships among insulin sensitivity, liver enzyme profiles, fat distribution, and risk of MASLD or metabolic dysfunction-associated fatty liver disease (MAFLD). Although earlier studies conducted under the NAFLD framework remain informative, MASLD and MAFLD represent distinct clinical entities with different patient populations and disease trajectories and therefore warrant separate consideration[93].
The biological links between liver health and insulin sensitivity are strongly influenced by sex. ALT, for example, may serve as a practical biomarker of insulin resistance and possibly cardiovascular disease risk in women, highlighting fundamental sex-related differences in metabolic disease pathophysiology[83]. This may explain why women are more likely than men to present with elevated ALT in the absence of clinically recognized liver disease, reinforcing the sex-specific patterns observed by Buday et al[83] and suggesting that metabolic and demographic factors must be carefully considered when interpreting liver enzyme profiles in clinical practice. Of concern, although men generally exhibit worse cardiometabolic risk profiles, women with MASLD appear more susceptible to adverse outcomes when metabolic dysfunction is present. This finding points to a paradox in which women may experience more severe complications of MASLD despite a lower overall prevalence of disease[84,87,89].
Handgrip strength (HGS) is another potential screening tool for T2D, especially in younger or obese individuals. Bae et al[85] reported that low adjusted HGS was associated with an increased risk of T2D across all groups, whereas low absolute HGS was mainly linked to T2D risk in older men. In addition, elevated circulating PCSK9 levels have been associated with higher risks of metabolic dysfunction, MASLD, and all-cause mortality, with these associations being more pronounced in women[56]. These findings further support the need for sex-specific risk assessments in metabolic disorders[56].
There is also growing awareness of the increased risk of CKD among individuals with MASLD. Zhang et al[88] showed that men particularly elderly individuals and those with MetS in the lowest SHBG quartile had nearly twice the odds of reduced eGFR. This finding offers valuable pathobiological insights and may inform triage practices.
Finally, clinical management should consider sex-specific differences in therapeutic response and access. Emerging data suggest possible sex-related differences in the efficacy of semaglutide for T2D[91] and underscore the importance of ensuring equitable treatment opportunities for women and men, particularly in relation to cardiovascular risk reduction[86].
Synthesizing these findings, several key themes emerge. First, sex substantially influences the relationships between liver enzymes, hepatic fat accumulation, and cardiometabolic risk, with women showing heightened vulnerability to the metabolic consequences of liver dysfunction. Second, although women generally have lower hepatic fat and metabolic risk in the absence of overt disease, this protection erodes in the presence of T2D, with fat accumulation and triglyceride export rates becoming similar to those of men. Third, composite risk indices, such as the cardiometabolic index, appear particularly effective in identifying at-risk women, supporting the use of sex-specific screening strategies. Fourth, innovative biomarkers, such as HSG, PCSK9, and SHBG, warrant further validation in sex-stratified analyses. Fifth, men and women may differ in both access to and response to therapeutic interventions. Collectively, these studies emphasize the importance of considering sex as a key biological variable in the assessment, diagnosis, and management of liver-related and cardiometabolic disorders. Future research should further clarify the mechanisms underlying these differences to enable more tailored and effective prevention and treatment strategies for both sexes.
The rs58542926C>T (E167K) variant in the TM6SF2 gene is associated with increased risk of both MASLD and T2D. Fan et al[45] performing sex-stratified analyses found that this variant was significantly associated with T2D, particularly among males. Experiments using E167K knock-in mice showed that impaired glucose tolerance occurred only in male mice. TM6SF2 was also found to interact with the endoplasmic reticulum stress sensor IRE1α, and male knock-in mice demonstrated reduced hepatic IRE1α signaling. Collectively, these findings suggest that the E167K variant contributes to glucose intolerance mainly in males, based on both human and animal data.
Jaruvongvanich et al[38] conducted a systematic review of 16 studies including 13721 men and 5840 women. They reported that men with MASLD had lower TT levels, and higher TT was associated with reduced MASLD odds in men but increased risk in women. SHBG levels were lower in MASLD patients of both sexes, with a stronger inverse association observed in women. Higher SHBG levels were linked to lower MASLD risk[38].
Reduced SHBG is seen in several metabolic conditions, including MASLD, and is a sensitive biomarker for insulin resistance and MetS. Low plasma SHBG levels are associated with an increased risk of hyperglycaemia and T2D, particularly among women[94]. Another study found that low serum SHBG is associated with insulin resistant in men with MASLD[95]. Evidence suggests that in women, SHBG, and in men, testosterone, may each be inversely associated with diabetes risk[96,97] analyzed NHANES data from 2013-2016 and found that the prevalence of low SHBG in the United States population was 3.3% in men, 2.7% in women, and 3.0% overall. Low SHBG was associated with higher body mass index (BMI), diabetes, certain ethnicities, chronic obstructive pulmonary disease, heart disease, and smoking. These findings may partly explain the higher prevalence of MASLD in men.
Sex chromosome aneuploidies such as Turner syndrome (TS) and Klinefelter syndrome (KS) provide useful clinical models to evaluate the role of X-chromosome in MASLD and systemic metabolic dysfunction[98]. In this context, Thomsen et al[99] studied individuals with KS and TS and examined the effects of sex steroid treatment. Testosterone in KS did not significantly reduce soluble cluster of differentiation 163 (sCD163), a marker of systemic inflammation[100]. However, hormone replacement therapy and oral contraceptives significantly reduced sCD163 levels in TS subjects and controls. These results prompt further investigation regarding the immune-metabolic effects of sex chromosome abnormalities.
Body fat distribution is strongly influenced by sex and age and plays a major role in ectopic intrahepatic fat accumulation and metabolic dysfunction. Naeem et al[101] analyzed data from 3628 adults and found that in women, visceral and liver fat were most strongly associated with OGTT results, whereas in men, the waist-to-height ratio showed the strongest association. Higher relative fat-free mass was linked to better OGTT outcomes in both sexes, with stronger effects in men. De Ritter et al[102], in a study of 7639 adults, reported clear sex differences in body composition related to prediabetes and T2D. Women showed greater increases in subcutaneous fat and hip circumference, while men had larger increases in visceral fat; however, liver fat percentage did not differ by sex. The clinical significance of these differences requires further investigation. Malpique et al[51] examined 86 prepubertal children and assessed BAT activation. Prepubertal girls showed greater BAT activation than boys, and higher BAT activity was linked to less visceral fat and better metabolic profiles. Long-term studies are needed to clarify the relevance of these findings to later metabolic disease[51]. Taken together, these studies confirm that sex-specific patterns of body fat distribution strongly influence the risk of intrahepatic fat accumulation and metabolic dysfunction.
FGF-21 is a liver-derived hormone that plays a key regulatory role in glucose and lipid metabolism and has been implicated in the pathogenesis of T2D[35,103]. Wang et al[35] conducted a prospective case-control study including 251 incident diabetes cases and 251 matched controls. Higher circulating FGF-21 levels were associated with increased T2D risk in women [odds ratio (OR) = 1.50, 95% confidence interval (CI): 1.00-2.25] but not in men (OR = 0.89, 95%CI: 0.52-1.53), with a significant interaction by sex (P = 0.029). These findings suggest that FGF-21 may be a sex-specific biomarker of diabetes risk, particularly among Chinese women, and highlight the need for further research in other ethnicities[35].
Sex disparities in MASLD are increasingly recognized as clinically relevant and require careful consideration. Di Bonito et al[104] studied obese children and adolescents and found that elevated ALT levels were more strongly associated with features of MetS in boys than in girls. Suzuki et al[105] reported that during intensive weight loss, ALT elevations were more pronounced in men with rapid weight loss and in women with higher baseline BMI, indicating sex-specific patterns of liver enzyme response. Wang et al[106] examined middle-aged adults and found that low vitamin D and low SHBG were independently associated with increased MASLD risk in both sexes, with particularly strong effects when both markers were low. Kastberg et al[107] performed a cross-sectional analysis of 743 rural and urban Kenyan individuals from diverse ethnic backgrounds and observed lower odds of MASLD in men than anticipated, highlighting the necessity for further investigation within Black African populations. Among people with human immunodeficiency virus, female sex appears to provide some protection against MASLD-related fibrosis, but menopause increases the risk of metabolic dysfunction-associated steatohepatitis and liver fibrosis. Women show faster worsening of metabolic and liver-related markers around the perimenopausal period, highlighting important age- and sex-specific vulnerabilities[108]. Taken together, these studies suggest that sex-specific factors, including hormonal status, body composition, and lifestyle habits, substantially modulate liver injury risk in the setting of metabolic dysfunction. Differences between pediatric and adult populations further suggest that age and developmental stage modify these relationships.
Genetics, hormones, SHBG, body fat, FGF-21, and sex all influence liver health, which is shaped by age, hormones, body composition, and lifestyle habits. More research is needed to create predictive models and validate them in future studies.
The clinical implications of the MASLD-MetS-T2D axis extend far beyond metabolic abnormalities and liver disease. As illustrated in Figure 2, sex differences within this interconnected axis ultimately translate into divergent long-term health outcomes in women and men. Although the core metabolic pathways are shared, their clinical expression differs according to sex, resulting in distinct risks for CVD, CKD, liver-related events, malignancies, mental health disorders, and other clinical disorders. These outcome disparities represent the final manifestation of the sex-specific interactions described in previous sections. In the following section, we review the available clinical evidence on how sex modifies the associations between metabolic dysfunction and major adverse outcomes, with particular focus on cancer and cardiovascular mortality.
A consistent body of evidence identifies T2D and MASLD as risk factors for several types of cancer. In this section, we examine how sex modulates these associations based on published studies. Diabetes increases the risk of all-site cancer in both women and men, but the excess risk appears to be slightly higher in women. A large systematic review and meta-analysis by Ohkuma et al[109], including 121 cohorts with more than 20 million individuals, found that women with diabetes have approximately a 6% higher overall cancer risk compared to men with diabetes [pooled relative risk ratio (RRR) = 1.06, 95%CI: 1.03-1.09]. The pooled RRRs were 1.10 (95%CI: 1.07-1.13) for all-site cancer incidence and 1.03 (95%CI: 0.99-1.06) for all-site cancer mortality. Diabetes was also associated with a significantly greater relative risk in women than in men for oral, stomach, and kidney cancers, as well as leukaemia; however, the relative risk for liver cancer was lower in women compared to men.
The large bowel is one of the organs at risk for cancerization in the context of metabolic dysfunction[13]. BardalesZuta et al[110] performed a retrospective case-control study of individuals undergoing colonoscopy in Peru and found that MASLD and the male sex were associated with colorectal adenoma (OR = 3.3, 95%CI: 1.8-6.1 for MASLD; OR = 2.2, 95%CI: 1.3-3.6 for male sex). These findings, together with other longitudinal studies[111] suggest that targeted colorectal cancer screening strategies may be useful in patients with MASLD[112].
Several studies have also evaluated sex differences in primary liver cancers (PLCs). Burton et al[113], analyzing United Kingdom cancer registry data including more than 82000 cases, reported that HCC was most common in Scottish men, while intrahepatic cholangiocarcinoma was more frequent in women. The study suggests that persistent obesity and diabetes in the United Kingdom are likely to sustain the future burden of liver cancer. In agreement, a large Korean cohort study by Yi et al[114], enrolling more than 500000 participants followed for 11 years, found that diabetes increased the risk of HCC by 80% after multivariable adjustment. Men had higher absolute risks of HCC, but women showed stronger associations between HCC and factors such age and cirrhosis. The authors pinpoint that diabetes increases HCC risk independent of cirrhosis. Consistent results were reported by Petrick et al[115] in a nested case-control study within the Multiphasic Health Checkup cohort of Kaiser Permanente Northern California members. Obesity was associated with a 2.4-fold increased risk of PLC, and overweight was associated with a 32% increased risk irrespective of sex, race/ethnicity, or birth cohort. Among women, obesity was linked to at least a 2-fold increase in liver cancer risk, indicating that elevated BMI increases risk across both sexes and all races/ethnicities.
A multicenter Austrian study analyzing more than 1000 patients undergoing hepatic resections for primary or metastatic liver tumors found that women with HCC tended to be younger and had fewer comorbidities than men. However, despite these favorable features, women experienced worse overall survival, possibly due to more advanced disease at diagnosis[116]. In addition, Chen et al[117] using national insurance data from more than diabetic patients, showed that diabetes doubled the risk of liver cancer compared with non-diabetic age- and sex-matched controls, though this risk decreased after adjusting for clinical factors, indicating a combined effect with diabetes-related disorders. This excess risk was particularly high in diabetic patients with cirrhosis, highlighting a strong pathogenic interaction between diabetes and CLD. Figure 3 provides an overview of the concepts previously discussed regarding sex-specific risks for cancer and pre-cancerous conditions in individuals diagnosed with diabetes.
Several studies have explored whether the cardiovascular risk associated with metabolic disorders differs between women and men[118]. Matsubayashi et al[119] analyzed data from more than 570000 individuals without previous cardiovascular disease. Over a median follow-up of 5.2 years, they found that MAFLD only (without Mets) had a greater impact on coronary artery disease risk in men, whereas, MetS only (without MAFLD) or a combination of both (MAFLD and Mets) had a greater impact in women. These findings imply that the sex-specific distinction between MetS and/or MAFLD, particularly in the presence or absence of T2D, may help to improve cardiovascular risk stratification. Similarly, Abbaszadeh et al[120], in a cohort of more than 7000 adults, reported that MAFLD was more prevalent in men at younger ages. However, women with MAFLD faced a greater risk of cardiovascular events and mortality. Among individuals with MAFLD, those with coexisting diabetes showed the highest risk for CVD and mortality[120]. It should, however, be pointed out that, compared to MASLD, MAFLD more accurately identifies adverse clinical outcomes[93] implying the different disease nomenclatures may provide different clinical findings. Desai et al[121] examined more than 400000 hospitalizations with MASLD. They found that MASLD was more frequently diagnosed in women, who were generally older and had higher rates of obesity and uncomplicated diabetes. Nevertheless, major adverse cardiovascular events, acute myocardial infarction, and cardiac arrest were more common in men, particularly at older ages. This study highlights that both age and male sex are important modifiers of cardiovascular outcomes in MASLD[121].
GGT is increasingly identified as a biomarker of cardiovascular risk rather than simply an indicator of liver health[122]. With this background, Jung et al[123] analyzing three large Korean cohorts (approximately 28000 CVD-free participants with a 50-month follow-up), found that higher GGT/HDL-cholesterol ratios were associated with increased risk of cardiovascular disease in women but not in men. This study supports the adoption of sex-specific biomarkers for cardiovascular risk assessment.
Women are generally considered to be relatively protected from CVD[4], but the presence of MASLD appears to abrogate this protection[12]. Ren and Zheng[124] using data from the United States NHANES survey (approximately 2600 MASLD cases), reported that men with MASLD had higher overall mortality than women (12.4% vs 7.7%; P = 0.005); however, women with MASLD aged ≤ 60 years had a higher risk of cardiovascular death than men of the same age group (adjusted hazard ratio = 0.214, 95%CI: 0.05-0.87). These results suggest that reproductive status and age strongly influence sex differences in cardiovascular mortality among patients with MASLD.
Finally, sex interacts with ethnicity in shaping cardiometabolic risk. Confirming this notion, Chan et al[125] in a study of healthy non-obese Asian adults, found that women had greater insulin sensitivity at the level of adipose tissue, whereas men had higher visceral fat and lower PPG control. These physiological differences may contribute to sex-specific patterns of metabolic and cardiovascular disease. Likewise, Goyal et al[126] analyzing nearly 1.9 million hospitalizations for heart failure with preserved ejection fraction (HFpEF), showed that sex and race both influenced clinical characteristics and outcomes. For example, atrial fibrillation was more strongly associated with mortality among women and Black patients, indicating that sex and ethnicity contribute to the heterogeneity of HFpEF phenotypes.
Sex-specific factors play a critical role in the development and progression of cancer, and cardio-metabolic and vascular disorders. Tailoring risk assessments and therapeutic interventions to an individual’s sex and ethnicity may substantially improve clinical outcomes.
Park et al[127] analyzed National Health Insurance claims data from approximately 49000 adult psychiatric inpatients in South Korea to assess the prevalence of physical comorbidities according to psychiatric diagnosis and sex. These authors found that liver disease was most prevalent in patients with mood disorders and ranked second, after neurologic conditions, among patients with schizophrenia-related disorders. In both diagnostic groups, liver disease and diabetes were more common in men, whereas hypothyroidism was more common in women. This study highlights the importance of screening and referring psychiatric patients for appropriate specialist management of their physical comorbidities.
Kaser et al[128] studied approximately 1100 patients who underwent non-traumatic lower-limb amputation. They found clear sex-related differences in outcomes and comorbidity patterns. Women most often died from diabetes, peripheral artery disease (PAD), and cerebrovascular disease, while men more commonly died from liver disease, renal disease, and myocardial infarction. Women underwent amputations at a significantly older age than men (78.9 years vs 68.1 years); however, major amputations were more likely to be their first surgical intervention in women, probably reflecting their older age at presentation. The study highlights the need for sex-specific strategies in risk-factor management and prevention of PAD.
Our review is limited by excluding non-English articles. With this limitation in mind, evidence shows sex-dependent links among MASLD, T2D, metabolic dysfunction, and biological sex. Research highlights connections between sex steroids, hepatokines, genetics, fat distribution, and cellular stress responses, leading to distinct metabolic profiles in men and women. Premenopausal women benefit from estrogen-driven insulin sensitivity, healthier fat storage, and stronger antioxidant defenses, slowing disease progression. Men, however, have more visceral and liver fat, greater oxidative stress, and unique hormonal and genetic risks, which drive earlier and faster metabolic disease development. After menopause, women’s protective mechanisms decline, equating their risk of advanced MASLD and cardiometabolic disease with men’s. The biological mechanisms described result in sex-specific clinical differences in conditions like cardiovascular disease, renal disease, cancers, mental disorders, and extra-hepatic complications related to diabetes and MASLD. Recognizing these processes explains the observed variations in morbidity and mortality in clinical practice. In this regard, robust evidence shows that ethnic discrepancies characterize the incidence of MASLD in the United States, with Hispanic populations exhibiting the highest disease prevalence, followed by non-Hispanic Whites, and lastly, non-Hispanic Black individuals[129]. The pathobiology of such differences needs to be fully revealed by in-depth mechanistic studies. Similarly, additional sex-specific biomarkers of MASLD, cardio-metabolic risk and their cut-off values need to be prospectively validated, with SHBG for MASLD[130], FGF-21 for incident T2D[34,53], higher PCSK9 for insulin resistance, MetS, MASLD, and all-cause and cancer mortality[54,55], ALT for insulin resistance[21] and higher GGT/HDL-cholesterol ratios for cardiovascular disease[122] being all more specific in women than in men.
While our understanding of the interconnections between menopausal hormonal replacement therapies and liver health increasingly expands[28,131-134] and similarly do the effects of sex and gender on response rates to pharmacotherapies used in the diabesity arena[135] significant knowledge gaps persist in this area. Although most mechanistic insights are obtained from experimental models, there remains a lack of validation in large, properly stratified human cohorts, particularly regarding sex differences. Based on the data presented in this review, we advocate for sex-stratified analyses in prospective patient populations and intervention trials aimed at targeting sex-specific pathways. More research is needed to understand how menopause, hormone exposure, and gender-affirming therapy affect T2D and MASLD risk. The roles of genetic variation and sex in treatment response and biomarker thresholds remain unexplored. For precision medicine to improve prevention, risk assessment, and therapy for MASLD and T2D, future studies must consistently integrate sex as a key biological variable across clinical research.
| 1. | Lu X, Xie Q, Pan X, Zhang R, Zhang X, Peng G, Zhang Y, Shen S, Tong N. Type 2 diabetes mellitus in adults: pathogenesis, prevention and therapy. Signal Transduct Target Ther. 2024;9:262. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 443] [Cited by in RCA: 327] [Article Influence: 163.5] [Reference Citation Analysis (2)] |
| 2. | Tabatabaei Yeganeh HS, Prokop LJ, Kiliaki SA, Gnanapandithan K, Yousufuddin M, Vella A, Montori VM, Dugani SB. Guidelines, position statements, and advisories for the primary prevention of type 2 diabetes, hypertension, and cardiovascular disease in rural populations: A systematic review protocol. PLoS One. 2023;18:e0288116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 3. | Zhang H, Zhou XD, Shapiro MD, Lip GYH, Tilg H, Valenti L, Somers VK, Byrne CD, Targher G, Yang W, Viveiros O, Opio CK, Mantzoros CS, Ryan JD, Kok KYY, Jumaev NA, Perera N, Robertson AG, Abu-Abeid A, Misra A, Wong YJ, Ruiz-Úcar E, Ospanov O, Kızılkaya MC, Luo F, Méndez-Sánchez N, Zuluaga M, Lonardo A, Al Momani H, Toro-Huamanchumo CJ, Adams L, Al-Busafi SA, Sharara AI, Chan WK, Abbas SI, Sookoian S, Treeprasertsuk S, Ocama P, Alswat K, Kong AP, Ataya K, Lim-Loo MC, Oviedo RJ, Szepietowski O, Fouad Y, Zhang H, Abdelbaki TN, Katsouras CS, Prasad A, Thaher O, Ali A, Molina GA, Sung KC, Chen QF, Lesmana CRA, Zheng MH. Global burden of metabolic diseases, 1990-2021. Metabolism. 2024;160:155999. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 225] [Article Influence: 112.5] [Reference Citation Analysis (0)] |
| 4. | Mauvais-Jarvis F, Bairey Merz N, Barnes PJ, Brinton RD, Carrero JJ, DeMeo DL, De Vries GJ, Epperson CN, Govindan R, Klein SL, Lonardo A, Maki PM, McCullough LD, Regitz-Zagrosek V, Regensteiner JG, Rubin JB, Sandberg K, Suzuki A. Sex and gender: modifiers of health, disease, and medicine. Lancet. 2020;396:565-582. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1682] [Cited by in RCA: 1482] [Article Influence: 247.0] [Reference Citation Analysis (3)] |
| 5. | Tramunt B, Smati S, Grandgeorge N, Lenfant F, Arnal JF, Montagner A, Gourdy P. Sex differences in metabolic regulation and diabetes susceptibility. Diabetologia. 2020;63:453-461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 264] [Cited by in RCA: 662] [Article Influence: 110.3] [Reference Citation Analysis (1)] |
| 6. | Palmisano BT, Zhu L, Eckel RH, Stafford JM. Sex differences in lipid and lipoprotein metabolism. Mol Metab. 2018;15:45-55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 454] [Cited by in RCA: 409] [Article Influence: 51.1] [Reference Citation Analysis (0)] |
| 7. | Wong RJ. Epidemiology of metabolic dysfunction-associated steatotic liver disease (MASLD) and alcohol-related liver disease (ALD). Metab Target Organ Damage. 2024;4:35. [DOI] [Full Text] |
| 8. | Lonardo A, Suzuki A. Sex differences in alcohol-related liver disease, viral hepatitis, metabolic dysfunction-associated steatotic liver disease, and hepatocellular carcinoma. Explor Dig Dis. 2025;4:1005101. [DOI] [Full Text] |
| 9. | Lonardo A, Weiskirchen R. Liver and obesity: a narrative review. Explor Med. 2025;6:1001334. [RCA] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (2)] |
| 10. | Cusi K, Abdelmalek MF, Apovian CM, Balapattabi K, Bannuru RR, Barb D, Bardsley JK, Beverly EA, Corbin KD, ElSayed NA, Isaacs S, Kanwal F, Pekas EJ, Richardson CR, Roden M, Sanyal AJ, Shubrook JH, Younossi ZM, Bajaj M. Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD) in People With Diabetes: The Need for Screening and Early Intervention. A Consensus Report of the American Diabetes Association. Diabetes Care. 2025;48:1057-1082. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 160] [Article Influence: 160.0] [Reference Citation Analysis (0)] |
| 11. | Shenoy A, Ahmed A, Kim D. Extrahepatic manifestation of metabolic dysfunction-associated steatotic liver disease. Metab Target Organ Damage. 2025;5:46. [RCA] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 12. | Jamalinia M, Zare F, Mantovani A, Targher G, Lonardo A. Metabolic dysfunction-associated steatotic liver disease and sex-specific risk of fatal and non-fatal cardiovascular events: A meta-analysis. Diabetes Obes Metab. 2025;27:5171-5181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 13. | Zhou BG, Jiang X, She Q, Ding YB. Association of MASLD with the risk of extrahepatic cancers: A systematic review and meta-analysis of 18 cohort studies. Eur J Clin Invest. 2024;54:e14276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 29] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 14. | Bril F, Elbert A. Metabolic dysfunction-associated steatotic liver disease and urinary system cancers: Mere coincidence or reason for concern? Metabolism. 2025;162:156066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 15. | Chung GE, Yu SJ, Park J, Kim YJ, Yoon JH, Han K, Cho EJ. Associations between changes in MASLD status and cancer development in young adults: A nationwide cohort study. Eur J Cancer. 2026;235:116228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 16. | Lonardo A, Jamalinia M, Weiskirchen R. Biological determinants and outcomes of sex discrepancies in MASLD. Metab Target Organ Damage. 2026;6:4. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 17. | Jamalinia M, Lonardo A, Weiskirchen R. Sex and Gender Differences in Liver Fibrosis: Pathomechanisms and Clinical Outcomes. Fibrosis. 2024;2:10006-10006. [DOI] [Full Text] |
| 18. | Garate-Carrillo A, Gonzalez J, Ceballos G, Ramirez-Sanchez I, Villarreal F. Sex related differences in the pathogenesis of organ fibrosis. Transl Res. 2020;222:41-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 56] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 19. | JBI. Welcome to the new edition of the JBI Manual for Evidence Synthesis. [cited March 19, 2026]. Available from: https://jbi-global-wiki.refined.site/space/MANUAL. |
| 20. | Joo SK, Kim W. Sex differences in metabolic dysfunction-associated steatotic liver disease: a narrative review. Ewha Med J. 2024;47:e17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 21. | Penmetsa R, Kapil S, VanWagner LB. Sex and gender differences in metabolic dysfunction-associated liver disease. Indian J Gastroenterol. 2025;45:192-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 22. | Jamalinia M, Saeian S, Nikkhoo N, Nazerian A, Lankarani KB. Sex and gender differences in MASLD: pathophysiological mechanisms, clinical implications, and future directions. Metab Target Organ Damage. 2025;5:60. [DOI] [Full Text] |
| 23. | Stefan N, Yki-Järvinen H, Neuschwander-Tetri BA. Metabolic dysfunction-associated steatotic liver disease: heterogeneous pathomechanisms and effectiveness of metabolism-based treatment. Lancet Diabetes Endocrinol. 2025;13:134-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 210] [Article Influence: 210.0] [Reference Citation Analysis (1)] |
| 24. | Varlamov O, Bethea CL, Roberts CT Jr. Sex-specific differences in lipid and glucose metabolism. Front Endocrinol (Lausanne). 2014;5:241. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 219] [Cited by in RCA: 264] [Article Influence: 24.0] [Reference Citation Analysis (1)] |
| 25. | Verhoog NJD, Africander D, Storbeck KH. Sex steroids, SHBG and type 2 diabetes in women: what do we really know? Diabetologia. 2026;69:837-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 26. | Bruns CM, Kemnitz JW. Sex hormones, insulin sensitivity, and diabetes mellitus. ILAR J. 2004;45:160-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 58] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 27. | Kim SW, Choi JW, Lee DS, Yun JW. Sex hormones regulate hepatic fetuin expression in male and female rats. Cell Physiol Biochem. 2014;34:554-564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 28. | Cazzagon N, Gambato M, Brunetto MR, Villa E, Burra P, Floreani A; Special Interest Group Gender in Hepatology of the Italian Association for the Study of the Liver (AISF). Sex Hormones and the Liver: Implications for Disease Progression and Hormonal Therapy. Liver Int. 2026;46:e70509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 29. | Milani I, Codini M, Guarisco G, Chinucci M, Gaita C, Leonetti F, Capoccia D. Hepatokines and MASLD: The GLP1-Ras-FGF21-Fetuin-A Crosstalk as a Therapeutic Target. Int J Mol Sci. 2024;25:10795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 25] [Reference Citation Analysis (1)] |
| 30. | Kemper EJ, Goossens GH, Blaak EE, Adriaens ME, Meex RCR. Fetuin B Is Related to Cytokine/Chemokine and Insulin Signaling in Adipose Tissue and Plasma in Humans. J Clin Endocrinol Metab. 2025;110:e3235-e3244. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 31. | Gustavsson C, Soga T, Wahlström E, Vesterlund M, Azimi A, Norstedt G, Tollet-Egnell P. Sex-dependent hepatic transcripts and metabolites in the development of glucose intolerance and insulin resistance in Zucker diabetic fatty rats. J Mol Endocrinol. 2011;47:129-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 32. | Yi M, Fashe M, Arakawa S, Moore R, Sueyoshi T, Negishi M. Nuclear receptor CAR-ERα signaling regulates the estrogen sulfotransferase gene in the liver. Sci Rep. 2020;10:5001. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 33. | Jiang M, He J, Kucera H, Gaikwad NW, Zhang B, Xu M, O'Doherty RM, Selcer KW, Xie W. Hepatic overexpression of steroid sulfatase ameliorates mouse models of obesity and type 2 diabetes through sex-specific mechanisms. J Biol Chem. 2014;289:8086-8097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 34. | Fashe M, Yi M, Sueyoshi T, Negishi M. Sex-specific expression mechanism of hepatic estrogen inactivating enzyme and transporters in diabetic women. Biochem Pharmacol. 2021;190:114662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 35. | Wang Y, Koh WP, Yuan JM, Pan A. Sex-specific association between fibroblast growth factor 21 and type 2 diabetes: a nested case-control study in Singapore Chinese men and women. Nutr Metab (Lond). 2017;14:63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 36. | Stangl TA, Wiepjes CM, Smit RAJ, van Hylckama Vlieg A, Lamb HJ, van der Velde JHPM, Winters-van Eekelen E, Boone SC, Brouwers MCGJ, Rosendaal FR, den Heijer M, Heijboer AC, de Mutsert R. Association Between Low Sex Hormone-Binding Globulin and Increased Risk of Type 2 Diabetes Is Mediated by Increased Visceral and Liver Fat: Results From Observational and Mendelian Randomization Analyses. Diabetes. 2024;73:1793-1804. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 37. | Simons PIHG, Valkenburg O, van de Waarenburg MPH, van Greevenbroek MMJ, Kooi ME, Jansen JFA, Schalkwijk CG, Stehouwer CDA, Brouwers MCGJ. Serum sex hormone-binding globulin is a mediator of the association between intrahepatic lipid content and type 2 diabetes: the Maastricht Study. Diabetologia. 2023;66:213-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 38. | Jaruvongvanich V, Sanguankeo A, Riangwiwat T, Upala S. Testosterone, Sex Hormone-Binding Globulin and Nonalcoholic Fatty Liver Disease: a Systematic Review and Meta-Analysis. Ann Hepatol. 2017;16:382-394. [PubMed] [DOI] [Full Text] |
| 39. | Xia C, Zhang X, Cao T, Wang J, Li C, Yue L, Niu K, Shen Y, Ma G, Chen F. Hepatic Transcriptome Analysis Revealing the Molecular Pathogenesis of Type 2 Diabetes Mellitus in Zucker Diabetic Fatty Rats. Front Endocrinol (Lausanne). 2020;11:565858. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 40. | Dungubat E, Kusano H, Mori I, Tawara H, Sutoh M, Ohkura N, Takanashi M, Kuroda M, Harada N, Udo E, Souda M, Furusato B, Fukusato T, Takahashi Y. Age-dependent sex difference of non-alcoholic fatty liver disease in TSOD and db/db mice. PLoS One. 2022;17:e0278580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (2)] |
| 41. | Cherubini A, Rosso C, Della Torre S. Sex-specific effects of PNPLA3 I148M. Liver Int. 2025;45:e16088. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 14] [Article Influence: 14.0] [Reference Citation Analysis (2)] |
| 42. | Weiskirchen R, Lonardo A. PNPLA3 as a driver of steatotic liver disease: navigating from pathobiology to the clinics via epidemiology. J Transl Genet Genom. 2024;8:355-77. [RCA] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 43. | Hochmuth L, Körner C, Ott F, Volke D, Cokan KB, Juvan P, Brosch M, Hofmann U, Hoffmann R, Rozman D, Berg T, Matz-Soja M. Sex-dependent dynamics of metabolism in primary mouse hepatocytes. Arch Toxicol. 2021;95:3001-3013. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 44. | Zhang H, Huang T, Jin X, Liu S, Yang Y, Liu L, Jiang X, Zhang R, Ye H, Qi X, Song T, Jin C, Feng B, Che L, Xu S, Lin Y, Fang Z, Luo T, Zhuo Y, Wu D, Hua L. Liver ERα mediates sex differences in metabolic pattern changes in response to time-restricted feeding. Life Metab. 2025;4:loaf011. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 45. | Fan Y, Wolford BN, Lu H, Liang W, Sun J, Zhou W, Rom O, Mahajan A, Surakka I, Graham SE, Liu Z, Kim H, Ramdas S, Fritsche LG, Nielsen JB, Gabrielsen ME, Hveem K, Yang D, Song J, Garcia-Barrio MT, Zhang J, Liu W, Zhang K, Willer CJ, Chen YE. Type 2 diabetes sex-specific effects associated with E167K coding variant in TM6SF2. iScience. 2021;24:103196. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 46. | Zhou P, Hummel AD, Pywell CM, Dong XC, Duffield GE. High fat diet rescues disturbances to metabolic homeostasis and survival in the Id2 null mouse in a sex-specific manner. Biochem Biophys Res Commun. 2014;451:374-381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 47. | Ching MEA, Hoyeck MP, Basu L, Merhi R, Poleo-Giordani E, van Zyl E, Crawley AM, Bruin JE. CYP1A1/1A2 enzymes mediate glucose homeostasis and insulin secretion in mice in a sex-specific manner. Am J Physiol Endocrinol Metab. 2025;328:E885-E898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 48. | Nejat S, Menikdiwela KR, Iskander O, Walker H, Scoggin S, Yosofvand M, Moussa H, Zu Y, Dufour JM, Chakroborty A, Vandanmagsar B, Thornalley PJ, Kalupahana NS, Dhanasekara CS, Dehbi M, Moustaid-Moussa N. DNAJB3 Deficiency in Mice, Sex-Dependently, Exacerbates High-Fat Diet-Induced Metabolic Dysfunctions. Obesity (Silver Spring). 2026;34:138-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 49. | Milani I, Parrotta ME, Colangeli L, Chinucci M, Palleschi S, Rossi B, Sbraccia P, Mantovani A, Leonetti F, Guglielmi V, Capoccia D. Sex Differences in MASLD After Age 50: Presentation, Diagnosis, and Clinical Implications. Biomedicines. 2025;13:2292. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 50. | Kaikaew K, Grefhorst A, Visser JA. Sex Differences in Brown Adipose Tissue Function: Sex Hormones, Glucocorticoids, and Their Crosstalk. Front Endocrinol (Lausanne). 2021;12:652444. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 74] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 51. | Malpique R, Gallego-Escuredo JM, Sebastiani G, Villarroya J, López-Bermejo A, de Zegher F, Villarroya F, Ibáñez L. Brown adipose tissue in prepubertal children: associations with sex, birthweight, and metabolic profile. Int J Obes (Lond). 2019;43:384-391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 52. | Gandhi A, Tang R, Seo Y, Bhargava A. Organ-Specific Glucose Uptake: Does Sex Matter? Cells. 2022;11:2217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 53. | Shin YK, Hsieh YS, Han AY, Kwon S, Kang P, Seol GH. Sex-specific susceptibility to type 2 diabetes mellitus and preventive effect of linalyl acetate. Life Sci. 2020;260:118432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 54. | Post A, Dam WA, Sokooti S, Groothof D, Gloerich J, van Gool AJ, Kremer D, Gansevoort RT, van den Born J, Kema IP, Franssen CFM, Dullaart RPF, Bakker SJL. Circulating FGF21 Concentration, Fasting Plasma Glucose, and the Risk of Type 2 Diabetes: Results From the PREVEND Study. J Clin Endocrinol Metab. 2023;108:1387-1393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 23] [Reference Citation Analysis (0)] |
| 55. | Jia F, Fei SF, Tong DB, Xue C, Li JJ. Sex difference in circulating PCSK9 and its clinical implications. Front Pharmacol. 2022;13:953845. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 56. | Yeh KH, Hsu LA, Tran NY, Wu S, Ko YL. High circulating PCSK9 concentration is associated with increased and sex-specific risks of metabolic disease, diabetes mellitus, MASLD, and long-term mortality in a Taiwanese population. Lipids Health Dis. 2025;25:31. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 57. | Yan T, Zhang X, Wong T, Cheung R, Nguyen MH. Sex Differences in Adverse Liver and Nonliver Outcomes in Steatotic Liver Disease. JAMA Netw Open. 2024;7:e2448946. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 58. | Abushamat LA, Shah PA, Eckel RH, Harrison SA, Barb D. The Emerging Role of Glucagon-Like Peptide-1 Receptor Agonists for the Treatment of Metabolic Dysfunction-Associated Steatohepatitis. Clin Gastroenterol Hepatol. 2024;22:1565-1574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 67] [Article Influence: 33.5] [Reference Citation Analysis (0)] |
| 59. | Narisada A, Shibata E, Hasegawa T, Masamura N, Taneda C, Suzuki K. Sex differences in the association between fatty liver and type 2 diabetes incidence in non-obese Japanese: A retrospective cohort study. J Diabetes Investig. 2021;12:1480-1489. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 60. | Yoshimoto M, Sakuma Y, Ogino J, Iwai R, Watanabe S, Inoue T, Takahashi H, Suzuki Y, Kinoshita D, Takemura K, Takahashi H, Shimura H, Babazono T, Yoshida S, Hashimoto N. Sex differences in predictive factors for onset of type 2 diabetes in Japanese individuals: A 15-year follow-up study. J Diabetes Investig. 2023;14:37-47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 61. | Masango B, Goedecke JH, Ramsay M, Storbeck KH, Micklesfield LK, Chikowore T. Postprandial glucose variability and clusters of sex hormones, liver enzymes, and cardiometabolic factors in a South African cohort of African ancestry. BMJ Open Diabetes Res Care. 2024;12:e003927. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 62. | Pina AF, Meneses MJ, Carli F, Ribeiro RT, Gardete-Correia L, Boavida JM, Raposo JF, Gastaldelli A, Macedo MP. Lipidomic profiling unveils sex differences in diabetes risk: Implications for precision medicine. Eur J Clin Invest. 2026;56:e70137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 63. | Crace CJ, Swenne I, Milner RD. Long-term follow-up after early protein-calorie malnutrition in young rats: sex difference in glucose tolerance and serum insulin levels. Metabolism. 1989;38:933-938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 64. | Hennes MM, McCune SA, Shrago E, Kissebah AH. Synergistic effects of male sex and obesity on hepatic insulin dynamics in SHR/Mcc-cp rat. Diabetes. 1990;39:789-795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 65. | Muthusamy T, Murugesan P, Balasubramanian K. Sex steroids deficiency impairs glucose transporter 4 expression and its translocation through defective Akt phosphorylation in target tissues of adult male rat. Metabolism. 2009;58:1581-1592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 61] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 66. | Gao J, He J, Shi X, Stefanovic-Racic M, Xu M, O'Doherty RM, Garcia-Ocana A, Xie W. Sex-specific effect of estrogen sulfotransferase on mouse models of type 2 diabetes. Diabetes. 2012;61:1543-1551. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 64] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 67. | Mathew D, Zhou P, Pywell CM, van der Veen DR, Shao J, Xi Y, Bonar NA, Hummel AD, Chapman S, Leevy WM, Duffield GE. Ablation of the ID2 gene results in altered circadian feeding behavior, and sex-specific enhancement of insulin sensitivity and elevated glucose uptake in skeletal muscle and brown adipose tissue. PLoS One. 2013;8:e73064. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 25] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 68. | Yokomizo H, Inoguchi T, Sonoda N, Sakaki Y, Maeda Y, Inoue T, Hirata E, Takei R, Ikeda N, Fujii M, Fukuda K, Sasaki H, Takayanagi R. Maternal high-fat diet induces insulin resistance and deterioration of pancreatic β-cell function in adult offspring with sex differences in mice. Am J Physiol Endocrinol Metab. 2014;306:E1163-E1175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 101] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 69. | Kruse MS, Vega MC, Rey M, Coirini H. Sex differences in LXR expression in normal offspring and in rats born to diabetic dams. J Endocrinol. 2014;222:53-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 70. | Babelova A, Burckhardt BC, Salinas-Riester G, Pommerenke C, Burckhardt G, Henjakovic M. Next generation sequencing of sex-specific genes in the livers of obese ZSF1 rats. Genomics. 2015;106:204-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 71. | Wells A, Barrington WT, Dearth S, May A, Threadgill DW, Campagna SR, Voy BH. Tissue Level Diet and Sex-by-Diet Interactions Reveal Unique Metabolite and Clustering Profiles Using Untargeted Liquid Chromatography-Mass Spectrometry on Adipose, Skeletal Muscle, and Liver Tissue in C57BL6/J Mice. J Proteome Res. 2018;17:1077-1090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 72. | Dong Q, Kuefner MS, Deng X, Bridges D, Park EA, Elam MB, Raghow R. Sex-specific differences in hepatic steatosis in obese spontaneously hypertensive (SHROB) rats. Biol Sex Differ. 2018;9:40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 73. | Zhang X, Xu H, Ning J, Ji H, Yan J, Zheng Y, Xu Q, Li C, Zhao L, Zheng H, Gao H. Sex-Specific Metabolic Changes in Peripheral Organs of Diabetic Mice. J Proteome Res. 2020;19:3011-3021. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 74. | Iena FM, Jul JB, Vegger JB, Lodberg A, Thomsen JS, Brüel A, Lebeck J. Sex-Specific Effect of High-Fat Diet on Glycerol Metabolism in Murine Adipose Tissue and Liver. Front Endocrinol (Lausanne). 2020;11:577650. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 75. | Kornicka-Garbowska K, Bourebaba L, Röcken M, Marycz K. Sex Hormone Binding Globulin (SHBG) Mitigates ER Stress in Hepatocytes In Vitro and Ex Vivo. Cells. 2021;10:755. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 76. | Chaix A, Deota S, Bhardwaj R, Lin T, Panda S. Sex- and age-dependent outcomes of 9-hour time-restricted feeding of a Western high-fat high-sucrose diet in C57BL/6J mice. Cell Rep. 2021;36:109543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 84] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 77. | Glavas MM, Lee AY, Miao I, Yang F, Mojibian M, O'Dwyer SM, Kieffer TJ. Developmental Timing of High-Fat Diet Exposure Impacts Glucose Homeostasis in Mice in a Sex-Specific Manner. Diabetes. 2021;70:2771-2784. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 78. | Kaare M, Mikheim K, Lilleväli K, Kilk K, Jagomäe T, Leidmaa E, Piirsalu M, Porosk R, Singh K, Reimets R, Taalberg E, Schäfer MKE, Plaas M, Vasar E, Philips MA. High-Fat Diet Induces Pre-Diabetes and Distinct Sex-Specific Metabolic Alterations in Negr1-Deficient Mice. Biomedicines. 2021;9:1148. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 79. | Gao L, Wang X, Guo L, Zhang W, Wang G, Han S, Zhang Y. Sex differences in diabetes-induced hepatic and renal damage. Exp Ther Med. 2024;27:148. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 80. | Makarova E, Kazantseva A, Dubinina A, Denisova E, Jakovleva T, Balybina N, Bgatova N, Baranov K, Bazhan N. Fibroblast Growth Factor 21 (FGF21) Administration Sex-Specifically Affects Blood Insulin Levels and Liver Steatosis in Obese A(y) Mice. Cells. 2021;10:3440. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 81. | Kokabiyan Z, Yaghmaei P, Jameie SB, Hajebrahimi Z. Effect of eugenol on lipid profile, oxidative stress, sex hormone, liver injury, ovarian failure, and expression of COX-2 and PPAR-α genes in a rat model of diabetes. Mol Biol Rep. 2023;50:3669-3679. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 82. | Ivić V, Zjalić M, Blažetić S, Fenrich M, Labak I, Scitovski R, Szűcs KF, Ducza E, Tábi T, Bagamery F, Szökő É, Vuković R, Rončević A, Mandić D, Debeljak Ž, Berecki M, Balog M, Seres-Bokor A, Sztojkov-Ivanov A, Hajagos-Tóth J, Gajović S, Imširović A, Bakula M, Mahiiovych S, Gaspar R, Vari SG, Heffer M. Elderly rats fed with a high-fat high-sucrose diet developed sex-dependent metabolic syndrome regardless of long-term metformin and liraglutide treatment. Front Endocrinol (Lausanne). 2023;14:1181064. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 83. | Buday B, Pach PF, Literati-Nagy B, Vitai M, Kovacs G, Vecsei Z, Koranyi L, Lengyel C. Sex influenced association of directly measured insulin sensitivity and serum transaminase levels: Why alanine aminotransferase only predicts cardiovascular risk in men? Cardiovasc Diabetol. 2015;14:55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 84. | Jesuthasan A, Zhyzhneuskaya S, Peters C, Barnes AC, Hollingsworth KG, Sattar N, Lean MEJ, Taylor R, Al-Mrabeh AH. Sex differences in intraorgan fat levels and hepatic lipid metabolism: implications for cardiovascular health and remission of type 2 diabetes after dietary weight loss. Diabetologia. 2022;65:226-233. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (4)] |
| 85. | Bae J, Kim SH, Won YJ, Kim YM. Age- and Sex-Specific Differences in Distribution of Cardiometabolic Diseases and Associations of Hand-Grip Strength Indices with Type 2 Diabetes in Korean Adults. Metab Syndr Relat Disord. 2022;20:199-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 86. | Drakul D, Sokolović D, Radanović M, Dukić N, Kunarac M, Ćurčić B, Pavlović D, Marić R. Sex Differences in the Use of Cardiovascular Drugs: A Survey of Patients in a Single Center. Acta Med Acad. 2021;50:351-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 87. | Petersen KF, Dufour S, Li F, Rothman DL, Shulman GI. Ethnic and sex differences in hepatic lipid content and related cardiometabolic parameters in lean individuals. JCI Insight. 2022;7:e157906. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 88. | Zhang H, Chen C, Zhang X, Wang Y, Wan H, Chen Y, Zhang W, Xia F, Lu Y, Wang N. Association between sex hormone-binding globulin and kidney function in men: results from the SPECT-China study. Chin Med J (Engl). 2022;135:2083-2088. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 89. | Fresneda S, Abbate M, Busquets-Cortés C, López-González A, Fuster-Parra P, Bennasar-Veny M, Yáñez AM. Sex and age differences in the association of fatty liver index-defined non-alcoholic fatty liver disease with cardiometabolic risk factors: a cross-sectional study. Biol Sex Differ. 2022;13:64. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 33] [Reference Citation Analysis (0)] |
| 90. | Kim RG, Khalili M. Undiagnosed abnormal alanine transaminase levels in vulnerable populations: Impact of sex, race/ethnicity, and body mass. Obes Sci Pract. 2023;9:190-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 91. | Piccione A, Bonsangue M, Barone M, Vigneri E, Mineo MI, Tomasello L, Maniscalco L, Pantò F, Arnaldi G, Guarnotta V. Sex specific responses to oral vs. subcutaneous semaglutide in type 2 diabetes mellitus: A 12-month real-world study. Nutr Metab Cardiovasc Dis. 2026;36:104462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 92. | Gu D, Lu Y, Xu B, Tang X. Sex-Specific Contribution of Cardiometabolic Index in Predicting Metabolic Dysfunction-Associated Fatty Liver Disease: Insights from a General Population. Diabetes Metab Syndr Obes. 2023;16:3871-3883. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 93. | Lonardo A, Zheng M, Eslam M. MASLD vs. MAFLD. A narrative review. Explor Dig Dis. 2025;4:100586. [DOI] [Full Text] |
| 94. | Pugeat M, Nader N, Hogeveen K, Raverot G, Déchaud H, Grenot C. Sex hormone-binding globulin gene expression in the liver: drugs and the metabolic syndrome. Mol Cell Endocrinol. 2010;316:53-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 97] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 95. | Ye J, Yao Z, Tan A, Gao Y, Chen Y, Lin X, He R, Tang R, Hu Y, Zhang H, Yang X, Wang Q, Jiang Y, Mo Z. Low Serum Sex Hormone-Binding Globulin Associated with Insulin Resistance in Men with Nonalcoholic Fatty Liver Disease. Horm Metab Res. 2017;49:359-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 96. | Goto A, Morita A, Goto M, Sasaki S, Miyachi M, Aiba N, Terauchi Y, Noda M, Watanabe S; Saku Cohort Study Group. Associations of sex hormone-binding globulin and testosterone with diabetes among men and women (the Saku Diabetes study): a case control study. Cardiovasc Diabetol. 2012;11:130. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 34] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 97. | Wang Y. Definition, Prevalence, and Risk Factors of Low Sex Hormone-Binding Globulin in US Adults. J Clin Endocrinol Metab. 2021;106:e3946-e3956. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 98. | Jamalinia M, Lonardo A, Weiskirchen R. Burden of Liver Disease Among Individuals With Turner Syndrome and Klinefelter Syndrome: A Comprehensive Perspective. Chron Dis Transl Med. 2026;12:39-48. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 99. | Thomsen HH, Møller HJ, Trolle C, Groth KA, Skakkebæk A, Bojesen A, Høst C, Gravholt CH. The macrophage low-grade inflammation marker sCD163 is modulated by exogenous sex steroids. Endocr Connect. 2013;2:216-224. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 100. | Plevriti A, Lamprou M, Mourkogianni E, Skoulas N, Giannakopoulou M, Sajib MS, Wang Z, Mattheolabakis G, Chatzigeorgiou A, Marazioti A, Mikelis CM. The Role of Soluble CD163 (sCD163) in Human Physiology and Pathophysiology. Cells. 2024;13:1679. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 31] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 101. | Naeem M, Bibi S, Bülow R, Bahls M, Schipf S, Töpfer P, Friedrich N, Rathmann W, Aghdassi A, Nauck M, Dörr M, Völzke H, Markus MR, Ittermann T. Sex-specific associations of anthropometric markers with prediabetes in the general population. Nutr Metab Cardiovasc Dis. 2026;36:104515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 102. | de Ritter R, Sep SJS, van Greevenbroek MMJ, Kusters YHAM, Vos RC, Bots ML, Kooi ME, Dagnelie PC, Eussen SJPM, Schram MT, Koster A, Brouwers MCG, van der Sangen NMR, Peters SAE, van der Kallen CJH, Stehouwer CDA. Sex differences in body composition in people with prediabetes and type 2 diabetes as compared with people with normal glucose metabolism: the Maastricht Study. Diabetologia. 2023;66:861-872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 103. | Li S, Zou T, Chen J, Li J, You J. Fibroblast growth factor 21: An emerging pleiotropic regulator of lipid metabolism and the metabolic network. Genes Dis. 2024;11:101064. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 104. | Di Bonito P, Sanguigno E, Di Fraia T, Forziato C, Boccia G, Saitta F, Iardino MR, Capaldo B. Association of elevated serum alanine aminotransferase with metabolic factors in obese children: sex-related analysis. Metabolism. 2009;58:368-372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 40] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 105. | Suzuki A, Binks M, Sha R, Wachholtz A, Eisenson H, Diehl AM. Serum aminotransferase changes with significant weight loss: sex and age effects. Metabolism. 2010;59:177-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 106. | Wang N, Zhai H, Zhu C, Li Q, Han B, Chen Y, Zhu C, Chen Y, Xia F, Lin D, Lu Y. Combined Association of Vitamin D and Sex Hormone Binding Globulin With Nonalcoholic Fatty Liver Disease in Men and Postmenopausal Women: A Cross-Sectional Study. Medicine (Baltimore). 2016;95:e2621. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 107. | Kastberg SE, Lund HS, De Lucia-Rolfe E, Kaduka LU, Boit MK, Corpeleijn E, Friis H, Bernard S, Paquette M, Baass A, Rasmussen JJ, Christensen DL. Hepatic steatosis is associated with anthropometry, cardio-metabolic disease risk, sex, age and urbanisation, but not with ethnicity in adult Kenyans. Trop Med Int Health. 2022;27:49-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 108. | Niedermayer F, Su Y, von Krüchten R, Thorand B, Peters A, Rathmann W, Roden M, Schlett CL, Bamberg F, Nattenmüller J, Rospleszcz S. Trajectories of glycaemic traits exhibit sex-specific associations with hepatic iron and fat content: Results from the KORA-MRI study. Liver Int. 2023;43:2153-2166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 109. | Ohkuma T, Peters SAE, Woodward M. Sex differences in the association between diabetes and cancer: a systematic review and meta-analysis of 121 cohorts including 20 million individuals and one million events. Diabetologia. 2018;61:2140-2154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 131] [Article Influence: 16.4] [Reference Citation Analysis (4)] |
| 110. | BardalesZuta VH, ReyesAroca S, VeronaEscurra MLS, Moya-Carranza HG, Fernández-Rodríguez LJ. Nonalcoholic Fatty Liver Disease and Male Sex are Risk Factors for Colorectal Adenoma: a Retrospective Analysis. Acta Gastroenterol Latinoam. 2024;54:239-246. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 111. | Allen AM, Hicks SB, Mara KC, Larson JJ, Therneau TM. The risk of incident extrahepatic cancers is higher in non-alcoholic fatty liver disease than obesity - A longitudinal cohort study. J Hepatol. 2019;71:1229-1236. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 280] [Cited by in RCA: 260] [Article Influence: 37.1] [Reference Citation Analysis (1)] |
| 112. | Wu CT, Targher G, Byrne CD, Younossi ZM, Armstrong MJ, Luu HN, Lesmana CRA, Rustgi VK, Thomas JA, Okabayashi K, Lee JWJ, El-Serag HB, Kim W, Zhang HJ, Lonardo A, Souza M, George ES, Yang W, Yilmaz Y, Papatheodoridis G, Sookoian S, Romero-Gómez M, Sebastiani G, Schattenberg JM, El-Kassas M, Chan WK, Ocama P, Lupșor-Platon M, Perera N, Alswat K, Méndez-Sánchez N, Wong VW, Chai J, Tilg H, Sung JJ, Zheng MH. Colorectal cancer in metabolic dysfunction-associated steatotic liver disease: an international Delphi consensus statement. Gut. 2026;gutjnl-2025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 113. | Burton A, Tataru D, Driver RJ, Bird TG, Huws D, Wallace D, Cross TJS, Rowe IA, Alexander G, Marshall A; HCC-UK/BASL/NCRAS Partnership Steering Group. Primary liver cancer in the UK: Incidence, incidence-based mortality, and survival by subtype, sex, and nation. JHEP Rep. 2021;3:100232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 114. | Yi SW, Choi JS, Yi JJ, Lee YH, Han KJ. Risk factors for hepatocellular carcinoma by age, sex, and liver disorder status: A prospective cohort study in Korea. Cancer. 2018;124:2748-2757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 72] [Article Influence: 9.0] [Reference Citation Analysis (4)] |
| 115. | Petrick JL, Freedman ND, Demuth J, Yang B, Van Den Eeden SK, Engel LS, McGlynn KA. Obesity, diabetes, serum glucose, and risk of primary liver cancer by birth cohort, race/ethnicity, and sex: Multiphasic health checkup study. Cancer Epidemiol. 2016;42:140-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 116. | Braunwarth E, Rumpf B, Primavesi F, Pereyra D, Hochleitner M, Göbel G, Gasteiger S, Gehwolf P, Öfner D, Starlinger P, Stättner S. Sex differences in disease presentation, surgical and oncological outcome of liver resection for primary and metastatic liver tumors-A retrospective multicenter study. PLoS One. 2020;15:e0243539. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 117. | Chen HF, Chen P, Li CY. Risk of malignant neoplasms of liver and biliary tract in diabetic patients with different age and sex stratifications. Hepatology. 2010;52:155-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 78] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 118. | Jamalinia M, Targher G, Lonardo A, Heydari ST, Bagheri Lankarani K. Application of machine learning for sex-specific risk prediction of coronary artery calcification. Nutr Metab Cardiovasc Dis. 2026;36:104579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 119. | Matsubayashi Y, Fujihara K, Yamada-Harada M, Mitsuma Y, Sato T, Yaguchi Y, Osawa T, Yamamoto M, Kitazawa M, Yamada T, Kodama S, Sone H. Impact of metabolic syndrome and metabolic dysfunction-associated fatty liver disease on cardiovascular risk by the presence or absence of type 2 diabetes and according to sex. Cardiovasc Diabetol. 2022;21:90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 120. | Abbaszadeh M, Hosseinpanah F, Tohidi M, Karimpour Reyhan S, Mahdavi M, Valizadeh M. Sex-Specific Impact of Metabolic Dysfunction-Associated Fatty Liver Disease on Incident Cardiovascular Diseases and Mortality. Endocrinol Diabetes Metab. 2025;8:e70035. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 121. | Desai R, Alvi AT, Vasavada A, Pulakurthi YS, Patel B, Mohammed AS, Doshi S, Ogbu I. Sex and racial disparities in non-alcoholic fatty liver disease-related cardiovascular events: National inpatient sample analysis (2019). World J Cardiol. 2024;16:137-148. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 122. | Lonardo A, Ndrepepa G. Concise review: gamma-glutamyl transferase - evolution from an indiscriminate liver test to a biomarker of cardiometabolic risk. Metab Target Organ Damage. 2022;2:17. [RCA] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 123. | Jung DH, Park B, Ryu HE, Lee YJ. Sex-specific associations of γ-glutamyltransferase to HDL-cholesterol ratio and the incident risk of cardiovascular disease: three Korean longitudinal cohorts from different regions. Front Endocrinol (Lausanne). 2023;14:1231502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 124. | Ren R, Zheng Y. Sex differences in cardiovascular and all-cause mortality in nonalcoholic fatty liver disease in the US population. Nutr Metab Cardiovasc Dis. 2023;33:1349-1357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 125. | Chan Z, Chooi YC, Ding C, Choo J, Sadananthan SA, Michael N, Velan SS, Leow MK, Magkos F. Sex Differences in Glucose and Fatty Acid Metabolism in Asians Who Are Nonobese. J Clin Endocrinol Metab. 2019;104:127-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 126. | Goyal P, Paul T, Almarzooq ZI, Peterson JC, Krishnan U, Swaminathan RV, Feldman DN, Wells MT, Karas MG, Sobol I, Maurer MS, Horn EM, Kim LK. Sex- and Race-Related Differences in Characteristics and Outcomes of Hospitalizations for Heart Failure With Preserved Ejection Fraction. J Am Heart Assoc. 2017;6:e003330. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 45] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 127. | Park S, Kim GU, Kim H. Physical Comorbidity According to Diagnoses and Sex among Psychiatric Inpatients in South Korea. Int J Environ Res Public Health. 2021;18:4187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 128. | Kaser S, Radlinger B, Blasinger J, Koellenberger N, Streitberger V, Kopp L, Bifano E, Aziz F, Sourij H, Goebel G, Klocker J. Non-Traumatic Lower-Limb Amputations: Outcome, Sex-Differences, Comorbidity Patterns and Temporal Trends from 2006 to 2022. J Clin Med. 2025;14:4030. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 129. | Gulati R, Moylan CA, Wilder J, Wegermann K. Racial and ethnic disparities in metabolic dysfunction-associated steatotic liver disease. Metab Target Organ Damage. 2024;4. [DOI] [Full Text] |
| 130. | Fodor Duric L, Čolak Romić Z, Pavičić D, Čurić J, Belčić V, Rajković I, Rajković I, Muslim J, Basic Jukic N, Vujicic B, Gulin T, Gulin M, Grgurević M, Oberiter Korbar A. The Role of Sex Hormone-Binding Globulin (SHBG) as a Marker of Metabolic Dysfunction-Associated Steatotic Liver Disease, with an Extended Analysis in Both Men and Women. J Clin Med. 2026;15:1301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 131. | Henney AE, Wilson J, Riley DR, Alam U, Cuthbertson DJ. Effect of Hormone Replacement Therapy on Liver and Cardiometabolic Outcomes in Peri-Menopausal MASLD. Liver Int. 2026;46:e70562. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 132. | Jancova P, Ismail K, Vistejnova L. Relationship between MASLD and women's health: A review. Womens Health (Lond). 2025;21:17455057251376883. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 133. | Miao MY, Han WW, Lyu JQ, Liu ZY, Jiang W, Zhang Z, Zhu Z, Qin LQ, Chen GC. Female reproductive factors and metabolic dysfunction-associated steatotic liver disease: an integrated analysis of population cohort, liver imaging, and genetic data. Am J Obstet Gynecol. 2025;233:469.e1-469.e54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 134. | Dong J, Dennis KMJH, Venkatakrishnan R, Hodson L, Tomlinson JW. The Impact of Estrogen Deficiency on Liver Metabolism: Implications for Hormone Replacement Therapy. Endocr Rev. 2025;46:790-809. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 26] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 135. | Weiskirchen R, Lonardo A. Treating obesity with GLP-1 RAs: does sex matter? A commentary on the meta-analysis by Yang et al. (J Diabetes 2025;17(3):e70063). Explor Endocr Metab Dis. 2026;3:101458. [DOI] [Full Text] |