INTRODUCTION
Colorectal cancer (CRC) is one of the most prevalent malignancies globally. Recent epidemiological studies indicate that its incidence ranks third among men and second among women, and it has become a leading cause of cancer-related deaths worldwide[1-3]. In China, CRC is the second most commonly diagnosed cancer and the fourth leading cause of cancer mortality[4]. Despite advances in targeted therapy, immunotherapy, surgery, chemotherapy, and radiotherapy, the prognosis for patients with advanced CRC remains poor, primarily due to rapid tumor growth and metastasis[5-7]. Therefore, exploring novel molecular targets and regulatory networks involved in CRC growth, invasion, and metastasis is of critical importance.
Epithelial-mesenchymal transition (EMT) is a key mechanism driving tumor cell invasion and migration in various cancers, including CRC[8]. This process is regulated by multiple signaling pathways, such as Wnt/β-catenin, Hedgehog, transforming growth factor-β, and Notch, which activate transcription factors like ZEB1/2 and Snail. Among these, the Wnt/β-catenin pathway plays a pivotal role in controlling EMT[9]. Aberrant Wnt/β-catenin activation significantly impacts tumor initiation, progression, local infiltration, distant metastasis, and even chemoresistance in CRC[10-12]. Targeting key proteins within this pathway represents a promising therapeutic strategy[5].
Icaritin, a prenylated flavonoid extracted from Epimedium, has demonstrated broad anti-cancer activity in numerous studies[6,7,13]. In CRC, icaritin induces cell death via the c-Jun N-terminal kinase (JNK)-dependent mitochondrial permeability transition pore necrosis pathway[14]. Furthermore, icaritin modulates the tumor immune microenvironment by activating innate and adaptive immunity, enhancing T-cell cytotoxicity, downregulating programmed death-ligand 1 expression, and reducing the number of myeloid-derived suppressor cells[15-19]. Approved in China as a first-line treatment for hepatocellular carcinoma, icaritin also inhibits tumor growth in Lewis lung carcinoma mouse models by enhancing CD8+ T-cell infiltration[20]. In CRC, inhibition of autophagy sensitizes cells to icaritin[20]. Icaritin suppresses tumor progression by inhibiting cyclin-dependent kinase 2 and exhibits anti-proliferative effects in liver and CRC models[21]. In colon cancer, it exerts anti-cancer effects by activating reactive oxygen species generation and inhibiting cyclin E, cyclin D1, and Bcl-2 signaling[22].
Icaritin exerts its biological effects by modulating EMT and the Wnt/β-catenin signaling pathway in various disease models. Studies have demonstrated that in models of skin and liver fibrosis, icaritin inhibits EMT through the Notch2/Hes-1 pathway, reduces mesenchymal marker expression, and upregulates epithelial markers to alleviate tissue fibrosis[23]. Additionally, icaritin upregulates epithelial markers in primary hematopoietic stem cells to suppress EMT[24]. Moreover, icaritin exerts anti-fibrotic effects by activating AMPK signaling and inhibiting the Wnt/β-catenin pathway[25]. In cervical cancer cells, icaritin disrupts the TLR4/MyD88/NF-κB and Wnt/β-catenin pathways, thereby suppressing inflammation, proliferation, migration, and invasion, and promoting apoptosis[26]. Although these studies indicate that icaritin can regulate EMT and Wnt/β-catenin signaling, its specific effects and underlying mechanisms in CRC remain unclear. Therefore, this study aims to investigate the effect of icaritin on CRC metastasis and its regulation of the EMT process and Wnt/β-catenin signaling pathway both in vitro and in vivo.
MATERIALS AND METHODS
Drugs and antibodies
Icaritin was obtained from Beijing Shennuoji Pharmaceutical Technology Co. Ltd. (Beijing, China) and was prepared as a 10 μM solution in dimethyl sulfoxide (DMSO). The Annexin V-FITC Apoptosis Kit was supplied by BestBio (Shanghai, China). Most antibodies, including matrix metalloproteinase 7 (MMP7) (ab181602), cyclin D1 (ab16663), adenomatous polyposis coli (APC) (ab40778), c-MYC (ab32072), Wnt3a (ab219412), N-cadherin (ab76057), MMP9 (ab76003), MMP2 (ab80737), and E-cadherin (ab76055) were obtained from Abcam. The antibodies for Snail transcription factor (3895) and β-catenin (8480) were obtained from Cell Signaling Technology, while the GAPDH antibody (AP0063) came from Bioworld Technology.
The human CRC cell lines HCT116 (CCL-247) and SW620 (CCL-227) were obtained from the American Type Culture Collection and underwent STR profiling for identification and mycoplasma contamination checks. HCT116 cells were grown in McCoy’s 5A medium, whereas SW620 cells were maintained in Dulbecco’s modified Eagle’s medium; both supplemented with 10% fetal bovine serum (Gibco; Thermo Fisher Scientific), 1% penicillin/streptomycin and 1% glutamine (Invitrogen; Thermo Fisher Scientific). The cells were incubated in a humidified environment at 37 °C with 5% CO2 and typically passaged every 2-3 days.
Cell counting kit-8 assay
Digitized microscopic images were used to evaluate cell morphology and viability after incubation with icaritin. HCT116 and SW620 cells were initially seeded in 96-well plates at 5000 cells per well. Following icaritin treatment (10 μM), the cell counting kit-8 (CCK-8) method (SAB, CP002) was applied to assess cell proliferation. The CCK-8 working solution (10 μL per well, Beyotime, Shanghai, China) was combined with serum-free culture medium (90 μL per well) and incubated for 0 hour, 24 hours, and 48 hours. After an additional hour at 37 °C, optical density was measured using a microplate reader (Bio-Rad) at 450 nm. Each experiment was conducted in triplicate over three separate trials. A dose-response curve was generated to determine the IC50 value, indicating the concentration of icaritin that resulted in 50% cell growth inhibition.
Flow cytometry analysis
Apoptosis was evaluated using Annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) double staining. HCT116 and SW620 cells were seeded and allowed to grow for 24 hours until reaching the logarithmic growth phase. The cells were collected by centrifugation and transferred to six-well plates, each containing 200 μL culture medium. They were incubated at 37 °C with 5% CO2 and high humidity for an additional 24 hours. Icaritin was added at 80 μg/mL to HCT116 cells and 100 μg/mL to SW620 cells.
After treatment, the cells were collected and rinsed with cold phosphate-buffered saline (PBS). To detect apoptosis, the cells were resuspended and incubated in the dark at room temperature for 15 minutes with Annexin V-FITC and PI. Apoptosis was analyzed using a BD FACSVerseTM flow cytometer (BD Biosciences, United States) with excitation at 488 nm and emission at 530 nm. The FITC signal detector was used for Annexin V-FITC, and a separate detector appropriate for PI fluorescence was employed.
Transwell cell migration and invasion assays
The invasion and migration of HCT116 and SW620 cells were assessed using a Transwell assay (8-μm pore; Corning Inc.), with Matrigel for the invasion assay. After 24 hours of icaritin treatment, cells were washed three times with PBS, resuspended in 200 μL serum-free medium and placed in the upper chamber. The lower chamber was filled with 500 μL medium containing 20% fetal bovine serum. After 24 hours of incubation, cells remaining on the upper side of the membrane were gently removed. The cells on the Transwell membrane were fixed with 4% paraformaldehyde for 15 minutes and subsequently stained with 0.1% crystal violet for 15 minutes at room temperature. Finally, cells in five random fields were counted using a BX53 upright microscope (Olympus Corp.) at 200 × magnification.
Western blotting
Western blotting was performed according to standard protocols. HCT116 and SW620 cells were cultured in six-well plates and treated with icaritin (10 μM) for the specified duration. To measure protein levels, a bicinchoninic acid protein assay kit (Beyotime Biotechnology, Shanghai, China) was used. SDS-PAGE was used to separate 35 μg total protein from each sample, which was subsequently transferred to a polyvinylidene difluoride membrane (PVDF; Solarbio, Beijing, China). Membranes were blocked with 5% skimmed milk in Tris-buffered saline-tween (TBS-T) for 1 hour at room temperature. After blocking, the membranes were incubated overnight at 4 °C with primary antibodies against N-cadherin, Wnt3a, β-catenin, c-MYC, cyclin D1, and MMP7. Following three washes (7-10 minutes each) with TBS-T, the membranes were incubated with secondary antibodies (KPL, Gaithersburg, MD, United States) for 1 hour at room temperature. After three additional washes (5 minutes each) with TBS-T, chemiluminescence detection was performed to visualize protein bands on autoradiographic film. Protein band intensity was analyzed using Image-Pro Plus (Version 11.0.2, Media a loading control.
Establishment of HCT116 nude mouse xenograft tumor model
Nude mouse xenograft models were established by subcutaneously injecting HCT116 cells (1 × 106 cells/mL to 5 × 107 cells/mL) into female athymic nude mice (BALB/c-nu/nu) aged 9-24 weeks, obtained from Beijing Vital River Laboratory Animal Technology Co. Ltd. The mice were kept in a specific pathogen-free environment and randomly assigned to the icaritin group or control group. Five mice in the icaritin group received daily intraperitoneal injections of icaritin at a concentration of 10 μM for 39 days. Five mice in the control group received an equal volume of normal saline. The mice were weighed twice weekly under specific pathogen-free conditions. During the experiment, the mice were anesthetized and placed in the supine position, and their abdomens were disinfected with 70% alcohol. An incision of less than 1 cm was made to expose the subcutaneous tumors. On day 39 post-injection, the mice were sacrificed by CO2 asphyxiation. Tumors were excised for measurement of weight and volume and photographed. Following fixation in formalin, paraffin sections were generated and stained using hematoxylin and eosin. This study was approved by the Ethics Committee of the Affiliated Zhongshan Hospital of Dalian University (No. DW2024-023-01).
Immunohistochemistry
Immunohistochemistry (IHC) was conducted following established protocols. Sections from xenograft tumors were dewaxed and rehydrated using the ElivisionTM Plus immunohistochemistry kit. Tissue sections were incubated overnight at 4 °C with primary antibodies against mouse anti-Ki67, MMP2, MMP9, β-catenin, and E-cadherin. A goat anti-mouse immunoglobulin G linked to horseradish peroxidase was used as the secondary antibody. Staining was performed with diaminobenzidine (Sigma, St. Louis, MO, United States), followed by hematoxylin. Positive cells were recognized by brown granules in the cytoplasm or nucleus. Following staining, an independent pathologist assessed immunohistochemical intensity and pathological features of all tumor specimens.
Statistical analysis
Data were presented as mean ± SD. Statistical analyses were conducted using Statistical Package for the Social Sciences version 16.0 (SPSS Inc., Chicago, IL, United States) and Excel 2016 (Microsoft, Redmond, WA, United States). Differences were evaluated using Student’s t-test or one-way analysis of variance (ANOVA) followed by Student-Newman-Keuls tests. GraphPad Prism version 6.0 (GraphPad Software Inc., San Diego, CA, United States) was utilized to generate graphs based on the relevant data, with P < 0.05 considered statistically significant.
RESULTS
Icaritin inhibits HCT116 and SW620 cell growth
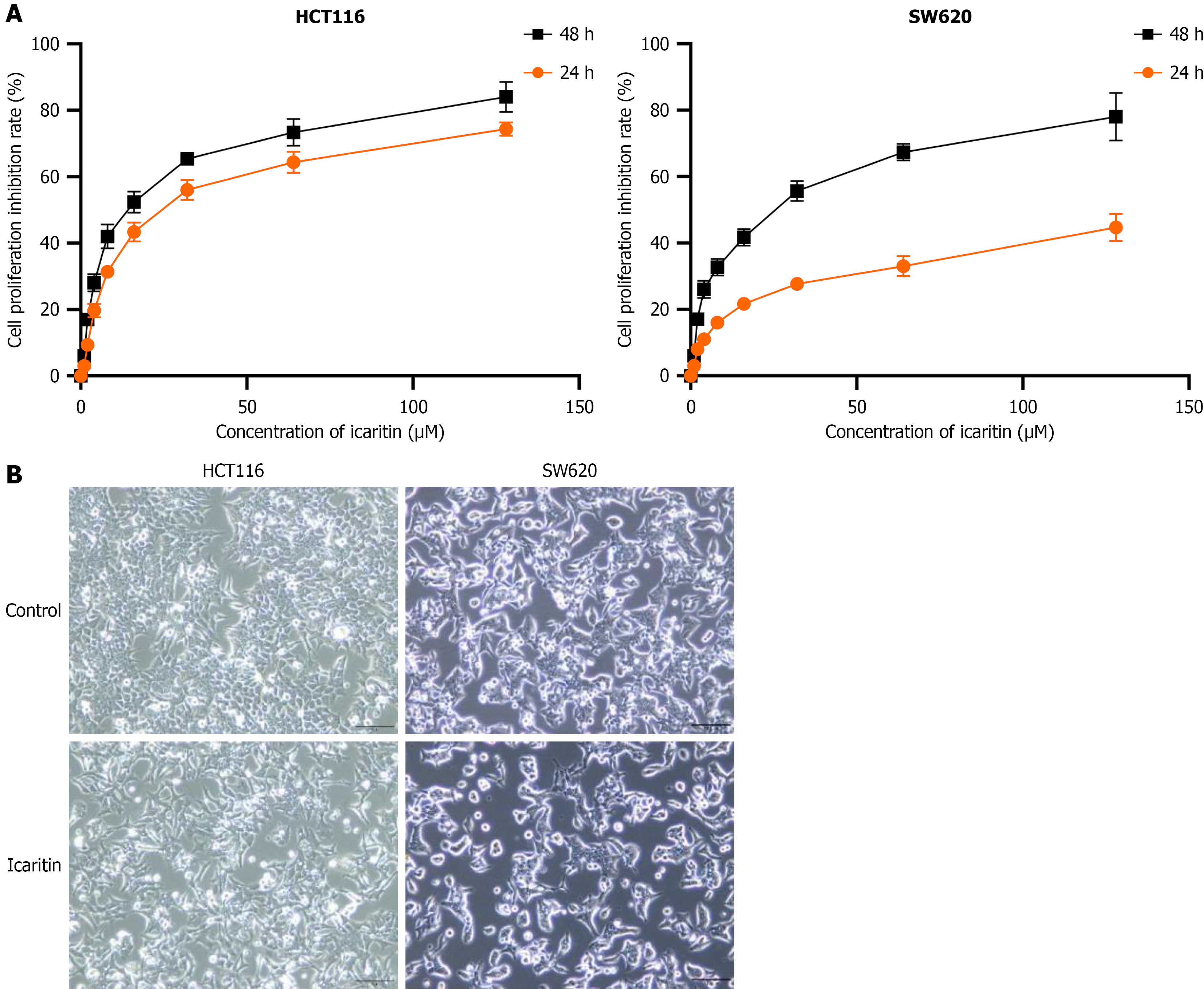
To evaluate the effect of icaritin on HCT116 and SW620 cell proliferation, we treated cells with various concentrations of icaritin (0-130 μM) for 24 hours and 48 hours, and assessed cell viability using the CCK-8 assay. The results showed that icaritin significantly inhibited the proliferation of both cell lines in dose-dependent manner and in a time-dependent manner (Figure 1). The IC50 for HCT116 cells was approximately 10 μM after 24 hours, while it was approximately 12 μM for SW620 cells (Figure 1A). Therefore, 10 μM for HCT116 and 12 μM for SW620 were used in subsequent experiments. Morphological assessments conducted 24 hours after icaritin treatment demonstrated that cells in the icaritin group displayed a more epithelial morphology compared to controls, suggesting a potential reduction in invasiveness (Figure 1B).
Figure 1 Icaritin inhibits growth of colorectal cancer cells.
A: Cell counting kit-8 assay confirmed the suppressive effect of icaritin on HCT116 and SW620 cell proliferation, clearly showing a significant time-dependent decrease in cell growth; B: Impact of icaritin on cell proliferation and morphological alterations of human colon cancer. Morphological transformations after icaritin treatment were observed under the microscope (200 × and 400 ×). HCT116 and SW620 cells lost their fibroblast-like shape and adopted an epithelial morphology.
Icaritin promotes HCT116 and SW620 cell apoptosis
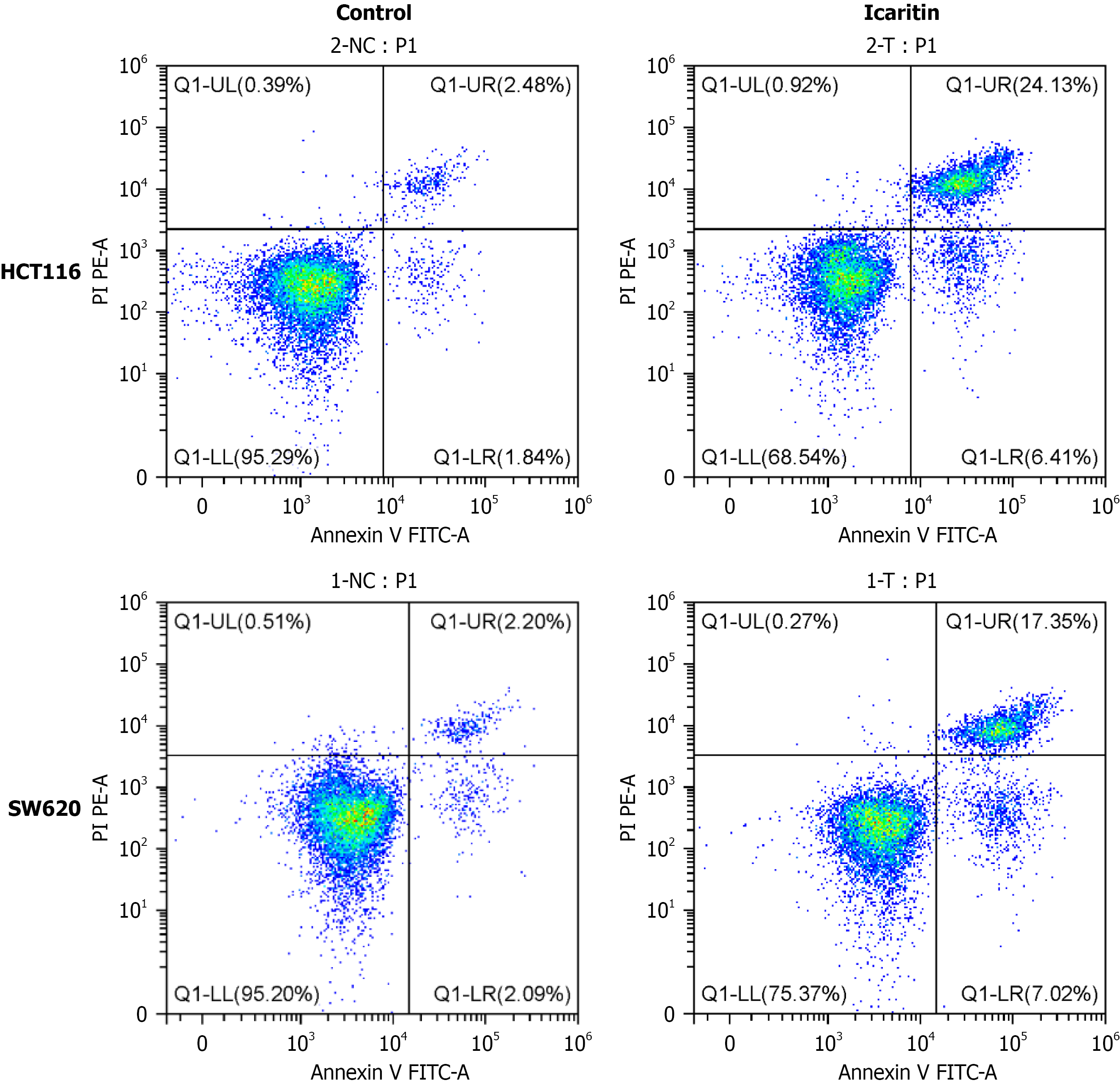
We used an Annexin V/propidium iodide staining assay to assess the impact of icaritin on CRC cell apoptosis. Flow cytometry analysis showed a significant increase in apoptosis of both HCT116 and SW620 cells following icaritin treatment compared with untreated controls (Figure 2; P < 0.01).
Figure 2 Annexin V/propidium iodide double staining.
Colorectal cancer cel (HCT116 and SW620) apoptosis markedly increased upon icaritin treatment compared with the control group (P < 0.01). The results are presented as mean ± SD (n = 3). FITC: Fluorescein isothiocyanate; PI: Propidium iodide.
Icaritin inhibited human colon cancer cell migration and invasion
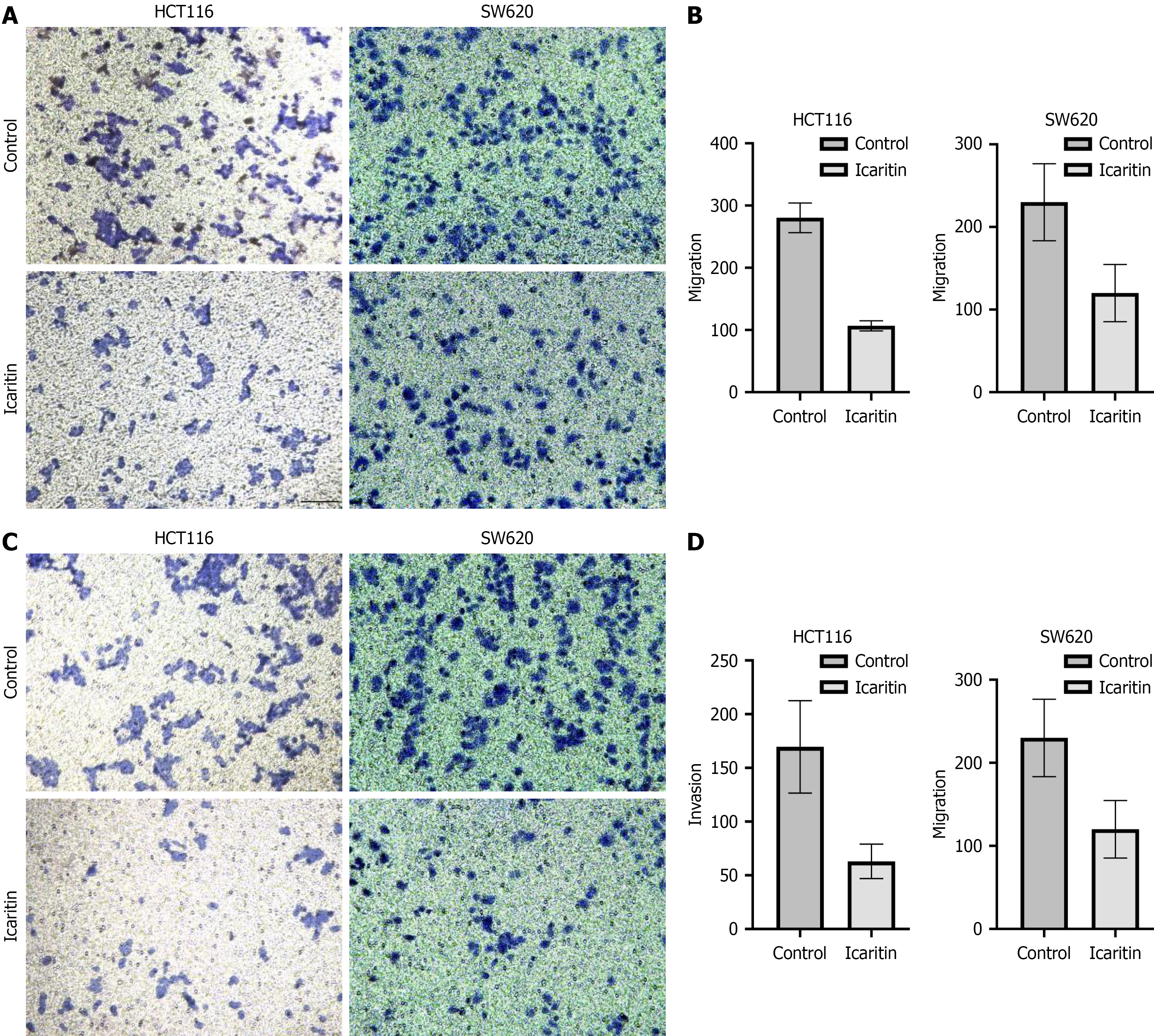
Transwell assays demonstrated that 48 hours of icaritin treatment significantly inhibited HCT116 and SW620 cell migration compared with control treatment (Figure 3A and B; P < 0.01). To evaluate the effect of icaritin on colon cancer cell invasion, we applied Matrigel was applied to the upper chamber of the Transwell. Icaritin treatment markedly reduced the number of cells that passed through the membrane (Figure 3C and D; P < 0.01).
Figure 3 Impact of icaritin on HCT116 and SW620 cell migration and invasive behavior.
A and B: Effect of icaritin on colon cancer cell migration (crystal violet 200 ×), assessed by Transwell assay without Matrigel. The number of migrating cells was quantified for HCT116 and SW620 cells; C and D: The impact of Clostridium butyricum on colon cancer cell invasion was evaluated using a Transwell assay incorporating Matrigel (crystal violet 200 ×). The number of invasive cells was quantified for HCT116 and SW620 cells. The results are displayed as the mean ± SD (n = 3), with P < 0.01 denoting a significant difference compared with the controls.
Icaritin inhibits cell invasion and metastasis by regulating EMT and Wnt/ß-catenin cell signaling
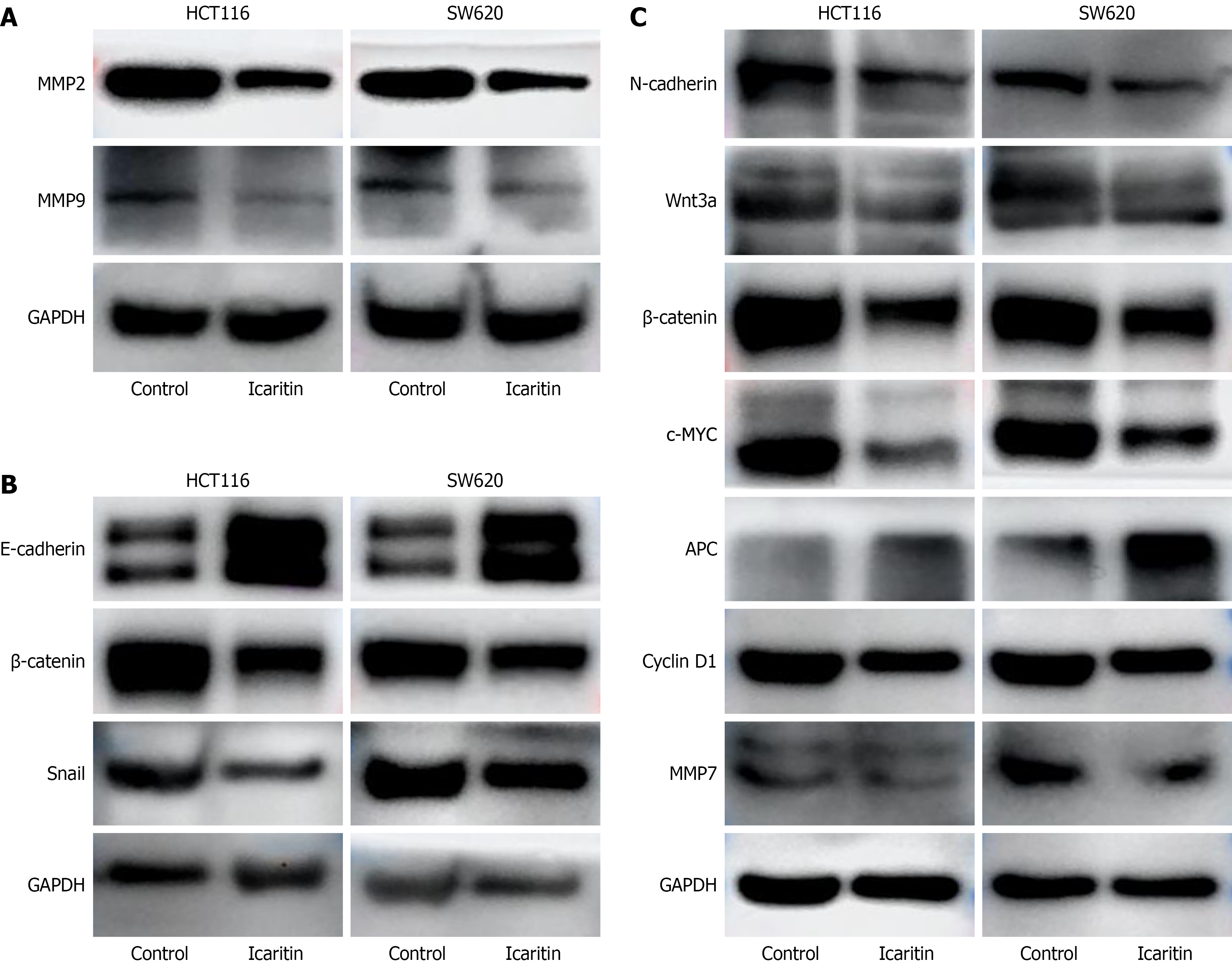
To investigate how icaritin affected colon cancer cell migration and invasion, we analyzed MMP2 and MMP9 protein expression levels by western blotting. Icaritin treatment significantly decreased their expression compared with untreated controls (Figure 4A; P < 0.05). We also examined the expression of EMT proteins following icaritin treatment and found that E-cadherin levels increased, while Snail, β-catenin (Figure 4B), and N-cadherin (Figure 4C) expression decreased compared with controls (P < 0.05).
Figure 4 Effects of icaritin on epithelial-mesenchymal transition and the Wnt/β-catenin signaling pathway.
A: Matrix metalloproteinase 2 (MMP2) and MMP9, associated with migration and invasion, showed decreased expression following icaritin treatment; B: Epithelial-mesenchymal transition-related gene expression of E-cadherin showed an increase and decrease in N-cadherin, Snail, and β-catenin; C: Expression of all the Wnt/β-catenin signaling-related genes, except adenomatous polyposis coli, decreased after icaritin treatment, including Wnt3a, β-catenin, c-MYC, cyclin D1, and MMP7. All data are displayed as mean ± SD (n = 3). P < 0.05 compared with the control group. MMP: Matrix metalloproteinase.
To clarify the mechanism by which icaritin inhibits EMT in CRC, we analyzed Wnt/β-catenin pathway gene expression. Western blotting demonstrated that icaritin treatment significantly decreased the levels of β-catenin, Wnt3a, c-MYC, cyclin D1, and MMP7, while increasing APC expression (Figure 4C; P < 0.05). Taken together, icaritin suppressed β-catenin activity and EMT in CRC. Thus, by modulating key components of the Wnt/β-catenin pathway and EMT markers, icaritin demonstrates significant therapeutic potential in CRC.
Effects of icaritin on metastasis and EMT-related genes in vivo
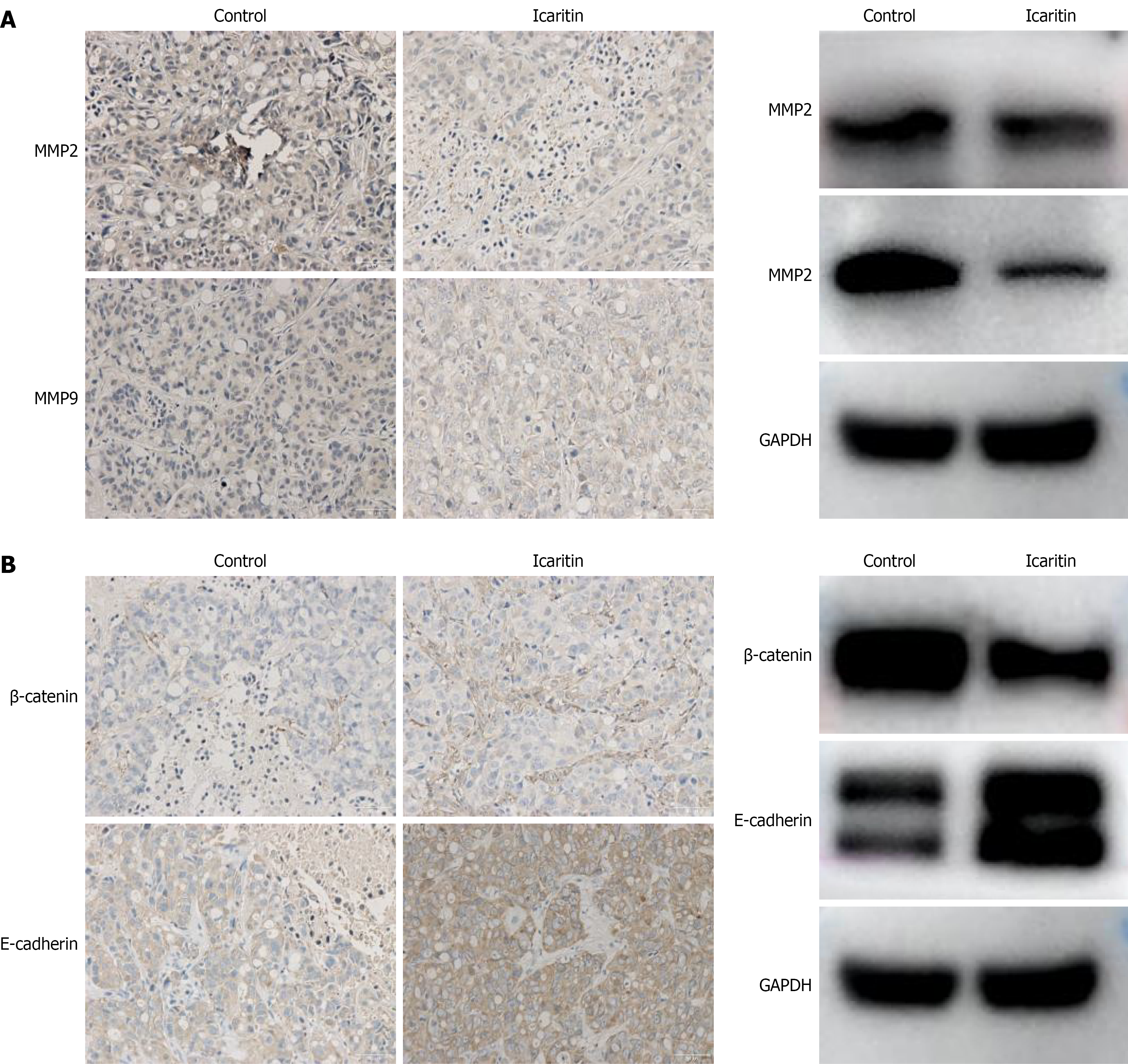
To investigate the impact of icaritin on EMT in CRC in vivo, we performed IHC and western blotting to analyze the expression of various EMT-related proteins in vivo. There was a significant reduction in MMP2, MMP9, and β-catenin in tumor tissues compared with controls (P < 0.05). Notably, E-cadherin expression increased after icaritin treatment (Figure 5), which is consistent with our in vitro findings.
Figure 5 Effects of icaritin on epithelial-mesenchymal transition-related genes in colorectal cancer in vivo.
A: Immunohistochemistry assay was used to examine the expression of tumor-metastasis-related genes matrix metalloproteinase 2, matrix metalloproteinase 9, and epithelial-mesenchymal transition-related genes β-catenin and E-cadherin. Positive cells were characterized by brown granules in the cytoplasm (immunohistochemistry 200 ×); B: Western blotting. GAPDH served as a reference for determining expression levels. The data are shown as mean ± SD (n = 10), P < 0.05 compared with the control group. MMP: Matrix metalloproteinase.
Icaritin inhibits colon cancer formation in vivo
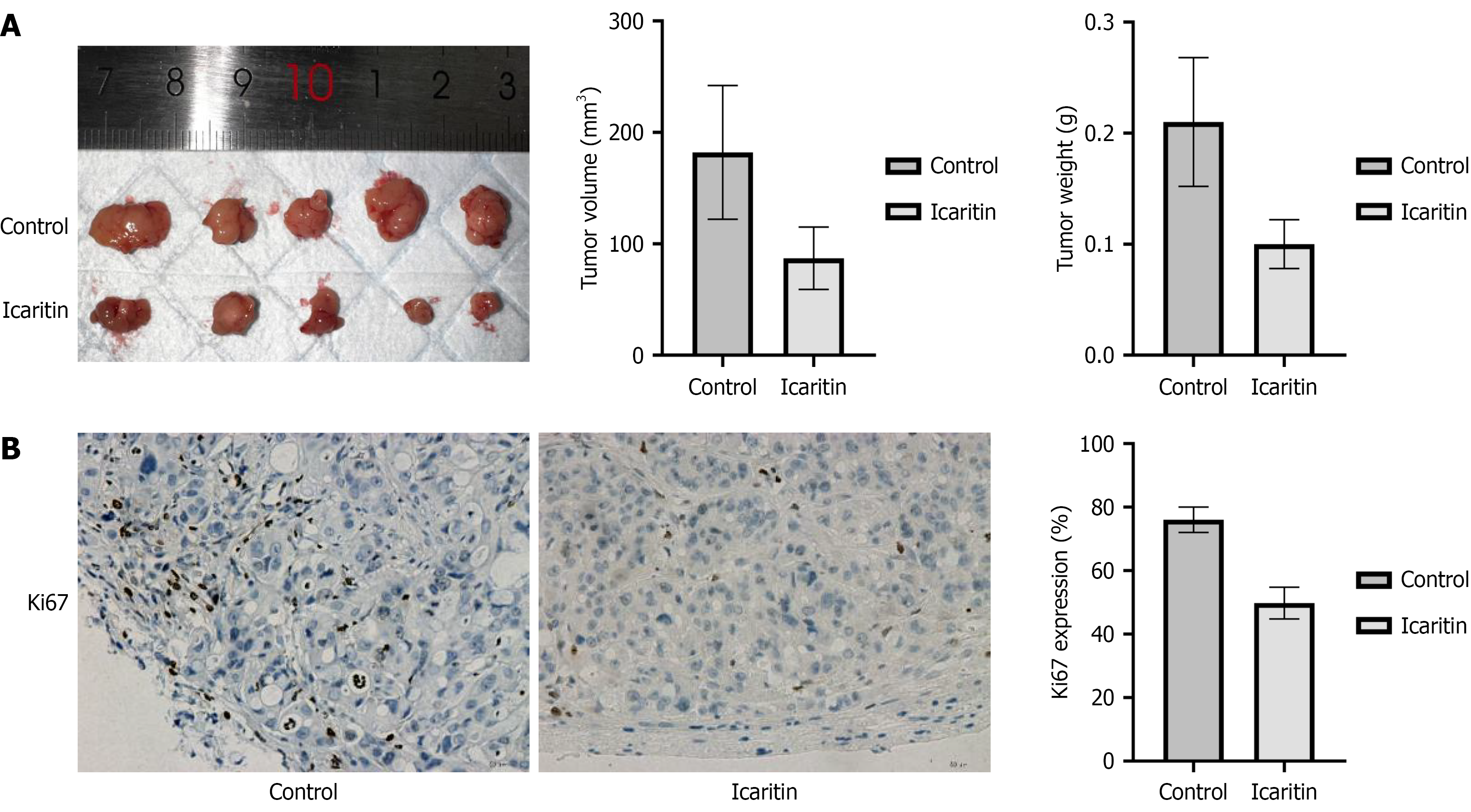
To further investigate the anti-cancer potential of icaritin in CRC, we established an orthotopic xenograft tumor model by injecting HCT116 cells into nude mice. After 39 days of observation, the tumor volume in the icaritin-treated group was reduced by approximately 54% and the tumor weight was reduced by approximately 50% compared with the control group (Figure 6A; P < 0.05), indicating that icaritin significantly inhibited tumor growth. IHC analysis revealed downregulation of Ki67 expression following icaritin treatment (Figure 6B; P < 0.05). These findings are consistent with the in vitro results and demonstrate that icaritin can suppress cell proliferation and tumor formation.
Figure 6 Effects of icaritin on colon cancer formation in vivo.
Thirty-nine days after establishment of the xenograft model, all mice were sacrificed and subcutaneous tumors were dissected. A: Volume and weight of dissected tumors; B: Positive cells, characterized by brown granules in the nuclei, were identified in the immunohistochemistry assay assessing Ki67. Indexes are shown as mean ± SD (n = 5), P < 0.05 compared with the control group.
DISCUSSION
At its advanced (metastatic) stage, CRC is a complex and heterogeneous disease associated with challenging outcomes. The incidence of CRC has remained notably high even in the most developed Western countries. Early diagnosis usually depends on endoscopic examination, but nearly 50% of patients present with signs suggestive of tumor metastasis during initial diagnosis. Metastasis remains a crucial factor and a significant clinical challenge contributing to CRC-associated mortality[21]. Notably, hepatic metastasis comprises approximately 70% of all metastases in patients with CRC. Additionally, some patients do not respond to conventional chemotherapy, leading to poor prognosis[22,27]. The clinical management and therapeutic strategies used in CRC are crucial for improving patient survival and treatment outcomes. Current treatment options include various drugs and approaches, such as local resection, combination therapy, and others[28]. Due to advances in CRC treatment, the survival duration for patients with advanced or recurrent CRC has extended from approximately 5 months to approximately 30 months or more. Despite these advances, the 5-year survival rate for patients with metastatic and advanced CRC remains below 10%. Moreover, each treatment modality is associated with significant side effects and complications[29,30]. Numerous studies across different cancers have shown that the Wnt/β-catenin pathway is constitutively activated, thereby promoting EMT[31]. EMT significantly contributes to the invasion and metastasis of various malignant tumors, including CRC[31-34]. In this study, we explored how icaritin regulates these pathways to inhibit CRC invasion and metastasis.
The occurrence of EMT is closely associated with MMPs, which are influenced by Wnt/β-catenin signaling[35-38]. A considerable body of research has indicated that MMP family proteins are important for cancer metastasis, owing to their capacity to destroy the extracellular matrix and facilitate cancer cell infiltration through the basement membrane[39,40]. Tumor tissue usually expresses high MMP levels, while normal healthy tissue does not[41,42]. Following icaritin administration, we observed a time-dependent decrease in MMP2 and MMP9 protein expression. These results indicate that icaritin suppresses CRC cell EMT by inhibiting MMP2 and MMP9.
In this study, we found that icaritin inhibited EMT in HCT116 and SW620 cells. MMP2 and MMP9 protein expression decreased, as shown by IHC and western blotting. Compared with the control group, E-cadherin expression increased; however, β-catenin and Snail levels decreased in HCT116 and SW620 cells upon icaritin treatment. In in vivo models, icaritin treatment decreased E-cadherin, β-catenin, MMP2, and MMP9 expression, consistent with previous studies[43]. EMT may play a vital role in tumor metastasis[44,45]. A notable feature of EMT in cancer is diminished E-cadherin function. Loss of cell polarity and adhesion complexes can result in loss of epithelial cell polarity, promoting development of EMT and enhancing the invasive potential of tumor cells[46]. In cancer cells, Snail protein can suppress E-cadherin expression while promoting the expression of mesenchymal markers, such as fibronectin and zinc-finger E-box binding homeobox 1[47,48]. In our study, icaritin decreased Snail protein expression, promoted E-cadherin expression, and induced EMT reversal, which resulted in HCT116 and SW620 cells losing their fibroblast-like appearance and gradually restoring their original epithelioid morphology. In contrast, the invasion and migration capacity of CRC cells in Transwell assays was markedly decreased after intervention with some drugs in vitro. Several studies have demonstrated that the Wnt/β-catenin signaling pathway can promote CRC cell EMT and metastasis[49,50]. Notably, Transwell assays revealed that icaritin treatment dramatically reduced HCT116 and SW620 cell migration and invasion. Therefore, our data suggest that icaritin inhibits CRC cell invasion and metastasis by regulating EMT and the Wnt/β-catenin pathway.
The Wnt/β-catenin pathway can facilitate communication between cells, which is important for processes such as cell proliferation, differentiation, and stem cell renewal during embryogenesis and adult tissue homeostasis[51]. There is evidence to suggest that Wnt/β-catenin has a role in CRC progression, with its activation known to promote extracellular matrix remodeling, facilitating CRC cell growth, proliferation, and metastasis[52]. The integrity and function of β-catenin rely on some special cell structures, such as the cytoplasmic APC/axin destruction complex. The major domains of the APC protein are three axin-binding sequence motifs distributed among approximately 15-amino and 20-amino acid repeats, which can bind to β-catenin. APC may play a crucial role in destruction complex activity in CRC. Its loss of function leads to abnormal cytoplasmic accumulation of β-catenin and subsequent translocation into the nucleus, further activating target genes, including Wnt3a, c-MYC, cyclin D1, and MMP7. All of these genes are involved in regulating cancer cell cycle, proliferation, EMT, and metastasis[53]. Consequently, we showed that icaritin treatment increased APC expression and decreased expression of N-cadherin, Wnt3a, β-catenin, c-MYC, cyclin D1, and MMP7. These data are consistent with a model in which icaritin inhibits CRC EMT and metastasis through inhibition of Wnt/β-catenin signaling.
Previous research indicates that natural compounds can impede CRC progression through various mechanisms, including induction of apoptosis, inhibition of migration and invasion, inhibition of pivotal cellular signaling pathways, and modulation of the immunosuppressive tumor microenvironment[54]. Numerous investigations have showcased the ability of icaritin to suppress cell proliferation and migration, induce apoptosis and differentiation, regulate autophagy, and disrupt the tumor cell cycle by modulating various signaling pathways[55]. Icaritin can markedly impede hepatocellular carcinoma growth by suppressing the proliferation of human liver cancer SMMC-7721 cells. The inhibitory effects are dose- and time-dependent[56]. Simultaneously, researchers have discovered that icaritin can induce tumor cell apoptosis via mitochondria-mediated apoptosis pathways. This process involves the loss of membrane potential of intracellular mitochondria and the alteration of the balance between the anti-apoptotic protein Bcl-2 and the pro-apoptotic protein Bax; specifically, it leads to decreased Bcl-2 expression and increased Bax expression. JNK activation plays an important role in apoptosis[57]. Moreover, the relevant research data confirmed its anti-cancer activity and indicated that the mechanism may be related to protein kinases (mitogen-activated protein kinase, protein kinase B, JNK, phosphatidylinositol 3-kinase, etc.), growth factors (vascular endothelial growth factor, transforming growth factor-β, epidermal growth factor, etc.), and transcription factors (nuclear factor-kappa B, signal transducer and activator of transcription 3, β-catenin, etc.)[41-44]. As described above, we found that icaritin promotes apoptosis and inhibits cell proliferation, demonstrating its therapeutic potential in CRC.
Icaritin has been approved for liver cancer treatment in China and shows particularly significant clinical effects when used in combination with other therapies. In our previous studies, including both in vivo and in vitro studies, all outcomes strongly demonstrated its positive effects in pancreatic cancer treatment. Although there has been growing insight into the biology of CRC and many therapeutic improvements that have emerged in recent decades, preclinical in vivo models are still indispensable for the research and development of new treatment approaches and modalities. We successfully established an orthotopic xenograft tumor model in nude mice using HCT116 cells; this model more closely resembles the true process of tumor growth and metastasis. Through an in vivo orthotopic xenograft tumor study, we found that icaritin treatment significantly decreased xenograft tumor volume and weight compared with the controls. In addition, combining the research results in vivo of HCT116 and SW620 cells demonstrated the effectiveness of icaritin in suppressing CRC invasion and metastasis.