Published online May 15, 2026. doi: 10.4251/wjgo.v18.i5.115303
Revised: November 26, 2025
Accepted: February 2, 2026
Published online: May 15, 2026
Processing time: 195 Days and 1 Hours
Postoperative complications in colorectal cancer surgery occur in 20%-50% of patients, significantly impacting recovery and prognosis. Traditional clinical prediction models have limited efficacy for preoperative risk assessment. Spectral computed tomography (CT) provides quantitative parameters including iodine concentration (IC) that may improve complication prediction, though systematic studies remain limited.
To investigate the value of preoperative spectral CT multi-parameters in pre
A retrospective cohort analysis was conducted on clinical data and preoperative spectral CT imaging data of 195 colorectal cancer patients from March 2022 to August 2025. Abdominal dual-phase enhanced scanning was performed using a GE Revolution CT spectral scanner, measuring parameters including tumor IC, normalized IC (NIC), effective atomic number, and spectral attenuation curve slope. Postoperative complications were assessed using the Clavien-Dindo classification system. Univariate and multivariate logistic regression analyses were used to screen independent predictive factors, establish a predictive model, and evaluate its performance.
Among 195 patients, 16 developed postoperative complications with an incidence rate of 8.2%. Patients in the complication group had significantly lower arterial phase and portal venous phase (PV) tumor IC, NIC, effective atomic number, and spectral attenuation curve slope compared to the non-complication group (P < 0.001). Multivariate analysis identified 5 independent predictive factors: Age [odds ratio (OR) = 1.042, P = 0.023], albumin (OR = 0.881, P = 0.007), PV tumor NIC (OR < 0.001, P < 0.001), tumor-nodes-metastasis stage (OR = 3.274, P = 0.030), and intraoperative blood loss (OR = 1.003, P = 0.009). The combined predictive model had an area under the curve of 0.843 (95% confidence interval: 0.789-0.897), with sensitivity of 75.3% and specificity of 80.3%. Bootstrap internal validation showed a bias-corrected C-index of 0.821, demonstrating good stability.
Preoperative spectral CT multi-parameters have important value in predicting postoperative complications in colorectal cancer. PV tumor NIC is the most valuable imaging predictive factor. The predictive model established by combining clinical indicators has good predictive efficacy and can be used for preoperative risk stratification and individualized treatment strategy formulation.
Core Tip: This study developed a preoperative model combining spectral computed tomography parameters and clinical factors to predict postoperative complications in colorectal cancer. The portal venous phase normalized iodine concentration was identified as the most valuable imaging biomarker. The integrated model showed good discrimination (area under the curve = 0.843) and calibration (C-index = 0.821), providing a noninvasive tool for preoperative risk stratification and individualized perioperative management in colorectal cancer patients.
- Citation: Duan R, Li HL, Deng L, Zhao Q, Yang RW, Wu ZL. Preoperative spectral computed tomography multi-parameter prediction of postoperative complications in colorectal cancer: A single-center retrospective cohort study. World J Gastrointest Oncol 2026; 18(5): 115303
- URL: https://www.wjgnet.com/1948-5204/full/v18/i5/115303.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i5.115303
Colorectal cancer is one of the most common malignant tumors worldwide, with incidence and mortality rates ranking among the top, seriously threatening human health[1]. With the continuous advancement of surgical techniques and optimization of perioperative management, the overall prognosis of colorectal cancer patients has improved, but postoperative complications remain an important factor affecting patient recovery, prolonging hospital stay, and increasing medical costs[2]. According to literature reports, the incidence of postoperative complications in colorectal cancer ranges from approximately 20%-50%, with severe complications potentially leading to significantly increased patient mortality and seriously affecting long-term survival and quality of life[3,4]. Therefore, accurate preoperative identification of high-risk patients and implementation of corresponding preventive measures has important clinical significance for improving patient prognosis.
Currently, prediction of postoperative complications mainly relies on traditional clinical indicators such as patients' general condition, comorbidities, tumor characteristics, and surgical factors, but the predictive efficacy of these indicators is limited and difficult to meet the requirements of precision medicine[5]. In recent years, the role of imaging technology in tumor diagnosis and treatment has become increasingly prominent, providing new approaches for preoperative risk assessment. Spectral computed tomography (CT), as an emerging imaging technology, can obtain material composition information that traditional CT cannot provide through rapid switching of X-rays with different energies, including various quantitative parameters such as iodine concentration (IC), effective atomic number, and spectral attenuation curves[6]. These parameters can more accurately reflect the pathophysiological characteristics of tissues and provide important information for individualized tumor diagnosis and treatment.
Previous studies have shown that spectral CT parameters are closely related to the biological behavior of various tumors and show good application prospects in tumor staging, efficacy evaluation, and prognosis prediction[7]. However, research on spectral CT parameters predicting postoperative complications in colorectal cancer is relatively limited, mostly consisting of small-sample preliminary explorations, lacking systematic multi-parameter analysis and predictive model construction[8]. Additionally, existing studies mostly focus on single parameter analysis and fail to fully utilize the advantages of spectral CT multi-parameter combined analysis, limiting their clinical application value[9].
Based on the above background, this study aims to retrospectively analyze preoperative spectral CT imaging data of 195 colorectal cancer patients to provide important scientific evidence for preoperative risk stratification and individualized treatment strategy formulation for colorectal cancer patients.
This study was a single-center retrospective cohort study, approved by the Medical Ethics Committee of our hospital and complied with the requirements of the Declaration of Helsinki. Due to its retrospective nature, patient informed consent was waived.
Inclusion criteria: (1) Colorectal cancer patients hospitalized in our hospital from March 2022 to August 2025; (2) Completion of abdominal dual-phase enhanced spectral CT examination within 1 week before surgery; (3) Receiving radical surgical treatment with pathological confirmation of colorectal adenocarcinoma; (4) Complete clinical data and complete postoperative follow-up data; and (5) Age ≥ 18 years.
Exclusion criteria: (1) Patients receiving preoperative neoadjuvant chemotherapy or radiotherapy; (2) Patients with previous abdominal surgery history; (3) Patients with other malignant tumors; (4) Patients with severe heart, liver, or kidney dysfunction; (5) Poor CT image quality affecting measurement; and (6) Loss to follow-up or postoperative follow-up time < 30 days.
All patients completed abdominal dual-phase enhanced spectral CT examination within 1 week before surgery. A GE Revolution CT spectral scanner was used for examination. Patients fasted for 8-12 hours before examination, and 30 minutes before examination, 800-1000 mL of 1.5%-2.0% iodinated contrast solution was taken orally to fill the gastroin
Scanning parameters: Spectral imaging mode, tube voltage rapid switching between 80 kVp and 140 kVp, tube current 350-600 mA (automatically adjusted according to patient body type), pitch 0.984:1, gantry rotation time 0.6 seconds, slice thickness 5 mm, slice interval 5 mm, reconstruction slice thickness 1.25 mm. Scanning range from diaphragm top to the level below the pubic symphysis.
Contrast agent used was non-ionic iodinated contrast agent iohexol (IC 350 mgI/mL), dose 1.5 mL/kg body weight, injected via antecubital vein at a flow rate of 3.0-3.5 mL/seconds, followed by injection of 30 mL normal saline at the same flow rate after completion of contrast injection.
Scanning phases: Contrast tracking technique was used, with the region of interest (ROI) set in the central lumen of the abdominal aorta at the renal artery level, trigger threshold 120 HU. Arterial phase scanning delay time was 18-25 seconds after triggering, portal venous phase (PV) scanning delay time was 65-70 seconds after triggering.
During examination, patients were instructed to hold their breath to ensure image quality. All image data were transmitted to a GE AW4.7 workstation for post-processing analysis.
All spectral CT raw data were transmitted to a GE AW4.7 workstation, and GSI Viewer software was used for image post-processing analysis. The following images were reconstructed using spectral imaging data: (1) Monoenergetic images: Optimal monoenergetic images in the 40-140 keV range were selected for analysis; (2) Material decomposition images: Iodine-based and water-based images were generated; (3) Effective atomic number maps; (4) Spectral attenuation curves; and (5) 120 kVp equivalent images: Reconstructed using weighted average algorithm of 0.5 kVp × 80 kVp image + 0.5 kVp × 140 kVp image, simulating traditional 120 kVp scanning images.
Two radiologists with more than 5 years of abdominal CT diagnostic experience independently performed image analysis and parameter measurement, and consensus was reached through discussion when opinions differed. ROIs were manually drawn on the maximum cross-sectional plane of the tumor, with specific requirements as follows: (1) Tumor ROI: Manually drawn along tumor margins, avoiding necrosis, cystic changes, calcification, and artifact areas, area ≥ 50 mm²; (2) Normal bowel wall ROI: Set on normal bowel wall > 5 cm from tumor margin, area approximately 30-50 mm²; (3) Abdominal aorta ROI: Set as circular ROI in the central lumen of abdominal aorta at renal artery level, area approximately 100 mm², avoiding wall calcification; and (4) Erector spinae ROI: Set as elliptical ROI on bilateral erector spinae at the same level, area approximately 150 mm², taking the average of both sides.
The following parameters were measured in arterial and PVs respectively: (1) IC: IC values of each ROI measured on iodine-based images, unit μg/cm³; (2) Normalized IC (NIC): Ratio of tumor IC to abdominal aorta IC in the same phase; (3) Effective atomic number (Zeff): Parameter reflecting tissue atomic number; (4) Spectral attenuation curve slope (λHU): Calculated in the 40-100 keV range, formula λHU = (CT40 keV-CT100 keV)/60; (5) CT value: CT values of each ROI measured on 120 kVp equivalent images; and (6) 40 keV monoenergetic CT value: CT values measured on 40 keV monoenergetic images.
Quality control included: (1) All measurements were repeated 3 times by the same observer at the same time point, taking the average; (2) ROI placement avoided motion artifacts and beam hardening artifact areas; (3) ROI positions were kept consistent between arterial and venous phases; and (4) Inter-observer consistency assessment: 30 cases were randomly selected for independent repeated measurement by two observers, calculating intraclass correlation coefficient (ICC).
The following derived parameters were calculated: (1) Enhancement degree: Enhanced CT value-plain CT value; (2) Relative enhancement rate: (Enhanced CT value-plain CT value)/plain CT value × 100%; (3) IC difference: PV IC-arterial phase IC; and (4) Tumor-muscle ratio: Tumor parameter value/erector spinae parameter value.
All measurement data were recorded in standardized data tables to ensure data accuracy and reproducibility. During measurement, measurers remained blinded to patient clinical information and postoperative complication status.
Clinical data for all patients were collected through the hospital information system and electronic medical record system.
General demographic data: (1) Age, gender, height, weight, body mass index; (2) Smoking history, alcohol history; and (3) Past medical history and comorbidities [hypertension, diabetes, coronary heart disease, chronic obstructive pulmonary disease (COPD), others].
Preoperative examination indicators: (1) Complete blood count: Hemoglobin (HGB), white blood cell count (WBC), neutrophil count (NEU), lymphocyte count (LYM), platelet count; (2) Biochemical indicators: Albumin (ALB), prealbumin (PA), total protein (TP), creatinine (CREA), blood urea nitrogen (BUN), alanine aminotransferase, aspartate aminotransferase (AST); (3) Tumor markers: Carcinoembryonic antigen (CEA), carbohydrate antigen 19-9 (CA19-9); and (4) Coa
Tumor-related indicators: (1) Tumor location: Colon or rectum; (2) Preoperative tumor-nodes-metastasis (TNM) staging: According to American Joint Committee on Cancer 8th edition criteria; (3) Tumor differentiation degree: Well differentiated, moderately differentiated, poorly differentiated, undifferentiated; (4) Tumor diameter: Maximum diameter mea
Surgery-related data: (1) Intraoperative blood loss; (2) Number of lymph nodes dissected; (3) Margin status: Positive or negative; (4) Whether prophylactic stoma was performed; and (5) American Society of Anesthesiologists (ASA) classification.
Postoperative complications were defined as any abnormal conditions deviating from normal postoperative recovery process occurring within 30 days after surgery, requiring drug treatment, interventional treatment, or reoperation. Follow-up time was calculated from the day of surgery to the 30th postoperative day.
The Clavien-Dindo classification system was used to grade postoperative complications: Grade I: Deviation from normal postoperative course without need for pharmacological treatment or surgical, endoscopic, radiological interventions, allowed treatments include only antiemetics, antipyretics, analgesics, diuretics, electrolytes, physiotherapy, and other bedside routine treatments (these routine treatments are not considered “pharmacological treatment” in this classification system); Grade II: Complications requiring pharmacological treatment, including blood transfusion, total parenteral nutrition, etc.; Grade III: Complications requiring surgical, endoscopic, or radiological intervention, subdivided into Grade IIIa (intervention not requiring general anesthesia) and Grade IIIb (intervention requiring general anesthesia); Grade IV: Life-threatening complications requiring intensive care unit monitoring, subdivided into Grade IVa (single organ dysfunction) and Grade IVb (multi-organ dysfunction); Grade V: Patient death.
Major complication types included: (1) Anastomotic leakage: Diagnosed by clinical symptoms combined with CT or endoscopic examination; (2) Intra-abdominal infection: Including intra-abdominal abscess, diffuse peritonitis; (3) Intestinal obstruction: Mechanical or functional intestinal obstruction; (4) Incision complications: Including incision infection, dehiscence, hernia; (5) Cardiopulmonary complications: Pneumonia, pulmonary embolism, arrhythmia, etc.; (6) Urinary system complications: Urinary retention, urinary tract infection, etc.; (7) Bleeding: Postoperative bleeding re
According to clinical severity, Clavien-Dindo grades I-II were defined as minor complications, and grades III-V were defined as major complications.
SPSS 26.0 software (IBM Corp., Armonk, NY, United States) and R language 4.3.0 were used for statistical analysis. Test level α = 0.05, P < 0.05 was considered statistically significant.
Descriptive statistics: The Shapiro-Wilk test was used to assess normality of continuous variables. Continuous variables following normal distribution were expressed as mean ± SD, non-normally distributed continuous variables were expressed as median (interquartile range) [M (P25, P75)]. Categorical variables were expressed as n (%).
Group comparisons: Patients were divided into complication and non-complication groups based on whether complications occurred within 30 days after surgery. For continuous variable group comparisons: Independent samples t-test was used for normally distributed data with equal variances, Mann-Whitney U test was used for non-normally distributed data or unequal variances. χ2 test or Fisher’s exact test was used for categorical variable group comparisons.
Inter-observer consistency assessment: ICC was used to assess consistency between two observers measuring spectral CT parameters. ICC values 0.75-1.00 indicated excellent consistency, 0.60-0.74 indicated good, 0.40-0.59 indicated fair, < 0.40 indicated poor.
Univariate analysis: Univariate logistic regression analysis was performed on all clinical variables and spectral CT parameters to screen risk factors related to postoperative complications.
Multivariate analysis: Variables with P < 0.10 in univariate analysis were included in multivariate logistic regression analysis, using forward stepwise method to establish a predictive model. Multicollinearity testing was performed simultaneously, with variance inflation factor (VIF) < 5 considered as no collinearity problem.
Predictive model construction and validation: (1) Model construction: Based on multivariate logistic regression results, a predictive model was constructed and a nomogram was established; (2) Model performance assessment: Receiver operating characteristic (ROC) curve was used to assess model discrimination, calculating area under the curve (AUC); Hosmer-Lemeshow goodness-of-fit test was used to assess model calibration; and (3) Internal validation: Bootstrap method (1000 resamples) was used for internal validation, calculating bias-corrected C-index.
This study included 195 colorectal cancer patients meeting the criteria, including 114 males and 81 females, with ages ranging from 35-82 years and median age of 62 years. Based on whether complications occurred within 30 days after surgery, patients were divided into complication group (73 cases) and non-complication group (122 cases).
The two groups showed statistical differences in age, HGB, WBC, NEU, LYM, ALB, PA, TP, CEA, CA19-9, PT, APTT, INR, TNM stage, tumor differentiation degree, tumor diameter, intraoperative blood loss, positive margin rate, and ASA classification (P < 0.05) (Table 1).
| Characteristic | Total (n = 195) | Non-complication group | Complication group | Statistic | P value |
| General demographic data | |||||
| Age (years), M (P25, P75) | 62 (54, 71) | 61 (53, 69) | 65 (57, 74) | Z = -2.146 | 0.032 |
| Gender | χ² = 2.315 | 0.128 | |||
| Male | 114 (58.5) | 67 (54.9) | 47 (64.4) | ||
| Female | 81 (41.5) | 55 (45.1) | 26 (35.6) | ||
| BMI (kg/m²), mean ± SD | 23.4 ± 3.2 | 23.6 ± 3.1 | 23.0 ± 3.4 | t = 1.164 | 0.245 |
| Smoking history | 78 (40.0) | 45 (36.9) | 33 (45.2) | χ² = 1.308 | 0.253 |
| Alcohol history | 52 (26.7) | 29 (23.8) | 23 (31.5) | χ² = 1.315 | 0.251 |
| Past medical history/comorbidities | |||||
| Hypertension | 89 (45.6) | 52 (42.6) | 37 (50.7) | χ² = 1.191 | 0.276 |
| Diabetes | 31 (15.9) | 16 (13.1) | 15 (20.5) | χ² = 1.864 | 0.173 |
| Coronary heart disease | 24 (12.3) | 12 (9.8) | 12 (16.4) | χ² = 1.810 | 0.178 |
| COPD | 18 (9.2) | 8 (6.6) | 10 (13.7) | χ² = 2.735 | 0.098 |
| Preoperative examination indicators | |||||
| Hemoglobin (g/L), mean ± SD | 125.3 ± 18.7 | 128.1 ± 17.2 | 120.9 ± 20.5 | t = 2.517 | 0.013 |
| White blood cell count (× 109/L), M (P25, P75) | 6.8 (5.4, 8.3) | 6.5 (5.2, 7.9) | 7.2 (5.8, 8.9) | Z = -2.014 | 0.045 |
| Neutrophil count (× 109/L), M (P25, P75) | 4.2 (3.1, 5.6) | 3.9 (2.9, 5.2) | 4.7 (3.5, 6.1) | Z = -2.364 | 0.018 |
| Lymphocyte count (× 109/L), M (P25, P75) | 1.8 (1.4, 2.3) | 1.9 (1.5, 2.4) | 1.6 (1.2, 2.1) | Z = -2.289 | 0.022 |
| Platelet count (× 109/L), mean ± SD | 285.4 ± 78.9 | 278.2 ± 75.1 | 297.8 ± 84.2 | t = -1.622 | 0.106 |
| Albumin (g/L), mean ± SD | 38.9 ± 4.8 | 40.1 ± 4.2 | 36.8 ± 5.2 | t = 4.624 | < 0.001 |
| Prealbumin (mg/L), mean ± SD | 268.7 ± 58.3 | 279.4 ± 52.9 | 250.1 ± 63.8 | t = 3.358 | 0.001 |
| Total protein (g/L), mean ± SD | 68.5 ± 7.2 | 69.8 ± 6.8 | 66.4 ± 7.6 | t = 3.158 | 0.002 |
| Creatinine (μmol/L), M (P25, P75) | 78.5 (65.2, 93.1) | 76.8 (63.4, 91.2) | 81.7 (68.9, 96.3) | Z = -1.714 | 0.087 |
| Blood urea nitrogen (mmol/L), M (P25, P75) | 5.8 (4.6, 7.2) | 5.6 (4.4, 6.9) | 6.2 (5.0, 7.8) | Z = -1.876 | 0.061 |
| ALT (U/L), M (P25, P75) | 25.3 (18.7, 34.2) | 24.1 (17.9, 32.5) | 27.8 (20.4, 37.1) | Z = -1.502 | 0.134 |
| AST (U/L), M (P25, P75) | 28.9 (22.1, 38.7) | 27.2 (21.3, 36.4) | 31.8 (24.2, 42.1) | Z = -1.705 | 0.089 |
| CEA (ng/mL), M (P25, P75) | 8.7 (3.2, 18.9) | 7.1 (2.8, 15.4) | 12.3 (4.7, 24.8) | Z = -2.954 | 0.003 |
| CA19-9 (U/mL), M (P25, P75) | 19.8 (8.4, 45.7) | 17.2 (7.6, 38.9) | 25.4 (10.8, 58.3) | Z = -2.052 | 0.041 |
| PT (s), mean ± SD | 12.8 ± 1.4 | 12.6 ± 1.3 | 13.2 ± 1.5 | t = -2.628 | 0.009 |
| APTT (s), mean ± SD | 31.7 ± 4.2 | 31.2 ± 3.9 | 32.6 ± 4.7 | t = -2.151 | 0.032 |
| INR, mean ± SD | 1.08 ± 0.12 | 1.06 ± 0.11 | 1.12 ± 0.13 | t = -3.367 | 0.001 |
| Tumor-related indicators | |||||
| Tumor location | χ² = 1.725 | 0.189 | |||
| Colon cancer | 118 (60.5) | 77 (63.1) | 41 (56.2) | ||
| Rectal cancer | 77 (39.5) | 45 (36.9) | 32 (43.8) | ||
| TNM stage | χ² = 12.238 | 0.002 | |||
| Stage I | 38 (19.5) | 29 (23.8) | 9 (12.3) | ||
| Stage II | 71 (36.4) | 49 (40.2) | 22 (30.1) | ||
| Stage III | 86 (44.1) | 44 (36.1) | 42 (57.5) | ||
| Tumor differentiation | χ² = 6.949 | 0.031 | |||
| Well differentiated | 42 (21.5) | 31 (25.4) | 11 (15.1) | ||
| Moderately differentiated | 123 (63.1) | 79 (64.8) | 44 (60.3) | ||
| Poorly differentiated | 30 (15.4) | 12 (9.8) | 18 (24.7) | ||
| Tumor diameter (cm), mean ± SD | 4.8 ± 2.1 | 4.4 ± 1.9 | 5.5 ± 2.3 | t = -3.389 | 0.001 |
| Surgery-related data | |||||
| Intraoperative blood loss (mL), M (P25, P75) | 180 (120, 280) | 160 (110, 240) | 220 (150, 350) | Z = -3.872 | < 0.001 |
| Number of lymph nodes dissected, mean ± SD | 18.7 ± 6.8 | 19.2 ± 6.5 | 17.8 ± 7.3 | t = 1.410 | 0.156 |
| Positive margin | 23 (11.8) | 9 (7.4) | 14 (19.2) | χ² = 6.072 | 0.014 |
| Prophylactic stoma | 28 (14.4) | 14 (11.5) | 14 (19.2) | χ² = 2.146 | 0.143 |
| ASA grade ≥ III | 47 (24.1) | 23 (18.9) | 24 (32.9) | χ² = 4.831 | 0.028 |
Using randomly selected 30 patients, the consistency of spectral CT parameter measurements between two observers was assessed. Results showed that ICC values for all parameters were > 0.75, indicating excellent inter-observer consistency. Arterial phase tumor IC ICC was 0.891, PV tumor IC ICC was 0.885, arterial phase NIC ICC was 0.876, PV NIC ICC was 0.868, effective atomic number ICC was 0.894, and spectral attenuation curve slope ICC was 0.882. ICC values for other parameters were all in the range of 0.75-0.95, ensuring the reliability of measurement results.
Significant differences existed between the two groups in multiple spectral CT parameters in both arterial and PVs. The complication group had significantly lower tumor IC, NIC, effective atomic number, spectral attenuation curve slope, CT value, 40 keV CT value, enhancement degree, and relative enhancement rate compared to the non-complication group (P < 0.001). Among derived parameters, IC difference, tumor-muscle IC ratio, and CT value ratio also showed significant differences between groups (P < 0.05) (Table 2).
| Parameter | Non-complication group | Complication group | Statistic | P value |
| Arterial phase parameters | ||||
| Tumor iodine concentration (μg/cm³) | 18.7 ± 6.8 | 13.4 ± 5.9 | t = 5.487 | < 0.001 |
| Normal bowel wall iodine concentration (μg/cm³) | 12.3 ± 4.2 | 11.8 ± 4.5 | t = 0.787 | 0.432 |
| Abdominal aorta iodine concentration (μg/cm³) | 210.4 ± 45.7 | 208.9 ± 48.2 | t = 0.214 | 0.831 |
| Erector spinae iodine concentration (μg/cm³) | 11.2 ± 3.8 | 10.8 ± 4.1 | t = 0.731 | 0.465 |
| Tumor normalized iodine concentration | 0.089 ± 0.025 | 0.064 ± 0.022 | t = 7.015 | < 0.001 |
| Tumor effective atomic number | 8.21 ± 0.47 | 7.86 ± 0.52 | t = 4.729 | < 0.001 |
| Tumor λHU | 2.84 ± 0.68 | 2.31 ± 0.59 | t = 5.425 | < 0.001 |
| Tumor plain CT value (HU) | 34.5 ± 8.7 | 35.1 ± 9.2 | t = -0.443 | 0.658 |
| Tumor CT value (HU) | 67.8 ± 18.9 | 54.2 ± 16.7 | t = 5.053 | < 0.001 |
| Abdominal aorta CT value (HU) | 268.4 ± 52.3 | 265.7 ± 48.9 | t = 0.363 | 0.717 |
| Erector spinae CT value (HU) | 40.8 ± 12.4 | 40.1 ± 13.7 | t = 0.350 | 0.726 |
| Tumor 40 keV CT value (HU) | 142.7 ± 38.4 | 115.3 ± 32.1 | t = 5.030 | < 0.001 |
| Tumor enhancement degree (HU) | 33.3 ± 15.2 | 19.1 ± 12.8 | t = 6.625 | < 0.001 |
| Tumor relative enhancement rate (%) | 98.7 ± 47.3 | 56.2 ± 39.1 | t = 6.352 | < 0.001 |
| Portal venous phase parameters | ||||
| Tumor iodine concentration (μg/cm³) | 22.9 ± 7.3 | 16.8 ± 6.4 | t = 5.879 | < 0.001 |
| Normal bowel wall iodine concentration (μg/cm³) | 15.7 ± 5.1 | 15.2 ± 5.4 | t = 0.639 | 0.524 |
| Abdominal aorta iodine concentration (μg/cm³) | 158.3 ± 32.1 | 155.9 ± 35.4 | t = 0.480 | 0.632 |
| Erector spinae iodine concentration (μg/cm³) | 10.4 ± 3.2 | 9.9 ± 3.6 | t = 1.000 | 0.318 |
| Tumor normalized iodine concentration | 0.145 ± 0.031 | 0.108 ± 0.027 | t = 8.406 | < 0.001 |
| Tumor effective atomic number | 8.45 ± 0.52 | 8.07 ± 0.58 | t = 4.659 | < 0.001 |
| Tumor λHU | 3.12 ± 0.74 | 2.58 ± 0.67 | t = 5.109 | < 0.001 |
| Tumor CT value (HU) | 89.4 ± 22.1 | 71.6 ± 19.8 | t = 5.690 | < 0.001 |
| Abdominal aorta CT value (HU) | 184.7 ± 38.9 | 181.2 ± 41.3 | t = 0.580 | 0.563 |
| Erector spinae CT value (HU) | 40.5 ± 12.1 | 39.8 ± 13.2 | t = 0.369 | 0.712 |
| Tumor 40 keV CT value (HU) | 178.3 ± 45.7 | 142.9 ± 38.9 | t = 5.498 | < 0.001 |
| Tumor enhancement degree (HU) | 54.9 ± 18.4 | 36.5 ± 15.7 | t = 7.105 | < 0.001 |
| Tumor relative enhancement rate (%) | 161.8 ± 58.2 | 106.3 ± 48.9 | t = 6.781 | < 0.001 |
| Derived parameters | ||||
| Iodine concentration difference (μg/cm³) | 4.2 ± 2.8 | 3.4 ± 2.1 | t = 2.152 | 0.032 |
| Arterial phase tumor-muscle iodine concentration ratio | 1.68 ± 0.42 | 1.34 ± 0.38 | t = 5.642 | < 0.001 |
| Portal venous phase tumor-muscle iodine concentration ratio | 2.21 ± 0.56 | 1.79 ± 0.47 | t = 5.339 | < 0.001 |
| Arterial phase tumor-muscle CT value ratio | 1.67 ± 0.38 | 1.36 ± 0.34 | t = 5.718 | < 0.001 |
| Portal venous phase tumor-muscle CT value ratio | 2.22 ± 0.51 | 1.83 ± 0.43 | t = 5.376 | < 0.001 |
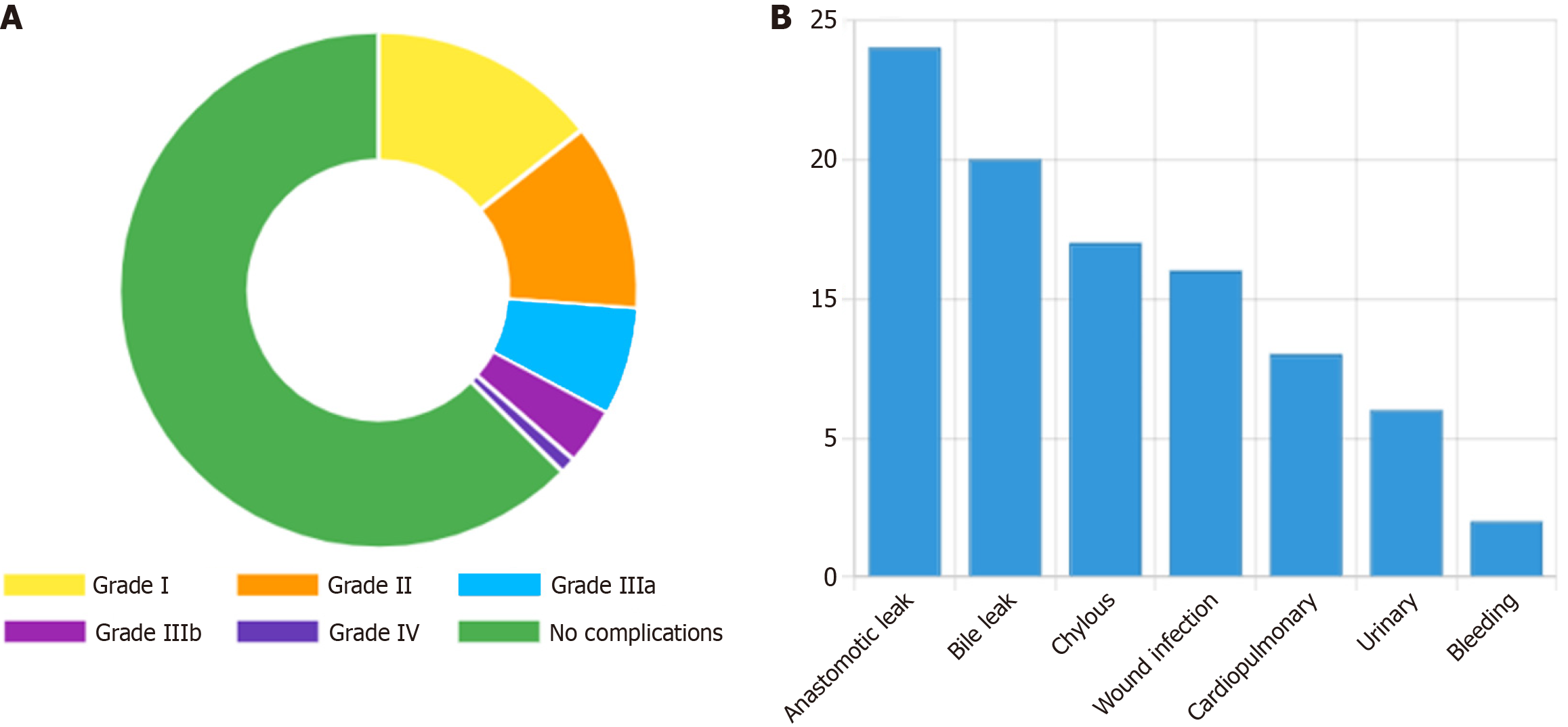
Among 195 patients, 16 developed postoperative complications with an incidence rate of 8.2%. According to the Clavien-Dindo classification system: Grade I 6 cases, Grade II 5 cases, Grade IIIa 3 cases, Grade IIIb 1 case, Grade IV 1 cases, no Grade V complications (death). According to severity classification, minor complications (Grades I-II) 11 cases, major complications (Grades III-V) 5 cases.
Complication type analysis showed: Anastomotic leakage 4 cases, intra-abdominal infection 3 cases, intestinal obstruction 3 cases, incision complications 2 cases, cardiopulmonary complications 2 cases, urinary system complications 1 cases, postoperative bleeding 1 cases. Among them, 2 patients experienced two or more complications simultaneously, classified according to the most severe complication (Figure 1).
Univariate logistic regression analysis results showed that among clinical variables, age, COPD, HGB, WBC, NEU, LYM, ALB, PA, TP, CREA, BUN, AST, CEA, CA19-9, PT, APTT, INR, TNM stage, tumor differentiation degree, tumor diameter, intraoperative blood loss, positive margin, and ASA classification were related to postoperative complications (P < 0.10) (Table 3).
| Variable | β | SE | χ² | P value | OR | 95%CI |
| Age | 0.028 | 0.013 | 4.768 | 0.029 | 1.028 | 1.003-1.054 |
| COPD | 0.822 | 0.483 | 2.897 | 0.089 | 2.271 | 0.880-5.862 |
| Hemoglobin | -0.020 | 0.008 | 6.018 | 0.014 | 0.980 | 0.964-0.996 |
| White blood cell count | 0.164 | 0.073 | 5.005 | 0.025 | 1.178 | 1.021-1.360 |
| Neutrophil count | 0.237 | 0.092 | 6.652 | 0.010 | 1.267 | 1.058-1.518 |
| Lymphocyte count | -0.491 | 0.214 | 5.268 | 0.022 | 0.612 | 0.402-0.932 |
| Albumin | -0.153 | 0.037 | 17.011 | < 0.001 | 0.858 | 0.799-0.922 |
| Prealbumin | -0.009 | 0.003 | 9.572 | 0.002 | 0.991 | 0.986-0.997 |
| Total protein | -0.061 | 0.021 | 8.421 | 0.004 | 0.941 | 0.903-0.981 |
| Creatinine | 0.016 | 0.008 | 3.702 | 0.054 | 1.016 | 1.000-1.033 |
| Blood urea nitrogen | 0.129 | 0.063 | 4.214 | 0.040 | 1.138 | 1.006-1.287 |
| AST | 0.018 | 0.009 | 3.969 | 0.046 | 1.018 | 1.000-1.036 |
| CEA | 0.012 | 0.004 | 7.236 | 0.007 | 1.012 | 1.003-1.021 |
| CA19-9 | 0.006 | 0.003 | 5.614 | 0.018 | 1.006 | 1.001-1.011 |
| PT | 0.326 | 0.123 | 7.019 | 0.008 | 1.385 | 1.086-1.767 |
| APTT | 0.075 | 0.034 | 4.702 | 0.030 | 1.078 | 1.007-1.154 |
| INR | 2.691 | 0.830 | 10.517 | 0.001 | 14.752 | 2.896-75.151 |
| TNM stage | ||||||
| Stage II vs Stage I | 0.141 | 0.429 | 0.108 | 0.743 | 1.153 | 0.493-2.696 |
| Stage III vs Stage I | 1.351 | 0.408 | 10.990 | 0.001 | 3.864 | 1.742-8.575 |
| Tumor differentiation | ||||||
| Moderate vs well differentiated | 0.449 | 0.391 | 1.315 | 0.251 | 1.565 | 0.726-3.373 |
| Poor vs well differentiated | 1.408 | 0.448 | 9.888 | 0.002 | 4.091 | 1.710-9.790 |
| Tumor diameter | 0.221 | 0.067 | 10.879 | 0.001 | 1.248 | 1.094-1.424 |
| Intraoperative blood loss | 0.004 | 0.001 | 17.842 | < 0.001 | 1.004 | 1.002-1.006 |
| Positive margin | 1.082 | 0.434 | 6.201 | 0.013 | 2.950 | 1.264-6.886 |
| ASA grade ≥ III | 0.745 | 0.316 | 5.563 | 0.018 | 2.108 | 1.139-3.900 |
Univariate analysis of spectral CT parameters showed that except for normal bowel wall IC, abdominal aorta IC, erector spinae IC, tumor plain CT value, abdominal aorta CT value, and erector spinae CT value, all other spectral CT parameters were related to postoperative complications (P < 0.05). Among them, PV tumor NIC showed the strongest predictive value (P < 0.001) (Table 4).
| Parameter | β | SE | χ² | P value | OR | 95%CI |
| Arterial phase parameters | ||||||
| Tumor iodine concentration | -0.127 | 0.027 | 22.157 | < 0.001 | 0.881 | 0.837-0.928 |
| Tumor normalized iodine concentration | -35.847 | 8.251 | 18.896 | < 0.001 | < 0.001 | < 0.001-0.018 |
| Tumor effective atomic number | -0.854 | 0.223 | 14.631 | < 0.001 | 0.426 | 0.275-0.661 |
| Tumor λHU | -0.658 | 0.183 | 12.949 | < 0.001 | 0.518 | 0.364-0.738 |
| Tumor CT value | -0.039 | 0.007 | 28.512 | < 0.001 | 0.962 | 0.948-0.976 |
| Tumor 40 keV CT value | -0.024 | 0.004 | 35.721 | < 0.001 | 0.976 | 0.968-0.984 |
| Tumor enhancement degree | -0.064 | 0.011 | 34.129 | < 0.001 | 0.938 | 0.917-0.959 |
| Tumor relative enhancement rate | -0.011 | 0.002 | 29.374 | < 0.001 | 0.989 | 0.985-0.993 |
| Portal venous phase parameters | ||||||
| Tumor iodine concentration | -0.135 | 0.024 | 32.411 | < 0.001 | 0.874 | 0.833-0.917 |
| Tumor normalized iodine concentration | -46.124 | 9.873 | 21.829 | < 0.001 | < 0.001 | < 0.001-0.012 |
| Tumor effective atomic number | -0.763 | 0.214 | 12.683 | < 0.001 | 0.467 | 0.307-0.710 |
| Tumor λHU | -0.616 | 0.163 | 14.344 | < 0.001 | 0.540 | 0.393-0.742 |
| Tumor CT value | -0.038 | 0.007 | 32.014 | < 0.001 | 0.963 | 0.950-0.976 |
| Tumor 40 keV CT value | -0.021 | 0.004 | 30.216 | < 0.001 | 0.979 | 0.972-0.986 |
| Tumor enhancement degree | -0.060 | 0.009 | 42.734 | < 0.001 | 0.942 | 0.925-0.960 |
| Tumor relative enhancement rate | -0.010 | 0.002 | 36.827 | < 0.001 | 0.990 | 0.987-0.993 |
| Derived parameters | ||||||
| Iodine concentration difference | -0.122 | 0.057 | 4.607 | 0.032 | 0.885 | 0.792-0.990 |
| Arterial phase tumor-muscle iodine concentration ratio | -1.294 | 0.283 | 20.824 | < 0.001 | 0.274 | 0.158-0.474 |
| Portal venous phase tumor-muscle iodine concentration ratio | -1.199 | 0.241 | 24.671 | < 0.001 | 0.302 | 0.188-0.485 |
| Arterial phase tumor-muscle CT value ratio | -1.316 | 0.286 | 21.133 | < 0.001 | 0.268 | 0.154-0.466 |
| Portal venous phase tumor-muscle CT value ratio | -1.289 | 0.246 | 27.474 | < 0.001 | 0.276 | 0.171-0.446 |
Univariate analysis screened 22 clinical variables and 19 spectral CT parameters related to postoperative complications (P < 0.10). Among them, PV tumor NIC showed the strongest predictive value.
Variables with P < 0.10 in univariate analysis were included in multivariate logistic regression analysis, using forward stepwise method to establish a predictive model. Multicollinearity testing showed that VIF values for all included variables were < 3.0, indicating no collinearity problems.
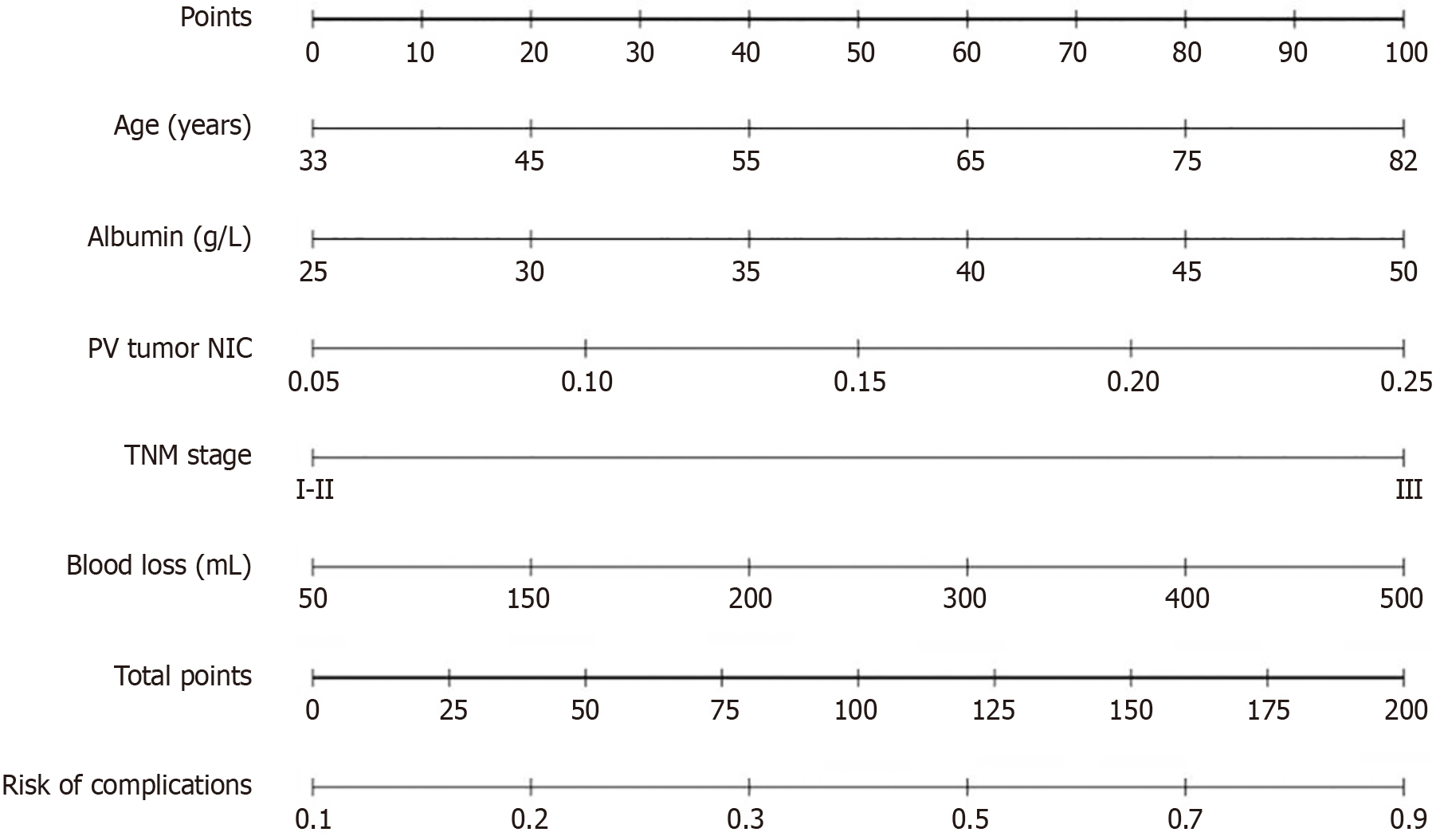
After forward stepwise screening, the final predictive model included 5 independent predictive factors: Age, ALB, PV tumor NIC, TNM stage, and intraoperative blood loss (P < 0.05) (Table 5).
| Predictive factor | β | SE | χ² | P value | OR | 95%CI | VIF |
| Age | 0.041 | 0.018 | 5.184 | 0.023 | 1.042 | 1.006-1.080 | 1.12 |
| Albumin | -0.127 | 0.047 | 7.289 | 0.007 | 0.881 | 0.803-0.967 | 1.34 |
| Portal venous phase tumor normalized iodine concentration | -28.734 | 8.125 | 12.511 | < 0.001 | < 0.001 | < 0.001-0.021 | 1.08 |
| TNM stage (stage III vs stage I) | 1.186 | 0.548 | 4.685 | 0.030 | 3.274 | 1.119-9.582 | 1.23 |
| Intraoperative blood loss | 0.003 | 0.001 | 6.841 | 0.009 | 1.003 | 1.001-1.005 | 1.15 |
| Constant | 1.287 | 1.952 | 0.434 | 0.510 | 3.621 |
Based on multivariate logistic regression results, a nomogram for postoperative complications prediction was established, integrating 5 predictive factors into a visualized predictive tool (Figure 2).
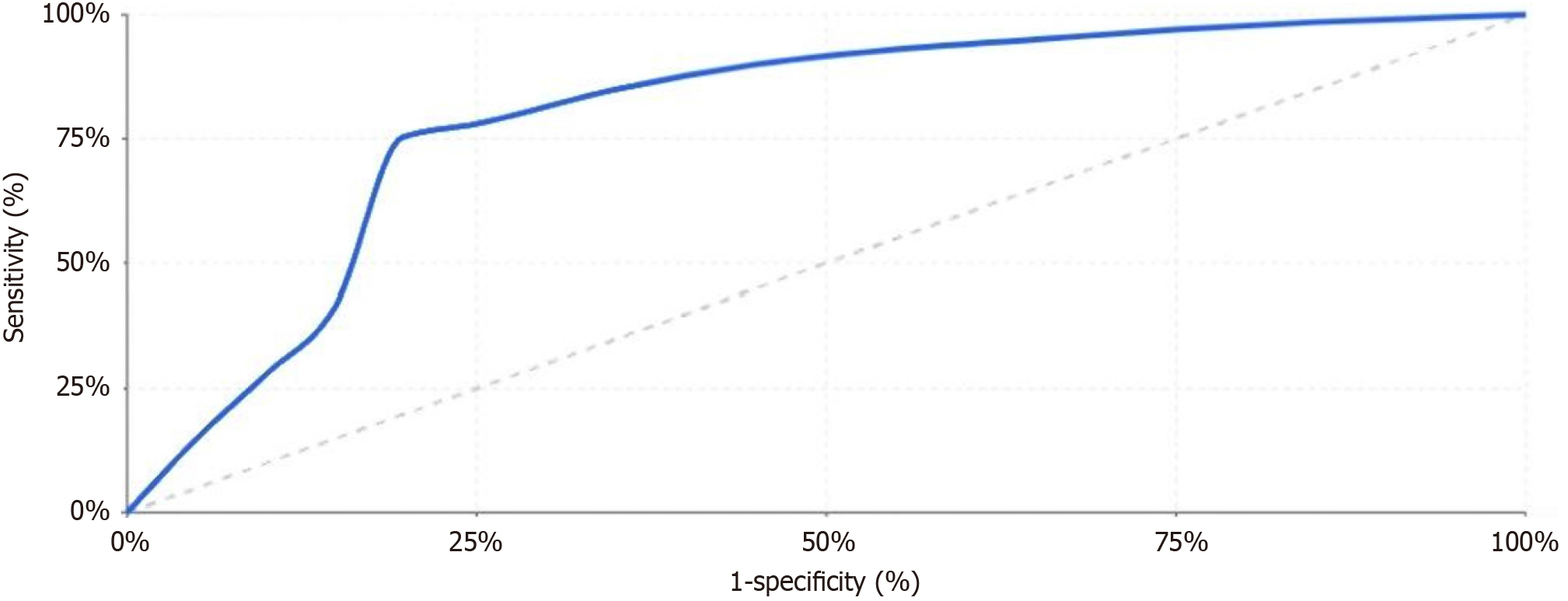
ROC curve analysis showed that the combined predictive model had an AUC of 0.843. When the optimal cutoff value was determined as 0.35, the model’s sensitivity was 75.3%, specificity was 80.3%, positive predictive value was 66.2%, negative predictive value was 86.1%, and accuracy was 78.5% (Figure 3).
Hosmer-Lemeshow goodness-of-fit test results showed χ² = 6.247, P = 0.619, indicating that the model had good calibration with good agreement between predicted and observed probabilities.
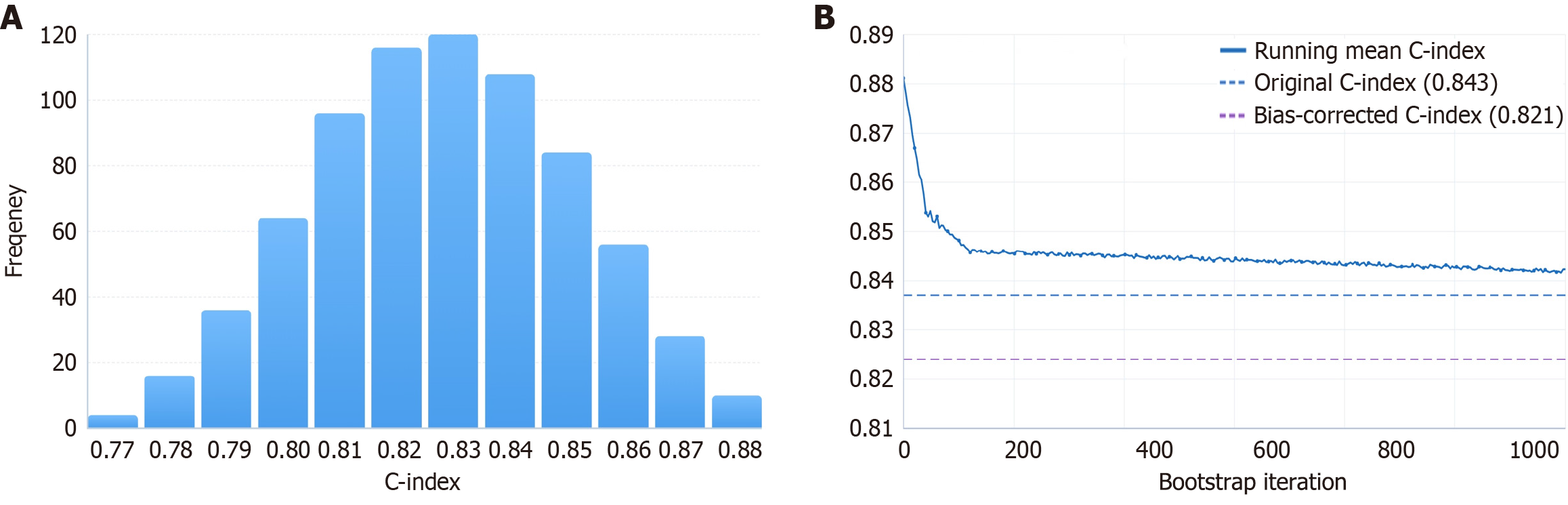
Bootstrap internal validation (1000 resamples) showed a bias-corrected C-index of 0.821, with a difference of 0.022 from the original C-index (0.843), indicating good internal consistency and stability of the model (Figure 4).
Postoperative complications are important factors affecting the recovery and prognosis of colorectal cancer patients. Accurate prediction of postoperative complication risk has important clinical significance for preoperative risk stratification, perioperative management optimization, and individualized treatment strategy formulation. This study analyzed preoperative spectral CT imaging data of 195 colorectal cancer patients and established a multivariate predictive model including age, ALB, PV tumor NIC, TNM stage, and intraoperative blood loss, providing a new imaging method for preoperative prediction of postoperative complications in colorectal cancer.
The postoperative complication rate of 8.2% in this study is basically consistent with domestic and international literature reports of 10%[10]. Among them, minor complications (Clavien-Dindo grades I-II) accounted for 69.9%, and major complications (grades III-V) accounted for 30.1%, with no perioperative deaths, reflecting the improvement in modern surgical techniques and perioperative management levels[11]. Anastomotic leakage, as the most common serious complication, had an incidence rate of 9.7%, slightly lower than the 10%-15% reported in literature, possibly related to improvements in surgical techniques and strengthened preoperative nutritional status assessment[12].
This study found that PV tumor NIC was the most valuable spectral CT parameter for predicting postoperative complications, showing the strongest independent predictive effect in multivariate analysis (P < 0.001). This finding has important pathophysiological significance. IC reflects tissue blood flow perfusion and microvascular density, and NIC, through the ratio to aortic IC, eliminates individual differences and contrast agent dosage effects, more accurately reflecting tumor tissue blood flow perfusion characteristics[13].
In this study, tumor IC in the complication group was significantly lower than in the non-complication group. The relationship between reduced PV NIC and increased postoperative complications operates through multiple interconnected biological pathways[14]. First, decreased iodine uptake directly reflects diminished tumor microvascular density and blood flow perfusion, which creates a hypoxic microenvironment. This chronic hypoxia triggers hypoxia-inducible factor-1α activation, promoting epithelial-mesenchymal transition and enhancing tumor invasiveness, which correlates with more extensive surgical resection and greater tissue trauma. Second, poor tumor perfusion indicates compromised regional blood supply that extends beyond the tumor itself, affecting the surrounding tissues including the anastomotic site. Inadequate oxygen and nutrient delivery to the anastomotic tissue impairs collagen synthesis, fibroblast proliferation, and neovascularization-all critical components of wound healing-thereby increasing anastomotic leakage risk. Third, tumors with low NIC often exhibit increased lactate production and acidic microenvironment, which impairs immune cell function, particularly neutrophil and macrophage activity, compromising the patient's ability to combat infection. Fourth, poor tumor perfusion may serve as a surrogate marker for systemic cardiovascular dysfunction and overall patient frailty, reflecting suboptimal tissue perfusion throughout the body and inadequate physiological reserve to withstand surgical stress. These mechanisms collectively explain why reduced NIC predicts not only anastomotic complications but also diverse postoperative adverse events including infections, cardiopulmonary complications, and delayed recovery[15].
In addition to IC, other spectral CT parameters such as effective atomic number and spectral attenuation curve slope also showed certain predictive value. Effective atomic number reflects tissue atomic composition, and decreased effective atomic number in tumor tissue may be related to changes in tissue structure and protein content[16]. Spectral attenuation curve slope reflects the attenuation difference of tissues to X-rays of different energies and is closely related to tissue density and atomic number[17]. The combined application of these parameters provides new approaches for comprehensive assessment of tumor tissue characteristics.
The other four independent predictive factors identified in this study all have sufficient pathophysiological basis. Age as a predictive factor reflects characteristics of elderly patients such as decreased body reserve function, reduced immune function, and increased comorbidities, increasing the risk of postoperative complications[18]. ALB level reflects patient nutritional status and liver synthesis function; hypoalbuminemia is closely related to postoperative complications and is an important factor affecting wound healing and immune function[19].
TNM stage reflects the degree of tumor invasion and metastasis status. Patients with advanced tumors often require more complex surgical procedures with greater intraoperative trauma, correspondingly increasing the risk of post
The predictive model established in this study had an AUC of 0.843, showing good discriminatory ability, with sensitivity and specificity of 75.3% and 80.3% respectively, having good clinical application value. Compared with previous predictive models based on traditional clinical indicators, this model significantly improved predictive efficacy by introducing spectral CT parameters[22]. Bootstrap internal validation results showed that the model had good stability and reproducibility.
This predictive model can be used for preoperative risk stratification to identify high-risk patients and guide formulation of perioperative management strategies. For patients predicted to be at high risk, preoperative nutritional support, immune function enhancement, comorbidity optimization control, and other interventional measures can be taken, with more refined surgical techniques used intraoperatively and strengthened monitoring and early intervention postoperatively, thereby reducing complication incidence[23].
Spectral CT, as an emerging imaging technology, has advantages that traditional CT cannot match. First, spectral CT can provide material composition information, and through material separation technology can quantitatively analyze tissue IC, water content, and other parameters, providing new biomarkers for disease diagnosis and prognosis asse
Additionally, spectral CT examination is simple and easy to perform, does not increase additional patient burden, and can be obtained during routine preoperative examination, having good clinical operability. Compared with other functional imaging examinations such as magnetic resonance imaging and positron emission tomography-CT, spectral CT has shorter examination time and lower cost, making it more suitable for clinical promotion[26].
This study has some limitations that need to be recognized. First, this is a single-center retrospective study with relatively limited sample size, possibly having selection bias. Second, measurement of spectral CT parameters may be affected by factors such as scanning techniques and reconstruction algorithms, requiring standardized operating procedures to ensure result reproducibility[27]. Third, this study did not include patients receiving preoperative chemotherapy or radiotherapy, limiting the model’s applicability. Fourth, follow-up time was only 30 days postoperatively, unable to assess the relationship between long-term complications and prognosis[28].
Future research should be further improved in the following aspects: First, conduct multicenter, large-sample prospective studies to validate external validity and universality of the predictive model[29]. Second, explore the relationship between spectral CT parameters and molecular biological markers to deeply understand the biological mechanisms of their predictive value[30]. Third, combine artificial intelligence and machine learning techniques to develop more precise predictive algorithms and improve predictive efficacy[31]. Fourth, study individualized inter
In conclusion, this study confirmed the value of preoperative spectral CT multi-parameters in predicting postoperative complications in colorectal cancer. The established combined predictive model has good predictive efficacy, providing new tools for clinical preoperative risk assessment and individualized treatment strategy formulation. PV tumor NIC, as the most valuable imaging predictive factor, reflects the close relationship between tumor blood flow perfusion status and postoperative complication risk, providing new evidence for the application of spectral CT in oncology. With continuous technological improvement and deepening research, spectral CT is expected to play a more important role in the era of precision medicine.
| 1. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 15061] [Article Influence: 7530.5] [Reference Citation Analysis (23)] |
| 2. | Antoniv M, Nikiforchin A, Sell NM, Bordeianou LG, Francone TD, Ahmed F, Rubin MS, Bleday R. Impact of Multi-Institutional Enhanced Recovery after Surgery Protocol Implementation on Elective Colorectal Surgery Outcomes. J Am Coll Surg. 2025;240:158-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 3. | Andras D, Lazar AM, Crețoiu D, Berghea F, Georgescu DE, Grigorean V, Iacoban SR, Mastalier B. Analyzing postoperative complications in colorectal cancer surgery: a systematic review enhanced by artificial intelligence. Front Surg. 2024;11:1452223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 4. | Weber MC, Berlet M, Stoess C, Reischl S, Wilhelm D, Friess H, Neumann PA. A nationwide population-based study on the clinical and economic burden of anastomotic leakage in colorectal surgery. Langenbecks Arch Surg. 2023;408:55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 5. | Hernandez MC, Chen C, Nguyen A, Choong K, Carlin C, Nelson RA, Rossi LA, Seth N, McNeese K, Yuh B, Eftekhari Z, Lai LL. Explainable Machine Learning Model to Preoperatively Predict Postoperative Complications in Inpatients With Cancer Undergoing Major Operations. JCO Clin Cancer Inform. 2024;8:e2300247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 6. | Greffier J, Villani N, Defez D, Dabli D, Si-Mohamed S. Spectral CT imaging: Technical principles of dual-energy CT and multi-energy photon-counting CT. Diagn Interv Imaging. 2023;104:167-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 194] [Reference Citation Analysis (0)] |
| 7. | Deng J, Zhang W, Xu M, Liu X, Ren T, Li S, Sun Q, Xue C, Zhou J. Value of spectral CT parameters in predicting the efficacy of neoadjuvant chemotherapy for gastric cancer. Clin Radiol. 2024;79:51-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 8. | Tan X, Yang X, Hu S, Chen X, Sun Z. Predictive modeling based on tumor spectral CT parameters and clinical features for postoperative complications in patients undergoing colon resection for cancer. Insights Imaging. 2023;14:155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 9. | Oliver CM, Wagstaff D, Bedford J, Moonesinghe SR; Peri-operative Quality Improvement Project delivery team and collaborative. Systematic development and validation of a predictive model for major postoperative complications in the Peri-operative Quality Improvement Project (PQIP) dataset. Anaesthesia. 2024;79:389-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 10. | Martín-Arévalo J, Moro-Valdezate D, García-Botello S, Pérez-Santiago L, Casado-Rodrigo D, Garzón-Hernández LP, Castillejos-Ibáñez F, Sánchez-Lara JS, Martínez-Ciarpaglini C, Pla-Martí V. Seasonal and cyclical variations in short-term postoperative outcomes of colorectal cancer: a time series analysis. Sci Rep. 2025;15:5161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 11. | Kannan V, Ullah N, Geddada S, Ibrahiam A, Munaf Shakir Al-Qassab Z, Ahmed O, Malasevskaia I. Impact of "Enhanced Recovery After Surgery" (ERAS) protocols vs. traditional perioperative care on patient outcomes after colorectal surgery: a systematic review. Patient Saf Surg. 2025;19:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 31] [Reference Citation Analysis (1)] |
| 12. | Tsalikidis C, Mitsala A, Mentonis VI, Romanidis K, Pappas-Gogos G, Tsaroucha AK, Pitiakoudis M. Predictive Factors for Anastomotic Leakage Following Colorectal Cancer Surgery: Where Are We and Where Are We Going? Curr Oncol. 2023;30:3111-3137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 77] [Article Influence: 25.7] [Reference Citation Analysis (0)] |
| 13. | Lennartz S, Kambadakone A. Reply to Letter to the Editor: "Intra‑patient variability of iodine quantification across different dual‑energy CT platforms: assessment of normalization techniques". Eur Radiol. 2024;34:7591-7592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 14. | Alilou M, Khorrami M, Prasanna P, Bera K, Gupta A, Viswanathan VS, Patil P, Velu PD, Fu P, Velcheti V, Madabhushi A. A tumor vasculature-based imaging biomarker for predicting response and survival in patients with lung cancer treated with checkpoint inhibitors. Sci Adv. 2022;8:eabq4609. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 15. | Ciepła J, Smolarczyk R. Tumor hypoxia unveiled: insights into microenvironment, detection tools and emerging therapies. Clin Exp Med. 2024;24:235. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 37] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 16. | Qin T, Wang M, Fan Y, Wang J, Gao Z, Wang F, Li R, Li K, Ruan C, Liang B. Multivendor comparison of quantification accuracy of effective atomic number by Dual-Energy CT: A phantom study. Eur J Radiol. 2024;180:111690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 17. | Ono K, Asada Y. Analysis of material composition and attenuation characteristics of anthropomorphic torso phantoms for dosimetry using dual energy CT technology. Phys Eng Sci Med. 2025;48:675-683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 18. | Bryant EA, Tulebaev S, Castillo-Angeles M, Moberg E, Senglaub SS, O'Mara L, McDonald M, Salim A, Cooper Z. Frailty Identification and Care Pathway: An Interdisciplinary Approach to Care for Older Trauma Patients. J Am Coll Surg. 2019;228:852-859.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 84] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 19. | Milaniuk A, Drabko K, Chojęta A. Role of albumin and prealbumin in assessing nutritional status and predicting increased risk of infectious complications during childhood cancer treatment. Acta Biochim Pol. 2024;71:13693. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 20. | Liu X, Chen XL, Yuan Y, Pu H, Li H. Dual-energy CT quantitative parameters for prediction of prognosis in patients with resectable rectal cancer. Eur Radiol. 2025;35:4945-4956. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 21. | Shi X, Cui Y, Wang S, Pan Y, Wang B, Lei M. Development and validation of a web-based artificial intelligence prediction model to assess massive intraoperative blood loss for metastatic spinal disease using machine learning techniques. Spine J. 2024;24:146-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 37] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 22. | Wei L, Wu Y, Bo J, Fu B, Sun M, Zhang Y, Xiong B, Dong J. Dual-Energy Computed Tomography Parameters Combined With Inflammatory Indicators Predict Cervical Lymph Node Metastasis in Papillary Thyroid Cancer. Cancer Control. 2024;31:10732748241262177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 23. | Bhave NM, Cibotti-Sun M, Moore MM. 2024 Perioperative Cardiovascular Management for Noncardiac Surgery Guideline-at-a-Glance. J Am Coll Cardiol. 2024;84:1970-1975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 24. | Narita K, Nakamura Y, Higaki T, Kondo S, Honda Y, Kawashita I, Mitani H, Fukumoto W, Tani C, Chosa K, Tatsugami F, Awai K. Iodine maps derived from sparse-view kV-switching dual-energy CT equipped with a deep learning reconstruction for diagnosis of hepatocellular carcinoma. Sci Rep. 2023;13:3603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 25. | Lund CM, Vistisen KK, Dehlendorff C, Rønholt F, Johansen JS, Nielsen DL. The effect of geriatric intervention in frail elderly patients receiving chemotherapy for colorectal cancer: a randomized trial (GERICO). BMC Cancer. 2017;17:448. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 32] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 26. | Borges AP, Antunes C, Curvo-Semedo L. Pros and Cons of Dual-Energy CT Systems: "One Does Not Fit All". Tomography. 2023;9:195-216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 58] [Reference Citation Analysis (0)] |
| 27. | Rizzo BM, Sidky EY, Schmidt TG. Dual energy CT reconstruction using the constrained one step spectral image reconstruction algorithm. Med Phys. 2024;51:2648-2664. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 28. | Downey CL, Bainbridge J, Jayne DG, Meads DM. Impact of in-hospital postoperative complications on quality of life up to 12 months after major abdominal surgery. Br J Surg. 2023;110:1206-1212. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 34] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 29. | Collins GS, Dhiman P, Ma J, Schlussel MM, Archer L, Van Calster B, Harrell FE Jr, Martin GP, Moons KGM, van Smeden M, Sperrin M, Bullock GS, Riley RD. Evaluation of clinical prediction models (part 1): from development to external validation. BMJ. 2024;384:e074819. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 435] [Cited by in RCA: 357] [Article Influence: 178.5] [Reference Citation Analysis (5)] |
| 30. | Moghiseh M, Searle E, Dixit D, Kim J, Dong YC, Cormode DP, Butler A, Gieseg SP, Mars Bioimaging Ltd. Spectral Photon-Counting CT Imaging of Gold Nanoparticle Labelled Monocytes for Detection of Atherosclerosis: A Preclinical Study. Diagnostics (Basel). 2023;13:499. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 31. | Bousse A, Kandarpa VSS, Rit S, Perelli A, Li M, Wang G, Zhou J, Wang G. Systematic Review on Learning-based Spectral CT. IEEE Trans Radiat Plasma Med Sci. 2024;8:113-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 32. | Desai RJ, Glynn RJ, Solomon SD, Claggett B, Wang SV, Vaduganathan M. Individualized Treatment Effect Prediction with Machine Learning - Salient Considerations. NEJM Evid. 2024;3:EVIDoa2300041. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 24] [Reference Citation Analysis (0)] |