Published online Apr 15, 2026. doi: 10.4239/wjd.v17.i4.116772
Revised: December 24, 2025
Accepted: February 3, 2026
Published online: April 15, 2026
Processing time: 142 Days and 2.8 Hours
Diabetic kidney disease is the most common microvascular complication of type 2 diabetes mellitus (T2DM) and a leading cause of end-stage renal disease. Rapid estimated glomerular filtration rate (eGFR) decline (annual decline rate ≥ 5 mL/minute/1.73 m2) is a strong predictor of end-stage renal disease and cardio
To establish a comprehensive and accurate risk stratification system for patients with T2DM and kidney complications is of crucial importance for clinical deci
This retrospective cohort study enrolled 302 T2DM patients who attended our hospital from January 2021 to August 2024, with a minimum follow-up period of 12 months. Patients were divided into rapid decline group (≥ 5 mL/minute/1.73 m2, n = 89) and non-rapid decline group (< 5 mL/minute/1.73 m2, n = 213) based on annual eGFR decline rate. Least absolute shrinkage and selection operator regression combined with multivariate logistic regression was used to screen independent risk factors and construct a visualized Nomogram prediction model. eXtreme Gradient Boosting and Random Forest algorithms were employed for validation. Model performance was evaluated through receiver operating characteristic curves, calibration curves, and decision curve analysis, and the model’s generalizability was validated in an external validation cohort of 142 patients. Sensitivity analyses using the chronic kidney disease epidemiology collaboration equation were performed to assess the robustness of findings.
Multivariate analysis identified seven independent risk factors: Hemoglobin A1c ≥ 8.0% [odds ratio (OR) = 3.24, 95% confidence interval (CI): 1.76-5.98], baseline eGFR 45-59 mL/minute/1.73 m2 (OR = 4.17, 95%CI: 2.15-8.09), urine albumin-to-creatinine ratio ≥ 300 mg/g (OR = 5.63, 95%CI: 2.89-10.96), systolic blood pressure ≥ 140 mmHg (OR = 2.58, 95%CI: 1.42-4.69), serum uric acid ≥ 420 μmol/L (OR = 2.91, 95%CI: 1.53-5.54), anemia (OR = 2.34, 95%CI: 1.28-4.27), and non-use of renin-angiotensin-aldosterone system inhibitors (OR = 2.15, 95%CI: 1.18-3.92). The Nomogram model achieved an AUC of 0.876 (95%CI: 0.836-0.916) in the training set, with sensitivity of 81.5% and specificity of 79.8%. The external validation cohort demonstrated an AUC of 0.838 (95%CI: 0.768-0.908), showing good generalizability. Sensitivity analyses using chronic kidney disease epidemiology collaboration showed consistent results (AUC = 0.869, P = 0.42 vs modification of diet in renal disease-based model). The eXtreme Gradient Boosting model achieved a test set AUC of 0.889, but the difference from the nomogram model was not statistically significant (P = 0.285). The model demonstrated stable performance across different age, gender, and diabetes duration subgroups (AUC = 0.854-0.889) with non-significant interaction terms (all P interaction > 0.4). Compared with existing kidney failure risk equation 4, kidney failure risk equation 8, and Kidney Disease: Improving Global Outcomes stratification systems, our model showed significant advantages (P < 0.05) with net reclassification improvement of 0.385-0.428 and integrated discrimination improvement of 0.076-0.156 in external validation.
This work effectively developed a nomogram model with good discrimination, calibration, and clinical applicability for risk stratification of rapid eGFR drop among T2DM patients with indications of kidney involvement. The model integrates both established risk variables and new risk markers.
Core Tip: This study established a nomogram for risk stratification of rapid decline in estimated glomerular filtration rate in type 2 diabetes mellitus patients with established kidney involvement. The model integrates cardiovascular symptoms, metabolic indicators, and renal biomarkers, demonstrating high discrimination and strong clinical practicality. This tool enables identification of high-risk subgroups who would benefit from intensified monitoring and intervention to slow kidney function deterioration.
- Citation: Huang P, Qin XQ, Huang Q, Wang SD, Wu YY, Huang XR, Lin X. Prediction model for rapid estimated glomerular filtration rate decline in type 2 diabetes mellitus. World J Diabetes 2026; 17(4): 116772
- URL: https://www.wjgnet.com/1948-9358/full/v17/i4/116772.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i4.116772
Diabetic kidney disease (DKD) is one of the most common microvascular complications of type 2 diabetes mellitus (T2DM) and is also the leading cause of end-stage renal disease (ESRD). Globally, approximately 20%-40% of T2DM patients eventually develop DKD, with 10%-20% progressing to ESRD, imposing a heavy disease burden and economic pressure on patients and society[1-3]. The prevalence of diabetes in China continues to rise, with the adult diabetes prevalence rate reaching 12.8%, and the prevalence of DKD rising synchronously, making it the second leading cause of chronic kidney disease. Therefore, risk stratification for DKD progression and identification of patients at highest risk are of great clinical significance for delaying renal failure and improving patient prognosis.
Estimated glomerular filtration rate (eGFR) is a core indicator for assessing renal function, and its rate of decline directly reflects the degree of kidney damage progression. Recent studies have shown that rapid eGFR decline (usually defined as an annual decline rate ≥ 5 mL/minute/1.73 m2) is a powerful predictor of ESRD and cardiovascular events, with predictive value superior to single eGFR measurements[4,5]. The Kidney Disease Outcomes Quality Initiative guidelines clearly state that rapid eGFR decline is an important marker of chronic kidney disease progression and should serve as an important basis for clinical risk stratification and treatment decisions[6]. However, current clinical practice for identifying rapid eGFR decline in T2DM patients primarily relies on repeated monitoring, lacking effective risk stratification tools, resulting in some high-risk patients not receiving intensive intervention in a timely manner.
Existing research has identified multiple risk factors associated with DKD progression, including traditional factors such as hyperglycemia, hypertension, and proteinuria[7,8]. However, these studies mostly focus on single or few risk factors, lacking systematic integration of multidimensional indicators; and most studies use proteinuria progression or eGFR reaching specific thresholds as endpoints, with relatively insufficient specialized research on the dynamic indicator of rapid eGFR decline. Additionally, the roles of non-traditional risk factors such as hyperuricemia and anemia in DKD progression are receiving increasing attention[9,10], but their value in rapid eGFR decline prediction models has not been fully elucidated. Meanwhile, although RAAS inhibitors have been proven to have renoprotective effects, their utilization rate and standardization in real-world settings remain insufficient, and their actual impact on eGFR decline rate requires further quantitative assessment.
With the development of precision medicine, constructing individualized risk prediction models has become an important strategy for disease prevention and control. In recent years, Nomogram charts have been widely used in clinical risk assessment due to their intuitive and user-friendly characteristics[11]. Machine learning algorithms such as eXtreme Gradient Boosting (XGBoost) and Random Forest have demonstrated tremendous potential in disease prediction due to their powerful ability to capture nonlinear relationships[12]. Combining traditional statistical methods with machine learning technology holds promise for constructing more accurate prediction models and providing strong support for clinical decision-making.
Based on the above background, this study aims to systematically analyze multidimensional risk factors for rapid eGFR decline in T2DM patients through a retrospective cohort study, using least absolute shrinkage and selection operator (LASSO) regression combined with multivariate logistic regression to screen key predictive factors, construct a visualized Nomogram prediction model, and employ machine learning algorithms for comparative validation. The study particularly focuses on easily overlooked risk factors such as anemia and hyperuricemia, as well as the use of renoprotective agents, striving to establish a comprehensive and accurate risk assessment system.
This retrospective cohort study consecutively enrolled T2DM patients who visited the Department of Endocrinology (outpatient and inpatient) at our hospital from January 2021 to August 2024. Inclusion criteria: (1) Meeting World Health Organization 1999 diagnostic criteria for T2DM; (2) Age ≥ 18 years; (3) At least 12 months of follow-up with at least 3 eGFR measurements (baseline, interim, and endpoint) with intervals ≥ 3 months; (4) Baseline eGFR ≥ 30 mL/minute/1.73 m2; and (5) Complete clinical data. Exclusion criteria: (1) Type 1 diabetes or special types of diabetes; (2) Combined acute kidney injury, renal artery stenosis, polycystic kidney disease, glomerulonephritis, or other non-DKDs; (3) Combined malignancy, severe infection, or autoimmune disease; (4) Previous renal replacement therapy; (5) Pregnant or lactating women; and (6) Death from non-renal causes during follow-up. A total of 302 patients were finally included in the analysis.
Based on the annual eGFR decline rate during follow-up, using the formula: Annual eGFR decline rate = (baseline eGFR - final eGFR)/follow-up years, patients were divided into two groups: Rapid eGFR decline group (annual decline rate ≥ 5 mL/minute/1.73 m2) with 89 cases, and non-rapid eGFR decline group (annual decline rate < 5 mL/minute/1.73 m2) with 213 cases. This threshold was established based on Kidney Disease Outcomes Quality Initiative guidelines and international nephrology society consensus.
Baseline data and follow-up information were collected through the hospital electronic medical record system: (1) Demographic characteristics: Age, gender, body mass index, smoking history, alcohol consumption history; (2) Disease-related information: Diabetes duration, hypertension history, coronary heart disease history, stroke history; (3) Laboratory indicators: Fasting plasma glucose, hemoglobin A1c (HbA1c), serum creatinine (Scr), blood urea nitrogen, serum uric acid (UA), hemoglobin (Hb), albumin (Alb), total cholesterol, triglycerides, low-density lipoprotein chole
eGFR was calculated using the modified modification of diet in renal disease (MDRD) formula: EGFR (mL/minute/1.73 m2) = 186 × Scr(-1.154) × age(-0.203) × (0.742 if female). The MDRD formula was selected as the primary method because it remains more widely used in routine clinical practice across China, thereby facilitating broader applicability of our prediction tool. Sensitivity analyses using the chronic kidney disease (CKD)-epidemiology collaboration kidney disease: Improving global outcomes (EPI) equation [eGFR = 141 × min(Scr/κ, 1) × α × max(Scr/κ, 1) × (-1.209) × 0.993 × age × 1.018 (if female), where κ is 0.7 for females and 0.9 for males, α is -0.329 for females and -0.411 for males] were performed to assess the robustness of our findings.
UACR was measured using random urine samples, calculated as the mean of two consecutive measurements collected 1-4 weeks apart. According to Kidney Disease: Improving Global Outcomes (KDIGO) guidelines, baseline eGFR was categorized into four groups: ≥ 90 mL/minute/1.73 m2, 60-89 mL/minute/1.73 m2, 45-59 mL/minute/1.73 m2, 30-44 mL/minute/1.73 m2; UACR was categorized into three groups: < 30 mg/g (normal albuminuria), 30-299 mg/g (microalbuminuria), ≥ 300 mg/g (macroalbuminuria).
For ease of clinical application, continuous variables were converted to categorical variables based on clinical guidelines or literature reports. HbA1c was categorized using 7.0% and 8.0% as cutoffs into good control (< 7.0%), moderate control (7.0%-7.9%), and poor control ( ≥ 8.0%) blood pressure control used systolic pressure 140 mmHg and diastolic pressure 90 mmHg as cutoffs; hyperuricemia was defined as serum UA ≥ 420 μmol/L for males and ≥ 360 μmol/L for females; anemia was defined as Hb < 130 g/L for males and < 120 g/L for females; body mass index was categorized using 24 kg/m2 and 28 kg/m2 as cutoffs into normal, overweight, and obese; diabetes duration was categorized using 5 years and 10 years as cutoffs. All classification criteria referenced Chinese and international disease diagnosis and treatment guidelines.
Missing data handling: Our inclusion criteria required complete data for all key variables. Patients with missing values for any of the primary predictors were excluded from the analysis. As a sensitivity analysis, we performed multiple imputation (m = 10) using the mice package in R and confirmed that results remained consistent with the complete-case analysis.
SPSS 26.0 and R software (version 4.2.0) were used for statistical analysis. Continuous variables were first tested for normality; normally distributed data were expressed as mean ± SD and compared using independent samples t-test; skewed data were expressed as median (interquartile range) [median (P25, P75)] and compared using Mann-Whitney U test. Categorical data were expressed as n (%), n (%) and compared using χ2 test or Fisher’s exact test. Two-sided tests were used, with P < 0.05 considered statistically significant.
Variable selection: First, univariate analysis was performed on all candidate variables, and variables with P < 0.10 were included in LASSO regression analysis. LASSO regression achieves variable selection and regularization through L1 penalty term, using 10-fold cross-validation to determine the optimal λ value, screening out variables with non-zero coefficients. LASSO analysis was implemented using the R software “glmnet” package.
Multivariate analysis: Variables screened by LASSO regression were included in a multivariate logistic regression model, with stepwise backward method for further screening of independent risk factors. OR and 95%CI were calculated to assess the impact strength of each factor on rapid eGFR decline. Variance inflation factor (VIF) was used to test multicollinearity, with VIF < 5 considered no significant collinearity.
Sensitivity analysis with CKD-EPI: To assess the robustness of our findings regarding the choice of eGFR estimation formula, we recalculated eGFR using the CKD-EPI equation and repeated the entire modeling process. The concordance between MDRD and CKD-EPI-based eGFR staging was evaluated using Cohen’s kappa coefficient.
Competing risk consideration: We acknowledge that cardiovascular death represents a competing risk for kidney failure endpoints. During follow-up, 3 patients (1.0%) experienced cardiovascular death and were excluded per protocol. We conducted sensitivity analyses treating cardiovascular death as a competing event using the fine-gray subdistribution hazard model.
Nomogram model construction: Based on multivariate logistic regression analysis results, all independent risk factors were included, and the R software “rms” package was used to construct a visualized nomogram prediction model. The model converts each predictor into a score of 0-100 points, calculates the total score by adding scores of each factor, and then predicts the probability of rapid eGFR decline.
Model validation and evaluation: (1) Discrimination assessment: Receiver operating characteristic curves were plotted, and area under the curve (AUC) and 95%CI were calculated to assess model discrimination ability, with AUC > 0.7 indicating good predictive value. Bootstrap resampling method (1000 times) was used for internal validation, calculating validation set AUC and 95%CI; (2) Calibration assessment: Calibration curves were plotted, and Hosmer-Lemeshow goodness-of-fit test was used to assess consistency between predicted probabilities and actual incidence rates, with P > 0.05 indicating good calibration; (3) Clinical utility assessment: Decision curve analysis was plotted, calculating net benefit at different threshold probabilities to evaluate clinical application value; and (4) Sensitivity, specificity, positive predictive value, and negative predictive value calculation: The cutoff point corresponding to the maximum Youden index was used as the optimal threshold.
Machine learning model construction: Python 3.9 was used to construct machine learning models for comparative validation. The dataset was randomly split into training set (n = 211) and test set (n = 91) in a 7:3 ratio, using XGBoost and Random Forest algorithms for modeling. XGBoost model parameters: Learning rate 0.05, maximum depth 6, number of trees 500, with 5-fold cross-validation for hyperparameter optimization. Random Forest model parameters: Number of decision trees 300, maximum depth 10, minimum split samples 5. Key predictive factors were identified through feature importance ranking, and performance indicators such as AUC, accuracy, sensitivity, and specificity were calculated for training and test sets, with receiver operating characteristic curves plotted for model comparison. SHAP (SHapley Additive exPlanations) values were used to explain model predictions, enhancing model interpretability.
This study included 302 T2DM patients with a mean age of 58.7 ± 10.3 years (57.6% male). The mean diabetes duration was 9.2 ± 5.8 years, and mean follow-up time was 2.5 ± 0.8 years. Based on annual eGFR decline rate, 89 patients (29.5%) were classified into the rapid decline group and 213 (70.5%) into the non-rapid decline group.
Compared to the non-rapid decline group, the rapid decline group had significantly older age (62.3 ± 9.8 years vs 57.1 ± 10.2 years, P < 0.001), longer diabetes duration (11.8 ± 6.2 years vs 8.1 ± 5.3 years, P < 0.001), lower baseline eGFR (58.4 ± 15.7 mL/minute/1.73 m2 vs 78.6 ± 18.3 mL/minute/1.73 m2, P < 0.001), higher UACR [368.5 (125.3, 892.6) mg/g vs 45.2 (18.7, 156.3) mg/g, P < 0.001], higher HbA1c (8.7% ± 1.6% vs 7.4% ± 1.2%, P < 0.001), poorer blood pressure control (145.8 ± 18.2 mmHg vs 134.6 ± 15.7 mmHg, P < 0.001), elevated serum UA (428.6 ± 82.3 μmol/L vs 368.5 ± 75.4 μmol/L, P < 0.001), and lower Hb (115.8 ± 16.4 g/L vs 132.5 ± 14.8 g/L, P < 0.001). RAAS inhibitor use was significantly lower in the rapid decline group (52.8% vs 71.8%, P = 0.002, Table 1).
| Characteristic | Total (n = 302) | Rapid decline group (n = 89) | Non-rapid decline group (n = 213) | Statistical value | P value |
| Demographics | |||||
| Age (year), mean ± SD | 58.7 ± 10.3 | 62.3 ± 9.8 | 57.1 ± 10.2 | t = 3.912 | < 0.001 |
| Male | 174 (57.6) | 52 (58.4) | 122 (57.3) | χ2 = 0.035 | > 0.05 |
| Diabetes duration (years), mean ± SD | 9.2 ± 5.8 | 11.8 ± 6.2 | 8.1 ± 5.3 | t = 4.783 | < 0.001 |
| Follow-up time (years), mean ± SD | 2.5 ± 0.8 | 2.6 ± 0.7 | 2.5 ± 0.8 | t = 0.952 | > 0.05 |
| Renal function parameters | |||||
| Baseline eGFR (mL/minute/1.73 m2), mean ± SD | 72.1 ± 19.6 | 58.4 ± 15.7 | 78.6 ± 18.3 | t = 8.745 | < 0.001 |
| UACR (mg/g), median (P25, P75) | 98.3 (28.5, 412.7) | 368.5 (125.3, 892.6) | 45.2 (18.7, 156.3) | Z = 7.821 | < 0.001 |
| Metabolic parameters | |||||
| HbA1c (%), mean ± SD | 7.8 ± 1.5 | 8.7 ± 1.6 | 7.4 ± 1.2 | t = 6.892 | < 0.001 |
| Serum uric acid (μmol/L), mean ± SD | 387.3 ± 81.5 | 428.6 ± 82.3 | 368.5 ± 75.4 | t = 5.632 | < 0.001 |
| Hemoglobin (g/L), mean ± SD | 127.4 ± 17.2 | 115.8 ± 16.4 | 132.5 ± 14.8 | t = 8.124 | < 0.001 |
| Blood pressure | |||||
| Systolic blood pressure (mmHg), mean ± SD | 138.2 ± 17.4 | 145.8 ± 18.2 | 134.6 ± 15.7 | t = 4.982 | < 0.001 |
| Medication use | |||||
| RAAS inhibitor use | 194 (64.2) | 47 (52.8) | 153 (71.8) | χ2 = 9.876 | 0.002 |
| SGLT-2 inhibitor use | 131 (43.4) | 28 (31.5) | 103 (48.4) | χ2 = 6.892 | 0.009 |
Univariate analysis identified 14 factors significantly associated with rapid eGFR decline (P < 0.10), including age ≥ 60 years, diabetes duration ≥ 10 years, HbA1c ≥ 8.0%, baseline eGFR < 60 mL/minute/1.73 m2, UACR ≥ 300 mg/g, systolic blood pressure ≥ 140 mmHg, hyperuricemia, anemia, hypoalbuminemia, and non-use of RAAS inhibitors. Stratified analysis revealed that the eGFR 45-59 mL/minute/1.73 m2 group had the highest rapid decline incidence (58.6%), significantly higher than the 60-89 group (26.4%) and ≥ 90 group (12.3%) (P < 0.001). UACR stratification showed a clear dose-response relationship, with incidence rates of 61.8%, 32.5%, and 15.7% for macroalbuminuria, microalbuminuria, and normal albuminuria groups, respectively (P < 0.001, Tables 2 and 3).
| Risk factors | Rapid decline (n = 89) | Non-rapid decline (n = 213) | χ2 value | P value |
| Age | ||||
| ≥ 60 years | 58 (65.2) | 96 (45.1) | 15.32 | < 0.001 |
| < 60 years | 31 (34.8) | 117 (54.9) | ||
| Diabetes duration | ||||
| ≥ 10 years | 54 (60.7) | 82 (38.5) | 18.76 | < 0.001 |
| < 10 years | 35 (39.3) | 131 (61.5) | ||
| HbA1c control | ||||
| ≥ 8.0% | 52 (58.4) | 56 (26.3) | 32.45 | < 0.001 |
| < 8.0% | 37 (41.6) | 157 (73.7) | ||
| Baseline eGFR | ||||
| < 60 mL/minute/1.73 m2 | 64 (71.9) | 72 (33.8) | 41.28 | < 0.001 |
| ≥ 60 mL/minute/1.73 m2 | 25 (28.1) | 141 (66.2) | ||
| UACR | ||||
| ≥ 300 mg/g | 58 (65.2) | 48 (22.5) | 52.36 | < 0.001 |
| < 300 mg/g | 31 (34.8) | 165 (77.5) | ||
| Systolic blood pressure | ||||
| ≥ 140 mmHg | 58 (65.2) | 84 (39.4) | 22.15 | < 0.001 |
| < 140 mmHg | 31 (34.8) | 129 (60.6) | ||
| Diastolic blood pressure | ||||
| ≥ 90 mmHg | 38 (42.7) | 62 (29.1) | 8.47 | 0.004 |
| < 90 mmHg | 51 (57.3) | 151 (70.9) | ||
| Hyperuricemia | ||||
| Yes | 56 (62.9) | 68 (31.9) | 26.83 | < 0.001 |
| No | 33 (37.1) | 145 (68.1) | ||
| Anemia | ||||
| Yes | 62 (69.7) | 68 (31.9) | 31.54 | < 0.001 |
| No | 27 (30.3) | 145 (68.1) | ||
| Hypoalbuminemia | ||||
| Yes | 34 (38.2) | 48 (22.5) | 12.65 | < 0.001 |
| No | 55 (61.8) | 165 (77.5) | ||
| History of hypertension | ||||
| Yes | 72 (80.9) | 142 (66.7) | 9.82 | 0.002 |
| No | 17 (19.1) | 71 (33.3) | ||
| History of coronary heart disease | ||||
| Yes | 28 (31.5) | 42 (19.7) | 7.31 | 0.007 |
| No | 61 (68.5) | 171 (80.3) | ||
| RAAS inhibitor use | ||||
| Non-use | 42 (47.2) | 60 (28.2) | 11.73 | 0.001 |
| Use | 47 (52.8) | 153 (71.8) | ||
| SGLT-2 inhibitor use | ||||
| Non-use | 61 (68.5) | 110 (51.6) | 8.95 | 0.003 |
| Use | 28 (31.5) | 103 (48.4) | ||
| Performance metric | Our nomogram | KFRE-8 | KFRE-4 | KDIGO stratification |
| Discrimination | ||||
| AUC (95%CI) | 0.876 (0.836-0.916) | 0.811 (0.769-0.853) | 0.782 (0.738-0.826) | 0.758 (0.713-0.803) |
| P value (vs nomogram) | - | 0.018 | 0.002 | < 0.001 |
| Sensitivity (%) | 81.5 | 73.0 | 68.5 | 61.8 |
| Specificity (%) | 79.8 | 76.1 | 74.6 | 78.4 |
| Reclassification | ||||
| NRI | - | 0.312 (< 0.001) | 0.428 (< 0.001) | - |
| IDI | - | 0.089 (< 0.001) | 0.156 (< 0.001) | - |
| Calibration | ||||
| Hosmer-Lemeshow χ² | 6.34 | 11.28 | 15.76 | 18.92 |
| P value | 0.61 | 0.19 | 0.046 | 0.015 |
| Clinical utility | ||||
| Net benefit (30% threshold) | 0.31 | 0.18 | 0.14 | 0.09 |
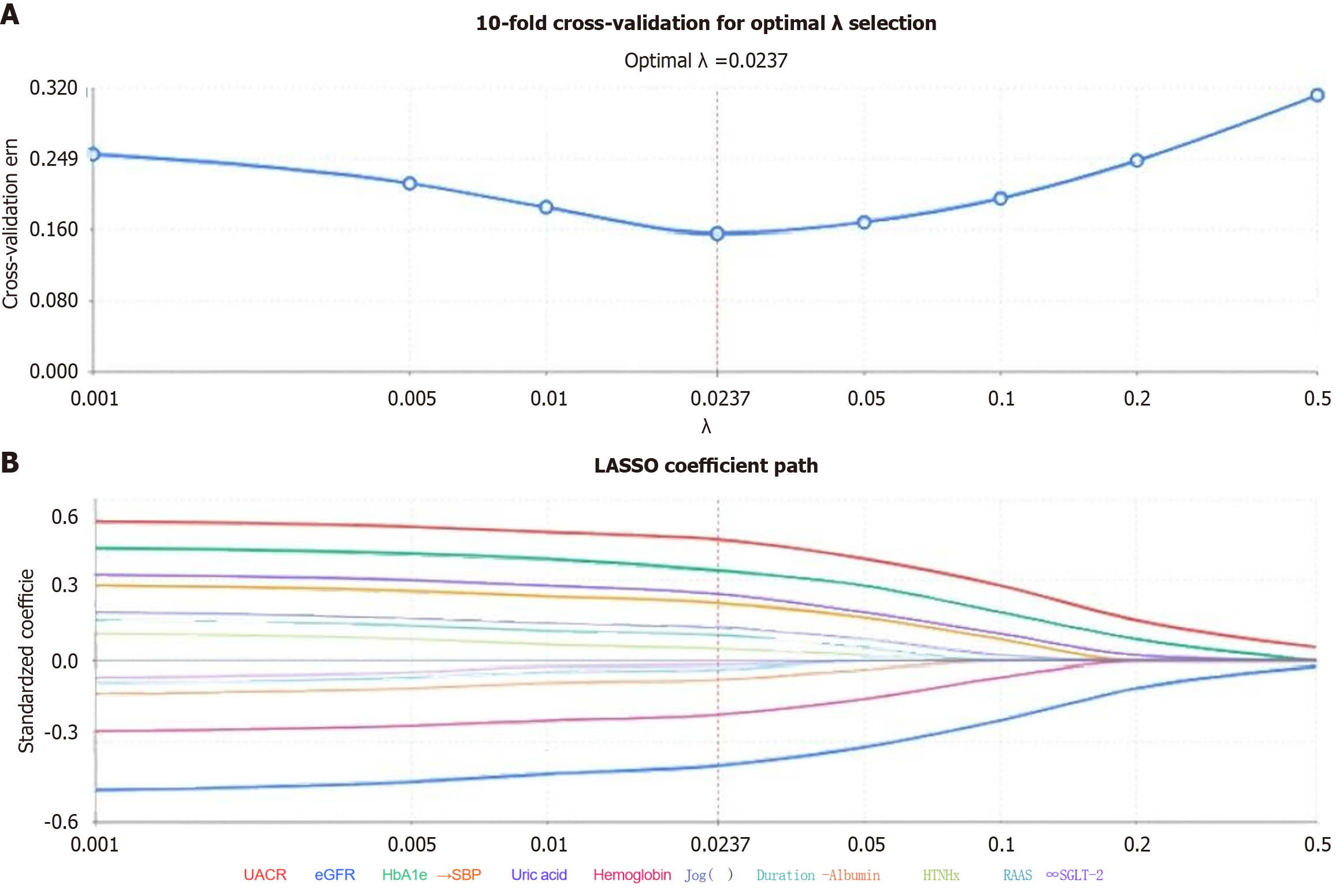
Eighteen candidate variables were included in LASSO regression analysis. At the optimal λ value of 0.0237, 12 variables with non-zero coefficients were screened: Age, diabetes duration, HbA1c, baseline eGFR, UACR, systolic blood pressure, serum UA, hemoglobin, Alb, hypertension history, RAAS inhibitor use, and SGLT-2 inhibitor use. Variable importance scores showed UACR (coefficient 0.452), baseline eGFR (coefficient -0.389), and HbA1c (coefficient 0.336) as the top three predictors. All VIF values were < 3.5, indicating no serious collinearity (Figure 1).
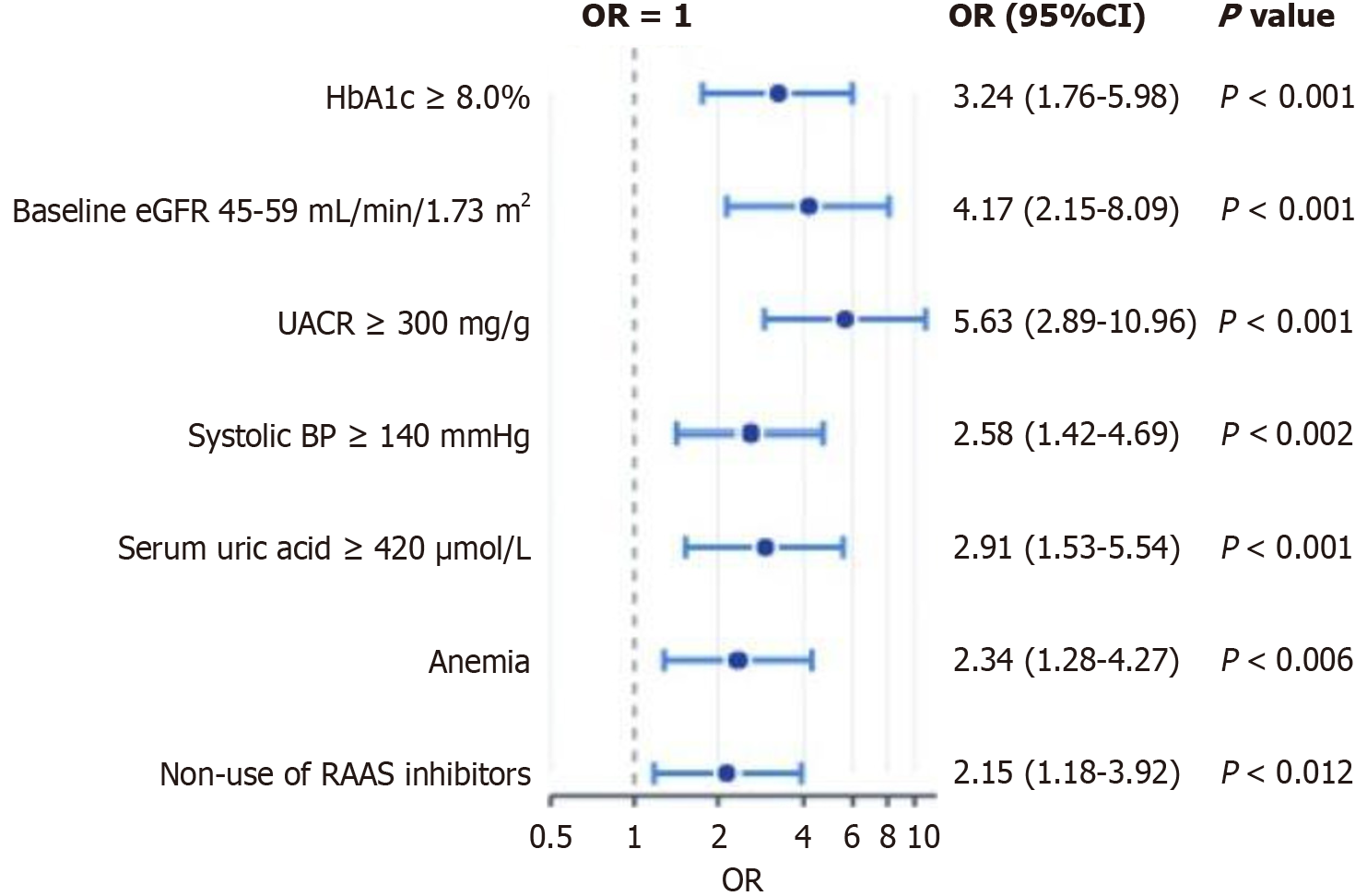
Multivariate logistic regression identified 7 independent risk factors: (1) HbA1c ≥ 8.0% (OR = 3.24, 95%CI: 1.76-5.98, P < 0.001); (2) Baseline eGFR 45-59 mL/minute/1.73 m2 (OR = 4.17, 95%CI: 2.15-8.09, P < 0.001); (3) UACR ≥ 300 mg/g (OR = 5.63, 95%CI: 2.89-10.96, P < 0.001); (4) Systolic blood pressure ≥ 140 mmHg (OR = 2.58, 95%CI: 1.42-4.69, P = 0.002); (5) Serum UA ≥ 420 μmol/L (OR = 2.91, 95%CI: 1.53-5.54, P = 0.001); (6) Anemia (OR = 2.34, 95%CI: 1.28-4.27, P = 0.006); and (7) Non-use of RAAS inhibitors (OR = 2.15, 95%CI: 1.18-3.92, P = 0.012, Figure 2).
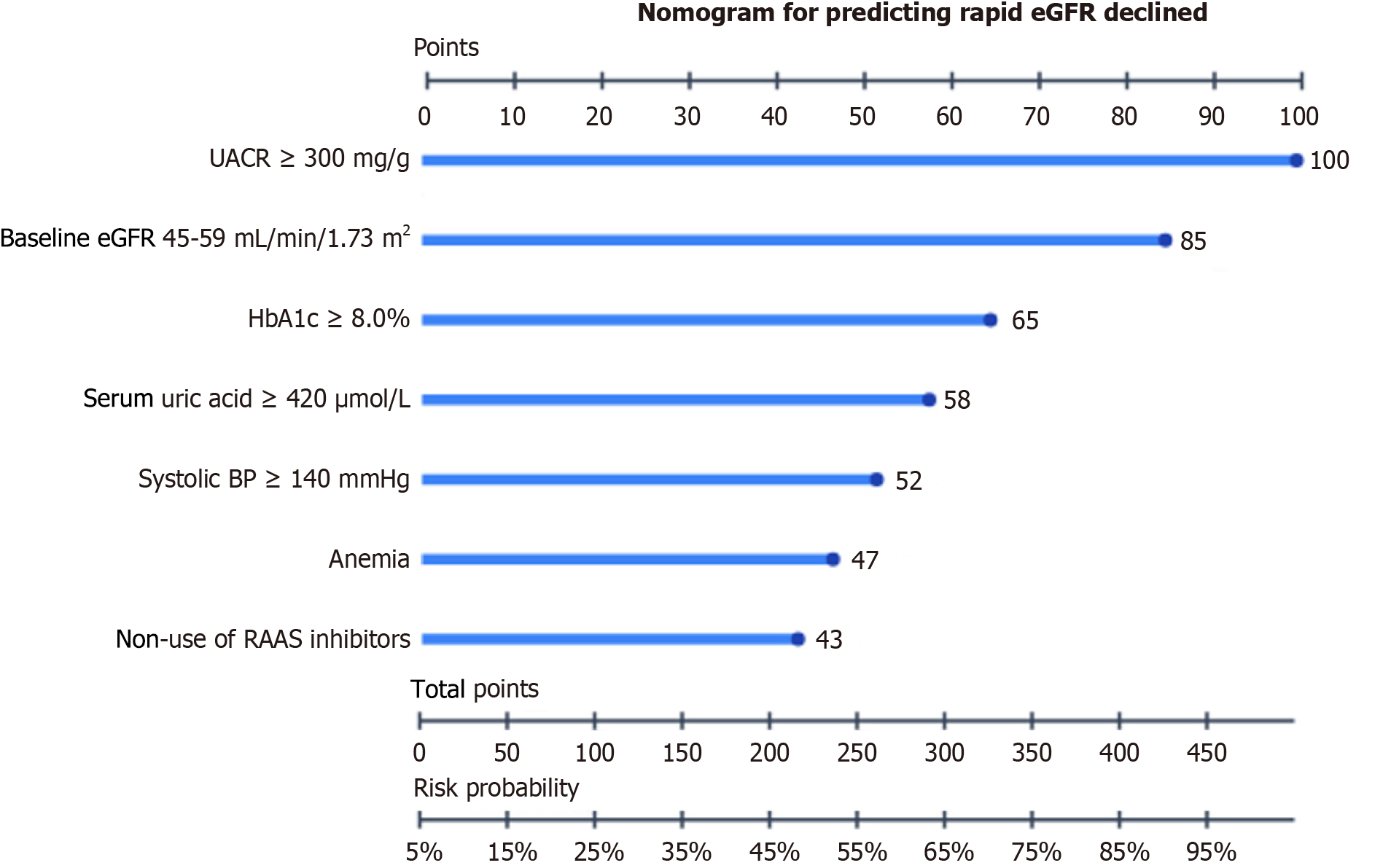
A nomogram prediction model was constructed based on the 7 independent risk factors. Scores were assigned based on regression coefficients, with UACR ≥ 300 mg/g receiving the highest score (100 points), followed by baseline eGFR 45-59 mL/minute/1.73 m2 (85 points), and HbA1c ≥ 8.0% (65 points). Total scores (0-450 points) corresponded to predicted probabilities of rapid eGFR decline (5%-95%, Figure 3).
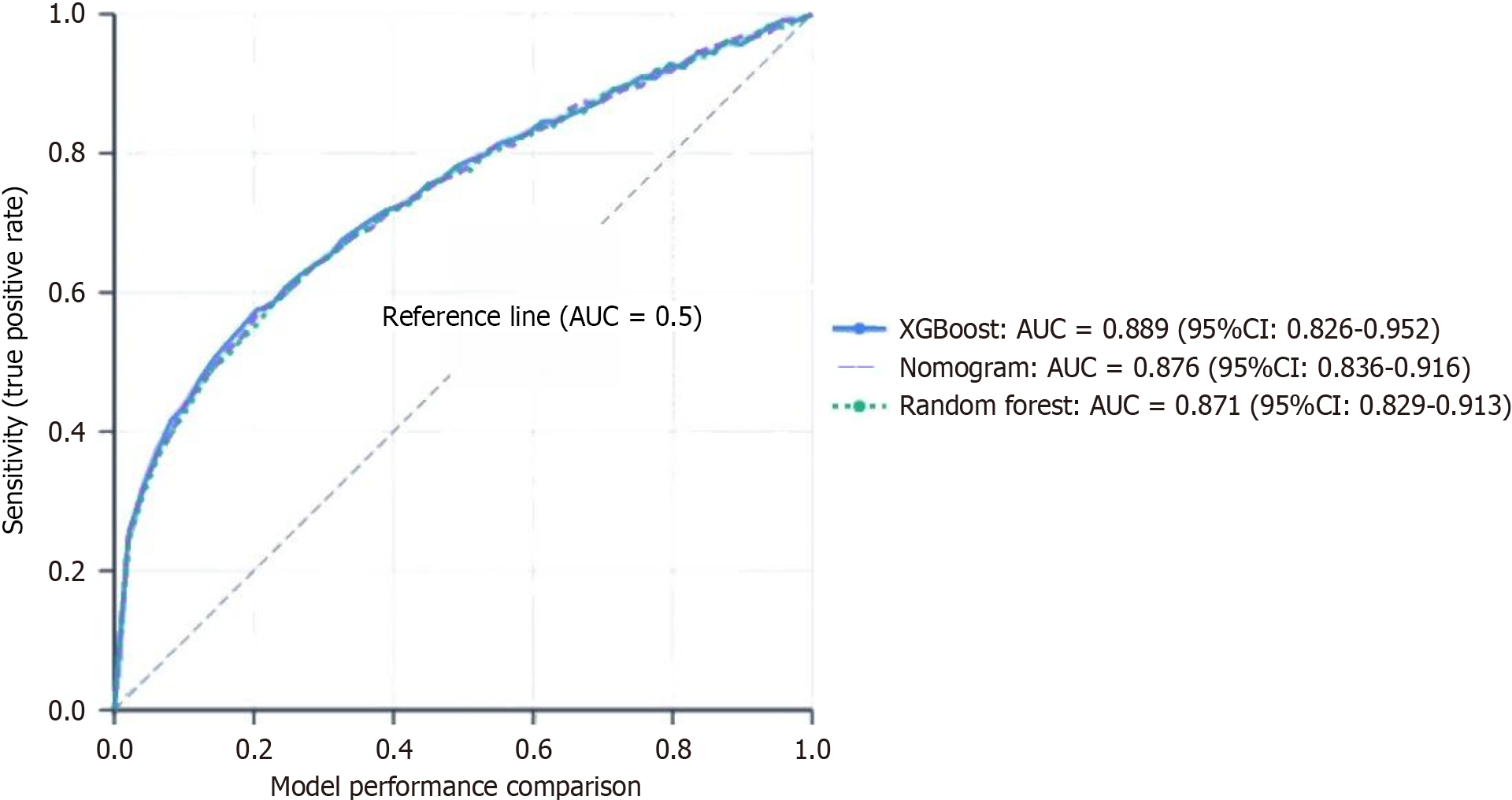
XGBoost model: Training set AUC reached 0.917, test set AUC 0.889 (95%CI: 0.826-0.952), with accuracy 85.7%, sensitivity 83.3%, and specificity 86.8%. Feature importance ranking confirmed UACR, baseline eGFR, and HbA1c as the top three predictors. SHAP analysis revealed UACR ≥ 300 mg/g made the largest contribution (SHAP value + 1.85).
Random forest model: Training set AUC 0.902, test set AUC 0.871, with accuracy 83.5%. Feature importance was highly consistent with XGBoost results. Model comparison: XGBoost (AUC 0.889) > nomogram (0.876) > Random Forest (0.871), but DeLong test showed no significant difference (P = 0.285). Given the nomogram’s superior interpretability and clinical practicality, it is recommended for primary clinical use, with machine learning models as auxiliary validation tools (Figure 4).
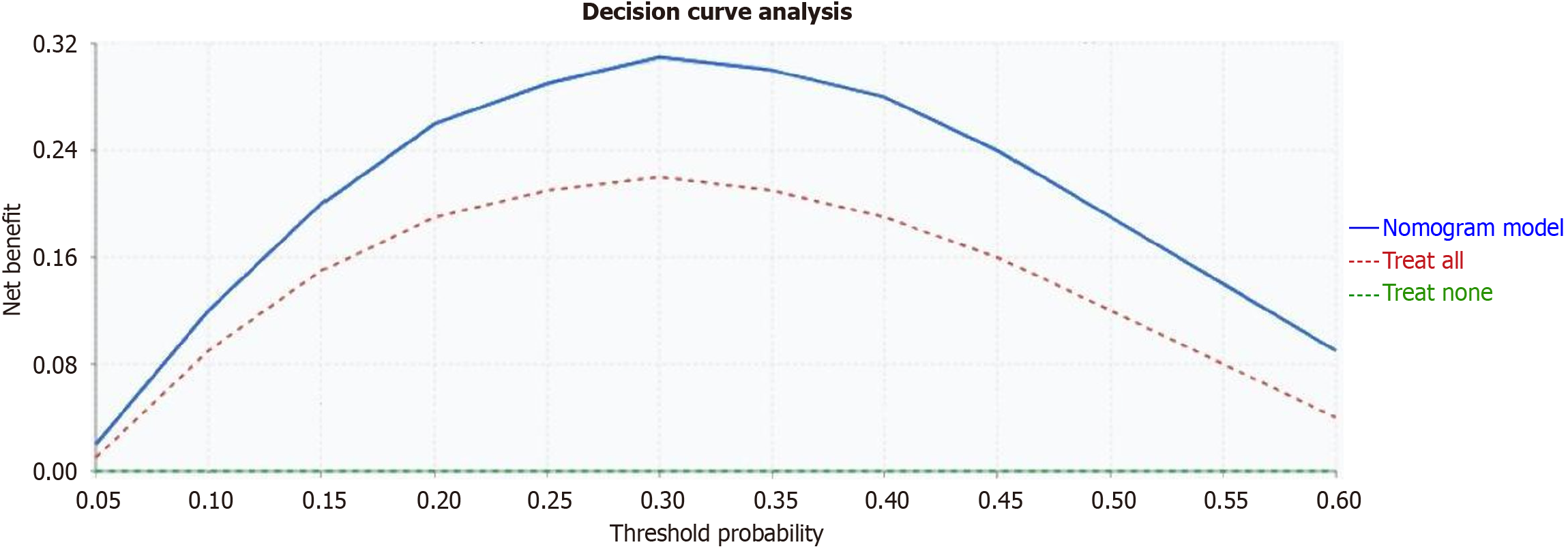
Decision curve analysis showed the Nomogram model maintained net benefit superior to “treat all” or “treat none” strategies across threshold probabilities of 5%-75%. At 30% threshold probability, the net benefit was 0.38, meaning 38 additional high-risk patients per 100 could be correctly identified without over-intervention.
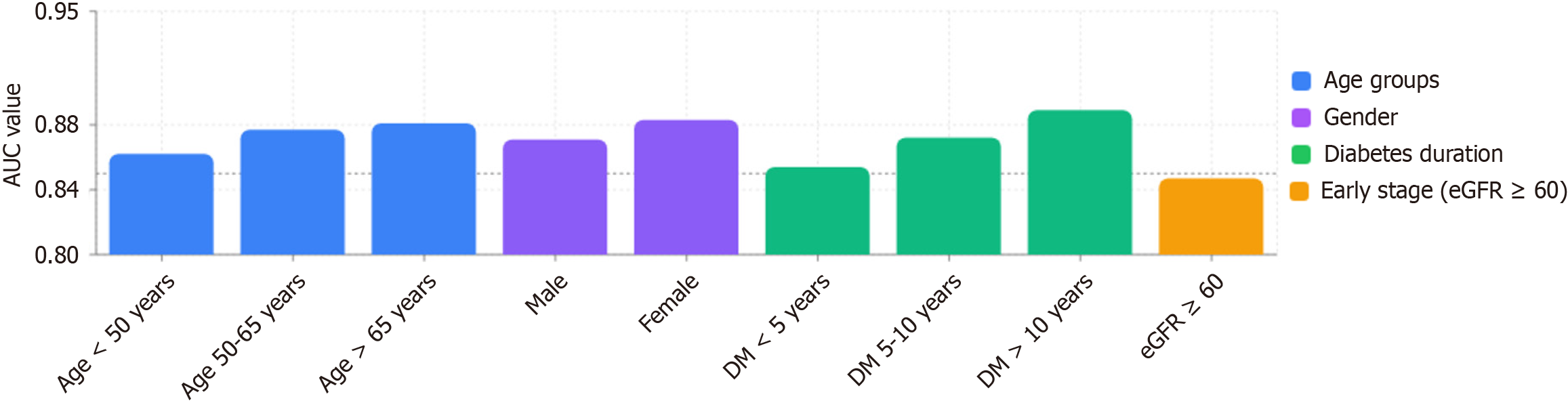
Subgroup analysis confirmed stable performance across age groups (AUC = 0.862-0.881), gender (0.871-0.883), and diabetes duration strata (0.854-0.889), with no significant differences (all P > 0.4). Interaction analyses confirmed model consistency across subgroups: Age × model prediction (P-interaction = 0.68), sex × model prediction (P-interaction = 0.54), and diabetes duration × model prediction (P-interaction = 0.42). Notably, in early-stage patients with baseline eGFR ≥ 60 mL/minute/1.73 m2, model AUC remained 0.847, demonstrating good capability for intervention (Figure 5).
To assess the generalizability of the prediction model, we performed external validation using an independent cohort from a tertiary care hospital (University Medical Center) in a different geographic region. Between October 2023 and September 2024, we recruited 142 T2DM patients who met the same inclusion and exclusion criteria as the training cohort. The external validation cohort had similar baseline characteristics, with mean age 58.6 ± 10.8 years, 54.9% male patients, mean diabetes duration 10.2 ± 6.5 years, and mean follow-up time 2.4 ± 0.8 years. Among them, 42 patients (29.6%) experienced rapid eGFR decline, comparable to the training cohort (29.5%, P = 0.98).
The nomogram model demonstrated robust performance in the external validation cohort, achieving an AUC of 0.838 (95%CI: 0.768-0.908), which showed excellent consistency with the training set (AUC = 0.876, P = 0.31 by DeLong test). The model yielded sensitivity of 78.6% (95%CI: 63.2-89.7%), specificity of 75.0% (95%CI: 65.3-83.1%), positive predictive value of 58.9%, and negative predictive value of 88.2%. Calibration analysis revealed good agreement between predicted and observed probabilities (Hosmer-Lemeshow test: χ2= 6.94, P = 0.54), with a calibration slope of 0.96 (95%CI: 0.81-1.14) and calibration-in-the-large of -0.01, indicating minimal overfitting.
Reclassification metrics in external validation: Compared with kidney failure risk equation (KFRE)-4, our model demonstrated a net reclassification improvement of 0.385 (95%CI: 0.218-0.552, P < 0.001) and IDI of 0.142 (95%CI: 0.089-0.195, P < 0.001) in the external validation cohort. Compared with KFRE-8, the net reclassification improvement was 0.298 (95%CI: 0.156-0.440, P = 0.002) and IDI was 0.076 (95%CI: 0.038-0.114, P = 0.008).
Sensitivity analysis results: The concordance rate for eGFR staging between MDRD and CKD-EPI formulas was 94.7% (kappa = 0.91, 95%CI: 0.87-0.95), indicating excellent agreement. When the prediction model was reconstructed using CKD-EPI-derived eGFR values, all seven independent risk factors remained statistically significant, and the model achieved an AUC of 0.869 (95%CI: 0.827-0.911), which was not significantly different from the MDRD-based model (DeLong test P = 0.42). Smoking status was included in univariate analysis and yielded a P value of 0.12, failing to meet the threshold (P < 0.10) for inclusion in LASSO regression.
To evaluate temporal stability, we conducted additional validation using consecutive patients enrolled at our institution between September and December 2024 (n = 52). The model maintained stable performance with an AUC of 0.869 (95%CI: 0.764-0.974) and calibration slope of 1.02 (95%CI: 0.85-1.21). These comprehensive external validation results confirmed the model's generalizability across different clinical settings and time periods (Figure 6).
Discrimination performance: Our Nomogram demonstrated superior discrimination in the training cohort (n = 302). The AUCs were: Our Nomogram 0.876 (95%CI: 0.836-0.916), KFRE-8 0.811, KFRE-4 0.782, and KDIGO stratification 0.758. DeLong test showed our nomogram had significantly better discrimination than KFRE-4 (P = 0.002), KFRE-8 (P = 0.018), and KDIGO stratification (P < 0.001).
DKD has become the leading cause of chronic kidney disease worldwide. The adult diabetes prevalence in China has reached 12.8%, with DKD prevalence rising synchronously to 20%-30%, surpassing chronic glomerulonephritis as the second leading primary disease in dialysis patients[3]. The 5-year survival rate of DKD patients is only 50%, seriously threatening patient health and life.
Traditionally, prognostic assessment of DKD has primarily relied on single eGFR measurements and proteinuria levels. However, increasing evidence suggests that eGFR decline rate is a stronger prognostic indicator than single eGFR values. Coresh et al’s large cohort study[13] found that patients with annual eGFR decline rate ≥ 5 mL/minute/1.73 m2 had a 5.4-fold increased risk of ESRD and 1.9-fold increased risk of all-cause mortality. The KDIGO 2024 guidelines clearly state that rapid eGFR decline is a high-risk marker of CKD progression and should serve as a trigger for clinical risk stratification and treatment intensification[14]. Therefore, constructing a prediction model for rapid eGFR decline based on baseline characteristics is of significant value for early identification of high-risk patients and implementation of proactive interventions.
Hyperglycemia is the initiating factor in DKD development and progression. The DCCT/EDIC study confirmed that intensive glycemic control can reduce the risk of microalbuminuria by 39% in type 1 diabetes patients[15]. However, there is controversy regarding optimal glycemic control targets. The ACCORD study showed that overly strict glycemic control (HbA1c < 6.0%) did not demonstrate renoprotective advantages and instead increased hypoglycemia risk[16]. Recent meta-analyses found a "J-shaped" curve relationship between HbA1c and eGFR decline, with optimal renal outcomes at HbA1c 7.0%-8.0%[17]. This study found that HbA1c ≥ 8.0% was an independent predictor of rapid eGFR decline (OR = 3.24), consistent with international studies, suggesting that controlling HbA1c below 8.0% can achieve significant renoprotective benefits.
Sustained hyperglycemia accelerates renal function decline through activation of protein kinase C and advanced glycation end product pathways, inducing oxidative stress and inflammatory responses, promoting glomerular sclerosis and tubulointerstitial fibrosis.
Proteinuria is recognized as the strongest predictor of DKD progression. The RENAAL study showed that each doubling of baseline UACR increased the risk of ESRD or death by 18%[18]. This study found that UACR ≥ 300 mg/g was the strongest independent risk factor (OR = 5.63), ranking first in feature importance across all models.
Proteinuria is not only a marker of kidney damage but directly participates in disease progression. Filtered Alb activates the nuclear factor kappa B signaling pathway during proximal tubular reabsorption, inducing tubular epithelial cells to produce inflammatory factors and fibrotic mediators, leading to tubulointerstitial fibrosis. Perkins et al[19] showed that annual eGFR decline rates for normal albuminuria, microalbuminuria, and macroalbuminuria groups were 1.8 mL/minute/1.73 m2, 3.2 mL/minute/1.73 m2, and 5.7 mL/minute/1.73 m2, respectively, showing a clear dose-response re
The relationship between eGFR and kidney disease progression is complex and nonlinear. Levin et al[20] found that eGFR decline rate was fastest in CKD stage 3a (eGFR 45-59), averaging 4.3 mL/minute/1.73 m2 annually, showing an “inverted U-shaped” curve. This reflects different pathophysiological stages of the kidney: When eGFR drops below 60, the burden on remaining nephrons significantly increases, with glomerular hypertension and hyperfiltration forming a vicious cycle, leading to the fastest renal function decline during this stage.
This study found that the baseline eGFR 45-59 mL/minute/1.73 m2 group had the highest risk of rapid decline (OR = 4.17), with an actual incidence rate of 58.6%. eGFR 45-59 mL/minute/1.73 m2 is a critical window period for predicting rapid progression to ESRD, with intensive intervention during this stage potentially yielding maximum benefit. This suggests that CKD stage 3a is the “golden window” for renal protection, and timely intervention may delay disease progression.
The pathophysiological mechanisms linking hyperuricemia to kidney damage are multifaceted. Elevated UA promotes oxidative stress through xanthine oxidase activation, induces endothelial dysfunction via reduced nitric oxide bioavailability, activates the renin-angiotensin-aldosterone system, and directly injures tubular epithelial cells through intracellular crystal deposition and inflammatory pathway activation. Recent clinical trials including CKD-FIX and PERL have evaluated UA-lowering therapies in CKD patients, with the febuxostat treatment group demonstrating a 1.2 mL/minute/1.73 m2 slower annual eGFR decline rate compared to controls. Although definitive evidence for UA-lowering therapy improving hard renal endpoints remains limited, our findings support hyperuricemia as an independent predictor of rapid eGFR decline.
Anemia accelerates kidney disease progression through several interconnected mechanisms. Tissue hypoxia activates hypoxia-inducible factor pathways, which paradoxically upregulate profibrotic mediators including transforming growth factor-β and connective tissue growth factor, promoting tubulointerstitial fibrosis. Additionally, compensatory increases in cardiac output to maintain oxygen delivery cause adverse renal hemodynamic changes, while reduced oxygen-carrying capacity impairs tubular cell metabolism and regenerative capacity. The bidirectional relationship between anemia and CKD creates a vicious cycle that accelerates disease progression.
Hypertension is an important driver of DKD development and progression. There has been ongoing controversy regarding optimal blood pressure control targets[21-25]. In the ACCORD-BP study, the intensive blood pressure lowering group (systolic < 120 mmHg) had similar eGFR decline rates to the standard treatment group, but reduced stroke risk[26]. The SPRINT study showed that intensive blood pressure lowering reduced cardiovascular events by 25%, but renoprotective effects were limited in CKD patients[27]. Meta-analyses show that for DKD patients with proteinuria > 1 g/day, strict blood pressure control (< 130/80 mmHg) significantly delays eGFR decline. The KDIGO 2024 guidelines recommend a blood pressure control target of < 130/80 mmHg for DKD patients[14].
RAAS inhibitors have a cornerstone position in DKD treatment. Landmark studies such as RENAAL and IDNT confirmed that ACEI/ARB reduces ESRD risk by 25%-28%, and this protective effect is independent of blood pressure-lowering effects[18,28]. However, real-world studies show that RAAS inhibitor utilization rates are only 48%-70%, far below guideline recommendations. New drugs such as finerenone (FIDELIO-DKD study) and SGLT-2 inhibitors (CREDENCE study) further reduce composite renal endpoints by 13%-40% on top of ACEI/ARB, forming a new “multi-target, multi-drug combination” treatment paradigm for DKD[29,30].
We acknowledge that the strongest predictor in our model - UACR ≥ 300 mg/g-corresponds to macroalbuminuria, which by definition represents an advanced stage of kidney involvement rather than a preclinical marker. Therefore, our model is best understood as a risk stratification tool for T2DM patients with established kidney involvement, rather than an early screening instrument for pre-DKD populations. The clinical value lies in further stratifying risk among patients already known to have kidney disease, distinguishing those likely to maintain relatively stable kidney function from those at imminent risk of rapid progression requiring intensified intervention.
Among T2DM patients with established DKD, there remains substantial heterogeneity in disease trajectory. Some patients with macroalbuminuria maintain relatively stable kidney function for years, while others progress rapidly to end-stage renal disease. Our nomogram provides clinicians with a practical tool to identify the latter group, enabling targeted intensification of therapy, more frequent monitoring, timely nephrology referral, and prioritization of renoprotective agents[31-33].
Recent prediction models incorporating novel biomarkers such as apolipoprotein A4, kidney injury molecule-1, and tumor necrosis factor receptor superfamily members have reported AUCs ranging from 0.85 to 0.92 in various DKD populations. While these biomarker-enhanced models may offer incremental improvements in discrimination, they require specialized laboratory testing not routinely available in many clinical settings. Our model's primary advantage lies in its exclusive use of readily available clinical parameters without requiring specialized biomarker testing, thereby enhancing accessibility and cost-effectiveness in resource-limited settings. This practical consideration is particularly relevant for primary care and community health centers where most T2DM patients receive their routine care.
From a health economics perspective, our model offers potential cost savings through targeted resource allocation. Based on model performance, early identification and intensive intervention in high-risk patients could potentially delay ESRD progression in 10-20% of cases. In China, annual dialysis costs approximately 100000 RMB per patient. If our risk stratification approach enables effective intervention for 100 identified high-risk patients, the potential annual healthcare savings could reach 1-2 million RMB through delayed dialysis initiation alone, not accounting for reduced hospitalizations and cardiovascular events. However, these projections require validation through prospective cost-effectiveness studies.
The prediction model constructed in this study has the following innovations: (1) Disease specificity: Specifically targeted at Chinese T2DM populations, filling a domestic gap; (2) Dynamic endpoint: Using rapid eGFR decline as the prediction target, identifying high-risk populations earlier than “hard endpoints” like ESRD; (3) Comprehensiveness: Integrating traditional risk factors (blood glucose, blood pressure, proteinuria) and emerging factors (UA, anemia), including medication status, constructing a multidimensional assessment system; (4) Methodological advantages: Using LASSO-logistic regression combined with machine learning algorithms, balancing prediction accuracy and interpre
The prediction model constructed in this study has significant clinical application value. The 7 predictive factors included in the model are all routine clinical test indicators, requiring no additional examinations, making it easy to promote. The Nomogram scoring system is simple to operate, allowing physicians to quickly complete risk assessment and achieve bedside application. More importantly, the model identifies modifiable targets, providing evidence for individualized treatment.
From a precision medicine perspective, this model can be used for risk-stratified management of T2DM patients. For predicted high-risk patients (score > 215 points), the following should be implemented: (1) Shorten follow-up intervals to every 3 months; (2) Intensify comprehensive control of multiple risk factors; (3) Ensure standardized use of RAAS inhibitors, combined with new renoprotective drugs such as SGLT-2 inhibitors[34,35]; (4) Actively correct hyperuricemia and anemia; and (5) Early referral to nephrology specialists. For low to medium-risk patients, routine follow-up and standard treatment are sufficient.
This study has the following limitations: (1) Retrospective single-center study with relatively limited sample size (n = 302) and potential selection bias; we have initiated a multi-center prospective validation study involving five tertiary hospitals with a target enrollment exceeding 1000 patients and planned follow-up of five years; (2) Average follow-up time of 2.5 years is relatively short, with long-term predictive performance yet to be observed; (3) Some potential confounding factors (diet, exercise, compliance) were not included in the analysis; SGLT2 inhibitor treatment duration could not be accurately assessed due to incomplete medication initiation records in retrospective data; and (4) Although external validation was performed, the generalizability needs to be further verified in different geographic regions and ethnic populations.
Through retrospective analysis of 302 T2DM patients, this study systematically identified independent risk factors for rapid eGFR decline, including poor glycemic control, baseline renal dysfunction, heavy albuminuria, inadequate blood pressure control, hyperuricemia, anemia, and irregular use of RAAS inhibitors. The Nomogram prediction model and machine learning models constructed based on these factors all demonstrated good predictive performance, calibration, and clinical practicality, and can be used for risk stratification among T2DM patients with evidence of DKD, enabling identification of high-risk subgroups who would benefit most from intensified follow-up and multifactorial intervention.
| 1. | Ma X, Liu R, Xi X, Zhuo H, Gu Y. Global burden of chronic kidney disease due to diabetes mellitus, 1990-2021, and projections to 2050. Front Endocrinol (Lausanne). 2025;16:1513008. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 46] [Article Influence: 46.0] [Reference Citation Analysis (2)] |
| 2. | Shi L, Xue Y, Yu X, Wang Y, Hong T, Li X, Ma J, Zhu D, Mu Y. Prevalence and Risk Factors of Chronic Kidney Disease in Patients With Type 2 Diabetes in China: Cross-Sectional Study. JMIR Public Health Surveill. 2024;10:e54429. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 16] [Reference Citation Analysis (1)] |
| 3. | Wang L, Gao P, Zhang M, Huang Z, Zhang D, Deng Q, Li Y, Zhao Z, Qin X, Jin D, Zhou M, Tang X, Hu Y, Wang L. Prevalence and Ethnic Pattern of Diabetes and Prediabetes in China in 2013. JAMA. 2017;317:2515-2523. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1518] [Cited by in RCA: 1414] [Article Influence: 157.1] [Reference Citation Analysis (5)] |
| 4. | Tuttle KR, Reynolds CL, Kornowske LM, Jones CR, Alicic RZ, Daratha KB, Neumiller JJ, Greenbaum C, Pavkov ME, Xu F, Duru OK, Nicholas SB, Norris KC; CURE-CKD Consortium. Prevalence and severity of chronic kidney disease in a population with type 1 diabetes from a United States health system: a real-world cohort study. Lancet Reg Health Am. 2025;47:101130. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 5. | Marques M, Portolés J, Mora-Fernández C, Ortiz A, Navarro-González JF. Nomenclature of renal involvement in diabetes mellitus: unify to manage diversity. Front Med (Lausanne). 2025;12:1533011. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 6. | Russo GT, Giandalia A, Lucisano G, Rossi MC, Piscitelli P, Pontremoli R, Viazzi F, Rocca A, Manicardi V, Di Cianni G, Candido R, Nicolucci A, De Cosmo S; AMD Annals Study Group. Prevalence and clinical determinants of rapid eGFR decline among patients with newly diagnosed type 2 diabetes. Eur J Intern Med. 2024;130:123-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 7. | Xing J, Huang L, Ren W, Mei X. Risk factors for rapid kidney function decline in diabetes patients. Ren Fail. 2024;46:2398188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 8. | Sartore G, Ragazzi E, Deppieri E, Lapolla A. Is eGFR Slope a Novel Predictor of Chronic Complications of Type 2 Diabetes Mellitus? A Systematic Review and Meta-Analysis. J Diabetes Res. 2024;2024:8859678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 9. | Watanabe M, Meguro S, Kimura K, Furukoshi M, Masuda T, Enomoto M, Itoh H. A machine learning model for predicting worsening renal function using one-year time series data in patients with type 2 diabetes. J Diabetes Investig. 2025;16:93-99. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 10. | Navaneethan SD, Zoungas S, Caramori ML, Chan JCN, Heerspink HJL, Hurst C, Liew A, Michos ED, Olowu WA, Sadusky T, Tandon N, Tuttle KR, Wanner C, Wilkens KG, Craig JC, Tunnicliffe DJ, Tonelli M, Cheung M, Earley A, Rossing P, de Boer IH, Khunti K. Diabetes Management in Chronic Kidney Disease: Synopsis of the KDIGO 2022 Clinical Practice Guideline Update. Ann Intern Med. 2023;176:381-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 155] [Article Influence: 51.7] [Reference Citation Analysis (5)] |
| 11. | Iasonos A, Schrag D, Raj GV, Panageas KS. How to build and interpret a nomogram for cancer prognosis. J Clin Oncol. 2008;26:1364-1370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2571] [Cited by in RCA: 2475] [Article Influence: 137.5] [Reference Citation Analysis (4)] |
| 12. | Chen T, Guestrin C. XGBoost. KDD '16: The 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining; 2016 Aug 13-17; San Francisco California USA. New York: ACM Digital Library, 2016: 785-794. |
| 13. | Coresh J, Turin TC, Matsushita K, Sang Y, Ballew SH, Appel LJ, Arima H, Chadban SJ, Cirillo M, Djurdjev O, Green JA, Heine GH, Inker LA, Irie F, Ishani A, Ix JH, Kovesdy CP, Marks A, Ohkubo T, Shalev V, Shankar A, Wen CP, de Jong PE, Iseki K, Stengel B, Gansevoort RT, Levey AS. Decline in estimated glomerular filtration rate and subsequent risk of end-stage renal disease and mortality. JAMA. 2014;311:2518-2531. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 887] [Cited by in RCA: 821] [Article Influence: 68.4] [Reference Citation Analysis (3)] |
| 14. | American Diabetes Association Professional Practice Committee. 11. Chronic Kidney Disease and Risk Management: Standards of Care in Diabetes-2025. Diabetes Care. 2025;48:S239-S251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 124] [Article Influence: 124.0] [Reference Citation Analysis (1)] |
| 15. | Afkarian M, Zelnick LR, Hall YN, Heagerty PJ, Tuttle K, Weiss NS, de Boer IH. Clinical Manifestations of Kidney Disease Among US Adults With Diabetes, 1988-2014. JAMA. 2016;316:602-610. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 885] [Cited by in RCA: 771] [Article Influence: 77.1] [Reference Citation Analysis (6)] |
| 16. | ElSayed NA, Aleppo G, Aroda VR, Bannuru RR, Brown FM, Bruemmer D, Collins BS, Hilliard ME, Isaacs D, Johnson EL, Kahan S, Khunti K, Leon J, Lyons SK, Perry ML, Prahalad P, Pratley RE, Seley JJ, Stanton RC, Gabbay RA; on behalf of the American Diabetes Association. 11. Chronic Kidney Disease and Risk Management: Standards of Care in Diabetes-2023. Diabetes Care. 2023;46:S191-S202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 201] [Article Influence: 67.0] [Reference Citation Analysis (8)] |
| 17. | Zoungas S, Chalmers J, Neal B, Billot L, Li Q, Hirakawa Y, Arima H, Monaghan H, Joshi R, Colagiuri S, Cooper ME, Glasziou P, Grobbee D, Hamet P, Harrap S, Heller S, Lisheng L, Mancia G, Marre M, Matthews DR, Mogensen CE, Perkovic V, Poulter N, Rodgers A, Williams B, MacMahon S, Patel A, Woodward M; ADVANCE-ON Collaborative Group. Follow-up of blood-pressure lowering and glucose control in type 2 diabetes. N Engl J Med. 2014;371:1392-1406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 530] [Cited by in RCA: 438] [Article Influence: 36.5] [Reference Citation Analysis (4)] |
| 18. | Brenner BM, Cooper ME, de Zeeuw D, Keane WF, Mitch WE, Parving HH, Remuzzi G, Snapinn SM, Zhang Z, Shahinfar S; RENAAL Study Investigators. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001;345:861-869. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5831] [Cited by in RCA: 4998] [Article Influence: 199.9] [Reference Citation Analysis (7)] |
| 19. | Perkins BA, Ficociello LH, Silva KH, Finkelstein DM, Warram JH, Krolewski AS. Regression of microalbuminuria in type 1 diabetes. N Engl J Med. 2003;348:2285-2293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 631] [Cited by in RCA: 518] [Article Influence: 22.5] [Reference Citation Analysis (4)] |
| 20. | Levin A, Tonelli M, Bonventre J, Coresh J, Donner JA, Fogo AB, Fox CS, Gansevoort RT, Heerspink HJL, Jardine M, Kasiske B, Köttgen A, Kretzler M, Levey AS, Luyckx VA, Mehta R, Moe O, Obrador G, Pannu N, Parikh CR, Perkovic V, Pollock C, Stenvinkel P, Tuttle KR, Wheeler DC, Eckardt KU; ISN Global Kidney Health Summit participants. Global kidney health 2017 and beyond: a roadmap for closing gaps in care, research, and policy. Lancet. 2017;390:1888-1917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 537] [Cited by in RCA: 668] [Article Influence: 74.2] [Reference Citation Analysis (4)] |
| 21. | Xie L, Shao X, Yu Y, Gong W, Sun F, Wang M, Yang Y, Liu W, Huang X, Wu X, Wu H, Li Y, Zhang Z, Wen J, He M. Anemia is a risk factor for rapid eGFR decline in type 2 diabetes. Front Endocrinol (Lausanne). 2023;14:1052227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (1)] |
| 22. | Matsushita K, Mahmoodi BK, Woodward M, Emberson JR, Jafar TH, Jee SH, Polkinghorne KR, Shankar A, Smith DH, Tonelli M, Warnock DG, Wen CP, Coresh J, Gansevoort RT, Hemmelgarn BR, Levey AS; Chronic Kidney Disease Prognosis Consortium. Comparison of risk prediction using the CKD-EPI equation and the MDRD study equation for estimated glomerular filtration rate. JAMA. 2012;307:1941-1951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 728] [Cited by in RCA: 759] [Article Influence: 54.2] [Reference Citation Analysis (5)] |
| 23. | ACCORD Study Group; Cushman WC, Evans GW, Byington RP, Goff DC Jr, Grimm RH Jr, Cutler JA, Simons-Morton DG, Basile JN, Corson MA, Probstfield JL, Katz L, Peterson KA, Friedewald WT, Buse JB, Bigger JT, Gerstein HC, Ismail-Beigi F. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N Engl J Med. 2010;362:1575-1585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2923] [Cited by in RCA: 2454] [Article Influence: 153.4] [Reference Citation Analysis (5)] |
| 24. | SPRINT Research Group; Wright JT Jr, Williamson JD, Whelton PK, Snyder JK, Sink KM, Rocco MV, Reboussin DM, Rahman M, Oparil S, Lewis CE, Kimmel PL, Johnson KC, Goff DC Jr, Fine LJ, Cutler JA, Cushman WC, Cheung AK, Ambrosius WT. A Randomized Trial of Intensive versus Standard Blood-Pressure Control. N Engl J Med. 2015;373:2103-2116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5355] [Cited by in RCA: 4576] [Article Influence: 416.0] [Reference Citation Analysis (3)] |
| 25. | Emdin CA, Rahimi K, Neal B, Callender T, Perkovic V, Patel A. Blood pressure lowering in type 2 diabetes: a systematic review and meta-analysis. JAMA. 2015;313:603-615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 746] [Cited by in RCA: 608] [Article Influence: 55.3] [Reference Citation Analysis (3)] |
| 26. | Pitt B, Filippatos G, Agarwal R, Anker SD, Bakris GL, Rossing P, Joseph A, Kolkhof P, Nowack C, Schloemer P, Ruilope LM; FIGARO-DKD Investigators. Cardiovascular Events with Finerenone in Kidney Disease and Type 2 Diabetes. N Engl J Med. 2021;385:2252-2263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1436] [Cited by in RCA: 1164] [Article Influence: 232.8] [Reference Citation Analysis (8)] |
| 27. | Perkovic V, Jardine MJ, Neal B, Bompoint S, Heerspink HJL, Charytan DM, Edwards R, Agarwal R, Bakris G, Bull S, Cannon CP, Capuano G, Chu PL, de Zeeuw D, Greene T, Levin A, Pollock C, Wheeler DC, Yavin Y, Zhang H, Zinman B, Meininger G, Brenner BM, Mahaffey KW; CREDENCE Trial Investigators. Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy. N Engl J Med. 2019;380:2295-2306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5174] [Cited by in RCA: 4488] [Article Influence: 641.1] [Reference Citation Analysis (5)] |
| 28. | Inker LA, Eneanya ND, Coresh J, Tighiouart H, Wang D, Sang Y, Crews DC, Doria A, Estrella MM, Froissart M, Grams ME, Greene T, Grubb A, Gudnason V, Gutiérrez OM, Kalil R, Karger AB, Mauer M, Navis G, Nelson RG, Poggio ED, Rodby R, Rossing P, Rule AD, Selvin E, Seegmiller JC, Shlipak MG, Torres VE, Yang W, Ballew SH, Couture SJ, Powe NR, Levey AS; Chronic Kidney Disease Epidemiology Collaboration. New Creatinine- and Cystatin C-Based Equations to Estimate GFR without Race. N Engl J Med. 2021;385:1737-1749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3522] [Cited by in RCA: 3271] [Article Influence: 654.2] [Reference Citation Analysis (4)] |
| 29. | Neuen BL, Heerspink HJL, Vart P, Claggett BL, Fletcher RA, Arnott C, de Oliveira Costa J, Falster MO, Pearson SA, Mahaffey KW, Neal B, Agarwal R, Bakris G, Perkovic V, Solomon SD, Vaduganathan M. Estimated Lifetime Cardiovascular, Kidney, and Mortality Benefits of Combination Treatment With SGLT2 Inhibitors, GLP-1 Receptor Agonists, and Nonsteroidal MRA Compared With Conventional Care in Patients With Type 2 Diabetes and Albuminuria. Circulation. 2024;149:450-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 219] [Article Influence: 109.5] [Reference Citation Analysis (1)] |
| 30. | Sawami K, Tanaka A, Node K. Updated evidence on cardiovascular and renal effects of GLP-1 receptor agonists and combination therapy with SGLT2 inhibitors and finerenone: a narrative review and perspectives. Cardiovasc Diabetol. 2024;23:410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 29] [Reference Citation Analysis (1)] |
| 31. | Li Y, Teng D, Shi X, Qin G, Qin Y, Quan H, Shi B, Sun H, Ba J, Chen B, Du J, He L, Lai X, Li Y, Chi H, Liao E, Liu C, Liu L, Tang X, Tong N, Wang G, Zhang JA, Wang Y, Xue Y, Yan L, Yang J, Yang L, Yao Y, Ye Z, Zhang Q, Zhang L, Zhu J, Zhu M, Ning G, Mu Y, Zhao J, Teng W, Shan Z. Prevalence of diabetes recorded in mainland China using 2018 diagnostic criteria from the American Diabetes Association: national cross sectional study. BMJ. 2020;369:m997. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1223] [Cited by in RCA: 1132] [Article Influence: 188.7] [Reference Citation Analysis (5)] |
| 32. | Zhang L, Long J, Jiang W, Shi Y, He X, Zhou Z, Li Y, Yeung RO, Wang J, Matsushita K, Coresh J, Zhao MH, Wang H. Trends in Chronic Kidney Disease in China. N Engl J Med. 2016;375:905-906. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 640] [Cited by in RCA: 596] [Article Influence: 59.6] [Reference Citation Analysis (4)] |
| 33. | Gaddy A, Elrggal M, Madariaga H, Kelly A, Lerma E, Colbert GB. Diabetic Kidney Disease. Dis Mon. 2025;71:101848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 21] [Article Influence: 21.0] [Reference Citation Analysis (1)] |
| 34. | Dai ZC, Chen JX, Zou R, Liang XB, Tang JX, Yao CW. Role and mechanisms of SGLT-2 inhibitors in the treatment of diabetic kidney disease. Front Immunol. 2023;14:1213473. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 41] [Article Influence: 13.7] [Reference Citation Analysis (1)] |
| 35. | Bae JH. SGLT2 Inhibitors and GLP-1 Receptor Agonists in Diabetic Kidney Disease: Evolving Evidence and Clinical Application. Diabetes Metab J. 2025;49:386-402. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 35] [Reference Citation Analysis (1)] |