Published online Aug 15, 2025. doi: 10.4239/wjd.v16.i8.108839
Revised: May 31, 2025
Accepted: July 4, 2025
Published online: August 15, 2025
Processing time: 112 Days and 15.2 Hours
Although recent studies have reported the reliability of triglyceride-glucose index (TyG) as a biomarker for evaluation of insulin resistance, the research exploring the association of TyG-body mass index (BMI) with major cardiovascular out
To ascertain whether TyG-BMI correlates with cardiovascular outcomes and mortality risk in individuals diagnosed with T2DM.
Data were drawn from the ACCORD trial and its subsequent follow-up (ACCORDION), which together included 10190 participants. To evaluate the associations of TyG-BMI with future cardiovascular outcomes and overall mortality risk among T2DM patients, Cox’s proportional-hazards model was employed. Non-linear associations and thresholds were investigated by per
Applying multivariable Cox regression analysis, we identified a significant correlation of TyG-BMI with both cardiovascular outcomes and overall mortality. Specifically, after adjusting for multiple confounders, the cardiovascular mortality, congestive heart failure (CHF) and overall mortality risks in the highest TyG-BMI quartile were 1.74, 2.65 and 1.42 times greater, respectively, when compared to the lowest quartile. The analysis revealed that TyG-BMI was linked nonlinearly to both types of mortality, while a two-stage linear regression discerned inflection points at 252.77 for cardiovascular mortality and 245.90 for overall mortality. TyG-BMI was found to correlate positively with future CHF. Additionally, introducing TyG-BMI into conventional prediction models resulted in a substantial improvement of their predictability for cardiovascular outcomes and overall mortality when compared to incorporating TyG or BMI alone.
Among T2DM individuals, a higher TyG-BMI is linked strongly to elevated risks of cardiovascular outcomes and overall mortality, highlighting its utility as a predictive marker for these risks.
Core Tip: This study uncovers a novel association between triglyceride-glucose index (TyG)-body mass index (BMI) and both cardiovascular outcomes and overall mortality in patients with type 2 diabetes mellitus (T2DM), based on the high-risk Action to Control Cardiovascular Risk in Diabetes cohort. It also marks the first identification of TyG-BMI as a robust predictor of future congestive heart failure, providing fresh perspectives on the risk stratification for complex T2DM patients.
- Citation: Liu MJ, Xiang SM, Hu XQ. Triglyceride glucose-body mass index is associated with cardiovascular outcomes and overall mortality in type-2 diabetes mellitus patients. World J Diabetes 2025; 16(8): 108839
- URL: https://www.wjgnet.com/1948-9358/full/v16/i8/108839.htm
- DOI: https://dx.doi.org/10.4239/wjd.v16.i8.108839
The past few decades have witnessed the emergence of type 2 diabetes mellitus (T2DM) as a major public health concern worldwide, with its prevalence rising alongside aging populations. T2DM is now among the world’s most rapidly expanding chronic diseases[1,2]. The International Diabetes Federation’s 2021 report states that 537 million people globally live with T2DM, a figure projected to rise by 46% to 783 million by 2045[3]. T2DM imposes a considerable social and economic burden on patients and their families, with its economic impact estimated at more than double that for individuals without diabetes[4]. T2DM is intricately associated with cardiovascular disease (CVD), placing diabetics at a markedly elevated risk of developing CVD compared to their non-diabetic counterparts. CVD is responsible for nearly half of all fatalities among individuals with diabetes[5-7]. Despite progress in diabetes management that has led to a reduction in the incidence of CVD, it continues to be the primary cause of mortality among diabetic individuals, un
A condition known as insulin resistance (IR) is marked by diminished insulin sensitivity and response[8-10]. Higher levels of IR are significantly linked to CVD risk in both diabetic and non-diabetic individuals. In those with diabetes, IR contributes to the development of CVD[11,12]. Hence, long-term IR monitoring and management may be beneficial in lowering cardiovascular mortality within the T2DM population. While hyperinsulinemic-euglycemic clamp (HEC) is widely considered the gold standard for evaluating IR, its complexity and high cost restrict its application in small-scale studies and broader clinical settings. Despite the frequent employment of the Homeostasis Model Assessment for Insulin Resistance (HOMA-IR) for assessing IR, it has limited accuracy in patients undergoing insulin therapy[13,14]. Recently, triglyceride-glucose index (TyG) has gained widespread acceptance as an IR assessment tool[15]. Guerrero-Romero et al[16] reported a high association between the TyG and the HEC standard for diagnosing IR, demonstrating strong sensitivity and specificity in assessing individual IR cases. Recent studies further corroborate the clinical utility of TyG in forecasting cardiovascular risk across diabetic and non-diabetic individuals[17-21].
TyG index, computed using the formula ln [fasting triglycerides (mg/dL) × fasting glucose (mg/dL)/2], is well-acknowledged as a surrogate marker for IR. TyG-body mass index (BMI), the product of TyG index and BMI (a standard anthropometric measure of general adiposity) incorporating both metabolic and anthropometric parameters, has recently been proposed as a novel and practical indicator for predicting metabolic disorders and cardiovascular risk. Evidence has suggested the particular effectiveness of TyG-BMI in diagnosing IR[22-25]. Nevertheless, its prognostic value for cardiovascular events and overall mortality in T2DM individuals remains understudied and debated. Existing research has predominantly focused on the general T2DM population, which often consists of younger individuals with relatively fewer comorbidities. In contrast, the ACCORD study cohort provides a unique opportunity to assess TyG-BMI’s predictive capacity within a higher-risk demographic. This cohort includes middle-aged and elderly individuals with T2DM, characterized by poor glycemic control, elevated cardiovascular risk, and a high prevalence of complex comorbidities such as heart failure and proteinuria. These distinct clinical features allow for a more comprehensive exploration of TyG-BMI’s relevance in real-world, high-risk scenarios. Leveraging data from the ACCORD trial and its ACCORDION follow-up, the present study attempts to evaluate the correlation of TyG-BMI with multiple cardiovascular outcomes, including cardiovascular mortality, congestive heart failure (CHF), and overall mortality. Our findings may offer important insights into the clinical utility of TyG-BMI in risk stratification and long-term prognosis for individuals with advanced-stage T2DM.
The present study employed a post-hoc analysis, drawing data from the ACCORD/ACCORDION Trial (Clini
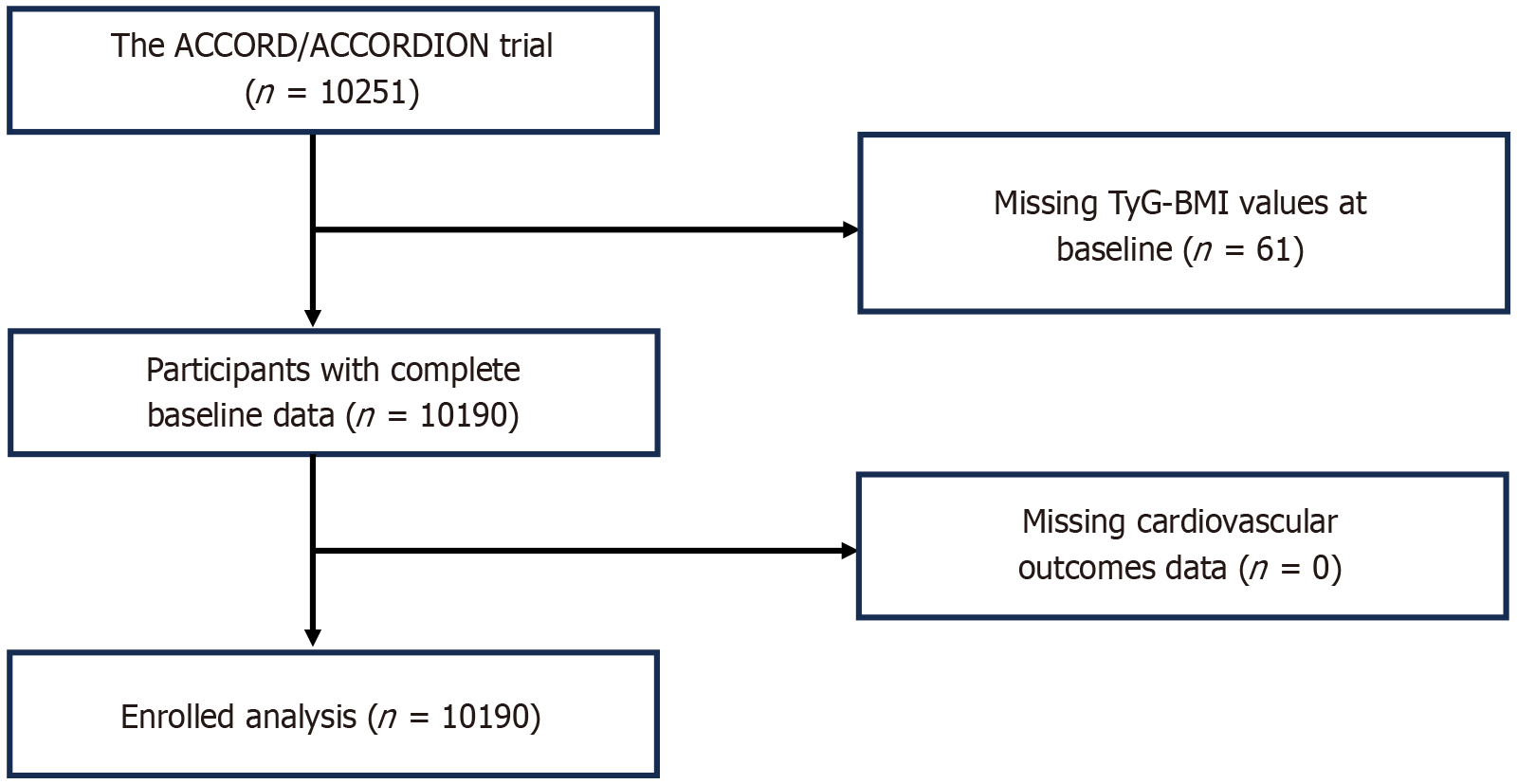
Data on demographic and clinical factors were collected, including age, sex, education, ethnicity, treatments, medical records, health checkups, laboratory test results, and past medication use. Out of the 10251 participants, 61 lacked baseline TyG-BMI data, leading to a final analysis sample of 10190 individuals (Figure 1). The respective formulas for calculating TyG and TyG-BMI were: TyG = ln[triglycerides (TG; mg/dL) × fasting blood glucose (FBG; mg/dL)/2]; BMI = weight/height2 (kg/m2); TyG-BMI = ln[TG (mg/dL) × FBG (mg/dL)/2] × weight/height2 (kg/m2).
The primary and secondary endpoints of this study were the severity of cardiovascular outcomes (including cardiovascular mortality and CHF), and the overall mortality.
SPSS 26.0 (IBM, Armonk, NY, United States), R software (The R Foundation, Vienna, Austria), combined with Empo
By applying the Cox’s proportional-hazards models, the correlations of TyG-BMI with cardiovascular outcomes and overall mortality were assessed. Prior to performing the multivariate adjusted Cox proportional hazards regression analysis, an association assessment between all collected variables and future cardiovascular mortality rates was conducted through univariate analysis to identify possible confounding factors. Every covariate was included based on a sound rationale or clinical significance. Variables deemed statistically significant in the univariate analysis (P < 0.05, Supplementary Table 1) were subsequently incorporated into the multivariate analysis. Additionally, we also incor
A range of sensitivity analyses were conducted to ascertain the robustness of our findings. Initially, we performed multiple imputation to address missing data and reduce the potential bias associated with incomplete covariate information. Subsequently, participants with a baseline history of CVD were excluded to minimize confounding effects from pre-existing vascular conditions. We also removed those with a history of heart failure at baseline, given its strong association with adverse cardiovascular outcomes. Lastly, to mitigate reverse causality and early mortality bias, we eliminated participants who either experienced cardiovascular events or passed away within the initial 3-year period of follow-up.
The baseline profile comprised 10190 patients, with 61.47% being males 62.81 ± 6.64 years in mean age. The mean baseline levels were as follows: TC: 183.30 ± 41.85, LDL-C: 104.90 ± 33.91, TG: 190.06 ± 148.27, fasting plasma glucose (FPG): 175.19 ± 56.18, TyG: 9.49 ± 0.73, and TyG-BMI: 306.18 ± 58.74. Depending on the quartiles of baseline TyG-BMI, we stratified the patients as follows: Quartile 1 (234.32 ± 21.93), Quartile 2 (282.91 ± 11.31), Quartile 3 (322.94 ± 12.37), and Quartile 4 (384.54 ± 30.17). Table 1 outlines the baseline clinical, and demographic traits, along with laboratory indicators for the four groups. We identified significant differences among the groups in the following aspects: Age, BMI, sex, race, living conditions, educational level, proteinuria, cardiac failure, depression, alcohol consumption, status of smoking, diabetes duration, previous hypertension, DBP, HR, FPG, HbA1c, TC, TG, high density lipoprotein, glomerular filtration rate, CCB, angiotensin receptor blocker, angiotensin converting enzyme inhibitors, beta-blockers, thiazolidinediones, insulins, biguanides, statins, sulfonylureas, and aspirin.
| Q1 (n = 2548; 131.06-263.12) | Q2 (n = 2547; 263.12-302.56) | Q3 (n = 2547; 302.56-345.60) | Q4 (n = 2548; 345.60-536.15) | P value | |
| TyG-BMI | 234.32 ± 21.93 | 282.91 ± 11.31 | 322.94 ± 12.37 | 384.54 ± 30.17 | < 0.001 |
| Age (years) | 64.32 ± 7.20 | 63.44 ± 6.57 | 62.37 ± 6.41 | 61.13 ± 5.89 | < 0.001 |
| Sex | < 0.001 | ||||
| Male | 1697 (66.60) | 1684 (66.12) | 1545 (60.66) | 1338 (52.51) | |
| Female | 851 (33.40) | 863 (33.88) | 1002 (39.34) | 1210 (47.49) | |
| Race | < 0.001 | ||||
| White | 1197 (46.98) | 1533 (60.19) | 1704 (66.90) | 1933 (75.86) | |
| Non-white | 1351 (53.02) | 1014 (39.81) | 843 (33.10) | 615 (24.14) | |
| Education | < 0.001 | ||||
| Less than high school graduate | 423 (16.62) | 418 (16.41) | 359 (14.11) | 301 (11.82) | |
| High school grad (or GED) | 689 (27.07) | 640 (25.13) | 683 (26.84) | 678 (26.63) | |
| Some college or technical school | 698 (27.43) | 807 (31.68) | 872 (34.26) | 964 (37.86) | |
| College graduate or more | 735 (28.88) | 682 (26.78) | 631 (24.79) | 603 (23.68) | |
| CVD history | 885 (34.73) | 929 (36.47) | 905 (35.53) | 863 (33.87) | 0.246 |
| Duration of diabetes (years) | 12.12 ± 8.16 | 10.96 ± 7.64 | 10.19 ± 7.19 | 9.93 ± 7.15 | < 0.001 |
| Previous hypertension | 1837 (72.10) | 1901 (74.64) | 1943 (76.29) | 1998 (78.41) | < 0.001 |
| Previous hyperlipidemia | 1742 (68.37) | 1792 (70.36) | 1776 (69.73) | 1816 (71.27) | 0.144 |
| Proteinuria | 433 (16.99) | 472 (18.53) | 525 (20.62) | 597 (23.43) | < 0.001 |
| Heart failure | 93 (3.65) | 113 (4.44) | 102 (4.01) | 179 (7.03) | < 0.001 |
| Depression | 394 (15.46) | 545 (21.40) | 612 (24.05) | 861 (33.79) | < 0.001 |
| Living alone | 2082 (81.71) | 2033 (79.82) | 2025 (79.57) | 1985 (77.90) | 0.009 |
| Smoking | < 0.001 | ||||
| Yes | 1394 (54.71) | 1512 (59.36) | 1532 (60.15) | 1495 (58.67) | |
| No | 1154 (45.29) | 1035 (40.64) | 1015 (39.85) | 1053 (41.33) | |
| Alcohol | < 0.001 | ||||
| Yes | 630 (24.73) | 664 (26.08) | 623 (24.49) | 515 (20.22) | |
| No | 1918 (75.27) | 1882 (73.92) | 1921 (75.51) | 2032 (79.78) | |
| BMI (kg/m2) | 26.08 ± 2.47 | 30.21 ± 2.23 | 33.76 ± 2.56 | 38.86 ± 3.30 | < 0.001 |
| SBP (mmHg) | 136.45 ± 17.19 | 136.34 ± 17.01 | 136.47 ± 16.97 | 136.13 ± 17.28 | 0.905 |
| DBP (mmHg) | 72.71 ± 10.55 | 74.41 ± 10.53 | 75.72 ± 10.42 | 76.70 ± 10.73 | < 0.001 |
| Heart rate (bpm) | 71.07 ± 11.54 | 71.71 ± 11.68 | 73.40 ± 11.70 | 74.52 ± 11.78 | < 0.001 |
| FPG (mg/dL) | 157.67 ± 54.17 | 170.58 ± 53.78 | 178.59 ± 53.77 | 193.92 ± 56.75 | < 0.001 |
| HbA1c (%) | 8.21 ± 1.07 | 8.26 ± 1.05 | 8.29 ± 1.01 | 8.45 ± 1.09 | < 0.001 |
| TC (mg/dL) | 174.85 ± 38.80 | 180.92 ± 40.15 | 183.96 ± 41.12 | 193.46 ± 44.91 | < 0.001 |
| TG (mg/dL) | 124.30 ± 73.92 | 170.07 ± 99.22 | 202.26 ± 129.90 | 263.60 ± 213.27 | < 0.001 |
| VLDL-C (mg/dL) | 24.67 ± 13.68 | 33.44 ± 18.32 | 39.08 ± 22.08 | 48.92 ± 32.34 | < 0.001 |
| LDL-C (mg/dL) | 104.50 ± 32.86 | 105.44 ± 33.44 | 104.23 ± 33.84 | 105.43 ± 35.45 | 0.558 |
| HDL-C (mg/dL) | 45.68 ± 12.99 | 42.04 ± 11.33 | 40.65 ± 10.81 | 39.12 ± 10.17 | < 0.001 |
| eGFR (mL/min/1.73 m2) | 91.07 ± 29.02 | 90.32 ± 28.28 | 91.68 ± 25.43 | 91.07 ± 25.62 | 0.041 |
| Medications | |||||
| ARB/ACEI | 1672 (65.62) | 1762 (69.18) | 1794 (70.44) | 1834 (71.98) | < 0.001 |
| CCB | 472 (18.52) | 452 (17.75) | 486 (19.08) | 541 (21.23) | 0.012 |
| β-blockers | 658 (25.89) | 729 (28.70) | 815 (32.09) | 862 (33.92) | < 0.001 |
| Biguanides | 1561 (61.26) | 1653 (64.90) | 1666 (65.44) | 1638 (64.29) | 0.009 |
| Thiazolidinediones | 496 (19.47) | 512 (20.10) | 590 (23.17) | 648 (25.43) | < 0.001 |
| Sulfonylureas | 1413 (55.46) | 1374 (53.95) | 1353 (53.14) | 1305 (51.22) | 0.023 |
| Insulins | 818 (32.10) | 853 (33.49) | 902 (35.41) | 988 (38.78) | < 0.001 |
| Statins | 1642 (64.75) | 1656 (65.33) | 1636 (64.41) | 1531 (60.28) | < 0.001 |
| Aspirin | 1351 (53.29) | 1375 (54.26) | 1451 (57.22) | 1371 (54.02) | 0.027 |
| Cholesterol absorption inhibitors | 45 (1.78) | 58 (2.29) | 50 (1.97) | 54 (2.13) | 0.608 |
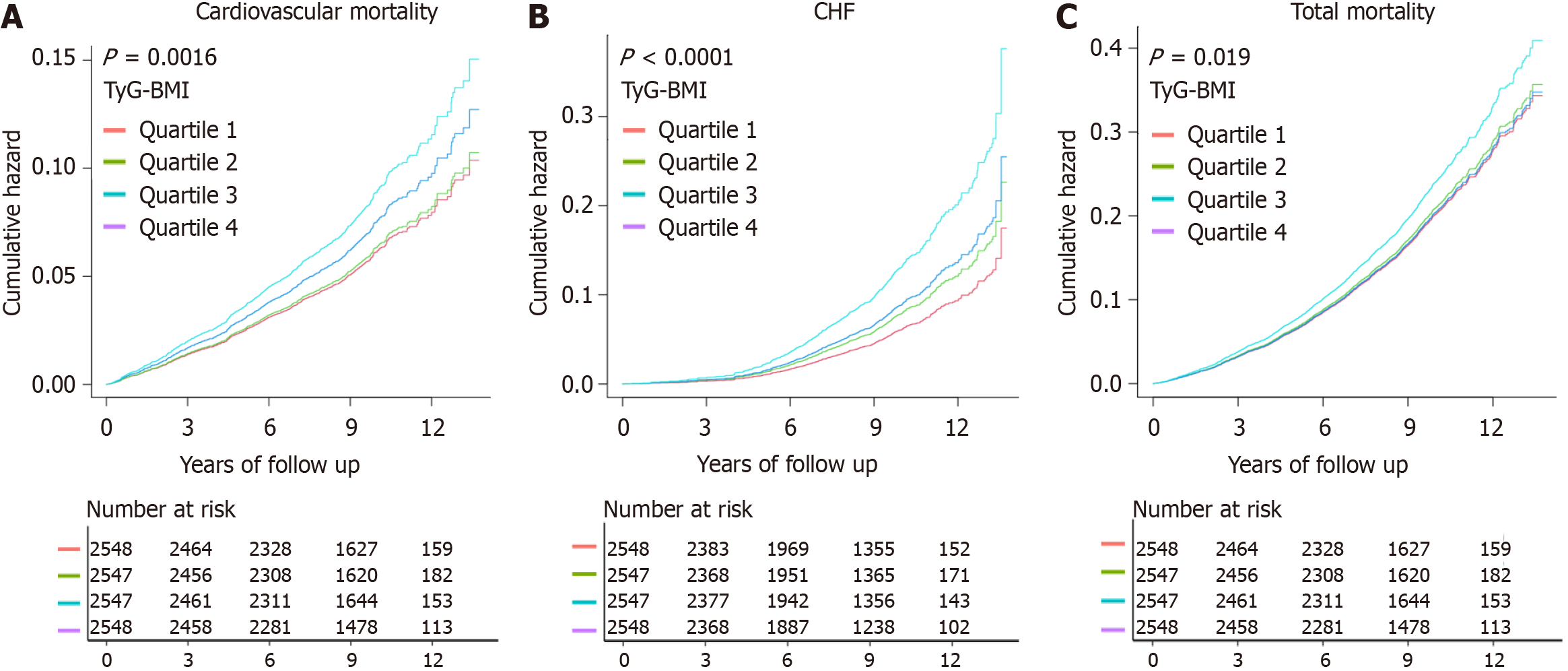
During a median follow-up period of 8.82 years, 662 patients with T2DM (6.50%) died from cardiovascular causes, while 690 (6.77%) patients died from CHF, contributing to an overall mortality count of 1942 (19.06%). Cumulative cardiovascular and overall mortality risks were determined by the Kaplan-Meier survival analysis, which revealed a link between elevated TyG-BMI and heightened cumulative risks for both parameters in the future (cardiovascular mortality: P = 0.0016; CHF: P < 0.0001; overall mortality: P = 0.019; Figure 2). Utilizing three multivariate regression models, the correlation of TyG-BMI with the incidence of outcome events was analyzed (Table 2). Model 1 was adjusted for gender, race, age, educational level, history of CVD, previous hypertension, previous hyperlipidemia, SBP, and DBP. Building upon Model 1, Model 2 further adjusted for the diabetes duration, proteinuria, cardiac failure, status of smoking, HbA1c, eGFR, TC, and LDL-C. Model 3, expanded upon Model 2, additionally adjusted for the use of such medications as CCB, beta-blockers, thiazolidinediones, cholesterol absorption blockers, insulins, biguanides, statins, and aspirin.
| Outcome | Non-adjusted | Model 1 | Model 2 | Model 3 | ||||
| HR (95%CI) | P value | HR (95%CI) | P value | HR (95%CI) | P value | HR (95%CI) | P value | |
| Cardiovascular mortality | ||||||||
| TyG-BMI | ||||||||
| Q1 | Reference | Reference | Reference | Reference | ||||
| Q2 | 1.03 (0.82, 1.30) | 0.7775 | 1.06 (0.84, 1.34) | 0.6247 | 1.05 (0.83, 1.33) | 0.6576 | 1.04 (0.82, 1.32) | 0.7537 |
| Q3 | 1.23 (0.98, 1.53) | 0.0710 | 1.39 (1.10, 1.74) | 0.0050 | 1.39 (1.10, 1.75) | 0.0056 | 1.36 (1.07, 1.71) | 0.0108 |
| Q4 | 1.45 (1.17, 1.80) | 0.0007 | 1.93 (1.53, 2.42) | < 0.0001 | 1.81 (1.42, 2.31) | < 0.0001 | 1.74 (1.36, 2.22) | < 0.0001 |
| Per 1 SD | 1.14 (1.05, 1.23) | 0.0009 | 1.27 (1.17, 1.38) | < 0.0001 | 1.24 (1.13, 1.35) | < 0.0001 | 1.22 (1.11, 1.33) | < 0.0001 |
| P for trend | 0.0002 | < 0.0001 | < 0.0001 | < 0.0001 | ||||
| TyG | ||||||||
| Q1 | Reference | Reference | Reference | Reference | ||||
| Q2 | 0.90 (0.71, 1.14) | 0.3720 | 0.89 (0.71, 1.13) | 0.3514 | 0.90 (0.71, 1.14) | 0.3819 | 0.89 (0.70, 1.13) | 0.3308 |
| Q3 | 1.19 (0.96, 1.48) | 0.1156 | 1.21 (0.96, 1.51) | 0.1015 | 1.17 (0.93, 1.49) | 0.1859 | 1.19 (0.93, 1.51) | 0.1608 |
| Q4 | 1.37 (1.11, 1.70) | 0.0033 | 1.46 (1.17, 1.82) | 0.0009 | 1.48 (1.12, 1.96) | 0.0058 | 1.43 (1.08, 1.89) | 0.0130 |
| Per 1 SD | 1.15 (1.07, 1.24) | 0.0003 | 1.19 (1.09, 1.28) | < 0.0001 | 1.23 (1.09, 1.39) | 0.0008 | 1.20 (1.07, 1.36) | 0.0024 |
| P for trend | 0.0003 | < 0.0001 | 0.0027 | 0.0046 | ||||
| BMI | ||||||||
| Q1 | Reference | Reference | Reference | Reference | ||||
| Q2 | 1.02 (0.82, 1.28) | 0.8488 | 1.03 (0.82, 1.30) | 0.7766 | 1.04 (0.83, 1.31) | 0.7508 | 1.02 (0.81, 1.29) | 0.8433 |
| Q3 | 1.15 (0.92, 1.43) | 0.2107 | 1.30 (1.04, 1.63) | 0.0201 | 1.30 (1.04, 1.64) | 0.0213 | 1.29 (1.03, 1.63) | 0.0266 |
| Q4 | 1.31 (1.06, 1.62) | 0.0143 | 1.69 (1.34, 2.11) | < 0.0001 | 1.61 (1.28, 2.02) | < 0.0001 | 1.56 (1.23, 1.97) | 0.0002 |
| Per 1 SD | 1.08 (1.00, 1.17) | 0.0403 | 1.20 (1.11, 1.31) | < 0.0001 | 1.18 (1.09, 1.29) | < 0.0001 | 1.17 (1.07, 1.27) | 0.0003 |
| P for trend | 0.0073 | < 0.0001 | < 0.0001 | < 0.0001 | ||||
| CHF | ||||||||
| TyG-BMI | ||||||||
| Q1 | Reference | Reference | Reference | Reference | ||||
| Q2 | 1.29 (1.02, 1.64) | 0.0331 | 1.34 (1.05, 1.70) | 0.0176 | 1.29 (1.01, 1.65) | 0.0376 | 1.29 (1.01, 1.66) | 0.0393 |
| Q3 | 1.46 (1.16, 1.84) | 0.0015 | 1.77 (1.40, 2.25) | < 0.0001 | 1.75 (1.37, 2.23) | < 0.0001 | 1.71 (1.34, 2.19) | < 0.0001 |
| Q4 | 2.15 (1.73, 2.68) | < 0.0001 | 3.03 (2.40, 3.83) | < 0.0001 | 2.70 (2.12, 3.45) | < 0.0001 | 2.65 (2.07, 3.40) | < 0.0001 |
| Per 1 SD | 1.32 (1.23, 1.42) | < 0.0001 | 1.52 (1.41, 1.65) | < 0.0001 | 1.46 (1.34, 1.58) | < 0.0001 | 1.45 (1.33, 1.58) | < 0.0001 |
| P for trend | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | ||||
| TyG | ||||||||
| Q1 | Reference | Reference | Reference | Reference | ||||
| Q2 | 1.22 (0.97, 1.52) | 0.0855 | 1.21 (0.97, 1.52) | 0.0902 | 1.22 (0.97, 1.53) | 0.0829 | 1.23 (0.98, 1.54) | 0.0788 |
| Q3 | 1.13 (0.90, 1.42) | 0.2804 | 1.16 (0.92, 1.47) | 0.1966 | 1.10 (0.86, 1.40) | 0.4420 | 1.12 (0.88, 1.43) | 0.3721 |
| Q4 | 1.56 (1.26, 1.93) | < 0.0001 | 1.76 (1.41, 2.20) | < 0.0001 | 1.62 (1.24, 2.13) | 0.0004 | 1.61 (1.22, 2.12) | 0.0007 |
| Per 1 SD | 1.18 (1.09, 1.27) | < 0.0001 | 1.25 (1.15, 1.35) | < 0.0001 | 1.26 (1.12, 1.42) | 0.0001 | 1.26 (1.12, 1.41) | 0.0002 |
| P for trend | 0.0002 | < 0.0001 | 0.0035 | 0.0042 | ||||
| BMI | ||||||||
| Q1 | Reference | Reference | Reference | Reference | ||||
| Q2 | 1.39 (1.10, 1.75) | 0.0054 | 1.44 (1.14, 1.82) | 0.0021 | 1.37 (1.09, 1.74) | 0.0082 | 1.40 (1.10, 1.77) | 0.0062 |
| Q3 | 1.32 (1.05, 1.67) | 0.0194 | 1.54 (1.21, 1.95) | 0.0004 | 1.53 (1.20, 1.94) | 0.0006 | 1.53 (1.20, 1.95) | 0.0006 |
| Q4 | 2.00 (1.61, 2.49) | < 0.0001 | 2.67 (2.12, 3.35) | < 0.0001 | 2.43 (1.92, 3.07) | < 0.0001 | 2.41 (1.89, 3.06) | < 0.0001 |
| Per 1 SD | 1.27 (1.18, 1.37) | < 0.0001 | 1.44 (1.33, 1.56) | < 0.0001 | 1.39 (1.28, 1.50) | < 0.0001 | 1.39 (1.28, 1.51) | < 0.0001 |
| P for trend | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | ||||
| Total mortality | ||||||||
| TyG-BMI | ||||||||
| Q1 | Reference | Reference | Reference | Reference | ||||
| Q2 | 1.04 (0.91, 1.18) | 0.5610 | 1.09 (0.96, 1.24) | 0.1740 | 1.07 (0.94, 1.22) | 0.3110 | 1.06 (0.93, 1.21) | 0.3553 |
| Q3 | 1.01 (0.89, 1.15) | 0.8545 | 1.18 (1.04, 1.35) | 0.0131 | 1.16 (1.01, 1.32) | 0.0337 | 1.13 (0.99, 1.30) | 0.0708 |
| Q4 | 1.19 (1.05, 1.35) | 0.0059 | 1.63 (1.42, 1.86) | < 0.0001 | 1.48 (1.29, 1.70) | < 0.0001 | 1.42 (1.23, 1.64) | < 0.0001 |
| Per 1 SD | 1.05 (1.00, 1.10) | 0.0316 | 1.19 (1.13, 1.25) | < 0.0001 | 1.14 (1.08, 1.20) | < 0.0001 | 1.12 (1.07, 1.18) | < 0.0001 |
| P for trend | 0.0123 | < 0.0001 | < 0.0001 | < 0.0001 | ||||
| TyG | ||||||||
| Q1 | Reference | Reference | Reference | Reference | ||||
| Q2 | 0.90 (0.79, 1.03) | 0.1319 | 0.91 (0.80, 1.04) | 0.1749 | 0.91 (0.80, 1.04) | 0.1570 | 0.91 (0.80, 1.04) | 0.1817 |
| Q3 | 1.03 (0.91, 1.17) | 0.6588 | 1.06 (0.93, 1.21) | 0.3577 | 1.00 (0.87, 1.15) | 0.9752 | 1.03 (0.89, 1.18) | 0.7184 |
| Q4 | 1.18 (1.05, 1.34) | 0.0076 | 1.31 (1.15, 1.49) | < 0.0001 | 1.21 (1.03, 1.41) | 0.0226 | 1.21 (1.03, 1.42) | 0.0212 |
| Per 1 SD | 1.07 (1.02, 1.12) | 0.0034 | 1.12 (1.07, 1.18) | < 0.0001 | 1.08 (1.01, 1.15) | 0.0315 | 1.08 (1.01, 1.15) | 0.0338 |
| P for trend | 0.0014 | < 0.0001 | 0.0270 | 0.0185 | ||||
| BMI | ||||||||
| Q1 | Reference | Reference | Reference | Reference | ||||
| Q2 | 1.03 (0.91, 1.17) | 0.6020 | 1.08 (0.95, 1.22) | 0.2541 | 1.06 (0.93, 1.21) | 0.3746 | 1.05 (0.92, 1.20) | 0.4544 |
| Q3 | 0.96 (0.84, 1.09) | 0.5209 | 1.10 (0.96, 1.25) | 0.1550 | 1.09 (0.96, 1.25) | 0.1869 | 1.07 (0.94, 1.23) | 0.2924 |
| Q4 | 1.11 (0.98, 1.25) | 0.1122 | 1.47 (1.29, 1.68) | < 0.0001 | 1.39 (1.22, 1.59) | < 0.0001 | 1.34 (1.17, 1.53) | < 0.0001 |
| Per 1 SD | 1.02 (0.98, 1.07) | 0.3059 | 1.15 (1.09, 1.20) | < 0.0001 | 1.12 (1.07, 1.18) | < 0.0001 | 1.11 (1.05, 1.16) | < 0.0001 |
| P for trend | 0.2515 | < 0.0001 | < 0.0001 | < 0.0001 | ||||
With TyG-BMI considered a continuous variable and after a comprehensive confounder adjustment, each 1-SD rise in TyG-BMI corresponded to a 22% elevated cardiovascular death risk (HR: 1.22, 95%CI: 1.11-1.33, P < 0.0001), a 45% higher risk of CHF (HR: 1.45, 95%CI: 1.33-1.58), and a 12% elevated risk of all-cause mortality (HR: 1.12, 95%CI: 1.07-1.18, P < 0.0001). In Model 3, following the potential confounder adjustment, TyG-BMI still correlated positively with the cumulative cardiovascular death risk (TyG-BMI Q2: HR: 1.04, 95%CI: 0.82-1.32, P = 0.7537; TyG-BMI Q3: HR: 1.36, 95%CI: 1.07-1.71, P = 0.0108; TyG-BMI Q4: HR: 1.74, 95%CI: 1.36-2.22, P < 0.0001). Moreover, baseline TyG-BMI exhibited significant associations with CHF (TyG-BMI Q2: HR: 1.29, 95%CI: 1.01-1.66, P = 0.0393; TyG-BMI Q3: HR: 1.71, 95%CI: 1.34-2.19, P < 0.0001; TyG-BMI Q4: HR: 2.65, 95%CI: 2.07-3.40, P < 0.0001) and overall mortality (TyG-BMI Q2: HR: 1.06, 95%CI: 0.93-1.21, P = 0.3553; TyG-BMI Q3: HR: 1.13, 95%CI: 0.99-1.30, P = 0.0708; TyG-BMI Q4: HR: 1.42, 95%CI: 1.23-1.64, P < 0.0001). An upward trend regarding the cardiovascular mortality, CHF and overall mortality risks was noted as TyG-BMI increased (cardiovascular mortality: P-trend < 0.0001; CHF: P-trend < 0.0001; overall mortality: P-trend < 0.0001). We also found that compared to TyG or BMI alone, TyG-BMI displayed more robust correlations with and hazard ratios concerning future cardiovascular outcomes and overall mortality risk in T2DM patients.
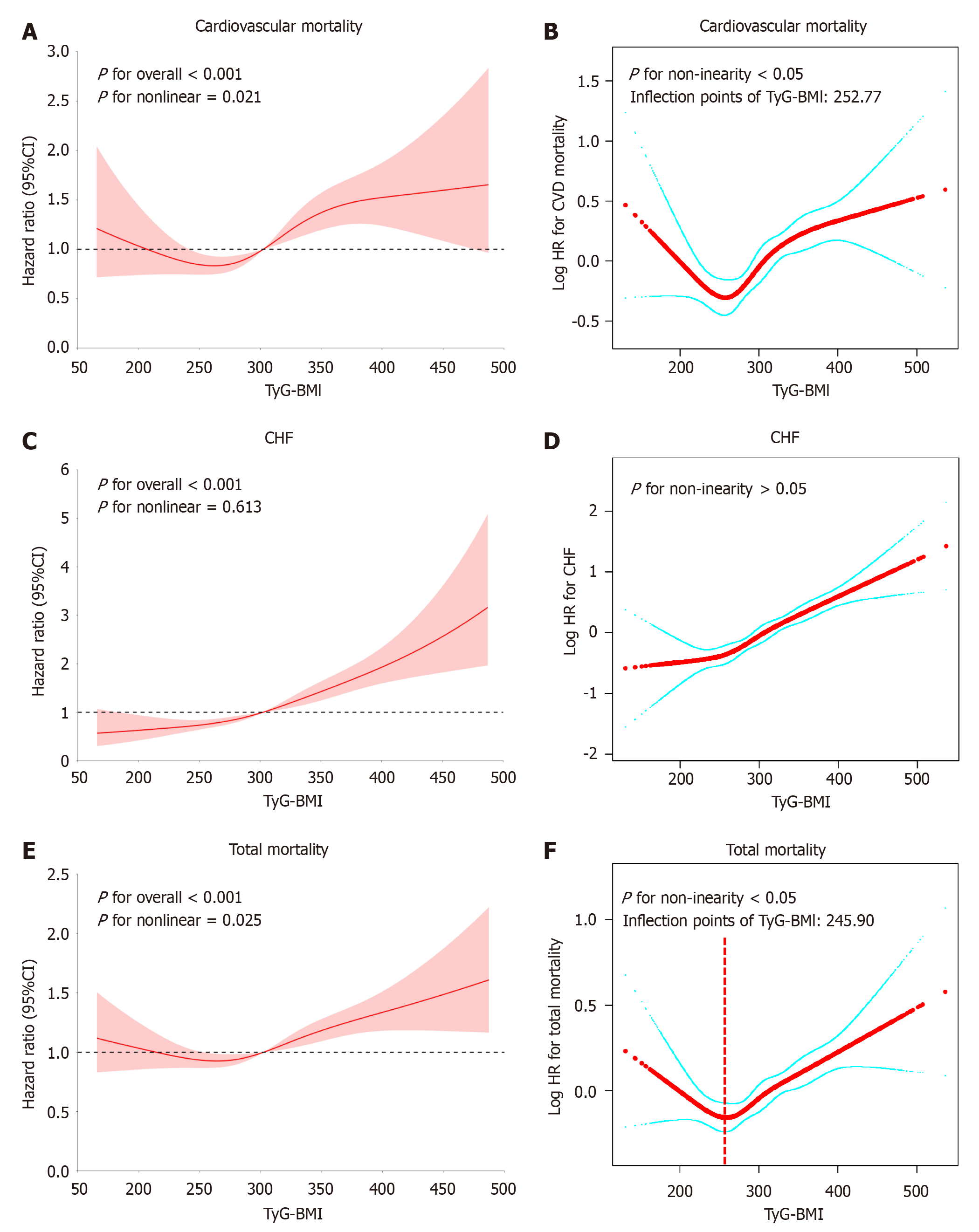
The study then evaluated a possible nonlinear association of TyG-BMI with cardiovascular and overall mortality risks among T2DM patients by the RCS analysis (cardiovascular mortality: P-overall < 0.001, P-nonlinear = 0.021; CHF: P-overall < 0.001, P-nonlinear > 0.050; overall mortality: P-overall < 0.001, P-nonlinear = 0.025; Figure 3). By fitting a smooth with a Cox’s model, a non-linear correlation of TyG-BMI with both mortality outcomes was confirmed. Log-likelihood test findings further supported this observation (P < 0.05). Additionally, a two-stage Cox’s model was utilized for evaluating the effects on either side of the inflection point along with their confidence intervals. Results suggested that beyond the inflection point, every 10-unit reduction in TyG-BMI corresponded to a 5% lowered cardiovascular mortality risk (HR: 1.05, 95%CI: 1.03-1.06). As for overall mortality, the HR values were 0.96 (95%CI: 0.92-1.00) and 1.03 (95%CI: 1.02-1.04) before and after the inflection point, respectively (Figure 3, Table 3).
| Outcomes | One linear-regression model | Inflection point (K) | < K, effect 1 | > K, effect 2 | P value for LRT |
| Cardiovascular mortality | 1.03 (1.02, 1.05), P < 0.0001 | 252.77 | 0.95 (0.89, 1.01), P = 0.1161 | 1.05 (1.03, 1.06), P < 0.0001 | 0.012 |
| Total mortality | 1.02 (1.01, 1.03), P < 0.0001 | 245.90 | 0.96 (0.92, 1.00), P = 0.0302 | 1.03 (1.02, 1.04), P < 0.0001 | 0.002 |
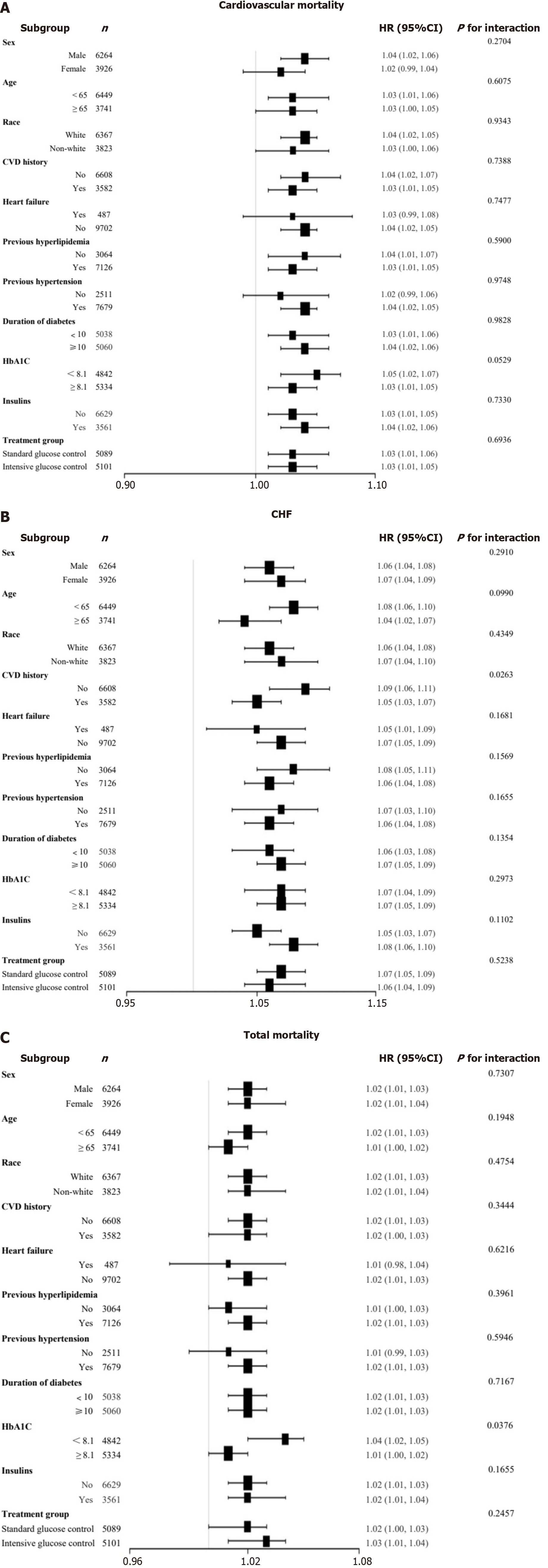
For the robustness examination of our findings and the exploration into association between TyG-BMI and outcome events, interaction and subgroup analyses were undertaken across varying age (< 65 years and ≥ 65 years), sex, race, CVD history, cardiac failure, prior hyperlipidemia, prior hypertension, diabetes duration (< 10 years and ≥ 10 years), and treatment subgroups (intensive vs standard glucose control). Further stratified analyses were also conducted based on insulin usage and HbA1c (< 8.1% vs ≥ 8.1%). Results indicated a significant interaction between TyG-BMI and the cardiovascular and overall mortality risks. Notably, in T2DM patients with HbA1c levels below 8.1%, TyG-BMI demonstrated a heightened predictive power for both mortality types (Figure 4).
For the robustness verification of our results, we conducted a range of sensitivity analyses. Through multiple imputation, missing data were handled to minimize the potential impact of data incompleteness. The results of multivariable Cox regression analysis, conducted following the elimination of subjects with missing values, were consistent with those obtained after multiple imputation (Supplementary Table 3), reinforcing the reliability of our conclusions. Following the elimination of subjects with a baseline CVD history, the final study cohort comprised 6608 individuals. The correlation of TyG-BMI with cardiovascular outcomes and overall mortality remained largely consistent with the primary analysis after adjusting for the entire covariates (Supplementary Table 4). Similarly, when subjects with a baseline cardiac failure history were eliminated, the resulting cohort comprised 9703 individuals, and the above correlation remained comparable to the main findings (Supplementary Table 5). Furthermore, after omitting subjects from the initial 3-year follow-up period, a total of 9496 individuals were included, and the above correlation persisted (Supplementary Table 6). Col
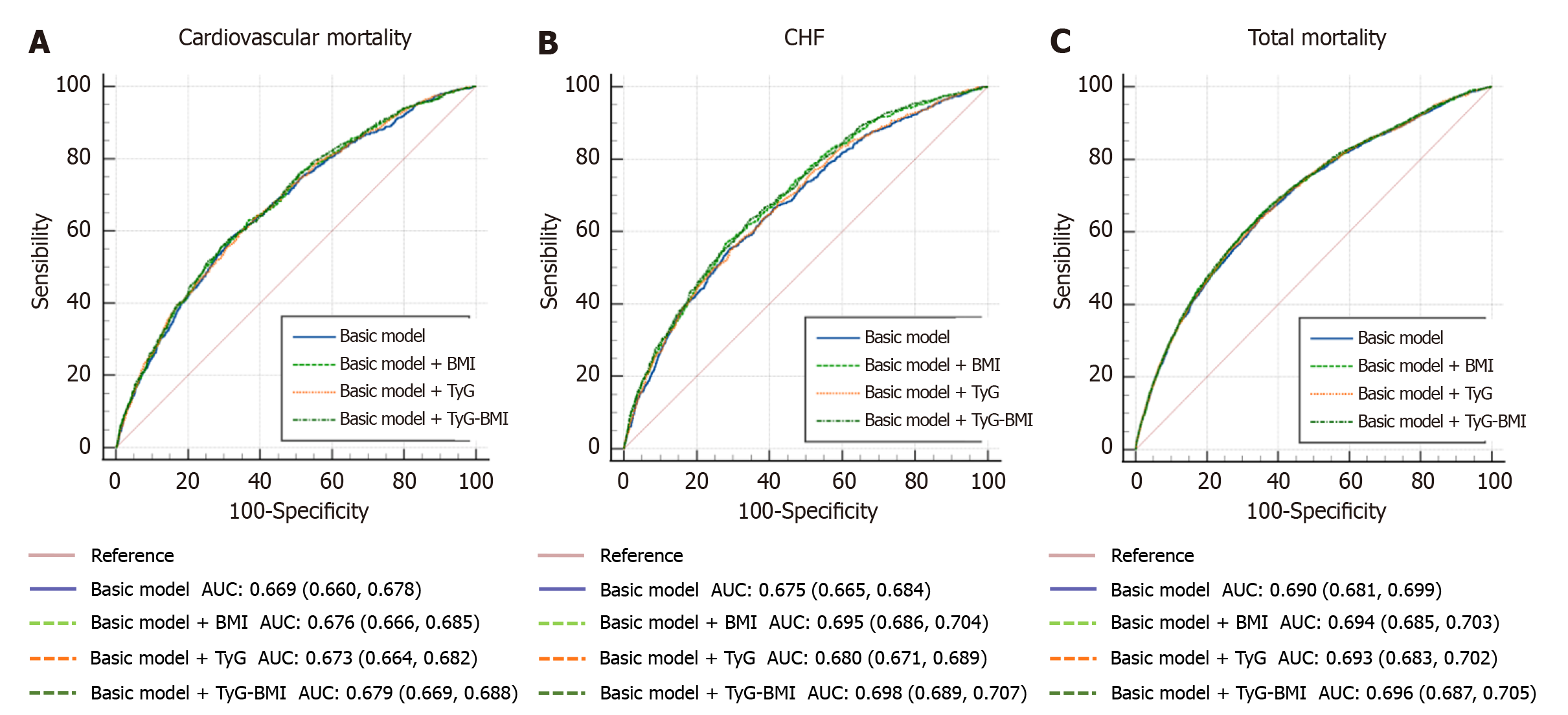
Through the ROC curve approach, we assessed TyG-BMI’s predictive value for future cardiovascular and overall mortality risks. Results showed an increase in the AUC for cardiovascular outcomes and overall mortality after incorporating TyG-BMI levels into conventional models, which accounted for factors like age, sex, educational background, diabetes duration, smoking status, history of hypertension/hyperlipidemia, SBP, DBP, proteinuria, depression, FPG, HbA1c, and LDL-C. Suggestively, the introduction of TyG-BMI into the classic models substantially enhances predictive precision for future cardiovascular outcomes and overall mortality among T2DM individuals. The base model incorporating TyG-BMI exhibited superior predictive power to those considering TyG or BMI alone (Figure 5).
This study conducted an initial investigation into the link of TyG-BMI to both cardiovascular outcomes and overall mortality among the T2DM population. The ACCORD/ACCORDION trial is a broad, population-based cohort study. As far as we are aware, this is the first research within this cohort to assess the correlations of TyG-BMI with cardiovascular outcomes and overall mortality. Our results indicate that TyG-BMI serves as a valid predictor for both overall and cardiovascular mortality risks in T2DM individuals. After comprehensive adjustment for confounding factors, we found that TyG-BMI elevations were independently linked to heightened risks of cardiovascular outcomes and overall mortality for this population. These findings may contribute to novel understandings about the prevention and management of cardiovascular and overall mortality in T2DM patients, serving as a reference for developing targeted strategies.
CVD continues to pose a major global health challenge, placing substantial strain on healthcare systems and families worldwide[28,29]. Diabetes has been acknowledged as a salient risk factor for CVD, with its poor outcomes being strongly linked to cardiovascular complications[30,31]. The median follow-up in our study spanned 8.82 years, with 6.50% of T2DM patients dying from cardiovascular causes, 6.77% dying from CHF, and 19.06% from any cause. The elevated incidence rates observed, relative to prior studies, may stem from the fact that the majority of ACCORD and ACCORDION participants were older, at a high cardiovascular risk, and exhibited poor glycemic control.
Prior research has shown that IR is pivotal in the development of atherosclerosis and recognized it as a major risk factor for CVD. Numerous investigations have emphasized the link between IR and CVD progression, underscoring the predictive value of IR for cardiovascular outcomes[10,11,32,33]. While the HEC is generally considered the gold standard for evaluating IR, its complexity and cost hinder its broader clinical application. Hence, it is crucial to identify a dependable and simpler surrogate indicator for the IR assessment.
Recently, TyG-BMI, a new and practical indicator combining TG, FBG, and BMI, has been recognized for its strong diagnostic value in detecting IR[22-25]. Its predictive power for clinical outcomes has been substantiated. For example, evidence suggests that TyG-BMI, either independently or in combination with sedentary behavior and physical activity, is associated with the risks of all-cause mortality and myocardial infarction[34]. In heart failure patients, a decreased TyG-BMI strongly correlated with an elevated 1-year mortality risk[35]. Among patients sustaining severe acute myocardial infarction, TyG-BMI was significantly correlated with overall mortality[36]. Among hypertensive patients, the incorporation of TyG-BMI into the basic predictive model significantly improved the cardiovascular mortality predictability[37]. As suggested by these results, TyG-BMI serves as a useful prognostic biomarker.
In our current study, the TyG-BMI index demonstrated a similar predictability for both cardiovascular and overall mortality among diabetic subjects, establishing a connection between them among T2DM individuals within the ACCORD trial cohort for the first time. In a cohort comprising 10090 patients with T2DM, our findings indicated that a higher baseline TyG-BMI correlated with greater future risks of both cardiovascular and overall mortality. Such an association remained statistically noteworthy even following the key confounder adjustment. Patients whose baseline TyG-BMI fell within the highest quartile exhibited a 1.74-fold higher cardiovascular mortality risk, a 2.65-fold elevated CHF risk, and a 1.42-fold higher overall mortality risk, respectively, in contrast to those within the lowest quartile. The RCS analysis, along with smooth-curve fitting, unveiled a nonlinear correlation of baseline TyG-BMI with future risks of cardiovascular and overall mortality in T2DM patients, whereas a positive linear association of TyG-BMI with CHF risk. The identified inflection points for cardiovascular and overall mortality were 252.77 and 245.90, respectively. These results are consistent with prior research. For instance, Xiao et al[38], drawing on the National Health and Nutrition Examination Survey data, also reported a U-shaped correlation of TyG-BMI with both mortality types among diabetic individuals. They identified the inflection points at 279.67 for overall mortality, whereas at 270.19 for cardiovascular mortality, both significantly exceeding the corresponding thresholds identified in our analysis (245.90 and 252.77, respectively). This discrepancy may reflect the unique characteristics of the ACCORD cohort, which consisted of older adults with poor glycemic control and high cardiovascular risk. Our findings suggest that in high-risk, late-stage T2DM sufferers, more stringent control of TyG-BMI may be necessary to reduce the long-term mortality risk. This further highlights the clinical importance of TyG-BMI as a tailored risk stratification tool for complex diabetic cohorts. The implications of this result are significant in clinical practice, offering a reference point for the cardiovascular mortality prevention among T2DM patients. Decreasing BMI, TG, and FPG through dietary and lifestyle modifications may contribute to lowered cardiovascular and overall mortality risks, particularly when TyG-BMI levels are maintained below 245.90. Additional stratified and interaction analyses provided results that largely corroborated the initial findings. These analyses indicated that TyG-BMI showed stronger predictive power for both cardiovascular and overall mortality in T2DM individuals with HbA1c levels below 8.1%. This finding may reflect distinct pathophysiological mechanisms operating across different glycemic control statuses. In individuals with poorly managed diabetes (HbA1c ≥ 8.1%), the direct vascular toxicity from sustained hyperglycemia—manifesting as endothelial dysfunction, oxidative stress, and pro-inflammatory states—may act as a more dominant driver of adverse cardiovascular outcomes, potentially attenuating the independent contribution of TyG-BMI. Conversely, in patients with relatively better glycemic control (HbA1c < 8.1%), where overt glucose toxicity is less pronounced, IR may become a more central driver of cardiovascular risk. In this scenario, TyG-BMI potentially serves as a more sensitive and integrative biomarker of metabolic disturbance, reflecting both dyslipidemia and IR burden. The above findings lend support to the assumption that TyG-BMI is particularly useful in identifying high-risk individuals with subclinical metabolic dysfunction, even in the absence of severe hyperglycemia, highlighting its potential clinical utility in early risk stratification. We also conducted a range of sensitivity analyses, yielding results that agreed with the main findings, thus confirming the robustness of our results.
As implied by the results of this study, TyG-BMI is a vital predictor for both cardiovascular outcomes and overall mortality among T2DM patients. Reducing TyG-BMI levels may substantially lower the risks associated with these outcomes and mortality for this population. Regular TyG-BMI assessment during the initial stages of diabetes diagnosis and treatment, coupled with effective weight management, is vital for minimizing cardiovascular comorbidities and mortality among patients with T2DM. Moreover, incorporating TyG-BMI into the conventional risk models greatly improves their prediction accuracy for cardiovascular and overall mortality risks.
The primary strength of this research lies in its pioneering evaluation within the ACCORD cohort of TyG-BMI’s predictive value for future cardiovascular outcomes and overall mortality among T2DM patients. Following adjustments of cardiovascular risk factors, an extensive, long-term follow-up was conducted on a substantial cohort of T2DM patients for cardiovascular and an all-cause mortality evaluation, aiming to objectively assess TyG-BMI’s potential as a biomarker for mortality risk prediction. This study introduces several important innovations. To our best knowledge, we are the first to reveal a significant positive association of TyG-BMI with the CHF risk among T2DM individuals, which broadens the predictive utility of this index beyond atherosclerotic cardiovascular outcomes to encompass heart failure-related endpoints. Furthermore, utilizing data from the ACCORD trial (a cohort characterized by older age, poor glycemic control, and high baseline cardiovascular risk), we are the first to uncover a U-shaped correlation of TyG-BMI with both cardiovascular and overall mortality, alongside a clinically relevant inflection point. This non-linear pattern may assist in refining risk stratification within the advanced-stage T2DM patients. In addition, our analyses validated that TyG-BMI has superior predictive power for adverse outcomes compared to either TyG or BMI alone, underscoring the importance of combining metabolic and anthropometric markers. These results provide a fresh perspective on the prognostic relevance of TyG-BMI, highlighting its potential clinical utility in high-risk diabetic cohorts with complex comorbidities.
Nevertheless, our study does presents certain limitations. Firstly, it is a secondary analysis of the data from the ACCORD and ACCORDION trials. Despite the multivariate Cox adjustment of potential confounders, some residual or overlooked confounders, such as lifelong dietary habits and lifestyle changes, might still persist. Secondly, being observational in nature, this study cannot confirm a causal link between baseline TyG-BMI and cardiovascular outcomes or overall mortality risks in T2DM patients. Consequently, future studies should include carefully designed, long-term prospective interventions with propensity score matching to further confirm our results.
Thirdly, the ACCORD and ACCORDION trial cohorts comprised T2DM patients at elevated cardiovascular risk, all of whom were exclusively from the United States and Canada. Variations in metabolic profiles, lipid levels, and cardiovascular risks exist across ethnicities, and healthcare systems in the United States and Canada differ from those in other regions regarding medical access and CVD management practices. Moreover, most participants in this study were older, had poorly controlled blood glucose, and were at a high risk for CVD, which may restrict the applicability of our findings to younger or better-controlled patients. Therefore, caution is warranted when generalizing these conclusions to other groups, and further research in more diverse cohorts is needed to confirm these findings. Lastly, relying solely on data mining to form conclusions could impact the generalizability. To make our results more generalizable and robust, we intend to collect cohort data from diverse subgroups and settings for further analyses. This strategy aims to reduce data bias and improve the credibility of our findings. Finally, the unavailability of fasting insulin data in the ACCORD database prevented us from comparing TyG-BMI with other established IR indices, such as HOMA-IR. This limitation hinders our ability to fully position TyG-BMI among conventional IR markers. To comprehensively evaluate the relative predictive value and clinical utility of TyG-BMI, future studies should incorporate direct measures of IR and compare them with this composite index.
Conclusively, this study has revealed that variations in TyG-BMI are independently linked to both cardiovascular outcomes and overall mortality among T2DM individuals. TyG-BMI exhibits U-shaped correlations with both car
The authors express their sincere gratitude to the ACCORD/ACCORDION study group and NHLBI BioLINCC. The views presented in this article are solely those of the authors and do not necessarily represent the perspectives of the ACCORD/ACCORDION study authors or NHLBI BioLINCC.
| 1. | Wong ND, Sattar N. Cardiovascular risk in diabetes mellitus: epidemiology, assessment and prevention. Nat Rev Cardiol. 2023;20:685-695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 380] [Cited by in RCA: 298] [Article Influence: 99.3] [Reference Citation Analysis (6)] |
| 2. | Khan MAB, Hashim MJ, King JK, Govender RD, Mustafa H, Al Kaabi J. Epidemiology of Type 2 Diabetes - Global Burden of Disease and Forecasted Trends. J Epidemiol Glob Health. 2020;10:107-111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2498] [Cited by in RCA: 1918] [Article Influence: 319.7] [Reference Citation Analysis (7)] |
| 3. | Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, Stein C, Basit A, Chan JCN, Claude Mbanya J, Pavkov ME, Ramachandaran A, Wild SH, James S, Herman WH, Zhang P, Bommer C, Kuo S, Boyko EJ, Magliano DJ. Erratum to "IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045" [Diabetes Res. Clin. Pract. 183 (2022) 109119]. Diabetes Res Clin Pract. 2023;204:110945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 104] [Article Influence: 34.7] [Reference Citation Analysis (1)] |
| 4. | American Diabetes Association. Economic Costs of Diabetes in the U.S. in 2017. Diabetes Care. 2018;41:917-928. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2135] [Cited by in RCA: 1786] [Article Influence: 223.3] [Reference Citation Analysis (1)] |
| 5. | Yun JS, Ko SH. Current trends in epidemiology of cardiovascular disease and cardiovascular risk management in type 2 diabetes. Metabolism. 2021;123:154838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 243] [Cited by in RCA: 180] [Article Influence: 36.0] [Reference Citation Analysis (0)] |
| 6. | Raghavan S, Vassy JL, Ho YL, Song RJ, Gagnon DR, Cho K, Wilson PWF, Phillips LS. Diabetes Mellitus-Related All-Cause and Cardiovascular Mortality in a National Cohort of Adults. J Am Heart Assoc. 2019;8:e011295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 461] [Cited by in RCA: 401] [Article Influence: 57.3] [Reference Citation Analysis (0)] |
| 7. | Rosengren A, Dikaiou P. Cardiovascular outcomes in type 1 and type 2 diabetes. Diabetologia. 2023;66:425-437. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 88] [Article Influence: 29.3] [Reference Citation Analysis (0)] |
| 8. | Aedh AI, Alshahrani MS, Huneif MA, Pryme IF, Oruch R. A Glimpse into Milestones of Insulin Resistance and an Updated Review of Its Management. Nutrients. 2023;15:921. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 9. | Kosmas CE, Bousvarou MD, Kostara CE, Papakonstantinou EJ, Salamou E, Guzman E. Insulin resistance and cardiovascular disease. J Int Med Res. 2023;51:3000605231164548. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 292] [Cited by in RCA: 220] [Article Influence: 73.3] [Reference Citation Analysis (0)] |
| 10. | Abdul-Ghani M, Maffei P, DeFronzo RA. Managing insulin resistance: the forgotten pathophysiological component of type 2 diabetes. Lancet Diabetes Endocrinol. 2024;12:674-680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 41] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 11. | Hill MA, Yang Y, Zhang L, Sun Z, Jia G, Parrish AR, Sowers JR. Insulin resistance, cardiovascular stiffening and cardiovascular disease. Metabolism. 2021;119:154766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 817] [Cited by in RCA: 696] [Article Influence: 139.2] [Reference Citation Analysis (1)] |
| 12. | Ormazabal V, Nair S, Elfeky O, Aguayo C, Salomon C, Zuñiga FA. Association between insulin resistance and the development of cardiovascular disease. Cardiovasc Diabetol. 2018;17:122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1770] [Cited by in RCA: 1478] [Article Influence: 184.8] [Reference Citation Analysis (0)] |
| 13. | Fahed G, Aoun L, Bou Zerdan M, Allam S, Bou Zerdan M, Bouferraa Y, Assi HI. Metabolic Syndrome: Updates on Pathophysiology and Management in 2021. Int J Mol Sci. 2022;23:786. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1163] [Cited by in RCA: 884] [Article Influence: 221.0] [Reference Citation Analysis (0)] |
| 14. | Sergi D, Spaggiari R, Dalla Nora E, Angelini S, Castaldo F, Omenetto A, Stifani G, Sanz JM, Passaro A; PANGEA study group. HOMA-IR and TyG index differ for their relationship with dietary, anthropometric, inflammatory factors and capacity to predict cardiovascular risk. Diabetes Res Clin Pract. 2025;222:112103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 15. | Simental-Mendía LE, Rodríguez-Morán M, Guerrero-Romero F. The product of fasting glucose and triglycerides as surrogate for identifying insulin resistance in apparently healthy subjects. Metab Syndr Relat Disord. 2008;6:299-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1837] [Cited by in RCA: 1559] [Article Influence: 86.6] [Reference Citation Analysis (0)] |
| 16. | Guerrero-Romero F, Simental-Mendía LE, González-Ortiz M, Martínez-Abundis E, Ramos-Zavala MG, Hernández-González SO, Jacques-Camarena O, Rodríguez-Morán M. The product of triglycerides and glucose, a simple measure of insulin sensitivity. Comparison with the euglycemic-hyperinsulinemic clamp. J Clin Endocrinol Metab. 2010;95:3347-3351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1667] [Cited by in RCA: 1465] [Article Influence: 91.6] [Reference Citation Analysis (0)] |
| 17. | He HM, Xie YY, Chen Q, Li YK, Li XX, Fu SJ, Li N, Han YR, Gao YX, Zheng JG. The synergistic effect of the triglyceride-glucose index and a body shape index on cardiovascular mortality: the construction of a novel cardiovascular risk marker. Cardiovasc Diabetol. 2025;24:69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 43] [Article Influence: 43.0] [Reference Citation Analysis (0)] |
| 18. | Lopez-Jaramillo P, Gomez-Arbelaez D, Martinez-Bello D, Abat MEM, Alhabib KF, Avezum Á, Barbarash O, Chifamba J, Diaz ML, Gulec S, Ismail N, Iqbal R, Kelishadi R, Khatib R, Lanas F, Levitt NS, Li Y, Mohan V, Mony PK, Poirier P, Rosengren A, Soman B, Wang C, Wang Y, Yeates K, Yusuf R, Yusufali A, Zatonska K, Rangarajan S, Yusuf S. Association of the triglyceride glucose index as a measure of insulin resistance with mortality and cardiovascular disease in populations from five continents (PURE study): a prospective cohort study. Lancet Healthy Longev. 2023;4:e23-e33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 291] [Cited by in RCA: 246] [Article Influence: 82.0] [Reference Citation Analysis (0)] |
| 19. | Yao Y, Wang B, Geng T, Chen J, Chen W, Li L. The association between TyG and all-cause/non-cardiovascular mortality in general patients with type 2 diabetes mellitus is modified by age: results from the cohort study of NHANES 1999-2018. Cardiovasc Diabetol. 2024;23:43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 75] [Article Influence: 37.5] [Reference Citation Analysis (0)] |
| 20. | Liu X, Tan Z, Huang Y, Zhao H, Liu M, Yu P, Ma J, Zhao Y, Zhu W, Wang J. Relationship between the triglyceride-glucose index and risk of cardiovascular diseases and mortality in the general population: a systematic review and meta-analysis. Cardiovasc Diabetol. 2022;21:124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 297] [Cited by in RCA: 254] [Article Influence: 63.5] [Reference Citation Analysis (0)] |
| 21. | Liu C, Liang D. The association between the triglyceride-glucose index and the risk of cardiovascular disease in US population aged ≤ 65 years with prediabetes or diabetes: a population-based study. Cardiovasc Diabetol. 2024;23:168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 49] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 22. | Bala C, Gheorghe-Fronea O, Pop D, Pop C, Caloian B, Comsa H, Bozan C, Matei C, Dorobantu M. The Association Between Six Surrogate Insulin Resistance Indexes and Hypertension: A Population-Based Study. Metab Syndr Relat Disord. 2019;17:328-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 92] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 23. | Ramírez-Vélez R, Pérez-Sousa MÁ, González-Ruíz K, Cano-Gutierrez CA, Schmidt-RioValle J, Correa-Rodríguez M, Izquierdo M, Romero-García JA, Campos-Rodríguez AY, Triana-Reina HR, González-Jiménez E. Obesity- and Lipid-Related Parameters in the Identification of Older Adults with a High Risk of Prediabetes According to the American Diabetes Association: An Analysis of the 2015 Health, Well-Being, and Aging Study. Nutrients. 2019;11:2654. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 88] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 24. | Er LK, Wu S, Chou HH, Hsu LA, Teng MS, Sun YC, Ko YL. Triglyceride Glucose-Body Mass Index Is a Simple and Clinically Useful Surrogate Marker for Insulin Resistance in Nondiabetic Individuals. PLoS One. 2016;11:e0149731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 499] [Cited by in RCA: 439] [Article Influence: 43.9] [Reference Citation Analysis (0)] |
| 25. | Park SY, Gautier JF, Chon S. Assessment of Insulin Secretion and Insulin Resistance in Human. Diabetes Metab J. 2021;45:641-654. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 214] [Cited by in RCA: 171] [Article Influence: 34.2] [Reference Citation Analysis (2)] |
| 26. | ACCORD Study Group; Gerstein HC, Miller ME, Genuth S, Ismail-Beigi F, Buse JB, Goff DC Jr, Probstfield JL, Cushman WC, Ginsberg HN, Bigger JT, Grimm RH Jr, Byington RP, Rosenberg YD, Friedewald WT. Long-term effects of intensive glucose lowering on cardiovascular outcomes. N Engl J Med. 2011;364:818-828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 817] [Cited by in RCA: 709] [Article Influence: 47.3] [Reference Citation Analysis (4)] |
| 27. | ACCORD Study Group; Buse JB, Bigger JT, Byington RP, Cooper LS, Cushman WC, Friedewald WT, Genuth S, Gerstein HC, Ginsberg HN, Goff DC Jr, Grimm RH Jr, Margolis KL, Probstfield JL, Simons-Morton DG, Sullivan MD. Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial: design and methods. Am J Cardiol. 2007;99:21i-33i. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 477] [Cited by in RCA: 444] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 28. | Raleigh V, Colombo F. Cardiovascular disease should be a priority for health systems globally. BMJ. 2023;382:e076576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 29. | Abushanab D, Marquina C, Morton JI, Al-Badriyeh D, Lloyd M, Magliano DJ, Liew D, Ademi Z. Projecting the Health and Economic Burden of Cardiovascular Disease Among People with Type 2 Diabetes, 2022-2031. Pharmacoeconomics. 2023;41:719-732. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 30. | Marx N, Federici M, Schütt K, Müller-Wieland D, Ajjan RA, Antunes MJ, Christodorescu RM, Crawford C, Di Angelantonio E, Eliasson B, Espinola-Klein C, Fauchier L, Halle M, Herrington WG, Kautzky-Willer A, Lambrinou E, Lesiak M, Lettino M, McGuire DK, Mullens W, Rocca B, Sattar N; ESC Scientific Document Group. 2023 ESC Guidelines for the management of cardiovascular disease in patients with diabetes. Eur Heart J. 2023;44:4043-4140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1212] [Cited by in RCA: 977] [Article Influence: 325.7] [Reference Citation Analysis (8)] |
| 31. | Ahmad A, Lim LL, Morieri ML, Tam CH, Cheng F, Chikowore T, Dudenhöffer-Pfeifer M, Fitipaldi H, Huang C, Kanbour S, Sarkar S, Koivula RW, Motala AA, Tye SC, Yu G, Zhang Y, Provenzano M, Sherifali D, de Souza RJ, Tobias DK; ADA/EASD PMDI, Gomez MF, Ma RCW, Mathioudakis N. Precision prognostics for cardiovascular disease in Type 2 diabetes: a systematic review and meta-analysis. Commun Med (Lond). 2024;4:11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 36] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 32. | Di Pino A, DeFronzo RA. Insulin Resistance and Atherosclerosis: Implications for Insulin-Sensitizing Agents. Endocr Rev. 2019;40:1447-1467. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 423] [Cited by in RCA: 367] [Article Influence: 52.4] [Reference Citation Analysis (0)] |
| 33. | Louie JZ, Shiffman D, McPhaul MJ, Melander O. Insulin resistance probability score and incident cardiovascular disease. J Intern Med. 2023;294:531-535. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 34. | Zhu Y, Yao T, Tian L, Zhang Y, Ke Q. Associations of triglyceride glucose-body mass index and the combination of sedentary behavior and physical activity with risks of all-cause mortality and myocardial infarction: a cohort study from the UK biobank. Cardiovasc Diabetol. 2025;24:102. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 35. | Dou J, Guo C, Wang Y, Peng Z, Wu R, Li Q, Zhao H, Song S, Sun X, Wei J. Association between triglyceride glucose-body mass and one-year all-cause mortality of patients with heart failure: a retrospective study utilizing the MIMIC-IV database. Cardiovasc Diabetol. 2023;22:309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 66] [Reference Citation Analysis (0)] |
| 36. | Luo C, Li Q, Wang Z, Duan S, Ma Q. Association between triglyceride glucose-body mass index and all-cause mortality in critically ill patients with acute myocardial infarction: retrospective analysis of the MIMIC-IV database. Front Nutr. 2024;11:1399969. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 37. | Li C, Zhang Z, Luo X, Xiao Y, Tu T, Liu C, Liu Q, Wang C, Dai Y, Zhang Z, Zheng C, Lin J. The triglyceride-glucose index and its obesity-related derivatives as predictors of all-cause and cardiovascular mortality in hypertensive patients: insights from NHANES data with machine learning analysis. Cardiovasc Diabetol. 2025;24:47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 58] [Article Influence: 58.0] [Reference Citation Analysis (0)] |
| 38. | Xiao S, Zhang Q, Yang HY, Tong JY, Yang RQ. The association between triglyceride glucose-body mass index and all-cause and cardiovascular mortality in diabetes patients: a retrospective study from NHANES database. Sci Rep. 2024;14:13884. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 29] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/