Copyright: ©Author(s) 2026.
World J Diabetes. May 15, 2026; 17(5): 119756
Published online May 15, 2026. doi: 10.4239/wjd.v17.i5.119756
Published online May 15, 2026. doi: 10.4239/wjd.v17.i5.119756
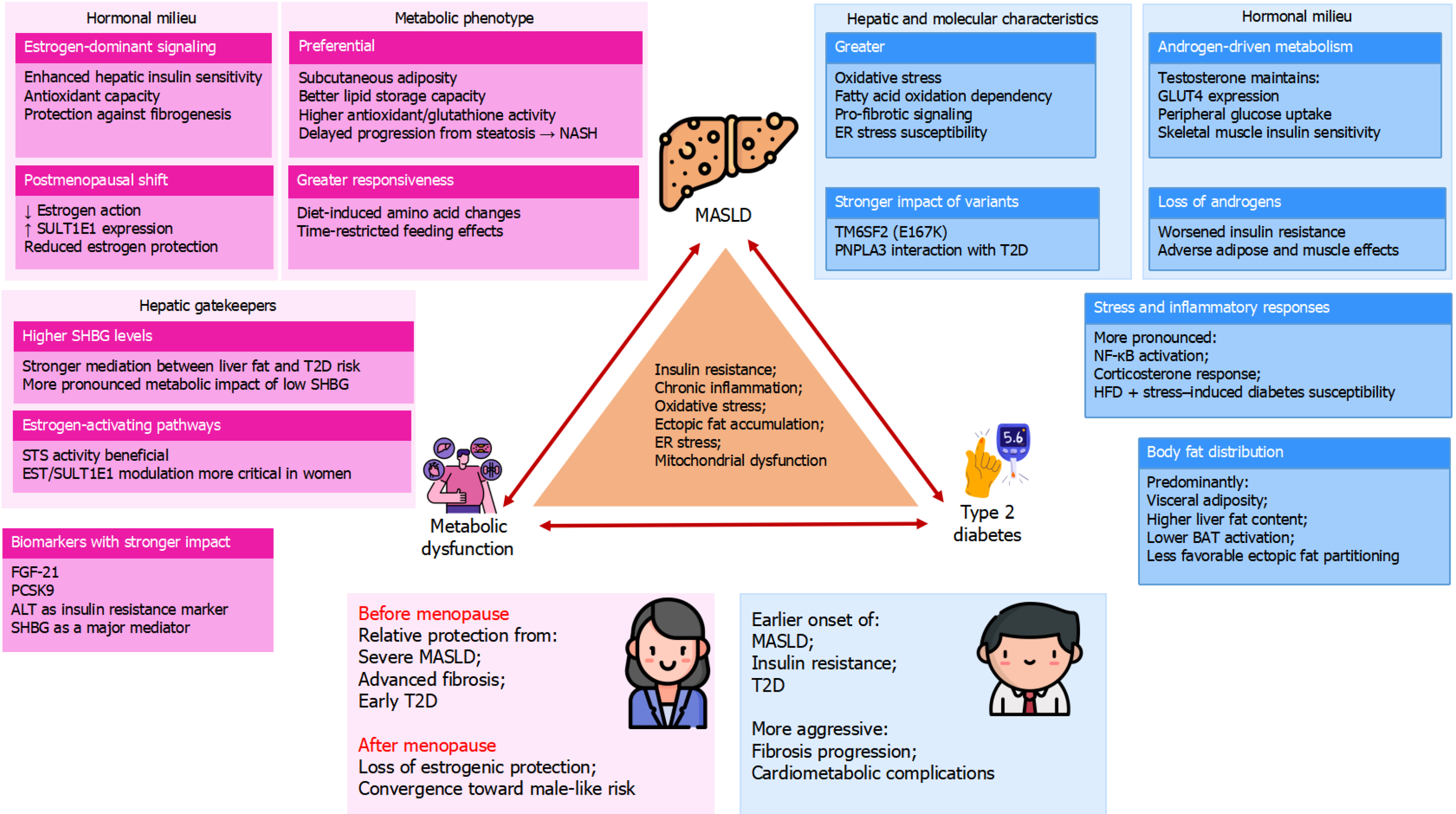
Figure 1 Integrative model summarizing sex-specific biological mechanisms linking metabolic dysfunction-associated steatotic liver disease and type 2 diabetes.
The central triangle depicts the bidirectional relationships among hepatic steatosis, systemic metabolic dysfunction, and diabetes. Surrounding domains illustrate key modifiers including sex steroids, sex hormone-binding globulin (SHBG), hepatic lipid handling, genetic variants, adipose tissue distribution, and inflammatory and endoplasmic reticulum stress pathways that operate differently in females and males. Pathways predominantly relevant to women are represented in pink, whereas pathways predominantly relevant to men are represented in blue. Premenopausal women are relatively protected through estrogen signaling, higher SHBG, more favorable fat distribution, and enhanced antioxidant capacity. In contrast, men exhibit greater visceral adiposity, increased oxidative stress, and stronger diabetogenic genetic susceptibility. These advantages progressively erode after menopause, leading to convergence of metabolic and hepatic risk between the sexes. NASH: Nonalcoholic steatohepatitis; SHBG: Sex hormone-binding globulin; STS: Steroid sulfatase; MASLD: Metabolic dysfunction-associated steatotic liver disease; T2D: Type 2 diabetes; FGF-21: Fibroblast growth factor 21; PCSK9: Proprotein convertase subtilisin/kexin type 9; ALT: Alanine aminotransferase; ER: Endoplasmic reticulum; BAT: Brown adipose tissue; GLUT4: Glucose transporter type 4; HFD: High-fat diet; NF-κB: Nuclear factor kappa-B.
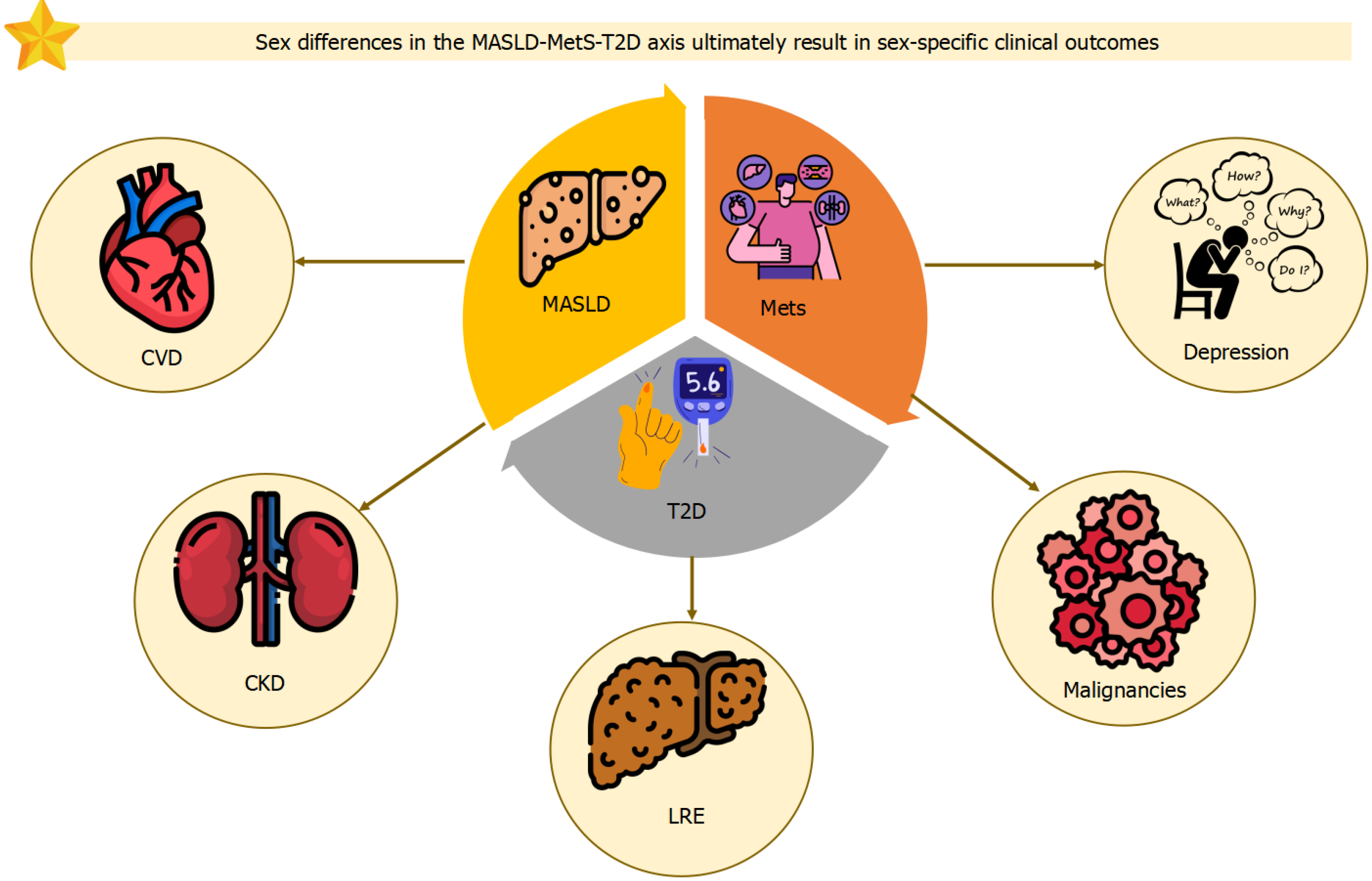
Figure 2 Conceptual model illustrating how sex differences in the metabolic dysfunction-associated steatotic liver disease-metabolic syndrome-type 2 diabetes axis translate into sex-specific clinical outcomes.
This figure shows that sex-specific relationships among metabolic dysfunction-associated steatotic liver disease (MASLD), metabolic syndrome (MetS), and type 2 diabetes (T2D) ultimately lead to sex-specific clinical consequences, including cardiovascular disease, chronic kidney disease, liver-related events, malignancies, and depression. The figure highlights that sex is a key determinant of how the MASLD-MetS-T2D continuum manifests in real-world clinical outcomes. MASLD: Metabolic dysfunction-associated steatotic liver disease; T2D: Type 2 diabetes; MetS: Metabolic syndrome; CVD: Cardiovascular disease; CKD: Chronic kidney disease; LRE: Liver-related events.
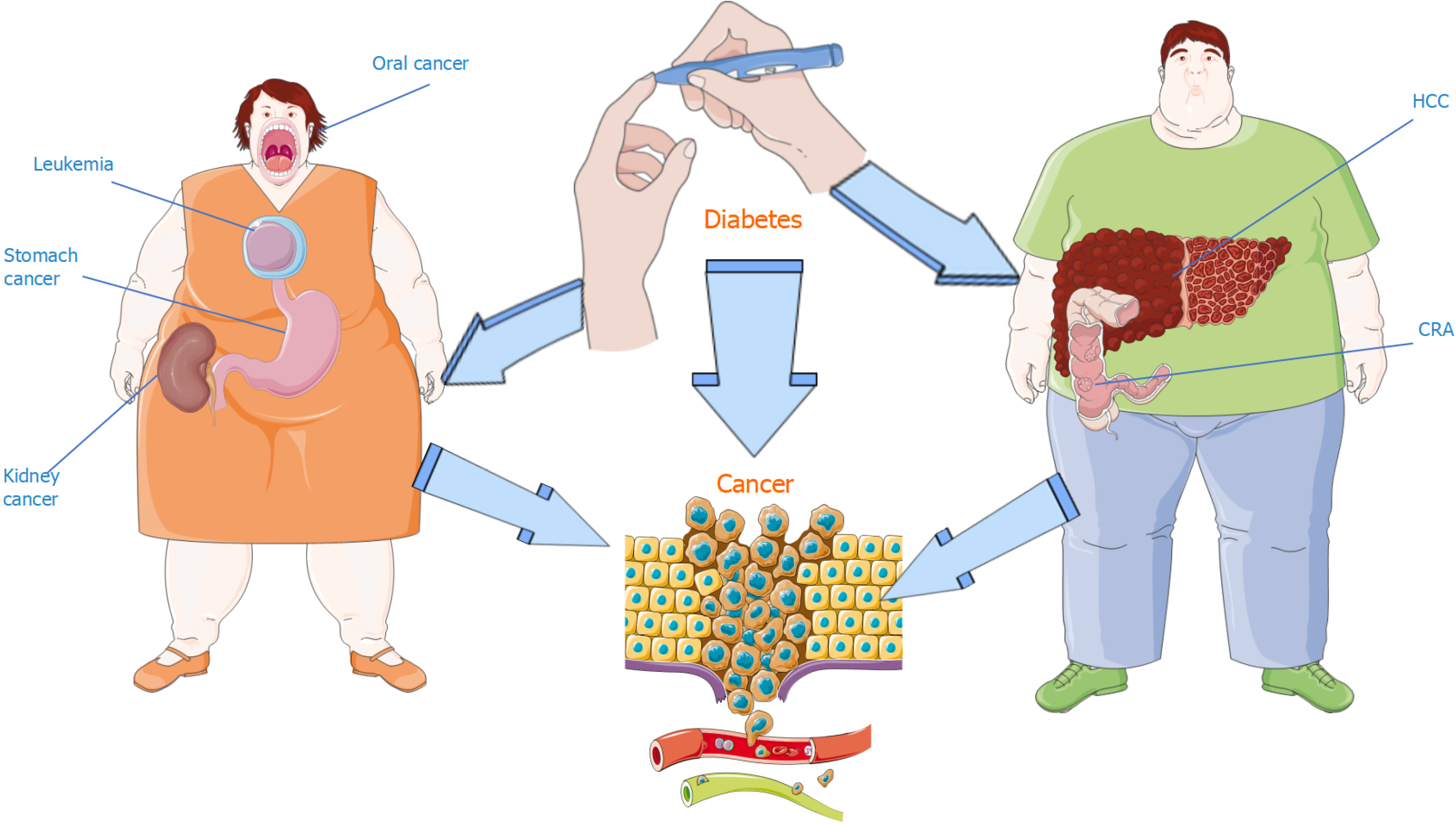
Figure 3 Sex-specific risks of cancer and pre-cancerous conditions among individuals with diabetes.
Women with diabetes, when compared to men with diabetes, show a 6% increased risk of overall cancer and significantly higher odds for oral, stomach, kidney cancers and leukemia. Diabetes increases the risk of hepatocellular carcinoma (HCC) by 80%, regardless of confounding factors, with men having a higher HCC risk than women. Additionally, both metabolic dysfunction-associated steatotic liver disease and male sex are associated with colorectal adenoma. HCC: Hepatocellular carcinoma; CRA: Colorectal adenoma.
- Citation: Lonardo A, Jamalinia M, Weiskirchen R. Type 2 diabetes, sex and metabolic dysfunction-associated steatotic liver disease. World J Diabetes 2026; 17(5): 119756
- URL: https://www.wjgnet.com/1948-9358/full/v17/i5/119756.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i5.119756