Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.114379
Revised: October 24, 2025
Accepted: January 22, 2026
Published online: March 15, 2026
Processing time: 176 Days and 6.4 Hours
Anti-EGFR therapy markedly improves outcomes in advanced colorectal cancer (CRC) patients. Nonetheless, resistance to this treatment is commonly observed even in patients with wild-type RAS tumors.
In this study, three patients with wild-type RAS metastatic CRC with anti-EGFR resistance were comprehensively assessed for genetic alterations (KRAS, NRAS, BRAF, and PIK3CA mutations; HER2 expression) in primary and metastatic lesions. Among the subjects, two exhibited acquired resistance to anti-EGFR therapy following an initial treatment response. One of these patients developed a KRAS mutation in metastatic lesions, while the other had HER2 overexpression in the primary tumor. The third patient demonstrated primary resistance, attributed to a preexisting KRAS mutation in a metastatic lesion that was not present in the primary tumor.
These cases underscore a critical need to understand the diverse mechanisms of resistance to anti-EGFR treatment in patients with wild-type RAS metastatic CRC. Despite the limited sample size, comprehensive genetic profiling of both primary and, when feasible, metastatic lesions is recommended prior to therapy initiation. Furthermore, HER2-targeted therapy could be beneficial for patients with HER2 amplification/overexpression. A multidisciplinary approach and ongoing molecular monitoring remain vital in the management of advanced CRC.
Core Tip: In this study, 3 wild-type RAS metastatic colorectal cancer patients with anti-EGFR resistance were analyzed. Key findings: Acquired resistance is associated with KRAS mutation/HER-2 amplification and primary resistance is associated with metastatic KRAS mutation (tumor heterogeneity). These findings suggest essential genetic profiling
- Citation: Wang J, Pei Q, Yu NH. Anti-EGFR treatment resistance in patients with wild-type RAS metastatic colorectal cancer: Three case reports. World J Gastrointest Oncol 2026; 18(3): 114379
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/114379.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.114379
Anti-EGFR therapy has emerged as a significant advancement in the treatment of advanced colorectal cancer (CRC), leading to improved patient outcomes[1]. Currently, its use is limited to patients with wild-type RAS tumors[2]. However, a substantial proportion of patients with wild-type RAS still develop resistance during anti-EGFR treatment[3]. This resistance can be either primary (innate) or acquired. Through the analysis of three clinical cases, this study aims to enhance our understanding of the underlying mechanisms driving anti-EGFR treatment resistance in patients with wild-type RAS metastatic CRC.
Case 1: A 58-year-old male with persistent abdominal discomfort, diagnosed with left-sided colon cancer and multiple liver metastases.
Case 2: A 48-year-old male with no obvious symptoms, diagnosed with right-sided colon cancer and multiple liver metastases during routine medical examination.
Case 3: A 68-year-old male with intermittent abdominal pain, diagnosed with rectal cancer and synchronous liver metastasis 11 months after radical resection of sigmoid colon cancer.
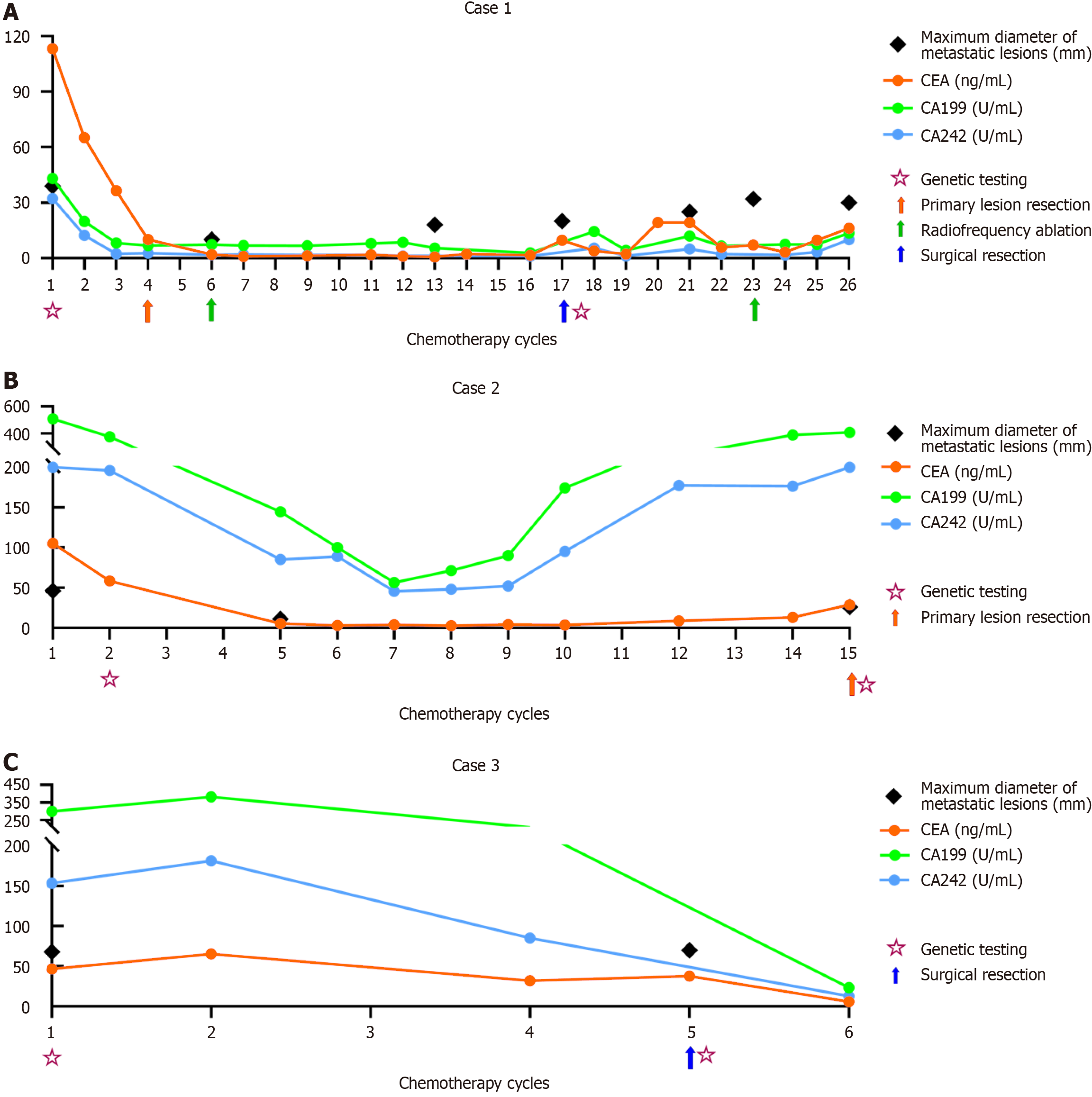
Case 1: In April 2020, abdominal ultrasound revealed multiple liver metastases, and subsequent colonoscopy confirmed left-sided colon cancer. Pyrosequencing showed wild-type RAS status. The patient received cetuximab combined with mFOLFOX6 chemotherapy. Initial response was observed, with decreased tumor markers (CA199, CA242) and reduced size of liver metastases. However, around the 17th cycle, tumor markers increased and liver metastatic lesions enlarged, indicating disease progression. The clinical timeline for case 1 is presented in Figure 1A.
Case 2: In March 2021, the diagnosis of right-sided colon cancer with multiple liver metastases was confirmed by colonoscopic biopsy and computed tomography (CT). Pyrosequencing of the primary tumor tissue confirmed wild-type RAS status. He was treated with cetuximab combined with FOLFIRI chemotherapy. Initial response was noted, with decreased CEA, CA199, and CA242 levels, and reduced metastatic lesion size. After 7-8 cycles, tumor markers rebounded and lesions enlarged, suggesting acquired resistance. By the 15th cycle, primary tumor resection was performed. The clinical timeline for case 2 is presented in Figure 1B.
Case 3: Eleven months prior to admission, the patient underwent “radical resection of sigmoid colon cancer” at a local hospital, with pyrosequencing confirming wild-type RAS status of the primary lesion. However, due to its large size and the associated high surgical risk, hepatic metastasis was not resected during the initial surgery. Postoperatively, he received cetuximab plus FOLFOX chemotherapy. Following this, serum tumor markers (CEA, CA199, CA242) remained persistently elevated. He was then admitted to our hospital for further management. During the first two cycles, tumor markers (CEA, CA199, CA242) and lesion sizes fluctuated but remained stable (stable disease). By the fifth cycle, imaging showed no significant reduction in metastatic lesions, indicating primary resistance. The clinical timeline for case 3 is presented in Figure 1C.
Case 1: Had a history of hypertension but was not on regular medication; no history of diabetes or other chronic diseases.
Case 2: No history of chronic diseases or surgical procedures before diagnosis.
Case 3: No significant past medical history or chronic disease.
The three patients reported no family history of malignancy or liver disease or other significant comorbidities. Case 1 and case 3 had a smoking history, with specific details unknown.
All three patients had normal vital signs. Abdominal examination showed no tenderness, rebound tenderness, or masses. Liver and spleen were not palpable. No abnormalities were found in cardiopulmonary, neurological, or other system examinations.
Case 1: CEA (ng/mL) was initially elevated at 113.26, CA199 (U/mL) at 43.10, and CA242 (U/mL) at 32.16. These tumor markers showed a progressive decrease as the chemotherapy cycles proceeded but began to increase to varying degrees around the 17th cycle of chemotherapy (Figure 1A).
Case 2: The patient’s tumor markers were initially elevated (CEA 105.34 ng/mL, CA199 299.14 U/mL, CA242 153.45 U/mL). They showed an initial decline with chemotherapy but began to rise again from approximately the 7-8 cycle onward (Figure 1B).
Case 3: Despite initial elevations (CEA 46.62 ng/mL, CA199 508.19 U/mL, CA242 > 200.00 U/mL), these tumors markers remained persistently elevated without a clear downward trend across five cycles of chemotherapy (before and after admission) (Figure 1C).
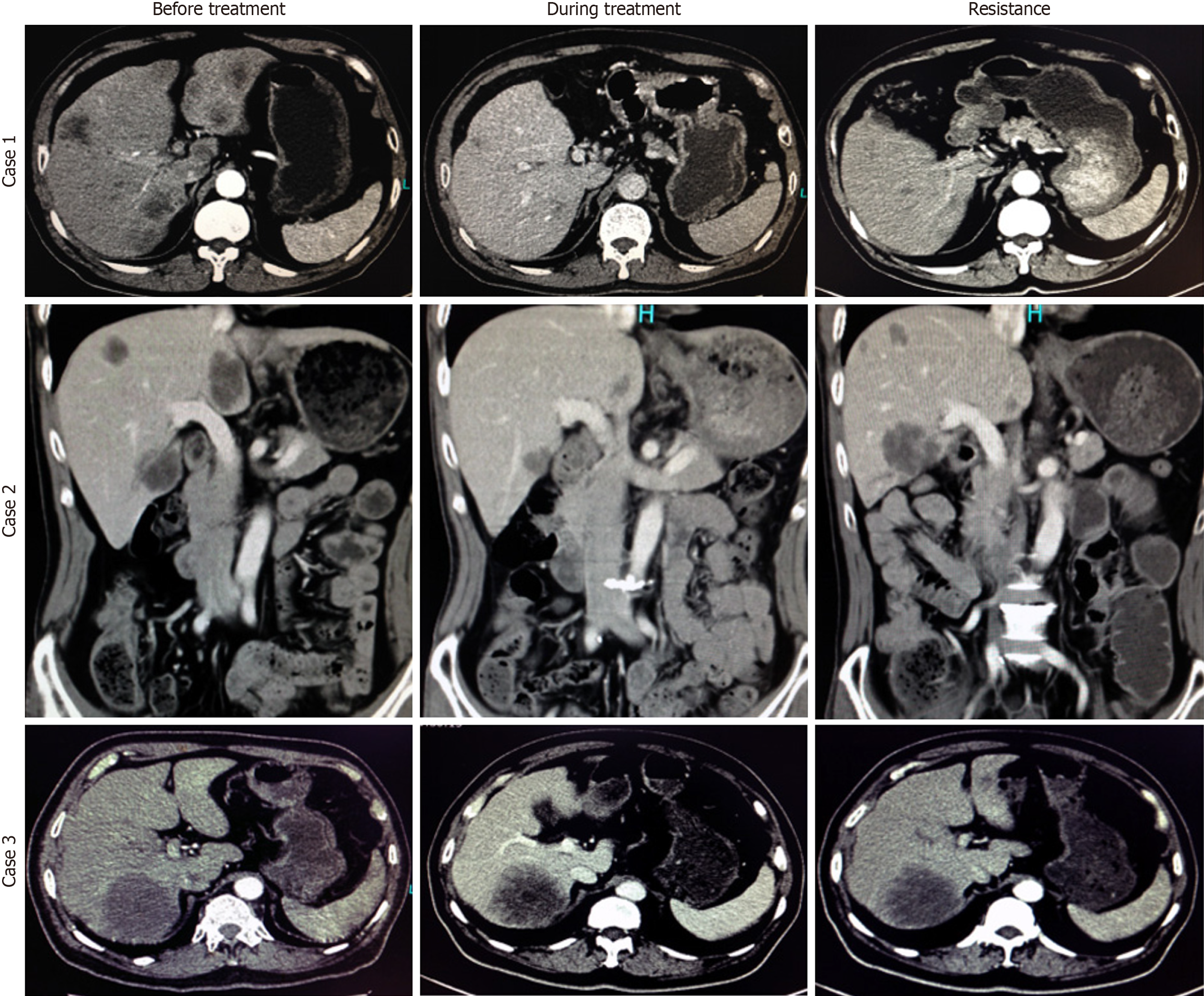
Case 1: Before treatment, CT imaging revealed a liver metastasis with a maximum diameter of 39 mm. During the initial phase of chemotherapy, the lesion demonstrated a significant reduction in size. Upon the development of acquired resistance, the maximum diameter of the lesion stabilized at 32 mm (Figure 2).
Case 2: Prior to therapy, the dominant liver metastasis measured 48 mm in maximum diameter. It showed a marked decrease in size during active treatment. However, following the emergence of treatment resistance, the maximum diameter increased to 46 mm, indicating radiographic progression (Figure 2).
Case 3: At diagnosis, a large liver metastasis with a maximum diameter of 68 mm was documented. Throughout the course of chemotherapy, serial imaging showed minimal change in the lesion size. Prior to surgical resection, the final maximum diameter was 70 mm, consistent with stable disease by imaging criteria (Figure 2).
Wild-type RAS metastatic left-sided colon cancer (liver metastases), anti-EGFR therapy acquired resistance (KRAS exon 2 mutation in metastatic lesions).
Wild-type RAS metastatic right-sided colon cancer (liver metastases), anti-EGFR therapy acquired resistance (HER2 overexpression/amplification in primary tumor).
Wild-type RAS metastatic rectal cancer (synchronous liver metastasis), anti-EGFR therapy primary resistance (KRAS exon 2 mutation in metastatic lesions, tumor heterogeneity).
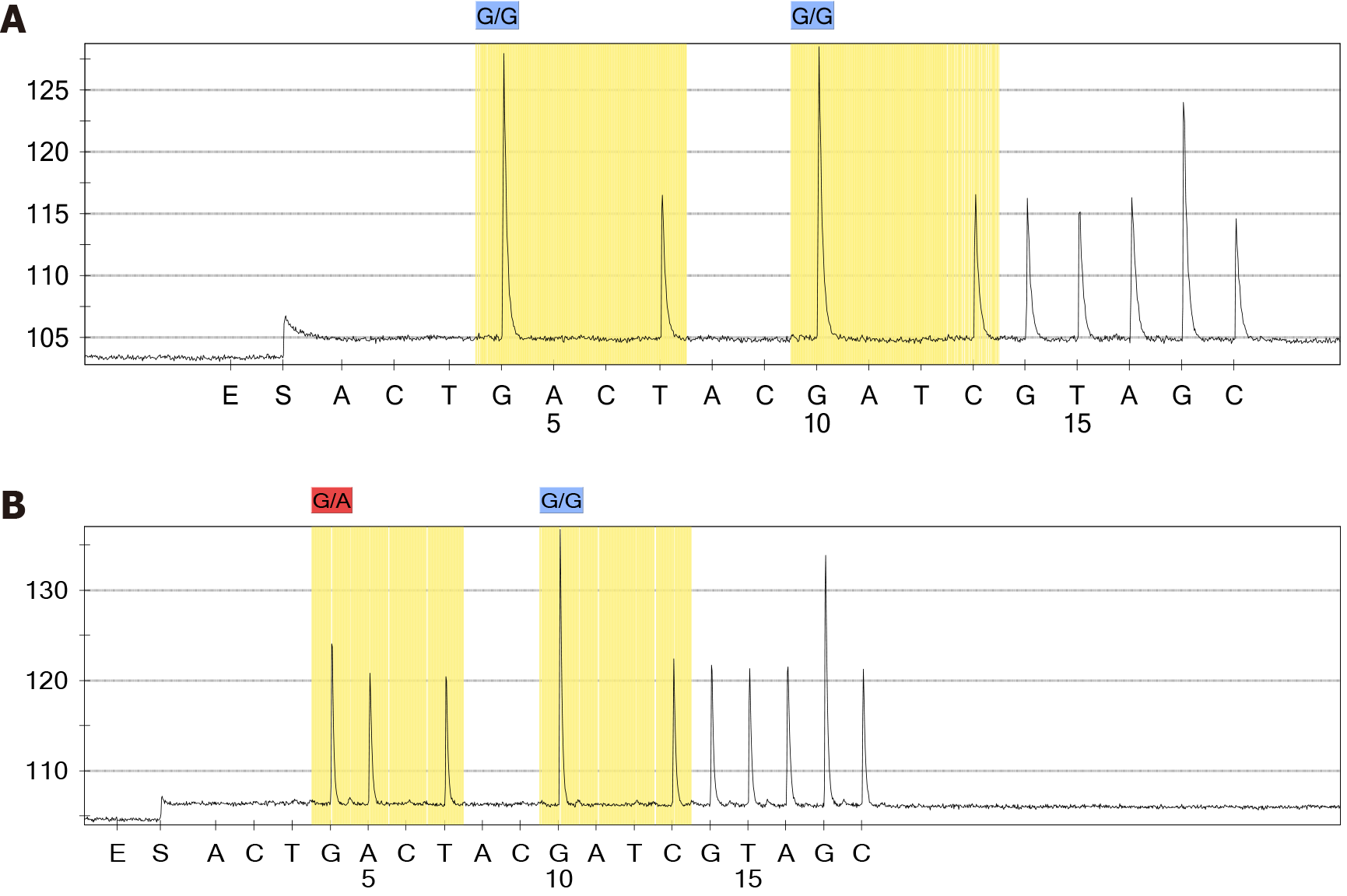
A 58-year-old male was diagnosed with leftsided colon cancer and multiple unresectable synchronous liver metastases. A colonoscopic biopsy confirmed wildtype RAS status. Initial treatment was cetuximab plus mFOLFOX6, with regular CEA and imaging surveillance for potential resectability. During the 4th cycle, he underwent palliative resection of the primary tumor. Pathology showed a pT3N0, welldifferentiated adenocarcinoma. Radiofrequency ablation of liver lesions was performed during the 7th and 23rd cycles. After the 12th cycle of cetuximab plus mFOLFOX6, treatment was modified to two cycles of cetuximab monotherapy followed by one cycle of cetuximab plus FOLFIRI. Following 15 cycles of targeted therapy, rising CEA and imaging progression of the liver metastases prompted an multidisciplinary team (MDT) discussion. Subsequently, during the 17th cycle, he underwent hepatic resection (segments II/III and V/VI/VII). Pathology of the resected liver confirmed metastatic, moderately differentiated adenocarcinoma. Genetic testing of this metastasis revealed an acquired KRAS exon 2 (codon 12) mutation (Figure 3 and Table 1).
| Case 1 | Case 2 | Case 3 | |
| Clinical baseline | |||
| Age | 58 | 48 | 68 |
| Gender | Male | Male | Male |
| Primary tumor location | Left-sided colon | Right-sided colon | Rectal |
| Metastatic site | Liver | Liver | Liver |
| Anti-EGFR therapy regimen | Cetuximab + mFOLFOX6 | Cetuximab + FOLFIRI | Cetuximab + FOLFOX |
| Cycle of resistance onset | 17 | 7-8 | 1-2 |
| Summary of resistance mechanism | Acquired resistance | Acquired resistance | Primary resistance |
| KRAS/NRAS/BRAF/PIK3CA status | KRAS exon 2 mt | - | KRAS exon 2 mt |
| HER2 expression/amplification | - | +++ | - |
| Clinical interventions | Palliative resection of the primary lesion, followed by resection of the hepatic metastatic lesion 12 months later | Palliative primary tumor resection was performed to resolve intestinal obstruction | Radical resection of sigmoid colon cancer, followed by resection of the hepatic metastatic lesion 14 months later |
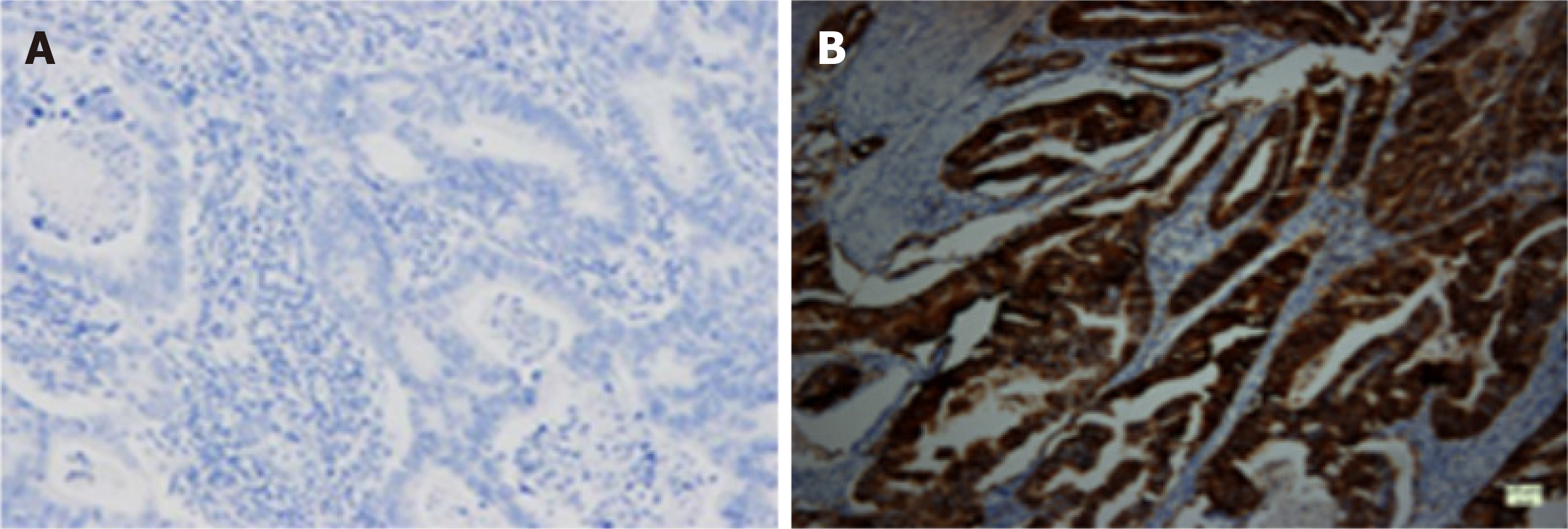
A 48-year-old male with right-sided colon cancer and unresectable synchronous liver metastases, confirmed to have wild-type RAS. Initial treatment comprised cetuximab plus mFOLFOX6, with regular monitoring of CEA and imaging for resectability reassessment. After 7-8 cycles, CEA rose significantly, imaging showed progression of both primary and metastatic lesions, and the patient developed incomplete bowel obstruction. Following MDT review, liver resection was not considered feasible. The patient subsequently underwent palliative right hemicolectomy at 15th cycles. Immunohistochemical (IHC) analysis of the primary tumor revealed HER2 protein overexpression, scored as 3+ (Figure 4). This IHC-positive result was further subjected to genetic testing via fluorescence in situ hybridization, which confirmed HER2 gene amplification (Table 1).
A 68-year-old male underwent a radical sigmoidectomy eleven months prior. Pathology confirmed a welldifferentiated adenocarcinoma (pT2, N0, margins negative). Due to the high surgical risk, resection of the synchronous liver metastasis was initially deferred. With a wildtype RAS primary tumor, he began cetuximab plus mFOLFOX6. Over four cycles, monitoring showed no clear decline in CEA and no significant reduction in the liver metastasis on imaging. Following an MDT discussion, he proceeded to hepatic metastasectomy. The liver lesion pathology revealed a moderately differentiated adenocarcinoma, consistent with metastatic disease. Subsequent genetic testing of this metastasis identified a KRAS exon 2 (codon 12) mutation (Table 1).
All three patients underwent their respective surgical procedures without immediate complications and returned for scheduled follow-up evaluations two months postoperatively.
Despite the established efficacy of anti-EGFR therapy in patients with RAS wild-type metastatic CRC, resistance remains a major clinical challenge. Our report delves into the underlying mechanisms by discussing three distinct cases of resistance.
In this series, two patients (cases 1 and 2) developed acquired resistance after an initial objective response (RECIST v.1.1)[4]. The molecular workup revealed two established mechanisms: A KRAS mutation in the progressing metastatic lesion (case 1) and HER2 amplification in the primary tumor (case 2).
Acquired resistance to anti-EGFR antibodies is driven primarily by molecular alterations that reactivate MAPK signaling. These mechanisms can be broadly categorized into two groups: (1) Mutations or amplifications within the EGFR-RAS-RAF-MEK-ERK pathway: This includes secondary mutations in KRAS, NRAS, or BRAF, which are the most common drivers. Amplification of KRAS or specific mutations in the extracellular domain of EGFR (e.g., S492R) also fall into this category; and (2) Activation of alternative receptor tyrosine kinases (RTKs): The amplification of other RTKs, such as HER2 (ERBB2) or MET, can activate downstream signaling (e.g., RAS-RAF-MEK) independent of EGFR, effectively bypassing therapeutic blockade. The prevalence of these mechanisms, as reported in the literature, is summarized in Table 2[5-12].
| Resistance mechanism | Detection source | Number of cases (%) | Ref. |
| KRAS gene mutation | Tissue | 1/10 (10) | Montagut et al[5] |
| Serum | 9/24 (37.5) | Diaz et al[6] | |
| Tissue and Serum | 5/11 (45.5) | Misale et al[7] | |
| Tissue and Serum | 3/7 (42.9) | Bardelli et al[8] | |
| Serum | 22/24 (91.7) | Bettegowda et al[9] | |
| Serum | 3/4 (75) | Misale et al[10] | |
| NRAS gene mutation | Serum | 9/24 (37.5) | Bettegowda et al[9] |
| Serum | 2/4 (50) | Misale et al[10] | |
| EGFR gene mutation | Tissue | 2/10 (20) | Montagut et al[5] |
| Serum | 2/24 (8.3) | Bettegowda et al[9] | |
| KRAS gene amplification | Tissue and Serum | 1/11 (9.1) | Misale et al[7] |
| Serum | 4/10 (40) | Mohan et al[11] | |
| HER-2 gene amplification | Tissue | 2/2 (100) | Yonesaka et al[12] |
| MET gene amplification | Tissue and Serum | 3/7 (42.9) | Bardelli et al[8] |
| Serum | 1/10 (10) | Mohan et al[11] |
Case 3 demonstrated primary resistance, with the best response being stable disease (per RECIST 1.1)[4]. Subsequent analysis revealed a KRAS mutation in the liver metastasis site that was not detected in the primary tumor. This case highlights the critical issue of tumor heterogeneity. While guidelines recommend RAS testing[13], and many studies have reported high concordance between primary and metastatic sites[5,14,15], significant discordance does occur[16-18]. Our initial assessment, which was based solely on the primary tumor, missed the resistant clone present in the metastasis. This finding suggests the need to genotype metastatic lesions whenever possible before initiating anti-EGFR therapy, especially in patients with equivocal or poor initial response.
Case 2 identifies HER2 amplification as the likely driver of acquired resistance. This is clinically relevant given the established efficacy of HER2-targeted therapies (e.g., trastuzumab) in HER2-positive breast[19] and gastric cancers[20]. A growing body of evidence suggests that dual HER2 blockade can be effective in patients with HER2-amplified, anti-EGFR-resistant CRC, making HER2 testing a crucial part of the resistance workup and opening avenues for subsequent targeted therapy[21].
The management of advanced CRC with targeted therapy aims to control disease, convert unresectable metastases to resectable, and guide surgical strategy. Given the complexity of resistance mechanisms and spatial heterogeneity, a MDT involving medical oncologists, liver surgeons, radiologists, and molecular pathologists is essential. This collaboration enables precise and dynamic assessment of treatment response and resistance patterns[6,9], informing critical decisions regarding therapy switches, local interventions (such as resection or ablation), and molecular reprofiling.
The resistance mechanisms observed in this study are consistent with and complement the literature. KRAS mutation resistance due to tumor heterogeneity in case 3, together with the KRAS copy number amplification case reported by Fang et al[22], collectively demonstrate that comprehensive genomic analysis beyond conventional RAS/BRAF point mutation testing, such as next-generation sequencing, is crucial for accurately identifying resistant populations. Conversely, a report by Xiao and Fakih[23] regarding the benefit in a right-sided colon cancer patient, along with the successful experience of Li et al[24] with combination targeted therapy, resonates with the findings of this study: In advanced late-line treatment, a precise molecular profile, particularly the confirmed absence of resistance mutations, is key to identifying potentially benefiting populations and guiding effective combination treatment strategies.
This study has a small sample size (3 patients). Although it illustrates the typical molecular mechanisms of anti-EGFR resistance, the generalizability of the conclusions is limited by the single-center, retrospective design. Future multicenter, large-sample studies are needed for further validation. Additionally, analyses covering more molecular targets, larger sample sizes, or more molecular levels (such as genomics, transcriptomics, and proteomics) can be conducted, incor
In conclusion, despite the preliminary nature of findings derived from a small case series, this research emphasizes KRAS mutations as the predominant mechanism, followed by NRAS and BRAF mutations, EGFR gene mutations, and RTK gene amplification. The identified molecular mechanisms contributing to acquired resistance include mutations or amplifications in EGFR-RAS-RAF-MEK-ERK pathway genes and RTKs.
In terms of primary resistance, this study suggests that tumor heterogeneity is a plausible cause. Comprehensive genetic testing of metastatic lesions is hence vital before starting anti-EGFR therapy. Moreover, HER-2 overexpression presents a potential mechanism for primary resistance, with HER-2-targeted therapy, such as trastuzumab, showing promise in the treatment of CRC.
In summary, this research highlights the critical need to understand anti-EGFR treatment resistance mechanisms in patients with wild-type RAS metastatic CRC. It advocates for thorough genetic profiling and consideration of HER-2-targeted therapy as an effective alternative. The role of multidisciplinary collaboration and regular molecular monitoring is underscored in the effective management of advanced CRC.
| 1. | Takeda M, Yoshida S, Inoue T, Sekido Y, Hata T, Hamabe A, Ogino T, Miyoshi N, Uemura M, Yamamoto H, Doki Y, Eguchi H. The Role of KRAS Mutations in Colorectal Cancer: Biological Insights, Clinical Implications, and Future Therapeutic Perspectives. Cancers (Basel). 2025;17:428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 37] [Article Influence: 37.0] [Reference Citation Analysis (0)] |
| 2. | Boilève A, Smolenschi C, Lambert A, Boige V, Delaye M, Camilleri GM, Tarabay A, Valéry M, Fuerea A, Pudlarz T, Mathieu JRR, Jaulin F, Hollebecque A, Ducreux M. KRAS, a New Target for Precision Medicine in Colorectal Cancer? Cancers (Basel). 2024;16:3455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 3. | Van Emburgh BO, Sartore-Bianchi A, Di Nicolantonio F, Siena S, Bardelli A. Acquired resistance to EGFR-targeted therapies in colorectal cancer. Mol Oncol. 2014;8:1084-1094. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 132] [Cited by in RCA: 133] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 4. | Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, Kaplan R, Lacombe D, Verweij J. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24191] [Cited by in RCA: 22884] [Article Influence: 1346.1] [Reference Citation Analysis (5)] |
| 5. | Montagut C, Dalmases A, Bellosillo B, Crespo M, Pairet S, Iglesias M, Salido M, Gallen M, Marsters S, Tsai SP, Minoche A, Seshagiri S, Serrano S, Himmelbauer H, Bellmunt J, Rovira A, Settleman J, Bosch F, Albanell J. Identification of a mutation in the extracellular domain of the Epidermal Growth Factor Receptor conferring cetuximab resistance in colorectal cancer. Nat Med. 2012;18:221-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 435] [Cited by in RCA: 395] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 6. | Diaz LA Jr, Williams RT, Wu J, Kinde I, Hecht JR, Berlin J, Allen B, Bozic I, Reiter JG, Nowak MA, Kinzler KW, Oliner KS, Vogelstein B. The molecular evolution of acquired resistance to targeted EGFR blockade in colorectal cancers. Nature. 2012;486:537-540. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1445] [Cited by in RCA: 1343] [Article Influence: 95.9] [Reference Citation Analysis (4)] |
| 7. | Misale S, Yaeger R, Hobor S, Scala E, Janakiraman M, Liska D, Valtorta E, Schiavo R, Buscarino M, Siravegna G, Bencardino K, Cercek A, Chen CT, Veronese S, Zanon C, Sartore-Bianchi A, Gambacorta M, Gallicchio M, Vakiani E, Boscaro V, Medico E, Weiser M, Siena S, Di Nicolantonio F, Solit D, Bardelli A. Emergence of KRAS mutations and acquired resistance to anti-EGFR therapy in colorectal cancer. Nature. 2012;486:532-536. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1620] [Cited by in RCA: 1500] [Article Influence: 107.1] [Reference Citation Analysis (4)] |
| 8. | Bardelli A, Corso S, Bertotti A, Hobor S, Valtorta E, Siravegna G, Sartore-Bianchi A, Scala E, Cassingena A, Zecchin D, Apicella M, Migliardi G, Galimi F, Lauricella C, Zanon C, Perera T, Veronese S, Corti G, Amatu A, Gambacorta M, Diaz LA Jr, Sausen M, Velculescu VE, Comoglio P, Trusolino L, Di Nicolantonio F, Giordano S, Siena S. Amplification of the MET receptor drives resistance to anti-EGFR therapies in colorectal cancer. Cancer Discov. 2013;3:658-673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 587] [Cited by in RCA: 558] [Article Influence: 42.9] [Reference Citation Analysis (0)] |
| 9. | Bettegowda C, Sausen M, Leary RJ, Kinde I, Wang Y, Agrawal N, Bartlett BR, Wang H, Luber B, Alani RM, Antonarakis ES, Azad NS, Bardelli A, Brem H, Cameron JL, Lee CC, Fecher LA, Gallia GL, Gibbs P, Le D, Giuntoli RL, Goggins M, Hogarty MD, Holdhoff M, Hong SM, Jiao Y, Juhl HH, Kim JJ, Siravegna G, Laheru DA, Lauricella C, Lim M, Lipson EJ, Marie SK, Netto GJ, Oliner KS, Olivi A, Olsson L, Riggins GJ, Sartore-Bianchi A, Schmidt K, Shih lM, Oba-Shinjo SM, Siena S, Theodorescu D, Tie J, Harkins TT, Veronese S, Wang TL, Weingart JD, Wolfgang CL, Wood LD, Xing D, Hruban RH, Wu J, Allen PJ, Schmidt CM, Choti MA, Velculescu VE, Kinzler KW, Vogelstein B, Papadopoulos N, Diaz LA Jr. Detection of circulating tumor DNA in early- and late-stage human malignancies. Sci Transl Med. 2014;6:224ra24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3960] [Cited by in RCA: 3732] [Article Influence: 311.0] [Reference Citation Analysis (4)] |
| 10. | Misale S, Arena S, Lamba S, Siravegna G, Lallo A, Hobor S, Russo M, Buscarino M, Lazzari L, Sartore-Bianchi A, Bencardino K, Amatu A, Lauricella C, Valtorta E, Siena S, Di Nicolantonio F, Bardelli A. Blockade of EGFR and MEK intercepts heterogeneous mechanisms of acquired resistance to anti-EGFR therapies in colorectal cancer. Sci Transl Med. 2014;6:224ra26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 227] [Cited by in RCA: 220] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 11. | Mohan S, Heitzer E, Ulz P, Lafer I, Lax S, Auer M, Pichler M, Gerger A, Eisner F, Hoefler G, Bauernhofer T, Geigl JB, Speicher MR. Changes in colorectal carcinoma genomes under anti-EGFR therapy identified by whole-genome plasma DNA sequencing. PLoS Genet. 2014;10:e1004271. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 159] [Cited by in RCA: 138] [Article Influence: 11.5] [Reference Citation Analysis (4)] |
| 12. | Yonesaka K, Zejnullahu K, Okamoto I, Satoh T, Cappuzzo F, Souglakos J, Ercan D, Rogers A, Roncalli M, Takeda M, Fujisaka Y, Philips J, Shimizu T, Maenishi O, Cho Y, Sun J, Destro A, Taira K, Takeda K, Okabe T, Swanson J, Itoh H, Takada M, Lifshits E, Okuno K, Engelman JA, Shivdasani RA, Nishio K, Fukuoka M, Varella-Garcia M, Nakagawa K, Jänne PA. Activation of ERBB2 signaling causes resistance to the EGFR-directed therapeutic antibody cetuximab. Sci Transl Med. 2011;3:99ra86. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 535] [Cited by in RCA: 535] [Article Influence: 35.7] [Reference Citation Analysis (5)] |
| 13. | Allegra CJ, Rumble RB, Hamilton SR, Mangu PB, Roach N, Hantel A, Schilsky RL. Extended RAS Gene Mutation Testing in Metastatic Colorectal Carcinoma to Predict Response to Anti-Epidermal Growth Factor Receptor Monoclonal Antibody Therapy: American Society of Clinical Oncology Provisional Clinical Opinion Update 2015. J Clin Oncol. 2016;34:179-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 207] [Article Influence: 20.7] [Reference Citation Analysis (1)] |
| 14. | Spindler KL, Pallisgaard N, Andersen RF, Jakobsen A. Changes in mutational status during third-line treatment for metastatic colorectal cancer--results of consecutive measurement of cell free DNA, KRAS and BRAF in the plasma. Int J Cancer. 2014;135:2215-2222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 66] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 15. | Therkildsen C, Bergmann TK, Henrichsen-Schnack T, Ladelund S, Nilbert M. The predictive value of KRAS, NRAS, BRAF, PIK3CA and PTEN for anti-EGFR treatment in metastatic colorectal cancer: A systematic review and meta-analysis. Acta Oncol. 2014;53:852-864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 324] [Cited by in RCA: 307] [Article Influence: 25.6] [Reference Citation Analysis (3)] |
| 16. | Jiang Y, Long G, Huang X, Wang W, Cheng B, Pan W. Single-cell transcriptomic analysis reveals dynamic changes in the liver microenvironment during colorectal cancer metastatic progression. J Transl Med. 2025;23:336. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (2)] |
| 17. | Joung JG, Oh BY, Hong HK, Al-Khalidi H, Al-Alem F, Lee HO, Bae JS, Kim J, Cha HU, Alotaibi M, Cho YB, Hassanain M, Park WY, Lee WY. Tumor Heterogeneity Predicts Metastatic Potential in Colorectal Cancer. Clin Cancer Res. 2017;23:7209-7216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 83] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 18. | Punekar SR, Velcheti V, Neel BG, Wong KK. The current state of the art and future trends in RAS-targeted cancer therapies. Nat Rev Clin Oncol. 2022;19:637-655. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 460] [Cited by in RCA: 394] [Article Influence: 98.5] [Reference Citation Analysis (0)] |
| 19. | Giordano SH, Franzoi MAB, Temin S, Anders CK, Chandarlapaty S, Crews JR, Kirshner JJ, Krop IE, Lin NU, Morikawa A, Patt DA, Perlmutter J, Ramakrishna N, Davidson NE. Systemic Therapy for Advanced Human Epidermal Growth Factor Receptor 2-Positive Breast Cancer: ASCO Guideline Update. J Clin Oncol. 2022;40:2612-2635. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 131] [Article Influence: 32.8] [Reference Citation Analysis (0)] |
| 20. | Wang H, Nie C, Xu W, Li J, Gou H, Lv H, Chen B, Wang J, Liu Y, He Y, Zhao J, Chen X. In era of immunotherapy: the value of trastuzumab beyond progression in patients with trastuzumab-resistant HER2-positive advanced or metastatic gastric cancer. Therap Adv Gastroenterol. 2024;17:17562848241245455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 21. | Khelwatty SA, Puvanenthiran S, Essapen S, Bagwan I, Seddon AM, Modjtahedi H. HER2 Expression Is Predictive of Survival in Cetuximab Treated Patients with RAS Wild Type Metastatic Colorectal Cancer. Cancers (Basel). 2021;13:638. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 22. | Fang T, Liang T, Wang Y, Wu H, Liu S, Xie L, Zhang Z, Liang J, Yao C, Tan Y, Wang C. An Early-Onset Advanced Rectal Cancer Patient With Increased KRAS Gene Copy Number Showed A Primary Resistance to Cetuximab in Combination With Chemotherapy: A Case Report. Front Oncol. 2021;11:755578. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 23. | Xiao A, Fakih M. Response to anti-EGFR therapy in chemo-refractory right-sided RAS wild-type metastatic colorectal cancer: a case report and literature review. J Gastrointest Oncol. 2025;16:292-300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 24. | Li Y, Chen X, Li W, Ye Y, Du X, Sun S, Liu L, Zhang H. Combination of Anti-EGFR and Anti-VEGF Drugs for the Treatment of Previously Treated Metastatic Colorectal Cancer: A Case Report and Literature Review. Front Oncol. 2021;11:684309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |