Published online Mar 15, 2026. doi: 10.4239/wjd.v17.i3.115433
Revised: December 26, 2025
Accepted: January 22, 2026
Published online: March 15, 2026
Processing time: 142 Days and 3.7 Hours
Diabetic retinopathy (DR) is a leading cause of adult blindness and involves Müller cell dysfunction, which contributes to neurovascular damage in the retina. PRDX2, a member of the thioredoxin family, possesses antioxidant properties and plays roles in cell survival and vascular remodeling. It also exhibits proinflammatory effects in diabetic nephropathy. However, its specific role in the pa
To investigate the role of PRDX2 in DR progression and to elucidate its mecha
A case-control study involving 220 participants (47 healthy controls and 173 individuals with diabetes) was conducted to evaluate plasma PRDX2 levels. Ocular PRDX2 expression was analyzed using vitreous humor samples from proliferative DR (PDR) patients, retinal tissues from DR patients (GSE53257), and retinas from streptozotocin (STZ)-induced diabetic mice (GSE178121). In vitro, PRDX2-knockdown rMC-1 cells (a rat Müller glial cell line) exposed to HG con
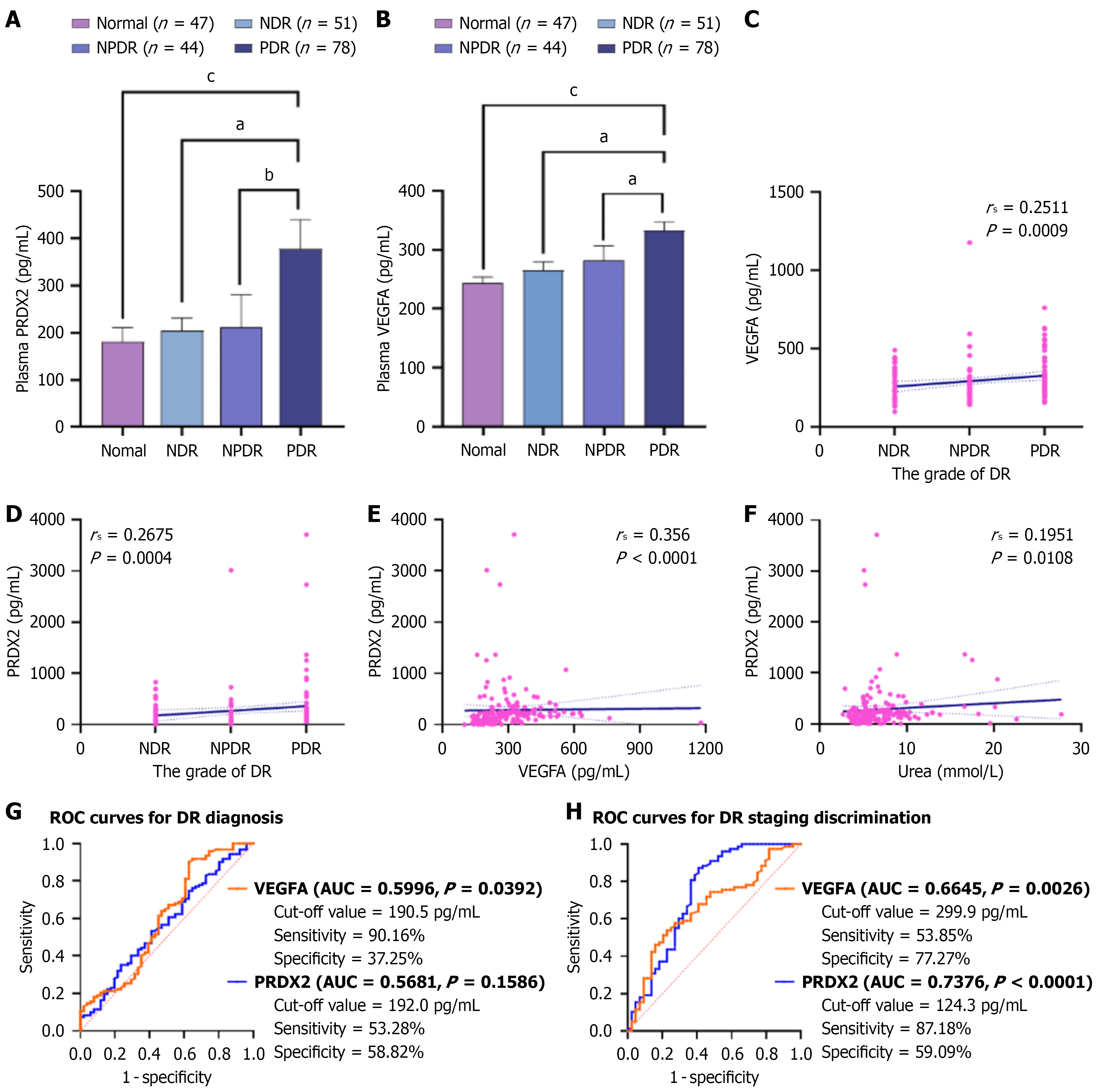
Plasma PRDX2 levels were positively correlated with DR severity (r = 0.267, P < 0.0001) and independently predicted PDR risk after adjustment for confounding factors, including hypertension, age, serum creatinine, urea, fasting blood glucose, and vascular endothelial growth factor A levels (cut-off > 124.3 pg/mL; adjusted odds ratio = 9.097; 95% confidence interval: 2.819-29.355; P = 0.0002). In alignment with its systemic elevation, PRDX2 was markedly upregulated in vitreous humor of PDR patients, retinas of DR patients (GSE53257), Müller cells from STZ-induced diabetic mice (GSE178121), and HG-treated rMC-1 cells. PRDX2 silencing attenuated HG-induced Müller cell hyperproliferation and reduced nestin ex
PRDX2 promotes Müller cell gliosis in DR via the RhoGDI1-RhoA/ROCK axis, making it a potential biomarker and therapeutic target.
Core Tip: This study identifies PRDX2 as a potential biomarker and mechanistic contributor in diabetic retinopathy (DR). PRDX2 was upregulated in both patient plasma and ocular tissues, with plasma levels showing a positive correlation with disease severity and serving as a significant predictor of proliferative DR. In vitro, PRDX2 silencing mitigated high-glucose-induced reactive hyperplasia in rat Müller cells. This effect was mediated by upregulation of RhoGDI1 and suppression of the downstream RhoA/ROCK signaling pathway. Targeting the PRDX2-RhoGDI1-RhoA axis may offer a novel therapeutic approach for alleviating neuroglial dysfunction in DR pathogenesis.
- Citation: Du XL, Ouyang S, Wang YS, Mao YS, Ren BC, Yu WH. PRDX2 silencing alleviates reactive hyperplasia of Müller glial cells in diabetic retinopathy by inhibiting the RhoA/ROCK signaling pathway. World J Diabetes 2026; 17(3): 115433
- URL: https://www.wjgnet.com/1948-9358/full/v17/i3/115433.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i3.115433
Diabetic retinopathy (DR) represents a significant microvascular complication associated with diabetes mellitus (DM)[1]. Globally, more than 140 million individuals are affected by DR[2]. Chronic hyperglycemia in DM leads to retinal vasculopathy and neuropathy, manifesting as microaneurysms, hemorrhage, edema, lipid exudates, neovascularization, vitreous hemorrhage, and fibrosis, ultimately resulting in ir
The structural and functional integrity of the eye is maintained by glial, vascular, and neuronal cells comprising the retinal neurovascular unit[8]. Reactive gliosis, characterized by Müller cell proliferation, activation, and hypertrophy under metabolic stress, is an early hallmark of DR[9,10]. Under sustained hyperglycemic conditions, Müller cells upregulate intermediate filaments (e.g., nestin, vimentin, and GFAP) and proinflammatory mediators (e.g., interleukin-6, interleukin-8, and VEGFA), thereby exacerbating neurovascular damage and promoting fibrovascular proliferation[11-13]. However, the precise molecular mechanisms underlying hyperglycemia-induced Müller cell gliosis in DR remain poorly understood.
PRDX2 is a thioredoxin-dependent antioxidant that scavenges reactive oxygen species and modulates cellular signaling pathways[14,15]. PRDX2 exhibits paradoxical regulatory effects on cell survival and proliferation. Wang et al[16] reported that PRDX2 boosts colorectal cancer proliferation through ubiquitin-dependent degradation of p53. Similarly, Zhao et al[17] demonstrated that PRDX2 promotes cell proliferation and angiogenesis by augmenting VEGF signaling in hepatocellular carcinoma. Conversely, Choi et al[18] reported that PRDX2 inhibits vascular smooth muscle cell proliferation and intimal thickening by disrupting platelet-derived growth factor signaling in a murine model of restenosis. Additionally, PRDX2 overexpression has been shown to suppress the growth of acute myeloid leukemia cells through blockade of the MAPK-ERK signaling pathway[19]. These findings suggest that PRDX2 functions as a context-dependent molecular switch, with its pleiotropic effects governed by the spatiotemporal dynamics of specific disease microenvironments.
Emerging evidence highlights the diverse regulatory roles of PRDX2 in diabetic pathophysiology. In a high-fat diet (HFD) mouse model characterized by insulin resistance, PRDX2 expression was markedly upregulated in skeletal muscle tissue[20]. In diabetic nephropathy, hyperglycemia induces PRDX2 overexpression in renal tubular epithelial cells, thereby activating macrophage-mediated inflammation via the TLR4/NF-κB signaling pathway[21]. Proteomic studies have revealed elevated PRDX2 levels in the vitreous humor of PDR patients[22,23], suggesting its potential involvement in the pathogenesis of DR. However, other PRDX isoforms display distinct protective roles in the retina. For instance, PRDX3 protects photoreceptor mitochondria from oxidative stress[24] and promotes microglial M2 polarization to suppress glial inflammation[25]. PRDX4 attenuates hyperglycemia-induced Müller cell gliosis[26], whereas PRDX5 and PRDX6 inhibit pericyte apoptosis[27] and oxidative damage in retinal pigment epithelial (RPE) cells[28], respectively. Despite these findings, the specific function of PRDX2 in DR pathogenesis, particularly in the context of Müller cell reactive gliosis, remains poorly defined.
Accordingly, the present study investigated the association between DR and PRDX2 to elucidate the role of PRDX2 in hyperglycemia-induced Müller cell responses, advance understanding of DR pathogenesis, and identify potential therapeutic targets.
Samples were collected between November 2020 and December 2023 at the Department of Ophthalmology, Peking Union Medical College Hospital, with approval from the Ethics Committee of Peking Union Medical College Hospital (Approval No. JS-3253). All participants provided informed consent and underwent comprehensive physical and ophthalmic examinations conducted by qualified physicians. Clinical data encompassing age, sex, duration of DM, hypertension, and pertinent medical history were gathered.
Plasma samples were obtained from patients with type 2 DM and healthy individuals. Exclusion criteria included pregnancy or lactation; presence of severe systemic conditions (such as significant cardiac, hepatic, or renal dysfunction), malignancies, severe infections, psychiatric disorders, or a history of PPV, specific retinal diseases, uveitis, advanced glaucoma, complex anterior segment surgeries, or retinal trauma. After applying the exclusion criteria, 220 participants were enrolled, comprising 47 healthy individuals and 173 patients with DM. The diabetic cohort was further stratified according to the international classification of DR[29] into non-DR (NDR; 51 cases), NPDR (44 cases), and PDR (78 cases; Table 1).
| Variables | Normal (n = 47) | DM (n = 173) | NDR (n = 51) | NPDR (n = 44) | PDR (n = 78) | P value1 | P value2 |
| Age (year) | 64 (58-73) | 63 (53-70) | 66 (60-73) | 67 (58-70.75) | 55 (49.75-62.25) | 0.297 | 0.0001 |
| Females | 30 (63.83) | 79 (45.7) | 24 (47.06) | 23 (52.27) | 32 (41.03) | 0.032 | 0.095 |
| Hypertension | 14 (29.79) | 102 (59) | 30 (58.82) | 28 (63.64) | 44 (56.41) | 0.0005 | 0.004 |
| FBG (mmol/L) | 5.7 (5.3-6.4) | 8.1 (7-9.9) | 7.9 (6.9-9.1) | 7.9 (7.025-9.8) | 8.850 (7.15-11.43) | 0.0002 | 0.0002 |
| ALT (U/L) | 16 (14-22) | 17 (14-25.25) | 18 (14-28.75) | 17 (12-19) | 17 (14-26.5) | 0.276 | 0.197 |
| AST (U/L) | 21.5 (19-24) | 19 (15-24) | 21 (16-24) | 19 (15-22.75) | 18.5 (14.75-25.52) | 0.008 | 0.052 |
| ALP (U/L) | 74 (62-92) | 76 (61.75-90) | 71 (56-90) | 77 (64-90) | 77 (62-90.25) | 0.773 | 0.804 |
| GGT (U/L) | 19 (14.5-29.5) | 20 (16-33) | 28 (19.25-36) | 22 (16-37) | 19 (14-30.5) | 0.311 | 0.015 |
| TP (g/L) | 71 (69-75) | 71 (67-74) | 71 (68.25-74) | 71 (67-74.75) | 71 (66-74) | 0.286 | 0.381 |
| Alb (g/L) | 45 (43-46) | 44 (41-46) | 45 (43-47) | 43.5 (41.25-46.75) | 43 (40.25-45) | 0.021 | < 0.0001 |
| A/G | 1.7 (1.575-1.8) | 1.6 (1.4-1.8) | 1.7 (1.5-1.875) | 1.6 (1.5-1.7) | 1.55 (1.4-1.7) | 0.042 | 0.018 |
| K (mmol/L) | 4.2 (4.1-4.4) | 4.4 (4.2-4.7) | 4.3 (4.15-4.5) | 4.4 (4.075-4.7) | 4.5 (4.2-2.75) | 0.027 | 0.058 |
| Na (mmol/L) | 141 (140-142) | 140 (139-141) | 140 (139-141) | 140 (139-141) | 140 (139-141) | 0.005 | 0.048 |
| Cl (mmol/L) | 105 (104-106) | 104 (102-106) | 104 (102-106.5) | 104 (102-106) | 105 (103-106) | 0.143 | 0.419 |
| P (mmol/L) | 1.18 (1.085-1.33) | 1.21 (1.12-1.36) | 1.145 (1.085-1.293) | 1.25 (1.04-1.36) | 1.25 (1.14-1.40) | 0.506 | 0.067 |
| Ca (mmol/L) | 2.374 ± 0.05416 | 2.373 ± 0.00872 | 2.405 ± 0.1060 | 2.37 ± 0.1024 | 2.353 ± 0.1146 | 0.779 | 0.037 |
| Urea (mmol/L) | 5.29 (4.56-6.63) | 6.14 (4.99-8.4) | 5.825 (4.285-7.21) | 5.38 (4.863-9.203) | 7.415 95.47-9.045) | 0.011 | 0.001 |
| SUA (mmol/L) | 301 (256-373) | 347 (284-423) | 340 (282.5-416.5) | 359 (287-423) | 349 (275-433.5) | 0.01 | 0.066 |
| SCr (μmol/L) | 67 (60-74) | 75 (63.75-91.25) | 71 (63-89.25) | 72.5 (62.75-86.25) | 78 (64.75-101.5) | 0.002 | 0.005 |
| VEGFA (pg/mL) | 233.8 (181.8-302.6) | 273.63 (203.84-358.34) | 237.7 (167.6-360.5) | 237.1 (204.5-297.8) | 308.6 (228.8-399.8) | 0.009 | 0.0001 |
| PRDX2 (pg/mL) | 128.6 (46.27-228.5) | 189.21 (96.42-259.31) | 180.8 (69.9-236.4) | 103.4 (33.57-232.7) | 225.6 (164.7-322.5) | 0.024 | 0.0001 |
Blood biochemical parameters measured included aspartate aminotransferase (AST), albumin, albumin/globulin (A/G) ratio, alkaline phosphatase (ALP), gamma-glutamyl transferase (GGT), alanine aminotransferase (ALT), urea, serum creatinine (SCr), serum uric acid (SUA), and fasting blood glucose (FBG). Physicians collected 2 mL of whole blood from each fasting participant in the morning. Samples were centrifuged, and plasma was stored at -80 °C for subsequent ELISA analysis.
Vitreous humor samples were collected from patients with PDR and from non-diabetic individuals undergoing primary PPV surgery. The PDR group (n = 9) consisted of patients diagnosed with PDR who required PPV. The non-diabetic control group (n = 6) included individuals without diabetes who underwent PPV for primary epiretinal membrane, rhegmatogenous retinal detachment, or idiopathic macular hole. Exclusion criteria for both groups included a history of prior vitrectomy, specific retinal diseases, uveitis, advanced glaucoma, complex anterior segment surgeries, or retinal trauma. Clinical characteristics of the 15 participants are summarized in Supplementary Table 1. Approximately 200 μL of vitreous humor was collected from each participant at the onset of PPV surgery, centrifuged at 4 °C to remove cellular debris, and stored undiluted at -80 °C for ELISA analysis.
ELISA kits were employed to quantify the concentrations of VEGFA (AX-998787H; ELK Biotechnology, Wuhan, China) and PRDX2 (JM-0909H1; JingMei Biotechnology, China) in human plasma and VH samples. Absorbance at 450 nm was measured using a microplate reader (Bio-Rad, United States).
Human retinal microarray datasets were obtained from the Gene Expression Omnibus database (GEO; https://www.ncbi.nlm.nih.gov/geo/) with the accession number GSE53257[30] to evaluate PRDX2 expression in retinal tissues from six DR patients vs five healthy controls (Supplementary Table 2). Raw data were subjected to log2 transformation and quantile normalization using the preprocessCore R package (v 3.4.1).
Cell-type-specific expression of PRDX2 in the human retina was analyzed using single-cell transcriptomic data (Supplementary Table 3) from the Human Protein Atlas (https://www.proteinatlas.org/ENSG00000167815-PRDX2/single+cell/eye.; accessed on July 1, 2022). The analysis included Müller glial cells, endothelial cells, horizontal cells, photoreceptors, bipolar cells, microglia, retinal ganglion cells, RPE cells, and fibroblasts.
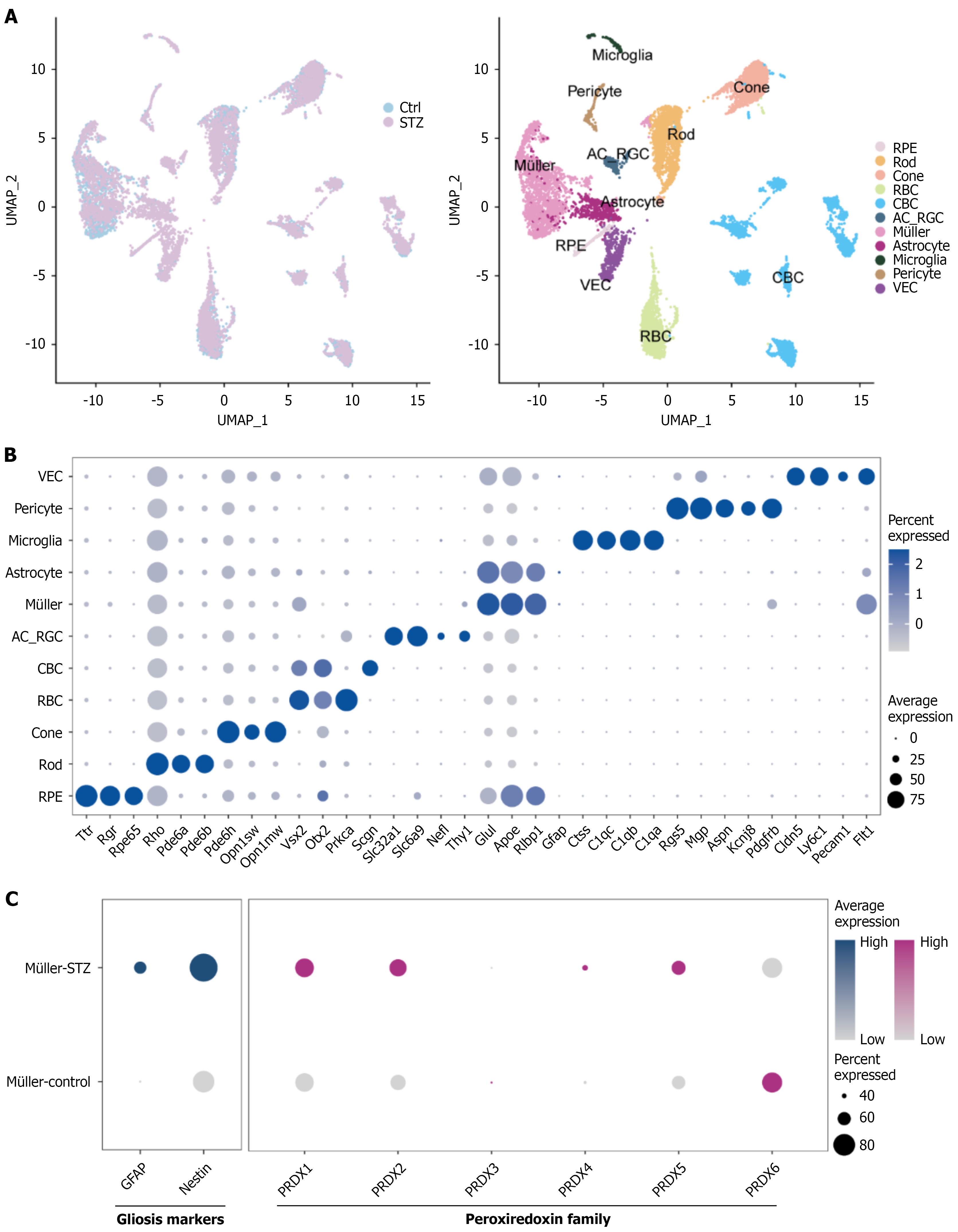
Single-cell RNA sequencing (scRNA-seq) data from the retinas of streptozotocin (STZ)-induced diabetic mice (6 months post-induction) were obtained from the GEO database under accession number GSE178121[31] to assess the gene expression of Müller gliosis markers (GFAP and nestin) and peroxiredoxin family genes (PRDX1-PRDX6) in diabetic murine retinal Müller cells. Subsequent analyses including quality control, normalization, dimensionality reduction, clustering, and annotation of cell types were performed via the Seurat package (v 4.3.0) in R. During quality control, cells expressing fewer than 200 genes or exhibiting mitochondrial gene content greater than 20% were excluded. The remaining retinal cells were annotated based on established marker genes commonly used in single-cell studies[31,32], as follows: RPE (Ttr, Rgr, and Rpe6), rod (Rho, Pde6a, and Pde6b), cone (Pde6h, Opn1sw, and Opn1mw), retinal bipolar cells (Vxs2, Otx2, and Prkca), cone bipolar cells (Vsx2, Otx2, and Scgn), amacrine/retinal ganglion cell mixed population (Slc32a1, Slc6a9, Nefl, and Thy1), Müller cells (Glul, Apoe, and Rlbp1), astrocytes (Glul, Apoe, and Gfap), microglia (Ctss, C1qc, C1qb, and C1qa), pericytes (Mgp, Aspn, Kcnj8, and Pdgfrb) and vascular endothelial cells (Cldn5, Ly6c1, Pecam1, and Flt1).
The transformed rat retinal Müller cell line (rMC-1), generously provided by Zhang et al[33] at Shanghai Jiao Tong University School of Medicine, was cultured in DMEM containing 5.5 mmol/L glucose (SH30021.01B; HyClone, United States), supplemented with 10% fetal bovine serum (FBS; 10099141C; Gibco, United States), and 1% penicillin-streptomycin (15140122; Gibco, United States). Cells were maintained at 37 °C in a humidified atmosphere with 5% CO2. The rMC-1 Müller cell line is a well-validated and widely used model in DR research, effectively recapitulating key pathogenic features, including gliotic activation, oxidative stress, and inflammatory cytokine release—processes conserved between rodent and human Müller cells[34,35].
Logarithmic-phase rMC-1 cells were treated with either normal glucose (NG; 5.5 mmol/L) or high glucose (HG; 25 mmol/L) for 24 hours or 48 hours. PRDX2 gene knockdown in rMC-1 cells was achieved using a lentiviral vector carrying PRDX2 knockdown (shPRDX2), whereas a non-targeting control lentivirus (shNC) was used for comparison. Both lentiviral constructs were obtained from Genomeditech (Shanghai, China), and stable rMC-1 cell lines were established through lentiviral transduction.
Cell viability was assessed using the Cell Counting Kit-8 (CCK-8; 40203ES60; Yeasen Biotechnology, China) and Hoechst staining (40732ES03; Yeasen Biotechnology, China). In the CCK-8 assay, absorbance was measured at 450 nm using a microplate reader (BioTek, United States). For Hoechst staining, cells cultured in six-well plates were incubated with 5 μg/mL dye at 37 °C for 20 minutes, followed by phosphate-buffered saline washes and visualization under a fluorescence microscope (Nikon, Japan).
Apoptosis in rMC-1 cells was assessed using the Annexin V-Alexa Fluor 647/PI Apoptosis Detection Kit (E-CK-A217; Elabscience, China). Cells cultured in six-well plates were stained with Annexin V-APC and propidium iodide for 10 minutes at room temperature. Following the addition of 400 μL Annexin-binding buffer, samples were analyzed by flow cytometry using the LSRFortessa system (BD Biosciences, United States).
Cell invasive capacity was evaluated using Transwell chambers with 8-μm pores (3422; Corning)[36]. Pretreated rMC-1 cells were seeded in the upper chamber at a density of 1 × 105 cells/well in serum-free medium. The lower chamber was filled with 675 μL of complete growth medium containing 30% FBS, serving as a chemoattractant. After a 24-hour incubation period, the cells that migrated to the bottom side of the membrane were stained with crystal violet and analyzed through imaging with a Nikon microscope. Invading cells were quantified in five randomly selected fields per membrane using ImageJ software (v 1.52, United States).
Cell migration was assessed using a wound-healing assay. Evenly spaced transverse lines were drawn on the bottom of six-well plates using a marker to guide scratch placement. rMC-1 cells (2 × 105 cells/well) were seeded and incubated overnight. After washing, a scratch was made perpendicular to the lines using a 10-μL pipette tip. The medium was then replaced with serum-free medium. Images were captured at 0-, 24-, and 48-hour using a Nikon microscope. The wound area was measured using ImageJ software (v 1.52).
rMC-1 cells (10000 cells/well) were seeded in a black, clear-bottom 96-well plate (6055300; PerkinElmer, United States) and treated as indicated. Cell proliferation was evaluated using the YF®647A Click-iT EdU Imaging Kits (40277ES60; Yeasen Biotechnology, China) and the CyQUANT NF assay kits (C350007; Thermo Fisher, United States). EdU incorporation was visualized using a High-Content Imaging System (PerkinElmer), and proliferation was quantified as the percentage of EdU-positive cells. The CyQUANT NF assay measured DNA content-associated fluorescence using a BioTek microplate reader.
Lactate assay kits (A019-2-1; Nanjing Jiancheng Bioengineering Institute, China) were used to evaluate lactate levels in rMC-1 cells following HG treatment. The rMC-1 cells in a six-well plate were washed, lysed, and lactate was determined by a microplate reader (BioTek, United States).
Total RNA was extracted using an RNAprep Pure Micro Kit (Solarbio, China) and reverse-transcribed into cDNA using FastKing RT (QIAGEN, China). Real-time PCR with SYBR Green (QIAGEN, China) was conducted on a LightCycler 480 II (Roche, Switzerland), using GAPDH as an internal reference. The sequences of the PRDX2 primers were F: TCCCTCTGCTTGCTGATGTGACT and R: CGTTGACTGTGATCTGGCGAAGG. The sequences of the PRDX6 primers were F: GCCACATCCGCTTCCACGATT and R: GCACCATTGTAAGCATTGATGTCCTT. The sequences of the GAPDH primers were F: TCAAGAAGGTGGTGAAGCAGG and R: GCGTCAAAGGTGGAGGAGTG.
Tandem mass tag (TMT) labeling and liquid chromatography-tandem mass spectrometry (LC-MS/MS) were employed to identify differentially expressed proteins (DEPs) in shPRDX2 (n = 3) and control (shNC, n = 3) rMC-1 cells. The procedure, conducted at TIANGEN BIOTECH in Beijing, China, included protein extraction, reductive alkylation, digestion, peptide desalting, TMT labeling, separation by high-pH reverse-phase liquid chromatography, LC-MS/MS analysis, protein identification, and bioinformatics analysis, as previously described[37]. Raw data were processed using ProteomeDiscoverer (v 3.0), with a false discovery rate below 1% and a peptide score above 40. Differential protein abundance was analyzed using paired t-tests with P value correction, and proteins with a fold change (FC) greater than 1.2 and P < 0.05 were considered DEPs. All identified proteins and DEPs from the shPRDX2 and shNC groups are presented in Supplementary Table 4. The enrichment analysis of Gene Ontology (GO) terms and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways for DEPs was performed and visualized using R. The interaction network of DEPs was analyzed with STRING (https://cn.string-db.org/), and hub proteins were identified using Cytoscape (v 3.9.1).
rMC-1 cells underwent harvesting and lysis utilizing RIPA buffer that was supplemented with protease inhibitors (Beyotime, China). Protein samples (30 μg per lane) were separated by electrophoresis and transferred onto polyvinylidene difluoride membranes (Millipore, United States) at 300 mA for 35-185 minutes. Membranes were blocked with QuickBlocTM buffer (Beyotime, China) for 20 minutes, followed by overnight incubation at 4 °C with the following primary antibodies: Anti-β-actin (1:10000, 60008-1-Ig; Proteintech, China), anti-tubulin (1:5000, M20005; Abmart, United States), anti-PRDX2 (1:2000, 10545-2-AP; Proteintech, China), anti-ROCK1/2 (1:2000, ab45171; Abcam, United States), anti-RhoGDI1 (1:1000, GB11726; Servicebio, China), and anti-RhoA (1:5000, ab187027; Abcam, United States). Secondary horseradish peroxidase-conjugated antibodies (1:5000; Proteintech, China) were applied, and protein expression was detected using a chemiluminescent kit (Epizyme Biotech, China) and quantified with ImageJ (v 1.52).
All analyses were carried out using SPSS version 19.0 alongside GraphPad Prism version 9.1. Categorical clinical data were presented as n (%) and compared between groups using the χ² test or Fisher’s exact test. Continuous data were reported as median [interquartile range (IQR)] or mean ± SD, whereas cell-based experimental data were expressed as mean ± SE. For comparisons between two groups, normally distributed data were analyzed using the Student’s t test, and nonparametric data were evaluated using the Mann-Whitney U test. For comparisons among three or more groups, one-way ANOVA with Tukey honestly significant difference test was used for parametric data, and the Kruskal-Wallis test with Dunn post hoc analysis was applied for nonparametric data. Bivariate correlations were assessed using Spearman's rank correlation. The diagnostic performance of VEGFA and PRDX2 was evaluated using receiver operating characteristic (ROC) curve analysis with area under the curve (AUC) calculations. Binary logistic regression was used to determine the independent effect of plasma PRDX2 on DR severity.
To systematically investigate the association between plasma PRDX2 and patients with DR, a total of 220 participants were enrolled, including 47 healthy individuals [median age: 64 years, IQR: 58-73; 30 females (63.83%)] and 173 patients with DM [median age: 63 years, IQR: 53-70; 79 females (45.7%)].
Compared with healthy controls (normal group), patients with diabetes (DM group) exhibited a lower proportion of females (P = 0.032), as well as meaningfully lower levels of serum AST (P = 0.008), albumin (P = 0.021), A/G ratio (P = 0.042), and Na (P = 0.005). In contrast, the DM group showed a higher prevalence of hypertension (P = 0.0005), along with elevated levels of FBG (P = 0.0002), serum urea (P = 0.011), SCr (P = 0.002), SUA (P = 0.01), K (P = 0.027), and VEGFA (P = 0.009; Table 1).
ELISA analysis indicated that plasma levels of PRDX2 were considerably greater in the DM group in comparison to the normal group (189.21 pg/L, IQR 96.42-259.31 vs 128.6 pg/L, IQR 46.27-228.5; P = 0.024). Other variables related to age, metabolism, and liver function—including ALT, ALP, GGT, total protein, pre-albumin, total bile acid, total bilirubin, direct bilirubin, lactate dehydrogenase, cholinesterase, Cl, P, TCO2, and Ca—did not differ significantly between the two groups (all P > 0.05).
We further examined the dynamics of plasma PRDX2 and VEGFA levels across different stages of DR. Compared with healthy controls and other DR stages, subjects with PDR were the youngest, had the highest prevalence of hypertension, and exhibited the lowest serum albumin and A/G ratios, along with the highest FBG, serum urea, and SCr levels (Table 1). Plasma concentrations of PRDX2 and VEGFA elevated in a stage-dependent manner, with peak levels displayed in the PDR group (Figure 1A and B). Spearman correlation analysis disclosed a significant positive association between DR severity and elevated plasma PRDX2 (r = 0.267, P < 0.0001) and VEGFA (r = 0.251, P = 0.001; Figure 1C and D). Additionally, plasma PRDX2 levels were positively correlated with VEGFA (r = 0.356, P < 0.0001) and serum urea (r = 0.195, P = 0.011), indicating that increased PRDX2 may be associated with retinal neovascularization and renal impairment (Table 2, Figure 1E and F).
| Variable | Spearman, rs | P value |
| Age | -0.083 | 0.278 |
| Females | 0.013 | 0.863 |
| Hypertension | 0.038 | 0.620 |
| FBG | -0.022 | 0.775 |
| ALT | -0.027 | 0.728 |
| AST | -0.044 | 0.582 |
| ALP | 0.085 | 0.287 |
| GGT | -0.138 | 0.096 |
| TP | -0.051 | 0.515 |
| Alb | -0.081 | 0.298 |
| A/G | -0.074 | 0.345 |
| K | -0.005 | 0.950 |
| Na | 0.056 | 0.476 |
| Cl | 0.075 | 0.332 |
| P | 0.070 | 0.384 |
| Ca | -0.061 | 0.442 |
| Urea | 0.195 | 0.011 |
| SUA | 0.088 | 0.264 |
| SCr | 0.144 | 0.061 |
| VEGFA | 0.356 | < 0.0001 |
| The severity of DR | 0.267 | < 0.0001 |
ROC curves were generated to evaluate plasma PRDX2 and VEGFA as diagnostic biomarkers for DR within the DM cohort (n = 173). VEGFA yielded an area AUC of 0.5996 [95% confidence interval (95%CI): 0.5004-0.6988; P = 0.0392], with an optimal cutoff threshold of 190.5 pg/mL, sensitivity of 90.16%, and specificity of 37.25% (Figure 1G). PRDX2 demonstrated an AUC of 0.5681 (95%CI: 0.4739-0.6622; P = 0.1568) at a cutoff value of 192.0 pg/mL, with a sensitivity of 53.28% and specificity of 58.82%. When assessed as a marker for DR progression stratification, PRDX2 showed significantly improved diagnostic performance (AUC = 0.7376; optimal cut-off = 124.3 pg/mL; sensitivity = 87.18%; specificity = 59.09%), surpassing VEGFA (AUC = 0.6645; sensitivity = 53.85%; specificity = 77.27%; P = 0.0026; Figure 1H). Multivariate logistic regression analysis, adjusted for clinical covariates (age, hypertension, VEGFA, FBG, serum albumin, A/G ratio, GGT, SCr, and urea), demonstrated that plasma PRDX2 > 124.3 pg/mL was independently associated with a 9.097-fold increased risk of PDR [adjusted odds ratio = 9.097, 95%CI: 2.819-29.355; P = 0.0002; Table 3]. These findings suggest that although VEGFA retains high sensitivity for initial DR detection, plasma PRDX2 serves as a promising biomarker for monitoring DR progression.
| Parameter | aOR (95%CI) | P value |
| Model 1 | 9.822 (4.012-24.049) | < 0.0001 |
| Model 2 | 9.097 (2.819-29.355) | 0.0002 |
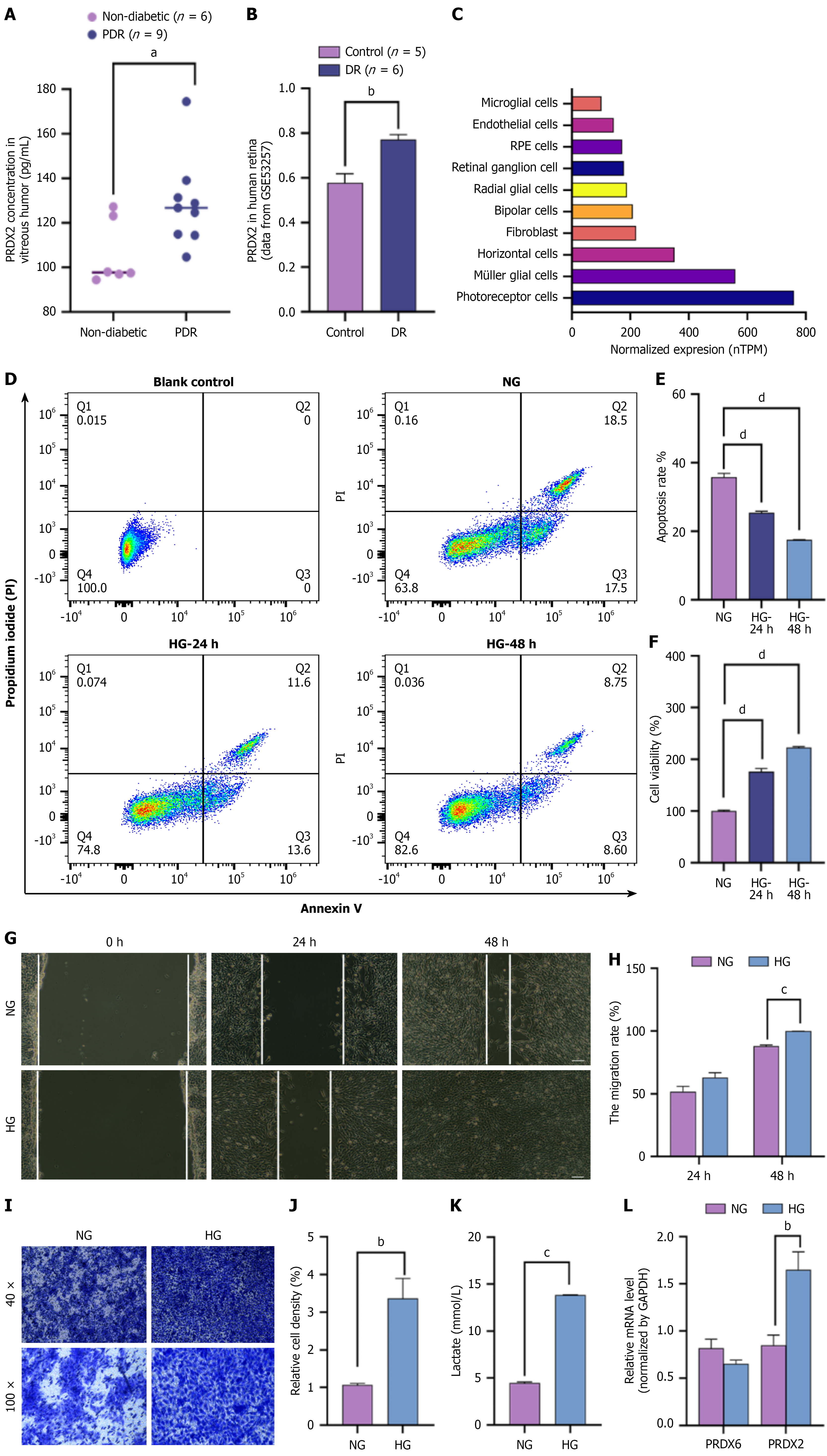
Building on previous proteomic evidence indicating elevated PRDX2 levels in the vitreous humor of PDR patients[22,23], we confirmed PRDX2 expression in vitreous humor samples from nine PDR patients and six non-diabetic controls (diagnosed with primary epiretinal membrane, rhegmatogenous retinal detachment, or idiopathic macular hole). Baseline characteristics showed significantly higher FBG levels in the PDR group (10.1 mmol/L, IQR: 8.45-14.05; P = 0.001) compared with the non-diabetic controls (5.4 mmol/L, IQR: 4.78-6.08; P = 0.001), whereas gender, age, blood pressure, and intraocular pressure were similarly distributed between groups (Supplementary Table 1). Quantification by ELISA revealed increased PRDX2 protein expression in the vitreous humor of PDR patients relative to non-diabetic controls (Figure 2A). Consistent with these findings, transcriptomic analysis of human retinal tissue from the GEO database[30] (GSE53257 dataset) demonstrated upregulation of PRDX2 in the DR group (n = 6) compared with healthy donors (n = 5; P < 0.05; Figure 2B, Supplementary Table 2).
Human retinal scRNA-seq data from the Human Protein Atlas demonstrated cell-type-specific transcript abundance of PRDX2 across human retinal populations, with predominant expression in photoreceptor cells, followed by Müller glial cells (Figure 2C and Supplementary Table 3). Given the central role of Müller glial cells in DR pathogenesis, we investigated PRDX2 function in diabetic Müller cells. The rat Müller glial cell line rMC-1 was subjected to HG (25 mmol/L) or NG (5 mmol/L) conditions for 24-48 hours to mimic diabetes-related stress. Compared with the NG group, HG exposure significantly decreased apoptosis (Figure 2D and E) and increased cell viability (Figure 2F), particularly at 48 hours. Wound-healing (Figure 2G and H) and Transwell chamber assay (Figure 2I and J) validated that HG exposure significantly promoted rMC-1 cell migration and invasion capacity. HG treatment increased lactate production (Figure 2K), indicating metabolic reprogramming. Consequently, a reactive proliferation model of diabetic Müller cells was established in vitro by treating rMC-1 cells with HG for 48 hours. Importantly, qPCR and western blotting revealed that the PRDX2 transcript and protein expression were upregulated in rMC-1 cells after HG treatment (Figures 2L and 3A), whereas PRDX6 transcript expression remained unchanged (Figure 2L).
We next examined PRDX2 expression in the retinas of STZ-induced diabetic mice (6 months post-induction) and non-diabetic controls using the scRNA-seq dataset GSE178121[31]. Uniform Manifold Approximation and Projection analysis identified 11 transcriptionally distinct retinal cell clusters based on established biomarkers. These included RPE, rod photoreceptor cells, cone photoreceptor cells, rod bipolar cells, cone bipolar cells, a mixed population of amacrine, retinal ganglion cells, Müller cells, astrocytes, microglia, pericytes, and vascular endothelial cells (Figure 3A and B). Dot plot visualization confirmed the cluster-specific expression of canonical markers (Figure 3B). Differential gene expression analysis in Müller cells revealed that PRDX1, PRDX2, PRDX4, and PRDX5 were upregulated, whereas PRDX3 and PRDX6 were downregulated in STZ-induced diabetic mice compared with controls (Figure 3C). Additionally, expression levels of reactive gliosis markers, including GFAP and nestin, were elevated (Figure 3C). These findings suggest that PRDX2 upregulation may represent a characteristic molecular alteration in the pathophysiology of diabetic Müller cells.
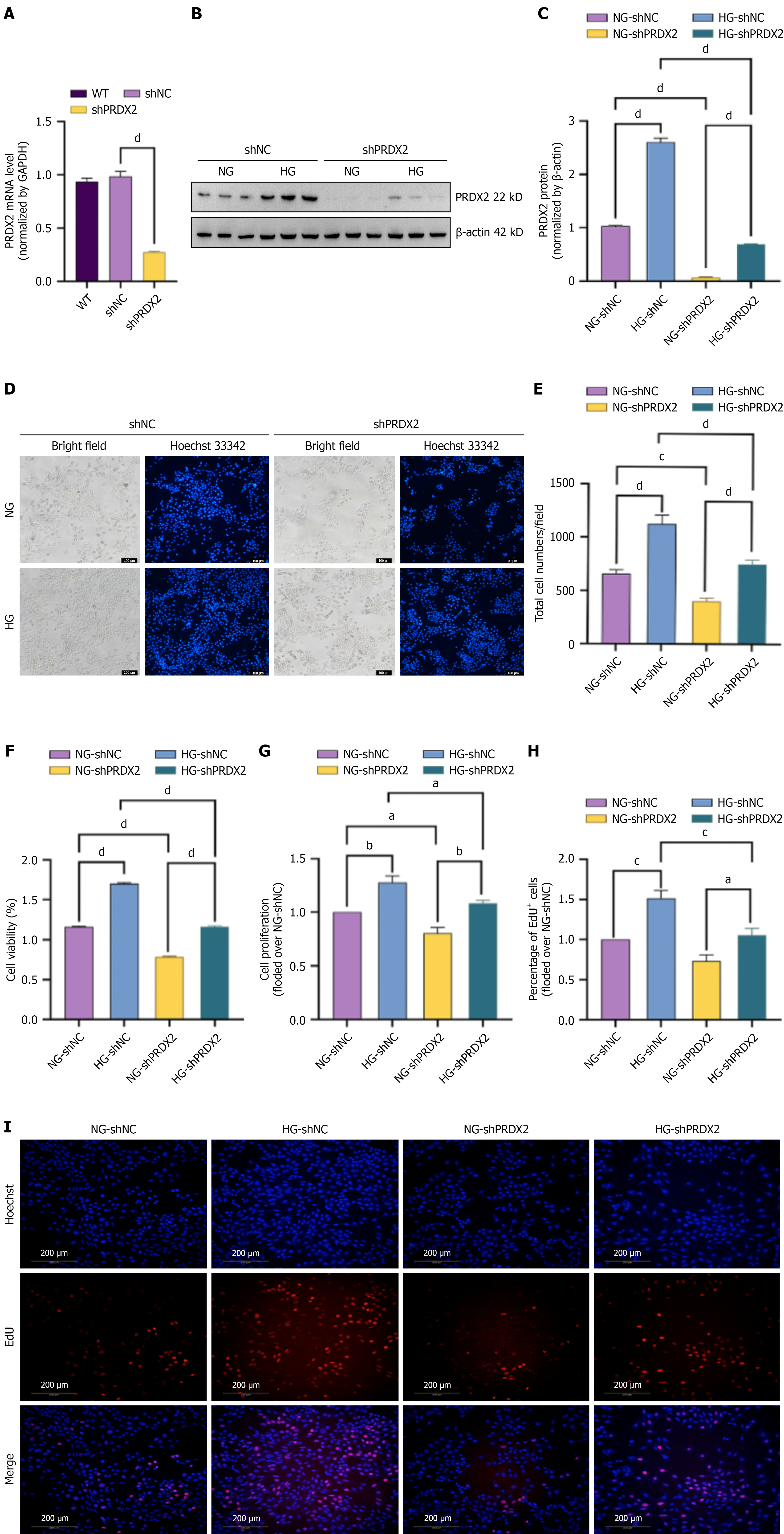
To clarify the functional role of PRDX2 in Müller cells, stable PRDX2-knockdown rMC-1 cells were generated via shRNA lentiviral transduction. qPCR confirmed significant suppression of PRDX2 transcript levels in shPRDX2 cells compared with both scrambled shNC and wild-type cells (Figure 4A). Western blot analysis demonstrated that HG (25 mmol/L) markedly induced PRDX2 protein expression, whereas shPRDX2 transduction effectively abolished this upregulation (Figure 4B and C).
Multiparametric assessments of proliferation, including Hoechst staining (Figure 4D and E), CCK-8 assay (Figure 4F), CyQUANT NF cell proliferation assay (Figure 4G), and EdU staining (Figure 4H and I), revealed that PRDX2 silencing significantly impaired Müller cell survival. Specifically, shPRDX2 reduced rMC-1 cell viability and inhibited proliferative capacity (P < 0.05). Notably, these antiproliferative effects were observed under both NG and HG conditions, indicating that PRDX2 contributes to basal Müller cell homeostasis and mediates pathological proliferation under diabetic stress (Figure 4D-I). Targeted suppression of HG-induced PRDX2 overexpression effectively attenuated aberrant glial expansion, supporting the role of PRDX2 as a critical driver of diabetic gliosis progression.
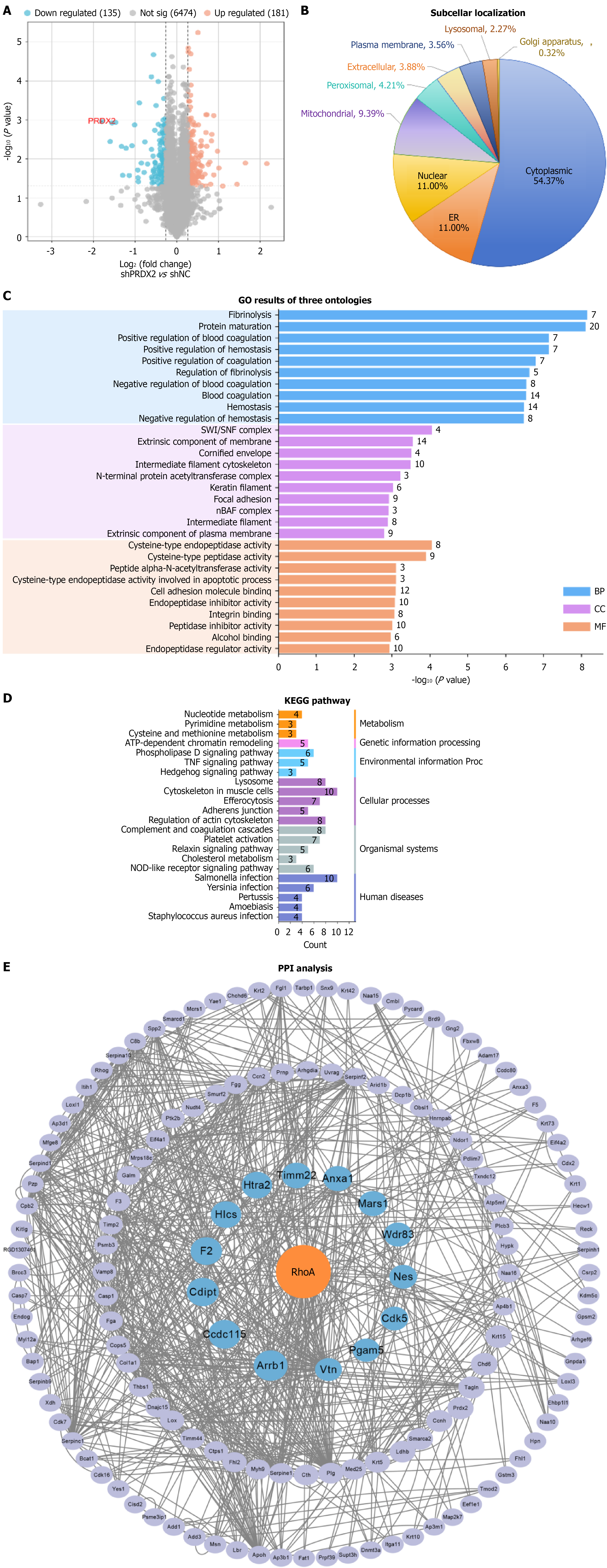
To investigate the molecular mechanism by which PRDX2 regulates reactive hyperplasia in Müller cells, TMT-based proteomics was employed to analyze protein expression changes in rMC-1 cells following PRDX2 knockdown. A total of 6790 proteins were identified and quantified in rMC-1 cells from the shPRDX2 group and shNC group. Comparative analysis revealed 316 DEPs, including 135 downregulated and 181 upregulated proteins, with a FC > 1.2 and P < 0.05 (Figure 5A). Details of all 6790 identified proteins and the 316 DEPs, including protein IDs, gene names, functional annotations, P values, and FCs, are provided in Supplementary Table 4.
Subcellular localization analysis showed that 54.37% of the DEPs were localized to the cytoplasm, 11.0% to the nucleus, 11.0% to the endoplasmic reticulum, and 9.39% to the mitochondria (Figure 5B). GO analysis indicated that the biological processes, cellular component, and molecular function terms of DEPs were mainly involved in fibrinolysis, protein maturation, SWI/SNF complex, extrinsic component of membrane, cysteine-type endopeptidase activity, cytoskeleton, cell adhesion, and hemostasis (Figure 5C). KEGG annotation revealed that 315 DPEs were enriched in multiple pathways, including those related to human diseases, genetic information processing, metabolism, environmental information processing, cellular processes, and organismal systems (Figure 5D). Among these, the most significantly affected KEGG pathways following PRDX2 silencing in retinal Müller cells were associated with cytoskeletal regulation and Salmonella infection (Figure 5D).
To further explore the relationships among these DEPs, a protein-protein interaction analysis was performed using the STRING database. Network topology was evaluated using four centrality measures—Betweenness, Degree, Subgraph, and Closeness—via the Cytoscape plugin cytoNAC. Across all analytical approaches, RhoA emerged as the central hub protein within the interaction network (Figure 5E). These findings suggest that PRDX2 knockdown primarily influences cytoskeleton organization and cellular homeostasis, potentially through modulation of RhoA signaling pathways.
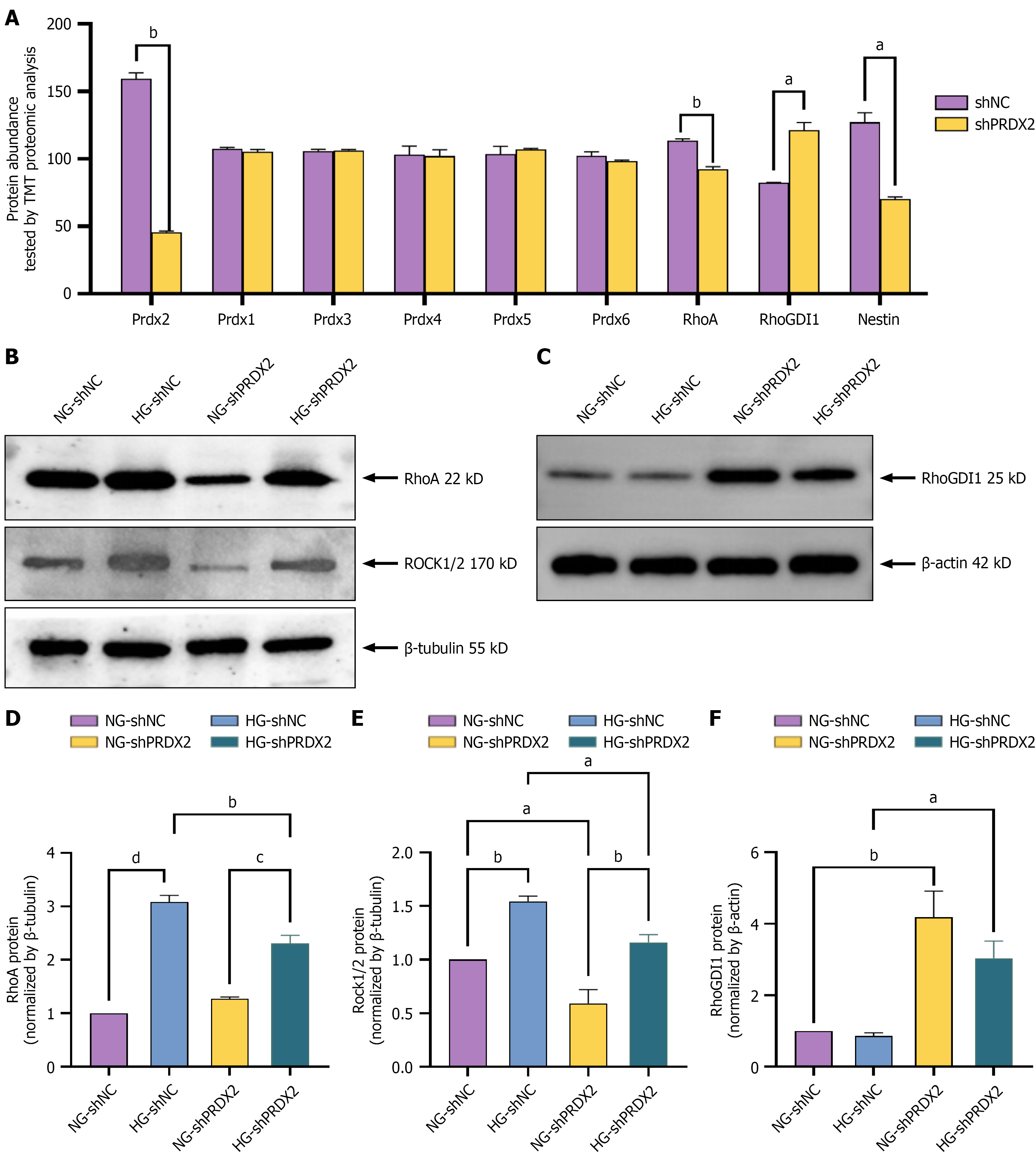
Activation of the RhoA/ROCK signaling pathway plays a critical role in cytoskeletal remodeling, contributing to Müller cell dysfunction and diabetic microvascular pathology[36,38]. TMT-based quantitative proteomic analysis demonstrated that PRDX2 silencing in rMC-1 cells significantly reduced the abundance of PRDX2 (P < 0.01), RhoA (P < 0.01), and the gliosis marker nestin (P < 0.05)[39] while increasing the expression of RhoGDI1 (P < 0.05; Figure 6A). PRDX2 knockdown did not alter the protein expression of its homologs (PRDX1, PRDX3-6; P > 0.05), confirming the specificity of the observed phenotype to PRDX2 ablation (Figure 6A).
Western blot analysis confirmed that HG treatment significantly increased RhoA and ROCK1/2 protein expression in rMC-1 cells (Figure 6B-D). PRDX2 knockdown significantly attenuated these increases under HG conditions (Figure 6B-D). Additionally, PRDX2 knockdown significantly upregulated RhoGDI1 protein expression independent of glucose concentration, whereas HG treatment alone did not affect RhoGDI1 expression (Figure 6E and F). These findings suggest that PRDX2 knockdown mitigates HG-induced reactive activation in Müller cells, likely by upregulating RhoGDI1 and thereby inhibiting the RhoA/ROCK signaling pathway.
Reactive gliosis of Müller cells is a key pathological hallmark of DR[1]. However, the mechanisms underlying diabetic Müller cell gliosis remain incompletely understood. This study demonstrated that PRDX2 expression was significantly elevated in DR plasma, PDR vitreous, DR retinas (GSE53257), STZ-induced diabetic murine retinal Müller cells (GSE178121), and HG-induced rat Müller cells. Plasma PRDX2 levels were positively correlated with DR severity and independently associated with disease progression. Mechanistically, PRDX2 silencing attenuated reactive gliosis by modulating the RhoA/ROCK signaling pathway through upregulation of RhoGDI1. These findings identify PRDX2 as a potential biomarker for DR staging and a promising therapeutic target for mitigating gliosis in DR pathogenesis.
PRDX2 is ubiquitously expressed across tissues and functions as a signaling modulator that regulates proliferation, apoptosis, inflammation, oxidative stress, and angiogenesis[14]. In diabetes research, PRDX2 has been reported to be upregulated in skeletal muscle cells of HFD mice and in renal tubular epithelial cells under hyperglycemic conditions[20,21], suggesting a role for PRDX2 as a hyperglycemic stress responder across diverse cell types. Despite these associations, its function in DR remains unexplored. Accordingly, plasma PRDX2 levels were assessed in a dedicated DR cohort. To minimize potential confounders, we excluded individuals with comorbidities including kidney disease, significant cardiac, malignancies, or severe infectious diseases. Nevertheless, PDR patients exhibited significantly elevated serum urea and Scr, indicating subclinical renal impairment prior to overt clinical diagnosis. Additionally, the prevalence of hypertension increased with DR severity. Plasma PRDX2 levels positively correlated with DR severity, plasma VEGFA, and serum urea. These findings suggest that the progressive elevation of plasma PRDX2, reaching peak levels in PDR, likely reflects an integrated stress response to chronic hyperglycemia across multiple cell types, including renal and vascular tissues. Importantly, although PRDX2 alone demonstrated moderate diagnostic value for overall DR detection, its utility in staging DR progression was substantial, surpassing that of VEGFA. Multivariate analysis confirmed that plasma PRDX2 levels greater than 124.3 pg/mL were associated with a 9.097-fold increased PDR risk after adjustment for potential confounders, including hypertension, VEGFA, SCr, urea, age, FBG, serum albumin, A/G, and GGT.
Although PRDX2 is ubiquitously expressed and its circulating levels may theoretically be influenced by systemic conditions such as nephropathy, cancer, or inflammation[14], several lines of evidence from this study support its specific relevance to DR. First, the cohort explicitly excluded individuals with major comorbidities, and the association between elevated plasma PRDX2 and PDR remained significant after rigorous adjustment for key confounders, including hypertension, renal function markers, and VEGFA. Second, and more critically, concordant upregulation of PRDX2 was observed within the ocular compartment: Its gene expression was elevated in DR retinas (GSE53257), and its protein level was increased in PDR vitreous humor, consistent with findings from prior proteomic studies[22,23]. This localized accumulation suggests that PRDX2 is not merely a systemic stress marker but may play an active role in DR pathogenesis.
Within the retina, PRDX2 is primarily localized to neurons of the inner nuclear layer and photoreceptors[40], with scRNA data from the Human Protein Atlas confirming predominant expression in photoreceptors, followed by Müller glia. Müller cells span the entire thickness of the retina and play a critical role in the progression of DR[41]. Exposure to hyperglycemia induces Müller cell proliferation, hypertrophy, and secretion of proinflammatory mediators, thereby disrupting neurovascular integrity[11,12,42]. Consistent with this, HG (25 mmol/L) treatment enhanced viability and proliferation while reducing apoptosis in rMC-1 cells. Given that suppression of pathological gliosis may mitigate neurovascular damage[9], this study focused on the role of PRDX2 in Müller cells. Further scRNA-seq analysis of STZ-diabetic mouse retinas (GSE178121) revealed that Müller cells significantly upregulated both classical gliosis markers (GFAP and nestin) and PRDX2. Additionally, the expression of other PRDX isoforms was differentially altered in these cells, with PRDX1/4/5 being elevated and PRDX3/6 downregulated, suggesting a complex, compensatory activation of antioxidant systems during chronic hyperglycemic stress. In vitro, HG exposure specifically induced PRDX2, but not PRDX6, in rMC-1 cells, while PRDX2 knockdown reversed HG-induced hyperproliferation. Quantitative TMT-based proteomics further confirmed that PRDX2 silencing downregulated nestin, a marker of Müller gliosis, without affecting other PRDX homologs in rMC-1 cells, thereby establishing the specific role of PRDX2 in Müller cell homeostasis. The upregulation of intermediate filament proteins, including GFAP, vimentin, and nestin, in conjunction with pathological hyperproliferation, is a hallmark of Müller cell gliosis[11-13]. Therefore, our data from diabetic murine retinal Müller cells (GSE178121) and HG-stimulated rMC-1 cells implicate PRDX2 as an important driver of gliosis, potentially acting through or in parallel with nestin upregulation.
The small Rho GTPase and its effector ROCK are activated in diabetic complications[43], where they influence cell proliferation, migration, and VEGF-mediated angiogenesis[44-46]. Previous studies have demonstrated that ROCK inhibitors (e.g., Y27632) suppress hypoxia- and oxidative stress-induced proliferation of Müller cells[36,38]. Our TMT-based proteomic and western blot analyses revealed that PRDX2 silencing abolished HG-induced upregulation of RhoA and ROCK1/2, while markedly increasing RhoGDI1 expression, a negative regulator that sequesters RhoA-GDP[47,48]. Notably, HG exposure alone did not alter RhoGDI1 levels, indicating that PRDX2 knockdown inhibits RhoA/ROCK signaling specifically through RhoGDI1 induction. These findings suggest that the suppression of reactive hyperplasia in diabetic Müller cells by PRDX2 knockdown is mediated by disruption of the RhoA/ROCK signaling pathway. Targeting the PRDX2-RhoGDI1-ROCK/RhoA axis represents a promising yet complex therapeutic strategy for DR. However, the ubiquitous expression and essential cytoprotective function of PRDX2 necessitate a highly specific approach. To mitigate potential off-target effects and preserve systemic redox homeostasis, future therapeutic efforts should focus on localized delivery (e.g., intravitreal administration) or the development of agents that selectively disrupt the signaling interaction between PRDX2 and RhoGDI1 specifically within retinal glial cells. This strategy aims to achieve a targeted therapeutic effect in the retina while minimizing systemic consequences.
Consistent with previous studies[49-52], our results indicate that markers of hypertension, liver disease, and kidney disease (such as elevated serum urea, creatinine, and uric acid) may also be associated with an increased DR risk. Reduced serum levels of AST, albumin, A/G ratio, and sodium in DR patients may reflect impaired hepatic synthesis, immune dysfunction, and microvascular damage[51,53]. These findings highlight the importance of comprehensive management of metabolic comorbidities in diabetes.
This study has several limitations. First, although the clinical sample size was adequate for initial analysis, it may not fully capture the heterogeneity of the diabetic population. Future longitudinal studies that systematically account for systemic variables are needed to conclusively establish the disease specificity of PRDX2. Second, while this study establishes a clear link between PRDX2 and Müller cell reactive gliosis, other reactive gliosis markers such as GFAP and vimentin were not examined. Investigating the relationship between PRDX2 and these additional gliotic markers will be valuable for fully delineating its regulatory mechanisms within this critical pathway. Third, the proposed link between PRDX2 and Rho/ROCK signaling requires further validation of upstream and downstream effectors. The precise mechanism by which PRDX2 regulates RhoGDI1—whether at the transcriptional or post-translational level—remains unclear and warrants investigation through approaches such as chromatin immunoprecipitation sequencing, RNA-binding proteomics, or pharmacological intervention. Fourth, while the HG-stimulated rMC-1 cell model is well established for studying Müller cell gliosis, it inherently lacks the multicellular interactions present in the intact retina. Therefore, extending this work to co-culture systems, human retinal organoids, or targeted in vivo approaches will be essential to determine whether PRDX2 knockdown exerts paracrine effects on other retinal cell types, such as neurons and endothelial cells. Finally, it is important to acknowledge that DR is a chronic condition driven by sustained processes, including metabolic memory, accumulation of advanced glycation end-products, and low-grade inflammation[54]. In contrast, our mechanistic insights are derived primarily from acute in vitro HG exposure and public datasets from STZ-induced diabetic mouse models, which may not fully reflect the chronic nature of disease progression. Thus, the PRDX2-RhoGDI1-RhoA/ROCK axis identified in this study likely represents an early maladaptive signaling node that may be progressively reinforced to sustain glial activation. To bridge this acute-to-chronic gap, future studies utilizing prolonged in vitro exposure models, long-term diabetic animal models, or Müller cell-specific conditional knockout systems will be essential to validate the contribution of this pathway to the sustained pathology of DR.
Our findings identify PRDX2 as a promising plasma biomarker for staging DR progression and as a key regulator of Müller cell dysfunction through the RhoGDI1-RhoA/ROCK axis. This study lays the groundwork for future investigations into targeted therapeutic interventions and elucidates a molecular mechanism contributing to DR pathogenesis. Targeting the PRDX2-RhoGDI1-RhoA axis may represent a potential therapeutic strategy for mitigating neuroglial dysfunction in DR.
We thank all the participants for their involvement. We also express our gratitude to the Biomedical Engineering Facility of the National Infrastructures for Translational Medicine, Institute of Clinical Medicine, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College for providing the necessary equipment.
| 1. | Zayed MG, Karsan W, Peto T, Saravanan P, Virgili G, Preiss D. Diabetic Retinopathy and Quality of Life: A Systematic Review and Meta-Analysis. JAMA Ophthalmol. 2024;142:199-207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 24] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 2. | Rorex C, Cardona SM, Church KA, Rodriguez D, Vanegas D, Saldivar R, Faz B, Cardona AE. Astrogliosis in the GFAP-Cre(ERT2):Rosa26(iDTR) Mouse Model Does Not Exacerbate Retinal Microglia Activation or Müller Cell Gliosis under Hypoxic Conditions. Biomolecules. 2024;14:567. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 3. | Jung KI, Han JS, Park CK. Neuroprotective Effects of Nicotinamide (Vitamin B(3)) on Neurodegeneration in Diabetic Rat Retinas. Nutrients. 2022;14:1162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 4. | Li Y, Mitchell W, Elze T, Zebardast N. Association Between Diabetes, Diabetic Retinopathy, and Glaucoma. Curr Diab Rep. 2021;21:38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 51] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 5. | Sadikan MZ, Abdul Nasir NA. Diabetic retinopathy: emerging concepts of current and potential therapy. Naunyn Schmiedebergs Arch Pharmacol. 2023;396:3395-3406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 6. | Martinez-Zapata MJ, Salvador I, Martí-Carvajal AJ, Pijoan JI, Cordero JA, Ponomarev D, Kernohan A, Solà I, Virgili G. Anti-vascular endothelial growth factor for proliferative diabetic retinopathy. Cochrane Database Syst Rev. 2023;3:CD008721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 7. | Morya AK, Ramesh PV, Nishant P, Kaur K, Gurnani B, Heda A, Salodia S. Diabetic retinopathy: A review on its pathophysiology and novel treatment modalities. World J Methodol. 2024;14:95881. [PubMed] [DOI] [Full Text] |
| 8. | Manogaran P, Samardzija M, Schad AN, Wicki CA, Walker-Egger C, Rudin M, Grimm C, Schippling S. Retinal pathology in experimental optic neuritis is characterized by retrograde degeneration and gliosis. Acta Neuropathol Commun. 2019;7:116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 58] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 9. | Conedera FM, Quintela Pousa AM, Presby DM, Mercader N, Enzmann V, Tschopp M. Diverse Signaling by TGFβ Isoforms in Response to Focal Injury is Associated with Either Retinal Regeneration or Reactive Gliosis. Cell Mol Neurobiol. 2021;41:43-62. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 10. | Yu X, Teng Q, Bao K, Chudhary M, Qi H, Zhou W, Che H, Liu J, Ren X, Kong L. Thioredoxin 1 overexpression attenuated diabetes-induced endoplasmic reticulum stress in Müller cells via apoptosis signal-regulating kinase 1. J Cell Biochem. 2023;124:421-433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 11. | Du J, Wang Y, Tu Y, Guo Y, Sun X, Xu X, Liu X, Wang L, Qin X, Zhu M, Song E. A prodrug of epigallocatechin-3-gallate alleviates high glucose-induced pro-angiogenic factor production by inhibiting the ROS/TXNIP/NLRP3 inflammasome axis in retinal Müller cells. Exp Eye Res. 2020;196:108065. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 46] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 12. | Sun Y, Wang D, Ye F, Hu DN, Liu X, Zhang L, Gao L, Song E, Zhang DY. Elevated cell proliferation and VEGF production by high-glucose conditions in Müller cells involve XIAP. Eye (Lond). 2013;27:1299-1307. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 13. | Qi Y, Yao R, Zhang W, Cui Q. KAT1 triggers YTHDF2-mediated ITGB1 mRNA instability to alleviate the progression of diabetic retinopathy. Pharmacol Res. 2021;170:105713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 14. | Liu J, Su G, Gao J, Tian Y, Liu X, Zhang Z. Effects of Peroxiredoxin 2 in Neurological Disorders: A Review of its Molecular Mechanisms. Neurochem Res. 2020;45:720-730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 15. | Zhang Y, Jiang J, Zhang J, Shen H, Wang M, Guo Z, Zang X, Shi H, Gao J, Cai H, Fang X, Qian H, Xu W, Zhang X. CircDIDO1 inhibits gastric cancer progression by encoding a novel DIDO1-529aa protein and regulating PRDX2 protein stability. Mol Cancer. 2021;20:101. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 135] [Cited by in RCA: 132] [Article Influence: 26.4] [Reference Citation Analysis (0)] |
| 16. | Wang W, Wei J, Zhang H, Zheng X, Zhou H, Luo Y, Yang J, Deng Q, Huang S, Fu Z. PRDX2 promotes the proliferation of colorectal cancer cells by increasing the ubiquitinated degradation of p53. Cell Death Dis. 2021;12:605. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 17. | Zhao Y, Liu J, Xiong Z, Gu S, Xia X. Exosome-derived miR-23a-5p inhibits HCC proliferation and angiogenesis by regulating PRDX2 expression: MiR-23a-5p/PRDX2 axis in HCC progression. Heliyon. 2024;10:e23168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 18. | Choi MH, Lee IK, Kim GW, Kim BU, Han YH, Yu DY, Park HS, Kim KY, Lee JS, Choi C, Bae YS, Lee BI, Rhee SG, Kang SW. Regulation of PDGF signalling and vascular remodelling by peroxiredoxin II. Nature. 2005;435:347-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 343] [Cited by in RCA: 305] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 19. | Agrawal-Singh S, Isken F, Agelopoulos K, Klein HU, Thoennissen NH, Koehler G, Hascher A, Bäumer N, Berdel WE, Thiede C, Ehninger G, Becker A, Schlenke P, Wang Y, McClelland M, Krug U, Koschmieder S, Büchner T, Yu DY, Singh SV, Hansen K, Serve H, Dugas M, Müller-Tidow C. Genome-wide analysis of histone H3 acetylation patterns in AML identifies PRDX2 as an epigenetically silenced tumor suppressor gene. Blood. 2012;119:2346-2357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 76] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 20. | Russell-Guzmán J, Américo-Da Silva L, Cadagan C, Maturana M, Palomero J, Estrada M, Barrientos G, Buvinic S, Hidalgo C, Llanos P. Activation of the ROS/TXNIP/NLRP3 pathway disrupts insulin-dependent glucose uptake in skeletal muscle of insulin-resistant obese mice. Free Radic Biol Med. 2024;222:187-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 24] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 21. | Li X, Long H, Peng R, Zou X, Zuo S, Yang Y, Chen M, Yuan H, Liu Z, Wang T, Zhao Q, Guo B, Liu L. A novel role of peroxiredoxin 2 in diabetic kidney disease progression by activating the classically activated macrophages. Sci Rep. 2024;14:28258. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 22. | Alli-Shaik A, Qiu B, Lai SL, Cheung N, Tan G, Neo SP, Tan A, Cheung CMG, Hong W, Wong TY, Wang X, Gunaratne J. System-wide vitreous proteome dissection reveals impaired sheddase activity in diabetic retinopathy. Theranostics. 2022;12:6682-6704. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 23. | Loukovaara S, Nurkkala H, Tamene F, Gucciardo E, Liu X, Repo P, Lehti K, Varjosalo M. Quantitative Proteomics Analysis of Vitreous Humor from Diabetic Retinopathy Patients. J Proteome Res. 2015;14:5131-5143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 105] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 24. | Moreira EF, Kantorow M, Rodriguez IR. Peroxiredoxin 3 (PDRX3) is highly expressed in the primate retina especially in blue cones. Exp Eye Res. 2008;86:452-455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 25. | Wang G, Li X, Li N, Wang X, He S, Li W, Fan W, Li R, Liu J, Hou S. Icariin alleviates uveitis by targeting peroxiredoxin 3 to modulate retinal microglia M1/M2 phenotypic polarization. Redox Biol. 2022;52:102297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 101] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 26. | Huang Y, Zhang Y, Liu Y, Jin Y, Yang H. PRDX4 mitigates diabetic retinopathy by inhibiting reactive gliosis, apoptosis, ER stress, oxidative stress, and mitochondrial dysfunction in Müller cells. J Biol Chem. 2025;301:108111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 27. | Kubo E, Singh DP, Fatma N, Akagi Y. TAT-mediated peroxiredoxin 5 and 6 protein transduction protects against high-glucose-induced cytotoxicity in retinal pericytes. Life Sci. 2009;84:857-864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 42] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 28. | Chen X, Tzekov R, Su M, Zhu Y, Han A, Li W. Hydrogen peroxide-induced oxidative damage and protective role of peroxiredoxin 6 protein via EGFR/ERK signaling pathway in RPE cells. Front Aging Neurosci. 2023;15:1169211. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 29. | Zhang Y, Lai Z, Yuan Z, Qu B, Li Y, Yan W, Li B, Yu W, Cai S, Zhang H. Serum disease-specific IgG Fc glycosylation as potential biomarkers for nonproliferative diabetic retinopathy using mass spectrometry. Exp Eye Res. 2023;233:109555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 30. | Govindarajan G, Mathews S, Srinivasan K, Ramasamy K, Periasamy S. Establishment of human retinal mitoscriptome gene expression signature for diabetic retinopathy using cadaver eyes. Mitochondrion. 2017;36:150-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 31. | Sun L, Wang R, Hu G, Liu H, Lv K, Duan Y, Shen N, Wu J, Hu J, Liu Y, Jin Q, Zhang F, Xu X. Single cell RNA sequencing (scRNA-Seq) deciphering pathological alterations in streptozotocin-induced diabetic retinas. Exp Eye Res. 2021;210:108718. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 67] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 32. | Gautam P, Hamashima K, Chen Y, Zeng Y, Makovoz B, Parikh BH, Lee HY, Lau KA, Su X, Wong RCB, Chan WK, Li H, Blenkinsop TA, Loh YH. Multi-species single-cell transcriptomic analysis of ocular compartment regulons. Nat Commun. 2021;12:5675. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 108] [Cited by in RCA: 85] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 33. | Zhang C, Luo D, Xie H, Yang Q, Liu D, Tang L, Zhang J, Li W, Tian H, Lu L, Sun X, Xu GT, Zhang J. Aquaporin 11 alleviates retinal Müller intracellular edema through water efflux in diabetic retinopathy. Pharmacol Res. 2023;187:106559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 34. | Deng L, Wang Y. MSCs-EXOs containing miR-21a-5p promoted cell proliferation in the rat retinal Müller cell rMC-1 via targeting LATS1. Stem Cell Res Ther. 2025;16:529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 35. | Sarthy VP, Brodjian SJ, Dutt K, Kennedy BN, French RP, Crabb JW. Establishment and characterization of a retinal Müller cell line. Invest Ophthalmol Vis Sci. 1998;39:212-216. [PubMed] |
| 36. | Zhang XH, Feng ZH, Wang XY. The ROCK pathway inhibitor Y-27632 mitigates hypoxia and oxidative stress-induced injury to retinal Müller cells. Neural Regen Res. 2018;13:549-555. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 37. | Qin W, Cheah JS, Xu C, Messing J, Freibaum BD, Boeynaems S, Taylor JP, Udeshi ND, Carr SA, Ting AY. Dynamic mapping of proteome trafficking within and between living cells by TransitID. Cell. 2023;186:3307-3324.e30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 114] [Article Influence: 38.0] [Reference Citation Analysis (0)] |
| 38. | Roda VMP, da Silva RA, Siqueira PV, Lustoza-Costa GJ, Moraes GM, Matsuda M, Hamassaki DE, Santos MF. Inhibition of Rho kinase (ROCK) impairs cytoskeletal contractility in human Müller glial cells without effects on cell viability, migration, and extracellular matrix production. Exp Eye Res. 2024;238:109745. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 39. | Jo YH, Choi GW, Kim ML, Sung KR. Statins Inhibit the Gliosis of MIO-M1, a Müller Glial Cell Line Induced by TRPV4 Activation. Int J Mol Sci. 2022;23:5190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 40. | Chidlow G, Wood JP, Knoops B, Casson RJ. Expression and distribution of peroxiredoxins in the retina and optic nerve. Brain Struct Funct. 2016;221:3903-3925. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 41. | Li Y, Zhu L, Cai MX, Wang ZL, Zhuang M, Tan CY, Xie TH, Yao Y, Wei TT. TGR5 supresses cGAS/STING pathway by inhibiting GRP75-mediated endoplasmic reticulum-mitochondrial coupling in diabetic retinopathy. Cell Death Dis. 2023;14:583. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 56] [Article Influence: 18.7] [Reference Citation Analysis (1)] |
| 42. | Russo B, D'Addato G, Salvatore G, Menduni M, Frontoni S, Carbone L, Camaioni A, Klinger FG, De Felici M, Picconi F, La Sala G. Gliotic Response and Reprogramming Potential of Human Müller Cell Line MIO-M1 Exposed to High Glucose and Glucose Fluctuations. Int J Mol Sci. 2024;25:12877. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 43. | Nagai Y, Matoba K, Yako H, Ohashi S, Sekiguchi K, Mitsuyoshi E, Sango K, Kawanami D, Utsunomiya K, Nishimura R. Rho-kinase inhibitor restores glomerular fatty acid metabolism in diabetic kidney disease. Biochem Biophys Res Commun. 2023;649:32-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 44. | Bryan BA, Dennstedt E, Mitchell DC, Walshe TE, Noma K, Loureiro R, Saint-Geniez M, Campaigniac JP, Liao JK, D'Amore PA. RhoA/ROCK signaling is essential for multiple aspects of VEGF-mediated angiogenesis. FASEB J. 2010;24:3186-3195. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 227] [Cited by in RCA: 221] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 45. | Qin X, He Y, Zhang Y, Li S, Li T, You F, Liu Y. Myosin regulates intracellular force and guides collective cancer cell migration via the FAK-Rho/ROCK feedback loop. Genes Dis. 2023;10:2199-2201. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 46. | Tang Y, He Y, Zhang P, Wang J, Fan C, Yang L, Xiong F, Zhang S, Gong Z, Nie S, Liao Q, Li X, Li X, Li Y, Li G, Zeng Z, Xiong W, Guo C. LncRNAs regulate the cytoskeleton and related Rho/ROCK signaling in cancer metastasis. Mol Cancer. 2018;17:77. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 152] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 47. | Mosaddeghzadeh N, Ahmadian MR. The RHO Family GTPases: Mechanisms of Regulation and Signaling. Cells. 2021;10:1831. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 323] [Cited by in RCA: 274] [Article Influence: 54.8] [Reference Citation Analysis (0)] |
| 48. | Sinha K, Kumawat A, Jang H, Nussinov R, Chakrabarty S. Molecular mechanism of regulation of RhoA GTPase by phosphorylation of RhoGDI. Biophys J. 2024;123:57-67. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 49. | Ceriello A, Prattichizzo F. Variability of risk factors and diabetes complications. Cardiovasc Diabetol. 2021;20:101. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 113] [Article Influence: 22.6] [Reference Citation Analysis (1)] |
| 50. | Reichert KP, Castro MFV, Assmann CE, Bottari NB, Miron VV, Cardoso A, Stefanello N, Morsch VMM, Schetinger MRC. Diabetes and hypertension: Pivotal involvement of purinergic signaling. Biomed Pharmacother. 2021;137:111273. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 51. | Hu F, Lou Y, Shi J, Cao L, Wang C, Ma J, Peng X, Xu S, Chen H, Zhao D, Zhao Y, Guo C, Liu D, Zhou Q, Li Q, Liu F, Tian G, Wu X, Qie R, Han M, Huang S, Zhao P, Zhang M, Hu D, Qin P. Baseline serum albumin and its dynamic change is associated with type 2 diabetes risk: A large cohort study in China. Diabetes Metab Res Rev. 2020;36:e3296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 52. | Song JJ, Han XF, Chen JF, Liu KM. Correlation between glycated hemoglobin A1c, urinary microalbumin, urinary creatinine, β2 microglobulin, retinol binding protein and diabetic retinopathy. World J Diabetes. 2023;14:1103-1111. [PubMed] [DOI] [Full Text] |
| 53. | Chen X, Hu G, He B, Cao Z, He J, Luo H, Li Y, Yu Q. Effect of brominated flame retardants exposure on liver function and the risk of non-alcoholic fatty liver disease in the US population. Ecotoxicol Environ Saf. 2024;273:116142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 54. | Zhu H, Li B, Huang T, Wang B, Li S, Yu K, Cai L, Ye Y, Chen S, Zhu H, Xu J, Lu Q, Ji L. Update in the molecular mechanism and biomarkers of diabetic retinopathy. Biochim Biophys Acta Mol Basis Dis. 2025;1871:167758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 20.0] [Reference Citation Analysis (0)] |