Published online May 15, 2026. doi: 10.4239/wjd.v17.i5.117132
Revised: January 20, 2026
Accepted: February 24, 2026
Published online: May 15, 2026
Processing time: 163 Days and 0.9 Hours
The loss of enteric neurons may contribute to the altered gastric emptying ob
To evaluate the impact of GES on enteric neurons in the antral myenteric plexus of diabetic beagle canines.
Adult female beagles were used to establish a diabetes mellitus (DM) (n = 6) model via intravenous injection of streptozotocin, with healthy canines as controls (n = 3). All canines were implanted with stimulation devices and received a real GES session (0.33 millisecond, 5 mA, 15 Hz, 0.2 second on, 3 seconds off) and a sham GES session (SGES) (stimulators off) of 4 weeks each in a randomized crossover design, separated by a 3-week washout period. Full-thickness antral biopsies were obtained after each session, and pancreatic biopsies were conducted at the end of the study. Hematoxylin-eosin staining was employed to measure histopathological changes in the pancreatic islets. Immunofluorescence staining of antral full-thickness sections was employed to evaluate the alterations in choline acetyltransferase (ChAT), vasoactive intestinal peptide, neuronal nitric oxide synthase (nNOS), or calretinin immunoreactive (IR) positive neurons in relation to the anti-human neuronal protein HuC/HuD. Based on different stimulations, the results were analyzed in DM + GES, DM + SGES, control + GES, and control + SGES subgroups.
Diabetic canines exhibited a significant reduction in the number and area of pancreatic islets compared with controls. The proportions of ChAT-IR and nNOS-IR neurons in the DM + SGES subgroup were significantly lower than those in the control + SGES subgroup (67.5% ± 1.8% vs 73.3% ± 1.8%, P = 0.003; 27.3% ± 1.4% vs 32.1% ± 0.7%, P = 0.001), with an upregulation of vasoactive intestinal peptide-IR neurons (51.9% ± 0.8% vs 46.8% ± 1.2%, P < 0.001). Following the 4-week GES intervention, the proportions of ChAT-IR and nNOS-IR neurons were significantly higher (73.3% ± 1.7% vs 67.5% ± 1.8%, P = 0.002; 32.1% ± 1.7% vs 27.3% ± 1.4%, P = 0.002) compared to SGES in diabetic canines. A similar increase was also observed in the proportion of nNOS-IR neurons between the GES and SGES subgroups in controls (37.5% ± 1.3% vs 32.1% ± 0.7%, P = 0.036).
Imbalanced regulation of excitatory and inhibitory neurons in the enteric nervous system occurred in streptozotocin-induced diabetic beagles, and GES improved the survival of cholinergic and nitrergic neurons in diabetic canines.
Core Tip: Experiments in small animal models have confirmed that damage to the enteric nervous system severely impairs physiological movement in the gastrointestinal system in diabetic gastroparesis. We found that the morphologic changes in gastric excitatory and inhibitory neurons are also present in streptozotocin-induced diabetic beagle canines, despite the absence of obvious manifestations of gastroparesis. High-frequency gastric electrical stimulation ameliorates the enteric nervous system in diabetic canines by upregulating choline acetyltransferase and neuronal nitric oxide synthase-immunoreactive positive neurons, these neuromodulatory effects on enteric neuronal plasticity could restore slow wave rhythms, enhance gastric emptying, and improve gastric accommodation.
- Citation: Tang P, Wu X, Xi JR, Fu MY, Xue XW, Jiang Y, Zhao Y, Yang HE, Fei GJ, Li XQ, Hu CH, Fang XC. Gastric electrical stimulation ameliorates antral myenteric neurons in streptozotocin-induced diabetic beagle canines. World J Diabetes 2026; 17(5): 117132
- URL: https://www.wjgnet.com/1948-9358/full/v17/i5/117132.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i5.117132
Diabetic gastroparesis (DGP) is a complication of diabetes mellitus (DM) characterized by delayed gastric emptying (GE) without mechanical pyloric obstruction. In a community-based study of individuals with diabetes, the 10-year incidence of gastroparesis in type 1 and type 2 diabetes were 5% and 1%, respectively[1]. DGP is manifested with symptoms such as early satiety, postprandial fullness, belching, nausea, vomiting, abdominal pain, and weight loss. These symptoms can result in dysglycemia, frequent hospitalizations, and a poor quality of life[2]. The pathophysiology of DGP is complex and appears to be multifactorial, such as hyperglycemia, vagal dysfunction, loss of neuronal nitric oxide synthase (nNOS) expression in the myenteric plexus (MP), abnormalities in the interstitial cells of Cajal (ICC) network, and oxidative stress[3,4]. Nevertheless, the exact pathophysiology of diabetic gastroenteropathy remains unknown. Dietary modifications and medications are the first-line treatment for gastroparesis, but approximately 30% of patients do not achieve symptom relief with conservative treatment[5].
Gastric electrical stimulation (GES) uses high-frequency, low-energy stimuli to improve symptoms of nausea, vomiting, and hyperglycemia in patients with gastroparesis and has a response rate of 50%-75%[6,7]. A prospective single-center study of 151 patients with refractory gastroparesis demonstrated that GES significantly improved symptoms in 75% of patients with 43% having at least moderate improvement[7]. Clinical improvement was observed in both diabetic and idiopathic patients, with a higher response rate in diabetics than in non-diabetic patients (85% vs 68%)[7]. However, the mechanisms by which GES improves the gastric motility and hyperglycemia, especially in DGP, need to be further elucidated.
Diabetic enteric neuropathy is associated with alterations in the size and number of excitatory and inhibitory enteric neurons, as well as neurodegenerative changes in the enteric nervous system (ENS)[8]. Excitatory enteric neurons, such as cholinergic neurons, primarily express choline acetyltransferase (ChAT) to augment slow waves and increase the amplitude of peristaltic waves. Du et al[9] found enteric neuropathy [characterized by decreased ChAT-immunoreactive (IR) and nNOS-IR positive neurons] in the proximal and distal colon of 8-week and 12-week diabetic rats compared with the control group. It has been reported that ChAT-IR positive neurons in the myenteric layer of the gastric antrum were also reduced in streptozotocin (STZ)-induced diabetic rats for more than 4 weeks[10]. Vasoactive intestinal peptide (VIP) together with nitric oxide (NO) are the primary inhibitory neurotransmitters, mediating smooth muscle relaxation and VIP, inhibiting gastric acid secretion in the stomach, and stimulating the release of intestinal juice and bile in the intestines[11]. Previous studies in the porcine diabetic model have shown that an increased population of VIP-IR positive neurons in the inner submucosal plexus and MP of the duodenum, jejunum and ileum for 6 weeks contributed to gastrointestinal (GI) dysfunctions[12]. The inhibitory nitrergic neurons, which mainly express nNOS, release NO to mediate the relaxation of smooth muscles in the gastric fundus, and are therefore important in gut motility[13]. Several studies have demonstrated that loss of nNOS-IR positive neurons in the MP of the antrum can result in delayed GE in STZ-induced diabetic rats or gastric relaxation in spontaneously diabetic rats[14,15]. Calretinin (CALR), a calcium-binding protein expressed in specific enteric interneurons, may play a role in sensory signal processing within the GI tract[16]. However, the effects of CALR-IR positive neurons in diabetes remain unknown.
According to available data, the therapeutic mechanisms of GES for DGP include improved gastric accommodation, enhanced vagal activity, alterations in the ENS, and activation of central neurons[17,18]. Chen et al[10] revealed that GES for 6 weeks increased the expression of ChAT-IR and nNOS-IR positive neurons located in the myenteric layer of diabetic rats partly through glial cell line-derived neurotrophic factor expression. Furthermore, Wang et al[19] found that synchronized dual-pulse GES [a long pulse (300 milliseconds, 4 mA) and 5 short pulses (0.33 millisecond, 4 mA, 100 Hz)] improved enteric neuronal survival in the antrum tissue, and enhanced GE in vagotomized rats, possibly via cell line-derived neurotrophic factor and the phosphatidylinositol 3-kinase/protein kinase B pathways. Several studies have focused on the physiology of canine ENS because its higher similarity to humans than other small animals[20], but to date, no information is available on the efficacy mechanisms of GES on ENS in diabetic canines. In this study, we aimed to evaluate the effects of GES in STZ-induced diabetic beagle canines in concert with morphological changes in the MP of the antrum.
Adult female beagle dogs (fifteen months) were purchased from Beijing Keyu Animal Breeding Co., Ltd (Beijing, China). They weighed 8.3-12.5 kg and were housed individually in cages in a standardized environment at 16-28 °C, with a relative humidity of 40%-70% and a 12-hour/12-hour light-dark cycle. The canines had free access to food and water and were acclimated for 7 days before the modeling experiments. All procedures involving animals complied with the ARRIVE guidelines and the NIH Guide for the Care and Use of Laboratory Animals and were approved by the Animal Welfare and Ethical Committee of Peking Union Medical College Hospital (No. XHDW-2020-047 and No. XHDW-2020-047-2).
The modeling procedures were conducted in batches with a healthy canine randomly assigned per batch, thereby dividing the cohort into experimental and control groups. During the initial modeling phase, experimental canines received intravenous injections of freshly dissolved STZ (30 mg/kg; Sigma-Aldrich, St. Louis, MO, United States) while controls were administered the same volume of sodium citrate buffer (0.25 mL/kg; Solarbio, Beijing, China). If experimental canines maintained normoglycemia (defined as fasting blood glucose levels < 5.5 mmol/L) within 1-4 months after the initial modeling, a supplemental dose of STZ (30 mg/kg) was administered to reinforce the model. A diabetic model was considered successfully established when the fasting blood glucose concentration measured by a portable blood glucose meter was consistently maintained at 11.1 mmol/L or higher[21]. Following successful model induction, the canines were divided into a DM group with confirmed hyperglycemia (n = 6) and a healthy control group (n = 3). The time from DM development to electrical stimulation device implantation was 0-24 (15.3 ± 10.5) months on average, and the body weights before the electrical stimulation device implantation were 9.5-14.0 kg and 11.9-14.1 kg in the two groups.
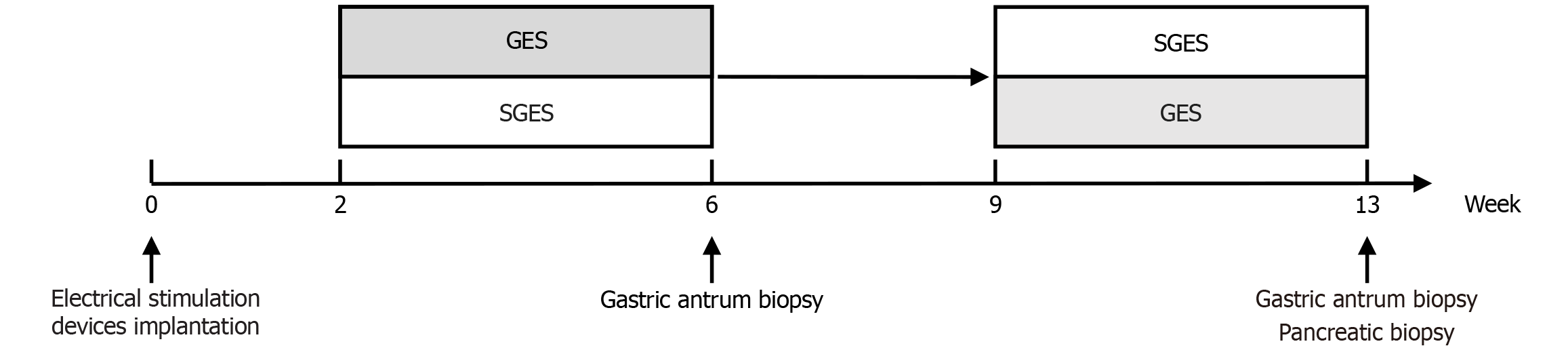
All canines underwent minor abdominal surgery to implant the electrical stimulation device under general anesthesia. A pair of stimulating electrodes was buried in the serous muscular layer of the antrum near the greater curvature 4 cm above the pylorus at a distance of 1 cm. A pulse generator (G101A, Beijing PINS Medical Co., Ltd., Beijing, China) was placed in the capsular bag in the myometrium-fascial space of the left upper abdominal wall which was bluntly separated. Following implantation, there was a 2-week postoperative recovery period (Figure 1).
This study was a randomized, controlled, crossover design. All beagle canines underwent a stimulation phase (labeled GES) or a sham GES (SGES) phase (labeled SGES) of 4 weeks each in a randomized order. At the end of the first 4-week phase of GES or SGES, there was a 3-week washout to avoid carry-over effects as well as post biopsy operative recovery, followed by crossover for 4 weeks of the alternate stimulation. GES was continuously applied with high-frequency, short-pulse stimulation (0.33 millisecond, 5 mA, 15 Hz, 0.2 second on, 3 seconds off). The procedures for SGES were the same as those for GES except the stimulators were not turned on. The timeline of the experiment is shown in Figure 1.
Full-thickness gastric antrum biopsies of 1 cm × 0.5 cm were obtained at the end of first phase treatment under general anesthesia, and the second antrum biopsies and pancreas tissue biopsies were conducted at the end of the study (Figure 1). Based on different stimulations, the gastric antrum tissues were grouped into the following four subgroups: (1) DM with GES group (DM + GES); (2) DM with SGES group (DM + SGES); (3) Control with GES group (control + GES); and (4) Control with SGES group (control + SGES).
Sections from the pancreatic tail, embedded in paraffin, were dewaxed using xylene, rehydrated with a series of graded ethanol, and rinsed with distilled water. The eosin dye solution was applied after rinsing with running water for 10 minutes and allowed to react for 2 minutes to stain the cytoplasm, and then dehydration and mounting were performed for microscopic examination. Pathological changes in the pancreatic islets of diabetic and control canines were detected using hematoxylin-eosin (HE) staining.
The full-thickness stomach tissue was slightly stretched and then fixed in 40 g/L formaldehyde, embedded in paraffin blocks, and sliced at a thickness of 4 μm. Consecutive sections were grouped into sets of 10 slices each, resulting in a total of 5 groups. From each group, the 1st, 2nd, 3rd, and 4th sections were used for four specific neuronal stainings (ChAT, VIP, nNOS and CALR), respectively. Immunofluorescence staining was employed to examine the distribution and proportions of neurons in the MP of diabetic canines (GES, SGES) and control groups (GES, SGES). Paraffin sections were kept in dewaxing agent and deparaffinized each for 3 × 20 minutes and gradually hydrated in a series of graded alcohol. Epitope retrieval was carried out by boiling sections in sodium citrate buffer (0.01M, pH 6.0) for 20 minutes and cooling down to room temperature. Sections were blocked with 10% normal goat serum in phosphate-buffered saline (PBS) containing 0.3% Triton X-100 at room temperature for 2 hours. The tissues were incubated overnight at 4 °C in a mixture of mouse anti-human neuronal protein HuC/HuD (anti-Hu) antiserum and a rabbit primary antibody for neuronal staining. Anti-Hu was utilized as a general neuronal marker for enteric neurons. Primary antibodies targeting ChAT, VIP, nNOS, and CALR were employed to identify specific neurons based on chemical markers: (1) ChAT indicating cholinergic excitatory motor neurons; (2) VIP for inhibitory secretomotor neurons; (3) The nNOS for inhibitory nitrergic neurons[22,23]; and (4) CALR for sensory neurons. The tissues were washed three times for 10 minutes each in PBS and then incubated with secondary antibodies conjugated with fluorescein isothiocyanate (FITC) and tetramethyl rhodamine isothiocyanate for 2 hours in the dark. To ensure proper labeling, the tissues were washed three times for 10 minutes each with PBS and then examined using a fluorescence microscope (Axio imager Z2, Carl Zeiss AG, Oberkochen, Germany). The details of the primary and secondary antibodies, including their dilutions and sources, are provided in Table 1. The outline of the ganglions in the MP was clear and distinguishable with cluster anti-Hu and specific chemical marker IR positive neurons under the fluorescence microscope. At a magnification of 10 × 20, non-overlapping fields-of-view were used across the sections, with 5 fields in the MP from each section and 25 identical fields from each biopsy to ensure that no less than 250 neurons with anti-Hu-IR positive staining were counted. The numbers of neurons and positive neurons for other specific antibodies in each of the 25 fields of the gastric tissue samples were counted by two researchers who had received uniform standard training in a blinded manner. The results were displayed as a percentage of the total neurons that were labeled with anti-Hu.
| Antigen | Host | Code | Dilution | Source |
| Anti-human neuronal protein HuC/HuD | Mouse | A-21271 | 1:200 | Invitrogen |
| Choline acetyltransferase | Rabbit | 20747-1-AP | 1:200 | Proteintech |
| Vasoactive intestinal peptide | Rabbit | Ab22736 | 1:1000 | Abcam |
| Neuronal nitric oxide synthase | Rabbit | T55342S | 1:200 | Abmart |
| Calretinin | Rabbit | AB5054 | 1:1000 | Chemicon |
| Mouse IgG | Goat tetramethyl rhodamine isothiocyanate | SA00013-3 | 1:200 | Proteintech |
| Rabbit IgG | Goat fluorescein isothiocyanate | SA00013-2 | 1:200 | Proteintech |
Results are shown as mean ± SD; n refers to the number of canines analyzed. Differences between the GES and SGES subgroups were evaluated using the paired-sample t-test in SPSS 25.0 (SPSS Inc., Chicago, IL, United States). For comparisons between the DM and control groups, Levene’s test was first applied to assess the homogeneity of variances. If the variances were equal, an independent samples t-test was used; if variances were unequal, Welch’s corrected t-test was employed. Multiple comparison correction was not applied as this was an exploratory study with only a few planned comparisons based on our protocol. Differences were considered statistically significant at P < 0.05. All graphs were created by commercial software (GraphPad Prism version 5.00 for Windows, GraphPad Software Inc., La Jolla, CA, United States).
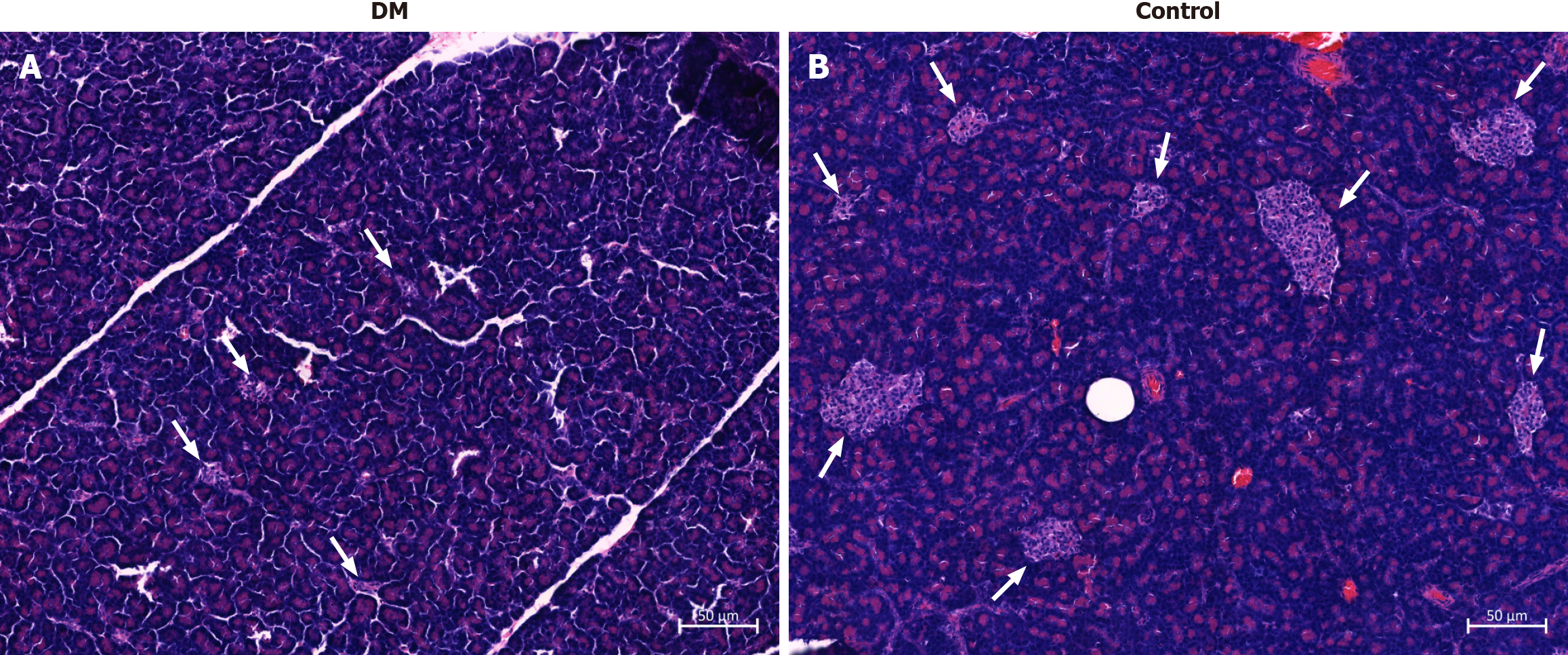
Six beagle canines developed DM following initial and subsequent booster modeling. We observed the shape of islets in the pancreatic tail tissues. The HE staining demonstrated that diabetic canines exhibited a significant reduction in the number and area of pancreatic islets with irregular, ill-defined contours and atrophied cells, which confirmed successful establishment of the DM model (Figure 2A). Representative HE staining of pancreatic tail tissues in healthy controls is shown in Figure 2B.
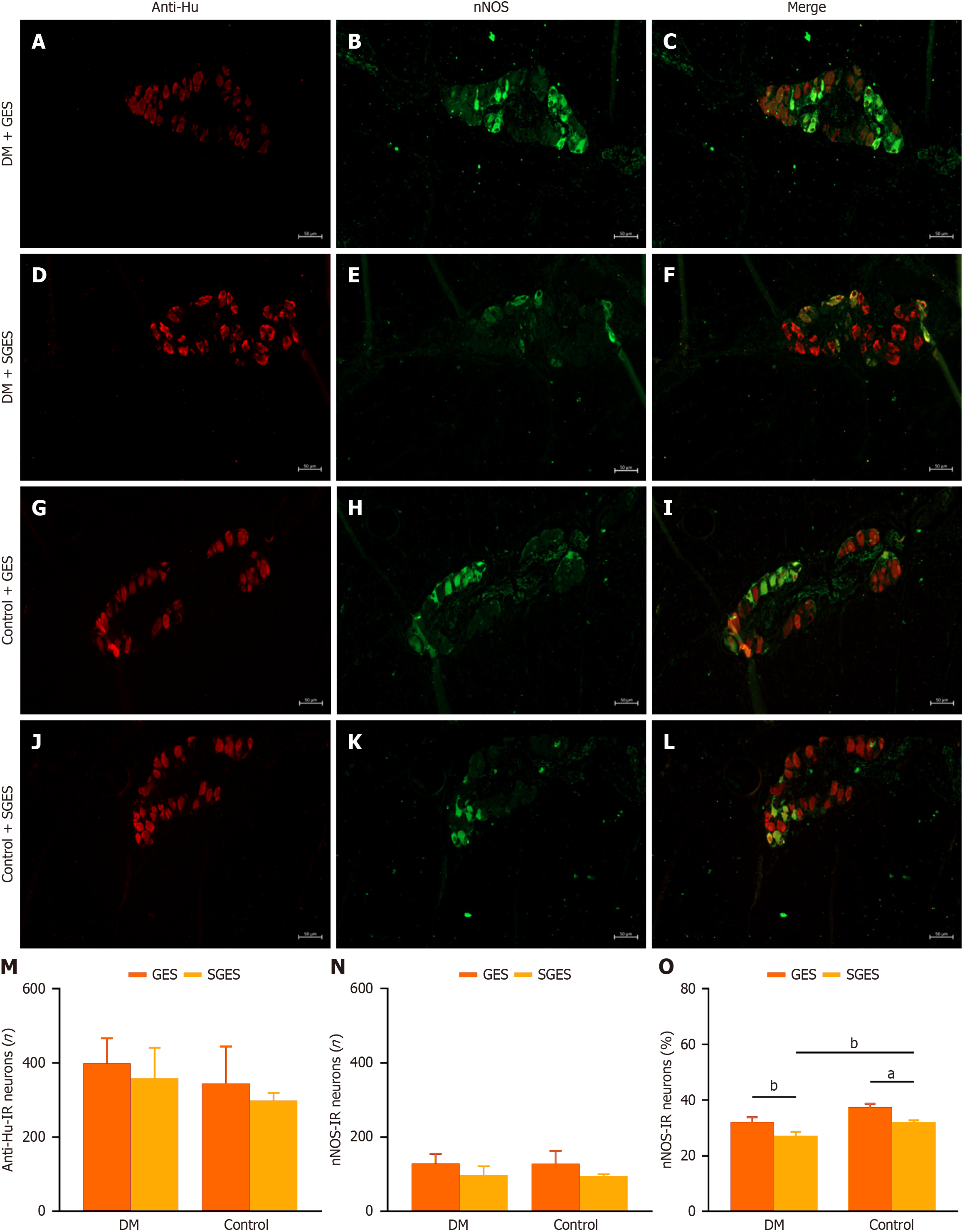
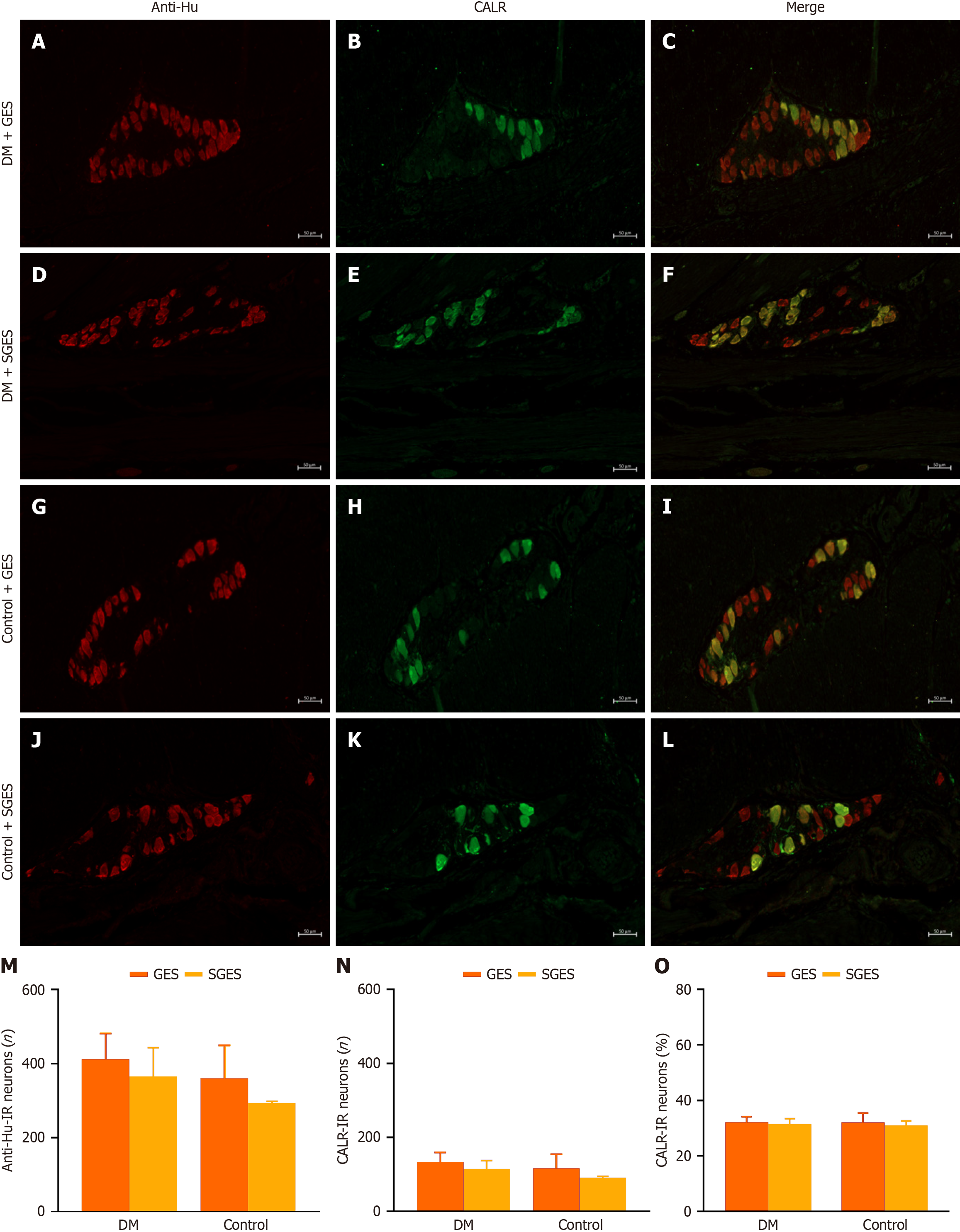
We observed the number of enteric ganglia, the total number of neurons (anti-Hu-IR positive neurons), and any specific markers (i.e., ChAT, VIP, nNOS, and CALR positive neurons) in the relatively consecutive and consistent MP of antral double-stained sections. There were no significant differences in the numbers of enteric ganglia between the GES and the SGES subgroups in diabetic canines or controls (all P > 0.05; Table 2). The total number of enteric neurons was not significantly different in anti-Hu with any specific markers double-staining analyses between the GES and the SGES subgroups, whether in the diabetic canines or the controls (all P > 0.05; Supplementary Table 1).
| Ganglia | Diabetes mellitus (n = 6) | Controls (n = 3) | ||||
| GES | SGES | P value | GES | SGES | P value | |
| Choline acetyltransferase | 16.8 ± 3.0 | 14.8 ± 2.8 | 0.292 | 13.8 ± 3.5 | 12.3 ± 0.8 | 0.538 |
| Vasoactive intestinal peptide | 16.9 ± 2.7 | 14.2 ± 3.2 | 0.130 | 14.1 ± 3.6 | 12.5 ± 0.8 | 0.435 |
| Neuronal nitric oxide synthase | 16.1 ± 2.8 | 14.5 ± 3.1 | 0.372 | 14.0 ± 3.9 | 12.0 ± 0.8 | 0.539 |
| Calretinin | 16.7 ± 2.7 | 14.8 ± 3.0 | 0.190 | 14.4 ± 3.6 | 11.9 ± 0.5 | 0.381 |
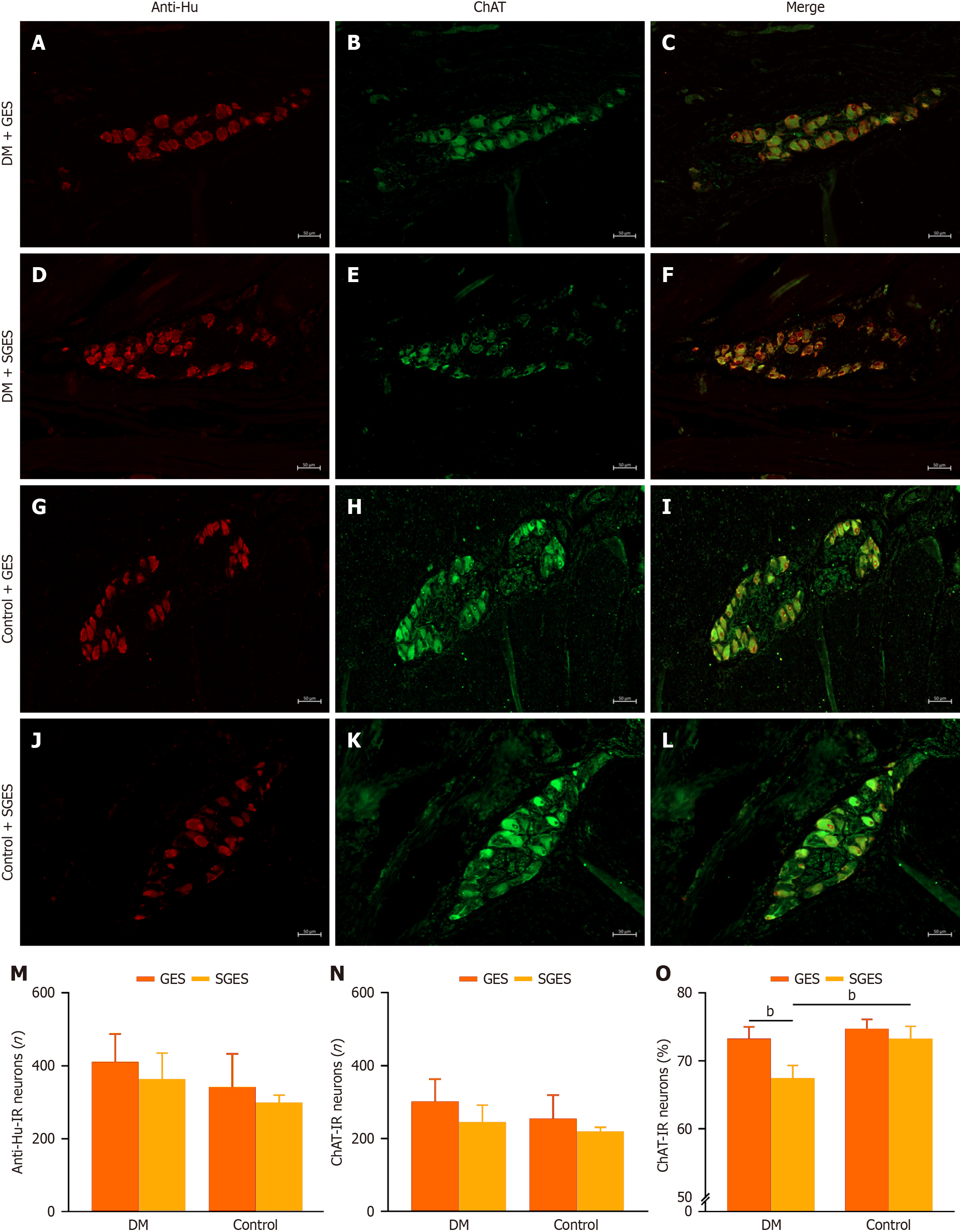
Myenteric ChAT-IR positive neurons were stained in the cytoplasm, they were the most important cellular component of the MP with a proportion of over 65% in the four subgroups. There was no significant difference in the total number of ChAT-IR positive neurons between the GES intervention group and SGES subgroup across both the diabetic group and control group (P = 0.127; P = 0.390, respectively; Figure 3A-N and Supplementary Table 1). Following GES, the proportion of ChAT-IR positive neurons was significantly higher compared to SGES (73.3% ± 1.7% vs 67.5% ± 1.8%, P = 0.002; Figure 3O and Supplementary Table 1) in diabetic canines, but this neuromodulatory effect was absent in normoglycemic canines (74.7% ± 1.4% vs 73.3% ± 1.8%, P = 0.255; Figure 3O and Supplementary Table 1). In the sham-stimulated subgroups, diabetic canines exhibited a significantly lower proportion of ChAT-IR positive neurons compared to controls (67.5% ± 1.8% vs 73.3% ± 1.8%, P = 0.003; Figure 3O and Supplementary Table 1).
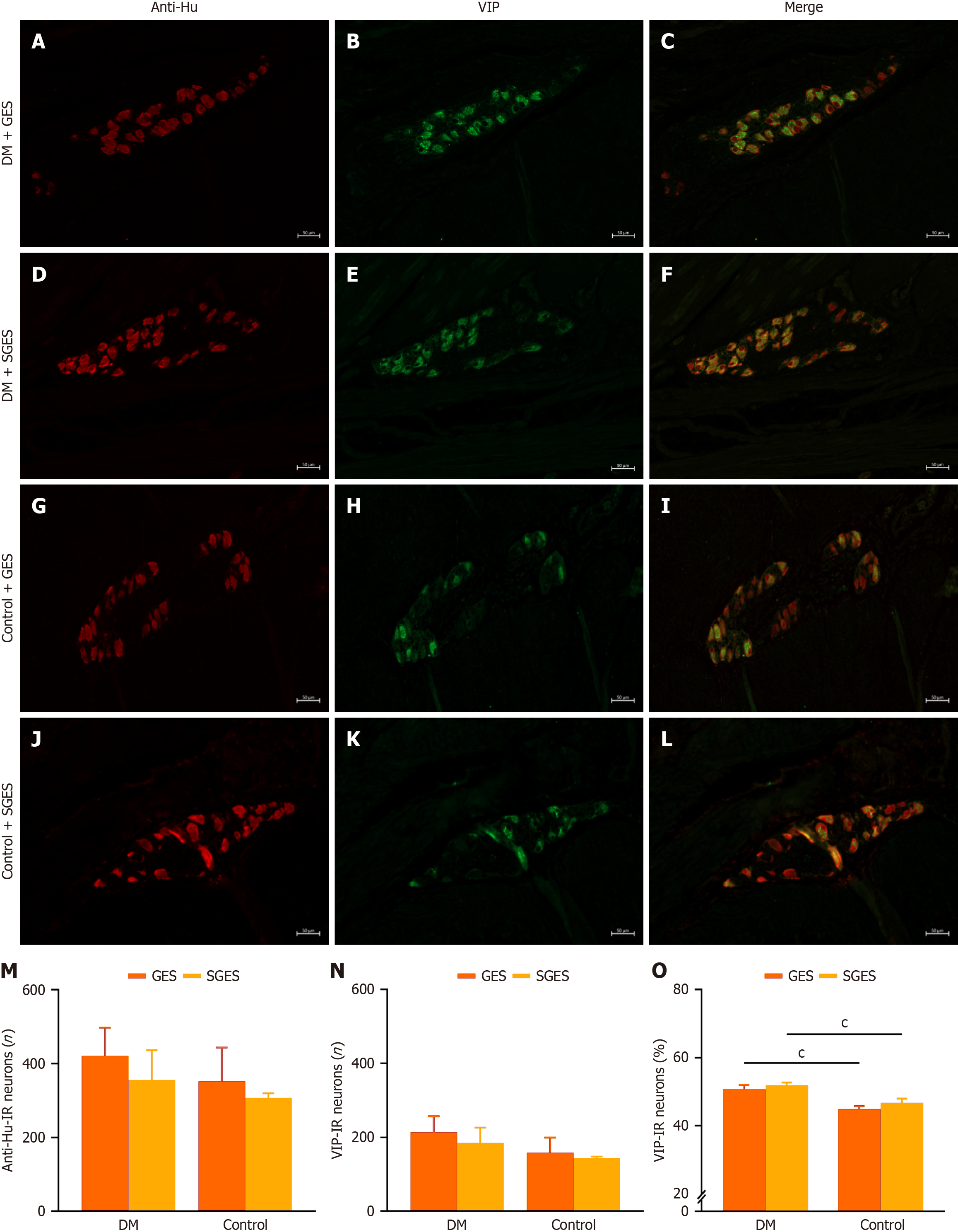
Myenteric VIP-IR positive neurons exhibited cytoplasmic dot-like staining and accounted for over 40% of the neuronal population in the four subgroups. No significant differences were observed in the count of VIP-IR positive neurons between the GES intervention and SGES across both the diabetic and control group (P = 0.223; P = 0.581, respectively; Figure 4A-N and Supplementary Table 1) as well as the proportion of VIP-IR positive neurons between GES and SGES among diabetic models or controls (P = 0.054; P = 0.227, respectively; Figure 4O and Supplementary Table 1). Within the GES subgroups, diabetic canines exhibited a significantly higher proportion of VIP-IR positive neurons compared to controls (50.8% ± 1.3% vs 44.9% ± 0.8%, P < 0.001; Figure 4O and Supplementary Table 1), and this phenomenon was also observed in the SGES subgroups (51.9% ± 0.8% in diabetic canines vs 46.8% ± 1.2% in controls, P < 0.001; Figure 4O and Supplementary Table 1).
Approximately 30% of myenteric neurons in the canine gastric antrum were nNOS-IR positive neurons with staining confined to the cytoplasm, and the number of these neurons was similar between the GES and SGES subgroups across both the DM models and controls (P = 0.070, P = 0.282, respectively; Figure 5A-N and Supplementary Table 1). GES intervention resulted in a significantly higher proportion of nNOS-IR positive neurons compared to the SGES subgroup (32.1% ± 1.7% vs 27.3% ± 1.4%, P = 0.002; Figure 5O and Supplementary Table 1) in diabetic canines; this neuromodulatory effect was also found in healthy canines (37.5% ± 1.3% vs 32.1% ± 0.7%, P = 0.036; Figure 5O and Supplementary Table 1), and there was no significant difference in this proportion between the two GES subgroups in diabetic and healthy canines. Under the same sham stimulation, the percentage of nNOS-IR positive neurons was markedly lower in the diabetic canines compared with controls (27.3% ± 1.4% vs 32.1% ± 0.7%, P = 0.001; Figure 5O and Supplementary Table 1).
Myenteric CALR-IR positive neurons exhibited intense cytoplasmic staining with low fluorescence intensity in the nuclear region, clearly delineating nuclear contours. No significant difference was detected in the total number of CALR-IR positive neurons after GES when compared with the SGES subgroup in both the DM group and the control group (P = 0.174, P = 0.326, respectively; Figure 6A-N and Supplementary Table 1). The percentage of CALR-IR positive neurons was similar between the GES and the SGES subgroups in the DM canines (27.3% ± 1.4% vs 31.5% ± 2.0%, P = 0.631; Figure 6O and Supplementary Table 1). This phenomenon was also observed in the controls (32.2% ± 3.3% in the GES subgroup vs 31.1% ± 1.6% in the SGES subgroup, P = 0.508; Figure 6O and Supplementary Table 1).
In this study, a diabetic model was established using large beagle canines. We observed a reduced proportion of ChAT and nNOS-IR positive neurons in the antrum of diabetic beagles. Furthermore, it was noted that the percentage of VIP-IR positive neurons was increased compared with the non-diabetic controls. GES partly reversed these pathological alterations and improved the survival of cholinergic and nitrergic neurons in both the DM groups and controls.
Gastroparesis is the most common GI complication of diabetes. The most accepted mechanisms of DGP are defects in the ICC and changes in enteric neurons, especially musculomotor neurons (i.e., ChAT) and inhibitory neurons (i.e., nNOS)[24]. ChAT affects the neurotransmitter acetylcholine, which activates smooth muscle M3 receptors, thereby increasing the frequency and amplitude of GI contractions[25]. VIP and NO are the key inhibitory neurotransmitters in the MP, which facilitate the receptive relaxation of the gastric fundus, modulate antral contractions, and trigger pyloric relaxation[26,27]. CALR is expressed in a subset of myenteric neurons-including cholinergic motor neurons, interneurons, and sensory neurons, and is crucial for detecting mucosal stimuli and coordinating peristaltic reflexes[28].
Previous studies demonstrated a time-dependent reduction in ChAT expression in the colon of STZ-induced diabetic rats, an increase in VIP-IR positive neurons in the duodenal, jejunal, and colonic tissues of the diabetic porcine model, and a decrease in nNOS expression in the antral MP of DGP rats[9,12,14]. However, there are no experimental data on the changes in CALR-IR positive neurons in the ENS of diabetic models. The impairments in gastric motor functions caused by a high-fat diet seem to be linked to: (1) A reduction in myenteric neurons; (2) Decreased nNOS immunoreactivity in the antrum and fundus; (3) A downregulation of antral ChAT expression; and (4) Increased VIP in the myenteric neurons of the antrum[29]. In diabetes, elevated reactive oxygen species cause the loss and degeneration of nNOS-IR positive neurons, collectively impairing NO-mediated inhibitory neurotransmission. This disruption results in antral hypercontractility, compromised coordination of pyloric sphincter relaxation, and ultimately delayed GE, along with gastric stasis and hypertrophic pyloric stenosis[30,31].
In this study, we discovered that the morphological changes in gastric musculomotor and inhibitory neurons are also present in STZ-induced diabetic beagles. These changes included a marked reduction in the proportions of ChAT- and nNOS-IR positive neurons and a significant increase in VIP-IR positive neurons within the antral MP, even though the diabetic canines did not show obvious manifestations of gastroparesis (according to the results of radionuclide GE scintigraphy). Some studies have shown that the quantity of VIP-IR positive neurons and the level of VIP in the antrum of diabetic animals are significantly reduced[32,33]. Our results are contrary to these findings but are consistent with the study by Bulc et al[12]. Our analysis revealed no significant differences in the proportion of CALR-IR positive neurons between the diabetic and non-diabetic groups. These results are speculated based on the comparison of the proportions of specific neurons between diabetic and control canines receiving the same SGES. The lack of baseline gastric antral histopathology is due to welfare and ethical considerations in animals.
GES is a treatment option for managing refractory gastroparesis, showing significant improvements in key symptoms such as nausea and vomiting, as well as in glucose regulation[34]. The therapeutic mechanisms of GES therapy may involve multifactorial neuromodulatory effects, including an increase in vagal efferent autonomic activity, an enhancement of gastric accommodation, and the potential restoration of ICC proliferation[6,18,35]. Can short-term GES therapy similarly ameliorate the damage to enteric neurons in diabetic beagle canines, as observed by Chen et al[10] in diabetic rats?
We found that GES increased the percentage of ChAT-IR positive neurons in DM models compared to SGES. These findings suggest that GES may regulate abnormal gastric motility by promoting smooth muscle contraction, providing non-pharmacological therapeutic evidence for GES treatment in DGP. However, this neuromodulatory effect was not observed in healthy canines. This pathology-dependent neuromodulation implies that GES counteracts hyperglycemia-induced transcriptional silencing (i.e., histone deacetylase activation) rather than broadly enhancing cholinergic plasticity. Our current study also demonstrated that 4-week GES restored the lost proportion of nNOS-IR positive neurons in both diabetic and control canines. A previous study indicated that nNOS dysregulation contributes to gastro-pyloric dysfunction in diabetic models, and insulin treatment restores its expression and function to normal levels[36]. Xing and Chen[17] provided clear evidence that long-pulse GES has an inhibitory impact on gastric tone and accommodation in healthy canines, with partial mediation through vagal and inhibitory nitrergic pathways. We propose that a selective increase in nNOS-IR neurons likely enhances receptive relaxation, gastric accommodation, and antro-pyloro-duodenal coordination. Notably, the proportions of VIP-IR and CALR-IR positive neurons remained unchanged after stimulation in both the diabetic and control groups. We consider that the stability of CALR-IR neuronal subtypes suggests that GES does not broadly alter the sensory neurons. Instead, its effect is highly selective neuromodulation on cholinergic and nitrergic neurons that regulate the abnormality of gastric motility and accommodation, without alterations in funda
This study provides the first evidence that GES significantly increased the proportions of cholinergic and nitrergic neurons within the gastric MP of diabetic beagle canines. As beagles have a higher physiological, anatomical, and metabolic similarity to humans than other small animals, we believe that the results obtained from canine models might be more representative in relation to humans. However, this study has several limitations. First, the limited sample size of animals (DM, n = 6; controls, n = 3) may reduce statistical power and limit the generalizability of the findings. The small sample size was mainly due to ethical constraints in large-animal research and difficulties in blood glucose management with long-term diabetic models. Secondly, the lack of exploration on molecular mechanisms related to neurotrophic signaling pathways, oxidative stress, and inflammatory markers precludes definitive conclusions about the molecular mechanisms responsible for the observed phenotypic changes. Thirdly, diabetic beagles did not exhibit delayed GE before GES, and no significant changes in GE were observed after GES (data not shown). Therefore, the relationship between the observed neuronal alterations and motility improvements needs to be confirmed in the gastroparesis models. Incorporating cellular mechanisms and functional assessments into our study is crucial for determining how GES promotes neuronal survival and whether GES affects gastric functions.
In summary, diabetic canines exhibited notable reductions in both ChAT-IR and nNOS-IR positive neuronal populations in the antral MP compared to controls, along with an upregulation of the proportion of VIP-IR positive neurons. Importantly, GES intervention had neuroprotective effects, effectively improving the survival of both cholinergic and nitrergic neurons in diabetic groups. These findings offer a theoretical foundation for the clinical application of GES in the treatment of DGP, especially through its neuromodulatory impacts on enteric neuronal plasticity.
The authors would like to acknowledge Beijing PINS Medical Co., Ltd., Beijing, China for their providing gastric electrical stimulation device and technical assistance.
| 1. | Choung RS, Locke GR 3rd, Schleck CD, Zinsmeister AR, Melton LJ 3rd, Talley NJ. Risk of gastroparesis in subjects with type 1 and 2 diabetes in the general population. Am J Gastroenterol. 2012;107:82-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 186] [Article Influence: 13.3] [Reference Citation Analysis (1)] |
| 2. | Krishnan B, Babu S, Walker J, Walker AB, Pappachan JM. Gastrointestinal complications of diabetes mellitus. World J Diabetes. 2013;4:51-63. [PubMed] [DOI] [Full Text] |
| 3. | Uppaluri S, Jain MA, Ali H, Shingala J, Amin D, Ajwani T, Fatima I, Patel N, Kaka N, Sethi Y, Kapoor N. Pathogenesis and management of diabetic gastroparesis: An updated clinically oriented review. Diabetes Metab Syndr. 2024;18:102994. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 4. | Concepción Zavaleta MJ, Gonzáles Yovera JG, Moreno Marreros DM, Rafael Robles LDP, Palomino Taype KR, Soto Gálvez KN, Arriola Torres LF, Coronado Arroyo JC, Concepción Urteaga LA. Diabetic gastroenteropathy: An underdiagnosed complication. World J Diabetes. 2021;12:794-809. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 33] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (4)] |
| 5. | McCurdy GA, Gooden T, Weis F, Mubashir M, Rashid S, Raza SM, Morris J, Cai Q. Gastric peroral endoscopic pyloromyotomy (G-POEM) in patients with refractory gastroparesis: a review. Therap Adv Gastroenterol. 2023;16:17562848231151289. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 24] [Reference Citation Analysis (0)] |
| 6. | Yin J, Abell TD, McCallum RW, Chen JD. Gastric neuromodulation with Enterra system for nausea and vomiting in patients with gastroparesis. Neuromodulation. 2012;15:224-31; discussion 231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 48] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 7. | Heckert J, Sankineni A, Hughes WB, Harbison S, Parkman H. Gastric Electric Stimulation for Refractory Gastroparesis: A Prospective Analysis of 151 Patients at a Single Center. Dig Dis Sci. 2016;61:168-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 59] [Article Influence: 5.9] [Reference Citation Analysis (3)] |
| 8. | Abdalla MMI. Enteric neuropathy in diabetes: Implications for gastrointestinal function. World J Gastroenterol. 2024;30:2852-2865. [PubMed] [DOI] [Full Text] |
| 9. | Du F, Wang L, Qian W, Liu S. Loss of enteric neurons accompanied by decreased expression of GDNF and PI3K/Akt pathway in diabetic rats. Neurogastroenterol Motil. 2009;21:1229-e114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 44] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 10. | Chen Y, Zhang S, Li Y, Yan H, Ba Y, Wang X, Shi N, Liu C. Gastric Electrical Stimulation Increases the Proliferation of Interstitial Cells of Cajal and Alters the Enteric Nervous System in Diabetic Rats. Neuromodulation. 2022;25:1106-1114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 11. | Chandrasekharan B, Srinivasan S. Diabetes and the enteric nervous system. Neurogastroenterol Motil. 2007;19:951-960. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 183] [Cited by in RCA: 130] [Article Influence: 6.8] [Reference Citation Analysis (4)] |
| 12. | Bulc M, Całka J, Palus K. Effect of Streptozotocin-Inducted Diabetes on the Pathophysiology of Enteric Neurons in the Small Intestine Based on the Porcine Diabetes Model. Int J Mol Sci. 2020;21:2047. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 13. | Groneberg D, Voussen B, Friebe A. Integrative Control of Gastrointestinal Motility by Nitric Oxide. Curr Med Chem. 2016;23:2715-2735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 52] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 14. | Wrzos HF, Cruz A, Polavarapu R, Shearer D, Ouyang A. Nitric oxide synthase (NOS) expression in the myenteric plexus of streptozotocin-diabetic rats. Dig Dis Sci. 1997;42:2106-2110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 74] [Article Influence: 2.6] [Reference Citation Analysis (3)] |
| 15. | Takahashi T, Nakamura K, Itoh H, Sima AA, Owyang C. Impaired expression of nitric oxide synthase in the gastric myenteric plexus of spontaneously diabetic rats. Gastroenterology. 1997;113:1535-1544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 148] [Article Influence: 5.1] [Reference Citation Analysis (3)] |
| 16. | Zetzmann K, Strehl J, Geppert C, Kuerten S, Jabari S, Brehmer A. Calbindin D28k-Immunoreactivity in Human Enteric Neurons. Int J Mol Sci. 2018;19:194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 17. | Xing JH, Chen JD. Effects and mechanisms of long-pulse gastric electrical stimulation on canine gastric tone and accommodation. Neurogastroenterol Motil. 2006;18:136-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 18. | Hasler WL. Methods of gastric electrical stimulation and pacing: a review of their benefits and mechanisms of action in gastroparesis and obesity. Neurogastroenterol Motil. 2009;21:229-243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 60] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 19. | Wang N, Li K, Song S, Chen J. Gastric electrical stimulation improves enteric neuronal survival. Int J Mol Med. 2017;40:438-446. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 20. | Kleinert M, Clemmensen C, Hofmann SM, Moore MC, Renner S, Woods SC, Huypens P, Beckers J, de Angelis MH, Schürmann A, Bakhti M, Klingenspor M, Heiman M, Cherrington AD, Ristow M, Lickert H, Wolf E, Havel PJ, Müller TD, Tschöp MH. Animal models of obesity and diabetes mellitus. Nat Rev Endocrinol. 2018;14:140-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 737] [Cited by in RCA: 639] [Article Influence: 79.9] [Reference Citation Analysis (0)] |
| 21. | Han Q, Sun J, Xie W, Bai Y, Wang S, Huang J, Zhou S, Li Q, Zhang H, Tang Z. Repeated Low-Dose Streptozotocin and Alloxan Induced Long-Term and Stable Type 1 Diabetes Model in Beagle Dogs. Biomed Res Int. 2022;2022:5422287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 22. | Sanders KM, Ward SM. Nitric oxide and its role as a non-adrenergic, non-cholinergic inhibitory neurotransmitter in the gastrointestinal tract. Br J Pharmacol. 2019;176:212-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 95] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 23. | Giancola F, Fracassi F, Gallucci A, Sadeghinezhad J, Polidoro G, Zini E, Asti M, Chiocchetti R. Quantification of nitrergic neurons in the myenteric plexus of gastric antrum and ileum of healthy and diabetic dogs. Auton Neurosci. 2016;197:25-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 24. | Azpiroz F, Malagelada C. Diabetic neuropathy in the gut: pathogenesis and diagnosis. Diabetologia. 2016;59:404-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 53] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 25. | Chiocchetti R, Giancola F, Mazzoni M, Sorteni C, Romagnoli N, Pietra M. Excitatory and inhibitory enteric innervation of horse lower esophageal sphincter. Histochem Cell Biol. 2015;143:625-635. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 26. | Grider JR, Makhlouf GM. Vasoactive intestinal peptide. Transmitter of inhibitory motor neurons of the gut. Ann N Y Acad Sci. 1988;527:369-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 27] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 27. | Shen GM, Zhou MQ, Xu GS, Xu Y, Yin G. Role of vasoactive intestinal peptide and nitric oxide in the modulation of electroacupucture on gastric motility in stressed rats. World J Gastroenterol. 2006;12:6156-6160. [PubMed] [DOI] [Full Text] |
| 28. | Sang Q, Young HM. Chemical coding of neurons in the myenteric plexus and external muscle of the small and large intestine of the mouse. Cell Tissue Res. 1996;284:39-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 200] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 29. | Kaya SD, Sinen O, Bülbül M. Gastric motor dysfunction coincides with the onset of obesity in rats fed with high-fat diet. Clin Exp Pharmacol Physiol. 2021;48:553-562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 30. | Korenaga K, Micci MA, Taglialatela G, Pasricha PJ. Suppression of nNOS expression in rat enteric neurones by the receptor for advanced glycation end-products. Neurogastroenterol Motil. 2006;18:392-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 40] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 31. | Sivarao DV, Mashimo H, Goyal RK. Pyloric sphincter dysfunction in nNOS-/- and W/Wv mutant mice: animal models of gastroparesis and duodenogastric reflux. Gastroenterology. 2008;135:1258-1266. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 56] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 32. | Spångéus A, El-Salhy M. Myenteric plexus of obese diabetic mice (an animal model of human type 2 diabetes). Histol Histopathol. 2001;16:159-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 33. | Adeghate E, Ponery AS, Sharma AK, El-Sharkawy T, Donáth T. Diabetes mellitus is associated with a decrease in vasoactive intestinal polypeptide content of gastrointestinal tract of rat. Arch Physiol Biochem. 2001;109:246-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 18] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 34. | Shada A, Nielsen A, Marowski S, Helm M, Funk LM, Kastenmeier A, Lidor A, Gould JC. Wisconsin's Enterra Therapy Experience: A multi-institutional review of gastric electrical stimulation for medically refractory gastroparesis. Surgery. 2018;164:760-765. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 35. | McCallum RW, Dusing RW, Sarosiek I, Cocjin J, Forster J, Lin Z. Mechanisms of symptomatic improvement after gastric electrical stimulation in gastroparetic patients. Neurogastroenterol Motil. 2010;22:161-167, e50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 76] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 36. | Watkins CC, Sawa A, Jaffrey S, Blackshaw S, Barrow RK, Snyder SH, Ferris CD. Insulin restores neuronal nitric oxide synthase expression and function that is lost in diabetic gastropathy. J Clin Invest. 2000;106:373-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 172] [Article Influence: 6.6] [Reference Citation Analysis (0)] |