Published online Nov 15, 2020. doi: 10.4251/wjgo.v12.i11.1364
Peer-review started: June 24, 2020
First decision: August 9, 2020
Revised: August 20, 2020
Accepted: September 22, 2020
Article in press: September 22, 2020
Published online: November 15, 2020
Processing time: 140 Days and 16.1 Hours
Fistula formation is a severe adverse event related to antiangiogenetic agents such as bevacizumab and inferior mesenteric arteriovenous fistula (IMAVF) is a result of acquired factor, especially colon surgery. However, IMAVF occurs very rarely and there are few reports in patients during chemotherapy. We report a case of a patient who developed IMAVF during treatment with bevacizumab in metastatic colorectal cancer (mCRC) after colon surgery.
An 81-year-old man was diagnosed with descending colon cancer and underwent left hemicolectomy without any complications. He was definitely diagnosed with high-risk stage 2 and received tegafur-uracil plus leucovorin as adjuvant chemotherapy. Three years and 6 mo after the operation, the cancer relapsed with peritoneal dissemination. The patient underwent CyberKnife radiosurgery targeting the recurrent tumor and received chemotherapy with S-1 plus bevacizumab. At 1 year after chemotherapy, he complained of severe diarrhea, which is suspected drug-induced colitis. As diarrhea worsened despite the termination of treatment, he underwent colonoscopy and computed tomography (CT) scans that revealed edematous change from sigmoid to rectosigmoid colon. CT scans also revealed an aneurysm adjacent to the inferior mesenteric vein and multidetector CT angiography showed the IMAVF. Elective angiography confirmed the diagnosis of an IMAVF and it was successfully treated by arterial embolization. The patient resumed chemotherapy with only S-1 6 mo after embolization.
Clinicians should keep in mind the probability of severe diarrhea arose from IMAVF in mCRC patients treated with bevacizumab.
Core Tip: Gastrointestinal perforation and fistula formation are s bevacizumab-induced serious adverse events (SAEs). Arteriovenous fistula may occur during bevacizumab treatment via the action of antiangiogenetic agent. Inferior mesenteric arteriovenous fistula (IMAVF) arises from acquired factors especially colon surgery, although there has been no report related to chemotherapy including bevacizumab. We report a case of IMAVF in metastatic colorectal cancer (mCRC) during bevacizumab treatment. This patient complained of severe diarrhea caused by ischemic colitis due to IMAVF. As fistula may be lethal complications, clinicians should pay attention to SAEs including IMAVF for mCRC patients during bevacizumab treatment.
- Citation: Doi A, Takeda H, Umemoto K, Oumi R, Wada S, Hamaguchi S, Mimura H, Arai H, Horie Y, Mizukami T, Izawa N, Ogura T, Nakajima TE, Sunakawa Y. Inferior mesenteric arteriovenous fistula during treatment with bevacizumab in colorectal cancer patient: A case report. World J Gastrointest Oncol 2020; 12(11): 1364-1371
- URL: https://www.wjgnet.com/1948-5204/full/v12/i11/1364.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v12.i11.1364
Angiogenesis is important for cancer proliferation and promotion since blood vessels in tumors supply oxygen and nutrients, which support cancer growth. Vascular endothelial growth factor (VEGF) is a key factor for angiogenesis, promoting new blood vessel formation[1-3]. Bevacizumab is a recombinant humanized monoclonal IgG1 antibody that acts as an antiangiogenic drug targeting VEGF-A related to angiogenesis and inhibits the growth of tumor vessels as well as tumor proliferation. Hence, it has survival benefits for patients with metastatic colorectal cancer (mCRC) and is a useful standard therapy not only in the first-line but also for second-line treatment of mCRC[4-6].
Major adverse events (AEs) of bevacizumab include secondary hypertension, wound healing complications, and proteinuria. Practitioners should also be aware of certain rare but fatal bevacizumab-induced AEs such as hemorrhage, thrombosis, gastrointestinal (GI), non-GI perforation, and fistula formation. Although perforation and fistula formation have been reported in various cancers, they have been predominantly observed in GI sites and very rarely in non-GI sites. Non-GI fistula formation has been reported at tracheoesophageal, bronchopleural, biliary, vaginal, renal, and bladder sites in patients treated with bevacizumab; however, few have been observed in patients with mCRC. Alternative drugs for mCRC targeting angiogenesis include aflibercept, ramucirumab, and regorafenib. The incidence of fistula formation during treatment with antiangiogenetic agents ranged from less than 1% to 1.5% across previous phase III clinical trials of mCRC, specifically 0.9% (6/694) during bevacizumab treatment[6], 1.5% (9/611) during aflibercept treatment[7], 0.8% (4/529) during ramucirumab treatment[8], and 0.4% (2/500) during regorafenib treatment[9].
To inform clinicians of the possibility of this rare AE, we herein report a patient who developed an inferior mesenteric arteriovenous fistula (IMAVF) during bevacizumab treatment.
An 81-year-old man complained of frequent diarrhea more than 10 times per day without bleeding and abdominal pain. The patient’s symptom started 1 year after the start of palliative chemotherapy with S-1 plus bevacizumab. The diarrhea worsened progressively, and we suspected S-1-induced AE. However, despite the termination of treatment for 1 mo, severe diarrhea did not improve; therefore, a careful investigation was conducted into the cause of the serious adverse event (SAE).
The patient was diagnosed with descending colon cancer and underwent laparoscopic left hemicolectomy without any complications at 5 years prior to this report. However, he was later pathologically diagnosed with high-risk stage 2 descending colon cancer with venous and lymph duct invasion for which he received tegafur-uracil plus leucovorin for 6 mo as an adjuvant chemotherapy. At 3 years and 6 mo after the operation, the cancer relapsed with peritoneal dissemination in front of the left kidney. Stereotactic CyberKnife radiosurgery targeting the limited margin of the recurrent tumor resulting in tumor shrinkage, and the patient went onto palliative chemotherapy with S-1 plus bevacizumab for the remaining tumors.
He underwent surgery for appendicitis in his 20s and received Helicobacter pylori eradication therapy for gastric ulcer in his 80s.
The patient’s temperature was 36.5 °C, heart rate was 86 beats per min, respiratory rate was 18 breaths per min, blood pressure was 122/71 mmHg, and oxygen saturation in room air was 98%. In clinical abdominal examination, his abdomen was soft, flat with spontaneous pain and tenderness in the left lower quadrant.
Laboratory test results were almost normal. Blood analysis revealed that hemoglobin and hematocrit were slightly decreased at 12.2 g/dL and 37.0%, respectively, with normal leukocyte and platelet count. Prothrombin percentage activity was slightly decreased at 71% with normal activated partial thromboplastin time and d-dimers were slightly increased at 0.9 μg/mL. Serum C-reactive protein was slightly increased at 1.58 mg/dL (normal range < 0.3 mg/dL) and erythrocyte sedimentation rate at 21 mm/h. The blood biochemistries were almost normal. Urine analysis revealed mild proteinuria at 50 mg/dL. Chest X-ray was normal and electrocardiogram showed QT prolongation without clinical importance.
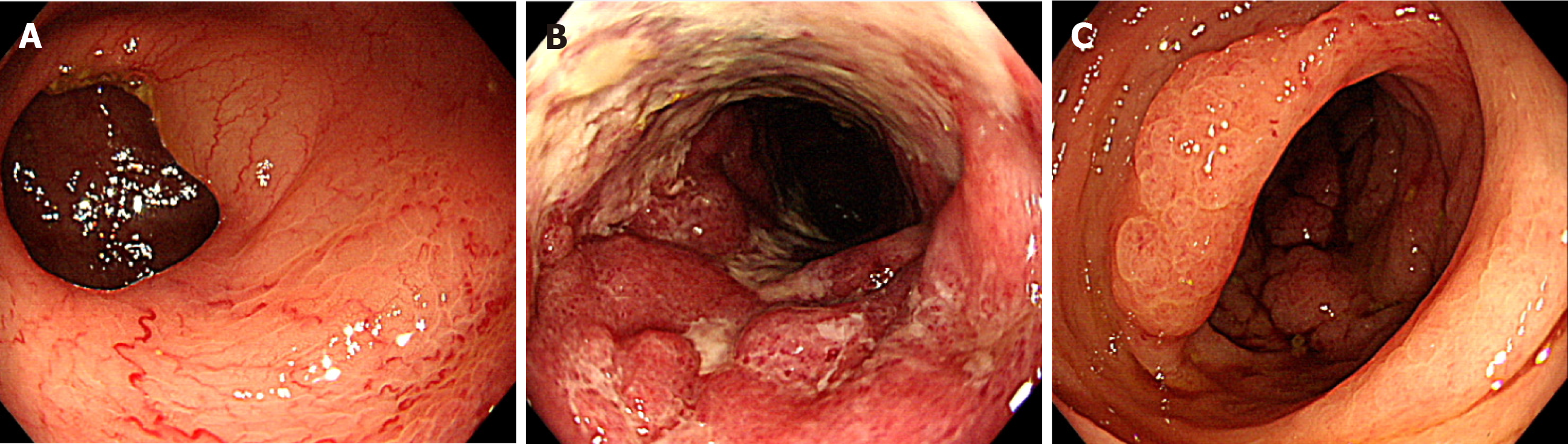
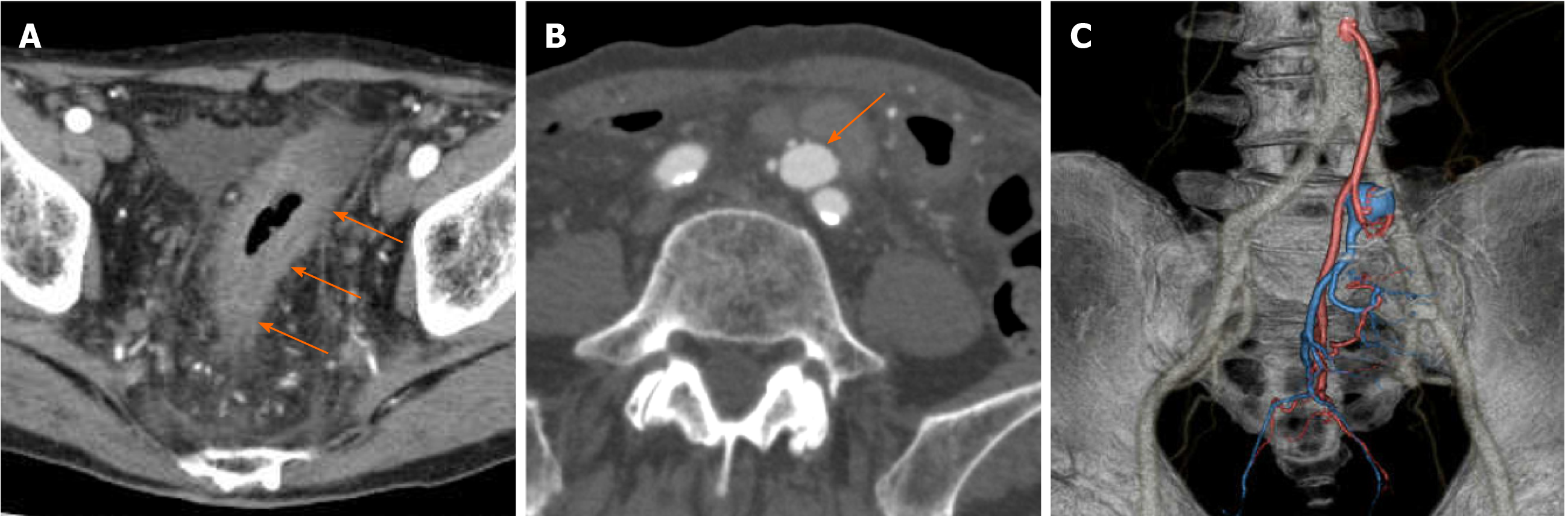
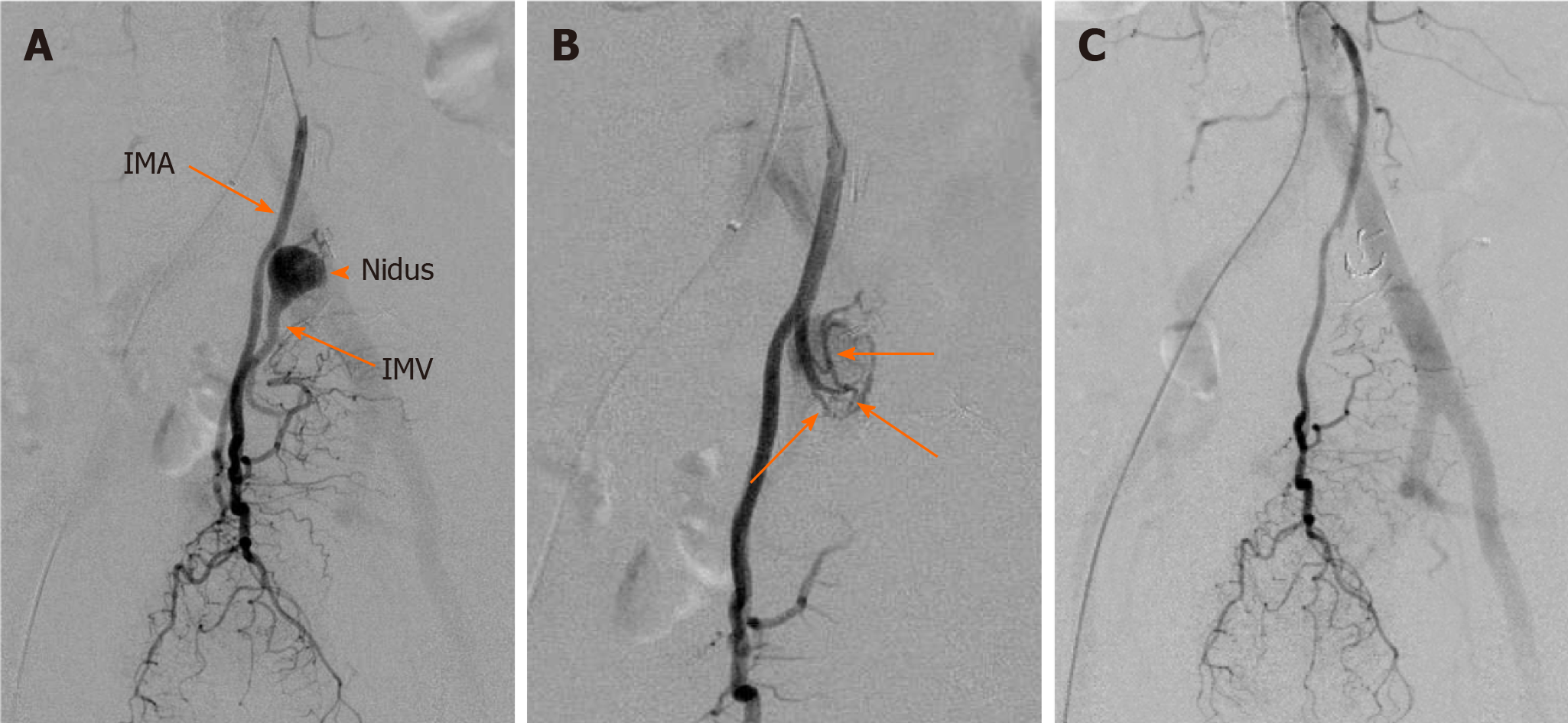
Colonoscopy showed abnormal colonic edematous mucosa with multiple ulcers stretching from the sigmoid colon (distal to the anastomotic site) to the rectosigmoid colon (Figure 1). Abdominal contrast-enhanced computed tomography (CT) scans revealed edematous change from the sigmoid colon to the rectum and a 1.8 cm × 1.3 cm aneurysm adjacent to the inferior mesenteric vein (IMV) (Figure 2A and B). Multidetector CT (MDCT) angiography showed the arteriovenous fistula with nidus involving the branches of the inferior mesenteric artery (IMA) and IMV (Figure 2C). An elective angiography of the IMA confirmed the existence of an IMAVF and defined several shunting points of the fistula from the branch of the IMA (Figure 3A and B).
The final diagnosis of the presented case was IMAVF with ischemic colitis.
This diagnosis was followed by treatment with interventional radiology (IR) on the same day. Abdominal angiography revealed that four small feeding arteries from the branch of the IMA were involved in AVF. Interventional radiologists judged that this case was candidate for transcatheter arterial embolization (TAE). TAE was performed by using detachable micro coils with 2 mm in diameter through the microcatheter, which was utilized to access the AVF. We did not use a liquid agent because there was a risk of rectal ischemia by accidental embolization of the superior rectal artery distal to AVF. Treatment succeeded without any complications and the AVF completely disappeared (Figure 3C).
Twelve days after fistula embolization, the patient’s symptom improved, and CT findings indicated an improvement in the edematous change of the colon. Reattempt colonoscopy also showed improvement of the colonic edematous mucosa. At 6 mo after the procedure, the patient recovered and resumed palliative chemotherapy with only S-1. He has continued the same treatment without any complications as of June 2020.
Previous studies[10,11] have reported the risk factors for GI perforation or fistula formation, which induce several types of cancer such as colorectal, ovarian, or renal cell cancer, as well as untreated primary tumor, colorectal surgery, abdominal irradiation, and chemotherapy-induced colitis. More than once surgery, irradiation, peritoneal dissemination, and cancer infiltration of the digestive tract were shown to be particularly high-risk factors[10,11]. The average period for the development of perforation or fistula formation after the start of treatment with bevacizumab was around 15 wk in several studies[11,12]. Most fistula formation occurs as a rectoperineal or colovesical fistulae in GI sites[12], while an AVF in CRC is extremely rare.
We report a case in which a patient with CRC developed an IMAVF during cancer treatment with bevacizumab after colorectal surgery. AVF including the IMA has been reported in only 35 cases so far. Among them, only seven articles available in the English language reported IMAVF following colorectal surgery from the PubMed database. Four of these patients were males and three females, with a median age of 63 years (range, 59–81 years) (Table 1)[13-17]. Most cases received surgical resection of the left colon involving the IMA and IMV. The median interval to diagnosis after the surgery was 5 years, and common symptoms were abdominal pain and GI bleeding. Only three cases of IMAVF have been observed in patients with CRC[16-18]. However, there has been no reported case of IMAVF related to chemotherapy, such as treatment with bevacizumab or other antiangiogenetic agents.
| Ref. | Sex | Age | Surgery | Interval to diagnosis | Symptoms | Treatment |
| Capron et al[14], 1984 | F | 60 | Colon resection | 11 yr | Abdominal pain, Meteorism | Embolization |
| Peer et al[15], 1989 | M | 63 | Anterior resection of rectum | 5 wk | Abdominal pain, Upper GI bleeding | Left colectomy |
| Pietri et al [13], 1990 | M | 72 | Left colectomy | Unknown | Abdominal mass | Left colectomy |
| F | 60 | Left colectomy | Unknown | Lower GI bleeding | Embolization | |
| Okada et al[16], 2002 | F | 69 | Sigmoidectomy | 8 yr | Abdominal mass | Left colectomy |
| Gorospe et al[17], 2012 | M | 59 | Right hemicolectomy | 5 yr | Abdominal pain, Lower GI bleeding, Diarrhea | Embolization, Total colectomy |
| Current case | M | 81 | Left hemicolectomy | 5 yr | Diarrhea | Embolization |
Angiography is useful for diagnosis of the IMAVF as it provides specific findings such as influent artery dilatation, abnormal blood vessel gathering, and early venous return. Recently, it has been shown that minimally invasive examinations including MR angiography and three- or four-dimensional CT angiography are also effective for diagnosis of IMAVF[18-20]. Treatment for the IMAVF includes surgery, IR, or both[13,14]. In the previous reported IMAVF cases, about half of the patients underwent surgical treatment or intra-arterial embolization, and only one patient underwent both treatments due to failure of the first embolization treatment[13-17]. Recently, IR has become the preferred method of treatment as it is easily repeatable and less invasive. It presents a good option in cases with identified fistula sites or when surgery is difficult due to complications. In the present case, the IMAVF was diagnosed by abdominal MDCT, and the diagnosis was confirmed by angiography. Since the location of the fistula and its feeding vessels were known, the IMAVF was treated by using arterial embolization. However, embolization may cause extensive arterial thrombosis followed by organ ischemia or recurrence in cases with more than one feeding vessel; therefore, it should not be performed in cases where the fistula has developed in a large vessel or has multiple feeding vessels[13,21]. In this case, an alternative possible treatment for ischemic colitis is resection of sigmoid colon and part of the rectum that are supplied by the IMA. Surgery may also be required in complications such as rebleeding, intestinal stenosis, or necrosis after IR.
To the best of our knowledge, the current case is the first report of an IMAVF in a patient with CRC who received palliative chemotherapy. The IMAVF occurred at 5 years after surgery (similar to the previously reported median time post-surgery) and 1 year after the initiation of bevacizumab treatment, which was later the average period of perforation or fistula formation after bevacizumab treatment initiation[12-17]. Also, although irradiation is considered to be a risk factor for perforation or fistula formation, it is unlikely to be the cause of the fistula in this case, because the recurrent site treated with CyberKnife stereotactic radiosurgery was distant from the site of the IMAVF and irradiation was limited to the margin of the tumor.
An AVF could occur due to acquired factors such as the spontaneous rupture of an aneurysm, trauma, or surgery. These are responsible for fistula formation in approximately 60% of cases, whereas 25% of fistula are due to congenital factors such as arteriovenous malformation[13]. AVF most often develops in the celiac artery or one of its branches, especially the hepatic artery (45%), or the splenic artery (30%). An IMAVF may cause ischemic colitis by steal syndrome causing a reduction in blood flow supply to the bowel wall because arterial blood directly flows into venous blood[13,21]. Several reports have indicated the IMAVF formation after colectomy to hematoma or infection followed by necrosis of the vessel wall with pseudoaneurysm formation, such as vessel injury and ligation, during surgical procedures[22,23]. However, the mechanisms by which surgery or chemotherapy with antiangiogenetic agent or both treatments cause IMAVF formation remain unclear. The risk of IMAVF formation during treatment with antiangiogenetic agent in CRC patients after colorectal surgery has significant implications for clinicians. Most patients with IMAVF complained of symptoms such as abdominal pain and GI bleeding followed by diarrhea[13-17], which overlapped in some cases. These symptoms are very common in mCRC patients due to cancer itself or other drug related adverse events and they are many mCRC patients during treatment with antiangiogenetic agent, which is most bevacizumab, after colorectal surgery. Therefore, this is an extremely important case report that clinician need to know. In order to avoid SAEs like this case, we should pay attention to abdominal related symptoms, such as pain, bloody stool and diarrhea, then diagnose and treat a patient as rapidly as possible.
We report on a case of IMAVF to have occurred during bevacizumab treatment in a patient with mCRC after colon surgery. There are many recurrent CRC patients after colon surgery and they routinely receive bevacizumab in clinical practice; however, as this case indicates, clinicians need to pay attention to SAEs related to bevacizumab. Formation of IMAVF and other fistula are potentially lethal complications causing massive bleeding and peritonitis. Accordingly, the clinician should keep in mind the probability of vascular-related AEs, including IMAVF, if patients complain of severe diarrhea during treatment with bevacizumab or other antiangiogenetic agents.
| 1. | Dvorak HF. Vascular permeability factor/vascular endothelial growth factor: a critical cytokine in tumor angiogenesis and a potential target for diagnosis and therapy. J Clin Oncol. 2002;20:4368-4380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1274] [Cited by in RCA: 1135] [Article Influence: 47.3] [Reference Citation Analysis (3)] |
| 2. | Hicklin DJ, Ellis LM. Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol. 2005;23:1011-1027. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2436] [Cited by in RCA: 2183] [Article Influence: 104.0] [Reference Citation Analysis (0)] |
| 3. | Folkman J. Tumor angiogenesis: therapeutic implications. N Engl J Med. 1971;285:1182-1186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6081] [Cited by in RCA: 5865] [Article Influence: 106.6] [Reference Citation Analysis (5)] |
| 4. | Giantonio BJ, Catalano PJ, Meropol NJ, O'Dwyer PJ, Mitchell EP, Alberts SR, Schwartz MA, Benson AB, Eastern Cooperative Oncology Group Study E3200. Bevacizumab in combination with oxaliplatin, fluorouracil, and leucovorin (FOLFOX4) for previously treated metastatic colorectal cancer: results from the Eastern Cooperative Oncology Group Study E3200. J Clin Oncol. 2007;25:1539-1544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1868] [Cited by in RCA: 1674] [Article Influence: 88.1] [Reference Citation Analysis (7)] |
| 5. | Kozloff M, Yood MU, Berlin J, Flynn PJ, Kabbinavar FF, Purdie DM, Ashby MA, Dong W, Sugrue MM, Grothey A; Investigators of the BRiTE study. Clinical outcomes associated with bevacizumab-containing treatment of metastatic colorectal cancer: the BRiTE observational cohort study. Oncologist. 2009;14:862-870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 218] [Article Influence: 12.8] [Reference Citation Analysis (3)] |
| 6. | Saltz LB, Clarke S, Díaz-Rubio E, Scheithauer W, Figer A, Wong R, Koski S, Lichinitser M, Yang TS, Rivera F, Couture F, Sirzén F, Cassidy J. Bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: a randomized phase III study. J Clin Oncol. 2008;26:2013-2019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2410] [Cited by in RCA: 2208] [Article Influence: 122.7] [Reference Citation Analysis (6)] |
| 7. | Van Cutsem E, Tabernero J, Lakomy R, Prenen H, Prausová J, Macarulla T, Ruff P, van Hazel GA, Moiseyenko V, Ferry D, McKendrick J, Polikoff J, Tellier A, Castan R, Allegra C. Addition of aflibercept to fluorouracil, leucovorin, and irinotecan improves survival in a phase III randomized trial in patients with metastatic colorectal cancer previously treated with an oxaliplatin-based regimen. J Clin Oncol. 2012;30:3499-3506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1173] [Cited by in RCA: 1045] [Article Influence: 74.6] [Reference Citation Analysis (4)] |
| 8. | Tabernero J, Yoshino T, Cohn AL, Obermannova R, Bodoky G, Garcia-Carbonero R, Ciuleanu TE, Portnoy DC, Van Cutsem E, Grothey A, Prausová J, Garcia-Alfonso P, Yamazaki K, Clingan PR, Lonardi S, Kim TW, Simms L, Chang SC, Nasroulah F; RAISE Study Investigators. Ramucirumab vs placebo in combination with second-line FOLFIRI in patients with metastatic colorectal carcinoma that progressed during or after first-line therapy with bevacizumab, oxaliplatin, and a fluoropyrimidine (RAISE): a randomised, double-blind, multicentre, phase 3 study. Lancet Oncol. 2015;16:499-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 805] [Cited by in RCA: 696] [Article Influence: 63.3] [Reference Citation Analysis (8)] |
| 9. | Grothey A, Van Cutsem E, Sobrero A, Siena S, Falcone A, Ychou M, Humblet Y, Bouché O, Mineur L, Barone C, Adenis A, Tabernero J, Yoshino T, Lenz HJ, Goldberg RM, Sargent DJ, Cihon F, Cupit L, Wagner A, Laurent D; CORRECT Study Group. Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381:303-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2394] [Cited by in RCA: 2210] [Article Influence: 170.0] [Reference Citation Analysis (6)] |
| 10. | Hapani S, Chu D, Wu S. Risk of gastrointestinal perforation in patients with cancer treated with bevacizumab: a meta-analysis. Lancet Oncol. 2009;10:559-568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 369] [Cited by in RCA: 312] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 11. | Sliesoraitis S, Tawfik B. Bevacizumab-induced bowel perforation. J Am Osteopath Assoc. 2011;111:437-441. [PubMed] |
| 12. | Ganapathi AM, Westmoreland T, Tyler D, Mantyh CR. Bevacizumab-associated fistula formation in postoperative colorectal cancer patients. J Am Coll Surg. 2012;214:582-8; discussion 588-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 25] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 13. | Pietri J, Remond A, Reix T, Abet D, Sevestre H, Sevestre MA. Arterioportal fistulas: twelve cases. Ann Vasc Surg. 1990;4:533-539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 20] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 14. | Capron JP, Gineston JL, Remond A, Lallement PY, Delamarre J, Revert R, Veyssier P. Inferior mesenteric arteriovenous fistula associated with portal hypertension and acute ischemic colitis. Successful occlusion by intraarterial embolization with steel coils. Gastroenterology. 1984;86:351-355. [PubMed] |
| 15. | Peer A, Slutzki S, Witz E, Abrahmsohn R, Bogokowsky H, Leonov Y. Transcatheter occlusion of inferior mesenteric arteriovenous fistula: a case report. Cardiovasc Intervent Radiol. 1989;12:35-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 16. | Okada K, Furusyo N, Sawayama Y, Ishikawa N, Nabeshima S, Tsuchihashi T, Kashiwagi S, Hayashi J. Inferior mesenteric arteriovenous fistula eight years after sigmoidectomy. Intern Med. 2002;41:543-548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 20] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 17. | Gorospe EC, Leggett CL, Sun G. Inferior mesenteric arteriovenous malformation: an unusual cause of ischemic colitis. Ann Gastroenterol. 2012;25:165. [PubMed] |
| 18. | Kai K, Sano K, Higuchi K, Uchiyama S, Sueta H, Nanashima A. A rare case of simultaneous rectal and gastric carcinomas accompanied with inferior mesenteric arterioportal fistula: case report. Surg Case Rep. 2019;5:82. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 19. | Lee S, Chung J, Ahn B, Lee S, Baek S. Inferior mesenteric arteriovenous fistula. Ann Surg Treat Res. 2017;93:225-228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 20. | Cheng L, Zhao R, Guo D, Cai K, Zou K, Yang J, Zhu L. Inferior mesenteric arteriovenous fistula with nonpulsatile abdominal mass: A case report and a mini-review. Medicine (Baltimore). 2017;96:e8717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 21. | Metcalf DR, Nivatvongs S, Andrews JC. Ischemic colitis: an unusual case of inferior mesenteric arteriovenous fistula causing venous hypertension. Report of a case. Dis Colon Rectum. 2008;51:1422-1424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 24] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 22. | Rossi P, Carillo FJ, Alfidi RJ, Ruzicka FF. Iatrogenic arteriovenous fistulas. Radiology. 1974;111:47-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 12] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 23. | Athanasiou A, Michalinos A, Alexandrou A, Georgopoulos S, Felekouras E. Inferior mesenteric arteriovenous fistula: case report and world-literature review. World J Gastroenterol. 2014;20:8298-8303. [PubMed] [DOI] [Full Text] |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See:
Manuscript source: Unsolicited manuscript
Specialty type: Oncology
Country/Territory of origin: Japan
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Jesus-Silva SG S-Editor: Ma YJ L-Editor: Filipodia P-Editor: Li JH