Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.115146
Revised: January 6, 2026
Accepted: February 5, 2026
Published online: April 15, 2026
Processing time: 181 Days and 13.9 Hours
The incidence and mortality of gastric cancer remain among the highest of all malignant tumors, and there is an urgent need for both standardized and indi
To develop a gastric cancer CDSS grounded in widely adopted international guidelines and to evaluate its clinical applicability.
This retrospective study included 156 patients with gastric cancer who were treated at Shenzhen Luohu People’s Hospital between January 1, 2015 and De
The overall concordance rate among the 156 patients was 90.4%. Concordance was 100% for patients with stage 0, I, and II; 89.8% for stage III; and 83.6% for stage IV. When comparing early-stage to mid-stage (0-III) with late-stage (IV) gastric can
CDSS demonstrates significant advantages in managing early-stage gastric cancer through standardized treatment approaches. However, improving concordance in complex and personalized cases remains a critical challenge.
Core Tip: The clinical decision support system (CDSS) is a cognitive computing platform designed to simulate clinician decision-making processes. Based on widely used international guidelines for the treatment of gastric cancer, a CDSS specifically for gastric cancer was developed. A single-center retrospective study was conducted at Shenzhen Luohu People’s Hospital. The results indicate that the CDSS demonstrates high concordance with multidisciplinary team recommendations in patients undergoing early-stage and standardized treatments. However, challenges remain in managing complex and personalized cases of gastric cancer, which will be a key focus for future system optimization.
- Citation: Chen TL, Yu XQ. Value of gastric cancer clinical decision support system in a single-center clinical application. World J Gastrointest Oncol 2026; 18(4): 115146
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/115146.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.115146
Gastric cancer ranks fifth in incidence and third in mortality in China, posing a significant threat to public health. With advancements in modern medicine, treatment options have expanded substantially, encompassing surgery, chemothe
Ping An Technology (Shenzhen) Co., Ltd., in collaboration with Shenzhen Luohu People’s Hospital, developed a CDSS specifically for gastric cancer. The system incorporates multiple authoritative guidelines, including those from the Chinese Society of Clinical Oncology, the Japanese Gastric Cancer Association, the European Society for Medical On
The gastric cancer CDSS was developed under the Shenzhen Collaborative Innovation Technology Plan through an international collaboration among Shenzhen Luohu People’s Hospital, Ping An Technology, and the National University of Singapore. The system integrates multi-source data, knowledge graphs, intelligent algorithms, and iterative validation. Electronic medical records from Shenzhen Luohu People’s Hospital – including information on staging, genetic testing, and treatment history – were systematically collected. Multiple international clinical guidelines, including those from the Japanese Gastric Cancer Association, the European Society for Medical Oncology, the National Comprehensive Cancer Network, and Chinese standards, were harmonized to construct a structured disease database.
Using natural language processing, key medical entities such as diseases, drugs, and surgical procedures were ex
Following prototype development at Shenzhen Luohu People’s Hospital, the system is scheduled for parameter optimization and validation across multiple hospitals to produce a clinically deployable version.
Patients with gastric cancer who were hospitalized at Shenzhen Luohu People’s Hospital between January 1, 2015, and December 31, 2024, were retrospectively included. The study received approval from the Ethics Committee of Shenzhen Luohu People’s Hospital on July 10, 2025.
Inclusion criteria: (1) Pathologically confirmed diagnosis of gastric cancer; (2) No concurrent malignant tumors origi
Exclusion criteria: (1) Diagnosis of two or more concurrent malignant tumors; (2) Enrollment in clinical trials during the study period; (3) Diagnosis of gastrointestinal stromal tumors; and (4) Poor compliance with treatment or follow-up re
The included patients with gastric cancer were classified into three groups: (1) New cases; (2) Previous cases; and (3) Recurrent cases. New cases were defined as patients newly diagnosed with gastric cancer who had not received treatment within the past year. Previous cases referred to patients who had been diagnosed with gastric cancer for more than one year or had already undergone treatment. Recurrent cases included patients who had previously received treatment and achieved clinical remission but subsequently experienced a recurrence of symptoms or confirmed re-diagnosis of gastric cancer.
The required clinical data for each patient was entered into the system, which subsequently generated one to three guideline-based treatment recommendations and recorded them for further analysis.
The CDSS-recommended treatment scheme was compared with the clinician-determined treatment plan. Concordance was defined as instances in which both treatment recommendations were identical. The concordance rate was calculated using the following formula: Concordance rate = consistent number of patients/total number of patients × 100%.
Data were analyzed using SPSS version 25. All statistical tests were two-sided, and a value of P < 0.05 was considered statistically significant. Patient characteristics were summarized descriptively. Categorical variables were expressed as percentages and compared using the χ2 test or Fisher’s exact test, as appropriate.
A total of 156 patients with gastric cancer were included in the study, comprising 110 newly diagnosed cases, 28 previously treated cases, and 18 cases of recurrence. The cohort consisted of 94 males and 62 females, aged 32-93 years, with a mean age of 61.76 years. Treatment modalities included surgery (82 patients), neoadjuvant chemotherapy (7 patients), palliative systemic therapy (45 patients), adjuvant therapy (11 patients), and supportive care (11 patients). According to the tumor, node, metastasis classification of gastric cancer, 1 patient was classified as stage 0, 24 as stage I, 21 as stage II, 49 as stage III, and 61 as stage IV (Table 1).
| Clinical features | |
| Gender | |
| Male | 94 (60.3) |
| Female | 62 (39.7) |
| Age (years) | |
| < 40 | 6 (3.8) |
| 40-50 | 24 (15.4) |
| 50-60 | 38 (24.4) |
| 60-70 | 45 (28.8) |
| 70-80 | 31 (19.9) |
| ≥ 80 | 12 (7.7) |
| Category of disease severity | |
| New cases | 110 (70.5) |
| Previous cases | 28 (17.9) |
| Recurrent cases | 18 (11.5) |
| Treatment | |
| Surgery | 82 (52.6) |
| Neoadjuvant chemotherapy | 7 (4.5) |
| Adjuvant therapy | 11 (7.1) |
| Palliative systemic therapy | 45 (28.8) |
| Supportive treatment | 11 (7.1) |
| Tumor, node, metastasis classification | |
| Stage 0 | 1 (0.6) |
| Stage I | 24 (15.4) |
| Stage II | 21 (13.5) |
| Stage III | 49 (31.4) |
| Stage IV | 61 (39.1) |
| Total | 156 |
Statistical analysis indicated that among the 156 patients with gastric cancer, 141 received treatment plans consistent with the CDSS recommendations, resulting in an overall concordance rate of 90.4%.
According to the type of case classification: The concordance rate was highest among new cases, exceeding 95%. Due to the greater complexity of treatment histories, concordance rates were significantly lower in previous and recurrent cases, with the previous case group exhibiting the lowest concordance rate (75.0%). Statistical analysis indicated that the differences in concordance rates among the three case types were statistically significant (P = 0.002 < 0.05; Table 2).
| Group | Consistent | Inconsistent | Concordance rate (%) |
| Category of disease severity | |||
| New cases | 105 | 5 | 95.5 |
| Previous case | 21 | 7 | 75.0 |
| Recurrent cases | 15 | 3 | 83.3 |
| Tumor, node, metastasis classification | |||
| Stage 0 | 1 | 0 | 100 |
| Stage I | 24 | 0 | 100 |
| Stage II | 21 | 0 | 100 |
| Stage III | 44 | 5 | 89.8 |
| Stage IV | 51 | 10 | 83.6 |
| Treatment | |||
| Surgery | 80 | 2 | 97.6 |
| Neoadjuvant chemotherapy | 7 | 0 | 100 |
| Adjuvant therapy | 8 | 3 | 72.7 |
| Palliative systemic therapy | 35 | 10 | 77.8 |
| Supportive treatment | 11 | 0 | 100 |
| Total | 141 | 15 | 90.4 |
According to tumor, node, metastasis staging division: Among the included patients, there was 1 case of stage 0, 24 cases of stage I, 21 cases of stage II, 49 cases of stage III, and 61 cases of stage IV gastric cancer. Across the new, previous, and recurrent cases, stage III and stage IV patients represented the largest proportions, indicating that gastric cancer in China is frequently diagnosed at an advanced stage.
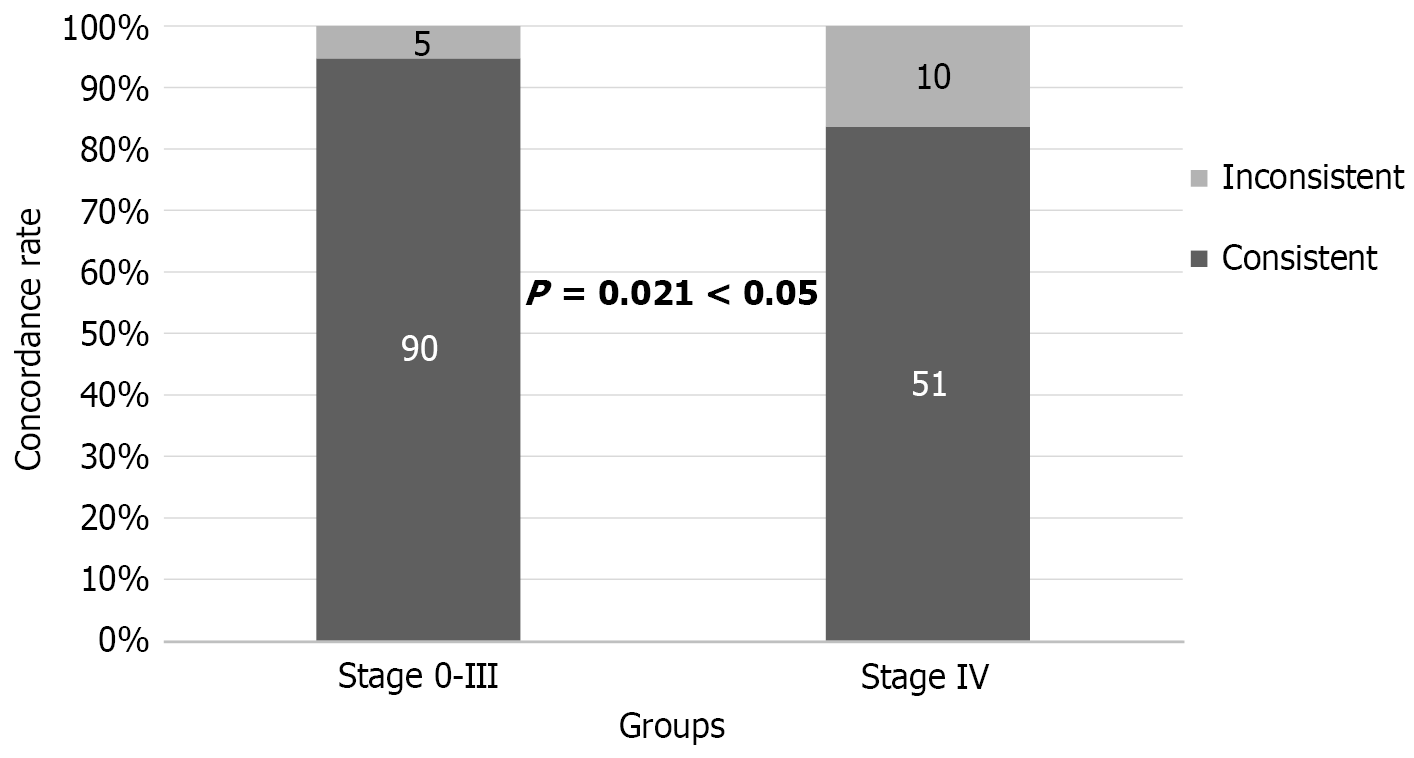
The concordance rate for patients with stage 0-II disease was 100%. Among patients with stage III disease, 44 of 49 were concordant (89.8%), and among those with stage IV disease, 51 of 61 were concordant (83.6%). When comparing early-stage to mid-stage (0-III) and late-stage (IV) disease, the concordance rate was 94.7% for stage 0-III and 83.6% for stage IV, with a statistically significant difference (χ2 = 5.295, P = 0.021; Figure 1).
The lower concordance observed in advanced gastric cancer likely reflects the increased clinical complexity of late-stage disease, as well as variability introduced by physician and patient preferences, which may extend beyond guideline-based recommendations.
Stratified by the treatment modality: Among the included patients, 82 underwent surgical treatment, of whom 80 received treatment plans consistent with the system recommendation (97.6%). All seven patients who received neoadjuvant chemotherapy were concordant (100%). Among the 45 patients who underwent palliative systemic therapy, 35 were concordant (77.8%). Of the 11 patients receiving adjuvant therapy, 8 were concordant (72.7%). All 11 patients who received supportive care were concordant (100%). Fisher’s exact test indicated that concordance rates differed significantly across treatment regimens (P = 0.001). However, due to the small sample size of patients receiving neo
In the era of rapid scientific and technological advancement, AI has become increasingly integrated into medicine. By leveraging machine learning and deep learning models, AI-based CDSS simulates clinician decision-making processes, offering valuable support for patients with gastric cancer. Existing CDSS for gastric cancer have demonstrated clinical utility in diagnostic decision-making, treatment planning, and predictive analysis. In clinical diagnosis, AI has the potential to enhance both detection rates and diagnostic efficiency, thereby reducing the incidence of missed diagnoses. For instance, the deep learning-based CDSS developed by Eun Jeong Gong for endoscopic diagnosis of gastric cancer achieved an internal test detection rate exceeding 95%[3]. Similarly, GastroMIL, a diagnostic model for gastric adenocarcinoma, exhibited diagnostic performance comparable to that of expert pathologists[4]. Watson for Oncology (WFO), developed by International Business Machines Corporation and trained at Memorial Sloan Kettering Cancer Center, has been applied to several types of cancer, including gastric, breast, lung, and colorectal cancers[5]. However, the clinical application of such systems remains constrained by factors including drug availability, variability in treatment options, physician and patient preferences, economic conditions, cultural influences, and medical insurance reimbursement policies[6-8]. Xu[9] compared WFO with MDT treatment plans for 30 patients with primary gastric cancer at the Second Hospital of Shaoxing, Zhejiang Province, and reported a concordance rate of only 53.3%, primarily due to differences in clinical guidelines. In contrast, the team at Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, developed a gastric cancer CDSS based on Chinese clinical guidelines, achieving concordance rates exceeding 90%, thereby highlighting its strong clinical utility[10]. In predictive applications, AI also supports prognosis and treatment opti
The gastric cancer CDSS developed in this study provides treatment recommendations tailored to patients with gastric cancer, based on multiple clinical guidelines. Unlike systems that rely on a single guideline, this multi-guideline approach not only compensates for the limitations of individual guidelines but also offers transparent explanations of the sources underlying treatment decisions, thereby presenting patients with a broader range of therapeutic options. By incorpo
The gastric cancer CDSS achieved an overall concordance rate of 90.4%, significantly surpassing single-guideline systems such as WFO (53.3%), thereby highlighting the advantage of multi-guideline integration. Concordance reached 100% among early-stage patients, demonstrating the system's reliability in supporting standardized treatment, particularly in primary care settings. In resource-limited regions, the CDSS may help mitigate disparities in healthcare access and quality[17]. Additionally, the Ping An Technology team’s repeated successes in international competitions (e.g., MEDIQA, COIN), along with its expertise in natural language processing and knowledge graphs, have provided a robust technical foundation for the system’s accuracy.
Among the 156 patients with gastric cancer, the overall concordance rate was 90.4%, indicating strong clinical utility, particularly in early-stage cases. However, subgroup analysis revealed that concordance rates in the previous and recurrent case groups were significantly lower than those in the new case group. Similarly, concordance rates in the palliative systemic treatment and adjuvant therapy groups were notably lower than the overall rate. These subgroups primarily consisted of patients with locally advanced or advanced diseases. Compared to early-stage patients, these individuals often present with more complex tumor characteristics and physical conditions. Additionally, patients and their families may experience a more negative psychological state and be burdened by financial constraints, reimbur
The treatment regimens received by 15 patients were not consistent with those recommended by the CDSS. All 15 patients were classified as having stage III or IV disease, reflecting the greater clinical complexity associated with advanced gastric cancer. In-depth analysis revealed that most discrepancies stemmed from physician or patient pre
One illustrative case involved a 66-year-old male patient diagnosed with moderately differentiated gastric adenocarcinoma with multiple bone marrow metastases. Routine blood tests revealed bone marrow suppression, with a platelet count below 50 × 109/L. The CDSS recommended symptomatic supportive care. However, the treating physician attri
Following a comprehensive assessment of bleeding risk and under close monitoring, the clinician elected to initiate oral chemotherapy with tegafur in combination with sintilimab immunotherapy. In another case, a 73-year-old male patient was diagnosed with gastric cancer with peritoneal metastasis. Given the patient’s poor general condition, the CDSS did not recommend chemotherapy and instead suggested symptomatic supportive care. However, considering the presence of peritoneal metastasis and malignant ascites, the clinician determined that local intraperitoneal therapy might help control ascites and improve the patient’s clinical status. As a result, intraperitoneal perfusion with cisplatin and bevacizumab was administered.
Both cases involved advanced gastric cancer with metastatic disease and poor overall physical condition. These complex clinical scenarios revealed limitations of the CDSS, highlighting the need for further optimization to accommo
Two patients declined surgical treatment at the request of their families, who considered advanced age a significant risk factor. In accordance with the patients’ wishes, palliative chemotherapy was ultimately adopted to prolong survival. Additionally, two patients underwent comprehensive preoperative evaluations and were initially deemed eligible for surgery, as no contraindications were identified. However, intraoperative exploration revealed extensive lymph node enlargement, fused into a mass, firmly adherent to adjacent blood vessels, rendering complete resection infeasible. Following intraoperative consultation with family members, the surgical plan was revised to palliative resection, followed by postoperative palliative chemotherapy.
Another case involved a female patient with gastric cancer and peritoneal metastasis who had previously received multimodal therapy. Her most recent regimen included oral tegafur in combination with toripalimab. Upon re-evaluation with gastroscopy and contrast-enhanced computed tomography, her disease status was assessed as stable. The CDSS recommended continuation of the current anti-tumor regimen. However, the treating physician observed recurrent thrombocytopenia associated with tegafur chemotherapy. After discussion with the patient and her family, the treatment plan was adjusted to abraxane chemotherapy in combination with toripalimab immunotherapy. These cases illustrate that patient preferences, intraoperative findings, and treatment-related adverse effects often necessitate clinical decisions that diverge from CDSS recommendations. Such scenarios highlight the importance of incorporating individualized factors and dynamic clinical judgment into future system optimization.
For patients with advanced gastric cancer, particularly those with a Karnofsky Performance Status score of 40-60, the system demonstrates limitations in thoroughly assessing treatment tolerance and balancing the potential benefits and risks of anti-tumor therapy. The CDSS lacks the capacity to dynamically interpret auxiliary examination results or monitor changes in patients’ physical conditions. Moreover, it is unable to integrate the values and preferences of patients and their families into the decision-making process. In contrast, MDT-based treatment planning enables the integration of perspectives from multiple clinical specialties, consideration of disease progression, and alignment with patient and family expectations. Therefore, AI-based CDSS should be regarded as an auxiliary tool that supports, rather than replaces, clinician judgment. Furthermore, the current system requires physicians to manually enter patient information, which reduces ease of use and limits clinical efficiency.
The results indicate that the CDSS demonstrates excellent performance in managing relatively early-stage gastric cancer, a pattern similar to that observed with WFO in gastric cancer management. In a meta-analysis of WFO applications across various cancer types – including breast, lung, colon, rectal, colorectal, and gastric cancers – the concordance for stage I-III tumors, except for lung cancer, was consistently higher than for stage IV disease[15]. However, CDSS is limited in its ability to provide comprehensive, individualized decision-making for patients with advanced and complex diseases. Therefore, while a high overall concordance rate is encouraging, it should not be overemphasized. Instead, valuable insights should be drawn from the cases in which treatment decisions diverged from CDSS recommendations.
Several limitations should be acknowledged in this study. First, the sample sizes for specific subgroups were small. For example, only one case of stage 0, seven cases of gastric cancer receiving neoadjuvant chemotherapy were included, and the corresponding statistical results should therefore be interpreted with caution. Second, although the study demon
Third, this was a single-center, retrospective study, which introduces inherent limitations regarding the representativeness of the patient population and the generalizability of the diagnostic and therapeutic approaches. For instance, treatment patterns may vary across institutions depending on institutional expertise and preferred protocols, potentially influencing the number and types of patients included. Therefore, the applicability of the findings requires further validation in a multi-center setting. Additionally, the retrospective nature of the study may introduce risks of incomplete medical records and selection bias that cannot be entirely avoided.
First, the treatment guidelines utilized by WFO are outdated, limiting their clinical practicality. This highlights the importance of ensuring timely updates to treatment guidelines in order to enhance the clinical applicability of AI-based decision support systems. Second, the system demonstrated low concordance in MDT discussions for relatively advanced-stage patients. These “inconsistent” cases provide valuable insights and highlight areas for system improvement. Future iterations of the system should incorporate tools for patient status assessment and symptom scoring and leverage real-world treatment experiences through deep learning to enhance the comprehensive evaluation of complex cases. In this study, some patients did not follow the optimal treatment recommendations proposed by clinicians, primarily due to the preferences of the patients and their families. To address this, future system development should include dedicated modules for patient and family input, enabling the incorporation of their values and pre
Third, this was a single-center, retrospective study, and the small sample sizes in certain subgroups pose notable limitations. In the future, prospective studies will be conducted at our institution to improve data quality and standardize the evaluation process. In addition, we have established cooperative agreements with several tertiary hospitals in Shenzhen to facilitate data collection from other institutions, thereby expanding the scope from a single-center to a multi-center study. The limited sample sizes in the neoadjuvant chemotherapy subgroup and among patients with stage 0 gastric cancer in this study contribute to the instability of the statistical results. Among the collaborating institutions, the Department of Gastroenterology at Shenzhen Hospital of Southern Medical University specializes in the detection of early-stage gastric cancer and Endoscopic Submucosal Dissection, which will help increase the sample size of patients with stage 0 disease. The Department of Oncology at Peking University Shenzhen Hospital, a leading center in cancer treatment, will support the expansion of sample sizes for patients receiving neoadjuvant chemotherapy, adjuvant treatment and those undergoing palliative systemic therapy.
Finally, in cases where CDSS recommendations differ from MDT decisions due to dynamic changes in a patient’s clinical condition, system optimization can be achieved by integrating a dynamic learning module with hospital information systems. This would enable real-time acquisition of laboratory results and imaging data, allowing the system to automatically update patient profiles and dynamically adjust treatment recommendations accordingly.
The CDSS demonstrated excellent performance in the management of early-stage gastric cancer and in supporting standardized treatment approaches, highlighting its potential as an effective tool to enhance the consistency and quality of diagnosis and therapy. This study provides valuable single-center validation data supporting the clinical translation of AI in oncology. Its broader application in multi-center settings holds significant promise for future development. However, the more important finding of this study is that the system cannot currently deliver dynamic, comprehensive, and personalized medical decisions for patients with complex presentations of gastric cancer. Therefore, future opti
We sincerely thank Yuan Ni for supporting this work.
| 1. | Tian Y, Liu X, Wang Z, Cao S, Liu Z, Ji Q, Li Z, Sun Y, Zhou X, Wang D, Zhou Y. Concordance Between Watson for Oncology and a Multidisciplinary Clinical Decision-Making Team for Gastric Cancer and the Prognostic Implications: Retrospective Study. J Med Internet Res. 2020;22:e14122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 2. | Chen J, Lu C, Huang H, Zhu D, Yang Q, Liu J, Huang Y, Deng A, Han X. Cognitive Computing-Based CDSS in Medical Practice. Health Data Sci. 2021;2021:9819851. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 3. | Gong EJ, Bang CS, Lee JJ, Baik GH, Lim H, Jeong JH, Choi SW, Cho J, Kim DY, Lee KB, Shin SI, Sigmund D, Moon BI, Park SC, Lee SH, Bang KB, Son DS. Deep learning-based clinical decision support system for gastric neoplasms in real-time endoscopy: development and validation study. Endoscopy. 2023;55:701-708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 46] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 4. | Huang B, Tian S, Zhan N, Ma J, Huang Z, Zhang C, Zhang H, Ming F, Liao F, Ji M, Zhang J, Liu Y, He P, Deng B, Hu J, Dong W. Accurate diagnosis and prognosis prediction of gastric cancer using deep learning on digital pathological images: A retrospective multicentre study. EBioMedicine. 2021;73:103631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 89] [Article Influence: 17.8] [Reference Citation Analysis (1)] |
| 5. | Zhou N, Zhang CT, Lv HY, Hao CX, Li TJ, Zhu JJ, Zhu H, Jiang M, Liu KW, Hou HL, Liu D, Li AQ, Zhang GQ, Tian ZB, Zhang XC. Concordance Study Between IBM Watson for Oncology and Clinical Practice for Patients with Cancer in China. Oncologist. 2019;24:812-819. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 151] [Cited by in RCA: 85] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 6. | Wang L, Chen X, Zhang L, Li L, Huang Y, Sun Y, Yuan X. Artificial intelligence in clinical decision support systems for oncology. Int J Med Sci. 2023;20:79-86. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 51] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 7. | Liang J, Li T, Zhang S, Chen C, Vanhouten C, Preininger A, Dankwa-Mullan I, Jackson GP. Reasons for discordance in treatment approaches between oncology practice and clinical decision support in China. J Clin Oncol. 2019;37:6555-6555. [DOI] [Full Text] |
| 8. | Suwanvecho S, Suwanrusme H, Jirakulaporn T, Issarachai S, Taechakraichana N, Lungchukiet P, Decha W, Boonpakdee W, Thanakarn N, Wongrattananon P, Preininger AM, Solomon M, Wang S, Hekmat R, Dankwa-Mullan I, Shortliffe E, Patel VL, Arriaga Y, Jackson GP, Kiatikajornthada N. Comparison of an oncology clinical decision-support system's recommendations with actual treatment decisions. J Am Med Inform Assoc. 2021;28:832-838. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 9. | Xu WX. [Application of Watson for Oncology in Comprehensive Treatment Decision of Gastric Cancer]. Zhejiang Shiyong Yixue. 2020;25:183-185. [DOI] [Full Text] |
| 10. | Li S, Li Z, Xue K, Zhou X, Ding C, Shao Y, Zhang S, Ruan T, Zheng M, Sun J. GC-CDSS: Personalized gastric cancer treatment recommendations system based on knowledge graph. Int J Med Inform. 2024;185:105402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 11. | Mahmoodi SA, Mirzaie K, Mahmoodi MS, Mahmoudi SM. A Medical Decision Support System to Assess Risk Factors for Gastric Cancer Based on Fuzzy Cognitive Map. Comput Math Methods Med. 2020;2020:1016284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 12. | Feng QX, Liu C, Qi L, Sun SW, Song Y, Yang G, Zhang YD, Liu XS. An Intelligent Clinical Decision Support System for Preoperative Prediction of Lymph Node Metastasis in Gastric Cancer. J Am Coll Radiol. 2019;16:952-960. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 47] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 13. | Hu C, Chen W, Li F, Zhang Y, Yu P, Yang L, Huang L, Sun J, Chen S, Shi C, Sun Y, Ye Z, Yuan L, Chen J, Wei Q, Xu J, Xu H, Tong Y, Bao Z, Huang C, Li Y, Du Y, Xu Z, Cheng X. Deep learning radio-clinical signatures for predicting neoadjuvant chemotherapy response and prognosis from pretreatment CT images of locally advanced gastric cancer patients. Int J Surg. 2023;109:1980-1992. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 31] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 14. | Zhang S, Xu H, Li W, Cui J, Zhao Q, Guo Z, Chen J, Yao Q, Li S, He Y, Qiao Q, Feng Y, Shi H, Song C. Development and validation of an inflammatory biomarkers model to predict gastric cancer prognosis: a multi-center cohort study in China. BMC Cancer. 2024;24:711. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 15. | Zhou J, Zeng Z, Li L. A meta-analysis of Watson for Oncology in clinical application. Sci Rep. 2021;11:5792. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 72] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 16. | Liu C, Liu X, Wu F, Xie M, Feng Y, Hu C. Using Artificial Intelligence (Watson for Oncology) for Treatment Recommendations Amongst Chinese Patients with Lung Cancer: Feasibility Study. J Med Internet Res. 2018;20:e11087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 83] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 17. | Emani S, Rui A, Rocha HAL, Rizvi RF, Juaçaba SF, Jackson GP, Bates DW. Physicians' Perceptions of and Satisfaction With Artificial Intelligence in Cancer Treatment: A Clinical Decision Support System Experience and Implications for Low-Middle-Income Countries. JMIR Cancer. 2022;8:e31461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |